This site displays a prototype of a “Web 2.0” version of the daily Federal Register. It is not an official legal edition of the Federal Register, and does not replace the official print version or the official electronic version on GPO’s govinfo.gov.
The documents posted on this site are XML renditions of published Federal Register documents. Each document posted on the site includes a link to the corresponding official PDF file on govinfo.gov. This prototype edition of the daily Federal Register on FederalRegister.gov will remain an unofficial informational resource until the Administrative Committee of the Federal Register (ACFR) issues a regulation granting it official legal status. For complete information about, and access to, our official publications and services, go to About the Federal Register on NARA's archives.gov.
The OFR/GPO partnership is committed to presenting accurate and reliable regulatory information on FederalRegister.gov with the objective of establishing the XML-based Federal Register as an ACFR-sanctioned publication in the future. While every effort has been made to ensure that the material on FederalRegister.gov is accurately displayed, consistent with the official SGML-based PDF version on govinfo.gov, those relying on it for legal research should verify their results against an official edition of the Federal Register. Until the ACFR grants it official status, the XML rendition of the daily Federal Register on FederalRegister.gov does not provide legal notice to the public or judicial notice to the courts.
AGENCY:
Centers for Medicare & Medicaid Services (CMS), Department of Health and Human Services (HHS).
ACTION:
Final rule with comment period.
SUMMARY:
This final rule with comment period revises the Medicare Hospital Outpatient Prospective Payment System (OPPS) and the Medicare Ambulatory Surgical Center (ASC) payment system for calendar year 2025 based on our continuing experience with these systems. We describe the changes to the amounts and factors used to determine the payment rates for Medicare services paid under the OPPS and those paid under the ASC payment system. Also, this final rule updates the requirements for the Hospital Outpatient Quality Reporting Program, Rural Emergency Hospital Quality Reporting Program, Ambulatory Surgical Center Quality Reporting Program, and Hospital Inpatient Quality Reporting Program. We also summarize information received in response to a Request for Information on potential modifications to the Safety of Care measure group in the Overall Hospital Quality Star Rating methodology. In this final rule, we are also finalizing our proposal to narrow the description of “custody” in the Medicare payment exclusion rule and to revise the special enrollment period criteria for formerly incarcerated individuals. We are also finalizing our Medicaid and Children's Health Insurance Program (CHIP) continuous eligibility provisions. We are also finalizing the proposal to reduce the review timeframe for standard prior authorization requests for certain covered outpatient department services paid under the OPPS from 10-business days to 7-calendar days. Further, this rule finalizes updates to the Conditions of Participation (CoPs) for hospitals and critical access hospitals (CAHs) in an effort to advance the health and safety of pregnant, birthing, and postpartum women. This rule also finalizes our proposed policy to separately pay Indian Health Service (IHS) and Tribal hospitals for high-cost drugs furnished in hospital outpatient departments through an add-on payment in addition to the all-inclusive rate (AIR) under the authorities used to calculate the AIR starting January 1, 2025. Finally, we are finalizing exceptions to the Medicaid clinic services four walls requirement for IHS and Tribal clinics, and, at state option, for behavioral health clinics and clinics located in rural areas.
DATES:
Effective date: The provisions of this rule are effective January 1, 2025.
Implementation date: Except as set forth in this section, the regulations at §§ 482.43(c), 482.55(c), and 485.618(e) must be implemented by July 1, 2025; the regulations at §§ 482.59(a) and (b) and 485.649(a) and (b) must be implemented by January 1, 2026; and the regulations at §§ 482.21(b)(4) and (e), 482.59(c), 485.641(d)(4) and (e)(2), and 485.649(c) must be implemented by January 1, 2027.
Comment period: To be assured consideration, comments must be received at one of the addresses provided below, by December 31, 2024.
ADDRESSES:
In commenting, please refer to file code CMS-1809-FC.
Comments, including mass comment submissions, must be submitted in one of the following three ways (please choose only one of the ways listed):
1. Electronically. You may submit electronic comments on this regulation to https://www.regulations.gov. Follow the “Submit a comment” instructions.
2. By regular mail. You may mail written comments to the following address ONLY: Centers for Medicare & Medicaid Services, Department of Health and Human Services, Attention: CMS-1809-FC, P.O. Box 8010, Baltimore, MD 21244-8010.
Please allow sufficient time for mailed comments to be received before the close of the comment period.
3. By express or overnight mail. You may send written comments to the following address ONLY: Centers for Medicare & Medicaid Services, Department of Health and Human Services, Attention: CMS-1809-FC, Mail Stop C4-26-05, 7500 Security Boulevard, Baltimore, MD 21244-1850.
For information on viewing public comments, see the beginning of the SUPPLEMENTARY INFORMATION section.
FOR FURTHER INFORMATION CONTACT:
Regulation coordination questions at OPPS-ASC-Rulemaking@cms.hhs.gov or Elise Barringer (410) 786, 9222.
Advisory Panel on Hospital Outpatient Payment (HOP Panel), contact the HOP Panel mailbox at APCPanel@cms.hhs.gov.
Ambulatory Surgical Center Quality Reporting (ASCQR) Program policies, contact Anita Bhatia via email at Anita.Bhatia@cms.hhs.gov.
Ambulatory Surgical Center Quality Reporting (ASCQR) Program measures, contact Marsha Hertzberg via email at Marsha.Hertzberg@cms.hhs.gov.
All-Inclusive Rate (AIR) Add-On Payment for High-Cost Drugs Provided by Indian Health Service (IHS) and Tribal Facilities, contact Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
Blood and Blood Products, contact Au'Sha Washington via email at AuShaWashington@cms.hhs.gov or Nicole Marcos via email at Nicole.Marcos@cms.hhs.gov.
Cancer Hospital Payments, contact Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
CMS Web Posting of the OPPS and ASC Payment Files, contact Gil Ngan via email at Gil.Ngan@cms.hhs.gov.
Medicaid Clinic Services Four Walls Exceptions, contact Sheri Gaskins via email at Sheri.Gaskins@cms.hhs.gov or Ryan Tisdale via email at Ryan.Tisdale@cms.hhs.gov.
Composite APCs (Multiple Imaging and Mental Health) and Comprehensive APCs (C-APCs), via email at Elise Barringer via email at Elise.Barringer@cms.hhs.gov.
Device-Intensive Status and No Cost/Full Credit and Partial Credit Devices, contact Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
Domestic Personal Protection Equipment RFI, contact Jesse Hawkins via email at jesse.hawkins@hhs.gov
Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals, contact The Clinical Standards Group, HealthandSafetyInquiries@cms.hhs.gov
Hospital Inpatient Quality Reporting (IQR) Program policies, contact Julia Venanzi via email at julia.venanzi@cms.hhs.gov. ( printed page 93913)
Hospital Inpatient Quality Reporting (IQR) Program measures, contact Melissa Hager via email at melissa.hager@cms.hhs.gov or Ngozi Uzokwe via email at ngozi.uzokwe@cms.hhs.gov.
Hospital Outpatient Quality Reporting (OQR) Program policies, contact Kimberly Go via email at Kimberly.Go@cms.hhs.gov.
Hospital Outpatient Quality Reporting (OQR) Program measures, contact Janis Grady via email at Janis.Grady@cms.hhs.gov.
Hospital Outpatient Visits (Emergency Department Visits and Critical Care Visits), contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov.
IHS Outpatient Encounter Rate available to all American Indian and Alaska Native (AI/AN) Outpatient Programs Request for Information, contact Lisa Parker via email at Lisa.Parker1@cms.hhs.gov.
Inpatient Only (IPO) Procedures List, contact Abigail Cesnik via email at Abigail.Cesnik@cms.hhs.gov.
Medicaid and CHIP Continuous Eligibility Policy, contact Cassie Lagorio via email at Cassandra.Lagorio@cms.hhs.gov.
New Technology Intraocular Lenses (NTIOLs), contact Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
No Legal Obligation to Pay Payment Exclusion, contact Frederick Grabau via email at Frederick.Grabau@cms.hhs.gov.
Non-Opioid Policy or Implementation of Section 4135 of the Consolidated Appropriations Act (CAA), 2023, contact Cory Duke via email at Cory.Duke@cms.hhs.gov.
OPPS Brachytherapy, contact Cory Duke via email at Cory.Duke@cms.hhs.gov and Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
OPPS Data (APC Weights, Conversion Factor, Copayments, Cost-to-Charge Ratios (CCRs), Data Claims, Geometric Mean Calculation, Outlier Payments, and Wage Index), contact Erick Chuang via email at Erick.Chuang@cms.hhs.gov or Scott Talaga via email at Scott.Talaga@cms.hhs.gov.
OPPS Dental Policy, contact Nicole Marcos via email at Nicole.Marcos@cms.hhs.gov.
OPPS Drugs, Radiopharmaceuticals, Biologicals, and Biosimilar Products, contact Gil Ngan via email at Gil.Ngan@cms.hhs.gov, Cory Duke via email at Cory.Duke@cms.hhs.gov, or Tonya Gierke via email at Tonya.Gierke@cms.hhs.gov.
OPPS New Technology Procedures/Services, contact the New Technology APC mailbox at NewTechAPCapplications@cms.hhs.gov.
OPPS Packaged Items/Services, contact Cory Duke via email at Cory.Duke@cms.hhs.gov.
OPPS Pass-Through Devices, contact the Device Pass-Through mailbox at DevicePTapplications@cms.hhs.gov.
OPPS Status Indicators (SI) and Comment Indicators (CI), contact Marina Kushnirova via email at Marina.Kushnirova@cms.hhs.gov.
Outpatient Department Prior Authorization Process, contact Kelly Wojciechowski via email at Kelly.Wojciechowski@cms.hhs.gov.
Overall Hospital Quality Star Rating Request for Information, contact Tyson Nakashima Sr. via email at Tyson.Nakashima@cms.hhs.gov.
Partial Hospitalization Program (PHP), Intensive Outpatient (IOP), and Community Mental Health Center (CMHC) Issues, contact the PHP Payment Policy Mailbox at PHPPaymentPolicy@cms.hhs.gov.
Payment Policy for Devices in Category B Investigational Device Exemption Clinical Trials Policy and Drugs with a Medicare Coverage with Evidence Development (CED) Designation, contact Cory Duke via email at Cory.Duke@cms.hhs.gov and Nicole Marcos via email at Nicole.Marcos@cms.hhs.gov.
Remote Services, contact Elise Barringer via email at Elise.Barringer@cms.hhs.gov or Nate Vercauteren via email at Nathan.Vercauteren@cms.hhs.gov.
Rural Emergency Hospital Quality Reporting (REHQR) Program policies, contact Anita Bhatia via email at Anita.Bhatia@cms.hhs.gov.
Rural Emergency Hospital Quality Reporting (REHQR) Program measures, contact Melissa Hager via email at Melissa.Hager@cms.hhs.gov.
Special Enrollment Period for Formerly Incarcerated Individuals, contact Steve Manning via email at Steve.Manning@cms.hhs.gov.
All Other Issues Related to Hospital Outpatient Payments Not Previously Identified, contact the OPPS mailbox at OutpatientPPS@cms.hhs.gov.
All Other Issues Related to the Ambulatory Surgical Center Payments Not Previously Identified, contact the ASC mailbox at ASCPPS@cms.hhs.gov.
SUPPLEMENTARY INFORMATION:
Inspection of Public Comments: All comments received before the close of the comment period are available for viewing by the public, including any personally identifiable or confidential business information that is included in a comment. We post all comments received before the close of the comment period on the following website as soon as possible after they have been received: https://www.regulations.gov. Follow the search instructions on that website to view public comments. CMS will not post on Regulations.gov public comments that make threats to individuals or institutions or suggest that the individual will take actions to harm the individual. CMS continues to encourage individuals not to submit duplicative comments. We will post acceptable comments from multiple unique commenters even if the content is identical or nearly identical to other comments.
Plain Language Summary: In accordance with 5 U.S.C. 553(b)(4), a plain language summary of this rule may be found at https://www.regulations.gov/.
Severability of Provisions: We are clarifying and emphasizing our intent that if any provision of this final rule is held to be invalid or unenforceable by its terms, or as applied to any person or circumstance, or stayed pending further action, it shall be severable from other parts of this final rule, and from rules and regulations currently in effect, and not affect the remainder thereof or the application of the provision to other persons not similarly situated or to other, dissimilar circumstances. If any provision is held to be invalid or unenforceable, the remaining provisions which could function independently should take effect and be given the maximum effect permitted by law. Through this rule, we adopt provisions that are intended to and will operate independently of each other, even if each serves the same general purpose or policy goal. Where a provision is necessarily dependent on another, the context generally makes that clear.
Addenda Available Only Through the Internet on the CMS Website
In the past, a majority of the Addenda referred to in our OPPS/ASC proposed and final rules were published in the Federal Register as part of the annual rulemakings. However, beginning with the calendar year (CY) 2012 OPPS/ASC proposed rule, all of the Addenda no longer appear in the Federal Register as part of the annual OPPS/ASC proposed and final rules to decrease administrative burden and reduce costs associated with publishing lengthy tables. Instead, these Addenda are published and available only on the CMS website. The Addenda relating to the OPPS are available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. The Addenda relating to the ASC payment ( printed page 93914) system are available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/asc-regulations-and-notices.
Current Procedural Terminology (CPT) Copyright Notice
Throughout this final rule with comment period, we use CPT codes and descriptions to refer to a variety of services. We note that CPT codes and descriptions are copyright 2024 American Medical Association (AMA). All Rights Reserved. CPT is a registered trademark of the AMA. Applicable Federal Acquisition Regulations and Defense Federal Acquisition Regulations apply.
Table of Contents
I. Summary and Background
A. Executive Summary of This Document
B. Legislative and Regulatory Authority for the Hospital OPPS
C. Excluded OPPS Services and Hospitals
D. Prior Rulemaking
E. Advisory Panel on Hospital Outpatient Payment (the HOP Panel or the Panel)
F. Public Comments Received on the CY 2024 Proposed Rule
G. Public Comments Received on the CY 2024 OPPS/ASC Final Rule With Comment Period
II. Updates Affecting OPPS Payments
A. Recalibration of APC Relative Payment Weights
B. Conversion Factor Update
C. Wage Index Changes
D. Statewide Average Default Cost-to-Charge Ratios (CCRs)
E. Adjustment for Rural Sole Community Hospitals (SCHs) and Essential Access Community Hospitals (EACHs) Under Section 1833(t)(13)(B) of the Act for CY 2025
F. Payment Adjustment for Certain Cancer Hospitals for CY 2025
G. Hospital Outpatient Outlier Payments
H. Calculation of an Adjusted Medicare Payment From the National Unadjusted
Medicare Payment
I. Beneficiary Copayments
III. OPPS Ambulatory Payment Classification (APC) Group Policies
A. OPPS Treatment of New and Revised HCPCS Codes
B. OPPS Changes—Variations Within APCs
C. New Technology APCs
D. Universal Low Volume APC Policy for Clinical and Brachytherapy APCs
E. APC-Specific Policies
IV. OPPS Payment for Devices
A. Pass-Through Payment for Devices
B. Device-Intensive Procedures
V. OPPS Payment for Drugs, Biologicals, and Radiopharmaceuticals
A. OPPS Transitional Pass-Through Payment for Additional Costs of Drugs, Biologicals, and Radiopharmaceuticals
B. OPPS Payment for Drugs, Biologicals, and Radiopharmaceuticals Without Pass-Through Payment Status
VI. Estimate of OPPS Transitional Pass-Through Spending for Drugs, Biologicals, Radiopharmaceuticals, and Devices
A. Amount of Additional Payment and Limit on Aggregate Annual Adjustment
B. Final Estimate of Pass-Through Spending for CY 2025
VII. OPPS Payment for Hospital Outpatient Visits and Critical Care Services
VIII. Payment for Partial Hospitalization and Intensive Outpatient Services
A. Background
B. Coding and Billing for PHP and IOP Services Under the OPPS
C. CY 2025 Payment Rates for PHP and IOP
D. Outlier Policy for CMHCs
IX. Services That Will Be Paid Only as Inpatient Services
A. Background
B. Changes to the Inpatient Only (IPO) List
X. Nonrecurring Policy Changes
A. Remote Services
B. Virtual Direct Supervision of Cardiac Rehabilitation (CR), Intensive Cardiac Rehabilitation (ICR), Pulmonary Rehabilitation (PR) Services and Diagnostic Services Furnished to Hospital Outpatients
C. All-Inclusive Rate (AIR) Add-On Payment for High-Cost Drugs Provided by Indian Health Service and Tribal Facilities
D. Coverage Changes for Colorectal Cancer (CRC) Screening Services
E. Request for Comment on Payment Adjustments under the IPPS and OPPS for Domestic Personal Protective Equipment
F. Payment for HIV Pre-Exposure Prophylaxis (PrEP) in Hospital Outpatient Departments
G. Payment Policy for Devices in Category B Investigational Device Exemption (IDE) Clinical Trials Policy and Drugs/Devices With a Medicare Coverage With Evidence Development (CED) Designation
XI. CY 2025 OPPS Payment Status and Comment Indicators
A. CY 2025 OPPS Payment Status Indicator Definitions
B. CY 2025 Comment Indicator Definitions
XII. MedPAC Recommendations
A. OPPS Payment Rates Update
B. Medicare Safety Net Index
C. ASC Cost Data
XIII. Updates to the Ambulatory Surgical Center (ASC) Payment System
A. Background, Legislative History, Statutory Authority, and Prior Rulemaking for the ASC Payment System
B. ASC Treatment of New and Revised Codes
C. Payment Policies Under the ASC Payment System
D. Additions to ASC Covered Surgical Procedures and Covered Ancillary Services Lists
E. ASC Payment Policy for Non-Opioid Post-Surgery Pain Management Drugs, Biologicals, and Devices
F. Final CY 2025 Non-Opioid Policy for Pain Relief Under the OPPS and ASC Payment System
G. New Technology Intraocular Lenses (NTIOLs)
H. Calculation of the ASC Payment Rates and the ASC Conversion Factor
XIV. Cross-Program Measures for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs
A. Background
B. CMS Commitment To Advancing Health Equity Using Quality Measurement
C. Modification of the Immediate Measure Removal Policy for the Hospital Outpatient Quality Reporting (OQR) and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs Beginning With CY 2025
XV. Hospital Outpatient Quality Reporting (OQR) Program
A. Background and Statutory Authority
B. Program Measure Set Policies
C. Program Measure Updates
D. Administrative Requirements
E. Form, Manner, and Timing of Data Submission
F. Public Reporting of Measure Data
G. Payment Reduction for Hospitals That Fail To Meet the Hospital OQR Program Requirements for the CY 2025 Payment Determination
XVI. Rural Emergency Hospital Quality Reporting (REHQR) Program
A. Background and Statutory Authority
B. Program Measure Set Policies
C. Program Measure Updates
D. Administrative Requirements
E. Form, Manner, and Timing of Data Submission
F. Public Reporting of Measure Data
XVII. Ambulatory Surgical Center Quality Reporting (ASCQR) Program
A. Background and Statutory Authority
B. Program Measure Set Policies
C. Program Measure Updates
D. Administrative Requirements
E. Form, Manner, and Timing of Data Submission
F. Public Reporting of Measure Data
G. Request for Information (RFI)—Development of Frameworks for Specialty Focused Reporting and Minimum Case Number for Required Reporting
H. Payment Reduction for ASCs That Fail To Meet the ASCQR Program Requirements
XVIII. Medicaid Clinic Services Four Walls Exceptions
A. Background
B. Summary of the Medicaid Clinic Services Four Walls Exceptions Proposed Provisions, Public Comments and Responses to Comments
XIX. Changes to the Review Timeframes for the Hospital Outpatient Department (OPD) Prior Authorization Process
XX. Provisions Related to Medicaid and the Children's Health Insurance Program (CHIP)
A. Continuous Eligibility in Medicaid and CHIP (42 CFR 435.926 and 457.342)
B. Summary of the Proposed Provisions, Public Comments and Responses to Comments on Medicaid and CHIP Continuous Eligibility ( printed page 93915)
XXI. Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals
A. Background and Statutory Authority
B. The U.S. Maternal Health Crisis
C. Summary of the Proposed Provisions, Public Comments and Responses to Comments on Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals
XXII. Hospital-Wide All-Cause Risk Standardized Mortality Measures in the Hospital Inpatient Quality Reporting Program
A. Background
B. Updates to the Form, Time, and Manner Requirements for the Hybrid Hospital-Wide All-Cause Readmission (HWR) and Hybrid Hospital-Wide All-Cause Risk Standardized Mortality (HWM) Measures for the FY 2026 and the FY 2027 Payment Determinations
XXIII. Individuals Currently or Formerly in the Custody of Penal Authorities
A. Medicare FFS No Legal Obligation to Pay Payment Exclusion and Incarceration (revisions to 42 CFR 411.4)
B. Revision to Medicare Special Enrollment Period for Formerly Incarcerated Individuals
XXIV. Overall Hospital Quality Star Rating Modification To Emphasize the Safety of Care Measure Group: Request for Information (RFI)
A. Summary
B. Background
C. Current Overall Hospital Quality Star Rating Methodology
D. Safety of Care in Star Ratings
E. Potential Future Options To Greater Emphasize Patient Safety in the Overall Hospital Quality Star Rating
F. Solicitation of Public Comment
XXV. Files Available to the Public via the Internet
XXVI. Collection of Information Requirements
A. ICRs for the Hospital Outpatient Quality Reporting (OQR) Program
B. ICRs for the Rural Emergency Hospitals Quality Reporting (REHQR) Program
C. ICRs for the Ambulatory Surgical Center Quality Reporting (ASCQR) Program
D. ICRs Related to Medicaid Clinic Services Four Walls Exceptions
E. ICRs for Changes to the Review Timeframes for Hospital Outpatient Department (OPD) Prior Authorization Process
F. ICRs for the Hospital Inpatient Quality Reporting (IQR) Program
G. ICRs for Continuous Eligibility (42 CFR 435.926 and 457.342)
H. ICRs Regarding Organization, Staffing and Delivery of Services for Hospitals (§ 482.59(a) and (b)) and CAHs (§ 485.649(a) and (b))
I. ICRs Regarding OB Staff Training for Hospitals (§ 482.59(c) and CAHs (§ 485.649(c))
J. ICRs Regarding Revisions to QAPI (§ 482.21) Standards for OB Services
K. ICRS Regarding Emergency Services Readiness in Emergency Services (§ 482.55) for Hospitals
L. Transfer Protocols in Discharge Planning (§ 482.43) for Hospitals
M. Total Costs for all ICRs Related to Maternal Health
XXVII. Response to Comments
XXVIII. Economic Analyses
A. Statement of Need
B. Overall Impact of Provisions of This Final Rule With Comment Period
C. Detailed Economic Analyses
D. Regulatory Review Cost Estimation
E. Regulatory Flexibility Act (RFA) Analysis
F. Unfunded Mandates Reform Act Analysis
G. Federalism
H. Conclusion
I. Waiver Fiscal Responsibility Act Requirements
I. Summary and Background
A. Executive Summary of This Document
1. Purpose
We are updating the payment policies and payment rates for services furnished to Medicare beneficiaries in hospital outpatient departments (HOPDs) and ambulatory surgical centers (ASCs), beginning January 1, 2025. Section 1833(t) of the Social Security Act (the Act) requires us to annually review and update the payment rates for services payable under the Hospital Outpatient Prospective Payment System (OPPS). Specifically, section 1833(t)(9)(A) of the Act requires the Secretary of the Department of Health and Human Services (the Secretary) to review certain components of the OPPS not less often than annually, and to revise the groups, the relative payment weights, and the wage and other adjustments to take into account changes in medical practice, changes in technology, and the addition of new services, new cost data, and other relevant information and factors. In addition, under section 1833(i)(D)(v) of the Act, we annually review and update the ASC payment rates. This final rule with comment period also includes additional policy changes made in accordance with our experience with the OPPS and the ASC payment system and recent changes in our statutory authority. We describe these and various other statutory authorities in the relevant sections of this final rule with comment period. Also, this final rule with comment period updates the requirements for the Hospital Outpatient Quality Reporting (OQR) Program, the Rural Emergency Hospital Quality Reporting (REHQR) Program, the Ambulatory Surgical Center Quality Reporting (ASCQR) Program, and the Hospital Inpatient Quality Reporting (IQR) Program. We summarize information received in response to a Request for Information on potential modifications of the Safety of Care measure group in the Overall Hospital Quality Star Rating methodology. Given that the maternal health crisis in the United States is among the highest in high-income countries and also disproportionately impacts racial and ethnic minorities, we are finalizing updates to the CoPs for hospitals and CAHs in an effort to advance the health and safety of pregnant, birthing, and post-partum women. We are also finalizing an implementation approach in phases that allows additional time for hospitals and CAHs to comply with the requirements of the rule. In addition, we are narrowing the description of “custody” for the purposes of Medicare's no legal obligation to pay payment exclusion at § 411.4(b), providing an illustrative list of individuals who are not considered to be in custody, adding a definition of “penal authority,” reorganizing the regulation, and making certain technical edits. This rule also revises the eligibility requirements in the special enrollment period (SEP) for formerly incarcerated individuals at §§ 406.27(d) (Premium Part A) and 407.23(d) (Part B) to tie the eligibility for this SEP to the determination made by the Social Security Administration (SSA) that they are no longer incarcerated for releases beginning on January 1, 2025, and limit the current eligibility criteria for the SEP, with reference to “custody” associated with § 411.4(b) to releases between January 1, 2023, and December 31, 2024. Additionally, we are finalizing our proposed revisions to Medicaid and CHIP regulations to codify the requirement within the CAA, 2023 to require States to provide 12 months of continuous eligibility to children under the age of 19 in Medicaid and CHIP, with limited exceptions.
Finally, we are finalizing our proposed exceptions to the Medicaid clinic services benefit four walls requirement, to authorize Medicaid payment for clinic services provided outside the four walls of the clinic for IHS/Tribal clinics, and at State option, behavioral health clinics and clinics located in rural areas. Our current regulation at 42 CFR 440.90(b) includes an exception to the four walls requirement under the Medicaid clinic services benefit only for clinic services furnished to individuals who are unhoused. We believe finalizing these additional exceptions will help maintain and improve access for the populations served by IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas. ( printed page 93916)
2. Summary of the Major Provisions
- OPPS Update: For CY 2025, we are increasing the payment rates under the OPPS by an Outpatient Department (OPD) fee schedule increase factor of 2.9 percent. This increase factor is based on the final inpatient hospital market basket percentage increase of 3.4 percent for inpatient services paid under the hospital inpatient prospective payment system (IPPS) reduced by a final productivity adjustment of 0.5 percentage point. Based on this update, we estimate that total payments to OPPS providers (including beneficiary cost sharing and estimated changes in enrollment, utilization, and case mix) for calendar year (CY) 2025 will be approximately $87.7 billion, an increase of approximately $4.7 billion compared to estimated CY 2024 OPPS payments. We are continuing to implement the statutory 2.0 percentage point reduction in payments for hospitals that fail to meet the hospital outpatient quality reporting requirements by applying a reporting factor of 0.9806 to the OPPS payments and copayments for all applicable services.
- ASC Payment Update: For CYs 2019 through 2023, we adopted a policy to update the ASC payment system using the hospital market basket update. In light of the impact of the COVID-19 public health emergency (PHE) on healthcare utilization, we extended our policy to update the ASC payment system using the hospital market basket update an additional 2 years—through CYs 2024 and 2025. Using the hospital market basket methodology, for CY 2025, we increased payment rates under the ASC payment system by 2.9 percent for ASCs that meet the quality reporting requirements under the ASCQR Program. This increase is based on a final hospital market basket percentage increase of 3.4 percent reduced by a productivity adjustment of 0.5 percentage point. Based on this final update, we estimate that total payments to ASCs (including beneficiary cost sharing and estimated changes in enrollment, utilization, and case-mix) for CY 2025 will be approximately $7.4 billion, an increase of approximately $308 million compared to estimated CY 2024 Medicare payments.
- Device Pass-Through Payment Applications: For CY 2025, we received 14 complete applications for device pass-through payments. We sought public comment on these applications and make final determinations on these applications in this final rule with comment period.
- Changes to the List of ASC Covered Surgical Procedures and Ancillary Services Lists: For CY 2025, we are adding 21 medical and dental procedures to the ASC covered procedures list (CPL) and ancillary services lists, based upon existing criteria at § 416.166.
- Changes to the Inpatient Only (IPO) List: For CY 2025, we are finalizing adding three liver allograft services for which codes were newly created by the AMA CPT Editorial Panel for CY 2025 to the IPO list. Additionally, we are finalizing removing a pelvic fixation code (CPT code 22848) from the IPO list for CY 2025.
- Remote Services: For CY 2025, we are clarifying our policies for remotely furnished outpatient therapy services, Diabetes Self-Management Training and Medical Nutrition Therapy services and mental health services furnished remotely to beneficiaries in their homes by hospital staff to maintain alignment across payment systems.
- Payment for High-Cost Drugs Provided by Indian Health Service and Tribal Hospitals: For CY 2025, we are finalizing our policy to separately pay IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through an add-on payment in addition to the AIR under the authorities used to calculate the AIR.
- Clinical Trials Coding and Payment: We are finalizing a clarification to our Category B clinical trials coding and payment policy for devices and procedures to specify that our policy applies only to IDE studies with a control arm and where a payment adjustment is necessary to preserve the scientific validity of such a study. We are not finalizing our proposal to extend our coding and payment policy to drugs and devices that are being studied in clinical trials under a Coverage with Evidence Development (CED) National Coverage Determination (NCD),[1] for which the trial includes a treatment and control arm for CY 2025. We are taking additional time to consider the broad implications of a payment methodology for clinical trials for CED drugs and devices.
- Payment for HIV Pre-Exposure Prophylaxis (PrEP) in Hospital Outpatient Departments: For CY 2025, we are finalizing paying for HIV PrEP drugs covered as an additional preventive service and related services under the OPPS. We are finalizing a site neutral policy where products are generally paid similar rates under the OPPS and Physician Fee Schedule.
- Diagnostic Radiopharmaceuticals Separate Payment: We are finalizing a policy to pay separately for diagnostic radiopharmaceuticals with per day costs above a threshold of $630, which is approximately two times the volume weighted average cost amount currently associated with diagnostic radiopharmaceuticals in the Nuclear Medicine APCs. We also are finalizing updating the $630 threshold in CY 2026 and subsequent years by the Producer Price Index (PPI) for Pharmaceutical Preparations. Finally, we are finalizing payment for separately payable diagnostic radiopharmaceuticals based on their Mean Unit Cost (MUC) derived from OPPS claims for CY 2025.
- Exclusion of Cell and Gene Therapies from Comprehensive Ambulatory Payment Classification (C-APC) Packaging: We are finalizing a policy to exclude qualifying cell and gene therapies from C-APC packaging.
- Add-on Payment for Radiopharmaceutical Technetium-99m (Tc-99m) Derived from Domestically Produced Mo-99: For CY 2025, an add-on payment applies to radiopharmaceuticals that use Tc-99m produced without use of highly enriched uranium (HEU). For CY 2026, we are finalizing replacing the add-on payment for radiopharmaceuticals produced without the use of Tc-99m derived from non-HEU sources with an add-on payment for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99.
- Changes to the Review Timeframes for the Hospital Outpatient Department (OPD) Prior Authorization Process: We are changing the current review timeframe for prior authorization requests for OPD services from 10-business days to 7-calendar days for standard reviews.
- Health Equity Measures for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs: We are finalizing: (1) adoption of the Hospital Commitment to Health Equity (HCHE) measure in the Hospital OQR and the REHQR Programs, and adoption of the Facility Commitment to Health Equity (FCHE) measure in the ASCQR Program, beginning with the CY 2025 reporting period/CY 2027 payment or program determinations; (2) adoption of the Screening for Social Drivers of Health (SDOH) measure in all three programs beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 ( printed page 93917) payment or program determinations; and (3) adoption of the Screen Positive Rate for SDOH measure in all three programs beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determinations.
- Modification of the Immediate Measure Removal Policy for the Hospital OQR and ASCQR Programs: We are finalizing modification of the immediate measure removal policy to an immediate measure suspension policy to increase transparency regarding the process for removing adopted measures in the Hospital OQR and ASCQR Programs beginning with CY 2025.
- Hospital Outpatient Quality Reporting (OQR) Program: In addition to the cross-program measures and policies, we are finalizing: (1) adoption of the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM) beginning with voluntary reporting for the CY 2026 reporting period followed by mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination; (2) removal of the MRI Lumbar Spine for Low Back Pain measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (3) removal of the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (4) the requirement that electronic health record (EHR) technology be certified to all electronic clinical quality measures (eCQMs) available to report beginning with the CY 2025 reporting period/CY 2027 payment determination; and (5) public reporting of the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients measure—Psychiatric/Mental Health Patients stratification on Care Compare beginning with CY 2025.
- Rural Emergency Hospital Quality Reporting (REHQR) Program: In addition to the cross-program measures, we are finalizing: (1) extension of the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure from 1 year to 2 years beginning with the CY 2027 program determination; and (2) when, after status conversion, REHs are required to report data under the REHQR Program.
- Ambulatory Surgical Center Quality Reporting (ASCQR) Program: In addition to finalizing the cross-program measures and policies, we summarize comments received on the potential development of frameworks for specialty focused reporting and minimum case number for required reporting under the ASCQR Program.
- Hospital Inpatient Quality Reporting (IQR) Program: We are finalizing the voluntary reporting of the core clinical data elements (CCDEs) and linking variables for both the Hybrid Hospital-Wide Readmission (HWR) and Hybrid Hospital-Wide Standardized Mortality (HWM) measures, for the performance period of July 1, 2023, through June 30, 2024, impacting the FY 2026 payment determination for the Hospital IQR Program. Additionally, we are extending voluntary reporting of CCDEs and linking variables for an additional year for the performance period of July 1, 2024, through June 30, 2025, impacting the FY 2027 payment determination for the Hospital IQR Program.
- Overall Hospital Quality Star Rating: We summarize comments received on potential modifications to the Safety of Care measure group in the Overall Hospital Quality Star Rating methodology.
- Medicare FFS No Legal Obligation to Pay Payment Exclusion and Incarceration: We are narrowing the description of “custody” for purposes of Medicare's no legal obligation to pay payment exclusion at § 411.4(b), stating explicitly in regulatory text that individuals who are released to the community pending trial (including those in pretrial community supervision and those released pursuant to cash bail), on parole, probation, or home detention, or required to reside in halfway houses are not considered to be in “custody” for purposes of the payment exclusion, adding a definition of “penal authority,” reorganizing the regulation, and making certain technical edits.
- Revision to Medicare Special Enrollment Period for Formerly Incarcerated Individuals: We are finalizing the eligibility requirements in the special enrollment period (SEP) for formerly incarcerated individuals at §§ 406.27(d) and 407.23(d) to remove the reference to “custody” associated with § 411.4(b) and instead tie the eligibility for this SEP to the determination made by SSA that they are no longer incarcerated. We are also expanding the SEP eligibility criteria to include individuals released from confinement to residency in halfway houses.
- Continuous Eligibility in Medicaid and CHIP: We are finalizing our proposed revisions to Medicaid and CHIP regulations to codify the requirement within the CAA, 2023 for States to provide 12 months of continuous eligibility to children under the age of 19 in Medicaid and CHIP, with limited exceptions. Specifically, we are finalizing the removal of the option to provide continuous eligibility to a subgroup of Medicaid and CHIP enrollees and for a time period of less than 12 months. For CHIP, we are finalizing the removal of the option to disenroll children from CHIP during a continuous eligibility period for failure to pay premiums.
- Medicaid Clinic Services Four Walls Exceptions: We are finalizing three exceptions to the Medicaid clinic services benefit four walls requirement at 42 CFR 440.90. Our current regulation at 42 CFR 440.90(b) allows for Medicaid payment for clinic services furnished outside of the four walls of the clinic only to individuals who are unhoused. Our final rule adds a mandatory exception to the four walls requirement for IHS/Tribal clinics at 42 CFR 440.90(c) and optional exceptions for behavioral health clinics and clinics located in rural areas at 42 CFR 440.90(d) and (e), respectively.
- Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals: We are finalizing new Conditions of Participation (CoPs) for hospitals and CAHs for obstetrical services, including new requirements for maternal quality assessment and performance improvement (QAPI), as well as baseline standards for the organization, staffing, and delivery of care within obstetrical units, and staff training on evidence-based best practices every 2 years. We are finalizing revisions to the emergency services CoP related to emergency readiness for hospitals and CAHs that provide emergency services. In addition, we are finalizing revisions to the Discharge Planning CoP for all hospitals related to transfer protocols. Further, we are finalizing a phased-in implementation to provide hospitals and CAHs with additional time to come into compliance with the requirements. Lastly, we sought comments in the proposed rule on whether these requirements should also apply to rural emergency hospitals (REHs), which we discuss below.
3. Summary of Costs and Benefits
In section XXVIII of this final rule with comment period, we set forth a detailed analysis of the regulatory and federalism impacts that the proposed ( printed page 93918) changes would have on affected entities and beneficiaries. Key estimated impacts are described below.
a. Impacts of All OPPS Changes
Table 201 in section XXVIII.C of this final rule with comment period displays the distributional impact of all the OPPS changes on various groups of hospitals and CMHCs for CY 2025 compared to all estimated OPPS payments in CY 2024. We estimate that the final policies in this final rule with comment period would result in a 3.0 percent overall increase in OPPS payments to providers. We estimate that total OPPS payments for CY 2025, including beneficiary cost-sharing, to the approximately 3,500 facilities paid under the OPPS (including general acute care hospitals, children's hospitals, cancer hospitals, and CMHCs) would increase by approximately $1.98 billion compared to CY 2024 payments, excluding our estimated changes in enrollment, utilization, and case-mix.
We estimated the isolated impact of our OPPS policies on CMHCs because CMHCs have historically only been paid for partial hospitalization services under the OPPS. Beginning CY 2024, they are also paid for intensive outpatient program (IOP) services under the OPPS. Continuing the provider-specific structure we adopted beginning in CY 2011, and basing payment fully on the type of provider furnishing the service, we estimate a 11.9 percent increase in CY 2025 payments to CMHCs relative to their CY 2024 payments.
b. Impacts of the Updated Wage Indexes
We estimate that our update of the wage indexes based on the fiscal year (FY) 2025 IPPS final rule wage indexes will result in a 0.1 percent increase for urban hospitals under the OPPS and a 0.9 percent increase for rural hospitals. These wage indexes include continued implementation of the Office of Management and Budget (OMB) labor market area delineations based on 2020 Decennial Census data, with updates, as discussed in section II.C of this final rule with comment period.
c. Impacts of the Rural Adjustment and the Cancer Hospital Payment Adjustment
For CY 2025, we are continuing to provide additional payments to cancer hospitals so that a cancer hospital's payment-to-cost ratio (PCR) after the additional payments is equal to the weighted average PCR for the other OPPS hospitals using the most recently submitted or settled cost report data. Section 16002(b) of the 21st Century Cures Act requires that this weighted average PCR be reduced by 1.0 percentage point. In light of the COVID-19 PHE impact on claims and cost data used to calculate the target PCR, we had maintained the CY 2021 target PCR of 0.89 through CYs 2022 and 2023. However, in CY 2024, we finalized a policy to reduce the target PCR by 1.0 percentage point each calendar year until the target PCR equals the PCR of non-cancer hospitals using the most recently submitted or settled cost report data. For CY 2024, we finalized a target PCR of 0.88. For CY 2025, we are finalizing a target PCR of 0.87 to determine the CY 2025 cancer hospital payment adjustment to be paid at cost report settlement. That is, the payment adjustments would be the additional payments needed to result in a PCR equal to 0.87 for each cancer hospital.
d. Impacts of the OPD Fee Schedule Increase Factor
For the CY 2025 OPPS/ASC, we are establishing an OPD fee schedule increase factor of 2.9 percent and applying that increase factor to the conversion factor for CY 2025. As a result of the OPD fee schedule increase factor and other budget neutrality adjustments, we estimate that urban hospitals will experience an increase in payments of approximately 3.2 percent and that rural hospitals will experience an increase in payments of 3.2 percent. Classifying hospitals by teaching status, we estimate non-teaching hospitals will experience an increase in payments of 3.3 percent, minor teaching hospitals will experience an increase in payments of 3.5 percent, and major teaching hospitals will experience an increase in payments of 2.7 percent. We also classified hospitals by the type of ownership. We estimate that hospitals with voluntary ownership will experience an increase of 3.1 percent in payments, while hospitals with government ownership will experience an increase of 2.6 percent in payments. We estimate that hospitals with proprietary ownership will experience an increase of 4.9 percent in payments.
e. Impacts of the ASC Payment Update
For impact purposes, the surgical procedures on the ASC covered surgical procedure list are aggregated into surgical specialty groups using CPT and HCPCS code range definitions. The percentage change in estimated total payments by specialty groups under the CY 2025 payment rates, compared to estimated CY 2024 payment rates, generally ranges between an increase of 3 percent and an increase of 4 percent, depending on the service, with some exceptions. We estimate the impact of applying the inpatient hospital market basket update to ASC payment rates will increase payments by $193 million under the ASC payment system in CY 2025.
f. Impacts of Medicaid Clinic Services Four Walls Exceptions
We estimate that the finalized exceptions to the four walls requirement under the Medicaid clinic services benefit for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas will increase total expenditures by $1.18 billion from FY 2025 through 2029. Our estimate includes a Federal impact of $1.15 billion and impact to States of $30 million. These estimates are discussed in more detail in section XXVIII of this final rule.
g. Impacts of Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals
We are finalizing maternal health focused revisions to the CoPs for hospitals and CAHs, which are estimated to increase burden on hospitals and CAHs by $423 million annually with total costs estimated at $4.23 billion over 10 years. We expect an average annual cost of $67,103 per hospital and CAH. As discussed in detail in section XXVIII of this final rule with comment period, we expect the benefits of these policies to include reduced maternal morbidity and mortality, leading to financial benefits for patients, their families, and payors. We also expect that the policies are likely to reduce inequality in maternal health outcomes among pregnant and postpartum women from different groups and lead to overall improvements in patient care.
h. Impacts of the Medicaid and CHIP Continuous Eligibility Requirements
We are finalizing our proposed revisions to Medicaid and CHIP regulations to codify the requirement within the CAA, 2023 for States to provide 12 months of continuous eligibility to children under the age of 19 in Medicaid and CHIP, with limited exceptions. For CHIP, we are finalizing the removal of the option to disenroll children from CHIP during a continuous eligibility period for failure to pay premiums. Requiring 12months of continuous eligibility in Medicaid and CHIP is estimated to increase annual average enrollment in Medicaid and CHIP by approximately 124,000 by 2028 (75,000 in Medicaid and 49,000 in CHIP). The total estimated impact of this requirement over 5 years is $2,466 million, including Federal impact of ( printed page 93919) $1,592 million and State impact of $874 million.
B. Legislative and Regulatory Authority for the Hospital OPPS
When Title XVIII of the Act was enacted, Medicare payment for hospital outpatient services was based on hospital-specific costs. In an effort to ensure that Medicare and its beneficiaries pay appropriately for services and to encourage more efficient delivery of care, the Congress mandated replacement of the reasonable cost-based payment methodology with a prospective payment system (PPS). The Balanced Budget Act of 1997 (BBA) (Pub. L. 105-33) added section 1833(t) to the Act, authorizing implementation of a PPS for hospital outpatient services. The OPPS was first implemented for services furnished on or after August 1, 2000. Implementing regulations for the OPPS are located at 42 CFR parts 410 and 419.
The Medicare, Medicaid, and SCHIP Balanced Budget Refinement Act of 1999 (BBRA) (Pub. L. 106-113) made major changes in the hospital OPPS. The following Acts made additional changes to the OPPS: the Medicare, Medicaid, and SCHIP Benefits Improvement and Protection Act of 2000 (BIPA) (Pub. L. 106-554); the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) (Pub. L. 108-173); the Deficit Reduction Act of 2005 (DRA) (Pub. L. 109-171), enacted on February 8, 2006; the Medicare Improvements and Extension Act under Division B of Title I of the Tax Relief and Health Care Act of 2006 (MIEA-TRHCA) (Pub. L. 109-432), enacted on December 20, 2006; the Medicare, Medicaid, and SCHIP Extension Act of 2007 (MMSEA) (Pub. L. 110-173), enacted on December 29, 2007; the Medicare Improvements for Patients and Providers Act of 2008 (MIPPA) (Pub. L. 110-275), enacted on July 15, 2008; the Patient Protection and Affordable Care Act (Pub. L. 111-148), enacted on March 23, 2010, as amended by the Health Care and Education Reconciliation Act of 2010 (HCERA, Pub. L. 111-152), enacted on March 30, 2010 (these two public laws are collectively known as the Affordable Care Act); the Medicare and Medicaid Extenders Act of 2010 (MMEA, Pub. L. 111-309); the Temporary Payroll Tax Cut Continuation Act of 2011 (TPTCCA, Pub. L. 112-78), enacted on December 23, 2011; the Middle Class Tax Relief and Job Creation Act of 2012 (MCTRJCA, Pub. L. 112-96), enacted on February 22, 2012; the American Taxpayer Relief Act of 2012 (Pub. L. 112-240), enacted January 2, 2013; the Pathway for SGR Reform Act of 2013 (Pub. L. 113-67) enacted on December 26, 2013; the Protecting Access to Medicare Act of 2014 (PAMA, Pub. L. 113-93), enacted on March 27, 2014; the Medicare Access and CHIP Reauthorization Act (MACRA) of 2015 (Pub. L. 114-10), enacted April 16, 2015; the Bipartisan Budget Act of 2015 (Pub. L. 114-74), enacted November 2, 2015; the Consolidated Appropriations Act, 2016 (Pub. L. 114-113), enacted on December 18, 2015, the 21st Century Cures Act (Pub. L. 114-255), enacted on December 13, 2016; the Consolidated Appropriations Act, 2018 (Pub. L. 115-141), enacted on March 23, 2018; the Substance Use Disorder—Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities Act (Pub. L. 115-271), enacted on October 24, 2018; the Further Consolidated Appropriations Act, 2020 (Pub. L. 116-94), enacted on December 20, 2019; the Coronavirus Aid, Relief, and Economic Security Act (Pub. L. 116-136), enacted on March 27, 2020; the Consolidated Appropriations Act, 2021 (Pub. L. 116-260), enacted on December 27, 2020; the Inflation Reduction Act, 2022 (Pub. L. 117-169), enacted on August 16, 2022; and the Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-238), enacted December 29, 2022.
Under the OPPS, we generally pay for hospital Part B services on a rate-per-service basis that varies according to the APC group to which the service is assigned. We use the Healthcare Common Procedure Coding System (HCPCS) (which includes certain Current Procedural Terminology (CPT) codes) to identify and group the services within each APC. The OPPS includes payment for most hospital outpatient services, except those identified in section I.C of this final rule with comment period. Section 1833(t)(1)(B) of the Act provides for payment under the OPPS for hospital outpatient services designated by the Secretary (which includes partial hospitalization services furnished by CMHCs), and certain inpatient hospital services that are paid under Medicare Part B.
The OPPS rate is an unadjusted national payment amount that includes the Medicare payment and the beneficiary copayment. This rate is divided into a labor-related amount and a nonlabor-related amount. The labor-related amount is adjusted for area wage differences using the hospital inpatient wage index value for the locality in which the hospital or CMHC is located.
All services and items within an APC group are comparable clinically and with respect to resource use, as required by section 1833(t)(2)(B) of the Act. In accordance with section 1833(t)(2)(B) of the Act, subject to certain exceptions, items and services within an APC group cannot be considered comparable with respect to the use of resources if the highest median cost (or mean cost, if elected by the Secretary) for an item or service in the APC group is more than 2 times greater than the lowest median cost (or mean cost, if elected by the Secretary) for an item or service within the same APC group (referred to as the “2 times rule”). In implementing this provision, we generally use the cost of the item or service assigned to an APC group.
For new technology items and services, special payments under the OPPS may be made in one of two ways. Section 1833(t)(6) of the Act provides for temporary additional payments, which we refer to as “transitional pass-through payments,” for at least 2 but not more than 3 years for certain drugs, biological agents, brachytherapy devices used for the treatment of cancer, and categories of other medical devices. For new technology services that are not eligible for transitional pass-through payments, and for which we lack sufficient clinical information and cost data to appropriately assign them to a clinical APC group, we have established special APC groups based on costs, which we refer to as New Technology APCs. These New Technology APCs are designated by cost bands which allow us to provide appropriate and consistent payment for designated new procedures that are not yet reflected in our claims data. Similar to pass-through payments, an assignment to a New Technology APC is temporary; that is, we retain a service within a New Technology APC until we acquire sufficient data to assign it to a clinically appropriate APC group.
C. Excluded OPPS Services and Hospitals
Section 1833(t)(1)(B)(i) of the Act authorizes the Secretary to designate the hospital outpatient services that are paid under the OPPS. While most hospital outpatient services are payable under the OPPS, section 1833(t)(1)(B)(iv) of the Act excludes payment for ambulance, physical and occupational therapy, and speech-language pathology services, for which payment is made under a fee schedule. It also excludes screening mammography, diagnostic mammography, and effective January 1, 2011, an annual wellness visit providing personalized prevention plan services. The Secretary exercises the authority granted under the statute to also exclude from the OPPS certain services that are ( printed page 93920) paid under fee schedules or other payment systems. Such excluded services include, for example, the professional services of physicians and nonphysician practitioners paid under the Medicare Physician Fee Schedule (MPFS); certain laboratory services paid under the Clinical Laboratory Fee Schedule (CLFS); services for beneficiaries with end-stage renal disease (ESRD) that are paid under the ESRD prospective payment system; and services and procedures that require an inpatient stay that are paid under the hospital IPPS. In addition, section 1833(t)(1)(B)(v) of the Act does not include applicable items and services (as defined in subparagraph (A) of paragraph (21)) that are furnished on or after January 1, 2017, by an off-campus outpatient department of a provider (as defined in subparagraph (B) of paragraph (21)). We set forth the services that are excluded from payment under the OPPS in regulations at 42 CFR 419.22.
Under § 419.20(b) of the regulations, we specify the types of hospitals that are excluded from payment under the OPPS. These excluded hospitals are:
- Critical access hospitals (CAHs);
- Hospitals located in Maryland and paid under Maryland's All-Payer or Total Cost of Care Model;
- Hospitals located outside of the 50 States, the District of Columbia, and Puerto Rico;
- Indian Health Service (IHS) hospitals; and
- Rural emergency hospitals (REH).
D. Prior Rulemaking
On April 7, 2000, we published in the Federal Register a final rule with comment period (65 FR 18434) to implement a prospective payment system for hospital outpatient services. The hospital OPPS was first implemented for services furnished on or after August 1, 2000. Section 1833(t)(9)(A) of the Act requires the Secretary to review certain components of the OPPS, not less often than annually, and to revise the groups, the relative payment weights, and the wage and other adjustments to take into account changes in medical practices, changes in technology, the addition of new services, new cost data, and other relevant information and factors.
Since initially implementing the OPPS, we have published final rules in the Federal Register annually to implement statutory requirements and changes arising from our continuing experience with this system. These rules can be viewed on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
E. Advisory Panel on Hospital Outpatient Payment (the HOP Panel or the Panel)
1. Authority of the Panel
Section 1833(t)(9)(A) of the Act, as amended by section 201(h) of Public Law 106-113, and redesignated by section 202(a)(2) of Public Law 106-113, requires that we consult with an expert outside advisory panel composed of an appropriate selection of representatives of providers to annually review (and advise the Secretary concerning) the clinical integrity of the payment groups and their weights under the OPPS. In CY 2000, based on section 1833(t)(9)(A) of the Act, the Secretary established the Advisory Panel on Ambulatory Payment Classification Groups (APC Panel) to fulfill this requirement. In CY 2011, based on section 222 of the Public Health Service Act (the PHS Act), which gives discretionary authority to the Secretary to convene advisory councils and committees, the Secretary expanded the panel's scope to include the supervision of hospital outpatient therapeutic services in addition to the APC groups and weights. To reflect this new role of the panel, the Secretary changed the panel's name to the Advisory Panel on Hospital Outpatient Payment (the HOP Panel). The HOP Panel is not restricted to using data compiled by CMS, and in conducting its review, it may use data collected or developed by organizations outside the Department.
2. Establishment of the Panel
On November 21, 2000, the Secretary signed the initial charter establishing the Panel, and, at that time, named the APC Panel. This expert panel is composed of appropriate representatives of providers (currently employed full-time, not as consultants, in their respective areas of expertise) who review clinical data and advise CMS about the clinical integrity of the APC groups and their payment weights. Since CY 2012, the Panel also is charged with advising the Secretary on the appropriate level of supervision for individual hospital outpatient therapeutic services. The Panel is technical in nature, and it is governed by the provisions of the Federal Advisory Committee Act (FACA). The current charter specifies, among other requirements, that the Panel—
- May advise on the clinical integrity of Ambulatory Payment Classification (APC) groups and their associated weights;
- May advise on the appropriate supervision level for hospital outpatient services;
- May advise on OPPS APC rates for ASC covered surgical procedures;
- Continues to be technical in nature;
- Is governed by the provisions of the FACA;
- Has a Designated Federal Official (DFO); and
- Is chaired by a Federal Official designated by the Secretary.
The Panel's charter was amended on November 15, 2011, renaming the Panel and expanding the Panel's authority to include supervision of hospital outpatient therapeutic services and to add critical access hospital (CAH) representation to its membership. The Panel's charter was also amended on November 6, 2014 (80 FR 23009), and the number of members was revised from up to 19 to up to 15 members. The Panel's current charter was approved on November 21, 2022, for a 2-year period.
The current Panel membership and other information pertaining to the Panel, including its charter, Federal Register notices, membership, meeting dates, agenda topics, and meeting reports, can be viewed on the CMS website at: https://www.cms.gov/Regulations-and-Guidance/Guidance/FACA/AdvisoryPanelonAmbulatoryPaymentClassificationGroups.html.
3. Panel Meetings and Organizational Structure
The Panel has held many meetings, with the last meeting taking place on August 26, 2024. The recommendations of the Panel for the most recent meeting are available on the CMS website at: https://www.cms.gov/medicare/regulations-guidance/advisory-committees/hospital-outpatient-payment. Prior to each meeting, we publish a notice in the Federal Register to announce the meeting, new members, and any other changes of which the public should be aware. Beginning in CY 2017, we have transitioned to one meeting per year (81 FR 31941). In CY 2022, we published a Federal Register notice requesting nominations to fill vacancies on the Panel (87 FR 68499). CMS is currently accepting nominations at: https://mearis.cms.gov.
In addition, the Panel has established an administrative structure that, in part, currently includes the use of three subcommittee workgroups to provide preparatory meeting and subject support to the larger panel. The three current subcommittees include the following:
- APC Groups and Status Indicator Assignments Subcommittee, which advises and provides recommendations to the Panel on the appropriate status ( printed page 93921) indicators to be assigned to HCPCS codes, including but not limited to whether a HCPCS code or a category of codes should be packaged or separately paid, as well as the appropriate APC assignment of HCPCS codes regarding services for which separate payment is made;
- Data Subcommittee, which is responsible for studying the data issues confronting the Panel and for recommending options for resolving them; and
- Visits and Observation Subcommittee, which reviews and makes recommendations to the Panel on all technical issues pertaining to observation services and hospital outpatient visits paid under the OPPS.
Each of these workgroup subcommittees was established by a majority vote from the full Panel during a scheduled Panel meeting, and the Panel recommended at the August 21, 2023, meeting that the subcommittees continue. We accepted this recommendation.
For discussions of earlier Panel meetings and recommendations, we refer readers to previously published OPPS/ASC proposed and final rules, the CMS website mentioned earlier in this section, and the FACA database at https://facadatabase.gov.
F. Public Comments Received on the CY 2025 Proposed Rule
We received approximately 3,500 timely pieces of correspondence on the CY 2025 OPPS/ASC proposed rule that appeared in the Federal Register on July 22, 2024 (89 FR 59186). We received comments from elected officials, providers and suppliers, practitioners, and advocacy groups. We provide summaries of the public comments, and our responses are set forth in the various sections of this final rule with comment period under the appropriate headings. We note that we received some public comments that were outside the scope of the CY 2025 OPPS/ASC proposed rule. Out-of-scope-public comments are not addressed in this CY 2025 OPPS/ASC final rule with comment period.
G. Public Comments Received on the CY 2024 OPPS/ASC Final Rule With Comment Period
We received approximately 180 timely pieces of correspondence on the CY 2024 OPPS/ASC final rule with comment period that appeared in the Federal Register on November 22, 2023 (88 FR 81540).
II. Updates Affecting OPPS Payments
A. Recalibration of APC Relative Payment Weights
1. Database Construction
a. Database Source and Methodology
Section 1833(t)(9)(A) of the Act requires that the Secretary review not less often than annually and revise the relative payment weights for Ambulatory Payment Classifications (APCs). In the April 7, 2000, OPPS final rule with comment period (65 FR 18482), we explained in detail how we calculated the relative payment weights that were implemented on August 1, 2000, for each APC group.
For the CY 2025 OPPS, we proposed to recalibrate the APC relative payment weights for services furnished on or after January 1, 2025, and before January 1, 2026 (CY 2025), using the same basic methodology that we described in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81549 through 81552), using CY 2023 claims data. That is, we proposed to recalibrate the relative payment weights for each APC based on claims and cost report data for hospital outpatient department (HOPD) services to construct a database for calculating APC group weights.
For the purpose of recalibrating the proposed APC relative payment weights for CY 2025, we began with approximately 145 million final action claims (claims for which all disputes and adjustments have been resolved and payment has been made) for HOPD services furnished on or after January 1, 2023, and before January 1, 2024, before applying our exclusionary criteria and other methodological adjustments. After the application of those data processing changes, we used approximately 73 million final action claims to develop the proposed CY 2025 OPPS payment weights. For exact numbers of claims used and additional details on the claims accounting process, we refer readers to the claims accounting narrative under supporting documentation for the CY 2025 OPPS/ASC proposed rule on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Addendum N to the CY 2025 OPPS/ASC proposed rule (which is available via the internet on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices) includes the proposed list of bypass codes for CY 2025. The proposed list of bypass codes contains codes that are reported on claims for services in CY 2023 and, therefore, includes codes that were in effect in CY 2023 and used for billing. We proposed to retain these deleted bypass codes on the proposed CY 2025 bypass list because these codes existed in CY 2023 and were covered OPD services in that period, and CY 2023 claims data were used to calculate proposed CY 2025 payment rates. Keeping these deleted bypass codes on the bypass list potentially allows us to create more “pseudo” single procedure claims for ratesetting purposes. “Overlap bypass codes” that are members of the proposed multiple imaging composite APCs are identified by asterisks (*) in the third column of Addendum N to the CY 2025 OPPS/ASC proposed rule. HCPCS codes that we proposed to add for CY 2025 are identified by asterisks (*) in the fourth column of Addendum N.
We did not receive any public comments on our general proposal to recalibrate the relative payment weights for each APC based on claims and cost report data for HOPD services or on our proposed bypass code process. We are finalizing as proposed the “pseudo” single claims process and the final CY 2025 list of bypass codes, as displayed in Addendum N to this final rule with comment period (which is available via the internet on the CMS website). For this final rule with comment period, for the purpose of recalibrating the final APC relative payment weights for CY 2025, we used approximately 78 million final action claims (claims for which all disputes and adjustments have been resolved and payment has been made) for HOPD services furnished on or after January 1, 2023, and before January 1, 2024. For the exact numbers of claims used and additional details on the claims accounting process, we refer readers to the claims accounting narrative under supporting documentation for this final rule with comment period on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
b. Calculation and Use of Cost-to-Charge Ratios (CCRs)
For CY 2025, we proposed to continue to use the hospital-specific overall ancillary and departmental cost-to-charge ratios (CCRs) to convert charges to estimated costs through application of a revenue code-to-cost center crosswalk. To calculate the APC costs on which the proposed CY 2025 APC payment rates are based, we calculated hospital-specific departmental CCRs for each hospital for which we had CY 2023 claims data by comparing these claims data to the most recently available hospital cost reports, which, in most ( printed page 93922) cases, are from CY 2022. For the proposed CY 2025 OPPS payment rates, we used the set of claims processed during CY 2023. We applied the hospital-specific CCR to the hospital's charges at the most detailed level possible, based on a revenue code-to-cost center crosswalk that contains a hierarchy of CCRs used to estimate costs from charges for each revenue code. To ensure the completeness of the revenue code-to-cost center crosswalk, we reviewed changes to the list of revenue codes for CY 2023 (the year of claims data we used to calculate the proposed CY 2025 OPPS payment rates) and updates to the National Uniform Billing Committee (NUBC) 2023 Data specifications Manual. That crosswalk is available for review and continuous comment on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
In accordance with our longstanding policy, similar to our finalized policy for CY 2024 OPPS ratesetting, we proposed to calculate CCRs for the standard cost centers—cost centers with a predefined label—and nonstandard cost centers—cost centers defined by a hospital—accepted by the electronic cost report database. In general, the most detailed level at which we calculate CCRs is the hospital-specific departmental level.
While we generally view the use of additional cost data as improving our OPPS ratesetting process, we have historically not included cost report lines for certain nonstandard cost centers in the OPPS ratesetting database construction when hospitals have reported these nonstandard cost centers on cost report lines that do not correspond to the cost center number. We believe it is important to further investigate the accuracy of these cost report data before including such data in the ratesetting process. Further, we believe it is appropriate to gather additional information from the public as well before including them in OPPS ratesetting. For CY 2025, we proposed not to include the nonstandard cost centers reported in this way in the OPPS ratesetting database construction.
We did not receive any public comments on the general CCR process and therefore, we are finalizing our proposal for CY 2025 to use the hospital-specific overall ancillary and departmental CRs to convert charges to estimated costs through application of a revenue code-to-cost center crosswalk and the proposed methodology.
2. Final Data Development and Calculation of Costs Used for Ratesetting
In this section of this final rule with comment period, we discuss the use of claims to calculate the OPPS payment rates for CY 2025. The Hospital OPPS page on the CMS website on which this final rule with comment period is posted ( https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient) provides an accounting of claims used in the development of the proposed payment rates. That accounting provides additional detail regarding the number of claims derived at each stage of the process. In addition, later in this section we discuss the file of claims that comprises the data set that is available upon payment of an administrative fee under a CMS data use agreement. The CMS website, https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient, includes information about obtaining the “OPPS Limited Data Set,” which now includes the additional variables previously available only in the OPPS Identifiable Data Set, including International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) diagnosis codes and revenue code payment amounts. This file is derived from the CY 2023 claims that are used to calculate the proposed payment rates for the CY 2025 OPPS/ASC proposed rule.
Previously, the OPPS established the scaled relative weights on which payments are based using APC median costs, a process described in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74188). However, as discussed in more detail in section II.A.2.f of the CY 2013 OPPS/ASC final rule with comment period (77 FR 68259 through 68271), we finalized the use of geometric mean costs to calculate the relative weights on which the CY 2013 OPPS payment rates were based. While this policy changed the cost metric on which the relative payments are based, the data process in general remained the same under the methodologies that we used to obtain appropriate claims data and accurate cost information in determining estimated service cost.
We used the methodology described in sections II.A.2.a through II.A.2.c of this final rule with comment period to calculate the costs we used to establish the proposed relative payment weights used in calculating the OPPS payment rates for CY 2025 shown in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices). We refer readers to section II.A.4 of this final rule with comment period for a discussion of the conversion of APC costs to scaled payment weights.
We note that under the OPPS, CY 2019 was the first year in which the claims data used for setting payment rates (CY 2017 data) contained lines with the modifier “PN,” which indicates nonexcepted items and services furnished and billed by off-campus provider-based departments (PBDs) of hospitals. Because nonexcepted items and services are not paid under the OPPS, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58832), we finalized a policy to remove those claim lines reported with modifier “PN” from the claims data used in ratesetting for the CY 2019 OPPS and subsequent years. For the CY 2025 OPPS, we proposed to continue to remove claim lines with modifier “PN” from the ratesetting process.
We did not receive any public comments on our proposal and are finalizing our proposal to continue to remove claim lines reported with modifier “PN” from the ratesetting process.
For details of the claims accounting process used in this CY 2025 OPPS/ASC final rule with comment period, we refer readers to the claims accounting narrative under supporting documentation for this final rule with comment period on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
a. Calculation of Single Procedure APC Criteria-Based Costs
(1) Blood and Blood Products
(a) Methodology
Since the implementation of the OPPS in August 2000, we have made separate payments for blood and blood products through APCs rather than packaging payment for them into payments for the procedures with which they are administered. Hospital payments for the costs of blood and blood products, as well as for the costs of collecting, processing, and storing blood and blood products, are made through the OPPS payments for specific blood product APCs.
In the CY 2025 OPPS/ASC proposed rule, we proposed to continue to establish payment rates for blood and blood products using our blood-specific CCR methodology (88 FR 49562), which utilizes actual or simulated CCRs from ( printed page 93923) the most recently available hospital cost reports to convert hospital charges for blood and blood products to costs. This methodology has been our standard ratesetting methodology for blood and blood products since CY 2005. It was developed in response to data analysis indicating that there was a significant difference in CCRs for those hospitals with and without blood-specific cost centers and past public comments indicating that the former OPPS policy of defaulting to the overall hospital CCR for hospitals not reporting a blood-specific cost center often resulted in an underestimation of the true hospital costs for blood and blood products. To address the differences in CCRs and to better reflect hospitals' costs, our methodology simulates blood CCRs for each hospital that does not report a blood cost center by calculating the ratio of the blood-specific CCRs to hospitals' overall CCRs for those hospitals that do report costs and charges for blood cost centers and applies this mean ratio to the overall CCRs of hospitals not reporting costs and charges for blood cost centers on their cost reports. We propose to calculate the costs upon which the proposed payment rates for blood and blood products are based using the actual blood-specific CCR for hospitals that reported costs and charges for a blood cost center and a hospital-specific, simulated, blood-specific CCR for hospitals that did not report costs and charges for a blood cost center.
We stated in the CY 2025 OPPS/ASC proposed rule that we continue to believe that the hospital-specific, simulated, blood-specific CCR methodology takes into account the unique charging and cost accounting structure of each hospital, as it better responds to the absence of a blood-specific CCR for a hospital than alternative methodologies, such as defaulting to the overall hospital CCR or applying an average blood-specific CCR across hospitals. This methodology also yields more accurate estimated costs for these products and results in payment rates for blood and blood products that appropriately reflect the relative estimated costs of these products for hospitals without blood cost centers and for these blood products in general.
We refer readers to Addendum B to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices) for the final CY 2025 payment rates for blood and blood products (which are generally identified with status indicator “R”).
For a more detailed discussion of payments for blood and blood products through APCs, we refer readers to:
- the CY 2005 OPPS proposed rule (69 FR 50524 and 50525) for a more comprehensive discussion of the blood-specific CCR methodology;
- the CY 2008 OPPS/ASC final rule with comment period (72 FR 66807 through 66810) for a detailed history of the OPPS payment for blood and blood products; and
- the CY 2015 OPPS/ASC final rule with comment period (79 FR 66795 and 66796) for additional discussion of our policy not to make separate payments for blood and blood products when they appear on the same claims as services assigned to a C-APC.
Comment: We received one comment regarding the cost calculations for blood and blood products. The commenter stated that current payment rates do not always cover the cost to process, store, and transport the unit of blood. The commenter recommended that CMS utilize the average wholesale price (AWP) as the payment rate for blood products, in addition to basing payment rates on the hospital CCRs, as they believed using AWP would help eliminate outliers and produce more accurate payment calculations. The commenter explained that cost reporting methodologies may vary among hospitals. For example, some cost centers may combine blood products with blood bank testing which could skew the CCRs for that cost center, which would then impact the payment rates.
Response: We thank the commenter for their input and will keep it in mind for future rulemaking. However, we note that reflecting the range and variation of billing patterns across hospitals, as reflected in their claims, is part of establishing prospective payment rates. Additionally, we do not believe at this time that using AWP to set the payment rate for blood products would necessarily result in more accurate payment calculations, because AWP does not factor in volume discounts or rebates and, therefore, may not reflect the true cost of the product.
Comment: One commenter supported our proposal to continue to pay separately for blood and blood products.
Response: We thank the commenter for their support.
After consideration of the public comments we received, we are finalizing our proposals for blood and blood products using our blood-specific CCR methodology without modification. Please refer to Addendum B to this final rule with comment period, which is available on the CMS website, for the final CY 2025 payment rates for blood and blood products.
(b) New HCPCS Code for Red Blood Cell Product (HCPCS Code P9027)
Effective October 1, 2024, the HCPCS workgroup created HCPCS code P9027 (Red blood cells, leukocytes reduced, oxygen/carbon dioxide reduced, each unit). HCPCS code P9027 may be used to bill for a blood product that utilizes a two-stage process to remove oxygen and maintain a deoxygenated state throughout its storage period, thereby preventing oxidative damage to red blood cells. We note that because HCPCS code P9027 was created after the CY 2025 OPPS/ASC proposed rule was issued, we did not include a proposal regarding the APC assignment for HCPCS code P9027 in the CY 2025 OPPS/ASC proposed rule. Due to its effective date, we assigned HCPCS code P9027 to APC 9541 with an SI = R (Blood and Blood Products) in Change Request 13784. The payment rate for HCPCS code P9027, effective October 1, 2024, is $252.48.
While we usually calculate payment rates for blood and blood products using our blood-specific CCR methodology, when there is no claims data on the charges and costs for blood products upon which to apply our blood-specific CCR methodology, we establish a payment rate for the new blood product based on a crosswalk to an existing blood product HCPCS code that we believe provides the best proxy for the costs of the new blood product. Because we did not have any claims data on HCPCS code P9027 ahead of its October 1, 2024, effective date, we used a crosswalk code to assign a payment rate for HCPCS code P9027. Specifically, we found that HCPCS code P9040 (Red blood cells, leukocytes reduced, irradiated, each unit) with a CY 2024 payment rate of $252.48 was sufficiently similar to P9027 to serve as a crosswalk for purposes of a payment rate assignment.
Comment: We received a comment from the manufacturer of the blood product described by P9027. HCPCS code P9027 may be used to bill for the blood product involving the Hemanext ONE System. The commenter provided estimated cost information regarding the blood product, including an expected average per-unit anticipated hospital cost of $510. The manufacturer provided the estimated costs based on total blood establishment manufacturing costs, including the costs to manufacture a single unit of the starting materials, the sales price for the Hemanext ONE System, and the blood establishment labor, overhead, ( printed page 93924) administrative, and shipping costs. The commenter noted that they believe their red blood cell product is potentially transformative and would provide improved health outcomes. The commenter stated that they believe the product whose CY 2024 Medicare payment rate reasonably approximates the projected average hospital acquisition cost for HCPCS P9027 is HCPCS code P9057 (Red blood cells, frozen/deglycerolized/washed/leukocytes reduced, irradiated, each unit), with a CY 2024 payment rate of $493.21 per unit. As such, the commenter recommends that CMS crosswalk the payment rate for HCPCS P9057 to HCPCS P9027. The commenter believes that a crosswalk to any other red blood cell product will result in a payment rate that does not cover the acquisition cost for HCPCS P9027 and creates patient access barriers.
Response: We thank the commenter for their comment. Based on the additional cost information provided by the manufacturer, we believe that HCPCS code P9040 is not the best proxy for the costs of the blood product described by HCPCS code P9027. However, we disagree with the commenter that HCPCS code P9057 is the most appropriate crosswalk for new HCPCS code P9027 because the payment rate for HCPCS code P9057 reflects additional processes that are not reflected in the code descriptor for HCPCS code P9027. Given this difference in process, and the associated differences in estimated costs, we believe HCPCS code P9035 is a more appropriate proxy for the costs associated with HCPCS code P9027. Therefore, we are establishing an interim payment rate for HCPCS code P9027 based on a crosswalk to existing blood product HCPCS code P9035. Table 1 lists the new HCPCS code P9027 and its payment crosswalk.
The interim payment rate for HCPCS code P9027 is open for public comment in this CY 2025 final rule with comment period. Specifically, HCPCS code P9027 is flagged with comment indicator “NI” in Addendum B to this final rule with comment period to indicate that we have assigned the codes an interim OPPS payment status for CY 2025 and are seeking public comments on the APC and status indicator assignments. Once we have claims data for this new HCPCS P-code, we will calculate a payment rate using the claims data that should be available for these new codes beginning in CY 2027, which is our practice for other blood products for which claims data have been available for 2 years.
(2) Brachytherapy Sources
Section 1833(t)(2)(H) of the Act mandates the creation of additional groups of covered OPD services that classify devices of brachytherapy—cancer treatment through solid source radioactive implants—consisting of a seed or seeds (or radioactive source) (“brachytherapy sources”) separately from other services or groups of services. The statute provides certain criteria for the additional groups. For the history of OPPS payment for brachytherapy sources, we refer readers to prior OPPS final rules, such as the CY 2012 OPPS/ASC final rule with comment period (77 FR 68240 and 68241). As we have stated in prior OPPS updates, we believe that adopting the general OPPS prospective payment methodology for brachytherapy sources is appropriate for a number of reasons (77 FR 68240). The general OPPS methodology uses costs based on claims data to set the relative payment weights for hospital outpatient services. This payment methodology results in more consistent, predictable, and equitable payment amounts per source across hospitals by averaging the extremely high and low values, in contrast to payment based on hospitals' charges adjusted to costs. We believe that the OPPS methodology, as opposed to payment based on hospitals' charges adjusted to cost, also would provide hospitals with incentives for efficiency in the provision of brachytherapy services to Medicare beneficiaries. Moreover, this approach is consistent with our payment methodology for the vast majority of items and services paid under the OPPS. We refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70323 through 70325) for further discussion of the history of OPPS payment for brachytherapy sources.
For CY 2025, except where otherwise indicated, we proposed to continue our policy and use the costs derived from CY 2023 claims data to set the proposed CY 2025 payment rates for brachytherapy sources because CY 2023 is the year of data we proposed to use to set the proposed payment rates for most other items and services that would be paid under the CY 2025 OPPS. With the exception of the proposed payment rate for brachytherapy source C2645 (Brachytherapy planar source, palladium-103, per square millimeter) and the proposed payment rates for low-volume brachytherapy APCs discussed in section III.D of the CY 2025 OPPS/ASC proposed rule, we proposed to base the payment rates for brachytherapy sources on the geometric mean unit costs for each source, consistent with the methodology that we proposed for other items and services paid under the OPPS, as discussed in section II.A.2 of the CY 2025 OPPS/ASC proposed rule. We also proposed for CY 2025 and subsequent years to continue the other payment policies for brachytherapy sources that we finalized and first implemented in the CY 2010 OPPS/ASC final rule with comment period (74 FR ( printed page 93925) 60537). For CY 2025 and subsequent years, we proposed to pay for the stranded and nonstranded not otherwise specified (NOS) codes, HCPCS codes C2698 (Brachytherapy source, stranded, not otherwise specified, per source) and C2699 (Brachytherapy source, non-stranded, not otherwise specified, per source), at a rate equal to the lowest stranded or nonstranded prospective payment rate for such sources, respectively, on a per-source basis (as opposed to, for example, per mCi), which is based on the policy we established in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66785). For CY 2025 and subsequent years, we also proposed to continue the policy we first implemented in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60537) regarding payment for new brachytherapy sources for which we have no claims data, based on the same reasons we discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66786; which was delayed until January 1, 2010, by section 142 of Pub. L. 110-275). Specifically, this policy is intended to enable us to assign new HCPCS codes for new brachytherapy sources to their own APCs, with prospective payment rates set based on our consideration of external data and other relevant information regarding the expected costs of the sources to hospitals. The proposed CY 2025 payment rates for brachytherapy sources are included on Addendum B to the CY 2025 OPPS/ASC proposed rule (which is available via the internet on the CMS website) and identified with status indicator “U.”
For CY 2018, we assigned status indicator “U” (Brachytherapy Sources, Paid under OPPS; separate APC payment) to HCPCS code C2645 (Brachytherapy planar source, palladium-103, per square millimeter) in the absence of claims data and established a payment rate using external data (invoice price) at $4.69 per mm2 for the brachytherapy source's APC-APC 2648 (Brachytx planar, p-103). For CY 2019, in the absence of sufficient claims data, we continued to establish a payment rate for C2645 at $4.69 per mm2 for APC 2648 (Brachytx planar, p103). Our CY 2018 claims data available for the CY 2020 OPPS/ASC final rule with comment period (84 FR 61142) included two claims with a geometric mean cost for HCPCS code C2645 of $1.02 per mm2 . In response to comments from interested parties, we agreed that, given the limited claims data available and a new outpatient indication for C2645, a payment rate for HCPCS code C2645 based on the geometric mean cost of $1.02 per mm2 may not adequately reflect the cost of HCPCS code C2645. In the CY 2020 OPPS/ASC final rule with comment period, we finalized our policy to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act, which states that the Secretary shall establish, in a budget neutral manner, other adjustments as determined to be necessary to ensure equitable payments, to maintain the CY 2019 payment rate of $4.69 per mm2 for HCPCS code C2645 for CY 2020. Similarly, in the absence of sufficient claims data to establish an APC payment rate, in the CY 2021, CY 2022, CY 2023, and CY 2024 OPPS/ASC final rules with comment period (85 FR 85879 through 85880, 86 FR 63469, 87 FR 71760-71761, and 88 FR 81553), we finalized our policy to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2019 payment rate of $4.69 per mm2 for HCPCS code C2645 for CYs 2021 through 2024.
There were no CY 2023 claims available that reported HCPCS code C2645 for the CY 2025 OPPS/ASC proposed rule. Therefore, in the absence of claims data, we proposed to continue to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2024 payment rate of $4.69 per mm2 for HCPCS code C2645, which we proposed to be assigned to APC 2648 (Brachytx planar, p-103), for CY 2025.
Additionally, for CY 2022 and subsequent calendar years, we adopted a Universal Low Volume APC policy for clinical and brachytherapy APCs. As discussed in further detail in section X.C of the CY 2022 OPPS/ASC final rule with comment period (86 FR 63743 through 63747), we adopted this policy to mitigate wide variation in payment rates that occur from year to year for APCs with low utilization. Such volatility in payment rates from year to year can result in even lower utilization and potential barriers to access. Brachytherapy APCs that have fewer than 100 single claims used for ratesetting purposes are designated as Low Volume APCs unless an alternative payment rate is applied, such as the use of our equitable adjustment authority under section 1833(t)(2)(E) of the Act in the case of APC 2648 (Brachytx planar, p-103), for which HCPCS code C2645 (Brachytherapy planar source, palladium-103, per square millimeter) is the only code assigned as discussed previously in this section.
For CY 2025, we proposed to designate six brachytherapy APCs as Low Volume APCs as these APCs meet our criteria to be designated as Low Volume APCs. For more information on the brachytherapy APCs we proposed to designate as Low Volume APCs, see section III.D of the CY 2025 OPPS/ASC proposed rule.
Comment: We received support from commenters for our proposal to use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2024 payment rate of $4.69 per mm2 for HCPCS code C2645, which we proposed to assign to APC 2648 (Brachytx planar, p-103) for CY 2025.
Response: We thank the commenters for their support.
After consideration of the public comment we received, we are finalizing, without modification, that we will use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to maintain the CY 2024 payment rate of $4.69 per mm2 for HCPCS code C2645, which we are assigning to APC 2648 (Brachytx planar, p-103), for CY 2025.
The final CY 2025 payment rates for brachytherapy sources are included in Addendum B to this final rule with comment period (which is available via the internet on the CMS website) and are identified with status indicator “U.” We continue to invite interested parties to submit recommendations for new codes to describe new brachytherapy sources. Such recommendations should be directed via email to outpatientpps@cms.hhs.gov or by mail to the Division of Outpatient Care, Mail Stop C4-01-26, Centers for Medicare and Medicaid Services, 7500 Security Boulevard, Baltimore, MD 21244. We will continue to add new brachytherapy source codes and descriptors to our systems for payment on a quarterly basis.
b. Comprehensive APCs (C-APCs) for CY 2025
(1) Background
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 74861 through 74910), we finalized a comprehensive payment policy that packages payment for adjunctive and secondary items, services, and procedures into the most costly primary procedure under the OPPS at the claim level. The policy was finalized in CY 2014, but the effective date was delayed until January 1, 2015, to allow additional time for further analysis, opportunity for public comment, and systems preparation. The comprehensive APC (C-APC) policy was implemented effective January 1, 2015, with modifications and clarifications in response to public ( printed page 93926) comments received regarding specific provisions of the C-APC policy (79 FR 66798 through 66810).
A C-APC is defined as a classification for the provision of a primary service and all adjunctive services provided to support the delivery of the primary service. We established C-APCs as a category broadly for OPPS payment and implemented 25 C-APCs beginning in CY 2015 (79 FR 66809 and 66810). We have gradually added new C-APCs since the policy was implemented beginning in CY 2015, with the number of C-APCs now totaling 72 (80 FR 70332; 81 FR 79584 and 79585; 83 FR 58844 through 58846; 84 FR 61158 through 61166; 85 FR 85885; 86 FR 63474; 87 FR 71769; and 88 FR 81562).
Under our C-APC policy, we designate a service described by a HCPCS code assigned to a C-APC as the primary service when the service is identified by OPPS status indicator “J1.” When such a primary service is reported on a hospital outpatient claim, taking into consideration the few exceptions that are discussed below, we make payment for all other items and services reported on the hospital outpatient claim as being integral, ancillary, supportive, dependent, and adjunctive to the primary service (hereinafter collectively referred to as “adjunctive services”) and representing components of a complete comprehensive service (78 FR 74865 and 79 FR 66799). Payments for adjunctive services are packaged into the payments for the primary services. This results in a single prospective payment for each of the primary, comprehensive services based on the costs of all reported services at the claim level. One example of a primary service would be a partial mastectomy and an example of a secondary service packaged into that primary service would be a radiation therapy procedure.
Services excluded from the C-APC policy under the OPPS include services that are not covered OPD services, services that cannot by statute be paid for under the OPPS, and services that are required by statute to be separately paid. This includes certain mammography and ambulance services that are not covered OPD services in accordance with section 1833(t)(1)(B)(iv) of the Act; brachytherapy seeds, which also are required by statute to receive separate payment under section 1833(t)(2)(H) of the Act; pass-through payment drugs and devices, which also require separate payment under section 1833(t)(6) of the Act; self-administered drugs (SADs) that are not otherwise packaged as supplies because they are not covered under Medicare Part B under section 1861(s)(2)(B) of the Act; and certain preventive services (78 FR 74865 and 79 FR 66800 and 66801). A list of services excluded from the C-APC policy is included in Addendum J to this final rule (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices). If a service does not appear on this list of excluded services, payment for it will be packaged into the payment for the primary C-APC service when it appears on an outpatient claim with a primary C-APC service.
The C-APC policy payment methodology set forth in the CY 2014 OPPS/ASC final rule with comment period and modified and implemented beginning in CY 2015 is summarized as follows (78 FR 74887 and 79 FR 66800):
Basic Methodology. As stated in the CY 2015 OPPS/ASC final rule with comment period, we define the C-APC payment policy as including all covered OPD services on a hospital outpatient claim reporting a primary service that is assigned to status indicator “J1,” [2] excluding services that are not covered OPD services or that cannot by statute be paid for under the OPPS. Services and procedures described by HCPCS codes assigned to status indicator “J1” are assigned to C-APCs based on our usual APC assignment methodology by evaluating the geometric mean costs of the primary service claims to establish resource similarity and the clinical characteristics of each procedure to establish clinical similarity within each APC.
In the CY 2016 OPPS/ASC final rule with comment period, we expanded the C-APC payment methodology to qualifying extended assessment and management encounters through the “Comprehensive Observation Services” C-APC (C-APC 8011). Services within this APC are assigned status indicator “J2.” [3] Specifically, we make a payment through C-APC 8011 for a claim that:
- Does not contain a procedure described by a HCPCS code to which we have assigned status indicator “T;”
- Contains 8 or more units of services described by HCPCS code G0378 (Hospital observation services, per hour);
- Contains services provided on the same date of service or one day before the date of service for HCPCS code G0378 that are described by one of the following codes: HCPCS code G0379 (Direct admission of patient for hospital observation care) on the same date of service as HCPCS code G0378; CPT code 99281 (Emergency department visit for the evaluation and management of a patient (Level 1)); CPT code 99282 (Emergency department visit for the evaluation and management of a patient (Level 2)); CPT code 99283 (Emergency department visit for the evaluation and management of a patient (Level 3)); CPT code 99284 (Emergency department visit for the evaluation and management of a patient (Level 4)); CPT code 99285 (Emergency department visit for the evaluation and management of a patient (Level 5)) or HCPCS code G0380 (Type B emergency department visit (Level 1)); HCPCS code G0381 (Type B emergency department visit (Level 2)); HCPCS code G0382 (Type B emergency department visit (Level 3)); HCPCS code G0383 (Type B emergency department visit (Level 4)); HCPCS code G0384 (Type B emergency department visit (Level 5)); CPT code 99291 (Critical care, evaluation and management of the critically ill or critically injured patient; first 30-74 minutes); or HCPCS code G0463 (Hospital outpatient clinic visit for assessment and management of a patient); and
- Does not contain services described by a HCPCS code to which we have assigned status indicator “J1.”
The assignment of status indicator “J2” to a specific set of services performed in combination with each other allows for all other OPPS payable services and items reported on the claim (excluding services that are not covered OPD services or that cannot by statute be paid for under the OPPS) to be deemed adjunctive services representing components of a comprehensive service and resulting in a single prospective payment for the comprehensive service based on the costs of all reported services on the claim (80 FR 70333 through 70336).
Services included under the C-APC payment packaging policy, that is, services that are typically adjunctive to the primary service and provided during the delivery of the comprehensive service, include diagnostic procedures, laboratory tests, and other diagnostic tests and treatments that assist in the delivery of the primary procedure; visits and evaluations performed in association with the procedure; uncoded services and supplies used during the service; durable medical equipment as well as prosthetic and ( printed page 93927) orthotic items and supplies when provided as part of the outpatient service; and any other components reported by HCPCS codes that represent services that are provided during the complete comprehensive service (78 FR 74865 and 79 FR 66800).
In addition, payment for hospital outpatient department services that are similar to therapy services, such as speech language pathology, and delivered either by therapists or nontherapists is included as part of the payment for the packaged complete comprehensive service. These services that are provided during the perioperative period are adjunctive services and are deemed not to be therapy services as described in section 1834(k) of the Act, regardless of whether the services are delivered by therapists or other nontherapist health care workers. We have previously noted that therapy services are those provided by therapists under a plan of care in accordance with section 1835(a)(2)(C) and section 1835(a)(2)(D) of the Act and are paid for under section 1834(k) of the Act, subject to annual therapy caps as applicable (78 FR 74867 and 79 FR 66800). However, certain other services similar to therapy services are considered and paid for as hospital outpatient department services. Payment for these nontherapy outpatient department services that are reported with therapy codes and provided with a comprehensive service is included in the payment for the packaged complete comprehensive service. We note that these services, even though they are reported with therapy codes, are hospital outpatient department services and not therapy services. We refer readers to the July 2016 OPPS Change Request 9658 (Transmittal 3523) for further instructions on reporting these services in the context of a C-APC service.
Items included in the packaged payment provided in conjunction with the primary service also include all drugs, biologicals, and radiopharmaceuticals, regardless of cost, except those drugs with pass-through payment status and SADs, unless they function as packaged supplies (78 FR 74868, 74869, and 74909 and 79 FR 66800). We refer readers to Section 50.2M, Chapter 15, of the Medicare Benefit Policy Manual for a description of our policy on SADs treated as hospital outpatient supplies, including lists of SADs that function as supplies and those that do not function as supplies.[4]
We define each hospital outpatient claim reporting a single unit of a single primary service assigned to status indicator “J1” as a single “J1” unit procedure claim (78 FR 74871 and 79 FR 66801). Line-item charges for services included on the C-APC claim are converted to line-item costs, which are then summed to develop the estimated APC costs. These claims are then assigned one unit of the service with status indicator “J1” and later used to develop the geometric mean costs for the C-APC relative payment weights. (We note that we use the term “comprehensive” to describe the geometric mean cost of a claim reporting “J1” service(s) or the geometric mean cost of a C-APC, inclusive of all the items and services included in the C-APC service payment bundle.) Charges for services that would otherwise be separately payable are added to the charges for the primary service. This process differs from our traditional cost accounting methodology only in that all such services on the claim are packaged (except certain services as described above). We apply our standard data trims, which exclude claims with extremely high primary units or extreme costs.
The comprehensive geometric mean costs are used to establish resource similarity and, along with clinical similarity, dictate the assignment of the primary services to the C-APCs. We establish a ranking of each primary service (single unit only) to be assigned to status indicator “J1” according to its comprehensive geometric mean costs. For the minority of claims reporting more than one primary service assigned to status indicator “J1” or units thereof, we identify one “J1” service as the primary service for the claim based on our cost-based ranking of primary services. We then assign these multiple “J1” procedure claims to the C-APC to which the service designated as the primary service is assigned. If the reported “J1” services on a claim map to different C-APCs, we designate the “J1” service assigned to the C-APC with the highest comprehensive geometric mean cost as the primary service for that claim. If the reported multiple “J1” services on a claim map to the same C-APC, we designate the most costly service (at the HCPCS code level) as the primary service for that claim. This process results in initial assignments of claims for the primary services assigned to status indicator “J1” to the most appropriate C-APCs based on both single and multiple procedure claims reporting these services and clinical and resource homogeneity.
Complexity Adjustments. We use complexity adjustments to provide increased payment for certain comprehensive services. We apply a complexity adjustment by promoting qualifying paired “J1” service code combinations or paired code combinations of “J1” services and certain add-on codes (as described further below) from the originating C-APC (the C-APC to which the designated primary service is first assigned) to the next higher paying C-APC in the same clinical family of C-APCs. We apply this type of complexity adjustment when the paired code combination represents a complex, costly form or version of the primary service according to the following criteria:
- Frequency of 25 or more claims reporting the code combination (frequency threshold); and
- Violation of the 2 times rule, as stated in section 1833(t)(2) of the Act and section III.B.2 of this final rule with comment period, in the originating C-APC (cost threshold).
These criteria identify paired code combinations that occur commonly and exhibit materially greater resource requirements than the primary service. The CY 2017 OPPS/ASC final rule with comment period (81 FR 79582) included a revision to the complexity adjustment eligibility criteria. Specifically, we finalized a policy to discontinue the requirement that a code combination (that qualifies for a complexity adjustment by satisfying the frequency and cost criteria thresholds described above) also not create a 2 times rule violation in the higher level or receiving APC.
After designating a single primary service for a claim, we evaluate that service in combination with each of the other procedure codes reported on the claim assigned to status indicator “J1” (or certain add-on codes) to determine if there are paired code combinations that meet the complexity adjustment criteria. For a new HCPCS code, we determine initial C-APC assignment and qualification for a complexity adjustment using the best available information, crosswalking the new HCPCS code to a predecessor code(s) when appropriate.
Once we have determined that a particular code combination of “J1” services (or combinations of “J1” services reported in conjunction with certain add-on codes) represents a ( printed page 93928) complex version of the primary service because it is sufficiently costly, frequent, and a subset of the primary comprehensive service overall according to the criteria described above, we promote the claim including the complex version of the primary service as described by the code combination to the next higher cost C-APC within the clinical family, unless the primary service is already assigned to the highest cost APC within the C-APC clinical family or assigned to the only C-APC in a clinical family. We do not create new APCs with a comprehensive geometric mean cost that is higher than the highest geometric mean cost (or only) C-APC in a clinical family just to accommodate potential complexity adjustments. Therefore, the highest payment for any claim including a code combination for services assigned to a C-APC would be the highest paying C-APC in the clinical family (79 FR 66802).
We package payment for all add-on codes into the payment for the C-APC. However, certain primary service add-on combinations may qualify for a complexity adjustment. As noted in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70331), all add-on codes that can be appropriately reported in combination with a base code that describes a primary “J1” service are evaluated for a complexity adjustment.
To determine which combinations of primary service codes reported in conjunction with an add-on code may qualify for a complexity adjustment for CY 2025, we apply the frequency and cost criteria thresholds discussed above, testing claims reporting one unit of a single primary service assigned to status indicator “J1” and any number of units of a single add-on code for the primary “J1” service. If the frequency and cost criteria thresholds for a complexity adjustment are met and reassignment to the next higher cost APC in the clinical family is appropriate (based on meeting the criteria outlined above), we make a complexity adjustment for the code combination; that is, we reassign the primary service code reported in conjunction with the add-on code to the next higher cost C-APC within the same clinical family of C-APCs. As previously stated, we package payment for add-on codes into the C-APC payment rate. If any add-on code reported in conjunction with the “J1” primary service code does not qualify for a complexity adjustment, payment for the add-on service continues to be packaged into the payment for the primary service and is not reassigned to the next higher cost C-APC. We list the complexity adjustments for “J1” and add-on code combinations for CY 2025, along with all the other final complexity adjustments, in Addendum J to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
Addendum J to this final rule with comment period includes the cost statistics for each code combination that would qualify for a complexity adjustment (including primary code and add-on code combinations). Addendum J to this final rule with comment period also contains summary cost statistics for each of the paired code combinations that describe a complex code combination that would qualify for a complexity adjustment and be reassigned to the next higher cost C-APC within the clinical family. The combined statistics for all proposed reassigned complex code combinations are represented by an alphanumeric code with the first four digits of the designated primary service followed by a letter. For example, the proposed geometric mean cost listed in Addendum J for the code combination described by complexity adjustment assignment 3320R, which is assigned to C-APC 5224 (Level 4 Pacemaker and Similar Procedures), includes all paired code combinations that will be reassigned to C-APC 5224 when CPT code 33208 is the primary code. Providing the information contained in Addendum J to this final rule with comment period allows interested parties the opportunity to better assess the impact associated with the assignment of claims with each of the paired code combinations eligible for a complexity adjustment.
Comment: We received support from commenters for a variety of existing and proposed complexity adjustments.
Response: We thank the commenters for their support.
Comment: Multiple commenters requested that CMS apply a complexity adjustment to additional code combinations. The specific C-APC complexity adjustment code combinations requested by the commenters for CY 2025 are listed in Table 2.
( printed page 93929) ( printed page 93930)Response: We reviewed each of the requested code combinations suggested by commenters, listed in Table 2, against our complexity adjustment ( printed page 93931) criteria. The code combination for primary HCPCS code 28296 with secondary HCPCS code 28270 meets our cost and frequency criteria, qualifying for a complexity adjustment for CY 2025. All the remaining code combinations listed failed to meet our cost or frequency criteria and do not qualify for complexity adjustments for CY 2025. Addendum J to this final rule with comment period includes the cost statistics for each code combination that was evaluated for a complexity adjustment.
Comment: Commenters requested that CMS modify, waive, or eliminate the established C-APC complexity adjustment eligibility criteria of 25 or more claims reporting the code combination (frequency) and a violation of the 2 times rule in the originating C-APC (cost) to allow additional code combinations to qualify for complexity adjustments. These commenters were concerned that C-APC packaging and a lack of complexity adjustment would limit access to procedures. Specifically, some commenters expressed concern that CMS's methodology for determining complexity adjustments is unnecessarily restrictive, particularly the 25-claim threshold, and suggested that CMS eliminate the 25-claim threshold and implement a complexity adjustment whenever a code pair exceeds the cost threshold.
Some commenters were concerned that when multiple “J1” primary services are reported on a claim, along with an add-on service, the add-on service is not evaluated for a complexity adjustment. Commenters cited examples where significant claims volume from add-on services may not be incorporated into the complexity adjustment evaluation. Commenters also reiterated requests to broaden the complexity adjustment policy and allow clusters of procedures, consisting of a “J1” code pair and multiple other associated add-on codes used in combination with that “J1” code pair, to qualify for complexity adjustments. Commenters stated that there are certain complex procedures that include numerous add-on codes and this approach would allow more accurate reflection of the cost of medical practice when multiple procedures are performed together. They noted that lack of additional payment for these code combinations can present a financial challenge for the providers who perform these more resource intensive services.
Response: We appreciate these comments. At this time, we do not believe changes to the C-APC complexity adjustment criteria are necessary or that we should make exceptions to the criteria to allow claims with the code combinations suggested by the commenters to receive complexity adjustments. As we stated in the CY 2017 OPPS/ASC final rule (81 FR 79582), we believe that the complexity adjustment criteria, which require a frequency of 25 or more claims reporting a code combination and a violation of the 2 times rule in the originating C-APC, are appropriate to determine if a combination of procedures represents a complex, costly subset of the primary service that should qualify for the adjustment and be paid at the next higher paying C-APC in the clinical family. As we also previously stated in the CY 2020 OPPS/ASC final rule with comment period (84 FR 61161), a minimum of 25 claims is already a very low threshold for a national payment system. Lowering the minimum of 25 claims further could lead to unnecessary complexity adjustments for service combinations that are rarely performed.
Comment: A few commenters suggested that to help maintain better stability and predictability of payments associated with complex procedures, CMS should revise the complexity adjustment policy by allowing qualifying codes to maintain the complexity adjusted payment for three calendar years before they are required to go through the eligibility review. For codes that do not qualify in a given year, they could be evaluated in the next calendar year. Commenters asserted that the updated cadence would allow more time for providers and coders to be educated on new code pairs that qualify for complexity adjustments and it would help promote payment stability and predictability for these complex procedures.
Response: For the OPPS ratesetting process, our goal is to use the best available data for ratesetting to accurately estimate the costs associated with furnishing outpatient services and to set appropriate payment rates. We evaluate code combinations each year against our complexity adjustment criteria using the latest available data. Potentially maintaining complexity adjustments that are not supported by the latest data could undermine our goals for the ratesetting process.
Comment: A few commenters requested that CMS provide additional information on the rationale for not counting claims that reflect code combinations of services that would otherwise meet the frequency criteria needed to receive a complexity adjustment. Commenters conducted their own data modeling using data reflecting all code combinations that met the frequency criteria needed to receive a complexity adjustment. Commenters asked for greater transparency on the methodology CMS used to evaluate the complexity adjustment frequency criteria for code combinations.
Response: We refer commenters to the claims accounting narrative under supporting documentation for this CY 2025 OPPS/ASC final rule on the CMS website at: https://www.cms.gov/files/document/2025-nprm-opps-claims-accounting.pdf. The claims accounting narrative provides a detailed overview of how CMS processes the CY 2023 claims data to produce the proposed prospective CY 2025 OPPS payment rates. We are updating the section on Comprehensive APCs in order to provide additional clarity on the claims accounting process used for determining complexity adjustments.
After consideration of the public comments we received, we are finalizing the C-APC complexity adjustment policy for CY 2025 as proposed. We are also finalizing the proposed complexity adjustments, with the addition of one new code combination ( i.e., primary HCPCS code 28296 with secondary HCPCS code 28270) suggested by commenters that meets our complexity adjustment criteria.
(2) Exclusion of Procedures Assigned to New Technology APCs From the C-APC Policy
Services that are assigned to New Technology APCs are typically new procedures that do not have sufficient claims history to establish an accurate payment for them. Beginning in CY 2002, we retain services within New Technology APC groups until we gather sufficient claims data to enable us to assign the service to an appropriate clinical APC. This policy allows us to move a service from a New Technology APC in less than 2 years if sufficient data are available. It also allows us to retain a service in a New Technology APC for more than 2 years if sufficient data upon which to base a decision for reassignment have not been collected (82 FR 59277).
The C-APC payment policy packages payment for adjunctive and secondary items, services, and procedures into the most costly primary procedure under the OPPS at the claim level. Prior to CY 2019, when a procedure assigned to a New Technology APC was included on the claim with a primary procedure, identified by OPPS status indicator “J1,” payment for the new technology service was typically packaged into the payment for the primary procedure. ( printed page 93932) Because the new technology service was not separately paid in this scenario, the overall number of single claims available to determine an appropriate clinical APC for the new service was reduced. This was contrary to the objective of the New Technology APC payment policy, which is to gather sufficient claims data to enable us to assign the service to an appropriate clinical APC.
To address this issue and ensure that there are sufficient claims data for services assigned to New Technology APCs, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58847), we finalized excluding payment for any procedure that is assigned to a New Technology APC (APCs 1491 through 1599 and APCs 1901 through 1908) from being packaged when included on a claim with a “J1” service assigned to a C-APC. In the CY 2020 OPPS/ASC final rule with comment period, we finalized that beginning in CY 2020, payment for services assigned to a New Technology APC would be excluded from being packaged into the payment for comprehensive observation services assigned status indicator “J2” when they are included on a claim with a “J2” service (84 FR 61167).
(3) Exclusion of Drugs and Biologicals Described by HCPCS Code C9399 (Unclassified Drugs or Biologicals) From the C-APC Policy
Section 1833(t)(15) of the Act, as added by section 621(a)(1) of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (Pub. L. 108-173), provides for payment under the OPPS for new drugs and biologicals until HCPCS codes are assigned. Under this provision, we are required to make payment for a covered outpatient drug or biological that is furnished as part of covered outpatient department services but for which a HCPCS code has not yet been assigned in an amount equal to 95 percent of average wholesale price (AWP) for the drug or biological.
In the CY 2005 OPPS/ASC final rule with comment period (69 FR 65805), we implemented section 1833(t)(15) of the Act by instructing hospitals to bill for a drug or biological that is newly approved by the Food and Drug Administration (FDA) and that does not yet have a HCPCS code by reporting the National Drug Code (NDC) for the product along with the newly created HCPCS code C9399 (Unclassified drugs or biologicals). We explained that when HCPCS code C9399 appears on a claim, the Outpatient Code Editor (OCE) suspends the claim for manual pricing by the Medicare Administrative Contractor (MAC). The MAC prices the claim at 95 percent of the drug or biological's AWP, using Red Book or an equivalent recognized compendium, and processes the claim for payment. We emphasized that this approach enables hospitals to bill and receive payment for a new drug or biological concurrent with its approval by the FDA. The hospital does not have to wait for the next quarterly release or for approval of a product specific HCPCS code to receive payment for a newly approved drug or biological or to resubmit claims for adjustment. We instructed that hospitals would discontinue billing HCPCS code C9399 and the NDC upon implementation of a product specific HCPCS code, status indicator, and appropriate payment amount with the next quarterly update. We also note that HCPCS code C9399 is paid in a similar manner in the ASC setting, as 42 CFR 416.171(b) outlines that certain drugs and biologicals for which separate payment is allowed under the OPPS are considered covered ancillary services for which the OPPS payment rate, which is 95 percent of AWP for HCPCS code C9399, applies. Since the implementation of the C-APC policy in 2015, payment for drugs and biologicals described by HCPCS code C9399 had been included in the C-APC payment when these products appear on a claim with a primary C-APC service. Packaging payment for these drugs and biologicals that appear on a hospital outpatient claim with a primary C-APC service is consistent with our C-APC packaging policy under which we make payment for all items and services, including all non-pass-through drugs, reported on the hospital outpatient claim as being integral, ancillary, supportive, dependent, and adjunctive to the primary service and representing components of a complete comprehensive service, with certain limited exceptions (78 FR 74869). It was our position that the total payment for the C-APC with which payment for a drug or biological described by HCPCS code C9399 is packaged includes payment for the drug or biological at 95 percent of its AWP.
However, we determined that in certain instances, drugs and biologicals described by HCPCS code C9399 are not being paid at 95 percent of their AWPs when payment for them is packaged with payment for a primary C-APC service. In order to ensure payment for new drugs, biologicals, and radiopharmaceuticals described by HCPCS code C9399 at 95 percent of their AWP, for CY 2023 and subsequent years, we finalized our proposal to exclude any drug, biological, or radiopharmaceutical described by HCPCS code C9399 from packaging when the drug, biological, or radiopharmaceutical is included on a claim with a “J1” service, which is the status indicator assigned to a C-APC, and a claim with a “J2” service, which is the status indicator assigned to comprehensive observation services. See Addendum J for the CY 2025 C-APC payment policy exclusions.
In the CY 2023 OPPS/ASC final rule with comment period, we finalized the proposal in section XI “CY 2023 OPPS Payment Status and Comment Indicators” to add a new definition to status indicator “A” to include unclassified drugs and biologicals that are reportable with HCPCS code C9399 (87 FR 72051). The definition, found in Addendum D1, would ensure the MAC prices claims for drugs, biologicals, or radiopharmaceuticals billed with HCPCS code C9399 at 95 percent of the drug or biological's AWP and pays separately for the drug, biological, or radiopharmaceutical under the OPPS when it appears on the same claim as a primary C-APC service.
(4) Exclusion of Cell and Gene Therapies From the C-APC Policy
As previously discussed in this section, and in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74865), the C-APC policy packages payment for items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to the primary service and provided during the delivery of the comprehensive service, including diagnostic procedures, laboratory tests and other diagnostic tests and treatments that assist in the delivery of the primary procedure. In the CY 2014 OPPS/ASC final rule (78 FR 74861), we finalized defining a comprehensive APC as a classification for the provision of a primary service and all adjunctive services provided to support the delivery of the primary service. Because a comprehensive APC treats all individually reported codes as representing components of the comprehensive service, we make a single prospective payment based on the cost of all individually reported codes that represent the provision of a primary service and all adjunctive services provided to support that delivery of the primary service.
As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59201 through 59204), we generally treat all items and services reported on a C-APC claim as integral, ancillary, supportive, dependent, and adjunctive to the primary service and representing ( printed page 93933) components of a comprehensive service. Historically, items packaged for payment provided in conjunction with the primary C-APC service also include all drugs, biologicals, and radiopharmaceuticals, regardless of cost, except those drugs with pass-through payment status and those drugs that are usually self-administered (SADs), unless they function as supplies (78 FR 74868 through 74869 and 74909).
Our intent has been to make a single prospective payment based on the cost of all individually reported codes that appear on a claim with the primary C-APC service, which we believe represent the provision of a primary service and all adjunctive services provided to support that delivery of the primary service. We discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59201 through 59204), there are rare instances where the cell and gene therapies listed in Table 3, which are usually separately payable under the OPPS, appear on the same claim as a primary C-APC service and therefore, have their payment packaged with payment for the primary C-APC service. The therapies in Table 3 are usually separately paid and priced using the ASP methodology when not on a C-APC claim. Given the unique nature of these therapies, we do not believe they function as integral, ancillary, supportive, dependent, or adjunctive to any of the current C-APCs primary services. The cell therapies described in Table 3 are primarily for the treatment of specific cancers and are administered through an intravenous infusion. The gene therapies listed in Table 3 are generally for the treatment of certain rare ocular or spinal conditions caused by specific genetic mutations and are also either intravenously infused or administered through a subretinal injection. When these products are administered, they are the primary treatment being administered to a patient and thus, are not integral, ancillary, supportive, dependent, or adjunctive to any primary C-APC services. Additionally, the current primary C-APC services describe common surgical procedures, such as breast/lymphatic surgery and musculoskeletal procedures. The cell and gene therapies listed in Table 3 are intended to treat a specific condition and would not be used to support the outcome of any primary C-APC procedure. For example, HCPCS code J3399 (Injection, onasemnogene abeparvovec-xioi, per treatment, up to 5x10‸15 vector genomes) may be used to describe the gene therapy Zolgensma. This product is FDA-approved as an adeno-associated virus (AAV) vector-based gene therapy indicated for the treatment of pediatric patients less than 2 years of age with spinal muscular atrophy (SMA) with bi-allelic mutations in the survival motor neuron 1 (SMN1) gene. The specified mechanism of onasemnogene abeparvovec is a recombinant AAV9-based gene therapy designed to deliver a copy of the gene encoding the human SMN protein.[5] The function of a product such as Zolgensma, is not intended to be integral, ancillary, supportive, dependent, and adjunctive to any C-APC as the gene therapy itself is an independent treatment.
Yescarta (HCPCS code Q2041) is an example of a cell therapy that functions as an independent treatment. Based on its FDA-approved indication,[6] this product's intended clinical use would not be integral, ancillary, supportive, dependent, or adjunctive to any current C-APC primary service. Yescarta is indicated as a CD19-directed genetically modified autologous T cell immunotherapy for the treatment of Adult patients with large B-cell lymphoma that is refractory to first-line chemoimmunotherapy or that relapses within 12 months of first-line chemoimmunotherapy and adult patients with relapsed or refractory large B-cell lymphoma after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL) not otherwise specified, primary mediastinal large B-cell lymphoma, high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma. Yescarta is the primary treatment being performed when administered for these FDA-approved indications and should not be packaged as supportive of any C-APC primary service even if the two services appear on the same claim.
We explained in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74868) that intravenous drugs, for example, are OPPS services that are considered adjunctive to the primary procedure because the correct administration of the drug either promotes a beneficial outcome, such as the use of intravenous pain medications, or prevents possible complications, such as the use of intravenous blood pressure medications to temporarily replace oral blood pressure medications and reduce the risk of a sudden rise in blood pressure when a normal daily medication is stopped. In the case of the cell and gene therapies described in Table 3, however, we do not believe the therapies “promote a beneficial outcome” or “prevent possible complications” of any of the procedures currently designated as primary C-APC services. While the cell and gene therapies in Table 3 may “promote a beneficial outcome” for the patient in general, we do not believe the provision of cell and gene therapies are “promoting a beneficial outcome” for any of the primary C-APC services themselves, as the cell and gene therapies are serving as independent therapies. These are distinguishable from the previous examples of intravenous pain medications that are directly related to the primary C-APC service and promote a beneficial outcome for that procedure. Further, in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74865), we stated that we proposed to package into C-APCs all of these integral, ancillary, supportive, dependent, and adjunctive services, hereinafter collectively referred to as “adjunctive services,” provided during the delivery of the comprehensive service. This includes the diagnostic procedures, laboratory tests and other diagnostic tests, and treatments that assist in the delivery of the primary procedure. In the CY 2025 OPPS/ASC proposed rule (89 FR 59201 through 59204), we stated we did not believe that the cell and gene therapies listed in Table 3 are assisting in the delivery of any primary procedure currently assigned to a C-APC.
Therefore, for CY 2025 only, we proposed not to package payment for the cell and gene therapies listed in Table 3 into the payment for the primary C-APC service when they appear on the same claim as primary C-APCs services. We proposed this policy for one year only in order to gather more information from interested parties as to whether the proposed policy appropriately captures all of the unique therapies, such as the cell and gene therapies listed in Table 3, that function as primary treatments and do not support C-APC primary services. As discussed later in this section, we welcomed comments from readers on the proposal and the potential need for a different, modified, expanded, or supplemental policy for future rulemaking. We stated we will assess whether to continue this policy, or a modified version of this policy, beyond one year in future rulemaking, taking into consideration the comments received.
We did not propose to include therapies that are on drug pass-through status for all of CY 2025 in Table 3 ( printed page 93934) because pass-through drugs are already excluded from C-APC packaging. We proposed that products for which pass-through status is expiring in CY 2025 would be excluded from C-APC packaging after their pass-through status expires. For example, the product described by HCPCS code Q2056 has pass-through status expiring June 30, 2025. Until its pass-through status expires, the product will be excluded from C-APC packaging due to the pass-through C-APC exclusion policy, but after its pass-through status expires, we propose that the therapy would continue to be excluded from C-APC packaging under our proposed exclusion for cell and gene therapies. For more information on drug pass-through status, including expiring and continuing pass-through status, please see section V.A of this final rule with comment period.
( printed page 93935)We proposed to exclude the therapies listed in Table 3 from C-APC packaging. We invited comment on the proposal, and we invited comment on whether there are any additional cell and gene therapies that may be appropriate to exclude from C-APC packaging for CY 2025. We asked commenters to explain why any additional cell and gene therapies that they believe should be excluded from C-APC packaging were not integral, ancillary, supportive, dependent, or adjunctive to any C-APC primary service. We invited comment on whether the proposal should be extended beyond 1 year or if a different, expanded, or supplemental policy approach may be warranted in future rulemaking. For example, we were interested in comments on whether there are other classes of drugs, biologicals, or other products that are not clearly integral, ancillary, adjunctive, or supportive of a primary C-APC service but could appear on the same claim as the C-APC for that primary service and for which payment would be packaged into the C-APC payment under our current policy. We stated that we would expect clinical evidence supporting commenters' assertion that other identified classes of drugs, biologicals, medical devices, or other products are not clearly supportive of a primary C-APC service but may nonetheless appear on the same claim as a primary C-APC procedure. Similarly, we invited comment on whether interested parties believe it is appropriate for these other classes of drugs, biologicals, or medical devices to be excluded from packaging with all C-APCs or only specific C-APCs, such as the Comprehensive Observation Services C-APC (SI = “J2”).
Finally, we invited comment on the following:
- Because the cell and gene therapies listed in Table 3 are not integral, ancillary, supportive, dependent, or adjunctive to any current C-APC procedure, how could CMS structure a new C-APC, or similar packaged payment policy, for the service to administer cell or gene therapies, such by creating as a Chimeric Antigen Receptor (CAR) T-cell therapy administration C-APC, with which the CAR-T or gene therapy would be integral, ancillary, supportive, dependent, or adjunctive to the primary C-APC service? What integral, ancillary, supportive, dependent, or adjunctive items and services are routinely provided as part of the administration of cell and gene therapies or in conjunction with cell and gene therapies generally?
We recognized that currently, for CY 2024, the following HCPCS codes are associated with CAR-T therapy: HCPCS code 0537T (Chimeric antigen receptor t-cell (car-t) therapy; harvesting of blood-derived t lymphocytes for development of genetically modified autologous car-t cells, per day), 0538T (Chimeric antigen receptor t-cell (car-t) therapy; preparation of blood-derived t lymphocytes for transportation (eg, cryopreservation, storage)), 0539T (Chimeric antigen receptor t-cell (car-t) therapy; receipt and preparation of car-t cells for administration), and 0540T (Chimeric antigen receptor t-cell (car-t) therapy; car-t cell administration, autologous) as discussed in previous OPPS rulemaking, including the CY 2022 OPPS/ASC final rule with comment period (86 FR 63550 through 63552).
Separately, we also invited comment on whether policy revisions to the C-APC policy may be appropriate in future rulemaking, such as a modified outlier payment policy specific to C-APCs to address related situations in the future. We listed all proposed C-APC exclusion categories for CY 2025 in Addendum J to the CY 2025 OPPS/ASC proposed rule (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
Comment: Commenters were generally very supportive of the proposal and thought this agency action was a positive step forward to ensure access to these new classes of drugs. They agreed with CMS's assessment that the cell and gene therapies listed in the CY 2025 OPPS/ASC proposed rule did not function as integral, ancillary, supportive, dependent, or adjunctive to any of the current C-APCs primary services. Commenters noted that they agreed that these treatments acted as independent therapies and were the reason for a Medicare beneficiary's visit to a hospital outpatient department. Additionally, commenters believed that this policy was necessary and important given the large number of cell and gene therapy products currently being researched that may ultimately be approved and available to Medicare beneficiaries in the coming years.
Response: We thank commenters for their support of our proposal and rationale.
Comment: Many commenters recommended CMS make this policy permanent as soon as possible. They noted that they agreed with CMS's assessment that it would be inappropriate to package cell and gene therapies into C-APCs for CY 2025 only, but that the same reasons that C-APC packaging would be inappropriate for CY 2025 would also apply to subsequent years. Other commenters recommended that CMS make this policy effective for an extended period of time, such as three years, in order to better understand how the policy is working.
Response: We thank commenters for their feedback. Based on the comments received, we are persuaded that the reasoning for excluding these cell and gene therapy products from C-APC packaging in CY 2025 would also apply to subsequent years. As such, we will be finalizing our proposal as later discussed in this section for CY 2025 and subsequent years.
Comment: One commenter recommended that its cell therapy be added to the list of qualifying exceptions to C-APC packaging as their product received FDA approval on August 1, 2024, during the public comment period. A few commenters recommended CMS apply this packaging exception to other categories of drugs that they believed fell into a similar clinical category as cell and gene therapies. For example, one commenter suggested that its drug product is one of a growing number of innovative drugs that are administered using surgical procedures that are mapped to a C-APC, resulting in packaging of that product. The commenter stated that the product was the primary treatment being delivered to the patient, and not the C-APC procedure. Another commenter stated they have a product in the pipeline that, according to guidance from a specialty society, requires a procedure corresponding to CPT code 51720 (bladder instillation of anticarcinogenic agent), which is assigned to C-APC 5372, in order to insert the investigational product into the bladder. This commenter believed their product is the primary treatment being administered to a patient and is not supportive to any primary C-APC services. Therefore, they requested this product not be packaged into a C-APC and believed C-APC packaging would be inappropriate. This commenter also requested that in order to prevent C-APC packaging, one option would be for CMS to reassign the code used to describe the administration of its product to a drug administration APC rather than a C-APC. Specifically, they recommended CMS reassign CPT code 51720, which corresponds to the procedure used to administer the investigational product, to a non C-APC, such as a drug administration APC. ( printed page 93936)
Some commenters recommended that CMS extend the proposed policy to cell and gene therapies assigned to status indicator “G”, which correlates to pass-through status. Similarly, commenters recommend CMS exclude a variety of products from C-APC packaging, such as Casgevy, Lyfgenia, Amtagvi, Omisurge, Zynteglo, Obe-cel, Hemgenix, Beqvez, Roctavian, Spinraza and Elevidys. A few commenters suggested CMS should review and identify any other products that may be administered as independent therapies in the hospital outpatient setting and stated that such products should be separately paid.
Response: We greatly thank commenters for their detailed comments. For new cell and gene therapy products that are not integral, ancillary, supportive, dependent, or adjunctive to any C-APC primary service that were approved during CY 2024 and that continue to be approved, CMS will add their product specific HCPCS codes, when created, to the C-APC exclusion list. For example, HCPCS code J3392 (Injection, exagamglogene autotemcel, per treatment) effective October 1, 2024, which may be used to describe the product Casgevy, was added to the list of qualifying products. We also thank commenters for their suggestions to add additional drugs and classes of drugs to this C-APC exclusion policy as well as those drugs that are administered during a C-APC procedure. Additionally, we thank the commenter for its suggestion to move some codes that describe the administration of a product from a C-APC to a drug administration APC. We will take this feedback into consideration for future rulemaking. As we stated in the proposed rule, we welcomed comments from readers on the proposal and the potential need for a different, modified, expanded, or supplemental policy for future rulemaking. We believe that cell and gene therapies are a unique class of products, but we believe we need additional evidence and engagement from interested parties concerning whether other identified classes of drugs, biologicals, medical devices, or other products are not supportive of a primary C-APC service but may nonetheless appear on the same claim as a primary C-APC procedure. We strongly believe in the principles of our prospective payment system and the C-APC policy; therefore, we want to ensure than any potential future changes to our C-APC packaging policy maintain these principles.
Commenters suggested that we exclude several additional products from C-APC packaging, such as Lyfgenia, Zynteglo, Hemgenix, Beqvez, Roctavian, and Elevidys. We note that these products are currently on OPPS pass-through status for the duration of CY 2025, and therefore will be excluded from C-APC packaging under that policy. Similarly, in response to commenters suggestion that we apply this policy to cell and gene therapy products assigned to status indicator “G”, we note that we specifically did not propose to include therapies that are on drug pass-through status, represented by status indicator “G” for all of CY 2025 because pass-through drugs are already excluded from C-APC packaging. Please see section V.A of this final rule with comment period for additional information on OPPS drug pass-through status.
Comment: Several commenters recommended that CMS unpackage all status indicator “K” drugs from C-APC packaging, which aligned with the 2024 Advisory Panel on Hospital Outpatient Payment recommendation to no longer package drugs with a status indicator of “K” into any C-APC and, instead, they recommended CMS provide separate payment for all drugs and biologicals above the drug packaging threshold. Commenters said these status indicator “K” drugs should be paid based on 1847A of the Social Security Act and reimbursed at Average Sales Price plus 6 percent rather than packaged. Similarly, a commenter suggested that since drugs on pass-through status are paid separately when billed with a C-APC, that CMS should continue that separately payable status after passthrough status expiration and the transition of that code's status indicator from “G” to “K.”
Commenters suggested that the proportion of status indicator “K” drug charges relative to all other charges appearing on C-APCs claims was less than 2.5 percent across all C-APCs. Meaning, in commenters' views, that C-APCs do not inherently involve the provision of status indicator “K” drugs. Another commenter recommended that CMS exclude high-cost primary therapy drugs across drug classes and not only to cell and gene therapies, apply the C-APC exclusion to primary therapy drugs that clinically align to C-APCs, and they included specific examples they researched that serve as the primary therapy drugs similar to cell and gene therapies.
Response: We appreciate the information submitted by commenters, but respectfully disagree with their conclusions. The C-APC policy aims to package payment for items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to the primary service and provided during the delivery of the comprehensive service, including diagnostic procedures, laboratory tests and other diagnostic tests and treatments that assist in the delivery of the primary procedure. We believe it is appropriate for most drugs and biologicals to be packaged into C-APCs as we believe they function to “promote a beneficial outcome” or “prevent possible complications” of C-APC services. We explained in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74868) that intravenous drugs, for example, are OPPS services that are considered adjunctive to the primary procedure because the correct administration of the drug either promotes a beneficial outcome, such as the use of intravenous pain medications, or prevents possible complications, such as the use of intravenous blood pressure medications to temporarily replace oral blood pressure medications and reduce the risk of a sudden rise in blood pressure when a normal daily medication is stopped. We are therefore not excluding all status indicator “K” drugs above the drug packaging threshold from C-APC packaging. Our packaging policies are a fundamental component of the OPPS and they support our strategic goal of using larger payment bundles in the OPPS to maximize hospitals' incentives to provide care in the most efficient manner. Additionally, we do not believe at this time that cost alone is a reason to exclude a product from packaging.
Comment: Many commenters responded to several of our requests for comment associated with the proposal. Commenters recommended no significant overall changes to the general methodology CMS proposed to exclude products from all C-APC packaging and pay separately based on the individual product's average sales price plus 6 percent. Specifically, commenters strongly conveyed that they did not see an appropriate reason for CMS to structure a new C-APC, or similar packaged payment policy, for the service to administer cell or gene therapies, such as by creating a CAR T-cell therapy administration C-APC. Commenters believed that there is no accurate way to create a bundled payment for a whole group of cell and gene therapies as the costs and resources required are unique and novel between each of the therapies, which also makes it difficult to determine appropriate items and services associated with each therapy for inclusion in C-APC packaging. Commenters broadly believed no ( printed page 93937) different or supplemental policy was warranted given the numerous challenges providers face related to cell and gene therapies currently. They saw any additional change to move towards a packaged payment policy as an additional challenge for the hospitals providing the therapies.
Similarly, commenters cautioned CMS against creating C-APCs for cell and gene therapies as this may cause inadequate Medicare reimbursement to hospitals, which, in their view, has the potential to jeopardize beneficiary access to CAR T-cell therapy and other high-cost therapies as commenters communicated that institutions weigh reimbursement challenges with their ability to provide this costly care. Commenters were concerned that creating a new packaged payment policy could lead to a financial loss and decrease the number of facilities offering high-cost cell and gene therapies in the hospital outpatient department.
Commenters emphasized that these therapies involve highly specialized procedures, intensive monitoring, and multidisciplinary care teams, all of which contribute to their substantial costs. Commenters were concerned by the fact that the administration of cell and gene therapies are multi-step processes that can take weeks if not months and entail multiple services that would not appear on the same claim. Additional commenters reiterated that all of the various and distinct clinical services for CAR T-cell therapy are furnished over multiple encounters on different dates—making CAR T-cell therapy inappropriate for C-APCs, which are typically specific to a single encounter in their view. Commenters believed a CAR T-cell therapy C-APC, or similar packaged payment policy, would be unprecedented and a huge policy departure for CMS and recommended CMS not pursue the idea any further.
Many commenters believed that separate payment based on average sales price plus 6 percent is the most appropriate payment methodology for cell and gene therapies due to its transparency, uniformity and predictability. A few commenters pointed out that current hospital cost data is lower than ASP for CAR T-cell therapy products, potentially leaving products underpaid if paid based on C-APC principles. Commenters similarly took the position that there is no appropriate method in which CMS could modify its outlier payment policy with respect to C-APCs to better pay for these types of products.
Response: We appreciate the insights provided by commenters and will take them into consideration for future rulemaking. At this time, we are not considering the creation of a new C-APC, similar packaged payment policy, or modified outlier policy, for services to administer cell or gene therapies.
After consideration of the public comments we received, we are finalizing our policy proposal with a modification with respect to how long this policy will apply. For CY 2025 and subsequent years, we are finalizing a policy not to package payment for cell and gene therapies into C-APCs, when those cell and gene therapies are not functioning as integral, ancillary, supportive, dependent, or adjunctive to the primary C-APC service. For new cell and gene therapy products that are not integral, ancillary, supportive, dependent, or adjunctive to any C-APC primary service, we will continue to add their product specific HCPCS codes, when created, to the C-APC exclusion list. The current list of qualifying products can be found in Table 4. We are not adding any additional drug classes to this policy exclusion at this time, and we are not currently considering any additional modifications to our policy, but will continue to consider additional refinements for future rulemaking.
We list all final C-APC exclusion categories for CY 2025 in Addendum J to this final rule (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
( printed page 93938)(5) Exclusion of Non-Opioid Products for Pain Relief Under Section 4135 of the Consolidated Appropriations Act, 2023 From the C-APC Policy
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), was signed into law on December 29, 2022. Section 4135(a) and (b) of the CAA, 2023, titled “Access to Non-Opioid Treatments for Pain Relief,” amended section 1833(t)(16) and section 1833(i) of the Social Security Act, respectively, to provide for temporary additional payments for non-opioid treatments for pain relief (as that term is defined in section 1833(t)(16)(G)(i) of the Act). In particular, section 1833(t)(16)(G) provides that with respect to a non-opioid treatment for pain relief furnished on or after January 1, 2025, ( printed page 93939) and before January 1, 2028, the Secretary shall not package payment for the non-opioid treatment for pain relief into payment for a covered OPD service (or group of services) and shall make an additional payment for the non-opioid treatment for pain relief as specified in clause (ii) of that section. Clauses (ii) and (iii) of section 1833(t)(16)(G) of the Act provide for the amount of additional payment and set a limitation on that amount. As stated earlier in this section, our current policy is to exclude from the packaged C-APC payment those items and services that are required by statute to be separately paid.
Accordingly, we proposed to exclude the non-opioid treatments for pain relief identified as satisfying the required criteria for payment under section 4135 of the CAA, 2023 from the C-APC policy to ensure payment is not packaged into any C-APC and that separate payment is made in accordance with the statute.
Comment: Commenters were generally supportive of our proposal not to package payment and to pay separately for qualifying drugs, biologicals, and medical devices that met the criteria for payment under section 4135 of the CAA, 2023. A commenter mentioned its support of CMS packaging policies, and noted reservations about paying separately, and not packaging, non-opioid treatments for pain relief; however, they ultimately conceded that CMS must implement section 4135 of the CAA, 2023, which requires separately payable status, and supported CMS's implementation methodology.
Response: We thank commenters for their support.
After consideration of the public comments we received, we are finalizing our policy as proposed, and will exclude the non-opioid treatments for pain relief identified as satisfying the required criteria for payment under section 4135 of the CAA, 2023 from the C-APC policy to ensure payment is not packaged into any C-APC and that separate payment is made in accordance with the statute. Please see section XIII.F of this final rule with comment period for a list of the products that we are finalizing would qualify for payment under the new payment policy for non-opioid drugs, biologicals, and devices for pain relief.
(6) C-APCs for CY 2025
For CY 2025 and subsequent years, we proposed to continue to apply the C-APC payment policy methodology. We refer readers to the CY 2017 OPPS/ASC final rule with comment period (81 FR 79583) for a discussion of the C-APC payment policy methodology and revisions.
Each year, in accordance with section 1833(t)(9)(A) of the Act, we review and revise the services within each APC group and the APC assignments under the OPPS. As a result of our annual review of the services and the APC assignments under the OPPS, we did not propose to convert any standard APCs to C-APCs in CY 2025; thus, we proposed that the number of C-APCs for CY 2025 would be the same as the number for CY 2024, which is 72 C-APCs.
Comment: A few commenters requested that CMS reconsider our packaging policies for C-APC 8011 (Comprehensive Observation Services). They requested that CMS remove the rule that the presence of a SI “T” procedure on a claim excludes payment of C-APC 8011 and instead package the payment of the SI “T” procedure into C-APC 8011, as is already done with SI “Q” procedures. Commenters stated that this requirement violates the basic tenet of the packaging concept in that when observation services are ordered and furnished, the observation services become the primary service provided to such patients and the SI “T” procedure is provided ancillary to that primary service. Commenters cited scenarios in which hospitals provide significant, resource-intensive services to a patient but are paid significantly less than if a SI “T” procedure was not done.
Response: We thank the commenters for bringing this to our attention. In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70334-70336), in response to commenters' concerns regarding packaging payment for potentially high-cost surgical procedures into the payment for an observation C-APC, we finalized a policy that claims reporting procedures assigned status indicator “T” do not qualify for payment through C-APC 8011, regardless of whether the procedure assigned status indicator “T” was furnished before or after observation services (described by HCPCS code G0378) were provided. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79562), we stated that services that would otherwise qualify for C-APC 8011 are not considered to be observation services when they are associated with a surgical procedure (assigned to status indicator “T”). Instead, they are considered to be perioperative recovery, which is always packaged in with the surgical procedure. We will continue to review the impacts of this issue and may revisit it in future rulemaking.
Comment: Two commenters expressed concerns with the C-APC methodology for surgical insertion codes for brachytherapy treatment, stating that these concerns impact beneficiary access to brachytherapy in the HOPD setting. These commenters stated that the C-APC methodology lacks the appropriate charge capture mechanisms to accurately reflect the services associated with the C-APC, that there are significant variations in the clinical practice and billing patterns in the hospital claims data used for ratesetting, and that the C-APC rates do not accurately or fully reflect the services and costs associated with the primary procedure. Commenters urged the agency to explore alternatives, including that CMS discontinue the C-APC policy for all brachytherapy insertion codes. Alternatively, one commenter suggested that CMS could continue to pay for “J1” brachytherapy insertion codes under the C-APC payment methodology but exclude and make separate payment for designated preparation and planning services in addition to the C-APC payment. Another commenter called for education on whether services, like brachytherapy, that are assigned to a J1 indicators and delivered over multiple patient encounters may be reported per encounter.
Response: We appreciate the comments on the C-APC methodology. However, we believe that the current C-APC methodology is appropriately applied to surgical insertion for brachytherapy treatment procedures and is accurately capturing costs, particularly as the brachytherapy sources used for these procedures are excluded from C-APC packaging and are separately payable. We will evaluate if provider education may be appropriate in this circumstance. We will continue to examine these concerns and will determine if any modifications on this policy are warranted in future rulemaking.
After consideration of the public comments we received, we are finalizing the C-APCs as proposed. Table 5 lists the final C-APCs for CY 2025. All C-APCs are displayed in Addendum J to this CY 2025 OPPS/ASC final rule with comment period (which is available via the internet on the CMS website). Addendum J to this final rule with comment period also contains all of the data related to the C-APC payment policy methodology, including the list of complexity adjustments and other information for CY 2025.
( printed page 93940) ( printed page 93941)C-APC Clinical Family Descriptor Key:
AENDO = Airway Endoscopy
AICDP = Automatic Implantable Cardiac Defibrillators, Pacemakers, and Related Devices.
BREAS = Breast Surgery
COCHL = Cochlear Implant
EBIDX = Excision/Biopsy/Incision and Drainage
ENTXX = ENT Procedures
EPHYS = Cardiac Electrophysiology/
EVASC = Endovascular Procedures
EXEYE = Extraocular Ophthalmic Surgery
GIXXX = Gastrointestinal Procedures
GYNXX = Gynecologic Procedures
INEYE = Intraocular Surgery
LAPXX = Laparoscopic Procedures
NERVE = Nerve Procedures
NSTIM = Neurostimulators
ORTHO = Orthopedic Surgery
PUMPS = Implantable Drug Delivery Systems
RADTX = Radiation Oncology
SCTXX = Stem Cell Transplant ( printed page 93942)
UROXX = Urologic Procedures
VASCX = Vascular Procedures
WPMXX = Wireless PA Pressure Monitor
c. Calculation of Composite APC Criteria-Based Costs
As discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66613), we believe it is important that the OPPS enhance incentives for hospitals to provide necessary, high-quality care as efficiently as possible. For CY 2008, we developed composite APCs to provide a single payment for groups of services that are typically performed together during a single clinical encounter and that result in the provision of a complete service. Combining payment for multiple, independent services into a single OPPS payment in this way enables hospitals to manage their resources with maximum flexibility by monitoring and adjusting the volume and efficiency of services themselves. An additional advantage to the composite APC model is that we can use data from correctly coded multiple procedure claims to calculate payment rates for the specified combinations of services, rather than relying upon single procedure claims which may be low in volume and/or incorrectly coded. Under the OPPS, we currently have composite policies for mental health services and multiple imaging services. We refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66611 through 66614 and 66650 through 66652) for a full discussion of the development of the composite APC methodology, and the CY 2012 OPPS/ASC final rule with comment period (76 FR 74163) and the CY 2018 OPPS/ASC final rule with comment period (82 FR 59241, 59242, and 59246 through 52950) for more recent background.
(1) Mental Health Services Composite APC
For CY 2025, we proposed to continue our longstanding policy of limiting the aggregate payment for specified less resource-intensive mental health services furnished on the same date to the payment for a day of partial hospitalization services provided by a hospital, which we consider to be the most resource-intensive of all outpatient mental health services (88 FR 49572). We refer readers to the April 7, 2000, OPPS final rule with comment period (65 FR 18452 through 18455) for the initial discussion of this longstanding policy and the CY 2012 OPPS/ASC final rule with comment period (76 FR 74168) for more recent background.
In the CY 2018 OPPS/ASC proposed rule and final rule with comment period (82 FR 33580 and 33581 and 82 FR 59246 and 59247), we proposed and finalized the policy for CY 2018 and subsequent years that, when the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, exceeds the maximum per diem payment rate for partial hospitalization services provided by a hospital, those specified mental health services will be paid through composite APC 8010 (Mental Health Services Composite). In addition, we set the payment rate for composite APC 8010 for CY 2018 at the same payment rate for APC 5863, which was the maximum partial hospitalization per diem payment rate for a hospital, and finalized a policy that the hospital would continue to be paid the payment rate for composite APC 8010. This policy applied in CYs 2018 through 2023.
In the CY 2024 OPPS/ASC proposed rule, we stated that APC 5863 was no longer the maximum partial hospitalization per diem payment rate for a hospital due to the creation of APC 5864, which is four or more hospital-based PHP services per day (88 FR 49572). We solicited comment on whether APC 5864 would be appropriate to use as the daily mental health cap, as we have historically set the daily mental health cap for composite APC 8010 at the maximum partial hospitalization per diem payment rate for a hospital (88 FR 49572). Based on public comments received and our longstanding policy, in CY 2024 OPPS/ASC final rule, we finalized APC 5864, four hospital-based PHP services per day, as the daily mental health cap (88 FR 81566).
We continue to believe that the costs associated with administering a partial hospitalization program represent the most resource intensive of all outpatient mental health services. For CY 2025 and subsequent years, we proposed to continue this policy that when the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, exceeds the per diem payment rate for 4 partial hospitalization services provided in a day by a hospital (the payment amount for APC 5864), those specified mental health services would be paid through composite APC 8010. In addition, we proposed to continue to set the payment rate for composite APC 8010 at the same payment rate that we propose for APC 5864, which is a partial hospitalization per diem payment rate for 4 partial hospitalization services furnished in a day by a hospital.
Under the proposed policy, the Integrated OCE (I/OCE) would continue to determine whether to pay for these specified mental health services individually, or to make a single payment at the same payment rate established for APC 5864 for all the specified mental health services furnished by the hospital on that single date of service by paying for the services through composite APC 5863.
Comment: One commenter recommended that CMS change the status indicator for two neuropsychological testing codes (HCPCS codes 96133 and 96137) from SI = N to SI = Q3 to allow separate payment for additional hours of testing or increase the payment rate for the primary testing procedure codes. Noting the multiple day structure of neuropsychological testing, they requested a daily payment for each date of service. The commenters noted that the payment rate for Composite APC 8010, which is capped at the maximum per diem partial hospitalization rate, is lower than the individual HCPCS code APC payment rates and does not provide sufficient payment for these procedures.
Response: After reviewing this issue, we believe the Composite APC methodology is being appropriately applied in this case. Composite APCs provide a single payment for groups of services that are typically performed together during a single clinical encounter and that result in the provision of a complete service.
The words “list separately in addition to code for primary procedure” are included in the long descriptors for HCPCS codes 96133 and 96137 to indicate that that the codes are considered “add-ons” to another primary code that cannot be reported independently. Specifically, add-on codes must always be reported with another primary code on the same day. The AMA states in the CPT 2024 Professional Edition (page xviii) that “add-on codes are always performed in addition to the primary service or procedure and must never be reported as a stand-alone code.” In most cases, add-on codes are typically ancillary and supportive to a primary diagnostic or therapeutic modality and are an integral part of the primary service they support. As specified under 42 CFR 419.2(b)(18), certain services described by add-on codes are generally packaged under the OPPS, and payments for the codes are bundled with the primary codes. ( printed page 93943) Consequently, HCPCS codes 96133 and 96137 are not paid separately under the OPPS, but instead, their payment is packaged into the primary code.
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59246 through 59247), we stated that the costs associated with administering a partial hospitalization program at a hospital represent the most resource intensive of all outpatient mental health services. We continue to believe that the Composite APC methodology, which limits the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, to the per diem payment rate for four partial hospitalization services provided in a day by a hospital (the payment amount for APC 5864), is appropriate.
We will continue to examine these concerns and will determine if any modifications to this policy are warranted in future rulemaking.
After consideration of the public comments we received, we are finalizing our proposal, without modification, that when the aggregate payment for specified mental health services provided by one hospital to a single beneficiary on a single date of service, based on the payment rates associated with the APCs for the individual services, exceeds the maximum per diem payment rate for four partial hospitalization services provided in a day by a hospital (the payment amount for APC 5864), those specified mental health services would be paid through composite APC 8010 for CY 2025. In addition, we are finalizing setting the payment rate for composite APC 8010 for CY 2025 at the same payment rate that we set for APC 5864, which is the maximum partial hospitalization per diem payment rate for a hospital.
(2) Multiple Imaging Composite APCs (APCs 8004, 8005, 8006, 8007, and 8008)
Effective January 1, 2009, we provide a single payment each time a hospital submits a claim for more than one imaging procedure within an imaging family on the same date of service, to reflect and promote the efficiencies hospitals can achieve when performing multiple imaging procedures during a single session (73 FR 41448 through 41450). We utilize three imaging families based on imaging modality for purposes of this methodology: (1) ultrasound; (2) computed tomography (CT) and computed tomographic angiography (CTA); and (3) magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA). The HCPCS codes subject to the multiple imaging composite policy and their respective families are listed in Table 6.
While there are three imaging families, there are five multiple imaging composite APCs due to the statutory requirement under section 1833(t)(2)(G) of the Act that we differentiate payment for OPPS imaging services provided with and without contrast. While the ultrasound procedures included under the policy do not involve contrast, both CT/CTA and MRI/MRA scans can be provided either with or without contrast. The five multiple imaging composite APCs established in CY 2009 are:
- APC 8004 (Ultrasound Composite);
- APC 8005 (CT and CTA without Contrast Composite);
- APC 8006 (CT and CTA with Contrast Composite);
- APC 8007 (MRI and MRA without Contrast Composite); and
- APC 8008 (MRI and MRA with Contrast Composite).
We define the single imaging session for the “with contrast” composite APCs as having at least one or more imaging procedures from the same family performed with contrast on the same date of service. For example, if the hospital performs an MRI without contrast during the same session as at least one other MRI with contrast, the hospital will receive payment based on the payment rate for APC 8008, the “with contrast” composite APC.
We make a single payment for those imaging procedures that qualify for payment based on the composite APC payment rate, which includes any packaged services furnished on the same date of service. The standard (noncomposite) APC assignments continue to apply for single imaging procedures and multiple imaging procedures performed across families. For a full discussion of the development of the multiple imaging composite APC methodology, we refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68559 through 68569).
For CY 2025, we proposed to continue to pay for all multiple imaging procedures within an imaging family performed on the same date of service using the multiple imaging composite APC payment methodology. We continue to believe that this policy would reflect and promote the efficiencies hospitals can achieve when performing multiple imaging procedures during a single session.
For CY 2025, except where otherwise indicated, we proposed to use the costs derived from CY 2023 claims data to set the proposed CY 2025 payment rates. Therefore, for CY 2025, the proposed payment rates for the five multiple imaging composite APCs (APCs 8004, 8005, 8006, 8007, and 8008) were based on proposed geometric mean costs calculated from CY 2023 claims available for the CY 2025 OPPS/ASC proposed rule that qualify for composite payment under the current policy (that is, those claims reporting more than one procedure within the same family on a single date of service). To calculate the proposed geometric mean costs, we used the same methodology that we used to calculate the geometric mean costs for these composite APCs since CY 2014, as described in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74918). The imaging HCPCS codes referred to as “overlap bypass codes” that we removed from the bypass list for purposes of calculating the proposed multiple imaging composite APC geometric mean costs, in accordance with our established methodology as stated in the CY 2014 OPPS/ASC final rule with comment period (78 FR 74918), are identified by asterisks in Addendum N to this final rule with comment period (which is available via the internet on the CMS website) and are discussed in more detail in section II.A.1.a of the this final rule with comment period.
For this final rule with comment period, we were able to identify approximately 0.95 million “single session” claims out of an estimated 2.1 million potential claims for payment through composite APCs from our ratesetting claims data, which represents approximately 45.0 percent of all eligible claims, to calculate the final CY 2025 geometric mean costs for the multiple imaging composite APCs. Table 6 lists the final HCPCS codes that would be subject to the multiple imaging composite APC policy and their respective families and approximate composite APC final geometric mean costs for CY 2025.
We did not receive any public comments on this policy. We are finalizing without modification our proposal to continue the use of multiple imaging composite APCs to pay for the provision of more than one imaging procedure from the same imaging family on the same date. Table 6 lists the HCPCS codes that will be subject to the multiple imaging composite APC policy and their respective imaging families and approximate composite APC final geometric mean costs for CY 2025.
( printed page 93944) ( printed page 93945) ( printed page 93946) ( printed page 93947)3. Changes to Packaged Items and Services
a. Background and Rationale for Packaging in the OPPS
Like other prospective payment systems, the OPPS relies on the concept of averaging to establish a payment rate for services. The payment may be more or less than the estimated cost of providing a specific service or a bundle of specific services for a particular beneficiary. The OPPS packages payments for multiple interrelated items ( printed page 93948) and services into a single payment to create incentives for hospitals to furnish services most efficiently and to manage their resources with maximum flexibility. Our packaging policies support our strategic goal of using larger payment bundles in the OPPS to maximize hospitals' incentives to provide care in the most efficient manner. For example, where there are a variety of devices, drugs, items, and supplies that could be used to furnish a service, some of which are more costly than others, packaging encourages hospitals to use the most cost-efficient item that meets the patient's needs, rather than to routinely use a more expensive item, which may occur if separate payment is provided for the item.
Packaging also encourages hospitals to effectively negotiate with manufacturers and suppliers to reduce the purchase price of items and services or to explore alternative group purchasing arrangements, thereby encouraging the most economical health care delivery. Similarly, packaging encourages hospitals to establish protocols that ensure that necessary services are furnished, while scrutinizing the services ordered by practitioners to maximize the efficient use of hospital resources. Packaging payments into larger payment bundles promotes the predictability and accuracy of payment for services over time. Finally, packaging may reduce the importance of refining service-specific payments because packaged payments include costs associated with higher cost cases requiring many ancillary items and services and lower cost cases requiring fewer ancillary items and services. Packaging encourages efficiency and is an essential component of a prospective payment system, therefore packaging payments for items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to a primary service has been a fundamental part of the OPPS since its implementation in August 2000. As we continue to develop larger payment groups that more broadly reflect services provided in an encounter or episode of care, we have expanded the OPPS packaging policies. Most, but not necessarily all, categories of items and services currently packaged in the OPPS are listed in 42 CFR 419.2(b). Our overarching goal is to make payments for all services under the OPPS more consistent with those of a prospective payment system and less like those of a per-service fee schedule, which pays separately for each coded item. As a part of this effort, we have continued to examine the payment for items and services provided under the OPPS to determine which OPPS services can be packaged to further achieve the objective of advancing the OPPS toward a more prospective payment system.
b. Final CY 2025 Policy on Packaged Items and Services
For CY 2025, we examined the items and services currently provided under the OPPS, reviewing categories of integral, ancillary, supportive, dependent, or adjunctive items and services for which we believe payment would be appropriately packaged into payment for the primary service that they support. Specifically, we examined the HCPCS code definitions (including CPT code descriptors) and hospital outpatient department billing patterns to determine whether there were categories of codes for which packaging would be appropriate according to existing OPPS packaging policies or a logical expansion of those existing OPPS packaging policies.
For CY 2025, we did not propose any changes to the overall packaging policy discussed. We proposed to continue to conditionally package the costs of selected newly identified ancillary services into payment for a primary service where we believe that the packaged item or service is integral, ancillary, supportive, dependent, or adjunctive to the provision of care that was reported by the primary service HCPCS code.
While we did not propose any changes to the overall packaging policy, we proposed potential modifications to our packaging policy as described in the following sections.
We did not receive any public comments on our overall packaging policy proposal and therefore, we are finalizing our proposal to continue to conditionally package the costs of selected newly identified ancillary services into payment for a primary service where we believe that the packaged item or service is integral, ancillary, supportive, dependent, or adjunctive to the provision of care that was reported by the primary service HCPCS code, as proposed for CY 2025.
We did receive specific recommendations regarding C-APC packaging of Cell and Gene Therapies and associated products, which are addressed in section II.b.4 of this final rule with comment period, and the packaging of non-opioid treatments for pain relief, which are addressed in section XIII.E of this final rule with comment period. Additionally, commenters made recommendations on our packaging policies in the context of our diagnostic radiopharmaceutical proposal, which is discussed in the next section.
c. Payment for Diagnostic Radiopharmaceuticals
(1) Background on OPPS Packaging Policy for Diagnostic Radiopharmaceuticals
Under the OPPS, we package several categories of nonpass-through drugs, biologicals, and radiopharmaceuticals, regardless of the cost of the products. Because the products are packaged according to the policies in § 419.2(b), we refer to them as “policy-packaged” drugs, biologicals, and radiopharmaceuticals. In particular, under § 419.2(b)(15), payment for drugs, biologicals, and radiopharmaceuticals that function as supplies when used in a diagnostic test or procedure is packaged with the payment for the related procedure or service. Packaging costs into a single aggregate payment for a service, encounter, or episode of care is a fundamental principle that distinguishes a prospective payment system from a fee schedule. In general, packaging the costs of supportive items and services into the payment for the primary procedure or service with which they are associated encourages hospital efficiencies and enables hospitals to manage their resources with maximum flexibility.
In the CY 2008 OPPS/ASC final rule with comment period we finalized the packaging status of diagnostic radiopharmaceuticals as part of our overall enhanced packaging approach for the CY 2008 OPPS and subsequent years (72 FR 66635 through 66641). Importantly, we believed diagnostic radiopharmaceuticals are always intended to be used with a diagnostic nuclear medicine procedure and function as supplies when used in a diagnostic test or procedure, making it appropriate to package the payment for the diagnostic radiopharmaceutical into the payment for the related nuclear medicine procedure. Diagnostic radiopharmaceuticals are one specific type of product that is policy packaged under the category described by § 419.2(b)(15). Since we implemented this policy in CY 2008, interested parties have raised concerns regarding policy packaging of diagnostic radiopharmaceuticals. In previous rulemaking (87 FR 71962 and 71963), commenters recommended that CMS always pay separately under the OPPS for diagnostic radiopharmaceuticals, not just when the products have pass-through payment status. Many of these commenters mentioned that pass- ( printed page 93949) through payment status helps the diffusion of new diagnostic radiopharmaceuticals into the market. However, commenters believe the packaged payment rate is often inadequate after pass-through status expires, especially in cases where the diagnostic radiopharmaceutical is high-cost and has low utilization.
We have heard from interested parties regarding alternative payment methodologies, such as subjecting diagnostic radiopharmaceuticals to the drug packaging threshold and creating separate APC payments for diagnostic radiopharmaceuticals with a per-day cost greater than $500. Interested parties have also recommended that we analyze our nuclear medicine APC structure and consider establishing additional nuclear medicine APCs to reflect the costs of diagnostic radiopharmaceuticals more accurately. Historically, commenters opposed incorporating the cost of diagnostic radiopharmaceuticals into the associated nuclear medicine APC as the nuclear medicine APCs are sometimes paid at a lower rate than the payment rate for the diagnostic radiopharmaceutical itself when it has pass-through payment status (87 FR 71962 and 71963).
Importantly, commenters historically have also been concerned that packaging payment for precision diagnostic radiopharmaceuticals in the outpatient hospital setting creates barriers to beneficiary access for safety net hospitals serving a high proportion of Medicare beneficiaries and hospitals serving underserved communities (87 FR 71962 and 71963). Commenters specified that certain populations, such as those with Alzheimer's disease, depend on the use of certain high-cost diagnostic radiopharmaceuticals. Commenters discussed difficulties enrolling hospitals in clinical studies due to OPPS packaging policies and suggested that we pay separately under the OPPS specifically for radiopharmaceuticals that are used for Alzheimer's disease. Additionally, commenters have recommended that CMS continue to apply radiolabeled product edits to the nuclear medicine procedures to ensure that all packaged costs are included on nuclear medicine claims to establish appropriate payment rates in the future. Beginning January 1, 2008, CMS implemented OPPS edits that require hospitals to include a HCPCS code for a radiolabeled product when a separately payable nuclear medicine procedure is present on a claim. This policy to require hospitals to include a HCPCS code for a radiolabeled product for a separately payable nuclear medicine procedure ended in CY 2014 (78 FR 75033 through 75034).
Many of these comments and our responses have been discussed in rulemaking since the policy to package diagnostic radiopharmaceuticals was adopted, and they prompted us to solicit comment on payment for diagnostic radiopharmaceuticals in the CY 2024 OPPS/ASC proposed rule (88 FR 49577). In that proposed rule, we stated we continue to believe that diagnostic radiopharmaceuticals are an integral component of many nuclear medicine and imaging procedures and charges associated with them should be reported on hospital claims to the extent they are used. Accordingly, we reiterated our belief that the payment for the diagnostic radiopharmaceutical should be reflected within the payment for the primary procedure with which it is used. We noted that ratesetting uses the geometric mean of reported procedure costs, which in the example of nuclear medicine procedures includes the costs of the reported diagnostic radiopharmaceutical, based on data submitted to CMS from all hospitals paid under the OPPS to set the payment rate for the service. The costs that are calculated by Medicare reflect the average costs of items and services that are packaged into a primary procedure and will not necessarily equal the sum of the cost of the primary procedure and the Average Sales Price (ASP) of the specific items and services used in the procedure in each case. Furthermore, we explained that the costs are based on the reported costs submitted to Medicare by the hospitals and not the list price established by the manufacturer. Claims data that include the diagnostic radiopharmaceutical packaged with the associated procedure should reflect the combined cost of the procedure and the radiopharmaceutical used in the procedure.
As we have reiterated over the years, we believe packaging policies are inherent principles of the OPPS and are essential to a prospective payment system. At the same time, we have explained that we are committed to ensuring beneficiary access to diagnostic radiopharmaceuticals while also ensuring the availability of new and innovative diagnostic tools for Medicare beneficiaries. Therefore, we sought public comments on potential modifications to our packaging policy for diagnostic radiopharmaceuticals to ensure equitable payment and continued beneficiary access.
As described in the CY 2024 OPPS/ASC proposed rule (88 FR 49578), we solicited comment on how the OPPS packaging policy for diagnostic radiopharmaceuticals has impacted beneficiary access, including whether there are specific patient populations or clinical disease states for whom this issue is especially critical.
In addition, we solicited comment on the following potential approaches that would enhance beneficiary access, while also maintaining the principles of the outpatient prospective payment system. These approaches included: (1) paying separately for diagnostic radiopharmaceuticals with per-day costs above the OPPS drug packaging threshold of $140; (2) establishing a specific per-day cost threshold that may be greater or less than the OPPS drug packaging threshold; (3) restructuring APCs, including by adding nuclear medicine APCs for services that utilize high-cost diagnostic radiopharmaceuticals; (4) creating specific payment policies for diagnostic radiopharmaceuticals used in clinical trials; and (5) adopting codes that incorporate the disease state being diagnosed or a diagnostic indication of a particular class of diagnostic radiopharmaceuticals.
Finally, we were interested in hearing from stakeholders how the suggested policy modifications might impact our overarching goal of utilizing packaging policies to better align OPPS policies with those of a prospective payment system rather than a fee schedule. We stated we would also like to know if making any of the suggested policy changes could have negative consequences for beneficiaries, such as unintentionally influencing clinical practice decisions, increasing beneficiary cost-sharing obligations, or inadvertently encouraging the use of higher-cost diagnostic radiopharmaceuticals over lower cost, but equally effective, diagnostic options.
We received a significant number of comments in response to the comment solicitation on potential issues caused by our current payment policy for diagnostic radiopharmaceuticals under the OPPS and on new approaches to payment for these products. Commenters expressed concerns regarding the CMS policy to package diagnostic radiopharmaceuticals and the financial implications this policy has for facilities. Commenters believe that, for newer, more innovative radiopharmaceuticals, the current OPPS packaging policy has led to a lack of patient access to the technologies after the radiopharmaceutical's pass-through status expires, especially if there is no clinical alternative to the radiopharmaceutical. Most commenters requested that CMS provide separate payment for diagnostic ( printed page 93950) radiopharmaceuticals. Some commenters believed paying separately for all diagnostic radiopharmaceuticals regardless of their per-day cost was the best methodology to avoid encouraging price inflation for diagnostic radiopharmaceuticals to reach a certain threshold. Other commenters thought that applying the existing OPPS per-day cost threshold ($135 for CY 2024) to the payment of diagnostic radiopharmaceuticals would be an adequate solution. Others supported a $500 threshold, and many cited the Facilitating Innovative Nuclear Diagnostics Act (FIND Act) of 2023 as their rationale for that number and recognized that the $500 threshold number may be a more targeted approach relative to the OPPS drug packaging threshold as the higher cost diagnostic radiopharmaceuticals are the most disadvantaged by the OPPS packaging policy in their view. For the full discussion on the comment solicitation summarized here, refer to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81573 through 81577).
(2) Packaging Threshold for Diagnostic Radiopharmaceuticals
As stated in the CY 2024 OPPS final rule with comment period (88 FR 81577), we agree with commenters that payment for diagnostic radiopharmaceuticals is a complex and important issue. We explained that we intended to further consider these points and take them into consideration for future notice and comment rulemaking. As described in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), after significant consideration and ongoing engagement from interested parties, we proposed a change to our current policy that packages diagnostic radiopharmaceuticals regardless of their cost.
We continue to believe diagnostic radiopharmaceuticals are always intended to be used with a diagnostic nuclear medicine procedure and function as supplies when used in a diagnostic test or procedure, generally making it appropriate to package payment for them with payment for the related nuclear medicine procedure. In the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we stated that while we continue to believe that this should be the policy for most diagnostic radiopharmaceuticals, we believe there are certain situations in which the packaged payment amount attributed to the diagnostic radiopharmaceutical used in an imaging procedure assigned to a nuclear medicine APC may not adequately account for the cost of a diagnostic radiopharmaceutical that has a significantly higher cost, but lower utilization relative to the other diagnostic radiopharmaceuticals that may be used with the procedure. In situations where a hospital may have to pay significantly more to purchase a diagnostic radiopharmaceutical than Medicare pays, a hospital may decide not to provide that specific diagnostic radiopharmaceutical imaging agent to Medicare beneficiaries. This could potentially deny access to diagnostic tools for which there is no clinical alternative. To ensure Medicare payment policy is not providing a financial disincentive to using high cost, low utilization diagnostic radiopharmaceuticals, especially when those agents may be the most clinically appropriate, and to ensure appropriate beneficiary access, we believe a subset of diagnostic radiopharmaceuticals with higher per day costs should be paid separately and not packaged into the diagnostic procedure with which the diagnostic radiopharmaceutical is used.
To address these concerns, we proposed to pay separately for any diagnostic radiopharmaceutical with a per day cost greater than $630. Any diagnostic radiopharmaceutical with a per day cost below that threshold would continue to be policy packaged under the current policy at § 419.2(b)(15). We discuss our proposed methodology for determining the proposed per day cost threshold of $630 in further detail in this section.
To determine an appropriate threshold for our proposal, we estimated the approximate payment that would typically be attributable to diagnostic radiopharmaceutical payment within each nuclear medicine APC (APCs 5591, 5592, 5593, and 5594). We did this by assessing the offsets associated with these APCs that were directly attributable to “policy packaged” drugs. The offset amounts used are correlated with the approximate portion of APC payment associated with these “policy packaged” drugs. For nuclear medicine APCs, the primary “policy packaged” drugs are diagnostic radiopharmaceuticals. To calculate this threshold, we calculated a volume weighted average of the offset dollar amount of each nuclear medicine APC. This involved taking the offset percentage for “policy packaged” drugs, multiplying it by the APC geometric mean to get an offset dollar amount, and then multiplying that offset amount by the number of single claims to get the total offset amount for each nuclear medicine APC level. We then calculated the sum of the total offset amount for all 4 of the nuclear medicine APCs. We divided this number by the total number of single claims for all 4 nuclear medicine APCs, resulting in $314.28, which represents the volume weighted average policy packaged offset amount for the nuclear medicine APC series. We then took that number and multiplied it by 2, and rounded it to the nearest $5 increment, which resulted in $630. See Table 7 for the values used to calculate this threshold amount. We noted that the data values in Table 7 were collected without unpackaging the set of diagnostic radiopharmaceuticals listed in Table 8. We noted that if we finalized our proposal and those diagnostic radiopharmaceuticals were unpackaged, it would change the APC geometric mean unit costs (MUCs) as well as the offset percentages. This is why the APC geometric mean cost values listed below are not the same as in the addenda to this rule.
( printed page 93951)The offset percentages used were updated based on the available data for the CY 2025 proposed rule and these offset percentages are usually the offsets that are published in the final rule. However, we included the data in Table 7 to illustrate how we arrived at the proposed $630 threshold. Additionally, the file and corresponding offset percentages used for the $630 threshold are similar to and consistent with the ones that can be found in the CY 2024 NFRM APC Offset File. These files are available via the internet on the CMS OPPS website.[7]
We proposed to multiply by two the volume weighted average amount of the offset to establish the threshold triggering separate payment because this amount would ensure that separate payment would apply only to diagnostic radiopharmaceuticals whose costs significantly exceed the approximate amount of payment already attributed to the product in the nuclear medicine APC payment. This is consistent with the principles of a prospective payment system where some payments are lower than hospitals' costs while other payments are greater than a hospitals' costs. However, diagnostic radiopharmaceuticals with costs more than double the volume weighted average amount of the offset could present a hospital with a significant financial loss. This is why the OPPS has several payment provisions that rely on a multiplier of costs as a threshold for modifying payment.
Our proposed approach to multiply the average offset amount by two is consistent with the two-times rule the OPPS uses to determine Ambulatory Payment Classification (APC) levels, where a significant service that has a cost greater than two times the lowest cost significant service in an APC is generally moved to a higher level APC in the series. The two-times rule requires that the highest calculated cost of an individual procedure categorized to any given APC cannot exceed two times the calculated cost of the lowest cost procedure categorized to that same APC. We note that the two-times rule does not apply to diagnostic radiopharmaceuticals themselves, but only to the procedures in which they are used, which is why we are proposing a diagnostic radiopharmaceutical packaging threshold utilizing a similar two-times methodology.
As described in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), our proposed approach to multiply the average offset amount by two is also generally consistent with the OPPS outlier policy applicable to certain high-cost procedures, where costs greater than 1.75 times the APC payment trigger an additional outlier payment. The OPPS provides outlier payments to hospitals to help mitigate the financial risk associated with high-cost and complex procedures, where a very costly service could present a hospital with significant financial loss. Outlier payments are provided on a service basis when the cost of a service exceeds the APC payment amount multiplier threshold (1.75) as well as the APC payment amount plus a fixed-dollar amount threshold. The proposed diagnostic radiopharmaceutical packaging threshold would serve a similar purpose as the outlier policy, in that it would provide payments to hospitals to help mitigate the financial risk associated with high-cost diagnostic radiopharmaceuticals, where a very costly diagnostic radiopharmaceutical could present a hospital with significant financial loss.
We proposed a multiplier of two and explained that we believe two is the most appropriate number for the multiplier for the volume weighted average amount of the offset, but we sought comment on the alternatives of using 1.75 times the volume weighted average amount of the offset as the threshold amount for triggering separate payment, or another appropriate multiplier amount. We stated that, for example, an interested party could present data that a financial disincentive to use diagnostic radiopharmaceuticals exists when costs are 1.75 times, or three times or five times, the volume weighted average offset amount. Since the hospital outpatient outlier payment policy is a longstanding policy familiar to most hospitals, we sought comment on utilizing elements of that policy for purposes of our proposed diagnostic radiopharmaceutical packaging policy in order to help hospitals mitigate the financial risk that may be associated with furnishing high-cost and complex diagnostic radiopharmaceuticals. As previously mentioned, we sought comment on the use of 1.75 times as the multiplier threshold rather than 2. Although the outlier policy uses both a 1.75 multiplier threshold and a fixed-dollar threshold, we sought comment regarding the use of 1.75 as the multiplier to set a fixed dollar threshold for the volume weighted average amount of the offset as the goals of the ( printed page 93952) outlier policy and the proposed diagnostic radiopharmaceutical policy are similar.
We also solicited comment on the alternative of using the standard drug packaging threshold, which was proposed to be $140 for CY 2025 and is being finalized as $140 for CY 2025 in section V.B of this final rule with comment period, as the threshold for separate payment for diagnostic radiopharmaceuticals. We believe that diagnostic radiopharmaceuticals are functioning as supplies to the nuclear medicine procedure in which they are used. Because diagnostic radiopharmaceuticals function as supplies in the diagnostic procedures in which they are used, they are serving as an item that is integral, ancillary, supportive, dependent, or adjunctive to the primary diagnostic service. This is in contrast to therapeutic drugs, biologicals, and therapeutic radiopharmaceuticals that are typically packaged under the standard drug packaging threshold. These products could be the only therapeutic modality provided to a patient during an encounter and may not serve as an item that is integral, ancillary, supportive, dependent, or adjunctive to the primary service. Due to this clinical difference, we did not believe that using the standard drug packaging threshold was appropriate for diagnostic radiopharmaceuticals, and therefore we proposed a threshold specific to diagnostic radiopharmaceuticals. As stated in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we were interested to hear from commenters whether they agreed or disagreed with this assessment.
Comment: A significant portion of commenters provided broad support for CMS's overall proposal regarding payment for diagnostic radiopharmaceuticals, stating that this policy will ensure access for Medicare beneficiaries, empower clinicians to make the best treatment decisions, and encourage innovation from industry. Commenters frequently stated that this policy will ensure access for underrepresented populations, which often have higher rates of disease but lower rates of detection.
Response: We thank commenters for their support.
Comment: Many commenters were supportive of CMS's proposal regarding separate payment for diagnostic radiopharmaceuticals, including the packaging threshold of $630. Commenters appreciated that CMS was targeting those products with costs that significantly exceeded the amount of the current packaged payment. These commenters supported the overall methodology and rationale for the calculation of the threshold, which was based on the approximate payment that would typically be attributable to diagnostic radiopharmaceuticals within each nuclear medicine APC by assessing the offsets associated with these APCs that were directly attributable to “policy packaged” drugs. Commenters generally agreed with using two times this amount to establish the threshold as they agreed it correlated with the statutory OPPS two times rule.
Response: We thank commenters for their support of our proposal to create a $630 threshold and the associated methodology used to calculate that threshold.
Comment: While many commenters were supportive of CMS's proposed $630 using the two times threshold multiplier, several said they also would not be opposed to a 1.75 multiplier, which would result in a $550 threshold. Other commenters advocated solely for a 1.75 multiplier citing consistency with the OPPS outlier policy, which they believed is a more appropriate comparator policy than the OPPS two times rule.
Response: The OPPS provides outlier payments to hospitals to help mitigate the financial risk associated with high-cost and complex procedures, where a very costly service could present a hospital with significant financial loss. Outlier payments are provided on a service basis when the cost of a service exceeds the APC payment amount multiplier threshold (1.75) as well as the APC payment amount plus a fixed-dollar amount threshold. An outlier payment is triggered when the cost of a service exceeds two different thresholds, a fixed dollar threshold and a multiplier threshold. A 1.75 multiplier is appropriate for the outlier payment policy as it is combined with a secondary fixed-dollar threshold, which is appropriate for the broader OPPS outlier policy which can apply to a large number of services with vastly different costs and payment amounts. For the OPPS outlier policy, two separate thresholds must be surpassed in order for separate payment to be made. We believe the lower 1.75 multiplier is appropriate for the outlier policy as there is also a secondary threshold that must be met to trigger outlier payments. The single threshold for separate payment for diagnostic radiopharmaceuticals would apply to a much narrower range of products; therefore, we do not believe the same two-threshold system used for OPPS outlier payments is practical. This makes a single threshold more appropriate, and because we are using a single threshold, we believe the higher two times multiplier is more appropriate, as there is not a secondary threshold that must also be surpassed. We believe our proposed approach to multiply the average offset amount by two to set the threshold is the most appropriate approach to determining this threshold, as this results in only those products whose costs significantly exceed, by two-times, the approximate amount of payment already attributed to the product in the nuclear medicine APC payment. This multiplier is consistent with the two-times rule the OPPS uses to determine Ambulatory Payment Classification (APC) levels, which also uses a single two times multiplier, rather than a two-threshold system like the OPPS outlier policy. The OPPS two times rule instructs that a significant service that has a cost greater than two times the lowest cost significant service in an APC is generally moved to a higher-level APC in the series. The two-times rule requires that the highest calculated cost of an individual procedure categorized to any given APC cannot exceed two times the calculated cost of the lowest cost procedure assigned to that same APC. A two times multiplier ensures that separate payment would apply only to diagnostic radiopharmaceuticals whose costs significantly exceed the approximate amount of payment already attributed to the product in the nuclear medicine APC payment.
Comment: A few commenters suggested other thresholds, such as the OPPS drug packaging threshold, in order to treat diagnostic radiopharmaceuticals in a similar manner as other drugs, biologicals, and therapeutic radiopharmaceuticals paid under the OPPS. Commenters also believed that using a lower threshold actually decreased the likelihood that a provider would be financially incentivized to use a product above the threshold, as more products would be paid separately using a lower threshold. They believed a lower threshold decreased the likelihood of a non-clinical financial incentive to use a higher cost radiopharmaceutical. Other commenters believed using a $500 threshold would be appropriate as it coincided with the figure used for separate payment for diagnostic radiopharmaceuticals in draft legislation, titled the Facilitating Innovative Nuclear Diagnostics (FIND) Act. Commenters also suggested using an alternative threshold multiplier, such as 1.51, 1.50, or 1.25. These commenters thought these lower thresholds would ( printed page 93953) allow for more products to be paid separately and argued a cost for a diagnostic radiopharmaceutical even 25 percent higher than the average diagnostic radiopharmaceutical cost could be considered significant and warrant separate payment. Two commenters suggested reassessing the threshold based on the changing costs of products over time or assessing which products are driving the APC payments and then setting the threshold higher than the cost of those high-volume products.
Response: We thank commenters for their comments; however, we believe the commenters' suggested thresholds are inconsistent with our OPPS two-times rule as described, which we believe is a longstanding and reasonable proxy for determining when costs exceed payment to a degree that warrants payment modification. The two-times rule utilizes a two times multiplier to determine APC levels, where a significant service that has a cost greater than two times the lowest cost significant service in an APC is generally moved to a higher-level APC in the series. The lower thresholds suggested by commenters would not follow this principle, and therefore, would not exclusively identify diagnostic radiopharmaceuticals whose costs significantly exceed the approximate amount of payment already attributed to the product in the nuclear medicine APC payment. We believe diagnostic radiopharmaceuticals are unique and therefore warrant their own specific threshold that is distinct from the broader drug packaging threshold used for therapeutic drugs, biologicals, and radiopharmaceuticals. This is because diagnostic radiopharmaceuticals are functioning as supplies to the nuclear medicine procedure in which they are used and are serving as an item that is integral, ancillary, supportive, dependent, or adjunctive to the primary diagnostic service. This is in contrast to therapeutic drugs, biologicals, and therapeutic radiopharmaceuticals that are typically packaged under the standard drug packaging threshold. These products could be the only therapeutic modality provided to a patient during an encounter and may not serve as an item that is integral, ancillary, supportive, dependent, or adjunctive to the primary service. Due to these differences, we believe a threshold specific to diagnostic radiopharmaceuticals is appropriate.
Comment: Some commenters advocated that CMS use a threshold of at least $630. Several commenters believed that focusing separate payment on those radiopharmaceuticals whose costs significantly exceed the payment already attributable to the product will help concentrate the effects of unbundling on only those products that are most likely to create access issues for beneficiaries while reducing the broader effects on the nuclear medicine APCs. One commenter stated that unpackaging radiopharmaceuticals with per day costs greater than $630 would decrease the nuclear medicine APC procedure payment rates, but that decrease would be exacerbated if CMS used a lower threshold.
Response: We thank commenters for their support. We agree a $630 threshold would appropriately identify only those diagnostic radiopharmaceuticals whose costs significantly exceed the approximate amount of payment already attributed to the product in the nuclear medicine APC payment.
Comment: Several commenters requested that CMS clarify that the proposal does not alter the qualifications for pass-through payment status or the pass-through payment methodology for those diagnostic radiopharmaceuticals that apply for pass-through status under the OPPS.
Response: That is correct. This policy is distinct from the policies described in section V.A of this final rule with comment period, which describe the qualifications and payment methodology for those products that qualify for OPPS pass-through status.
Comment: A few commenters recommended CMS monitor the effects of this policy if finalized. For example, one commenter supported the proposal, but suggested CMS carefully monitor the affected nuclear medicine APCs and address any larger than expected alterations to their reimbursement or other unintended consequences. An additional commenter encouraged CMS to continue to evaluate this methodology as new products enter the market. This commenter recommended CMS ensure no unintended consequences of this policy occur, such as manufacturers purposefully pricing their products just above the payment threshold.
Response: We thank the commenter for their support. We will monitor the effects of this policy and will consider proposing modifications in future rulemaking if appropriate.
Comment: One commenter sought clarification from CMS that, if finalized, its proposal to provide separate payment for diagnostic radiopharmaceutical products would be implemented in a budget neutral manner.
Response: Yes. This policy will be implemented in a budget neutral manner.
After consideration of the public comments we received, we are finalizing this aspect of our proposal without modification and will pay separately for any diagnostic radiopharmaceutical with a per day cost greater than $630 for CY 2025. Any diagnostic radiopharmaceutical with a per day cost at or below that threshold will continue to be policy packaged under the current policy at § 419.2(b)(15).
(3) Calculating the Per Day Cost of Diagnostic Radiopharmaceuticals
We proposed to calculate the per day costs for diagnostic radiopharmaceuticals using a methodology similar to the one we use for determining the per day costs of drugs and biologicals for comparison to the OPPS drug packaging threshold, proposed to be $140 for CY 2025.
We proposed to calculate the per day cost based on the methodology described in section V.B.1.b of the CY 2025 OPPS/ASC proposed rule, which relies on the methodology in the CY 2006 OPPS/ASC proposed rule (70 FR 42723 and 42724) and finalized in the CY 2006 OPPS final rule with comment period (70 FR 68636 through 68638). Though the clinical use of the drugs, biologicals, and therapeutic radiopharmaceuticals differs from diagnostic radiopharmaceuticals, we believe the method of determining how much of that item is used per day should be similar. Therefore, we proposed to use a similar methodology for determining the per day costs of diagnostic radiopharmaceuticals, as we do drugs, biologicals, and therapeutic radiopharmaceuticals. This methodology consists of nine steps:
Step 1. After application of the CCRs, we aggregated all line-items for a single date of service on a single claim for each product. This resulted in creation of a single line-item with the total number of units and the total cost of a diagnostic radiopharmaceutical given to a patient in a single day.
Step 2. We then created a separate record for each diagnostic radiopharmaceutical by date of service, regardless of the number of lines on which the diagnostic radiopharmaceutical was billed on each claim. For example, “diagnostic radiopharmaceutical X” is billed on a claim with two different dates of service, and for each date of service, the diagnostic radiopharmaceutical is billed on two line-items with a cost of $10 and 5 units for each line-item. In this case, ( printed page 93954) the computer program would create two records for this diagnostic radiopharmaceutical, and each record would have a total cost of $20 and 10 units of the product.
Step 3. We trimmed records with unit counts per day greater or less than 3 standard deviations from the geometric mean.
Step 4. For each remaining record for a diagnostic radiopharmaceutical, we calculated the cost per unit of the diagnostic radiopharmaceutical. If the HCPCS descriptor for “diagnostic radiopharmaceutical X” is “per 1 millicurie” and one record was created for a total of 10 millicurie (as indicated by the total number of units for the diagnostic radiopharmaceutical on the claim for each unique date of service), the computer program divided the total cost for the record by 10 to give a per unit cost. We then weighted this unit cost by the total number of units in the record. We did this by generating a number of line-items equivalent to the number of units in that particular claim. Thus, a claim with 100 units of “diagnostic radiopharmaceutical X” and a total cost of $200 would be given 100 line-items, each with a cost of $2, while a claim of 50 units with a cost of $50 would be given 50 line items, each with a cost of $1.
Step 5. We trimmed the unit records with cost per unit greater or less than 3 standard deviations from the geometric mean.
Step 6. We aggregated the remaining unit records to determine the mean cost per unit of the diagnostic radiopharmaceutical.
Step 7. Using only the records that remained after records with unit counts per day greater or less than 3 standard deviations from the geometric mean were trimmed (step 3), we determined the total number of units billed for each item and the total number of unique per-day records for each item. We divided the count of the total number of units by the total number of unique per day records for each item to calculate an average number of units per day.
Step 8. We used the payment rate (the mean unit cost (MUC) derived from the CY 2023 hospital claims data) for each diagnostic radiopharmaceutical and multiplied the payment rate by the average number of units per day for each diagnostic radiopharmaceutical to arrive at its per day cost.
Step 9. We packaged the items with per day costs less than or equal to $630 and designated items with per day costs greater than $630 as separately payable.
As just described, to determine the proposed CY 2025 packaging status for all nonpass-through diagnostic radiopharmaceuticals, we proposed to use the per day cost, calculated on a HCPCS code-specific basis, of each diagnostic radiopharmaceutical that had a HCPCS code in CY 2023 and was paid (via packaged or separate payment) under the OPPS. We used data from CY 2023 claims processed through December 31, 2023, for this calculation.
We proposed to continue to package payment for diagnostic radiopharmaceuticals with per day costs less than or equal to $630 under our existing packaging policy for diagnostic radiopharmaceuticals that function as surgical supplies under § 419.2(b)(15). Similar to our policy for the drug packaging threshold, we proposed to use updated claims data to make final determinations of the packaging status of HCPCS codes for diagnostic radiopharmaceuticals for each OPPS/ASC final rule with comment period. We proposed to make an annual packaging determination for each diagnostic radiopharmaceutical HCPCS code only when we develop the OPPS/ASC final rule with comment period for the update year. We proposed that only diagnostic radiopharmaceutical HCPCS codes that are identified as separately payable in this final rule with comment period would be subject to quarterly updates.
Consequently, the packaging status of some HCPCS codes for diagnostic radiopharmaceuticals in the OPPS/ASC proposed rule may differ from the same HCPCS codes' packaging status determined based on the data used for the final rule with comment period. Under these circumstances, we proposed to follow the established policies for the OPPS drug packaging threshold, which were initially adopted for the CY 2005 OPPS (69 FR 65780), to more equitably pay for those diagnostic radiopharmaceuticals whose costs fluctuate relative to the proposed CY 2025 OPPS diagnostic radiopharmaceutical packaging threshold in a way that affects the product's payment status (packaged or separately payable). Our policy for the OPPS drug packaging threshold has not changed for many years and is the same as described in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70434). We believe these same policies should apply to diagnostic radiopharmaceuticals in order to ensure payment consistency for those diagnostic radiopharmaceuticals whose costs fluctuate relative to the proposed CY 2025 OPPS diagnostic radiopharmaceutical packaging threshold. For CY 2025, similar to our historical practice for the drug packaging threshold, we proposed to apply the following policies to those HCPCS codes for diagnostic radiopharmaceuticals whose relationship to the diagnostic radiopharmaceutical packaging threshold changes based on the final updated data: HCPCS codes for diagnostic radiopharmaceuticals that are proposed for separate payment in CY 2025, and that then have per day costs equal to or less than the CY 2025 final rule diagnostic radiopharmaceutical packaging threshold, based on the updated hospital claims data used for the CY 2025 final rule, would remain packaged in CY 2025. HCPCS codes for diagnostic radiopharmaceuticals for which we proposed packaged payment in CY 2025 but that then have per-day costs greater than the CY 2025 final rule drug packaging threshold, based on updated hospital claims data used for the CY 2025 final rule, would receive separate payment in CY 2025.
Comment: Most commenters were supportive of the methodology used to calculate the per day costs, and many commenters were able to analyze the cost data published with the proposed rule and calculate the same list of products with per day costs exceeding $630.
Response: We thank commenters for their support of our proposal, and for their agreement with our calculations of the per day costs.
Comment: A few commenters recommended CMS consider alternative methods of calculating per day costs and make refinements in the future as we gain more experience with this policy.
Response: We thank commenters for their input. Our methods for calculating per day cost are consistent with our longstanding policies for calculating per day costs for purposes of the OPPS drug packaging threshold and we believe they should be finalized for CY 2025 and subsequent years. We could address this methodology in future rulemaking if the need arises; however, this methodology has worked effectively for calculating per day costs of drugs, biologicals and radiopharmaceuticals for purposes of the OPPS drug packaging threshold, and we believe this well-known process will work equally well for diagnostic radiopharmaceuticals to create a trimmed data set from which from per day costs can be calculated.
Comment: Many commenters recommended that CMS not apply the proposed packaging rules in 2025 and pay separately for any product that was proposed for separate payment or that exceeds the cost threshold in the final rule data. Commenters stated that CMS ( printed page 93955) should only consider applying its proposed methodology after the separate payment threshold has been in effect for a reasonable period of time. Specifically, commenters recommended that HCPCS codes for diagnostic radiopharmaceuticals that are proposed for separate payment in CY 2025, and that then have per day costs equal to or less than the threshold for the final rule, be separately paid. Similarly, commenters believed HCPCS codes for diagnostic radiopharmaceuticals for which CMS proposed packaged payment in CY 2025 but that then have per-day costs greater than the CY 2025 final rule drug packaging threshold, based on updated hospital claims data used for the CY 2025 final rule, receive separate payment in CY 2025.
Commenters stated that the claims data and the cost report data are updated between the proposed and final rule. Specifically, commenters noted that the determination of whether a diagnostic radiopharmaceutical will qualify for separate payment uses both units and mean cost calculated from Medicare claims data to determine the per day cost. As the mean cost estimate relies on cost-to-charge ratios, in the commenter's view, the diagnostic radiopharmaceutical packaging determination is susceptible to fluctuations in the mean cost estimate and those fluctuations are not necessarily driven by variation in the actual price of the product. The commenter stated that this is in contrast to ASP based payment rates, as ASP is an external estimate of product cost.
Response: We thank commenters for their comments and understand their concerns about potential cost data fluctuations changing packaging determinations between the proposed and final rule. However, based on updated data for the final rule, the diagnostic radiopharmaceuticals that have per day costs that exceed $630 are the same 26 diagnostic radiopharmaceuticals that exceeded the $630 threshold using proposed rule data, meaning, for purposes of determining whether a product's per day cost exceeded $630, the updated data for the final rule did not change the list of qualifying products.
After consideration of the public comments we received, we are finalizing our proposal regarding the calculation of per day costs of diagnostic radiopharmaceuticals without modification. Specifically, we are finalizing a policy to calculate the per day costs for diagnostic radiopharmaceuticals for purposes of determining whether a diagnostic radiopharmaceutical qualifies for separate payment using a methodology similar to the one we use for determining the per day costs of drugs and biologicals for comparison to the OPPS drug packaging threshold. We calculated the per day cost based on the methodology described in section V.B.1.b of this final rule with comment period, which relies on the methodology in the CY 2006 OPPS/ASC proposed rule (70 FR 42723 and 42724) and finalized in the CY 2006 OPPS final rule with comment period (70 FR 68636 through 68638).
(4) Updating the Diagnostic Radiopharmaceutical Packaging Threshold in CY 2026
Starting in CY 2026 and subsequent years, we proposed to update the proposed threshold amount of $630 by the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IHS Global, Inc (IGI). IGI is a nationally recognized economic and financial forecasting firm with which CMS contracts to forecast the various price indexes including the Producer Price Index (PPI) Pharmaceuticals for Human Use (Prescription). This is the same as the update factor used for the OPPS drug packaging threshold, where we originally used the four-quarter moving average PPI levels for Pharmaceutical Preparations (Prescription) to trend the $50 threshold forward from the third quarter of CY 2005 (when the Pub. L. 108-173 mandated threshold became effective) to the third quarter of the applicable calendar year. We believe it is appropriate to use the same PPI for Pharmaceuticals for Human Use (Prescription) for the diagnostic radiopharmaceutical packaging threshold, as diagnostic radiopharmaceuticals are also prescription pharmaceuticals for human use. We proposed that starting for CY 2026, we would use the most recently available four quarter moving average PPI levels to trend the final CY 2025 threshold forward from the third quarter of CY 2024 to the third quarter of CY 2025 and round the resulting dollar amount to the nearest $5 increment. We explained that the proposal to update the diagnostic radiopharmaceutical packaging threshold would maintain consistency with our longstanding methodology for updating the OPPS drug packaging threshold, which is discussed in more detail in section V.B.1.a. of this final rule with comment period and also in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68085 and 68086).
Comment: The majority of commenters supported our proposal to update the final threshold by the PPI for Pharmaceuticals for Human Use (Prescription) in CY 2026 and future years. They stated this was appropriate to reflect the increasing cost of diagnostic radiopharmaceuticals annually.
Response: We thank commenters for their support.
Comment: A small number of commenters were not in favor of our proposal to update the threshold by the PPI for Pharmaceuticals for Human Use (Prescription). One commenter believed CMS should take a broader approach and comprehensively review the appropriateness of the threshold amount yearly based on data for that year.
Response: We thank commenters for their insights; however, we continue to believe it is appropriate to subject the diagnostic radiopharmaceutical packaging threshold to the same update factor that is used for the OPPS drug packaging threshold as supported by the majority of commenters. Updating the finalized threshold by the PPI for Pharmaceuticals for Human Use (Prescription) is consistent with our longstanding policy to update the OPPS drug packaging threshold annually. This PPI update factor provides aggregate changes in the selling prices of pharmaceuticals, which makes it an appropriate factor with which to update the diagnostic radiopharmaceutical packaging threshold to ensure that as diagnostic radiopharmaceuticals' costs change over time, the threshold continues to identify products with costs that significantly exceed the otherwise applicable APC payment amounts as determined in this rule and that therefore should be eligible for separate payment.
After consideration of the public comments we received, we are finalizing our proposal to update the finalized packaging threshold without modification. Specifically, starting in CY 2026 and for subsequent years, we will update the threshold amount of $630 by the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IHS Global, Inc (IGI).
(5) Amount of Separate Payment for Diagnostic Radiopharmaceuticals Exceeding the Threshold
As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), once we determine that the per day cost of a nonpass-through diagnostic radiopharmaceutical exceeds the cost threshold of $630 per day for CY 2025, we then proposed to assign that radiopharmaceutical to an APC, ( printed page 93956) making it a specified covered outpatient drug (SCOD) per section 1833(t)(14)(B) of the Act. Accordingly, we proposed to pay for those nonpass-through, separately payable diagnostic radiopharmaceuticals based on our authority under section 1833(t)(14)(A)(iii)(II) of the Act. While under this authority we would ordinarily use the ASP methodology under section 1847A of the Act, we find that the ASP data we have is not usable for payment purposes. As previously mentioned, radiopharmaceuticals are not required to report ASP under section 1847A of the Act, and as such, there are very few manufacturers reporting ASP for their products currently. As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), of those few manufacturers reporting ASP, the ASP values that we do have generally do not align with the ASP we would expect based on the cost and Mean Unit Cost (MUC) data submitted to CMS by hospitals. For example, a frequently used diagnostic radiopharmaceutical had a reported ASP that is over 23,000 percent higher than the MUC derived from claims data. As manufacturers of diagnostic radiopharmaceuticals may be unaware of the correct reporting requirements, we explained that we believe it would be inappropriate to propose to pay for separately payable diagnostic radiopharmaceuticals based on their ASPs as currently reported, without giving manufacturers the opportunity to submit, certify, or restate the ASPs of their products. We believed MUC would be an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we stated that under our policy for therapeutic radiopharmaceuticals (74 FR 60520), there are several requirements for reporting ASP. For example, ASP data submitted would need to be provided for a patient-specific dose, or patient-ready form, of the radiopharmaceutical in order to properly calculate the ASP amount for a given HCPCS code if that HCPCS code dose descriptor was per study dose or similar. ASP data submitted should align with the code's dose descriptor and billing unit. We stated we would expect that the ASP data reported by a manufacturer would be representative of the item(s) sold by the manufacturer. We used the term “patient-ready” in that rule to ensure that ASP data submitted for OPPS payment purposes for separately payable radiopharmaceuticals reflect the costs of all the component materials of the finished radiopharmaceutical product. We expected that the ASP data would represent the sales price of all of the component materials of the finished radiopharmaceutical product sold by the manufacturer in terms that reflect the applicable HCPCS code descriptor such as “per study dose”, “per millicurie” and “up to XX millicuries.” For the few manufacturers that reported ASP data for their diagnostic radiopharmaceuticals, we believed it may be possible that they are not aware of the reporting requirements or are unaware of how to properly report ASP for their product, as CMS has not used ASP as the basis of payment for nonpass-through diagnostic radiopharmaceuticals before. Therefore, we explained that we believe a reasonable alternative for CY 2025 for separate payment of diagnostic radiopharmaceuticals that exceed the per day cost threshold is to use of their mean unit cost from claims data. This is consistent with our current practice for therapeutic radiopharmaceuticals when ASP data is not available. For diagnostic radiopharmaceuticals, we believe that ASP data is effectively not available for purposes of determining a payment amount and, therefore, payment based on MUC is a reasonable alternative.
We previously acknowledged (74 FR 35335), and continue to acknowledge, the complexities associated with reporting ASP for radiopharmaceuticals. We encourage manufacturers to submit ASP information for diagnostic radiopharmaceuticals, if possible. While we proposed to use MUC to pay for separately payable diagnostic radiopharmaceuticals in CY 2025, manufacturers can begin, or continue, to report ASP data for potential future use in paying for diagnostic radiopharmaceuticals. For CY 2025, ASP reporting is voluntary for diagnostic radiopharmaceuticals paid under the OPPS. We encouraged interested parties to submit comments regarding potential issues that may arise that prevent appropriate ASP reporting for diagnostic radiopharmaceuticals. If manufacturers choose to report ASP data, the data must meet reporting requirements to be used for payment under the OPPS.
As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), manufacturers that choose to report ASP data for their diagnostic radiopharmaceuticals would need to provide comprehensive data for CMS to calculate an ASP amount for a given HCPCS code. In instances where there is more than one manufacturer of a particular diagnostic radiopharmaceutical, we proposed that all manufacturers would need to submit ASP information for payment to be made based on ASP. This is because it would be inappropriate for Medicare payment for a HCPCS code to be based on the payment information submitted by one manufacturer, if that payment is used for a product made by different manufacturers. This is because the ASP information reported by one manufacturer might not reflect the ASP of the same product made by other manufacturers.
We note that ASP submissions for radiopharmaceutical payment under the OPPS would need to meet all of the existing regulatory and subregulatory requirements of the ASP reporting process under sections 1847A and 1927(b)(3) of the Act.
Specifically, we reiterated our ASP reporting requirements outlined in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60520) for products for which we are encouraging the reporting of ASP, but for which reporting is not statutorily required. The ASP data submitted would need to be provided for a patient-specific dose, or patient-ready form, of the diagnostic radiopharmaceutical in order to properly calculate the ASP amount that aligns with the dose descriptor for a given HCPCS code. When reporting an ASP for a separately payable radiopharmaceutical, we expect that the ASP data reported by a manufacturer would be representative of the item(s) sold by the manufacturer. In the CY 2010 OPPS/ASC final rule with comment period (74 FR 60520), we used the term “patient-ready” to ensure that ASP data submitted for OPPS payment purposes for separately payable radiopharmaceuticals reflect the costs of all the component materials of the finished radiopharmaceutical product. We expect that the ASP data would represent the sales price of all of the component materials of the finished radiopharmaceutical product sold by the manufacturer in terms that reflect the applicable HCPCS code descriptor, such as “per study dose” or “millicurie.” We defined a “patient-ready” dose for OPPS purposes as including all component materials of the radiopharmaceutical, at a minimum, and any other processing the manufacturer requires to produce the radiopharmaceutical that it sells that are reflected in the sales price, including radiolabeling, as long as any fees paid for such processing done on behalf of ( printed page 93957) the manufacturer meet the definition of “bona fide service fees” under § 414.802 (74 FR 60525).
As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we understand that manufacturers of separately payable radiopharmaceuticals produce radiopharmaceuticals that require a variety of processing steps in order to prepare the product for administration to a beneficiary. To be used for separate OPPS radiopharmaceutical payment, the ASP data reported by a manufacturer must represent sales of all of the component materials associated with the radiopharmaceutical. For our full policy on which factors to incorporate into ASP pricing, please see the CY 2010 OPPS/ASC final rule with comment period (74 FR 60520 through 60521).
In order to be used for payment under the OPPS, all radiopharmaceutical ASP submissions should meet the existing regulatory and subregulatory requirements of the ASP submission process under sections 1847A and 1927(b)(3) of the Act. In particular, we explained that we believe the “bona fide service fee” test in the ASP regulations is instructive here, and we refer readers to the CY 2010 OPPS/ASC final rule with comment period for our discussion on radiopharmaceutical ASP reporting (74 FR 60521).
To summarize our CY 2010 policy for ASP reporting on radiopharmaceuticals for OPPS payment purposes (74 FR 60521), a patient-specific dose or patient-ready form in the context of OPPS ASP submission for radiopharmaceutical payment means that the ASP reflecting manufacturer sales must represent sales of all of the component materials for the radiopharmaceutical, including a minimum of a cold kit and a radioisotope, and be reported in terms that reflect the applicable HCPCS code descriptor, such as “treatment dose” or “millicurie.” The ASP would not necessarily take into account the preparation of the final form of the radiopharmaceutical for patient administration, including radiolabeling, which may be conducted by the manufacturer, freestanding radiopharmacy, hospital pharmacy, or other entity. With respect to the latter, fees paid by the manufacturer for these services would be excluded from the ASP calculation (that is, would not be considered price concessions that reduce the ASP) only if they are “bona fide service fees” as defined in the regulations governing ASP. Thus, if the manufacturer pays a “bona fide service fee” for the services of the freestanding radiopharmacy, hospital pharmacy, or other entity, and reflects that fee in its price for the radiopharmaceutical, the amount of the “bona fide service fee” would be taken into account in the reported ASP data. However, manufacturers are not required to pay for the preparation of a radiopharmaceutical (including radiolabeling) in a freestanding radiopharmacy, hospital pharmacy, or other entity after sale of all of the component materials, and in that case, the cost of those services would not be reflected in the ASP data submitted to CMS. Manufacturers should submit ASP data for a separately payable radiopharmaceutical that incorporates prices for sales of all of the component materials by the manufacturer. We sought comment on these ASP reporting requirements outlined in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60521) and previous CMS guidance on the guidelines for the Submission of OPPS ASP Data for Nonpass-Through Separately Payable Therapeutic Radiopharmaceuticals and Radiopharmaceuticals with Pass-Through Status.[8] We continue to believe that these ASP reporting practices should be applicable to radiopharmaceuticals, including both therapeutic and diagnostic radiopharmaceuticals, but sought comment from interested parties in this space to ensure that these reporting guidelines are clear and reflective of clinical practice today.
We mentioned in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222) that we still see the potential value in the use of ASP data for payment purposes for diagnostic radiopharmaceuticals when reported correctly and by all manufacturers who manufacture a product that is described by a given HCPCS code. We stated that we believe that the use of ASP information for OPPS payment could provide an opportunity to improve payment accuracy for separately payable diagnostic radiopharmaceuticals by applying an established methodology that has already been successfully implemented under the OPPS for other separately payable drugs and biologicals, as well as therapeutic radiopharmaceuticals. Because the per day cost calculations determine whether a diagnostic radiopharmaceutical qualifies for separate payment, using the most accurate pricing information is paramount. The use of ASP information could provide an opportunity to further improve the accuracy of the per day cost calculations and the separate payment amounts for diagnostic radiopharmaceuticals. As previously mentioned, we do not believe that the limited amount of ASP information submitted currently is adequate for the purpose of determining separate payment for those few products that currently do report ASP, which is why we proposed to pay diagnostic radiopharmaceuticals with per day costs above the $630 threshold at each diagnostic radiopharmaceutical's mean unit cost. However, we were still interested in the potential to use ASP for the purpose of determining a diagnostic radiopharmaceutical's per day cost and payment amount in the future. Therefore, we wanted to engage with interested parties to learn about the unique aspects and challenges that may be associated with reporting ASP for diagnostic radiopharmaceuticals, and radiopharmaceuticals in general. We specifically sought comment as to whether interested parties believe CMS should require payment for diagnostic radiopharmaceuticals to be based on ASP in the future, such as in CY 2026 rulemaking, if interested parties are confident in their reporting ability.
We discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222) that we do believe that there could be situations in which it is appropriate to use ASP currently. For example, in section V.A.4 of the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we proposed to utilize ASP in payment for diagnostic radiopharmaceuticals on OPPS transitional pass-through status. In this situation, we believed the use of ASP is appropriate as the manufacturer of that diagnostic radiopharmaceutical is actively involved in the radiopharmaceutical's pass-through application, and CMS can ensure that pricing is reported appropriately for purposes of the drug pass-through cost significance tests and for purposes of payment if the pass-through status is approved. Typically, there is only one manufacturer for a diagnostic radiopharmaceutical applying for pass-through status, so CMS does not have to ensure all manufacturers are reporting ASP for that particular HCPCS code prior to establishing a separate payment amount based on ASP. Additionally, as discussed in section V.B.5 of the CY 2025 OPPS/ASC proposed rule (Proposed Payment for Nonpass-Through Drugs, Biologicals, and Radiopharmaceuticals with HCPCS Codes but Without OPPS Hospital ( printed page 93958) Claims Data), we proposed to base the initial payment for new diagnostic radiopharmaceuticals with HCPCS codes, but which do not have pass-through status and are without claims data, on ASP, and on WAC if ASP data for these diagnostic radiopharmaceuticals are not available. If the WAC also is unavailable, we proposed to make payment for new diagnostic radiopharmaceuticals at 95 percent of the products' most recent AWP. We believe the volume of products in this category will typically be very low; however, in these rare situations, we believe it would be appropriate to use ASP until a MUC is available. Similar to drugs applying for pass-through status, there is typically only one manufacturer for a diagnostic radiopharmaceutical that is new and described by a HCPCS code, but without claims data, so CMS does not have to ensure all manufacturers are reporting ASP for that particular HCPCS code prior to establishing a separate payment amount based on ASP. Additionally, although reporting of ASP is not a condition of CMS approving a HCPCS application, CMS has the opportunity to actively engage with the manufacturer, or sponsor of a HCPCS application, during the HCPCS application process. This allows for ongoing dialogue and education regarding the unique ASP reporting requirements that may be associated with a particular product, including how to ensure the reported ASP aligns with the dose descriptor for the newly assigned HCPCS code.
We sought comments on additional unique situations in which it still may be appropriate for CMS to use ASP information to assess per day costs and payment amounts for diagnostic radiopharmaceuticals for CY 2025. For example, we stated that one such unique situation could be continuing the use of ASP for a particular HCPCS code once its pass-through status has ended, if the HCPCS code was actively being paid based on ASP while on pass-through status. Under our proposal, payment for a diagnostic radiopharmaceutical would be based on MUC once its pass-through status ends. We sought comment on this potential unique situation, as well as others of which readers may be aware. We stated that we may finalize utilizing ASP in additional situations that commenters bring to our attention in the final rule as policies for CY 2025 depending on comments received.
As discussed, we proposed to base the payment rate for diagnostic radiopharmaceuticals on mean unit cost data derived from hospital claims. We did not propose to use ASP data for determining payment rates of nonpass-through diagnostic radiopharmaceuticals with claims data but are seeking comment on its use for determining the per day cost and setting the payment rate for diagnostic radiopharmaceuticals in the future. Additionally, we did not propose to use WAC or AWP as a basis for payment for diagnostic radiopharmaceuticals. Similar to our reasoning for payment of therapeutic radiopharmaceuticals in the CY 2012 OPPS/ASC final rule with comment period (77 FR 68390), we believe that paying for diagnostic radiopharmaceuticals using mean unit cost would appropriately pay for the average price of nonpass-through separately payable diagnostic radiopharmaceuticals for the applicable year. We believe MUC is an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS.
As we stated in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60523), we believe that WAC or AWP is not an appropriate proxy to provide OPPS payment for average therapeutic radiopharmaceutical acquisition cost and associated handling costs when manufacturers are not required to submit ASP data because payment based on WAC or AWP for separately payable drugs and biologicals is usually temporary for a calendar quarter until a manufacturer is able to submit the required ASP data in accordance with the quarterly ASP submission timeframes for reporting under section 1847A of the Act. However, WAC and AWP reported to compendia may not be reflective of a patient ready dose. We are additionally concerned about the use of WAC and AWP since ASP reporting for OPPS payment of separately payable diagnostic radiopharmaceuticals would not be required for CY 2025. The absence of appropriate ASP reporting could result in payment for a separately payable diagnostic radiopharmaceutical based on WAC or AWP indefinitely, a result which we believe would be inappropriate, as these pricing metrics do not capture all of the pricing discounts that may be reflected in the ASP. In the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222), we stated that given all of the concerns we had with other pricing methodologies for diagnostic radiopharmaceuticals, we proposed to rely on CY 2023 mean unit cost data derived from hospital claims data for payment rates for diagnostic radiopharmaceuticals for CY 2025.
Our proposed payment methodology for diagnostic radiopharmaceuticals that have costs above a $630 threshold would be similar, but not the same, as the methodology adopted for therapeutic radiopharmaceuticals as described in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60518). Although therapeutic radiopharmaceuticals are generally targeted at treating a certain disease or condition, and diagnostic radiopharmaceuticals are generally targeted at diagnosing a certain disease or condition, we believe they are clinically very similar products, manufactured in a similar manner, and should generally be paid using a similar payment methodology when paid separately. As such, we stated that we believe the same payment method as is used for therapeutic radiopharmaceuticals should apply to diagnostic radiopharmaceuticals above the cost threshold. However, as previously discussed, given our concerns with current ASP reporting patterns, we proposed to use MUC as the basis of payment for nonpass-through diagnostic radiopharmaceuticals for CY 2025. Therefore, we explained that we believe it is appropriate for the methodology to determine the proposed payment amounts to differ between diagnostic and therapeutic radiopharmaceuticals, at least for CY 2025. We stated that we will consider aligning the payment methodologies between therapeutic and diagnostic radiopharmaceuticals, either based on ASP or MUC, in future rulemaking.
We explained that we believe that mean unit cost data is an appropriate and adequate proxy for the average price for diagnostic radiopharmaceuticals and associated handling costs for these products. Mean unit cost data is reflective of the actual cost data that hospitals submit to CMS. The MUC payment methodology is consistent with our payment policy for therapeutic radiopharmaceuticals as stated in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60523) and is currently the basis of payment for many therapeutic radiopharmaceuticals when ASP is unavailable.
As previously discussed, we find that the ASP data we have is not usable for the purpose of paying for diagnostic radiopharmaceuticals and, therefore, we proposed to pay for qualifying nonpass-through diagnostic radiopharmaceuticals with claims data based on MUC. However, we also sought comment on how we could ( printed page 93959) potentially use our equitable adjustment authority at section 1833(t)(2)(E) of the Act to make an adjustment to the ASP data that has been reported in order to make it usable for the purpose of paying equitably for these products. For example, we sought comment as to whether CMS could use its equitable adjustment authority to adjust payment for diagnostic radiopharmaceuticals based on an adjusted ASP value when the ASP amounts reported to CMS deviate by a given threshold, such as two times the MUC calculated for the diagnostic radiopharmaceutical using claims data. Alternatively, the adjusted payment rate could be an average of the reported ASP and MUC, or other methodologies suggested by commenters. We broadly sought comment on this potential use of equitable adjustment authority to make the limited ASP data reported for diagnostic radiopharmaceuticals usable for purposes of setting payment rates for qualifying products.
We note, if readers do not believe it is appropriate for CMS to base the payment amount for diagnostic radiopharmaceuticals on MUC for CY 2025, we proposed in the alternative to maintain our current policy of unconditionally policy packaging all diagnostic radiopharmaceuticals regardless of their cost until an appropriate payment methodology can be established to determine a separate payment amount for diagnostic radiopharmaceuticals.
We stated that HCPCS codes that describe diagnostic radiopharmaceuticals with per day costs that meet or exceed the proposed diagnostic radiopharmaceutical packaging threshold would be assigned to a status indicator of “K”, indicating separate payment. An APC and a payment rate would be assigned as shown in Addendum B to the proposed rule. HCPCS codes that describe diagnostic radiopharmaceuticals with per day costs that are at or below the proposed diagnostic radiopharmaceutical packaging threshold would continue to be assigned to a status indicator of “N”, indicating packaged payment. We welcomed comment on these determinations. The proposed list of diagnostic radiopharmaceuticals that we calculated as having per day costs that exceed $630 and their proposed status indicators can be found in Table 8.
( printed page 93960)Proposed definitions of status indicators were included in Addendum D1 to the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222). Addenda to that proposed rule can be found on the CMS OPPS web page.
We proposed corresponding regulation text edits at § 419.2(b)(15) to only package diagnostic radiopharmaceuticals when their per day cost is at or below the per day diagnostic radiopharmaceutical packaging threshold for the applicable year. Specifically, we proposed to add the text “at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year” to qualify the packaging of diagnostic radiopharmaceuticals. We also proposed corresponding regulation text edits at § 419.41 (Calculation of national beneficiary copayment amounts and national Medicare program payment amounts) to codify our proposed payment policy for diagnostic radiopharmaceuticals and our existing policy for therapeutic radiopharmaceuticals.
Comment: Commenters were largely supportive of CMS's proposal to pay diagnostic radiopharmaceuticals based on their mean unit cost for CY 2025. Commenters applauded CMS for recognizing that the uniqueness of diagnostic radiopharmaceuticals could lead to some difficulty or uncertainty around reporting other pricing data, such as Average Sales Price (ASP), for these products. Additionally, commenters generally seemed to understand CMS's concerns with using ASP as a basis for payment broadly for CY 2025 given the limited information available.
Response: We thank commenters for their support of our proposal.
Comment: Although the majority of commenters were generally supportive of using mean unit cost as the basis for diagnostic radiopharmaceutical payment for CY 2025, many of these commenters saw the use of MUC as a transitional step, and recommended CMS consider Average Sales Price as the ( printed page 93961) methodology to determine the payment amount as soon as possible. Other commenters suggested specific unique situations in which they believed it would be appropriate for CMS to use ASP, such as when a product's OPPS pass-through status expires if the product was being paid based on ASP while on OPPS pass-through status. Similarly, some commenters expressed some drawbacks to using MUC as a payment amount rather than ASP, such as that mean unit cost depends on hospital cost to charge ratios and uses cost data from prior years, rather than from the current payment year. Similarly, a commenter communicated that the MUC methodology underestimates the actual costs to hospitals for higher priced technologies due to charge compression which, in their view, may result in an inaccurate payment rate for a product compared to the actual acquisition cost for a product by a given hospital.
Commenters stated that using ASP information was consistent with payment for drugs under the OPPS, and they also stated that ASP information was an established methodology that could provide an opportunity to improve payment accuracy.
Commenters believed there was value in using ASP as the basis of payment, and although they acknowledged CMS may not have access to ASP data for patient ready doses for all diagnostic radiopharmaceuticals, commenters recommended we transition to an ASP-based payment.
Some commenters stated their belief that separate payment will incentivize reporting of ASP data. CMS was urged to work with manufacturers to obtain ASP data and only consider basing payment on ASP when such data are determined to be reliable. Commenters also recommended that CMS provide at least one year notice of its intent to base payment on ASP to facilitate the most complete reporting and consistent payment methodologies across products.
Response: We thank commenters for their feedback and considerations on our proposal. Our concerns from the proposed rule remain for the final rule, in that there are very few manufacturers reporting ASP for their products currently, and of those few manufacturers reporting ASP, the ASP values that we do have generally do not align with the ASP we would expect based on the cost data submitted to CMS by hospitals. While a small number of manufacturers may be reporting ASP correctly, such as those recently on OPPS pass-through status, we believe a consistent payment methodology is most appropriate for nonpass-through diagnostic radiopharmaceuticals with per day costs above the threshold. We do agree that there could be some value in the use of ASP for determining a payment amount in the future; however, if we were to use the ASP methodology, we believe there must be more consistent, validated, and universal reporting in order for ASP to be a viable payment methodology. To maintain a consistent payment methodology for CY 2025, we believe it is appropriate to treat all nonpass-through diagnostic radiopharmaceuticals with claims data the same and pay using MUC without exception, such as for products with recent pass-through expiration.
Comment: Commenters expressed a broad interest in using ASP data and learning how to report that data appropriately to CMS, including regarding topics such as “bona fide service fees.” Commenters indicated a desire to learn more about how they could report ASP for their specific products. Many manufacturers expressed their intent to initiate discussions with CMS in order to potentially begin to report ASP data.
Response: We thank commenters for expressing their interest in use of ASP data and appropriate ASP reporting. As we have previously stated, and as commenters have noted, we recognize the complexities associated with reporting ASP for radiopharmaceuticals. We encourage ongoing engagement with CMS in order for manufacturers to provide information regarding their products' unique reporting complexities.
Comment: Some commenters recommended CMS continue to consider the appropriateness of other sources of cost data for future years, such as wholesale acquisition cost (WAC).
Response: We continue to believe that neither WAC nor AWP is an appropriate proxy to provide OPPS payment for average radiopharmaceutical acquisition costs and associated handling costs when manufacturers are not required to submit ASP data. This is because payment based on WAC or AWP for separately payable drugs and biologicals is usually temporary for a calendar quarter until a manufacturer is able to submit the required ASP data in accordance with the quarterly ASP submission timeframes for reporting under section 1847A of the Act. WAC and AWP reported to compendia may not be reflective of a patient ready dose. The absence of an ASP reporting requirement and inappropriate or no reporting of ASP could result in payment for a separately payable diagnostic radiopharmaceutical based on WAC or AWP indefinitely, a result which we believe would be inappropriate, as these pricing metrics do not capture all of the pricing discounts that may be reflected in the ASP.
Comment: Some commenters were concerned about potential fluctuations in MUC data and the effect on payment for separately payable diagnostic radiopharmaceuticals. Several commenters recommend CMS apply its low volume APC policy to diagnostic radiopharmaceutical APCs, stating that CMS's policy for setting payment rates for low volume APCs, defined as those with fewer than 100 claims, by using up to four years of claims data and basing the rate on the highest value among the arithmetic mean, geometric mean, or median improves payment rate stability for low volume APCs.
Response: We thank commenters for their recommendations. We did not propose to subject low volume diagnostic radiopharmaceutical APCs to the broader OPPS low volume policy, and we note that the low volume APC policy does not apply to APCs to which single drugs, biologicals, or radiopharmaceuticals are assigned, even if there is a low volume of claims for these items. We understand the commenter's concerns; however, we do not believe it would be appropriate to finalize this policy modification without further engagement from interested parties. While we agree this suggested policy modification could result in greater payment stability, we want to ensure that there are no unintended consequences of such a policy, such as an unintended decrease in the overall MUC payment rate as a result of using 4 years of cost data. We encourage additional engagement on this issue.
Comment: A commenter noted that we proposed that only diagnostic radiopharmaceutical HCPCS codes that are identified as separately payable in the final rule with a comment period would be subject to quarterly updates. The commenter was unsure why quarterly updates would be necessary for MUC-based payment, which they expect would be established annually as part of the rulemaking process and thus not be subject to quarterly updates.
Response: We thank the commenter for this comment and confirm that their understanding is correct. MUC will be calculated on an annual basis for the payment of nonpass-through diagnostic radiopharmaceuticals with per day costs exceeding $630. Because we are not adopting an ASP-based payment approach at this time, there is no need to update the payment amounts ( printed page 93962) quarterly as the MUC will be set in this final rule for the entire calendar year.
Comment: A few commenters believed that the product Magtrace, which may be described by HCPCS code A9697 (Injection, carboxydextran-coated superparamagnetic iron oxide, per study dose), was inappropriately assigned to a packaged payment status for the proposed rule and should be subject to this policy and designated for separate payment in the final rule. One commenter stated that the product is a magnetic tracer indicated to assist in localizing lymph nodes draining a tumor site, as part of a sentinel lymph node biopsy procedure in patients with breast cancer undergoing mastectomy or lumpectomy. The commenter stated that when HCPCS code A9697 was first incorporated in the October 2023 OPPS Update Transmittal, it was in a table titled “Table 15.—Newly Established HCPCS Codes for Drugs, Biologicals, and Radiopharmaceuticals as of October 1, 2023” and that Medicare Administrative Contractors (MACs) have indicated that HCPCS codes in the A9500-A9700 range, which the Magtrace code falls within, are for radiopharmaceuticals.
Response: We note that this product is not radioactive [9 10] and is regulated by the FDA as a medical device under the product name “Magtrace and Sentimag Magnetic Localization System” with a premarket approval.[11] Accordingly, we do not believe that this product qualifies as a diagnostic radiopharmaceutical, and do not believe that it should be paid separately under this policy. We believe this product functions as a supply when used in a diagnostic test or procedure and should continue to be packaged under 42 CFR 419.2(b)(15). Please see section V.A. of this final rule with comment period for the payment status of HCPCS code A9697 for CY 2025.
Comment: Broadly, commenters suggested that CMS subject additional classes of drugs, such as contrast agents to this policy. For example, a commenter discussed one such product, Cysview, which may be described by HCPCS code A9589 (Instillation, hexaminolevulinate hydrochloride, 100 mg), and believed CMS's reasoning in the proposed rule for unpackaging and paying separately for diagnostic radiopharmaceuticals applies equally to their product. The commenter believed lack of separate payment was a barrier to beneficiary access and recommended CMS pay for products like theirs when the products' costs exceeded a certain threshold.
Response: We thank the commenters for their feedback. We continue to believe that our packaging policies are a fundamental principle that distinguishes a prospective payment system from a fee schedule. In general, packaging the costs of supportive items and services into the payment for the primary procedure or service with which they are associated encourages hospital efficiencies and enables hospitals to manage their resources with maximum flexibility. We will take the information commenters provided into consideration as appropriate for possible future rulemaking.
After consideration of the public comments we received, we are finalizing our proposal without modification. In summary, for CY 2025 we are paying separately for any diagnostic radiopharmaceutical with a per day cost greater than $630 using the methodology described. Any diagnostic radiopharmaceutical with a per day cost at or below that threshold would continue to be policy packaged under our longstanding policy at § 419.2(b)(15). We are finalizing our proposal regarding updating the selected packaging threshold without modification. Specifically, starting in CY 2026 and for subsequent years, we will update the threshold amount of $630 by the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IHS Global, Inc (IGI). We are finalizing our proposal to pay for those nonpass-through, separately payable diagnostic radiopharmaceuticals based on our authority under section 1833(t)(14)(A)(iii)(II) of the Act, and as we find that the ASP data we have is not usable for the purpose of paying for diagnostic radiopharmaceuticals, we are paying for qualifying nonpass-through diagnostic radiopharmaceuticals with claims data based on mean unit cost data derived from hospital claims. We believe that paying for diagnostic radiopharmaceuticals using mean unit cost would appropriately pay for the average price of nonpass-through separately payable diagnostic radiopharmaceuticals for the applicable year. We believe MUC is an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS.
The finalized list of diagnostic radiopharmaceuticals that have calculated per day costs that exceed $630 and their status indicators can be found in Table 9.
( printed page 93963)Definitions of status indicators can be found in Addendum D1 to this final rule with comment period. Addenda to this final rule with comment period can be found on the CMS OPPS web page.
Additionally, we are finalizing with slight modification corresponding regulation text edits at § 419.2(b)(15) to only package diagnostic radiopharmaceuticals when their per day cost is at or below the per day diagnostic radiopharmaceutical packaging threshold for the applicable year. We are adding the language “with per-day costs at or” to the regulation text as a technical change to conform with our final policy as the prior text did not make it clear that the per day cost of a diagnostic radiopharmaceutical must be at or below the per-day diagnostic radiopharmaceutical packaging threshold in order for the product to be packaged. Overall, this regulation text is changed by adding the text “with per-day costs at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year” rather than only “below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year” to qualify the packaging of diagnostic radiopharmaceuticals. Aside from those changes described in V.B.5 of this CY 2025 OPPS/ASC final rule with comment period to conform with our payment policy for nonpass-through diagnostic radiopharmaceuticals in situations in which claims data are not available, we are finalizing without modification corresponding regulation text edits at § 419.41 (Calculation of national beneficiary copayment amounts and national Medicare program payment amounts) to codify our proposed payment policy for diagnostic radiopharmaceuticals and our existing policy for therapeutic radiopharmaceuticals. ( printed page 93964)
4. Implementation of Section 4135 of the Consolidated Appropriations Act (CAA)
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), was signed into law on December 29, 2022. Section 4135(a) and (b) of the CAA, 2023, titled Access to Non-Opioid Treatments for Pain Relief, amended sections 1833(t)(16) and 1833(i) of the Act, respectively, to provide for temporary additional payments for non-opioid treatments for pain relief (as that term is defined in section 1833(t)(16)(G)(i) of the Act). In particular, section 1833(t)(16)(G) of the Act provides that with respect to a non-opioid treatment for pain relief furnished on or after January 1, 2025, and before January 1, 2028, the Secretary shall not package payment for the non-opioid treatment for pain relief into payment for a covered OPD service (or group of services) and shall make an additional payment for the non-opioid treatment for pain relief as specified in clause (ii) of that section. Clauses (ii) and (iii) of section 1833(t)(16)(G) of the Act provide for the amount of additional payment and set a limitation on that amount, respectively.
As the additional payments are required to begin on January 1, 2025, we proposed and are finalizing a policy to implement the CAA, 2023 section 4135 amendments in this final rule with comment period. Our finalized policy to implement section 4135 of CAA, 2023 can be found in section XIII.E of this final rule with comment period.
5. Calculation of OPPS Scaled Payment Weights
We established a policy in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68283) of using geometric mean-based APC costs to calculate relative payment weights under the OPPS. In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81577 through 81578), we applied this policy and calculated the relative payment weights for each APC for CY 2024 that were shown in Addenda A and B of the CY 2024 OPPS/ASC final rule with comment period (which were made available via the internet on the CMS website) using the APC costs discussed in sections II.A.1 and II.A.2 of the CY 2024 OPPS/ASC final rule with comment period (88 FR 81549 through 81572). For CY 2025, as we did for CY 2024, we proposed to continue to apply the policy established in CY 2013 and calculate relative payment weights for each APC for CY 2025 using geometric mean-based APC costs.
For CY 2012 and CY 2013, outpatient clinic visits were assigned to one of five levels of clinic visit APCs, with APC 0606 representing a mid-level clinic visit. In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75036 through 75043), we finalized a policy that created alphanumeric HCPCS code G0463 (Hospital outpatient clinic visit for assessment and management of a patient), representing all clinic visits under the OPPS. HCPCS code G0463 was assigned to APC 0634 (Hospital Clinic Visits). We also finalized a policy to use CY 2012 claims data to develop the CY 2014 OPPS payment rates for HCPCS code G0463 based on the total geometric mean cost of the levels one through five CPT Evaluation or Assessment and Management (E/M) codes for clinic visits previously recognized under the OPPS (CPT codes 99201 through 99205 and 99211 through 99215). In addition, we finalized a policy to no longer recognize a distinction between new and established patient clinic visits.
For CY 2016, we deleted APC 0634 and reassigned the outpatient clinic visit HCPCS code G0463 to APC 5012 (Level 2 Examinations and Related Services) (80 FR 70372). For CY 2025, as we did for CY 2024, we proposed to continue to standardize all of the relative payment weights to APC 5012. We believe that standardizing relative payment weights to the geometric mean of the APC to which HCPCS code G0463 is assigned maintains consistency in calculating unscaled weights that represent the cost of some of the most frequently provided OPPS services. For CY 2025, as we did for CY 2024, we proposed to assign APC 5012 a relative payment weight of 1.00 and to divide the geometric mean cost of each APC by the geometric mean cost for APC 5012 to derive the unscaled relative payment weight for each APC. The choice of the APC on which to standardize the relative payment weights does not affect payments made under the OPPS because we scale the weights for budget neutrality.
Section 1833(t)(9)(B) of the Act requires that APC reclassification and recalibration changes, wage index changes, and other adjustments be made in a budget neutral manner. Budget neutrality ensures that the estimated aggregate weight under the OPPS for CY 2025 is neither greater than nor less than the estimated aggregate weight that would have been calculated without the changes. To comply with this requirement concerning the APC changes, we proposed to compare the estimated aggregate weight using the CY 2024 scaled relative payment weights to the estimated aggregate weight using the proposed CY 2025 unscaled relative payment weights.
For CY 2024, we multiplied the CY 2024 scaled APC relative payment weight applicable to a service paid under the OPPS by the volume of that service from CY 2023 claims to calculate the total relative payment weight for each service. We then added together the total relative payment weight for each of these services in order to calculate an estimated aggregate weight for the year. For CY 2025, we proposed to apply the same process using the estimated CY 2025 unscaled relative payment weights rather than scaled relative payment weights. We proposed to calculate the weight scalar by dividing the CY 2024 estimated aggregate weight by the unscaled CY 2025 estimated aggregate weight.
For a detailed discussion of the weight scalar calculation, we refer readers to the OPPS claims accounting document available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. Click on the link labeled “Hospital Outpatient Prospective Payment—Notice of Proposed Rulemaking with Comment Period (NPRM)” for 2025, which can be found under the heading “Hospital Outpatient Regulations and Notices” and open the claims accounting document link, which is labeled “2025 NPRM OPPS Claims Accounting (PDF).”
We proposed to compare the estimated unscaled relative payment weights in CY 2025 to the estimated total relative payment weights in CY 2024 using CY 2023 claims data, holding all other components of the payment system constant to isolate changes in total weight. Based on this comparison, we proposed to adjust the calculated CY 2025 unscaled relative payment weights for purposes of budget neutrality. We proposed to adjust the estimated CY 2025 unscaled relative payment weights by multiplying them by a proposed weight scalar of 1.4452 to ensure that the proposed CY 2025 relative payment weights are scaled to be budget neutral. The proposed CY 2025 relative payment weights listed in Addenda A and B to the proposed rule (which are available via the internet on the CMS website) are scaled and incorporate the recalibration adjustments discussed in sections II.A.1 and II.A.2 of this final rule with comment period.
Section 1833(t)(14) of the Act provides the payment rates for certain specified covered outpatient drugs (SCODs). Section 1833(t)(14)(H) of the ( printed page 93965) Act provides that additional expenditures resulting from this paragraph shall not be taken into account in establishing the conversion factor, weighting, and other adjustment factors for 2004 and 2005 under paragraph (9) but shall be taken into account for subsequent years. Therefore, the cost of those SCODs (as discussed in section V.B.2 of this final rule with comment period) is included in the budget neutrality calculations for the CY 2025 OPPS.
We did not receive any public comments on the proposed weight scalar calculation, and we are finalizing our proposal to use the calculation process described in the CY 2025 OPPS/ASC proposed rule, without modification, for CY 2025. For CY 2025, as we did for CY 2024, we will continue to apply the policy established in CY 2013 and calculate relative payment weights for each APC for CY 2025 using geometric mean-based APC costs. For CY 2025, as we did for CY 2024, we will assign APC 5012 a relative payment weight of 1.00; and we will divide the geometric mean cost of each APC by the geometric mean cost for APC 5012 to derive the unscaled relative payment weight for each APC. To comply with this requirement concerning the APC changes, we will compare the estimated aggregate weight using the CY 2024 scaled relative payment weights to the estimated aggregate weight using the CY 2025 unscaled relative payment weights.
Using updated final rule claims data, we are updating the estimated CY 2025 unscaled relative payment weights by multiplying them by a weight scalar of 1.4452 to ensure that the final CY 2025 relative payment weights are scaled to be budget neutral. The final CY 2025 relative payments weights listed in Addenda A and B of this final rule with comment period (available via the internet on the CMS website) were scaled and incorporate the recalibration adjustments discussed in sections II.A.1 and II.A.2 of this final rule with comment period.
B. Conversion Factor Update
1. OPD Fee Schedule Increase Factor
Section 1833(t)(3)(C)(ii) of the Act requires the Secretary to update the conversion factor used to determine the payment rates under the OPPS on an annual basis by applying the OPD fee schedule increase factor. For purposes of section 1833(t)(3)(C)(iv) of the Act, subject to sections 1833(t)(17) and 1833(t)(3)(F) of the Act, the OPD fee schedule increase factor is equal to the hospital inpatient market basket percentage increase applicable to hospital discharges under section 1886(b)(3)(B)(iii) of the Act. In the FY 2025 IPPS/Long Term Care Hospital (LTCH) PPS proposed rule (89 FR 36204), consistent with current law, based on IHS Global, Inc.'s fourth quarter 2023 forecast, the proposed FY 2025 IPPS market basket percentage increase was 3.0 percent. In the CY 2025 OPPS/ASC proposed rule, we noted that under our regular process for the CY 2025 OPPS/ASC final rule with comment period, we would use the market basket update for the FY 2025 IPPS/LTCH PPS final rule (89 FR 68986). We explained that if that forecast is different than the IPPS market basket percentage increase used for the proposed rule, the CY 2025 OPPS/ASC final rule with comment period OPD fee schedule increase factor would reflect that updated forecast of the market basket percentage increase.
For CY 2025, we proposed to use the estimate of the hospital inpatient market basket percentage increase of 3.0 percent as one component to calculate the OPD fee schedule increase factor.
Comment: Two commenters expressed their support for the proposed hospital inpatient market basket increase of 3.0 percent.
Response: We appreciate the support of the commenters.
Comment: Several commenters expressed concerns that the proposed CY 2025 market basket update does not adequately reflect the rising inflation and costs that hospitals have faced over the last few years. Commenters stated that economy-wide inflation grew by 12.4 percent from 2021 through 2023 (as measured by the Consumer Price Index (CPI)), more than two times faster than Medicare reimbursement for hospital outpatient services, which increased by 5.9 percent during the same time. A commenter noted that the US Health Care Inflation Rate (USHCIR) for June 2024 was 3.3 percent and long-term USHCIR was 5.1 percent. Another commenter noted that inflation for hospital services in July 2024 was 6.1 percent. The commenters observed these rates are substantially higher that the proposed market basket increase.
Many commenters stated that rapid and sustained growth in labor costs have put persistent cost pressure on hospitals. They also noted increases in drug prices, citing a recent study and a report by the Health and Human Services (HHS) Assistant Secretary for Planning and Evaluation which found that in 2022 and 2023, prices for nearly 2,000 drugs increased faster than the rate of general inflation, with an average price increase of 15.2 percent. Several commenters also stated that hospitals have seen significant growth in administrative costs due to what they described as inappropriate practices by large commercial health insurers, including Medicare Advantage and Medicaid managed care plans, such as automatic claim denials and onerous prior authorization requirements. Commenters also discussed the continued costs of addressing past and preventing future cyberattacks and a commenter stated they have seen significant increases in capital costs, particularly since the pandemic. Some commenters stated that hospitals will continue to face increased costs due to the Change Healthcare cyberattack, such as interest costs on loan payments for loans acquired during the cyberattack, expected denials that will require additional administrative costs, and manual processing of claims. Commenters also urged CMS to consider the changing health care environment, which they state is putting enormous financial strain on hospitals and health systems and is expected to continue through 2025.
Several commenters proposed CMS apply a payment increase of at least 4.1 percent which is aligned with MedPAC's March 2024 Report to Congress, which recommended a 1.5 percentage points increase over the FY 2025 payment update. These commenters noted that this was the second year that MedPAC made a recommendation of increasing the market basket update. Several commenters suggested various higher market basket increases, which they believe better reflects hospitals' input prices and the contract labor staffing challenge. A commenter encouraged CMS to consider, at a minimum, matching the 3.7 percent increase that the commenter stated Medicare Advantage will receive.
Several commenters recommended CMS look to alternative data sources that they asserted better reflect true labor and input cost increases in a timely manner. The commenters stated that the proposed payment update does not recognize these challenges, nor does it factor in the realities of inflation impacting operating costs. Commenters also stated CMS must use data that better reflects the input price inflation that hospitals have experienced and are projected to experience in CY 2025. Multiple commenters recommended CMS use more recent data to update adjustments to 2025 OPPS rates.
Many commenters noted their financial pressures due to the PHE, aging, more complex patients, negative Medicare margins of −12.7 percent as ( printed page 93966) estimated by MedPAC, and reliance on public payers. Several commenters urged CMS to consider and assess the financial position of hospitals, particularly those with low margins. Multiple commenters asked for CMS to increase rates to cover the cost of care for Medicaid patients. Many commenters noted financial hardships, particularly in 2022, with high inflation and workforce shortages. They noted that MedPAC found that all-payment operating and overall Medicare margins both fell to record lows, estimating Medicare hospital margins of negative 12.7 percent for FY 2022. MedPAC's FY 2024 recommendation was to increase the market basket update by one percentage point and, for FY 2025, that Congress increase the acute hospital market basket by 1.5 percentage points over current law.
Response: We acknowledge commenters' concerns, however, as we stated in the CY 2025 OPPS/ASC proposed rule, section 1833(t)(3)(C)(iv) of the Act requires the OPD fee schedule increase factor for a year to equal the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year. Accordingly, we are unable to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2025 IPPS/LTCH PPS final rule. We refer commenters to that final rule for responses regarding the issues commenters raised (89 FR 69340).
Comment: Many commenters expressed concerns with the Employment Cost Index (ECI) used to measure changes in labor compensation in the market basket, which they state may no longer accurately capture the changing composition and cost structure of the hospital labor market given the large increases in short-term contract labor use and its growing costs. The commenters stated labor costs have increased by more than 18 percent from CY 2020 to CY 2023. They attributed this increase to expensive contract labor costs (as a result of higher utilization rates and higher costs per hour) and faster growth in salaries for employed workers (reflecting sign-on and retention bonuses). They further stated that while salaries for contract nurses have decreased some from a peak in certain geographical areas, they still remained nearly 60 percent higher at the end of FY 2023 compared to the start of FY 2020. They further stated that CMS recognizes that the ECI does not capture shifts in composition of labor, and the commenters stated that by design, the ECI is not capturing the shifts that have occurred as hospitals have had to turn to contract labor to meet patient demand. Several commenters recommended that CMS use its exceptions and adjustments authority to adopt new or supplemental data sources, to ensure labor costs are adequately reflected in the payment update in the final rule. They further requested CMS utilize supplemental data sources to evaluate the accuracy of the ECI proxy and to modify methodologies, including adopting new or supplemental data, to calculate the payment update if its analysis determines that the ECI is not adequately capturing labor costs.
Response: We refer commenters to the FY 2025 IPPS/LTCH PPS final rule, where we responded to similar comments regarding the use of the ECI and related issues raised by commenters (89 FR 69340 through 69341). We further note that, as we stated in the CY 2025 OPPS/ASC proposed rule, section 1833(t)(3)(C)(iv) of the Act requires the OPD fee schedule increase factor for a year to equal the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year. Accordingly, we are unable to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2025 IPPS/LTCH PPS final rule, which would occur if we used a different methodology to measure changes in labor compensation in the OPPS market basket.
Comment: Several commenters recommended that CMS reevaluate the data sources it uses for rebasing its market basket and calculating the annual market basket update, including labor costs. They strongly encouraged CMS to adopt new or supplemental data sources in future rulemaking that more accurately reflect the costs to hospitals, such as through use of more real time data from the hospital community. They stated that they believe that the current market basket does not account for the higher costs of contract labor, which has become more common in hospitals in an era of clinical labor shortages. A commenter requested that CMS rebase the market baskets more frequently and at least every 3 years to ensure the market basket reflects the appropriate mix of services provided to Medicare beneficiaries.
Response: We refer commenters to the FY 2025 IPPS/LTCH PPS final rule, where we responded to similar comments (89 FR 69341). As we stated in the CY 2025 OPPS/ASC proposed rule, section 1833(t)(3)(C)(iv) of the Act requires the OPD fee schedule increase factor for a year to equal the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year. Accordingly, we are unable to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2025 IPPS/LTCH PPS final rule.
Comment: Commenters stated that since the COVID-19 PHE, IHS Global, Inc (IGI) has shown a consistent 3-year trend of under-forecasting the market basket growth and expressed concern this may indicate a more systematic issue with IGI's forecasting. They stated that these missed forecasts are permanently established in the standard payment rate for IPPS and will continue to compound, which they estimate to be $4 billion. Several commenters, including many associations, urged CMS to use its special exceptions and adjustments authority under section 1886(d)(5)(i) of the Act to implement a retrospective one-time adjustment for CY 2025 to account for the underestimation of the market basket updates over the last several years. Commenters recommended that CMS implement various one-time adjustments to account for underpayments in one or more years between CY 2021 and CY 2023 as well as for forecasted underpayments for CY 2024. The commenters stated the underestimation is, in large part, because the market basket is a time-lagged estimate that cannot fully account for unexpected changes that occur, such as historic inflation and increased labor and supply costs. They stated this is exactly what occurred at the end of the CY 2021 into CY 2022, which resulted in a large forecast error in the CY 2022 market basket update.
Several commenters noted that CMS currently implements a capital IPPS market basket forecast error adjustment as well as SNF PPS market basket forecast error adjustment policy, which resulted in FY 2024 and FY 2025 SNF forecast error adjustments of 3.6 percentage points and 1.7 percentage points, respectively. They stated while CMS has not developed an analogous policy for the OPPS operating update, they believe such a forecast error adjustment to the CY 2025 OPPS operating update could be adopted under CMS' existing authority. They noted the forecast errors for CY 2021 through CY 2023 for OPPS exceeded the 0.5 percentage point threshold that is used for the SNF forecast error adjustment policy. A commenter recommended CMS establish a forecast ( printed page 93967) error threshold of 1.5 percentage points, and retroactively adjust payments for that year.
Response: We refer commenters to the FY 2025 IPPS/LTCH PPS final rule, where we responded to similar comments (89 FR 69342). As we stated in the CY 2025 OPPS/ASC proposed rule, section 1833(t)(3)(C)(iv) of the Act requires the OPD fee schedule increase factor for a year to equal the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year. Accordingly, we are unable to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2025 IPPS/LTCH PPS final rule.
Comment: Multiple commenters supported the proposed 3.0 percent market basket increase believing the market basket increase would help rural hospitals. However, the commenters believe inflation for outpatient hospital services is substantially higher than the market basket increase, and they would prefer a higher market basket increase to help rural providers. The commenters stated that the operating margin for independent rural hospitals is -2.2 percent and that the operating margins for system-affiliated rural hospitals is 1.7 percent. Other commenters believe the low market basket increases lead to challenges for rural providers. Those commenters noted that the operating margins for rural hospitals are not sustainable, and they noted that eleven hospitals in rural and underserved areas have closed in the past two years even with the establishment of a new Medicare provider type that allows such hospitals to become rural emergency hospitals. The commenters believe a higher market basket increase would help rural hospitals stay open and provide access to quality care especially maternal care in the areas that they serve.
Response: We appreciate the support of the commenters of the proposed market basket increase, and we appreciate the commenters' raising their concerns regarding rural hospitals. As we stated in the CY 2025 OPPS/ASC proposed rule, section 1833(t)(3)(C)(iv) of the Act requires the OPD fee schedule increase factor for a year to equal the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year. Accordingly, we are unable to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2025 IPPS/LTCH PPS final rule.
Comment: Multiple commenters claim that section 1833(t)(3)(C)(iv) of the Act, which defines the outpatient hospital department market basket increase factor, has provisions that give CMS flexibility to use a different market basket increase factor for outpatient hospital services than the market basket increase factor used for inpatient hospital services. The commenters requested that CMS take advantage of section 1833(t)(3)(C)(iv) to establish a market basket increase for CY 2025 for the OPPS that would be larger than the market basket increase that was finalized for FY 2025 for the IPPS.
Response: Section 1833(t)(3)(C)(iv) of the Act permits the Secretary to substitute for the market basket percentage increase an annual percentage increase that is computed and applied with respect to covered OPD services furnished in a year in the same manner as the market basket percentage increase is determined and applied to inpatient hospital services for discharges occurring in a fiscal year. We would only anticipate utilizing a specific outpatient hospital market basket factor if the rate of change in the cost of covered OPD services was substantially different than the rate of change in the cost of inpatient hospital services for the previous fiscal year. We did not find evidence that the rate of change for covered OPD services was substantially different that the rate of changes for inpatient hospital services. Therefore, we are adopting an OPD fee schedule increase factor for CY 2025 equal to the IPPS market basket percentage increase factor applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year, as required by section 1833(t)(3)(C)(iv) of the Act. We decline to adopt a final OPD fee schedule increase factor different than the IPPS market basket percentage increase factor finalized in the FY 2025 IPPS/LTCH PPS final rule.
2. Productivity Adjustment
Section 1833(t)(3)(F)(i) of the Act requires that, for 2012 and subsequent years, the OPD fee schedule increase factor under subparagraph (C)(iv) be reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act. Section 1886(b)(3)(B)(xi)(II) of the Act defines the productivity adjustment as equal to the 10-year moving average of changes in annual economy-wide, private nonfarm business multifactor productivity (MFP) (as projected by the Secretary for the 10-year period ending with the applicable fiscal year, year, cost reporting period, or other annual period) (the “productivity adjustment”). In the FY 2012 IPPS/LTCH PPS final rule (76 FR 51689 through 51692), we finalized our methodology for calculating and applying the productivity adjustment. The U.S. Department of Labor's Bureau of Labor Statistics (BLS) publishes the official measures of private nonfarm business productivity for the U.S. economy. We note that previously the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) of the Act was published by BLS as private nonfarm business multifactor productivity. Beginning with the November 18, 2021, release of productivity data, BLS replaced the term multifactor productivity (MFP) with total factor productivity (TFP). BLS noted that this is a change in terminology only and will not affect the data or methodology. As a result of the BLS name change, the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) of the Act is now published by BLS as private nonfarm business total factor productivity. However, as mentioned, the data and methods are unchanged. Please see www.bls.gov for the BLS historical published TFP data. A complete description of IHS Global, Inc.'s (IGI) TFP projection methodology is available on the CMS website at https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information. In addition, we note that beginning with the FY 2022 IPPS/LTCH PPS final rule, we refer to this adjustment as the productivity adjustment rather than the MFP adjustment to more closely track the statutory language in section 1886(b)(3)(B)(xi)(II) of the Act. We note that the adjustment continues to rely on the same underlying data and methodology. In the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36204), the proposed productivity adjustment for FY 2025 was 0.4 percentage point.
Therefore, we proposed that the productivity adjustment for the CY 2025 OPPS/ASC would be 0.4 percentage point. We also proposed that if more recent data subsequently became available after the publication of the CY 2025 OPPS/ASC proposed rule (for example, a more recent estimate of the market basket percentage increase and/or the productivity adjustment), we would use such updated data, if appropriate, to determine the CY 2025 hospital inpatient market basket update and the productivity adjustment, which are components in calculating the OPD fee schedule increase factor under ( printed page 93968) sections 1833(t)(3)(C)(iv) and 1833(t)(3)(F) of the Act.
We note that section 1833(t)(3)(F) of the Act provides that application of this subparagraph may result in the OPD fee schedule increase factor under section 1833(t)(3)(C)(iv) of the Act being less than 0.0 percent for a year and may result in OPPS payment rates being less than rates for the preceding year. As described in further detail below, we proposed for CY 2025 an OPD fee schedule increase factor of 2.6 percent for the CY 2025 OPPS/ASC (which is the proposed estimate of the hospital inpatient market basket percentage increase of 3.0 percent, less the proposed 0.4 percentage point productivity adjustment).
Comment: Several commenters expressed concerns about the application of the productivity adjustment, particularly given the extreme pressures in which hospital and health systems operate. They stated the use of the private nonfarm business TFP is meant to capture gains from new technologies, economies of scale, business acumen, managerial skills and changes in productions. Thus, they stated this measure effectively assumes the hospital sector can mirror productivity gains from the private nonfarm business sector. They stated, however, in an economy marked by great uncertainty due to the PHE and labor and other productivity shocks, this assumption is significantly flawed. Other commenters requested that productivity adjustment be eliminated for CY 2025 without any additional reasons. A commenter also requested that CMS use its “special exceptions and adjustments” authority to reduce the productivity adjustment for CY 2025. Another commenter asked that the productivity adjustment not apply to small rural and sole community hospitals for CY 2025.
Response: Section 1833(t)(3)(F)(i) requires that after determining the OPD fee schedule increase factor under subparagraph (C)(iv), the Secretary shall reduce such increase factor by the productivity adjustment described in section 1886(b)(3)(B)(xi) of the Act. As required by statute, the FY 2025 productivity adjustment is derived based on the 10-year moving average growth in economy-wide productivity for the period ending FY 2025.
We thank the commenters for their comments. After consideration of the comments received and consistent with our proposal, we are finalizing an OPD fee schedule increase factor of 2.9 percent for CY 2025, which consists of the IPPS market basket increase factor of 3.4 percent less a 0.5 percentage point productivity adjustment.
3. Other Conversion Factor Adjustments
To set the OPPS conversion factor for 2025, we proposed to increase the CY 2024 conversion factor of $87.382 by 2.6 percent. In accordance with section 1833(t)(9)(B) of the Act, we proposed further to adjust the conversion factor for CY 2025 to ensure that any revisions made to the wage index and rural adjustment are made on a budget neutral basis. We proposed to calculate an overall budget neutrality factor of 1.0026 for wage index changes by comparing proposed total estimated payments from our simulation model using the proposed FY 2025 IPPS wage indexes to those payments using the FY 2024 IPPS wage indexes, as adopted on a calendar year basis for the OPPS. We further proposed to calculate an additional budget neutrality factor of 0.9982 to account for our proposed policy to cap wage index reductions for hospitals at 5 percent on an annual basis.
For CY 2025, we proposed to maintain the current rural adjustment policy, as discussed in section II.E of this final rule with comment period. Therefore, the proposed budget neutrality factor for the rural adjustment was 1.0000.
We proposed to calculate a CY 2025 budget neutrality adjustment factor for the cancer hospital payment adjustment by transitioning from the target PCR of 0.89 we finalized for CYs 2020 through 2023 (which included the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act) and incrementally reducing the target PCR by an additional 1.0 percentage point for each calendar year, beginning with CY 2024, until the target PCR equals the PCR of non-cancer hospitals calculated using the most recent data minus 1.0 percentage point as required by section 16002(b) of the 21st Century Cures Act. Therefore, we proposed to apply a budget neutrality adjustment factor of 1.0006 to the conversion factor for the cancer hospital payment adjustment. In accordance with section 1833(t)(18)(C) of the Act, as added by section 16002(b) of the 21st Century Cures Act (Pub. L. 114-255), we reduce the target PCR by 0.01, which brings the proposed target PCR to 0.87. This is 0.02 less than the target PCR of 0.89 from CY 2021 through CY 2023, which was held at the pre-PHE target.
For the CY 2025 OPPS/ASC proposed rule, we estimated that proposed pass-through spending for drugs, biologicals, and devices for CY 2025 would equal approximately $625 million, which represented 0.71 percent of total projected CY 2025 OPPS spending. Therefore, we stated that the proposed conversion factor would be adjusted by the difference between the 0.27 percent estimate of pass-through spending for CY 2024 and the 0.71 percent estimate of proposed pass-through spending for CY 2025, resulting in a proposed decrease to the conversion factor for CY 2025 of 0.44 percent.
We proposed that estimated payments for outliers would remain at 1.0 percent of total OPPS payments for CY 2025. We estimated for the proposed rule that outlier payments would be approximately 0.85 percent of total OPPS payments in CY 2024; the 1.00 percent for proposed outlier payments in CY 2025 would constitute a 0.15 percent increase in payment in CY 2025 relative to CY 2024.
For CY 2025, we proposed to use a conversion factor of $87.382 in the calculation of the national unadjusted payment rates for those items and services for which payment rates are calculated using geometric mean costs; that is, the proposed OPD fee schedule increase factor of 2.6 percent for CY 2025, the required proposed wage index budget neutrality adjustment of approximately 1.0026, the proposed 5 percent annual cap for individual hospital wage index reductions adjustment of approximately 0.9982, the proposed cancer hospital payment adjustment of 1.0006, and the proposed adjustment of a decrease of 0.44 percentage point of projected OPPS spending for the difference in pass-through spending, which resulted in a proposed conversion factor for CY 2025 of $89.379.
For CY 2025, we also proposed that hospitals that fail to meet the reporting requirements of the Hospital OQR Program would continue to be subject to a further reduction of 2.0 percentage points to the OPD fee schedule increase factor. For hospitals that fail to meet the requirements of the Hospital OQR Program, we proposed to make all other adjustments discussed above, but use a reduced OPD fee schedule update factor of 0.6 percent (that is, the proposed OPD fee schedule increase factor of 2.6 percent further reduced by 2.0 percentage points). This would result in a proposed reduced conversion factor for CY 2025 of $87.636 for hospitals that fail to meet the Hospital OQR Program requirements (a difference of −1.743 in the conversion factor relative to hospitals that met the requirements). For further discussion of the Hospital OQR Program, we refer readers to section XV of this final rule with comment period. For 2025, we proposed ( printed page 93969) to use a reduced conversion factor of $87.636 in the calculation of payments for hospitals that fail to meet the Hospital OQR Program requirements (a difference of −1.743 in the conversion factor relative to hospitals that met the requirements).
We received no comments on our proposed adjustments to the conversion factor for CY 2025. For this CY 2025 OPPS/ASC final rule with comment period, based on more recent data available, the OPD fee schedule increase factor for the CY 2025 OPPS is 2.9 percent (which reflects the 3.4 percent final estimate of the hospital inpatient market basket percentage increase reduced by a 0.5 percentage point productivity adjustment). For CY 2025, we are using a conversion factor of $89.169 in the calculation of the national unadjusted payment rates for those items and services for which payment rates are calculated using geometric mean costs; that is, the OPD fee schedule increase factor of 2.9 percent for CY 2025, the required wage index budget neutrality adjustment of 0.9927, the 5 percent annual cap for individual hospital wage index reductions of 0.9995, the cancer hospital payment adjustment of 1.0005, and the adjustment of 0.10 (or 0.37 less 0.27) percentage point of projected OPPS spending for the difference in pass-through spending that results in a conversion factor for CY 2024 of $89.169. We are also finalizing a reduced conversion factor of $87.439 in the calculation of payments for hospitals that fail to meet the Hospital OQR Program requirements (a difference of −1.730 in the conversion factor relative to hospitals that met the requirements).
The calculations we performed to determine the CY 2025 final conversion factor are shown in Table 10.
( printed page 93970) ( printed page 93971)C. Wage Index Changes
Section 1833(t)(2)(D) of the Act requires the Secretary to determine a wage adjustment factor to adjust the portion of payment and coinsurance attributable to labor-related costs for relative differences in labor and labor-related costs across geographic regions in a budget neutral manner (codified at 42 CFR 419.43(a)). This portion of the OPPS payment rate is called the OPPS labor-related share. Budget neutrality is discussed in section II.A.5 of this final rule with comment period.
The OPPS labor-related share is 60 percent of the national OPPS payment. This labor-related share is based on a regression analysis that determined that, for all hospitals, approximately 60 percent of the costs of services paid under the OPPS were attributable to wage costs. We confirmed that this labor-related share for outpatient services is appropriate during- our regression analysis for the payment adjustment for rural hospitals in the CY 2006 OPPS final rule with comment period (70 FR 68553). We proposed to continue this policy for the CY 2025 OPPS/ASC. We refer readers to section II.C of this final rule with comment period for a description and an example of how the wage index for a particular hospital is used to determine payment for the hospital.
We did not receive any public comments on our proposed labor-related share, and we are finalizing our proposal without modification.
As discussed in the claims accounting narrative included with the supporting documentation for this final rule with comment period (which is available via the internet on the CMS website ( https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices)), for estimating APC costs, we would standardize 60 percent of estimated claims costs for geographic area wage variation using the same FY 2025 pre-reclassified wage index that we use under the IPPS to standardize costs. This standardization process removes the effects of differences in area wage levels from the determination of a national unadjusted OPPS payment rate and copayment amount.
Under 42 CFR 419.41(c)(1) and 419.43(c) (published in the OPPS April 7, 2000, final rule with comment period (65 FR 18495 and 18545)), the OPPS adopted the final fiscal year IPPS post-reclassified wage index as the calendar year wage index for adjusting the OPPS standard payment amounts for labor market differences. Therefore, in general, the wage index that applies to a particular acute care, short-stay hospital under the IPPS has also applied to that hospital under the OPPS. As initially explained in the September 8, 1998, OPPS/ASC proposed rule (63 FR 47576), we believe that using the IPPS wage index as the source of an adjustment factor for the OPPS is reasonable and logical, given the inseparable, subordinate status of the HOPD within the hospital overall. In accordance with section 1886(d)(3)(E) of ( printed page 93972) the Act, the IPPS wage index is updated annually.
The Affordable Care Act contained several provisions affecting the wage index. These provisions were discussed in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74191). Section 10324 of the Affordable Care Act added section 1886(d)(3)(E)(iii)(II) to the Act, which defines a frontier State and amended section 1833(t) of the Act to add paragraph (19), which requires a frontier State wage index floor of 1.00 in certain cases, and states that the frontier State floor shall not be applied in a budget neutral manner. We codified these requirements at § 419.43(c)(2) and (3) of our regulations. For CY 2025 we proposed to implement this provision in the same manner as we have since CY 2011. Under this policy, the frontier State hospitals would receive a wage index of 1.00 if the otherwise applicable wage index (including reclassification, the rural floor, and rural floor budget neutrality) is less than 1.00. Because the HOPD receives a wage index based on the geographic location of the specific inpatient hospital with which it is associated, the frontier State wage index adjustment applicable for the inpatient hospital also would apply for any associated HOPD. We refer readers to the FY 2011 through FY 2024 IPPS/LTCH PPS final rules for discussions regarding this provision, including our methodology for identifying which areas meet the definition of “frontier States” as provided for in section 1886(d)(3)(E)(iii)(II) of the Act: for FY 2011, 75 FR 50160 through 50161; for FY 2012, 76 FR 51793, 51795, and 51825; for FY 2013, 77 FR 53369 and 53370; for FY 2014, 78 FR 50590 and 50591; for FY 2015, 79 FR 49971; for FY 2016, 80 FR 49498; for FY 2017, 81 FR 56922; for FY 2018, 82 FR 38142; for FY 2019, 83 FR 41380; for FY 2020, 84 FR 42312; for FY 2021, 85 FR 58765; for FY 2022, 86 FR 45178; FY 2023, 87 FR 49006; and for FY 2024, 88 FR 58977.
In addition to the changes required by the Affordable Care Act, we note that the proposed FY 2025 IPPS wage indexes continue to reflect a number of adjustments implemented in past years, including, but not limited to, reclassification of hospitals to different geographic areas, the rural floor provisions, the imputed floor wage index adjustment in all-urban states, an adjustment for occupational mix, an adjustment to the wage index based on commuting patterns of employees (the out-migration adjustment), and the permanent 5-percent cap on any decrease to a hospital's wage index from its wage index in a prior FY. Beginning with FY 2024, we include hospitals with § 412.103 reclassification along with geographically rural hospitals in all rural wage index calculations, and exclude “dual reclass” hospitals (hospitals with simultaneous § 412.103 and Medicare Geographic Classification Review Board (MGCRB) reclassifications) implicated by the hold harmless provision at section 1886(d)(8)(C)(ii) of the Act (88 FR 58971 through 58973). We also proposed to continue the low wage index hospital policy, under which we increase the wage index for hospitals with a wage index value below the 25th percentile wage index value for a fiscal year by half the difference between the otherwise applicable final wage index value for a year for that hospital and the 25th percentile wage index value for that year across all hospitals. We refer readers to the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36181 through 36186) for a detailed discussion of all proposed changes to the FY 2025 IPPS wage indexes.
We note that in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49018 through 49021), we finalized a permanent approach to smooth year-to-year decreases in hospitals' wage indexes. Specifically, for FY 2023 and subsequent years, we apply a 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline. That is, a hospital's wage index for FY 2025 would not be less than 95 percent of its final wage index for FY 2024. Except for newly opened hospitals, we apply the cap for a fiscal year using the final wage index applicable to the hospital on the last day of the prior fiscal year. A newly opened hospital would be paid the wage index for the area in which it is geographically located for its first full or partial fiscal year (subject to any reclassification), and it would not receive a cap for that first year, because it would not have been assigned a wage index in the prior year (in accordance with 42 CFR 419.41(c)(1) and 419.43(c), as noted previously).
We delineate hospital labor market areas based on Core-Based Statistical Areas (CBSAs) established by the Office of Management and Budget (OMB). As discussed in the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36139 through 36174), OMB issued revisions to the current labor market area delineations on July 21, 2023, that included a number of significant changes such as new CBSAs, urban counties that become rural, rural counties that become urban, and existing CBSAs that are split apart (OMB Bulletin 23-01). This bulletin can be found at: https://www.whitehouse.gov/wp-content/uploads/2023/07/OMB-Bulletin-23-01.pdf. The proposed changes to the IPPS wage index based on the newest CBSA delineations are available in the FY 2025 IPPS/LTCH PPS proposed rule. We proposed that corresponding changes would be adopted in the OPPS, which uses the IPPS wage index, based on the new OMB delineations in the CY 2025 OPPS/ASC proposed rule, consistent with any proposals in the FY 2025 IPPS/LTCH PPS proposed rule. We believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the OPPS wage index system by creating a more accurate representation of current geographic variations in wage levels. We refer readers to proposed changes based on the new OMB delineations in the FY 2025 IPPS/LTCH PPS proposed rule at 89 FR 36139 through 36174, which includes a discussion of the effects of implementation of the proposal to adopt the revised OMB labor market area delineations on reclassified hospitals.
CBSAs are made up of one or more constituent counties. Each CBSA and constituent county has its own unique identifying codes. The FY 2018 IPPS/LTCH PPS final rule (82 FR 38130) discussed the two different lists of codes to identify counties: Social Security Administration (SSA) codes and Federal Information Processing Standard (FIPS) codes. Historically, CMS listed and used SSA and FIPS county codes to identify and crosswalk counties to CBSA codes for purposes of the IPPS and OPPS wage indexes. However, the SSA county codes are no longer being maintained and updated, although the FIPS codes continue to be maintained by the U.S. Census Bureau. The Census Bureau's most current statistical area information is derived from ongoing census data from 2020. The Census Bureau maintains a complete list of changes to counties or county equivalent entities on the website at: https://www.census.gov/geo/reference/county-changes.html (which, as of May 6, 2019, migrated to: https://www.census.gov/programs-surveys/geography.html). In the FY 2018 IPPS/LTCH PPS final rule (82 FR 38130), for purposes of cross walking counties to CBSAs for the IPPS wage index, we finalized our proposal to discontinue the use of the SSA county codes and begin using only the FIPS county codes. Similarly, for the purposes of cross walking counties to CBSAs for the OPPS wage index, in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59260), we finalized our proposal to discontinue ( printed page 93973) the use of SSA county codes and begin using only the FIPS county codes. For CY 2025, under the OPPS, we are continuing to use only the FIPS county codes for purposes of cross walking counties to CBSAs.
We proposed to use the FY 2025 IPPS post-reclassified wage index for urban and rural areas as the wage index for the OPPS to determine the wage adjustments for both the OPPS payment rate and the copayment rate for CY 2025. We note that the proposed FY 2025 IPPS wage indexes reflect several proposed changes as a result of the revised OMB delineations, including proposed policies to accommodate changes in rural or urban status for existing counties, as well as addition or removal of certain individual CBSAs compared to the previous delineations. Therefore, policies and adjustments that were finalized for the FY 2025 IPPS post-reclassified wage index would be reflected in the final CY 2025 OPPS wage index beginning on January 1, 2025, if appropriate. We refer readers to the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36181 through 36186) and the proposed FY 2025 hospital wage index files posted on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2025-ipps-proposed-rule-home-page. Regarding budget neutrality for the CY 2025 OPPS wage index, we refer readers to section II.C of this final rule with comment period.
As a general matter, we continue to believe that using the IPPS post-reclassified wage index as the source of an adjustment factor for the OPPS is reasonable and logical, given the inseparable, subordinate status of the HOPD within the hospital overall. We acknowledge, however, that an interim final action with comment period (IFC) entitled Changes to the Fiscal Year 2025 Hospital Inpatient Prospective Payment System (IPPS) Rates Due to Court Decision appeared in the October 3, 2024 Federal Register (89 FR 80405), which makes certain changes to the FY 2025 IPPS post-reclassified wage index for some hospitals that were not and could not have been anticipated or accounted for in the OPPS proposed rule. For reasons discussed in detail later in this section, we believe it is more appropriate and consistent with CMS' authority to use the post-reclassified wage index established in the FY 2025 IPPS final rule (89 FR 68986) as corrected in the FY 2025 IPPS final rule correction (89 FR 80098) for purposes of the CY 2025 OPPS wage index than it would be to incorporate the exceptional changes implemented by the IFC.
Hospitals that are paid under the OPPS, but not under the IPPS, do not have an assigned hospital wage index under the IPPS. Therefore, for non-IPPS hospitals paid under the OPPS, it is our longstanding policy to assign the wage index that would be applicable if the hospital was paid under the IPPS, based on its geographic location and any applicable wage index policies and adjustments. We proposed to continue this policy for CY 2025. We refer readers to the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36181 through 36186) for a detailed discussion of the proposed changes to the FY 2025 IPPS wage indexes.
It has been our longstanding policy to allow non-IPPS hospitals paid under the OPPS to qualify for the out-migration adjustment if they are located in a section 505 out-migration county (section 505 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA)) (Pub. L. 108-173). Applying this adjustment is consistent with our general approach to adopt IPPS wage index policies for hospitals paid under the OPPS. We note that, because non-IPPS hospitals cannot reclassify, they are eligible for the out-migration wage index adjustment if they are located in a section 505 out-migration county. This is the same out-migration adjustment policy that would apply if the hospital were paid under the IPPS. For CY 2025, we proposed to continue our policy of allowing non-IPPS hospitals paid under the OPPS to qualify for the outmigration adjustment if they are located in a section 505 out-migration county (section 505 of the MMA) (88 FR 49585 and 49586). Furthermore, we proposed that the wage index that would apply for CY 2025 to non-IPPS hospitals paid under the OPPS would continue to include the rural floor adjustment and any policies and adjustments applied to the IPPS wage index to address wage index disparities. In addition, we proposed that the wage index that would apply to non-IPPS hospitals paid under the OPPS would include the 5-percent cap on wage index decreases.
Comment: One commenter supported CMS's decision to utilize the updated OMB delineations and agreed with the CBSA assignments made in Connecticut based on the regional planning areas.
Response: We appreciate the commenter's support.
Comment: A commenter did not agree with CMS's adoption of OMB's CBSA delineation revisions. The commenter stated that OMB cautions that CBSAs are not intended for any non-statistical uses and should only be used for other purposes with full consideration of the effects of using the delineations for such purposes. Further, the commenter stated that the Metropolitan Areas Protection and Standardization Act (MAPS Act) bars the automatic propagation of OMB revisions in CBSA delineations to geographic area determinations in nonstatistical Federal programs, and stated that any change to the standards of CBSA delineations shall propagate for any non-statistical use by any domestic assistance program only if the relevant agency determines that such a propagation supports the purposes of the program, is in the public interest, and adopts the change through notice-and-comment rulemaking. The commenter contends that if CMS chooses to adopt new OMB delineations, CMS must fully explain why reliance on the updated CBSAs as set forth by OMB is appropriate for purposes of the FY 2025 hospital wage index adjustments. The commenter stated that CMS has not provided an appropriate rationale for relying on the updated CBSAs and proposed to adopt the revised CBSAs by default. The commenter contends that CMS must make a fact-specific determination of those CBSAs' suitability for Medicare reimbursement purposes, including whether it would be appropriate to use additional data to modify OMB's delineations to ensure that such changes are appropriate for purposes of defining regional labor markets for hospital workers.
Response: CMS acknowledges that the CBSA definitions and delineations were not specifically created for the purpose of determining a hospital wage index. However, based on the reasons stated in prior rulemaking, we continue to believe that these definitions and delineations, which are regularly reviewed and updated by OMB, are the best proxy for CMS to use to adjust hospital payment rates based on geographic variations in labor costs in accordance with the statute. We note that the OPPS wage index has historically adopted the IPPS wage index on a calendar year basis, and thus the same structure for organizing geographic areas applied to the IPPS through CBSAs has applied in the OPPS.
Section 1886(d)(3)(E) of the Act requires that, as part of the methodology for determining prospective payments to hospitals through the IPPS, the Secretary must adjust the standardized amounts “for area differences in hospital wage levels by a factor (established by the Secretary) reflecting the relative hospital wage level in the ( printed page 93974) geographic area of the hospital compared to the national average hospital wage level.” Section 1833(t)(2)(D) of the Act provides that for the OPPS, “the Secretary shall determine a wage adjustment factor to adjust the portion of payment and coinsurance attributable to labor-related costs for relative differences in labor and labor-related costs across geographic regions in a budget neutral manner.” We refer readers to the FY 1985 IPPS final rule (50 FR 24375 through 24377) and the FY 1995 IPPS final rule (60 FR 29218 through 29220) for a history of the outreach, consultation, and discussion of the challenges faced in defining appropriate labor market areas for purposes of the wage index methodology. As with any classification system in which boundaries must be established, it is impossible to designate boundaries that will be completely satisfactory to all concerned. There was no consensus among the interested parties on a choice for new labor market areas, and CMS concluded the adoption and continuation of an MSA-based framework was the most prudent course of action. We also refer readers to the FY 2005 rule (69 FR 49027 through 49028) for further discussion regarding the process and outreach CMS undertook before initially adopting OMB CBSAs as the basis for the wage index methodology. We found that the CBSA framework offered a useful proxy for labor market area delineations and that none of the alternative labor market areas that were studied provided a distinct improvement over the use of MSAs.
As stated previously, CMS continued to evaluate other potential methods to calculate variations in geographic labor markets in a manner that maintains or improves consistency and equity in hospital payments in response to recommendations from MedPAC. However, as stated in the 2012 Report to Congress: Plan to Reform the Medicare Wage Index (on the web at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/Wage-IndexReform), CMS has concluded that implementing any of the recommended revisions to wage index methodologies would require Congressional action. The commenter did not suggest any alternative method for defining geographic labor market areas and, given our decades long history of using OMB CBSA (and the prior Primary MSA) definitions and delineations for wage index purposes, we continue to believe adopting OMB revisions in a timely manner is essential to the wage index by creating a more accurate representation of geographic variations in wage levels. CMS is aware of the MAPS Act requirements for the adoption of CBSA definitions for nonstatistical use and believes that we have provided an adequate rationale to support our proposed adoption through notice and comment rulemaking. As we stated in the proposed rule (89 FR 36140), we believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the wage index by creating a more accurate representation of current geographic variations in wage levels. While the adoption of the revised delineations will have both positive and negative impacts on specific hospitals and labor markets, we believe that periodically updating the labor market delineations using objective criteria and based on the most recently available commuting data will serve the public's interest in ensuring accurate Medicare payments to hospitals under the OPPS by more accurately reflecting current geographic variations in labor costs in the hospital payment methodology. While some CBSAs would be modified in significant ways, the criteria for MSA, Micropolitan Statistical Area, and Metropolitan Division definitions finalized by OMB are generally consistent with past updates, and we do not find that the adoption of these delineations will create extreme variations in payments to hospitals, especially when considering the impact of the policy to cap annual wage index reductions at 5 percent. On this basis, and for the reasons we stated in prior rulemaking, we have determined that their use supports the purpose of adjusting hospital payment rates based on geographic variations in labor costs in accordance with the statute. We have reviewed our findings and impacts relating to the new OMB delineations and find no compelling reason to delay implementation. Therefore, we are finalizing the proposed policies implementing the revised OMB delineations, including the policy for the treatment of Micropolitan Statistical areas, Metropolitan divisions, and the change to county-equivalent definitions for the State of Connecticut.
Comment: A commenter requested that in addition to the permanent cap policy, CMS implement a 3-year wage index transition period consistent with prior updates to the CBSA categorizations made due to OMB updates.
Response: We note that when we previously adopted revised OMB delineations, the majority of negatively impacted hospitals received a wage index adjustment for only one fiscal year via a 50/50 blend of wage index values using the then-current and newly adopted delineations (79 FR 66827). Hospitals that were reassigned from an urban to rural area as a result of our adoption of the revised OMB delineations received a 3-year transition period from their previous urban area wage index, as long as they did not obtain a new MGCRB reclassification during that time period. As discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49018 through 49021), the 5 percent cap on annual wage index reductions was intended to make unnecessary any future transitions in connection with wage index policy implementations, including the adoption of revised labor market area definitions. Based on our analysis of wage index differences between FY 2024 and FY 2025, we estimate that only 136 hospitals (less that 4 percent) will receive a wage index cap that did not receive the cap in FY 2024. This indicates any impact on overall wage index values that could be caused by the adoption of the revised delineations would be relatively small. Furthermore, given the iterative and interactive effects of different reclassification and wage index hold-harmless policies, it is difficult to isolate the effects on wage index values (both positive and negative) that are due solely to the adoption of the revised delineations. That is, hospitals may make different reclassification decisions based on the transition policy, rather than the actual impacts of the revised delineations. We believe that any attempt to tailor a transition policy specifically to the impacts of adopting revised labor market delineations is not likely to yield results that more accurately reflect current differences in area wages than the 5 percent cap policy. We believe the 5 percent cap policy ensures that hospitals will not experience large payment reductions as a result of annual changes in their wage index value, allows adequate time for hospitals to evaluate reclassification options, and provides consistency and predictability in wage index values. Largely due to the modification of the rural wage index calculation finalized in FY 2024 IPPS/LTCH PPS final rule (88 FR 58971 through 58977), a much larger number of urban and rural hospitals within the same state (nearly 60 percent) receive identical wage index values (prior to the application of other policies, such as the outmigration adjustment, 5 percent cap on annual wage index decreases, and low-wage index hospital policy). This ( printed page 93975) fact suggests that there is even less need for separate transition policies for urban and rural hospitals in response to changes in geographic delineations than there was previously. Furthermore, we did not receive a comment from any hospital (urban or rural) citing specific negative impacts due solely or primarily to the proposed adoption of the revised OMB delineations. For these reasons, we do not believe it is necessary to implement any additional or alternative transition policy to the 5 percent cap on wage index decrease policy.
Comment: A commenter supported our policy to treat urban hospitals reclassified as rural hospitals under § 412.103 as rural hospitals for purposes of the rural wage indexes and the rural floor.
Response: We appreciate the commenter's support of our policy.
Comment: Multiple commenters supported our proposed low-wage index policy, which, for hospitals with a wage index value below the 25th percentile, increases the hospital's wage index by half the difference between the otherwise applicable wage index value for that hospital and the 25th percentile wage index value for all hospitals. Some commenters stated that the policy helps to address unjustified disparities in Medicare reimbursement. One commenter supported the continuation of this policy and noted the court decision in Bridgeport Hospital v. Becerra, 108 F.4th 882 (D.C. Cir. 2024) that determined that CMS lacked the statutory authority to adopt this policy in the IPPS.
One commenter opposed the proposed low wage index hospital policy. They believe that it has the effect of harming hospitals in high wage areas while having unclear impacts on the hospitals in low wage areas that receive higher payments. The commenter further noted the recent court decision in Bridgeport Hospital v. Becerra. As a result, they urged CMS to discontinue the low wage index policy in the OPPS, but advocated that if CMS maintained the policy, it do so in a non-budget neutral manner.
Response: We appreciate these comments.
Since the establishment of the OPPS, we have adopted the IPPS wage index on a calendar year basis in the OPPS. As we have noted across the years and earlier in this section, using the IPPS wage index as the source of an adjustment factor for the OPPS is reasonable and logical, given the inseparable, subordinate status of the HOPD within the hospital overall. In cases where CMS has had statutory authority to implement certain policies under the IPPS in a non-budget neutral manner, to apply the same wage index values in the OPPS we have budget neutralized those adjustments in accordance with section 1833(t)(2)(D) of the Act, such as with the outmigration adjustment (69 FR 65842 through 65844).
On July 23, 2024, the Court of Appeals for the D.C. Circuit held that the Secretary lacked authority under section 1886(d)(3)(E) of the Act or under the “adjustments” language of section 1886(d)(5)(I)(i) of the Act to adopt the low wage index hospital policy for FY 2020 for the IPPS, and that the policy and related budget neutrality adjustment in the IPPS must be vacated. Bridgeport Hosp. v. Becerra, 108 F.4th 882, 887-91 & n.6 (D.C. Cir. 2024). In consideration of the court decision, CMS subsequently issued the interim final action with comment period (IFC) entitled Changes to the Fiscal Year 2025 Hospital Inpatient Prospective Payment System (IPPS) Rates Due to Court Decision (CMS-1808-IFC) to remove the low wage index hospital policy for FY 2025 IPPS purposes. At the same time, the IFC included a payment exception under section 1886(d)(5)(I)(i) of the Act that corresponded mathematically to a 5 percent cap on wage index reductions from FY 2024 for certain low wage index hospitals most significantly impacted by the removal of the low wage index policy.
We note that we proposed to include the low wage index hospital policy as part of the CY 2025 OPPS wage index and believe that the statutory authority provided under section 1833(t)(2)(D) of the Act allows us to finalize a similar policy for the CY 2025 OPPS, as we have done each year beginning in CY 2020. Specifically, section 1886(d)(3)(E) of the Act requires that CMS base the IPPS wage index on a comparison of “the relative hospital wage level in the geographic area of the hospital compared to the national average hospital wage level,” and on “a survey conducted by the Secretary (and updated as appropriate) of the wages and wage-related costs of subsection (d) hospitals in the United States,” whereas section 1833(t)(2)(D) contains no such prescriptions for the manner of determining the OPPS wage index. (We continue to effectuate relevant prescriptions, such as the requirement in sections 1833(t)(2)(D) and 1833(t)(9) of the Act that OPPS wage index policies are generally budget neutral.) In addition, CMS has authority to apply the low wage index hospital policy to the OPPS wage index under section 1833(t)(2)(E) of the Act, which allows the Secretary to “establish, in budget neutral manner, . . . other adjustments as determined to be necessary to ensure equitable payments, such as adjustments for certain classes of hospitals. We believe implementing the low wage index hospital policy is a valid exercise of the Secretary's authority to adopt “adjustments” to OPPS payments under the OPPS statute, where the provision regarding calculation of the OPPS wage adjustment factor does not contain the same language that the D.C. Circuit found to be prescriptive for the calculation of the IPPS wage index found at section 1886(d)(3)(E) of the Act. Additionally, section 1833(t)(2)(E) of the Act—unlike the adjustment authority at section 1886(d)(5)(I)—authorizes us to make adjustments to “ensure equitable payments.” In light of the unique circumstances presented by the timing of D.C. Circuit's decision, application of the low wage-index-hospital policy under the OPPS for CY 2025 avoids unexpected and arguably unfair payment consequences for hospitals that were not plaintiffs in the Bridgeport case and so falls within our equitable adjustment authority.
While this would create a divergence between the OPPS wage index values in CY 2025 and the ultimate, effective FY 2025 IPPS wage index values for some hospitals, we continue to believe that the concerns related to the wage index that led to the application of this policy to the OPPS wage index in previous years continue to apply, and that the OPPS authority for wage adjustment factors continues to give CMS the authority to implement a budget neutral low wage index hospital policy. Additionally, we believe that the same reasons underlying adoption of the IFC policies for the FY 2025 IPPS wage index weigh against incorporating those policies for purposes of the CY 2025 OPPS wage index. Specifically, we noted in the IFC that the intention of the policies implemented therein is to “promote certainty regarding. . .payments” and “provide for payment stability and promote predictability,” in light of the court's decision in the Bridgeport Hospital case (89 FR 80408). Given the differences noted in the previous paragraph between the IPPS and OPPS in CMS's statutory authorities related to the wage index, those interests are better served by finalizing the OPPS wage index methodology as proposed, including the low wage index hospital policy.
Finally, we acknowledge that in the IFC, we implemented under section 1886(d)(5)(I)(i) of the Act a transitional payment exception for hospitals that benefitted from the FY 2024 low wage ( printed page 93976) index hospital policy in the IPPS without budget neutralizing that exception. But that payment exception serves a different purpose from that of the low wage index hospital policy. And, in any event, unlike the statute authorizing the transitional payment exception, CMS's authority to implement a low wage index hospital policy in the OPPS requires it to budget neutralize that policy. That is, while section 1886(d)(5)(I)(i) contains no budget neutrality requirement, both section 1833(t)(2)(D) and (t)(2)(E) do. Thus, in the context of the OPPS, we will continue to budget neutralize the low wage index hospital policy. For these reasons, and those discussed above, we are finalizing in the CY 2025 OPPS the low wage index hospital policy as we had proposed in the CY 2025 OPPS/ASC proposed rule (89 FR 59227).
We recognize that creating different effective wage index values for a hospital under the OPPS than the wage index value for the hospital under the IPPS for CY 2025 differs in some respects from how we have implemented the OPPS wage index previously, where we have used the IPPS wage index value in effect throughout the FY as the OPPS wage index value for all hospitals. However, we believe this is the appropriate approach for the CY 2025 OPPS given the unusual circumstances wherein an appellate court ruled that CMS lacked authority under the IPPS statute for a policy under the FY 2020 IPPS wage index that the OPPS proposed rule had already proposed to include in the OPPS wage index. We further believe the approach in this final rule is consistent with § 419.43(c), which states: “CMS uses the hospital inpatient prospective payment system wage index established in accordance with Part 412 of this chapter to make the adjustment specified under paragraph (a) of this section.” The OPPS wage index values will match the IPPS wage index values in the FY 2025 IPPS final rule, as corrected in the FY 2025 IPPS final rule correction (89 FR 80098), which were “established in accordance with Part 412”; we are merely declining to incorporate certain modifications to those values made in the IFC, which reflect extraordinary steps taken due to the timing of the court's decision in Bridgeport Hospital.
CMS will explore options for realigning the IPPS and OPPS wage index values through future rulemaking.
Comment: Multiple commenters supported our policy to apply a 5 percent cap on any decrease to a hospital's wage index from its wage index in the prior FY. Commenters also requested that the proposed 5-percent cap policy be applied in a non-budget neutral manner, which the commenter stated would allow the cap to be applied while avoiding decreases to the wage index in areas with high wage indexes.
Response: We appreciate the commenters' support of our policy to apply a 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY. For the OPPS, section 1833(t)(2)(D) of the Act requires the Secretary to determine a wage adjustment factor to adjust the portion of payment and coinsurance attributable to labor-related costs for relative differences in labor and labor-related costs across geographic regions in a budget neutral manner, which is inconsistent with the commenters' request to exclude the wage index cap policy from budget neutrality.
After consideration of the public comments we received, we are finalizing our proposal without modification to use the FY 2025 IPPS post-reclassified wage index for urban and rural areas as finalized in the FY 2025 IPPS final rule and corrected in the FY 2025 IPPS final rule correction as the wage index for the OPPS, which includes the OPPS low wage index hospital policy, to determine the wage adjustments for both the OPPS payment rate and the copayment rate for CY 2025. In addition to the low wage index hospital policy, which will only apply in the OPPS in CY 2025, certain FY 2025 post-reclassified wage index policies will be reflected in the final CY 2025 OPPS wage index beginning on January 1, 2025, including, but not limited to, reclassification of hospitals to different geographic areas, the rural floor provisions, the imputed floor wage index adjustment in all-urban states, an adjustment for occupational mix, an adjustment to the wage index based on commuting patterns of employees (the out-migration adjustment), and a 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY. We refer readers to the FY 2025 IPPS/LTCH PPS final rule (89 FR 69252 through 69310) and the FY 2025 hospital wage index files posted on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2025-ipps-final-rule-home-page. Regarding budget neutrality for the CY 2025 OPPS wage index, we refer readers to section II.B. of this final rule with comment period.
For CMHCs, for CY 2025, we proposed to continue to calculate the wage index by using the post-reclassification IPPS wage index based on the CBSA where the CMHC is located. Furthermore, we proposed that the wage index that would apply to a CMHC for CY 2025 would continue to include the rural floor adjustment and any policies and adjustments applied to the IPPS wage index to address wage index disparities. In addition, the wage index that would apply to CMHCs would include the 5-percent cap on wage index decreases. Also, we proposed that the wage index that would apply to CMHCs would not include the outmigration adjustment because that adjustment only applies to hospitals.
We did not receive any public comments on our proposals, and we are finalizing our proposals regarding CMHC wage index calculations without modification.
Table 4A associated with the FY 2025 IPPS/LTCH PPS final rule (available via the internet on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2025-ipps-final-rule-home-page) identifies counties that would be eligible for the out-migration adjustment. Table 2 associated with the FY 2025 IPPS/LTCH PPS final rule as corrected by the FY 2025 IPPS final rule correction identifies IPPS hospitals that would receive the out-migration adjustment for FY 2025. We are including the outmigration adjustment information from Table 2 associated with the FY 2025 IPPS/LTCH PPS final rule as Addendum L to this final rule with comment period, with the addition of non-IPPS hospitals that would receive the section 505 outmigration adjustment under this final rule. Addendum L is available via the internet on the CMS website. We refer readers to the CMS website for the OPPS at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices. At this link, readers will find a link to the FY 2025 IPPS final rule wage index tables and the CY 2025 OPPS final rule Addendum L.
D. Statewide Average Default Cost-to-Charge Ratios (CCRs)
In addition to using CCRs to estimate costs from charges on claims for ratesetting, we use overall hospital-specific CCRs calculated from the hospital's most recent cost report (OMB control number: 0938-0050 for Form CMS-2552-10) to determine outlier payments, payments for pass-through devices, and monthly interim transitional corridor payments under the OPPS during the PPS year. For certain hospitals, under the regulations at 42 CFR 419.43(d)(5)(iii), we use the ( printed page 93977) statewide average default CCRs to determine the payments mentioned earlier if it is not possible to determine an accurate CCR for a hospital in certain circumstances. This includes hospitals that are new, hospitals that have not accepted assignment of an existing hospital's provider agreement, and hospitals that have not yet submitted a cost report. We also use the statewide average default CCRs to determine payments for hospitals whose CCR falls outside the predetermined ceiling threshold for a valid CCR or for hospitals in which the most recent cost report reflects an all-inclusive rate status (Medicare Claims Processing Manual (Pub. L. 100-04), Chapter 4, Section 10.11).
We discussed our policy for using default CCRs, including setting the ceiling threshold for a valid CCR, in the CY 2009 OPPS/ASC final rule with comment period (73 FR 68594 through 68599) in the context of our adoption of an outlier reconciliation policy for cost reports beginning on or after January 1, 2009. For details on our process for calculating the statewide average CCRs, we refer readers to the Claims Accounting Narrative for this final rule with comment period, which is posted on our website. We proposed to calculate the default ratios for CY 2025 using the most recent cost report data.
We did not receive any public comments on our proposal, and we are finalizing our proposal without modification to calculate the default ratios for CY 2025 using the most recent cost report data, which are from a June 2024 HCRIS cost report extract.
We no longer publish a table in the Federal Register containing the statewide average CCRs in the annual OPPS/ASC proposed rule and final rule with comment period. These CCRs and the upper limit CCR value at which we would apply statewide CCRs will be available for download with each OPPS/ASC CY proposed rule and final rule on the CMS website. We refer readers to our website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices; click on the link on the left of the page titled “Annual Policy Files” and then select the relevant year to download the statewide CCRs and upper limit in the “Downloads” section of the web page.
E. Adjustment for Rural Sole Community Hospitals (SCHs) and Essential Access Community Hospitals (EACHs) Under Section 1833(t)(13)(B) of the Act for CY 2025
In the CY 2006 OPPS final rule with comment period (70 FR 68556), we finalized a payment increase for rural sole community hospitals (SCHs) of 7.1 percent for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, items paid at charges reduced to costs, and devices paid under the pass-through payment policy, in accordance with section 1833(t)(13)(B) of the Act, as added by section 411 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) (Pub. L. 108-173). Section 1833(t)(13) of the Act provides the Secretary the authority to make an adjustment to OPPS payments for rural hospitals, effective January 1, 2006, if justified by a study of the difference in costs by APC between hospitals in rural areas and hospitals in urban areas. Our analysis showed a difference in costs for rural SCHs. Therefore, for the CY 2006 OPPS, we finalized a payment adjustment for rural SCHs of 7.1 percent for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, items paid at charges reduced to costs, and devices paid under the pass-through payment policy, in accordance with section 1833(t)(13)(B) of the Act.
In the CY 2007 OPPS/ASC final rule with comment period (71 FR 68010 and 68227), for purposes of receiving this rural adjustment, we revised our regulations at § 419.43(g) to clarify that essential access community hospitals (EACHs) are also eligible to receive the rural SCH adjustment, assuming these entities otherwise meet the rural adjustment criteria. Currently, two hospitals are classified as EACHs, and as of CY 1998, under section 4201(c) of the Balanced Budget Act of 1997 (BBA) (Pub. L. 105-33), a hospital can no longer become newly classified as an EACH.
This adjustment for rural SCHs is budget neutral and applied before calculating outlier payments and copayments. We stated in the CY 2006 OPPS final rule with comment period (70 FR 68560) that we would not reestablish the adjustment amount on an annual basis, but we may review the adjustment in the future and, if appropriate, would revise the adjustment. We provided the same 7.1 percent adjustment to rural SCHs, including EACHs, again in CYs 2008 through 2024.
For CY 2025, we proposed to continue the current policy of a 7.1 percent payment adjustment for rural SCHs, including EACHs, for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, brachytherapy sources, items paid at charges reduced to costs, and devices paid under the pass-through payment policy, applied in a budget neutral manner.
Comment: Many commenters supported our policy to continue the 7.1 percent payment adjustment for rural SCHs and EACHs.
Response: We thank the commenters for their support of our policy.
Comment: Multiple commenters stated that the 7.1 percent payment adjustment should be extended to urban SCHs and Medicare dependent hospitals (MDHs). The commenters suggested CMS perform another study to look at the costs that MDHs incur and that CMS has the authority to extend the adjustment to MDHs and urban SCHs without legislation.
Response: We reiterate that our study of the cost difference by APC's between hospitals in rural areas and urban areas only showed a significant difference in costs for rural SCHs. CMS results did not identify significant cost differences between hospitals in urban areas and MDHs. In addition, our authority under section 1833(t)(13) of the Act only extends to rural hospitals. Therefore, we are not expanding the types of hospitals eligible for the 7.1 percent payment adjustment at this time.
After consideration of the public comments we received, we are finalizing our proposal, without modification, to continue our current policy of utilizing a budget neutral 7.1 percent payment adjustment for rural SCHs, including EACHs, for all services and procedures paid under the OPPS, excluding separately payable drugs and biologicals, devices paid under the pass-through payment policy, and items paid at charges reduced to costs.
F. Payment Adjustment for Certain Cancer Hospitals for CY 2025
1. Background
Since the inception of the OPPS, which was authorized by the Balanced Budget Act of 1997 (BBA) (Pub. L. 105-33), Medicare has paid the 11 hospitals that meet the criteria for cancer hospitals identified in section 1886(d)(1)(B)(v) of the Act under the OPPS for covered outpatient department services. These cancer hospitals are exempted from payment under the IPPS. With the Medicare, Medicaid and SCHIP Balanced Budget Refinement Act of 1999 (Pub. L. 106-113), the Congress added section 1833(t)(7), “Transitional Adjustment to Limit Decline in Payment,” to the Act, which requires the Secretary to determine OPPS payments to cancer and children's hospitals based on their pre-BBA ( printed page 93978) payment amount (these hospitals are often referred to under this policy as “held harmless” and their payments are often referred to as “hold harmless” payments).
As required under section 1833(t)(7)(D)(ii) of the Act, a cancer hospital receives the full amount of the difference between payments for covered outpatient department services under the OPPS and a “pre-BBA amount.” That is, cancer hospitals are permanently held harmless to their “pre-BBA amount,” and they receive transitional outpatient payments (TOPs) or hold harmless payments to ensure that they do not receive a payment that is lower in amount under the OPPS than the payment amount they would have received before implementation of the OPPS, as set forth in section 1833(t)(7)(F) of the Act. The “pre-BBA amount” is the product of the hospital's reasonable costs for covered outpatient department services occurring in the current year and the base payment-to-cost ratio (PCR) for the hospital defined in section 1833(t)(7)(F)(ii) of the Act. The “pre-BBA amount” and the determination of the base PCR are defined at § 419.70(f). TOPs are calculated on Worksheet E, Part B, of the Hospital Cost Report or the Hospital Health Care Complex Cost Report (Form CMS-2552-96 or Form CMS-2552-10 (OMB NO: 0938-0050), respectively), as applicable each year. Section 1833(t)(7)(I) of the Act exempts TOPs from budget neutrality calculations.
Section 3138 of the Affordable Care Act (Pub. L. 111-148) amended section 1833(t) of the Act by adding a new paragraph (18), which instructs the Secretary to conduct a study to determine if, under the OPPS, outpatient costs incurred by cancer hospitals described in section 1886(d)(1)(B)(v) of the Act with respect to APC groups exceed outpatient costs incurred by other hospitals furnishing services under section 1833(t) of the Act, as determined appropriate by the Secretary. Section 1833(t)(18)(A) of the Act requires the Secretary to take into consideration the cost of drugs and biologicals incurred by cancer hospitals and other hospitals. Section 1833(t)(18)(B) of the Act provides that, if the Secretary determines that cancer hospitals' costs are higher than those of other hospitals, the Secretary shall provide an appropriate adjustment under section 1833(t)(2)(E) of the Act to reflect these higher costs. In 2011, after conducting the study required by section 1833(t)(18)(A) of the Act, we determined that outpatient costs incurred by the 11 specified cancer hospitals were greater than the costs incurred by other OPPS hospitals. For a complete discussion regarding the cancer hospital cost study, we refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74200 and 74201).
Based on these findings, we finalized a policy to provide a payment adjustment to the 11 specified cancer hospitals that reflects their higher outpatient costs, as discussed in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74202 through 74206). Specifically, we adopted a policy to provide additional payments to the cancer hospitals so that each cancer hospital's final PCR for services provided in a given calendar year is equal to the weighted average PCR (which we refer to as the “target PCR”) for other hospitals paid under the OPPS. The target PCR is set in advance of the calendar year and is calculated using the most recently submitted or settled cost report data that are available at the time of final rulemaking for the calendar year. The amount of the payment adjustment is made on an aggregate basis at cost report settlement. We note that the changes made by section 1833(t)(18) of the Act do not affect the existing statutory provisions that provide for TOPs for cancer hospitals. The TOPs are assessed, as usual, after all payments, including the cancer hospital payment adjustment, have been made for a cost reporting period. Table 11 displays the target PCR for purposes of the cancer hospital adjustment for CY 2012 through CY 2024.
2. Policy for CY 2025
Section 16002(b) of the 21st Century Cures Act (Pub. L. 114-255) amended section 1833(t)(18) of the Act by adding subparagraph (C), which requires that in applying § 419.43(i) (that is, the payment adjustment for certain cancer hospitals) for services furnished on or after January 1, 2018, the target PCR adjustment be reduced by 1.0 percentage point less than what would ( printed page 93979) otherwise apply. Section 16002(b) also provides that, in addition to the percentage reduction, the Secretary may consider making an additional percentage point reduction to the target PCR that takes into account payment rates for applicable items and services described under section 1833(t)(21)(C) of the Act for hospitals that are not cancer hospitals described under section 1886(d)(1)(B)(v) of the Act. Further, in making any budget neutrality adjustment under section 1833(t) of the Act, the Secretary shall not take into account the reduced expenditures that result from application of section 1833(t)(18)(C) of the Act.
We proposed to provide additional payments to the 11 specified cancer hospitals so that each cancer hospital's proposed PCR is equal to the weighted average PCR (or “target PCR”) for the other OPPS hospitals, generally using the most recent submitted or settled cost report data that are available, reduced by 1.0 percentage point, to comply with section 16002(b) of the 21st Century Cures Act, and adjusted by the proposed post-Public Health Emergency (PHE) transition as described later in this section. We are not proposing an additional reduction beyond the 1.0 percentage point reduction required by section 16002(b) of the 21st Century Cures Act for CY 2025.
To calculate the proposed CY 2025 target PCR, we proposed to use the same extract of cost report data from HCRIS used to estimate costs for the CY 2025 OPPS which, in most cases, would be the most recently available hospital cost reports. Using these cost report data, we included data from Worksheet E, Part B, for each hospital, using data from each hospital's most recent cost report, whether as submitted or settled.
We then limited the dataset to the hospitals with CY 2023 claims data that we used to model the impact of the proposed CY 2025 APC relative payment weights (3,448 hospitals) because it is appropriate to use the same set of hospitals that are being used to calibrate the modeled CY 2025 OPPS. The cost report data for the hospitals in this dataset were from cost report periods with fiscal year ends ranging from 2019 to 2023; however, the cost reporting periods were predominantly from fiscal years ending in 2022 and 2023. We then removed the cost report data of the 49 hospitals located in Puerto Rico from our dataset because we did not believe their cost structure reflected the costs of most hospitals paid under the OPPS, and, therefore, their inclusion may bias the calculation of hospital-weighted statistics. We also removed the cost report data of 16 hospitals because these hospitals had cost report data that were not complete (missing aggregate OPPS payments, missing aggregate cost data, or missing both), so that all cost reports in the study would have both the payment and cost data necessary to calculate a PCR for each hospital, leading to a proposed analytic file of 3,421 hospitals with cost report data.
Using this smaller dataset of cost report data, we estimated that, on average, the OPPS payments to other hospitals furnishing services under the OPPS were approximately 87 percent of reasonable cost (weighted average PCR of 0.87). Therefore, after applying the 1.0 percentage point reduction, as required by section 16002(b) of the 21st Century Cures Act, using our standard process the payment amount associated with the cancer hospital payment adjustment to be determined at cost report settlement would be the additional payment needed to result in a target PCR equal to 0.86 for each cancer hospital.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81586 through 81589), we explained that we believe we should begin to take into consideration the PCR of non-cancer hospitals based on the most recently available data for calculating the target PCR. We noted that we do not know if the changes in the data that have yielded lower PCRs for non-cancer hospitals are likely to continue in future years or if, when data from after the PHE is available, we will see the target PCR increase toward its historical norm. Therefore, in the CY 2024 OPPS/ASC final rule with comment period, we finalized our proposal to transition from the target PCR of 0.89 we finalized for CYs 2020 through 2024 (which included the 1.0 percentage point reduction as required by section 16002(b) of the 21st Century Cures Act) and incrementally reduce the target PCR by an additional 1.0 percentage point for each calendar year, beginning with CY 2024, until the target PCR equals the PCR of non-cancer hospitals calculated using the most recent data minus 1.0 percentage point as required by section 16002(b) of the 21st Century Cures Act. Therefore, utilizing this methodology for the CY 2025 OPPS/ASC proposed rule, since the target PCR that would otherwise apply under our standard process would be a target PCR of 0.86, we proposed to reduce the CY 2024 target PCR of 0.88 by 1 percentage point and proposed a cancer hospital target PCR of 0.87 for CY 2025.
Comment: Commenters supported the proposed target PCR of 0.87 for CY 2025. One commenter requested that CMS ensure that repayments to 340B hospitals are appropriately reflected in the final 2025 PCR calculation.
Response: We thank the commenters for their support. With respect to the commenter's request that the final 2025 PCR calculation include repayments to 340B hospitals, we refer to our response to that same comment in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81588).
After consideration of the public comments we received, we are finalizing without modification our proposal to reduce the CY 2024 target PCR by 1 percentage point and apply a target PCR of 0.87 to the 11 specified cancer hospitals for CY 2025.
Table 12 shows the estimated percentage increase in OPPS payments to each cancer hospital for CY 2025, due to the cancer hospital payment adjustment policy. The actual, final amount of the CY 2025 cancer hospital payment adjustment for each cancer hospital will be determined at cost report settlement and will depend on each hospital's CY 2025 payments and costs from the settled CY 2025 cost report. We note that the requirements contained in section 1833(t)(18) of the Act do not affect the existing statutory provisions that provide for TOPs for cancer hospitals. The TOPs will be assessed, as usual, after all payments, including the cancer hospital payment adjustment, have been made for a cost reporting period.
( printed page 93980)G. Hospital Outpatient Outlier Payments
1. Background
The OPPS provides outlier payments to hospitals to help mitigate the financial risk associated with high-cost and complex procedures, where a very costly service could present a hospital with significant financial loss. As explained in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66832 through 66834), we set our projected target for aggregate outlier payments at 1.0 percent of the estimated aggregate total payments under the OPPS for the prospective year. Outlier payments are provided on a service-by-service basis when the cost of a service exceeds the APC payment amount multiplier threshold (the APC payment amount multiplied by a certain amount) as well as the APC payment amount plus a fixed-dollar amount threshold (the APC payment plus a certain dollar amount). In CY 2024, the outlier threshold was met when the hospital's cost of furnishing a service exceeded 1.75 times the APC payment amount (the multiplier threshold) and exceeded the APC payment amount plus $7,750 (the fixed-dollar amount threshold) (88 FR 81589 through 81591). If the hospital's cost of furnishing a service exceeds both the multiplier threshold and the fixed-dollar threshold, the outlier payment is calculated as 50 percent of the amount by which the hospital's cost of furnishing the service exceeds 1.75 times the APC payment amount. Beginning with CY 2009 payments, outlier payments are subject to a reconciliation process similar to the IPPS outlier reconciliation process for cost reports, as discussed in the CY 2009 OPPS/ASC final rule with comment period (73 FR 68594 through 68599).
It has been our policy to report the actual amount of outlier payments as a percent of total spending in the claims being used to model the OPPS. Our estimate of total outlier payments as a percent of total CY 2023 OPPS payments, using CY 2023 claims available for this CY 2025 OPPS/ASC final rule with comment period, is approximately 0.65 percent. Therefore, for CY 2023, we estimate that we did not meet the outlier target by 0.35 percent of total aggregated OPPS payments.
For this CY 2025 OPPS/ASC final rule with comment period, using CY 2023 claims data and CY 2024 payment rates, we estimate that the aggregate outlier payments for CY 2024 would be approximately 0.83 percent of the total CY 2024 OPPS payments. We provide estimated CY 2025 outlier payments for hospitals and CMHCs with claims included in the claims data that we used to model impacts in the Hospital-Specific Impacts—Provider-Specific Data file on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
2. Outlier Calculation for CY 2025
For CY 2025, we proposed to continue our policy of estimating outlier payments to be 1.0 percent of the estimated aggregate total payments under the OPPS. We proposed that a portion of that 1.0 percent, an amount equal to less than 0.01 percent of outlier payments (or 0.0001 percent of total OPPS payments), would be allocated to CMHCs for partial hospitalization program (PHP) and intensive outpatient program (IOP) outlier payments. This is the amount of estimated outlier payments that would result from the proposed CMHC outlier threshold as a proportion of total estimated OPPS ( printed page 93981) outlier payments. We proposed to continue our outlier policy that if a CMHC's cost for PHP and IOP services exceeds 3.40 times the APC payment rate, the outlier payment would be calculated as 50 percent of the amount by which the cost exceeds 3.40 times the proposed APC payment rate.
For further discussion of CMHC outlier payments, we refer readers to section VIII.C of this final rule with comment period.
To ensure that the estimated CY 2025 aggregate outlier payments would equal 1.0 percent of estimated aggregate total payments under the OPPS, we proposed that the hospital outlier threshold be set so that outlier payments would be triggered when a hospital's cost of furnishing a service exceeds 1.75 times the APC payment amount and exceeds the APC payment amount plus the fixed-dollar threshold.
We calculated the proposed fixed-dollar threshold using the standard methodology most recently used for CY 2024 (88 FR 81589 through 81591). For purposes of estimating outlier payments for CY 2025, we use the hospital-specific overall ancillary CCRs available in the April 2024 update to the Outpatient Provider-Specific File (OPSF). The OPSF contains provider-specific data, such as the most current CCRs, which are maintained by the MACs and used by the OPPS Pricer to pay claims. The claims that we generally use to model each OPPS update lag by 2 years.
In order to estimate the CY 2025 hospital outlier payments, we inflated the charges on the CY 2023 claims using the same proposed charge inflation factor of 1.084555 that we used to estimate the IPPS fixed-loss cost threshold for the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36572). We used an inflation factor of 1.04142 to estimate CY 2024 charges from the CY 2023 charges reported on CY 2023 claims before applying CY 2024 CCRs to estimate the percent of outliers paid in CY 2024. The proposed methodology for determining these charge inflation factors is discussed in the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36572). As we stated in the CY 2005 OPPS final rule with comment period (69 FR 65844 through 65846), we believe that the use of the same charge inflation factors is appropriate for the OPPS because, with the exception of the inpatient routine service cost centers, hospitals use the same ancillary and cost centers to capture costs and charges for inpatient and outpatient services.
As noted in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68011), we are concerned that we could systematically overestimate the OPPS hospital outlier threshold if we did not apply a CCR inflation adjustment factor. Therefore, we proposed to apply the same CCR adjustment factor that we proposed to apply for the FY 2025 IPPS outlier calculation to the CCRs used to simulate the proposed CY 2025 OPPS outlier payments to determine the fixed-dollar threshold. Specifically, for CY 2025, we proposed to apply an adjustment factor of 1.03331 to the CCRs that were in the April 2024 OPSF to trend them forward from CY 2024 to CY 2025. The methodology for calculating the proposed CCR adjustment factor is discussed in the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36572 through 36573).
To model hospital outlier payments for the CY 2025 proposed rule, we applied the overall CCRs from the April 2024 OPSF after adjustment (using the proposed CCR inflation adjustment factor of 1.03331 to approximate CY 2025 CCRs) to charges on CY 2023 claims that were adjusted (using the proposed charge inflation factor of 1.084555 to approximate CY 2025 charges). We simulated aggregated CY 2023 hospital outlier payments using these costs for several different fixed-dollar thresholds, holding the 1.75 multiplier threshold constant and assuming that outlier payments would continue to be made at 50 percent of the amount by which the cost of furnishing the service would exceed 1.75 times the APC payment amount, until the total outlier payments equaled 1.0 percent of aggregated estimated total CY 2025 OPPS payments. We estimated that a proposed fixed-dollar threshold of $8,000, combined with the proposed multiplier threshold of 1.75 times the APC payment rate, would allocate 1.0 percent of aggregated total OPPS payments to outlier payments. For CMHCs, we proposed that, if a CMHC's cost for partial hospitalization or intensive outpatient services exceeds 3.40 times the APC payment rate, the outlier payment would be calculated as 50 percent of the amount by which the cost exceeds 3.40 times the APC payment rate.
Section 1833(t)(17)(A) of the Act, which applies to hospitals, as defined under section 1886(d)(1)(B) of the Act, requires that hospitals that fail to report data required for the quality measures selected by the Secretary, in the form and manner required by the Secretary under section 1833(t)(17)(B) of the Act, incur a 2.0 percentage point reduction to their OPD fee schedule increase factor; that is, the annual payment update factor. The application of a reduced OPD fee schedule increase factor results in reduced national unadjusted payment rates that would apply to certain outpatient items and services furnished by hospitals that are required to report outpatient quality data and that fail to meet the Hospital Outpatient Quality Reporting (OQR) Program requirements. For hospitals that fail to meet the Hospital OQR Program requirements, we proposed to continue the policy that we implemented in CY 2010 that the hospitals' costs would be compared to the reduced payments for purposes of outlier eligibility and payment calculation. For more information on the Hospital OQR Program, we refer readers to section XV of this final rule with comment period.
Comment: One commenter noted that they believe the CCR adjustment factors used for the IPPS and OPPS proposed rules overstate the effect of cost increases that took place from 2022 to 2023. The commenter argues that the CCR adjustment factor used in each final rule has been below 1.0; however, we proposed a CCR adjustment factor of 1.03331. The commenter recommended that we substitute a different CCR adjustment factor or cap the CCR adjustment factor at 1.0.
Response: As discussed in greater detail in the FY 2025 IPPS/LTCH PPS final rule (89 FR 69955 through 69962), consistent with our usual practice, we are using more recent CCR data for this final rule with comment period which yields a CCR adjustment factor of 1.015123. We do not believe it is necessary to deviate from our usual practice of using the CCR adjustment factor for our calculation, which was finalized under the IPPS for FY 2025. Therefore, we are also not accepting this recommendation under the OPPS.
After consideration of the public comments we received, we are finalizing our proposal, without modification, to continue our policy of estimating outlier payments to be 1.0 percent of the estimated aggregate total payments under the OPPS and to use our established methodology to set the OPPS outlier fixed-dollar loss threshold for CY 2025.
3. Final Outlier Calculation
Consistent with historical practice, we used updated data for this final rule with comment period for outlier calculations. For CY 2025, we are applying the overall ancillary CCRs from the July 2024 OPSF file after adjustment (using the CCR adjustment factor of 1.015123 to approximate CY 2025 CCRs) to charges on CY 2023 claims that were adjusted using a charge inflation factor of 1.08406 to ( printed page 93982) approximate CY 2025 charges. These are the same CCR adjustment and charge inflation factors that were used to set the IPPS fixed-dollar thresholds for the FY 2025 IPPS/LTCH PPS final rule (89 FR 69955 through 69962). We simulated aggregated CY 2025 hospital outlier payments using these costs for several different fixed-dollar thresholds, holding the 1.75 multiple-threshold constant and assuming that outlier payments will continue to be made at 50 percent of the amount by which the cost of furnishing the service would exceed 1.75 times the APC payment amount, until the total outlier payment equaled 1.0 percent of aggregated estimated total CY 2025 OPPS payments. We estimate that a fixed-dollar threshold of $7,175 combined with the multiple threshold of 1.75 times the APC payment rate, will allocate the 1.0 percent of aggregated total OPPS payments to outlier payments.
For CMHCs, if a CMHC's cost for partial hospitalization or intensive outpatient services exceeds 3.40 times the APC payment rate, the outlier payment will be calculated as 50 percent of the amount by which the cost exceeds the 3.40 times the ACP payment rate.
H. Calculation of an Adjusted Medicare Payment From the National Unadjusted Medicare Payment
The national unadjusted payment rate is the payment rate for most APCs before accounting for the wage index adjustment or any applicable adjustments. The basic methodology for determining prospective payment rates for HOPD services under the OPPS is set forth in existing regulations at 42 CFR part 419, subparts C and D. For this CY 2025 OPPS/ASC final rule with comment period, the payment rate for most services and procedures for which payment is made under the OPPS is the product of the conversion factor calculated in accordance with section II.B of this final rule with comment period and the relative payment weight described in section II.A of this final rule with comment period. The national unadjusted payment rate for most APCs contained in Addendum A to this final rule with comment period (which is available via the CMS website https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/addendum-a-b-updates) and for most HCPCS codes to which separate payment under the OPPS has been assigned in Addendum B to this final rule with comment period (which is available on the CMS website link above) is calculated by multiplying the final CY 2025 scaled weight for the APC by the CY 2025 conversion factor.
We note that section 1833(t)(17) of the Act, which applies to hospitals, as defined under section 1886(d)(1)(B) of the Act, requires that hospitals that fail to submit data required to be submitted on quality measures selected by the Secretary, in the form and manner and at a time specified by the Secretary, incur a reduction of 2.0 percentage points to their OPD fee schedule increase factor, that is, the annual payment update factor. The application of a reduced OPD fee schedule increase factor results in reduced national unadjusted payment rates that apply to certain outpatient items and services provided by hospitals that are required to report outpatient quality data and that fail to meet the Hospital OQR Program requirements. For further discussion of the payment reduction for hospitals that fail to meet the requirements of the Hospital OQR Program, we refer readers to section XIV of this final rule with comment period.
Below we demonstrate the steps used to determine the APC payments that will be made in a CY under the OPPS to a hospital that fulfills the Hospital OQR Program requirements and to a hospital that fails to meet the Hospital OQR Program requirements for a service that has any of the following status indicator assignments: “J1,” “J2,” “P,” “Q1,” “Q2,” “Q3,” “Q4,” “R,” “S,” “T,” “U,” or “V” (as defined in Addendum D1 to this final rule with comment period, which is available via the internet on the CMS website), in a circumstance in which the multiple procedure discount does not apply, the procedure is not bilateral, and conditionally packaged services (status indicator of “Q1” and “Q2”) qualify for separate payment. We note that, although blood and blood products with status indicator “R” and brachytherapy sources with status indicator “U” are not subject to wage adjustment, they are subject to reduced payments when a hospital fails to meet the Hospital OQR Program requirements.
Individual providers interested in calculating the payment amount that they would receive for a specific service from the national unadjusted payment rates presented in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website) should follow the formulas presented in the following steps. For purposes of the payment calculations below, we refer to the national unadjusted payment rate for hospitals that meet the requirements of the Hospital OQR Program as the “full” national unadjusted payment rate. We refer to the national unadjusted payment rate for hospitals that fail to meet the requirements of the Hospital OQR Program as the “reduced” national unadjusted payment rate. The reduced national unadjusted payment rate is calculated by multiplying the reporting ratio of 0.9806 times the “full” national unadjusted payment rate. The national unadjusted payment rate used in the calculations below is either the full national unadjusted payment rate or the reduced national unadjusted payment rate, depending on whether the hospital met its Hospital OQR Program requirements to receive the full CY 2025 OPPS fee schedule increase factor.
Step 1. Calculate 60 percent (the labor-related portion) of the national unadjusted payment rate. Since the initial implementation of the OPPS, we have used 60 percent to represent our estimate of that portion of costs attributable, on average, to labor. We refer readers to the April 7, 2000 OPPS/ASC final rule with comment period (65 FR 18496 through 18497) for a detailed discussion of how we derived this percentage. During our regression analysis for the payment adjustment for rural hospitals in the CY 2006 OPPS final rule with comment period (70 FR 68553), we confirmed that this labor-related share for hospital outpatient services is appropriate.
The formula below is a mathematical representation of Step 1 and identifies the labor-related portion of a specific payment rate for a specific service.
X is the labor-related portion of the national unadjusted payment rate.
X = .60 * (national unadjusted payment rate).
Step 2. Determine the wage index area in which the hospital is located and identify the wage index level that applies to the specific hospital. The wage index values assigned to each area would reflect the geographic statistical areas (which are based upon OMB standards) to which hospitals are assigned for FY 2025 in the FY 2025 IPPS, as corrected in the FY 2025 IPPS final rule correction (89 FR 80098), reclassifications through the Medicare Geographic Classification Review Board (MGCRB), section 1886(d)(8)(B) “Lugar” hospitals, and reclassifications under section 1886(d)(8)(E) of the Act, as implemented in § 412.103 of the regulations. We are continuing to apply for the CY 2025 OPPS wage index any adjustments for the FY 2025 IPPS final rule post-reclassified wage index, including, but not limited to, the rural floor adjustment and a wage index floor of 1.00 in frontier states, in accordance with section 10324 of the Affordable ( printed page 93983) Care Act of 2010. We are also including the low wage index hospital policy as part of the CY 2025 OPPS wage index. The adoption of this policy for CY 2025 for the OPPS wage index will create different effective wage index values for hospitals that benefit from the policy under the OPPS than the wage index value for those hospitals under the IPPS for FY 2025 as modified by the interim final action with comment period “Changes to the Fiscal Year 2025 Hospital Inpatient Prospective Payment System (IPPS) Rates Due to Court Decision” (89 FR 80405). For further discussion of the wage index we are applying for the CY 2025 OPPS, including the low wage index hospital policy, we refer readers to section II.C of this final rule with comment period.
Step 3. Adjust the wage index of hospitals located in certain qualifying counties that have a relatively high percentage of hospital employees who reside in the county, but who work in a different county with a higher wage index, in accordance with section 505 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (Pub. L. 108-173). Addendum L to this final rule with comment period (which is available via the internet on the CMS website) contains the qualifying counties and the associated wage index increase developed for the final FY 2025 IPPS wage index, which are listed in Table 3 associated with the FY 2025 IPPS final rule and available via the internet on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps. (Click on the link on the left side of the screen titled “FY 2025 IPPS Final Rule Home Page” and select “FY 2025 Final Rule Tables.”) This step is to be followed only if the hospital is not reclassified or redesignated under section 1886(d)(8) or section 1886(d)(10) of the Act.
Step 4. Multiply the applicable wage index determined under Steps 2 and 3 by the amount determined under Step 1 that represents the labor-related portion of the national unadjusted payment rate.
The formula below is a mathematical representation of Step 4 and adjusts the labor-related portion of the national unadjusted payment rate for the specific service by the wage index.
Xais the labor-related portion of the national unadjusted payment rate (wage adjusted).
Xa = labor-portion of the national unadjusted payment rate * applicable wage index.
Step 5. Calculate 40 percent (the nonlabor-related portion) of the national unadjusted payment rate and add that amount to the resulting product of Step 4. The result is the wage index adjusted payment rate for the relevant wage index area.
The formula below is a mathematical representation of Step 5 and calculates the remaining portion of the national payment rate, the amount not attributable to labor, and the adjusted payment for the specific service.
Y is the nonlabor-related portion of the national unadjusted payment rate.
Y = 0.40 * (national unadjusted payment rate).
Step 6. If a provider is an SCH, as set forth in the regulations at § 412.92, or an EACH, which is considered to be an SCH under section 1886(d)(5)(D)(iii)(III) of the Act, and located in a rural area, as defined in § 412.64(b), or is treated as being located in a rural area under § 412.103, multiply the wage index adjusted payment rate by 1.071 to calculate the total payment.
The formula below is a mathematical representation of Step 6 and applies the rural adjustment for rural SCHs.
Adjusted Medicare Payment (SCH or EACH) = Adjusted Medicare Payment * 1.071.
Step 7. The adjusted payment rate is the sum of the wage adjusted labor-related portion of the national unadjusted payment rate and the nonlabor-related portion of the national unadjusted payment rate.
Xais the labor-related portion of the national unadjusted payment rate (wage adjusted).
Y is the nonlabor-related portion of the national unadjusted payment rate.
Adjusted Medicare Payment = Xa + Y
We are providing examples below of the calculation of both the full and reduced national unadjusted payment rates that would apply to certain outpatient items and services performed by hospitals that meet and that fail to meet the Hospital OQR Program requirements, using the steps outlined previously. For purposes of this example, we are using a provider that is located in Brooklyn, New York that is assigned to CBSA 35614. This provider bills one service that is assigned to APC 5071 (Level 1 Excision/Biopsy/Incision and Drainage). The final CY 2025 full national unadjusted payment rate for APC 5071 is $703.59. The final reduced national adjusted payment rate for APC 5071 for a hospital that fails to meet the Hospital OQR Program requirements is $689.94. This reduced rate is calculated by multiplying the reporting ratio of 0.9806 by the full unadjusted payment rate for APC 5071.
Step 1. The labor-related portion of the final full national unadjusted payment is approximately $422.15 (0.60 * $703.59). The labor-related portion of the final reduced national adjusted payment is approximately $413.96 (0.60 * $689.94).
Step 2 & 3. The FY 2025 wage index for a provider located in CBSA 35614 in New York, which includes the adoption of the final IPPS 2025 wage index policies, is 1.3056.
Step 4. The wage adjusted labor-related portion of the final full national unadjusted payment is approximately $551.16 ($422.15 * 1.3056). The wage adjusted labor-related portion of the final reduced national adjusted payment is approximately $540.47 ($413.96 * 1.3056).
Step 5. The nonlabor-related portion of the final full national unadjusted payment is approximately $281.44 (0.40 * $703.59). The nonlabor-related portion of the final reduced national adjusted payment is approximately $275.98 (0.40 * $689.94).
Step 6. For this example of a provider located in Brooklyn, New York, the rural adjustment for rural SCHs does not apply.
Step 7. The sum of the labor-related and nonlabor-related portions of the final full national unadjusted payment is approximately $832.60 ($551.16 + $281.44). The sum of the portions of the final reduced national adjusted payment is approximately $816.45 ($540.47 + $275.98) as shown in Table 13.
( printed page 93984)We did not receive any public comments on these steps under the methodology that we included in the proposed rule to determine the APC payments for CY 2025. Therefore, we are using the steps in the methodology specified above to demonstrate the calculation of the final CY 2025 OPPS payments using the same parameters.
I. Beneficiary Copayments
1. Background
Section 1833(t)(3)(B) of the Act requires the Secretary to set rules for determining the unadjusted copayment amounts to be paid by beneficiaries for covered OPD services. Section 1833(t)(8)(C)(ii) of the Act specifies that the Secretary must reduce the national unadjusted copayment amount for a covered OPD service (or group of such services) furnished in a year in a manner so that the effective copayment rate (determined on a national unadjusted basis) for that service in the year does not exceed a specified percentage. As specified in section 1833(t)(8)(C)(ii)(V) of the Act, the effective copayment rate for a covered OPD service paid under the OPPS in CY 2006, and in CYs thereafter, shall not exceed 40 percent of the APC payment rate.
Section 1833(t)(3)(B)(ii) of the Act provides that, for a covered OPD service (or group of such services) furnished in a year, the national unadjusted copayment amount cannot be less than 20 percent of the OPD fee schedule amount. However, section 1833(t)(8)(C)(i) of the Act limits the amount of beneficiary copayment that may be collected for a procedure (including items such as drugs and biologicals) performed in a year to the amount of the inpatient hospital deductible for that year.
Section 4104 of the Affordable Care Act eliminated the Medicare Part B coinsurance for preventive services furnished on and after January 1, 2011, that meet certain requirements, including flexible sigmoidoscopies and screening colonoscopies, and waived the Part B deductible for screening colonoscopies that become diagnostic during the procedure. For a discussion of the changes made by the Affordable Care Act with regard to copayments for preventive services furnished on and after January 1, 2011, we refer readers to section XII.B of the CY 2011 OPPS/ASC final rule with comment period (75 FR 72013).
Section 122 of the Consolidated Appropriations Act (CAA) of 2021 (Pub. L. 116-260), Waiving Medicare Coinsurance for Certain Colorectal Cancer Screening Tests, amended section 1833(a) of the Act to offer a special coinsurance rule for screening flexible sigmoidoscopies and screening colonoscopies, regardless of the code that is billed for the establishment of a diagnosis as a result of the test, or for the removal of tissue or other matter or other procedure, that is furnished in connection with, as a result of, and in the same clinical encounter as the colorectal cancer screening test. We refer readers to section X.B, “Changes to Beneficiary Coinsurance for Certain Colorectal Cancer Screening Tests,” of the CY 2022 OPPS/ASC final rule with comment period for the full discussion of this policy (86 FR 63740 through 63743). Under the regulation at 42 CFR 410.152(l)(5)(i)(B), the Medicare Part B payment percentage for colorectal cancer screening tests described in the regulation at § 410.37(j) that are furnished in CY 2023 through CY 2026 is 85 percent, with beneficiary coinsurance equal to 15 percent.
On August 16, 2022, the Inflation Reduction Act of 2022 (IRA) (Pub. L. 117-169) was signed into law. Section 11101(a) of the IRA amended section 1847A of the Act by adding a new subsection (i), which requires the payment of rebates into the Supplementary Medical Insurance Trust Fund for Part B rebatable drugs if the payment limit amount exceeds the inflation-adjusted payment amount, which is calculated as set forth in section 1847A(i)(3)(C) of the Act. The provisions of section 11101 of the IRA thus far have primarily been implemented through program instruction, as permitted under section 1847A(c)(5)(C) of the Act. As such, we issued guidance for the computation of inflation-adjusted beneficiary coinsurance under section 1847A(i)(5) of the Act and amounts paid under section 1833(a)(1)(EE) of the Act on February 9, 2023.[12 13] For additional information regarding implementation of section 11101 of the IRA, please see the inflation rebates resources page at https://www.cms.gov/inflation-reduction-act-and-medicare/inflation-rebates-medicare.
Section 11101(b) of the IRA amended sections 1833(i) and 1833(t)(8) of the Act by adding a new paragraph (9) and subparagraph (F), respectively. Section 1833(i)(9) requires under the ASC payment system that, in the case of a Part B rebatable drug, in lieu of calculation of coinsurance that would otherwise apply under the ASC payment system, the provisions of section 1847A(i)(5) of the Act shall, as determined appropriate by the Secretary, apply for calculation of beneficiary coinsurance in the same manner as the provisions of section 1847A(i)(5) of the Act apply under that section. Similarly, section 1833(t)(8)(F) of the Act requires under the OPPS that in the case of a Part B rebatable drug (except for a drug that has no copayment applied under subparagraph (E) of such section or for which payment is packaged into the payment for a covered OPD service or group of services), in lieu of the calculation of the copayment amount that would otherwise apply under the OPPS, the provisions of section 1847A(i)(5) of the Act shall, as determined appropriate by the Secretary, apply in the same manner as the provisions of section 1847A(i)(5) of the Act apply under that section. Section 1847A(i)(5) of the Act requires that for Part B rebatable drugs, as defined in section 1847A(i)(2)(A) of the Act, furnished on or after April 1, 2023, in quarters in which the payment ( printed page 93985) amount described in section 1847A(i)(3)(A)(ii)(I) of the Act (or, in the case of selected drugs described under section 1192(c) of the Act, the payment amount described in section 1847A(b)(1)(B) of the Act), exceeds the inflation-adjusted payment amount determined in accordance with section 1847A(i)(3)(C) of the Act, the coinsurance will be 20 percent of the inflation-adjusted payment amount for such quarter (hereafter, the inflation-adjusted coinsurance amount). This inflation-adjusted coinsurance amount is applied as a percent, as determined by the Secretary, to the payment amount that would otherwise apply for such calendar quarter in accordance with section 1847A(b)(1)(B) or (C) of the Act, as applicable, including in the case of a selected drug.
Paragraph (9) of section 1833(i) of the Act and subparagraph (F) of section 1833(t)(8) of the Act, as added by section 11101(b) of the IRA, also provide that in lieu of the amounts of payment otherwise applicable under the ASC payment system and the OPPS, the provisions of paragraph (1)(EE) of subsection (a) of section 1833 of the Act shall apply, as determined appropriate by the Secretary. Section 11101(b) of the IRA amended section 1833(a)(1) of the Act by adding a new subparagraph (EE), which requires that if the payment amount under section 1847A(i)(3)(A)(ii)(I) of the Act or, in the case of a selected drug, the payment amount described in section 1847A(b)(1)(B) of the Act, for that drug exceeds the inflation-adjusted payment amount for a Part B rebatable drug, the Part B payment amount would, subject to the Part B deductible and sequestration, equal the difference between such payment amount and the inflation-adjusted coinsurance amount. Consistent with the policy adopted in section 40 of the revised Medicare Part B Drug Inflation Rebate Guidance, the calculation to determine the applicable beneficiary coinsurance amount would not be adjusted for sequestration. CMS codified the Medicare payment for Part B rebatable drugs in the CY 2024 PFS final rule by adding new paragraph (m) to § 410.152.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81594), we codified the OPPS program payment and cost sharing amounts for Part B rebatable drugs as required by section 1833(t)(8)(F) by adding a new paragraph (e) to § 419.41, which cross-references the regulations adopted in the CY 2024 PFS final rule (§§ 410.152(m) and 489.30(b)(6)). We also amended the regulation text to reflect our longstanding policies for calculating the Medicare program payment and cost sharing amounts for separately payable drugs and biologicals by adding a new paragraph (d) to § 419.41. Similarly, we codified the ASC cost sharing amounts for Part B rebatable drugs as required by section 1833(i)(9) of the Act by revising § 416.172(d) to include a cross-reference to 42 CFR 489.30(b)(6), which codified the cost sharing amounts for Part B rebatable drugs with prices increasing at a rate faster than inflation.
In the CY 2025 PFS proposed rule (89 FR 61956), CMS proposed to adopt new provisions at §§ 427.200 and 427.201 to codify the policies regarding the computation of the inflation-adjusted beneficiary coinsurance, defined in § 427.200, for Part B rebatable drugs as required by section 1847A(i)(5) of the Act. The proposed new provision at § 427.201 includes references to the existing provisions at §§ 410.152(m), 419.41(e), and 489.30(b)(6). CMS further proposed at § 427.201(c) that any category of products that is excluded from the identification of Part B rebatable drugs at § 427.101(b) is not subject to the inflation-adjusted beneficiary coinsurance. Examples of these excluded products include separately payable radiopharmaceuticals, skin substitute products, and qualifying biosimilar biological products.
Additionally, CMS proposed at § 427.201(b) that CMS would use the published payment amount in quarterly pricing files [14 15 16] to determine if a Part B rebatable drug should have an adjusted beneficiary coinsurance equal to 20 percent of the inflation-adjusted payment amount as described in section 1847A(i)(3)(C) for a calendar quarter. The proposed approach deviates from the rebate calculation approach proposed in § 427.302, which relies on the specified amount defined at § 427.20 even when the specified amount and the published payment amount in quarterly pricing files differ. The approach proposed at § 427.201(b) will be used only to determine whether there should be a coinsurance adjustment and will not impact the applicability or calculation of inflation rebates. CMS believes this approach is consistent with the statutory language and appropriately reflects the differences in the statutory text of section 1847A(i)(5) of the Act, which sets forth the payment amount that is used to determine whether coinsurance should be adjusted, and section 1847A(i)(3)(A) of the Act, which sets forth the “specified amount” used to determine rebate amounts.
We did not receive any public comments related to inflation-adjusted beneficiary coinsurance or program payment amounts.
2. OPPS Copayment Policy
For CY 2025, we proposed to determine copayment amounts for new and revised APCs using the same methodology that we implemented beginning in CY 2004. We refer readers to the November 7, 2003 OPPS final rule with comment period for a discussion of that methodology (68 FR 63458). In addition, we proposed to use the same standard rounding principles that we have historically used in instances where the application of our standard copayment methodology would result in a copayment amount that is less than 20 percent and cannot be rounded, under standard rounding principles, to 20 percent. We refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66687) in which we discuss our rationale for applying these rounding principles. The final national unadjusted copayment amounts for services payable under the OPPS that will be effective January 1, 2025, are included in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website).
As discussed in section XIV.E of this final rule with comment period, for CY 2025, the Medicare beneficiary's minimum unadjusted copayment and national unadjusted copayment for a service to which a reduced national unadjusted payment rate applies will equal the product of the reporting ratio and the national unadjusted copayment, or the product of the reporting ratio and the minimum unadjusted copayment, respectively, for the service.
We note that OPPS copayments may increase or decrease each year based on changes in the calculated APC payment rates, due to updated cost report and claims data, and any changes to the OPPS cost modeling process. However, as described in the CY 2004 OPPS final rule with comment period, the development of the copayment methodology generally moves beneficiary copayments closer to 20 percent of OPPS APC payments (68 FR 63458 through 63459).
In the CY 2004 OPPS final rule with comment period (68 FR 63459), we adopted a new methodology to calculate unadjusted copayment amounts in situations including reorganizing APCs, ( printed page 93986) and we finalized the following rules to determine copayment amounts in CY 2004 and subsequent years.
- When an APC group consists solely of HCPCS codes that were not paid under the OPPS the prior year because they were packaged or excluded or are new codes, the unadjusted copayment amount would be 20 percent of the APC payment rate.
- If a new APC that did not exist during the prior year is created and consists of HCPCS codes previously assigned to other APCs, the copayment amount is calculated as the product of the APC payment rate and the lowest coinsurance percentage of the codes comprising the new APC.
- If no codes are added to or removed from an APC and, after recalibration of its relative payment weight, the new payment rate is equal to orgreater than the prior year's rate, the copayment amount remains constant (unless the resulting coinsurance percentage is less than 20 percent).
- If no codes are added to or removed from an APC and, after recalibration of its relative payment weight, the new payment rate isless than the prior year's rate, the copayment amount is calculated as the product of the new payment rate and the prior year's coinsurance percentage.
- If HCPCS codes are added to or deleted from an APC and, after recalibrating its relative payment weight, holding its unadjusted copayment amount constant results in a decrease in the coinsurance percentage for the reconfigured APC, the copayment amount would not change (unless retaining the copayment amount would result in a coinsurance rate less than 20 percent).
- If HCPCS codes are added to an APC and, after recalibrating its relative payment weight, holding its unadjusted copayment amount constant results in an increase in the coinsurance percentage for the reconfigured APC, the copayment amount would be calculated as the product of the payment rate of the reconfigured APC and the lowest coinsurance percentage of the codes being added to the reconfigured APC.
We noted in the CY 2004 OPPS final rule with comment period that we would seek to lower the copayment percentage for a service in an APC from the prior year if the copayment percentage was greater than 20 percent. We noted that this principle was consistent with section 1833(t)(8)(C)(ii) of the Act, which accelerates the reduction in the national unadjusted coinsurance rate so that beneficiary liability will eventually equal 20 percent of the OPPS payment rate for all OPPS services to which a copayment applies, and with section 1833(t)(3)(B) of the Act, which achieves a 20-percent copayment percentage when fully phased in and gives the Secretary the authority to set rules for determining copayment amounts for new services. We further noted that the use of this methodology would, in general, reduce the beneficiary coinsurance rate and copayment amount for APCs for which the payment rate changes as the result of the reconfiguration of APCs and/or recalibration of relative payment weights (68 FR 63459).
We did not receive any public comments on our proposal and we are finalizing our proposal to determine copayment amounts for new and revised APCs using the same methodology that we implemented beginning in CY 2004. The finalized national unadjusted copayment amounts for services payable under the OPPS that will be effective January 1, 2025, are included in Addenda A and B to the CY 2025 OPPS/ASC final rule (which are available on the CMS website).
3. Calculation of an Adjusted Copayment Amount for an APC Group
Individuals interested in calculating the national copayment liability for a Medicare beneficiary for a given service provided by a hospital that met or failed to meet its Hospital OQR Program requirements should follow the formulas presented in the following steps.
Step 1. Calculate the beneficiary payment percentage for the APC by dividing the APC's national unadjusted copayment by its proposed payment rate. For example, using APC 5071, $140.72 is approximately 20 percent of the full national unadjusted payment rate of $703.59. For APCs with only a minimum unadjusted copayment in Addenda A and B to this final rule with comment period (which are available via the internet on the CMS website), the beneficiary payment percentage is 20 percent.
The formula below is a mathematical representation of Step 1 and calculates the national copayment as a percentage of national payment for a given service.
B is the beneficiary payment percentage.
B = National unadjusted copayment for APC/national unadjusted payment rate for APC.
Step 2. Calculate the appropriate wage-adjusted payment rate for the APC for the provider in question, as indicated in Steps 2 through 4 under section II.H of this final rule with comment period. Calculate the rural adjustment for eligible providers, as indicated in Step 6 under section II.H of this final rule with comment period.
Step 3. Multiply the percentage calculated in Step 1 by the payment rate calculated in Step 2. The result is the wage-adjusted copayment amount for the APC.
The formula below is a mathematical representation of Step 3 and applies the beneficiary payment percentage to the adjusted payment rate for a service calculated under section II.H of this final rule with comment period, with and without the rural adjustment, to calculate the adjusted beneficiary copayment for a given service.
Wage-adjusted copayment amount for the APC = Adjusted Medicare Payment * B.
Wage-adjusted copayment amount for the APC (SCH or EACH) = (Adjusted Medicare Payment * 1.071) * B.
Step 4. For a hospital that failed to meet its Hospital OQR Program requirements, multiply the copayment calculated in Step 3 by the reporting ratio of 0.9806.
The unadjusted copayments for services payable under the OPPS that will be effective January 1, 2025, are shown in Addenda A and B to this final rule with comment period (which are available via the CMS website). We note that the final national unadjusted payment rates and copayment rates shown in Addenda A and B to this final rule with comment period reflect the CY 2025 OPD fee schedule increase factor discussed in section II.B of this final rule with comment period.
In addition, as noted earlier, section 1833(t)(8)(C)(i) of the Act limits the amount of beneficiary copayment that may be collected for a procedure performed in a year to the amount of the inpatient hospital deductible for that year.
Comment: One commenter suggested beneficiaries should not be responsible for drug pricing increases and that CMS should limit or eliminate cost-sharing for beneficiaries. The commenter also stated that cost-sharing has unintended consequences and negative effects on access to health care and health outcomes, including increased use of emergency rooms. The commenter also acknowledged that CMS is limited by statute and that our copayment methodologies ensure that copayments are minimal.
Response: We thank the commenter for the input. We note that the beneficiary copayment is established by section 1833(t)(3)(B) of the Act and § 419.41(d). Section 1847A(i)(5) of the Act provides for an adjustment to the ( printed page 93987) beneficiary coinsurance for Part B rebatable drugs with prices that have increased faster than the rate of inflation beginning April 1, 2023. In the CY 2024 OPPS/ASC OPPS final rule and the CY 2024 Physician Fee Schedule final rule, CMS codified this inflation-adjusted coinsurance amount at §§ 419.41(e), 410.152(m), and 489.30(b)(6), respectively. For these drugs and biologicals, the beneficiary coinsurance is 20 percent of the inflation-adjusted payment amount, which is less than what the beneficiary would pay in coinsurance otherwise. Therefore, beneficiaries are insulated from coinsurance amounts calculated based on drug prices that outpace inflation. More information about the beneficiary coinsurance adjustment and the Medicare Part B Inflation Rebate Program is available at https://www.cms.gov/inflation-reduction-act-and-medicare/inflation-rebates-medicare.
III. OPPS Ambulatory Payment Classification (APC) Group Policies
A. OPPS Treatment of New and Revised HCPCS Codes
Payments for OPPS procedures, services, and items are generally based on medical billing codes, specifically, Healthcare Common Procedure Coding System (HCPCS) codes, that are reported on hospital outpatient department (HOPD) claims. HCPCS codes are used to report surgical procedures, medical services, items, and supplies under the hospital OPPS. The HCPCS is divided into two principal subsystems, referred to as Level I and Level II of the HCPCS. Level I is comprised of CPT (Current Procedural Terminology) codes, a numeric and alphanumeric coding system that is established and maintained by the American Medical Association (AMA), and consists of Category I, II, III, MAAA, and PLA CPT codes. Level II, which is established and maintained by CMS, is a standardized coding system that is used primarily to identify products, supplies, and services not included in the CPT codes. Together, Level I and II HCPCS codes are used to report procedures, services, items, and supplies under the OPPS payment system. Specifically, we recognize the following codes on OPPS claims:
- Category I CPT codes, which describe surgical procedures, diagnostic and therapeutic services, and vaccine codes;
- Category III CPT codes, which describe new and emerging technologies, services, and procedures;
- MAAA CPT codes, which describe laboratory multianalyte assays with algorithmic analyses (MAA);
- PLA CPT codes, which describe proprietary laboratory analyses (PLA) services; and
- Level II HCPCS codes (also known as alpha-numeric codes), which are used primarily to identify drugs, devices, supplies, temporary procedures, and services not described by CPT codes.
The codes are updated and changed throughout the year. CPT and Level II HCPCS code changes that affect the OPPS are published through the annual rulemaking cycle and through the OPPS quarterly update Change Requests (CRs). Generally, these code changes are effective January 1, April 1, July 1, or October 1. CPT code changes are released by the AMA (via their website) while Level II HCPCS code changes are released to the public via the CMS HCPCS website. CMS recognizes the release of new CPT and Level II HCPCS codes outside of the formal rulemaking process via OPPS quarterly update CRs. Based on our review, we assign the new codes to interim status indicators (SIs) and APCs. These interim assignments are finalized in the OPPS/ASC final rules. This quarterly process offers hospitals access to codes that more accurately describe the items or services furnished and provides payment for these items or services in a timelier manner than if we waited for the annual rulemaking process. We solicit public comments on the new CPT and Level II HCPCS codes, status indicators, and APC assignments through our annual rulemaking process.
We note that, under the OPPS, the APC assignment determines the payment rate for an item, procedure, or service. The items, procedures, or services not exclusively paid separately under the hospital OPPS are assigned to appropriate status indicators. Certain payment status indicators provide separate payment while other payment status indicators do not. In section XI (CY 2025 Payment Status and Comment Indicators) of this final rule with comment period, we discuss the various status indicators and comment indicators used under the OPPS. We also provide a complete list of the status indicators and their definitions in Addendum D1 to this final rule with comment period.
1. April 2024 HCPCS Codes Proposed Rule Comment Solicitation
For the April 2024 update, 73 new HCPCS codes were established and made effective on April 1, 2024. Through the April 2024 OPPS quarterly update CR (Transmittal 12552, Change Request 13568, dated March 21, 2024), we recognized several new HCPCS codes for payment under the OPPS. We solicited public comments on the proposed APC and status indicator assignments for the codes listed in Table 10 (New HCPCS Codes Effective April 1, 2024) of the CY 2025 OPPS/ASC proposed rule (89 FR 59239 through 59241, which are also displayed in Table 14.
We received some public comments on the proposed OPPS APC and SI assignments for the new Level II HCPCS codes that were effective on April 1, 2024. The comments and our responses are addressed in the applicable sections of this final rule with comment period, which include, but are not limited to: sections III.C. (New Technology APCs); III.E. (OPPS APC-Specific Policies); and IV. (OPPS Payment for Devices). For those April 2024 codes for which we received no comments, we are finalizing the proposed APC and status indicator assignments as proposed. We note that several of the HCPCS C-codes have been replaced with HCPCS J-codes, effective January 1, 2025. Their replacement codes are listed in Table 14. In addition, in prior years we included the final OPPS status indicators and APC assignments in the coding preamble tables, however, because the same information can be found in Addendum B, we are no longer including them in Table 14. Therefore, readers are advised to refer to the OPPS Addendum B for the final OPPS status indicators, APC assignments, and payment rates for all codes reportable under the hospital OPPS. These new codes that were effective April 1, 2024, were assigned to comment indicator “NP” in Addendum B to the CY 2025 OPPS/APC proposed rule to indicate that the codes are assigned to an interim APC assignment and comments would be accepted on their interim APC assignments. The complete list of status indicators and definitions used under the OPPS can be found in Addendum D1 to this final rule with comment period, while the complete list comment indicators and definitions can be found in Addendum D2 to this final rule with comment period. We note that OPPS Addendum B (OPPS payment file by HCPCS code), Addendum D1 (OPPS Status Indicators), and Addendum D2 (OPPS Comment Indicators) are available via the internet on the CMS website.
( printed page 93988) ( printed page 93989) ( printed page 93990) ( printed page 93991)2. July 2024 HCPCS Codes Proposed Rule Comment Solicitation
For the July 2024 update, 130 new codes were established and made effective July 1, 2024. Through the July 2024 OPPS quarterly update CR (Transmittal 12665, Change Request 13632, dated May 31, 2024), we recognized several new codes for payment and assigned them to appropriate interim OPPS status indicators and APCs. We solicited public comments on the proposed APC and status indicator assignments for the codes listed in Table 11 (New HCPCS Codes Effective July 1, 2024) of the CY 2025 OPPS/ASC proposed rule (89 FR 59242 through 59249), which are also listed in Table 15.
We received some public comments on the proposed OPPS APC and SI assignments for the new Level II HCPCS codes implemented on July 1, 2024. The comments and our responses are addressed in pertinent sections of this final rule with comment period, which include, but are not limited to: sections III.C (New Technology APCs); III.E (OPPS APC-Specific Policies); and IV (OPPS Payment for Devices). For those July 1, 2024, codes for which we received no comments, we are finalizing the proposed APC and status indicator assignments. Additionally, we note that in prior years we included the final OPPS status indicators and APC assignments in the coding preamble tables, however, because the same information can be found in Addendum B, we are no longer including them in Table 15. Therefore, readers are advised to refer to the OPPS Addendum B for the final OPPS status indicators, APC assignments, and payment rates for all codes reportable under the hospital OPPS. These new codes that were effective July 1, 2024, were assigned to comment indicator “NP” in Addendum B to the CY 2025 OPPS/ASC proposed rule to indicate that the codes are assigned to an interim APC assignment and comments would be accepted on their interim APC assignments. The complete list of status indicators and definitions used under the OPPS can be found in Addendum D1 to this final rule with comment period, while the complete list of comment indicators and definitions can be found in Addendum D2 to this final rule with comment period. We note that OPPS Addendum B (OPPS payment file by HCPCS code), Addendum D1 (OPPS Status Indicators), and Addendum D2 (OPPS Comment Indicators) are available via the internet on the CMS website.
( printed page 93992) ( printed page 93993) ( printed page 93994) ( printed page 93995) ( printed page 93996) ( printed page 93997) ( printed page 93998) ( printed page 93999) ( printed page 94000) ( printed page 94001)3. October 2024 HCPCS Codes Final Rule Comment Solicitation
For the October 2024 update, 107 codes were established and made effective October 1, 2024. Through the October 2024 OPPS quarterly update CR (Transmittal 12816, Change Request 13784, dated August 29, 2024), we recognized several new codes for separate payment and assigned them to appropriate interim OPPS status indicators and APCs. For CY 2025, consistent with our established policy, we proposed in the CY 2025 OPPS/ASC proposed rule (89 FR 59250) that the HCPCS codes that would be effective October 1, 2024, would be flagged with comment indicator “NI” in Addendum B to the CY 2025 OPPS/ASC final rule with comment period to indicate that we have assigned the codes to interim OPPS status indicators for CY 2025. Table 16 lists the codes that were effective October 1, 2024. We note that one of the temporary C-codes has been replaced with a permanent J-code effective January 1, 2025. We are inviting public comments in this final rule with comment period on the interim payment indicators, which will be finalized in the CY 2026 OPPS/ASC final rule with comment period. We note the proposed APC assignments and status indicators for these same codes will be subject to comment in the CY 2026 OPPS/ASC proposed rule with comment period, and will be finalized in the CY 2026 OPPS/ASC final rule with comment period.
( printed page 94002) ( printed page 94003) ( printed page 94004) ( printed page 94005) ( printed page 94006) ( printed page 94007) ( printed page 94008) ( printed page 94009)4. January 2025 HCPCS Codes
a. New Level II HCPCS Codes Final Rule Comment Solicitation
Consistent with past practice, we are soliciting comments on the new Level II HCPCS codes that will be effective January 1, 2025, in this CY 2025 OPPS/ASC final rule with comment period, thereby allowing us to finalize the status indicators and APC assignments for the codes in the CY 2026 OPPS/ASC final rule with comment period. Unlike the CPT codes that are effective January 1 and are included in the OPPS/ASC proposed rules, and except for the proposed new C-codes and G-codes listed in Addendum O of the CY 2025 OPPS/ASC proposed rule, most Level II HCPCS codes are not released until sometime around November to be effective January 1. Because these codes are not available until November, we are unable to include them in the OPPS/ASC proposed rules. Consequently, for CY 2025, we propose to include the new Level II HCPCS codes that will be effective January 1, 2025 (that would be incorporated in the January 2025 OPPS quarterly update CR), in Addendum B to the CY 2025 OPPS/ASC final rule with comment period. Specifically, for CY 2025, we propose to continue our established policy of assigning comment indicator “NI” in Addendum B to the OPPS/ASC final rule with comment period to the new HCPCS codes that will be effective January 1, 2025, to indicate that we are assigning them an interim status indicator, which is subject to public comment. We are inviting public comments in this final rule with comment period on the status indicators and APC assignments, which would then be finalized in the CY 2026 OPPS/ASC final rule with comment period. Similar to the codes effective October 1, 2024, the proposed APC assignments and status indicators for these new Level II HCPCS codes that will be effective January 1, 2025, will also be subject to comment in the CY 2026 OPPS/ASC proposed rule with comment period, and will be finalized in the CY 2026 OPPS/ASC final rule with comment period.
b. New CPT Codes Proposed Rule Comment Solicitation
In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66841 through 66844), we finalized a revised process of assigning APCs and status indicators for new and revised Category I and III CPT codes that would be effective January 1. Specifically, for the new/revised CPT codes that we receive in a timely manner from the AMA's CPT Editorial Panel, we finalized our proposal to include the codes that would be effective January 1 in the OPPS/ASC proposed rules, along with proposed APC and status indicator assignments for them, and to finalize the APC and status indicator assignments in the OPPS/ASC final rules beginning with the CY 2016 OPPS update. For those new/revised CPT codes that were received too late for inclusion in the OPPS/ASC proposed rule, we finalized our proposal to establish and use ( printed page 94010) HCPCS G-codes that mirror the predecessor CPT codes and retain the current APC and status indicator assignments for a year until we can propose APC and status indicator assignments in the following year's rulemaking cycle. We note that even if we find that we need to create HCPCS G-codes in place of certain CPT codes for the PFS proposed rule, we do not anticipate that these HCPCS G-codes will always be necessary for OPPS purposes. We will make every effort to include proposed APC and status indicator assignments for all new and revised CPT codes that the AMA makes publicly available in time for us to include them in the proposed rule, and to avoid resorting to use of HCPCS G-codes and the resulting delay in utilization of the most current CPT codes. Also, we finalized our proposal to make interim APC and status indicator assignments for CPT codes that are not available in time for the proposed rule and that describe wholly new services (such as new technologies or new surgical procedures), to solicit public comments in the final rule, and to finalize the specific APC and status indicator assignments for those codes in the following year's final rule.
For the CY 2025 OPPS update, we received the CPT codes that will be effective January 1, 2025, from the AMA in time to be included in the CY 2025 OPPS/ASC proposed rule. The new, revised, and deleted CPT codes were included in Addendum B to the proposed rule (which is available via the internet on the CMS website). We note that the new and revised CPT codes were assigned to comment indicator “NP” in Addendum B of the proposed rule to indicate that the code is new for the next calendar year or the code is an existing code with substantial revision to its code descriptor in the next calendar year as compared to the current calendar year with a proposed APC assignment, and that comments would be accepted on the proposed APC assignment and status indicator.
Further, we noted that the CPT code descriptors that appeared in Addendum B were short descriptors and did not accurately describe the complete procedure, service, or item described by the CPT code. Therefore, we included the 5-digit placeholder codes and the long descriptors for the new and revised CY 2025 CPT codes in Addendum O to the proposed rule (which is available via the internet on the CMS website) so that the public could adequately comment on the proposed APCs and SI assignments. The 5-digit placeholder codes were included in Addendum O to the proposed rule, specifically under the column labeled “CY 2025 OPPS/ASC Proposed Rule 5- Digit AMA Placeholder Code”. We noted that the final CPT code numbers would be included in this CY 2025 OPPS/ASC final rule with comment period. We also noted that not every code listed in Addendum O is subject to public comment. For the new and revised Category I and III CPT codes, we requested public comments on only those codes that are assigned comment indicator “NP.”
In summary, in the CY 2025 OPPS/ASC proposed rule, we solicited public comments on the proposed CY 2025 SI and APC assignments for the new and revised Category I and III CPT codes that would be effective January 1, 2025. The CPT codes were listed in Addendum B to the proposed rule with short descriptors only. We listed them again, with long descriptors, in Addendum O to the proposed rule. We also proposed to finalize the SI and APC assignments for these codes (with their final CPT code numbers) in the CY 2025 OPPS/ASC final rule with comment period. The proposed SI and APC assignments for these codes were included in Addendum B to the proposed rule (which is available via the internet on the CMS website). We received comments on several of the new CPT codes that were assigned to comment indicator “NP” in Addendum B to the CY 2025 OPPS/ASC proposed rule. We have responded to those public comments in sections III.C, III.E, and IV of this CY 2025 OPPS/ASC final rule with comment period.
The final SIs, APC assignments, and payment rates for the new CPT codes that are effective January 1, 2025, can be found in Addendum B to this final rule with comment period. In addition, the SI definitions can be found in Addendum D1 (OPPS Payment Status Indicators for CY 2024) to this final rule with comment period. Addenda B and D1 are available via the internet on the CMS website.
Finally, Table 17, which is a reprint of Table 12 from the CY 2025 OPPS/ASC proposed rule (89 FR 59251), shows the comment timeframe for new and revised HCPCS codes. The table provides information on our current process for updating codes through our OPPS quarterly update CRs, seeking public comments, and finalizing the treatment of these codes under the OPPS.
( printed page 94011)B. OPPS Changes—Variations Within APCs
1. Background
Section 1833(t)(2)(A) of the Act requires the Secretary to develop a classification system for covered hospital outpatient department services. In addition, section 1833(t)(2)(B) of the Act provides that the Secretary may establish groups of covered OPD services within this classification system, so that services classified within each group are comparable clinically and with respect to the use of resources. In accordance with these provisions, we developed a grouping classification system, referred to as Ambulatory Payment Classifications (APCs), as set forth in the regulation at 42 CFR 419.31. We use Level I (also known as CPT codes) and Level II HCPCS codes (also known as alphanumeric codes) to identify and group the services within each APC. The APCs are organized such that each group is homogeneous both clinically and in terms of resource use. Using this classification system, we have established distinct groups of similar services. We also have developed separate APC groups for certain medical devices, drugs, biologicals, therapeutic radiopharmaceuticals, and brachytherapy devices that are not packaged into the payment for the procedure.
We have packaged into the payment for each procedure or service within an APC group, the costs associated with those items and services that are typically ancillary and supportive to a primary diagnostic or therapeutic modality and, in those cases, are an integral part of the primary service they support. Therefore, we do not make separate payment for these packaged items or services. In general, packaged items and services include, but are not limited to, the items and services listed in the regulation at 42 CFR 419.2(b). A further discussion of packaged services is included in section II.A.3 of this final rule with comment period.
Under the OPPS, we generally pay for covered hospital outpatient services on a rate-per-service basis, where the service may be reported with one or more HCPCS codes. Payment varies according to the APC group to which the independent service or combination of services is assigned. For CY 2025, we proposed that each APC relative payment weight represents the hospital cost of the services included in that APC, relative to the hospital cost of the services included in APC 5012 (Clinic Visits and Related Services). The APC relative payment weights are scaled to APC 5012 because it is the hospital clinic visit APC and clinic visits are among the most frequently furnished services in the hospital outpatient setting.
2. Application of the 2 Times Rule
Section 1833(t)(9)(A) of the Act requires the Secretary to review, not less ( printed page 94012) often than annually, and revise the APC groups, the relative payment weights, and the wage and other adjustments described in paragraph (2) to consider changes in medical practice, changes in technology, the addition of new services, new cost data, and other relevant information and factors. Section 1833(t)(9)(A) of the Act also requires the Secretary to consult with an expert outside advisory panel composed of an appropriate selection of representatives of providers to review (and advise the Secretary concerning) the clinical integrity of the APC groups and the relative payment weights. We note that the Advisory Panel on Hospital Outpatient Payment (also known as the HOP Panel or the Panel) recommendations for specific services for the CY 2025 OPPS update will be discussed in the relevant specific sections throughout this CY 2025 OPPS/ASC final rule with comment period.
In addition, section 1833(t)(2) of the Act provides that, subject to certain exceptions, the items and services within an APC group cannot be considered comparable regarding the use of resources if the highest cost for an item or service in the group is more than 2 times greater than the lowest cost for an item or service within the same group (referred to as the “2 times rule”). The statute authorizes the Secretary to make exceptions to the 2 times rule in unusual cases, such as for low-volume items and services (but the Secretary may not make such an exception in the case of a drug or biological that has been designated as an orphan drug under section 526 of the Federal Food, Drug, and Cosmetic Act). In determining the APCs with a 2 times rule violation, we consider only those HCPCS codes that are significant based on the number of claims. We note that, for purposes of identifying significant procedure codes for examination under the 2 times rule, we consider procedure codes that have more than 1,000 single major claims or procedure codes that both have more than 99 single major claims and contribute at least 2 percent of the single major claims used to establish the APC cost to be significant (75 FR 71832). This longstanding definition of when a procedure code is significant for purposes of the 2 times rule was selected because we believe that a subset of 1,000 or fewer claims is negligible within the set of approximately 100 million single procedure or single session claims we use for establishing costs. Similarly, a procedure code for which there are fewer than 99 single claims and that comprises less than 2 percent of the single major claims within an APC will have a negligible impact on the APC cost (75 FR 71832). In the CY 2025 OPPS/ASC proposed rule, for CY 2025, we proposed to make exceptions to this limit on the variation of costs within each APC group in unusual cases, such as for certain low-volume items and services.
For the CY 2025 OPPS update, we identified the APCs with violations of the 2 times rule, and we proposed changes to the procedure codes assigned to these APCs (with the exception of those APCs for which we proposed a 2 times rule exception) in Addendum B to CY 2025 OPPS/ASC proposed rule. We note that Addendum B does not appear in the printed version of the Federal Register as part of this final rule with comment period. Rather, it is published and made available via the internet on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
To eliminate a violation of the 2 times rule and improve clinical and resource homogeneity in the APCs for which we did not propose a 2 times rule exception, we proposed to reassign these procedure codes to new APCs that contain services that are similar with regard to both their clinical and resource characteristics. In many cases, the proposed procedure code reassignments and associated APC reconfigurations for CY 2025 included in the CY 2025 OPPS/ASC proposed rule are related to changes in costs of services that were observed in the CY 2023 claims data available for CY 2025 ratesetting. Addendum B to the CY 2025 OPPS/ASC proposed rule identifies with a comment indicator “CH” those procedure codes for which we proposed a change to the APC assignment or status indicator, or both, that were initially assigned in the July 1, 2024, OPPS Addendum B Update, which is available via the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/addendum-a-b-updates.
3. APC Exceptions to the 2 Times Rule
While considering the APC changes that we proposed for CY 2025, we reviewed all of the APCs for which we identified 2 times rule violations to determine whether any of the APCs would qualify for an exception. We used the following criteria to evaluate whether to propose exceptions to the 2 times rule for affected APCs:
- Resource homogeneity;
- Clinical homogeneity;
- Hospital outpatient setting utilization;
- Frequency of service (volume); and
- Opportunity for upcoding and code fragments.
For a detailed discussion of these criteria, we refer readers to the April 7, 2000 final rule (65 FR 18457 and 18458).
Based on the CY 2023 claims data available for the CY 2025 OPPS/ASC proposed rule, we found 23 APCs with violations of the 2 times rule. We applied the criteria as described above to identify the APCs for which we proposed to make exceptions under the 2 times rule for CY 2025 and found that all of the 23 APCs we identified met the criteria for an exception to the 2 times rule based on the CY 2023 claims data available for the CY 2025 OPPS/APC proposed rule. We note that, on an annual basis, based on our analysis of the latest claims data, we identify violations to the 2 times rule and propose changes when appropriate. Those APCs that violate the 2 times rule are identified and appear in Table 18. In addition, we did not include in that determination those APCs where a 2 times rule violation was not a relevant concept, such as APC 5401 (Dialysis), which only has two HCPCS codes assigned to it that have similar geometric mean costs and do not create a 2 times rule violation. Therefore, we have only identified those APCs, including those with criteria-based costs, such as device-dependent CPT/HCPCS codes, with violations of the 2 times rule, where a 2 times rule violation is a relevant concept.
Table 13 of the CY 2025 OPPS/ASC proposed rule (89 FR 59253) listed the 23 APCs for which we proposed to make an exception under the 2 times rule for CY 2025 based on the criteria cited above and claims data submitted between January 1, 2023, and December 31, 2023, and CCRs, if available. The proposed geometric mean costs for covered hospital outpatient services for these and all other APCs that were used in the development of the proposed rule can be found on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
Based on the updated final rule CY 2023 claims data used for this CY 2025 final rule with comment period, we found a total of 28 APCs with violations of the 2 times rule. Of these 28 total APCs, 23 were identified in the proposed rule and five are newly identified in this final rule with comment period. The following two APCs appeared in Table 13 of the CY 2025 OPPS/ASC proposed rule (89 FR ( printed page 94013) 59253) as violating the 2 times rule, however, after conducting our data analysis for this final rule with comment period, we found that the APCs no longer violate the 2 times rule:
- APC 5303 (Level 3 Upper GI Procedures)
- APC 5822 (Health and Behavior Services)
- In addition, the following five APCs are newly identified with 2 times rule violations using updated data for this final rule with comment period: APC 5024 (Level 4 Type A ED Visits)
- APC 5501 (Level 1 Extraocular, Repair, and Plastic Eye Procedures)
- APC 5613 (Level 3 Therapeutic Radiation Treatment Preparation)
- APC 5674 (Level 4 Pathology)
- APC 5722 (Level 2 Diagnostic Tests and Related Services)
Although we did not receive any comments on Table 13 of the CY OPPS/ASC proposed rule, we did receive comments on APC assignments for specific HCPCS codes. The comments, and our responses, can be found in section III.E. of this final rule with comment period.
( printed page 94014) ( printed page 94015)We did not receive any public comments on our proposal. Based on our analysis of the CY 2023 costs from hospital claims and cost report data available for this CY 2025 final rule with comment period, we are finalizing our proposals with some modifications. Specifically, we are finalizing our proposal to except the 23 proposed APCs that continue to have 2 times violations in the final rule data from the 2 times rule for CY 2023 claims data and also excepting five additional APCs (APCs 5024, 5501, 5613, 5674, 5722) that did not violate the 2 times rule in the proposed rule data, but do violate the 2 times rule in the final rule data, for a total of 28 APCs for which we identified 2 times rule violations but that qualify for exceptions.
In summary, Table 18 lists the 28 APCs that we are excepting from the 2 times rule for CY 2025 based on the criteria described earlier and a review of updated claims data for dates of service between January 1, 2023, and December 31, 2023, that were processed on or before June 30, 2024, and updated CCRs, if available. We note that, for cases in which a recommendation by the HOP Panel appears to result in or allow a violation of the 2 times rule, we generally accept the HOP Panel's recommendation because those recommendations are based on explicit consideration of resource use, clinical homogeneity, site of service, and the quality of the claims data used to determine the APC payment rates. The geometric mean costs for hospital outpatient services for these and all other APCs that were used in the development of this final rule with comment period can be found on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
C. New Technology APCs
1. Background
In the CY 2002 OPPS final rule (66 FR 59903), we finalized changes to the time period in which a service can be eligible for payment under a New Technology APC. Beginning in CY 2002, we retain services within New Technology APC groups until we gather sufficient claims data to enable us to assign the service to an appropriate clinical APC. This policy allows us to move a service from a New Technology APC in less than 2 years if sufficient data are available. It also allows us to retain a service in a New Technology APC for more than 2 years if sufficient data upon which to base a decision for reassignment have not been collected.
We also adopted in the CY 2002 OPPS final rule the following criteria for assigning a complete or comprehensive service to a New Technology APC: (1) the service must be truly new, meaning it cannot be appropriately reported by an existing HCPCS code assigned to a clinical APC and does not appropriately fit within an existing clinical APC; (2) the service is not eligible for transitional pass-through payment (however, a truly new, comprehensive service could qualify for assignment to a new technology APC even if it involves a device or drug that could, on its own, qualify for pass-through payment); and (3) the service falls within the scope of Medicare benefits under section 1832(a) of the Act and is reasonable and necessary in accordance with section 1862(a)(1)(A) of the Act (66 FR 59898 through 59903). For additional information about our New Technology APC policy, we refer readers to https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/pass-through-payment-status-new-technology-ambulatory-payment-classification-apc on the CMS website and then follow the instructions to access the MEARIS[TM] system for OPPS New Technology APC applications.[17]
In the CY 2004 OPPS final rule with comment period (68 FR 63416), we restructured the New Technology APCs to make the cost intervals more consistent across payment levels and refined the cost bands for these APCs to retain two parallel sets of New Technology APCs: one set with a status indicator of “S” (Significant Procedures, Not Discounted when Multiple. Paid under OPPS; separate APC payment) and the other set with a status indicator of “T” (Significant Procedure, Multiple Reduction Applies. Paid under OPPS; separate APC payment). These current New Technology APC configurations allow us to price new technology services more appropriately and consistently.
For CY 2024, there were 52 New Technology APC levels, ranging from the lowest cost band assigned to APC 1491 (New Technology—Level 1A ($0-$10)) to the highest cost band assigned to APC 1908 (New Technology—Level 52 ($145,001-$160,000)). We note that the cost bands for the New Technology APCs, specifically, APCs 1491 through 1599 and 1901 through 1908, vary with increments ranging from $10 to $14,999. These cost bands identify the APCs to which new technology procedures and services with estimated service costs that fall within those cost bands are assigned under the OPPS. Payment for each APC is made at the mid-point of the APC's assigned cost band. For example, payment for New Technology APC 1507 (New Technology—Level 7 ($501-$600)) is made at $550.50.
Under the OPPS, one of our goals is to make payments that are appropriate for the services that are necessary for the treatment of Medicare beneficiaries. The OPPS, like other Medicare payment systems, is budget neutral and increases are limited to the annual hospital market basket increase reduced by the productivity adjustment. We believe that our payment rates reflect the costs that are associated with providing care to Medicare beneficiaries and are adequate to ensure access to services (80 FR 70374). For many emerging technologies, there is a transitional period during which utilization may be low, often because providers are first learning about the technologies and their clinical utility. Quite often, parties request that Medicare make higher payments under the New Technology APCs for new procedures in that transitional phase. These requests, and their accompanying estimates for expected total patient utilization, often reflect very low rates of patient use of expensive equipment, resulting in high per-use costs for which requesters believe Medicare should make full payment. Medicare does not, and we believe should not, assume responsibility for more than its share of the costs of procedures based on projected utilization for Medicare beneficiaries and does not set its payment rates based on initial projections of low utilization for services that require expensive capital equipment. For the OPPS, we rely on hospitals to make informed business decisions regarding the acquisition of high-cost capital equipment, taking into consideration their knowledge about their entire patient base (Medicare beneficiaries included) and an understanding of Medicare's and other payers' payment policies. We refer readers to the CY 2013 OPPS/ASC final rule with comment period (77 FR 68314) for further discussion regarding this payment policy.
Some services assigned to New Technology APCs have low annual volume, which we consider to be fewer than 100 claims in the year of claims data used for ratesetting (86 FR 63528). Where utilization of services assigned to a New Technology APC is low, it can lead to wide variation in payment rates from year to year, resulting in even lower utilization and potential barriers to access to new technologies, which ( printed page 94016) ultimately limits our ability to assign the service to the appropriate clinical APC. To mitigate these issues, we finalized a policy in the CY 2019 OPPS/ASC final rule with comment period to utilize our equitable adjustment authority at section 1833(t)(2)(E) of the Act to adjust how we determine the costs for low-volume services assigned to New Technology APCs (83 FR 58892 through 58893). Specifically, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58893), we established that, in each of our annual rulemakings, we would calculate and present the result of each statistical methodology (arithmetic mean, geometric mean, and median) based on up to 4 years of claims data and solicit public comment on which methodology should be used to establish the payment rate for the low-volume new technology service. In the CY 2022 OPPS/ASC final rule (86 FR 63529), we replaced the New Technology APC low volume policy with the universal low volume APC policy. Unlike the New Technology APC low volume policy, the universal low volume APC policy applies to clinical APCs and brachytherapy APCs, in addition to procedures assigned to New Technology APCs, and uses the highest of the geometric mean, arithmetic mean, or median based on up to 4 years of claims data to set the payment rate for the APC. We refer readers to the CY 2022 OPPS/ASC final rule with comment period (86 FR 63529) for further discussion regarding this policy.
Finally, we note that, in a budget-neutral system, payments may not fully cover hospitals' costs in a particular circumstance, including those for the purchase and maintenance of capital equipment. We rely on hospitals to make their decisions regarding the acquisition of high-cost equipment with the understanding that the Medicare program must be careful to establish its initial payment rates, including those made through New Technology APCs, for new services that lack hospital claims data based on realistic utilization projections for all such services delivered in cost-efficient hospital outpatient settings. As the OPPS acquires claims data regarding hospital costs associated with new procedures, we regularly examine the claims data and any available new information regarding the clinical aspects of new procedures to confirm that our OPPS payments remain appropriate for procedures as they transition into mainstream medical practice (77 FR 68314). For CY 2025, we included the proposed payment rates for New Technology APCs 1491 to 1599 and 1901 through 1908 in Addendum A to the proposed rule (which is available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
Comment: We received a comment requesting that CMS revise the New Technology APC criteria to accommodate for forms of algorithm-based healthcare services (ABHS). The commenter explained that certain ABHS, that provide new clinical outputs that can impact the diagnosis or treatment of a patient's disease, and represent additional costs to hospitals not currently accounted for in the provision of an underlying service, would most likely not meet the New Technology APC criteria because the service provided by the technology is occurring at the same time as the performance of an underlying service. The commenter also requested that the New Technology APC process allow for the creation of new procedural C-codes when needed.
Response: We thank the commenter for their comment. As part of the existing New Technology APC process, CMS creates C-codes when we find that there is no existing code that can appropriately describe a service. CMS will assign a service to a New Technology APC when certain criteria are met, including the criterion that the service is truly new, meaning that it cannot be appropriately reported by an existing HCPCS code assigned to a clinical APC and does not appropriately fit within an existing clinical APC. In the past, we have found that several SaaS procedures, including those that are algorithm-based, meet the existing New Technology APC criteria. For CY 2025, we proposed to assign several SaaS procedures that meet our criteria to New Technology APCs, many of which have been assigned to New Technology APCs for several years. However, we recognize that software-based technologies are rapidly evolving and, therefore, we are considering for future rulemaking whether specific adjustments to payment policies, including our New Technology APC policies, are needed to more accurately and appropriately pay for these products and services across settings of care.
For CY 2025, we did not propose to make any changes to the existing New Technology APC criteria. After consideration of the public comments we received, we are not finalizing any changes to the New Technology APC criterion at this time.
2. Policy To Exempt Services With Very Low Claims Volume From APC Reassignment Based on the Universal Low Volume Policy
We continue to be concerned about payment stability for services assigned to New Technology APCs, specifically services with very low claims volume of fewer than 10 claims in the 4-year lookback period used under the universal low volume APC policy. Historically, we have used our equitable adjustment authority at section 1833(t)(2)(E) of the Act to exempt a number of services with very low claims volume from the universal low volume APC policy in instances where application of the universal low volume policy would lead to significant fluctuations in payment. Given the frequency with which we have needed to utilize our equitable adjustment authority to address significant fluctuations in payment for very low volume services, we noted in the proposed rule that we believed that refinements to our universal low volume policy for services assigned to New Technology APCs may be necessary. We also recognized that determining initial cost estimates for these services may be particularly challenging, given the lack of cost information for new and innovative technologies.
To allow time for us to consider these issues, we proposed for CY 2025 to exempt services assigned to New Technology APCs with fewer than 10 claims over the 4-year lookback period used for the universal low volume policy. Instead of assigning these services to a different New Technology APC based on the very few claims available, we proposed that we would maintain the New Technology APC assignment for each service from the prior year, which in this case would be the New Technology APC assignment for CY 2024. We explained that we believed it was appropriate to apply this policy to New Technology APCs because services assigned to New Technology APCs represent new technologies for which it may be more challenging to determine an appropriate cost than for other, more established services. We believed 10 claims was an appropriate ceiling for exempting services from reassignment based on the universal low volume policy because we believed that at 10 claims a rough standard distribution begins to appear. We also believed that services with so few claims over the 4-year lookback period would be especially vulnerable to large changes in payment rates year-to-year as a result of one or two new ( printed page 94017) claims being available or one or two claims from what was previously the fourth year of the lookback period no longer being included in that period.
Consistent with our overall policy regarding use of updated claims data in the final rule, we proposed to perform a similar analysis for the final rule using updated claims data, including determining whether specific HCPCS codes continue to meet the criteria for our universal low volume APC policy or our proposal to exempt services with fewer than 10 claims in the 4-year lookback period from the universal low volume APC policy and maintain their CY 2024 New Technology APC assignment. We noted we would update the APC placement as needed in the final rule.
Comment: Commenters agreed with our concerns that the application of the universal low volume APC policy may result in payment instability for certain services assigned to New Technology APCs, but offered different perspectives on how to address the issue. One commenter, representing artificial intelligence companies, supported our proposal to exempt certain services from the universal low volume APC policy and recommended that we utilize 25 claims, rather than 10 claims when determining whether to maintain a service's New Technology APC assignment from the previous year. Another commenter, representing manufacturers of medical imaging equipment, did not believe that the proposal sufficiently addressed the policy concern of payment stability for services assigned to New Technology APCs. This commenter also believed that the proposal would create a disincentive for hospitals to furnish the service and, therefore, discourage claims utilization where it would be more prudent to have no claims than to risk being assigned to a lower paying New Technology APC in future years by exceeding the 10-claim threshold.
Response: We agree with the commenters' concerns that applying the universal low volume APC policy can result in payment fluctuations from year-to-year. We also recognize that major payment fluctuations that do not reflect the actual cost of the service can have the effect of discouraging usage and keeping claims volume low, which prevents CMS from gathering sufficient claims data upon which to transition the service from a New Technology APC to an appropriate clinical APC. We disagree with one commenter's assertion that our proposal to maintain the New Technology APC placement of a service with extremely low claims volume would disincentivize usage of the service. By maintaining the APC placement for services with fewer than 10 claims in the four-year lookback period, we are removing the potential for a major payment change based on a handful of claims over multiple years, that may not reflect the actual cost of the service. We believe that this policy is an incremental step towards more stable and predictable payments while providers learn about these new technology services, which should have the effect of encouraging utilization in the long-term. We also disagree with the commenter's suggestion to increase the claims threshold from 10 to 25 claims. We proposed to exempt less than 10 claims from the universal low volume APC policy because at 10 claims a rough standard distribution begins to appear, and services with an extremely low number of claims are most vulnerable to significant payment fluctuations.
Our proposal is meant to serve as a first step to addressing the issue of payment stability for services assigned to New Technology APCs. As we stated in the proposed rule, we continue to believe that refinements to the universal low volume APC policy with respect to its application to services in New Technology APCs may be necessary. We are finalizing our policy as proposed for CY 2025 and will consider additional changes in future years.
Comment: One commenter expressed support for the universal low volume APC policy.
Response: We thank the commenter for their input.
After consideration of the public comments we received, we are finalizing our proposal to exempt services assigned to New Technology APCs with fewer than 10 claims in the four-year lookback period from the universal low volume APC policy and maintain their CY 2024 New Technology APC assignment as proposed.
3. Procedures Assigned to New Technology APCs for CY 2025
As we described in the CY 2002 OPPS final rule (66 FR 59902), we generally retain a procedure in the New Technology APC to which it is initially assigned until we have obtained sufficient claims data to justify reassignment of the procedure to a clinically appropriate APC. In addition, in cases where we find that our initial New Technology APC assignment was based on inaccurate or inadequate information (although it was the best information available at the time), where we obtain new information that was not available at the time of our initial New Technology APC assignment, or where the New Technology APCs are restructured, we may, based on more recent resource utilization information (including claims data) or the availability of refined New Technology APC cost bands, reassign the procedure or service to a different New Technology APC that more appropriately reflects its cost (66 FR 59903).
Consistent with our current policy, for CY 2025, we proposed to retain services within New Technology APC groups until we obtain sufficient claims data to justify reassignment of the service to an appropriate clinical APC. The flexibility associated with this policy allows us to reassign a service from a New Technology APC in less than 2 years if we have obtained sufficient claims data. It also allows us to retain a service in a New Technology APC for more than 2 years if we have not obtained sufficient claims data upon which to base a reassignment decision (66 FR 59902).
a. Administration of Subretinal Therapies Requiring Vitrectomy (APC 1563)
Effective January 1, 2021, CMS established HCPCS code C9770 (Vitrectomy, mechanical, pars plana approach, with subretinal injection of pharmacologic/biologic agent) and assigned it to a New Technology APC based on the geometric mean cost of CPT code 67036 (Vitrectomy, mechanical, pars plana approach) due to similar resource utilization. For CY 2021, HCPCS code C9770 was assigned to APC 1561 (New Technology—Level 24 ($3001-$3500)). This code may be used to describe the administration of HCPCS code J3398 (Injection, voretigene neparvovec-rzyl, 1 billion vector genomes). This procedure was previously discussed in depth in the CY 2021 OPPS/ASC final rule with comment period (85 FR 85939 through 85940). For CY 2022, we maintained the APC assignment of APC 1561 (New Technology—Level 24 ($3001-$3500)) for HCPCS code C9770 (86 FR 63531 through 63532).
( printed page 94018)HCPCS code J3398 (Injection, voretigene neparvovec-rzyl, 1 billion vector genomes) is for a gene therapy product indicated for a rare mutation-associated retinal dystrophy. Voretigene neparvovec-rzyl (Luxturna®) was approved by FDA in December of 2017 and is an adeno-associated virus vector-based gene therapy indicated for the treatment of patients with confirmed biallelic RPE65 mutation-associated retinal dystrophy.18 This therapy is administered through a subretinal injection, which interested parties describe as an extremely delicate and sensitive surgical procedure. The FDA-approved package insert describes one of the steps for administering Luxturna as, “after completing a vitrectomy, identify the intended site of administration. The subretinal injection can be introduced via pars plana.”
Interested parties, including the manufacturer of Luxturna®, recommended CPT code 67036 (Vitrectomy, mechanical, pars plana approach) for the administration of the gene therapy.[19] However, the manufacturer previously contended the administration was not accurately described by any existing codes as CPT code 67036 (Vitrectomy, mechanical, pars plana approach) does not account for the administration itself.
CMS recognized the need to accurately describe the unique procedure that is required to administer the therapy described by HCPCS code J3398. Therefore, in the CY 2021 OPPS/ASC proposed rule (85 FR 48832), we proposed to establish a new HCPCS code, C97X1 (Vitrectomy, mechanical, pars plana approach, with subretinal injection of pharmacologic/biologic agent) to describe this process. We stated that we believed this new HCPCS code accurately described the unique service associated with intraocular administration of HCPCS code J3398. We recognized that CPT code 67036 represents a clinically similar procedure and process that approximates similar resource utilization to C97X1. However, we also recognized that it is not prudent for the code that describes the administration of this unique gene therapy, C97X1, to be assigned to the same C-APC to which CPT code 67036 is assigned, as this would package the primary therapy, HCPCS code J3398, into the code that represents the process to administer the gene therapy.
Therefore, for CY 2021, we proposed to assign the services described by C97X1 to a New Technology APC with a cost band that contains the geometric mean cost for CPT code 67036. The placeholder code C97X1 was replaced by HCPCS code C9770. For CY 2021, we finalized our proposal to create HCPCS code C9770 (Vitrectomy, mechanical, pars plana approach, with subretinal injection of pharmacologic/biologic agent), and we assigned this code to APC 1561 (New Technology—Level 24 ($3001-$3500)) using the geometric mean cost of CPT code 67036. For CY 2022, we continued to assign HCPCS code C9770 to APC 1561 (New Technology—Level 24 ($3001-$3500)) using the geometric mean cost of CPT code 67036.
CY 2023 was the first year that claims data were available for HCPCS code C9770; therefore, we proposed and finalized a policy to base the payment rate of HCPCS code C9770 on claims data for that code rather than on the geometric mean cost of CPT code 67036. Given the low number of claims for this procedure, we designated HCPCS code C9770 as a low volume procedure under our universal low volume APC policy and used the greater of the geometric mean, arithmetic mean, or median cost calculated based on the available claims data to calculate an appropriate payment rate for purposes of assigning HCPCS code C9770 to a New Technology APC.
Based on the claims data available for the CY 2023 OPPS/ASC final rule with comment period, we found the median was the statistical methodology that estimated the highest cost for the service. The payment rate calculated using this methodology fell within the cost band for New Technology APC 1562 (New Technology—Level 25 ($3501-$4000)). Therefore, we finalized our proposal to assign HCPCS code C9770 to APC 1562 for CY 2023.
For CY 2024, we proposed and finalized that we would delete HCPCS code C9770 effective December 31, 2023, and recognize CPT code 0810T (Subretinal injection of a pharmacologic agent, including vitrectomy and 1 or more retinotomies) starting January 1, 2024 (88 FR 81617 through 81619). We determined the payment rate for CPT code 0810T using the claims data for HCPCS code C9770 and designated CPT code 0810T as a low volume procedure under our universal low volume APC policy and used the greater of the geometric mean, arithmetic mean, or median cost calculated based on the available claims data for HCPCS code C9770 to calculate an appropriate payment rate for purposes of assigning CPT code 0810T to a New Technology APC. For CY 2024, we finalized assignment of CPT code 0810T to APC 1563 (New Technology—Level 26 ($4001-$4500)) (88 FR 81617 through 81619).
Since CMS recognized CPT code 0810T starting January 1, 2024, we do not have claims data for CPT code 0810T available for CY 2025 rulemaking. However, as HCPCS code C9770 was still in use until December 31, 2023, we proposed to determine the payment rate for CPT code 0810T using the claims data for HCPCS code C9770. This is similar to the policy we finalized for CY 2024. For CY 2025, we proposed to designate CPT code 0810T as a low volume procedure under our universal low volume APC policy, given that there were only 34 claims available for HCPCS code C9770 and none for CPT code 0810T. This is below the threshold of 100 claims for a service within a year required to designate a service as a low volume service and apply our universal low volume APC policy. Therefore, we proposed to use the greater of the geometric mean, arithmetic mean, or median cost calculated based on the available claims data for HCPCS code C9770 to calculate an appropriate payment rate for purposes of assigning CPT code 0810T to a New Technology APC.
Using all available claims from the 4-year lookback period, which was 34 claims, we determined the geometric mean cost to be $3,934, the arithmetic mean cost to be $4,173, and the median cost to be $4,103. Because the arithmetic mean is the statistical methodology that estimated the highest cost for the service, we proposed to use this cost to determine the New Technology APC placement. The arithmetic mean of $4,173 falls within the cost band for New Technology APC 1563 (New Technology—Level 26 ($4001-$4500)). Therefore, we proposed to assign CPT code 0810T to APC 1563 for CY 2025. Additionally, we proposed to perform a similar analysis using updated claims data in this final rule with comment period, including determining if CPT code 0810T continues to meet the criteria for our universal low volume APC policy, and update the APC assignment as needed.
Please refer to Table 19 below for the final CY 2024 and proposed CY 2025 OPPS New Technology APC and status indicator assignments for CPT code 0810T. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
( printed page 94019)We did not receive any public comments on our proposal to continue to assign HCPCS code 0810T to APC 1563 for CY 2025.
Additional claims for CY 2023 have been processed since the proposed rule. Our analysis of the updated claims data found that the greater of the geometric mean, arithmetic mean, or median cost calculated for HCPCS code C9770 is approximately $4,240. This continues to fall into the cost band of New Technology APC 1563. Therefore, we are finalizing our proposal without modification to continue to assign HCPCS code 0810T to APC 1563. Please refer to Table 20 for the final OPPS New Technology APC and status indicator assignment for HCPCS codes 0810T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
b. BgRT (APCs 1521 and 1525)
Biology Guided Radiation Therapy (BgRT) uses positron-emitting radiopharmaceuticals to control delivery of radiation therapy to treat primary and metastatic lung or bone tumors. During radiation treatment delivery, the same system applies these firing filters to the real-time positron emission tomography (PET) data collected by the radiation treatment delivery machine. Effective January 1, 2024, CMS created HCPCS codes C9794 (Therapeutic radiology simulation-aided field setting; complex, including acquisition of PET and CT imaging data required for radiopharmaceutical-directed radiation therapy treatment planning ( i.e., modeling) and C9795 (Stereotactic body radiation therapy, treatment delivery, per fraction to 1 or more lesions, including image guidance and real-time positron emissions-based delivery adjustments to 1 or more lesions, entire course not to exceed 5 fractions) to describe the modeling and treatment delivery portions of the BgRT service. We assigned HCPCS code C9794 to APC 1521 (New Technology—Level 21 ($1901-$2000)) and HCPCS code C9795 to APC 1525 (New Technology—Level 25 ($3501-$4000)) for CY 2024.
For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data. As HCPCS codes C9794 and C9795 were effective January 1, 2024, we do not have any claims data for the service. Therefore, for CY 2025, we proposed to continue to assign HCPCS code C9794 to APC 1521 (New Technology—Level 21 ($1901-$2000)) with a payment rate of $1,950.50 and HCPCS code C9795 to APC 1525 (New Technology—Level 25 ($3501-$4000)) with a payment rate of $3,750.50. Please refer to Table 21 below for the proposed ( printed page 94020) OPPS New Technology APC and status indicator assignment for HCPCS codes C9794 and C9795 for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
Comment: We received several comments supporting our proposal to maintain the APC assignments for HCPCS code C9794 and C9795 until claims data are received.
Response: We thank the commenters for their support.
After consideration of the public comments we received, we are finalizing our proposal without modification. Specifically, for CY 2025 we are finalizing our proposal to assign C9794 and C9795 to APCs 1521 (New Technology—Level 21 ($1901-$2000)) and 1525 (New Technology—Level 25 ($3501-$4000)), respectively.
We note that, effective January 1, 2025, two new G-codes are replacing HCPCS codes C9794 and C9795. Specifically, effective January 1, 2025, HCPCS codes C9794 and C9795 are being deleted and replaced by G0562 and G0563, respectively, to allow for payment in settings other than hospital outpatient departments for CY 2025. The descriptors for the new G-codes are the same as existing HCPCS codes C9794 and C9795.
Please refer to Table 22 for the final OPPS New Technology APC and status indicator assignments for HCPCS codes G0562 and G0563 for CY 2025. The CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94021)c. Blinded Procedure for NYHA Class III/IV Heart Failure (APC 1590)
A randomized, double-blinded, controlled IDE study is currently in progress for the V-Wave interatrial shunt. The V-Wave interatrial shunt is for patients with severe symptomatic heart failure and is designed to regulate left atrial pressure in the heart. All participants who passed initial screening for the study receive a right heart catheterization procedure described by CPT code 93451 (Right heart catheterization including measurement(s) of oxygen saturation and cardiac output, when performed). Participants assigned to the experimental group also receive the V-Wave interatrial shunt procedure while participants assigned to the control group only receive right heart catheterization. The developer of V-Wave was concerned that the current coding of these services by Medicare would reveal to the study participants whether they had received the interatrial shunt because an additional procedure code, CPT code 93799 (Unlisted cardiovascular service or procedure), would be included on the claims for participants receiving the interatrial shunt. Therefore, for CY 2020, we created a temporary HCPCS code to describe the V-Wave interatrial shunt procedure for both the experimental group and the control group in the study. Specifically, we established HCPCS code C9758 (Blinded procedure for NYHA class III/IV heart failure; transcatheter implantation of interatrial shunt or placebo control, including right heart catheterization, trans-esophageal echocardiography (TEE)/intracardiac echocardiography (ICE), and all imaging with or without guidance (for example, ultrasound, fluoroscopy), performed in an approved investigational device exemption (IDE) study) to describe the service, and we assigned the service to New Technology APC 1589 (New Technology—Level 38 ($10,001-$15,000)) with a payment rate of $12,500.50.
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 85946), we stated that we believe similar resources and device costs are involved with the V-Wave interatrial shunt procedure and the Corvia Medical interatrial shunt procedure (HCPCS code C9760), except that payment for HCPCS codes C9758 and C9760 differs based on how often the interatrial shunt is implanted when each code is billed. An interatrial shunt is implanted one-half of the time HCPCS code C9758 is billed, whereas an interatrial shunt is implanted every time HCPCS code C9760 is billed. Accordingly, for CY 2021, we reassigned HCPCS code C9758 to New Technology APC 1590 (New Technology—Level 39 ($15,001-$20,000)), which reflects the cost of furnishing the interatrial shunt one-half of the time the procedure is performed.
For CY 2022, we used the same claims data from CY 2019 that we did for the CY 2021 OPPS/ASC final rule with comment period. Because there were no claims reporting HCPCS code C9758, we continued to assign HCPCS code C9758 to New Technology APC 1590 with a payment rate of $17,500.50 for CY 2022. For CY 2023 we used claims data from CY 2019 through CY 2022. Because there were no claims reporting HCPCS code C9758 in CY 2023 or CY 2024, we continued to assign HCPCS code C9758 to New Technology APC 1590 with a payment rate of $17,500.50 for CY 2023 and 2024.
For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data. There were only three claims for HCPCS code C9758 within this time period. As this is below the threshold of 100 claims for a service within a year, we would designate C9758 as a low volume service and apply our universal low volume APC policy. Under this policy, we would use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign HCPCS code C9758 to the appropriate New Technology APC. Given our proposal, which we are finalizing, to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, we proposed to continue assigning HCPCS code C9758 to New Technology APC 1590 with a proposed payment rate of $17,500.50.
Please refer to Table 23 for the proposed OPPS New Technology APC and status indicator assignments for HCPCS code C9758 for CY 2025.
( printed page 94022)Comment: We received one comment supporting our proposal to continue to assign HCPCS code C9758 to New Technology APC 1590.
Response: We thank the commenter for their support. Our updated claims data for the 4-year lookback period for the universal low volume APC policy continues to show only 3 claims for HCPCS code C9758. Therefore, we are finalizing our proposal without modification to continue assigning HCPCS code C9758 to New Technology APC 1590. Please refer to Table 24 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9758 for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
d. Bronchoscopy With Transbronchial Ablation of Lesion(s) by Microwave Energy (APC 1562)
Effective January 1, 2019, CMS established HCPCS code C9751 (Bronchoscopy, rigid or flexible, transbronchial ablation of lesion(s) by microwave energy, including fluoroscopic guidance, when performed, with computed tomography acquisition(s) and 3-D rendering, computer-assisted, image-guided navigation, and endobronchial ultrasound (EBUS) guided transtracheal and/or transbronchial sampling ( e.g., aspiration[s]/biopsy[ies]) and all mediastinal and/or hilar lymph node stations or structures and therapeutic intervention(s)). This microwave ablation procedure utilizes a flexible catheter to access the lung tumor via a working channel and may be used as an alternative procedure to a percutaneous microwave approach. Based on our review of the New Technology APC application for this service and the service's clinical similarity to existing services paid under the OPPS, we estimated the likely cost of the procedure would be between $8,001 and $8,500. We assigned the procedure to New Technology APC 1571 (New Technology—Level 34 ($8001-$8500)) for CY 2019.
In claims data available from CY 2019 for the CY 2021 OPPS/ASC final rule with comment period, there were four claims reported for bronchoscopy with transbronchial ablation of lesions by microwave energy. Given the low volume of claims for the service, we proposed for CY 2021 to apply the universal low volume APC policy we adopted in CY 2019, under which we utilize our equitable adjustment ( printed page 94023) authority under section 1833(t)(2)(E) of the Act to calculate the geometric mean, arithmetic mean, and median costs to determine an appropriate payment rate for purposes of assigning bronchoscopy with transbronchial ablation of lesions by microwave energy to a New Technology APC. We found the geometric mean cost for the service to be approximately $2,693, the arithmetic mean cost to be approximately $3,086, and the median cost to be approximately $3,708. The median was the statistical methodology that estimated the highest cost for the service. The payment rate calculated using this methodology fell within the cost band for New Technology APC 1562 (New Technology—Level 25 ($3501-$4000)). Therefore, we assigned HCPCS code C9751 to APC 1562 for CY 2021.
In CY 2022, we again used the claims data from CY 2019 for HCPCS code C9751. Because the claims data was unchanged from when it was used in CY 2021, the values for the geometric mean cost ($2,693), the arithmetic mean cost ($3,086), and the median cost ($3,708) for the service described by HCPCS code C9751 remained the same. The highest cost metric using these methodologies was again the median and within the cost band for New Technology APC 1562 (New Technology—Level 25 ($3,501-$4,000)). Therefore, we continued to assign HCPCS code C9751 to APC 1562 (New Technology—Level 25 ($3501-$4000)), with a payment rate of $3,750.50 for CY 2022.
There were no new claims for HCPCS code C9751 for the CY 2024 OPPS/ASC final rule with comment period. Therefore, we continued to use claims from CY 2019 to determine the payment rate for the service; and continued to assign HCPCS code C9751 to APC 1562 (New Technology—Level 25 ($3501-$4000)), with a payment rate of $3,750.50.
There continue to be no separately payable claims for HCPCS code C9751 since 2019. Consistent with our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, we proposed, for CY 2025, to continue to assign HCPCS code C9751 to APC 1562 (New Technology—Level 25 ($3501-$4000)), with a payment rate of $3,750.50.
Please refer to Table 25 for the proposed OPPS New Technology APC and status indicator assignments for HCPCS code C9751 for CY 2025.
We did not receive any public comments on our proposal to continue to assign HCPCS code C9751 to APC 1562 (New Technology—Level 25 ($3501-$4000)). Additionally, our updated claims data continues to show that there are no new claims for C9751 for CY 2025. Therefore, we are finalizing our proposal without modification to continue to assign HCPCS code C9751 to APC 1562. Please refer to Table 26 for the final OPPS New Technology APC and status indicator assignment for HCPCS codes C9751 for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94024)e. Cardiac Positron Emission Tomography (PET)/Computed Tomography (CT) Studies (APCs 1520 and 1522)
For CY 2025, the OPPS payment rates for the service described by CPT codes 78431, 78432, and 78433 were proposed to be based on available CY 2023 claims data. CPT code 78431 had over 26,000 single frequency claims in CY 2023. The geometric mean cost for CPT code 78431 was approximately $2,350. The geometric mean falls within APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50, which is the current APC assignment for this service. Therefore, we proposed, for CY 2025, to continue to assign CPT code 78431 to APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50.
There were only 19 single frequency claims in CY 2023 for CPT code 78432. As this is below the threshold of 100 claims for a service within a year, we proposed to apply our universal low volume New Technology APC policy and used the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT code 78432 to the appropriate New Technology APC. Using available claims data from CY 2021, CY 2022, and CY 2023, our analysis found the geometric mean cost of the service was approximately $1,760, the arithmetic mean cost of the service was approximately $1,920, and the median cost of the service was approximately $1,540. The arithmetic mean was the statistical methodology that estimated the highest cost for the service. The arithmetic mean cost of $1,923, is an amount that is above the cost band for APC 1520 (New Technology—Level 20 ($1801-$1900)), where the procedure is currently assigned for CY 2024. Therefore, we proposed, for CY 2025, to assign CPT code 78432 to APC 1521 (New Technology—Level 21 ($1901-$2000)) with a payment rate of $1950.50.
There were over 1,400 single frequency claims for CPT code 78433 in CY 2023. The geometric mean cost for CPT code 78433 was approximately $2,010, which is an amount that is above the current New Technology APC cost band for APC 1521 (New Technology—Level 21 ($1901-$2000)) to which it is assigned for CY 2024. Therefore, for CY 2025, we proposed to reassign CPT code 78433 to APC 1522 (New Technology—Level 22 ($2001-$2500)) with a payment rate of $2,250.50.
Please refer to Table 27 below for the proposed OPPS New Technology APC and status indicator assignments for CPT codes 78431, 78432, and 78433 for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
( printed page 94025)Comment: We received comments supporting our proposals to assign CPT codes 78431 and 78433 to APC 1522 (New Technology—Level 22 ($2001-$2500)) and to assign CPT code 78432 to APC 1521 (New Technology—Level 21 ($1901-$2000)). One commenter noted that the geometric mean costs and hospital reporting for PET/CT services is stabilizing and that the claims data will support stable payment rates in future years.
Response: We thank the commenters for their support. We agree that the claims data for these services have been stabilizing in recent years, and any payment adjustments have been, generally, minor. As we have begun to gather adequate claims data, we will consider whether to assign CPT codes 78431 and 78433 to clinical APCs in future rulemaking.
Comment: One commenter supported our proposals to assign CPT codes 78431 and 78433 to APC 1522 but did not support our proposal to assign CPT code 78432 to APC 1521. The commenter explained that CPT code 78431 should be assigned to APC 1522 or higher because, from a clinical workflow perspective, CPT code 78432 requires more resources than CPT code 78431. The commenter explained that they believe the low claims volume is responsible for the lower payment rate for CPT codes 78432.
Response: We thank the commenter for their input. We note that New Technology APCs are cost bands rather than clinical groupings. Unlike when we assign a service to a clinical APC and consider resource and clinical similarities to other services in a clinical APC, we assign services to New Technology APCs based on cost. While we appreciate the commenter's information regarding the clinical differences between CPT codes 78431 and 78432, we adjust New Technology APC assignments based on the claims ( printed page 94026) data available rather than clinical characteristics of a service.
We note that additional claims for CPT codes 78431 through 78433 have been processed since the proposed rule. Based on updated claims data, CPT code 78431 has an updated geometric mean cost of approximately $2,300. Because the geometric mean cost of CPT code 78431 is still within the range for APC 1522, the proposed APC assignment for CPT code 78431 for CY 2025, we are finalizing the proposed APC assignment of CPT code 78431 without modification.
There were close to 15 additional single frequency claims for CY 2023 processed for CPT code 78432 since the proposed rule, bringing the total number of single frequency claims to approximately 33 for CPT code 78432 for CY 2023. Based on the updated claims data for CPT code 78432, the geometric mean cost is approximately $1,750; the arithmetic mean cost is approximately $1,890; and the median cost is approximately $1,590. Of these, the highest statistical methodology is the arithmetic mean cost of $1,890. Since the updated arithmetic mean cost for CPT code 78432 is outside of the cost band for APC 1521 (New Technology—Level 21 ($1901-$2000), we are not finalizing our proposal to assign CPT code 78432 to APC 1521 for CY 2025. Based on the updated statistical methodologies, we are assigning CPT code 78432 to APC 1520 for CY 2025 (New Technology—Level 20 ($1801-$1900)) with a payment rate of $1,850.50.
For CPT code 78433, the geometric mean cost based on the updated claims data has changed to approximately $1964, down from approximately $2010 at the time of the proposed rule. Therefore, we are not finalizing the assignment of CPT code 78433 to APC 1522 as proposed because the geometric mean cost falls outside of the cost band for proposed APC 1522 (New Technology—Level 22 ($2001-$2500)). We are assigning CPT code 78433 to APC 1521 (New Technology—Level 21 ($1901-$2000)) for CY 2025, which is the same New Technology APC to which it is currently assigned for CY 2024.
Please refer to Table 28 for the final OPPS New Technology APC and status indicator assignments for CPT codes 7843, 78432, and 78433 for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94027)f. CardiAMP (APC 1590)
The CardiAMP cell therapy IDE studies are two randomized, double-blinded, controlled IDE studies: the CardiAMP Cell Therapy Chronic Myocardial Ischemia Trial [20] and the CardiAMP Cell Therapy Heart Failure Trial.[21] The two trials are designed to investigate the safety and efficacy of autologous bone marrow mononuclear cell treatment for the following: (1) patients with medically refractory and symptomatic ischemic cardiomyopathy; and (2) patients with refractory angina pectoris and chronic myocardial ischemia. On April 1, 2022, we established HCPCS code C9782 to describe the CardiAMP cell therapy IDE studies and assigned HCPCS code C9782 to APC 1574 (New Technology—Level 37 ($9,501-$10,000)) with the status indicator “T.” We subsequently revised the descriptor for HCPCS code C9782 to: (Blinded procedure for New York Heart Association (NYHA) Class II or III heart failure, or Canadian Cardiovascular Society (CCS) Class III or IV chronic refractory angina; transcatheter intramyocardial transplantation of autologous bone marrow cells ( e.g., mononuclear) or placebo control, autologous bone marrow harvesting and preparation for transplantation, left heart catheterization including ventriculography, all laboratory services, and all imaging with or without guidance ( e.g., transthoracic ( printed page 94028) echocardiography, ultrasound, fluoroscopy), all device(s), performed in an approved Investigational Device Exemption (IDE) study) to clarify the inclusion of the Helix trans endocardial injection catheter device in the descriptor. Additionally, we determined that APC 1590 (New Technology—Level 39 ($15,001-$20,000)) most accurately accounted for the resources associated with furnishing the procedure described by HCPCS code C9782.
For CY 2025, the proposed OPPS payment rates were based on available CY 2023 claims data. We identified three single frequency paid claims for C9782 for ratesetting for CY 2025. As this is below the threshold of 100 claims for a service within a year, we would designate C9782 as a low volume service and apply our universal low volume APC policy. Under this policy, we would use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes C9782 to the appropriate New Technology APC. Our analysis of the data found the geometric mean cost of the service is approximately $18,045, the arithmetic mean cost of the service is approximately $18,332, and the median cost of the service is approximately $20,394. The median was the statistical methodology that estimated the highest cost for the service. However, because there are only three claims for HCPCS code C9782 from the CY 2023 claims data, we had concerns that the universal low volume APC policy calculations would not accurately capture the cost of the service. Given our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed to continue to assign HCPCS code C9782 to New Technology APC 1590 with a payment rate of $17,050.50.
Please refer to Table 29 below for the proposed OPPS New Technology APC and status indicator assignments for HCPCS code C9782 for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
We did not receive any public comments on our proposal and are finalizing it without modification. HCPCS code C9782 will be assigned to New Technology APC 1590 with a status indication of “T” for CY 2025. Please refer to Table 30 for the final OPPS New Technology APC and status indicator assignment for CPT code C9782. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment via the internet on the CMS website.
( printed page 94029)g. Atherosclerosis Imaging-Quantitative Computer Tomography (AI-QCT) (APC 1511)
Atherosclerosis Imaging-Quantitative Computer Tomography (AI-QCT) is a Software as a Service (SaaS) that assesses the extent of coronary artery disease severity. This procedure is performed to quantify the extent of coronary plaque and stenosis in patients who have undergone coronary computed tomography analysis (CCTA). The AMA CPT Editorial Panel established the following four codes associated with this service, effective January 1, 2021:
0623T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; data preparation and transmission, computerized analysis of data, with review of computerized analysis output to reconcile discordant data, interpretation, and report.
0624T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; data preparation and transmission.
0625T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; computerized analysis of data from coronary computed tomographic angiography.
0626T: Automated quantification and characterization of coronary atherosclerotic plaque to assess severity of coronary disease, using data from coronary computed tomographic angiography; review of computerized analysis output to reconcile discordant data, interpretation and report.
Of these four CPT codes, only CPT code 0625T was determined to be separately payable in the OPPS and was assigned to status indicator = “S” (Procedure or Service, Not Discounted When Multiple) starting October 1, 2022. We assigned CPT code 0625T to a separately payable status indicator based on the technology and its potential utilization in the HOPD setting, our evaluation of the service, as well as input from our medical advisors. The procedure was assigned to APC 1511 (New Technology—Level 11 ($900-$1000)) with a payment rate of $950.50.
For CY 2024, the OPPS payment rates were proposed to be based on available CY 2022 claims data. There were 37 claims for CPT code 0625T during this time period. As this was below the threshold of 100 claims for a service within a year, we explained that we could propose to designate CPT code 0625T as a low volume service under our universal low volume New Technology APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign code 0625T to the appropriate New Technology APC. We found the geometric mean cost for the service to be approximately $3.70, the arithmetic mean cost to be approximately $4.10, and the median cost to be approximately $3.50. Under our universal low volume New Technology APC policy, we would use the greatest of the statistical methodologies, the arithmetic mean, to assign CPT code 0625T to New Technology 1491 (New Technology Level 1A—(0-$10)) with a payment rate of $5.00. However, we acknowledged that, because CPT code 0625T was only made separately payable as part of the OPPS in October 2022, and, therefore, the claims available only reflect two months of data, we were concerned that we did not have sufficient claims data to justify reassignment to another New Technology APC (66 FR 69902). Therefore, consistent with our current policy to retain services within New Technology APC groups until we obtain sufficient claims data to justify reassignment (66 FR 59902), for CY 2024 we adopted as final our proposal ( printed page 94030) to maintain CPT code 0625T's current assignment to APC 1511 (New Technology—Level 11 ($901-$1000) with a payment rate of $950.50.
For setting CY 2025 payment rates, there were only three separately payable claims in the CY 2023 data reported for CPT code 0625T indicating a geometric mean cost of approximately $180, which is substantially lower than the code's current payment rate of $950.50. Additionally, in CY 2022 and CY 2023, there were a total of 40 separately payable claims reported for CPT code 0625T, but it is unlikely that a service with a current payment rate of $950.50 would have a geometric mean of $4.20, an arithmetic mean of $6.60, and a median of $3.52. These findings lead to uncertainty about the appropriate payment rate for the service described by CPT code 0625T. A review of the evidence submitted by the developer of the procedure when this procedure was originally assigned to a New Technology APC, and before any claims data were available, indicated the procedure had a cost between $901 and $1,000. Claims for CPT code 0625T from CY 2021 and CY 2022 indicate that the cost of the procedure is less than $10, which would not appear to cover the basic cost of this procedure, including computing time, generating a report, and having medical personnel interpret the report. For CY 2023, the geometric mean cost of approximately $180 based on three claims may better reflect the cost of the procedure described by CPT code 0625T, but there were not enough claims to be confident about the result. Therefore, we proposed to use our authority under section 1833(t)(2)(E) for CY 2025 to continue to assign CPT code 0625T to APC 1511 (New Technology—Level 11 ($901-$1000) with a payment rate of $950.50 based on the data currently available to us, which we believe best reflects the cost of the service as described by the New Technology APC application.
Refer to Table 31 for the proposed OPPS New Technology APC and status indicator assignments for CPT code 0625T for CY 2025.
Comment: We received several comments supporting the assignment of CPT code 0625T to APC 1511 (New Technology—Level 11 ($901-$1000) for CY 2025.
Response: We thank the commenters for their support. We note that since the CY 2025 OPPS/ASC proposed rule was issued, two additional claims for CPT code 0625T have been processed, with the updated geometric mean cost increasing to approximately $498. Although we have two additional claims, we are not certain that we have enough claims data to be confident in the calculated geometric mean cost for CPT code 0625T. Therefore, we are finalizing our proposal, without modification, to continue to assign CPT code 0625T to APC 1511 (New Technology—Level 11 ($901-$1000). Please refer to Table 32 for the final OPPS New Technology APC and status indicator assignment for CPT code 0625T. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 94031)h. Corvia Medical Interatrial Shunt Procedure (APC 1592)
On July 1, 2020, we established HCPCS code C9760 (Non-randomized, non-blinded procedure for nyha class ii, iii, iv heart failure; transcatheter implantation of interatrial shunt, including right and left heart catheterization, transeptal puncture, trans-esophageal echocardiography (tee)/intracardiac echocardiography (ice), and all imaging with or without guidance (for example, ultrasound, fluoroscopy) performed in an approved investigational device exemption (ide) study, performed in an approved investigational device exemption (ide) study) to facilitate payment for the implantation of the Corvia Medical interatrial shunt.
As we stated in the CY 2021 OPPS final rule with comment period (85 FR 85947), we believe that similar resources and device costs are involved with the Corvia Medical interatrial shunt procedure and the V-Wave interatrial shunt procedure. Unlike the V-Wave interatrial shunt, which is implanted half the time the associated interatrial shunt procedure described by HCPCS code C9758 is billed, the Corvia Medical interatrial shunt is implanted every time the associated interatrial shunt procedure (HCPCS code C9760) is billed. Therefore, for CY 2021, we assigned HCPCS code C9760 to New Technology APC 1592 (New Technology—Level 41 ($25,001-$30,000)) with a payment rate of $27,500.50. We also modified the code descriptor for HCPCS code C9760 to remove the phrase “or placebo control,” from the descriptor. In CY 2022, we generally used the same claims data as was used in the CY 2021 OPPS final rule to set the payment rates for that year. Accordingly, because there were no claims for this service in CY 2019, we continued to assign HCPCS code C9760 to New Technology APC 1592 in CY 2022. There continued to be no claims data for this service in CY 2021, so we continued to assign HCPCS code C9760 to New Technology APC 1592 in CY 2023, the year for which we used CY 2021 data for ratesetting. While there was one claim in CY 2022, we continued to assign HCPCS code C9760 to New Technology APC 1592 for CY 2024 due to the extremely limited claims data available.
For CY 2025, the OPPS payment rates were proposed to be based on available CY 2023 claims data. There were no claims for HCPCS code C9760 in CY 2023. Therefore, we proposed to continue assigning HCPCS code C9760 to New Technology APC 1592.
Please refer to Table 33 for the proposed OPPS New Technology APC and status indicator assignments for HCPCS code C9760. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
( printed page 94032)We did not receive any public comments on our proposal, and we still do not have any claims for the service. Therefore, for CY 2025, we are finalizing our proposal without modification. Specifically for CY 2025, we are assigning HCPCS code C9760 to APC 1592 (New Technology—Level 41 ($25,001-$30,000)) with a payment rate of $27,500.50.
Please refer to Table 34 below for the final OPPS New Technology APC and status indicator assignments for HCPCS code C9760 for CY 2025. The CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
i. DARI Motion Procedure (APC 1505)
Effective January 1, 2022, CPT code 0693T (Comprehensive full body computer-based markerless 3D kinematic and kinetic motion analysis and report) is associated with the DARI Motion Procedure, a service that provides human motion analysis to aid clinicians in pre- and post-operative surgical intervention and in making other treatment decisions, including selecting the best course of physical therapy and rehabilitation. The technology consists of eight cameras that surround a patient, which send live video to a computer workstation that analyzes the video to create a 3D reconstruction of the patient without the need for special clothing, markers, or devices attached to the patient's clothing or skin. For CY 2022, we assigned CPT code 0693T to New Technology APC 1505 (New Technology—Level 5 ($301-$400)). For CY 2023, the OPPS payment rates were based on claims submitted between January 1, 2021, and December 31, 2021, processed through June 30, 2022. Due to its effective date of January 1, 2022, there were no claims available for CPT ( printed page 94033) code 0693T for rate setting in CY 2023. Therefore, in CY 2023, we continued to assign CPT code 0693T to New Technology APC 1505. For CY 2024, there were no claims available, so we again continued to assign CPT code 0693T to New Technology APC 1505.
For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data. Although CPT code 0693T was effective January 1, 2022, we have no claims data at this time. Because we have no claims data, for CY 2025, we proposed to continue to assign CPT code 0693T to APC 1505 with a proposed payment rate of $350.50.
Refer to Table 35 for the proposed OPPS New Technology APC and status indicator assignments for CPT code 0693T for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
We did not receive any public comments on our proposal, and we still do not have any claims for the service. Therefore, for CY 2025, we are finalizing our proposal without modification. Specifically, for CY 2025, we are assigning CPT code 0693T to APC 1505 with a status indicator of “S”. The final New Technology APC and status indicator assignments for CPT code 0693T for CY 2025 are found in Table 36. The CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
j. Instillation of Anti-Neoplastic Pharmacologic/Biologic Agent Into Renal Pelvis (APC 1558)
Effective October 1, 2023, CMS established HCPCS code C9789 (Instillation of anti-neoplastic pharmacologic/biologic agent into renal pelvis, any method, including all imaging guidance, including volumetric measurement if performed) and assigned it to New Technology APC 1559 (New Technology—Level 22 ($2001-$2500)), with a payment rate of $2,250.50 based on our review of the clinical and resource characteristics of this service.
This code may be used to describe the unique procedure associated with the administration of the drug described by HCPCS code J9281 (Mitomycin pyelocalyceal instillation, 1 mg) or similar products. HCPCS code J9281 may be used to describe the product, Jelmyto (mitomycin for pyelocalyceal solution). The FDA approved Jelmyto in 2020, and the FDA approved indication and usage for Jelmyto is as an alkylating drug indicated for the treatment of adult patients with low-grade Upper Tract Urothelial Cancer (LG-UTUS).[22]
Because we created HCPCS code C9789 effective October 1, 2023, we have limited claims data from CY 2023 available for CY 2025 rulemaking. Specifically, we only had 6 claims available. Given our proposal to ( printed page 94034) maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed to continue to assign HCPCS code C9789 to New Technology APC 1559 (New Technology—Level 22 ($2001-$2500)).
Please refer to Table 37 for the proposed OPPS New Technology APC and status indicator assignments for CPT code C9789 for CY 2025.
Comment: We received one comment in support of our proposal.
Response: We thank the commenter for their support.
Our updated claims data during the 4-year lookback period applicable for the universal low-volume APC policy continues to show only 6 claims for HCPCS code C9789. Therefore, we are finalizing our proposal without modification to continue assigning HCPCS code C9758 to New Technology APC 1559 (New Technology—Level 22 ($2001-$2500)). Please refer to Table 38 below for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9789 for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
k. LimFlow TADV Procedure CPT Code 0620T (APC 1579)
The LimFlow TADV procedure, which is described by CPT code 0620T (Endovascular venous arterialization, tibial or peroneal vein, with transcatheter placement of intravascular stent graft(s) and closure by any method, including percutaneous or open vascular access, ultrasound guidance for vascular access when performed, all catheterization(s) and intraprocedural roadmapping and imaging guidance necessary to complete the intervention, all associated radiological supervision and interpretation, when performed), is an endovascular procedure that is used ( printed page 94035) to treat patients with chronic limb-threatening ischemia. According to the developer, these patients are no longer eligible for conventional endovascular or open bypass surgery to treat their artery blockage, and without this procedure, they are likely to face limb amputation.
CPT code 0620T was established in January 2021 and was assigned to APC 5194 (Level 4 Endovascular Procedures) with a payment rate of approximately $17,400, which is the highest-paying APC for endovascular procedures. While we proposed to continue to assign CPT code 0620T to APC 5194 for CY 2024, we finalized a reassignment from a clinical APC to a New Technology APC with a higher payment rate based on comments received expressing concern that the low payment rate of the procedure would discourage providers from performing the procedure and deny access to the procedure. To determine the appropriate New Technology APC assignment for CY 2024, we looked at the available cost information. There were only 15 claims for the procedure for CY 2021 and CY 2022, so the LimFlow TADV procedure was subject to our new technology procedure low volume APC policy. An analysis of the median, arithmetic mean, and geometric mean of CPT code 0620T for CY 2024 rulemaking found that the median was approximately $25,800, the arithmetic mean was approximately $28,600, and the geometric mean was approximately $26,700. Because the arithmetic mean had the highest value of the three cost statistics, for CY 2024, we assigned CPT code 0620T to New Technology APC 1578 (New Technology—Level 41 ($25,001-$30,000)) with a payment rate of $27,500.50.
For CY 2025, the OPPS payment rates were proposed to be based on available CY 2023 claims data. We identified only six single frequency claims for CPT code 0620T in the CY 2023 claims data. As this is below the threshold of 100 claims for a service within a year, we proposed to again apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign the service to the appropriate New Technology APC. Based on our review of the available claims, we determined that the arithmetic mean was approximately $35,000; the median was approximately $36,000; and the geometric mean cost was approximately $33,000. Of these, the median was the statistical methodology that estimated the highest cost for the service. The payment rate calculated using this methodology fell within the cost band for New Technology APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50. Therefore, for CY 2025, we proposed to designate this service as a low volume service under our universal low volume APC policy and to assign HCPCS code 0620T to New Technology APC 1579.
Comment: We received a comment from the manufacturer supporting our proposal to designate CPT code 0620T as a low volume service under the universal low volume APC policy and assign the service to New Technology APC 1579 for CY 2025, as they believe the proposed assignment more accurately reflects the resources used in the procedure.
Response: We thank the commenter for their support.
We note that there are an additional five claims for CPT code 0620T for CY 2023 that have been processed since the proposed rule. Based on the 11 single frequency claims available for CY 2023, in addition to the other 26 claims in the 4-year lookback period, we determined that the arithmetic mean is $35,700; the median is $36,400; and the geometric mean cost is $33,900. Since the total number of claims for CPT code 0620T for CY 2023 is still below the 100-claim threshold, we are finalizing our proposal to designate 0620T as a low volume service under the universal low volume APC policy. The payment rate calculated using the highest of these methodologies, the median, continues to fall within the cost band for New Technology APC 1579 (New Technology—Level 42 ($30,001-$40,000)) with a payment rate of $35,000.50. Therefore, we are also finalizing our proposal without modification to assign CPT code 0620T to New Technology APC 1579 for CY 2025.
Please refer to Table 40 for the final OPPS New Technology APC and status indicator assignments for CPT code ( printed page 94036) 0662T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
l. Liver Histotripsy Service (APC 1576)
CPT code 0686T (Histotripsy ( i.e., non-thermal ablation via acoustic energy delivery) of malignant hepatocellular tissue, including image guidance) was first effective July 1, 2021, and describes the histotripsy service associated with the use of the HistoSonics system. Histotripsy is a non-invasive, non-thermal, mechanical process that uses a focused beam of sonic energy to destroy cancerous liver tumors and is currently in a non-randomized, prospective clinical trial to evaluate the efficacy and safety of the device for the treatment of primary or metastatic tumors located in the liver.[23] When HCPCS code 0686T was first effective, the histotripsy procedure was designated as a Category A IDE clinical study (NCT04573881). Since devices in Category A IDE studies are excluded from Medicare payment, payment for CPT code 0686T only reflected the cost of the service that is performed (absent the cost of the device) each time it is reported on a claim. On March 2, 2023, the histotripsy IDE clinical study was re-designated as a Category B (Non-experimental/Investigational) IDE study. Due to this new designation, payment for CPT code 0686T in CY 2024 reflects payment for both the service that is performed and the device used each time it is reported on a claim. For CY 2024, we assigned CPT code 0686T to New Technology APC 1576 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of $17,500.50.
For CY 2025, proposed OPPS payment rates were based on available CY 2023 claims data. We have identified one claim for CPT code 0686T within the CY 2023 claims data. As the available claims data is below the threshold of 100 claims for a service within a year, we would propose to designate CPT code 0686T as a low volume service under our universal low volume APC policy, and use the highest of the geometric mean cost, arithmetic mean cost, or median cost to assign CPT code 0686T to the appropriate New Technology APC. However, because there is only a single claim in the CY 2023 data, we have concerns that the universal low volume APC policy calculations do not accurately capture the cost of the service.
Given our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, and based on the fact that there have only been 3 claims for CPT code 0686T in the prior 4-year period, we proposed to continue to assign CPT code 0686T to APC 1576 (New Technology—Level 39 ($15,001-$20,000)) with a payment rate of $17,500.50 as shown in Table 25.
Please refer to Table 41 for the proposed OPPS New Technology APC and status indicator assignments for CPT code 0686T for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule.
( printed page 94037)We did not receive any public comments on our proposal and are finalizing it without modification. HCPCS code 0686T will be assigned to New Technology APC 1576 with a status indication of “S” for CY 2025. Please refer to Table 42 below for the final OPPS New Technology APC and status indicator assignment for CPT code 0686T. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
m. LiverMultiScan Service (APC 1511)
Effective July 1, 2021, CPT codes 0648T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained without diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure) during the same session; single organ) and 0649T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained with diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure); single organ (list separately in addition to code for primary procedure)) are associated with the LiverMultiScan service. LiverMultiScan is a Software as a medical Service (SaaS) that is intended to aid the diagnosis and management of chronic liver disease, the most prevalent of which is Non-Alcoholic Fatty Liver Disease (NAFLD). It provides standardized, quantitative imaging biomarkers for the characterization and assessment of inflammation, hepatocyte ballooning, and fibrosis, as well as steatosis, and iron accumulation. LiverMultiScan receives MR images acquired from patients' providers and analyzes the images using their proprietary Artificial Intelligence (AI) algorithms. It then sends the providers a quantitative metric report of the patient's liver fibrosis and inflammation. In accordance with our SaaS add-on codes policy (87 FR 72032 to 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Thus, CPT code 0649T, the add-on code for LiverMultiScan, is assigned to the identical APC and status indicator as CPT code 0648T, the standalone code for the same service. For CY 2024, we assigned CPT codes 0648T and 0649T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000) with a payment rate of $950.50.
For CY 2025, the OPPS payment rates were proposed to be based on available CY 2023 claims data. We identified 71 claims for CPT code 0648T and 72 claims for CPT code 0649T for CY 2023. As this is below the threshold of 100 claims for each code within a year, we ( printed page 94038) proposed to utilize our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes 0648T and 0649T to the appropriate New Technology APC. There are available claims data from CY 2021 and CY 2022 for CPT codes 0648T and 0649T. Our analysis of the combined data, 114 claims for CPT code 0648T and 115 claims for CPT code 0649T, yielded a geometric mean cost of approximately $180, an arithmetic mean cost of approximately $234, and a median cost of approximately $197. We explained that we believed it was appropriate to utilize our universal low volume APC policy to assign the LiverMultiScan service to a New Technology APC because we stated that we believed that the combined claims data from CY 2021 to CY 2023 provided sufficient claims to capture the cost of the service. The arithmetic mean was the statistical methodology that estimated the highest cost for CPT codes 0648T and 0649T. Therefore, we proposed to reassign CPT codes 0648T and 0649T to New Technology APC 1504 (New Technology—Level 4 ($201-$300)) with a payment rate of $250.50 as shown in Table 43.
Comment: We received several comments, including a comment from the manufacturer, requesting that we maintain the APC assignment for the CPT codes 0648T and 0649T at APC 1511. Many commenters stated that the cost of each of the services is at least $950, and that the proposed payment rate of $250 would be inadequate to ensure patient access. Commenters noted that the claims data used to determine the CY 2025 OPPS proposed APC assignment for this service represent an insufficient number of claims to estimate the cost for this service. One commenter expressed concern about the wide data variability present in the claims data for CPT codes 0648T and 0649T. One commenter requested that we develop a payment policy for SaaS like CPT codes 0648T and 0649T that results in appropriate and consistent payment for innovative new services. We also received one comment signed by ten radiologists stating that the current payment rate for the procedures was too high.
Response: We recognize that software-based technologies, like those described by CPT codes 0648T and 0649T, are rapidly evolving and, therefore, we are considering for future rulemaking whether specific adjustments to payment policies, including our New Technology APC policies, are needed to more accurately and appropriately pay for these products and services across settings of care. For CY 2025, we recognize that the few claims available for CPT codes 0648T and 0649T may not truly represent the cost of this service. While we received a comment expressing concern over the potential overpayment of the service, we do not believe we have sufficient claims to justify a reassignment at this time. Therefore, we agree with the commenters that, for both CPT codes 0648T and 0649T, we should maintain the payment rates for CY 2025 until we have additional claims data.
Comment: One commenter supported the proposed reassignment of CPT codes 0648T and 0649T to New Technology APC 1504. The commenter stated that the payment rate of $950 created a paradigm where the third-party automated quantitative analytic service was valued greater than the MRI procedure that included technologist staffing, professional interpretation and liability. The commenter believed that the payment of the service should be ( printed page 94039) based on the true cost of these automated digital quantitative services at New Technology APC 1504.
Response: We thank the commenter for its support of our proposal. We reiterate that the few claims for CPT codes 0648T and 0649T may not truly represent the cost of this service or justify a reassignment at this time.
After consideration of the public comments we received, we are finalizing our proposal with modifications. We are using our equitable adjustment authority under section 1833(t)(2)(E) to continue to assign CPT codes 0648T and 0649T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000) with a payment rate of $950.50 for CY 2025. Please refer to Table 44 below for the final OPPS New Technology APC and status indicator assignments for CPT codes 0648T and 0649T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
n. Optellum Lung Cancer Prediction (LCP) (APC 1508)
CPT codes 0721T (Quantitative computed tomography (CT) tissue characterization, including interpretation and report, obtained without concurrent CT examination of any structure contained in previously acquired diagnostic imaging) and 0722T (Quantitative computed tomography (CT) tissue characterization, including interpretation and report, obtained with concurrent CT examination of any structure contained in the concurrently acquired diagnostic imaging dataset (list separately in addition to code for primary procedure)) became effective July 1, 2022, and are associated with the Optellum LCP technology. The Optellum LCP applies an algorithm to a patient's CT scan to produce a raw risk score for a patient's pulmonary nodule. The physician uses the risk score to quantify the risk of lung cancer and to determine what the next management step should be for the patient (for example, CT surveillance versus invasive procedure). In accordance with our SaaS add-on codes policy (87 FR 72032 and 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Thus, CPT code 0722T, the add-on code for the Optellum LCP service, is assigned to the identical APC and status indicator as CPT code 0721T, the standalone code for the same service. For CY 2024, we assigned CPT codes 0721T and 0722T to APC New Technology 1508 (New Technology—Level 8 ($601-$700)).
For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data. We identified three claims for CPT codes 0721T and 0722T for ratesetting for CY 2025. As this is below the threshold of 100 claims for a service within a year, we would usually propose to apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes 0721T and 0722T to the appropriate New Technology APC. There are available claims data only from CY 2023 for CPT codes 0721T and 0722T. Our analysis of the data for CPT code 0721T found the geometric mean cost of the service is approximately $84, the arithmetic mean cost of the service is approximately $98, and the median ( printed page 94040) cost of the service is approximately $130. We did not identify any reported claims for CPT code 0722T. However, because there were only three claims for the Optellum LCP service and these claims show a much lower cost than would be expected based on the current APC assignment of this service, we have concerns that the universal low volume APC policy calculations do not accurately capture the cost of the service. Given our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low-volume APC policy, we proposed to continue to assign CPT codes 0721T and 0722T to New Technology APC 1508 (New Technology—Level 8 ($601-$700)) with a proposed payment rate of $650.50.
Please refer to Table 45 below for the proposed OPPS New Technology APC and status indicator assignments for HCPCS codes 0721T and 0722T for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
We did not receive any public comments on our proposal, and we are finalizing our proposal without modification. We are assigning HCPCS codes 0721T and 0722T to New Technology APC 1508 with a status indication of “S” for CY 2025. Please refer to Table 46 below for the final OPPS New Technology APC and status indicator assignment for CPT codes 0721T and 0722T. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94041)o. Quantitative Magnetic Resonance (QMR) for Analysis of Tissue Composition (APC 1511)
Effective January 1, 2022, CPT codes 0697T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained without diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure) during the same session; multiple organs) and 0698T (Quantitative magnetic resonance for analysis of tissue composition ( e.g., fat, iron, water content), including multiparametric data acquisition, data preparation and transmission, interpretation and report, obtained with diagnostic mri examination of the same anatomy ( e.g., organ, gland, tissue, target structure); multiple organs (list separately in addition to code for primary procedure)) are associated with the CoverScan Software as a medical Service (SaaS). This service is a medical image management and processing software package that analyzes MR data and provides quantified metrics of multiple organs such as the heart, lungs, liver, spleen, pancreas, and kidney. In accordance with our SaaS add-on codes policy (87 FR 72032 to 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Thus, CPT code 0698T, the add-on code for CoverScan is be assigned to the same APC and status indicator as CPT code 0697T, the standalone code for the same service. For CY 2024, we assigned CPT codes 0697T and 0698T to New Technology APC 1511 (New Technology—Level 11 ($900-$1,000)).
For CY 2025, the proposed OPPS payment rates were based on available CY 2023 claims data. We identified 48 claims for CPT code 0698T and no claims for CPT code 0697T in CY 2023. As this is below the threshold of 100 claims for a service within a year, we proposed to apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes 0697T and 0698T to the appropriate New Technology APC. There are also available claims data from CY 2022 for CPT code 0698T. Our analysis of the combined data, approximately 80 claims, yielded a geometric mean cost of approximately $444, an arithmetic mean cost of approximately $622, and a median cost of approximately $786. The median cost is the statistical methodology that estimates the highest cost for CPT codes 0697T and 0698T. Therefore, we proposed, for CY 2025, to reassign CPT codes 0697T and 0698T to APC 1509 (New Technology—Level 9 ($701-$800)) with a payment rate of $750.50.
Refer to Table 47 below for the proposed OPPS New Technology APC and status indicator assignments for CPT codes 0697T and 0698T for CY 2025. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
( printed page 94042)Comment: Commenters requested that we maintain the APC assignment for the CPT codes 0697T and 0698T at APC 1511 for CY 2025. Many commenters stated that the cost of each of the services is at least $950, and that the proposed payment rate of $750 does not cover the cost of this service. One commenter noted that the claims data used to determine the CY 2025 OPPS proposed rate for this service represent an insufficient number of claims to estimate the cost for this service. Additionally, the commenter stated that wide variability in claims data distorts the applicability of the universal low volume APC policy and skews the arithmetic mean value. Commenters were concerned that the proposed payment rate would hinder patient access to these services.
Response: We recognize that software-based technologies, like those described by CPT codes 0697T and 0698T, are rapidly evolving and, therefore, we are considering for future rulemaking whether specific adjustments to payment policies, including our New Technology APC policies, are needed to more accurately and appropriately pay for these products and services across settings of care. For CY 2025, we recognize that the few claims available for CPT codes 0697T and 0698T may not truly represent the cost of this service. We agree with the commenters that, for both CPT codes 0697T and 0698T, we should maintain the payment rates for CY 2025 until we have additional claims data.
After consideration of the public comments we received, we are finalizing our proposal with modifications. We will use our equitable adjustment authority under section 1833(t)(2)(E) to continue to assign CPT codes 0697T and 0698T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000) with a payment rate of $950.50 for CY 2025. Please refer to Table 48 below for the final OPPS New Technology APC and status indicator assignments for CPT codes 0697T and 0698T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94043)p. Quantitative Magnetic Resonance Cholangiopancreatography (QMRCP) (APC 1511)
Effective July 1, 2022, CPT codes 0723T (Quantitative magnetic resonance cholangiopancreatography (QMRCP) including data preparation and transmission, interpretation and report, obtained without diagnostic magnetic resonance imaging (MRI) examination of the same anatomy ( e.g., organ, gland, tissue, target structure) during the same session) and 0724T (Quantitative magnetic resonance cholangiopancreatography (QMRCP), including data preparation and transmission, interpretation and report, obtained with diagnostic magnetic resonance imaging (MRI) examination of the same anatomy ( e.g., organ, gland, tissue, target structure) (list separately in addition to code for primary procedure)) are associated with the QMRCP Software as a medical Service (SaaS). The service performs quantitative assessment of the biliary tree and gallbladder. It uses a proprietary algorithm that produces a three-dimensional reconstruction of the biliary tree and pancreatic duct and also provides precise quantitative information of biliary tree volume and duct metrics. In accordance with our SaaS add-on codes policy (87 FR 72032 and 72033), SaaS CPT add-on codes are assigned to the same APCs and status indicators as their standalone codes. Consistent with our SaaS add-on codes policy, CPT code 0724T, the add-on code for QMRCP is assigned to the identical APC and status indicator as CPT code 0723T, the standalone code for the same service. For CY 2024, we assigned CPT codes 0723T and 0724T to New Technology APC 1511 (New Technology—Level 11 ($900-$1,000)).
For CY 2025, the proposed OPPS payment rates were based on available CY 2023 claims data. We identified 3 claims for CPT code 0724T and no claims for CPT code 0723T in CY 2023. As this is below the threshold of 100 claims for a service within a year, we would usually propose to apply our universal low volume APC policy and use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign CPT codes 0723T and 0724T to the appropriate New Technology APC. There is only one single claim from CY 2022 for CPT code 0724T and no claims for CPT code 0723T. For CY 2023, we received 3 claims for CPT code for CPT 0724T and no claims for CPT code 0723T. Our analysis of the combined CY 2022 and CY 2023 data for CPT code 0723T and 0724T found the geometric mean cost of the service is approximately $26, the arithmetic mean cost of the service is approximately $26, and the median cost of the service is approximately $27. Given our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, we proposed to continue to assign the CPT codes 0723T and 0724T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000)) with a payment rate of $950.50.
Refer to Table 49 for the proposed OPPS New Technology APC and status indicator assignments for CPT codes 0723T and 0724T for CY 2025.
( printed page 94044)We did not receive any public comments on our proposal to continue to assign the CPT codes 0723T and 0724T to New Technology APC 1511 (New Technology—Level 11 ($901-$1,000)). Our updated claims data did not show any additional claims for CY 2023 for CPT codes 0723T and 0724T. Therefore, we are finalizing our proposal without modification to continue to assign CPT codes 0723T and 0724T to APC 1511. Please refer to Table 50 for the final OPPS New Technology APC and status indicator assignment for CPT codes 0723T and 0724T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94045)q. Scalp Cooling (APC 1519)
CPT code 0662T (Scalp cooling, mechanical; initial measurement and calibration of cap) became effective on July 1, 2021, to describe initial measurement and calibration of a scalp cooling device for use during chemotherapy administration to prevent hair loss. According to Medicare's National Coverage Determination (NCD) policy, specifically, NCD 110.6 (Scalp Hypothermia During Chemotherapy to Prevent Hair Loss), the scalp cooling cap itself is classified as an incident to supply to a physician service, and would not be paid under the OPPS; however, interested parties have indicated that there are substantial resource costs of around $1,900 to $2,400 associated with calibrating and fitting the cap. CPT guidance states that CPT code 0662T should be billed once per chemotherapy session, which we interpret to mean once per course of chemotherapy. Therefore, if a course of chemotherapy involves, for example, 6 or 18 sessions, HOPDs should report CPT 0662T only once for that 6 or 18 therapy sessions.
For CY 2022, we assigned CPT code 0662T to APC New Technology 1520 (New Technology—Level 20 ($1,801-$1,900)) with a payment rate of $1,850.50. For CY 2023, we did not have any claims data, so we continued to assign CPT code 0662T to APC 1520. For CY 2024 we finalized reassignment of CPT code 0662T to APC 1514 (New Technology—Level 14 ($1,201-$1,300)) with a payment rate of $1,250.50 based on 11 single frequency claims.
For CY 2025, the OPPS payment rates were proposed to be based on available CY 2023 claims data. The Scalp Cooling service became effective in the OPPS in CY 2022, and we identified 38 single frequency paid claims for CPT code 0662T for CY 2023. As this is below the threshold of 100 claims for a service within a year, we proposed to designate CPT code 0662T as a low volume service under our universal low volume APC policy and to use the highest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data to assign the service to the appropriate New Technology APC. Based on our review of the available claims, we determined that the geometric mean cost for CPT code 0662T was approximately $841; the median was approximately $1,351; and the arithmetic mean was approximately $1,361. Therefore, for CY 2025, we proposed to designate this service as a low volume service under our universal low volume APC policy and to reassign CPT code 0662T to APC 1515 (New Technology—Level 15 ($1301-$1400)) with a payment rate of $1,350.50 for CY 2025 based on the arithmetic mean of approximately $1,361.Please refer to Table 51 below for the proposed OPPS New Technology APC and status indicator assignment for CPT code 0662T. The proposed CY 2025 payment rates can be found in Addendum B CY 2025 OPPS/ASC proposed rule via the internet on the CMS website.
( printed page 94046)We did not receive any public comments on our proposal. We note that additional claims for CPT code 0662T have been processed since the proposed rule. Specifically, there are 50 single frequency claims for CPT code 0662T for CY 2023. Since this is below the threshold of 100 claims, we are finalizing our proposal to designate CPT code 0662T as a low volume service under our universal low volume APC policy and use the greater of the geometric mean, arithmetic mean, or median cost based on the available claims data for CPT code 0662T to calculate an appropriate payment rate for purposes of assigning CPT code 0662T to a New Technology APC. Based on the updated claims data available for this final rule, we found the geometric mean cost to be approximately $1,110, the median to be $1,750, and the arithmetic mean to be $1,420. The median was the statistical methodology that estimated the highest cost for the service. The payment rate calculated using this methodology falls within the cost band for New Technology APC 1519 (New Technology—Level 19 ($1701-$1800)). Therefore, we are assigning CPT code 0662T to APC 1519 for CY 2025.
Please refer to Table 52 for the final OPPS New Technology APC and status indicator assignments for CPT code 0662T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
r. Supervised Visits for Esketamine Self-Administration (APCs 1513 and 1518)
On March 5, 2019, FDA approved SpravatoTM (esketamine) nasal spray, used in conjunction with an oral antidepressant, for treatment of depression in adults who have tried other antidepressant medicines but have not benefited from them (treatment-resistant depression (TRD)). This is the first FDA approval of esketamine for any use.
Esketamine is a noncompetitive N-methyl D-aspartate (NMDA) receptor antagonist. It is a nasal spray supplied as an aqueous solution of esketamine hydrochloride in a vial with a nasal spray device. Each device delivers two sprays containing a total of 28 mg of esketamine. Patients would require either two (2) devices (for a 56 mg dose) or three (3) devices (for an 84 mg dose) per treatment.
Because of the risk of serious adverse outcomes resulting from sedation and dissociation caused by esketamine nasal spray administration, and the potential for misuse of the product, it is only available through a restricted distribution system under a Risk Evaluation and Mitigation Strategy (REMS). A REMS is a drug safety program that the FDA can require for certain medications with serious safety concerns to help ensure the benefits of the medication outweigh its risks. The SpravatoTM REMS program requires the esketamine nasal spray to be dispensed and administered to enrolled patients in health care settings that are certified in the REMS. See www.fda.gov for more information regarding the SpravatoTM REMS program compliance requirements.
A treatment session of esketamine consists of instructed nasal self-administration by the patient followed by a period of at least 2 hours post-administration observation of the patient under direct supervision of a health care professional in the certified health care setting. Refer to the CY 2020 PFS final rule and interim final rule for ( printed page 94047) more information about supervised visits for esketamine nasal spray self-administration (84 FR 63102 through 63105).
To facilitate prompt beneficiary access to the new, potentially life-saving treatment for TRD using esketamine, we created two new HCPCS G codes, G2082 and G2083, effective January 1, 2020. HCPCS code G2082 is for an outpatient visit for the evaluation and management of an established patient who requires the supervision of a physician or other qualified health care professional and provision of up to 56 mg of esketamine through nasal self-administration and includes 2 hours of post-administration observation. For CY 2020, HCPCS code G2082 was assigned to New Technology APC 1508 (New Technology—Level 8 ($601-$700)) with a payment rate of $650.50. HCPCS code G2083 describes a similar service to HCPCS code G2082 but involves the administration of more than 56 mg of esketamine. For CY 2020, HCPCS code G2083 was assigned to New Technology APC 1511 (New Technology—Level 11 ($901-$1000)) with a payment rate of $950.50. Updates to the APC assignments for G2082 and G2083 have been made in past rules. See the CY 2021 OPPS/ASC final rule with comment period (85 FR 85948), CY 2022 OPPS/ASC final rule with comment period (86 FR 63538), CY 2023 OPPS/ASC final rule with comment period (87 FR 71816-71817), and CY 2024 OPPS/ASC final rule with comment period (88 FR 81628—81630) for these updates.
For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data as the available single frequency claims exceed the 100 claims threshold generally used for our universal low volume policy. Therefore, for CY 2025, we proposed to assign HCPCS codes G2082 and G2083 to New Technology APCs based on the codes' geometric mean costs. Specifically, we proposed to assign HCPCS code G2082 to New Technology APC 1512 (New Technology—Level 12 ($1001-$1100)) with a payment rate of $1,050.50 based on its geometric mean cost of $1,087, which was calculated using the available 424 single frequency claims from CY 2023 claims data. We also proposed to assign HCPCS code G2083 to New Technology APC 1518 (New Technology—Level 18 ($1601-$1700)) with a payment rate of $1,650.50 based on its geometric mean cost of $1,643, which was calculated using the available 2,482 single frequency claims from CY 2023 claims data.
Refer to Table 53 for the New Technology APC and status indicator assignments for HCPCS codes G2082 and G2083. The proposed CY 2025 payment rates for these HCPCS codes can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule.
( printed page 94048)We did not receive any public comments on our proposal. We note however that the geometric mean costs for both HCPCS code G2082 and HCPCS code G2083 have changed since the proposed rule. Based on the updated claims data available for this final rule, the geometric mean cost for HCPCS code G2082 is $1,104.71 and the geometric mean cost for G2083 is $1,448.83. Based on updated claims data available for this final rule with comment period, we are finalizing a New Technology APC assignment for HCPCS code G2082 to APC 1513 (New Technology—Level 13 ($1101-$1200)) with a payment of $1150.50 and we are finalizing a New Technology APC assignment for HCPCS code G2083 to APC 1516 (New Technology—Level 16 ($1401-$1500)) with a payment of $1450.50.
Please refer to Table 54 for the CY 2025 proposed and final APC and SI assignments for HCPCS codes G2082 and G2083. The CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
Finally, we note that because we have gathered additional claims data and seen increases in claims volume, we are considering placing HCPCS codes G2082 and G2083 into clinical APCs through future rulemaking.
( printed page 94049)s. Surfacer® Inside-Out® Access Catheter System (APC 1534)
HCPCS code C9780 (Insertion of central venous catheter through central venous occlusion via inferior and superior approaches ( e.g., inside-out technique), including imaging guidance) describes the procedure associated with the use of the Surfacer® Inside-Out® Access Catheter System that is designed to address central venous occlusion. HCPCS code C9780 was established on October 1, 2021, and since its establishment the code has been assigned to New Technology APC 1534 (New Technology—Level 34 ($8001-$8500)).
For CY 2025, there were only 3 single frequency claims in CY 2023 for HCPCS code C9780. There were no available claims from CY 2021 or CY 2022. Given our proposal to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, we proposed to continue to assign HCPCS code C9780 to APC 1534 (New Technology—Level 34 ($8001-$8500)) with a payment rate of $8,250.50. Refer to Table 55 for the proposed New Technology APC and status indicator assignments for HCPCS code C9780.
( printed page 94050)We did not receive any public comments on our proposal to continue to assign CPT code C9870 to APC 1534 (New Technology—Level 34 ($8001-$8500)). We note that there was one additional claim in our updated claims data, however, the total number of claims for CY 2023 is still under 10. Therefore, given our new policy to maintain current New Technology APC assignments for CY 2025 for New Technology APC services with fewer than 10 claims in the 4-year lookback period applicable for the universal low volume APC policy, we are finalizing as proposed to continue to assign CPT code C9870 to APC 1534. Please refer to Table 56 below for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9780. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
t. TASS (APC 1537)
The Transcatheter Atrial Shunt System (TASS) is a nitinol self-expanding cardiovascular implant consisting of four arms including two left atrial (LA) arms and two coronary sinus (CS) arms placed between the left atrium and coronary sinus to create a 7mm flow diameter channel for blood to flow from the high pressure region of the left atrium to the lower pressure region of the right atrium via the coronary sinus. TASS is currently in a Category B IDE clinical trial. Effective October 1, 2023 CMS created HCPCS code C9792 (Blinded or nonblinded procedure for symptomatic New York Heart Association (NYHA) Class II, III, IV a heart failure; transcatheter implantation of left atrial to coronary sinus shunt using jugular vein access, including all imaging necessary to intra procedurally map the coronary sinus for optimal shunt placement ( e.g., TEE or ICE ultrasound, fluoroscopy), performed under general anesthesia in an approved investigational device exemption (IDE) study) to describe the TASS service and assigned it to APC 1537 (New Technology—Level 37 ($9501-$10000)) with a payment rate of $9,750.50.
For CY 2025, the proposed OPPS payment rates were based on available CY 2023 claims data. Due to the effective date of the code of October 1, 2023, there were no claims available for HCPCS code C9792 for rate setting in CY 2024. Therefore, in CY 2025, we proposed to continue to assign HCPCS code C9792 to APC 1537.
Please refer to Table 57 below for the current and proposed OPPS New Technology APC and status indicator assignments for HCPCS code C9792. The proposed CY 2025 payment rates can be found in Addendum B to the proposed rule via the internet on the CMS website.
( printed page 94051)Comment: We received a comment supporting our proposal to continue to assign HCPCS code C9792 to APC 1537.
Response: We appreciate the commenter's support for our proposal.
After consideration of the public comments we received, we are finalizing our proposal without modification to assign CPT code C9792 to APC 1537 (New Technology—Level 37 ($9501-$10000)) with a payment rate of $9,750.50 for CY 2025. Please refer to Table 58 for the final OPPS New Technology APC and status indicator assignment for HCPCS code C9792. The final CY 2024 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
u. Magnetic Resonance Imaging With Inhaled Hyperpolarized Xenon-129 Contrast Agent (APC 1551)
HCPCS code C9791 (Magnetic resonance imaging with inhaled hyperpolarized xenon-129 contrast agent, chest, including preparation and administration of agent) was established on October 1, 2023. For CY 2023, we assigned HCPCS code C9791 to New Technology APC 1551 (New Technology—Level 14 ($1201-$1300)). For CY 2024, the OPPS payment rates were based on claims submitted between January 1, 2022, and December 31, 2022, processed through June 30, 2023. Due to the effective date of the service of October 1, 2023, there were no claims available for HCPCS code C9791 for rate setting in CY 2024. Therefore, in CY 2024, we continued to assign HCPCS code C9791 to New Technology APC 1551.
For CY 2025, the proposed OPPS payment rates were based on available CY 2023 claims data. Although HCPCS code C9791 was effective October 1, 2023, we did not have any claims data for the service for the proposed rule. Therefore, for CY 2025, we proposed to continue to assign HCPCS code C9791 to New Technology APC 1551 (New Technology—Level 14) with a proposed payment rate of $1,250.50. ( printed page 94052)
Please refer to Table 59 for the proposed OPPS New Technology APC and status indicator assignment for HCPCS code C9791 for CY 2025.
We did not receive any public comments on our proposal, and we still do not have any claims for the service. Therefore, for CY 2025, we are finalizing our proposal without modification to assign HCPCS code C9791 to New Technology APC 1551 (New Technology—Level 14 ($1201-$1300)) and SI “T”. The final New Technology APC and status indicator assignments for HCPCS code C9791 for CY 2025 are found in Table 60. The CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
v. SAINT Neuromodulation System
The SAINT Neuromodulation System is a non-invasive repetitive transcranial magnetic stimulation (rTMS) system that identifies an individualized target and delivers navigationally directed repetitive magnetic pulses to that individualized target located within the left dorsolateral prefrontal cortex to treat major depressive disorder (MDD). The patient first receives structural MRI and functional MRI scans that are analyzed by the provider to identify and localize the personalized stimulation target in the patient's dorsolateral prefrontal cortex. Once the areas targeted for treatment are identified, the patient receives non-invasive magnetic stimulation in the targeted area. The patient has 10 treatment sessions per day with each treatment session lasting 10 minutes followed by 50 minutes of rest before another treatment session occurs. The treatment is administered over five days for a total of 50 sessions of non-invasive magnetic stimulation therapy. There are four CPT codes listed in Table 61 that describe the MRI scans that are used to target the treatment and describe the administration of the non-invasive magnetic stimulation therapy.
( printed page 94053)For CY 2025, the OPPS payment rates were proposed based on available CY 2023 claims data. However, CPT codes 0889T, 0890T, 0891T, and 0892T did not become effective until July 1, 2024, which means there was no claims data for the procedures described these CPT codes. We assigned our proposed rates for these services based on our evaluation of the resources needed to perform these services. Table 62 lists CPT codes 0889T, 0890T, 0891T, and 0892T and our proposed status indicator and APC assignment for CY 2025.
( printed page 94054)Comment: Several commenters, including clinicians and other interested parties, believe that the payment rate of $2,250.50 for the proposed APC assignment to New Technology APC 1522 (New Technology—Level 22 ($2001-$2500)) for CPT codes 0890T, 0891T, and 0892T that describes the administration of the non-invasive magnetic stimulation therapy is too low. The commenters are concerned that the proposed payment rate will discourage providers from using the SAINT neuromodulation system to treat MDD. The commenters supported assigning CPT codes 0890T, 0891T, and 0892T to New Technology APC 1528 (New Technology—Level 28 ($5001-$5500)) with a payment rate of $5,250.50 to better reflect the resource cost of each of these three services. Multiple commenters also felt that the IPPS new technology add-on payment amount for the SAINT neuromodulation system acknowledged the cost of the SAINT system.
Response: We do not have any claims data to directly determine the cost of the procedures described by CPT codes 0890T, 0891T, and 0892T. Therefore, we make our assessment of the resource costs to perform the procedure based on our judgment of the clinical and resource information presented to us about the SAINT neuromodulation system and the treatment approach in the manufacturer's OPPS New Technology APC application and through comments on the proposed rule. We agree with the commenters that the cost per case used for purposes of determining the IPPS maximum new technology add-on payment amount (88 FR 58938) was a more appropriate estimate of the cost of the SAINT neuromodulation system than our original cost estimate for the procedure in the CY 2025 OPPS proposed rule. For purposes of determining the new technology add-on payment amount, the cost estimate for the entire SAINT course of treatment, which included fifty sessions over five days of the non-invasive magnetic stimulation therapy and related operating costs, was $19,500.
Our review has determined that the cost estimate of the procedure described by CPT code 0899T should remain the same and CPT code 0899T should ( printed page 94055) continue to be assigned to New Technology APC 1511 (New Technology—Level 11 ($901-$1000)) with a payment rate of $950.50. However, we now have determined that the per day cost of the services described by CPT codes 0890T, 0891T, and 0892T is around $3,710 instead of our proposed estimate of around $2,250. We are changing the APC assignment for CPT codes 0890T, 0891T, and 0892T to New Technology APC 1525 (New Technology—Level 25 ($3501-$4000)) with a payment rate of $3,750.50.
Comment: Several commenters described their experiences with MDD and receiving treatment for their MDD using the SAINT neuromodulation system. All of these commenters claimed that the SAINT therapy was beneficial to them and helped to reduce the symptoms of MDD, which substantially improved their mental health. Many of these commenters were able to receive SAINT because they participated in clinical trials for the therapy or were able to afford the treatment even if it was not covered by health insurance. The commenters want us to ensure that SAINT will be affordable to patients who receive treatment, and that SAINT will be widely available throughout the United States, so those who have MDD are able to access this therapy.
Response: We thank commenters for their input, and we are committed to establishing appropriate payment for the services described by CPT codes 0890T, 0891T, and 0892T.
After consideration of the public comments we received, we are finalizing our proposal with modification for CY 2025 to assign CPT code 0899T to New Technology APC 1511 (New Technology—Level 11 ($901-$1000)) and SI of “S” and to assign CPT codes 0890T, 0891T and 0892T to New Technology APC 1525 (New Technology—Level 25 ($3501-$4000)) and SI of “S”. The final New Technology APC and status indicator assignments for CPT codes 0889T, 0890T, 0891T, and 0892T for CY 2025 are found in Table 63. The CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addendum D1 can also be found via the internet on the CMS website.
( printed page 94056)D. Universal Low Volume APC Policy for Clinical and Brachytherapy APCs
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63743 through 63747), we adopted a policy to designate clinical and brachytherapy APCs as low volume APCs if they have fewer than 100 single claims that can be used for ratesetting purposes in the claims year used for ratesetting for the prospective year. For the CY 2025 OPPS/ASC proposed rule, CY 2023 claims are generally the claims used for ratesetting; and clinical and brachytherapy APCs with fewer than 100 single claims from CY 2023 that can be used for ratesetting would be low volume APCs subject to our universal low volume APC policy. As we stated in the CY 2022 OPPS/ASC final rule with comment period, we adopted this policy to reduce the volatility in the payment rate for those APCs with fewer than 100 single claims. Where a clinical or brachytherapy APC has fewer than 100 single claims that can be used for ratesetting, under our low volume APC payment adjustment policy, we determine the APC cost as the greatest of the geometric mean cost, arithmetic mean cost, or median cost based on up to 4 years of claims data. We excluded APC 5853 (Partial Hospitalization for CMHCs) and APC 5863 (Partial Hospitalization for Hospital-based PHPs) from our universal low volume APC policy given the different nature of policies that affect the partial hospitalization program. We also excluded APC 2698 (Brachytx, stranded, nos) and APC 2699 (Brachytx, non-stranded, nos) as our current methodology for determining payment rates for non-specified brachytherapy sources is appropriate.
Based on claims data available for the CY 2025 OPPS/ASC proposed rule, we proposed to designate six brachytherapy APCs and five clinical APCs as low volume APCs under the OPPS. The six brachytherapy APCs and five clinical APCs meet our criteria of having fewer than 100 single claims in the claims' year used for ratesetting (CY 2023 for the CY 2025 OPPS/ASC proposed rule). Nine of the 11 APCs were designated as low volume APCs in CY 2024. Based on data for the CY 2025 OPPS/ASC proposed rule, APC 2645 (Brachytx, ( printed page 94057) non-stranded, gold-198) and APC 5881 (Ancillary Outpatient Services When Patient Dies) met our criteria to be designated a Low Volume APCs; and we proposed to designate those APCs as such for CY 2025.
We did not receive any public comments on our proposal. Based on CY 2023 claims data available for this final rule with comment period, we are finalizing our proposal to designate six brachytherapy APCs and five clinical APCs as low volume APCs under the OPPS. Table 64 includes the CY 2023 claims available for ratesetting for each of the APCs we are designating as Low Volume APCs for CY 2025. The final cost statistics for our CY 2025 Low Volume APCs, such as the median, arithmetic mean, and geometric mean cost are available for download with this final rule with comment period on the CMS website. We refer readers to our website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices; click on the relevant regulation to download the low volume APC cost statistics under the comprehensive (OPPS) ratesetting methodology in the downloads section of the web page.
E. APC-Specific Policies
1. Cardiac CT Services, CPT Codes 75572, 75573, and 75574 (APC 5572)
For the 2006 coding update, the AMA's CPT Editorial Panel established six Category III CPT codes to describe cardiac computed tomography angiography with contrast materials (cardiac CT services) effective January 1, 2006. The codes were active and separately payable under the OPPS between January 1, 2006, and December 31, 2009. The CPT Editorial Panel deleted the Category III CPT codes and replaced them with Category I CPT codes 75572 through 75574 effective January 1, 2010. With the deletion of the Category III CPT codes on December 31, 2009, we crosswalked the APC assignments from the Category III CPT codes (predecessor codes) to the new Category I CPT codes effective January 1, 2010. Since 2010, the Category I CPT codes describing cardiac CT services are CPT codes 75572, 75573, and 75574. The codes and their long descriptors are listed below.
- 75572: Computed tomography, heart, with contrast material, for evaluation of cardiac structure and morphology (including 3D image postprocessing, assessment of cardiac function, and evaluation of venous structures, if performed).
- 75573: Computed tomography, heart, with contrast material, for evaluation of cardiac structure and morphology in the setting of congenital heart disease (including 3D image postprocessing, assessment of left ventricular (LV) cardiac function, right ventricular (RV) structure and function and evaluation of vascular structures, if performed).
- 75574: Computed tomographic angiography, heart, coronary arteries and bypass grafts (when present), with contrast material, including 3D image postprocessing (including evaluation of cardiac structure and morphology, assessment of cardiac function, and evaluation of venous structures, if performed).
Like the predecessor codes, CPT codes 75572, 75573, and 75574 have been paid separately under the OPPS since 2010. From CY 2015 through CY 2024, the OPPS payment rate, based on the geometric mean cost for the cardiac CT codes, has ranged between $175 and $265 for these codes, as listed in Table 65 below.
( printed page 94058)We note that the OPPS payment rate applies only to the hospital outpatient facility and does not include the physician service payment. Physician services are paid under Medicare's Physician Fee Schedule (PFS).
We update the OPPS payment rates on an annual basis consistent with the requirements set forth in section 1833(t)(9)(A) of the Act, which requires the HHS Secretary to review, not less often than annually, and revise the APC groups, the relative payment weights, and the wage and other adjustments to take into account changes in medical practice, changes in technology, the addition of new services, new cost data, and other relevant information and factors. Because of the annual updates, OPPS payment rates for services may fluctuate from year to year. We note that we generally use the latest claims data available to set the annual payment rates. Payment rates for this CY 2025 OPPS/ASC final rule with comment period are based on claims with dates of service between January 1, 2023, and December 31, 2023, processed through June 30, 2024.
Over the years we have received comments noting that the payment for these codes has declined since 2017. Comments on previous OPPS proposed rules have indicated that the payment amount is insufficient to cover the cost of providing the service and have stated that the payment amount does not consider the hospital resources required to perform these services, including the use of the equipment, medication administration, staff time, and scanner time. We have maintained over the years that an analysis of our claims data for these three codes have shown geometric mean costs consistent with the geometric mean cost for APC 5571 (Level 1 Imaging with Contrast), the APC to which the cardiac CT codes have been assigned since 2015.
We have also received comments in the past urging CMS to allow hospitals the flexibility to submit charges for cardiac CT services with a revenue code other than CT scan (035X) and Radiology Diagnostic (032X) revenue codes, implying that MACs had applied edits to the cardiac CT codes that prevented hospitals from reporting a cardiology (048X) revenue code when appropriate. It is longstanding CMS policy that hospital outpatient facilities are responsible for reporting the appropriate cost centers and revenue codes on claims. As stated in section 20.5 in Chapter 4 (Part B Hospital) of the Medicare Claims Processing Manual, CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignments of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.” We have consistently stated that hospital outpatient facilities must determine the most appropriate cost center and revenue code for the cardiac CT codes (87 FR 71849, 88 FR 81664).
After we issued the CY 2024 OPPS/ASC final rule, interested parties notified us of a specific claims edit that may have limited the revenue codes reported with the cardiac CT codes in prior years' claims data. CMS removed the revenue code edit in early December 2023 to allow for the cardiac CT codes to be billed with any appropriate revenue code. We informed the public of our findings and the changes that we made in the January 2024 OPPS Update (Transmittal 12421, Change Request 13488), dated December 21, 2023. We believe the edit may have prevented some providers from reporting the cardiology revenue code (048X), which maps to the cardiology cost center (03140), when billing for cardiac CT services. In the past, commenters have indicated that the cardiology cost center has a higher cost-to-charge ratio (CCR) than the imaging cost centers, and they believe the inability to report the cardiology revenue code has resulted in a lower payment rate for cardiac CT services. Since the OPPS ratesetting process utilizes the applicable cost center's CCR to reduce the charges on the claim to estimated cost, utilizing cost centers with lower CCRs results in a lower OPPS payment compared to utilizing cost centers with higher CCRs. With the edit no longer in place, hospitals may bill for cardiac CT services with whichever revenue code they believe appropriate for CY 2024, including cardiology revenue code 048X, and the CY 2026 OPPS payment rates (which most likely will be based on CY 2024 claims) will reflect those updated revenue code billing patterns. We note that for CY 2025, based on our standard ratesetting methodology using claims submitted during CY 2023, our analysis reveals that the angiocardiography and CT scan revenue codes were reported with CPT codes 75572 through 75574, which were mapped to angiography and CT scan cost centers, as shown in Table 66.
( printed page 94059)Because we wanted to determine whether the revenue edit could have affected the geometric mean costs for CPT codes 75572 through 75574, we conducted studies prior to the CY 2025 proposed rule and during the comment period to calculate HCPCS geometric mean costs for these codes based on a simulation that assumed that differing numbers of HOPDs (specifically 25 percent, 50 percent, and 75 percent of the total number of HOPDs billing for these services) would have assigned these services to the cardiology revenue code (048X) and cardiology cost center (03140). Based upon the results of the studies, we found that if 50 percent or more of HOPDs had billed these services with the cardiology revenue code (048X) and cardiology cost center (03140), the geometric mean cost for these codes would have increased and would have resulted in a revised APC assignment from APC 5571 (Level 1 Imaging with Contrast) to APC 5572 (Level 2 Imaging with Contrast). As noted in Table 67, under our standard ratesetting methodology, the geometric mean cost for the cardiac CT codes would be approximately $182, which maps to APC 5571, while an assumption that 50 percent of HOPDs billed with the cardiology revenue code (048X) and cardiology cost center (03140) on CY 2023 claims would result in a geometric mean cost of about $386, which maps to APC 5572.
We also reviewed the limited CY 2024 claims data for cardiac CT services available to determine the percentage of HOPDs that are utilizing the cardiology revenue code (048X) and cardiology cost center (03140) with the understanding that many HOPDs may still be updating their current billing practices. We did not find any appreciable change in current billing practices.
( printed page 94060)Because the RTP edit associated with the cardiac CT codes may have affected the CY 2023 data we have available to establish the CY 2025 OPPS payment rates for these services, in the CY 2025 OPPS/ASC proposed rule, we requested information on the following topics regarding hospitals' billing practices for cardiac CT services:
(1) Where are cardiac CT services performed in a hospital? Are cardiac CT services performed in a dedicated cardiology department, radiology department, or some other hospital outpatient department?
(2) What factors determine the revenue code assignment for cardiac CT services ( i.e., the department in which the service is performed, the type of service that is performed, or some other factor)?
(3) What revenue codes are HOPDs reporting for these services in CY 2024? Are HOPDs using the cardiology revenue code on claims for cardiac CT services now that they are no longer restricted from using this revenue code?
We stated in the CY 2025 OPPS/ASC proposed rule that we would use the comments received and the information from the CY 2024 claims data to help us identify whether the current OPPS payment is appropriate for the cardiac CT codes, or whether we should revise the payment methodology for the CY 2025 OPPS. We received a significant number of comments in response to our Request for Information. While a major portion of the comments were part of a large write-in campaign, commenters provided information specific to their experiences in their facilities. We appreciate all the feedback and address the comments below.
Comment: We received a significant number of comments in response to our Request for Information about cardiac CT services and the billing practices for these services. Commenters ranged from specialty societies to healthcare providers who practice in a variety of settings of different sizes and in various locations throughout the United States.
In terms of where cardiac CT services are performed, commenters shared that cardiac CT services may be performed in a radiology department, an interventional radiology department, a cardiology department, or a clinic setting. Resources to furnish the services, such as staff, are utilized from both the cardiology and radiology departments. Many commenters described the test being performed in radiology departments utilizing cardiology nursing staff to prepare the patient, administer medications (such as IV beta blockers and nitroglycerin), and monitor the patient during and after the procedure.
In terms of the factors that determine the revenue code assignment for cardiac CT services, most commenters stated that the resources used for these services are very similar to other cardiac procedures such as cardiac stress tests or even cardiac catheterizations, which are assigned to the cardiology revenue code. Commenters described cardiac CT services as resource intensive, stating that the cross-department coordination between cardiology and radiology, the skill level of staff (technicians, nurses, and physicians), the expense of up-to-date CT equipment, and the amount of testing time involved is comparable to other more expensive and invasive cardiac tests.
In response to our request for the revenue codes hospital outpatient departments are reporting with cardiac CT services now that there is not an edit in place prohibiting them from using the cardiology revenue code, commenters stated that they want to use the cardiology revenue code when billing cardiac CT services, but they are unable to due to third-party edits (billing and payer) in place from clearinghouses, billing companies, or billing software companies. Some stated that they have continued to use the radiology revenue codes to prevent receiving claims denials. Another commenter stated their coding staff expressed compliance concerns with assigning cardiac CT services to a cardiology revenue code versus the CT revenue code. Commenters requested that CMS provide education about revenue code use for providers, healthcare facilities, clearinghouses, and health plans and indicated that explicit education would facilitate use of the cardiac revenue code with cardiac CT services.
Many commenters shared how many cardiac CT services are completed per year at their facilities. They expressed concern about the declining payment for such a complex, resource-intensive test that is used to accurately diagnose and manage coronary artery disease and the impact that insufficient payment could have on beneficiary access. Commenters noted that cardiac CT has been identified by the American College of Cardiology as a first line approach to manage patients with chest pain and asserted that cardiac CT is more clinically useful than more expensive, invasive tests such as cardiac catheterizations.
Response: We thank the commenters for their feedback regarding the clinical value cardiac CT services provide for clinicians diagnosing and managing patients with chest pain and coronary artery disease. We also thank commenters for describing the process of cardiac CT tests and all the resources used in providing those services and sharing information about selection of revenue code assignment when billing for CT services.
We acknowledge commenters' concerns that, even with the revenue code edit removed for CY 2024, there are a number of procedural and logistical hurdles to providers billing with the cardiology revenue codes in CY 2024 and that it would not be realistic to expect a majority of providers to be billing with the cardiology revenue codes at this point due to those issues. We acknowledge that process changes often happen slowly and that it may take time for facilities to update their billing practices, even if the majority of providers would prefer to bill these codes with the cardiology revenue code.
We appreciate commenters sharing their concerns about third-party edits that are still affecting the billing process and understand their request for additional guidance from CMS. We remind readers that we informed the public of our findings and the changes that we made in the January 2024 OPPS Update (Transmittal 12421, Change Request 13488), dated December 21, 2023. Specifically, we stated the following: “We recently identified an outdated return-to-provider (RTP) Healthcare Common Procedure Coding System-to-revenue code edit that resulted in certain claims submissions being limited to specific revenue codes for CPT codes 75572, 75573, and 75574. These claims were returned to the providers for resubmission. The outdated edit has been removed; and providers, when appropriate, may begin billing these codes with any appropriate revenue code.” We have also stated in section 20.5 in Chapter 4 (Part B Hospital) of the Medicare Claims Processing Manual, CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignments of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.” We have consistently stated that hospital outpatient facilities must determine the most appropriate cost center and revenue code for the cardiac CT codes (87 FR 71849, 88 FR 81664). While we believe that this information should suffice for education to third parties who may have edits in place that affect the selection of a cardiology ( printed page 94061) revenue code when billing for cardiac CT services and for other interested parties who have compliance concerns, we will be providing public education and instruction through the CMS Medicare Learning Network (MLN).
We are persuaded after a review of the comments submitted that a majority of providers who bill these codes would prefer to bill them with the cardiology revenue code but have not been able to do so due to the prior revenue code edit and remaining procedural hurdles that flow from the prior revenue code edit. As stated above, if there had not been a systems edit in place preventing providers from choosing the cardiology revenue code (048X) and cardiology cost center (03140) and 50 percent or more of HOPDs had billed these services with the cardiology revenue code and cardiology cost center, the geometric mean cost for these codes would have increased and would have resulted in a revised APC assignment from APC 5571 (Level 1 Imaging with Contrast) to APC 5572 (Level 2 Imaging with Contrast). Therefore, for CY 2025, we are using our equitable adjustment authority under section 1833(t)(2)(E) of the Act to utilize an alternative methodology to calculate the payment for the cardiac CT services in CY 2025 and subsequent years. Specifically, we are finalizing a temporary reassignment of the cardiac CT codes (CPT code 75572 through 75574) to APC 5572 (Level 2 Imaging with Contrast). See Table 68 for the CY 2025 Proposed and Final OPPS SI and APC assignments for CPT codes 75572 through 75574. We anticipate that it may take 3 to 4 years to see an impact from changes in billing practices from providers based on the comments we received on the proposed rule. If we do not see a significant change in the geometric mean costs after several years, we would revert payment for these services to the standard OPPS payment methodology and assign the cardiac CT codes to appropriate APCs based on their geometric mean costs. We note that this APC reassignment will not involve reprocessing of claims with dates prior to January 1, 2025.
( printed page 94062)The final CY 2025 payment with rates for the cardiac CT CPT codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
2. Neurostimulator and Related Procedures (APCs 5461 Through 5465)
In the CY 2021 OPPS/ASC final rule with comment period, we finalized a five-level APC structure for the Neurostimulator and Related Procedures series (85 FR 85968 through 85970). For a detailed discussion of the history of neurostimulators policy, we refer readers to the CY 2015, CY 2020, CY 2021, CY 2023, and CY 2024 OPPS/ASC final rules with comment period (79 FR 66807 through 66808; 84 FR 61162 through 6116, 85 FR 85968 through 85970; 87 FR 71869; 88 FR 81645 through 81658).
In the CY 2024 OPPS/ASC final rule with comment period, we did not make any changes to the 5 level APC structure for the Neurostimulator and Related Procedures series. However, we made temporary changes to services previously assigned to the neurostimulator APCS: reassigning HCPCS code 33276 (Insertion of phrenic nerve stimulator system (pulse generator and stimulating lead[s]), including vessel catheterization, all imaging guidance, and pulse generator initial analysis with diagnostic mode activation, when performed), with predecessor code 0424T, and 33287 (Removal and replacement of phrenic nerve stimulator, including vessel ( printed page 94063) catheterization, all imaging guidance, and interrogation and programming, when performed; pulse generator), from New Technology APC 1581 to New Technology APC 1580 (88 FR 81645 through 81647) and assigning HCPCS code 0266T (Implantation or replacement of carotid sinus baroreflex activation device; total system (includes generator placement, unilateral or bilateral lead placement, intra-operative interrogation, programming, and repositioning, when performed)) to New Tech APC 1580 (88 FR 81658).
For the CY 2025 OPPS/ASC proposed rule, we proposed to maintain the 5 level APC structure for the Neurostimulator series. We proposed to assign HCPCS codes 0266T and 33276 to the APC 5465 (Level 5 Neurostimulator and Related Procedures), given the cost and coding changes associated with those codes. We noted that while we have claims data available for HCPCS codes in the neurostimulator APC series that are no longer active, there would also be cost and coding changes associated with new CPT codes as their claims data become available.
While we continued to believe that a five-level structure for the Neurostimulator and Related Procedures APC series remained appropriate, we also solicited comments from interested parties in CY 2025 OPPS/ASC proposed rule on the need for a Level 6 APC, given the clinical and estimated cost characteristics of the services currently assigned to APC 5465 (Level 5 Neurostimulator and Related Procedures).
In summary, for the CY 2025 OPPS, we proposed to maintain the current 5-level structure for the Neurostimulator and Related Procedure APC series, assign HCPCS codes 0266T and 33276 to APC 5465, and solicited comment creating an additional Level 6 Neurostimulator APC.
See Table 69 for the services that we proposed to reassign to the Level 5 APC and Table 70 below for the proposed CY 2025 Neurostimulator and Related Procedures APCs.
( printed page 94064)At the August 26, 2024, HOP Panel Meeting, several presenters provided information to the Panel regarding HCPCS codes 33276 (Insertion of phrenic nerve stimulator system (pulse generator and stimulating lead[s]), including vessel catheterization, all imaging guidance, and pulse generator initial analysis with diagnostic mode activation, when performed), 64568 (Open implantation of cranial nerve ( e.g., vagus nerve) neurostimulator electrode array and pulse generator), and 0266T (Implantation or replacement of carotid sinus baroreflex activation device; total system (includes generator placement, unilateral or bilateral lead placement, intra-operative interrogation, programming, and repositioning, when performed)). The presenters advised the Panel to request that CMS either create a new Level 6 Neurostimulator and Related Procedures APC and consider including the three codes mentioned above in the Level 6 Neurostimulator APC, or alternatively assign the three codes mentioned above to New Technology APC 1580 (New Technology—Level 43 ($40,001-$50,000)). The HOP Panel agreed with the presenter and made that recommendation.
Comment: Many commenters requested the creation of a Level 6 Neurostimulator and Related Procedures APC, noting that creating an additional APC would align payment more closely with costs incurred while maintaining access to services assigned to that APC. Many of these commenters requested that if a Level 6 APC could not be created, that HCPCS code 0266T be maintained in New Technology APC 1580 for an additional year. Other commenters asked that CMS reconsider the proposed assignment of HCPCS code 33276 to APC 5465 and place the code in an APC consistent with the resources required for the procedure. One of those commenters specifically requested an APC assignment to New Technology APC 1580 until a more permanent solution to address disparity between payment and resource costs could be determined. Some commenters requested that HCPCS code 64568 be moved from APC 5465 (Level 5 Neurostimulator and Related Procedures) to New Technology APC 1580. Two commenters supported maintaining the current 5 level APC structure for the Neurostimulator series. One of those commenters recommended that HCPCS codes 33276 and 0266T continue to be assigned to New Technology APC 1580 to maintain beneficiary access while additional claims data are collected, while the other recommended more broadly that New Technology APCs be used for similar scenarios where codes had high cost but low volume. One commenter believed that HCPCS code 64568 should remain assigned to APC 5465 (Level 5 Neurostimulator and Related Procedures) since it is a long-established procedure and would not be expected to have changes in cost or volume the way “new” technologies would.
Response: We appreciate the thoughtful responses commenters have provided with regard to the proposed policy and our requests for comments. While we understand the concerns and issues commenters have raised, we believe that the 5 level APC structure for the series continues to remain appropriate in grouping together procedures with similar cost and clinical characteristics. Therefore, we are finalizing the 5 level APC structure for the Neurostimulator and Related Procedures APCs.
However, we also recognize the concerns commenters have raised regarding HCPCS codes 0266T and 33276 and in particular, the request that we return those codes to their current New Technology APC placements should we establish a final policy of not creating an additional APC level. Thus, we are assigning these codes to New Tech APC 1580, the same APC these codes were assigned to in CY 2024, in this CY 2025 OPPS/ASC final rule with comment period.
For HCPCS code 64568, we believe its current assignment to the Level 5 APC remains appropriate. The code has been assigned to the Neurostimulator and Related Procedures APC series for many years and the claims volume we have for the procedure allow for an estimate of the associated resource costs. Therefore, we do not believe a New Tech APC assignment is appropriate at this time, as New Technology APC placements are typically used when insufficient cost and claims data are available to make appropriate clinical APC assignments. We will continue to monitor the data as additional claims become available for these codes.
After consideration of the public comments we received, we are finalizing our proposal with modification. We are finalizing the ( printed page 94065) proposed 5 level APC structure for the Neurostimulator and Related Procedures APC series. However, we are continuing to assign HCPCS codes 33276 and 0266T to New Technology APC 1580 (New Technology—Level 43 ($40,001-$50,000)) in CY 2025 rather than finalizing our proposal to assign them to APC 5465. See Table 71 for the status indicators and APC assignments for HCPCS codes 0266T and 33276. We note that we have previously discussed the placement of these codes in New Technology APCs until such time that sufficient claims data were available to assign those codes to clinical APCs (87 FR 81769, 88 FR 81645 through 81647, 88 FR 81658). We will continue to monitor the data as more claims for these codes become available. Finally, we are maintaining the APC assignment of HCPCS code 64568 to APC 5465 (Level 5 Neurostimulator and Related Procedures). Please see Table 74 for the final CY 2025 OPPS APC and SI assignments for HCPCS codes 0266T and 33276.
3. Focal Laser Ablation, CPT Code 0655T (APC 5375)
Focal laser ablation is an MRI directed and image guided, minimally invasive procedure that targets prostate cancer tissue. The focal laser ablation procedure, represented by CPT code 0655T (Transperineal focal laser ablation of malignant prostate tissue, including transrectal imaging guidance, with mr-fused images or other enhanced ultrasound imaging), became effective July 1, 2021, and describes the destruction of localized prostate cancer tissue with the high energy source of focal laser radiation. The procedure utilizes real-time intraoperative prostate ultrasound fused with MRI guidance to allow the surgeon to plan the ablation and guide the laser targeting as well as providing real-time feedback to minimize changes to the tissues outside of the targeted ablation zone. This procedure offers another therapy option for select patients with localized intermediate risk prostate cancer.
For the CY 2025 OPPS/ASC proposed rule, we reviewed the CY 2023 claims submitted between January 1, 2023, through December 31, 2023, that were processed on or before December 31, 2023, for CPT code 0655T and found seven single frequency claims available for ratesetting, with a resulting geometric mean cost of $12,777. Additionally, for the CY 2025 OPPS/ASC proposed rule, we examined the procedures assigned to the Urology Procedures APCs. Based on our examination of the procedures assigned to Urology and Related Procedures APCs and the available CY 2023 claims data, we stated that we believe it is appropriate to move CPT code 0655T to APC 5375 (Level 5 Urology and Related Services) from APC 5374 (Level 4 Urology and Related Services) because 0655T shares more resource cost and clinical homogeneity with procedures in APC 5375. Specifically, we explained that we believe CPT code 0655T shares resources and is clinical homogeneous with CPT code 0714T (Transperineal laser ablation of benign prostatic hyperplasia, including imaging guidance), and CPT code 52648 (Laser vaporization of prostate, including control of postoperative bleeding, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, internal urethrotomy and transurethral resection of prostate are included if performed)). We noted that the seven available CY 2023 single frequency claims for CPT code 0655T would not significantly impact the geometric mean cost calculations for APC 5374 and APC 5375. Therefore, for CY 2025, we proposed to reassign CPT code 0655T from APC 5374 (Level 4 Urology and Related Services) to APC 5375 (Level 5 Urology and Related Services).
Comment: One commenter supported our proposal to reassign CPT code 0655T from APC 5374 (Level 4 Urology and Related Services) to APC 5375 (Level 5 Urology and Related Services), citing the clinical and resource homogeneity with CPT codes 52648 and 0714T. Furthermore, the commenter suggested we monitor the impact of the ( printed page 94066) reassignment on the geometric mean cost for APCs 5374 and 5375 to ensure payment stability.
Response: We thank the commenter for the support. Based on evaluation of their clinical and resource similarities, and taking into account updated claims data for this final rule with comment period, we continue to believe it is appropriate to reassign CPT code 0655T to APC 5375.
After consideration of the public comment we received, we are finalizing our policy without modification for CPT code 0655T. Please refer to Table 72 below for the final OPPS APC and status indicator assignments for HCPCS code 0655T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
4. Bone Mass Measurement: Biomechanical Computed Tomography (BCT) Analysis With and Without Vertebral Fracture Assessment, CPT Codes 0554T-0558T, 0743T (APCs 5521, 5523, and 5731)
CPT code 0743T (Bone strength and fracture risk using finite element analysis of functional data and bone mineral density (BMD), with concurrent vertebral fracture assessment, utilizing data from a computed tomography scan, retrieval and transmission of the scan data, measurement of bone strength and BMD and classification of any vertebral fractures, with overall fracture-risk assessment, interpretation and report) became effective January 1, 2023. This code describes the service associated with biomechanical computed tomography (BCT) analysis with concurrent vertebral fracture assessment (VFA).
In the CY 2023 OPPS/ASC final rule (87 FR71844 through 71846), we stated that, based on our review and understanding of the service, BCT analysis does not meet Medicare's definition of bone mass measurement, as specified in § 410.31(a), which specifies the coverage of, and payment for, bone mass measurements for Medicare beneficiaries. Therefore, we assigned the BCT codes (CPT codes 0554T-0558T) and CPT code 0743T to status indicator “E1” to indicate that these codes are not covered by Medicare, and not paid by Medicare when submitted on outpatient claims (any outpatient bill type). The complete long descriptors for the codes can be found below:
0554T: Bone strength and fracture risk using finite element analysis of functional data and bone-mineral density utilizing data from a computed tomography scan; retrieval and transmission of the scan data, assessment of bone strength and fracture risk and bone-mineral density, interpretation and report.
0555T: Bone strength and fracture risk using finite element analysis of functional data and bone-mineral density utilizing data from a computed tomography scan; retrieval and transmission of the scan data.
0556T: Bone strength and fracture risk using finite element analysis of functional data and bone-mineral density utilizing data from a computed tomography scan; assessment of bone strength and fracture risk and bone mineral density.
0557T: Bone strength and fracture risk using finite element analysis of functional data and bone-mineral density utilizing data from a computed tomography scan; interpretation and report.
0558T: Computed tomography scan taken for the purpose of biomechanical computed tomography analysis.
In the CY 2024 OPPS/ASC proposed rule, we proposed to continue to assign the codes to status indicator “E1” to indicate non-coverage and non-payment for the services. (See Addendum B for CY 2024/ASC proposed rule via the internet on the CMS website.) However, as discussed in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81660 through 81661), based on comments received and further review of the issue, we did not finalize our proposal. We instead assigned CPT code 0555T to APC 5731 (Level 1 Minor Procedures) and SI of “S,” (Procedure or Service, Not Discounted When Multiple; Paid under OPPS; separate APC payment), CPT code 0556T to APC 5523 (Level 3 Imaging without Contrast) and SI of “S,” and CPT code 0558T to APC 5521 (Level 1 Imaging without Contrast) with SI of “S,” which were the same APC assignments for the codes between CY 2019 and CY 2022. In addition, we assigned CPT codes 0554T, 0557T, and 0743T to SI of “M” (Items and Services Not Billable to the MAC. Not paid under OPPS) to indicate that these codes are not payable under the OPPS because they describe physician-only services. The final payment rates for these codes were listed in the OPPS Addendum B that was released with the CY 2024 OPPS/ASC final rule via the internet on the CMS website.
For the CY 2025 OPPS/ASC proposed rule, we proposed to assign CPT codes 0554T and 0557T to status indicator of “M” as the codes include or describe a professional component of the service ( printed page 94067) that is provided by a physician as evidenced by “interpretation and report” in the descriptor. It is important to note that CPT code 0554T is a comprehensive code (or “parent code”) that includes both technical and professional components. Because there are additional CPT codes (“child codes”) that facilities can use to describe the technical components of BCT analysis, we explained that we believed it was appropriate for the comprehensive code that includes the professional component to be assigned a SI of “M”. In addition, we proposed to assign CPT code 0555T to APC 5731 (Level 1 Minor Procedures) and a SI of “S,” CPT code 0556T to APC 5523 (Level 3 Imaging without Contrast) and a SI of “S,” and CPT code 0558T to APC 5521 (Level 1 Imaging without Contrast) with a SI of “S.”
For the CY 2025 OPPS/ASC proposed rule, we explained that we believe the service described by CPT code 0743T is a comprehensive code and involves both a technical component and a professional component that are performed by hospital outpatient facilities. Unlike CPT 0554T, there are no additional codes to describe the technical component(s) of this service (BCT analysis and VFA) and there is a parenthetical note instructing facilities to not report the BCT analysis codes (0554T-0557T) with CPT code 0743T. Consequently, we proposed to assign CPT code 0743T to APC 5523 (Level 3 Imaging without Contrast) and we proposed to change the status indicator for 0743T from “M” to “S” (Procedure or Service, Not Discounted When Multiple; Paid under OPPS; separate APC payment) for CY 2025. See Table 73 below.
Comment: We received several comments describing support for the use of BCT services to assess bone strength, bone mineral density, and fracture risk indicating that these services were cost-effective and provided valuable clinical information. Commenters noted the significant impact that osteoporosis has on Medicare beneficiaries, especially women. They shared that BCT is effective in clinical practice when determining use and approval of anti-osteoporotic medications that are dependent on bone density assessments. They also noted that DXA (dual-energy X-ray absorptiometry) scans (most common test used to measure bone mineral density) may be difficult to do when patients have scoliosis or extensive degenerative diseases and thus prefer to use BCT.
Response: We appreciate the feedback supporting BCT services.
Comment: We received one comment requesting that we assign payable status indicators for CPT codes 0554T and 0557T.
Response: We thank the commenter for their request. As stated above, we proposed to continue to assign CPT codes 0554T and 0557T a status indicator of “M” as the codes include or describe a professional component of the service that is provided by a physician as evidenced by “interpretation and report” in the descriptor. CPT code 0554T is a comprehensive code (or “parent code”) that includes both technical and professional components. Because there are additional CPT codes (“child codes”) that facilities can use to describe the technical components of BCT analysis, it is appropriate for the comprehensive code that includes the professional component to be assigned a SI of “M”. CPT code 0557T describes only the professional component of the BCT analysis code and therefore, is appropriately assigned a SI of “M”.
Comment: We received several comments expressing support for the proposed APC and status indicator assignments.
Response: We thank the commenters for their support.
Comment: Several commenters believe that Medicare has limited access to some BCT services in the hospital outpatient setting by assigning non-payable status indicators. Consequently, they are requesting that the pertinent codes be allowed through national coverage.
Response: We appreciate these comments. In Transmittal 2362 dated September 19, 2019, we note the BCT codes, specifically CPT codes 0554T, 0555T, 0556T, 0557T, and 0558T were listed as covered effective July 1, 2019 under NCD 150.3 Bone Mineral Density Studies. These services were added in error and were retracted due to “added in error” in Transmittal 11952 dated April 12, 2023. We apologize for the confusion this has caused and the mistaken belief that “coverage was revoked” when in fact these services had not been approved for national coverage.
While CMS further considers the issue of national coverage, we will finalize our proposed status indicator and (if applicable) APC assignments for CPT codes 0554T-0558T and CPT code 0743T as listed in Table G42 below.
The regulation at 42 CFR 410.31(f) (Bone mass measurement: Conditions for coverage and frequency standards) recognizes that CMS may use the Medicare National Coverage Determination (NCD) process to examine additional BMM systems for coverage. The public may request an NCD for a particular item or service by following the procedures established at 78 FR 48164 (August7, 2013). The commenter may request a new NCD (or seek reconsideration of an existing NCD). As a reminder to our readers, National Coverage Determinations do not include coding and payment decisions.
In summary, after consideration of the public comments, we are finalizing our proposed status indicator and (if applicable) APC assignments for CPT codes 0554T-0558T and CPT code 0743T without modification. The final payment rates for the separately payable codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator (SI) meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
( printed page 94068) ( printed page 94069)5. 3D Contour Simulation, CPT Code 0944T (APC 5523)
CAS-One® IR is Software as a Service (SaaS) that assists a physician or other qualified health care professional during percutaneous liver ablation treatments by providing 3D images that allow the physician to visualize the liver lesions, as well as define suitable margins and pathways to perform the ablation procedure. The AMA CPT Editorial Panel established CPT code 0944T effective January 1, 2025:
The long descriptor for CPT code 0944T is as follows: 3D contour simulation of target liver lesion(s) and margin(s) for image-guided percutaneous microwave ablation.
In the CY 2025 OPPS/ASC proposed rule with comment period, we proposed to assign 0944T to status indicator “E1” to indicate that the code is not payable by Medicare when submitted on outpatient claims because the service had not received FDA clearance at the time of the assignment.
Comment: We received one comment from the manufacturer. The commenter states that the 3D simulation is always performed with CPT code 47382 (Ablation, 1 or more liver tumor(s), percutaneous, radiofrequency) which is assigned to APC 5361 (Level 1 Laparoscopy and Related Services) and has a payment rate of approximately $5,800. The manufacturer asserts the costs for the 3D simulation service are approximately $3,000 which include a CT of the abdomen with contrast, utilization of the CAS-One® system, sterile accessories (patient markers, instrument markers, drapes, and marker templates), and annual services for the system (including maintenance, warranty, software subscription, and clinical support). They have requested that we reassign CPT code 0944T to APC 5362 (Level 2 Laparoscopy and Related Service) with a payment rate of approximately $10,400 and a status indicator of J1, representing a comprehensive service that includes both the ablation procedure (CPT code 47382) and the 3D simulation service (CPT code 0944T).
Response: We thank the commenter for their input. We disagree that we should consider adding the costs of both the ablation procedure (CPT code 47382) and the 3D simulation service (CPT code 0944T) to determine the appropriate payment for the 3D simulation service. The ablation procedure (CPT code 47382) is a primary service as identified by the OPPS status indicator “J1” (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS). The ablation procedure can be done with or without 3D simulation services, but 3D simulation services are never provided without the primary ablation services. Under our C-APC policy, we designate a service described by a HCPCS code assigned to a C-APC as the primary service when the service is identified by OPPS status indicator “J1”. Because the 3D simulation service is always performed with the ablation service, we believe the 3D simulation could be considered an adjunctive service. (See 78 FR 74865 and 79 FR 66799.) Payments for adjunctive services are generally packaged into the payments for the primary services.
After review of the procedure description for 3D simulation and input from our CMS medical officers, we do not believe it would be appropriate to always package the costs of the 3D simulation procedure into the ablation procedure. Based on the technology and its potential use in the HOPD setting, we used CPT code 75557 (Cardiac magnetic resonance imaging for morphology and function without contrast material) as a comparator code to determine an APC and status indicator assignment for CPT 0944T. While we realize that there are significant differences between these two codes, we believe that both codes use a type of imaging service to provide valuable information for the clinician to assist in further diagnosing and treating patients. CPT code 75557 is assigned to APC 5523 (Level 3 Imaging without Contrast) and a status indicator of “Q1” (STV-Packaged Codes; Paid under OPPS). We also note that the CAS-One® IR has received 510(k) Premarket Notification from the FDA and should no longer be assigned status indicator “E1”.
In summary, we are revising the status indicator for CPT code 0944T from status indicator “E1” to status indicator “Q1” (STV-Packaged Codes; Paid under OPPS), indicating a conditionally packaged procedure. CPT code 0944T will be packaged when it is provided with a significant procedure but will be separately paid when the service appears on the claim without a significant procedure (see Table 74). We are assigning CPT code 0944T to APC 5523 (Level 3 Imaging without Contrast) with a final payment rate of around $240.00. Please refer to Table G43 for the code descriptor, APC assignment, and status indicator assignment for CPT code 0944T for CY 2025. The final payment rate for 0944T can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator (SI) meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
( printed page 94070)6. Administration of Lacrimal Ophthalmic Insert Into Lacrimal Canaliculus, CPT Code 68841(APC 5503)
Dextenza, which is described by HCPCS code J1096 (Dexamethasone, lacrimal ophthalmic insert, 0.1 mg), is a drug indicated for “the treatment of ocular inflammation and pain following ophthalmic surgery” and for “the treatment of ocular itching associated with allergic conjunctivitis.” [24] The drug manufacturer previously asserted that this drug is administered and described by CPT code 0356T (Insertion of drug-eluting implant (including punctal dilation and implant removal when performed) into lacrimal canaliculus, each). Interested parties also previously stated that Dextenza is inserted in a natural opening in the eyelid (called the punctum) and that the drug is designed to deliver a tapered dose of dexamethasone to the ocular surface for up to 30 days. CPT code 0356T was deleted December 31, 2021, and replaced with CPT code 68841 (Insertion of drug-eluting implant, including punctal dilation when performed, into lacrimal canaliculus, each), effective January 1, 2022.
Interested parties currently assert that the drug, Dextenza, is administered and described by CPT code 68841. We refer readers to the CY 2023 OPPS/ASC final rule with comment period for a detailed history of CMS payment assignments for CPT code 0356T and CPT code 68841 (87 FR 71840). See section XIII.F. of this final rule with comment period for the CY 2025 payment status of the drug, Dextenza, which may be described by HCPCS code J1096.
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81651 through 81653), we stated that based on the claims data available at the time, we continued to believe that the assignment of CPT code 68841 to APC 5503 (Level 3 Extraocular, Repair, and Plastic Eye Procedures) was appropriate based on the geometric mean costs. We also reiterated our reasoning that CPT code 68841 was appropriately assigned to an OPPS status indicator of “Q1” and ASC payment indicator of “N1.” We continued to believe that CPT code 68841 is mostly performed during ophthalmic surgeries, such as cataract surgeries. A status indicator “Q1,” indicating a conditionally packaged procedure, describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indictor “N1”) under the ASC payment system. Although stakeholders stated this is an independent surgical procedure and should not be packaged into the primary ophthalmic procedure in which the drug and drug administration are associated, we did not agree based on observed clinical patterns of how the drug is used. Based on CY 2024 claims data, out of over 7,000 total frequency claims, CPT code 68841 was used independently only about 2 percent of the time, meaning that the other 98 percent of the time CPT code 68841 had its payment packaged into the primary procedure with which it is associated.
These data reinforced our belief that Dextenza and CPT code 68841 are not furnished independently of a surgical procedure and should be packaged into the primary ophthalmic procedure with which the drug and drug administration are associated. While we recognized that there are some claims that may only include CPT code 68841 without a primary ophthalmic surgery on the claim, we did not believe that this is a frequent occurrence based on our claims data and clinical use patterns; as previously mentioned, our claims data showed that only 2 percent of claims are performed independently of another primary procedure.
For CY 2025, we proposed to continue to assign CPT code 68841 to APC 5503 (Level 3 Extraocular, Repair, and Plastic Eye Procedures). We also proposed to continue to assign CPT code 68841 to OPPS status indicator “Q1” and an ASC payment indicator of “N1.” The issue of payment for CPT code 68841 was brought to the Advisory Panel on Hospital Outpatient Payment (also known as HOP Panel) in 2024 for CY 2025 rulemaking. At the August 2024 meeting, based on the information presented, the Panel made no recommendation to CMS regarding changing the proposed status indicator assignment, Q1, for HCPCS code 68841 for CY 2025.
Comment: We received input from several interested parties contending that increased and separate payment for CPT code 68841, the code that can be used to describe the administration of the drug Dextenza, is required to ensure continued beneficiary access to Dextenza (HCPCS code J1096) in both the HOPD and ASC settings. Commenters emphasized that the lack of payment disproportionately and negatively affects the ASC setting. Several commenters pointed to the clinical importance of providing Dextenza to patients, noting that it reduces ocular pain and inflammation and reduces the burden of topical eyedrop application. Additionally, commenters stated that they usually perform the procedure to administer Dextenza in conjunction with ophthalmic surgeries.
Commenters generally did not disagree with the proposed APC assignment, but rather recommended CMS pay separately for the procedure. ( printed page 94071) Many of these commenters recommended CMS assign CPT code 68841 to a status indicator of “J1,” indicating a comprehensive APC procedure. Commenters believed that “J1” would be the most accurate status indicator assignment and would generate consistency within APC 5503 (Level 3 Extraocular, Repair, and Plastic Eye Procedures), as all other codes in APC 5503 are assigned to status indicator “J1.” Commenters stated that CPT code 68841 required an additional 5 minutes of operating time and the assistance of an ophthalmic technician, and without payment for those costs, it was difficult to provide the drug to Medicare beneficiaries.
Commenters did not agree with CMS that the fact that CPT code 68841 was performed as a standalone procedure only two percent of the time was an adequate justification for assigning a “Q1” status indicator. They believed, and provided examples, that there are many other procedures that are performed independently at that frequency or a lower percentage of the time, that are assigned to a payable status indicator for hospital outpatient purposes and thus also are paid separately in the ASC setting. For example, commenters mentioned CPT code 64415 (Injection(s), anesthetic agent(s) and/or steroid; brachial plexus, including imaging guidance when performed) as an example of a procedure with a lower percentage of single frequency claims and that is assigned to a payable status indicator; a status indicator of “T” in this example. Commenters also believed that CPT code 68841 was similar to other procedures for introducing a product to the eye, such as CPT code 66020 (Injection, anterior chamber of eye (separate procedure); air or liquid) and should be paid in a similar manner. Commenters believed the procedure described by CPT code 68841 is a distinct surgical procedure that requires additional operating room time and resources. These commenters believed that the cataract surgery is conducted and concluded, and then the procedure described by CPT code 68841 begins.
Response: We thank commenters for their feedback. We have long maintained that Dextenza is a drug that functions as a surgical supply and should be packaged under our packaging policy at 42 CFR 419.2(b), which lists the types of items and services for which payment is packaged under the OPPS. Specifically, § 419.2(b)(16) includes drugs and biologicals that function as supplies when used in a surgical procedure as packaged costs. Historically, we have stated that we consider all items related to the surgical outcome and provided during the hospital stay in which the surgery is performed, including postsurgical pain management drugs, to be part of the surgery for purposes of our drug and biological surgical supply packaging policy (79 FR 66875). As such, the drug administration procedure, CPT code 68841, is also supporting the main ocular procedure being performed. CPT code 68841 should, therefore, be packaged as an intraoperative service under § 419.2(b)(14).
Based on the rationale provided in the CY 2024 OPPS/ASC final rule with comment period, (88 FR 81651 through 81653), the procedure described by CPT code 68841 had its payment packaged into another procedure 98 percent of the time, meaning that it is not an independent surgical procedure as commenters assert. Rather, CPT code 68841 is used to support the primary procedure being performed and is a supportive intraoperative service that is a part of that primary procedure. We reiterate that both Dextenza and its administration impact the surgical outcome of the primary procedure and are provided during the hospital stay in which the surgery is performed, which we consider to be part of the surgery for purposes of our drug and biological surgical supply packaging policy. Although interested parties state this is an independent surgical procedure and should not be packaged into the primary ophthalmic procedure in which the drug and drug administration are associated, based on expected and observed clinical patterns as to how the drug is used, we do not agree.
When revaluating the claims data available for CY 2025, CPT code 68841 has 94 single frequency claims in the OPPS out of over 4200 total frequency claims, meaning that 98 percent of the time CPT code 68841 was furnished with another primary procedure into which CPT code 68841's payment was packaged. This confirms our belief that CPT code 68841 is a component of another primary procedure, supportive of that procedure, and should have its payment packaged into that procedure.
Additionally, as commenters were most concerned about a financial disincentive, we note that utilization has been increasing year over year based on utilization patterns of Dextenza in the ASC setting. For example, from claims year CY 2022 to claims year CY 2023, the number of units of Dextenza billed increased from approximately 260,000 to approximately 320,000 units, which reflects a nearly 20 percent year over year increase in utilization. Therefore, we do not believe that our packaging policies are hindering appropriate utilization as suggested by commenters. We note that HCPCS code J1096, which may be used to describe the drug, Dextenza, is a qualifying product for separate payment in both the OPPS and ASC under our policy to implement section 4135 of the CAA, 2023. Please see section XIII.F. of this final rule with comment period, for additional information.
For the reasons discussed, we continue to believe that it is appropriate to assign CPT code 68841 to a status indicator “Q1,” indicating a conditionally packaged procedure, which describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indictor “N1”) under the ASC payment system. We do not believe the HCPCS codes suggested by commenters that are not conditionally packaged are analogous to CPT code 68841. Therefore, our analysis was based on claims data specific to CPT code 68841 in order to confirm the appropriateness of the assignment to an OPP SI = “Q1” and ASC PI = “N1.”
After consideration of the public comments, we are finalizing our proposal, without modification, to assign CPT code 68841 to APC 5503 with OPPS status indicator “Q1” (STV Packaged Codes) for CY 2025, which typically means there will be a packaged APC payment if this code is billed on the same claims as a HCPCS code assigned to status indictor “S,” “T,” or “V” (Clinic or Emergency Department Visit). In addition, based on the OPPS assignments, we are finalizing an ASC payment indicator of “N1” (Packaged service/item; no separate payment made) for CPT code 68841 for CY 2025.
For the final CY 2025 OPPS payment rates, we refer readers to OPPS Addendum B to this final rule with comment period. In addition, we refer readers to OPPS Addendum D1 to this final rule with comment period for the status indicator definitions for all codes reported under the OPPS. For the final CY 2025 ASC payment rates and payment indicators, we refer readers to Addendum AA and Addendum BB for the ASC payment rates, and Addendum DD1 for the ASC payment indicator and their definitions. The OPPS Addenda B and D1 and ASC Addenda AA, BB, and ( printed page 94072) DD1 are available via the internet on the CMS website.
Please refer to Table 75 for the code descriptor, APC assignment, status indicator assignment, and payment indicator assignment for CPT code 68841 for CY 2025.
7. Application of Rigid Total Contact Leg Cast, CPT Code 29445 (APC 5102)
CPT code 29445 describes the procedure of applying a rigid leg cast. This code specifically refers to the process of immobilizing a leg fracture or injury by encasing the affected area in a hard cast, which helps to stabilize the bone and promote healing.
In the CY 2025 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 29445 to APC 5102 (Level 2 Strapping and Cast Application) with status indicator “T” (Procedure or service, multiple procedure reduction applies; with a separate APC payment under OPPS) and proposed payment rate of $266.86.
Payment for CPT code 29445 was brought to the Advisory Panel on Hospital Outpatient Payment (also known as HOP Panel) at the August 2024 meeting. Based on the information presented, the HOP Panel recommended that “CMS consider HCPCS code 29445, (Application of rigid total contact leg cast), a separately payable code when performed concurrently on the same date of service as any of the following: HCPCS codes 11402, 11403,11404, 11405, 11406, 11407, 97597, 97598, 15271, 15272, 15273, 15274, 15275, 15276, 15277, 15278.”
We received two public comments endorsing the recommendation of the HOP Panel.
Comment: One commenter requested that CMS ensure that hospital outpatient departments are able to receive separate payment for the CPT code 29445 when a debridement or graft is performed on the same date of service, which they are currently are unable to do because of a National Correct Coding Initiative (NCCI) edit that has been put in place.
Another commenter stated that if a wound debridement is performed (codes 11042-11047, 97597-97598), any primary or secondary dressing materials used to cover the wound would be included in the debridement and would not be separately reported. However, a TCC is not considered a wound dressing and is not included in the debridement procedure. Therefore, the commenter believed the cast application should be reported in addition to the code for the appropriate level of debridement, if performed, and should be mapped to APC 5102.
Response: We thank the commenters for providing this information related to CPT code 29445. We will take commenters' suggestions into consideration for the future rulemaking.
8. Aquabeam Waterjet Ablation Procedure, CPT Code 0421T (APC 5376)
CPT code 0421T (Transurethral waterjet ablation of prostate, including control of post-operative bleeding, including ultrasound guidance, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy are included when performed) describes the Aquabeam waterjet ablation procedure. According to the manufacturer, Aquabeam is for treating lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) by using a high-velocity water stream to ablate and remove tissue from enlarged prostates.
For the CY 2025 proposed rule, we estimated the geometric mean cost for CPT code 0421T to be $10,438 based on 4,743 single claims and proposed to assign the service to APC 5376 (Level 6 Urology and Related Services), which has a geometric mean cost of $9,356.
Comment: One commenter supported our proposal to assign CPT code 0421T to APC 5376 due to the clinical and resource similarities to other procedures assigned to APC 5376.
Response: We appreciate the commenter's feedback on CPT code 0421T and we agree with the commenter's recommendation to finalize the APC assignment.
After consideration of the public comment we received, we are finalizing our proposal without modification to assign CPT code 0421T to APC 5376. Please refer to Table 76 below for the final OPPS APC and status indicator assignment for HCPCS code 0421T for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94073)9. Aqueous Shunt to Extraocular Plate Reservoir Procedure, CPT Code 66180 (APC 5493)
For CY 2024, we assigned CPT code 66180 (Aqueous shunt to extraocular equatorial plate reservoir, external approach; with graft) to APC 5492 (Level 2 Intraocular Procedures). For CY 2025, as shown in OPPS Addendum B that was released with the CY 2025 OPPS/ASC proposed rule with comment period, we proposed to maintain the APC assignment to APC 5492 with a payment rate of $3,873.90 for CPT code 66180.
Comment: We received two comments requesting that we reassign CPT code 66180 to APC 5493 (Level 3 Intraocular Procedures) due to the code's similarity to CPT code 66179 (Aqueous shunt to extraocular equatorial plate reservoir, external approach; without graft), which we proposed to assign to APC 5493 for CY 2025. The commenters explained that the procedures are the same except that CPT code 66180 requires additional clinical work, time, and resources, associated with suturing a patch graft to the eye. The commenters also explained that the available claims data from the CY 2025 OPPS/ASC proposed rule support assigning CPT code 66180 to APC 5493. For example, per the commenters, the geometric mean cost for CPT code 66180 ($5,026.75) is closer to the geometric mean cost of APC 5493 ($5,298.63) than the geometric mean cost of APC 5492 ($4,159.32).
Response: We agree with the commenters that APC 5493 is a more appropriate placement for CPT code 66180 due to the additional clinical work and resources utilized to furnish the service due to its similarity to CPT code 66179. We also agree with the commenters that the geometric mean cost of CPT code 66180 confirms that the resources necessary to furnish the service more closely match the geometric mean cost of APC 5493 than APC 5492.
After consideration of the public comments received, we are finalizing an APC assignment for CPT code 66180 to APC 5493 for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
10. Arteriovenous Fistula (AVF) Creation Procedures, CPT Codes 36836 and 36837 (APC 5194)
CPT codes 36836 (Percutaneous arteriovenous fistula creation, upper extremity, single access of both the peripheral artery and peripheral vein, including fistula maturation procedures ( e.g., transluminal balloon angioplasty, coil embolization) when performed, including all vascular access, imaging guidance and radiologic supervision and interpretation) and 36837 (Percutaneous arteriovenous fistula creation, upper extremity, separate access sites of the peripheral artery and peripheral vein, including fistula maturation procedures ( e.g., transluminal balloon angioplasty, coil embolization) when performed, including all vascular access, imaging guidance and radiologic supervision and interpretation) describe a percutaneous arteriovenous fistula creation of an upper extremity. CPT code 36836 replaced HCPCS codes C9754 and G2170, while CPT code 36837 replaced HCPCS codes C9755 and G2171.
In the CY 2020 OPPS/ASC final rule with comment period (84 FR 61246 through 61247), in response to public comment, input from our medical advisors, and the available claims data, we reassigned C9754 and C9755 from APC 5193 to APC 5194. In the CY 2021 OPPS/ASC final rule with comment period (85 FR 85954 through 95955), we used our equitable adjustment authority to maintain the assignment of HCPCS codes G2170 and G2171 to APC 5194 (Level 4 Endovascular Procedures), given that both procedures are for ESRD patients that need dialysis, the predecessor codes had very similar median costs, and there were low claims data available. We continued the assignment of CPT codes 36836 and 36837 to APC 5194, with commenter support, for CY 2022.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71863 through 71864), we assigned the newly established [25] CPT codes 36836 and 36837 to APC 5194 based on our assessment of the CY 2023 geometric mean cost of predecessor codes HCPCS codes G2170 ($12,055.90) and G2171 ($13,486.08) and their APC assignment. For CY 2024, we continued assignment to APC 5194 for HCPCS codes G2170 and G2171, with commenter support.
For CY 2025, we proposed to continue assignment of both CPT codes 36836 and 36837 to APC 5194 (Level 4 Endovascular) with a proposed payment rate of around $17,956. For the CY 2025 proposed rule, CPT code 36836 had227 single claims for ratesetting, a geometric mean cost (GMC) of around $11,234, and CPT code 36837 had 121 single claims for ratesetting and a GMC of around $16,770.
Comment: A few commenters supported our assignment of CPT code 36837 to APC 5194.
Response: We thank the commenters for their support.
Comment: Several commenters requested that we avoid a proposed payment reduction for CPT code 36837 by maintaining its CY 2024 APC assignment.
Response: We note that for CY 2025 we proposed to maintain the assignment ( printed page 94074) of CPT code 36837 to APC 5194 and did not propose to reassign CPT code 36837 to a lower-paying APC.
Comment: Several commenters indicated that CPT codes 36836 and 36837 do not have similar resource costs and that CPT code 36836 would be more appropriately assigned to APC 5193, which had a proposed payment rate of $11,478.26, as opposed to continuing to assign the code to APC 5194, which had a proposed payment rate of $18,167.36. A commenter further suggested that the volume of current claims data is sufficient to support reassigning CPT code 36836 to APC 5193. The commenter believed that the proposed rule geometric mean cost data illustrated distinct differences in required resources to perform the procedures described CPT code 36836 (GMC: $10,123.79) and CPT code 36837 (GMC: $16,849.60) and that CPT code 36836 is significantly overpaid in APC 5194 and would be overpaid in APC 5193, but to a lesser degree.
Response: We thank the commenters for their comments. We reiterate that we review, on an annual basis, the APC assignments for all services and items paid under the OPPS based on our analysis of the latest claims data and review of the clinical characteristics of the procedure. After further clinical review, we continue to believe that CPT code 36836 is more similar clinically to services in APC 5194, which includes more complex endovascular procedures, than those in APC 5193, which primarily contains less complex revascularization and embolization procedures. Further, while CPT code 36836 does have more available claims than what was available for the initial assignment of CPT code 36836 to APC 5194, we believe additional claims and clinical data would be useful before proposing to move this code to a different APC.
However, we note that we review the APC assignments for all items and services paid under the OPPS on an annual basis. We will reevaluate the APC assignment for CPT code 36836 in the next rulemaking cycle.
In summary, after consideration of the public comments we received, we are finalizing our proposal, without modification, to continue to assign CPT code 36836 and CPT code 36837 to APC 5194 for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rate for all codes paid under the OPPS.
11. Arthroscopic Subacromial Balloon Implant, HCPCS Code C9781 (APC 5115)
HCPCS code C9781 describes the implantation of a subacromial spacer, such as a balloon, in the shoulder to treat a torn rotator cuff. The procedure may also include debridement, acromioplasty, subacromial decompression, and biceps tenodesis.
In the CY 2025 OPPS/ASC proposed rule, we proposed to continue to assign HCPCS code C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer ( e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed) to APC 5115 (Level 5 Musculoskeletal Procedures) with a proposed payment rate of $12,755.58.
We received two comments in support of our proposal.
Comment: Both commenters endorsed our proposal to assign HCPCS code C9781, which describes InSpaceTM Subacromial Tissue Spacer System, to APC 5115 for CY 2025. They encouraged CMS to maintain this assignment in the final rule.
Response: Based on our analysis of the latest CY 2023 claims data available for CY 2025 OPPS ratesetting, the geometric mean cost associated with HCPCS code C9781 is $12,757.71based on 493 single claims, which is consistent with the geometric mean cost of $13,022.88for APC 5115.
In summary, after consideration of the public comments we received, we are finalizing our proposal to assign HCPCS code C9781 to APC 5115 without modification. The final CY 2025 OPPS payment rate for this code can be found in Addendum B to this final rule with comment period.
12. Artificial Iris Insertion Procedures, CPT Code 66683 (APC 5496)
For the CY 2025 OPPS proposed rule, we proposed to assign placeholder CPT code 6X004 (Implantation of iris prosthesis, including suture fixation and repair or removal of iris, when performed) to APC 5496 (Level 6 Intraocular Procedures) with a payment rate of around $16,416. CPT code 66683 is the final code for 6X004 and will replace CPT code 0616T (Insertion of iris prosthesis, including suture fixation and repair or removal of iris, when performed; without removal of crystalline lens or intraocular lens, without insertion of intraocular lens); CPT code 0617T (Insertion of iris prosthesis, including suture fixation and repair or removal of iris, when performed; with removal of crystalline lens and insertion of intraocular lens); and CPT code 0618T (Insertion of iris prosthesis, including suture fixation and repair or removal of iris, when performed; with secondary intraocular lens placement or intraocular lens exchange).
Comment: One commenter supported our proposal to assign placeholder CPT code 6X004 (CPT code 66683) to APC 5496.
Response: We appreciate the support of the commenter for our proposal.
After consideration of the public comments we received, we are implementing our proposal without modification to assign CPT code 66683 to APC 5496 (Level 6 Intraocular Procedures). Table 77 shows the finalized status indicator and APC assignment for CPT code 66683. We refer readers to Addendum B of this final rule for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94075)13. Bronchoscopy With Needle Biopsy, CPT Code 31629 (APC 5154)
CPT code 31629 describes the use of diagnostic rigid or flexible bronchoscope to obtain one or more biopsies from the trachea, main stem, and/or lobar bronchus using a transbronchial needle aspiration technique. In the CY 2025 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 31629 to APC 5154 (Level 4 Airway Endoscopy) with a proposed payment rate of $3,681.38.
Comment: We received one public comment that requested CMS reassign CPT code 31629 from APC 5154 (Level 4 Airway Endoscopy) to APC 5155 (Level 5 Airway Endoscopy) in the CY 2025 ASC/OPPS final rule. The commenter stated that CPT code 31629 is the highest cost significant procedure in APC 5154 with a geometric mean cost of $5,123.33. The geometric mean cost of CPT code 31629 exceeds the geometric mean cost of the lowest cost significant procedure in APC 5155, CPT code 31267 with a geometric mean cost of $5,031.74.
According to the commenter, given the intraoperative service time and physician skill level required for the procedure, this procedure is aligned with procedures already assigned to APC 5155 including balloon dilation and stenting procedures. The commenter stated that the reassignment of CPT code 31629 from APC 5154 to APC 5155 would better support resource homogeneity within the Airway Endoscopy Procedures APC series.
Response: Although the geometric mean cost of the CPT code 31629 is $1,300 higher than the geometric mean cost of APC 5154, it is $1,974 lower than the geometric mean cost of APC 5155. CPT code 31629 also does not violate the 2 times rule in its current assignment in APC 5154. In addition, we believe that CPT code 31629 fits appropriately in APC 5154 based on clinical similarity and resource homogeneity with the procedures in that APC.
Therefore, after consideration of the public comment we received, we are finalizing our proposal without modification, to assign CPT code 31629 to APC 5154 for CY 2025. We refer readers to Addendum B of this final rule with comment period for the CY 2025 final payment rate for this code. Addendum B is available via the internet on the CMS website.
14. CADScor System, CPT Code 0716T (APC 5733)
The CADScor System is intended as a diagnostic aid in symptomatic patients suspected of stable coronary artery disease (CAD) and is designed to help reduce invasive and costly diagnostic procedures. The CADScor System is a diagnostic aid that uses sensitive acoustics and advanced computational processing to analyze coronary blood flow to rule out obstructive CAD. CPT code 0716T describes the service that utilizes the CADScor System: Cardiac acoustic waveform recording with automated analysis and generation of coronary artery disease risk score. For CY 2025, we proposed to assign CPT 0716T to APC 5733—Level 3 Minor Procedures, with a payment rate of $59.07 and a status indicator of “Q1” (conditionally packaged).
Comment: Commenters disagreed with our proposal to assign CPT code 0716T to APC 5733 with a status indicator of “Q1.” Commenters suggested that CPT code 0716T should be assigned to APC 5722 based on clinical and resource homogeneity with other services assigned to APC 5722. Commenters also expressed that status indicator “S” (separately payable) would be most appropriate for CPT code 0716T. They contended that status indicator “Q1” essentially unconditionally packages the code because CPT code 0716T is never furnished without an emergency department visit.
Response: Although CPT code 0716T was effective July 1, 2022, we have no claims data for the code. The OPPS payment rates are proposed based on available CY 2023 claims data. Because we have no claims data, we believe that we should continue to assign CPT code 0716T to APC 5733 for CY 2024. We review, on an annual basis, the APC assignments for all services and items paid under the OPPS. As a result, we will reevaluate the APC assignment for CPT code 0716T for the next rulemaking cycle.
In addition, as listed in OPPS Addendum D1 of the CY 2025 OPPS/ASC proposed rule, codes assigned to status indicator “Q1” may be packaged, assigned to a composite APC, or paid separately under the OPPS. Specifically, a “Q1” status indicator may indicate a:
- Packaged APC payment if billed on the same claim as a HCPCS code assigned status indicator “S”, “T”, or “V”; or
- Composite APC payment if billed with specific combinations of services based on OPPS composite-specific payment criteria. Payment is packaged into a single payment for specific combinations of services; or
- In other circumstances, payment is made through a separate APC payment
After reviewing the public comments, we believe that it is appropriate for the procedure described by CPT code 0716T to be paid separately. We agree with commenters that it is unlikely that CPT code 0716T would be furnished without an emergency department (ED) visit and should not be always be packaged with an ED visit. Therefore, we are revising the status indicator for the code from “Q1” (conditionally packaged) to “S” (Procedure or Service, Not Discounted When Multiple) to indicate that the service is paid separately.
In summary, after consideration of the public comments, we are finalizing the APC and status indicator assignment for ( printed page 94076) CPT code 0716T with modification. Specifically, we are finalizing the assignment of CPT code 0716T to APC 5733 and we are revising the status indicator assignment for CPT code 0716T to “S” (separately payable). Please refer to Table 78 for the code descriptor, APC assignment, status indicator assignment, and payment indicator assignment for CPT code 0716T for CY 2025.
15. Cardiac Magnetic Resonance Imaging Procedures, CPT Codes 75561 and 75563 (APCs 5572 and 5573)
CPT code 75561 (Cardiac magnetic resonance imaging for morphology and function without contrast material(s), followed by contrast material(s) and further sequences) and CPT code 75563 (Cardiac magnetic resonance imaging for morphology and function without contrast material(s), followed by contrast material(s) and further sequences; with stress imaging) both describe cardiac MRI procedures.
For CY 2024, we assigned CPT code 75561 to APC 5572 (Level 2 Imaging with Contrast), and CPT code 75563 to APC 5573 (Level 3 Imaging with Contrast). For the CY 2025 OPPS/ASC proposed rule, we proposed to maintain the current APC assignments for both cardiac MRI codes.
Comment: We received one comment requesting that we reassign CPT code 75561 to APC 5573 (Level 3 Imaging with Contrast). The commenter indicated that the other services in APC 5572 (Level 2 Imaging with Contrast) are not clinically similar or similar in resource use to the service described by CPT code 75561.
Response: We thank the commenter for their input. The OPPS payment rates are proposed based on available CY 2023 claims data. Our claims data show a geometric mean cost of about $450 for CPT code 75561 based on 27,543 single claims. We believe that the geometric mean cost of about $450 for CPT code 75561 is more consistent with the geometric mean cost of approximately $361 for APC 5572, than the geometric mean cost of approximately $800 for APC 5573. We continue to believe that the clinical and resource characteristics of CPT code 75561 are sufficiently like other procedures assigned to APC 5572 and should continue to be assigned to APC 5572.
Comment: The same commenter requested that CPT code 75563 (Cardiac magnetic resonance imaging for morphology and function without contrast material(s), followed by contrast material(s) and further sequences; with stress imaging) be reassigned to APC 5593 (Level 3 Nuclear Medicine and Related Services) because of lack of clinical homogeneity and resource utilization with other services currently assigned to APC 5573. The commenter notes that CPT code 75563 was previously assigned to the Nuclear Medicine APC family until the imaging services were restructured during a comprehensive review of the APC series that took place in CY 2016. The purpose of the restructuring of the OPPS APC groupings for imaging services was to improve the clinical and resource homogeneity of the services classified within the imaging APCs (80 FR 70392 and 81 FR 7928).
The commenter cites the following reasons to support the reassignment of 75563 to APC 5593:
(1) the clinical labor and resources (acquisition time, staffing, medications, and perfusion agents) are very similar to CPT code 78452 which describes myocardial perfusion imaging;
(2) both tests are indicated for patients with cardiac angina or coronary artery disease; and
(3) both tests provide the same clinical information.
We disagreed with a similar argument in the past (81 FR 79630) where commenters asserted that cardiac MRI with stress imaging is very similar to myocardial perfusion imaging because both tests are performed under a stress protocol and therefore should be assigned to APC 5593. APC 5593 contains procedures that describe nuclear medicine tests, and CPT code 75563 is a specific type of MRI and not a nuclear medicine test. Additionally, the geometric mean cost of CPT code 75563 is approximately $887 and the geometric mean cost of the APC to which it is assigned, APC 5573, is approximately $800. These geometric mean costs are very similar. However, the geometric mean cost of APC 5593 is approximately $1,321, which is significantly higher than the geometric mean cost of CPT code 75563. Therefore, assigning CPT code 75563 to APC 5593 would assign the procedure to an APC with clinically dissimilar nuclear medicine tests that have a geometric mean cost of $1,321 (as compared to the $887 geometric mean cost of CPT code 75563).
In summary, after consideration of the public comment, we are finalizing our proposal without modification to assign CPT code 75561 to APC 5572 and CPT code 75563 to APC 5573 for CY 2025. The final CY 2025 payment rates for the codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website. ( printed page 94077)
16. CardioMEMS, HCPCS Code GMEM1, CPT Code 93264 (APCs 5724 and 5741)
The CardioMEMS Heart Failure System allows practitioners treating heart failure patients to wirelessly monitor and measure pulmonary artery pressure and heart rate in patients with heart failure and transmit the information to the physician to inform the treatment plan for the patient. The system includes two critical components: first, a miniaturized, wireless monitor, which is implanted into a patient's pulmonary artery, and second, a smart pillow (the CardioMEMS Patient Electronics System), which captures and transmits readings via safe radio frequency from the patient's implanted CardioMEMS Heart Failure System. The CardioMEMS Heart Failure System enables patients to transmit critical heart failure status information to clinicians regularly, potentially eliminating the need for frequent clinic or hospital visits. The CardioMEMS Heart Failure System is described by CPT code 33289 (Transcatheter implantation of wireless pulmonary artery pressure sensor for long-term hemodynamic monitoring, including deployment and calibration of the sensor, right heart catheterization, selective pulmonary catheterization, radiological supervision and interpretation, and pulmonary artery angiography, when performed).
Interested parties have met with CMS and highlighted the critical importance of the device for heart failure patients who require close monitoring of weight and blood pressure to prevent fluid buildup around the heart and have requested that CMS establish coding to describe when practitioners and hospitals incur costs during clinical scenarios when crucial components of the system require replacement. Given that these components are crucial for system functionality and there is no existing coding framework to address their replacement, we believe that establishing appropriate coding and payment mechanisms can facilitate the provision of these services more effectively in the office and hospital settings. In the CY 2025 Physician Fee Schedule (PFS) proposed rule, we proposed a new code, HCPCS code GMEM1 (Provision of replacement patient electronics system (for example, system pillow) for home pulmonary artery pressure monitoring including provision of materials for use in the home and reporting of test results to physician or qualified health care professional). The CY 2025 PFS proposed rule (89 FR 61956) sought comment from interested parties regarding direct costs from invoices for the replacement component referenced above, utilization estimates, and potential indicators.
CPT Code 93264 (Remote monitoring of a wireless pulmonary artery pressure sensor for up to 30 days, including at least weekly downloads of pulmonary artery pressure recordings, interpretation(s), trend analysis, and report(s) by a physician or other qualified health care professional) can be used by hospitals to bill for the ongoing analysis and monitoring performed with CardioMEMS.
While we did not discuss the GMEM1 code in the CY 2025 OPPS proposed rule preamble it was included in Addendum B with an assignment to APC 5741 (Level 1 Electronic Analysis of Devices) with a payment rate of $36.90 and assigned a status indicator of Q1. CPT code 93264 was included in Addendum B with a status indicator of “M” to indicate that this code is not payable under the OPPS.
The issue of payment for GMEM1 was brought to the Advisory Panel on Hospital Outpatient Payment (also known as HOP Panel) in 2024 for CY 2025 rulemaking. At the August 2024 meeting, based on the information presented, the Panel did not make any recommendations on GMEM1.
We note that HCPCS code G0555 is replacing the placeholder code GMEM1.
Comment: In their comment letters on the CY 2025 OPPS proposed rule, commenters indicated that the G0555 (placeholder codeGMEM1) code descriptor proposed in the CY 2025 PFS proposed rule introduces confusion by also referencing reporting test results to the physician, which is a separate function furnished by a different party than the replacement Patient Electronics System (PES). Commenters requested that CMS revise the proposed code descriptor for G0555 (placeholder code GMEM1) to clarify that it is for the replacement PES only and remove the following language from the descriptor: “and reporting of test results to physician or qualified health care professional”). Commenters requested that if the code is finalized under OPPS (and PFS) with the revised descriptor, that CMS assign it to APC 1528, New Technology—Level 28 ($5001-$5500), which they believe is reflective of the cost of the replacement PES.
Response: We agree that assignment to APC 5741 is likely not reflective of the cost of the replacement PES. However, assignment to New Technology APCs usually occurs when there is no existing clinical APC that is appropriate in terms of clinical similarity or resources. Based on the nature of the procedure, we believe that the service associated with GMEM1 is more appropriately in APC 5724 (Level 4 Diagnostic Tests and Related Services). We believe that the service described by HCPCS code G0555 (placeholder code GMEM1) shares similar resource costs and clinical characteristics with services included in APC 5724. Assignment to APC 5741 may not have sufficiently covered the cost of the replacement of the PES. Therefore, for CY 2025, we are revising the assignment for HCPCS code G0555 (placeholder code GMEM1) to APC 5724, with a status indicator of “S”.
Comment: Commenters recommend that CMS revise the OPPS status indicator for CPT 93264, which CMS proposed to designate as “M” (not payable under the OPPS). They argued that this classification is contrary to CMS's treatment of similar monitoring procedures ( e.g., CPT codes 93297 and 93298), which have status indicator “Q1” and are assigned to APC 5741 (Level 1 Electronic Analysis of Devices).
Response: As listed in OPPS Addendum D1 of the CY 2025 OPPS/ASC proposed rule, codes assigned to status indicator “Q1” may be packaged, assigned to a composite APC, or paid separately under the OPPS. Specifically, a “Q1” status indicator may indicate a:
- Packaged APC payment if billed on the same claim as a HCPCS code assigned status indicator “S”, “T”, or “V”; or
- Composite APC payment if billed with specific combinations of services based on OPPS composite-specific payment criteria. Payment is packaged into a single payment for specific combinations of services; or
- In other circumstances, payment is made through a separate APC payment
After reviewing the public comments, we agree with commenters that it is appropriate for the procedure described by CPT code 93264 to be assigned to status indicator Q1(conditionally packaged). Therefore, we are revising the status indicator for the code from “M” (not payable under the OPPS”) to “Q1” (conditionally packaged). The conditional packaging of this code supports our overarching goal to make payments for all services paid under the OPPS and ASC payment system more consistent with those of a prospective payment system and less like those of a per-service fee schedule. We believe that packaging encourages efficiency and is an essential component of a prospective payment system, and that packaging payments for items and services that are typically integral, ancillary, supportive, dependent, or adjunctive to a primary service is a ( printed page 94078) fundamental part of the OPPS. We therefore believe packaging of CPT code 93264 is appropriate.
Because CPT code 93264 was assigned status indicator “M” (not payable under the OPPS) we have no claims data to use for making an APC assignment for CPT code 93264 in CY 2025. We agree with commenters that the code is similar in clinical and resource use to CPT codes 93297 and 93298. Therefore, we will be assigning CPT code 93264 to APC 5741 (Level 1 Electronic Analysis of Devices) for CY 2025.
In summary, after consideration of the public comment, we are finalizing the APC assignment for HCPCS code G0555 (placeholder code GMEM1) with modification. Specifically, we are revising the APC assignment for HCPCS code G0555 (placeholder code GMEM1) to APC 5724 and assigning the code to status indicator “S” for CY 2025. We are revising the status indicator for CPT 93264 and assigning a status indicator of “Q1” for CY 2025. Lastly, we are assigning CPT 93264 to APC 5741 for CY 2025. The final CY 2025 OPPS payment rate for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
We refer readers to Table 79 for the code descriptor, APC assignment, status indicator assignment, and payment indicator assignment for CPT codes G0555 (placeholder code GMEM1) and 93264 for CY 2025.
17. Caregiver Training Services, HCPCS Codes GCTD1, GCTD2, and GCTD3 (APC 5731)
The CY 2025 Physician Fee Schedule (PFS) proposed rule (89 FR 61956) proposed to establish new coding and payment for caregiver training for direct care services and supports. The topics of training could include, but would not be limited to, techniques to prevent decubitus ulcer formation, wound dressing changes, and infection control. Unlike other caregiver training codes that are currently paid under the PFS, the caregiver training codes for direct care services and supports focus on specific clinical skills aimed at the caregiver effectuating hands-on treatment, reducing complications, and monitoring the patient when the patient is not capable to do so themselves. Three new HCPCS codes were proposed: GCTD1 ( Caregiver training in direct care strategies and techniques to support care for patients with an ongoing condition or illness and to reduce complications (including, but not limited to, techniques to prevent decubitus ulcer formation, wound dressing changes, and infection control) (without the patient present), face-to-face; initial 30 minutes), GCTD2 ( Caregiver training in direct care strategies and techniques to support care for patients with an ongoing condition or illness and to reduce complications (including, but not limited to, techniques to prevent decubitus ulcer formation, wound dressing changes, and infection control) (without the patient present), face-to-face; each additional 15 minutes (List separately in addition to code for primary service) (Use GCTD2 in conjunction with GCTD1)), and GCTD3 ( Group caregiver training in direct care strategies and techniques to support care for patients with an ongoing condition or illness and to reduce complications (including, but not limited to, techniques to prevent ( printed page 94079) decubitus ulcer formation, wound dressing changes, and infection control) (without the patient present), face-to-face with multiple sets of caregivers) ). We did not propose APC or status indicator assignments for these G codes in the CY 2025 OPPS proposed rule (89 FR 59186).
We note that the HCPCS codes G0541, G0542, and G0543 are replacing their respective placeholder codes GCTD1, GCDT2, and GCTD3.
Comment: We received several comments related to the CY 2025 Physician Fee Schedule (PFS) proposal suggesting that we begin providing payment for Caregiver Training Services (CTS) G0541, G0542, and G0543placeholder codes GCTD1, GCTD2, and GCTD3) under the OPPS. Commenters asked for clarification on whether these codes would be given the OPPS status indicator “A,” which would limit their use for outpatient hospital payment purposes to therapists only, or whether CMS will assign separately payable status indicators to those codes. Commenters stated that assigning separately payable status would recognize the different types of hospital staff that can render appropriate CTS. Commenters requested that CMS make the newly proposed CTS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3) payable under the OPPS.
Response: HCPCS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3) will be effective on January 1, 2025. Based on our evaluation of these new codes, we are proposing an interim assignment of HCPCS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3) to status indicator “A” to indicate that these codes are payable under a fee schedule or payment system other than the OPPS. These services are covered Medicare services and will be assigned payable indicators under the Physician Fee Schedule (PFS). While these services are not payable under the OPPS, they are payable under the PFS; therefore, we are proposing to assign them status indicator of “A.” Sections 1834(k) and 1833(a)(8) of the Act require that payment be made for outpatient therapy services (physical therapy, speech language pathology, and occupational therapy) furnished by providers of therapy services, including a hospital (to its outpatients and inpatients not under a Part A stay), be made under the PFS.
In summary, we are proposing an interim assignment of HCPCS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3) to status indictor “A” to indicate that the code is payable under a fee schedule or payment system other than the OPPS. Because we were not able to propose APC and status indicator assignments in the CY 2025 OPPS proposed rule, we will be assigning HCPCS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3) comment indicator “NI” in Addendum B of this final rule. This comment indicator is for new HCPCS codes that will be effective January 1, 2025, to indicate that we are assigning them an interim status indicator and APC assignment, which is subject to public comment. We invite public comment on the interim proposed status indicators for HCPCS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3), which will then be finalized in the CY 2026 OPPS/ASC final rule with comment period.
Please refer to Table 80 below for the proposed interim status indicator assignments for HCPCS codes G0541, G0542, and G0543 (placeholder codes GCTD1, GCTD2, and GCTD3) for CY 2025.
( printed page 94080)18. Chimeric Antigen Receptor Therapy (CAR-T), CPT Codes 0537T, 0538T, 0539T, and 0540T (APC 5694)
Chimeric Antigen Receptor T-Cell (CAR T-cell) therapy is a cell-based gene therapy in which T-cells are collected and genetically engineered to express a chimeric antigen receptor that will bind to a certain protein on a patient's cancerous cells. The CAR T-cells are then administered to the patient to attack certain cancerous cells, and the individual is observed for potential serious side effects that would require medical intervention. We refer readers to previous discussions in the OPPS/ASC final rules with comment period for background regarding the specific CAR T-cell products, in both the CY 2020 OPPS/ASC final rule with comment period (84 FR 61231 through 61234) and the CY 2019 OPPS/ASC final rule with comment period (83 FR 58904 through 58908). In addition, for discussion about CY 2022 OPPS payment policies for separately paid drugs with pass-through status expiring or continuing in CY 2022, please see sections V.A.4 and V.A.5 of this final rule with comment period. The AMA created four Category III CPT codes that are related to CAR T-cell therapy, effective January 1, 2019. As discussed in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58904 through 58908), the CY 2020 OPPS/ASC final rule with comment period (84 FR 61231 through 61234), and the CY 2021 OPPS/ASC final rule with comment period (85 FR 85949 through 85951) we finalized our proposal to assign procedures described by CPT codes 0537T, 0538T, and 0539T to status indicator “B” (Codes that are not recognized by OPPS when submitted on an outpatient hospital Part B bill type (12x and 13x)) to indicate that the services are not paid under the OPPS. The procedures described by CPT codes 0537T, 0538T, and 0539T describe the various steps required to collect and prepare the genetically modified T-cells, and Medicare does not generally pay separately for each step used to ( printed page 94081) manufacture a drug or biological. We also finalized that the procedures described by CPT code 0540T would be assigned status indicator “S” (Procedure or Service, Not Discounted when Multiple) and APC 5694 (Level 4 Drug Administration) for CY 2019 through CY 2024 and made no proposal to change the assignment for CY 2025. Additionally, the National Uniform Billing Committee (NUBC) established CAR T-cell-related revenue codes and a value code to be reportable on Hospital Outpatient Department (HOPD) claims effective for claims received on or after April 1, 2019.
Category III CPT codes 0537T, 0538T, 0539T, and 0540T are being replaced by Category I CPT codes 38225, 38226, 38227, and 38228, which were previously represented by placeholder codes 3X018, 3X019, 3X020, and 3X021. As such, we made no specific proposal related to the CAR T-cell preparation codes but proposed to apply the existing APC and status indicator assignments for the predecessor Category III CPT codes to the replacement Category I CPT codes.
As listed in Addendum B of the CY 2025 OPPS/ASC proposed rule, we proposed to continue to assign procedures described by these CPT codes, 3X018, 3X019, and 3X020, to status indicator “B” (Codes that are not recognized by OPPS when submitted on an outpatient hospital Part B bill type (12x and 13x)) to indicate that the services are not paid under the OPPS. We proposed to continue to assign CPT code 3X021 to status indicator “S” (Procedure or Service, Not Discounted when Multiple) and APC 5694 (Level IV Drug Administration).
Comment: Some commenters provided input on the proposed policies in the physician office setting regarding the dose preparation procedures associated with CAR T-cell therapy and other billing topics in the office setting, such as the expansion of digits on MAC claims processing screens for physician office claims.
Response: Comments related to the CY 2025 PFS payment policies are out of scope.
Comment: Several commenters opposed our proposal to continue to assign status indicator “B” to CPT codes 38225, 38226, and 38227, for CY 2025. Many commenters provided alternate status indicator assignment recommendations, such as “S”, as well as APC assignment recommendations. Commenters stated that by allowing these codes to be billable and paid, CMS could use claims data collected to assign the codes to more accurate APCs. Some commenters explained that CMS's overall practice and billing process should be reevaluated, with some recommending revisions to, or the retirement of, MLN Matters Number SE19009 [26] which they believed caused confusion, raised numerous program integrity concerns, including confusion regarding what can be included in ASP reporting, increased inconsistencies with Medicare requirements for under arrangement services and with charges reported and hospital billing, and commenters believed the elimination of this guidance, including separate payment of 38225, 38226, and 38227, would reduce hospital burden. One commenter raised the concern that some manufacturers will pay hospitals for the dose preparation services, while other manufacturers will not. In their view, this raises potential program integrity concerns and could influence medical decision making.
Commenters stated that the cell collection required as part of the CAR T-cell therapy process is done by leukapheresis, a costly and labor-intensive procedure in commenters' view. Commenters also communicated that the hospital in which the preparation procedure occurs is not always the same facility in which the final infusion of the therapy occurs. In their view, this could disincentivize hospitals from providing the preparation procedures if they are not the hospital ultimately providing the final product. A few commenters emphasized that manufacturers do not provide these services, hospitals do. Commenters believed “under-arrangements” made between hospitals were infeasible long term given the likely growth in the volume of these therapies and the costs incurred. Commenters also discussed that the preparation procedures could occur months apart from the final administration of the product.
Many of these commenters also stated that 10-15 percent of the time a finalized CAR T-cell therapy product is never administered to a beneficiary, which could occur for a multitude of reasons, such as a manufacturing failure, or disease progression potentially resulting in death or ineligibility for treatment while the CAR T-cell therapy product is being manufactured.
Commenters also recommended CMS revise the product specific Q-codes to remove “leukapheresis and dose preparation procedures” and they believe it is inappropriate for these services to be described by the HCPCS Level II codes when they can be described by Level I HCPCS codes. Additional commenters recommended CMS transition the product specific Q-codes to permanent J-codes.
Several commenters discussed other therapies, such as stem cell transplant, and therapies such as those described by HCPCS codes J3394 (Injection, lovotibeglogene autotemcel, per treatment) and J3393 (Injection, betibeglogene autotemcel, per treatment), which in commenter's view is similar to CAR T-cell therapy and requires similar dose preparation procedures.
Commenters supported the 2024 Advisory Panel on Hospital Outpatient Payment recommendation to assign CPT code 38225/3X018, Chimeric antigen receptor T-cell therapy; harvesting of blood-derived T lymphocytes for development of genetically modified autologous CAR-T cells, per day, an SI of S and place the code in APC 5242, Level 2 Blood Product Exchange and Related Services. The Panel further recommended that CMS assign CPT code 38226/3X019, Chimeric antigen receptor T-cell therapy; preparation of blood-derived T lymphocytes for transportation (e.g., cryopreservation, storage); and CPT code 38227/3X020, Chimeric antigen receptor T-cell therapy; receipt and preparation of CAR-T cells for administration; an SI of S and place these codes in APC 5241, Level 1 Blood Product Exchange and Related Services.
Response: We thank the commenters for their feedback. CMS continues to believe that the procedures described by CPT codes 0537T, 0538T, and 0539T and replacement CPT codes 38225, 38226, and 38227, describe the various steps required to collect and prepare the genetically modified T-cells, and Medicare does not generally pay separately for each step used to manufacture a drug or biological product. Therefore, CMS does not believe that separate or packaged payment under the OPPS is necessary for the procedures described by CPT codes 38225, 38226, and 38227, for CY 2025. However, we thank commenters for providing their unique perspectives and experiences in situations where the manufacturing process does not result in a final product being administered to a beneficiary.
We note that the current HCPCS coding for the currently approved CAR T-cell therapies include leukapheresis and dose preparation procedures, as these services are included in the manufacturing of these biologicals. Therefore, payment for these services is ( printed page 94082) incorporated into the drug codes. CMS is not revising the descriptors for the Q-codes to remove leukapheresis and dose preparation procedures nor are we transitioning the Q-codes to J-codes at this time.
We note that, although there is no payment associated with CPT codes 38225, 38226, and 38227, for reasons stated previously, these codes can still be reported to CMS for tracking purposes. We thank commenters for their feedback and for raising concerns related to our guidance contained in MLN Matters Article SE19009. We are not revising this document at this time as we believe these instructions are consistent with our longstanding policies, but we understand the feedback provided. Accordingly, we are not accepting the recommendations at this time to revise the status indicators for procedures described by CPT codes 38225, 38226, and 38227. We will continue to evaluate and monitor payment for CAR T-cell therapies. In summary, after consideration of the public comments we received, we are finalizing our proposal to assign status indicator “B” to CPT codes 38225, 38226, and 38227 for CY 2025 without modification.
Additionally, we are continuing our policy from CY 2019 to assign status indicator “S” to CPT code 38228 for CY 2025. Table 81 shows the final SI and APC assignments for HCPCS codes 38225, 38226, 38227, and 38228 for CY 2025. For more information on CY 2025 OPPS final status indicators, APC assignments, and payment rates for HCPCS codes, including the CAR T-cell drug codes, we refer readers to Addendum B to this final rule with comment period. In addition, the status indicator definitions can be found in Addendum D1 (OPPS Payment Status Indicators for CY 2025) to this final rule with comment period. Both Addendum B and D1 are available via the internet on the CMS website.
19. Complex Bunion Correction Procedure CPT Code 28297 (APC 5115)
CPT code 28297 describes a surgical procedure to correct a bunion, or hallux valgus, in the foot. The procedure involves:
- Removing the bunion
- Realigning the big toe
- Fusing the joint between the first metatarsal bone and the medial cuneiform bone
- Removing the sesamoid bones, which are pea-shaped bones under the big toe joint
Correcting the bony prominence at the base of the toe ( printed page 94083)
In the CY 2025 OPPS/ASC proposed rule, we proposed to move CPT code 28297 (Correction, hallux valgus with bunionectomy, with sesamoidectomy when performed; with first metatarsal and medial cuneiform joint arthrodesis, any method) from APC 5114 (Level 4 Musculoskeletal Procedures) to APC 5115 (Level 5 Musculoskeletal Procedures) with the proposed payment rate of $12,755.58.
We received several comments supporting the proposed reassignment of CPT code 28297 from APC 5114 (Level 4 Musculoskeletal Procedures) to APC 5115 (Level 5 Musculoskeletal Procedures).
Comment: One commenter stated that CPT code 28297 clearly meets the criteria for reassignment. The total frequency of CPT code 28297 exceeded the significance threshold of 1,000 procedures. The geometric mean cost of CPT code 28297 exceeded two times the geometric mean cost of the lowest significant procedure in APC 5114 (Level 4 Musculoskeletal Procedures). Furthermore, the geometric mean cost of CPT code 28297 was nearly equal to the geometric mean cost of APC 5115 (Level 5 Musculoskeletal Procedures). Several other commenters also supported assigning CPT code 28297 to APC 5114 ((Level 5 Musculoskeletal Procedures).
Response: We agree with the commenters, and we thank them for their support. In summary, after consideration of the public comments that we received, we are finalizing our proposal without modification and are assigning CPT code 28297 to APC 5115 (Level 5 Musculoskeletal Procedures) for CY 2025. The final CY 2025 payment rate for this code can be found in Addendum B of this final rule with comment period.
20. Computational Electrocardiogram (ECG) Analysis System (vMap), CPT Code 0897T (APC 5724)
CPT code 0897T (Noninvasive augmentative arrhythmia analysis derived from quantitative computational cardiac arrhythmia simulations, based on selected intervals of interest from 12-lead electrocardiogram and uploaded clinical parameters, including uploading clinical parameters with interpretation and report) utilizes ECG data to identify potential arrhythmia focal points for patients. The vMap provides augmented information which enables physicians to characterize arrhythmia and assists in triage and treatment of abnormal rhythm. CPT code 0897T became effective July 1, 2024 and since its establishment, the code has been assigned to APC 5724 (Level 4 Diagnostic Tests and Related Services) for CY 2024. Because this is a new code for CY 2024, there are no claims available. For CY 2025, we proposed to maintain the assignment to APC 5724, with a payment rate of $1,012.27.
Comment: One commenter supported our proposal to assign CPT code 0897T to APC 5724, due to the resource similarity of this technology to other procedures assigned to APC 5724.
Response: We appreciate the commenter's feedback on CPT code 0897T and we agree with the commenter's recommendation to finalize the APC assignment.
After consideration of the public comment we received, we are finalizing our proposal without modification for CPT code 0897T. Please refer to Table 82 below for the final OPPS APC and status indicator assignment for HCPCS code 0897T for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
21. Computed Tomographic Colonography, CPT Code 74263 (APC 5523)
For CY 2024, we assigned CPT code 74263 (Computed tomographic (CT) colonography, screening, including image postprocessing) to status indicator “E1” indicating that the service was not covered and not payable by Medicare under the OPPS. For the CY 2025 OPPS/ASC proposed rule, we proposed to assign CPT code 74263 to APC 5522 (Level 2 Imaging without Contrast) and a status indicator of “S” (Procedure or Service, Not Discounted When Multiple. Paid under OPPS; separate APC payment) to indicate that the code is separately payable under OPPS based on the proposed coverage changes for the colorectal cancer screening. (We refer our readers to section X.E. of this final rule with comment period for a full discussion of the coverage changes for colorectal cancer screening services.)
For the CY 2025 OPPS/ASC proposed rule, we believed the time and resources associated with performing a screening virtual colonoscopy is similar to those necessary to furnish a diagnostic virtual colonoscopy, which is described by CPT code 74261 (Computed tomographic (ct) colonography, diagnostic, including image postprocessing; without contrast material). Consequently, we proposed to ( printed page 94084) assign CPT code 74263 to APC 5522 (Level 2 Imaging without Contrast) based on its clinical and resource homogeneity to CPT code 74261, which is assigned to the same clinical APC.
Comment: We received one comment requesting to reassign CPT code 74263 to APC 5571 (Level 1 Imaging with Contrast). The commenter is concerned that the proposed payment for the screening CT colonography is insufficient to cover the costs of providing the service. The commenter believed that other services within that APC family such as CT abdomen with contrast have greater clinical homogeneity with CPT code 74263.
Response: We thank the commenter for their input. We disagree with the commenter that CPT code 74263 should be placed in APC 5571 (Level 1 Imaging with Contrast) as it is a procedure that is completed without contrast. However, we do believe that this newly covered screening test for colorectal cancer should be assigned to an APC where payment is more comparable to the purported resource costs. Given that any claims data in CY 2023 would be from before the service was payable in the OPPS, it would be difficult to rely on the claims data to determine the APC assignment for this code. Based on our review of the service (CT colonography) compared to other services assigned to the Imaging without Contrast APC series, we believe CPT code 74176 (Computed tomography, abdomen and pelvis; without contrast material) is an appropriate crosswalk code for CPT code 74263.
In summary, after consideration of the public comment, we are finalizing our proposal with modification to assign CPT code 74263 to APC 5523 (Level 3 Imaging without Contrast) and a status indicator of “S” for CY 2025. The final CY 2025 payment with rates for this code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI definitions for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
22. Concurrent Optical and Magnetic Stimulation (COMS) Therapy, CPT Codes 0906T and 0907T (APC 5051)
On June 20, 2023, CMS approved for Medicare coverage the Category B Investigational Device Exemption (IDE) study associated with concurrent optical and magnetic stimulation (Study Title: [27] Concurrent Optical and Magnetic Stimulation (COMS) for Treatment of Refractory Diabetic Foot Ulcer; a Prospective Randomized, Sham-controlled, Double-blinded, Pivotal Clinical Trial; NCT number NCT05758545; IDE number G220277 [28] ). For the July 2024 update, the AMA CPT Editorial Panel established 0906T (placeholder code XX18T) (Concurrent optical and magnetic stimulation therapy, wound assessment and dressing care; first application, total wound(s) surface area less than or equal to 50 sq cm) and 0907T (placeholder code XX37T) (Concurrent optical and magnetic stimulation (COMS) therapy, wound assessment and dressing care; each additional application, total wound(s) surface area less than or equal to 50 sq cm (List separately in addition to code for primary procedure)) for COMS, effective January 1, 2025.
For the CY 2025 OPPS/ASC proposed rule, CMS proposed to assign the SIs and APCs listed in Table 83 below for the new codes based on the clinical characteristics of the procedures.
We note that at the August 28, 2024, HOP Panel Meeting, a presentation was made requesting the reassignment to APC 5053 for CPT code 0906T (placeholder code XX18T). Based on the information presented at the meeting, the Panel made no recommendation on the APC assignment for the code. ( printed page 94085)
Comment: We received a comment from the manufacturer of COMS on our proposed assignments for the codes listed in Table 83. The commenter stated that the COMS procedures include extensive wound examination, wound bed preparation, and dressing management. With the inclusion of standard wound care administered before and after each active COMS device treatment, the commenter estimates that the total time required for a COMS procedure is 35-50 minutes, during which considerable medical supplies and facility resources are utilized. The manufacturer suggested that the total resource code for CPT code 0906T is $627, with $392 in hospital facility resource costs and $235 specifically for the COMS device-associated costs. The commenter maintains that the assignment to APC 5051, which has a proposed CY 2025 payment rate of $201.14, does not cover the costs of the procedure.
The commenter claims that CPT code 0906T is sufficiently similar in terms of resources and time to CPT codes 11043 (Debridement, muscle and/or fascia (includes epidermis, dermis, and subcutaneous tissue, if performed); first 20 sq cm or less) and 12044 (Repair, intermediate, wounds of neck, hands, feet and/or external genitalia; 7.6 cm to 12.5 cm), both assigned to APC 5053 ((Level 3 Skin Procedures, proposed CY 2025 payment rate of $619.63), to warrant the reassignment of CPT code 0906T to APC 5053. The commenter further stated that assigning these procedures to APC 5053 would align with other wound care procedures with similar resource costs and procedural time.
Response: We thank the commenter for their input. We have no claims data for CPT code 0906T available for this final rule. However, based on this code's similarity to CPT code 0521T (Extracorporeal shock wave for integumentary wound healing, including topical application and dressing care; initial wound) and CPT code 97610 (Low frequency, non-contact, non-thermal ultrasound, including topical application(s), when performed, wound assessment, and instruction(s) for ongoing care, per day) which are both assigned to APC 5051, we continue to believe CPT code 0906T has clinical and resource similarities to procedures assigned to APC 5051. Therefore, for CY 2025, we are finalizing our proposal without modification and assigning CPT code 0906T to APC 5051. Table 84 lists the final CY 2025 OPPS payment rate for 0906T and 0907T.
In summary, after consideration of the public comment that we received, we are finalizing the APC and SI assignments for CPT codes 0906T to APC 5051. As we do every year, we will reevaluate the APC assignments for these codes in the next rulemaking cycle. We remind hospitals that we review, on an annual basis, the APC assignments for all items and services paid under the OPPS. The final payment rates for the codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Both Addendum B and D1 are available via the internet on the CMS website.
( printed page 94086)23. Cystourethroscopy With Temporarily Implanted Nitinol Device Procedure, HCPCS Code C9769 (APC 5376)
Cystourethroscopy temporarily implanted nitinol device (iTIND) is a minimally invasive surgery to treat bothersome lower urinary tract symptoms (LUTS) caused by benign prostate enlargement. The iTIND serves to re-model the bladder neck and the prostatic urethra. The HCPCS Code C9769 (Cystourethroscopy, with insertion of temporary prostatic implant/stent with fixation/anchor and incisional struts) became effective October 1, 2020.
For CY 2025, the proposed OPPS payment rates are based on available CY 2023 claims data. For CY 2025, based on our analysis of claims data, we found a total of 96 single frequency claims and a geometric mean cost of approximately $4,902 for HCPCS code C9769. For CY 2025, we proposed to continue to assign HCPCS code C9769 to APC 5376 (Level 6 Urology and Related Services) with a proposed payment rate of approximately $9,209.
Comment: One commenter pointed out that the AMA CPT created a new code, CPT code 5XX05 effective January 1, 2025, to describe the cystourethroscopy with iTIND service represented by HCPCS code C9769. CPT code 5XX05 (Cystourethroscopy with insertion of temporary device for ischemic remodeling ( i.e., pressure necrosis) of bladder neck and prostate) becomes effective January 1, 2025. The commenter stated that CMS may either sunset HCPCS code C9769 and reassign the underlying claims to CPT code 5XX05 or maintain HCPCS code C9769 active and assign CPT code 5XX05 to a non-payable status in the OPPS.
Response: We thank the commenter for pointing out that CPT code 5XX05 (53865) will become effective January 1, 2025. We note that we will replace HCPCS code C9769 with CPT code 5XX05 (53865) effective January 1, 2025, as well as assign the underlying claims associated with HCPCS code C9769 to CPT code 5XX05 (53865). Therefore, for CY 2025, we are reassigning CPT code 5XX05 (53865) to Level 6 Urology and Related Services (APC 5376). We note that we will continue to evaluate and monitor the cost for CPT code 5XX05 (53865) for future rulemaking.
After consideration of the public comment we received, we are finalizing our policy to assign CPT code 5XX05 (53865) to APC 5376 (Level 6 Urology and Related Services) and we will be sunsetting HCPCS code C9769. Please refer to Table 85 for the final OPPS APC and status indicator assignment for CPT code 5XX05 (53865) for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
24. Dental Alveoloplasty and Exostosis Removal Procedures, CDT Codes D7320, D7321, and D7471 (APCs 5163, 5164)
Effective January 1, 2024, we made 229 additional dental codes payable under the OPPS when payment and coverage requirements are met, as provided in the relevant PFS payment rules regarding Medicare Part B payment for dental services (88 FR 81540-82185). For CY 2025, we did not propose to make additional dental codes payable under the OPPS. As a result, we proposed to continue to assign many dental services to non-payable status indicators, including CDT codes D7320 (Alveoloplasty not in conjunction with extractions—four or more teeth or tooth spaces, per quadrant), D7321(Alveoloplasty not in conjunction with extractions—one to three teeth or tooth spaces, per quadrant), and D7471 (Removal of lateral exostosis (maxilla or mandible), which we proposed to assign to status indicator “B,” indicating the service is not paid under OPPS, for CY 2025.
Comment: We received a comment from an association representing oral and maxillofacial surgeons requesting that we make CDT codes D7320, D7321, and D7471 payable under the OPPS. The commenter stated that these codes may contribute to the elimination of an oral or dental infection. The commenter explained that alveoloplasty and the removal of lateral exostosis are often necessary procedures for medically compromised patients undergoing tooth removal prior to radiation treatment. Failure to perform these procedures, per the commenter, can put patients at risk for osteoradionecrosis following radiation therapy. In addition, the commenter explained that not performing an alveoloplasty or removing the exostosis when clinically indicated may impede the patient's ability to receive a removable prosthesis due to the risk of tissue trauma. The procedures may also be required to address common sequelae following dental extractions. Per the commenters, excluding the services described by CDT codes D7320, D7321, and D7471 from OPPS payment places patients at considerable risk and diminishes the effectiveness of initial treatments aimed at eliminating oral or dental infections. Finally, the commenters stated that there are existing CPT codes that describe these procedures. Specifically, per the commenters, CPT code 41874 (Alveoloplasty, each quadrant (specify)) may describe alveoloplasty procedures and CPT code 41823 (Excision of osseous tuberosities, dentoalveolar structures) may describe the removal of exostosis.
Response: We appreciate the commenter's input. We note that the commenter submitted a similar public comment to the CY 2024 OPPS/ASC final rule to assign several codes, including CDT codes D7320, D7321, and ( printed page 94087) D7471, to APCs for CY 2024. In the CY 2024 OPPS/ASC final rule, we agreed to assign to clinical APCs the codes for which we agreed with the commenter may be considered medically necessary diagnostic and treatment services immediately necessary to eliminate or eradicate an oral or dental infection prior to, or contemporaneously with certain Medicare-covered medical services specified in the CY 2023 PFS final rule (42 CFR 411.15). We also specified we needed more information on how certain codes the commenter recommended for APC assignment, including CDT codes D7320, D7321, and D7471, were consistent with existing dental payment polices.
The comment to the CY 2025 OPPS/ASC proposed rule provided additional information regarding how dental surgical procedures including surgical interventions to facilitate tooth eruption, alveoloplasty or ridge preparation and removal of lateral exostosis of the jaw may contribute to the elimination of an oral or dental infection. Based on the additional information provided, we agree with the commenter that t CDT codes D7320, D7321 and D7471 should be assigned to clinical APCs for CY 2025 as we believe there may be instances where these services may be considered medically necessary diagnostic and treatment services immediately necessary to eliminate or eradicate an oral or dental infection prior to, or contemporaneously with, certain Medicare-covered medical services specified in the CY 2023 and CY 2024 PFS final rules. For CY 2025, we are assigning CDT codes D7320 and D7321 to APC 5163 (Level 3 ENT Procedures), based on clinical and resource similarities to other alveoloplasty codes currently assigned to the same clinical APC, specifically CDT codes D7310 (Alveoloplasty in conjunction with extractions—four or more teeth or tooth spaces, per quadrant) and D7311 (Alveoloplasty in conjunction with extractions—one to three teeth or tooth spaces, per quadrant). We are also assigning CDT code D7471 to APC 5164 (Level 4 ENT Procedures) based on clinical and resource similarities to other exostosis services assigned to the same clinical APC, specifically CPT codes 21031 (Remove exostosis mandible) and 21032 (Remove exostosis maxilla).
We note that Medicare Part B does not pay for removable prostheses, such as dentures. While we understand that services like alveoloplasty and the removal of the exostosis can improve the fit and stability of dentures and other dental prosthetics as the commenter explained, we are not finalizing APC assignments for CPT codes D7320, D7321 and D7471 so that these services may be furnished in preparation for a patient's denture or dental implant, and billing Medicare for that purpose would not meet our payment and coverage requirements. As we have consistently stated in past rules (87 FR 71879) and quarterly change requests to assign new codes to APCs (see, e.g., Pub 100-04 Medicare Claims Processing, Transmittal 11937), the fact that a drug, device, procedure or service is assigned a HCPCS code and a payment rate under the OPPS does not imply coverage by the Medicare program, but indicates only how the product, procedure, or service may be paid if covered by the program. Medicare Administrative Contractors (MACs) determine whether a drug, device, procedure, or other service meets all program requirements and conditions for coverage and payment. Accordingly, we emphasize that HOPDs would only receive payment for a dental service assigned to an APC when the appropriate MAC determines that the service meets the relevant conditions for coverage and payment.
After consideration of the public comment received, for CY 2025, we are finalizing clinical APC assignments for CDT codes D7320, D7321, and D7471. Please refer to Table 86for the final APC assignments and status indicators for CDT codes D7320, D7321, and D7471. We refer readers to Addendum D1 to this final rule with comment period for a complete list of status indicators and their definitions. Addendum D1 is available via the internet on the CMS website.
25. Digital Mental Health Treatment Devices, HCPCS Codes GMBT1, GMBT2, and GMBT3 (APC 5012)
In CY 2025 Medicare Physician Fee Schedule proposed rule (89 FR 61956) we stated that we recognize that digital therapeutics may offer innovative means to access certain behavioral health care services. We noted that given nationwide behavioral health workforce shortages combined with increasing demand for behavioral health care services, some Medicare beneficiaries may have limited access to these services. To that end, we proposed to create three new HCPCS codes for Digital Mental Health Treatment Devices (DHMT): GMBT1 ( Supply of digital mental health treatment device and initial education and onboarding, per course of treatment that augments a behavioral therapy plan) for furnishing a DMHT device; GMBT2 ( First 20 ( printed page 94088) minutes of monthly treatment management services directly related to the patient's therapeutic use of the digital mental health treatment (DMHT) device that augments a behavioral therapy plan, physician/other qualified health care professional time reviewing data generated from the DMHT device from patient observations and patient specific inputs in a calendar month and requiring at least one interactive communication with the patient/caregiver during the calendar month ) and GMBT3 ( Each additional 20 minutes of monthly treatment management services directly related to the patient's therapeutic use of the digital mental health treatment (DMHT) device that augments a behavioral therapy plan, physician/other qualified health care professional time reviewing data generated from the DMHT device from patient observations and patient specific inputs in a calendar month and requiring at least one interactive communication with the patient/caregiver during the calendar month). We did not address the new proposed DMHT device G-codes or propose APC or Status Indicator assignments in the CY 2025 OPPS proposed rule (89 FR 59186).
The HCPCS codes G0552, G0553, and G0554 are replacing the respective placeholder codes GMBT1, GMBT2, and GMBT3.
Comment: We received several comments on the DHMT codes with different suggestions on how these codes be treated in the OPPS. One commenter recommended that CMS assign code G0552 (placeholder code GMBT1) to APC 1511—New Technology Level 11—with a CY 2025 proposed payment rate of $950.50. This commenter went on to say that CMS could then update the applicable APC assignment for DMHT devices over time after it obtains hospital claims data. Commenters who suggested CMS assign HCPCS code G0552 (placeholder code GMBT1) to a New Technology APC explained that this approach is similar to ones the agency has taken in the past—most notably for reimbursing for the technical/facility components of remote therapeutic monitoring (RTM) and remote physiological monitoring (RPM) codes. One commenter supported contractor pricing for HCPCS code G0552 (placeholder code GMBT1). Commenters urged CMS to establish a payment for the DMHT device supply in the hospital outpatient setting in the CY 2025 OPPS final rule.
Response: HCPCS codes G0552, G0553, and G0554 (placeholder codes GMBT1, GMBT2, and GMBT3) will be effective on January 1, 2025. Based on our evaluation of these new codes and in response to commenters' requests, we are proposing an interim assignment of HCPCS code G0552 (placeholder code GMBT1) to status indicator “V” (Clinic or Emergency Department Visit; Paid under OPPS; separate APC payment) and APC 5012 (Clinic Visits and Related Services). We are proposing an interim assignment of HCPCS code G0553 (placeholder code GMBT2) to status indicator “V” (Clinic or Emergency Department Visit; Paid under OPPS; separate APC payment) and APC 5012 (Clinic Visits and Related Services). Finally, we are proposing an interim assignment of HCPCS code G0554 (placeholder code GMBT3) to status indictor “N” to indicate that the code is packaged since it is an add-on service to the primary code, and payment for add-on codes is always packaged with their associated primary service codes under regulation at 419.2(b)(18).
Because we were not able to propose APC and status indicator assignments in the CY 2025 OPPS proposed rule, we will be assigning HCPCS codes G0552, G0553, and G0554 (placeholder codes GMBT1, GMBT2, and GMBT3) comment indicator “NI” in Addendum B of this final rule. This comment indicator is for new HCPCS codes that will be effective January 1, 2025, to indicate that we are assigning them an interim status indicator and APC assignment, which is subject to public comment. We invite public comment on the interim proposed status indicators and APC assignments for HCPCS codes G0552, G0553, and G0554 (placeholder codes GMBT1, GMBT2, and GMBT3), which will then be finalized in the CY 2026 OPPS/ASC final rule with comment period.
Please refer to Table 87 for the proposed interim APC and status indicator assignments for HCPCs codes G0552, G0553, and G0554 (placeholder codes GMBT1, GMBT2, and GMBT3) for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period.
( printed page 94089)26. Drug-Coated Balloon for Esophageal and Bowel Strictures, CPT Codes 0884T, 0885T, and 0886T (APC 5331)
ProTractX3TM TTS is a drug-coated balloon that is used to treat strictures in the esophagus, small intestine, and large intestine. For the CY 2025 OPPS proposed rule, we proposed to assign CPT code 0884T (Esophagoscopy, flexible, transoral, with initial transendoscopic mechanical dilation ( e.g., nondrug-coated balloon) followed by therapeutic drug delivery by drug-coated balloon catheter for esophageal stricture, including fluoroscopic guidance, when performed) with no claims data for CY 2025 to APC 5303 (Level 3 Upper GI Procedures) with a payment rate of around $3,805804. In addition, we proposed to assign both CPT code 0885T (Colonoscopy, flexible, with initial transendoscopic mechanical dilation ( e.g., nondrug-coated balloon) followed by therapeutic drug delivery by drug-coated balloon catheter for colonic stricture, including fluoroscopic guidance, when performed) and CPT code 0886T (Sigmoidoscopy, flexible, with initial transendoscopic mechanical dilation ( e.g., nondrug-coated balloon) followed by therapeutic drug delivery by drug-coated balloon catheter for colonic stricture, including fluoroscopic guidance, when performed) with no claims data for CY 2025 to APC 5313 (Level 3 Lower GI Procedures) with a payment rate of around $2,742.
Comment: One commenter, the manufacturer of the ProTractX3TM TTS, requested that the procedures described CPT codes 0884T-0886T be reassigned to APC 5331 (Complex GI Procedures) with a payment rate of around $5,953. The commenter stated that the cost of the ProTractX3TM TTS is $3,100. The commenter based their cost estimate for the ProTractX3TM TTS on similar drug-coated balloons used for other medical procedures. The commenter believes the cost of ProTractX3TM TTS should be added to the geometric mean cost of the existing procedures that correspond to the procedures described by CPT codes 0884T-0886T, but do not describe the use of a drug-coated balloon with the procedures. These procedures are CPT code 43220 (Esophagoscopy, flexible, transoral; with transendoscopic balloon dilation (less than 30 mm diameter) with a geometric mean cost of around $2,175 that corresponds to procedure described by CPT code 0884T; CPT code 45386 (Colonoscopy, flexible; with transendoscopic balloon dilation) with a geometric mean cost of around $1,662 that corresponds to the procedure described by CPT code 0885T; and CPT code 45340 (Sigmoidoscopy, flexible; with transendoscopic balloon dilation) with a geometric mean cost of around $1,518 that corresponds to the procedure described by CPT code 0886T. The commenter also noted that additional operating room time needed to use the drug-coated balloon adds another $806 to the cost of each of these procedures.
According to the commenter, when the cost of ProTractX3TM TTS drug-coated balloon and the cost of the additional operating room time to use the drug-coated balloon is added to the geometric cost of CPT code 43220, the estimated cost of CPT code 0884T is $6,081. The commenter performed similar calculations for CPT code 0885T reporting an estimated procedure cost of $5,568, and for CPT code 0886T, the commenter reported an estimated procedure cost is $5,424. The commenter made the case that these procedure costs are much closer to the payment rate of APC 5331 of $5,953, than the payment rate of $3,805 for APC 5303 where we proposed to assign CPT code 0884T. Likewise, the commenter showed that the procedure costs for CPT codes 0885T and 0886T are much closer to the payment rate of APC 5331 of $5,953, than the payment rate of APC 5313 of $2,742 where we had proposed to assign those CPT codes.
Response: We appreciate the detailed analysis by the commenter which demonstrated the additional costs of the ProTractX3TM TTS drug-coated balloon, as well as the use of the drug-coated ( printed page 94090) balloon when compared to the corresponding procedures where no drug-coated balloon is used. Based on the additional cost data provided by the commenter, we agree with the commenter that CPT codes 0884T-0886T should be assigned to APC 5331.
After consideration of the public comments we received, we are implementing our proposal with modification to assign CPT codes 0884T, 0885T, and 0886T to APC 5331 (Complex GI Procedures). Table 88 shows the finalized status indicator and APC assignment for all of the procedure codes. We refer readers to Addendum B of this final rule for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
27. EchoGo Echocardiography Image Processing Service, HCPCS Code C9786 (APC 5743)
Effective July 1, 2023, based on a New Technology application received by CMS for an echocardiography image processing service, CMS established HCPCS code C9786 (Echocardiography image post processing for computer aided detection of heart failure with preserved ejection fraction, including interpretation and report) and assigned it to APC 5742 (Level 2 Electronic Analysis of Devices). In CY 2024, CMS reassigned HCPCS code C9786 to APC 5743 (Level 3 Electronic Analysis of Devices).
For CY 2025, CMS is deleting HCPCS code C9786 because the CPT Editorial Panel established new Category III CPT code, specifically, 0932T (Noninvasive detection of heart failure derived from augmentative analysis of an echocardiogram that demonstrated preserved ejection fraction, with interpretation and report by a physician or other qualified health care professional) to describe the same service, which will be effective January 1, 2025. Because the final CY 2025 CPT code number was not available when we published the CY 2025 OPPS/ASC proposed rule, the code was listed as placeholder code XX68T in the OPPS Addendum B of the CY 2025 OPPS/ASC proposed rule.
In the CY 2025 OPPS/ASC proposed rule, we proposed to assign CPT code XX68T (Noninvasive detection of heart failure derived from augmentative analysis of an echocardiogram that demonstrated preserved ejection ( printed page 94091) fraction, with interpretation and report by a physician or other qualified health care professional) to APC 5743 (Level 3 Electronic Analysis of Devices), the same APC to which HCPCS code C9786 is assigned.
We note that because both HCPCS code C9786 and CPT code 0932T describe the same service, effective January 1, 2025, CMS will delete HCPCS code C9786 and only CPT code 0932T will be used to bill for the service. We received one public comment in support of our proposal.
Comment: The commenter noted that the assignment of XX68T to APC 5743 appropriately reflects the resources required to perform this advanced artificial intelligence (AI) analysis, as well as the clinical value it provides. and stated that this APC assignment would help ensure adequate reimbursement for hospitals, supporting broader access to this important diagnostic tool for Medicare beneficiaries. The commenter appreciated CMS's consideration of this new technology and its potential impact on patient care.
Response: We thank the commenter for the support.
In summary, after consideration of the public comment we received, we are finalizing our proposal, without modification, to assign CPT code 0932T to APC 5743 for CY 2025. We note that the final CY 2025 payment rate for the code can be found in Addendum B of this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
28. Endoscopic Submucosal Dissection (ESD) Procedure, HCPCS Code C9779 (APC 5303)
CMS established HCPCS code C9779 (Endoscopic submucosal dissection (ESD), including endoscopy or colonoscopy, mucosal closure, when performed) effective October 1, 2021, to describe the endoscopic submucosal dissection (ESD) performed during an endoscopy or colonoscopy. HCPCS code C9779 was established based on a New Technology application that was submitted to CMS for New Technology consideration under the OPPS. Based on our assessment, we assigned the code to APC 5313 (Level 3 Lower GI Procedures) because we believed the ESD procedure had similar clinical characteristics and resource costs as the surgical procedures assigned to APC 5313. We announced the assignment to APC 5313 in the October 2021 OPPS quarterly update CR (Transmittal 10997, Change Request 12436, dated September 16, 2021). In CY 2022, we continued to assign the code to APC 5313.
For CY 2023, we assigned HCPCS code C9779 to APC 5303 (Level 3 Upper GI Procedures) after receiving public comments that stated that the ESD procedure's resource requirements and geometric mean cost were more similar to the resource requirements and geometric mean costs of procedures found in APC 5303. Further, commenters noted that the ESD procedure is technically more demanding, requires advanced skills to perform, and is clinically similar to CPT code 43497 (Lower esophageal myotomy, transoral ( i.e., peroral endoscopic myotomy [POEM])), which was assigned to APC 5303. For CY 2025, we proposed to maintain HCPCS code C9779 (with a geometric mean cost (GMC) of around $5,093) in APC 5303 (Level 3 Upper GI Procedures).
Comment: We received one comment requesting that we reassign HCPCS code C9779 from APC 5303 (Level 3 Upper GI Procedures) to APC 5361 (Level 1 Laparoscopy and Related Procedures). The commenter states that resource requirements for HCPCS code C9779 are well-aligned with other procedures in APC 5361, noting that the geometric mean cost of HCPCS code C9779 is higher than 4 procedures with significant volume assigned to APC 5361. The commenter believes that based on resource cost and clinical homogeneity, HCPCS code C9779 should be reassigned to APC 5361.
Response: We thank the commenter for their input. We disagree with the commenter that HCPCS code C9779 should be assigned to APC 5361. Specifically, we do not believe that endoscopic submucosal dissection is a laparoscopic procedure and should not be assigned to a laparoscopic APC family. Additionally, we believe HCPCS code C9779 is appropriately assigned, based on both clinical similarity and resource costs, to APC 5303 (Level 3 Upper GI Procedures).
In summary, after consideration of the public comment, we are finalizing our proposal without modification to assign HCPCS code C9779 to APC 5303 (Level 3 Upper GI Procedures). We note that we review, on an annual basis, the APC assignments for all services and items paid under the OPPS based on our analysis of the latest claims data. The final CY 2025 OPPS payment rate for the code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator (SI) meanings for all codes reported under the OPPS. Both Addendum B and D1 are available via the internet on the CMS website.
29. Esophageal Balloon Distention Study, CPT Code 91040 (APC 5723)
Esophageal balloon distension studies are used to diagnose conditions of the esophagus and may be used to determine the source of certain types of pain, such as chest pain. The esophageal balloon study is often performed in conjunction with esophagogastroduodenoscopy procedures. For CY 2025, we proposed to assigned CPT code 91040 (Esophageal balloon distension study, diagnostic, with provocation when performed) to APC 5723 (Level 3 Diagnostic Tests and Related Services) with a proposed payment rate of around $530.
Comment: We received one public comment requesting that CPT code 91040 be reassigned from APC 5723 (Level 3 Diagnostic Tests and Related Services) to APC 5724 (Level 4 Diagnostic Tests and Related Services), stating that the costs of this procedure significantly exceed the APC 5723 assignment. The commenter suggested that a higher payment would more closely align with the costs of the resources utilized to perform this test.
Response: We thank the commenter for their input. We note that the geometric mean cost (GMC) for CPT code 91040 is around $1,500, and the GMC for APC 5723 is around $537. While this is a significant difference, our claims data show that approximately 70 percent of the costs for this procedure are made up of higher cost items (equipment, drugs, and supplies) that are packaged with the procedure costs.
In addition, claims data for CY 2025, show that out of all the claims billed with CPT code 91040 (3,429 total frequency), only 2.7 percent (approximately 95) of those claims were billed with only CPT code 91040. We believe that in addition to the higher costs of the items that are packaged, the costs from the other procedures that are performed with CPT code 91040 have driven up the GMC of CPT code 91040.
Based on review of the service and other services in the same APC family, input from CMS medical advisors, and the claims data, we believe that the clinical and resource characteristics of CPT code 91040 are sufficiently like other procedures assigned to APC 5723 and should continue to be assigned to APC 5723. For CY 2025, based on our evaluation, we are finalizing our proposal, without modification, to assign CPT code 91040 to APC 5723. ( printed page 94092) The final CY 2025 payment rate for the code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
30. Esophagogastroduodenoscopy With Optical Endomicroscopy, CPT Code 43252 (APC 5302)
An esophagogastroduodenoscopy is a procedure to view the esophagus, stomach, and duodenum by means of a flexible endoscope passed through the mouth. The provider uses an endomicroscopy system to evaluate the structures during this procedure. This procedure is reported with CPT code 43252 (Esophagogastroduodenoscopy, flexible, transoral; with optical endomicroscopy).
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81685), we reassigned CPT code 43252 from APC 5303 (Level 3 Upper GI Procedures) to APC 5302 (Level 2 Upper GI Procedures). In the 2025 OPPS/ASC proposed rule, we proposed to continue to assign this code to APC 5302 with a proposed payment rate of $1,884.11. We received two public comments that disagreed with our proposal.
Comment: Both commenters noted that the significant payment reduction for this service could impede access to this service for beneficiaries. One commenter noted that just a few hospital outpatient facilities are offering this procedure and that over 70 percent of cases are performed in ASCs.
One commenter stated that CPT code 43252 is similar in clinical intensity, resource utilization and costs to CPT code 0654T (Esophagogastroduodenoscopy, flexible, transnasal; with insertion of intraluminal tube or catheter) which is assigned to APC 5303. This commenter also claimed that some hospitals are underreporting their costs for CPT code 43252. Both commenters requested that CMS reassign CPT code 43252 to APC 5303.
Response: We note that the CY 2025 OPPS payment rates are proposed based on available CY 2023 claims data. Based on our latest review of the claims data, we found the geometric mean cost of approximately $1,839.38for CPT code 43252 based on 246 single claims, is consistent with the geometric mean cost of about $1,920.00for APC 5302. We note that the geometric mean cost of APC 5303 is approximately $3,850.67which is about $2,011 more than the geometric mean cost of CPT code 43252.
In summary, we are finalizing without modification our proposal to assign CPT code 43252 to APC 5302 for CY 2025. The CY 2025 final payment rate for this code can be found in Addendum B of this final rule with comment period.
31. Extracorporeal Shock Wave Lithotripsy (SWL), CPT Code 50590 (APC 5374)
Extracorporeal shock wave lithotripsy is a procedure used to break up stone in the urinary tract using directed shock wave therapy. Shock waves are generated by a lithotripter which is a machine and capital equipment for the provider. The procedure is described by CPT code 50590 (Lithotripsy, extracorporeal shock wave).
For CY 2025, the OPPS payment rates are proposed based on available CY 2023 claims data. For the CY 2025 OPPS/ASC proposed rule, we found a total of 26,669 single frequency claims and a geometric mean cost of approximately $3,536 for CPT code 50590. For CY 2025, we proposed to continue to assign CPT code 50590 to APC 5374 (Level 4 Urology and Related Services) with a proposed payment rate of approximately $3,438.
Comment: One commenter disagreed with our APC assignment of CPT code 50590 to APC 5374 (Level 4 Urology and Related Services) and suggested that CPT code 50590 should be assigned to APC 5375 (Level 5 Urology and Related Services) like CPT code 52353 (Cystourethroscopy, with ureteroscopy and/or pyeloscopy; with lithotripsy (ureteral catheterization is included)) and CPT code 52356 (Cystourethroscopy, with ureteroscopy and/or pyeloscopy; with lithotripsy including insertion of indwelling ureteral stent ( e.g., gibbons or double-j type)). The commenter stated that higher payment for CPT codes 52353 and 52356 has driven service volume toward those codes and away from CPT code 50590.
Response: We thank the commenter for the input. We note that, with over 25,000 single claims in CY 2025, the geometric mean cost of CPT code 50590 very closely aligns with the geometric mean cost of the APC to which it is assigned, APC 5374. Furthermore, CPT code's geometric mean cost is well below that of CPT code 52356, which is the primary driver of the geometric mean cost of APC 5375.
After consideration of the public comment we received, we are finalizing our proposal without modification for CPT code 50590. Please refer to Table 89 for the final OPPS APC and status indicator assignment for CPT code 50590 for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94093)32. Female Intraurethral Valve-Pump, Insertion and Replacement, CPT Codes 0596T and 0597T (APC 5372)
For CY 2025, we proposed to assign CPT codes 0596T (Temporary female intraurethral valve-pump ( i.e., voiding prosthesis); initial insertion, including urethral measurement) and 0597T (Temporary female intraurethral valve-pump ( i.e., voiding prosthesis); replacement) to status indicator “J1”, APC 5372 (Level 2 Urology and Related Services) with a proposed payment rate of $675.64.
Comment: Commenters shared support for the proposal to assign CPT code 0597T to APC 5372 and agreed that the proposed assignment to APC 5372 aligns with costs incurred by hospitals to perform this service.
Response: We thank commenters for their support.
Comment: Commenters, the manufacturer and a trade association, expressed concerns that the proposed assignment of CPT code 0597T to APC 5372 does not sufficiently consider the targeted device cost of $1,885 and is not appropriate from a resource perspective, which the commenters feel may limit access to the procedures. The commenters requested reassignment of CPT code 0596T to APC 5373 (Level 3 Urology and Related Services) with a proposed payment rate of $2,074.53 as they believe that APC 5373 would be appropriate from a clinical and resource perspective and would more appropriately reflect the device costs to hospitals to furnish the service.
Response: We thank the commenter for their comment. For this final rule with comment period, we have no claims data for CPT code 0596T for OPPS ratesetting purposes. We continue to believe that CPT code 0596T shares similar clinical characteristics and resource costs to the services assigned to APC 5372.
In summary, after consideration of the public comments we received, we are finalizing our proposal, without modification, to continue to assign CPT codes 0596T and 0597T to APC 5372 for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reported under the OPPS. In addition, we refer readers to Addendum A of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Both Addendum A and Addendum B are available via the internet on the CMS website.
33. Fractional Flow Reserve (FFR) With 3D Coronary Mapping, CPT Code 0523T
The fractional flow reserve (FFR) with 3D coronary mapping procedure is described by CPT code 0523T (Intraprocedural coronary fractional flow reserve (ffr) with 3d functional mapping of color-coded ffr values for the coronary tree, derived from coronary angiogram data, for real-time review and interpretation of possible atherosclerotic stenosis(es) intervention (list separately in addition to code for primary procedure)). CPT code 0523T became effective January 1, 2019. For CY 2024, CPT code 0523T was assigned the status indicator “N” (packaged).
Comment: One commenter requested that the packaging exemption for SaaS add-on codes policy should be applied to CPT code 0523T. The commenter further requested that CPT code 0523T be assigned to status indicator “J1” (Comprehensive APC) and assigned to APC 5191 (Level 1 Endovascular Procedures) for CY 2025.
Response: We note that CPT code 0523T is an add-on code. Add-on codes represent services that are always performed in addition to the primary service or procedure and must never be reported as a stand-alone code. As specified under 42 CFR 419.2(b)(18), add-on codes are generally packaged under the OPPS, and payment for the codes are bundled with the primary codes. Consequently, CPT code 0523T is not paid separately under the OPPS, but instead, has its payment packaged into the payment for the primary code.
Under the packaging exemption for SaaS add-on codes (87 FR 72027 to 72035), we adopted a policy that services represented by SaaS add-on codes are not among the “certain services described by add-on codes” for which we package payment with the related procedures or services under the regulation at 42 CFR 419.2(b)(18). Specifically, the SaaS CPT add-on codes will be assigned to identical APCs and have the same status indicator assignments as their standalone SaaS codes. As such, the policy exempts SaaS add-on codes for which a standalone SaaS code is separately payable under the OPPS for the identical SaaS service furnished without concurrent imaging and described by the standalone CPT code. In summary, the packaging exemption for SaaS add-on codes policy only exempts SaaS add-on codes when the identical SaaS procedure is also represented by a standalone SaaS code and payable under the OPPS because the procedure requires the same resources regardless of whether it is furnished with or without the imaging service.
After consideration of the public comments we received, we are finalizing our proposal without modification for CPT codes 0523T. Please refer to Table 90 below for the final OPPS APC and status indicator assignment for CPT codes 0523T for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94094)34. Fractional Flow Reserve Derived From Computed Tomography (FFRCT), CPT Code 75580 (APC 5724)
Fractional Flow Reserve Derived from Computed Tomography (FFRCT), also known by the trade name HeartFlow®, is a noninvasive diagnostic service that allows physicians to measure coronary artery disease in a patient through the use of coronary CT scans. The HeartFlow® service is indicated for clinically stable symptomatic patients with coronary artery disease, and, in many cases, may avoid the need for an invasive coronary angiogram procedure. HeartFlow® uses a proprietary data analysis process performed at a central facility to develop a three-dimensional image of a patient's coronary arteries, which allows physicians to identify the fractional flow reserve to assess whether patients should undergo further invasive testing (that is, a coronary angiogram).
HeartFlow® is currently described by CPT code 75580 (Noninvasive estimate of coronary fractional flow reserve (FFR) derived from augmentative software analysis of the data set from a coronary computed tomography angiography, with interpretation and report by a physician or other qualified health care professional). On January 1, 2024, CPT code 75580 replaced CPT code 0503T ((Noninvasive estimated coronary fractional flow reserve (ffr) derived from coronary computed tomography angiography data using computation fluid dynamics physiologic simulation software analysis of functional data to assess the severity of coronary artery disease; analysis of fluid dynamics and simulated maximal coronary hyperemia, and generation of estimated ffr model).
HeartFlow® was assigned to APC 5724 (Level 4 Diagnostic Tests and Related Services) for CY 2024, and we proposed for CY 2025 to continue to assign HeartFlow® (CPT code 75580) to APC 5724.
Comment: We received one comment stating that several of the Medicare Administrative Contractors (MACs) have an edit in place that prohibits the use of the cardiology revenue code (0480) when billing CPT code 75580 as evidenced by claims denials with “Invalid Revenue Code” errors. The commenter asked that we provide guidance that explicitly allows CPT code 75580 to be billed with any appropriate revenue code just as we did for cardiac CT services. (See January 2024 Update (Transmittal 12421, Change Request 13488, dated December 21, 2023.)
Response: Based on the information the commenter provided, we were able to identify the outdated edit and remove it. Facilities may bill CPT 75580 with any appropriate revenue code. As a reminder, it is longstanding CMS policy that hospital outpatient facilities are responsible for reporting the appropriate cost centers and revenue codes on claims. As stated in section 20.5 in Chapter 4 (Part B Hospital) of the Medicare Claims Processing Manual, CMS “does not instruct hospitals on the assignment of HCPCS codes to revenue codes for services provided under OPPS since hospitals' assignment of cost vary. Where explicit instructions are not provided, HOPDs should report their charges under the revenue code that will result in the charges being assigned to the same cost center to which the cost of those services are assigned in the cost report.” In addition to referring readers to the Medicare Claims Processing Manual, we will be providing public education and instruction through the CMS Medicare Learning Network (MLN).
Comment: Multiple commenters supported our proposal to continue to assign CPT code 75580 to APC 5724 for CY 2025.
Response: We thank the commenters for their input and for their support of our policy.
After consideration of the public comments we received, we are finalizing our proposal without modification. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
35. Gastric Electrophysiology Mapping With Simultaneously Validated Patient System Profiling (GEMS) Service, CPT Code 0868T (APC 5723)
Effective July 1, 2023, based on a New Technology application received by CMS for the GEMS service, CMS established HCPCS code C9787 (Gastric electrophysiology mapping with simultaneous patient symptom profiling) and assigned it to APC 5723 (Level 3 Diagnostic Tests and Related Services). Effective July 1, 2024, HCPCS code C9787 was deleted and replaced by CPT code 0868T (High-resolution gastric electrophysiology mapping with simultaneous patient symptom profiling, with interpretation and report). CMS assigned CPT code 0868T to APC 5723, the same APC to which its predecessor code, HCPCS code C9787, was assigned. For CY 2025, CMS proposed to continue to assign CPT code 0868T to APC 5723 with a proposed payment rate of $527.44 for CY 2025.
Comment: We received several comments, including a comment from the manufacturer, requesting that we reassign CPT code 0868T from APC 5723 to APC 1520 (New Technology—Level 20 ($1801-$1900)) with a payment rate of $1850.50, based on the purported cost of the technology and the ( printed page 94095) fact that it is a new service with a low volume of claims. The commenters explained that the proposed payment rate for CPT code 0868T would not cover the costs of the device, supplies, and clinical staff expenses. One commenter explained that the available claims data supports the conclusion that the proposed APC placement is inadequate. Specifically, the commenter pointed to one single frequency claim that shows a geometric mean cost of approximately $1,800 to demonstrate that CPT code 0868T should be assigned to APC 1520. The commenters suggested that if CMS did not agree to finalize an assignment to APC 1520, they would then suggest that CPT code 0868T be assigned to APC 5724 (Level 4 Diagnostic Tests and Related Services) with a proposed payment rate of approximately $1,000 or APC 5302 (Level 2 Upper GI Procedures) with a proposed payment rate of approximately $1,800 for CY 2025. Finally, one commenter drew differences between the clinical characteristics of CPT code 0868T and the other services assigned to APC 5723, including CPT code 0799T. For example, the commenter stated that CPT code 0799T involves a low-resolution electrode patch generating data comparable to traditional legacy electrogastrography, whereas C9787/0868T uses a high-resolution 64-lead array and sophisticated software to provide detailed multimodal diagnostic data.
Response: We appreciate the commenters' input. We disagree with the APC assignments recommended by commenters based on the purported costs of the service. Since we issued the proposed rule, additional claims data have been processed which, we believe, confirm that the proposed APC assignment is appropriate for HCPCS code C9787/CPT code 0868T for CY 2025. Our analysis of the available claims data demonstrates that the geometric mean cost for HCPCS code C9787 is approximately $310 based on 5 single frequency claims, which is below the geometric mean cost of $530 for APC 5723, but within the range of geometric mean costs of services assigned to APC 5723 which vary from approximately $150 to $2000. We note that given the effective date of payment, we have only one year of claims data for HCPCS code C9787/CPT code 0868T with an extremely low level of single frequency claims.
We will continue to monitor the claims data and adjust the APC placement for CPT code 0868T, based on the claims data in future rulemaking. Additionally, based on our review of the technology used as part of the service, input from CMS medical advisors, and review of all other information available to us, we continue to believe that CPT code 0868T is similar to services assigned to APC 5723. Based on the resource and clinical similarities to other services assigned to APC 5723, including CPT code 0779T (Gi myoelectrical actv study), as well as the available claims data, we believe that our proposal to assign CPT code 0868T is appropriate for CY 2025.
After consideration of the public comments, we are finalizing our proposal without modification to continue to assign CPT code 0868T to APC 5723. The final CY 2025 payment rate for the code can be found in Addendum B to this final rule with comment period. We also refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
36. Hernia Repair Procedures, CPT Codes 49593, 49595, and 49615 (APCs 5342 and 5361)
Effective January 1, 2023, the AMA created new abdominal hernia repair CPT codes 49593 (Repair of anterior abdominal hernia(s) ( i.e., epigastric, incisional, ventral, umbilical, spigelian), any approach ( i.e., open, laparoscopic, robotic), initial, including implantation of mesh or other prosthesis when performed, total length of defect(s); 3 cm to 10 cm, reducible), 49595 (Repair of anterior abdominal hernia(s) ( i.e., epigastric, incisional, ventral, umbilical, spigelian), any approach ( i.e., open, laparoscopic, robotic), initial, including implantation of mesh or other prosthesis when performed, total length of defect(s); greater than 10 cm, reducible), and 49615 (Repair of anterior abdominal hernia(s) ( i.e., epigastric, incisional, ventral, umbilical, spigelian), any approach ( i.e., open, laparoscopic, robotic), recurrent, including implantation of mesh or other prosthesis when performed, total length of defect(s); 3 cm to 10 cm, reducible). For CY 2023, we assigned these codes to APC 5341 (Level 1 Abdominal/Peritoneal/Biliary and Related Procedures). For CY 2025, we proposed to reassign these codes to APC 5342 (Level 2 Abdominal/Peritoneal/Biliary and Related Procedures) to resolve a 2-times violation for APC 5341.
Comment: We received two comments supporting the reassignment of CPT codes 49593, 49595, and 49615 to APC 5342 for CY 2025.
Response: We thank the commenters for their support.
Comment: The two commenters recommended that we also reassign CPT codes 49650 (Laparoscopy, surgical; repair initial inguinal hernia) and 49651 (Laparoscopy, surgical; repair recurrent inguinal hernia) from APC 5361 (Level 1 Laparoscopy and Related Services) to APC 5342. The commenters noted that with the addition of the three ventral hernia procedures, APC 5342 would now contain both open and laparoscopic surgical procedures.
Response: We thank the commenter for their suggestions. After further clinical review, we continue to believe that CPT codes 49650 and 49651 are still appropriately assigned to APC 5361 based on clinical and resource homogeneity to the procedures in the APC 5361.
After consideration of the public comment we received, we are finalizing our proposal without modification to reassign CPT codes 49593, 49595 and 49615 to APC 5342 (Level 2 Abdominal/Peritoneal/Biliary and Related Procedures). We are also finalizing our proposal without modification to continue to assign CPT codes 49650 and 49651 to APC 5361 (Level 1 Laparoscopy and Related Services). Table 91 shows the finalized status indicator and APC assignment for the procedure code. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94096)37. Imaging of Retina for Detection or Monitoring of Disease, CPT Code 92229 (APC 5733)
CPT code 92229 (Imaging of retina for detection or monitoring of disease; point-of-care autonomous analysis and report, unilateral or bilateral) is performed to screen patients with diabetes for signs of diabetic retinopathy and other eye diseases. The code was established on January 1, 2021 and assigned to APC 5733 (Level 3 Minor Procedures). The code was assigned to Level 3 Minor Procedures because the service had clinical and resource similarity to long-established CPT code 92227 (Imaging of retina for detection or monitoring of disease; with remote clinical staff review and report, unilateral or bilateral) which also is assigned to APC 5733 (Level 3 Minor Procedures).
In the CY 2023 claims data, there were 370 (single frequency) claims for CPT code 92229 and the geometric mean for the service was $28.53. For CY 2025, we proposed to maintain CPT code 92229 in APC 5733 (Level 3 Minor Procedures) and wait for more claims data to be available before adjusting the payment rates for this service.
Comment: We received several comments supporting our decision to maintain CPT 92229 in APC 5733 (Level 3 Minor Procedures). Commenters noted that this service provides potentially sight saving technology to beneficiaries with diabetes in rural and underserved communities.
Response: We thank the commenters for their input.
Comment: We received one comment requesting that CPT code 92229 be removed from the Deficit Reduction Act (DRA) cap.
Response: We thank the commenter for their input, but we note that this comment is out of scope for purposes of this CY 2025 OPPS/ASC final rule with comment period.
After consideration of the public comments we received, we are finalizing our proposal without modification to assign CPT code 92229 to APC 5733. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
38. Implantable Cardiac Alert System, CPT Codes 0525T and 0527T (APCs 5224 and 5222)
The implantable cardiac alert system is a cardiac monitoring and alerting device to detect acute coronary syndrome (ACS) events and alerts the patient to seek urgent medical attention. The CPT codes 0525T (Insertion or replacement of intracardiac ischemia monitoring system, including testing of the lead and monitor, initial system programming, and imaging supervision and interpretation; complete system (electrode and implantable monitor)) and 0527T (Insertion or replacement of intracardiac ischemia monitoring system, including testing of the lead and monitor, initial system programming, and imaging supervision and interpretation; implantable monitor only) became effective January 1, 2019.
For CY 2025, the OPPS payment rates are proposed based on available CY 2023 claims data. For the CY 2025 OPPS/ASC proposed rule, based on our ( printed page 94097) analysis of claims data, we found a total of 15 single frequency claims and an estimated geometric mean cost of $3,244 for CPT code 0525T and a total of three single frequency claims and an estimated geometric mean cost of $3,433 for CPT code 0527T. For CY 2025, we proposed to continue to assign CPT code 0525T to Level 3 Pacemaker and Similar Procedures (APC 5223) and CPT code 0527T to Level 2 Pacemaker and Similar Procedures (APC 5222).
Comment: One Commenter disagreed with our APC's assignments of CPT codes 0525T and 0527T, suggesting that we did not account for the cost of the device represented by HCPCS code C1833 in the proposed rule. The commenter also stated that we erroneously assigned the status indicator of “H” to HCPCS code C1833 in the CY 2025 OPPS/ASC proposed rule. The commenter noted the Transitional Pass-Through period for C1833 expires December 31, 2024, and that HCPCS code C1833 should have been assigned the status indicator of “N” for CY 2025.
Additionally, the commenter argued that the proposed payment rates for CPT codes 0525T and 0527T are insufficient to cover the procedures' cost because we did not account for the device cost in the proposed CY 2025 payment rates. They claimed that the procedures represented by 0525T and 0527T are similar procedures and should both be assigned to APC 5231 (Level 1 ICD and Similar Procedures).
Response: We appreciate the commenter pointing out that HCPCS code C1833's Transitional Pass-through Status expires December 31, 2024. Therefore, we are revising the HCPCS code C1833's status indicator to “N” (packaged) for CY 2025.
We agree with the commenter the CY 2025 proposed APC assignments for 0525T and 0527T erroneously omitted the device cost (HCPCS code C1833) associated with these procedures. With the updated final rule data including the HCPCS code C1833 device cost, the revised geometric mean cost for CPT code 0525T is approximately $15,167 and the geometric mean cost for 0527T is approximately $7,242. Therefore, we believe an appropriate APC assignment for CPT code 0525T would be APC 5224 (Level 4 Pacemaker and Similar Procedures); and an appropriate assignment for CPT code 0527T would be APC 5222 (Level 2 Pacemaker and Similar Procedures).
We disagree with the commenter that CPT code 0525T is similar clinically and in resource costs to CPT code 0527T. We note that CPT code 0525T is for insertion of the complete system while CPT code 0527T is for insertion of the implantable monitor only.
After consideration of the public comments we received, we are finalizing a revision to our proposed APC assignment for CPT code 0525T and assigning this code to APC 5224 (Level 4 Pacemaker and Similar Procedures). We are finalizing the assignment of CPT code 0527T to APC 5222 (Level 2 Pacemaker and Similar Procedures) as proposed. Please refer to Table 92 for the final OPPS APC and status indicator assignment for CPT codes 0525T, 0527T and C1833 for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 94098)39. Implantable Glucose Monitoring System, CPT Codes 0446T and 0448T (APC 5054)
In 2017, the AMA CPT Editorial Panel established CPT codes 0446T and 0448T. CPT code 0446T (Creation of subcutaneous pocket with insertion of implantable interstitial glucose sensor, including system activation and patient training) and CPT code 0448T (Removal of implantable interstitial glucose sensor with creation of subcutaneous pocket at different anatomic site and insertion of new implantable sensor, including system activation) describe an implantable glucose sensor for diabetic patients. The current codes are used to describe both 90-day and 180-day sensors. The FDA recently approved a 365-day sensor that is not currently on the market.
For CY 2025, we proposed to maintain both CPT codes in APC 5054 (Level 4 Skin Procedures) with a status indicator of “T” (Procedure or Service, Multiple Procedure Reduction Applies; Paid under OPPS; separate APC payment.).
Comment: We received one comment from the manufacturer requesting that we reassign CPT codes 0446T and 0448T from APC 5054 (Level 4 Skin Procedures) to APC 1531 (New Technology—Level 31 ($6501-$7000)) with a payment of $6,750.50 to reflect the increased expense of the implantable sensor that has quadrupled the life of the 90-day sensor. The manufacturer reiterated the value of the longer sensor duration, including fewer insertion and removal procedures and increased adherence to therapy.
Response: We appreciate the request of the commenter and understand the implicated value that a longer life sensor brings to Medicare beneficiaries. Because the current CPT codes do not designate the increased battery life of the sensor, we believe it is more appropriate to create 2 new HCPCS G codes effective January 1, 2025 to describe the implantable interstitial glucose sensor with a 365-day battery life than to make APC reassignments and potentially overpay for the current codes that could be used to describe the 90-day, 180-day, or 365-day sensor. Specifically, we created the following G codes to describe the 365-day sensor:
- G0564 (Creation of subcutaneous pocket with insertion of 365 day implantable interstitial glucose sensor, including system activation and patient training); and
- G0565 (Removal of implantable interstitial glucose sensor with creation of subcutaneous pocket at different anatomic site and insertion of new 365 day implantable sensor, including system activation).
The manufacturer asserts the supply costs of the 365-day system will be equivalent to two 180-day systems or four 90-day systems. While we agree that there would be inherently increased costs to the new technology of a 365-day system, we do not agree that those costs would be 4 times more than the 90-day system.
After reviewing the device and procedure and obtaining input from our medical officers, we are assigning HCPCS codes G0564 and G0565 to APC 1561 (New Technology—Level 24 ($3001-$3500)) with a payment rate of $3,250.50. ( printed page 94099)
We currently have very little claims information available for CPT codes 0446T and 0448T as there has only been 1 single frequency claim for CPT 0448T (with a geometric mean cost of around $1,320) and no single frequency claims for 0446T. The geometric mean cost for APC 5054 (Level 4 Skin Procedures) is around $1,850 so we believe APC 5054 is an appropriate assignment. We will continue to monitor these codes as we gather adequate claim information and other relevant clinical information that would be useful in determining appropriate APC assignments.
In summary, after consideration of the public comment, we are finalizing our proposal, without modification, to assign CPT code 0446T and 0448T to APC 5054 (Level 4 Skin Procedures) for CY 2025 and finalizing APC assignments for G0564 and G0565. The final CY 2025 payment rate for these codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
40. Integrated Sacral Neurostimulators, CPT Code 0786T
Effective January 1, 2024, the CPT Editorial Panel separated integrated from non-integrated ( i.e., traditional) sacral neurostimulator procedure by establishing new CPT code, 0786T to report procedures using integrated sacral neurostimulator devices, while CPT code 64590 was updated to reflect the use of traditional technology:
- 0786T: Insertion or replacement of percutaneous electrode array, sacral, with integrated neurostimulator, including imaging guidance, when performed.
- 64590: Insertion or replacement of peripheral, sacral, or gastric neurostimulator pulse generator or receiver, requiring pocket creation and connection between electrode array and pulse generator or receiver
In the CY 2025 OPPS/ASC proposed rule Addendum B, we proposed to continue to assign CPT code 0786T to APC 5463 (Level 3 Neurostimulator and Related Procedures) with a proposed payment rate of $13,029.81. We received several comments related to the proposal.
Comment: One commenter disagreed with our proposal. The commenter stated that the proposed assignment for this technology would reduce reimbursement and limit access for Medicare and Medicaid patients seeking care in the outpatient surgery setting. The commenter thought that this decision may have been based on the concept that the Neuspera system is an integrated system, requiring no intra-operative connection. Although physical connections are not required, they stated a considerable amount of time and effort is expended on creating wireless connections and re-positioning the device once partially deployed.
One commenter asserted that the integrated sacral neuromodulation system (SNM) represents a disruptive technology that should improve patient satisfaction, with similar efficacy, lower risk, and as a “one and done” procedure that will reduce the need for 2-3 devices per patient to only one Neuspera devices per patient. The commenter stated the lead placement takes the majority of the procedure time, while the implantable pulse generator (IPG) placement requires very little time and expertise. Given their claim that the integrated system is nearly identical to the current non-integrated systems, they requested it should be paid on par with the current non-integrated system.
Other commenters maintained that the integrated SNM system anticipated to reach the market in 2025 has time and resources substantially equivalent to those for traditional SNM systems. Given this, they noted keeping CPT code 0786T in a lower APC with a lower device-intensive designation than traditional SNM systems will create an economic barrier and restrict hospitals and ASCs from offering this new less invasive form of SNM to patients.
Several commenters urged CMS to reassign CPT code 0786T to APC 5464 (Level 4 Neurostimulator and Related Procedures) to reflect the most accurate procedure and cost information related to this novel sacral neuromodulation system. They stated that there is a high degree of parity and similarity in time, work, staffing, costs, and resources required for the Neuspera procedure, compared to traditional sacral neuromodulation systems, which are currently assigned to APC 5464. The commenters also pointed out that if CMS is not willing to reassign CPT code 0786T to APC 5464, status indicator “E1” may provide CMS the ability to postpone APC assignment until there is FDA cleared technology to make a decision. Currently no FDA approved “integrated” sacral neuromodulation system exists.
Response: We proposed to continue to assign CPT code 0786T to APC 5463 for CY 2025, based on input from our clinicians and based on our evaluation of the code. We inadvertently listed Neuspera integrated sacral nerve stimulation system as receiving FDA-approval. However, it is still pending FDA approval and therefore, we agree with most of the commenters who recommended to reassign CPT code 0786T to status indicator “E1” until FDA approval has been received.
In summary, after consideration of the public comments that we received, we are finalizing our proposal with modification and are reassigning CPT code 0786T from APC 5463 to status indicator “E1” (Not covered by any Medicare outpatient benefit category; Statutorily excluded by Medicare; Not reasonable and necessary; Not paid by Medicare when submitted on outpatient claims (any outpatient bill type)) for CY 2025 because this service is not yet FDA approved.
We refer readers to Addendum D1 of this final rule with comment period for the status indicator (SI) meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
41. Laparoscopic Appendectomy, CPT Code 44970 (APC 5361)
CPT code 44970 (Laparoscopy, surgical, appendectomy) describes the procedure for laparoscopic appendectomy. Using CY 2023 claims data, CPT code 44970 had a geometric mean cost of $6,499.86 in the CY 2025 OPPS/ASC proposed rule. For CY 2025, we believed this was still appropriately assigned to APC 5361 (Level 1 Laparoscopy and Related Services), which had a proposed payment rate of $5,798.13. Therefore, we proposed to continue assigning CPT code 44970 to APC 5361.
Comment: We received one comment requesting that we reassign CPT code 44970 to APC 5342 (Level 2 Abdominal/Peritoneal/Biliary and Related Procedures). The commenter argued that CPT code 44970 had clinical homogeneity and resource utilization similar to CPT code 44950 (Appendectomy), which is currently assigned to APC 5342.
Response: We thank the commenter for their input. CPT code 44970 has an updated GMC of $6,404.30 in the final rule data. While this is closer to the payment rate for APC 5342, after clinical review of this procedure, we did not find APC 5342 to be the clinically appropriate APC family for CPT code 44970. We continue to believe that this procedure belongs in the Laparoscopy and Related Procedures family.
After consideration of the public comment we received, we are finalizing our proposal without modification to continue to assign CPT code 44970 to APC 5361 (Level 1 Laparoscopy and Related Services). Table 93 shows the ( printed page 94100) finalized status indicator and APC assignment for the procedure code. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
42. Litholapaxy Procedure, CPT Code 52318 (APC 5374)
Litholapaxy is a procedure that involves crushing bladder stones and extracting the fragmented stones. CPT code 52318 (Litholapaxy: crushing or fragmentation of calculus by any means in bladder and removal of fragments; complicated or large (over 2.5 cm)) became effective January 1, 1985.
For CY 2025, OPPS payment rates are proposed based on available CY 2023 claims data. For CY 2025, based on our analysis of claims data, we found a total of 4,215 single frequency claims and a geometric mean cost of approximately $4,298 for CPT code 52318. For CY 2025, we proposed to continue to assign CPT code 52318 to Level 4 Urology and Related Services (APC 5374) with a proposed payment rate of approximately $3,438.
Comment: One commenter requested the APC assignment of CPT code 52318 to Level 5 Urology and Related Services (APC 5375). The commenter stated that the complexity and resources required for CPT code 52318 is comparable to CPT code 52601 (Transurethral electrosurgical resection of prostate, including control of postoperative bleeding, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy are included)), which is assigned to APC 5375. Moreover, the commenter alleged that CPT code 52318 is two times greater than the lowest geometric mean of CPT code 54670 (Suture or repair of testicular injury) in APC 5375, which is a two times rule violation.
Response: We appreciate the commenter's recommendation regarding the APC assignment of CPT code 52318, but we disagree that the complexity and resources required for CPT code 52318 are comparable to CPT code 52601. Based on the claims data, we believe that CPT code 52318 fits more appropriately in APC 5374, rather than in APC 5375, based on resource cost and clinical similarity to the procedures in APC 5374.
Furthermore, CPT codes 52318 and 54670 are currently assigned to APC 5374 (Level 4 Urology and Related Services). We note that APC 5374 does not currently have a 2 times rule violation in the final rule data. In addition, both CPT codes 52318 and 54670 do not meet the requirements for cost significance for 2 times rule violation purposes, under the requirements described in section III.B.2. of this final rule with comment period. We have reviewed the codes' geometric mean cost based on the available CY 2023 claims data within APC 5374 and believe that their current APC assignment continues to be appropriate.
After consideration of the public comment we received, we are finalizing our proposal without modification for CPT code 52318 to be reassigned to APC 5374 (Level 4 Urology and Related Services). Please refer to Table 94 for the final OPPS APC and status indicator assignment for CPT code 52318 for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
43. LIXELLE® Apheresis
LIXELLE® β2-microglobulin Apheresis Column is indicated for use in the treatment of dialysis-related amyloidosis (DRA), a disease that affects people with end-stage renal disease (ESRD). DRA is a metabolic disorder from the failure of the kidney to filter and remove β2-microglobulin, typically from chronic hemodialysis (typically 5 years or longer). The LIXELLE® device is used in an apheresis procedure that selectively removes β2-microglobulin from circulating blood and is used pursuant to a physician prescription in conjunction with hemodialysis. It is intended to be used at each hemodialysis session (that is, frequency ( printed page 94101) of treatment is expected to be 3 times per week). In March 2015, FDA approved LIXELLE® as a Class III Humanitarian Use Device (HUD) with an approved Humanitarian Device Exemption (HDE). For CY 2025, there are currently no specific HCPCS or CPT codes that represent the LIXELLE® apheresis service.
Comment: We received one comment, from the manufacturer, requesting CMS provide reimbursement for LIXELLE® apheresis procedure under the ESRD payment system or the OPPS to benefit patients with DRA. Specifically, the commenter requested that CMS provide separate payment under the OPPS for the procedure, and offered the following options:
(1) establish a new HCPCS C code or G code for the LIXELLE® apheresis procedure and assign the code to APC 5242 (Level 2 Blood Product Exchange and Related Services); or
(2) pay separately for the apheresis procedure used with the LIXELLE® device through CPT code 36516 (Therapeutic apheresis with extracorporeal immunoadsorption, selective adsorption or selective filtration and plasma reinfusion), assigned to APC 5243 (Level 3 Blood Product Exchange and Related Services) for CY 2025, and require the use of a modifier or add-on code when the LIXELLE® apheresis procedure is billed to reduce the procedure payment APC 5242 (Level 2 Blood Product Exchange and Related Services) payment rate; or
(3) allow separate payment for the dialysis performed as part of LIXELLE® apheresis procedure through HCPCS code G0257 (Unscheduled or emergency dialysis treatment for an ESRD patient in a hospital outpatient department that is not certified as an ESRD facility), which is assigned to APC 5401 (Dialysis) for CY 2025 and require the use of a modifier or add-on code to provide additional payment beyond that provided for APC 5401; or
(4) assign CPT 90947 (Dialysis procedure other than hemodialysis ( e.g., peritoneal dialysis, hemofiltration, or other continuous renal replacement therapies) requiring repeated evaluations by a physician or other qualified health care professional, with or without substantial revision of dialysis prescription) to APC 5242 (Level 2 Blood Product Exchange and Related Services).
The commenter also noted the FDA has approved a clarification to the Instructions for Use (IFU) since last year's comment and rulemaking. The IFU for LIXELLE® expands the list of personnel who can supervise the patient while connected to the extracorporeal system to include not only a physician, but a qualified health care professional such as a physician assistant, nurse practitioner, or registered nurse who is qualified to supervise hemodialysis procedures.
Response: We appreciate the thoughtful recommendations and will consider them for future rulemaking. We note this complex, ongoing issue is still under consideration and continues to merit a thorough evaluation to ensure an appropriate Medicare benefit category and payment pathway for the service is determined.
44. Low Ejection Fraction AI-ECG Service, CPT Codes 0764T and 0765T (APC 5734)
The low ejection fraction AI-ECG Service is intended to aid in screening for Left Ventricular Ejection Fraction (LVEF) less than or equal to 40 percent in adults at risk for heart failure. The low ejection fraction AI-ECG Service is an algorithmic SaaS service that aims to detect hidden diseases using algorithms to enhance and improve interventional procedures through real-time AI insights. CPT codes 0764T (Assistive algorithmic electrocardiogram risk-based assessment for cardiac dysfunction ( e.g., low ejection fraction, pulmonary hypertension, hypertrophic cardiomyopathy); related to concurrently performed electrocardiogram (List separately in addition to code for primary procedure)) and 0765T (Assistive algorithmic electrocardiogram risk-based assessment for cardiac dysfunction ( e.g., low ejection fraction, pulmonary hypertension, hypertrophic cardiomyopathy); related to previously performed electrocardiogram) became effective January 1, 2023 and were assigned to status indicator “E1” (Not paid by Medicare when submitted on outpatient claims). For CY 2025, we proposed that CPT codes 0764T and 0765T continue to be assigned to status indicator E1.
Comment: One Commenter provided documentation that AI-ECG has been FDA approved on September 28, 2023. The commenter requested separate payment for the low ejection fraction AI-ECG service for CY 2025. In addition, the commenter referenced our CY 2023 packaging exemption for SaaS add-on codes policy and requested separate payment for the add-on CPT code 0764T.
Response: We appreciate the commenter providing the FDA documentation for the AI-ECG approval. We agree that CPT code 0765T should be payable for CY 2025. Consistent with our SaaS Add-on Codes policy, CPT code 0764T, the add-on code for the low ejection fraction AI-ECG service would be assigned to the identical APC and status indicator to CPT code 0765T, the standalone code for the same service.
After consideration of the public comments we received, we are finalizing revised status indicator and APC assignments for CPT codes 0764T and 0765T. Please refer to Table 95 for the final OPPS APC and status indicator assignment for HCPCS codes 0764T and 0765T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 94102)45. Lower Esophageal Myotomy (POEM), CPT 43497 (APC 5331)
CPT code 43497 (Lower esophageal myotomy, transoral ( i.e., peroral endoscopic myotomy [POEM]) describes a minimally invasive endoscopic procedure that treats swallowing disorders caused by muscle problems in the esophagus.
For CY 2025, we proposed to assign CPT code 43497 to APC 5331 (Complex GI Procedures) with a proposed payment rate of around $5,838.
Comment: We received one comment requesting that we reassign CPT code 43497 to APC 5361 (Level 1 Laparoscopy and Related Services) with a proposed payment of around $5,798. The commenter stated that the procedure utilizes techniques that are surgical in nature and that APC 5361 would be a more appropriate APC assignment, based on clinical homogeneity and resource utilization with other services in APC 5361.
Response: The OPPS is a prospective payment system that provides payment for groups of services that share clinical and resource use characteristics. Based on our understanding of the service and input from our medical advisors, we do not agree that CPT code 43497 has clinical and resource homogeneity to other services in APC 5361 such that it should be assigned to APC 5361.
In summary, after consideration of the public comment, we are finalizing our proposal, without modification, to assign CPT code 43497 to APC 5331 for CY 2025. The final CY 2025 payment rate for the code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
46. Magnetic Resonance Exam Safety Procedures, CPT Codes 76014 Through 76019 (APCs 5521, 5523, 5731, 5733, and 5742)
For CY 2025, the AMA CPT Editorial Panel created six codes to report magnetic resonance (MR) examination safety procedures, effective January 1, 2025. The new codes did not have a predecessor code or a one-to-one match to an existing code. When determining the proposed status indicators and APC assignments for CY 2025, we reviewed the clinical and resource characteristics of the procedures, considered input from our medical advisors, and reviewed existing APC classifications to identify similar and closely related procedures. The codes, their long descriptors, and their proposed CY 2025 OPPS status indicators and APC assignments are listed in Table 96.
We note that CPT codes 76014, 76015, 76016, 76017, 76018, and 76019 were listed as placeholder codes 7XX00, 7XX01, 7XX02, 7XX03, 7XX04, and 7XX05 respectively, in OPPS Addendum B and Addendum O that were released with the CY 2025 OPPS/ASC proposed rule. Because we had not received the final CPT code numbers from AMA for the new codes that would be effective January 1, 2025, in time for the publication of the proposed rule, we listed the new CPT codes with their respective placeholder codes in OPPS Addendum B and Addendum O.
( printed page 94103)Comment: Some commenters disagreed with the proposed assignments for the five separately payable codes and provided ( printed page 94104) recommendations for APC reassignments. They stated that the proposed APC assignments for the new codes would be insufficient to cover the cost of furnishing the procedures and would impact beneficiary access. Commenters detailed that the proposed APC assignments for CPT codes 76017, 76018, and 76019 did not reflect the facility costs associated with these procedures, including additional staff time, MR room time and clinical resources for planning, preparation, and patient positioning.
The commenters requested that CMS reassign CPT codes 76014 (placeholder code 7XX00) and 76016 (placeholder code 7XX05) to APC 5611(Level 1 Therapeutic Radiation Treatment Preparation) with a proposed payment rate of $89.12. Commenters requested that CMS reassign CPT codes 76017 (placeholder code 7XX03) and 76019 (placeholder code 7XX05) to APC 5612 (Level 2 Therapeutic Radiation Treatment Preparation; proposed payment rate of around $370). Commenters shared that CPT codes 76017 (placeholder code 7XX03) and 76019 (placeholder code 7XX05) require additional staff time and clinical resources for planning, preparation, and positioning which warrants reassignment to APC 5612.
One commenter requested that CMS reassign CPT code 76018 (placeholder code 7XX04) to APC 5612 (Level 2 Therapeutic Radiation Treatment Preparation; proposed payment rate of around $370), Another commenter requested a reassignment of CPT code 76018 to APC 5742 (Level 2 Electronic Analysis of Devices; proposed payment rate of around $92). The commenter stated that CPT code 76018 involves interrogation and programming of an implanted device to protect the device and patient against interactions with the MRI scanner and would therefore fit well within APC 5612.
Response: We thank the commenters for their feedback and recommendations. In determining the appropriate APC placement for new codes, we rely on input from a variety of sources, including, but not limited to, review of the resource costs and clinical similarity of the service to existing procedures; input from CMS medical advisors; and information from interested specialty societies. We evaluated the recommendations, modeled the suggestions, analyzed the cost results of the suggested APC reassignments, and received additional input from our medical advisors. Because we believe that the services assigned to the Therapeutic Radiation Treatment Preparation APC family have higher degrees of technicality, require higher levels of trained professional staff, and therefore have additional facility costs beyond those described by the CPT codes 76014 through 76019, we disagree with the commenter requests to assign these CPT codes to APCs 5611 and 5612. And while we recognize that there is not currently a one-to-one match to crosswalk to the new codes, we based the proposed APC assignments for CPT codes 76014, 76016, and 76019 on crosswalks to CPT codes that have similar service and resource elements, as well as required staff, to the new codes.
Specifically, we continue to believe that:
- CPT code 76014 is similar to the service described by CPT code 0521T (Interrogation device evaluation (in person) with analysis, review and report, includes connection, recording, and disconnection per patient encounter, wireless cardiac stimulator for left ventricular pacing), which is assigned to APC 5731.
- CPT code 76016 is similar to the service described by CPT code 77091 (Trabecular bone score (tbs), structural condition of the bone microarchitecture; technical calculation only), which is assigned to APC 5521.
- CPT code 76019 is similar to CPT code 29520 (Strapping; hip), which is assigned to APC 5733.
After consideration of the public comments, our review of CPT code 76017 identified procedures in APC 5523 (Level 3 Imaging without Contrast) that we believe better align with the clinical characteristics and resources required for CPT code 76017, including CPT code 73718 (Magnetic resonance ( e.g., proton) imaging, lower extremity other than joint; without contrast material(s)). After further input from CMS medical advisors, we believe that it may be appropriate to assign CPT code 76017 to APC 5523.
Additionally, we believe that we have identified additional procedures in APC 5742 that share similar resource and clinical characteristics with CPT code 76018. Specifically, we believe that CPT code 76018 shares resource and clinical similarities to the service described by CPT code 29125 (Application of short arm splint (forearm to hand); static). Based on our assessment, we believe it appropriate to assign CPT code 76018 to APC 5742, based on shared resource and clinical characteristics.
In summary, we believe that the below assignment of the new codes in Table 97 are the most appropriate, in terms of clinical and resource homogeneity. We note that we review our claims data on an annual basis to establish the OPPS payment rates and we will reevaluate the status indicator and APC assignments annually.
After consideration of the public comments we received, we are finalizing our proposal with modification for CPT codes 76014 through 76019. Table 97 shows the finalized status indicators and APC assignments for all of the procedure codes. The final CY 2025 payment rates for these codes can be found in Addendum B to this final rule with comment period. We also refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
( printed page 94105) ( printed page 94106)47. MindMotion GO Neurorehabilitative Remote Therapy Service, CPT Code 0733T (APC 1505)
Effective July 1, 2022, the AMA established CPT code 0733T (Remote real-time, motion capture-based neurorehabilitative therapy ordered by a physician or other qualified health care professional; supply and technical support, per 30 days) to describe the procedure associated with the use of MindMotion GO, which provides remote real-time, motion capture-based neurorehabilitative therapy. In the CY 2025 OPPS/ASC proposed rule, CMS proposed to continue to assign CPT code 0733T to APC 5741 (Level 1 Electronic Analysis of Devices) with a proposed payment rate of $36.90, due to its similarity to CPT code 98976 (Remote therapeutic monitoring ( e.g., therapy adherence, therapy response); device(s) supply with scheduled ( e.g., daily) recording(s) and/or programmed alert(s) transmission to monitor respiratory system, each 30 days).
Comment: We received one comment from the manufacturer of MindMotion GO requesting that CMS reassign CPT code 0733T to APC 1510 (New Technology—Level 10 ($801-$900)). The commenter stated that the resource costs of this service are $849 and that the MindMotion GO technology will not be commercially available until early 2025, so there is currently no claims data available to assign this service to a clinical APC. The commenter also believed that CPT code 0733T was not clinically homogeneous with APC 5741 as the procedures in the APC are for monitoring services, while CPT code 0733T also describes a device used primarily for treatment of neurological conditions.
Response: We thank the commenter for their input. We agree with the commenter that the current assignment of APC 5741 would result in too low of a payment for CPT code 0733T. After further review, we believe this procedure is similar to the procedure described by CPT code 0693T (Comprehensive full body computer-based markerless 3d kinematic and kinetic motion analysis and report), which describes technology for DARI Motion. CPT code 0693T is currently assigned to APC 1505 (New Technology—Level 5 ($301—$400)). Therefore, we are reassigning CPT code 0733T to APC 1505 until we are able to obtain claims data for this procedure.
After consideration of the public comment we received, we are finalizing our proposal with modification to reassign CPT code 0733T to APC 1505 (New Technology—Level 5 ($301-$400)). Table 98 shows the finalized status indicator and APC assignment for the procedure code. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
48. Musculoskeletal Procedures (APCs 5111 Through 5116)
Prior to the CY 2016 OPPS, payment for musculoskeletal procedures was primarily divided according to anatomy and the type of musculoskeletal procedure. As part of the CY 2016 reorganization to better structure the OPPS payments to utilize prospective payment packages, we consolidated these individual APCs so that they became a general Musculoskeletal APC series (80 FR 70397 and 70398).
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59300), we continued to apply a six-level structure for the Musculoskeletal APCs because doing so provided an appropriate distinction for resource costs at each level and provided clinical homogeneity. However, we indicated that we would continue to review the structure of these APCs to determine whether additional granularity would be necessary. In the CY 2019 OPPS proposed rule (83 FR 37096), we recognized that commenters had previously expressed concerns regarding the granularity of the current APC levels and, therefore, requested comment on the establishment of additional levels. Specifically, we solicited comments on the creation of a new APC level between the current Level 5 and Level 6 within the Musculoskeletal APC series. While some commenters suggested APC reconfigurations and requested changes to APC assignments, many commenters requested that we maintain the current six-level structure and continue to monitor the claims data as they become available. Therefore, in the CY 2019 OPPS/ASC final rule with comment period, we maintained the six-level APC structure for the Musculoskeletal Procedures APCs (83 FR 58920 and 58921).
For CY 2025, based on the claims data available for the CY 2024 OPPS/ASC proposed rule, we continued to believe that the six-level APC structure for the Musculoskeletal Procedures APC series is appropriate, and we proposed to maintain it for the CY 2025 OPPS update.
Comment: Several commenters supported the assignment of HCPCS code 0737T (Xenograft implantation into the articular surface) to APC 5115 (Level 5 Musculoskeletal Procedures) and recommended that we finalize the assignment in the CY 2025 OPPS.
Response: We appreciate the commenters' support of our proposal. We agree and are finalizing the proposed assignment of HCPCS code 0737T (Xenograft implantation into the articular surface) to APC 5115 (Level 5 Musculoskeletal Procedures). We refer ( printed page 94107) readers to section III.E.67 of this final rule with comment period for a detailed discussion of this issue.
Comment: A commenter supported the continued assignment of HCPCS code C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer ( e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed) to APC 5115 (Level 5 Musculoskeletal Procedures).
Response: We appreciate the commenter's support of our proposal. We agree and are finalizing the proposed assignment of HCPCS code C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer ( e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed) to APC 5115 (Level 5 Musculoskeletal Procedures). We refer readers to section III.E.11. of this final rule with comment period for a detailed discussion of this issue.
Comment: A few commenters recommended that CMS consider the creation of additional musculoskeletal APC levels. Commenters noted that additional APCs should be considered with the goal of reducing payment gaps and volatility, while aligning cost and clinical homogeneity in the APCs. One commenter recommended that an additional APC level be considered between the current levels 4 and 5 with costs ranging from $7,000 to $12,000. Another commenter requested that we create an additional Musculoskeletal APC level from codes currently in APC 5116 (Level 6 Musculoskeletal Procedures) with geometric mean costs above $22,000, while moving codes with geomean costs above $14,000 in the current level 5 APC to the level 6 APC.
Response: We appreciate the commenters' recommendations. We also appreciate the goal of developing APC levels that properly group procedures by clinical and resource similarity. At this time, we believe the six-level structure for the Musculoskeletal APCs that we proposed continues to provide an appropriate distinction between the resource costs at each level and clinical homogeneity and are therefore finalizing that six-level structure for the Musculoskeletal Procedures APC series in the CY 2025 OPPS. However, we will take these comments into consideration for future rulemaking and continue to monitor the claims data as they are available.
After consideration of the public comments, we are finalizing our proposal to maintain the six-level Musculoskeletal Procedures APC structure. We are also finalizing the assignment of HCPCS code 0737T (Xenograft implantation into the articular surface) to APC 5115 and HCPCS code C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer ( e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed) to APC 5115 for the CY 2025 OPPS.
49. Noncontact Near-Infrared (NIR) Spectroscopy, CPT 0640T (APC 5732)
Effective CY 2024, there are three codes that describe the service related to NIR spectroscopy: CPT codes 0640T (Noncontact near-infrared spectroscopy ( e.g., for measurement of deoxyhemoglobin, oxyhemoglobin, and ratio of tissue oxygenation), other than for screening for peripheral arterial disease, image acquisition, interpretation, and report; first anatomic site, 0859T (Noncontact near-infrared spectroscopy ( e.g., for measurement of deoxyhemoglobin, oxyhemoglobin, and ratio of tissue oxygenation), other than for screening for peripheral arterial disease, image acquisition, interpretation, and report; each additional anatomic site (list separately in addition to code for primary procedure)), and 0860T (Noncontact near-infrared spectroscopy ( e.g., for measurement of deoxyhemoglobin, oxyhemoglobin, and ratio of tissue oxygenation), for screening for peripheral arterial disease, including provocative maneuvers, image acquisition, interpretation, and report, one or both lower extremities). Only CPT code 0640T is separately paid under the OPPS. For CY 2025, CMS proposed to assign CPT code 0640T to APC 5732 (Level 2 Minor Procedures) and status indicator “S” with a $39 payment rate.
Comment: We received one comment regarding the proposed APC assignment for CPT code 0640T. The commenter requested that we reassign 0640T to APC 5722 (Level 2 Imaging without contrast) with a proposed payment rate of approximately $310 for CY 2025 based on a crosswalk to CPT code 0598T (Noncontact real-time fluorescence wound imaging, for bacterial presence, location, and load, per session; first anatomic site ( e.g., lower extremity)). The commenter explained that CPT code 0598T is an appropriate crosswalk code because it yields diagnostic images without contrast, is newer, and utilizes a Category III CPT code.
Response: We thank the commenter for their input. First, we note that prior to the AMA's CPT Editorial Panel descriptor revision for CPT code 0640T, the technical service associated with NIR spectroscopy was described by CPT code 0641T, which was assigned to APC 5732 for CY 2023, prior to being deleted for CY 2024. Under the OPPS, the predecessor code for CPT code 0640T is CPT code 0641T. We note for CY 2025, OPPS payment rates are proposed based on available CY 2023 claims data. Based on our analysis of the claims data for this final rule with comment period, we found a geometric mean cost of about $16 for predecessor CPT code 0641T, based on 32 single frequency claims (out of 445 total claims). In contrast, we found a geometric mean cost of approximately $170 for CPT code 0598T based on 1109 single frequency claims (out of 2531 total claims). Based on the data, the resource cost associated with noncontact real-time fluorescence imaging (CPT code 0598T), is significantly higher compared to noncontact near-infrared (NIR) spectroscopy (CPT code 0640T/0641T). We disagree that the resource cost for NIR spectroscopy is similar to noncontact real-time fluorescence imaging. Therefore, we do not agree that both technologies should be placed in the same APC. We believe that the code describing NIR spectroscopy, specifically, CPT code 0640T, is appropriately placed in APC 5732.
In consideration of the public comment we received, we are finalizing our proposal without modification for CPT code 0640T. Specifically, for CY 2025, we are finalizing our proposal to assign CPT code 0640T to APC 5732. The final CY 2025 OPPS payment rate for all the codes payable under the OPPS can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendum D1 is available via the internet on the CMS website.
50. Percutaneous Coronary Revascularization Services by Intracoronary Antiproliferative Drug Delivery, CPT Codes 0913T and 0914T (APC 5192)
For the July 2024 update, the AMA's CPT Editorial Panel established two Category III codes to describe percutaneous coronary revascularization services by intracoronary antiproliferative drug delivery, performed for occlusive disease of the coronary vessels (major coronary arteries, coronary artery branches) using ( printed page 94108) drug-delivery balloon ( e.g., drug-coated, drug-eluting) for intracoronary antiproliferative drug delivery:
- CPT code 0913T (placeholder code XX23T) (Percutaneous transcatheter therapeutic drug delivery by intracoronary drug-delivery balloon (e.g., drug-coated, drug-eluting), including mechanical dilation by nondrug-delivery balloon angioplasty, endoluminal imaging using intravascular ultrasound (IVUS) or optical coherence tomography (OCT) when performed, imaging supervision, interpretation, and report, single major coronary artery or branch)); and,
- CPT code 0914T (placeholder code XX40T) (Percutaneous transcatheter therapeutic drug delivery by intracoronary drug-delivery balloon (e.g., drug-coated, drug-eluting) performed on a separate target lesion from the target lesion treated with balloon angioplasty, coronary stent placement or coronary atherectomy, including mechanical dilation by nondrug-delivery balloon angioplasty, endoluminal imaging using intravascular ultrasound (IVUS) or optical coherence tomography (OCT) when performed, imaging supervision, interpretation, and report, single major coronary artery or branch (List separately in addition to code for percutaneous coronary stent or atherectomy intervention)).
In the CY 2025 OPPS/ASC proposed rule, we proposed to assign CPT code 0913T (placeholder code XX23T) to APC 5192 (Level 2 Endovascular Procedures; geometric mean cost of $5,771.29) based on clinical and resource similarity to other Percutaneous Coronary Intervention (PCI) procedures, including CPT code 92920 (Percutaneous transluminal coronary angioplasty; single major coronary artery or branch). Additionally, as an add-on code, we proposed to assign CPT code 0914T to status indicator “N.”
We note that a transitional device pass-through application was submitted for the AGENTTM Paclitaxel-Coated Balloon Catheter for CY 2025 to request separate payment for the device under the OPPS, in addition to the surgical CPT codes. We direct readers to section IV.A (Pass-Through Payment for Devices) of this final rule with comment period for a more detailed discussion of the transitional device pass-through applications and the status of the CY 2025 device pass-through application for the AGENTTM Paclitaxel-Coated Balloon Catheter.
We also note that a presenter provided information to the Panel on the description of the service and the cost of the AgentTM device and procedure at the August 28, 2024, HOP Panel Meeting. At the conclusion of the presentation, the presenter asked the Panel to request that CMS reassign CPT code 0913T from APC 5192 to APC 5193 (Level 3 Endovascular Procedures). Based on the information presented at the HOP Panel meeting, the HOP Panel recommended reassigning CPT 0913T to APC 5193.
Comment: We received several comments from interested parties, including the manufacturer, a trade association, and several practitioners. One commenter reiterated information shared during the 2024 HOP Panel. Commenters stated that coronary drug-delivery by drug-coated balloon (DCB) is a clinically similar procedure to a drug-eluting stent (DES) implant, which is reported with HCPCS code C9600 (Percutaneous transcatheter placement of drug eluting intracoronary stent(s), with coronary angioplasty when performed; single major coronary artery or branch) and is assigned to APC 5193 (Level 3 Endovascular Procedures). The manufacturer also provided a cost analysis of one facility's Medicare outpatient claims for coronary DCB. Their provided cost analysis included eleven claims and indicated that the geometric mean cost of those eleven claims was $18,935.69.
Response: We thank the commenters for their comments. After consideration of the public comments, we continue to believe that CPT code 0913T is appropriately assigned to APC 5192. We note that the long descriptor for CPT code 0913T describes a service similar to other codes in APC 5192. After review of the service and input from our CMS medical officers, we believe that CPT code 92920 (Percutaneous transluminal coronary angioplasty; single major coronary artery or branch) which is assigned to APC 5192 is a good crosswalk code for CPT code 0913T because it is clinically similar.
In summary, we are finalizing our proposal to assign CPT code 0913T to status indicator “J1,” and APC 5192. The final CY 2025 payment rate for CPT code 0913T can be found in Addendum B to this final rule with comment period. We also refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addendums B and D1 are available via the internet on the CMS website.
51. Potential Two-Times Rule Violations (APCs 5302, 5415, 5092, and 5114)
We received comments on the APC assignments we proposed in the CY 2025 OPPS/ASC proposed rule, which commenters believe result in 2-times rule violations.
Endoscopic Esophageal Procedures
CPT code 43254 (Esophagogastroduodenoscopy, flexible, transoral; with endoscopic mucosal resection) with a geometric mean cost of around $2,632; CPT code 43270 (Esophagogastroduodenoscopy, flexible, transoral; with ablation of tumor(s), polyp(s), or other lesion(s) (includes pre- and post-dilation and guide wire passage, when performed)) with a geometric mean cost of around $2,985; and CPT code 43275 (Endoscopic retrograde cholangiopancreatography (ERCP); with removal of foreign body(s) or stent(s) from biliary/pancreatic duct(s)) with a geometric mean cost of around $2,886 describe endoscopic procedures that are performed on the esophagus. For the CY 2025 OPPS/ASC proposed rule, we proposed to assign all 3 procedures to APC 5302 (Level 2 Upper GI Procedures), which had a proposed a payment rate of around $1,884.
Comment: One commenter requested that we reassign CPT codes 43254, 43270, and 43275 to APC 5303 (Level 3 Upper GI Procedures) with a proposed payment rate of around $3,648, due to the geometric mean costs of these codes being more than 2 times that of CPT code 0653T (Esophagogastroduodenoscopy, flexible, transnasal; with biopsy, single or multiple). CPT code 0653T has a geometric mean cost of around $1,212.
Response: We appreciate the commenter's request. We did not identify a 2-times rule violation for APC 5302. Since the release of the CY 2025 OPPS proposed rule, we have updated our 2-times analysis of claims from CY 2023 that are used to set rates for CY 2025. Our updated claims data found CPT code 0653T has fewer than 100 claims and does not meet the significance threshold for the two times rule evaluation, and therefore is not considered the lowest cost significant procedure for APC 5302.
Vaginal Hysterectomy Procedures
CPT code 58260 (Vaginal hysterectomy, for uterus 250 g or less) with a geometric mean cost of around $6,083 and CPT code 58262 (Vaginal hysterectomy, for uterus 250 g or less; with removal of tube(s), and/or ovary(s)) with a geometric mean cost of around $6,753 describe vaginal hysterectomy procedures. For the CY 2025 OPPS/ASC proposed rule, we proposed to assign both procedures to APC 5415 (Level 5 Gynecologic Procedures) with a ( printed page 94109) proposed payment rate of around $4,902.
Comment: A commenter requested that we reassign CPT codes 58260 and 58262 to APC 5416 (Level 6 Gynecologic Procedures) with a proposed payment rate of around $7,312, citing the geometric mean cost of CPT code 57550 (Excision of cervical stump, vaginal approach) which is around $1,879.
Response: We appreciate the commenter's request. We did not identify a 2-times rule violations for APC 5415. Our updated claims data found CPT code 57550 has fewer than 100 claims and does not meet the significance threshold for the 2-times rule evaluation, and therefore is not considered the lowest cost significant procedure for APC 5415.
Mastectomy Procedures
CPT code 19303 (Mastectomy, simple, complete) with a geometric mean cost of around $7,236 and CPT code 19307 (Mastectomy, modified radical, including axillary lymph nodes, with or without pectoralis minor muscle, but excluding pectoralis major muscle) with a geometric mean cost of around $7,412 describe mastectomy procedures. For the CY 2025 OPPS/ASC proposed rule, we proposed to assign both procedures to APC 5092 (Level 2 Breast/Lymphatic Surgery and Related Procedures) with a proposed payment rate of around $6,473.
Comment: Another commenter requested that we reassign CPT code 19303 and 19307 to APC 5093 (Level 3 Breast/Lymphatic Surgery and Related Procedures) with a proposed payment rate of around $9,417, citing the geometric mean cost of CPT code 38555 (Excision of cystic hygroma, axillary or cervical; with deep neurovascular dissection) which is around $3,475.
Response: We appreciate the commenter's request. We did not identify a 2-times rule violations for APC 5092. Our updated claims data found CPT code 38555 has fewer than 100 claims and does not meet the significance threshold for the two times rule evaluation and therefore is not considered the lowest cost significant procedure for APC 5092.
Arthrodesis Procedure
CPT code 28740 (Arthrodesis, midtarsal or tarsometatarsal, single joint), which has a geometric mean cost of around $11,058, describes an arthrodesis procedure performed in the foot. For the CY 2025 OPPS/ASC proposed rule, we proposed to assign this procedure to APC 5114 (Level 4 Musculoskeletal Procedures) with a proposed payment rate of around $7,090.
Comment: A commenter requested that CPT code 28740 be reassigned to APC 5115 (Level 5 Musculoskeletal Procedures) with a proposed payment rate of around $12,755, citing the geometric mean cost of CPT code 27385 which is around $5,616.
Response: We appreciate the commenter's request. Our updated claims data did not identify a 2-times rule violation for APC 5115. We also found that the geometric mean cost for CPT code 28740 is $11,074. This is less than two times the updated cost of the suggested code CPT code 27385, which is $5,616.
In summary, after consideration of these public comments, we are finalizing our proposal without modification to assign CPT codes 43254, 43270, and 43275 to APC 5302 (Level 2 Upper GI Procedures); CPT codes 58260 and 58262 to APC 5415 (Level 5 Gynecologic Procedures); CPT codes 19303 and 19307 to APC 5092 (Level 2 Breast/Lymphatic Surgery and Related Procedures); and CPT code 28740 to APC 5114 (Level 4 Musculoskeletal Procedures). We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
52. Prostate Laser Enucleation Procedure, CPT Code 52649 (APC 5375)
Prostate laser enucleation procedure utilizes laser to remove the prostate tissue to treat benign prostatic hyperplasia. The procedure is described by CPT code 52649 (Laser enucleation of the prostate with morcellation, including control of postoperative bleeding, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, internal urethrotomy and transurethral resection of prostate are included if performed)).
For CY 2025, the OPPS payment rates are proposed based on available CY 2023 claims data. For the CY 2025 proposed rule, based on our analysis of claims data, we found a total of 7,494 single frequency claims and a geometric mean cost of approximately $6,812 for CPT code 52649. For CY 2025, we proposed to continue to assign CPT code 52649 to Level 5 Urology and Related Services (APC 5375) with a proposed payment rate of approximately $5,057.
Comment: One commenter disagreed with the proposed APC assignment to APC 5375 (Level 5 Urology and Related Services) and requested the reassignment of CPT code 52649 to APC 5376 (Level 6 Urology and Related Services). The commenter argued that CPT code 52649 shared more clinical and resource similarities with CPT code 50562 (Renal endoscopy through established nephrostomy or pyelostomy, with or without irrigation, instillation, or ureteropyelography, exclusive of radiologic service; with resection of tumor) and CPT code 55873 (Cryosurgical ablation of the prostate (includes ultrasonic guidance and monitoring)), which are currently assigned to APC 5376. Moreover, the commenter argued that there is more work involved in CPT code 52649 than CPT code 52601 (Transurethral electrosurgical resection of prostate, including control of postoperative bleeding, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy are included)) which is currently assigned to APC 5375 (Level 5 Urology and Related Services).
Response: The CY 2025 OPPS payment rates were proposed based on available CY 2023 claims data. We analyzed the claims data for this final rule, and based on our review, we found a geometric mean cost of approximately $6,812 for CPT code 52649 based on 7,494 single claims (out of 7,564 total claims), is consistent with the geometric mean cost of about $5,145 for APC 5375, rather than the geometric mean cost of approximately $9,356 for APC 5376. Based on the claims data, we believe that CPT code 52649 fits more appropriately in APC 5375, rather than in APC 5376, based on resource cost and clinical similarity to CPT code 52601 in APC 5375. We note that we review, on an annual basis, the APC assignments for all services and items paid under the OPPS based on our analysis of the latest claims data.
We also disagree that CPT code 52649 is sufficiently clinically similar to CPT code 50562 because the latter code involves a surgical treatment of the kidney rather than the prostate. Furthermore, we disagreed that CPT code 52649 shares resource similarity with CPT code 55873, as cryosurgical ablation requires many more resources than the prostate laser enucleation procedure.
In summary, after consideration of the public comment, we are finalizing our proposal, without modification, to assign CPT code 52649 to APC 5375 for CY 2025. Please refer to Table 99 below for the final OPPS APC and status indicator assignment for HCPCS code 52649 for CY 2025. The final CY 2025 payment rate for the code can be found in Addendum B to this final rule with comment period. In addition, we refer ( printed page 94110) readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Both Addendum B and D1 are available via the internet on the CMS website.
53. Remote Uroflowmetry Service, CPT Code 0812T (APC 5721)
Remote uroflowmetry is a service that allows patients to measure urine output while remotely monitored by health professionals. This service aids in the diagnosis and management of lower urinary tract issues, such as benign prostatic hyperplasia. It is described by CPT code 0812T (Remote multi-day complex uroflowmetry ( e.g., calibrated electronic equipment); device supply with automated report generation, up to 10 days) which became effective January 1, 2024. For CY 2025, we proposed to assign CPT code 0812T to APC 5741 (Level 1 Electronic Analysis of Devices).
Comment: One commenter disagreed with our proposed APC assignment of APC 5741 (Level 1 Electronic Analysis of Devices), citing higher equipment and labor cost associated with the CPT code 0812T than services assigned to APC 5741. The commenter requested CPT code 0812T be reassigned to APC 5722 (Level 2 Diagnostic Tests and Related Services), citing resource similarity to CPT code 51741(Complex uroflowmetry ( e.g., calibrated electronic equipment)).
Response: We appreciate the commenter's recommendation regarding the APC assignment for CPT code 0812T. However, we disagree with the commenter that CPT code 51741 shares similar resource requirements with CPT code 0812T, although both CPT codes describe the uroflowmetry service. CPT code 51741 is a more complex procedure than CPT code 0812T, which is why we proposed to assign CPT code 51741 to APC 5722 (Level 2 Diagnostic Tests and Related Services)). However, we also believe CPT code 0812T shares more resource similarity with CPT code 51703 (Insertion of temporary indwelling bladder catheter; complicated ( e.g., altered anatomy, fractured catheter/balloon)) than CPT code 51741. Therefore, for CY 2025, we are reassigning CPT code 0812T to APC 5721 (Level 1 Diagnostic Tests and Related Services).
After consideration of the public comment we received, we are finalizing our proposal with modification and reassigning CPT code 0812T to APC 5721 (Level 1 Diagnostic Tests and Related Services). Please refer to Table 100 below for the final OPPS APC and status indicator assignment for CPT code 0821T for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94111)54. Skin Cell Suspension Autograft (SCSA) Procedures, CPT Codes 15011 Through 15018 (APCs 5051, 5054, and 1567)
The RECELL System is a device used for skin cell suspension autograft (SCSA) procedures. Skin cell suspension autograft procedures treat acute thermal burn injuries by adhering skin cell solutions to the wound site without use of fixation. There are three steps to perform a SCSA procedure: (1) skin harvesting, (2) SCSA preparation (utilizing the RECELL System), and (3) SCSA application to the wound bed.
The RECELL System is on transitional pass-through (TPT) effective January 1, 2022, to December 31, 2024. While on TPT, each time the RECELL device is used, providers must bill device code HCPCS C1832 (Autograft suspension, including cell processing and application, and all system components) and either CPT code 15110 (Epidermal autograft, trunk, arms, legs; first 100 sq cm or less, or 1% of body area of infants and children) or CPT code 15115 (Epidermal autograft, face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits; first 100 sq cm or less, or 1% of body area of infants and children),as provided in the January 2022 OPPS Change Request 12552 (Transmittal R11150CP).
While HCPCS code C1832 describes the RECELL System that prepares the SCSA, CPT codes 15110 and 15115 describe the autograft procedure, which includes both the skin harvesting and SCSA application components of the service. For CY 2024, CPT codes 15510 and 15115 were assigned to APC 5054 (Level 4 Skin Procedures), with a payment rate of approximately $1,800. HCPCS code C1832 can also be billed with CPT codes 15100 (Split-thickness autograft, trunk, arms, legs; first 100 sq cm or less, or 1% of body area of infants and children (except 15050)) or 15120 (Split-thickness autograft, face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits; first 100 sq cm or less, or 1% of body area of infants and children (except 15050)), so long as it is billed with one of the two preceding codes (CPT codes 15110 or 15115).
In September 2023, AMA's CPT Editorial Panel created eight new Category 1 CPT codes to describe SCSA procedures, effective January 1, 2025. The eight new codes are provided in Table 101.
( printed page 94112)In creating these eight new CPT codes, the harvesting, preparation, and application components of the service are each split into separate codes. The harvesting of the skin is described by CPT codes 15011 and 15012, the preparation of the SCSA is described by CPT codes 15013 and 15014, and the application of the SCSA is described by CPT codes 15015 through 15018. While the RECELL System is on TPT, it is sufficient to bill the device code HCPCS C1832 and only one procedure code (either CPT code 15110 or CPT 15115) to describe all three components of the service. However, with the creation of the eight new CPT codes, at least three CPT codes will need to be billed each time the RECELL System is used for a SCSA procedure: CPT code 15011 with or without CPT code 15012 for the skin harvesting, CPT code 15013 with or without CPT code 15014 for the SCSA preparation, and CPT code 15015 with or without CPT code 15016 or CPT code 15017 with or without CPT code 15018 for the SCSA application.
For CY 2025, we proposed to assign the eight new CPT codes to various clinical APCs and status indicators provided in Table 102.
( printed page 94113)Comment: We received two comments requesting that we finalize our proposal to assign 15011, 15015, and 15017 to ( printed page 94114) APC 5054 (Level 4 Skin Procedures) for CY 2025.
Response: We thank the commenters for their support.
Comment: We received two comments, including one comment from the manufacturer of the RECELL System, requesting that we reassign CPT code 15013 from APC 5051 (Level 1 Skin Procedures) to APC 1575 (New Technology—Level 38 ($10,001-$15,000) with a payment rate of $12,500.50 for CY 2025. The commenters expressed concern that the proposed APC assignment for CPT code 15013 to APC 5051 does not accurately reflect the hospital-reported costs associated with the RECELL System. The commenters requested that we consider the hospital costs reported with device code C1832 during the TPT period when finalizing the APC assignment for CPT 15013. The comments estimated a combined total cost of $10,188.96 for CPT code 15013 by adding the purported mean cost for device code C1832 ($7,220) with supply and operating room time costs of approximately $2,900. Based on these estimates, the commenters believe assigning CPT code 15013 to APC 1575 with a payment rate of $12,500.50 would sufficiently pay for the preparation step of the SCSA procedure.
Response: We thank the commenters for their input. Under the OPPS, once pass-through status ends for a device, the cost of the device is packaged into its associated procedure. For the RECELL System, once TPT expires on December 31, 2024, the associated procedure will be CPT code 15013. We agree with the commenters that we need to use the claims data available while the RECELL System is on TPT to determine the appropriate APC placement for CPT code 15013.
However, given the restructuring of how SCSA procedures utilizing the RECELL System will be billed effective January 1, 2025, we also believe we need to consider the costs of the skin harvesting and application steps of the procedure, which are currently billed by only one procedure code (either CPT Code 15110 or CPT code 15115), paid at approximately $1,800, to determine the appropriate payment for the RECELL System and SCSA application step. In other words, we believe that the sum of the payment rates for the new codes describing the three steps of the SCSA procedure should approximate the cost of the RECELL System on TPT and CPT codes 15110 or 15115. Based on our review of the claims data for device code C1832 during the TPT period, as well as the payment rates for CPT codes 15110 and 15115, we believe assigning CPT code 15013 to APC 1567 (New Technology—Level 30 ($6001-$6500)) with a $6,250.50 payment rate would be appropriate for CY 2025, considering that it will be billed with additional procedure codes that describe the other steps of the SCSA procedure. Rather than assigning CPT code 15013 to APC 5051, as proposed, which has a payment rate of approximately $200 for CY 2025, we believe assigning CPT code 15013 to APC 1567 with a payment rate of $6,250.50 will more appropriately pay for the preparation part of the service, which uses the device. When the full service is performed, meaning that the harvesting, preparation, and application steps are performed, the total cost of the service for CY 2025 will be approximately $10,000, an amount that we believe is appropriate based on the data we have collected while the device has had transitional pass-through status.
In consideration of the public comments received, we are finalizing our proposal with modification. For CY 2025, we are finalizing the APC assignments for CPT codes 15011, 15015, and 15017 to APC 5054 (Level 4 Skin Procedures) as proposed. However, we are not finalizing our proposal to assign CPT code 15013 to APC 5051 (Level 1 Skin Procedures) as proposed. For CY 2025, we are assigning CPT code 15013 to APC 1567 ((New Technology—Level 30 ($6001-$6500)).
The final 2025 payment rates for the codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the status indicator meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
55. Surgical Pathology Examination, CPT Code 88309 (APC 5674)
CPT code 88309 describes a level VI examination of a surgical pathology specimen performed by the physician, typically a pathologist. The examination includes accessioning the specimen into the lab, which involves formally receiving the specimen, examining gross and microscopic features of the specimen, and reporting findings.
In the CY 2025 OPPS/ASC proposed rule, we proposed to reassign CPT code 88309 from APC 5674 (Level 4 Pathology) to APC 5673 (Level 3 Pathology) with a proposed payment rate of $356.00.
Comment: Three commenters asked CMS to maintain the assignment of CPT code 88309 in APC 5674.
One commenter recommended that CMS maintain the current assignment of CPT 88309 (Level VI—Surgical pathology, gross and microscopic examination) in APC 5674, (Level 4 Pathology), rather than assigning the code to APC 5673, (Level 3 Pathology), to promote resource and clinical cohesion.
Two commenters stated that the proposed reassignment is based on claims data that do not reflect the actual costs of performing the test. They stated that the CY 2025 geometric mean cost for this code is over 25% lower than the geometric mean cost for the prior calendar years and that the complexity of specimens and technical work associated with these services requires the maintenance of the code in APC 5674. Therefore, to ensure resource cohesion within the clinical APCs, the commenters asked CMS to maintain the assignment of CPT code 88309 to APC 5674 for CY 2025 and to not finalize an APC reassignment that would result in a greater than 56% reduction in the payment rate for this important service for Medicare beneficiaries.
Response: We thank the commenters for their input. We acknowledge a significant drop in the geometric mean cost for CPT code 88309, which is almost 25% lower than the geometric mean cost in previous years.
For this final rule with comment period, we once again reviewed the available CY 2023 claims data and observed a wide variation in claims used for ratesetting purposes for CY 2025. We note that the claims included in ratesetting for this code reflect a wide range of estimated costs and providers furnishing this service along with their associated clinical patterns. We believe these claims are an accurate reflection of those changing costs. However, we are sympathetic to commenters' concerns regarding the 25% reduction in the proposed payment rate for CPT code 88309 from CY 2024 to CY 2025. Therefore, we will use our equitable adjustment authority under section 1833(t)(2)(E) of the Act to continue to assign CPT code 88309 to APC 5674 for CY 2025.
In summary, after consideration of the public comments that we received, we are finalizing our proposal with modification to assign CPT code 88309 to APC 5674. However, we will continue to monitor the claims data as they are updated and available.
We refer readers to Addendum B to this final rule with comment period for the final CY 2025 OPPS payment rate for this code. Addendum B is available via the internet on the CMS website. ( printed page 94115)
56. Therapeutic Ultrafiltration, CPT 0692T (APC 5242)
In the CY 2025 OPPS/ASC proposed rule, we proposed to continue to assign CPT code 0692T (Therapeutic ultrafiltration), which became effective January 1, 2022, to APC 5241 (Level 1 Blood Product Exchange and Related Services) with a proposed payment rate of $431.37.
Therapeutic ultrafiltration reported with CPT code 0692T, is an apheresis procedure for fluid-overloaded heart failure patients through which plasma water and sodium are removed from the blood using the Aquadex SmartFlow System.
We received several comments requesting CMS reassign CPT code 0692T from APC 5241 (Level 1 Blood Product Exchange and Related Services) to APC 5242 (Level 2 Blood Product Exchange and Related Services).
Comment: Several commenters claimed that the payment rate for APC 5241 is inadequate for CPT code 0692T and that APC 5242 better aligns with the resource costs and clinical aspects of the service. According to the commenters, the resources required, and risks associated with ultrafiltration are the same as plasmapheresis, which is assigned to APC 5242. Ultrafiltration ( e.g., apheresis) is the extracorporeal removal of isotonic plasma water (water and salt/electrolytes) and is very similar to plasmapheresis according to the commenters, which involves the extracorporeal removal, return, or exchange of blood plasma or components and is assigned to APC 5242. Commenters suggested that reassigning the ultrafiltration therapy code 0692T to APC 5242 would align OPPS payment with the cost of providing the service.
Response: We thank commenters for their input. We have limited claims data (9 total claims) for CPT code 0692T available for CY 2025 ratesetting. However, based on the code's clinical similarity and expected resource cost similarity to CPT code 36514 (Therapeutic apheresis; for plasma pheresis), which we have assigned to APC 5242 (Level 2 Blood Product Exchange and Related Services) for CY 2025, we believe that we should reassign CPT code 0692T to APC 5242.
In summary, after consideration of the public comments that we received, we are finalizing our proposal with modification, and assigning CPT code 0692T to APC 5242 for CY 2025. The CY 2025 final payment rate for this code can be found in Addendum B of this final rule with comment period.
57. Thyroid Ablation, CPT Codes 60660 and 60661 (APC 5072)
The AMA established CPT codes 60660 (Ablation of 1 or more thyroid nodule(s), one lobe or the isthmus, percutaneous, including imaging guidance, radiofrequency) and 60661 (Ablation of 1 or more thyroid nodule(s), additional lobe, percutaneous, with imaging guidance, radiofrequency (List separately in addition to code for primary service), effective January 1, 2025. These codes were previously described by the placeholder codes 6XX01 and 6XX02. In the CY 2025 OPPS/ASC proposed rule, CMS crosswalked CPT code 60660 to CPT code 0673T (Ablation, benign thyroid nodule(s), percutaneous, laser, including imaging guidance) and proposed to assign it to APC 5072 (Level 2 Excision/Biopsy/Incision and Drainage) for CY 2025. CMS also proposed to assign CPT code 60661 to status indicator “N” (packaged), as it is an add-on code.
Comment: We received two comments requesting reassignment for CPT codes 60660 and 60661. One comment requested we reassign CPT code 60660 to APC 5073 (Level 3 Excision/Biopsy/Incision and Drainage), due to its clinical similarity and resource homogeneity with the other procedures in APC 5073. A second comment requested we reassign both 60660 and 60661 to APC 5164 (Level 4 ENT Procedures), due to their similarity to CPT code 30140 (Submucous resection inferior turbinate, partial or complete, any method).
Response: We thank the commenters for their suggestions. After further clinical review, we continue to believe that CPT code 0673T is the appropriate crosswalk for CPT code 60660 and that CPT code 30140 would not be the correct clinically comparable code. CPT code 0673T has a geometric mean cost of around $1,578 and is assigned to APC 5072, which has a payment rate of around $1,620. We believe that 0673T is appropriately placed in APC 5072, and therefore, CPT code 60660 is appropriately assigned to APC 5072 and should not be reassigned to APC 5073 at this time. Additionally, because CPT code 60661 is an add-on code, we believe that the current status indicator assignment of “N” is correct.
After consideration of the public comments we received, we are finalizing our proposal without modification to assign CPT code 60660 to APC 5072 (Level 2 Excision/Biopsy/Incision and Drainage) and 60661 to status indicator “N” for CY 2025. Table 103 shows the finalized status indicator and APC assignment for the procedure code. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94116)58. Thyroid Removal, CPT Code 60240 (APC 5361)
In the CY 2025 OPPS/ASC proposed rule, CPT code 60240 (Thyroidectomy, total or complete), which describes thyroid removal, was proposed to be assigned to APC 5361 (Level 1 Laparoscopy and Related Services) with a proposed geometric mean cost of $7,150.89 and proposed APC payment rate of $5,798.13.
Comment: We received one comment requesting that we reassign CPT code 60240 to APC 5362 (Level 2 Laparoscopy and Related Services), which had a proposed payment rate of $10,378.45. The commenter argued that keeping CPT code 60240 in APC 5361 would be a 2 times rule violation because CPT code 60240 has a geometric mean cost that is greater than 2 times the geometric mean value of CPT code 60281(Excision of thyroglossal duct cyst or sinus; recurrent), which had a proposed geometric mean cost of around $3,462 and is also in APC 5361.
Response: We thank the commenter for their input. Our review found that APC 5361 was not in violation of the 2 times rule. Because CPT code 60821 has only 2 claims for CY 2025, it does not meet the significance threshold for the 2 times rule evaluation. Additionally, the updated geometric mean cost for CPT code 60240 of $7,022.82 is closer to the payment rate of APC 5361 ($5,834.36) compared to APC 5362 ($10,411.39).
After consideration of the public comment we received, we are finalizing our proposal without modification to assign CPT code 60240 to 5361 (Level 1 Laparoscopy and Related Services) for CY 2025. Table 104 shows the finalized status indicator and APC assignment for the procedure code. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
59. Trabecular Bypass Procedures, CPT Codes 66989, 66991, 0660T, 0661T, and 0671T (APCs 5492 and 5493)
Trabecular bypass procedures are used for the treatment of glaucoma and may be performed along with cataract surgery or as a stand-alone procedure. Another glaucoma treatment involves the implantation of a device that releases a drug into the eye. For the CY 2025 OPPS final rule, we proposed to assign CPT code 66989 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1-stage procedure), manual or mechanical technique ( e.g., irrigation and aspiration or phacoemulsification), complex, requiring devices or techniques not generally used in routine cataract surgery ( e.g., iris expansion device, suture support for intraocular lens, or primary posterior capsulorrhexis) or performed on patients in the amblyogenic developmental stage; with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more) with a geometric mean cost around $5,440; CPT code 66991 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1 stage procedure), manual or mechanical technique ( e.g., irrigation and aspiration or phacoemulsification); with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more) with a geometric mean cost around $4,990; and CPT code 0671T (Insertion of anterior segment aqueous drainage device into the trabecular meshwork, without external ( printed page 94117) reservoir, and without concomitant cataract removal, one or more) with a geometric mean cost around $5,527 to APC 5493 (Level 3 Intraocular Procedures) with a payment rate around $5,160.
Also, for the CY 2025 OPPS final rule, we proposed to assign CPT code 0660T (Implantation of anterior segment intraocular nonbiodegradable drug-eluting system, internal approach) with no claims data for CY 2025, and CPT code 0661T (Removal and reimplantation of anterior segment intraocular nonbiodegradable drug-eluting implant) with no claims data for CY 2025 to APC 5492 (Level 2 Intraocular Procedures) with a payment rate of around $4,023.
Comment: Multiple commenters supported our proposal to assign CPT codes 66989, 66991, and 0671T to APC 5493 (Level 3 Intraocular Procedures) with a payment rate around $5,160. The commenters also supported our proposal to assign CPT codes 0660T and 0661T to APC 5492 (Level 2 Intraocular Procedures) with a payment rate of around $4,023.
Response: We appreciate the commenters' support for our proposals.
After consideration of the public comments we received, we are finalizing our proposal without modification for CPT codes 66989, 66991, 0660T, 0661T, 0671T to assign these procedures to APC 5493 (Level 3 Intraocular Procedures). We also are implementing our proposal without modification for CPT codes 0660T and 0661T to assign these procedures to APC 5492 (Level 2 Intraocular Procedures). Table 105 shows the finalized status indicator and APC assignment for all of the procedure codes. We refer readers to Addendum B of this final rule for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94118) ( printed page 94119)60. Transcutaneous Magnetic Peripheral Nerve Stimulation CPT Codes 0766T and 0767T (APC 5722)
Transcutaneous Magnetic Peripheral Nerve Stimulation is intended to stimulate peripheral nerves for relief of chronic intractable pain, post‐traumatic pain, post‐surgical pain and/or for relief of chronic painful diabetic peripheral neuropathy in the lower extremities for patients 18 and older. CPT code 0766T (Transcutaneous magnetic stimulation by focused low-frequency electromagnetic pulse, peripheral nerve, with identification and marking of the treatment location, including noninvasive electroneurographic localization (nerve conduction localization), when performed; first nerve) became effective January 1, 2023. For CY 2024, CPT code 0766T has been assigned to APC 5721 (Level 1 Diagnostic Tests and Related Services) with the status indicator “Q1” (STV-Packaged Codes). There are no claims available for CPT codes 0766T in CY 2023. For CY 2025, we proposed to continue assigning CPT codes 0766T to APC 5721.
CPT code 0767T (Transcutaneous magnetic stimulation by focused low-frequency electromagnetic pulse, peripheral nerve, with identification and marking of the treatment location, including noninvasive electroneurographic localization (nerve conduction localization), when performed; each additional nerve (list separately in addition to code for primary procedure)) became effective January 1, 2023. CPT code 0767T is an add-on code and we proposed to assign status indicator “N” (packaged) for this code.
Comment: Commenters disagreed with the proposed assignment of APC 5721 (Level 1 Diagnostic Tests and Related Services) and the status indicator “Q1” for CPT code 0766T, claiming that CPT code 0766T is not clinically similar nor uses comparable resources to other services in APC 5721. The commenters stated that the HOP Panel recommended that 0766T be reassigned to APC 5431 (Level 1 Nerve Procedures) with a status indicator “S” (Significant Procedures, Not Discounted when Multiple. Paid under OPPS; separate APC payment). Additionally, commenters disagreed with the status indicator of “N” for CPT code 0767T and requested the 0767T be reassigned status indicator “S” and assigned to APC 5431 (Level 1 Nerve Procedures).
Response: We thank commenters for their input. We disagree that CPT code 0766T is comparable clinically and in resource costs to the services assigned to APC 5431. However, after further evaluation, we believe CPT code 0766T is more comparable clinically and from a resource cost perspective to CPT code 90867 (Therapeutic repetitive transcranial magnetic stimulation (tms) treatment; initial, including cortical mapping, motor threshold determination, delivery and management), which is assigned to APC 5722 (Level 2 Diagnostic Tests and Related Services), because these services share similar magnetic stimulation and nerve localization processes. Additionally, we believe that patients may receive this service as a primary standalone service. Therefore, for CY 2025, we are finalizing the reassignment of CPT code 0766T to APC 5722 (Level 2 Diagnostic Tests and Related Services) and its status indicator to “S” (Significant Procedures, Not Discounted when Multiple. Paid under OPPS; separate APC payment).
We note that CPT code 0767T is an add-on code. Add-on codes are always performed in addition to the primary service or procedure and must never be reported as a stand-alone code. As specified under regulation 42 CFR 419.2(b)(18), add-on codes are generally packaged under the OPPS, and payment for the codes are bundled with the primary codes. Consequently, CPT code 0767T is not paid separately under the OPPS, but instead, its payment is packaged into payment for the primary code.
After consideration of the public comments we received, we are finalizing reassigned status indicator and APC assignments for CPT codes 0766T and finalizing as proposed the status indicator assignment for 0767T. Please refer to Table 106 below for the final OPPS APC and status indicator assignment for CPT codes 0766T and 0767T for CY 2025. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
( printed page 94120)61. Transurethral Ultrasound Ablation (TULSA) Procedure, HCPCS Code C9734 and CPT Code 55882 (APC 5377)
The TULSA procedure is a minimally invasive procedure that, under MRI guidance, delivers precise doses of therapeutic ultrasound to treat prostate cancer. HCPCS code C9734 (Focused ultrasound ablation/therapeutic intervention, other than uterine leiomyomata, with magnetic resonance (mr) guidance) became effective April 1, 2013.
For CY 2025, the OPPS payment rates are proposed based on available CY 2023 claims data. For CY 2025, based on our analysis of claims data, we found a total of 89 single frequency claims and an estimated geometric mean cost of $10,056 for HCPCS code C9734. For CY 2025, we proposed to continue to assign HCPCS code C9734 to APC 5115 (Level 5 Musculoskeletal Procedures) with a proposed payment rate of approximately $12,756.
Comment: Commenters pointed out that the AMA CPT created a new code, CPT code 5X008, to describe the TULSA procedure represented by HCPCS code C9734. CPT code 5X008 (Ablation of prostate tissue, transurethral, using thermal ultrasound, including magnetic resonance imaging guidance for, and monitoring of, tissue ablation; with insertion of transurethral ultrasound transducer for delivery of thermal ultrasound, including suprapubic tube placement and placement of an endorectal cooling device, when performed) becomes effective January 1, 2025.
Commenters also recommended that the TULSA procedure be reassigned to the Urology and Related Services APC series, which is more clinically consistent with other urology related procedures. Furthermore, commenters stated TULSA is more similar clinically and in terms of resources to the procedures in APC 5377 (Level 7 Urology and Related Services) than procedures in APC 5376 (Level 6 Urology and Related Services). Commenters referenced the use of a MRI technology, specialized staffing, and the longer procedural time that increases the resources needed to perform the TULSA procedure relative to other prostate ablation procedures in Level 6 Urology and Related Services (APC 5376). Commenters stated that HCPCS code C9734's geometric mean cost aligns more appropriately with APC 5377 (Level 7 Urology and Related Services) with a proposed APC geometric mean cost of $13,217.
Response: We thank the commenters for pointing out that CPT code 5X008 (55882) will become effective January 1, 2025. We note that we will replace HCPCS code C9734 with CPT code 5X008 (55882) effective January 1, 2025, as well as assign the underlying claims associated with C9734 to CPT code 5X008 (55882).
We agree with the commenters that the TULSA procedure, described by HCPCS code C9734 (CPT code 55882) should be reassigned to the Urology and Related Services APC series to be consistent with other urological related procedures. We also believe that the TULSA procedure is clinically similar to CPT code 55880 (Ablation of malignant prostate tissue, transrectal, with high intensity-focused ultrasound (hifu), including ultrasound guidance) or CPT code 55873 (Cryosurgical ablation of the prostate (includes ultrasonic guidance and monitoring)) which are assigned to APC 5376. However, we acknowledge that the TULSA procedure requires greater resources from MRI equipment and MRI technologists, and requires a longer procedure time than CPT codes 55880 and 55873. Therefore, for CY 2025, we are reassigning CPT code 5X008 (55882) to APC 5377 (Level 7 Urology and Related Services). We note that we will continue to evaluate and monitor the cost of CPT code 5X008 (55882) for future rulemaking.
After consideration of the public comments we received, we are finalizing our policy to assign CPT code 5X008 (55882) to APC 5377 (Level 7 Urology and Related Services). Please refer to Table 107 below for the final OPPS APC and status indicator assignment for CPT code 5X008 (55882) for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
( printed page 94121)62. Unfold AI Service, CPT Code 0898T (APC 5724)
Unfold AI is an algorithm-based test that analyzes multiple clinical data inputs and multi-modal images to create a newly computed 3D visualization of the precise location and dimensions of clinically significant prostate cancer. The generated output assists physicians in identifying the cancer margins for treatment. The Unfold AI service is represented by CPT code 0898T (Noninvasive prostate cancer estimation map, derived from augmentative analysis of image-guided fusion biopsy and pathology, including visualization of margin volume and location, with margin determination and physician interpretation and report) which became effective July 1, 2024. As this is a new code in 2024, we have no claims data for CPT code 0898T. For CY 2025, we proposed to assign CPT Code 0898T to APC 5724 (Level 4 Diagnostic Tests and Related Services).
Comment: Commenters supported our proposal to assign CPT code 0898T to Level 4 Diagnostic Tests and Related Services (APC 5724).
Response: We thank the commenters for their support on our proposal to assign CPT code 0898T to Level 4 Diagnostic Tests and Related Services (APC 5724).
After consideration of the public comments we received, we are finalizing our proposal without modification for CPT code 0898T. Please refer to Table 108 below for the final OPPS APC and status indicator assignment for HCPCS code 0898T for CY 2025. The final CY 2025 payment rates can be found in Addendum B to this final rule with comment period via the internet on the CMS website.
63. Ureteroscopy, HCPCS Code C9761 (APC 5376)
The ureteroscopy procedure addresses kidney stones and may apply catheterization to vacuum aspirate the fragmented kidney stones. HCPCS code C9761 (Cystourethroscopy, with ureteroscopy and/or, with lithotripsy, and ureteral catheterization for steerable vacuum aspiration of the kidney, collecting system, ureter, bladder, and urethra if applicable (must use a steerable ureteral catheter) became effective October 1, 2020.
For CY 2025, the OPPS payment rates are proposed based on available CY ( printed page 94122) 2023 claims data. For the CY 2025 OPPS/ASC proposed rule, we found a total of 95 single frequency claims and a geometric mean cost of approximately $9,718 for HCPCS code C9761. For CY 2025, we proposed to continue to assign HCPCS code C9761 to APC 5376 (Level 6 Urology and Related Services) with a proposed payment rate of approximately $9,208.
Comment: One commenter requested CMS provide coding guidance if HCPCS code C9761 should be reported when the ureteroscopy procedure utilizes the flexible ureteroscopy (fURS) with the flexible and navigable suction (FANS) sheath.
Response: This comment is out of scope of this OPPS/ASC final rule with comment period. We note that if hospitals have questions about appropriate coding that they cannot resolve on their own, the appropriate first step would be to review the HCPCS codes or consult a Medicare Administrative Contractor (MAC).
64. V-LAP System Left Atrial Pressure Monitoring Procedure, CPT Code 0933T (APC 5191)
On January 1, 2025, CPT code 0933T (Transcatheter implantation of wireless left atrial pressure sensor for long-term left atrial pressure monitoring, including sensor calibration and deployment, right heart catheterization, transseptal puncture, imaging guidance, and radiological supervision and interpretation) will become effective to report the implantation and remote monitoring of the V-LAP System. The system is used to measure and monitor left atrial pressure in patients with advanced congestive heart failure (CHF).
The V-LAP system was designated as a Category A IDE clinical study (NCT06147336) on January 30, 2024. Since devices in Category A IDE studies are not covered by Medicare during the study, the payment for CPT code 0933T while in the Category A IDE study will reflect only the cost of the service that is performed each time it is reported on a claim. For CY 2025, we proposed to assign CPT code 0933T to APC 5191 (Level 1 Endovascular Procedures) and status indicator of “J1” (Hospital Part B Services Paid Through a Comprehensive APC) with a proposed payment of around $3,210.
Comment: We received one comment from the manufacturer requesting that we reassign CPT code 0933T from APC 5191 (Level 1 Endovascular Procedures) to APC 1532 (New Technology—Level 32 ($7001-$7500)), as it more accurately reflects the costs of this procedure.
Response: We appreciate the commenter's input. The commenter asserts that the current APC assignment of 5191 (Level 1 Endovascular Procedures) does not adequately reflect the complex nature and resource demands of the V-LAP procedure even though the procedure is described as a standard minimally invasive catheterization. We are unclear what procedural steps makes this procedure more complex than the other procedures assigned to APC 5191. Based on our understanding of the service and input from our medical advisors, we do not agree and believe that CPT code 93451 (Right heart catheterization including measurement(s) of oxygen saturation and cardiac output, when performed) is an appropriate crosswalk code while the V-LAPTM procedure is in a Category A IDE trial. Once the service receives appropriate FDA approval outside of the Category A IDE study ( e.g., PMA approval or 510(k)), we will work to assign CPT code 0933T to an APC that is more reflective of the cost of the service, beyond routine care items and services.
We remind the readers that for Category A IDE studies, Medicare only provides payment for routine care items and services furnished in the FDA-approved Category A study. We refer readers to the Medicare Benefit Policy Manual, See 20.1—Medicare Requirements for Coverage of Items and Services in FDA-approved Category A and B IDE Studies.
After consideration of the public comment we received, we are finalizing our proposal without modification to assign CPT code 0933T to APC 5191. We refer readers to Addendum B of this final rule with comment period for the payment rates for all codes reportable under the OPPS. Addendum B is available via the internet on the CMS website.
65. Vagal Nerve Neurostimulator System, CPT Codes 0908T Through 0912T
The CPT Editorial Panel created 5 new Category III CPT codes, effective January 1, 2025, which describe the implantation, replacement, removal, interrogation, and programming of the integrated vagus nerve stimulation device used for the treatment of patients who have rheumatoid arthritis (RA) and are not responsive to pharmacological therapy. The patient is placed under general anesthesia (for about 60-90 minutes) while the neurosurgeon dissects the tissue on the left side of the neck to access the cervical vagus nerve within the carotid sheath. The rechargeable integrated neurostimulator is then placed directly on the cervical vagus nerve. The implant is charged weekly with an external charger. A rheumatologist is responsible for prescribing and making changes to the stimulation therapy via an app. See Table 109 for the CPT codes, their long descriptors, and the proposed payment assignments.
( printed page 94123)For CY 2025, we proposed to assign 0908T and 0909T to APC 5462 (Level 2 Neurostimulator and Related Procedures) and status indicator “J1” (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS.) based on their similarity to CPT code 0587T (Percutaneous implantation or replacement of integrated single device neurostimulation system for bladder dysfunction including electrode array and receiver or pulse generator, including analysis, programming, and imaging guidance when performed, posterior tibial nerve) and because CPT 0587T also describes an integrated system.
We proposed to assign 0910T to APC 5461 (Level 1 Neurostimulator and Related Procedures) and status indicator “J1” based on its similarity to CPT code 0588T (Revision or removal of percutaneously placed integrated single device neurostimulation system for bladder dysfunction including electrode array and receiver or pulse generator, including analysis, programming, and imaging guidance when performed, posterior tibial nerve) as both codes describe the removal of an integrated neurostimulation system.
We also proposed to assign CPT codes 0911T to APC 5733 (Level 3 Minor Procedures) and status indicator “Q1” (STV-Packaged Codes; Paid Under OPPS) using CPT code 95981(Electronic analysis of implanted neurostimulator pulse generator system ( e.g., rate, pulse amplitude and duration, configuration of wave form, battery status, electrode selectability, output modulation, cycling, impedance and patient measurements) gastric neurostimulator pulse generator/transmitter; subsequent, without reprogramming) as a crosswalk code and 0912T to APC 5742 (Level 2 Electronic Analysis of Devices) and status indicator of “S” (Procedure or Service, Not Discounted When Multiple; Paid under OPPS; separate APC payment.) using CPT code 0589T (Electronic analysis with simple programming of implanted integrated neurostimulation system for bladder dysfunction ( e.g., electrode array and receiver), including contact group(s), amplitude, pulse width, frequency (Hz), on/off cycling, burst, dose lockout, patient-selectable parameters, responsive neurostimulation, detection algorithms, closed-loop parameters, and passive parameters, when performed by physician or other qualified health care professional, posterior tibial nerve, 1-3 parameters) as a crosswalk code.
Comment: We received several comments from stakeholders requesting that CPT codes XX32T (0908T) and XX33T (0909T) be reassigned from APC 5462 (Level 2 Neurostimulator and Related Procedures) to APC 5465 (Level 5 Neurostimulator and Related Procedures) because the proposed APC assignments do not reflect the costs of the device and procedure or consider the clinical intensity and risk of the procedure. Commenters provided CPT code 64568 (Open implantation of cranial nerve ( e.g., vagus nerve) neurostimulator electrode array and pulse generator) as an appropriate comparator code, noting that the integrated devices do not require the creation of an additional subcutaneous pocket. While there are some differences in the two surgical procedures described by CPT codes XX32T (0908T) and XX33T (0909T) compared to CPT code 64568, the commenter asserted that ( printed page 94124) inserting the integrated system is a complex procedure to perform and is similar in cost, resources, and required skill.
Another commenter requested that we reassign XX34T (0910T) from APC 5461 (Level 1 Neurostimulator and Related Procedures) to APC 5463 (Level 3 Neurostimulator and Related Procedures) stating that CPT code 61888 (Revision or removal of cranial neurostimulator pulse generator or receiver) is an appropriate comparator code.
Commenters noted that the vagal nerve neurostimulation system is a novel treatment for rheumatoid arthritis patients who have exhausted other pharmacological options to manage their disease. They stated that biological therapies are expensive and may cost over $50,000/per year, while this neurostimulation system is expected to have a shelf life of 10 years. They asserted that APC reassignments of XX32T (0908T), XX33T (0909T), and XX34T (0910T) would support CMS' commitment to promoting new innovative treatment options and expanding access for beneficiaries with chronic conditions.
In addition to the comments requesting APC reassignments, the manufacturer stated they were expecting FDA approval, specifically Premarket Approval (PMA), in the 2nd quarter of 2025 with an expected statement “for the treatment of adult patients with moderately to severely active RA who have had an inadequate response, loss of response or intolerance to one (1) or more biological or targeted synthetic DMARDs (disease-modifying antirheumatic drugs)”. They acknowledged that, in the past, CMS has assigned non-FDA approved technologies a status indicator of “E1” and addressed the APC assignments once FDA approval is received. The manufacturer asserted the lack of payment assignments (APCs and status indicators) would significantly hinder commercialization of their neurostimulation system.
Response: We thank the commenters for all their valuable input. Because this vagal nerve neurostimulation system has not yet received FDA approval, we believe that we should reassign CPT codes 0908T, 0909T, 0910T, 0911T, and 0912T to status indicator “E1” to indicate that the code is not payable by Medicare when submitted on outpatient claims (any outpatient bill type). When we are notified by stakeholders that the FDA has approved this neurostimulation system, we will reassess the status indicator and APC assignments for these codes based on the comments we have received, in a future quarterly update and/or rulemaking cycle.
In summary, after consideration of the public comments received, we are finalizing our proposal, with modification, to assign status indicator “E1” to CPT codes 0909T through 0912T. The final status indicator assignment for the codes is listed in Table 110. We refer readers to Addendum D1 of this final rule with comment period for the complete list of the OPPS payment status indicators and their definitions for CY 2025. Addendum D1 is available via the internet on the CMS website.
( printed page 94125)66. VisONE® Synchronized Diaphragmatic StimulationTM (SDS®) System CPT Codes 0674T Through 0685T
The VisONE® Synchronized Diaphragmatic StimulationTM (SDS®) System is a technology being studied to treat select patients with chronic heart failure by delivering electrical stimulation to the diaphragm to modulate intrathoracic pressure pulses on the cardiovascular system. The SDS® System is comprised of an implantable pulse generator (IPG), sensing and therapy lead electrodes, a laparoscopic implant tool to place the leads, and an external programmer and telemetry wand.
There are 12 Category III CPT codes that may describe the various procedures related to the procedures involving the SDS® System: CPT codes 0674T-0685T. For CY 2025, as the SDS® System did not have FDA approval at the time of the proposed rule, we proposed to assign CPT codes 0674T-0685T to status indicator “E1” to indicate that the services are not paid by Medicare when submitted on outpatient claims (any outpatient bill type). Please refer to Table 111 for the full list of CPT codes and the proposed status indicators for CY 2025.
( printed page 94126)Comment: One commenter, specifically, the manufacturer of the SDS® System, requested clinical APC assignments for 9 of the 12 CPT codes ( printed page 94127) describing the various procedures involving the SDS® System. The commenter requested assignments to packaged status indicator “N” for the remaining three add-on CPT codes for which they did not request a clinical APC assignment.
Specifically, the commenter requested that, for CY 2025, CMS assign:
- CPT code 0674T to APC 5465 (Level 5 Neurostimulator and Related Procedures) with SI “J1” based on a crosswalk to CPT code 63685.
- CPT code 0675T to APC 5463 (Level 3 Neurostimulator and Related Procedures) with SI “J1” based on a crosswalk to CPT code 43647.
- CPT codes 0677T and 0679T to APC 5362 (Level 2 Laparoscopy and Related Services) with SI “J1” based on a crosswalk to CPT code 43648.
- CPT code 0680T to APC 5464 (Level 4 Neurostimulator and Related Procedures) with SI “J1” based on a crosswalk to CPT code 64590.
- CPT code 0681T and 0682T to APC 5461 (Level 1 Neurostimulator and Related Procedures) with SI “J1” based on a crosswalk to CPT code 64595.
- CPT code 0683T and 0685T to APC 5741 (Level 1 Electronic Analysis of Devices) with SI “Q1” based on crosswalks to CPT codes 93281 and 93288, respectively.
- SI “N” to CPT codes 0676T, 0678T, and 0684T.
The commenter also provided additional information regarding the FDA approval status of the SDS® System. Per the commenter, on July 26, 2024, the SDS® System received FDA approval for the Investigational Device Exemption (IDE) regarding the RECOVER-HF trial, which was anticipated to begin in late 2024. The request for Medicare coverage of the RECOVER-HF trial was pending when the comment was submitted. The commenter requested that CMS make the 12 codes payable effective January 1, 2025, in anticipation of Medicare's approval of their Category B IDE trial application.
Response: We thank the commenter for their input and agree with the suggested CPT crosswalk codes provided by the commenter.
We note that the RECOVER-HF trial was approved for Medicare coverage on September 23, 2024. Because the Category B IDE trial meets the CMS standards for coverage, and appears on the CMS approved IDE list on the CMS website, we are not finalizing our proposal to assign the 12 codes that describe the various procedures associated with the SDS® System to status indicator “E1.” In light of the recent Category B IDE study approval for the RECOVER-HF clinical trial, and based on our review of the comment received, more specifically the crosswalk codes recommended by the commenter, we are finalizing the following clinical APC and status indicator assignments:
( printed page 94128) ( printed page 94129)The final CY 2025 payment rate for the codes can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 to this final rule with comment period for the complete list of the OPPS payment status indicators and their definitions for CY 2025. Addendum D1 is available via the internet on the CMS website.
67. Xenograft Implantation Into the Articular Surface, CPT Code 0737T (APC 5115)
For the July 1, 2022, update, the AMA CPT Editorial Panel established CPT code 0737T (Xenograft implantation into the articular surface) to describe xenograft implantation for use in cartilage and osteochondral defects in joints. Since July 1, 2022, CPT code 0737T has been assigned to status indicator “E1” (Not paid by Medicare when submitted on outpatient claims (any outpatient bill type)) to indicate that the code was not payable under the OPPS. In the OPPS Quarterly Change Request (Transmittal 12816, CR 13784) dated August 29, 2024, we noted that the device associated with this code (Agili-CTM) had become available, and therefore we reassigned CPT code 0737T to status indicator “J1” (Hospital Part B Services Paid Through a Comprehensive APC; Paid under OPPS) and APC 5115 (Level 5 Musculoskeletal Procedures) effective July 1, 2024. For CY 2025, we proposed to continue to assign CPT code 0737T to APC 5115 with a proposed payment assignment of around $12,755.
Comment: We received a couple of comments expressing support for assigning CPT code 0737T to APC 5115 and status indicator “J1”. Commenters noted that the reimbursement was consistent with the costs associated with the procedure and would ensure equitable patient access, even allowing some beneficiaries to possibly avoid more costly knee replacement procedures. They requested that we maintain the APC and status indicator assignments for several years to gather claims data as they anticipate the patient volume will be limited due to the patient selection criteria.
Response: We thank the commenters for their input. Under the OPPS, we review our claims data on an annual basis to determine payment rates and will continue to monitor the claims data and the appropriateness of the APC assignment for CPT code 0737T to APC 5115.
In summary, we are finalizing our proposal, without modification, to assign CPT code 0737T to APC 5115 for CY 2025. The final CY 2025 payment rate for the code can be found in Addendum B to this final rule with comment period. In addition, we refer readers to Addendum D1 of this final rule with comment period for the SI meanings for all codes reported under the OPPS. Addenda B and D1 are available via the internet on the CMS website.
68. OPPS Payment for Software as a Service
New clinical software, which includes clinical decision support software, clinical risk modeling, and computer aided detection (CAD), is becoming increasingly available to providers. These technologies often perform data analysis of diagnostic images from patients. While many of these technologies are new, we note that clinical software, particularly CAD, has been used to aid or augment clinical decision making for decades. These technologies rely on complex algorithms or statistical predictive modeling to aid in the diagnosis or treatment of a patient's condition. We refer to these algorithm-driven services that assist practitioners in making clinical assessments, and that providers pay for either on a subscription or per-use basis, as Software as a Service (SaaS).
We note the proliferation of SaaS procedures approved by the FDA and their subsequent assignment of CPT codes by the AMA has led the agency to seek a workable SaaS payment strategy. We are seeking a strategy that would provide equitable payment for these new technologies while also preserving the integrity of the OPPS payment bundles, an essential component of a prospective payment system. We are seeking a payment strategy that aligns with our agency's mission to improve quality, health, reduce cost, and strengthen the health care system. We note that the manufacturer-supplied cost data of ( printed page 94130) these SaaS services is often high and the true return in investment for these services is still unknown. Moreover, many SaaS services often do not share clinical and resource similarity to existing medical services. Thus, it generally has been challenging to compare a new SaaS service to existing medical services for the purpose of determining a fair payment rate for these new services.
Although some have recently received separate payment for these SaaS procedures under the OPPS, we recognize that certain clinical decision support software, including machine learning or “AI,” has been available for many years. In the past ten years, clinical decision support software has been commonly used alongside electronic medical records by medical practitioners. Nonetheless, the number of FDA approved or cleared “machine learning” or “AI” clinical software programs has rapidly increased in the past few years. We note that the FDA has approved many SaaS procedures for similar functions: there are at least six software products that purport to detect findings in Computed Tomography (CT) studies of the chest.[29] Additionally, we note some clinical software developers are now using alternative licensing that charges per use rather than using the traditional annual subscription or bulk use subscription. As a result of these variables and potentially others, there is significant price variation within the SaaS procedure space.
We recognize that, as described in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72027), SaaS procedures are a heterogenous group of services, which presents challenges when it comes to adopting payment policy for SaaS procedures as a whole. In the CY 2023 OPPS/ASC final rule, we solicited public comment on a payment approach that would broadly apply to SaaS procedures and have received some valuable feedback. Some of the feedback includes incorporating AI taxonomy across agencies and interested parties, or not to establish a single policy that would apply to all SaaS-type technology but instead separately evaluate each new technology to determine the appropriate HCPCS coding, including whether or not a potential CPT code can be used to support payment for the separate and distinct service under the OPPS.
For CY 2025, we are continuing to work to formulate a workable SaaS payment policy that would enhance our ability to provide equitable payment for SaaS procedures while responsibly managing cost and protecting the Medicare trust fund. Specifically, we are working to address the following:
- Identifying a payment strategy that is applicable across the settings of care (physician offices, for example);
- Identifying the fair costs associated for SaaS services;
- Distinguishing services that should be paid separately versus services that should be packaged under a prospective payment system; and
- Identifying a payment strategy for SaaS services that are part of other medical devices versus those that are distinct services.
We did not make any proposals on OPPS payment for SaaS for CY 2025 but received a number of comments on the topic.
Comment: Commenters suggested CMS formulate a SaaS payment policy that would incentivize adoption of new AI and ensure access and provide stability for its adoption of SaaS technologies. Commenters stated that CMS payment decisions have not uniformly and consistently ensured appropriate levels of payment. Some commenters recommended that we adopt the term “Algorithm-based healthcare services” (ABHS), which include SaaS procedures. ABHS includes these services regardless of whether they are delivered by a third party, or a health care professional using the AI software through the cloud, the web, a workstation, or embedded in imaging acquisition hardware. Commenters also recommended that ABHS codes be initially assigned to New Technology APCs for at least 5 years based on manufacture-supply cost data, until sufficient claims data are available. Some commenters also recommend that we codify the 2023 packaging exemption for SaaS add-on codes policy.
Response: We appreciate the commenters' valuable input on a SaaS payment policy approach, and we will consider their input for any future SaaS payment policy. We note that the suggestion for any new SaaS services to remain in the initial New Technology APC assignment for at least 5 years is in contrast to our current Transitional Pass-Through policy of three years for device or drugs. Moreover, we would need to examine the feasibility of payment determination based on manufacture-supplied cost data, which has historically not been comparable when the claims data become available.
Comment: One commenter requested CMS clarify that SaaS codes created by the AMA CPT are to be vendor neutral, and that these codes do not represent any single vendor's services. The commenter stated the focus on a single commercial platform for a code is not accurate, confuses the applicability, and limits adoption of other tools for which the code was intended. The commenter recommended removing the current language that these codes are “associated with” a specific service, which may result in an increase in claims, therefore allowing for a more accurate understanding of actual costs associated with these services.
Response: We thank the commenter for the valuable input. We recognize that the proliferation of SaaS procedures has created a need for the agency to continue to update our payment approach. We are continuing to work to improve SaaS payment policy in future rulemakings to enhance our ability to provide equitable payment for SaaS procedures while responsibly managing cost and protecting the Medicare trust fund.
69. APC and Status Indicator Review Process
Each year, CMS receives a high volume of requests from stakeholders and other interested parties to make certain OPPS payment policy changes, including many requesting changes to the APC and status indicator assignments of new or revised codes. Current APC groupings, APC assignments, and status indicators are reflected in the various addenda to the proposed and final OPPS payment rules. In addition, comments within the scope of the proposed rule, are summarized and responded to in the annual OPPS final rule with comment period.
Comment: We received one comment requesting that CMS outline a process that describes the criteria used to determine which stakeholder requests regarding APC assignments are included in the OPPS proposed rule. The commenter suggested that all APC assignment requests, including APC structure requests, be included in the OPPS proposed rule and that we should follow a process similar to the IPPS, which includes an annual deadline, and requests discussed in the following year's proposed rule.
Response: We appreciate the feedback and will consider this comment for future rulemaking. As a reminder, codes are updated and changed throughout the year. CPT and Level II HCPCS code changes that affect the OPPS are published through the annual rulemaking cycle and through the OPPS quarterly update Change Requests (CRs). Generally, these code changes are effective January 1, April 1, July 1, or ( printed page 94131) October 1. CPT code changes are released by the AMA (via their website) while Level II HCPCS code changes are released to the public via the CMS HCPCS website. CMS recognizes the release of new CPT and Level II HCPCS codes outside of the formal rulemaking process via OPPS quarterly update CRs. Based on our review, we assign the new codes to interim status indicators (SIs) and APCs. These interim assignments are finalized in the OPPS/ASC final rules. This quarterly process offers hospitals access to codes that more accurately describe the items or services furnished and provides payment for these items or services in a timelier manner than if we waited for the annual rulemaking process. We solicit public comments on the new CPT and Level II HCPCS codes, status indicators, and APC assignments through our annual rulemaking process.
Comment: We received one comment requesting information about our process for assigning status indicator “E1” to CPT or HCPCS codes that are not FDA approved. The commenter asserts that the delay in timing (from FDA approval to APC and SI assignment) affects beneficiary access to new technologies and services and impacts how other payers will pay for the services.
Response: We appreciate the feedback. We believe for items and services that are not FDA approved, it is appropriate to assign the codes to status indicator “E1” to indicate that they are not payable by Medicare when submitted on outpatient claims (any outpatient bill type) because the services are either not covered by any Medicare outpatient benefit category, statutorily excluded by Medicare, or not reasonable and necessary. We rely on stakeholders to notify us upon FDA approval of their device and/or service and we work to ensure a timely transition and assign the applicable codes to APCs that reflect clinical and resource similarity to the new codes. We accept information regarding the device and/or service that the stakeholder can provide to us to assist us in our review. We remind readers that OPPS payment policies are updated quarterly through a sub-regulatory process.
IV. OPPS Payment for Devices
A. Pass-Through Payment for Devices
1. Beginning Eligibility Date for Device Pass-Through Status and Quarterly Expiration of Device Pass-Through Payments
a. Background
The intent of transitional device pass-through payment, as implemented at § 419.66, is to facilitate access for beneficiaries to the advantages of new and truly innovative devices by allowing for adequate payment for these new devices while the necessary cost data is collected to incorporate the costs for these devices into the procedure APC rate (66 FR 55861). Under section 1833(t)(6)(B)(iii) of the Act, the period for which a device category eligible for transitional pass-through payments under the OPPS can be in effect is at least 2 years but not more than 3 years. Prior to CY 2017, our regulation at § 419.66(g) provided that this pass-through payment eligibility period began on the date CMS established a particular transitional pass-through category of devices, and we based the pass-through status expiration date for a device category on the date on which pass-through payment was effective for the category. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79654), in accordance with section 1833(t)(6)(B)(iii)(II) of the Act, we amended § 419.66(g) to provide that the pass-through eligibility period for a device category begins on the first date on which pass-through payment is made under the OPPS for any medical device described by such category.
In addition, prior to CY 2017, our policy was to propose and finalize the dates for expiration of pass-through status for device categories as part of the OPPS annual update. This means that device pass-through status would expire at the end of a calendar year when at least 2 years of pass-through payments had been made, regardless of the quarter in which the device was approved. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79655), we changed our policy to allow for quarterly expiration of pass-through payment status for devices, beginning with pass-through devices approved in CY 2017 and subsequent calendar years, to afford a pass-through payment period that is as close to a full 3 years as possible for all pass-through payment devices. We also have an established policy to package the costs of the devices that are no longer eligible for pass-through payments into the costs of the procedures with which the devices are reported in the claims data used to set the payment rates (67 FR 66763).
We refer readers to the CY 2017 OPPS/ASC final rule with comment period (81 FR 79648 through 79661) for a full discussion of the current device pass-through payment policy.[30]
In the CY 2023 OPPS/ASC final rule with comment period, we finalized our policy to publicly post online OPPS device pass-through applications received on or after March 1, 2023, beginning with the issuance of the CY 2025 proposed rule and for each OPPS rulemaking thereafter. We refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 71934 through 71938) for a full discussion of the policy to publicly post OPPS device pass-through applications.
b. Expiration of Transitional Pass-Through Payments for Certain Devices
As stated earlier, section 1833(t)(6)(B)(iii) of the Act requires that, under the OPPS, a category of devices be eligible for transitional pass-through payments for at least 2 years, but not more than 3 years. Currently, there are 13 device categories eligible for pass-through payment. These devices are listed in Table 112 of this final rule where we detail the expiration dates of pass-through payment status for each of the 13 devices currently receiving device pass-through payment.
( printed page 94132) ( printed page 94133)As discussed in section IV.A.2. New Device Pass-Through Applications for CY 2024 of the CY 2024 OPPS/ASC final rule with comment period, we approved HCPCS code C1601 (Endoscope, single-use ( i.e. disposable), pulmonary, imaging/illumination device (insertable)), as a new device category for pass-through status under the OPPS, with an effective date of January 1, 2024. For the full discussion of the criteria used to evaluate device pass-through applications, refer to the CY 2024 OPPS/ASC final rule with comment period, which was published in the Federal Register on November 22, 2023 (88 FR 81729 through 81743). We note that HCPCS code C1601 was established for a bronchoscope that can only be used for a single procedure and cannot be reprocessed. As such, HCPCS code C1601 only describes devices that cannot be reprocessed.
In addition, as discussed in section IV.A.2 (New Device Pass-Through Applications for CY 2023) of the CY 2023 OPPS/ASC final rule with comment period, we approved HCPCS code C1747 (Endoscope, single-use ( i.e. disposable), urinary tract, imaging/illumination device (insertable)), as a new device category for pass-through status under the OPPS, with an effective date of January 1, 2023. For the full discussion on the criteria used to evaluate device pass-through applications, refer to the CY 2023 OPPS/ASC final rule with comment period, which was published in the Federal Register on November 23, 2022 (87 FR 71929 through 71934). We note that HCPCS code C1747 was established for a ureteroscope that can only be used for a single procedure and cannot be reprocessed. As such, HCPCS code C1747 only describes devices that cannot be reprocessed.
2. New Device Pass-Through Applications for CY 2025
a. Background
Section 1833(t)(6) of the Act provides for pass-through payments for devices, and section 1833(t)(6)(B) of the Act requires CMS to use categories in determining the eligibility of devices for pass-through payments. As part of implementing the statute through regulations, we have continued to believe that it is important for hospitals to receive pass-through payments for devices that offer substantial clinical improvement in the treatment of Medicare beneficiaries to facilitate access by beneficiaries to the advantages of the new technology. Conversely, we have noted that the need for additional payments for devices that offer little or no clinical improvement over previously existing devices is less apparent. In such cases, these devices can still be used by hospitals, and hospitals will be paid for them through appropriate APC payment. Moreover, a goal is to target pass-through payments for those devices where cost considerations are most likely to interfere with patient access (66 FR 55852; 67 FR 66782; and 70 FR 68629).
As specified in regulations at § 419.66(b)(1) through (3), to be eligible for transitional pass-through payment under the OPPS, a device must meet the following criteria:
- If required by FDA, the device must have received FDA approval or clearance and FDA marketing authorization (except for a device that has received an FDA investigational device exemption (IDE) and has been classified as a Category B device by FDA), or meet another appropriate FDA exemption; and the pass-through payment application must be submitted within 3 years from the date of the initial FDA marketing authorization, if required, unless there is a documented, verifiable delay in U.S. market availability after FDA marketing authorization is granted, in which case CMS will consider the pass-through payment application if it is submitted within 3 years from the date of market availability;
- The device is determined to be reasonable and necessary for the diagnosis or treatment of an illness or injury or to improve the functioning of a malformed body part, as required by section 1862(a)(1)(A) of the Act; and
- The device is an integral part of the service furnished, is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted (either permanently or temporarily), or applied in or on a wound or other skin lesion.
In addition, according to § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker).
Separately, we use the following criteria, as set forth under § 419.66(c), to determine whether a new category of pass-through payment devices should be established. The device to be included in the new category must—
- Not be appropriately described by an existing category or by any category previously in effect established for transitional pass-through payments, and was not being paid for as an outpatient service as of December 31, 1996;
- Have an average cost that is not “insignificant” relative to the payment amount for the procedure or service with which the device is associated as determined under § 419.66(d) by demonstrating: (1) the estimated average reasonable cost of devices in the category exceeds 25 percent of the applicable APC payment amount for the service related to the category of devices; (2) the estimated average reasonable cost of the devices in the category exceeds the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent; and (3) the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device exceeds 10 percent of the APC payment amount for the related service (with the exception of brachytherapy and temperature-monitored cryoablation, which are exempt from the cost requirements as specified at § 419.66(c)(3) and (e)); and
- Demonstrate a substantial clinical improvement, that is, substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment, or, for devices for which pass-through payment status will begin on or after January 1, 2020, as an alternative pathway to demonstrating substantial clinical improvement, a device is part of the FDA's Breakthrough Devices Program and has received marketing authorization for the indication covered by the Breakthrough Device designation.
Beginning in CY 2016, we changed our device pass-through evaluation and determination process. Device pass-through applications are still submitted to CMS through the quarterly subregulatory process, but the applications are subject to notice and comment rulemaking in the next applicable OPPS annual rulemaking cycle. Under this process, all applications that are preliminarily approved upon quarterly review will automatically be included in the next applicable OPPS annual rulemaking cycle, while submitters of applications that are not approved upon quarterly review will have the option of being ( printed page 94134) included in the next applicable OPPS annual rulemaking cycle or withdrawing their application from consideration. Under this notice-and-comment process, applicants may submit new evidence, such as clinical trial results published in a peer-reviewed journal or other materials, for consideration during the public comment process for the proposed rule. This process allows those applications that we are able to determine meet all of the criteria for device pass-through payment under the quarterly review process to receive timely pass-through payment status, while still allowing for a transparent, public review process for all applications (80 FR 70417 through 70418).
In the CY 2020 annual rulemaking process, we finalized an alternative pathway for devices that are granted a Breakthrough Device designation (84 FR 61295) and receive FDA marketing authorization for the indication covered by the Breakthrough Device designation. Under this alternative pathway, devices that are granted an FDA Breakthrough Device designation are not evaluated in terms of the current substantial clinical improvement criterion at § 419.66(c)(2) for the purposes of determining device pass-through payment status, but do need to meet the other requirements for pass-through payment status in our regulation at § 419.66. Devices that are part of the Breakthrough Devices Program, have received FDA marketing authorization for the indication covered by the Breakthrough Devices designation, and meet the other criteria in the regulation can be approved through the quarterly process and announced through that process (81 FR 79655). Proposals regarding these devices and whether pass-through payment status should continue to apply are included in the next applicable OPPS rulemaking cycle. This process promotes timely pass-through payment status for innovative devices, while also recognizing that such devices may not have a sufficient evidence base to demonstrate substantial clinical improvement at the time of FDA marketing authorization.
More details on the requirements for device pass-through payment applications are included on the CMS website in the application form itself at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/passthrough_payment.html, in the “Downloads” section. In addition, CMS is amenable to meeting with applicants or potential applicants to facilitate information sharing to support the evaluation of an OPPS device pass-through payment application or discuss general application criteria, including the substantial clinical improvement criterion.
b. Applications Received for Device Pass-Through Status for CY 2025
We received 14 complete applications by the March 1, 2024, quarterly deadline, which was the last quarterly deadline for applications to be received in time to be included in this final rule. Of the complete applications, we received two applications in the second quarter of 2023, two application in the third quarter of 2023, three applications in the fourth quarter of 2023, and seven applications in the first quarter of 2024. Three of the applications were approved for device pass-through payment during the quarterly review process: The DETOURTM System, which was preliminarily approved upon quarterly review under the alternative pathway effective January 1, 2024, and the AVEIRTM DR Dual Chamber Leadless Pacemaker System and the EndoSound Vision SystemTM (EVSTM) which both were preliminarily approved upon quarterly review under the alternative pathway effective July 1, 2024. As previously stated, all applications that are preliminarily approved upon quarterly review will automatically be included in the next applicable OPPS annual rulemaking cycle. Therefore, the DETOURTM System, the AVEIRTM DR Dual Chamber Leadless Pacemaker System, and the EndoSound Vision SystemTM (EVSTM) are discussed in the following section IV.2.b.1.
We note that the quarterly application process and requirements have not changed because of the addition of rulemaking review. Detailed instructions on submission of a quarterly device pass-through payment application are included on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/Downloads/catapp.pdf.
Discussions of the applications we received by the March 1, 2024, deadline are included below. We did not include the description and discussion of new device category for transitional pass-through payment status applications which were included in the proposed rule that were withdrawn for consideration for the upcoming calendar year. We also do not summarize or respond to public comments received regarding these withdrawn or ineligible applications.
(1) Alternative Pathway Device Pass-Through Applications
We received 10 device pass-through applications by the March 2024 quarterly application deadline for devices that have received Breakthrough Device designation from FDA and FDA marketing authorization for the indication for which they have a Breakthrough Device designation, and therefore were eligible to apply under the alternative pathway.
(a) AGENTTM Paclitaxel-Coated Balloon Catheter
Boston Scientific Corporation submitted an application for a new device category for transitional pass-through payment status for the AGENTTM Paclitaxel-Coated Balloon Catheter for CY 2025. Per the applicant, the AGENTTM Paclitaxel-Coated Balloon Catheter is a device/drug combination product consisting of a semi-compliant intracoronary balloon catheter with a paclitaxel/acetyl tributyl citrate drug coating on the balloon component. The applicant asserted that the AGENTTM Paclitaxel-Coated Balloon Catheter delivers paclitaxel, an antiproliferative drug, directly to the arterial tissue which inhibits the proliferation of neointimal smooth muscle cells without introducing an additional stent layer, thereby reducing the rate of restenosis. According to the applicant, the AGENTTM Paclitaxel-Coated Balloon Catheter is intended for use in adult patients, after appropriate vessel preparation, undergoing percutaneous coronary intervention (PCI) in coronary arteries 2.0 mm to 4.0 mm in diameter and lesions up to 26 mm in length for the purpose of improving myocardial perfusion when treating in-stent restenosis (ISR) and the management of atherosclerotic coronary artery disease.
Please refer to the online application posting for the AGENTTM Paclitaxel-Coated Balloon Catheter, available at https://mearis.cms.gov/public/publications/device-ptp/DEP2402295H2TU, for additional detail describing this device and the disease treated by the device.
Comment: Many commenters expressed their support for the approval of transitional pass-through payment for the AGENTTM Paclitaxel-Coated Balloon Catheter. The commenters, including the applicant, emphasized their positive views on the device's transformational nature as a treatment for ISR, the device's ability to demonstrate clinical improvement relative to other available treatments, and the device's safety profile. In addition, many commenters expressed their concerns that without ( printed page 94135) adequate reimbursement many medical facilities may not adopt widespread use of the AGENTTM Paclitaxel-Coated Balloon Catheter.
Response: We appreciate the commenters' input and support for the AGENTTM Paclitaxel-Coated Balloon Catheter application for transitional pass-through payment. We have taken these comments into consideration in our final determination for pass-through status for the AGENTTM Paclitaxel-Coated Balloon Catheter.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the AGENTTM Paclitaxel-Coated Balloon Catheter received FDA Breakthrough Device designation effective January 22, 2021, as a combination product indicated for percutaneous transluminal coronary angioplasty (PTCA) in coronary arteries 2.0 mm to 4.0 mm in diameter to treat ISR, up to 26 mm in length, for the purpose of improving myocardial perfusion. FDA approved the premarket approval application (PMA) for the AGENTTM Paclitaxel-Coated Balloon Catheter on February 29, 2024, as indicated for use after appropriate vessel preparation in adult patients undergoing PCI in coronary arteries 2.0 mm to 4.0 mm in diameter and lesions up to 26 mm in length for the purpose of improving myocardial perfusion when treating ISR. We noted that while the indication for the FDA Breakthrough Device designation and the indication for the FDA premarket approval vary slightly, we stated that we believe that FDA premarket approval indication is the indication covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the AGENTTM Paclitaxel-Coated Balloon Catheter on February 29, 2024, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the AGENTTM Paclitaxel-Coated Balloon Catheter meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the AGENTTM Paclitaxel-Coated Balloon Catheter received FDA premarket approval on February 29, 2024, for use after appropriate vessel preparation in adult patients undergoing PCI in coronary arteries 2.0 mm to 4.0 mm in diameter and lesions up to 26 mm in length for the purpose of improving myocardial perfusion when treating ISR. In addition, a few other commenters expressed their belief that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the newness criterion at § 419.66(b)(1).
Response: We appreciate the applicant's and commenters' input. The AGENTTM Paclitaxel-Coated Balloon Catheter received FDA Breakthrough Device designation effective January 22, 2021. We received the application for a new device category for transitional pass-through payment status for the AGENTTM Paclitaxel-Coated Balloon Catheter on February 29, 2024, which is within 3 years of February 29, 2024, the date of FDA premarket approval application. Based on our review of the application, we have determined that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criterion at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not explicitly indicate whether the AGENTTM Paclitaxel-Coated Balloon Catheter is integral to the service provided. The applicant stated that the AGENTTM Paclitaxel-Coated Balloon Catheter is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted as required by § 419.66(b)(3).
We invited public comment on whether the AGENTTM Paclitaxel-Coated Balloon Catheter meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant and another commenter asserted that the AGENTTM Paclitaxel-Coated Balloon Catheter meets all the requirements of § 419.66(b)(3). The applicant submitted a comment clarifying that the AGENTTM Paclitaxel-Coated Balloon Catheter is integral to the service provided and meets the eligibility criterion at § 419.66(b)(3) because the service cannot be furnished without the AGENTTM Paclitaxel-Coated Balloon Catheter. Specifically, the applicant commented that that the AGENTTM Paclitaxel-Coated Balloon Catheter is a device/drug combination product consisting of a semi-compliant intracoronary balloon catheter with a drug coating consisting of paclitaxel/acetyl tributyl citrate on the balloon component. In addition, the applicant provided that the AGENTTM Paclitaxel-Coated Balloon Catheter is inserted transiently in a coronary artery suffering from ISR and transfers a long-dwelling, therapeutic dose of paclitaxel to the vessel wall of the artery to prevent ISR reoccurrence. The applicant reiterated that the AGENTTM Paclitaxel-Coated Balloon Catheter is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted.
Response: We appreciate the applicant's and commenter's clarification, and we agree that the AGENTTM Paclitaxel-Coated Balloon Catheter is integral to the service furnished. We agree with the applicant and commenter that the AGENTTM Paclitaxel-Coated Balloon Catheter is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion. After consideration of the public comments received and our review of the application, we have determined that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not indicate whether the AGENTTM Paclitaxel-Coated Balloon Catheter is equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or if the AGENTTM Paclitaxel-Coated Balloon Catheter is a supply or material furnished incident to a service.
We invited public comment on whether the AGENTTM Paclitaxel-Coated Balloon Catheter meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant and commenter submitted a comment clarifying that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the eligibility criterion at § 419.66(b)(4) because it is not equipment, an instrument, apparatus, implement or any type of item for which depreciation and financing expenses are recovered, and because it is the primary therapeutic intervention and is not a supply or material furnished incident to a service.
Response: We appreciate the applicant's and commenter's clarification. We agree with the ( printed page 94136) applicant and commenter that the AGENTTM Paclitaxel-Coated Balloon Catheter is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. After consideration of the public comments received and our review of the application, we have determined that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe the AGENTTM Paclitaxel-Coated Balloon Catheter. Per the applicant, while device category C2623 (Catheter, transluminal angioplasty, drug-coated, non-laser) describes related or similar products to the AGENTTM Paclitaxel-Coated Balloon Catheter, the AGENTTM Paclitaxel-Coated Balloon Catheter is not appropriately described by C2623 because the devices in this category differ from the AGENTTM Paclitaxel-Coated Balloon Catheter in both size and indicated use, and therefore, the device is not appropriately described by C2623. The applicant further claimed that the devices described by C2623 are approved for use in the femoral or popliteal arteries in vessels with a diameter of at least 4.0 mm, whereas the AGENTTM Paclitaxel-Coated Balloon Catheter is indicated for use in coronary arteries that are between 2.0 mm to 4.0 mm in diameter. In addition, the applicant also noted that the length of the lesions (up to 180 mm) treated with devices in this device category greatly exceeds the maximum lesion size of 26 mm for the AGENTTM Paclitaxel-Coated Balloon Catheter. Moreover, the applicant asserted that the devices described by C2623 are used to treat peripheral arterial disease and are contraindicated for use in coronary arteries. Per the applicant, the AGENTTM Paclitaxel-Coated Balloon Catheter is used in conjunction with transluminal PCIs which are described by different procedure codes than the percutaneous transluminal angioplasty services used for the devices in C2623. Lastly, the applicant stated that an analysis of claims found that the devices described by C2623 are typically reported with femoral or popliteal revascularization procedures (HCPCS codes from 37224 to 37227).
We noted that, based on the description the applicant provided, the AGENTTM Paclitaxel-Coated Balloon Catheter is a device/drug combination product consisting of a semi-compliant intracoronary balloon catheter with a paclitaxel/acetyl tributyl citrate drug coating on the balloon component and thus could be appropriately described by C2623. Specifically, we stated that we believe that C2623 may appropriately describe the AGENTTM Paclitaxel-Coated Balloon Catheter because it is a non-laser, drug-coated catheter used for transluminal angioplasty procedures. In this context, we noted that we believe the AGENTTM Paclitaxel-Coated Balloon Catheter may be similar to the devices described by C2623, and therefore, the AGENTTM Paclitaxel-Coated Balloon Catheter may also be appropriately described by C2623.
In addition, while we acknowledged that when C2623 was established as a device category code effective April 1, 2015, the procedure codes with which C2623 could be reported (HCPCS code 37224 and HCPCS code 37226) were limited to use in the femoral or popliteal arteries. However, based on the subsequent changes that were made to the procedure codes with which C2623 could be reported, we did not agree that C2623 is limited to use with femoral or popliteal revascularization procedures. First, we noted that effective August 25, 2017, while C2623 was in device pass-through payment status, CMS added two procedure codes with which C2623 could be reported that were for procedures other than femoral popliteal revascularization procedures. Specifically, based on the FDA approval of a new indication for an existing device (a drug-coated balloon catheter for use with dialysis circuit procedures for the treatment of patients with dysfunctional arteriovenous fistulae),[31] CMS added two procedure codes, HCPCS codes 36902 and 36903 (transluminal balloon angioplasty procedures in peripheral dialysis segments),[32] with which C2623 could be reported effective August 25, 2017. The devices used with these two added HCPCS codes, 36902 and 36903, which are also described by C2623, are drug-coated balloon catheters used for dialysis circuit procedures in the upper extremities. We stated that we believe the inclusion of these additional reportable procedure codes illustrates that devices that may be described by C2623 were neither intended to be restricted to the treatment of vascular lesions of a specified dimension nor anatomically limited to femoral or popliteal revascularization procedures and is inconsistent with the applicant's assertion that the AGENTTM Paclitaxel-Coated Balloon Catheter is not appropriately described by C2623 because the category is only applicable for devices used in femoral or popliteal arteries with a diameter of at least 4.0 mm, and not smaller coronary arteries.
Further, beginning January 1, 2018, upon the expiration of device pass-through payment status for C2623, CMS packaged the payment for the costs of each of the devices described by C2623 into the payment for the costs related to the procedure with which each device is reported in the hospital claims data (FR 82 59321 through 59323). We further noted that upon becoming packaged for payment, C2623 effectively became reportable with other transluminal angioplasty procedure codes, including procedure codes for percutaneous coronary transluminal angioplasty services. Finally, we noted that while, per the applicant, the devices described by C2623 are typically reported with femoral or popliteal revascularization procedures, other procedure codes, including procedure codes for other percutaneous transluminal angioplasty services and ( printed page 94137) other related coronary procedure codes can and have been performed with devices described by C2623. As such, we stated that we believe the procedures with which the AGENTTM Paclitaxel-Coated Balloon Catheter is utilized could be reported with C2623.
In this context, based on the description the applicant provided, we stated that we believe the AGENTTM Paclitaxel-Coated Balloon Catheter may be similar to the devices described by C2623, and therefore, the AGENTTM Paclitaxel-Coated Balloon Catheter may also be appropriately described by C2623.
We invited public comment on whether the AGENTTM Paclitaxel-Coated Balloon Catheter meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concerns that the AGENTTM Paclitaxel-Coated Balloon Catheter may be appropriately described by C2623, the applicant and many commenters asserted that the AGENTTM Paclitaxel-Coated Balloon Catheter is not described by C2623 because the device is not used to perform transluminal angioplasties. Specifically, these commenters explained that the AGENTTM Paclitaxel-Coated Balloon Catheter is used to effectively deliver its drug to a lesion after the vessel wall has been prepared, typically through an angioplasty. They also asserted that C2623 is used to describe drug-coated balloons indicated for use in the peripheral vasculature which are capable of both angioplasty and drug-delivery; however, according to the commenters, the AGENTTM Paclitaxel-Coated Balloon Catheter is used solely for drug-delivery and cannot be used for performing an angioplasty. In addition, the applicant commented that peripheral drug-coated balloon catheters, like those described by C2623, are primarily used for treating typically diffused atherosclerotic/restenotic lesions in the comparatively elastic femoral or popliteal arteries, whereas coronary drug-coated balloons are used for treatment of substantially smaller, stented (and therefore, relatively inelastic) coronary segments, with largely focal lesions. Similarly, many commenters underlined the differences between the AGENTTM Paclitaxel-Coated Balloon Catheter and peripheral drug-coated balloon catheters, including design, size, deployment location and time length, clinical indication, and procedural complexity.
The applicant and a few commenters noted that CMS has historically included specific anatomy in several device categories, including those differentiating coronary and non-coronary devices. The applicant offered the following as examples of device categories that are specific to a particular type of intervention: C1759 (Catheter, intracardiac echocardiography), C1761 (Catheter, transluminal intravascular lithotripsy, coronary), and C1888 (Catheter, ablation, non-cardiac, endovascular). A few commenters highlighted device categories that are more granular in description than C2623, such as C1714 (Catheter, transluminal atherectomy, directional), C1724 (Catheter, transluminal atherectomy, rotational), C1732 (Catheter, electrophysiology, diagnostic/ablation, 3D or vector mapping), and C1733 (Catheter, electrophysiology, diagnostic/ablation, other than 3D or vector mapping, other than cool-tip), and suggested that CMS similarly acknowledge the differences between the AGENTTM Paclitaxel-Coated Balloon Catheter and those devices in device category C2623.
Response: We appreciate the applicant's and commenters' input. We note that while the FDA Breakthrough Device designation for the AGENTTM Paclitaxel-Coated Balloon Catheter stated a proposed indication for PTCA in coronary arteries 2.0 mm to 4.0 mm in diameter to treat ISR, up to 26 mm in length, for the purpose of improving myocardial perfusion, the FDA PMA for the AGENTTM Paclitaxel-Coated Balloon Catheter on February 29, 2024, stated the combination drug/device is indicated for use after appropriate vessel preparation in adult patients undergoing PCI in coronary arteries 2.0 mm to 4.0 mm in diameter and lesions up to 26 mm in length for the purpose of improving myocardial perfusion when treating ISR, removing the PTCA indication. This appears to be consistent with the applicant's and commenters' assertions that the AGENTTM Paclitaxel-Coated Balloon Catheter is not utilized in the treatment of PTCA. After consideration of the public comments received, we agree that the AGENTTM Paclitaxel-Coated Balloon Catheter is not indicated for use in transluminal angioplasty; rather, it is intended for improving myocardial perfusion when treating in-stent restenosis after appropriate vessel preparation. Further, we agree that C2623 describes drug-coated balloons which are capable of angioplasty, while the AGENTTM Paclitaxel-Coated Balloon Catheter appears to be used solely for drug-delivery and cannot be used for performing an angioplasty, including PCTAs. As such, we do not believe that the AGENTTM Paclitaxel-Coated Balloon Catheter is described by C2623.
We acknowledge that CMS has included specific anatomic language in the descriptors of previous device category codes when such language was necessary based on the unique circumstances surrounding the establishment of a particular device category code. We note that CMS does not establish pass-through device categories for the purposes of describing specific devices, but rather, device categories which are intended to encompass all devices that can be appropriately described by a category. As such, all device categories, regardless of the language utilized in the descriptors, are established in such a way that no medical device is described by more than one category in accordance with section 1833(t)(6)(B)(ii)(II) of the Act.
After consideration of the public comments received and our review of the application, we agree there is no existing category or category previously in effect that appropriately describes the AGENTTM Paclitaxel-Coated Balloon Catheter. Based on this information, we have determined that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The AGENTTM Paclitaxel-Coated Balloon Catheter has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in the discussion of the newness criterion), and therefore, is not evaluated for substantial clinical improvement.
We invited public comment on whether the AGENTTM Paclitaxel-Coated Balloon Catheter meets the device category criterion at § 419.66(c)(2)(ii).
Comment: The applicant and one commenter confirmed that the ( printed page 94138) AGENTTM Paclitaxel-Coated Balloon Catheter has received both Breakthrough Device designation and marketing authorization from FDA, meeting the device category criterion at § 419.66(c)(2).
Response: We appreciate the applicant's and commenter's input. The AGENTTM Paclitaxel-Coated Balloon Catheter has a Breakthrough Device designation effective January 22, 2021, and marketing authorization from FDA effective February 29, 2024, for the indication covered by the Breakthrough Device designation. Therefore, the AGENTTM Paclitaxel-Coated Balloon Catheter meets the criterion at 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application, we have determined that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the AGENTTM Paclitaxel-Coated Balloon Catheter would be reported with the HCPCS codes as shown in Table 113.
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant utilized the CY 2024 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5192, which had a CY 2024 payment rate of $5,445.84 at the time the application was received. HCPCS code 92920 in APC 5192 had a device offset amount of $1,662.61 at the time the application was received. According to the applicant, the cost of the AGENTTM Paclitaxel-Coated Balloon Catheter is $5,500.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $5,500.00 for the AGENTTM Paclitaxel-Coated Balloon Catheter is 101.00 percent of the applicable APC payment amount for the service related to the category of devices of $5,445.84 (($5,500.00/$5,445.84) × 100 = 101.00 percent). Therefore, we stated that we believe the AGENTTM Paclitaxel-Coated Balloon Catheter meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $5,500.00 for the AGENTTM Paclitaxel-Coated Balloon Catheter is 330.81 percent of the cost of the device-related portion of the APC payment amount for the related service of $1,662.61 (($5,500.00/$1,662.61) × 100 = 330.81 percent). Therefore, we stated that we believe the AGENTTM Paclitaxel-Coated Balloon Catheter meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $5,500.00 for the AGENTTM Paclitaxel-Coated Balloon Catheter and the portion of the APC payment amount for the device of $1,662.61 is 70.46 percent of the APC payment amount for the related service of $5,445.84 ((($5,500.00 − ( printed page 94139) $1,662.61)/$ 5,445.84) × 100 = 70.46 percent). Therefore, we stated that we believe the AGENTTM Paclitaxel-Coated Balloon Catheter meets the third cost significance requirement.
We invited public comment on whether the AGENTTM Paclitaxel-Coated Balloon Catheter meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to cost significance criteria, the applicant and a commenter reiterated that the AGENTTM Paclitaxel-Coated Balloon Catheter meets all three of the cost significance criteria.
Response: We appreciate the applicant's and commenter's input. After consideration of the public comments received and our findings from the first, second, and third cost significance tests, we agree that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments received and our review of the device pass-through application, we have determined that the AGENTTM Paclitaxel-Coated Balloon Catheter meets the requirements for device pass-through status described at § 419.66. We are finalizing approval for device pass-through payment status for the AGENTTM Paclitaxel-Coated Balloon Catheter effective January 1, 2025 under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(b) AveirTM DR Dual Chamber Leadless Pacemaker System
Abbott Laboratories submitted an application for a new device category for transitional pass-through payment status for the AveirTM DR Dual Chamber Leadless Pacemaker System (AveirTM DR System) for CY 2025. Per the applicant, the AveirTM DR System is comprised of two leadless pacemakers, one atrial and one ventricular with each containing a generator and electrodes, that provide dual-chamber pacing therapy after being placed within the heart's myocardium through a minimally invasive catheter-based procedure. According to the applicant, the AveirTM DR System is without the need for traditional wire electrodes and can provide beat-to-beat communication and synchrony between the two pacemakers for the treatment of arrhythmia/bradycardia. Per the applicant, patients with indication for dual-chamber pacing would benefit from a dual-chamber leadless pacemaker system that provides atrial and ventricular bradycardia therapy, while eliminating the complications associated with conventional pacing systems.
Please refer to the online application posting for the AveirTM DR System, available at https://mearis.cmsgov/public/publications/device-ptp/DEP230831B8DX0, for additional detail describing the device and the disease treated by the device.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the AveirTM DR System received FDA Breakthrough Device designation effective March 27, 2020, as a pacemaker implantation indicated in one or more of the following permanent conditions: syncope, presyncope, fatigue, disorientation due to arrhythmia/bradycardia, or any combination of these symptoms. FDA approved the premarket approval application (PMA) for the AveirTM DR System on June 29, 2023, for the indication covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the AveirTM DR System on March 23, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the AveirTM DR System meets the newness criterion at § 419.66(b)(1).
We did not receive public comments regarding whether the AveirTM DR System meets the newness criterion at § 419.66(b)(1). The AveirTM DR System received FDA Breakthrough Device designation effective March 27, 2020. We received the application for a new device category for transitional pass-through payment status for the AveirTM DR System on March 23, 2023, which is within 3 years of June 29, 2023, the date of FDA PMA. Based on our review of the application, we have determined that the AveirTM DR System meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. As noted in the proposed rule, the applicant did not indicate whether the AveirTM DR System is integral to the service furnished. The applicant also did not explicitly state that the AveirTM DR System is single-use; however, the applicant did state that one AveirTM DR System is required per patient per procedure. While the applicant did not explicitly state whether the AveirTM DR System comes in contact with human tissue or is surgically inserted or implanted, the applicant noted that the two AveirTM Delivery Catheters are inserted into the peripheral vasculature and the cardiovascular system to deliver and implant the AveirTM AR Atrial Leadless Pacemaker and the AveirTM VR Ventricular Leadless Pacemaker into the right atrium and right ventricle of the heart, respectively.
We invited public comment on whether the AveirTM DR System meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant submitted a comment confirming that the AveirTM DR System is an integral part of the service provided, is used for one patient only, comes in contact with human tissue, and is surgically inserted or implanted using two delivery catheters that are inserted into the peripheral vasculature and cardiovascular system to deliver and implant the AveirTM AR Atrial Leadless Pacemaker and the AveirTM VR Ventricular Leadless Pacemaker into the right atrium and right ventricle of the heart, respectively. The applicant also stated that therefore, it believes the AveirTM DR System meets the eligibility criterion at § 419.66(b)(3).
Response: We appreciate the applicant's input. We agree with the applicant that the AveirTM DR System is an integral part of the service furnished, is used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion. After consideration of the public comment received and our review of the application, we have determined that the AveirTM DR System meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not indicate whether the AveirTM DR System is equipment, an instrument, apparatus, implement, or item of this type for which depreciating and financing expenses are recovered, ( printed page 94140) or if the AveirTM DR System is a supply or material furnished incident to a service.
We invited public comment on whether the AveirTM DR System meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant confirmed that the AveirTM DR System is not an instrument, apparatus, implement, or similar item for which depreciation and financing expenses are recovered. The applicant also stated that the AveirTM DR System is not a supply or material furnished incident to a service, and therefore, meets the eligibility criterion at § 419.66(b)(4).
Response: We appreciate the applicant's input. With respect to the eligibility criterion at § 419.66(b)(4), we agree with the applicant that the AveirTM DR System is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. After consideration of the public comment received and our review of the application, we have determined that the AveirTM DR System meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. The applicant asserted that the AveirTM DR System is the only dual-chamber leadless pacemaker authorized by FDA and indicated for implantation in patients with one or more of the following permanent conditions: syncope, presyncope, fatigue, disorientation due to arrhythmia/bradycardia, or any combination of those conditions. Per the applicant, the AveirTM DR System is a modular dual-chamber leadless pacemaker system with bidirectional implant-to-implant communication that can accommodate all pacing indications. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe the AveirTM DR System. The applicant stated that device categories C1785 (Pacemaker, dual-chamber, rate-responsive (implantable)) and C1889 (Insertable/implantable device, not otherwise classified) do not appropriately describe the AveirTM DR System because the AveirTM DR System received Breakthrough Device designation from FDA and has specific functionality and capabilities that are new to the market. The applicant also asserted that the AveirTM DR system is modular, such that a single device can be implanted in a heart chamber initially, and the second pacemaker added to the other heart chamber in the future should the clinical need arise; and therefore, it is not appropriately described by either C1785 or C1889.
We did not identify an existing pass-through payment category that describes the AveirTM DR System. We invited public comment on whether the AveirTM DR System meets the device category criterion at § 419.66(c)(1).
Comment: The applicant agreed with CMS's assessment that there are no existing pass-through payment categories that describe the AveirTM DR System.
Response: We appreciate the applicant's input. After consideration of the public comment received and our review of the application, we continue to believe that there is no existing category or category previously in effect that appropriately describes the AveirTM DR System. Therefore, we have determined that the AveirTM DR System meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The AveirTM DR System has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail in the discussion of the newness criterion), and therefore, is not evaluated for substantial clinical improvement.
We invited public comment on whether the AveirTM DR System meets the device category criterion at § 419.66(c)(2)(ii).
We did not receive public comment regarding whether the AveirTM DR System meets the device category criterion at § 419.66(c)(2)(ii). However, the AveirTM DR System has a Breakthrough Device designation effective March 27, 2020, and a marketing authorization from FDA effective June 29, 2023, for the indication covered by the Breakthrough Device designation. Therefore, the AveirTM DR System meets the criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application, we have determined that the AveirTM DR System meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the AveirTM DR System would be reported with HCPCS codes as shown in Table 114.
( printed page 94141)To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2023 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5224, which had a CY 2023 payment rate of $18,672.01 at the time the application was received. We used the CY 2023 APC level device offset amount of $11,739.09 for APC 5224, as HCPCS codes 0795T and 0801T provided by the applicant were not included in Addendum P to the CY 2023 OPPS/ASC final rule with comment period and no CY 2023 HCPCS/CPT code level device offset amount was available at the time the application was received.[33] According to the applicant, the cost of the AveirTM DR System is $24,000.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $24,000.00 for the AveirTM DR System is 128.54 percent of the applicable APC payment amount for the service related to the category of devices of $18,672.01 (($24,000.00/$18,672.01) × 100 = 128.54 percent). Therefore, we stated that we believe the AveirTM DR System meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $24,000.00 for the AveirTM DR System is 204.45 percent of the cost of the device-related portion of the APC payment amount for the related service of $11,739.09 (($24,000.00/$11,739.09) × 100 = 204.45 percent). Therefore, we stated that we believe the AveirTM DR System meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $24,000.00 for the AveirTM DR System and the portion of the APC payment amount for the device of $11,739.09 is 65.66 percent of the APC payment amount for the related service of $18,672.01 ((($24,000.00−$11,739.09)/$ 18,672.01) × 100 = 65.66 percent). Therefore, we stated that we believe the AveirTM DR System meets the third cost significance requirement.
We invited public comment on whether the AveirTM DR System meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to cost significance criteria, the applicant reiterated that the AveirTM DR System meets all three of the cost significance criteria.
Response: We appreciate the applicant's input. After consideration of the public comment we received and our findings from the first, second, and third cost significance tests, we agree that the AveirTM DR System meets the cost significance criteria specified at § 419.66(d). ( printed page 94142)
Comment: The applicant submitted a comment requesting that CMS revise the proposed device offset for HCPCS code 0795T (Transcatheter insertion of permanent dual-chamber leadless pacemaker, including imaging guidance ( e.g., fluoroscopy, venous ultrasound, right atrial angiography, right ventriculography, femoral venography) and device evaluation ( e.g., interrogation or programming), when performed; complete system ( i.e., right atrial and right ventricular pacemaker components)) used to report dual-chamber leadless pacemaker procedures.
Response: We appreciate the applicant's input regarding the device offset amount associated with HCPCS code 0795T. We note that we have addressed this comment in Section IV.B of this final rule with comment period.
As discussed, the AveirTM DR System pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective July 1, 2024. After consideration of the public comment we received and our review of the device pass-through application, we have determined that the AveirTM DR System meets the requirements for device pass-through status described at § 419.66 and we are finalizing approval for device pass-through payment status for the AveirTM DR System under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(c) The DETOURTM System
Endologix, LLC submitted an application for a new device category for transitional pass-through payment status for the DETOURTM System for CY 2025. According to the applicant, the DETOURTM System is an implantable component, used to create a femoropopliteal bypass routed through the femoral vein. The DETOURTM System is comprised of two main components: (1) the TORUSTM Stent Graft System, which is comprised of the TORUSTM Stent Graft and the TORUSTM Stent Graft Delivery System, and (2) the ENDOCROSSTM Device. Per the applicant, the DETOURTM System is used to treat patients with advanced peripheral vascular disease, specifically those with long complex femoropopliteal artery stenoses and occlusions resulting in lifestyle limiting claudication or severe lower limb threatening ischemia. According to the applicant, the DETOURTM System can restore arterial blood flow to the lower limb around the blocked femoral artery and allows for venous blood flow around the conduit for normal venous return, to reduce signs and symptoms of lower limb ischemia and prevent amputation.
Please refer to the online application posting for the DETOURTM System, available at https://mearis.cms.gov/public/publications/device-ptp/DEP23090164QL2, for additional detail describing the device and the disease treated by the device.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the DETOURTM System received FDA Breakthrough Device designation effective September 2, 2020, under the name the PQ Bypass System, as a device intended for percutaneous revascularization of symptomatic femoropopliteal lesions 200mm to 460mm with a chronic total occlusion 100mm to 425mm, and/or moderate-to-severe calcification, and/or in-stent-restenosis in patients with severe peripheral arterial disease. FDA approved the premarket approval application (PMA) for the DETOURTM System on June 7, 2023, indicated for use for percutaneous revascularization in patients with symptomatic femoropopliteal lesions from 200 mm to 460 mm in length with chronic total occlusions (100 mm to 425 mm) or diffuse stenosis >70 percent who may be considered suboptimal candidates for surgical or alternative endovascular treatments. The DETOURTM System, or any of its components, is not for use in the coronary and cerebral vasculature. We noted that while the indication for the FDA Breakthrough Device designation and the indication for the FDA premarket approval vary slightly, we stated we believe that FDA premarket approval indication is the indication covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the DETOURTM System on September 1, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the DETOURTM System meets the newness criterion at § 419.66(b)(1).
We did not receive public comments regarding whether the DETOURTM System meets the newness criterion at § 419.66(b)(1). The DETOURTM System received FDA Breakthrough Device designation effective September 2, 2020. We received the application for a new device category for transitional pass-through payment status for the DETOURÔ System on September 1, 2023, which is within 3 years of June 7, 2023, the date of FDA PMA. Based on our review of the application, we have determined that the DETOURTM System meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, the DETOURTM System is integral to the service provided and is used for one patient only. While the applicant did not indicate whether the DETOURTM System comes in contact with human tissue, the applicant did specify that both components of the DETOURTM System, the TORUSTM Stent Graft System and the ENDOCROSSTM Device, are inserted or implanted during the percutaneous transmural femoropopliteal bypass procedure, as required by § 419.66(b)(3).
We invited public comments on whether the DETOURTM System meets the eligibility criterion at § 419.66(b)(3).
We did not receive public comments regarding whether the DETOURTM System meets the eligibility requirements of § 419.66(b)(3). As noted in the proposed rule, the applicant did not indicate that the DETOURTM System comes in contact with human tissue. However, because both components of the DETOURTM System, the TORUSTM Stent Graft System and the ENDOCROSSTM Device, are inserted or implanted during the percutaneous transmural femoralpopliteal bypass procedure, we believe that the DETOURTM System comes into contact with human tissue. After consideration of the information in the application, we have determined that the DETOURTM System meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant asserted that the DETOURTM System meets the device eligibility ( printed page 94143) requirements because it is not an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, and it is not a supply or material furnished incident to a service.
We invited public comment on whether the DETOURTM System meets the exclusion criterion at § 419.66(b)(4).
We did not receive public comments regarding whether the DETOURTM System meets the eligibility requirements of § 419.66(b)(4). Based on our review of the application, we agree with the applicant that the DETOURTM System is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. Therefore, we have determined that the DETOURTM System meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. The applicant described that the DETOURTM System is a minimally invasive, single-use device with an implantable component, used to create a femoropopliteal bypass routed through the femoral vein. The applicant provided a list of existing and previous device categories for pass-through payment for other stents and explained why they do not believe any of the categories describe the DETOURTM System. In summary, the applicant asserted that the referenced device categories do not adequately describe the DETOURTM System because, in contrast to the DETOURTM System, the referenced device categories do not have: (1) a crossing device with long needle for transmural access, (2) a crossing device with high pressure needle delivery for heavily calcified and atherosclerotic arteries, (3) a high radial strength transmural stent graft capable of self-support and sustaining blood flow through conduit bridging artery to vein and back to artery, (4) a percutaneous stent graft delivery catheter, (5) a covered stent graft to allow for arterial blood flow within the conduit as venous blood flows around it in the vein, or (6) a permanent implant to maintain arterial and venous blood flow. The reasons the applicant asserted for why the DETOURTM System is not adequately described by each of the device categories are shown in Table 115.
( printed page 94144)We did not identify an existing pass-through payment category that describes the DETOURTM System.
We invited public comment on whether the DETOURTM System meets the device category criterion at § 419.66(c)(1).
We did not receive any comments regarding whether the DETOURTM System meets the eligibility requirements at § 419.66(c)(1). Based on our review of the application, we continue to believe there is no existing category or category previously in effect that appropriately describes the DETOURTM System. Therefore, we have determined that the DETOURTM System meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The DETOURTM System has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail in the discussion of the newness criterion), and therefore, is not evaluated for substantial clinical improvement.
We invited public comment on whether the DETOURTM System meets the device category criterion at § 419.66(c)(2).
We did not receive public comment regarding whether the DETOURTM System meets the device category criterion at § 419.66(c)(2). However, the DETOURTM System has a Breakthrough Device designation effective September 2, 2020, and marketing authorization from FDA effective June 7, 2023, for the indication covered by the Breakthrough Device. Therefore, the DETOURTM System meets the criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application, we have determined that the DETOURTM System meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the DETOURTM System would be reported with the HCPCS code as shown in Table 116.
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2023 payment rates for the three tests of the cost criterion. For our calculations, like the applicant, we used APC 5193, which had a CY 2023 payment rate of $10,615.31 at the time the application was received. HCPCS code 0505T in APC 5193 had a CY 2023 device offset amount of $5,229.10 at the time the application was received. According to the applicant, the cost of the DETOURTM System is $25,000.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $25,000.00 for the DETOURTM System is 235.51 percent of the applicable APC payment amount for the service related to the category of devices of $10,615.31 (($25,000.00/$10,615.31) × 100 = 235.51 percent). Therefore, we stated that we believe the DETOURTM System meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $25,000.00 for the DETOURTM System is 478.09 percent of the cost of the device-related portion of the APC payment amount for the related service of $5,229.10 (($25,000.00/$5,229.10) × 100 = 478.09 percent). Therefore, we stated that we believe the DETOURTM System meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related ( printed page 94145) service. The difference between the estimated average reasonable cost of $25,000.00 for the DETOURTM System and the portion of the APC payment amount for the device of $5,229.10 is 186.25 percent of the APC payment amount for the related service of $10,615.31 ((($25,000.00−$5,229.10)/$10,615.31) × 100 = 186.25 percent). Therefore, we stated that we believe the DETOURTM System meets the third cost significance requirement.
We invited public comment on whether the DETOURTM System meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
We did not receive any comments regarding whether the DETOURÔ System meets the cost significance criteria as described at § 419.66(d). Based on our findings from the first, second, and third cost significance tests, we have determined that the DETOURÔ System meets the cost significance criteria specified at § 419,66(d).
Comment: The applicant commented that the device offset amount assigned to HCPCS code 0505T should remain at $0.00 to align with the precedent set for similarly situated devices and ensure that hospitals continue to receive full and fair reimbursement for C1604.
Response: We appreciate the applicant's input regarding the device offset amount associated with HCPCS code 0505T. We note that we have addressed this comment in section IV.B of this final rule with comment period.
As discussed, the DETOURTM System pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective January 1, 2024. After our review of the device pass-through application, we have determined that the DETOURÔ System meets the requirements for device pass-through status described at § 419.66 and we are finalizing approval for device pass-through payment status for the DETOURÔ System under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(d) EndoSound Vision SystemTM (EVSTM)
EndoSound, Inc. submitted an application for a new device category for transitional pass-through payment status for the EVSTM for CY 2025. The applicant is only seeking a new device category for transitional pass-through payment status for the Ultrasound Disposable Kit—Diagnostic/Therapeutic (UDK-T) component (hereinafter referred to as “UDK-T”) of the EVSTM . According to the applicant, the EVSTM is an ultrasound system designed to externally attach to an upper gastrointestinal (GI) endoscope (gastroscope/upper (EGD) endoscope). Per the applicant, the EVSTM is a device that, once attached to an EGD endoscope, temporarily converts the EGD endoscope to a fully capable endoscopic ultrasound (EUS) endoscope. The applicant asserted that the EVSTM can be coupled with an upper GI endoscope device to enable real-time ultrasound imaging, ultrasound guided needle aspiration, and other EUS guided procedures within the upper GI tract and surrounding organs. According to the applicant, the EVSTM consists of: (1) the EVS Scanner, a beamformer/scanner that performs ultrasound signal processing; (2) the Ultrasound Transducer Module (UTM), a reusable transducer assembly that converts the electrical signals from the scanner into ultrasound energy; (3) the Transducer Extension Cable (TEC), a cable/connector to interface the UTM to the EVS Scanner; and (4) the UDK-T, a disposable mounting kit with an operator control mechanism used to externally affix the EVSTM to a standard EGD endoscope and to provide needle and transducer angulation while maintaining the native gastroscope controls.
Please refer to the online application posting for the EVSTM, available at https://mearis.cms.gov/public/publications/device-ptp/DEP240228GJT0X, for additional detail describing this device and the disease treated by the device.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the EVSTM, which includes the UDK-T, received FDA Breakthrough Device designation effective July 29, 2021, as a device intended to provide optical visualization of, ultrasonic visualization of, and therapeutic access to, the upper GI tract including but not restricted to the organs, tissues, and subsystems: esophagus, stomach, duodenum, and underlying areas. FDA granted the applicant 510(k) clearance for the EVSTM on December 27, 2023, indicated for use such that when affixed to an endoscope, is intended to provide ultrasonic visualization of, and ultrasound guided therapeutic access to the upper GI tract including but not restricted to the organs, tissues, and subsystems: esophagus, stomach, duodenum, and underlying areas. The EVSTM, mounted on an endoscope, is introduced orally when indications consistent with the requirement for a GI procedure are met. The EVSTM is a prescription-only device to be used by a qualified physician. The clinical environments where the system can be used include clinics, hospitals, and ambulatory surgery centers. We noted that while the indication for the FDA Breakthrough Device designation and the indication for the FDA 510(k) clearance vary slightly, we stated we believe that the FDA 510(k) clearance indication is the one covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the EVSTM on February 28, 2024, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the EVSTM, inclusive of the UDK-T component, meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant clarified that the EVSTM received FDA market authorization for 510(k) clearance. The applicant asserted that, according to FDA guidance,[34] 510(k) clearance is permissible for pre-determined Breakthrough Device designation and stated there were no changes specific to device components, mechanism of use, or indications of use for either the Breakthrough Device designation or the 510(k) clearance. The applicant stated the 510(k) clearance occurred within 3 years of the submission of the pass-through application such that the EVSTM meets the newness criterion at 42 CFR 419.66(b)(1).
Response: We appreciate the commenter's input. The EVSTM received Breakthrough Device designation effective July 29, 2021. We received the application for a new device category for transitional pass-through payment status for the EVSTM on February 28, 2024, which is within 3 years of December 27, 2023, the date of FDA 510(k) clearance. Based on our review of the application, we have determined that the EVSTM meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criterion at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in ( printed page 94146) contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not indicate whether the UDK-T component of the EVSTM is integral to the service furnished; however, the applicant did indicate that the UDK-T is single-use, comes in contact with human tissue, and is inserted as part of an endoscopy procedure. We preliminarily approved the EVSTM HCPCS code C1606 (Adapter, single-use ( i.e., disposable), for attaching ultrasound system to upper gastrointestinal endoscope) upon quarterly review under the alternative pathway with an effective date of July 1, 2024. We noted that C1606 was established for an adapter for attaching an ultrasound system to an upper GI endoscope that can only be used for a single procedure and cannot be reprocessed. As such, C1606 only describes devices that cannot be reprocessed.
We invited public comment on whether the UDK-T component of the EVSTM meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant submitted a comment reiterating that the EVSTM is used for one patient, comes into contact with human tissue, and is surgically implanted or inserted. The applicant clarified that the C1606 device component ( i.e., the UDK-T component) of the EVSTM meets the § 419.66(b)(3) criteria and is integral to the function and operation of the EVSTM . The applicant further commented that C1606 was established for an adapter for attaching an ultrasound system to an upper GI endoscope that can only be used for a single procedure and cannot be reprocessed. The applicant asserted that the C1606 device component of the EVSTM meets the eligibility criterion at 42 CFR 419.66(b)(3).
Response: We appreciate the applicant's clarification. We agree with the applicant that the UDK-T component of the EVSTM is integral to the function and operation of the EVSTM and that the device is used for one patient only, comes into contact with human tissue, and is surgically implanted or inserted. After consideration of the applicant's comments and our review of the application we have determined that the UDK-T component of the EVSTM meets the eligibility requirement at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant claimed that the UDK-T meets the device eligibility requirements because it is not equipment or an item for which depreciation and financing expenses are recovered. In addition, the applicant asserted that the UDK-T is not a supply or material.
We invited public comment on whether the UDK-T component of the EVSTM meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant submitted a comment reiterating that the UDK-T component of the EVSTM is not equipment or an item for which depreciation and financing expenses are recovered and is not a supply or material.
Response: We appreciate the applicant's input. We agree with the applicant that the UDK-T component of the EVSTM is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. After consideration of the applicant comments we received and our review of the application, we have determined that the UDK-T component of the EVSTM meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. According to the applicant, the EVSTM is an ultrasound system designed to externally attach to an upper GI endoscope (gastroscope/upper (EGD) endoscope). According to the applicant, no previous or existing device categories for pass-through payment appropriately describe the UDK-T. Per the applicant, device category C1748 (Endoscope, single-use ( i.e., disposable), Upper GI, imaging/illumination device (insertable)) does not appropriately describe the EVSTM, inclusive of the UDK-T, because: (1) the EVSTM, inclusive of the UDK-T, enables an endoscope that a hospital has to have added functionalities such as the ability to perform an EUS procedure, but is not an endoscope like the devices in C1748; (2) the EVSTM, inclusive of the UDK-T, when used with an endoscope allows EUS procedures to be done without an elevator, unlike the other devices described in C1748; and (3) the EVSTM, inclusive of the UDK-T, and the devices described in C1748 are used in different procedures. The applicant explained that CMS indicated that C1748 should always be billed with a HCPCS code in the ranges of 43260-43265 and 43274-43278, but there is no overlap between those HCPCS codes billed with C1748 and the HCPCS codes the applicant stated that the EVSTM would be reported with as shown in Table 48 of the CY 2025 OPPS/ASC proposed rule.
We did not identify an existing pass-through payment category that describes the UDK-T component of the EVSTM.
We invited public comment on whether the UDK-T component of the EVSTM meets the device category criterion at § 419.66(c)(1).
Comment: The applicant agreed with CMS' assessment that there are no existing pass-through payment categories that describe the UDK-T component of the EVSTM . The applicant further asserted that there is no other product similar in how the EVSTM assembles, operates, and functions and that, therefore, the device meets the criteria in 42 CFR 419.66(c)(1).
Response: We appreciate the applicant's input. After consideration of the comment and our review of the application, we continue to believe that there is no existing category or category previously in effect that appropriately describes the UDK-T component of the EVSTM . Therefore, we have determined that the UDK-T component of the EVSTM meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough ( printed page 94147) Device designation. The EVSTM, inclusive of the UDK-T, has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail in the discussion of the newness criterion) and therefore is not evaluated for substantial clinical improvement.
We invited public comment on whether the EVSTM, inclusive of the UDK-T component, meets the device category criterion at § 419.66(c)(2)(ii).
Comment: The applicant reiterated that the EVSTM and its components were granted Breakthrough Device designation and received FDA marketing authorization for the indication covered by the Breakthrough Device designation, and therefore, meets the criteria at 42 CFR 419.66(c)(2)(ii).
Response: We appreciate the applicant's comment. The EVSTM and its components have a Breakthrough Device designation effective July 29, 2021, and marketing authorization from FDA effective December 27, 2023, for the indication covered by the Breakthrough designation. Therefore, the EVSTM meets the criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application and the applicant's comments, we have determined that the EVSTM meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the EVSTM would be reported with HCPCS codes as shown in Table 117.
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2024 payment rates for the three tests of the cost criterion. For our calculations, ( printed page 94148) we used APC 5302, which had a CY 2024 payment rate of $1,812.99 at the time the application was received. HCPCS code 43232 in APC 5302 had a CY 2024 device offset amount of $14.50 at the time the application was received.[35] According to the applicant, the cost of the disposable, single-use UDK-T component of the EVSTM is $500.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $500.00 for the UDK-T is 27.59 percent of the applicable APC payment amount for the service related to the category of devices of $1,812.99 (($500.00/$1,812.99) × 100 = 27.59 percent). Therefore, we stated that we believe the UDK-T component of the EVSTM meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $500.00 for the UDK-T is 3,448.28 percent of the cost of the device-related portion of the APC payment amount for the related service of $14.50 (($500.00/$14.50) × 100 = 3,448.28 percent). Therefore, we stated that we believe the UDK-T component of the EVSTM meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $500.00 for the UDK-T and the portion of the APC payment amount for the device of $14.50 is 26.78 percent of the APC payment amount for the related service of $1,812.99 ((($500.00 − $14.50)/$1,812.99) × 100 = 26.78 percent). Therefore, we stated that we believe the UDK-T component of the EVSTM meets the third cost significance requirement.
We invited public comment on whether the UDK-T component of the EVSTM meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to the cost criterion, the applicant agreed that the UDK-T component of the EVSTM meets all three of the cost criterion outlined in 42 CFR 419.66(c)(3) and (d)(1) through (3).
Response: We appreciate the applicant's input. After consideration of the comment we received and our findings from the first, second, and third cost significance tests, we agree that the UDK-T component of the EVSTM meets the cost significance criteria specified at § 419.66(d).
As discussed, the UDK-T component of the EVSTM pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective July 1, 2024. After considering the applicant comment received and our review of the device pass-through application, we have determined that the UDK-T component of EVSTM meets the requirements for device pass-through status described at § 419.66. We are finalizing approval for device pass-through payment status for the UDK-T component of the EVSTM under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
Comment: The applicant commented that HCPCS codes 43240 and 43253 should be added to the list of procedures that can be reported with C1606. The applicant stated that in Table 48 of the CY 2025 OPPS/ASC proposed rule, there are 6 primary HCPCS procedures codes reported with the nominated device, including HCPCS codes 43231, 43232, 43237, 43238, 43242, and 43259. The applicant stated that in the July 2024 ASC code pairs file,[36] dated June 14, 2024, 8 primary HCPCS procedures codes were listed with HCPCS code C1606, which included the HCPCS codes listed included those listed in proposed rule, with the addition of HCPCS codes 43240 and 43253. The applicant agreed with the preliminary approval of pass-through status for the nominated device and requested that it should be made clear that the 8 procedures codes on the July 2024 ASC code pairs file for C1606 are the procedures that can be reported with C1606.
Response: We thank the applicant for the comment. We will evaluate this request and add HCPCS codes 43240 and 43253 to the list of procedures that can be reported with C1606 if appropriate.
(e) iFuse Bedrock GraniteTM Implant System
SI-BONE submitted an application for a new device category for transitional pass-through payment status for the iFuse Bedrock GraniteTM Implant System for CY 2025. According to the applicant, the iFuse Bedrock GraniteTM Implant System consists of iFuse GraniteTM implants of various lengths and diameters and associated instruments sets. The titanium (Ti-6Al-4V ELI) iFuse GraniteTM implant consists of a porous fusion sleeve with threaded length attached to a solid post that has connection and implant placement features of a typical pedicle fixation screw. The iFuse GraniteTM implant is intended to provide sacropelvic fusion of the sacroiliac joint (when placed in the sacral-alar-iliac (SAI) trajectory) and fixation to the pelvis when used in conjunction with commercially available pedicle screw fixation systems as a foundational element for segmental spinal fusion only when performing both a lumbar and a sacroiliac joint (SIJ) fusion procedure in the same operative session. The applicant asserted that joint fusion occurs as a result of the device's porous surface and interstices and fixation occurs through the device's helical threaded design and traditional posterior fixation rod connection.
Per the applicant, the device can be placed into the pelvis in two trajectories: the SAI trajectory ( i.e., into the sacrum, across the SIJ and into the ilium), or directly into the ilium. The applicant explained that the iFuse GraniteTM implant is typically placed in the SAI trajectory, bilaterally, and ( printed page 94149) oftentimes stacked to achieve two points of fusion and fixation/stabilization across each SIJ. According to the applicant, the iFuse GraniteTM implant may also be used in a single, but bilateral, configuration, where only two implants may be required when replacing traditional pedicle screws in either a SAI trajectory or iliac trajectory. The applicant asserted that the iFuse Bedrock GraniteTM Implant System is always used in addition to lumbar fusion instrumentation when used to perform lumbar and SIJ fusion at the same time.
Please refer to the online application posting for the iFuse Bedrock GraniteTM Implant System, available at https://mearis.cms.gov/public/publications/device-ptp/DEP240220LPFNM, for additional detail describing the device and the disease treated by the device.
Comment: One commenter strongly encouraged CMS to consider the iFuse Bedrock GraniteTM Implant System as having met all requirements for transitional pass-through payment status for CY 2025 and noting that that the device has Breakthrough Device designation for lumbar spinal deformity indications and was granted with New Technology Add-on Payment status effective in FY 2023 (87 FR 48780).
Response: We appreciate the commenters' input and support for the iFuse Bedrock GraniteTM Implant System application for transitional pass-through payment. We have taken these comments into consideration in our final determination for pass-through status for the iFuse Bedrock GraniteTM Implant System.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the iFuse Bedrock GraniteTM Implant System received FDA Breakthrough Device designation effective November 23, 2021, as a treatment of the acute and chronic instabilities or deformities of the thoracic, lumbar, and sacral spine, including: (1) degenerative disc disease (DDD), as defined by back pain of discogenic origin with degeneration of the disc confirmed by patient history and radiographic studies; (2) severe spondylolisthesis (Grades 3 and 4) of the L5-S1 vertebra; (3) skeletally mature patients receiving fusions by autogenous bone graft having implants attached to the lumbar and sacral spine (L3 to sacrum) with removal of the implants after the attainment of a solid fusion; (4) spondylolisthesis; (5) trauma ( i.e., fracture or dislocation); (6) spinal stenosis; (7) deformities or curvatures ( i.e., scoliosis, kyphosis, and/or lordosis); (8) spinal tumor; and (9) pseudarthrosis, and/or failed previous fusion. Subsequently, FDA also granted the applicant 510(k) clearance for the iFuse Bedrock GraniteTM Implant System on May 26, 2022, and December 22, 2022, for the indication covered by the Breakthrough Device designation with one additional indication for use: SIJ dysfunction that is a direct result of SIJ disruption and degenerative sacroiliitis, including conditions whose symptoms began during pregnancy or in the peripartum period and have persisted postpartum for more than 6 months. We noted that the 510(k) clearance dated December 22, 2022, expanded the previously cleared indication of the iFuse Bedrock GraniteTM Implant System to include general compatibility with certain compatible pedicle screw systems, whereas the indications under the May 26, 2022, 510(k) clearance only addressed compatibility of the iFuse Bedrock GraniteTM Implant System with the SeaSpine Mariner Pedicle Screw System. Each 510(k) clearance, the May 26, 2022, and the December 22, 2022, are covered by the November 23, 2021, Breakthrough Device designation for the iFuse Bedrock GraniteTM Implant System. We received the application for a new device category for transitional pass-through payment status for the iFuse Bedrock GraniteTM Implant System on February 20, 2024, which is within 3 years of the dates of the May 26, 2022, and December 22, 2022, FDA marketing authorizations for the iFuse Bedrock GraniteTM Implant System.
We invited public comment on whether the iFuse Bedrock GraniteTM Implant System meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the iFuse Bedrock GraniteTM Implant System received FDA marketing authorization on May 26, 2022. The applicant requested that CMS begin the newness period for the iFuse Bedrock GraniteTM Implant System using the FDA Breakthrough Device designation of November 23, 2021, and the earlier market availability date of May 26, 2022, for the 510(k) marketing authorization.
Response: We appreciate the applicant's input. We agree with the applicant that the market availability date for the iFuse Bedrock GraniteTM Implant System should be May 26, 2022, rather than December 22, 2022, because that is when the device became commercially available. We received the application for a new device category for transitional pass-through payment status for the iFuse Bedrock GraniteTM Implant System on February 20, 2024, which is within 3 years of May 26, 2022, the date of 510(k) clearance. After consideration of the public comments received and our review of the application, we have determined that the iFuse Bedrock GraniteTM Implant System meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criterion at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not indicate if the iFuse Bedrock GraniteTM Implant System is integral to the service furnished. The applicant provided that the iFuse Bedrock GraniteTM Implant System is single-use, permanently implanted, and surgically inserted into the patient. However, we noted that we do not have sufficient information to determine if the associated instruments sets included in the iFuse Bedrock GraniteTM Implant System meet the eligibility criterion at § 419.66(b)(3).
We invited public comment on whether the iFuse Bedrock GraniteTM Implant System meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant submitted a comment clarifying that the iFuse Bedrock GraniteTM Implant System, including the associated instrument sets included in the iFuse Bedrock GraniteTM Implant System, meet the eligibility criterion at § 419.66(b)(3). Specifically, the applicant stated that the iFuse Bedrock GraniteTM Implant System technology, including the associated instrument sets is integral to the service furnished, is single-use, permanently implanted, and surgically inserted into the patient, aligning fully with § 419.66(b)(3).
Response: We thank the applicant for the additional information. We agree with the applicant that the iFuse Bedrock GraniteTM Implant System, including the associated instrument sets, is an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion. After consideration of the public comments received and our review of the application, we have determined that the iFuse Bedrock GraniteTM Implant System meets the criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through ( printed page 94150) payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not indicate whether the iFuse Bedrock GraniteTM Implant System is equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or if the iFuse Bedrock GraniteTM Implant System is a supply or material furnished incident to a service.
We invited public comment on whether the iFuse Bedrock GraniteTM Implant System meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant submitted a comment clarifying that the iFuse Bedrock GraniteTM Implant System is not a material or supply furnished incident to a service and meets the eligibility criterion at § 419.66(b)(4) because neither the implant nor its associated instrument sets are a material or supply furnished incident to a service, and that the iFuse Bedrock GraniteTM Implant System operates like a vast majority of spine and orthopedic hardware typically used in surgery of this type. The applicant also confirmed that the iFuse Bedrock GraniteTM Implant System is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets.
Response: We appreciate the applicant's input. We agree with the applicant that the iFuse Bedrock GraniteTM Implant System, including the associated instruments, is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply such as a suture or clip furnished incident to a service. After consideration of the public comments received and our review of the application, we have determined that the iFuse Bedrock GraniteTM Implant System meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe the iFuse Bedrock GraniteTM Implant System. Per the applicant, the device category C1821 (Interspinous process distraction device) does not appropriately describe the iFuse Bedrock GraniteTM Implant System because the iFuse Bedrock GraniteTM Implant System is used to fixate and fuse, while the devices described in C1821 are interspinous spacers which, after implantation, are opened or expanded to distract the neural foramina and decompress the nerves. The applicant asserted that device category C1713 (Anchor/screw for opposing bone-to-bone or soft tissue-to-bone (implantable)) also does not appropriately describe the iFuse Bedrock GraniteTM Implant System because the iFuse Bedrock GraniteTM Implant System allows for simultaneous fusion of the SIJ and fixation of the pelvis by connecting via the Tulip Connector to the base of the stabilizing rods within the lumbosacral spinal construct, while C1713 includes implantable pins and/or screws that are used to oppose soft tissue-to-bone, tendon-to-bone, or bone-to-bone. Per the applicant, the device category C1889 (Implantable/insertable device, not otherwise classified) also does not appropriately describe the iFuse Bedrock GraniteTM Implant System because it does not describe any specific device category, and therefore does not uniquely describe the device category proposed for the iFuse Bedrock GraniteTM Implant System.
We noted that, according to the applicant, the iFuse Bedrock GraniteTM Implant System is intended to provide sacropelvic fusion of the sacroiliac joint (when placed in the SAI trajectory) and fixation to the pelvis when used in conjunction with commercially available pedicle screw fixation systems as a foundational element for segmental spinal fusion only when performing both a lumbar and SIJ fusion procedure in the same operative session. The applicant asserted that joint fusion occurs as a result of the device's porous surface and interstices and fixation occurs through the device's helical threaded design and traditional posterior fixation rod connection. We stated that we believe that the device category C1713 may appropriately describe the iFuse Bedrock GraniteTM Implant System and questioned whether a transfixing device utilizing the Tulip Connector is sufficiently distinguishable from traditional implantable pins or screws that it is meant to replace. In this context, based on the description the applicant provided, we stated that we believe the iFuse Bedrock GraniteTM Implant System may be similar to the devices described by C1713, and therefore, the iFuse Bedrock GraniteTM Implant System may also be appropriately described by C1713.
In addition, we stated that we believe the device category C1889 may appropriately describe the iFuse Bedrock GraniteTM Implant System because C1889 may be used to describe any implantable/insertable device that is not otherwise described by a more specific device category and is, therefore, sufficiently broad to include implantable devices that allow for simultaneous fusion of the SIJ and fixation of the pelvis. We noted that in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79562), CMS created C1889 with the specific intent to recognize devices furnished during a device intensive procedure that are not described by a specific Level II HCPCS Category C-code. In this context, we stated that we believe the iFuse Bedrock GraniteTM Implant System may be appropriately described by either C1713 or C1889.
We invited public comment on whether the iFuse Bedrock GraniteTM Implant System meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concerns that the iFuse Bedrock GraniteTM Implant System may be appropriately described by the device pass-through category C1713, the applicant and a few other commenters asserted that the iFuse Bedrock GraniteTM Implant System is not described by C1713. Specifically, the applicant relied upon the C1713 descriptor and supporting guidance: C1713: Anchor for opposing bone-to-bone or soft tissue-to-bone—Implantable pins and/or screws that are used to oppose soft tissue-to-bone, tendon-to-bone, or bone-to-bone. Screws oppose tissues via drilling as follows: soft tissue-to-bone, tendon-to-bone, or bone to-bone fixation. Pins are inserted or drilled into bone, principally with the intent to facilitate stabilization or oppose bone-to-bone. This may include orthopedic plates with accompanying washers and nuts. This category also applies to synthetic bone substitutes that may be used to fill bony void or gaps ( i.e., bone substitute implanted into a bony defect created from trauma or surgery). The applicant and commenters clarified that, unlike ( printed page 94151) devices described by C1713, the iFuse Bedrock GraniteTM Implant System's intended use is not to anchor bone to bone, or soft tissue, tendons, or ligaments to bone, but to promote simultaneous pelvic stabilization and fusion across the SI joint space, via a unique and proprietary Tulip Connector that attaches to the spinal fusion construct. The applicant and another commenter further provided that the typical use scenarios for reporting C1713 include orthopedic procedures involving bone sutures, anchors, small plastic polyether ether ketone (PEEK) screws for bone-to-bone or bone-to-tendon/tissue connections, often in anatomic areas outside of the spine (including the extremities, shoulders, knees and small bones). Furthermore, the applicant and commenter noted that C1713 is used to report procedures that are mostly non-fusion applications with the intent to restore motion or function to anatomy(ies) and that C1713 can also include bone void filler or bone substitute materials ( e.g., demineralized bone matrix, Subchondroplasty, etc.).
The applicant and commenters specified that other implanted devices in this category restore motion while the iFuse Bedrock GraniteTM Implant System immobilizes; and that the iFuse Bedrock GraniteTM Implant System is intended to traverse an articulating joint space and fuse two bones via bridging the joint to immobilize it so bony fusion may occur.
Response: We appreciate the applicant and commenters' clarifications and the additional information. We agree that the ability to simultaneously fuse the SIJ and fixate the pelvis to the spinal fusion construct distinguishes the iFuse Bedrock GraniteTM Implant System from other implant devices described by C1713 that anchor bone-to-bone, or soft tissue-to-bone, tendons-to-bone, or ligaments-to-bone. We also agree that the iFuse Bedrock GraniteTM Implant System is distinguishable from other implant devices described by C1713 because it secures bone to a metal rod via the Tulip Connector, and that typical C1713 implants do not include joint fusion technologies, and are not specific to anatomic areas, nor do they function similarly to the iFuse Bedrock GraniteTM.
After consideration of the public comments we received and our review of the application, we agree that existing device category C1713 does not appropriately describe the iFuse Bedrock GraniteTM.
Comment: In response to our concerns that the iFuse Bedrock GraniteTM Implant System may be appropriately described by C1889, the applicant and a few commenters noted that C1889 is not, and has not been, a device pass-through category code, and that it is not included in the complete list of device pass-through category codes.
Response: We appreciate the commenters' input. We agree that C1889 is not a device pass-through category code, and therefore, agree that it is not relevant to our determination at § 419.66(c)(1).
After consideration of the public comments received and our review of the application, we agree there is no existing category or category previously in effect that appropriately describes the iFuse Bedrock GraniteTM Implant System. Based on this information, we have determined that the iFuse Bedrock GraniteTM Implant System meets the eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category at § 419.66(c)(2) provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The iFuse Bedrock GraniteTM Implant System has a Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in the discussion of the newness criterion) and therefore is not evaluated for substantial clinical improvement.
We invited public comment on whether the iFuse Bedrock GraniteTM Implant System meets the device category criterion at § 419.66(c)(2)(ii).
We did not receive any public comment regarding whether the iFuse Bedrock GraniteTM Implant System meets the device category criterion at § 419.66(c)(2)(ii). The iFuse Bedrock GraniteTM Implant System has a Breakthrough Device designation effective November 23, 2021, and marketing authorization from FDA effective May 26, 2022, for the indication covered by the Breakthrough Device designation. Based on our review of the application, we have determined that the iFuse Bedrock GraniteTM Implant System meets the device category criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.636(c)(2)(i).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the iFuse Bedrock GraniteTM Implant System would be reported with HCPCS codes shown in Table 118.
( printed page 94152)According to the applicant, the iFuse Bedrock GraniteTM Implant System is only used when both a SIJ fusion procedure and a lumbar fusion procedure are performed in the same operative session. The applicant stated that the iFuse Bedrock GraniteTM Implant System is not utilized when only a SIJ fusion procedure is performed (HCPCS code 27279) or when only a lumbar fusion procedure is performed (HCPCS code 22612, 22630, or 22633). Rather, per the applicant, the appropriate coding of the procedure where the iFuse Bedrock GraniteTM Implant System is used should include the HCPCS code for SIJ fusion (HCPCS code 27279) and a HCPCS code for lumbar fusion (HCPCS code 22612, 22630, or 22633). Per the applicant, the selection of the primary lumbar fusion HCPCS code (HCPCS code 22612, 22630, or 22633) is dependent on the procedure performed.
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant utilized the CY 2024 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5116, which had a CY 2024 payment rate of $17,756.28 at the time the application was received. The applicant stated the iFuse Bedrock GraniteTM Implant System device should be reported with the SIJ fusion procedure HCPCS code 27279 along with one of the three lumbar fusion procedures (HCPCS code 22612, 22630 or 22633). While the applicant utilized HCPCS code 22612 for the device offset amount for test two of the cost criterion, we stated that we believe that HCPCS code 27279 is the appropriate HCPCS code for the offset and subsequent calculation. Specifically, we understood that code 27279 is always reported when the iFuse Bedrock GraniteTM Implant System is used along with only one of the three specified lumbar fusion codes and that the SIJ fusion procedure described by code 27279 is always performed when the iFuse Bedrock GraniteTM is used along with just one of three possible lumbar procedures, depending on the specific surgical approach used. Therefore, we stated that we believe that neither HCPCS codes 22612, 22630, nor 22633 is appropriate to use for the cost criterion calculation. As such, we used HCPCS code 27279, the code that should always be reported with the iFuse Bedrock GraniteTM Implant System, for our calculations. HCPCS code 27279 in APC 5116 had a CY 2024 device offset amount of $12,264.26 at the time the application was received. According to the applicant, the cost of the iFuse Bedrock GraniteTM Implant System is $11,689.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $11,689.00 for the iFuse Bedrock GraniteTM Implant System is 65.83 percent of the applicable APC payment amount for the service related to the category of devices of $17,756.28 (($11,689.00/$17,756.28) × 100 = 65.83 percent). Therefore, we stated that we believe the iFuse Bedrock GraniteTM Implant System meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $11,689.00 for the iFuse Bedrock GraniteTM Implant System is 95.31 percent of the cost of the device-related portion of the APC payment amount for the related service of $12,264.26 (($11,689.00/$12,264.26) × 100 = 95.31 percent). Therefore, we stated that we believe the iFuse Bedrock GraniteTM Implant System does not meet the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $11,689.00 for the iFuse Bedrock GraniteTM Implant System and the portion of the APC payment amount for the device of $12,264.26 is negative 3.24 percent of the APC payment amount for the related service of $17,756.28 ((($11,689.00−$12,264.26)/$17,756.28) × 100 =−3.24 percent). Therefore, we stated that we believe the iFuse Bedrock ( printed page 94153) GraniteTM Implant System does not meet the third cost significance requirement.
We invited public comment on whether the iFuse Bedrock GraniteTM Implant System meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: In response to our proposal to utilize HCPCS code 27279 for evaluation of cost criteria for the iFuse Bedrock GraniteTM Implant System, the applicant and another commenter asserted that the most appropriate HCPCS code to evaluate the criterion for the iFuse Bedrock GraniteTM Implant System is HCPCS code 22612. The applicant and another commenter provided several reasons: (1) The intended indication and use of the nominated device in the hospital outpatient and ASC setting will be in support of the lumbar spinal construct for lumbar fusion cases; (2) The typical patient for the nominated device has severe lumbar spinal pathology, such as scoliosis or kyphosis of the lumbar spine, as the primary diagnosis, whereas sacroiliac joint disease or dysfunction is not the primary diagnosis or procedure indication for these patients; and (3) As evidence of the consensus on this type of pelvic fixation to support lumbar patients, numerous commercial payers across the U.S. agree and have published medical coverage policies stating that SI joint fusion at the bottom of a long construct is covered for treatment of lumbar disease or dysfunction.[37] The applicant claimed that in the outpatient setting, the iFuse Bedrock GraniteTM Implant System would always be used in conjunction with one of three spinal fusion procedures and would always be considered as a secondary procedure. The applicant stated that of the four relevant HCPCS codes in its application (HCPCS codes 22612, 22630, 22633, and 27279), the threshold for HCPCS code 22612 is the most appropriate for evaluating the cost criterion for the iFuse Bedrock GraniteTM Implant System. The applicant provided the following reasons: (1) HCPCS code 22612 is the only lumbar spinal fusion procedure payable by Medicare in both hospital outpatient and ASC settings; (2) HCPCS code 22612 has a lower device offset, consistent with CMS' policies; and (3) HCPCS code 22612 is the primary code in spinal fusion procedures in which the iFuse Bedrock GraniteTM will always be used as an adjunctive technology, when connected to compatible posterior spinal rods, to improve the potential outcome of the lumbar fusion case. Finally, the applicant argued that HCPCS code 22612 would also be appropriate from a primary assignment rank perspective, stating that since the nominated device will have multiple J1 HCPCS codes, it would be appropriate to determine which HCPCS code has the highest primary assignment ranking. The applicant referenced Addendum J of the CY 2024 final rule and the CY 2025 proposed rule stating that HCPCS code 22612 has a higher ranking for primary assignment purposes than HCPCS code 27279. The applicant claimed that it would be consistent with CMS' policy on assignment of primary procedures to select the cost threshold for HCPCS code 22612, as opposed to HCPCS code 27279, to assess the cost criterion for the iFuse Bedrock GraniteTM Implant System.
Response: We thank the commenters for the additional information. Based on the updated information provided in the comments, we agree HCPCS code 22612 is an appropriate procedure code for the evaluation of the cost criterion for the iFuse Bedrock GraniteTM Implant System. As such, we have updated our calculations accordingly for the second and third cost significance requirements, at § 419.66(d)(2) and (3), respectively as below.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The average reasonable cost of $11,689 for the iFuse Bedrock GraniteTM Implant System is 136.75 percent of the cost of the device-related portion of the APC payment amount for the related service of $8,547.88 (($11,689.00/$8,547.88) × 100 = 136.75 percent). Therefore, we agree that the iFuse Bedrock GraniteTM Implant System meets the second cost significant requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $11,689.00 for the iFuse Bedrock GraniteTM Implant System and the portion of the APC payment amount for the device of $8,547.88 is 17.70 percent of the APC payment amount for the related service of $17,756.28 ((($11,689.00−$8,547.88)/$17,756.28) × 100 = 17.70 percent). Therefore, we agree that the iFuse Bedrock GraniteTM Implant System meets the third cost significance requirement.
Comment: The applicant and another commenter stated that they believe it is appropriate for CMS to use the lowest APC payment rate applicable for use with the nominated device regarding the iFuse Bedrock GraniteTM Implant System, stating that this is consistent with increasing probability that the device will pass the cost significance test. The applicant and another commenter asserted that CMS should likewise use the offset amount that gives the nominated device the best opportunity to pass the test, arguing that HCPCS code 22612 is the most appropriate code for assessing the cost threshold in this context.
Response: We thank the commenters for their input. As we explained in the CY 2005 OPPS/ASC final rule with comment period (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion. We note that in both the original evaluation of cost in the proposed rule as well as in the re-evaluation in this final rule, we followed this procedure by selecting the lowest, and in this case only, APC payment rate applicable for APC 5116 of $17,756. While we note that since 2017, we calculate the device offset amount at the HCPCS/CPT® code level instead of the APC level (89 FR 79657), we disagree that we are bound by the same policy to select the HCPCS code with the lowest offset amount. We believe that doing so would potentially conflict with our stated policy of selecting the lowest APC payment rate, which is intended to increase the probability the device will pass the cost significance test. We do, however, agree to use HCPCS code 22612 and its device offset amount for the evaluation of the cost criterion for the iFuse Bedrock GraniteTM Implant System, based on the updated information we received in the comments.
Comment: The applicant requested that the iFuse Bedrock GraniteTM Implant System be approved for the transitional pass-through status and the ( printed page 94154) device offset amount should be set to $0. The applicant asserted that the costs of the iFuse Bedrock GraniteTM Implant System are not included as part of the device-related portion of the APC 5116 payment supporting hospitals performing a primary lumbar fusion procedure. The applicant stated that hospital outpatient departments and ASCs do not typically support combination lumbar fusion with SI joint fusion procedures performed in the same operative session, and claims data reveals an extremely low volume of these codes being reported together each year in the HOPD setting relative to other code pairings. In addition, the applicant stated that the iFuse Bedrock GraniteTM Implant System cannot be used as a stand-alone device; rather, it will only be used in conjunction with one of three lumbar spinal fusion procedures. The applicant referred to the CY 2024 and CY 2025 Addendum J files and stated that all three HCPCS codes describing lumbar fusion which are eligible for payment in the hospital outpatient setting (HCPCS 22612, 22630, and 22633) are all ranked higher for primary assignment purposes than HCPCS code 27279. The applicant therefore argued that the device costs of the iFuse Bedrock GraniteTM Implant System have not been appropriately included in the device offset of any of the fusion procedures. The applicant further stated that, since the costs of the iFuse Bedrock GraniteTM Implant System are purely additive for hospitals currently performing lumbar spinal fusion procedures (HCPCS codes 22612, 22630, or 22633) mapping to Comprehensive APC 5116, the applicant requested that CMS set the device offset to $0 when granting transitional pass-through status for the iFuse Bedrock GraniteTM Implant System for CY 2025.
Response: After further review, we are persuaded by commenters that the iFuse Bedrock GraniteTM Implant System cannot be used as a stand-alone device and that it is only used in conjunction with the existing device costs for the lumbar spinal fusion procedures. Therefore, we agree that the device is not included in the existing APC payment amount for these procedures and a device offset amount under 42 CFR 419.66(h) would not be appropriate at this time. For CY 2025, for the procedures we described that are performed with this new transitional pass-through device category, we are accepting the commenter's recommendation to apply a $0 offset for the iFuse Bedrock GraniteTM Implant System. We refer the reader to Addendum B of this CY 2025 OPPS/ASC final rule with comment period for APC payment rates.
After consideration of the public comments we received and our findings from the first, second, and third cost significance tests, we agree that the iFuse Bedrock GraniteTM meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments we received and our review of the device pass-through application, we have determined that the iFuse Bedrock GraniteTM Implant System meets the criteria for device pass-through status described at § 419.66. We are finalizing approval for device pass-through payment status for the iFuse Bedrock GraniteTM Implant System effective January 1, 2025, under the alternative pathway for devices that have an FDA Breakthrough Device Designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(f) Paradise® Ultrasound Renal Denervation (RDN) System
ReCor Medical, Inc. submitted an application for a new device category for transitional pass-through payment status for the Paradise® Ultrasound RDN System for CY 2025. Per the applicant, the Paradise® Ultrasound RDN System is a catheter-based system that delivers ultrasound energy in the location of sympathetic nerves surrounding the renal arteries. The applicant explained that the Paradise® Ultrasound RDN System is indicated to reduce blood pressure as an adjunctive treatment in patients with hypertension in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure. According to the applicant, the Paradise® Catheter, when used with the other Paradise® Ultrasound RDN System components, provides complete 360-degree energy delivery and targeted ablation depth with each energy emission with the goal of disrupting the nerves and consequently achieving a reduction in systemic arterial blood pressure. The applicant asserted that the Paradise® Catheter protects the artery walls using a cooling system during periods of ultrasound energy emission (also called sonications).
Per the applicant's instructions for use, the Paradise® Ultrasound RDN System includes the following components: (1) Paradise® Generator, which circulates coolant fluid and electrical energy to the Paradise® Catheter via the Paradise® Cable and Paradise® Cartridge; (2) Paradise® Catheter, which connects with the Paradise® Generator and has a distal balloon (available in six different diameters that correspond to varying artery diameter ranges) that is pressurized using coolant fluid; (3) Paradise® Cartridge, which controls the fluid flow into and out of the Paradise® Catheter when used in conjunction with the Paradise® Generator; (4) Paradise® Connection Cable, which transfers electrical energy from the Paradise® Generator to the Paradise® Catheter; (5) Paradise® Remote, included with the Paradise® Generator for optional use; and (6) Paradise® Cart, an optional wheeled cart to which the Paradise® Generator can be mounted to stabilize the Paradise® Generator during a procedure and to transport the Paradise® Generator from one location to another. Per the applicant, additional items required for the procedure include: (1) a bag of coolant fluid for inflation and cooling of balloon; (2) a 0.014 inch guidewire to track the Paradise® Catheter into position for delivery of ultrasound energy; (3) a push/pull style hemostasis valve; (4) a 6 French (Fr) or larger guide sheath; and (5) a 7 Fr or larger guide catheter. According to the applicant, the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable are single-patient, one-time use components of the system. According to the applicant, key steps for operating the Paradise® Ultrasound RDN System include: (1) gaining access to the femoral artery using standard interventional techniques and placing a 7 Fr (or larger) guide catheter; (2) advancing the 7 Fr guide catheter into the left or right renal artery under fluoroscopic guidance; (3) performing an angiogram to verify the patency of the left or right renal artery; (4) measuring the distal, mid, and proximal artery diameters and selecting the appropriate Paradise® Catheter balloon size; (5) preparing and attaching the Paradise® Cartridge, Paradise® Connection Cable, and sterile water supply, and connecting the Paradise® Cartridge extension tubing to the Paradise® Catheter; (6) preparing and flushing the Paradise® Catheter; removing access devices from the lumen of the guide catheter and inserting a 0.014 inch guidewire; (7) verifying the balloon on the Paradise® Catheter is deflated; (8) tracking the Paradise® Catheter over the guidewire and gently inserting the Paradise® Catheter into the push/pull style hemostasis valve and guide catheter; (9) advancing and positioning the Paradise® Catheter in desired locations within the renal arteries; (10) inflating the balloon via ( printed page 94155) the Paradise® Generator; (11) verifying the position of the balloon and catheter transducer via fluoroscopy and contrast injection; (12) performing denervation of the left and/or right renal artery by delivery of ultrasound energy; (13) verifying balloon deflation via fluoroscopy before moving to the next location; (14) withdrawing the Paradise® Catheter back into the guide catheter prior to moving the device into an alternate artery or accessory vessel, continuing to another position and exchanging the balloon catheter, as needed; (15) removing the Paradise® Catheter, ensuring that Paradise® Catheter balloon is in a deflated state prior to removal, by slowly withdrawing the Paradise® Catheter through the guide catheter, until completely withdrawn, and removing the guidewire and guide catheter; and (16) closing the wound per standard of practice.
Please refer to the online application posting for the Paradise® RDN System, available at https://mearis.cms.gov/public/publications/device-ptp/DEP231128137E1, for additional detail describing the device and the disease treated by the device.
Comment: Many commenters expressed their support for the approval of the Paradise® Ultrasound RDN System for transitional pass-through payment. In addition, many commenters shared their positive experiences with the Paradise® Ultrasound RDN System.
Response: We appreciate the commenters' input and support for the Paradise® Ultrasound RDN System application for transitional pass-through payment. We have taken these comments into consideration in our final determination for pass-through status for the Paradise® Ultrasound RDN System.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the Paradise® Ultrasound RDN System received FDA Breakthrough Device designation effective December 4, 2020, as a device with the indicated use to reduce blood pressure in adult (≥22 years of age) patients with uncontrolled hypertension, who may be inadequately responsive to, or who are intolerant to anti-hypertensive medications. FDA approved the premarket approval application (PMA) for the Paradise® Ultrasound RDN System on November 7, 2023, for the indicated use to reduce blood pressure as an adjunctive treatment in hypertension patients in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure. We noted that while the indication for the FDA Breakthrough Device designation and the indication for the FDA premarket approval vary slightly, we believed that FDA premarket approval indication is the one covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for the Paradise® Ultrasound RDN System on November 28, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the Paradise® Ultrasound RDN System meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the Paradise® Ultrasound RDN System received Breakthrough Device designation and later FDA approval on November 7, 2023, and CMS received the application for a new device category for transitional pass-through payment status on November 28, 2023, which is within 3 years of FDA approval. A few commenters stated the Paradise® Ultrasound RDN System meets the newness criterion, with one commenter specifically noting that the Paradise® Ultrasound RDN System meets the newness criterion based on its FDA Breakthrough Device designation.
Response: We appreciate the commenters' input. We agree with the applicant and commenters that the Paradise® Ultrasound RDN System meets the device eligibility requirements of § 419.66(b)(1); however, while we agree that the Paradise® Ultrasound RDN System meets the eligibility criterion specified at § 419.66(b)(1), we note that a device's uniqueness or FDA designation does not alone qualify a device for transitional pass-through payment. Specifically, when we adopted the alternative pathway for device pass-through payments under the OPPS, we stated that applications for devices that have received FDA marketing authorization and are part of the FDA Breakthrough Devices Program would not be evaluated in terms of the current substantial clinical improvement criterion at § 419.66(c)(2) for purposes of determining device pass-through payment status, but would continue to need to meet the other requirements for pass-through payment status in our regulations at § 419.66(c)(1) (84 FR 61295).
The Paradise® Ultrasound RDN System received Breakthrough Device designation on December 4, 2020. We received the application for a new device category for transitional pass-through payment status for the Paradise® Ultrasound RDN System on November 28, 2023, which is within 3 years of November 7, 2023, the date of FDA PMA. Based on our review of the application, we have determined that the Paradise® Ultrasound RDN System meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, be used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not explicitly state whether the Paradise® Ultrasound RDN System is integral to the service furnished. With respect to whether the Paradise® Ultrasound RDN System is used for one patient only, the applicant asserted that the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable are single-patient use. However, the Paradise® Generator, Paradise® Remote, and Paradise® Cart are reusable and are capital equipment. While the applicant did not explicitly state whether the Paradise® Ultrasound RDN System comes into contact with human tissue, the applicant provided that the Paradise® Catheter is placed within the renal artery. According to the applicant, the Paradise® Ultrasound RDN System is an implantable device. We noted that the Paradise® Generator, Paradise® Remote, and Paradise® Cart are reusable, do not come in contact with the patient's tissue, are not surgically implanted or inserted, or applied in or on a wound or other skin lesion, as required by § 419.66(b)(3).
We invited public comment on whether the Paradise® Ultrasound RDN System meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant submitted a comment clarifying that the Paradise® Ultrasound RDN System is an integral part of the service furnished and meets the eligibility criterion at § 419.66(b)(3) because the Paradise® Ultrasound RDN System performs the ablations via ultrasound which produce the therapeutic effect to reduce blood pressure. The applicant also clarified that the Paradise® Ultrasound RDN System comes into contact with human tissue and meets the eligibility criterion at § 419.66(b)(3) because the Paradise® Ultrasound RDN System's ablation catheter comes in contact with human tissue and is surgically inserted into the body and removed at the end of the procedure. Finally, the applicant reiterated that the Paradise® Connector ( printed page 94156) Cable, Paradise® Cartridge, and Paradise® Catheter components of the Paradise® Ultrasound RDN System are single-use only and the Paradise® Catheter is surgically inserted into the body and removed at the end of the procedure.
Response: We appreciate the applicant's input. We agree with the applicant that the Paradise® Ultrasound RDN System is an integral part of the service furnished, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion. In addition, we agree that the Paradise® Connector Cable, Paradise® Cartridge, and Paradise® Catheter components of the Paradise® Ultrasound RDN System are used for one patient only and come in contact with human tissue. We continue our discussion and final determination of the criterion at § 419.66(b)(3) in our response below.
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). While the applicant did not explicitly state whether the Paradise® Ultrasound RDN System, or select components, is equipment, an instrument, apparatus, implement, or item for which depreciation and financing expenses are recovered, per the applicant, the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable are single-use only. The applicant further explained that the Paradise® Generator, Paradise® Cart, and Paradise® Remote are capital equipment; as such, they are excluded from device pass-through payment eligibility under § 419.66(b)(4). The applicant did not explicitly state whether the Paradise® Ultrasound RDN System is a supply or material furnished incident to a service.
The applicant requested pass-through payment for the Paradise® Ultrasound RDN System, but we questioned whether only the Paradise® Catheter component of the Paradise® Ultrasound RDN System, as opposed to the whole system, is eligible for pass-through payments under § 419.66(b)(3) or at § 419.66(b)(4). We stated that we do not believe the Paradise® Generator, Paradise® Cable, Paradise® Cartridge, Paradise® Connection Cable, Paradise® Remote, or Paradise® Cart meet the eligibility requirements under § 419.66(b)(3) or at § 419.66(b)(4), and, as such, are not eligible for pass-through payments.
We invited public comment on whether the Paradise® Ultrasound RDN System meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant submitted a comment clarifying that the Paradise® Cartridge and Paradise® Connector Cable meet the eligibility criterion at § 419.66(b)(4) because they are critical parts of a single piece of equipment, they are not sold without the Paradise® Catheter, and because these two components are not separable from the Paradise® Catheter component. With regard to our question as to whether only the Paradise® Catheter component of the Paradise® Ultrasound RDN System is eligible for pass-through payments under § 419.66(b)(3) or at § 419.66(b)(4), the applicant stated that the Paradise® Cartridge and Paradise® Connector Cable are eligible for pass-through payment because the procedure cannot be performed without all three components, i.e., the Paradise® Catheter, the Paradise® Cartridge, and the Paradise® Connector Cable. The applicant further stated that while the Paradise® Connector Cable and Paradise® Cartridge are not inserted into the body, the Paradise® Catheter that is inserted into the body will not function without them. Therefore, the applicant asserted that the Paradise® Connector Cable and Paradise® Cartridge should also be eligible for transitional pass-through status along with the Paradise® Catheter. The applicant also confirmed that the Paradise® Generator, Paradise® Remote, and Paradise® Cart components of the Paradise® Ultrasound RDN System are capital equipment and are not eligible for pass-through payment. With regard to our question about whether the Paradise® Ultrasound RDN System is a supply or material furnished incident to a service, the applicant submitted a comment clarifying that the Paradise® Ultrasound RDN System is not a supply or material furnished incident to a service because the purpose of the procedure is to use the Paradise® Ultrasound RDN System to treat the patient with renal denervation; therefore, it is integral and essential and it is not a supply that is used incident to or for the purpose of doing another procedure.
Response: We thank the applicant for the clarification and agree with the applicant that the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable components of the Paradise® Ultrasound RDN System meet the eligibility requirements under § 419.66(b)(3) and (4) because the Paradise® Connector Cable and Paradise® Cartridge are required parts of a single piece of equipment, they are not sold without the Paradise® Catheter, they are single-use and they are not separable. We also agree with the applicant that the Paradise® Generator, Paradise® Remote, or Paradise® Cart are capital equipment and do not meet the eligibility requirements under § 419.66(b)(3) or (4). Finally, we agree with the applicant that the Paradise® Ultrasound RDN System is not a material or supply furnished incident to a service. After consideration of the public comments we received and our review of the application, we have determined that the applicable components of the Paradise® Ultrasound RDN System meet the eligibility criteria at § 419.66(b)(3) and (4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. Per the applicant, the Paradise® Ultrasound RDN System is a catheter-based system that delivers ultrasound energy in the location of sympathetic nerves surrounding the renal arteries. According to the applicant, no previous or existing device categories for pass-through payment have encompassed the Paradise® Ultrasound RDN System. Per the applicant, device categories C1753 (Catheter, intravascular ultrasound) and C1888 (Catheter, ablation, noncardiac, endovascular (implantable)) do not appropriately describe the Paradise® Ultrasound RDN System. The applicant asserted that C1753 does not appropriately describe the Paradise® Ultrasound RDN System because the Paradise® Ultrasound RDN System is used to treat a disease and denervates renal nerves, whereas C1753 was created to describe ultrasound catheter devices that are not used to treat a disease and are used for imaging of the vessel. According to the applicant, C1888 does not appropriately describe the Paradise® Ultrasound RDN System because the Paradise® Ultrasound RDN System is intended to denervate renal nerves by using ultrasound energy that does not otherwise affect the blood vessel tissue, whereas C1888 was ( printed page 94157) created to describe devices that use radiofrequency or laser technologies to occlude or obliterate blood vessels.[38]
We did not identify an existing pass-through payment category that describes the Paradise® Ultrasound RDN System. We invited public comment on whether the Paradise® Ultrasound RDN System meets the device category criterion at § 419.66(c)(1).
Comment: The applicant and a few other commenters agreed with CMS' assessment that there are no existing pass-through payment categories that describe the Paradise® Ultrasound RDN System.
Response: We appreciate the commenters' input. After consideration of the public comments we received and our review of the application, we continue to believe that there is no existing category or category previously in effect that appropriately describes the Paradise® Ultrasound RDN System. Therefore, we have determined that the Paradise® Ultrasound RDN System meets the device category eligibility criterion at § 419.66(c)(1).
We noted that the applicant indicated the Paradise® Ultrasound RDN System is the only device authorized by FDA with an indication for renal denervation using ultrasound energy to achieve reductions in blood pressure. However, we noted that the Symplicity SpyralTM Catheter (Symplicity SpyralTM RDN System) device, for which we also received an application for transitional pass-through payments for CY 2025 as discussed in the proposed rule (89 FR 59312 through 59315) and this final rule with comment, is also authorized by FDA with an indication for renal denervation using a radiofrequency modality to achieve reductions in blood pressure. Accordingly, we noted that while the Paradise® Ultrasound RDN System device may have a different modality ( i.e., ultrasound compared to radiofrequency) than that of the Symplicity SpyralTM Catheter device, the Paradise® Ultrasound RDN System device may have a similar mechanism of action to that of the Symplicity SpyralTM Catheter device. We questioned whether the device descriptions provided in the respective applications support establishing two modality specific pass-through payment device categories or a single device category that would encompass both RDN device modalities. We address this question in section (i) in detail immediately following the full discussion of all other applicable eligibility criteria for both the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System applications.
For the summary of public comments received and our final decision on the question of whether the device descriptions provided in the respective applications support establishing two modality-specific pass-through payment device categories or a single device category that would encompass both RDN device modalities, please see section (i) immediately following the Symplicity SpyralTM RDN System application determination.
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. As previously stated, the Paradise® Ultrasound RDN System has Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail in the discussion of the newness criterion) and therefore is not evaluated for substantial clinical improvement.
We invited public comment on whether the Paradise® Ultrasound RDN System meets the device category criterion at § 419.66(c)(2)(ii).
Comment: A few commenters stated that the Paradise® Ultrasound RDN System meets the eligibility criterion at § 419.66(c)(2)(ii), with one commenter asserting that the Paradise® Ultrasound RDN System meets the clinical improvement criterion based on its FDA Breakthrough Device designation.
Response: We appreciate the commenter's input. We agree with the commenters that the Paradise® Ultrasound RDN System has a Breakthrough Device designation effective December 4, 2020, and marketing authorization from FDA effective November 7, 2023, for the indication covered by the Breakthrough Device designation and therefore, meets the criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application and consideration of the public comments we received, we have determined that the Paradise® Ultrasound RDN System meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the Paradise® Ultrasound RDN System would be reported with HCPCS codes shown in Table 119.
( printed page 94158)To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2023 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5192, which had a CY 2023 payment rate of $5,215.40 at the time the application was received. We used the CY 2023 APC level device offset amount of $1,491.08 for APC 5192, since HCPCS codes 0338T and 0339T were not included in Addendum P to the CY 2023 OPPS/ASC final rule with comment period and no CY 2023 HCPCS/CPT code level device offset amount was available at the time the application was received.[39] According to the applicant, the operating cost [40] of the Paradise® Ultrasound RDN System is $23,000.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $23,000.00 for the Paradise® Ultrasound RDN System is 441.00 percent of the applicable APC payment amount for the service related to the category of devices of $5,215.40 (($23,000.00/$5,215.40) × 100 = 441.00 percent). Therefore, we stated that we believe the Paradise® Ultrasound RDN System meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $23,000.00 for the Paradise® Ultrasound RDN System is 1,542.51 percent of the cost of the device-related portion of the APC payment amount for the related service of $1,491.08 (($23,000.00/$1,491.08) × 100 = 1,542.51 percent). Therefore, we stated that we believe the Paradise® Ultrasound RDN System meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $23,000.00 for the Paradise® Ultrasound ( printed page 94159) RDN System and the portion of the APC payment amount for the device of $1,491.08 is 412.41 percent of the APC payment amount for the related service of $5,215.40 ((($23,000.00−$1,491.08)/$5,215.40) × 100 = 412.41 percent). Therefore, we stated that we believe the Paradise® Ultrasound RDN System meets the third cost significance requirement.
We invited public comment on whether the Paradise® Ultrasound RDN System meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to cost significance criteria, the applicant and a few other commenters reiterated that the Paradise® Ultrasound RDN System meets all three of the cost significance criteria. The applicant further clarified that the Paradise® Ultrasound RDN System meets the cost criteria regardless of whether the Paradise® Cartridge and Paradise® Connection Cable are deemed eligible for pass-through or excluded from the calculation. The applicant stated that the Paradise® Cartridge and Paradise® Connection Cable should be included when evaluating the cost criteria because they are both single-use items that are required for the Paradise® Ultrasound RDN System to function as intended and are essential to complete each patient treatment.
Response: We appreciate the applicant's and commenters' input. We agree with the applicant that the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable are single-use components that are required for the Paradise® Ultrasound RDN System to function as intended and are essential to complete each patient treatment and should be included when evaluating the cost criteria. After consideration of the public comments we received and our findings from the first, second, and third cost significance tests, we agree that the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable components of the Paradise® Ultrasound RDN System meet the cost significance criteria specified at § 419.66(d).
After consideration of the public comments we received and our review of the device pass-through application, we have determined that the Paradise® Catheter, Paradise® Cartridge, and Paradise® Connection Cable components of the Paradise® Ultrasound RDN System meet the requirements for device pass-through status described at § 419.66. Therefore, effective beginning January 1, 2025, we are finalizing approval for device pass-through payment status for the applicable components of the Paradise® Ultrasound RDN System under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(g) Precision GI
Limaca Medical submitted an application for a new device category for transitional pass-through payment status for Precision GI for CY 2025. According to the applicant, Precision GI is a motorized, battery operated, single-use, fully disposable endoscopic ultrasound-guided (EUS) fine needle biopsy device used to obtain biopsies of tissue for definitive diagnosis of pancreatic cancer and other life-threatening gastrointestinal (GI) abnormalities. Per the applicant, Precision GI is untethered and battery operated with an internally powered and controlled motor, featuring a long flexible shaft transferring the proximal force of the motor through the inserted endoscope to the needle circumferential cutting tip. The device is controlled by a physician, who inserts the device into the patient's GI tract via the ultrasound endoscope. Upon reaching the designated biopsy site, the physician operates the device's motorized mechanism that automatically rotates the needle (which is included in the device's package) to cut and extract tissue. The biopsy site is accessed through the instrument channel of an ultrasound imaging endoscope that detects the device's echogenic needle tip.
Please refer to the online application posting for Precision GI, available at https://mearis.cms.gov/public/publications/device-ptp/DEP23113023REE, for additional detail describing the device and the disease treated by the device.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), Precision GI received FDA Breakthrough Device designation effective March 24, 2022, as a device used with an ultrasound endoscope for fine needle biopsy of submucosal lesions, mediastinal masses, lymph nodes, and intraperitoneal masses within or adjacent to the GI tract. FDA granted the applicant 510(k) clearance for Precision GI on August 28, 2023, for the indication covered by the Breakthrough Device designation. We received the application for a new device category for transitional pass-through payment status for Precision GI on November 30, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether Precision GI meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant and one commenter agreed with CMS' assessment and the applicant noted that CMS raised no concerns about Precision-GI meeting the criterion at § 419.66(b)(1).
Response: We appreciate the applicant's and the commenter's input. We received the application for a new device category for transitional pass-through payment status for Precision GI on November 30, 2023, which is within 3 years of August 28, 2023, the date of FDA 510(k) clearance. Based on our review of the application and public comments received, we have determined that Precision GI meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not indicate whether Precision GI is an integral part of the service furnished. The applicant stated that the device is intended for single use. While the applicant did not explicitly state whether Precision GI comes in contact with human tissue and is surgically inserted or implanted, the applicant noted that Precision GI is used to sample targeted submucosal lesions, mediastinal masses, lymph nodes, and intraperitoneal masses within or adjacent to the GI tract and this is achieved by a physician who inserts Precision GI into the patient's GI tract using an ultrasound endoscope. However, we noted that, while the needle (which is a component of the device and is included in the device's package) does come into contact with human tissue and is surgically inserted, the motorized mechanism of the Precision GI device itself may not come in contact with human tissue and may not be surgically implanted or inserted (either permanently or temporarily), or applied in or on a wound or other skin lesion, as required at § 419.66(b)(3).
We invited public comment on whether Precision GI meets the eligibility criterion at § 419.66(b)(3).
Comment: In response to our concern about whether Precision GI is integral to the service furnished and meets the eligibility criterion at § 419.66(b)(3), the ( printed page 94160) applicant clarified that Precision GI is an integral part of the service furnished because the Precision GI device is an endoscopic ultrasound-guided fine needle biopsy (EUS-FNB) medical device and is the essential device that physicians must use to enable the procedures for the services the applicant identified. The applicant clarified that physicians use Precision GI for procedures that are reported using the following HCPCS codes: 43238 (Esophagogastroduodenoscopy, flexible, transoral; with transendoscopic ultrasound-guided intramural or transmural fine needle aspiration/biopsy(s), (includes endoscopic ultrasound examination limited to the esophagus, stomach or duodenum, and adjacent structures), and 43242 (Esophagogastroduodenoscopy, flexible, transoral with transendoscopic ultrasound-guided intramural or transmural fine needle aspiration/biopsy(s), (includes endoscopic ultrasound examination limited to the esophagus stomach and either the duodenum or a surgically altered stomach where the jejunum is examined distal to the anastomosis)) and Precision GI is clearly integral to performing both of these services, given that these procedures are intended to extract suspected cancerous tissue and that Precision GI is the essential device used to obtain and remove the tissue. A commenter stated that based on the Precision GI application, they believe Precision GI would be integral to the procedures with which it is associated. The applicant reiterated that Precision GI is a single-use device and clarified that it is used for one patient only.
Response: We appreciate the applicant's and the commenter's input. Based on the information provided in the comments we agree with the applicant and commenter that Precision GI is an essential device that physicians must use to enable the procedures for the services the applicant identified and, as such, Precision GI is an integral part of the service furnished. After consideration of the public comments received and our review of the application, we have determined that Precision GI is integral to the service furnished and is used for one patient only. We have taken this information into consideration in making our final determination of whether Precision GI meets the requirements of § 419.66(b)(3).
Comment: In response to our concerns about whether Precision GI comes in contact with human tissue and meets the eligibility criterion at § 419.66(b)(3), the applicant and a commenter submitted comments clarifying that Precision GI comes in contact with human tissue when the device enters the patient and extracts suspected tumor tissue from the body. The applicant and commenter explained that after the Precision GI device is advanced through the mouth via the working channel of the upper GI endoscope to the section of the gastric wall located closest to the mass or tumor as identified by endoscopic ultrasound, the rotating needle component of Precision GI goes through the gastric wall to acquire tissue from the tumor. The applicant clarified that Precision GI operates as a single unit, which includes the needle and emphasized that Precision GI is not provided in separate components, does not require assembly, and does not require any additional supplies or materials to operate. As such, the applicant and the commenter asserted that Precision GI comes into contact with human tissue as required.
In response to our concerns about whether Precision GI is surgically inserted or implanted the applicant and another commenter clarified that Precision GI is surgically inserted when Precision GI is inserted through a natural orifice (the mouth) to access the biopsy site, and therefore it meets the eligibility criterion at § 419.66(b)(3). Specifically, the applicant clarified that the surgical path to the tumor is created by the endoscope which is passed through the mouth (natural orifice) where Precision GI is then used to access the site and to extract tissue in or adjacent to the GI tract, meeting the requirements of § 419.66(b)(3).
Response: We appreciate the applicant's and the commenter's input. Based on the clarifying information provided in the comments, we agree with the applicant that Precision GI comes in contact with human tissue and is surgically implanted or inserted as required by § 419.66(b)(3). Specifically, we relied upon the clarification that Precision GI functions collectively as a single unit with no separate components and the device enters the patient via an endoscope to extract human tissue from the body, and as such, we believe that Precision GI comes into contact with human tissue. In addition, we agree with the applicant and commenter that Precision GI is surgically inserted when it is advanced through a natural orifice ( i.e., the mouth) via an endoscope to create a surgical path to the tumor. After consideration of the public comments received and our review of the application, we have determined that Precision GI comes in contact with human tissue and is surgically inserted in accordance with the eligibility criterion at § 419.66(b)(3).
We agree with the applicant and commenter that Precision GI is an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion. After consideration of the public comments received and our review of the application, we have determined that Precision GI meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant indicated that Precision GI is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets; the applicant noted that Precision GI is a motorized, single-use, fully disposable EUS fine needle biopsy device that functions with no related capital component. The applicant stated that Precision GI is not a material or supply furnished incident to a service. However, based on the applicant's description of Precision GI as a biopsy device, we questioned whether Precision GI may be considered a supply or material furnished incident to a service and excluded from device pass-through payment eligibility under § 419.66(b)(4).
Specifically, in the CY 2001 OPPS interim final rule with comment period (65 FR 67804 through 67805), we explained how we interpreted § 419.43(e)(4)(iv). We stated that we consider a device to be surgically implanted or inserted if it is introduced into the human body through a surgically created incision. We also stated that we do not consider an item used to cut or otherwise create a surgical opening to be a device that is surgically implanted or inserted. We consider items used to create incisions, such as scalpels, electrocautery units, biopsy apparatuses, or other commonly used operating room instruments, to be supplies or capital equipment not eligible for transitional pass-through payments. We stated that we believe the function of these items is different and distinct from that of devices that are ( printed page 94161) used for surgical implantation or insertion. Finally, we stated that, generally, we would expect that surgical implantation or insertion of a device occurs after the surgeon uses certain primary tools, supplies, or instruments to create the surgical path or site for implanting the device. Further, in the CY 2006 OPPS final rule with comment period (70 FR 68629 through 68630), we adopted as final our interpretation that the surgical insertion or implantation criterion can be met by devices that are surgically inserted or implanted via a natural or surgically created orifice, as well as those devices that are inserted or implanted via a surgically created incision. We reiterated that we maintain all of the other criteria in § 419.66 of the regulations, namely, that we do not consider an item used to cut or otherwise create a surgical opening to be a device that is surgically implanted or inserted. We reiterated this interpretation in the CY 2024 OPPS final rule (88 FR 81543, 81743).
We noted that Precision GI, is inserted into the patient's GI tract via the ultrasound endoscope to reach the designated biopsy site where the device's motorized mechanism is then used for cutting and extraction of tissue endoscopically from within or adjacent to the patient's GI tract. However, we questioned whether Precision GI, which is described as a biopsy device, may be considered a supply or material furnished incident to a service consistent with our previous interpretation of § 419.43(e)(4)(iv) and therefore excluded from device pass-through payment eligibility under § 419.66(b)(4).
We welcomed additional evidence regarding whether Precision GI should be considered a material or supply incident to a service based on our previous interpretation of § 419.43(e)(4)(iv) as it has been applied to biopsy devices and we invited public comments on whether Precision GI meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant submitted a comment stating that there is agreement that Precision GI is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets.
Response: We appreciate the applicant's input. We agree with the applicant that Precision GI is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets. We have taken this information into consideration in making our final determination of whether Precision GI meets the requirements of § 419.66(b)(4).
Comment: The applicant submitted a comment clarifying that Precision GI is not a material or supply incident to a service and meets the eligibility criterion at § 419.66(b)(4) because Precision GI does not create incisions and is not used to create a surgical opening, rather Precision GI is used to capture and extract a diagnostically relevant portion of the tumor. The applicant stated that Precision GI's cutting function does not make the device a commodity supply and that Precision GI should not be placed in the same category as simple cutting or basic commodity tools ( e.g., such as scalpel, biopsy forceps, simple exchangeable needles, etc.).
Additionally, the applicant stated that Precision GI is not a generic biopsy device as defined by CMS for pass-through devices, referencing section 60.4.3, Chapter 4 of the Medicare Claims Processing Manual. In support, the applicant compared Precision GI to devices described by C1782 (Morcellator), which like Precision GI are also used for tissue removal. The applicant explained that because C1782 devices are used for laparoscopic tissue removal, per the CMS guidance in the Medicare Claims Processing Manual, they are not considered generic biopsy devices.[41] Thus, the applicant asserted its belief that, similar to C1782 devices, Precision GI is not a generic biopsy device because it is used for tissue removal. Furthermore, the applicant noted that C1782 would not appropriately describe Precision GI because C1782 is specific to laparoscopic removal of tissue, whereas Precision GI is endoscopically inserted to remove tissue within or adjacent to the GI tract.
The applicant asserted that CMS previously created a pass-through device category C1830 (Powered bone marrow biopsy needle), effective October 1, 2011, and that the device in this category functions similarly to Precision GI in complexity of operation and is not a biopsy needle as the applicant stated CMS defines this term. The applicant further commented that suggesting that Precision GI is a supply ignores the complexity of the device compared to commonly used endoscopic biopsy forceps and interchangeable commodity operating instruments.
One commenter stated that, per the application, Precision GI is a single, disposable, mechanized unit that would be integral to the procedures with which it is associated and for this reason, it should not be categorized with scalpels, needles, and other commonly used operating room instruments as a supply furnished incident to a service and should not be excluded from device pass-through eligibility under § 419.66(b)(4).
The applicant asserted that the FDA Breakthrough Designation implicitly recognizes the innovative nature of Precision GI and that it would be unlikely to have received this designation if it were deemed a supply. The applicant also asserted that Precision GI obtains tumor tissue in a superior method compared to today's non-motorized EUS-FNB devices, providing consistently higher quality core tumor tissue for the anatomic pathologist to more confidently diagnose.
Response: We appreciate the applicant's and commenter's input and the additional information. We agree with the applicant that Precision GI is not a supply or material furnished incident to a service because the device does not appear to be an item used to cut or otherwise create a surgical opening, rather it is inserted through a natural orifice ( i.e., the mouth) via an endoscope to create a surgical path to reach the designated biopsy site where the device's motorized mechanism is used for cutting and extraction of tissue endoscopically from within or adjacent to the patient's GI tract.
We also agree with the applicant that similar to devices described by C1782 and C1830, Precision GI appears to be distinct from biopsy devices as defined by section 60.4.3, Chapter 4 of the Medicare Claims Processing Manual.
In response to the applicant's comment that FDA Breakthrough Designation implicitly recognizes the innovative nature of Precision GI and that it would be unlikely to have received this designation if it were deemed a supply, we note that a device's FDA designation does not alone qualify a device for transitional device pass-through payment; the criteria FDA utilizes to grant medical device approvals differ from the criteria CMS has established to evaluate device eligibility for OPPS device pass-through payments. As such, CMS must evaluate all information submitted for each device pass-through application for each applicable criterion as it applies to the nominated device. In response to the ( printed page 94162) applicant's comment that Precision GI obtains tumor tissue in a superior method compared to today's non-motorized EUS-FNB devices, providing consistently higher quality core tumor tissue for the anatomic pathologist to more confidently diagnose, we note that § 419.66(b)(4) does not require that a device is superior to other available devices.
Based on the additional information provided in the comments, we agree with the applicant and commenter that Precision GI is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. After consideration of the public comments received and our review of the application, we have determined that Precision GI meets the eligibility criterion at § 419.66(b)(4).
Comment: With respect to our concerns about the eligibility criterion at § 419.66(b)(3), the applicant sought to distinguish Precision GI from the CytoCore device, which was denied pass-through status in CY 2024 (88 FR 81744). The applicant stated that Precision GI differs from CytoCore in two ways: unlike the CytoCore device, Precision GI does not have a separate needle component and Precision GI comes into contact with human tissue. Specifically, the applicant commented that the needle attached to the CytoCore is a separate needle, and not part of the CytoCore device and the needle is the only portion of the device that comes in contact with human tissue, whereas Precision GI's needle is an integral part of the Precision GI device that works in conjunction with Precision GI's long flexible shaft fully driven by its control system and motor, to transfer the proximal driving force of the motor to the highly specialized circumferential cutting and capturing tip to access the tumor tissue and perform the biopsy. The applicant further clarified that the Precision GI's needle is not only an integrated part of the device, but it is also a specifically and uniquely designed cannula with circumferential internal sharpening to enable superior tumor tissue capture when driven by Precision GI as a motor and microchip control program. The applicant stated that Precision GI enables the physician to accomplish motorized, automated, and synchronized axial advancement and rotational cutting and extraction of tissue at the target tissue site. Finally, the applicant noted that the needle used with Precision GI cannot be swapped out for other generic needles.
Response: We appreciate the applicant's input and agree that because Precision GI does not have separate components, functions collectively as a single device, and is surgically inserted when it is advanced through a natural orifice ( i.e., the mouth) via an endoscope, creating a surgical path to the tumor, the device comes into contact with human tissue and is surgically inserted as required by § 419.66(b)(3).
Comment: With regard to our discussion about the eligibility criterion specified at § 419.66(b)(4), a commenter stated that the Precision GI transitional pass-through payment application raises an important policy question about the definition of a biopsy apparatus. According to the commenter, items used to create incisions, such as biopsy apparatuses, or other commonly used operating room instruments, that CMS classified as supplies in in the CY 2006 OPPS final rule (70 FR, 68629 through 68630) [42] are substantially different from the targeted mechanical core biopsies of today. The commenter explained that technological advances in incision devices began in the late 2010s with various alterations to needle shapes and have continued to improve and progress to the current, mechanical, EUS-guided devices, which do not merely create incisions and should not be classified along with biopsy forceps or even aspiration needles. Therefore, the commenter requested that CMS consider the definition of a supply and consider re-classification of mechanical tissue extraction apparatuses such as Precision GI into a new category of devices outside of the existing definition. The commenter stated their belief that this would enable CMS to determine the benefits and costs of single-use devices and future innovations in this space and determine their role in therapeutic decision-making and associated improvements in patient outcomes.
Response: We thank the commenter for their feedback. We will continue to consider issues related to the definition of a biopsy apparatus as it relates to the criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. The applicant asserted that Precision GI is authorized by FDA with an indication to sample targeted submucosal lesions, mediastinal masses, lymph nodes, and intraperitoneal masses within or adjacent to the GI tract. Per the applicant, the device category C1830 (Powered bone marrow biopsy needle) does not appropriately describe Precision GI because Precision GI is not targeting bone marrow and instead is targeting sub‐mucosal and extramural GI lesions. According to the applicant, the device category C1782 (Morcellator) also does not appropriately describe Precision GI because that device category, per Medicare Claims Processing Manual, Ch. 4, section 60.4.3, is only for laparoscopic procedures. The applicant added that Precision GI cuts and extracts tissue endoscopically, not laparoscopically.
We did not identify an existing pass-through payment category that describes Precision GI. We invited public comment on whether Precision GI meets the device category criterion at § 419.66(c)(1).
Comment: With respect to the device category criterion at § 419.66(c)(1), the applicant agreed with our assessment that there are no existing pass-through payment categories that describe Precision GI.
Response: We appreciate the applicant's input. After consideration of the public comments received and our review of the application, we continue to believe there is no existing category or category previously in effect that appropriately describes Precision GI. Therefore, we have determined that Precision GI meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. Precision GI has a Breakthrough Device designation and marketing authorization from FDA for ( printed page 94163) the indication covered by the Breakthrough Device designation (as explained in more detail in the discussion of the newness criterion) and therefore is not evaluated for substantial clinical improvement.
We invited public comment on whether Precision GI meets the device category criterion at § 419.66(c)(2)(ii).
Comment: With respect to the device category criterion at § 410.66(c)(2)(ii), the applicant reiterated that Precision GI received FDA Breakthrough Device designation and marketing authorization for the same indication. The applicant agreed with CMS that Precision GI meets the device category criterion at § 410.66(c)(2)(ii), and therefore, is not evaluated for substantial clinical improvement.
Response: We appreciate the applicant's input. FDA granted Precision GI Breakthrough Device designation effective March 24, 2022, and a marketing authorization effective August 28, 2023, for the indication covered by the Breakthrough Device designation. Therefore, Precision GI meets the criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). After consideration of public comments received and our review of the application, we have determined that Precision GI meets the device category criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that Precision GI would be reported with HCPCS codes as shown in Table 120.
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant utilized the CY 2024 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5302, which had a CY 2024 payment rate of $1,812.99 at the time the application was received. HCPCS code 43242 in APC 5302 had a CY 2024 device offset amount of $23.75 at the time the application was received.[43] According to the applicant, the cost of Precision GI is $1,400.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $1,400.00 for Precision GI is 77.22 percent of the applicable APC payment amount for the service related to the category of devices of $1,812.99 (($1,400.00/$1,812.99) × 100 = 77.22 percent). Therefore, we stated that we believe Precision GI meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount. The estimated average reasonable cost of $1,400.00 for Precision GI is 5894.74 percent of the cost of the device-related portion of the APC payment amount for the related service of $23.75 (($1,400.00/$23.75) × 100 = 5894.74 percent). Therefore, we stated that we believe Precision GI meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of ( printed page 94164) $1,400.00 for Precision GI and the portion of the APC payment amount for the device of $23.75 is 75.91 percent of the APC payment amount for the related service of $1,812.99 ((($1,400−$23.75)/$1,812.99) × 100 = 75.91 percent). Therefore, we stated that we believe Precision GI meets the third cost significance requirement.
We invited public comment on whether Precision GI meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to cost significance criteria, the applicant stated that they agree with CMS that Precision GI satisfies the cost criteria.
Response: We appreciate the applicant's input. After consideration of the public comment received and our findings from the first, second, and third cost significance tests, we have determined that Precision GI meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments received, and our review of the device pass-through application, we have determined that Precision GI meets the requirements for device pass-through status described at § 419.66, and we are finalizing approval for device pass-through payment status for Precision GI effective January 1, 2025, under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(h) Symplicity SpyralTM Renal Denervation (RDN) System
Medtronic submitted an application for a new device category for transitional pass-through payment status for the Symplicity SpyralTM RDN System for CY 2025. The applicant is seeking a new device category for transitional pass-through payment status only for the Symplicity SpyralTM Multi-Electrode RDN Catheter (hereinafter Symplicity SpyralTM Catheter) component of the Symplicity SpyralTM RDN System. According to the applicant, the Symplicity SpyralTM RDN System consists of the Symplicity SpyralTM Catheter and the Symplicity G3TM generator; however, the applicant requested device pass-through status for the catheter component of the system only. The applicant further explained that the Symplicity SpyralTM RDN System is indicated to reduce blood pressure as an adjunctive treatment in hypertension patients in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure. Per the applicant, the Symplicity SpyralTM Catheter, when used with the Symplicity G3TM generator, delivers radiofrequency (RF) energy through the wall of the renal artery to disrupt the surrounding renal nerves with the aim of modulating or suppressing sympathetic nerve hyperactivity. According to the applicant, the Symplicity SpyralTM Catheter is a single-use catheter used to deliver multiple ablations in both kidneys, in the renal main, accessory, and branch arteries, based on a patient's artery anatomy and size.
Per the applicant, the Symplicity SpyralTM Catheter is designed to be used with the Symplicity G3TM generator and includes the following components: (1) four gold radiopaque electrodes at the spiral (helical) distal end that are deployed into a spiral (helical) shape by partially retracting the guidewire proximal to the spiral section of the catheter; (2) self-expanding electrode array assembly which radially spaces the four gold electrodes for quadratic ablation; (3) rapid exchange port; (4) straightening tool intended to facilitate safe insertion of the guidewire into the catheter; (5) cable connector attached to the catheter handle that connects the catheter to the generator; (6) catheter handle; and (7) femoral marker. Per the applicant, additional components of the Symplicity SpyralTM RDN System include the: (1) Symplicity G3TM generator, which is only compatible with the Symplicity SpyralTM Catheter, which includes a remote control, power cable, and output for the Symplicity SpyralTM Catheter; (2) Symplicity G3TM generator cart, an optional mobile cart; and (3) foot switch, an optional component. Per the applicant, additional items required for the procedure include: (1) a 0.36 mm (0.014 in) guidewire, preferably without hydrophilic coating; (2) a dispersive electrode; (3) a sterile bag to cover the remote control if used in the sterile field; (4) a 6 French (Fr) guide-catheter; (5) an introducer sheath; (6) a stopcock sidearm; (7) a Tuohy-Borst adapter; and (8) other standard items used to aid percutaneous transluminal catheterization in renal arteries.
According to information submitted by the applicant, key steps for operating the Symplicity SpyralTM Catheter include: (1) connecting the Symplicity SpyralTM Catheter to the Symplicity G3TM generator; (2) inserting the Symplicity SpyralTM Catheter through a small femoral incision and guiding it to the renal artery via the abdominal aorta; (3) advancing the Symplicity SpyralTM Catheter until the distal electrode is located in the desired position within the renal artery; (4) retracting the guidewire, allowing the self-expanding catheter to expand and fit the renal arterial vessel walls; (5) delivering the treatment of RF energy to ablate the renal nerves through the activation of the catheter electrodes, which is controlled using the generator; (6) if treating another vessel, repositioning the guide catheter within the next vessel and repeating the procedure for positioning the catheter and delivering treatments; (7) upon completion of all treatments, straightening the distal end by advancing the guidewire and withdrawing both the guidewire and the straightened catheter from the guide catheter; (8) retracting the guide catheter from the sheath and removing the introducer sheath from the artery; (9) using standard of care procedures to achieve hemostasis at the puncture site; and (10) disposing of the devices.
Please refer to the online application posting for the Symplicity SpyralTM RDN System, available at https://mearis.cms.gov/public/publications/device-ptp/DEP231130WPU4J, for additional detail describing the device and the disease treated by the device.
Comment: Many commenters expressed their support for the approval of the Symplicity SpyralTM RDN System for transitional pass-through payment.
Response: We appreciate the commenters' input and support for the Symplicity SpyralTM RDN System application for transitional pass-through payment. We have taken these comments into consideration in our final determination for pass-through status for the Symplicity SpyralTM RDN System.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), the Symplicity SpyralTM RDN System received FDA Breakthrough Device designation effective March 27, 2020, as a device with the following indicated use: the Symplicity SpyralTM multi-electrode renal denervation catheter and the Symplicity G3TM RF Generator are indicated for the reduction of blood pressure in patients with uncontrolled hypertension despite the use of anti-hypertensive medications or in patients who may have documented intolerance to anti-hypertensive medications. FDA approved the premarket approval application (PMA) for the Symplicity SpyralTM RDN System on November 17, 2023, for the indicated use to reduce blood pressure as an adjunctive treatment in patients with hypertension ( printed page 94165) in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure. We received the application for a new device category for transitional pass-through payment status for the Symplicity SpyralTM Catheter on November 30, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the Symplicity SpyralTM Catheter meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the Symplicity SpyralTM Catheter received FDA marketing authorization on November 17, 2023, and the Symplicity SpyralTM RDN System was granted Breakthrough Device designation effective March 27, 2020. A few other commenters also stated that they believed the Symplicity SpyralTM RDN System meets the newness criterion.
Response: We appreciate the applicant's and commenters' input. The Symplicity SpyralTM RDN System received FDA Breakthrough Device designation on March 27, 2020. We received the application for a new device category for transitional pass-through payment status for the Symplicity SpyralTM Catheter on November 30, 2023, which is within 3 years of November 17, 2023, the date of FDA PMA. After consideration of the public comments received and our review of the application, we have determined that the Symplicity SpyralTM Catheter meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not indicate whether the Symplicity SpyralTM Catheter is integral to the service furnished. Per the applicant, the Symplicity SpyralTM Catheter is intended for single-patient use only. While the applicant did not explicitly state whether the Symplicity SpyralTM Catheter comes into contact with human tissue, the applicant asserted that the SymplicityTM Catheter ablates renal nerve tissue by positioning the catheter within the renal artery, which expands and fits the renal arterial vessel walls. While the applicant did not explicitly state if the Symplicity SpyralTM Catheter is surgically inserted or implanted, per the device description, the Symplicity SpyralTM Catheter is inserted through a small femoral incision, after which it is inserted into the renal arterial vessel.
We invited public comment on whether the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant submitted a comment stating that the Symplicity SpyralTM catheter meets all eligibility criteria as laid out at § 419.66(b)(3). The applicant clarified that the Symplicity SpyralTM Catheter is required to ablate the renal sympathetic nerves in this procedure, is integral to the service furnished, and meets the eligibility criterion at § 419.66(b)(3). The applicant further clarified that the Symplicity SpyralTM Catheter comes into contact with human tissue because it is inserted through the skin and into the arterial system, and is then introduced into the renal artery.
Response: We appreciate the applicant's input. We agree with the applicant that the Symplicity SpyralTM Catheter is an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion because the Symplicity SpyralTM Catheter is a single-use catheter and is inserted through a small femoral incision, after which it is inserted into the renal arterial vessel. After consideration of the public comments we received and our review of the application, we have determined that the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not state whether the Symplicity SpyralTM Catheter is equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or whether the Symplicity SpyralTM Catheter is a supply or material furnished incident to a service.
We invited public comment on whether the Symplicity SpyralTM Catheter meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant submitted a comment confirming that the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(b)(4) because it is not equipment, an instrument, apparatus, implement, or item for which depreciation and financing expenses are recovered as defined in Chapter 1 of the Medicare Provider Reimbursement Manual. The applicant clarified that the Symplicity SpyralTM Catheter is not a supply or material furnished incident to a service. The applicant commented that the Symplicity SpyralTM Catheter is used with the Symplicity G3TM generator, which may be considered depreciating equipment as defined at § 419.66(b)(4); however, the applicant noted that it is only seeking pass-through status for the single-use Symplicity SpyralTM Catheter.
Response: We thank the applicant for the clarification and agree with the applicant that the Symplicity SpyralTM Catheter is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. After consideration of the public comments we received and our review of the application, we have determined that the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. According to the applicant, the Symplicity SpyralTM Catheter is a single-use catheter used to deliver multiple ablations in both kidneys, in the renal main, accessory, and branch arteries, based on a patient's artery anatomy and size. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe the Symplicity SpyralTM Catheter. The applicant asserted that two categories, C1886 (Catheter, extravascular tissue ablation, any modality (insertable)) and C1888 (Catheter, ablation, noncardiac, endovascular (implantable)), do not appropriately describe the Symplicity SpyralTM Catheter. Per the applicant, C1886 does not appropriately describe the Symplicity SpyralTM Catheter because the Symplicity SpyralTM ( printed page 94166) Catheter ablates renal nerve tissue via an endovascular approach by positioning the catheter within the renal artery and C1886 was created to describe devices that ablate extravascular tissue (via an extravascular approach).[44] According to the applicant, C1888 does not appropriately describe the Symplicity SpyralTM Catheter because the Symplicity SpyralTM Catheter does not ablate or otherwise affect the blood vessel tissue and C1888 was created to describe devices designed to occlude or obliterate blood vessels.[45]
We did not identify an existing pass-through payment category that describes the Symplicity SpyralTM Catheter. We invited public comment on whether the Symplicity SpyralTM Catheter meets the device category criterion at § 419.66(c)(1).
Comment: The applicant and a few other commenters agreed with CMS' assessment that there are no existing pass-through payment categories that describe the Symplicity SpyralTM Catheter.
Response: We appreciate the commenters' input. After consideration of the public comments we received and our review of the application, we continue to believe that there is no existing category or category previously in effect that appropriately describes the Symplicity SpyralTM Catheter. Therefore, we have determined that the Symplicity SpyralTM Catheter meets the device category eligibility criterion at § 419.66(c)(1).
We noted that the applicant indicated the Symplicity SpyralTM Catheter is not the only device authorized by FDA with an indication for renal denervation to achieve reductions in blood pressure. Paradise® Ultrasound RDN System, for which we also received an application for transitional pass-through payments for CY 2025 as discussed in more detail in the proposed rule (89 FR 59304 through 59307) and this final rule with comment, is also authorized by FDA with an indication for renal denervation using ultrasound energy to achieve reductions in blood pressure. Per the applicant, the Paradise® Ultrasound RDN System would also be described by the applicant's proposed pass-through payment category: Ablation catheter, renal nerve, via endovascular approach, any modality. Accordingly, we noted that while the Symplicity SpyralTM Catheter device may have a different modality ( i.e., radiofrequency compared to ultrasound), the Symplicity SpyralTM Catheter device may have a similar mechanism of action to that of the Paradise® Ultrasound RDN System device. We questioned whether the device descriptions provided in the respective applications support establishing two modality specific pass-through payment device categories or a single device category that would encompass both RDN device modalities. We address this question in section (i) in detail immediately following the full discussion of all other applicable eligibility criteria for the Symplicity SpyralTM RDN System application.
For the summary of public comments received and our final decision on the question of whether the device descriptions provided in the respective applications support establishing two modality-specific pass-through payment device categories or a single device category that would encompass both RDN device modalities, please see section (i) immediately following the Symplicity SpyralTM RDN System application determination.
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. As previously stated, the Symplicity SpyralTM Catheter has Breakthrough Device designation and marketing authorization from FDA for the indication covered by the Breakthrough Device designation (as explained in more detail in the discussion of the newness criterion) and therefore is not evaluated for substantial clinical improvement.
We invited public comment on whether the Symplicity SpyralTM Catheter meets the device category criterion at § 419.66(c)(2)(ii).
Comment: The applicant submitted a comment confirming that the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(c)(2)(ii) because as an FDA Breakthrough Device, it meets the device category criterion as described at § 419.66(c)(2)(ii). A few commenters also stated that the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(c)(2)(ii).
Response: We appreciate the applicant's and commenters' input. The Symplicity SpyralTM RDN System has a Breakthrough Device designation, as stated by the applicant, effective March 27, 2020, and marketing authorization from FDA effective November 17, 2023, for the indication covered by the Breakthrough Device designation and therefore, meets the criterion at § 419.66(c)(2)(ii) and is not evaluated for substantial clinical improvement at § 419.66(c)(2)(i). Based on our review of the application and consideration of the public comments we received, we have determined that the Symplicity SpyralTM Catheter meets the eligibility criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the Symplicity SpyralTM Catheter would be reported with HCPCS codes shown in Table 121.
( printed page 94167)To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2024 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5192, which had a CY 2024 payment rate of $5,445.84 at the time the application was received. HCPCS code 0339T in APC 5192 had a CY 2024 device offset amount of $3,362.26 at the time the application was received.[46] According to the applicant, the cost of the Symplicity SpyralTM Catheter is $16,000.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $16,000.00 for the Symplicity SpyralTM Catheter is 293.80 percent of the applicable APC payment amount for the service related to the category of devices of $5,445.84 (($16,000.00/$5,445.84) × 100 = 293.80 percent). Therefore, we stated that we believe the Symplicity SpyralTM Catheter meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $16,000.00 for the Symplicity SpyralTM Catheter is 475.87 percent of the cost of the device-related portion of the APC payment amount for the related service of $3,362.26 (($16,000.00/$3,362.26) × 100 = 475.87 percent). Therefore, we stated that we believe the Symplicity SpyralTM Catheter meets the second cost significance requirement.
The third cost significance requirement at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $16,000.00 for the Symplicity SpyralTM Catheter and the portion of the APC payment amount for the device of $3,362.26 is 232.06 percent of the APC payment amount for the related service of $5,445.84 ((($16,000.00−$3,362.26)/$5,445.84) × 100 = 232.06 percent). Therefore, we stated that we believe the Symplicity SpyralTM Catheter meets the third cost significance requirement.
We invited public comment on whether the Symplicity SpyralTM Catheter meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to cost significance criteria, the applicant and a few other commenters reiterated that the Symplicity SpyralTM Catheter meets all three of the cost significance criteria.
Response: We appreciate the applicant's and commenters' input. After consideration of the public comments we received and our findings from the first, second, and third cost significance tests, we agree that the Symplicity SpyralTM Catheter meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments we received and our review of the device pass-through application, we have determined that the Symplicity ( printed page 94168) SpyralTM Catheter meets the requirements for device pass-through status described at § 419.66. Therefore, effective beginning January 1, 2025, we are finalizing approval for device pass-through payment status for the Symplicity SpyralTM Catheter under the alternative pathway for devices that have an FDA Breakthrough Device designation and have received FDA marketing authorization for the indication covered by the Breakthrough Device designation.
(i) Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System Device Category Code Establishment
As previously discussed, we received two applications for transitional device pass-through status for RDN devices: the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System (Symplicity SpyralTM Catheter). Per the applicants, both are RDN devices that use an endovascular approach to enter the renal arteries and ablate renal sympathetic nerves to achieve reductions in blood pressure. We questioned whether the information provided for each nominated device supported establishing two modality-specific device pass-through payment device categories or establishing a single device category that would encompass both RDN devices.
We noted that the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System are both authorized by FDA for the indicated use to reduce blood pressure as an adjunctive treatment in patients with hypertension in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure. In addition, based on the information provided in the respective applications, we stated that it appears that both devices use the same procedure (renal sympathetic denervation, also called renal sympathetic nerve ablation) with the same underlying mechanism of action (thermal ablation), treat the same disease (hypertension) in the same patient population (patients in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure), and aim to achieve the same therapeutic outcome (to reduce blood pressure). We noted that in addition to having the same indicated use, per the applicants, both devices may be used with the same HCPCS procedure codes: 0338T [47] or 0339T.[48]
We noted several differences in procedural technique with use of the Paradise® Ultrasound RDN System device compared to the Symplicity SpyralTM RDN System device. Per the applicants, the Paradise® Ultrasound RDN System delivers ablation while positioned in the main renal arteries only, whereas the Symplicity SpyralTM RDN System may deliver ablation while positioned in the main renal, accessory, and branch arteries and therefore may require advancing the catheter beyond the main renal arteries. According to the applicants, the Paradise® Ultrasound RDN System procedural technique requires the measurement of the main renal artery diameter to select an appropriate size cooling balloon catheter, whereas the Symplicity SpyralTM RDN System does not require a cooling balloon catheter nor this measurement. Similarly, per the applicants, the Paradise® Catheter's cooling balloon catheter requires additional procedural techniques to ensure the balloon is appropriately inflated and deflated during the procedure, but the Symplicity SpyralTM Catheter does not require this.
The two applicants proposed different device category descriptions. Based on the information submitted, we acknowledged that the device category proposed by the applicant for the Paradise® Ultrasound RDN System would not appropriately describe the Symplicity SpyralTM RDN System, but that the device category proposed by the applicant for the Symplicity SpyralTM RDN System would appropriately describe the Paradise® Ultrasound RDN System. Specifically, the applicant for the Paradise® Ultrasound RDN System proposed the following device category: Catheter, intravascular renal denervation, ultrasound, with balloon cooling ,[49] while the applicant for the Symplicity SpyralTM RDN System proposed the following device category: Ablation catheter, renal nerve, via endovascular approach, any modality. The device category descriptor the applicant proposed for the Paradise® Ultrasound RDN System specifies the device's treatment or procedure (catheter renal denervation), the ablation modality ( i.e., ultrasound), and an attribute of the catheter ( i.e., the cooling balloon). We noted that while the device category descriptor the applicant proposed for the Symplicity SpyralTM RDN System device also specifies the device's treatment or procedure (catheter ablation of the renal nerves), it does not specify an ablation modality or any additional attributes of the catheter; rather, the proposed device category for the Symplicity SpyralTM RDN System more broadly describes any ablation modality ( e.g., radiofrequency or ultrasound).
The applicant for the Symplicity SpyralTM RDN System asserted that the use of a common thermal mechanism of action in both systems supported the establishment of a single device category irrespective of modality. However, we noted the applicant of the Paradise® Ultrasound RDN System stated a separate device category associated with ultrasound denervation is supported by possible differences in clinical efficacy between RDN devices using ultrasound and those using radiofrequency ablation. However, we noted that we did not evaluate the validity or generalizability of these claims, nor was it clear if the two different ablation modalities ( i.e., ultrasound and radiofrequency) would render different clinical results in larger studies or in the long term.[50 51] We further noted that we discussed these claims solely for the purpose of ( printed page 94169) determining whether the information provided supports establishing two modality-specific pass-through payment device categories or establishing a single device category that may encompass both RDN devices.
In seeking comment, we noted that in accordance with section 1833(t)(6)(B)(ii)(II) of the Act, new categories must be established in such a way that no medical device is described by more than one category. We further noted that CMS does not establish pass-through device categories for the purposes of describing specific devices, but rather, device categories which are intended to encompass all devices that can be appropriately described by a category. However, there are instances where CMS has defined specific ablation modalities ( e.g., high intensity ultrasound, microwave, and cryoablation) in HCPCS codes for use with the OPPS. In addition, we noted the existence of several modality-specific tissue ablation procedure codes in the Current Procedural Terminology (CPT®) 2024. However, there are examples where CMS has established device categories with additional granularity to differentiate similar devices with special characteristics ( e.g., implantable neurostimulators). Further, there are examples where CMS has created HCPCS codes specifying the modality of ablation ( e.g., ultrasound, microwave). Finally, we noted that the intent of transitional device pass-through payment, as implemented at § 419.66, is to facilitate access for beneficiaries to the advantages of new and truly innovative devices by allowing for adequate payment for these new devices while the necessary cost data is collected to incorporate the costs for these devices into the procedure APC rate (66 FR 55861). We questioned whether two modality-specific device category codes may facilitate the collection of more accurate data for incorporating the costs of these two devices into the procedure APC rate as well as foster the tracking of efficacy data for these two ablation modalities.
As such, we invited public comment on whether the device descriptions provided in the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System applications support establishing two modality-specific pass-through payment device categories or a single device category that would encompass both RDN device modalities.
Comment: Commenters, including the Paradise® Ultrasound RDN System applicant, overwhelmingly supported establishing two pass-through payment device categories. However, some commenters suggested that the device category codes be specific to the physical device characteristics while other commenters suggested the device category codes be specific to modality. Specifically, the applicant for the Paradise® Ultrasound RDN System and many other commenters stated that two device categories are more appropriate for many reasons such as documented procedural differences between the two treatment modalities, differences in the prices between the two technologies, CMS's previous creation of separate and distinct device categories for similar technologies, CMS's recent creation of different ICD-10-PCS codes for radiofrequency and ultrasound renal denervation modalities in the FY 2025 IPPS final rule, and to facilitate the tracking of each device's efficacy data.
We received a few comments, including from the Symplicity SpyralTM RDN System applicant, who suggested that CMS establish a single device category that would encompass both RDN device modalities. The Symplicity SpyralTM RDN System applicant and a few other commenters supported the establishment of an energy modality-agnostic RDN device pass-through payment device category for several reasons. Specifically, the Symplicity SpyralTM RDN System applicant emphasized the similarities between the two devices and procedures, opinions from the physician community, existing policy and coding standards, and potential future consequences of this determination. The Symplicity SpyralTM RDN System applicant also disagreed with many of the reasons for separate pass-through payment device categories provided by the Paradise® Ultrasound RDN System applicant that were outlined in the proposed rule.
Response: We appreciate the feedback provided by the applicants and the commenters on this topic. We have taken the points raised into consideration in our final decision.
Comment: Commenters, including the Paradise® Ultrasound RDN System applicant noted many procedural differences between the two treatment modalities. For example, the radiofrequency system ( i.e., Symplicity SpyralTM RDN System) requires ablation of the main renal artery and branches of the main renal artery, whereas the Paradise® Ultrasound RDN System normally requires ablation of only the main renal artery, leading to significantly fewer ablations [52 53] and the Paradise® Ultrasound RDN System requires the estimation of the size of the patient's renal artery to ensure the correct balloon size is utilized, which may require additional equipment or procedural steps at the discretion of the operator compared to the Symplicity SpyralTM RDN System procedure, where this is not required. In addition, the applicant and many commenters noted that the Symplicity SpyralTM RDN System procedure takes longer than the overall procedure for the Paradise® Ultrasound RDN System, and the Paradise® Ultrasound RDN System applicant specified this difference in procedure time as approximately 30 percent.[54 55 56 57 58] Many other commenters also indicated additional potential procedural differences between the two devices, including variation in the number of catheters used, pre- and post-procedure scheduling and workflow protocols, complexity of the procedure, depth of ablation, specialized training requirements, patient selection and management issues, and recovery time.
With respect to procedural and device similarities, the Symplicity SpyralTM RDN System applicant and another commenter reiterated that both devices share the same intended use, for the same patient population, with identical ( printed page 94170) or nearly identical FDA-approved indications, and are deployed similarly, on the same anatomy. The Symplicity SpyralTM RDN System applicant further stated that the two devices have the same mechanism of action (thermal ablation of renal sympathetic nerves using heat energy), a similar form factor and product composition (single use endovascular catheters, connected to a generator, and delivered over a wire to the renal arteries, with distal ends containing heat energy emitting implements which ablate nerve tissue), and that the RDN procedure performed with either technology is fundamentally the same. The Symplicity SpyralTM RDN System applicant also asserted that even though there may be small differences in the placement of each device along the renal arterial vasculature, they believe these small differences can be expected for devices that are inherently similar but not identical in form factor, size, and deliverability. The Symplicity SpyralTM RDN System applicant asserted that the RDN procedure performed by both devices is also fundamentally the same, as both include catheter-based femoral access and closure, intraprocedural imaging, physician wire skills and expertise, and the ablation targeting of the sympathetic renal nerves to achieve the identical therapeutic goal of reducing blood pressure.
The Symplicity SpyralTM RDN System applicant reiterated that the Paradise® Catheter's cooling balloon requires specific procedural techniques to ensure the balloon is appropriately inflated and deflated during the procedure, noting that the Symplicity SpyralTM Catheter does not have this requirement. The applicant stated that the Symplicity SpyralTM Catheter does not require a cooling balloon as the non-occlusive spiral design of the radiofrequency catheter allows for continued collateral blood flow to cool the artery wall during ablation. Further, while use of the Paradise® Catheter's cooling balloon does require some different considerations when compared to the Symplicity SpyralTM Catheter, particularly related to vessel size, the cooling balloon itself is not providing the therapeutic effect ( i.e. ablation of the nerves); rather, the balloon is preventing vessel damage from heat energy. The applicant stated this distinction makes it similar to many medical devices which provide the same treatment but require slightly different considerations based on manufacturer-specific equipment differences, and while the energy modality has driven some differences in the Symplicity SpyralTM Catheter and the Paradise® Catheter designs, the overall procedure is substantially similar between the two technologies.
With respect to the differences in procedural technique described in the proposed rule that the Paradise® Ultrasound RDN System only ablates in the main renal arteries, while the Symplicity SpyralTM RDN System typically involves ablation in the main renal arteries, accessories, and branches, the applicant stated that according to the Paradise® Ultrasound RDN System instructions for use (IFU),[59] the Paradise® Ultrasound RDN System is recommended for treatment of branches and accessory arteries, provided they are of appropriate size to accommodate the balloon and meet specific spacing requirements. The Symplicity SpyralTM RDN System applicant explained that the Symplicity SpyralTM RDN System IFU [60] do not specify or require that distal branches be treated (only a size range for vessel diameters is specified). Furthermore, per the applicant, a thorough treatment strategy typically including distal branches is recommended for the Symplicity SpyralTM RDN System. The applicant noted that this recommendation is based on available knowledge of renal sympathetic nerve anatomy, to increase the likelihood of thorough and complete denervation up to the size limitations of the device, given that the current procedure and technology do not yet have an intraprocedural way to validate successful denervation. Additionally, the Symplicity SpyralTM RDN System applicant noted that several studies, including a sub-analysis of the Paradise® Ultrasound RDN System applicant's RADIANCE-HTN SOLO [61] study, point to the importance of thorough ablation, including appropriate branch and accessory renal arteries, to achieving optimal nerve ablation and blood pressure reduction with RDN.[62 63] Further, the Symplicity SpyralTM RDN System applicant stated that proceduralists with both devices seek to denervate to the greatest extent possible up to the limitations of the RDN technology used, and there are many considerations in how proceduralists may determine where to denervate, and with how many energy emissions, based on clinical experience and patient anatomical considerations.
The Symplicity SpyralTM RDN System applicant disagreed with some assertions provided in the Paradise application and discussed in some of the comments supporting the creation of two device category codes. Primarily, the applicant asserted that the differences in intraprocedural metrics cited in the proposed rule noted are not accurate comparisons. For example, the Symplicity SpyralTM RDN System applicant stated that the average procedure times from respective pooled analyses suggest that the total procedure time is 85 minutes for the Symplicity SpyralTM RDN System [64] and 78 minutes for the Paradise® Ultrasound RDN but the total procedure times attributed to using either device are variable across studies, and, due to differing trial procedural requirements and physician experience, it is difficult to compare procedure times across studies. In addition, the applicant further stated other intraprocedural metrics, such as total ablation time and ablation time per cycle, are not necessarily reflective of additional resources involved in the procedures. The Symplicity SpyralTM RDN System applicant noted that in the proposed rule CMS stated that the Symplicity SpyralTM Catheter required more ablations per patient at 46.9, compared to 5.4 ablations for the Paradise® Ultrasound RDN System. However, the applicant stated that as a quadripolar device, each of the Symplicity SpyralTM Catheter's four electrodes may be activated independently for focal treatments, causing the difference in the number of ablations. The applicant clarified that the SPYRAL HTN-OFF MED ( printed page 94171) randomized controlled trial [65] counted ablations from each of the four electrodes of the Symplicity SpyralTM Catheter as a separate ablation, although most ablation cycles would have counted the four ablations simultaneously as a single ablation. The applicant indicated that given this design difference, it is misleading to draw comparisons between those two data points for each system. The applicant also clarified that regarding ablation depth, cadaveric studies found that >90 percent of the renal nerves lie within 6.39 mm of the arterial lumen.[66] While the applicant stated that it cannot independently confirm the 1-6 mm ablation depth cited for the Paradise® Ultrasound RDN System, a histological study of radiofrequency RDN in porcine models using the Symplicity SpyralTM RDN System found a comparable mean lesion depth of 6.26 mm (+/- 1.62) in the main renal segment.[67] The Symplicity SpyralTM RDN System applicant also stated that regardless of modality, ablation depth is not uniform and occurs asymmetrically as a result of heating different tissue types surrounding the renal artery (given the presence of other organs surrounding the renal artery, and anatomical variability between patients, this heat energy is variably dissipated). Therefore, the applicant asserted that ablation depth, by itself, is an incomplete two-dimensional measure of a three-dimensional phenomenon.
Response: We thank the commenters and the applicants for the information. We agree with commenters that the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System have the same underlying thermal mechanism of action, have the same intended use for the same patient population, and have identical FDA-approved primary indications. However, we continue to believe that, while there are similarities, the devices are not identical and warrant separate device categories. We note that commenters, including the applicants, addressed potential clinical impacts, efficacy, and outcomes in the comments. While we have included some of those details in our discussion for the purposes of illustrating the procedural comparison of the two devices and determining whether the information provided supports establishing two modality-specific pass-through payment device categories or establishing a single device category that may encompass both RDN devices, we note that we did not evaluate the validity or generalizability of these claims, nor was it is clear if the two different ablation modalities ( i.e., ultrasound and radiofrequency) would render different clinical results in larger studies or in the long term. In regard to comments on the differences in clinical trial data between the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System, we are not evaluating any evidence submitted in relation to the devices' clinical outcomes or the comparison of clinical outcomes to other similar devices because both devices meet the requirements at § 419.66(c)(2)(ii) and are not required to prove substantial clinical improvement for pass-through payment, as discussed in this final rule.
With regard to the procedural differences we noted in the proposed rule, we agree with commenters that there are some procedural differences between the two RDN technologies and that the long-term impact of these differences on patient outcomes is unknown. We appreciate that differences between the two treatment modalities may result in different resource requirements such as operating room time, more anesthetic, and more contrast medium. We also agree with the many commenters who stated the Paradise® Ultrasound RDN System may require additional equipment or procedural steps to determine the estimation of the size of the patient's renal artery to ensure the correct balloon size is utilized.
With regards to comments stating the cooling balloon itself is not providing the therapeutic effect ( i.e. ablation of the nerves); rather, the balloon is preventing vessel damage from heat energy, we appreciate that the cooling balloon does not constitute a difference in mechanism of action. However, we believe the different considerations of resources due to the differences of the cooling balloon may be considered a significant difference in modalities that could reasonably suggest a need for separate device codes.
Comment: The Paradise® Ultrasound RDN System applicant and many commenters stated that the difference in price between the two technologies also warrants two device categories. The Paradise® Ultrasound RDN System applicant and many other commenters stated that while the transitional pass-through payment calculation is based on hospital charges, having only one device category could lead to undercharging for one technology and overcharging for the other. The Paradise® Ultrasound RDN System applicant and many other commenters further stated that having two distinct device categories would allow hospitals to set charges that accurately reflect the cost of each procedure, thereby enabling CMS to more accurately calculate the costs of each procedure. The applicant and a few commenters further stated two distinct device categories would provide additional flexibility for CMS to more fully evaluate both modalities over the pass-through period to determine the most appropriate APC assignment when transitional pass-through payment expires.
The Symplicity SpyralTM RDN System applicant commented that it appreciates CMS' intent to facilitate access for beneficiaries to innovative technologies by allowing for adequate payment while cost data is collected for APC assignments, and that the payment calculation methodology for transitional device pass-through provides for adequate payments based on charges on individual hospital claims adjusted to costs during the 3-year eligibility period, regardless of device category assignment. The applicant further commented that when the pass-through period expires and CMS packages the procedures into an APC, payment will be calculated at the level of the CPT code, which both RDN technologies share. The applicant claimed that the RDN device costs for both technologies will be packaged into the payment for the same CPT code, whether these costs are similar or not. The Symplicity SpyralTM RDN System applicant therefore questioned the utility of using separate device categories for cost tracking. The Symplicity SpyralTM RDN System applicant also opined that tracking efficacy data for the two devices is beyond the fundamental purpose of establishing device categories and should not be a primary consideration. The applicant stated that both manufacturers are engaged in post- ( printed page 94172) market research to continue building the body of evidence for RDN specific to each technology, and the research will continue independent of the establishment of pass-through device categories. The applicant urged CMS to prioritize and maintain an OPPS policy-based rationale in establishing new device categories, with focus on the payment-driven mechanisms and purposes of the device category codes.
Response: In response to comments on the difference in price between the two technologies and whether that difference warrants two device categories, we note that while price difference considerations are generally outside the scope of the traditional pass-through payment program, we agree with commenters that two distinct device categories will provide an easier mechanism for hospitals to set charges that reflect the price differences between the two device categories which will enable CMS to more accurately calculate the costs of each procedure for future rulemaking.
Comment: The Paradise® Ultrasound RDN System applicant and many commenters asserted that CMS has previously created separate and distinct device categories for similar technologies. The applicant suggested the most notable of these are the neurostimulators, where CMS created categories to differentiate rechargeable, non-rechargeable, high-frequency, and closed feedback loop neurostimulators for the treatment of chronic pain.[68] The Paradise® Ultrasound RDN System applicant stated that other neurostimulator categories have also been created for phrenic nerve stimulation, paired stimulation, and carotid sinus baroreceptor stimulation. Further, the applicant stated that there are also currently five device categories [69] that are all electrophysiology catheters. The applicant stated that they believe that these device categories appropriately capture the characteristics among these groups of technologies to allow for the most accurate reporting of device costs. The Paradise® Ultrasound RDN System applicant opined that the situation with respect to the Paradise® Ultrasound RDN System and the SymplicityTM Spryal RDN System is similar to the examples presented, and therefore, it is appropriate to have two device categories to differentiate tracking of ultrasound and radiofrequency renal denervation. Several other commenters also referenced device categories that were differentiated due to their unique characteristics, including examples related to cardiology, neurostimulators, catheters, and ablation modalities ( i.e., high-intensity ultrasound, microwave, and cryoablation), to support their position that the Paradise® Ultrasound RDN System and the SymplicityTM Spryal RDN System should be classified within two different pass-through payment device categories.
Further, the Paradise® Ultrasound RDN System applicant and many commenters noted that CMS recently created different ICD-10-PCS codes that differentiate between radiofrequency and ultrasound renal denervation modalities for the new technology add-on payments confirmed in the IPPS final rule for FY 2025. The applicant and several commenters opined that it is important to be consistent across inpatient and outpatient settings as the same rationale for separation and independent tracking of costs applies in both instances. A few commenters stated that the use of separate ICD-10-PCS codes for the two devices reflect their unique contributions to the treatment of hypertension.
The applicant for the Symplicity SpyralTM RDN System argued that pass-through device categories are not established for the purpose of describing specific devices; rather, device categories are intended to encompass all devices that can be appropriately described by a category (89 FR 59316). The applicant acknowledged that, as stated in the proposed rule, CMS has established energy-specific device categories in the past; however, the applicant asserted that the establishment of device categories requires a nuanced approach, balancing appropriate granularity without becoming manufacturer- or device-specific. The applicant asserted that CMS must assess each situation on a case-by-case basis, in the context of the procedure and device in question, the existing code set, and established policy. The applicant asserted its belief that a single device category for the two RDN technologies is consistent with the considerations put forth by CMS in the proposed rule and consistent with past precedent, whereas the creation of two categories may conflict with these policy standards. Another commenter stated a preference for a simple single code to be used on claims when performing RDN, regardless of which device is chosen.
With regard to broader device category definitions, the Symplicity SpyralTM RDN System applicant asserted that RDN ablation catheters in the future may come with several combinations of modalities and cooling mechanisms, noting that there are other RDN devices currently in development, such as a radiofrequency RDN catheter with balloon cooling under study by Metavention, with an approved Investigational Device Exemption (IDE) in the United States. The applicant stated the belief that other manufacturers may eventually gain FDA authorization with various device structures and features employing either ultrasound or radiofrequency RDN, and thus believed that the specific category description proposed by the Paradise® Ultrasound RDN System applicant would establish a framework in which different iterations of RDN technology could be considered new by virtue of their brand-specific characteristics. The Symplicity SpyralTM RDN System applicant asserted that, in contrast, a broader device category definition for RDN technologies currently eligible for pass-through would provide flexibility for future RDN devices to be evaluated on their own merits and that, if those devices meet the newness and other criteria for pass-through payment status, a broad category code for RDN can be easily adapted to reflect the distinguishing characteristic that merits such recognition.
The Symplicity SpyralTM RDN System applicant noted that the RDN procedure used with either device is currently reported using the same energy modality-agnostic Category III CPT® codes. This applicant further opined that the broader physician community views RDN as fundamentally a single procedure with multiple technologies ( printed page 94173) available in the same class.[70 71 72 73] Specifically, the applicant commented that in a Society for Cardiovascular Angiography & Interventions (SCAI) expert consensus statement published in August 2023, the operator training and experience recommended for the RDN procedure is the same, regardless of which technology is used, with the only device-specific training noted in the statement being operator familiarity with the functionality of each technology's generator and knowledge regarding balloon sizing with ultrasound RDN,[74] while the European Society of Cardiology/European Society of Hypertension (ESC/ESH) Guidelines refer only to RDN as a class and do not distinguish between radiofrequency and ultrasound technologies.[75 76] A commenter stated that from a physician work perspective, the two systems are similar in time and technique. The applicant further asserted that this issue was addressed at the September 2023 American Medical Association CPT® Editorial Panel meeting, where a request for a CPT® code for an energy modality-specific ultrasound RDN procedure was ultimately rejected by the Panel.
Response: As commenters stated, we have established precedent for creating both broad and distinct device categories for similar technologies. In fact, there have been many scenarios is which we have established distinct device categories to differentiate between various neurostimulators, electrophysiology catheters, and ablation modalities to ensure that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect due as required by § 419.66(c)(1). We agree with the SymplicityTM Spryal RDN System applicant that the establishment of device categories requires a nuanced approach, and that CMS must assess each situation on a case-by-case basis in accordance with established policy. We believe that we have followed not only our established policy, but our precedent in our evaluation of the device category code determination for the two nominated RDN devices. We agree with commenters that this situation with respect to the Paradise® Ultrasound RDN System and the SymplicityTM Spryal RDN System is similar to the examples presented.
The SymplicityTM Spryal RDN System applicant noted that manufacturers may gain FDA authorization for other RDN devices with various device characteristics employing either ultrasound or radiofrequency RDN and establishing device category codes that are device characteristic specific, like the code descriptor proposed by the Paradise® Ultrasound RDN System applicant. This, according to the SymplicityTM Spryal RDN System applicant, would establish a framework in which different iterations of RDN technology could be considered new by virtue of their brand-specific characteristics. We acknowledge that concern and agree that the device category codes established for the nominated devices should not typically be differentiated by the specific device characteristics, rather, consistent with the options in the proposed rule, we believe that in this instance the device pass-through payment device categories should be modality-specific.
With respect to a comment on the September 2023 AMA CPT® Editorial Panel meeting, where the SymplicityTM Spryal RDN System applicant stated that a request for a CPT® code specific to an energy modality ultrasound RDN procedure was ultimately rejected, as of the date this final rule with comment period is issued, we are not aware of any decision made by the AMA CPT® Editorial Panel regarding this issue.
Comment: The Paradise® Ultrasound RDN System applicant and many commenters also stated creating two categories may facilitate tracking modality-specific efficacy data and shared their belief that having the ability to identify and track which RDN device modality Medicare patients receive via claims data may be beneficial for CMS in the future.
The Symplicity SpyralTM RDN System applicant stated that tracking efficacy data for the two devices is beyond the fundamental purpose of establishing device categories and should not be a primary consideration.
Response: In response to comments on tracking of long-term efficacy data for the nominated devices, while we note that collecting long-term efficacy data is always a priority of CMS and other interested parties, we agree with the Symplicity SpyralTM RDN System applicant that we do not believe the tracking of long-term efficacy is a fundamental purpose of establishing ( printed page 94174) device category codes, as such, we did not consider this topic in our final decision.
We thank the commenters for their feedback and took into account all of the points raised in our final decision.
Comment: The applicant for the Symplicity SpyralTM RDN System, Medtronic, submitted another application for its device, PulseSelectTM, for transitional pass-through payment during CY 2025. The proposed rule stated that PulseSelectTM is used to achieve catheter ablation to treat atrial fibrillation, and thus, could be appropriately described by C1733. The Symplicity SpyralTM RDN System applicant stated that the PulseSelectTM application involved the interpretation of an existing device category description, but the applicant stated CMS's conclusion for that application is consistent with the logic for establishing a single device category to describe the two RDN devices. Following the publication of the proposed rule, the applicant withdrew its application for transitional pass-through payment for PulseSelectTM.
Response: With respect to the application for the PulseSelectTM included in the CY 2025 notice of proposed rulemaking, we note that we invited comment on whether the PulseSelectTM PFA System was described by C1733 (89 FR 59310 through 59311). We noted that based on the description the applicant provided, the PulseSelectTM PFA System is used to achieve catheter ablation to treat atrial fibrillation, and thus could be appropriately described by C1733. However, because the application was withdrawn prior to making a final determination on the device category criterion at § 419.66(c)(1), we are unable to address whether we would have ultimately determined that the PulseSelectTM PFA System was described by C1733.
We thank both applicants and the many commenters for their input. We note that we received overwhelming support in favor of establishing two device category codes to reflect the nominated devices. After review of the applications and the comments we received, we agree with majority of the commenters that we should establish two pass-through payment device categories. We believe that there are procedural differences and potential resource requirement differences between the two treatment modalities that warrant separate device categories. In addition, we believe that the circumstances presented by the nominated devices are sufficiently similar to the previous scenarios in which we established device category codes to differentiate similar devices with different modalities. However, we disagree with the suggestion that the device category codes should be specific to the physical device characteristics rather than modality-specific device categories. As such, consistent with our proposal and our final determination to approve both the Paradise® Ultrasound RDN System and the Symplicity SpyralTM RDN System for device pass-through payment status effective January 1, 2025, we are finalizing our decision to create two modality-specific pass-through payment device categories for RDN devices: radiofrequency and ultrasound.
(2) Traditional Device Pass-Through Applications
(a) Ambu® aScopeTM Gastro
Ambu Inc. submitted an application for a new device category for transitional pass- through payment status for the Ambu® aScopeTM Gastro for CY 2025. Per the applicant, the Ambu® aScopeTM Gastro is a sterile, single-use, flexible gastroscope intended to be used for: (1) endoscopic access to and examination of the upper gastrointestinal (GI) anatomy; and (2) upper GI endoscopy or esophagogastroduodenoscopy (EGD) to diagnose and treat problems in the upper GI tract, including dysphagia, gastroesophageal reflux disease, narrowing or blockages, esophageal varices, inflammation, ulcers, tumors, hiatal hernia, Celiac disease, Crohn's disease, and infections of the upper GI tract in adult patients.
According to the applicant, the Ambu® aScopeTM Gastro works with the Ambu® aBoxTM 2, a compatible, reusable displaying unit. The Ambu® aScopeTM Gastro endoscope is inserted into the upper GI anatomy airway through the mouth, while the Ambu® aBoxTM 2 is a non-sterile digital monitor intended to display live imaging data from Ambu visualization devices. The applicant is only seeking a new device category for transitional pass-through payment status for the Ambu® aScopeTM Gastro.
Please refer to the online application posting for the Ambu® aScopeTM Gastro, available at https://mearis.cms.gov/public/publications/device-ptp/DEP2305305795M, for additional detail describing this device and the disease treated by the device.
Comment: A few commenters expressed general support for the application for transitional pass-through payments for the Ambu® aScopeTM Gastro.
Response: We appreciate the commenters' input and recognize the commenters' support for the approval of the Ambu® aScopeTM Gastro for transitional pass-through payment.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), on February 3, 2022, the applicant received 510(k) clearance from FDA for the Ambu® aScopeTM Gastro, Ambu® aBoxTM 2, as a sterile, single-use, flexible gastroscope intended to be used for endoscopic access to and examination of the upper GI anatomy. The Ambu® aScopeTM Gastro is intended to provide visualization via a compatible Ambu displaying unit and to be used with endotherapy accessories and other ancillary equipment. We received the application for a new device category for transitional pass-through payment status for the Ambu® aScopeTM Gastro on May 30, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether the Ambu® aScopeTM Gastro meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the Ambu® aScopeTM Gastro meets the newness requirement for transitional pass-through payment.
Response: We appreciate the applicant's input. We received the application for a new device category for transitional pass-through payment status for the Ambu® aScopeTM Gastro on May 30, 2023, which is within 3 years of February 3, 2022, the date of FDA 510(k) approval to market for the Ambu® aScopeTM Gastro. As such we have concluded that the Ambu® aScopeTM Gastro meets the newness criterion.
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not indicate whether the Ambu® aScopeTM Gastro is integral to the service furnished. The applicant stated that the device was single-use and is intended to be used with one patient only. We noted that the Ambu® aScopeTM Gastro, based on the device description provided by the applicant and the evidence provided in support of the substantial clinical improvement as discussed in detail in the § 419.66(c)(2) analysis in this ( printed page 94175) application summary write-up, explicitly provides that the nominated device is intended to be used on one patient, for a single procedure and then disposed of. As such, we noted that our evaluation and final decision as it relates to this potential category of devices (gastroscopes) would be based on the understanding that devices included in this device category (gastroscopes) can only be used for a single procedure, on a single patient, and cannot be reprocessed. While the applicant did not explicitly state whether the device comes in contact with human tissue or is surgically inserted, per the device description, the Ambu® aScopeTM Gastro is a flexible gastroscope intended to be used for endoscopic access to and examination of the upper GI anatomy.
We invited public comment on whether the Ambu® aScopeTM Gastro meets the eligibility criterion at § 419.66(b)(3).
Comment: With respect to the eligibility criterion at § 419.66(b)(3), the applicant submitted a comment confirming that the Ambu® aScopeTM Gastro is used for one patient only, does come in contact with human tissue, and is surgically inserted during applicable procedures, and that the Ambu® aScopeTM Gastro is a device that is an integral part of the service furnished.
Response: We appreciate the applicant's input. We agree with the applicant that the Ambu® aScopeTM Gastro is an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and is surgically inserted. After consideration of the public comment received and based on our review of the application, we have determined that the Ambu® aScopeTM Gastro meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant indicated that the Ambu® aScopeTM Gastro is single-use equipment, not intended for use in multiple patients, for which depreciation and financing expenses are not recovered. The applicant explained that the Ambu® aScopeTM Gastro is purely an operating cost and is not subject to capitalization or a depreciation schedule.
We noted that the applicant stated in the application that the Ambu® aScopeTM Gastro is a supply furnished incident to a service rendered, as described, the Ambu® aScopeTM Gastro would be considered a supply or material furnished incident to a service and excluded from device pass-through payment eligibility under § 419.66(b)(4).
We invited public comment on whether the Ambu® aScopeTM Gastro meets the exclusion criterion at § 419.66(b)(4).
Comment: The applicant submitted a comment clarifying that the Ambu® aScopeTM Gastro is not a material or supply furnished incident to the service and meets the eligibility criterion at § 419.66(b)(4) because it must be purchased for each patient and is a device that is integral to the procedure. The applicant reiterated that as a single-use scope, it is not subject to capital equipment depreciation schedules.
Response: We appreciate the applicant's input. We agree with the applicant that the Ambu® aScopeTM Gastro is not a material or supply furnished incident to the service because it is single-use equipment, not intended for use in multiple patients, for which depreciation and financing expenses are not recovered. In addition, we agree that the Ambu® aScopeTM Gastro is not a material or supply furnished incident to the service. After consideration of the public comment received and our review of the application, we have determined that the Ambu® aScopeTM Gastro meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determine that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. Per the applicant, the Ambu® aScopeTM Gastro is a sterile, single-use, flexible, imaging/illumination gastroscope device that uses an integrated camera module and built-in dual light-emitting diode (LED) illumination to provide access to, illumination, and imaging of the upper GI anatomy for diagnostic and therapeutic purposes for a GI patient. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe the Ambu® aScopeTM Gastro. Per the applicant, the two device categories, C1747 (Endoscope, single-use ( i.e., disposable), urinary tract, imaging/illumination device (insertable)) and C1748 (Endoscope, single-use ( i.e., disposable), upper gastrointestinal tract (GI), imaging/illumination device, (insertable)), do not appropriately describe the Ambu® aScopeTM Gastro. Specifically, the applicant asserted that the urinary tract scopes described in C1747 are not indicated for use in the GI system and therefore, do not appropriately describe Ambu® aScopeTM Gastro. The applicant further asserted that while C1748 describes a single-use endoscopic device, C1748 is only appropriate for single-use duodenoscopes and endoscopic retrograde cholangiopancreatography (ERCP) services. While the long descriptor of C1748 describes disposable endoscopes with imaging and illumination capabilities intended for use in the upper GI and the applicant describes the Ambu® aScopeTM Gastro as a single-use, gastroscope with illumination and imaging intended for use in the upper GI anatomy, we noted that C1748 only describes single-use duodenoscopes and ERCP services. As such, the Ambu® aScopeTM Gastro is not described by C1748.
We did not identify an existing pass-through payment category that describes the Ambu® aScopeTM Gastro. We invited public comment on whether the Ambu® aScopeTM Gastro meets the device category criterion at § 419.66(c)(1).
Comment: The applicant and a commenter agreed with CMS' assessment that there are no existing pass-through payment categories that describe the Ambu® aScopeTM Gastro. The applicant submitted a comment with supporting documents reiterating that C1747 for single-use ureteroscopes and C1748 for single-use duodenoscopes do not describe the Ambu® aScopeTM Gastro because the Ambu® aScopeTM Gastro is a gastroscope and not a duodenoscope.[77] The applicant also submitted a July 2020 CMS Medicare Learning Network memorandum (MM11842) as a supporting document to confirm that C1748 is specific to ERCP procedures, which does not apply to the Ambu® ( printed page 94176) aScopeTM Gastro.[78] The other commenter indicated that endoscopic procedure codes, including those for EGD, were mostly created prior to the invention of single-use endoscopes. The commenter provided a detailed list of procedure codes for different types of endoscopes, including duodenoscopes and gastroscopes, to illustrate the differences in their applications. The commenter recommended the establishment of a new category to allow reporting specifically for gastroscopes, as their indications for use differ from other types of endoscopes like duodenoscopes.
However, a few other commenters stated that they believed that the existing code C1748 appropriately describes the Ambu® aScopeTM Gastro technology. One of the commenters stated that this code was established to cover all single-use scopes for upper GI procedures, regardless of the camera orientation (forward-facing or side-facing). Another commenter noted that C1748 was established in 2020, following an application from Boston Scientific for its EXALTTM Model D Single-Use Duodenoscope.[79] The commenter noted that while the EXALTTM Model D Single-Use Duodenoscope is used primarily in ERCP, CMS updated C1748 by assigning additional transnasal procedure codes to C1748 to include devices like the EvoEndo Model LE Single-Use Gastroscope.[80] The commenter stated that the description of C1748 reflects the use of an upper GI imaging/illumination device and is not specific only to duodenoscopes.
Response: We appreciate the applicant's and commenters' input. We agree with the applicant that C1747 does not describe the Ambu® aScopeTM Gastro because C1747 is intended to describe devices used in a different anatomical area (the urethra) of the patient. With regard to C1748, we continue to believe that C1748 does not describe the Ambu® aScopeTM Gastro because C1748 describes devices intended to perform ERCP services and limited transnasal endoscopy services. Unlike the devices described by C1748, according to the description provided by the applicant, the Ambu® aScopeTM Gastro is intended to be used for transoral endoscopy and is not indicated for transnasal endoscopy or ERCP services. Specifically, while we agree that C1748 describes upper GI imaging/illumination devices, which could describe the Ambu® aScopeTM Gastro, C1748 does not include the types of procedures that the Ambu® aScopeTM Gastro can perform, such as those described by HCPCS code 43205 (Esophagoscopy, flexible, transoral; with bandligation of esophageal varices). We acknowledge that a list of procedure codes associated with HCPCS code C1748 were updated in 2022 to include transnasal services and were assigned to APC 5301 (Level 1 Upper GI Procedures) and APC 5302 (Level 2 Upper GI Procedures), however, we do not believe that the addition of those codes to the codes for which C1748 can be reported describe the Ambu® aScopeTM Gastro because, as previously indicated, it is our understanding that the Ambu® aScopeTM Gastro is intended for transoral services only.[81] We note that the EvoEndo ® Model LE Single-use Gastroscope is indicated for use both as a transoral and transnasal gastroscope, while the Ambu® aScopeTM Gastro is only indicated for transoral use. While we believe the devices are comparable for the transoral functions of the devices for the purposes of evaluating substantial clinical improvement as discussed below, we do not believe that adding a subset of applicable procedure codes to C1748 expanded the device category code in such a way that it describes the Ambu® aScopeTM Gastro. After consideration of the public comments received and our review of the application, we agree that there is no previous or existing pass-through payment category that appropriately describes the Ambu® aScopeTM Gastro because no current category appropriately describes a single-use, transoral gastroscope with illumination and imaging intended for use in the upper GI anatomy. Therefore, we have determined that the Ambu® aScopeTM Gastro meets the device eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The applicant claimed that the Ambu® aScopeTM Gastro represents a substantial clinical improvement over existing technologies in the diagnosis and management of endoscopic procedures and examination within the upper GI anatomy. The applicant outlined the following areas in which it claimed the Ambu® aScopeTM Gastro would provide a substantial clinical improvement: (1) elimination of the risk of cross-contamination between patients and scopes, (2) elimination of the risk of cross-contamination for reusable gastroscopes, (3) elimination of the risk of resistant infections that originate from reusable gastroscopes, (4) avoidance of scope damage and debris after reprocessing, (5) avoidance of damaged and contaminated scopes from being used on patients, (6) elimination of the risk of patient-to-patient infections associated with contaminated scopes, and (7) avoidance of infection and death associated with reusable gastroscope contamination.
The applicant provided seven background articles about reusable GI endoscopes to support its claims. Table 122 summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for the Ambu® aScopeTM Gastro for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.
( printed page 94177) ( printed page 94178)After review of the information provided by the applicant, we stated we had the following concerns regarding whether the Ambu® aScopeTM Gastro meets the substantial clinical improvement criterion.
First, we noted that the applicant identified 11 other devices that it believed are most like the Ambu® aScopeTM Gastro: (1) Olympus GIF-HQ 190; (2) Olympus GIF-1TH190; (3) Olympus GIF-H190; (4) Olympus GIF-CP190N; (5) Fujifilm EG-760R; (6) Fujifilm EG-760CT; (7) Fujifilm EG-760Z; (8) Fujifilm EG-740N; (9) Pentax HD Video Gastroscope EG34 i10; (10) Pentax MagniView EG 2990Zi; and (11) Pentax G EYE. According to the applicant, these devices are used during the same specific procedure(s) and/or services with which the nominated device is used. The applicant stated that the nominated device's single-use feature is unique among the comparators because its single-use feature eliminates gastroscope reprocessing. The applicant also indicated that there are no HCPCS Level I and/or Level II code(s) used to identify these existing devices. While the evidence provided demonstrated that the Ambu® aScopeTM Gastro may be different than the other 11 closely related devices, it does not provide any comparative data that demonstrates that the Ambu® aScopeTM Gastro offers a substantial clinical improvement when compared to the other 11 devices.
Second, we noted that the nominated device was determined to be substantially equivalent to a predicate device: OLYMPUS EVIS EXERA II Gastrointestinal Videoscope GIF H180 (K100584). The FDA 510(k) summary indicated that both devices share the same technological characteristics such as insertion portion length, working channel diameter, direction of view and bending angles. We noted that the 510(k) summary indicated that, unlike the predicate device, the Ambu® aScopeTM Gastro is a sterile, single-use device and not intended to be reprocessed. Again, while this demonstrated that the Ambu® aScopeTM Gastro may be different than the predicate device, it is unclear whether this difference demonstrates substantial clinical improvement. No comparative data demonstrating that the Ambu® aScopeTM Gastro provides a substantial clinical improvement when compared to the Olympus EVIS EXERA II Gastrointestinal Videoscope GIF-H180 was provided. We stated we would be interested in additional information to demonstrate whether the nominated device demonstrates a substantial clinical benefit in comparison to other existing devices.
Further, the applicant indicated that while other single-use endoscopes are available, there are no known competitive devices on the market that are single-use, transoral, and marketed in the U.S. The applicant compared the Ambu® aScopeTM Gastro to the following two existing devices: (1) EndoFresh Single-Use Gastroscope; and (2) EvoEndo Model LE Single-Use Gastroscope. Specifically, the applicant noted that although EndoFresh Single-Use Gastroscope is FDA-cleared and a similar device that could also become eligible for transitional pass-through payment under the proposed additional category, it has no commercial activity in the U.S. According to the applicant, while EvoEndo® Model LE Single-Use Gastroscope is used during the same specific procedure(s) and/or services as the Ambu® aScopeTM Gastro, the Ambu® aScopeTM Gastro is different ( printed page 94179) from EvoEndo® Model LE Single-Use Gastroscope because the Ambu® aScopeTM Gastro is a transoral scope, not transnasal. The applicant also indicated that there are no HCPCS Level I and/or Level II code(s) used to identify EvoEndo Model LE Single-Use Gastroscope. However, we noted that EvoEndo® Model LE Single-Use Gastroscope is both transoral and transnasal, which is indicated on the EvoEndo, Inc.'s website and on its FDA 510(k) clearance letter. We also noted that the applicant did not compare the Ambu® aScopeTM Gastro to another single-use, FDA-cleared endoscope available on the market—EXALTTM Model D, Single-Use Duodenoscope—which we stated that we believe may be similar. We stated we were interested in additional information to demonstrate whether the nominated device demonstrates a substantial clinical improvement in comparison to similar single-use competitive devices such as the EvoEndo® Model LE Single-Use Gastroscope and the EXALTTM Model D, Single-Use Duodenoscope. In addition, we noted that the applicant's self-sponsored studies, which are background articles by Muscarella, L. F. (2022),[82] Muscarella, L.F. (2023),[83] and Ofstead, et. al. (2022),[84] lack direct comparison of the nominated device to other devices, and do not directly show any clinical improvement that results from the use of the nominated device compared to the use of other devices. In order to demonstrate substantial clinical improvement over currently available treatments, we consider supporting evidence, preferably published peer-reviewed clinical trials, that shows improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process compared to the standard of care. Additional supporting evidence, preferably published peer-reviewed clinical trials, that shows these improved clinical outcomes would help inform our assessment of whether the Ambu® aScopeTM Gastro demonstrates substantial clinical improvement over existing technologies.
Moreover, while the details provided in the application and all the articles submitted as evidence of substantial clinical improvement discuss potential adverse events from reusable gastroscope procedures, they do not appear to directly show any clinical improvement that results from the use of the Ambu® aScopeTM Gastro. Rather, the applicant provided evidence which seems to rely on indirect inferences from other sources of data. Specifically, the applicant included an FDA Manufacturer and User Facility Device Experience (MAUDE) report [85] which provides the details of multiple adverse event reports associated with the contamination or suspected contamination of reusable gastroscopes but does not directly show any clinical improvement that results from the use of the Ambu® aScopeTM Gastro.
While the applicant claimed that the Ambu® aScopeTM Gastro eliminates cross-contamination associated with reusable gastroscopes and eliminates the risk of infections that originate from reusable gastroscopes, we stated that we do not believe that we have sufficient information on the prevalence of infection to evaluate the applicant's substantial clinical improvement claims for the Ambu® aScopeTM Gastro. We noted the analyses on adverse event reports and the FDA MAUDE report appear to apply to flexible, reprocessed gastroscope or endoscopes, broadly, but not to disposable, single-use devices comparable to the nominated device.[86 87 88] Therefore, we questioned the direct relevance of these background articles to the nominated device and the applicant's substantial clinical improvement claims. Further, we noted that many of the applicant's substantial clinical improvement claims rely on an assumption that inadequate reprocessing of reusable endoscopes is positively correlated with heightened risk of infection. We noted that the applicant's self-sponsored analyses of FDA adverse event reports and studies and the FDA MAUDE report do not provide evidence on the prevalence of infection, establish a clear relationship between infection risk and reprocessing procedures, or substantiate that single-use disposable scopes, or the nominated device specifically, would be a substantial clinical improvement over currently available devices.[89 90 91 92] We stated that we would be interested in more information on the prevalence of infection due to incomplete/inadequate processing for gastroscopes in the U.S. and whether single-use gastroscopes reduce the infection rate in patients to identify the extent of the problem with existing technologies.
We invited public comment on whether the Ambu® aScopeTM Gastro meets the device category criterion at § 419.66(c)(2).
Comment: In response to our concerns that the Ambu® aScopeTM Gastro lacked comparative data demonstrating substantial clinical improvement compared to other available treatments, the applicant submitted a comment along with a multitude of articles and background documents.[93] The applicant submitted these documents as evidence to support the claim that the Ambu® aScopeTM Gastro is as a single-use device which offers substantial clinical improvements compared to reusable gastroscopes by eliminating infection risks and ensuring consistent functionality, which they assert are significant issues with reusable gastroscopes. The applicant stated that the Ambu® aScopeTM Gastro reduces infection risks and ensures consistent functionality. To support this claim, the applicant cited over 10,000 ( printed page 94180) Manufacturer and User Facility Device Experience (MAUDE) adverse event database reports for reusable scopes since May 2023 that include 1,500 related to contamination issues. The applicant noted that none of the MAUDE reports include adverse event reports for the Ambu® aScopeTM Gastro. In addition, the applicant referenced studies showing problems with reusable scopes, such as inadequate cleaning and performance degradation.[94 95 96 97 98] The applicant claimed that single-use scopes could be used in various settings without reprocessing delays, potentially expanding access to care. Although no specific studies on the Ambu® aScopeTM Gastro's impact on patient throughput exist, the applicant cited research on other single-use scopes indicating increased efficiency and reduced wait times, suggesting similar benefits.[99 100 101] In response to our concern about the lack of information identifying the extent of the problem with existing technologies, including the prevalence of infection due to incomplete and/or inadequate processing for gastroscopes in the U.S., and whether single-use gastroscopes reduce the infection rate in patients, the applicant stated that contamination issues are primarily relevant to reusable gastroscopes due to their complex reprocessing requirements, which are prone to human error and design flaws. The applicant referenced the studies and MAUDE reports again, reiterating that they show significant contamination rates and adverse events for reusable gastroscopes.[102 103] The applicant indicated that one study found a 19.98 percent contamination rate in reprocessed endoscopes. The applicant argued that single-use devices, like the Ambu® aScopeTM Gastro, eliminate cross-contamination risks, and noted there have been no MAUDE reports including the Ambu® aScopeTM Gastro.[104] The applicant anticipated that FDA might recommend single-use gastroscopes, citing severe infections linked to reusable ones, including deaths from Escherichia coli (E. coli) and Carbapenem-resistant Enterobacteriaceae (CRE). The applicant emphasized that single-use gastroscopes would particularly benefit immunosuppressed patients by eliminating these risks.
In response to our concern about the lack of comparative data demonstrating that the Ambu® aScopeTM Gastro provides substantial clinical improvement over the OLYMPUS EVIS EXERA II Gastrointestinal Videoscope GIF H180, the applicant submitted a study by Billy, et al. (2024), conducted at a Community Memorial Health Systems-Ventura, CA's endoscopic GI lab, that examined the difference in the degrees of retroflexion articulation achieved between an inventory of reusable diagnostic gastroscopes (RUDG: OLYMPUS GIF-HQ190 & H190) compared to that of two, single-use diagnostic gastroscopes (SUDG: Ambu® aScopeTM Gastro).[105] Per the applicant, the study demonstrated that the Ambu® aScopeTM Gastro has superior retroflexion compared to the OLYMPUS GIF-HQ190. The applicant believed that this is significant because articulation is crucial for the procedures these devices perform and reduced effectiveness in RUDGs can hinder optimal patient care.
However, one commenter stated that the applicant has not demonstrated substantial clinical improvement over existing technologies. The commenter stated that FDA clearance for the Ambu® aScopeTM Gastro is based on equivalence to the earlier generation Olympus EVIS Exera II model 180 series. The commenter believed that the applicant has not provided comparative clinical documentation to support its claim.
In response to our concern about the lack of information demonstrating substantial clinical improvement with the use of the Ambu® aScopeTM Gastro device compared to similar single-use competitive devices, such as the EXALTTM Model D, Single-Use Duodenoscope and the EvoEndo Model LE Single-Use Gastroscope, the applicant submitted a comment asserting that there are key differences between the Ambu® aScopeTM Gastro and the EXALTTM Model D Single-Use Duodenoscope and the EvoEndo Model LE Single-Use Gastroscope. The applicant stated that the Ambu® aScopeTM Gastro cannot be compared to the EXALTTM Model D because it is not indicated for ERCP procedures. The applicant noted that, compared to the EvoEndo Model LE Single-Use Gastroscope, the Ambu® aScopeTM Gastro has a larger working channel (2.8 mm vs. 2.0 mm), allowing for more endoscopic accessories and broader capabilities. The applicant also claimed that while the EvoEndo® Model LE Single-Use Gastroscope can perform some of the same procedures, it is limited compared to the Ambu® aScopeTM Gastro, which can perform 30 HCPCS procedures versus 10 for the EvoEndo® Model LE Single-Use Gastroscope.
A commenter agreed with CMS, noting that the EvoEndo® Model LE Single-Use Gastroscope is for both transoral and transnasal use, as stated on EvoEndo® Model LE Single-Use Gastroscope website and its FDA 510(k) clearance summary. The commenter highlighted similarities in FDA clearances between the Ambu® ( printed page 94181) aScopeTM Gastro and the EvoEndo® Model LE Single-Use Gastroscope and noted that the applicant has not shown substantial clinical improvement of the nominated device over this existing device.
To bolster its claims that single-use gastroscopes have improved outcomes compared to reusable gastroscopes, the applicant submitted background articles and other documents about the potential adverse events from reusable gastroscope procedures as well as from other reusable types of endoscopes for different anatomy ( e.g., bronchoscopy). Many commenters noted numerous challenges and risks associated with the reprocessing of reusable endoscopes, emphasizing the need for single-use alternatives like the Ambu® aScopeTM Gastro to improve patient safety. A few commenters pointed out that delays in reprocessing due to staffing shortages, union-related issues, and the need to transport scopes across hospitals are common. Several commenters stated that the cleaning process for endoscopes involves many complex steps, making the cleaning process prone to human error and creating difficulty in training staff effectively. One commenter stated that despite proper education, human error remains a significant barrier to successful reprocessing with observations of missed steps and shortcuts due to assumptions or mimicking others. A few commenters further explained that inadequate training and preparation, especially during after-hours procedures, contribute to poor cleaning practices and increased infection risks. Several commenters further noted that continuous cleaning without breaks due to staff shortages and high demand increases the likelihood of errors.
Many commenters identified specific reprocessing gaps, including blood dripping from reprocessed scopes, improper handling of Creutzfeldt-Jakob Disease (CJD) cases, non-compliance with cleaning guidelines, and failed competencies among reprocessing technicians. One commenter indicated that problems with automatic preprocessors, inconsistent reprocessing locations, and confusion over Automated Endoscope Reprocessors (AERs) filters add to the complexity and risk. One commenter stated that there is a positive correlation between inadequate reprocessing and increased infection risk. Specifically, the commenter asserted that single-use gastroscopes eliminate this risk, providing a higher standard of care. The commenter cited several FDA reports and peer-reviewed studies asserting that they link reusable gastroscopes to infections and outbreaks, thereby supporting the need for single-use alternatives. The commenter acknowledged that linking or associating a reusable gastroscope with an infection or outbreak does not confirm the gastroscope transmitted or otherwise caused the infection, as one or more other factors could be, in part or solely, responsible. The commenter suggested more data would be required to conclude more definitively that the endoscope caused an infection. The commenter also acknowledged that FDA's MAUDE database has limitations and that its housed adverse event reports may be incomplete, inaccurate, untimely, unverified, or biased.
The applicant and several commenters advocated for the adoption of disposable gastroscopes, such as the Ambu® aScopeTM Gastro in healthcare settings, emphasizing their benefits in terms of efficiency, patient safety, and cost-effectiveness. Several commenters stated that single-use gastroscopes are always ready for use, eliminating delays caused by the need to reprocess reusable scopes because they can be set up quickly, facilitating continuous care, especially in critical situations. Multiple commenters stated that single-use gastroscopes eliminate the risk of cross-infection associated with reusable scopes, which can remain contaminated even after reprocessing. One commenter indicated that sterile single-use devices lower the infection risk for patients with compromised immune systems. Another commenter provided a scenario in which single-use gastroscopes are crucial for patients with diseases like CJD, where reusable scopes might need to be destroyed. Several commenters pointed out that single-use gastroscopes remove the financial and logistical burdens associated with reprocessing, sampling, culturing, and infection surveillance. One commenter considered the single-use gastroscopes serve as a bridge when reusable scopes are out for repair, ensuring uninterrupted patient care. Several commenters stated that the single-use scopes provide consistent performance as they are used only once, avoiding issues related to scope degradation. One commenter believed that single-use scopes can incorporate rapid design improvements, enhancing their clinical capabilities.
Response: We appreciate the applicant's and commenters' input and insights on efficiency, patient safety, and cost-effectiveness. However, we wish to reiterate the statement we made in CY 2024 (88 FR 81736) where we encouraged applicants to submit all relevant supporting evidence with their device pass-through application to allow us to adequately evaluate and include the data in the notice of proposed rulemaking. Further, we note that the information submitted by the applicant in support of its CY 2025 application during the comment period did not appear to provide information that demonstrates that the use of the Ambu® aScopeTM Gastro improves clinical outcomes when compared to the use of similar reusable or single-use gastroscope devices. In order to evaluate substantial clinical improvement over currently available treatments to meet the transitional pass-through payment criterion at § 419.66(c)(2), we consider supporting evidence, preferably published peer-reviewed clinical trials, that demonstrates improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process comparing the nominated device to the standard of care.
Based on the information provided and our review, we note that the Ambu® aScopeTM Gastro and the EvoEndo Model LE Single Use Gastroscope do perform at least 10 of the same procedures. We remain concerned with the lack of evidence comparing these devices' respective clinical outcomes. We further note that the applicant referenced the study by Billy, et al (2024), which compared the nominated device against OLYMPUS EVIS EXERA III GIF-HQ190, not the predicate device, OLYMPUS EVIS EXERA II Gastrointestinal Videoscope GIF H180 for retroflexion articulation (not clinical outcome). We also note that the Billy, et al. (2024) article is not a peer-reviewed publication and does not provide evidence of the Ambu® aScopeTM Gastro's clinical improvement compared to other single-use gastroscopes. In addition to this study, the applicant submitted numerous background articles; however, these articles did not include evidence that demonstrated or supported the Ambu® aScopeTM Gastro's substantial clinical improvement compared to other single-use gastroscopes. We also have concerns regarding the submitted articles' relevance and appropriateness. For example, we have concerns about the validity of the findings in the Haislip, et al. (2023) poster submitted as evidence by the applicant because it lacks a full description of the methods, and it is unknown where it was presented and the extent to which it was peer reviewed for the validity of its findings. In the Tomlinson publication, which primarily ( printed page 94182) discusses the complexity of Instructions For Use (IFUs) for noncritical devices but not semi-critical devices like endoscopes that require high-level disinfection, we note that the problematic IFUs mentioned pertain to physical therapy and ophthalmology, not endoscopy.[106] As such, we question the relevancy of the submitted materials and do not believe that the information provided supported the applicants claims of substantial clinical improvement.
Based on our review of the extensive MAUDE reports provided by one of the commenters, we found that the vast majority did not conclusively link reusable gastroscopes to infections and those with proven or highly likely cross-contamination were few in number. We also note that the references provided by the commenter were about bronchoscopes and duodenoscopes, which are different anatomical areas that may be more vulnerable to infection.
Comment: The applicant and a commenter urged CMS to follow precedents set with Uretero1, aScope5 Broncho HD, and other single-use endoscopes and to approve the Ambu® aScopeTM Gastro for transitional pass-through payment status. The applicant argued that the existing evidence is consistent with the level of evidence provided for previously approved single-use endoscopes like Uretero1 and aScope 5 Broncho HD.
Response: We appreciate the applicant's and commenter's input. We evaluate documentation submitted for each application as it applies to that specific device. Due to inherent differences in the devices themselves and/or the supporting documentation submitted, we may have different concerns. In addition, we are not precluded from evaluating and expressing concerns regarding documentation submitted with an application because we have evaluated the document as part of a previous application. We approved the Uretero1 and the aScope5 Broncho HD applications for device pass-through status because the applicants submitted documentation of studies that directly demonstrated the nominated device's improved clinical outcomes compared to other devices. We do not believe that the applicant has submitted documentation that demonstrates a substantial clinical improvement with use of the Ambu® aScopeTM Gastro compared to the use of other comparable devices.
Comment: In regard to our concern about using other sources of data and relying on indirect inferences as evidence, the applicant commented that we should call our attention to the volumes of literature concerning reprocessing issues. The applicant shared MAUDE reports to show that reusable devices have had outbreaks, while disposable gastroscopes, like the Ambu® aScopeTM Gastro, have not. The applicant also noted a pattern of FDA advisories and subsequent CMS approvals for single-use endoscopes (single-use aScope 5 Broncho HD) suggesting that single-use gastroscopes offer substantial clinical improvements. Additionally, the applicant argued that single-use gastroscopes enhance availability and access to care, especially during equipment failures or staffing shortages.
In response to our concern about lack of studies that directly compare the Ambu® aScopeTM Gastro device to other single-use devices, the applicant argued that due to the nature and relative newness of single-use endoscopes, extensive data is not yet available on the Ambu® aScopeTM Gastro device specifically. The applicant emphasized reprocessing issues, submitting reports documenting challenges and risks, including bacterial transmission, even with meticulous adherence to instructions. The applicant further argued that conducting a full-scale randomized trial comparing single-use and reusable gastroscopes would be time-prohibitive. The applicant again cited the study by Billy, et al (2024) showing superior retroflexion of single-use devices over reusable ones. The applicant emphasized the sterile nature of the Ambu® aScopeTM Gastro, eliminating infection risks associated with reprocessing reusable devices. The applicant reiterated that single-use endoscopes save time, improve morale, and reduce reprocessing risks, leading to increased patient throughput and reduced wait times, demonstrating clinical improvements over reusable options.
Response: We thank the applicant for its input. However, we continue to have concerns about the studies and evidence submitted, as most of these are background articles that do not directly assess, evaluate, or review the Ambu® aScopeTM Gastro. When there are currently available treatment options for a patient population (as is the case for the Ambu® aScopeTM Gastro), substantial clinical improvement is demonstrated when the candidate device demonstrates significantly improved clinical outcomes compared to the currently available treatments.[107] In this context, the submitted evidence in support of the Ambu® aScopeTM Gastro's substantial clinical improvement must demonstrate that Ambu® aScopeTM Gastro results in substantial clinical improvement when compared to available reusable and single-use devices for the treatment of the patient population. Inferences that the device may improve clinical outcomes because it may obviate complications associated with other available treatments are insufficient to demonstrate substantial clinical improvement. Therefore, the inferences derived from submitted evidence, including analysis of adverse event reports and MAUDE reports, do not establish substantial clinical improvement. Specifically, we are concerned that the details provided in the application and the documents submitted as evidence of substantial clinical improvement are background documents that discuss potential adverse events from reusable gastroscope procedures and rely on indirect inferences from other sources of data. In addition, while we acknowledge that we have considered FDA advisories in the evaluation and subsequent CMS approval for single-use endoscopes in the past, we note that the applicant did not submit an FDA safety communication for reusable gastroscope, rather the applicant speculated that FDA will issue a safety communication similar to the safety communications they issued on bronchoscopes, ureteroscope, and duodenoscopes. We are not aware of an FDA safety communication for gastroscope at this time. We do not believe that the documents provided by the applicant and commenters demonstrate any clinical improvements that result from the use of the Ambu® aScopeTM Gastro when compared to available reusable or single-use devices that are similar to the Ambu® aScopeTM Gastro. As such, we continue to have the concerns articulated in the CY 2025 OPPS/ASC proposed rule and discussed above.
We appreciate the commenters' input but remain concerned that there has not been an adequate comparison of clinical outcomes between the Ambu® aScopeTM Gastro and other available reuseable and/or single-use devices used for similar indications. Because of the reasons discussed above, we do not believe that the Ambu® aScopeTM ( printed page 94183) Gastro represents a substantial clinical improvement relative to existing therapies currently available. Therefore, we have determined that the Ambu® aScopeTM Gastro does not meet the substantial clinical improvement criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that the Ambu® aScopeTM Gastro would be reported with HCPCS codes shown in Table 123.
( printed page 94184) ( printed page 94185)To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2023 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5301, which had a CY 2023 payment rate of $825.51 at the time the application was received. HCPCS code 43239 in APC 5301 had a CY 2023 device offset amount of $2.64 at the time the application was received.[108] According to the applicant, the cost of the Ambu® aScopeTM Gastro is $799.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $799.00 for the Ambu® aScopeTM Gastro is 96.79 percent of the applicable APC payment amount for the service related to the category of devices of $825.51 (($799.00/$825.51) × 100 = 96.79 percent). Therefore, we stated that we believe the Ambu® aScopeTM Gastro meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $799.00 for the Ambu® aScopeTM Gastro is 30,265.15 percent of the cost of the device-related portion of the APC payment amount for the related service of $2.64 (($799.00/$2.64) × 100 = 30,265.15 percent). Therefore, we stated that we believe the Ambu® aScopeTM Gastro meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $799.00 for the Ambu® aScopeTM Gastro and the portion of the APC payment amount for the device of $2.64 is 96.47 percent of the APC payment amount for the related service of $825.51 ((($799.00−$2.64)/$825.51) × 100 = 96.47 percent). Therefore, we stated that we believe the Ambu® aScopeTM Gastro meets the third cost significance requirement.
We invited public comment on whether the Ambu® aScopeTM Gastro meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: With respect to cost significance criteria, the applicant reiterated that the Ambu® aScopeTM Gastro meets all three of the cost significance criteria.
Response: We appreciate the applicant's input. After consideration of ( printed page 94186) the public comment received and our findings from the first, second, and third cost significance tests, we agree that the Ambu® aScopeTM Gastro meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments received and our review of the device pass-through application, we are not approving the Ambu® aScopeTM Gastro for transitional pass-through payment status in CY 2024 because the product does not meet the substantial clinical improvement criterion at § 419.66(c)(2).
(b) OMEZA Wound Care Matrix (OCMTM)
OMEZA LLC submitted an application for a new device category for transitional pass-through payment status for OCMTM for CY 2025. According to the applicant, OCMTM is an amorphous, solid, malleable sheet comprised of hydrolyzed fish peptides infused with cod liver oil which acts as an anhydrous skin protectant. Per the applicant, OCMTM is indicated for the management of wounds. The applicant asserted that, when applied to a clean wound surface, OCMTM is naturally incorporated into the wound over time. Per the applicant, OCMTM 's cold water fish peptides provide building blocks for tissue regeneration and cell signaling molecules stimulate tissue growth. Additionally, OCMTM 's matrix-like device also contains active pharmaceutical ingredient(s) (API) and nutrients that continuously reduce biofilm impact, reduce inflammation, increase tissue proliferation, and support remodeling of tissue.
Please refer to the online application posting for the OCMTM, available at https://mearis.cms.gov/public/publications/device-ptp/DEP2403016HWP6, for additional detail describing the device and the disease treated by the device.
Comment: Several commenters with experience treating patients with OCMTM stated their belief that it meets all the criteria outlined in the rule and should be granted pass-through status so that patients with stalled or non-healing wounds can be treated before the wound increases in size or becomes infected.
Response: As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), on September 1, 2021, the applicant received 510(k) clearance from FDA for OCMTM as a device to be used for the management of wounds including: (1) partial and full-thickness wounds, (2) pressure ulcers, (3) venous ulcers, (4) diabetic ulcers, (5) chronic vascular ulcers, (6) tunneled/undermined wounds, (7) surgical wounds (donor sites/grafts, post-Moh's surgery, post-laser surgery, podiatric, wound dehiscence), (8) trauma wounds (abrasions, lacerations, superficial partial thickness burns, skin tears), and (9) draining wounds. We received the application for a new device category for transitional pass-through payment status for OCMTM on March 1, 2024, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether OCMTM meets the newness criterion at § 419.66(b)(1).
Comment: The applicant thanked CMS for agreeing that OCMTM meets the newness criteria.
Response: We appreciate the applicant's comment. We received the application for a new device category for transitional pass-through payment status for OCMTM on March 1, 2024, which is within 3 years of September 1, 2021, the date of FDA 510(k) clearance. Based on our review of the application, we have determined that OCMTM meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not indicate whether OCMTM is integral to the service furnished. In the CY 2014 final rule with comment period (78 FR 75005), we stated that we have interpreted the term “integral” to mean that the device is necessary to furnish or deliver the primary procedure with which it is used. For example, a pacemaker is integral to the procedure of implantation of a pacemaker. We noted that OCMTM does not appear to be necessary to furnish or deliver the primary procedure with which it is used, specifically debridement. Rather, we noted the use of OCMTM following the debridement procedure, including the duration of treatment and the reapplication frequency, seems to be based entirely on provider discretion. As such, we stated that we do not believe that OCMTM is integral to the service furnished as required by § 419.66(b)(3). The applicant stated that OCMTM is classified for one-time use and is designed for intimate contact with both regular and irregular wound beds, and as such, it is applied in or on a wound.
We invited public comment on whether OCMTM meets the eligibility criterion at § 419.66(b)(3).
Comment: In response to our concern that OCMTM may not meet the eligibility criteria under § 419.66(b)(3), the applicant asserted that OCMTM is integral to services furnished in the outpatient setting for non-healing wounds. Specifically, the applicant stated that OCMTM is integral to both the active wound care management furnished for HCPCS codes 97597, 97598, 97602, and 97605-97608, and surgical debridement services furnished under HCPCS codes 11000-11012 and 11042-11047. The applicant added that for hard-to-heal, non-healing wounds that fail to respond to four weeks of standard wound care (including debridement), adjunctive application of advanced wound therapies, like OCMTM, is the next recommended wound management step. The applicant stated that OCMTM is indicated for application following initial standard of care failure and may replace the need for negative-pressure wound therapy, placental membranes, bioengineered skin substitutes, several acellular matrices, autologous fibrin, and leukocyte platelet patches, which are all typically used in the hospital outpatient setting. The applicant further explained that OCMTM is not a skin protectant, but instead, a bioactive matrix that conforms to the wounds allowing for more complete coverage of the wound in a safe and effective manner; thus, it supplements the missing necessary components for the natural healing to occur. The applicant asserted that OCMTM performs a similar function to certain collagen-based implantable devices used in internal surgeries to promote healing by improving the structural integrity of joints, soft tissues, and nerves. Per the applicant, debridement alone is insufficient for managing refractory wounds, necessitating adjunctive advanced wound therapies, such as OCMTM . The applicant, therefore, stated that classification of OCMTM as specifically an incident to supply is inconsistent with its necessitated clinical use. Several non-applicant commenters agreed with the applicant that OCMTM does not always require debridement, and therefore, the device is integral to the service performed.
As support for the integral function of OCMTM , the applicant quoted the CY 2014 OPPS/ASC final rule [109] (78 FR ( printed page 94187) 75005) statement that skin substitutes are integral to, dependent on, and supportive to the surgical procedures in which they are used. Specifically, the applicant asserted that while OCMTM is not a skin substitute, it has a distinct composition and mechanism, as well as a higher degree of regulatory oversight, exemplified by its FDA 510(k) clearance for wound management. The applicant stated that, like skin substitutes, OCMTM meets the integral to service criterion for wound management, as outlined in the CY 2014 OPPS final rule, given that OCMTM not only matches but also exceeds the clinical utility of skin substitutes as an advanced wound therapy. The applicant concluded by asserting that OCMTM 's indicated use, per its FDA 510(k) clearance, supports the device's classification as integral to the services furnished.
Response: We appreciate the applicant's input. Based on the additional information provided in the comments, we agree that OCMTM is integral to advanced wound therapy because it is used for active wound care management after the patient has received standard of care services with or without debridement including in the management of refractory wounds. Specifically, we agree that OCMTM is integral to active wound care management furnished for HCPCS codes 97597, 97598, 97602, and 97605-97608 and surgical debridement services furnished under HCPCS codes 11000-11012 and 11042-11047. Additionally, we agree with the applicant that OCMTM is used for one patient only, comes in contact with human tissue, and is applied in or on a wound. After consideration of the public comments received and our review of the application, we have determined that OCMTM meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criteria at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not indicate whether OCMTM is equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or if OCMTM is a supply or material furnished incident to a service. However, in the CY 2014 final rule, we described skin substitutes as a type of supply used in a surgical procedure (78 FR 74929 through 74930). As explained in the CY 2014 final rule, supplies are a large category of items that typically are either for single-patient use or have a shorter life span in use than equipment. Supplies can be anything that is not equipment and include not only minor, inexpensive, or commodity-type items but also include a wide range of products used in the hospital outpatient setting, including certain implantable medical devices, which we have considered supplies since the inception of the OPPS (78 FR 74929 through 74930). We clarified that we believe skin substitutes are supplies used in a surgical procedure because, as a part of a surgical repair procedure, they reinforce and aid the healing of tissue like implantable biologicals, but with skin substitutes, the tissue is skin instead of internal connective tissues (78 FR 74931). As such, we questioned whether OCMTM would be considered a supply, and as such it would be excluded from device pass-through payments under § 419.66(b)(4).
We invited public comment on whether OCMTM meets the exclusion criterion at § 419.66(b)(4).
Comment: In response to our concern that OCMTM would be excluded under § 419.66(b)(4), the applicant asserted that OCMTM is not an item for which depreciation and financing expenses are recovered and that, like an implantable biologic or medical device, is used as an integral and necessary supply in a surgical procedure. The applicant stated that OCMTM does not fit the classification of an incident to supply, defined as a material or supply furnished incident to a service because it aids in the management of wounds by supplementing the missing necessary components for the natural function of healing to occur; and is necessary to the wound care procedure itself when debridement alone is insufficient. The applicant asserted that classification of OCMTM as an incident to supply is not consistent with its necessitated clinical use. The applicant asserted that given that OCMTM exceeds the clinical utility of skin substitutes as an advanced wound therapy and that skin substitutes as outlined in the CY 2014 OPPS final rule meet the integral to service criteria for wound management, the applicant believes that OCMTM does not meet the disqualifying criteria of being an incidental supply under § 419.66(b)(4).
Response: We appreciate the clarification from the applicant. Based on the additional information provided in the comments, we agree with the applicant that OCMTM is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or are a material or supply furnished incident to a service. Specifically, we believe that OCMTM is necessary to the wound care procedure itself when debridement alone is insufficient and is, therefore, not a material or supply furnished incident to a service. We note that while the applicant did not furnish further detail to explain why OCMTM is not a depreciable asset, we believe that the applicant provided enough additional information about OCMTM to conclude that it is like other cellular and tissue-based products for wound management, and therefore, OCMTM is not an asset subject to depreciation or used in a normal or standby capacity by the provider per the Medicare Reimbursement Manual (CMS Pub. 15-1). After consideration of the public comment received and our review of the application, we have determined that OCMTM meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. The applicant asserted that OCMTM is indicated for the comprehensive treatment of advanced wounds and provides continuous delivery of pharmaceutical grade products through an amorphous, anhydrous solid, which reduces biofilm while simultaneously promoting tissue proliferation and remodeling. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe OCMTM.
We did not identify an existing pass-through payment category that describes OCMTM . We invited public comment on whether OCMTM meets the device category criterion at § 419.66(c)(1).
We did not receive any comments regarding whether OCMTM meets the eligibility requirements at § 419.66(c)(1). Based on our review of the application, we continue to believe there is no ( printed page 94188) existing category or category previously in effect that appropriately describes OCMTM . Therefore, we have determined that OCMTM meets the device category eligibility criterion at § 419.66(c)(1).
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The applicant claimed that OCMTM represents a substantial clinical improvement over existing technologies in the treatment of hard to heal or chronic wounds which require advanced wound care procedures such as venous leg ulcers, diabetic foot ulcers, pressure ulcers, and wound dehiscence where proper wound preparation, product application, and proper secondary dressings are a requirement. Specifically, the applicant claimed that OCMTM demonstrates: (1) superior clinical outcomes and healing for Diabetic Foot Ulcers (DFU) compared to standard of care; (2) faster healing rates than standard of care for Venous Leg Ulcers (VLUs); (3) superior clinical outcomes for patients who could not qualify for clinical trials due to comorbidities; (4) improved results when compared to results with standard of care for patients who failed prior treatment; (5) in vitro/in vivo antimicrobial properties and patient safety; and (6) improved patient safety.
The applicant provided the following clinical trial data and case studies to support these claims: (1) two randomized controlled trials (a single-site trial of patients with DFUs to evaluate percent area reduction, and a randomized, multicenter, open label study for a patient group with VLUs); (2) two real-world trials comprised of two separate case studies of patients receiving follow-up care at two different wound treatment centers; (3) one in vitro study; (4) one in vivo porcine study; and (5) one consumer research study assessing the safety of OCMTM using the skin prick method. Table 124 summarizes the applicant's assertions regarding the substantial clinical improvement criterion. We noted that there are multiple variations in poster presentations for the same study; these posters are identified by study number and presentation number in parentheses. Please see the online posting for OCMTM for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.
( printed page 94189) ( printed page 94190) ( printed page 94191)After review of the information provided by the applicant, we stated we had the following concerns regarding whether the applicant presents clinical data to suggest that OCMTM provides a substantial clinical improvement over other similar skin protectant and wound healing products to meet the criterion at § 419.66(c)(2)(i). Based on the evidence submitted in the application, we noted the following concerns: (1) lack of direct comparison between the nominated device and the predicate or reference devices for skin substitutes, particularly with respect to treatment of deep or persistent chronic wounds in people with DFU and VLU; (2) reliance on non-peer-reviewed studies, such as unpublished abstracts or conference posters, the results of which are only presented in a final data table; and (3) reliance on studies which were sponsored by the device manufacturer rather than independent research. We noted that the unpublished abstract for OCMTM lacked a detailed discussion of study limitations, patient population, and assurances that studies have been thoroughly peer-reviewed and free from implicit bias. Furthermore, the abstract does not state if or how standard of care treatment was administered within the same time period to control groups, and therefore, we stated we were unsure if there was a direct comparison between OCMTM and its predicate or reference ( printed page 94192) devices. Furthermore, we noted that the two randomized controlled trials [110 111] and two real world studies [112 113] submitted by the applicant to support its claims had relatively small sample sizes, some investigating only two patients total, which potentially limits the statistical significance of the results.
We noted that the applicant did not provide a comparison of OCMTM to other devices it identified are closely related or similar to OCMTM . Specifically, in the FDA authorization letter dated September 1, 2021, FDA identified one predicate device, SweetBio Apis (K182725), and three reference devices, INTEGRATM Flowable Wound Matrix (K072113), Kerecis MariGen Wound Dressing (K132343), and Southwest Technologies Stimulen Collagen (K030774) to which OCMTM may be compared. We noted that we did not approve transitional device pass-through payment for Kerecis MariGen Wound Dressing (K132343) for CY 2018 after determining that the clinical data provided by the applicant did not support the claim that Kerecis Omega3 Wound Dressing provides a substantial clinical improvement over other similar skin substitute products (82 FR 59330 through 59332). The FDA authorization letter noted that OCMTM and the predicate device SweetBio Apis have similar indications and the same intended use, namely, to manage wounds by providing an animal-derived collagen product that is biodegradable and incorporates into the surrounding tissue during the body's natural wound healing processes. Both products supplement the collagen constituent with additional biocompatible materials to achieve a final product that covers and protects the wound, assists in managing wound exudate, and maintains a moist wound environment. Further, the substantial equivalence table included in the FDA authorization letter indicated that OCMTM raised no new questions of safety or effectiveness when compared to the predicate and reference devices.
In the first claim, the applicant asserted OCMTM has superior clinical outcomes and healing for DFU compared to the standard of care. Based on the evidence submitted by the applicant, we noted the following concerns: (1) lack of a direct comparison to the predicate or reference devices in the two randomized controlled trials and the two real world clinical studies; (2) reliance on unpublished studies; (3) reliance on manufacturer sponsored studies; and (4) small sample sizes. First, Simman, et al. (2023) describes the results of a single-site trial in patients with DFUs to evaluate percent area reduction in wound healing. The stated goal of this study was to demonstrate that a combination therapy, using OCMTM plus standard of care treatment, moves chronic DFUs from a stalled state to a healing state in a 4-week period. The study enrolled 25 patients, five of whom did not complete the study, and one of whom died during the study from comorbidities related to their underlying condition. Study group DFUs were managed with combination therapy from 4-12 weeks, and control group DFUs (comprised of six total study participants) were managed with standard of care treatment involving cleaning and debridement only.
Interim analyses presented as a poster (Bell, et al., 2023) as evidence to support the first claim was limited to 12 total study participants. The interim analysis concluded that healing rates showed an average of 63 percent area reduction for the remaining participants at 4 weeks following standard of care treatment, and an average of 91 percent area reduction at 12 weeks following the treatment in patients with DFUs managed with the combination therapy. The interim analysis study further showed that one patient with a 12-week percent area reduction of 73 percent continued to improve through week 14 while three patients' wounds had not healed at the time of analysis for those receiving combination therapy.
In the final results presented in Simman, et al. (2023), the average 4-week percent area reduction was 60 percent, with three patients experiencing 100 percent closure with combination therapy. At 12 weeks, the median wound size was 0.0 cm2 (range, 0-2.59), and the average percent area reduction was 93 percent, with five additional patients experiencing 100 percent closure with combination therapy. The average 4- and 12-week percent area reductions with standard of care alone were 42 percent and 45 percent, respectively. According to the final analysis study abstract, every wound treated with OCMTM combination therapy was reduced by more than 70 percent in 12 weeks; and all wounds previously failed treatments.
We noted that the Simman, et al. (2023) study abstract and the interim analysis do not provide any direct comparison to standard of care treatment with another collagen-based wound matrix or device that is otherwise similar to the indications for use of OCMTM in nonhealing wounds. In addition, we noted that it is unclear if any of the control group patients received collagen-based treatments including the predicate or reference devices to draw comparisons to collagen-based skin products that perform similarly to OCMTM . While we recognized, given the number of skin substitute products on the U.S. market, it is not possible to compare OCMTM to each product, we stated that we believe studies comparing the product against other powder, liquid, or gel skin substitute products could provide more evidence demonstrating the clinical superiority of OCMTM . In addition to the lack of comparison to other collagen-based wound matrix devices, we noted that the standard of care treatment in this study was limited to cleaning and debridement, which, based on the applicant's description for methods for administering OCMTM, is a step prior to administering OCMTM.
In reference to the applicant's statements that debridement combined with application of OCMTM is more effective in the removal of biofilm compared to the standard of care of debridement alone, we noted that neither the abstract nor the interim analysis for this study analyzed results on removal or prevention of biofilm in isolation from the overall metric on wound percent area reduction. We noted that FDA recommends sharp debridement alone as an effective method to remove the biofilm and necrotic tissue in a chronic wound (Bettle, et al., 2023). We questioned whether the results describing the average percent area reduction in wounds transitioning from a nonhealing state to a healing state are sufficient to show substantial clinical improvement in removal or prevention of biofilm. We further questioned whether the results in percent area reduction can be attributed to debridement combined with application of OCMTM as opposed to debridement alone because it is unclear if debridement was performed on all participants in the retrospective control group. ( printed page 94193)
Furthermore, we noted that only the effects of historic standard of care treatments administered prior to the start of the study to patients in the control group were included for analysis. While one of the selection criteria for study participants was having failed prior treatment, neither the abstract nor interim analysis discussed how other variables, such as age and comorbidities, may have contributed to treatment failure, or which specific treatments failed in each of the six control group participants. We noted that not all patients in the control group received, or were eligible to receive, the same standard of care treatments prior to the study and did not receive any skin substitute or wound dressing treatments during the study as a comparison to the test group patients that were treated with OCMTM . Due to the stated limitations in the study as previously described, we stated that we do not believe that the applicant has demonstrated that OCMTM offers a substantial clinical improvement over existing treatments.
Finally, we noted that the sample size of 19 individuals (six of whom were assigned to the control group) in the Simman, et al. (2023) study limits the generalizability of the findings. Therefore, we questioned whether OCMTM has superior clinical outcomes and healing for DFUs compared to the standard of care or the predicate or reference devices. Additionally, we noted that the Simman, et al. (2023) study abstract and interim analysis were sponsored by the manufacturer and have not been published, and therefore are not based on independent and peer-reviewed findings.
In addition to the Simman, et al. (2023) study (including the abstract and interim results), in support of its first claim, the applicant submitted two posters presenting results from limited case studies investigating two patients who received treatment using OCMTM combination therapy at the point of care. The first poster (Black, et al., 2023) discussed the treatment of two patients seeking treatment for DFUs at a wound care clinic: (1) a 75-year-old female patient who developed a DFU on her right third toe whose DFU wound progressed from nonhealing to healing after one application of the combination OCMTM therapy; and (2) a 65-year-old female patient with a history of diabetes and a blister of 3-month duration that progressed from nonhealing (after treatment with collagen powder, a gauze covering, an absorbent dressing, and a protective bandage) to completely closed after nine applications of the combination OCMTM therapy over 63 days. The study authors concluded that these case studies demonstrated: (1) rapid and durable healing of chronic/nonhealing wounds in two patients with diabetes who received the combination therapy for their chronic wounds; (2) significantly faster closure of a DFU within 1 week using OCMTM combination therapy than the average healing rate of 84 days for a 1-3 cm2 plantar ulcer managed using standard care practices; and (3) that early treatment of chronic/nonhealing wounds with OCMTM combination therapy improves outcomes and can lead to complete closure.
Similarly, the second poster (Barrett, et al., 2023) presented results from a case study of two patients who received follow-up care at an outpatient wound center: (1) a 58-year-old male patient with a distal plantar lateral ulceration with infection, which required hospitalization; and (2) a 56-year-old male patient with leg trauma that had obliterated the patient's anterior tibial and peroneal arteries, leaving him with single vessel runoff to the left foot via the posterior tibial artery. In the first patient, after 5 weeks of initial negative pressure wound therapy following surgery, the percentage area reduction of the wound was 19 percent. In comparison, after three weekly follow-up applications of OCMTM combination therapy, the patient's percentage area reduction was 95 percent. In the second patient, the amputation site was noted as completely necrotic, and therefore not a candidate for standard of care negative pressure wound therapy due to poor skin condition, ischemia, and hyperalgesia. It was noted that after three applications of OCMTM combination therapy, there was a significant improvement in the wound depth and tissue color, with visible epithelialization at the wound edges despite the patient's obvious ischemia. It was noted that the wound size improved and completely healed between the fourth and fifth application of OCMTM combination therapy and after the seventh application of OCMTM combination therapy. The researchers concluded that the case studies demonstrate complete and rapid healing of refractory DFUs in two patients with diabetes who had previously undergone lower extremity amputations and that early use of OCMTM combination therapy has the potential to reduce the rate of amputations and improve patients' quality of life.
We noted that both case studies (Barrett, et al., 2023, and Black, et al., 2023) were sponsored by the manufacturer and only had two study participants treated with OCMTM combination therapy, which limits the generalizability of the findings. Although the studies suggest that the two participants treated with OCMTM combination therapy showed transition to a healing state subsequent to the application of OCMTM, the results varied widely in terms of number of applications needed to achieve positive results and treatment duration. Further, we noted that these case studies provide no direct comparison to the standard of care treatment or the predicate or reference devices. We noted that eligibility for standard of care treatments also varied across patients and resulted in varying degrees of percent area reduction or wound closure from prior treatments before application of OCMTM combination therapy. While in one patient, the study showed an improved clinical outcome in percentage area reduction (19 percent to 95 percent) with treatment utilizing OCMTM combination therapy, we noted that the treatment including OCMTM was not only completed subsequent to standard of care treatment with a collagen wound protectant, but also delivered to the same individual rather than as a comparison to standard of care treatments in a control group.
We questioned whether the submitted evidence adequately supports the claim that OCMTM has superior clinical outcomes and healing for DFU compared to the standard of care. We stated our interest in additional information to demonstrate whether the nominated device demonstrates a substantial clinical improvement in comparison to similar collagen-based matrix devices.
In the second claim, the applicant asserted that OCMTM provides faster healing rates than standard of care for VLUs. However, based on the evidence submitted, we noted the following concerns: (1) reliance on unpublished studies; and (2) a lack of any documentation indicating the study authors, study description, methods, limitations, information on standard of care treatment for the comparison of control groups, analysis, or discussion. The only data provided were in the form of two tables. One table [114] provided demographic information for the study participants, such as race, age, gender, presence of VLUs, comorbidities, wound area, and wound age; however, there is no indication of how many initial study participants were included in the final results or how many were assigned to either the treatment or control group receiving the standard of ( printed page 94194) care. The other table [115] presented one row of data from the final results of a randomized controlled trial on VLUs showing an average percent area reduction of 66 percent at 12 weeks in OCMTM treatment group (there was no comparison to the standard of care treatment group) and an average percent area reduction of 34 percent at four weeks in the OCMTM treatment group compared to an average percent area reduction of 31 percent at four weeks in the standard of care group. Due to the lack of a study report, we stated that we have insufficient information to adequately assess this study or make a determination as to whether the study supports the claim that OCMTM provides faster healing rates than standard of care for VLU.
In order to demonstrate substantial clinical improvement over currently available treatments, we noted that we consider supporting evidence, preferably published peer-reviewed clinical trials, that shows improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process compared to the standard of care. We noted that additional supporting evidence, preferably published peer-reviewed clinical trials, that shows these improved clinical outcomes would help inform our assessment of whether OCMTM demonstrates substantial clinical improvement over existing technologies.
In the third claim, the applicant asserted that OCMTM provides superior clinical outcomes for patients who could not qualify for clinical trials due to comorbidities, and in the fourth claim, the applicant stated that OCMTM improved results when compared to results with standard of care for patients who failed prior treatment. The applicant used the same pair of documents as supporting evidence for both the third and fourth claims: (1) a case study by 16 independent investigators (Bettle, et al., 2023), and (2) a final summary table [116] of the results of that case study. In the case study by the 16 independent investigators, OCMTM combination therapy was administered to 65 patients with wound ages ranging from 12 weeks to 15 years who failed prior treatment, including six patients with prior failed cellular tissue product therapies. Patients were not otherwise subjected to inclusion or exclusion criteria. According to the applicant, the findings by the 16 independent investigators showed 77 percent of wounds were closed by 12 weeks and the average area reduction was 90 percent. Wounds treated included DFUs, VLUs, pressure injuries, arterial, pyoderma, hematomas, surgical, and trauma. We noted that the study lacked direct comparison to a standard of care treatment. Rather, the study compared patient data to standardized data on wound closure and mean time to total wound closure by wound type based on standardized data from the U.S. Wound Registry.
We questioned whether the submitted evidence adequately supports the claims that OCMTM provides superior clinical outcomes for patients who could not qualify for clinical trials, due to comorbidities, or that OCMTM improved results when compared to results with standard of care for patients who failed prior treatment. We welcomed further investigation with comparators to help determine whether the device demonstrates substantial clinical improvement over currently available treatments in the clinical setting where it is most likely to be used.
In its fifth claim, the applicant asserted that in vitro (Davis, et al., 2023) and in vivo (Davis, Jozic, et al., 2023) study results demonstrate antimicrobial properties and patient safety. The applicant further asserted that OCMTM addresses an unmet medical need, stating that no other product demonstrates antimicrobial properties and leads to complete wound healing. In the in vivo study (Davis, Jozic, et al., 2023), researchers made 31 deep reticular wounds across the paravertebral and thoracic areas on each of specific pathogen-free pigs. Pathogenic strains of Methicillin-Resistant Staphylococcus Aureus (USA300) or Pseudomonas Aeruginosa (ATCC 27312), prepared as 106 CFU/ml inoculum suspensions, were used to inoculate all wounds within 20 minutes after wounding followed by application of polyurethane dressings (Tegaderm, 3M, USA) for 72 hours before being treated. Subsequent treatment consisted of OCMTM alone in one test group, OCMTM plus a skin protectant in another test group, Aquacel Ag Advantage in the positive control group, or the wounds were left untreated in the negative control group. We noted that the only in vivo study (Davis, Jozic, et al., 2023) with direct comparison to a skin protectant was conducted on non-human subjects (pigs). We questioned whether these data can be extrapolated to demonstrate significant clinical improvement in humans. In addition, according to the applicant, the in vitro study (Davis, et al., 2023) showed OCMTM significantly inhibiting Methicillin-resistant Staphylococcus aureus and Pseudomonas aeruginosa compared to negative controls. We noted that the in vitro study (Davis, Jozic, et al., 2023) lacked a direct comparison to performance of other similar skin protectant products or wound therapies besides infection control methods such as silver sulfadiazine or Mupirocin antibiotic. We further noted that both the in vitro and in vivo studies were submitted as poster presentations and that the studies had not been published and peer-reviewed in full.
We questioned whether the submitted evidence adequately supports the claims that OCMTM demonstrates antimicrobial properties and patient safety. We noted that additional supporting evidence, preferably published peer-reviewed clinical trials, that demonstrates improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process, would help inform our assessment of whether OCMTM demonstrates substantial clinical improvement over the standard of care and existing technologies.
For its sixth claim, the applicant asserted that study results demonstrated patient safety of OCMTM . In support of this claim, the applicant provided one consumer research study (Princeton Consumer Research Corp., 2019) [117] of 25 subjects showing no immediate allergic reaction to OCMTM . We noted that, similar to our previously stated concerns, the study did not include a direct comparison to predicate or reference devices despite claiming an improvement over standard of care treatment. We noted that the submitted evidence does not adequately support the claims that OCMTM demonstrates substantial clinical improvement in product safety in comparison to similar products.
Finally, we noted that OCMTM may not demonstrate that it substantially improves the diagnosis or treatment of an illness when compared to the benefits of other available treatments. OCMTM was determined to be substantially equivalent to a legally marketed device, the SweetBio Apis, which received 510(k) clearance on ( printed page 94195) April 29, 2019. The FDA 510(k) summary for OCMTM indicated that both devices share similar technological characteristics. Per FDA, the main differences between OCMTM and the predicate are the specific collagen source (OCMTM uses whitefish skin-derived collagen, while the SweetBio Apis uses porcine skin-derived collagen) and the specific identity of the supplemental components, which serve the same fundamental purpose in enabling each wound dressing to achieve the shared intended use.[118]
We invited public comment on whether OCMTM meets the substantial clinical improvement criterion at § 419.66(c)(2)(i).
Comment: With respect to our concern about the lack of independent peer-reviewed or published clinical evidence, the applicant commented that they recognize the importance of peer-reviewed research. Further, the applicant noted that while at the time of the initial application, none of the studies had been submitted for peer-review or published in indexed journals, the clinical evidence previously referenced has now been published, is in press for an indexed journal, or has been submitted for review at an indexed journal and is publicly available on a preprint server. The applicant submitted these as part of its comment.[119 120 121] The applicant also acknowledged that it sponsored the provided studies but asserted that, due to the newness of the product, cost, and unclear coverage status, the lack of investigator-sponsored trials is not unexpected.
Response: We thank the applicant for their response to our concerns regarding the lack of independent and peer-reviewed studies. After consideration of the applicant's comments, we believe the applicant has addressed our concern about the lack of peer-reviewed and published studies by submitting two studies (Bettle III, et al., 2024, and Simman, et al., 2024) that appear to support the applicant's claims of substantial clinical improvement that have been accepted for publication in peer-reviewed medical journals.
Comment: In response to our concerns that the applicant did not provide a comparison of OCMTM to similar wound closure products, the applicant reiterated that OCMTM catalyzes wound closure in patients with nonhealing wounds treated with other advanced wound therapies. The applicant also asserted that OCMTM offers a more rapid beneficial resolution over the best available therapies, including the predicate and reference devices. Specifically, the applicant asserted that in comparison to the protective dressing application, OCMTM application in conjunction with debridement qualifies as an active wound procedure within the scope of active wound care management services. The applicant also noted that OCMTM application following initial standard of care failure may replace the need for negative-pressure wound therapy, placental membranes, bioengineered skin substitutes, several acellular matrices, autologous fibrin, and leukocyte platelet patches, all of which are currently used in the hospital outpatient setting.
The applicant clarified that it has not advocated for OCMTM to be used in routine management of wounds as it believes the current standard of care, which includes four weeks of routine therapy, should be utilized prior to the application of OCMTM . Specifically, the applicant asserted that once wounds become refractory, then OCMTM can be applied to re-initiate the healing process, as demonstrated in the submitted studies.
The applicant further stated that FDA has specifically acknowledged the lack of innovative products aimed at the treatment of non-healing chronic wounds. The applicant noted that patients with significant medical comorbidities presenting with non-healing wounds of any size have not been enrolled in advanced wound therapy randomized controlled trials, even though these patients represent a high-risk group that most clinicians believe need active treatment beyond the routine standard of care to prevent infection, amputation, and even death. The applicant asserted that OCMTM addresses an unmet need for patients with multiple comorbidities who have refractory wounds of at least four weeks duration, including at least two weeks of care by a wound specialist, who would have likely been excluded from the few randomized control trials in this field.
Response: We thank the applicant for its input and clarification. While we appreciate that FDA has specifically acknowledged the lack of innovative products aimed at the treatment of non-healing chronic wounds, we remain concerned with the lack of clinical studies and comparative studies demonstrating whether the use of OCMTM, when applied following initial standard of care failure as an active wound procedure, results in substantial clinical improvement over other available active wound care management services. With respect to replacing the need for negative pressure wound therapy, placental membranes, bioengineered skin substitutes, several acellular matrices, autologous fibrin and leukocyte platelet patches, we address specific substantial clinical improvement claims and responses to our concerns below.
Comment: Several commenters stated that they believed for wound infections that could progress to sepsis or necessitate an amputation, OCMTM could prevent patient complications, including disability secondary to the complications of a non-healing wound, or death. The commenters stated that for chronic non-healing and hard-to-heal wounds OCMTM offers several advantages including that its effects can be seen quickly, even though other products used prior to OCMTM failed, and its amorphous property enables optimal use of the product, even in wounds with non-perfect, irregular wound beds. The commenters stated that this amorphous structure allows the product to fill unique areas within wounds, such as tunneling and undermining, that would otherwise be unmanageable using a standard advanced wound graft product.
Response: We thank the commenters for their input. We maintain our concerns that the submitted evidence does not adequately support the applicant's claims that OCMTM demonstrates substantial clinical improvement compared to other available treatments. We appreciate that commenters provided information about the unique uses for OCMTM 's amorphous structure; however, we note that the commenters did not supply additional data in support of these claims. Furthermore, we note that the applicant did not make any specific claims about the consequential benefits directly attributable to any specific or isolated mechanism of OCMTM.
Comment: With respect to our concerns about the lack of direct comparison to the predicate or reference devices in the studies used to support its first three claims, the applicant noted ( printed page 94196) that while OCMTM and the predicate device SweetBio Apis have similar indications and the same intended use, they differ in the chemical and physical properties of the derived collagen product as well as the additional biocompatible materials that supplement the product. The applicant also asserted that SweetBio Apis does not have any published data on its effectiveness in healing wounds in a similar population, as in OCMTM 's most recently completed study. The applicant stated that SweetBio Apis's study of real-world evidence featured a small sample size (n = 12) with likely singular etiology (implied to be DFU given diabetic population) and limited transparency into the selection of study participants. The applicant asserted that, in comparison, OCMTM has been investigated in a patient group that is significantly larger, has more variability in ulcer etiology, and has more comorbidities, which is representative of the overall patient population for whom OCMTM is indicated. Additionally, the applicant highlighted that SweetBio Apis has published only one randomized controlled trial involving participants with Mohs Surgical Defects, which demonstrated no significant difference in re-epithelization.
The applicant asserted that, as such, a direct comparison of OCMTM 's and SweetBio Apis's published evidence indicates that OCMTM has more substantially proven capability to deliver clinical improvement for these hard-to-treat wounds. In addition, the applicant stated that while OCMTM and SweetBio Apis have similar indications and the same intended use, their derived collagen products differ in the chemical and physical properties. The applicant further stated that OCMTM differs from SweetBio Apis in terms of additional biocompatible materials that supplement the product. The applicant asserted that this supplement material ( i.e., the additional biocompatible materials) raises no new questions of safety or effectiveness when OCMTM is compared to the predicate and reference devices but does confer additional benefits to OCMTM . The applicant asserted that OCMTM 's additional benefits warrant the product's consideration for transitional device pass-through payment. The applicant stated they are planning to work with the HCPCS workgroup to define a more accurate description of OCMTM since it is unlike other collagen products on the market.
The applicant also clarified that their most recent clinical studies demonstrate the multi-faceted impact of OCMTM 's multiple components on wound healing. Specifically, the applicant asserted that other collagen wound matrix devices' similar compositions confer some, but not all, of the wound healing properties characteristic of OCMTM . The applicant further asserted that a head-to-head comparison of OCMTM to any of these other products would only represent the marginal benefits of OCMTM 's supplemental biomaterials. In addition, the applicant stated that their clinical study relies on the metric of wound percent area reduction in hard-to-heal wounds in order to demonstrate OCMTM 's total clinical benefit, rather than the consequential benefits directly attributable to any specific mechanism in isolation. The applicant asserted it has not made individual claims about OCMTM 's clinical superiority over similar wound healing products in regards to any specific mechanisms that contribute to the totality of OCMTM 's clinical benefits conferred to patients with hard-to-heal wounds as it has not performed such independent or multi-factorial statistical analyses.
Response: We thank the applicant for the additional information. After reviewing the provided information, we note that the applicant's points regarding the differences between OCMTM and SweetBio Apis are comparisons of volume of data, study design, and test results between studies that each evaluate OCMTM and SweetBio Apis separately and without a direct comparison. Therefore, we remain concerned about the lack of direct comparison of OCMTM to other similar devices, particularly those that are animal-derived collagen-based products in test arms or cohorts within the same study. We note that we were not able to verify the applicant's claims that there is more scientific data supporting the clinical effectiveness of OCMTM while there is none supporting the clinical effectiveness of SweetBio Apis, especially since there appears to be a high volume of data showing clinical effectiveness of SweetBio Apis available on the manufacturer's website.[122] We also note that even if the applicant could demonstrate that there is comparatively more data on effectiveness in wound healing from treatment with OCMTM than for SweetBio Apis, clinical effectiveness is not an equivalent standard to substantial clinical improvement over existing technologies. With respect to the applicant's argument that SweetBio Apis may not be a suitable comparator because OCMTM 's derived collagen products differ in chemical and physical properties as well as the additional biocompatible materials that supplement the products, as we noted previously, the FDA made no distinction between porcine or fish-derived collagen in wound dressings,[123] or the chemical and physical properties of any additives used to anchor this collagen to the wound.[124] We do not believe that the applicant has demonstrated that the physical and chemical properties result in substantial clinical improvement because of the lack of direct comparison of OCMTM to these similar products.
In response to the applicant's assertion that a head-to-head comparison of OCMTM to any other products would only represent the marginal benefits of OCMTM 's supplemental biomaterials, we believe that this suggests that other products would perform similarly to OCMTM in the metric of percent area reduction across different test arms in patients with the same or similar hard-to-heal wounds. While we appreciate the need to study OCMTM 's effect on the total clinical benefits in hard-to-heal wounds in isolation, we continue to believe evaluation of the applicant's first three claims of substantial clinical improvement ( i.e., OCMTM 's superior outcomes and faster healing rates in comparison to the standard of care) requires a direct comparison of OCMTM 's clinical outcomes to those similar products. For the reasons discussed, we do not believe that OCMTM represents a substantial clinical improvement relative to similar currently available therapies.
Comment: In regard to the applicant's claim that OCMTM provides a treatment option for a patient population unresponsive to, or ineligible for currently available treatments, the applicant submitted additional information to clarify that OCMTM not ( printed page 94197) only matches but exceeds the clinical utility of skin substitutes as advanced wound therapy for patients with hard-to-heal wounds. Specifically, the applicant asserted that OCMTM 's superiority over other forms of wound management is evident in its ability to reduce wound size in patients with non-healing wounds that were failed by alternative wound products, such as gentian violet/methylene blue foam, manuka honey, cellular tissue products (CTPs), Negative Pressure Wound Therapy (NPWT), and dressings consisting of alginate, collagen, or silver. The applicant stated that the study protocol in Dhillon, et al. (2024) [125] ensured that patients had wounds that had not responded to therapy for at least eight weeks and had undergone high-quality wound care for a minimum of two weeks that included the use of advanced therapies, such as the predicate and reference devices. Furthermore, the applicant asserted that the OCMTM clinical trial design was intentional in evaluating the device's clinical utility in a patient population that was not served by other alternative wound products.
Response: We appreciate the applicant's clarification. We agree with the applicant that the Dhillon, et al. (2024) study demonstrated OCMTM 's effectiveness in treating a group of 53 patients who did not improve after receiving alternative wound products prior to the study. However, we note that, of the 111 patients that entered the study's screening phase, only 53 patients and 54 wounds (which included 18 DFUs, 19 VLUs, two pressure injuries, one surgical, one lower extremity wound, and 12 unclassified etiology) received treatment and were eligible for the data set. We also note that the study's final analysis excluded the 58 patients who responded to the standard of care ( i.e., cleaning and debridement) with wound reduction of more than 30 percent. We are concerned that Dhillon, et al. (2024) fails to provide details about the specific previous wound management treatments that the study participants received to demonstrate that it exceeds the clinical utility of skin substitutes as advanced wound therapy for hard-to-heal wounds as the applicant claims above. Absent this data, we are unable to verify that OCMTM treats a patient population unresponsive to currently available multimodal wound management treatments.
Comment: In response to our concerns that the studies provided on OCMTM 's antimicrobial properties do not demonstrate results in live human subjects, the applicant commented that OCMTM can play a preventative role in wound management through the inherent antimicrobial properties of its anhydrous matrix. The applicant asserted that a small-scale clinical study signaled OCMTM 's antimicrobial capacity and that its antimicrobial properties could enable preventative care of hard-to-heal wounds. Specifically, the applicant stated that OCMTM 's non-antibiotic composition may reduce the probability of bacterial colonization progressing to wound infection, which could result in antibiotic resistance or even sepsis and limb amputation. In addition, the applicant asserted that OCMTM has clinical utility as a non-antibiotic decolonization agent in clinically uninfected wounds for which antibiotic prophylaxis is deemed inappropriate, per International Working Group on the Diabetic Foot/Infectious Diseases Society of America (IWGDF/IDSA) Guidelines. The applicant also commented that it is committed to working with FDA to evaluate the extent to which the OCMTM 's antimicrobial properties confer substantial clinical benefit over the standard of care treatment.
Furthermore, the applicant suggested that transitional device pass-through payments would enable it to continue developing evidence that OCMTM 's decolonization function provides substantial clinical benefit over the standard of care. The applicant compared OCMTM 's decolonization function to topical prophylactic decolonization methods, for which clinical significance has historically been difficult to substantiate. The applicant asserted that this mechanism of infection prevention and control has been widely practiced for decades in accordance with CDC guidance, as exemplified by chlorhexidine bathing and administration of intranasal mupirocin or Iodophor. Similarly, several non-applicant commenters stated that OCMTM has some unique and validated anti-microbial properties that can potentially reduce the risk of infection, as many hard-to-heal wounds are colonized by bacteria but not yet infected. These commenters also asserted that this may lead to a further reduction in antibiotic prescriptions during treatment of hard-to-heal wounds.
Response: We thank the applicant and commenters for their input. After consideration of the comments received, we remain concerned that the applicant has not provided sufficient clinical evidence to demonstrate OCMTM 's antimicrobial properties in human subjects. The provided case study of OCMTM 's decolonization effects includes only three patients with hard-to-heal wound etiologies and compares OCMTM to non-treatment of periwound skin, rather than wound beds of similar etiology treated with the standard of care.[126] Therefore, we do not believe this demonstrates that OCMTM application to the wound bed results in fewer bacterial infections.
Concerning the applicant and other commenters' statements about the claim about antimicrobial properties, we note that these claims appear to be conditional. While the applicant compared OCMTM 's decolonization function to topical prophylactic decolonization methods, its claims that OCMTM has the potential to reduce the probability of bacterial colonization, play a preventative role in wound management, and reduce the risk of infection are not proven. In addition, we note that the applicant did not provide the referenced joint CDC/FDA workshop materials describing this study's methods, so we are unable to verify whether topical prophylactic decolonization methods are analogous to OCMTM . Therefore, we remain concerned about the lack of evidence demonstrating that OCMTM antimicrobial properties improve outcomes by reducing the risk of infection.
In regard to the applicant's plan to pursue further studies to demonstrate substantial clinical improvement in this area, we are unable to consider future evidence for OCMTM 's current transitional pass-through payment application.
Comment: In response to our concerns that the submitted evidence does not adequately support the applicant's claim that OCMTM demonstrates substantial clinical improvement in product safety in comparison to similar products, the applicant stated that FDA's clearance of OCMTM through the 510(k) application process involves a comprehensive review of the device's safety and performance data. Specifically, the applicant asserted that ( printed page 94198) OCMTM is among the minority of advanced wound therapies that have passed rigorous FDA review of safety and effectiveness. In addition, the applicant stated that Bettle, et. al. (2024) demonstrates that OCMTM has no potential allergenicity, no potential sensitization, and resulted in no known product-related adverse events. The applicant asserted that this was further corroborated by OCMTM 's most recent, real-world evidence dataset presented in Dhillon, et. al. (2024), which showed that treatment with OCMTM resulted in only eight potential product-related adverse events and no serious adverse events.
Response: We appreciate the applicant's input. However, we maintain our concern that the submitted evidence does not demonstrate OCMTM 's substantial clinical improvement in product safety in comparison to similar products. We acknowledge that FDA's 510(k) clearance process includes a comprehensive review to evaluate the safety and effectiveness of a new product, however, demonstrating substantial clinical improvement for device pass-through payment is different from FDA's 510(k) process. While the applicant has shown that OCMTM is safe, we continue to believe that the applicant has not shown that OCMTM demonstrates substantial clinical improvement in product safety in comparison to currently available therapies. For the reasons discussed, we do not believe that OCMTM represents a substantial clinical improvement relative to existing therapies currently available as discussed in the summary above. Therefore, we have determined that OCMTM does not meet the substantial clinical improvement criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that OCMTM would be reported with HCPCS codes as shown in Table 125.
To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant utilized the CY 2024 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5052, which had a CY 2024 payment rate of $379.92 at the time the application was received. HCPCS code 11042 in APC 5052 had a device offset amount of $0.04 at the time the application was received. According to the applicant, the cost of OCMTM is $1,320.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 ( printed page 94199) percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $1,320.00 of OCMTM is 347.44 percent of the applicable APC payment amount for the service related to the category of devices of $379.92 (($1,320.00/$379.92) × 100 = 347.44 percent). Therefore, we stated that we believe OCMTM meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $1,320.00 for OCMTM is 3,300,000.00 percent of the cost of the device-related portion of the APC payment amount for the related service of $0.04 (($1,320.00/$0.04) × 100 = 3,300,000.00 percent). Therefore, we stated that we believe OCMTM meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $1,320.00 for OCMTM and the portion of the APC payment amount for the device of $0.04 is 347.43 percent of the APC payment amount for the related service of $379.92 ((($1,320.00−$0.04)/$379.92) × 100 = 347.43 percent). Therefore, we stated that we believe OCMTM meets the third cost significance requirement.
We invited public comment on whether OCMTM meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
Comment: Several non-applicant commenters asserted that OCMTM warrants transitional pass-through payments because the current construction of the APC bundle includes the services for hard-to-heal wounds but its associated payment rate ( i.e., the reimbursement amount for the services assigned to the APC) is not sufficient to cover the cost of more advanced treatments, such as the OCMTM, which are needed to manage complex wounds. The commenters stated that they strongly support the use of OCMTM in the outpatient hospital setting; however, they asserted that the costs are too high given the current APC assignment and its payment rate. The commenters stated that without access to OCM in the hospital outpatient setting patients may be treated in other settings, which can be costlier since these other settings bill separately for advanced wound therapy. The commenters stated that OCMTM represents a clinically effective solution for hard-to-heal wounds that, at present, cannot be used in the outpatient hospital setting given that the cost of using OCMTM is not adequately reflected in the payment rate for the APC to which the product is assigned under the OPPS.
Response: We thank the commenters for their additional input. We acknowledge the commenters' concerns about the cost of OCMTM as a treatment option in the hospital outpatient department; however, we note that in order to be eligible for device pass-through, a device must meet all requirements for pass-through payment status in our regulation at § 419.66, in addition to determining that the cost of the device is not insignificant, as described in § 419.66(d).
Comment: The applicant thanked CMS for agreeing that it met the cost criterion.
Response: We appreciate the applicant's comment. After consideration of the public comment received and our findings from the first, second, and third cost significance tests, we agree that OCMTM meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments received and our review of the device pass-through application, we are not approving OCMTM for transitional pass-through payment status in CY 2025 because the product does not meet the substantial clinical improvement criterion at § 419.66(c)(2).
(c) OPN NC
SIS Medical AG submitted an application for a new device category for transitional pass-through payment status for OPN NC for CY 2025. Per the applicant, OPN NC percutaneous transluminal coronary angioplasty (PTCA) dilatation catheter is a sterile, single-use, rapid exchange catheter with a distal non-compliant double layer balloon attached to a flexible distal polymer shaft. The applicant explained that OPN NC is intended for balloon dilatation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion. Per the applicant, the balloon dilatation catheter is also indicated for post deployment expansion of balloon expandable coronary stents. The applicant asserted that the device is inserted to position a balloon in a calcified coronary lesion where super-high pressure is used with the intention of achieving acceptable expansion of the lesion. Per the applicant, radiopaque balloon marker bands enable accurate positioning of the device, and shaft markers for brachial and femoral techniques are also in place. According to the applicant, OPN NC is intended for all patient populations.
Please refer to the online application posting for OPN NC, available at https://mearis.cms.gov/public/publications/device-ptp/DEP231214L8XQC, for additional detail describing the device and the disease treated by the device.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), on March 14, 2022, the applicant received 510(k) clearance from FDA for OPN NC as a device intended for balloon dilatation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion. The balloon dilatation catheter is also indicated for post deployment expansion of balloon expandable coronary stents. We received the application for a new device category for transitional pass-through payment status for OPN NC on December 14, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether OPN NC meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), one commenter stated that they believe OPN NC meets the newness criterion at § 419.66(b)(1) because unlike prior balloons including noncompliant balloons, it is specially designed to achieve 35-55 atmospheres (atm) of pressure, which is far beyond the 20 atm of conventional balloons. The commenter further noted that this can be particularly and uniquely useful for under-expanded stents from fibrotic or calcified vessels, which even new technologies such as lithotripsy and older technologies such as laser are unable to expand.
Response: We appreciate the commenter's input; however, the comment relates to an assessment of whether the technology meets the substantial clinical improvement criterion rather than the newness criterion. As such, we have addressed ( printed page 94200) this comment in the substantial clinical improvement criterion discussion below.
We received the application for a new device category for transitional pass-through payment status for OPN NC on December 14, 2023, which is within 3 years of March 14, 2022, the date of FDA 510(k) clearance. Based on our review of the application, we have determined that OPN NC meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. Per the applicant, OPN NC is integral to the service provided and is used for one patient only. While the applicant did not explicitly state whether the device is surgically inserted or comes in contact with human tissue, per the device description, OPN NC is inserted into the patient for balloon dilation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion.
We invited public comment on whether OPN NC meets the eligibility criterion at § 419.66(b)(3).
Comment: One commenter submitted a comment stating that they believe that OPN NC meets the § 419.66(b)(3) criterion because it is integral to the service provided and used for one patient only, that is, it is disposable.
Response: We appreciate the commenter's input. With respect to the eligibility criterion at § 419.66(b)(3), as noted in the proposed rule, the applicant did not indicate that OPN NC is surgically inserted or comes in contact with human tissue; however, because the device is inserted into the patient for balloon dilation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion, we believe that OPN NC is surgically inserted and comes in contact with human tissue. In addition, we agree with the applicant that OPN NC is an integral part of the service furnished and used for one patient only. After consideration of the public comment we received and our review of the application, we have determined that OPN NC meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not address whether OPN NC is equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or if OPN NC is a supply or material furnished incident to a service.
We invited public comments on whether OPN NC meets the exclusion criterion at § 419.66(b)(4).
Comment: With regards to criterion § 419.66(b)(4), one commenter stated that OPN NC is not equipment.
Response: We appreciate the commenter's input. With respect to the eligibility criterion at § 419.66(b)(4), as noted in the proposed rule, the applicant did not indicate that OPN NC is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or if OPN NC is a supply or material furnished incident to a service; however, because the device is inserted into a calcified coronary lesion where super-high pressure is used for the purpose of improving myocardial perfusion, we believe that OPN NC is not a supply or material furnished incident to a service. In addition, OPN NC is a single-use PTCA dilatation catheter with a distal non-compliant double layer balloon attached to a flexible distal polymer shaft, and therefore does not appear to be equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets. After consideration of the public comment we received and our review of the application, we have determined that OPN NC meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. The applicant described OPN NC as a PTCA dilatation catheter with a distal non-compliant double layer balloon attached to a flexible distal polymer shaft. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe OPN NC. Per the applicant, the device category, C1725 (Catheter, transluminal angioplasty, non-laser (may include guidance, infusion/perfusion capability)) does not appropriately describe OPN NC, because OPN NC is a super high pressure, non-compliant double (twin) layer balloon. Based on the description the applicant provided, OPN NC is a transluminal vascular dilatation catheter with a balloon intended for dilatation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion, which is consistent with the devices described by C1725. In this context, we stated that we believe OPN NC may be similar to the devices described by C1725, and therefore, OPN NC may also be appropriately described by C1725.
We invited public comment on whether OPN NC meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concerns that OPN NC may be appropriately described by C1725, one commenter commented that OPN NC is similar in purpose and function to other devices under C1725; however, the double-layer construction is unique in the coronary space, and enables the high-pressure inflation needed for angioplasty of resistant lesions. The commenter further commented that while this is similar in mechanism to conventional angioplasty balloons (dilatation through pressure, rather than through intravascular lithotripsy (IVL)), it creates a unique functionality of using pressure to dilate lesions and stents that would otherwise be resistant to dilatation. Another commenter stated that it thinks OPN NC should be reimbursed and assigned the same codes as coronary IVL (C1761) as it is used for similar patients/treatments.
Response: We appreciate the commenters' input. We do not agree that OPN NC is described by the C1761 (Catheter, transluminal intravascular lithotripsy, coronary) device category as suggested. We note that C1761 is used to describe coronary IVL devices that are used to perform IVL, a methodology that delivers sonic pressure waves to break calcium deposits in a coronary vessel, which is inconsistent with the function and purpose of OPN NC. As such, we do not believe that C1761 describes OPN NC. However, we continue to believe OPN NC is similar to the devices described by C1725. Specifically, C1725 (Catheter, transluminal angioplasty, non-laser (may include guidance, infusion/ ( printed page 94201) perfusion capability)) is used to describe devices that rely on inflation of a balloon to directly apply pressure to plaque in a vessel during an angioplasty procedure. The applicant stated that OPN NC is a transluminal vascular dilatation catheter with a balloon intended for dilatation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion, which is consistent with the devices described by C1725. While we recognize that OPN NC may vary in construction from other devices described by C1725, we nevertheless continue to believe that OPN NC is appropriately described by C1725. Therefore, we have determined that OPN NC does not meet the device category eligibility criterion at § 419.66(c)(1) because it is appropriately described by an existing category or a category previously in effect.
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. According to the applicant, OPN NC represents a substantial clinical improvement over existing technologies in the management of patients with highly calcified coronary lesions by providing optimal lumen expansion and demonstrating better outcomes in lesion treatment compared to other devices.
The applicant provided the following evidence to support its claim: three peer-reviewed studies; a PowerPoint presenting an indirect comparison of OPN NC versus another device, Shockwave Intravascular Lithotripsy (IVL) System with Shockwave C2 Coronary Intravascular Lithotripsy (IVL) Catheter (Shockwave),[127] that uses IVL to treat calcium lesions; a spreadsheet summarizing the data presented in the PowerPoint document comparing OPN NC and Shockwave; and a background article providing an expert consensus statement from the Society for Cardiovascular Angiography & Interventions on management of in-stent restenosis and stent thrombosis.[128] Table 126 summarizes the applicant's assertion regarding the substantial clinical improvement criterion. Please see the online posting for OPN NC for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.
( printed page 94202) ( printed page 94203)After review of the information provided by the applicant, we stated that we have the following concerns regarding whether OPN NC meets the substantial clinical improvement criterion. The applicant presented the published results of one study of 50 patients undergoing optical coherence tomography (OCT)-guided percutaneous coronary interventions, including OPN NC, to treat calcified lesions (Natalia Pinilla-Echeverri, et al., 2023). The retrospective study aimed to gain a better understanding of OPN NC calcium modification mechanisms, such as creating deep and wide calcium fractures during percutaneous coronary interventions with the intended clinical outcome of improving myocardial perfusion. Per the applicant, the study showed a primary efficacy endpoint of ≥ 80 percent expansion of the mean reference lumen area achieved in 80 percent of the patients treated. The applicant also presented a retrospective study evaluating 326 highly resistant coronary lesions that had failed to achieve adequate post-dilatation luminal gain with conventional NC-balloons (Secco, et al., 2019). Per the study authors, an OPN NC balloon was inflated to achieve a uniform balloon expansion after the failed attempts with conventional NC-balloons. According to the authors, 413 OPN NC balloons were used (1.26 per lesion), and angiographic success was achieved in 318 lesions (97.5 percent), procedural success was achieved in 315 lesions (96.6 percent), and technical success was achieved in 288 patients (90.5 percent). The study authors also reported that the OPN NC balloon alone was able to achieve adequate expansion in 288 cases (90.5 percent), while in 30 patients, rotational atherectomy was needed and performed because of the impossibility to cross the lesion with a proper sized OPN NC balloon. The applicant presented a third study focused on patients needing treatment of in-stent restenosis (ISR) (Seiler, et at., 2023). According to the authors, 208 ISR lesions were treated in 188 patients. The study authors concluded that the use of OPN NC for treatment of ISR lesions was safe (primary endpoint of the study) and may lead to a low rate of target lesion/vessel failure (TLF/TVF) during long-term follow-up. We noted that these studies were not randomized clinical trials with a comparator to demonstrate clinical improvement. Instead, the applicant presented results from registries using non-randomized, retrospective study designs without a control group, which we stated that we believe may reduce the strength of the evidence presented to support the claim. The authors noted in all three studies that randomized trials may be needed to compare OPN NC to other similar devices.
Further, we also noted that in one of the studies (Natalia Pinilla-Echeverri, et al., 2023), the study authors indicated that use of other calcium lesion modification devices prior to applying OPN NC to the patients in that study is a potential confounder that could result in overestimation of OPN NC's effectiveness. The study authors stated that this was controlled by having an exclusive OCT pullback pre-OPN NC but indicated that calcium plaque modification caused by other devices may not be evident on OCT. The study authors further noted that since other devices were used before OPN NC, they could not comment on calcium modification from OPN NC use upfront or an OPN NC-only strategy. We welcomed any additional evidence supporting the claim that that OPN NC provides optimal lumen expansion and the impact of using other calcium lesion modification devices prior to applying OPN NC to a patient.
With regard to safety, in the Natalia Pinilla-Echeverri, et al. (2023) study, one patient was found to have had a flow limiting dissection requiring stent deployment; however, no coronary perforations or no-reflow were reported. In the Secco, et al. (2019) study, three patients (0.9 percent) were reported to have experienced coronary rupture after balloon inflation and were successfully treated with stent implantation. In the Seiler, et al. (2023) study, coronary perforation was reported to have occurred twice (0.96 percent) with both successfully treated by balloon inflation and implantation of a covered stent; a total of nine (4.3 percent) locally limited, but flow limiting dissections were reported to have occurred and were successfully treated with implantation of a drug-eluting stent; 4 (1.9 percent) cases of flow deterioration due to embolization of thrombotic material (no-reflow) were found; and one patient (0.5 percent) was reported to have suffered from immediate vessel closure after stent implantation. The application did not address whether the use of the device is safe beyond the data on safety endpoints presented in the studies provided. We welcomed additional studies or evidence discussing the risk of adverse events with the use of these types of non-compliant balloons.
Finally, we expressed concern that the evidence may not demonstrate that ( printed page 94204) OPN NC substantially improves the treatment of an illness when compared to the benefits of other available treatments. The applicant asserted in a supporting document included in the application, that OPN NC is not the only FDA-authorized device with an indication for balloon dilatation of the stenotic portion of a coronary artery or bypass graft stenosis for the purpose of improving myocardial perfusion and also an indication for post deployment expansion of balloon expandable coronary stents. OPN NC was determined to be substantially equivalent to a legally marketed device, the NC Euphora Rapid Exchange Balloon Dilatation Catheter (Medtronic Inc; K141090), which received 510(k) clearance on August 15, 2014. The FDA 510(k) summary for OPN NC indicated that the devices share similar technological characteristics. In fact, the FDA 510(k) summary indicated that OPN NC differs only in the rated burst pressure of the balloon. We noted that the applicant did not compare the nominated device with the NC Euphora Rapid Exchange Balloon Dilatation Catheter, which we believe may be similar. While the applicant asserted that OPN NC is the only super-high pressure, non-compliant twin layer balloon dilatation catheter available in the U.S. and the only device on the market of this nature and capability, we stated we would be interested in additional information to demonstrate whether the nominated device demonstrates a substantial clinical benefit in comparison to other similar NC balloon devices.
We invited public comment on whether OPN NC meets the substantial clinical improvement criterion at § 419.66(c)(2).
Comment: We received multiple comments supporting the approval of OPN NC based on personal experience with the product and opinions on the clinical benefit of utilizing OPN NC. The commenters described their experiences using OPN NC and asserted that it works where other interventional strategies, such as traditional non-compliant balloons, atherectomy or intravascular lithotripsy have failed. The commenters added that they believe OPN NC is unique in allowing full expansion of extremely resistant lesions, particularly in the treatment of previous stents with restenosis due to under-expansion of underlying calcium and where the presence of the stent limits the use of technology such as atherectomy to modify the calcium. In addition, the commenters stated that in some cases OPN NC may be used as a stand-alone treatment, particularly in cases involving previously under-expanded stents. Further, the commenters asserted that they believe OPN NC is unique in achieving these results because it is a super high-pressure noncompliant balloon consisting of a double balloon layer and therefore, the rated burst pressure is substantially higher than that of a traditional non-compliant balloon. One commenter stated that the rated burst pressure for OPN NC is 35 atms of pressure, which is significantly higher than traditional non-compliant angioplasty balloons that have rated burst pressures of 20-24 atms of pressure. One commenter stated that OPN NC is specially designed to achieve 35-55 atms of pressure.[129] While this commenter addressed this device feature in reference to the § 419.66(b)(1) in their comment, we believe the comment was intended to address to address the substantial clinical improvement criterion, and therefore is included in the substantial clinical improvement discussion.
One commenter acknowledged that while the OPN NC device has not been tested in randomized trials, the commenter believes the device has demonstrated utility in cases of prior device failure, i.e., where conventional balloons have failed to dilate a lesion and effectively demonstrates its use case in selected circumstances. The commenter further commented that, as a result, they believe that the OPN NC balloon is substantially different from the predicate NC Euphora Rapid Exchange Balloon Dilatation Catheter.
Response: While we appreciate the commenters input regarding their experiences with OPN NC, we did not receive any additional data to address our concerns related to the applicant's claims of substantial clinical improvement. As such, we maintain our concerns listed in the CY 2025 OPPS/ASC proposed rule regarding the lack of randomized trials data, the lack of information about comparators, potential confounders not being accounted for, potential risk of adverse events, limited data supporting substantial improvement in the treatment of an illness, and insufficient evidence comparing OPN NC to its predicate. Several commenters asserted that OPN NC as an intervention yields results where other interventions failed. These commenters stated that OPN NC is unique in allowing full expansion of extremely resistant lesions. Commenters further stated that OPN NC is unique in treatment compared to previous stents with restenosis due to under-expansion of underlying calcium where the presence of the stent limits the use of technology such as atherectomy to modify the calcium. We note that we did not receive comments from the applicant. While we appreciate the information provided by the commenters, we note that no additional evidence was submitted to support the applicant's claims and we continue to have the same concerns discussed in the proposed rule.
After consideration of the public comments we received and our review of the application, we do not believe that OPN NC represents a substantial clinical improvement relative to existing therapies currently available and have determined that OPN NC does not meet the device eligibility criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that OPN NC would be reported with HCPCS codes shown in Table 127.
( printed page 94205)To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant did not provide details regarding the payment rates it applied for the three tests of the cost criterion. For our calculations, we used APC 5192, which had a CY 2023 payment rate of $5,215.40 at the time the application was received. HCPCS code 92920 in APC 5192 had a CY 2023 device offset amount of $1609.99 at the time the application was received.
We noted that the applicant provided two cost amounts for OPN NC: (1) a price list showing the cost of OPN NC as $2,200.00; and (2) a product list that lists the cost of OPN NC as $1,200.00. We further noted that the cost included on the product list provided by the applicant for OPN NC ($1,200.00) does not pass any of the three tests of the cost criterion, but the cost included on the price list for OPN NC ($2,200.00) passes all three tests of the cost criterion.
When performed with the price list cost for OPN NC of $2,200.00, we noted the following calculation outcomes: § 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $2,200.00 for OPN NC is 42.18 percent of the applicable APC payment amount for the service related to the category of devices of $5,215.40 (($2,200.00/$5,215.40) × 100 = 42.18 percent). Therefore, when utilizing the price list cost of $2,200.00 provided, we stated that we believe OPN NC meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $2,200.00 for OPN NC is 136.65 percent of the cost of the device-related portion of the APC payment amount for the related service of $1,609.99 ($2,200.00/$1,609.99) × 100 = 136.65 percent). Therefore, when utilizing the price list cost of $2,200.00 provided, we stated that we believe OPN NC meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $2,200.00 for OPN NC and the portion of the APC payment amount for the device of $1,609.99 is 11.31 percent of the APC payment amount for the related service of $5,215.40 ((($2,200.00−$1,609.99)/$5,215.40) × 100 = 11.31 percent). Therefore, when utilizing the price list cost of $2,200.00 provided, we stated that we believe OPN NC meets the third cost significance requirement.
When performed with the product list cost for OPN NC of $1,200.00, we noted the following calculation outcomes: § 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The average reasonable cost of $1,200.00 for OPN NC is 23.01 percent of the applicable APC payment amount for the service related to the category of devices of $5,215.40 (($1,200.00/$5,215.40) × 100 = 23.01 percent). Therefore, when utilizing the product list cost of $1,200.00 provided, we stated that we believe OPN NC does not meet the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $1,200.00 for OPN NC is 74.53 percent of the cost of the device-related portion of the APC payment amount for the related service of $1,609.99 ($1,200.00/$1,609.99) × 100 = 74.53 percent). Therefore, when utilizing the product list cost of $1,200.00 provided, we stated that we believe OPN NC does not meet the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $1,200.00 for OPN NC and the portion of the APC payment amount for the device of $1,609.99 is negative 7.86 percent of the APC payment amount for the related service of $5,215.40 ((($1,200.00−$1,609.99)/$5,215) × 100 = ( printed page 94206) −7.86 percent). Therefore, when utilizing the product list cost of $1,200.00 provided, we stated that we believe OPN NC does not meet the third cost significance requirement.
Based on the conflicting amounts provided for the reasonable cost of OPN NC, we questioned whether OPN NC meets the cost significance criterion. We welcomed additional information regarding this inconsistency on the estimated average reasonable cost of OPN NC.
We invited public comment on whether the OPN NC meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
We did not receive any comments regarding whether OPN NC meets the cost significance criteria as described at § 419.66(d). As noted in the proposed rule, the applicant provided two different amounts for the reasonable cost of OPN NC. For this final rule with comment period, we will use the submitted device cost of $2,200 stated in the price list in the proposed rule to perform our cost calculations. As such, based on our findings from the first, second, and third cost significance tests, we believe OPN NC meets the eligibility criterion at § 419.66(d).
After consideration of the public comments we received and our review of the device pass-through application, we are not approving OPN NC for transitional pass-through payment status in CY 2025 because the product does not meet eligibility criteria at § 419.66(c)(1) and (2).
(d) OSCAR® Peripheral Multifunctional Catheter
Biotronik, Inc. submitted an application for a new device category for transitional pass-through payment status for OSCAR® Peripheral Multifunctional Catheter (OSCAR®) for CY 2025. According to the applicant, OSCAR® is a tool used to simplify the treatment of peripheral artery disease (PAD), a disease process characterized by the narrowing of arteries that supply blood to the limbs, usually the legs. In severe cases, PAD can cause tissue death and gangrene, leading to amputation. Per the applicant, OSCAR® can simplify the process of peripheral interventions, reduce the time required to perform the procedure and the need for repeat procedures, reduce the risk of complications associated with changing out multiple medical devices, minimize radiation exposure, and enhance patient comfort.
Please refer to the online application posting for OSCAR®, available at https://mearis.cms.gov/public/publications/device-ptp/DEP230601F6NM2, for additional detail describing the device and the disease treated by the device.
Comment: A few commenters stated that OSCAR® meets the criteria for transitional pass-through payment. Commenters stated that they believe OSCAR® is highly effective and safe in crossing multiple occluded vessels of different morphologies above and below the knee, and is a necessary tool to treat patient with critical limb ischemia and calcific blockages of the arteries in the legs. One commenter asserted that OSCAR® will save money for hospitals because the device will result in less equipment use, faster procedure times, and higher rates of success and case completion.
Response: We appreciate the commenters' input and support for the OSCAR® application for transitional pass-through payment. We have taken these comments into consideration in our final determination regarding pass-through status for OSCAR®.
As stated previously, to be eligible for transitional pass-through payment under the OPPS, a device must meet the criteria at § 419.66(b)(1) through (4). With respect to the newness criterion at § 419.66(b)(1), on July 5, 2022, the applicant received 510(k) clearance from FDA for OSCAR® as a device to be used for percutaneous transluminal interventions in the peripheral vasculature to provide support during access into, and to dilate stenoses in, femoral, popliteal and infrapopliteal arteries. The product is also intended for injecting radiopaque contrast media for angiography. We received the application for a new device category for transitional pass-through payment status for OSCAR® on June 1, 2023, which is within 3 years of the date of the initial FDA marketing authorization.
We invited public comment on whether OSCAR® meets the newness criterion at § 419.66(b)(1).
Comment: With respect to the newness criterion at § 419.66(b)(1), the applicant reiterated that the pass-through payment application for OSCAR® is within the timeframe of 3 years post-FDA approval; therefore, the newness criterion at § 419.66(b)(1) is met.
Response: We appreciate the applicant's input. We received the application for a new device category for transitional pass-through payment status for OSCAR® on June 1, 2023, which is within 3 years of July 5, 2022, the date of FDA 510(k) clearance. Based on our review of the application, we have determined that OSCAR® meets the newness criterion at § 419.66(b)(1).
With respect to the eligibility criteria at § 419.66(b)(3), the device must be an integral part of the service furnished, used for one patient only, come in contact with human tissue, and be surgically inserted or implanted, or applied in or on a wound or other skin lesion. The applicant did not explicitly state whether OSCAR® is integral to the service provided. While the applicant did not explicitly state whether the device is used for one patient only or whether it comes in contact with human tissue, per the device description, OSCAR® is surgically inserted into the lower extremity peripheral vascular system and is single-use.
We invited public comments on whether OSCAR® meets the eligibility criterion at § 419.66(b)(3).
Comment: The applicant submitted a comment confirming that the OSCAR® meets the eligibility criterion at § 419.66(b)(3) because it is an integral part of the service furnished, is used for one patient only, comes into contact with human tissue, and is surgically inserted or implanted.
Response: We appreciate the applicant's input. We agree with the applicant that OSCAR® is an integral part of the service furnished, used for one patient only, comes in contact with human tissue, and is surgically implanted or inserted, or applied in or on a wound or other skin lesion. After consideration of the public comments received and our review of the application, we have determined that OSCAR® meets the eligibility criterion at § 419.66(b)(3).
With respect to the exclusion criterion at § 419.66(b)(4), a device is not eligible to be considered for device pass-through payment if it is any of the following: (1) equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or (2) a material or supply furnished incident to a service (for example, a suture, customized surgical kit, or clip, other than a radiological site marker). The applicant did not indicate whether OSCAR® is equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered, or if OSCAR® is a supply or material furnished incident to a service.
We invited public comment on whether OSCAR® meets the exclusion criterion at § 419.66(b)(4). ( printed page 94207)
Comment: The applicant submitted a comment confirming that OSCAR® meets the eligibility criterion at § 419.66(b)(4) because it is not equipment, an instrument, apparatus, implement, or item for which depreciation and financing expenses are recovered. The applicant also clarified that OSCAR® is not a supply or material furnished incident to a service.
Response: We appreciate the applicant's input. We agree with the applicant that OSCAR® is not equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciation assets, or a material or supply furnished incident to a service. After consideration of the public comments received and our review of the application, we have determined that OSCAR® meets the eligibility criterion at § 419.66(b)(4).
In addition to the criteria at § 419.66(b)(1) through (4), the criteria for establishing new device categories are specified at § 419.66(c). The first criterion, at § 419.66(c)(1), provides that CMS determines that a device to be included in the category is not appropriately described by any of the existing categories or by any category previously in effect, and was not being paid for as an outpatient service as of December 31, 1996. The applicant asserted that OSCAR® is a combination device authorized by FDA with an indication to diagnose and treat peripheral vascular lesions, identify obstructions, and cross the areas of obstruction and restore blood flow using a single system. According to the applicant, no previous or existing device categories for pass-through payment appropriately describe OSCAR®. Per the applicant, OSCAR® has the functionality of multiple devices currently used during lower extremity peripheral vasculature interventions. The applicant provided multiple HCPCS codes that could describe some of the components of OSCAR®; however, only one of the codes provided, C1725 (Catheter, transluminal angioplasty, non-laser (may include guidance, infusion/perfusion capability)), is a pass-through device category HCPCS code, and, therefore, C1725 is the only device category we evaluated for this criterion. Per the applicant, the device category C1725 does not appropriately describe OSCAR® because OSCAR® can cover the functionality of support catheters, several sizes of angioplasty balloons, chronic total occlusion crossing devices, reentry catheters, resistant lesion preparation devices, and dissection-reducing devices. According to the applicant, current pass-through coding does not adequately capture OSCAR®'s full functionality and the added clinical and economic value derived from its simplification of peripheral vascular interventions.
We noted, based on the description the applicant provided, that when the OSCAR® support catheter and OSCAR® dilator are combined with the OSCAR® PTA balloon, the device is used to complete a transluminal angioplasty, which is consistent with the devices described by C1725. In this context, we stated that we believe OSCAR® may be similar to the devices described by C1725 and, therefore, may be appropriately described by C1725.
We invited public comment on whether OSCAR® meets the device category criterion at § 419.66(c)(1).
Comment: In response to our concerns that OSCAR® may be appropriately described by C1725, the applicant commented that while some functionality of OSCAR® is captured in C1725, OSCAR®'s functionality of crossing and lesion treatment is not described by C1725. According to the applicant, C1725 does not adequately capture OSCAR®'s multi-functionality because while C1725 describes non-laser catheters used for transluminal angioplasty, which may include guidance, infusion, or perfusion capability, OSCAR® facilitates the steps of percutaneous transluminal angioplasty (PTA) procedures through lesion access, lesion crossing, and lesions of different length treatment, achieved using only a single device for the entire procedure. The applicant reiterated that OSCAR® consists of a support catheter, dilator, and PTA, providing the possibility for several different configurations during peripheral vasculature intervention. Further, the applicant noted that with this versatility, OSCAR® also provides features that are not available with other devices, such as user-adjustable guidewire support for accessing and crossing lesions and a length-adjustable balloon for lesion-specific angioplasty.
Response: We appreciate the applicant's input. After consideration of the public comments received and our review of the application, we continue to believe that C1725 appropriately describes OSCAR® because, as described by the applicant, when the OSCAR® support catheter and OSCAR® dilator are combined with the OSCAR® PTA balloon, the device is used to complete a transluminal angioplasty, which is consistent with the function of devices that may appropriately be described by C1725. The applicant asserted that OSCAR® provides features that are not available with other devices, such as a user-adjustable guidewire for accessing and crossing lesions and a length-adjustable balloon for lesion-specific angioplasty. However, based on the information provided, it remains unclear whether some of these additional items are simply supplies incidental to the service. If these additional items are supplies incidental to a service, the inclusion of these supplies along with a device to streamline a service does not materially differentiate the device from similar devices. Therefore, the multi-functionality differences of OSCAR® described by the applicant would not distinguish it from other devices described by the pass-through device category C1725.
After consideration of the public comments received and our review of the application, we continue to believe that C1725 appropriately describes OSCAR®. Therefore, we have determined that OSCAR® does not meet the device category eligibility criterion at § 419.66(c)(1) because it is appropriately described by an existing category or a category previously in effect.
Comment: The applicant asserted that device category C1725 does not appropriately describe OSCAR® because the functionality of OSCAR® allows the operator to perform several procedural steps with only one device, which improves procedural efficiency, reduces radiation exposure for patient and operator, and reduces the number of devices used to complete the procedure. The applicant also stated that OSCAR® adds clinical and economic value derived from its simplification of peripheral vascular interventions.
Response: We appreciate the applicant's input. We believe these comments are more appropriately related to the substantial clinical improvement criterion at § 419.66(c)(2), and, therefore, we will take these comments into consideration in that discussion below.
The second criterion for establishing a device category, at § 419.66(c)(2), provides that CMS determines either of the following: (i) that a device to be included in the category has demonstrated that it will substantially improve the diagnosis or treatment of an illness or injury or improve the functioning of a malformed body part compared to the benefits of a device or devices in a previously established category or other available treatment; or (ii) for devices for which pass-through status will begin on or after January 1, 2020, as an alternative to the substantial clinical improvement criterion, the ( printed page 94208) device is part of the FDA's Breakthrough Devices Program and has received FDA marketing authorization for the indication covered by the Breakthrough Device designation. The applicant claimed that OSCAR® represents a substantial clinical improvement over existing technologies in the diagnosis and management of peripheral artery disease because it uses less equipment, cuts down procedure time, and mitigates risks like vascular damage, infections, and radiation exposure, thereby enhancing clinical efficiency and safety.
The applicant provided four background documents supporting its substantial clinical improvement claim. Table 128 summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for OSCAR® for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.
After review of the information provided by the applicant, we stated we had the following concerns regarding whether OSCAR® meets the substantial clinical improvement criterion. First, the applicant did not submit peer-reviewed or published clinical evidence to substantiate clinical improvement over existing devices. The applicant submitted four background documents in support of OSCAR®: (1) a clinical benefit table, (2) a presentation on the Evaluation of Market Acceptance, (3) the OSCAR® U.S. Evaluation of Market Acceptance Report, and the (4) OSCAR® Clinical Evaluation Report. All four of these documents rely on data from the Evaluation of Market Acceptance. We noted these documents are not published or peer-reviewed, and reflect data collected for marketing purposes rather than clinical improvement purposes. The data included appear to be opinion-based survey questions asked of physicians recruited by the applicant for the Evaluation of Market Acceptance and noted that these documents suggest an implicit bias. We questioned the link between these documents and the claims the applicant made that OSCAR® shows substantial clinical improvement because it uses less equipment, cuts down procedure ( printed page 94209) time, and mitigates risks like vascular damage, infections, and radiation exposure, thereby enhancing clinical efficiency and safety. We requested clarification on how the supporting documents directly relate to the substantial clinical improvement claims.
Further, we questioned how a collection of devices currently available on the market consolidated into a single packaged product demonstrates substantial clinical improvement. According to the applicant, with OSCAR®, some procedures may be performed with a single device which cuts down procedure time and mitigates risks like vascular damage, infections, and radiation exposure, thereby enhancing clinical efficiency and safety. The applicant asserted several benefits of using OSCAR® over multiple devices, including reducing (1) the need to remove and replace multiple devices, which may reduce the incidence of complications like infection and vessel damage; (2) the need to use ill-fitting devices; (3) the need for multiple guidewires in several procedures; and (4) the incidence of complications, such as infections and vessel damage. However, we did not receive comparative data supporting the claim that OSCAR® offers superiority over currently available treatments in terms of clinical benefit or safety. The evidence provided did not discuss any advantages of using a single system of devices rather than multiple individual devices with diverse functionalities. We welcomed any additional evidence supporting these claims.
Furthermore, per the applicant, OSCAR® is effective in preparing intravascular lesions for advanced interventions, particularly stenting, and by ensuring optimal lesion preparation, OSCAR® elevates the success rate of these procedures, enhances patient safety, and streamlines institutional operations. According to the applicant, OSCAR® can treat chronic total occlusions (CTOs) and incorporates reentry capabilities, features traditionally found in standalone devices. The applicant asserted this integration enhances patient safety, simplifies procedures, and elevates the efficiency of operations. However, we noted that the applicant did not provide clinical information in support of these claims. Again, we welcomed any additional evidence supporting these claims.
In addition, we questioned whether OSCAR® can be sufficiently distinguished from similar existing technologies to demonstrate substantial clinical improvement. OSCAR® was determined to be substantially equivalent to a legally marketed device, the INFINITY Angioplasty Balloon CatheterTM, which received 510(k) clearance on May 20, 2020. The FDA 510(k) summary for OSCAR® indicated that the devices share similar technological characteristics and that OSCAR® differs only in that it combines support catheters to be used with the dilator and balloon catheter. We did not receive data demonstrating how OSCAR® offers a substantial clinical improvement compared to the INFINITY Angioplasty Balloon CatheterTM . We stated that we would be interested in additional information to demonstrate whether the nominated device demonstrates a substantial clinical benefit in comparison to INFINITY Angioplasty Balloon CatheterTM.
Finally, per the applicant, there are six device types that it believed OSCAR® is most closely related to: (1) workhorse guidewires (Abbott, Boston Scientific, Terumo, Medtronic, Biotronik, Cook Medical, Cordis); (2) premium guidewires (Abbott, Asahi Intecc, Boston Scientific, Cook Medical and more); (3) workhorse & premium support catheters (Philips, Boston Scientific, Cook Medical, Medtronic, Asahi Intecc, Teleflex and more); (4) angioplasty balloons (Abbott, BD Interventional, Biotronik, Cook Medical, Medtronic and more); (5) lesion preparation balloons (Philips, Medtronic, BD Interventional and Cagent Vascular); and (6) chronic total occlusion and reentry devices. We stated that we do not believe that OSCAR® is similar to the workhorse guidewires and premium guidewires listed because OSCAR® does not include guidewires. We welcomed additional information illustrating how OSCAR® is similar to the listed workhorse guidewires and premium guidewires and evidence demonstrating the benefits of OSCAR® over these other devices.
While we noted that OSCAR® is comparable to other available technologies, we did not receive data demonstrating how OSCAR® offers a substantial clinical improvement compared to the workhorse guidewires and premium support catheters, angioplasty balloons, lesion preparation balloons, or chronic total occlusion and reentry devices and stated that we would be interested in additional evidence demonstrating the substantial clinical benefits of OSCAR® over these other devices.
We stated that additional evidence comparing OSCAR® to existing technologies would be particularly helpful to determine whether the device demonstrates substantial clinical improvement over currently available treatments in the clinical setting where it is most likely to be used. Specifically, we welcomed published peer-reviewed clinical trials that show improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process compared to the standard of care.
We invited public comment on whether OSCAR® meets the device category criterion at § 419.66(c)(2)(i).
Comment: In response to our concerns that the applicant did not provide sufficient peer-reviewed or published evidence to substantiate OSCAR®'s clinical improvement over existing devices, the applicant commented that other data sources are acceptable for transitional pass-through payment applications to prove substantial clinical improvement. Specifically, the applicant asserted that the provided documents show two sources of evidence: internal benchmark tests and a real-world user evaluation.
Per the applicant, the clinical benefit table provided summarizes the industry's state-of-the-art device procedural success and complication rates from published literature about similarly marketed devices. The applicant asserted that the real-world market evaluation compares OSCAR®'s internal benchmark tests to these published state-of-the-art benchmarks to determine whether OSCAR® met or exceeded the benchmarks. To evaluate market acceptance, the applicant explained that it conducted internal benchmark tests of OSCAR® in a real-world setup, without any exclusion criteria in terms of lesion characteristic and patient population. The applicant noted that it did this to obtain the opinion of different users—a method of user experience testing that is well-accepted in the field. Furthermore, the applicant stated that the real-world user experience data showed that OSCAR® has the potential to simplify procedures with one platform, which could reduce device escalations and lead to procedural efficiencies. According to the applicant, this potential simplification is especially important as staffing shortages and availability issues continue to plague U.S. facilities.
Response: We appreciate the applicant's input. We acknowledge that data sources other than peer-reviewed or published studies may be submitted for the purpose of supporting substantial clinical improvement in ( printed page 94210) transitional pass-through payment applications. CMS considers all submitted evidence when reviewing transitional pass-through payment applications, however, we note that higher levels of evidence, such as peer-reviewed controlled studies, will typically be more supportive and in some cases may be required to establish a substantial clinical improvement argument. We continue to believe that the predominance of the data submitted appears to be opinion-based survey questions asked of physicians for marketing purposes, is likely subject to implicit bias, and falls significantly short of demonstrating substantial clinical improvement. In addition, we do not believe that the documents submitted by the applicant demonstrate that OSCAR® mitigates risks like vascular damage, infections, and radiation exposure as claimed. Finally, we do not believe that the applicant has provided an appropriate level of evidence that the use of OSCAR® reduces procedure time, nor has the applicant established that a potential reduction in procedure time and/or the use of less equipment translates to a substantial improvement in treatment of the affected patient population. After consideration of the public comment received and our review of the application, we continue to have concerns about whether OSCAR® demonstrates substantial clinical improvement, and we believe that additional peer-reviewed evidence would be helpful.
Comment: In response to our concern whether OSCAR® can be sufficiently distinguished from a legally marketed device, the INFINITY Angioplasty Balloon CatheterTM, to demonstrate substantial clinical improvement, the applicant commented that OSCAR® is not identical to the INFINITY Angioplasty Balloon CatheterTM . Specifically, the applicant asserted that, in its 510(k) approval summary, FDA stated that OSCAR® is comparable to the predicate device rather than equivalent. According to the applicant, the term comparable indicates that OSCAR® has similar characteristics and intended use as the predicate device, but it does not imply that the two devices are identical. The applicant asserted that the INFINITY Angioplasty Balloon CatheterTM is indicated for PTA in the peripheral vasculature, including iliac, femoral, popliteal, infra-popliteal arteries, and for the treatment of obstructive lesions of native or synthetic arteriovenous dialysis fistulae; whereas OSCAR® facilitates the steps of PTA procedures (lesion access, cross and treat lesions of different length) using a single device for the entire procedure to reduce procedural time and simplify steps.
Per the applicant, OSCAR® was designed to combine features of other marketed devices, such as support catheters and PTA balloon catheters. The applicant asserted the main benefits of OSCAR® are (1) the device's variable shaft stiffness which allows for lesion-adapted access and crossing, and (2) its length adjustability features which allow for precise adjustment of the balloon length to match the target lesion's length. The applicant further explained that these features allow OSCAR® to treat residual stenosis using a single balloon, rather than the typical treatment's multiple balloons, resulting in the potential benefits of lower device consumption, fewer device exchanges, reduced overall procedural time, and fewer stock-keeping units needed in the catheterization laboratory.
Response: We appreciate the applicant's input. While FDA determination of substantial equivalence cannot alone be used to conclude that a device demonstrates substantial clinical improvement as required by the regulation at 42 CFR 419.66(c)(2), we note that the FDA 510(k) summary provided by the applicant indicated that both the nominated and predicate devices share similar technological characteristics and that OSCAR® differs only in that it combines support catheters to be used with the dilator and balloon catheter. Specifically, OSCAR® was determined to be substantially equivalent to a legally marketed device, the INFINITY Angioplasty Balloon CatheterTM, which received 510(k) clearance on May 20, 2020. The FDA 510(k) summary for OSCAR® indicated that the devices share similar technological characteristics and that OSCAR® differs only in that it combines support catheters to be used with the dilator and balloon catheter. While the applicant asserted that treatment of complex lesions with OSCAR® reduces device exchanges and procedural steps, procedure time, radiation exposure, contrast media, and crossing attempts, the applicant did not provide evidence that demonstrated how these reductions result in improved clinical outcomes. We did not receive data demonstrating how OSCAR® offers a substantial clinical improvement compared to the INFINITY Angioplasty Balloon CatheterTM . We expressed concern in the proposed rule regarding the language in the FDA 510(k) summary because we could not determine whether OSCAR® could be distinguished from the INFINITY Angioplasty Balloon CatheterTM sufficiently to demonstrate substantial clinical improvement. Neither could we determine exactly how the nominated device is superior to its earlier legally marketed device, as per the applicant's assertion. After consideration of the public comment received and our review of the application, we maintain our concern that OSCAR® does not demonstrate a substantial clinical improvement compared to the predicate device, INFINITY Angioplasty Balloon CatheterTM.
Comment: In response to our concerns that OSCAR® may be substantially similar to six device types that the applicant indicated OSCAR® is most closely related to, the applicant commented that OSCAR® reduces the need for multiple devices and may lower procedural costs. Specifically, the applicant clarified that OSCAR® requires a guidewire but replaces the need for support catheters and percutaneous balloons and may replace the need for chronic total occlusion devices, reentry catheters, or lesion preparation devices. In response to our request that the applicant submit additional evidence comparing OSCAR®'s clinical outcomes to those of existing technologies to help us determine whether the device demonstrates substantial clinical improvement, the applicant commented that this request is unreasonable with these types of tools in percutaneous transluminal interventions. Specifically, the applicant asserted that, in below-the-knee interventions, global user experience has shown OSCAR® to be definitive as it allows length adjustability, which they believe is an improvement over other balloon catheters used for angioplasty. The applicant concluded that real-world user experience has shown that OSCAR® aids in the treatment of complex lesions through the reduction of device exchanges and procedural steps, reduction of procedure time, reduction of radiation exposure, reduction of contrast media, and fewer crossing attempts.
Response: We appreciate the applicant's input. While the applicant asserted, both for this § 419.66(c)(2) criterion and the § 419.66(c)(1) criterion discussed above, that OSCAR® may reduce the need for support catheters, percutaneous balloons, chronic total occlusion devices, reentry catheters, or lesion preparation devices during percutaneous transluminal interventions, the applicant did not provide evidence demonstrating that OSCAR®'s potential to reduce the ( printed page 94211) number of devices improved clinical outcomes for patients with PAD. We maintain that these potential reductions in procedural time and cost compared to other devices do not translate to improved clinical outcomes, such as reduction in mortality, complications, subsequent interventions, future hospitalizations, recovery time, pain, or a more rapid beneficial resolution of the disease process, required to demonstrate substantial clinical improvement.
Regarding the applicant's statements that OSCAR® will save money for hospitals because hospitals will use less equipment, procedure times will be faster, and procedures will have higher success rates and case completion and, as a result, patients will avoid limb amputation, we appreciate the commenters' input. However, while reducing costs to the healthcare system is important, it is not a consideration for substantial clinical improvement for purposes of transitional pass-through payments. With respect to the higher success rates, case completion, and decrease in limb amputations, which could demonstrate substantial clinical improvement, the commenters did not provide evidence to support these claims. Similarly, regarding faster procedure times, no link was demonstrated between this and a substantial clinical improvement outcome, nor was evidence provided to support this claim. As such, we maintain our concern about whether the use of OSCAR® has a substantial clinical benefit compared to the six existing device types noted in the application and over other similar existing technologies.
After consideration of the public comments received and our review of the application, we continue to believe the applicant has not demonstrated substantial clinical improvement over existing technologies in the treatment of PAD. Therefore, we have determined that OSCAR® does not meet the substantial clinical improvement criterion at § 419.66(c)(2).
The third criterion for establishing a device category, at § 419.66(c)(3), requires us to determine that the cost of the device is not insignificant, as described in § 419.66(d). Section 419.66(d) includes three cost significance criteria that must each be met. The applicant provided the following information in support of the cost significance requirements. The applicant stated that OSCAR® would be reported with HCPCS codes as shown in Table 129.
( printed page 94212) ( printed page 94213)To meet the cost criterion for device pass-through payment status, a device must pass all three tests of the cost criterion for at least one APC. As we explained in the CY 2005 OPPS final rule (69 FR 65775), we generally use the lowest APC payment rate applicable for use with the nominated device when we assess whether a device meets the cost significance criterion, thus increasing the probability the device will pass the cost significance test. Beginning in CY 2017, we calculate the device offset amount at the HCPCS/CPT code level instead of the APC level (81 FR 79657). We noted that the applicant used the CY 2023 payment rates for the three tests of the cost criterion. For our calculations, we used APC 5183, which had a CY 2023 payment rate of $2,978.97 at the time the application was received. HCPCS code 75625 in APC 5183 had a CY 2023 device offset amount of $530.85 at the time the application was received.[130] According to the applicant, the cost of OSCAR® is $2,020.00.
Section 419.66(d)(1), the first cost significance requirement, provides that the estimated average reasonable cost of devices in the category must exceed 25 percent of the applicable APC payment amount for the service related to the category of devices. The estimated average reasonable cost of $2,020.00 for OSCAR® is 67.81 percent of the applicable APC payment amount for the service related to the category of devices of $2,978.97 (($2,020.00/$2,978.97) × 100 = 67.81 percent). Therefore, we stated that we believe OSCAR® meets the first cost significance requirement.
The second cost significance requirement, at § 419.66(d)(2), provides that the estimated average reasonable cost of the devices in the category must exceed the cost of the device-related portion of the APC payment amount for the related service by at least 25 percent, which means that the device cost needs to be at least 125 percent of the offset amount (the device-related portion of the APC found on the offset list). The estimated average reasonable cost of $2,020.00 for OSCAR® is 380.52 percent of the cost of the device-related portion ( printed page 94214) of the APC payment amount for the related service of $530.85 (($2,020.00/$530.85) × 100 = 380.52 percent). Therefore, we stated that we believe OSCAR® meets the second cost significance requirement.
The third cost significance requirement, at § 419.66(d)(3), provides that the difference between the estimated average reasonable cost of the devices in the category and the portion of the APC payment amount for the device must exceed 10 percent of the APC payment amount for the related service. The difference between the estimated average reasonable cost of $2,020.00 for OSCAR® and the portion of the APC payment amount for the device of $530.85 is 49.99 percent of the APC payment amount for the related service of $2978.97 ((($2,020.00−$530.85)/$ 2,978.97) × 100 = 49.99 percent). Therefore, we stated that we believe OSCAR® meets the third cost significance requirement.
We invited public comment on whether OSCAR® meets the device pass-through payment criteria discussed in this section, including the cost criterion for device pass-through payment status.
We did not receive any comments regarding whether OSCAR® meets the cost significance criteria as described at § 419.66(d). Based on our findings from the first, second, and third cost significance tests, we have determined that OSCAR® meets the cost significance criteria specified at § 419.66(d).
After consideration of the public comments, we received and our review of the device pass-through application, we are not approving OSCAR® for transitional pass-through payment status in CY 2025 because the product does not meet the device category eligibility criterion at § 419.66(c)(1) or the substantial clinical improvement criterion at § 419.66(c)(2).
Comment: The commenter recommended CMS calculate the device-related portion of APCs for purposes of determining device pass-through payment status eligibility and the device offset using only the cost of the devices replaced by the proposed device pass-through payment status device category. The commenter stated that many procedures with transitional pass-through devices require the use of multiple other devices, and the transitional pass-through device may replace only some (and in some cases none) of the devices included in the device-related portion of the APC. The commenter asserted that, if the device offset is calculated to include other devices that continue to be required to perform the procedure safely and successfully, without properly offsetting the cost of the nominated device, the final payment would fail to adequately reimburse facilities. The commenter requested that CMS also apply this methodology to the TPT applications for AGENTTM Paclitaxel-Coated Balloon Catheter, iFuse Bedrock GraniteTM Implant System, Paradise® Ultrasound RDN System, and Symplicity SpyralTM RDN System applications.
Response: We thank the commenter's recommendation regarding the revision of the methodology for calculating the device-related portion of APCs for the purpose of determining the eligibility of transitional pass-through status of a nominated device. We will continue to consider this issue and any additional public comments related to it.
B. Device-Intensive Procedures
1. Background
Under the OPPS, prior to CY 2017, device-intensive status for procedures was determined at the APC level for APCs with a device offset percentage greater than 40 percent (79 FR 66795). Beginning in CY 2017, CMS began determining device-intensive status at the HCPCS code level. In assigning device-intensive status to an APC prior to CY 2017, the device costs of all the procedures within the APC were calculated and the geometric mean device offset of all of the procedures had to exceed 40 percent. Almost all of the procedures assigned to device-intensive APCs utilized devices, and the device costs for the associated HCPCS codes exceeded the 40-percent threshold. The no cost/full credit and partial credit device policy (79 FR 66872 through 66873) applies to device-intensive procedures and is discussed in detail in section IV.B.4 of this final rule with comment period. A related device policy was the requirement that certain procedures assigned to device-intensive APCs require the reporting of a device code on the claim (80 FR 70422) and is discussed in detail in section IV.B.3 of this final rule with comment period. For further background information on the device-intensive APC policy, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70421 through 70426).
a. HCPCS Code-Level Device-Intensive Determination
As stated earlier, prior to CY 2017, under the device-intensive methodology we assigned device-intensive status to all procedures requiring the implantation of a device that were assigned to an APC with a device offset greater than 40 percent and, beginning in CY 2015, that met the three criteria as listed. Historically, the device-intensive designation was at the APC level and applied to the applicable procedures within that APC. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79658), we changed our methodology to assign device-intensive status at the individual HCPCS code level rather than at the APC level. Under this policy, a procedure could be assigned device-intensive status regardless of its APC assignment, and device-intensive APC designations were no longer applied under the OPPS or the ASC payment system.
We believe that a HCPCS code-level device offset is, in most cases, a better representation of a procedure's device cost than an APC-wide average device offset based on the average device offset of all of the procedures assigned to an APC. Unlike a device offset calculated at the APC level, which is a weighted average offset for all devices used in all of the procedures assigned to an APC, a HCPCS code-level device offset is calculated using only claims for a single HCPCS code. We believe that this methodological change results in a more accurate representation of the cost attributable to implantation of a high-cost device, which ensures consistent device-intensive designation of procedures with a significant device cost. Further, we believe a HCPCS code-level device offset removes inappropriate device-intensive status for procedures without a significant device cost that are granted such status because of their APC assignment.
Under our existing policy, procedures that meet the criteria listed in section IV.C.1.b of this final rule with comment period are identified as device-intensive procedures and are subject to all the policies applicable to procedures assigned device-intensive status under our established methodology, including our policies on device edits and no cost/full credit and partial credit devices discussed in sections IV.C.3 and IV.C.4 of this final rule with comment period.
b. Use of the Three Criteria To Designate Device-Intensive Procedures
We clarified our established policy in the CY 2018 OPPS/ASC final rule with comment period (82 FR 52474), where we explained that device-intensive procedures require the implantation of a device and additionally are subject to the following criteria:
- All procedures must involve implantable devices that would be reported if device insertion procedures were performed. ( printed page 94215)
- The required devices must be surgically inserted or implanted devices that remain in the patient's body after the conclusion of the procedure (at least temporarily); and
- The device offset amount must be significant, which is defined as exceeding 40 percent of the procedure's mean cost.
We changed our policy to apply these three criteria to determine whether procedures qualify as device-intensive in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66926), where we stated that we would apply the no cost/full credit and partial credit device policy—which includes the three criteria listed previously—to all device-intensive procedures beginning in CY 2015. We reiterated this position in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70424), where we explained that we were finalizing our proposal to continue using the three criteria established in the CY 2007 OPPS/ASC final rule with comment period for determining the APCs to which the CY 2016 device intensive policy will apply. Under the policies we adopted in CYs 2015, 2016, and 2017, all procedures that require the implantation of a device and meet the previously described criteria are assigned device-intensive status, regardless of their APC placement.
2. Device-Intensive Procedure Policy
As part of our effort to better capture costs for procedures with significant device costs, in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58944 through 58948), for CY 2019, we modified our criteria for device-intensive procedures. We had heard from interested parties that the criteria excluded some procedures that interested parties believed should qualify as device-intensive procedures. Specifically, we were persuaded by interested party arguments that procedures requiring expensive surgically inserted or implanted devices that are not capital equipment should qualify as device-intensive procedures, regardless of whether the device remains in the patient's body after the conclusion of the procedure. We agreed that a broader definition of device-intensive procedures was warranted, and made two modifications to the criteria for CY 2019 (83 FR 58948). First, we allowed procedures that involve surgically inserted or implanted single-use devices that meet the device offset percentage threshold to qualify as device-intensive procedures, regardless of whether the device remains in the patient's body after the conclusion of the procedure. We established this policy because we no longer believe that whether a device remains in the patient's body should affect a procedure's designation as a device-intensive procedure, as such devices could, nonetheless, comprise a large portion of the cost of the applicable procedure. Second, we modified our criteria to lower the device offset percentage threshold from 40 percent to 30 percent, to allow a greater number of procedures to qualify as device intensive. We stated that we believe allowing these additional procedures to qualify for device-intensive status will help ensure these procedures receive more appropriate payment in the ASC setting, which will help encourage the provision of these services in the ASC setting. In addition, we stated that this change would help to ensure that more procedures containing relatively high-cost devices are subject to the device edits, which leads to more correctly coded claims and greater accuracy in our claims data. Specifically, for CY 2019 and subsequent years, we finalized that device-intensive procedures will be subject to the following criteria:
- All procedures must involve implantable devices assigned a CPT or HCPCS code;
- The required devices (including single-use devices) must be surgically inserted or implanted; and
- The device offset amount must be significant, which is defined as exceeding 30 percent of the procedure's mean cost (83 FR 58945).
In addition, to further align the device-intensive policy with the criteria used for device pass-through payment status, we finalized, for CY 2019 and subsequent years, that for purposes of satisfying the device-intensive criteria, a device-intensive procedure must involve a device that:
- Has received FDA marketing authorization, has received an FDA investigational device exemption (IDE), and has been classified as a Category B device by FDA in accordance with §§ 405.203 through 405.207 and 405.211 through 405.215, or meets another appropriate FDA exemption from premarket review;
- Is an integral part of the service furnished;
- Is used for one patient only;
- Comes in contact with human tissue;
- Is surgically implanted or inserted (either permanently or temporarily); and
- Is not either of the following:
++ Equipment, an instrument, apparatus, implement, or item of the type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or
++ A material or supply furnished incident to a service (for example, a suture, customized surgical kit, scalpel, or clip, other than a radiological site marker) (83 FR 58945).
In addition, for new HCPCS codes describing procedures requiring the implantation of devices that do not yet have associated claims data, in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79658), we finalized a policy for CY 2017 to apply device-intensive status with a default device offset set at 41 percent for new HCPCS codes describing procedures requiring the implantation or insertion of a device that did not yet have associated claims data until claims data are available to establish the HCPCS code-level device offset for the procedures. This default device offset amount of 41 percent was not calculated from claims data; instead, it was applied as a default until claims data were available upon which to calculate an actual device offset for the new code. The purpose of applying the 41-percent default device offset to new codes that describe procedures that implant or insert devices was to ensure ASC access for new procedures until claims data become available.
As discussed in the CY 2019 OPPS/ASC proposed rule and final rule with comment period (83 FR 37108 through 37109 and 58945 through 58946, respectively), in accordance with our policy stated previously to lower the device offset percentage threshold for procedures to qualify as device-intensive from greater than 40 percent to greater than 30 percent, for CY 2019 and subsequent years, we modified this policy to apply a 31-percent default device offset to new HCPCS codes describing procedures requiring the implantation of a device that do not yet have associated claims data until claims data are available to establish the HCPCS code-level device offset for the procedures. In conjunction with the policy to lower the default device offset from 41 percent to 31 percent, we continued our current policy of, in certain rare instances (for example, in the case of a very expensive implantable device), temporarily assigning a higher offset percentage if warranted by additional information such as pricing data from a device manufacturer (81 FR 79658). Once claims data are available for a new procedure requiring the implantation or insertion of a device, device-intensive status is applied to the code if the HCPCS code-level device offset is greater than 30 percent, ( printed page 94216) according to our policy of determining device-intensive status by calculating the HCPCS code-level device offset.
In addition, in the CY 2019 OPPS/ASC final rule with comment period, we clarified that since the adoption of our policy in effect as of CY 2018, the associated claims data used for purposes of determining whether or not to apply the default device offset are the associated claims data for either the new HCPCS code or any predecessor code, as described by CPT coding guidance, for the new HCPCS code. Additionally, for CY 2019 and subsequent years, in limited instances where a new HCPCS code does not have a predecessor code as defined by CPT, but describes a procedure that was previously described by an existing code, we use clinical discretion to identify HCPCS codes that are clinically related or similar to the new HCPCS code but are not officially recognized as a predecessor code by CPT, and to use the claims data of the clinically related or similar code(s) for purposes of determining whether or not to apply the default device offset to the new HCPCS code (83 FR 58946). Clinically related and similar procedures for purposes of this policy are procedures that have few or no clinical differences and use the same devices as the new HCPCS code. In addition, clinically related and similar codes for purposes of this policy are codes that either currently or previously describe the procedure described by the new HCPCS code. Under this policy, claims data from clinically related and similar codes are included as associated claims data for a new code, and where an existing HCPCS code is found to be clinically related or similar to a new HCPCS code, we apply the device offset percentage derived from the existing clinically related or similar HCPCS code's claims data to the new HCPCS code for determining the device offset percentage. We stated that we believe that claims data for HCPCS codes describing procedures that have minor differences from the procedures described by new HCPCS codes will provide an accurate depiction of the cost relationship between the procedure and the device(s) that are used, and will be appropriate to use to set a new code's device offset percentage, in the same way that predecessor codes are used. If a new HCPCS code has multiple predecessor codes, the claims data for the predecessor code that has the highest individual HCPCS-level device offset percentage is used to determine whether the new HCPCS code qualifies for device-intensive status. Similarly, in the event that a new HCPCS code does not have a predecessor code but has multiple clinically related or similar codes, the claims data for the clinically related or similar code that has the highest individual HCPCS level device offset percentage is used to determine whether the new HCPCS code qualifies for device-intensive status.
a. Change to the Device-Intensive Status Default Offset Methodology for New HCPCS Codes
As described above, under our existing policies for assigning a device offset percentage to new HCPCS codes, we first rely on the associated claims data for new HCPCS codes. For new HCPCS codes that do not have available claims data yet, we rely on any available claims data from a predecessor code for the new HCPCS code, as described by CPT coding guidance. We assign the device offset percentage to the new HCPCS code that is the device offset percentage of the predecessor code for which we have available claims data. If claims data from the new HCPCS or any predecessor code is unavailable, we use clinical discretion to identify HCPCS codes that are clinically related or similar to the new HCPCS code but are not officially recognized as a predecessor code by CPT, and to use the claims data of the clinically related or similar code(s) for purposes of determining a device offset percentage to the new HCPCS code (83 FR 58946). Clinically related and similar procedures for purposes of this policy are procedures that have few or no clinical differences and use the same devices. If a clinically similar procedure that uses the same devices is not available, then for new HCPCS codes describing procedures requiring the insertion or implantation of devices that do not yet have claims data (from either the new HCPCS code or any predecessor code), we apply a default device offset set at 31 percent.
As we stated previously, the purpose of applying the default device offset to new codes that describe procedures that implant or insert devices is to ensure access in the ASC setting for new procedures until claims data become available. Also, under the OPPS, the default device offset is useful for establishing a device amount for new device-intensive procedures. For example, under our policy for no cost/full credit or partial credit devices, we reduce the OPPS payment for device-intensive procedures by the lesser of the full or partial credit a hospital receives for a replaced device or the device offset amount. Additionally, we may remove the device offset amount from the OPPS payment for procedures that are terminated prior to administering anesthesia (since the device was not used for the procedure).
While we do allow for additional information in consideration of a higher offset percentage than the default device offset, it would be extremely rare that the appropriate determination of a device offset percentage would rely on pricing data or invoices from a device manufacturer rather than the default device offset percentage. However, we are aware that there may be certain situations where the default device offset percentage would not adequately reflect the existing device portion of the procedure's costs when compared to the cost of similar devices. This difference could impede our ability to accurately remove device offset amounts from new device-intensive procedures under the OPPS. As HOPDs and ASCs perform new procedures with significant device costs, we believe it is appropriate to modify our default device offset methodology to pay HOPDs and ASCs more appropriately when we lack claims data for these newer procedures. Therefore, for CY 2025 and subsequent calendar years, we proposed to modify our default device offset percentage policy for new device-intensive procedures. Specifically, for new HCPCS codes that both describe a procedure that requires the implantation or insertion of a single-use device that meets our requirements of a device as described above and lack claims data (from either the new HCPCS code or any predecessor code), we would apply a default device offset percentage that is the greater of 31 percent or the device offset percentage of the APC to which the procedure has been assigned. We proposed this methodological change for both the OPPS and ASC Payment System for CY 2025 and subsequent calendar years. We explained that we still believe that a HCPCS code-level device offset is, in most cases, a more accurate representation of a procedure's device cost than an APC-wide average device offset based on the average device offset of all the procedures assigned to an APC. However, because newer device-intensive procedures lack claims data, we believe the APC-wide average device offset percentage is, in many cases, a better reflection of the estimated device costs of the procedure than a default 31 percent offset. Additionally, there can be instances where the typical device costs of procedures in an APC can be significantly greater than the 31 percent default device offset. For these reasons, we proposed to modify our default device offset percentage for new device- ( printed page 94217) intensive procedures that describe the implantation or insertion of a single-use device that meets our requirements of a device (as described above) and that do not yet have associated claims data, by applying a default device offset percentage that is the greater of 31 percent or the device offset percentage of the APC to which the procedure has been assigned. We proposed to apply this policy to new procedures assigned to clinical APCs, but not to new procedures assigned to New Technology APCs.
As we indicated in the CY 2019 OPPS/ASC proposed rule and final rule with comment period, we may consider additional information for an offset percentage greater than the default offset percentage (which, for the proposed rule, is the greater of 31 percent or the APC-level offset percentage) for new HCPCS codes describing procedures requiring the implantation (or, in some cases, the insertion) of a device that do not yet have associated claims data, such as pricing data or invoices from a device manufacturer. This would be for our consideration in extremely rare circumstances, such as an extremely high-cost implantable device. While we explained that we believe our proposed modification to the default device offset policy would improve payment under the OPPS and ASC payment system, we would continue to accept additional information in consideration of an alternative offset percentage. This information would be directed to the Division of Outpatient Care, Mail Stop C4-01-26, Centers for Medicare & Medicaid Services, 7500 Security Boulevard, Baltimore, MD 21244-1850, or electronically at outpatientpps@cms.hhs.gov. Additional information would be submitted prior to issuance of an OPPS/ASC proposed rule or as a public comment in response to an issued OPPS/ASC proposed rule. Device offset percentages would be set in each year's final rule with comment period.
Comment: While commenters supported our proposed change to determining default device offset percentages under the ASC payment system, many commenters recommended that we delay implementing our proposed change under the OPPS as we have not fully discussed the implications of such a change under the OPPS. Some stakeholders were concerned that our proposed default device offset methodology could inappropriately inflate the device offset amount for transitional pass-through cost significance tests, which utilize the device offset amounts under the OPPS.
Response: Under the OPPS, our proposal, if finalized, would apply whenever a default device offset percentage and the associated device offset amount are utilized. This includes determining eligibility for transitional pass-through payment status for devices, setting the payment amount for transitional pass-through devices, determining the payment amount for discontinued device-intensive procedures, and determining the maximum credit amount for a device-intensive procedure under our device credits policy. The purpose of our proposal was to improve the accuracy of the way in which we determine the device portion for device-intensive procedures in the absence of claims data. We believe the average device cost of the APC can better reflect the expected device portion of a new procedure without claims data than a default device offset of 31 percent since procedures assigned to a clinical APC often have similar device costs. We believe this improvement in the accuracy of determining the device portion of an APC payment should be applied in both settings rather than just one setting as the commenters recommend. We understand that the increased device portion that would result from this change in policy may reduce payment in the OPPS setting if such newer procedures are reported with a transitional pass-through device category or in situations with a device credit or a terminated procedure, but we do not believe it would be appropriate to assign a device offset amount that we believe is less accurate to avoid such a payment reduction under the OPPS. Since our policy has been to keep device costs constant between the OPPS and ASC payment system for device-intensive procedures to appropriately account for the device costs of certain surgical procedures and maintain beneficiary access in the ASC setting for device-intensive procedures, for which interested parties have expressed strong support, we are not accepting the commenter's recommendation to delay implementing our proposed change under the OPPS.
Comment: Several commenters noted that the device offset percentage for HCPCS code C9757 (Laminotomy (hemilaminectomy), with decompression of nerve root(s), including partial facetectomy, foraminotomy and excision of herniated intervertebral disc, and repair of annular defect with implantation of bone anchored annular closure device, including annular defect measurement, alignment and sizing assessment, and image guidance; 1 interspace, lumbar) was inaccurate and did not reflect the total costs for the procedure. The manufacturer of the Barricaid device—a device which is used with HCPCS code C9757—noted that there was an abundance of misreported claims for HCPCS code C9757 which they believe has undermined the integrity of the claims data. The manufacturer and other commenters recommended that we assign a device offset percentage based on claims that reported the use of the Barricaid device, or invoices for the Barricaid device, or using the proposed default device offset methodology that would assign the greater of the APC wide device offset percentage or 31 percent. Commenters also recommended we implement a device-specific edit for this procedure and issue a technical direction letter to ensure appropriate device reporting when billing for HCPCS code C9757. Without such hospital guidance, the commenters believe hospitals may continue to not report the associated device, which they believe could impede our ability to properly set payment rates for device-intensive procedures.
Response: We appreciate commenters bringing this issue to our attention. We have taken these comments into consideration with respect to our modification of our device edits policy for CY 2025 in section IV.B.3 of this final rule with comment period. While we continue to rely on hospital claims data rather than external invoices or our default device offset methodology in determining device offset percentages when claims data is available, we believe the stakeholder raises an important issue regarding the nonreporting of device charges and potentially “missing” device costs from certain hospital outpatient claims. We are persuaded by commenters that a lack of a device edit for device-intensive procedures, particularly new technologies, might lead to an underreporting of device costs and total procedure costs and impede beneficiary access to such new technologies. Therefore, in conjunction with the modification to our finalized device edits policy as explained further in section IV.B.3 of this final rule with comment period, for procedures subject to our modified device edits policy for CY 2025 that cannot report modifier “CG” to bypass this claims processing edit, the device offset percentages calculated for CY 2025 (for the CPT/HCPCS code or its predecessor code) are based on hospital claims that reported a device code. We believe that hospital ( printed page 94218) outpatient claims that report a device code with device-intensive procedures provide, in general, a more accurate representation of the procedures' total costs. Therefore, based on claims data available for CY 2025 subject to this device-reporting requirement, HCPCS code C9757 has a device offset percentage that exceeds the device-intensive threshold and is assigned device-intensive status for CY 2025. Final CY 2025 device offset percentages based on claims data used for this CY 2025 OPPS/ASC final rule with comment period can be found in Addendum P.
Comment: Several commenters recommended that we assign device-intensive status to CPT code 52284 (Cystourethroscopy, with mechanical urethral dilation and urethral therapeutic drug delivery by drug-coated balloon catheter for urethral stricture or stenosis, male, including fluoroscopy, when performed) based on the cost of the device or using the default device offset percentage. Further, the manufacturer of the Optilume® device recommended we create a new HCPCS code to describe the Optilume® device or a claims processing edit as hospital claims data do not accurately report the device code and device cost.
Response: We continue to hear from stakeholders regarding hospital nonreporting of device charges. We note that CPT code 52284 was previously described by CPT code 0499T (Cystourethroscopy, with mechanical dilation and urethral therapeutic drug delivery for urethral stricture or stenosis, including fluoroscopy, when performed), which was effective from January 1, 2018, until January 1, 2024. Therefore, as explained further in section IV.B.3 of this final rule with comment period, in conjunction with our modified device edits policy for CY 2025, we are utilizing the device offset percentages calculated for CY 2025 based on hospital claims that reported a device code. Based on claims data available for CY 2025 subject to this device-reporting requirement, CPT code 0499T has a device offset percentage that exceeds our device-intensive threshold. Therefore, we are assigning device-intensive status to successor CPT code 52284 for CY 2025. Final CY 2025 device offset percentages based on claims data used for this CY 2025 OPPS/ASC final rule with comment period can be found in Addendum P.
Comment: One commenter recommended that we assign device-intensive status to CPT codes 0884T, 0885T, and 0886T, which became effective July 1, 2024.
Response: We proposed to assign these procedures device-intensive status in the July 2024 OPPS update and in Addendum P to the CY 2025 OPPS/ASC proposed rule. Since these are new procedures with no predecessor or clinically similar codes that use the same device, we are finalizing our proposal to assign device-intensive status to these three procedures and to use our default offset methodology to assign a device offset percentage to CPT codes 0884T, 0885T, and 0886T.
Comment: One commenter recommended that we restore device-intensive status to CPT code 27412 (Autologous chondrocyte implantation, knee). The commenter stated the procedure is always performed with HCPCS Code J7330 (Autologous cultured chondrocytes, implant), which they argue is a device. Further, the commenter argues the procedure has historically been device-intensive and should continue to have that designation.
Response: We disagree with the commenter. We believe we inadvertently assigned this procedure device-intensive status based on hospitals reporting significant device costs. However, as the commenter noted, CPT code 27412 is performed with J7330 which is categorized as a drug/biological and may have been incorrectly reported as an implantable device by certain hospitals. We note that J7330 is regulated as Human Cells, Tissues, and Cellular and Tissue-Based Products (HTC/Ps) by the FDA under 21 CFR part 1271 and is not regulated as a medical device by the FDA. Since CPT code 27412 does not involve a device that meets our requirement, we are not assigning device-intensive status to CPT code 27412.
Comment: One commenter was concerned that Addendum P did not reflect the device offset amount that is applied to the transitional pass-through device performed with CPT code 0505T (Endovenous femoral-popliteal arterial revascularization, with transcatheter placement of intravascular stent graft(s) and closure by any method, including percutaneous or open vascular access, ultrasound guidance for vascular access when performed, all catheterization(s) and intraprocedural roadmapping and imaging guidance necessary to complete the intervention, all associated radiological supervision and interpretation, when performed, with crossing of the occlusive lesion in an extraluminal fashion). The commenter suggested that the device offset amount is $0.00 and Addendum P should reflect this amount.
Response: While we agree that the device offset amount for CPT code 0505T is $0.00 as we stated in the July 2024 OPPS Change Request, the amount in Addendum P reflects the total device-related costs of the procedure. In certain situations where an approved transitional pass-through device does not replace existing APC device costs, the amount reflected in Addendum P does not reflect the actual device offset amount that is applied against the separate payment for the transitional pass-through device.
Comment: Some commenters recommended that we assign device-intensive status to CPT code 0621T (Trabeculostomy ab interno by laser;) since the procedure was assigned a status indicator of “E1”—Items, codes, and services not covered by any Medicare outpatient benefit category; statutorily excluded; not reasonable and necessary—in CY 2023 and the claims that would be used to determine device-intensive status are invalid and not usable for this purpose. Similarly, one commenter argued that our device offset percentage for CPT code 0795T (Transcatheter insertion of permanent dual-chamber leadless pacemaker, including imaging guidance ( e.g., fluoroscopy, venous ultrasound, right atrial angiography, right ventriculography, femoral venography) and device evaluation ( e.g., interrogation or programming), when performed; complete system ( i.e., right atrial and right ventricular pacemaker components)) should be revised to be set using the default device offset methodology as CPT code 0795T was also assigned status indicator of “E1” in CY 2023.
Response: We agree with the commenters that claims with a status indicator of “E1” may not be appropriate for determining device offset percentages and device offset amounts as this status indicator can be used for procedures that involve a device where such device has not received FDA marketing authorization or received FDA investigational device exemption. For purposes of determining device offset percentages, we will not use claims data from procedures that had a status indicator of “E1” for determining device offset percentages. We will revise our claims accounting narrative to reflect this change.
Comment: One commenter recommended that we evaluate, as part of rulemaking, procedures that utilize pass-through devices expiring mid-year using the latest final rule claims data available to determine the appropriate APC assignment when the device no ( printed page 94219) longer has transitional pass-through status.
Response: We thank the commenter for this recommendation and note we already conduct such an evaluation in our proposed and final rule ratesetting process. Devices for which pass-through status is expiring at any point in the prospective year are packaged in our ratesetting process for that prospective year so that we may reevaluate the APC assignments of these technologies as the commenter noted.
Comment: One commenter recommended that we update the device offset percentages from predecessor codes annually rather than just in the first year we calculate the device offset percentage from the predecessor code.
Response: We agree with the commenter and will refine our process for applying device offset percentages to use available claims data from predecessor codes annually rather than just in the first year.
Comment: Commenters supported the proposed device offset percentage for CPT codes:
- 0421T (Transurethral waterjet ablation of prostate, including control of post-operative bleeding, including ultrasound guidance, complete (vasectomy, meatotomy, cystourethroscopy, urethral calibration and/or dilation, and internal urethrotomy are included when performed));
- 0737T (Xenograft implantation into the articular surface);
- 0816T (Open insertion or replacement of integrated neurostimulation system for bladder dysfunction including electrode(s) (e.g., array or leadless), and pulse generator or receiver, including analysis, programming, and imaging guidance, when performed, posterior tibial nerve; subfascial);
- 0817T (Revision or removal of integrated neurostimulation system for bladder dysfunction, including analysis, programming, and imaging, when performed, posterior tibial nerve; subcutaneous);
- 66989 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1-stage procedure), manual or mechanical technique (e.g., irrigation and aspiration or phacoemulsification), complex, requiring devices or techniques not generally used in routine cataract surgery ( e.g., iris expansion device, suture support for intraocular lens, or primary posterior capsulorrhexis) or performed on patients in the amblyogenic developmental stage; with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more);
- 66991 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1 stage procedure), manual or mechanical technique (e.g., irrigation and aspiration or phacoemulsification); with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more);
- 0671T (Insertion of anterior segment aqueous drainage device into the trabecular meshwork, without external reservoir, and without concomitant cataract removal, one or more);
- 66683 (Implantation of iris prosthesis, including suture fixation and repair or removal of iris, when performed);
- C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer (e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed).
Response: We thank the commenters for their support.
After consideration of the public comments we received, we are finalizing our proposal with modification to apply a default device offset percentage that is the greater of 31 percent or the device offset percentage of the APC to which the procedure has been assigned for a new HCPCS code that describes a procedure that requires the implantation or insertion of a single-use device that meets the definition of a device for purposes of our device offset policy and for which the procedure lacks claims data (from either the new HCPCS code or any predecessor code). Also, in conjunction with the modification to our finalized device edits policy as explained further in section IV.B.3 of this final rule with comment period, for procedures subject to our modified device edits policy for CY 2025 that cannot report modifier “CG” to bypass this claims processing edit, the device offset percentages calculated for CY 2025 (for the CPT/HCPCS code or its predecessor code) are based on hospital claims that reported a device code, as we believe that hospital outpatient claims that report a device code for these device-intensive procedures provide, in general, a more accurate representation of the procedures' total costs. We are also finalizing, for purposes of determining device offset percentages, that we will not use claims data from procedures that had a status indicator of “E1” during the calendar year we are using for ratesetting and determining device offset percentages. Lastly, we are refining our process for applying device offset percentages to use available claims data from predecessor codes annually, rather than the first year of the successor code's activation date, until we have available claims data from the successor code.
The full listing of the final CY 2025 device-intensive procedures, which utilize the revised default device offset policy we are finalizing, can be found in Addendum P to the proposed rule (which is available via the internet on the CMS website). Further, our claims accounting narrative contains a description of our device offset percentage calculation. Our claims accounting narrative for this final rule can be found under supporting documentation for this CY 2025 OPPS/ASC final rule with comment period on our website at: https://www.cms.gov/medicare/medicare-fee-for-service-payment/hospitaloutpatientpps.
3. Device Edit Policy
In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66795), we finalized a policy and implemented claims processing edits that require any of the device codes used in the previous device-to-procedure edits to be present on the claim whenever a procedure code assigned to any of the APCs listed in Table 5 of the CY 2015 OPPS/ASC final rule with comment period (the CY 2015 device-dependent APCs) was reported on the claim. In addition, in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70422), we modified our previously existing policy and applied the device coding requirements exclusively to procedures that require the implantation of a device assigned to a device-intensive APC. In the CY 2016 OPPS/ASC final rule with comment period, we also finalized our policy that the claims processing edits are such that any device code, when reported on a claim with a procedure assigned to a device-intensive APC (listed in Table 42 of the CY 2016 OPPS/ASC final rule with comment period (80 FR 70422)), will satisfy the edit.
In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79658 through 79659), we changed our policy for CY 2017 and subsequent years to apply the CY 2016 device coding requirements to the newly defined device-intensive procedures. For CY 2017 and subsequent years, we also specified that any device code, when reported on a claim with a device-intensive procedure, will satisfy the ( printed page 94220) edit. In addition, we created HCPCS code C1889 to recognize devices furnished during a device-intensive procedure that are not described by a specific Level II HCPCS Category C-code. Reporting HCPCS code C1889 with a device-intensive procedure will satisfy the edit requiring a device code to be reported on a claim with a device-intensive procedure. In the CY 2019 OPPS/ASC final rule with comment period, we revised the description of HCPCS code C1889 to remove the specific applicability to device-intensive procedures (83 FR 58950). For CY 2019 and subsequent years, the description of HCPCS code C1889 is “Implantable/insertable device, not otherwise classified.”
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81758 through 81759), we finalized our proposal to establish a procedure-to-device edit for the procedures assigned to APC 5496 (Level 6 Intraocular Procedures) and require hospitals to report the correct device HCPCS codes when reporting any of the four procedures—CPT codes 0308T, 0616T, 0617T, and 0618T. While we noted that interested parties have previously recommended in past rulemaking that we reestablish all of our previous procedure-to-device edits, we did not expect to extend this policy beyond the procedures assigned to APC 5496 (Level 6 Intraocular Procedures). This APC represents a unique situation—the APC (which was the Level 5 Intraocular APC in previous years) had been a Low Volume APC (fewer than 100 claims in a claims year) since we established our Low Volume APC policy, the procedures associated with this APC have significant procedure costs often greater than $15,000, and the procedures associated with this APC require the implantation of a high-cost intraocular device. Additionally, in our review of claims data for some of the procedures, we noticed unusual coding, charge, and cost data. These claims had an outsized impact because of the low volume of claims for the APC which impeded our ability to determine a payment rate accurately and appropriately for APC 5496 (Level 6 Intraocular Procedures). Further, because of the low volume of procedures assigned to this APC, we did not believe the reinstatement of procedure-to-device edits for the four procedures assigned to this APC would be administratively burdensome to hospitals. We finalized our proposal to modify our device edits policy to require a procedure-to-device edit for procedures assigned to APC 5496 (Level 6 Intraocular Procedures) for CY 2024. We proposed to continue this policy for APC 5496 (Level 6 Intraocular Procedures) for CY 2025 and subsequent CYs in the CY 2025 OPPS/ASC proposed rule and noted that new CPT placeholder code 6X004 (Implantation of iris prosthesis, including suture fixation and repair or removal of iris, when performed) is replacing CPT code 0616T (Insertion of iris prosthesis, including suture fixation and repair or removal of iris, when performed; without removal of crystalline lens or intraocular lens, without insertion of intraocular lens) effective January 1, 2025. Additionally, CPT codes 0617T and 0618T currently assigned to APC 5496 (Level 6 Intraocular Procedures) will be deleted effective January 1, 2025. Therefore, for CY 2025, the procedure-to-device edit for procedures assigned to APC 5496 (Level 6 Intraocular Procedures) would apply to CPT code 0308T and 66683 (placeholder code 6X004). We did not propose any other changes to our device edit policy for CY 2025.
Comment: One commenter supported our proposal to continue our procedure-to-device edit for the insertion of the Artificial Iris—from CPT code 0616T, which will be deleted on January 1, 2025, to CPT code 66683 will become active on January 1, 2025.
Response: We thank the commenters for their support. We are finalizing our proposal to apply the procedure-to-device edit for predecessor code 0616T to successor code 66683.
Comment: Several commenters recommended reinstating a claims processing edit to improve hospital reporting of device costs. In particular, the manufacturer of the device Barricaid recommended we implement a device-specific edit for this procedure and issue a technical direction letter to ensure correct device code reporting with HCPCS code C9757 (Laminotomy (hemilaminectomy), with decompression of nerve root(s), including partial facetectomy, foraminotomy and excision of herniated intervertebral disc, and repair of annular defect with implantation of bone anchored annular closure device, including annular defect measurement, alignment and sizing assessment, and image guidance; 1 interspace, lumbar). Additionally, the manufacturer of the Optilume® device recommended we create a new HCPCS code to describe the Optilume® device or a claims processing edit as hospital claims data do not accurately report the device code and device cost on hospital outpatient claims with the appropriate procedure code—CPT code 52284 (Cystourethroscopy, with mechanical urethral dilation and urethral therapeutic drug delivery by drug-coated balloon catheter for urethral stricture or stenosis, male, including fluoroscopy, when performed). A few commenters requested that we implement a device edit for CPT code 30469 (Repair of nasal valve collapse with low energy, temperature-controlled ( i.e., radiofrequency) subcutaneous/submucosal remodeling) to allow additional years of claims data to determine its device-intensive status as they claim hospitals have been slow to update their chargemasters. Further, one device manufacturer reiterated a longstanding recommendation that we reinstate all our previous device-to-procedure edits.
Response: We appreciate commenters bringing to our attention their concerns regarding our assignment of device offset percentages and hospital reporting of device costs. We believe commenters raise an important issue that there may have been instances when a newly created procedure code loses device-intensive status if the device cost fluctuates below the device-intensive threshold of 30 percent of the total procedure cost and then is never able to regain device-intensive status because hospitals are no longer required to report a device code on the claim with that procedure. The commenters have indicated to us that the presence of the device edit requirement can have a significant impact on the device portion and geometric mean cost of a procedure. For example, the geometric mean cost was $9,218.47 for hospitals that reported a device code with HCPCS code C9757 and $6,287.47 for hospitals that did not report a device code. The geometric mean cost was $5,277.22 for hospitals that reported a device code with CPT code 52284 and $3,791.95 for hospitals that did not report a device code. Additionally, removing claims from hospitals that do not report a device code with these procedures increased the device offset percentage for HCPCA code C9757 from 23.62 percent to 43.21 percent and from 32.17 percent to 32.225 percent for CPT code 52284. These results highlight the impact that hospital reporting of device codes can have on our assignment of device offset percentages, device-intensive status, and the geometric mean cost of a procedure and these results impacted our final CY 2025 device-intensive policy as described in section IV.B.2 of this final rule with comment period. We are persuaded by commenters that are our existing device edits policy may be insufficient for ( printed page 94221) certain procedures, particularly newer technologies with device costs that fluctuate around our device-intensive threshold.
After consideration of the public comments we received, we are finalizing our proposal with modification. While historically our device edits policy has only applied to procedures that are device-intensive based on the most recent claims data available, for CY 2025 and subsequent calendar years, we are finalizing a policy to apply our device edit policy permanently once a procedure is designated as a device-intensive procedure in a given year. For the procedures designated as device-intensive for CY 2025, we propose that the device edit requirement would apply in CY 2025 and subsequent calendar years as well. We are concerned that the loss of an applicable device edit may impact hospital reporting of device costs and impede our ability to properly set payment rates and determine appropriate device offset percentages for device-intensive procedures. Additionally, we are finalizing a policy to reinstate our device edits policy for procedures that have been device-intensive since we began assigning device-intensive status at the HCPCS code level on January 1, 2017. We believe that by applying our device edit policy to procedures that were device-intensive on or after January 1, 2017, we might continue to receive device cost information for relatively new procedures with limited claims data, which may have been impacted by our policy to require that only existing device-intensive procedures be subject to our device edits policy. For CY 2025, under our modified device edits policy, our device edits requirement will apply to procedures that are device-intensive in CY 2025 as well as procedures that have been device-intensive on or after January 1, 2017.
4. Adjustment to OPPS Payment for No Cost/Full Credit and Partial Credit Devices
a. Background
To ensure equitable OPPS payment when a hospital receives a device without cost or with full credit, in CY 2007, we implemented a policy to reduce the payment for specified device-dependent APCs by the estimated portion of the APC payment attributable to device costs (that is, the device offset) when the hospital receives a specified device at no cost or with full credit (71 FR 68071 through 68077). Hospitals were instructed to report no cost/full credit device cases on the claim using the “FB” modifier on the line with the procedure code in which the no cost/full credit device is used. In cases in which the device is furnished without cost or with full credit, hospitals were instructed to report a token device charge of less than $1.01. In cases in which the device being inserted is an upgrade (either of the same type of device or to a different type of device) with a full credit for the device being replaced, hospitals were instructed to report as the device charge the difference between the hospital's usual charge for the device being implanted and the hospital's usual charge for the device for which it received full credit. In CY 2008, we expanded this payment adjustment policy to include cases in which hospitals receive partial credit of 50 percent or more of the cost of a specified device. Hospitals were instructed to append the “FC” modifier to the procedure code that reports the service provided to furnish the device when they receive a partial credit of 50 percent or more of the cost of the new device. We refer readers to the CY 2008 OPPS/ASC final rule with comment period for more background information on the “FB” and “FC” modifiers payment adjustment policies (72 FR 66743 through 66749).
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75005 through 75007), beginning in CY 2014, we modified our policy of reducing OPPS payment for specified APCs when a hospital furnishes a specified device without cost or with a full or partial credit. For CY 2013 and prior years, our policy had been to reduce OPPS payment by 100 percent of the device offset amount when a hospital furnishes a specified device without cost or with a full credit and by 50 percent of the device offset amount when the hospital receives partial credit in the amount of 50 percent or more of the cost for the specified device. For CY 2014, we reduced OPPS payment, for the applicable APCs, by the full or partial credit a hospital receives for a replaced device. Specifically, under this modified policy, hospitals are required to report on the claim the amount of the credit in the amount portion for value code “FD” (Credit Received from the Manufacturer for a Replaced Device) when the hospital receives a credit for a replaced device that is 50 percent or greater than the cost of the device. For CY 2014, we also limited the OPPS payment deduction for the applicable APCs to the total amount of the device offset when the “FD” value code appears on a claim. For CY 2015, we continued our policy of reducing OPPS payment for specified APCs when a hospital furnishes a specified device without cost or with a full or partial credit and to use the three criteria established in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68072 through 68077) for determining the APCs to which our CY 2015 policy will apply (79 FR 66872 through 66873). In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70424), we finalized our policy to no longer specify a list of devices to which the OPPS payment adjustment for no cost/full credit and partial credit devices would apply and instead apply this APC payment adjustment to all replaced devices furnished in conjunction with a procedure assigned to a device-intensive APC when the hospital receives a credit for a replaced specified device that is 50 percent or greater than the cost of the device.
b. Policy for No Cost/Full Credit and Partial Credit Devices
In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79659 through 79660), for CY 2017 and subsequent years, we finalized a policy to reduce OPPS payment for device-intensive procedures, by the full or partial credit a provider receives for a replaced device, when a hospital furnishes a specified device without cost or with a full or partial credit. Under our current policy, hospitals continue to be required to report on the claim the amount of the credit in the amount portion for value code “FD” when the hospital receives a credit for a replaced device that is 50 percent or greater than the cost of the device.
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75005 through 75007), we adopted a policy of reducing OPPS payment for specified APCs when a hospital furnishes a specified device without cost or with a full or partial credit by the lesser of the device offset amount for the APC or the amount of the credit. We adopted this change in policy in the preamble of the CY 2014 OPPS/ASC final rule with comment period and discussed it in subregulatory guidance, including Chapter 4, section 61.3.6 of the Medicare Claims Processing Manual. Further, in the CY 2021 OPPS/ASC final rule with comment period (85 FR 86017 through 86018, 86302), we made conforming changes to our regulations at § 419.45(b)(1) and (2) that codified this policy.
We did not propose any changes, and we did not receive any public comments related to our policies regarding ( printed page 94222) payment for no cost/full credit and partial credit devices for CY 2025.
V. OPPS Payment for Drugs, Biologicals, and Radiopharmaceuticals
A. OPPS Transitional Pass-Through Payment for Additional Costs of Drugs, Biologicals, and Radiopharmaceuticals
1. Background
Section 1833(t)(6) of the Act provides for temporary additional payments or “transitional pass-through payments” for certain drugs and biologicals. Throughout this final rule with comment period, the term “biological” is used because this is the term that appears in section 1861(t) of the Act. A “biological” as used in this final rule with comment period includes (but is not necessarily limited to) a “biological product” or a “biologic” as defined under section 351 of the PHS Act. As enacted by the Medicare, Medicaid, and SCHIP Balanced Budget Refinement Act of 1999 (BBRA) (Pub. L. 106-113), this pass-through payment provision requires the Secretary to make additional payments to hospitals for: current orphan drugs for rare diseases and conditions, as designated under section 526 of the Federal Food, Drug, and Cosmetic Act; current drugs and biologicals and brachytherapy sources used in cancer therapy; and current radiopharmaceutical drugs and biologicals. “Current” refers to those types of drugs or biologicals mentioned above that are hospital outpatient services under Medicare Part B for which transitional pass-through payment was made on the first date the hospital OPPS was implemented.
Transitional pass-through payments also are provided for certain “new” drugs and biologicals that were not being paid for as an HOPD service as of December 31, 1996, and whose cost is “not insignificant” in relation to the OPPS payments for the procedures or services associated with the new drug or biological. For pass-through payment purposes, radiopharmaceuticals are included as “drugs.” As required by statute, transitional pass-through payments for a drug or biological described in section 1833(t)(6)(C)(i)(II) of the Act can be made for a period of at least 2 years, but not more than 3 years, after the payment was first made for the drug as a hospital outpatient service under Medicare Part B. Final CY 2025 pass-through drugs and biologicals and their designated APCs are assigned status indicator “G” in Addenda A and B to this final rule with comment period (which are available on the CMS website).[131]
Section 1833(t)(6)(D)(i) of the Act specifies that the pass-through payment amount, in the case of a drug or biological, is the amount by which the amount determined under section 1842(o) of the Act for the drug or biological exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the drug or biological. The methodology for determining the pass-through payment amount is set forth in regulations at 42 CFR 419.64. These regulations specify that the pass-through payment equals the amount determined under section 1842(o) of the Act minus the portion of the APC payment that CMS determines is associated with the drug or biological.
Section 1847A of the Act establishes the average sales price (ASP) methodology, which is used for payment for drugs and biologicals described in section 1842(o)(1)(C) of the Act furnished on or after January 1, 2005. The ASP methodology, as applied under the OPPS, uses several sources of data as a basis for payment, including the ASP, the wholesale acquisition cost (WAC), and the average wholesale price (AWP). In this final rule with comment period, the term “ASP methodology” and “ASP-based” are inclusive of all data sources and methodologies described therein. Additional information on the ASP methodology can be found on our website at: https://www.cms.gov/medicare/payment/fee-for-service-providers/part-b-drugs/average-drug-sales-price.
The pass-through application [132] and review process for drugs and biologicals is described on our website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/pass-through-payment-status-new-technology-ambulatory-payment-classification-apc.
2. Transitional Pass-Through Payment Period for Pass-Through Drugs, Biologicals, and Radiopharmaceuticals and Quarterly Expiration of Pass-Through Status
As required by statute, transitional pass-through payments for a drug or biological described in section 1833(t)(6)(C)(i)(II) of the Act can be made for a period of at least 2 years, but not more than 3 years, after the payment was first made for the drug or biological as a hospital outpatient service under Medicare Part B. Our current policy is to accept pass-through applications on a quarterly basis and to begin pass-through payments for approved pass-through drugs and biologicals on a quarterly basis through the next available OPPS quarterly update after the approval of a drug's or biological's pass-through status. However, prior to CY 2017, we expired pass-through status for drugs and biologicals on an annual basis through notice-and-comment rulemaking (74 FR 60480). In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79662), we finalized a policy change, beginning with pass-through drugs and biologicals approved in CY 2017 and subsequent calendar years, to allow for a quarterly expiration of pass-through payment status for drugs, biologicals, and radiopharmaceuticals to afford a pass-through payment period that is as close to a full 3 years as possible for all pass-through drugs, biologicals, and radiopharmaceuticals.
This change eliminated the variability of the pass-through payment eligibility period, which previously varied based on when a particular application was initially received. We adopted this change for pass-through approvals beginning on or after CY 2017, to allow, on a prospective basis, for the maximum pass-through payment period for each pass-through drug without exceeding the statutory limit of 3 years. Notice of drugs for which pass-through payment status is ending during the calendar year is included in the quarterly OPPS Change Request transmittals.
Comment: One commenter expressed support for our policy of including radiopharmaceuticals as drugs eligible for pass-through status in the OPPS. In addition, this commenter supported our policy to allow drug pass-through status to expire on a quarterly basis to allow all drugs to have as close to three years of pass-through status as possible.
Response: We appreciate the support of the commenter for our policies.
Comment: Several commenters requested that we confirm that our proposal to pay separately for diagnostic radiopharmaceuticals with a per day cost greater than $630 that is described in section II.A.3.c of this final rule with comment period would not alter the qualification or payment methodology for those diagnostic radiopharmaceuticals applying for pass-through status under the OPPS. ( printed page 94223)
Response: The commenter is correct. This policy, which we are finalizing in section II.A.3.c of this final rule with comment period, is separate from the policies described in this section of this final rule with comment period, which describes the payment methodology for products that qualify for OPPS pass-through status.
3. Drugs and Biologicals With Expiring Pass-Through Payment Status in CY 2024
There are 25 drugs and biologicals for which pass-through payment status expires by December 31, 2024, as listed in Table 130. These drugs and biologicals will have received OPPS pass-through payment for 3 years during the period of April 1, 2021, through December 31, 2024. In accordance with the policy finalized in CY 2017 and described earlier, pass-through payment status for drugs and biologicals approved in CY 2017 and subsequent years will expire on a quarterly basis, with a pass-through payment period as close to 3 years as possible.
With the exception of those groups of drugs and biologicals that are always packaged when they do not have pass-through payment status (specifically, anesthesia drugs; drugs, biologicals, and radiopharmaceuticals [133] that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure), our standard methodology for providing payment for drugs and biologicals with expiring pass-through payment status in an upcoming calendar year is to determine the product's estimated per day cost and compare it with the OPPS drug packaging threshold for that calendar year, which will be $140 for CY 2025 for all drugs, biologicals, and therapeutic radiopharmaceuticals (for diagnostic radiopharmaceuticals we will provide separate payment when their per day cost exceeds the threshold we are adopting of $630). These policies are discussed further in section V.B.1 of this final rule with comment period. If the estimated per day cost for the drug or biological is less than or equal to the applicable OPPS drug packaging threshold, we package payment for the drug or biological into the payment for the associated procedure in the upcoming calendar year. If the estimated per day cost of the drug or biological is greater than the OPPS drug packaging threshold, we provide separate payment at the applicable ASP methodology-based payment amount (which is generally ASP plus 6 percent), as discussed further in section V.B.2 of this final rule with comment period.
( printed page 94224) ( printed page 94225)4. Drugs, Biologicals, and Radiopharmaceuticals With Pass-Through Payment Status Expiring in CY 2025
We proposed to end pass-through payment status in CY 2025 for 28 drugs and biologicals. These drugs and biologicals, which were initially approved for pass-through payment status between April 1, 2022, and January 1, 2023, are listed in Table 131. The APCs and HCPCS codes for these drugs and biologicals, which have pass-through payment status that will end by December 31, 2025, are assigned status indicator “G” (Pass-Through Drugs and Biologicals) in Addenda A and B to this final rule with comment period (which are available on the CMS website).[134] The APCs and HCPCS codes for these drugs and biologicals are assigned status indicator “G” only for the duration of their pass-through status.
Section 1833(t)(6)(D)(i) of the Act sets the amount of pass-through payment for pass-through drugs and biologicals (the pass-through payment amount) as the difference between the amount authorized under section 1842(o) of the Act and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is associated with the drug or biological. For CY 2025, we are continuing our policy to pay for pass-through drugs and biologicals using the ASP methodology, meaning a ( printed page 94226) payment rate based on ASP, WAC, or AWP, as applicable. This payment rate is generally ASP plus 6 percent, equivalent to the payment rate these drugs and biologicals would receive in the physician's office setting in CY 2025. We note that, under the OPD fee schedule, separately payable drugs assigned to an APC are generally payable at ASP plus 6 percent. Therefore, a $0 pass-through payment amount would continue to be paid for pass-through drugs and biologicals under the CY 2025 OPPS because the difference between the amount authorized under section 1842(o) of the Act, which is generally ASP plus 6 percent, and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is appropriate, which is generally ASP plus 6 percent, is $0.
In the case of policy-packaged drugs (which include the following: anesthesia drugs; drugs, biologicals, and radiopharmaceuticals [135] that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure), their pass-through payment amount will continue to be equal to a payment rate calculated using the ASP methodology, meaning a payment rate based on ASP, WAC, or AWP. This payment rate will generally continue to be ASP plus 6 percent for CY 2025, minus a payment offset for the portion of the otherwise applicable OPD fee schedule that the Secretary determines is associated with the drug or biological. We note that if not for the pass-through payment status of these policy-packaged products, payment for these products would be packaged into the associated procedure and therefore, there are associated OPD fee schedule amounts for them.
We will continue our policy to update pass-through payment rates on a quarterly basis on the CMS website during CY 2025 if later quarter ASP submissions (or more recent WAC or AWP information, as applicable) indicate that adjustments to the payment rates for these pass-through payment drugs or biologicals are necessary. For a full description of this policy, we refer readers to the CY 2006 OPPS/ASC final rule with comment period (70 FR 68632 through 68635).
For CY 2025, consistent with our CY 2024 policy for diagnostic and therapeutic radiopharmaceuticals, we would continue to provide payment for both diagnostic and therapeutic radiopharmaceuticals that are granted pass-through payment status based on the ASP methodology. As stated earlier, for purposes of pass-through payment, we consider radiopharmaceuticals to be drugs under the OPPS. Therefore, if a diagnostic or therapeutic radiopharmaceutical receives pass-through payment status during CY 2025, we would continue to follow the standard ASP methodology to determine the pass-through payment rate that drugs receive under section 1842(o) of the Act, which is generally ASP plus 6 percent. If ASP data are not available for a radiopharmaceutical, we would continue to provide pass-through payment at WAC plus 3 percent (consistent with our policy in section V.B.2.a of this final rule with comment period), the equivalent payment provided for pass-through drugs and biologicals without ASP information. Additional detail on the WAC plus 3 percent payment policy can be found in section V.B.2.a of this final rule with comment period. If WAC information also is not available, we would continue to provide payment for the pass-through radiopharmaceutical at 95 percent of its most recent AWP.
We refer readers to Table 131 below for the list of drugs and biologicals with pass-through payment status expiring during CY 2025.
( printed page 94227) ( printed page 94228) ( printed page 94229)We did not receive any public comments on our proposal and therefore, we are finalizing our proposal to end pass-through payment status in CY 2025 for 28 drugs and biologicals.
5. Drugs, Biologicals, and Radiopharmaceuticals With Pass-Through Payment Status Continuing Through CY 2025
We proposed to continue pass-through payment status in CY 2025 for 80 drugs and biologicals. These drugs and biologicals, which were approved for pass-through payment status with effective dates beginning between April 1, 2023, and January 1, 2025, are listed in Table 132. The APCs and HCPCS codes for these drugs and biologicals, which have pass-through payment status that would continue after December 31, 2025, are assigned status indicator “G” in Addenda A and B to this final rule with comment period (which are available on the CMS website).[136]
Section 1833(t)(6)(D)(i) of the Act sets the amount of pass-through payment for pass-through drugs and biologicals (the pass-through payment amount) as the difference between the amount authorized under section 1842(o) of the Act and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is associated with the drug or biological. For CY 2025, we are continuing our policy to pay for pass-through drugs and biologicals at a payment rate based on the ASP methodology, which may be based on ASP, WAC, or AWP, but is generally ASP plus 6 percent, which is equivalent to the payment rate these drugs and biologicals would receive in the physician's office setting in CY 2025. We will continue with our policy of paying a $0 pass-through payment amount for pass-through drugs and biologicals that are not policy-packaged under the CY 2025 OPPS, because the difference between the amount authorized under section 1842(o) of the Act, which would generally be ASP plus 6 percent, and the portion of the otherwise applicable OPD fee schedule that the Secretary determines is appropriate, which would also generally be ASP plus 6 percent, is $0.
In the case of policy-packaged drugs (which include the following: anesthesia drugs; drugs, biologicals, and radiopharmaceuticals [137] that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure), their pass-through payment amount would continue to be equal to a payment rate based on the ASP methodology, which may be based on ASP, WAC, or AWP, but would generally be ASP plus 6 percent for CY 2025, minus a payment offset for any predecessor drug products contributing to the pass-through payment. We note if not for the pass-through payment status of these policy-packaged products, payment for these products would be ( printed page 94230) packaged into the associated procedure and therefore, there are associated OPD fee schedule amounts for them.
We are continuing our policy to update pass-through payment rates on a quarterly basis on our website during CY 2025 if later quarter ASP submissions (or more recent WAC or AWP information, as applicable) indicate that adjustments to the payment rates for these pass-through payment drugs or biologicals are necessary. For a full description of this policy, we refer readers to the CY 2006 OPPS/ASC final rule with comment period (70 FR 68632 through 68635).
For CY 2025, consistent with our CY 2024 policy for diagnostic and therapeutic radiopharmaceuticals, we proposed to continue our policy to provide payment for both diagnostic and therapeutic radiopharmaceuticals that are granted pass-through payment status based on the ASP methodology. As stated earlier, for purposes of pass-through payment, we consider radiopharmaceuticals to be drugs under the OPPS. Therefore, if a diagnostic or therapeutic radiopharmaceutical receives pass-through payment status during CY 2025, we will continue to follow the standard ASP methodology to determine the pass-through payment rate that drugs receive under section 1842(o) of the Act, which would generally be ASP plus 6 percent. If ASP data are not available for a radiopharmaceutical, we would provide pass-through payment at WAC plus 3 percent (consistent with our policy in section V.B.2.a of this final rule with comment period), the equivalent payment provided for pass-through drugs and biologicals without ASP information. Additional detail on the WAC plus 3 percent payment policy can be found in section V.B.2.a of this final rule with comment period. If WAC information also is not available, we would provide payment for the pass-through radiopharmaceutical at 95 percent of its most recent AWP.
The drugs and biologicals that would have pass-through payment status expire after December 31, 2025, are shown in Table 132.
( printed page 94231) ( printed page 94232) ( printed page 94233) ( printed page 94234) ( printed page 94235) ( printed page 94236) ( printed page 94237)We did not receive any public comments on our proposal to continue pass-through payment status in CY 2025 for 80 drugs and biologicals and we are finalizing it as proposed. Additionally, we are finalizing our proposal to continue our policy to provide payment for both diagnostic and therapeutic radiopharmaceuticals that are granted pass-through payment status based on the ASP methodology.
B. OPPS Payment for Drugs, Biologicals, and Radiopharmaceuticals Without Pass-Through Payment Status
1. Criteria for Packaging Payment for Drugs, Biologicals, and Radiopharmaceuticals
a. Packaging Threshold
In accordance with section 1833(t)(16)(B) of the Act, the threshold for establishing separate APCs for payment of drugs and biologicals was set to $50 per administration during CYs 2005 and 2006. In CY 2007, we used the four-quarter moving average Producer Price Index (PPI) levels for Pharmaceutical Preparations (Prescription) to trend the $50 threshold forward from the third quarter of CY 2005 (when the Pub. L. 108-173 mandated threshold became effective) to the third quarter of CY 2007. We then rounded the resulting dollar amount to the nearest $5 increment in order to determine the CY 2007 threshold amount of $55. Using the same methodology as that used in CY 2007 (which is discussed in more detail in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68085 through 68086)), we set the packaging threshold for establishing separate APCs for drugs and biologicals at $135 for CY 2024 (88 FR 81776 through 81777).
Following the CY 2007 methodology, for the CY 2025 OPPS/ASC proposed rule, we used the most recently available four quarter moving average PPI levels to trend the $50 threshold forward from the third quarter of CY 2005 to the third quarter of CY 2025 and round the resulting dollar amount ($140.81) to the nearest $5 increment, which yielded a figure of $140. In performing this calculation, we used the most recent forecast of the quarterly index levels for the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IGI. IGI is a nationally recognized economic and financial forecasting firm with which CMS contracts to forecast the various price indexes including the PPI Pharmaceuticals for Human Use (Prescription). Based on these calculations using the CY 2007 OPPS methodology, we proposed a packaging threshold for CY 2025 of $140 for drugs, biologicals, and therapeutic radiopharmaceuticals.
We proposed in section II.A.3.c of the CY 2025 OPPS/ASC proposed rule (89 FR 59213 through 59222) to pay separately for diagnostic radiopharmaceuticals with a per-day cost above the proposed packaging threshold for CY 2025 of $630. We also proposed that starting in CY 2026 and subsequent years, we would update this threshold by the PPI for Pharmaceuticals for Human Use (Prescription) (Bureau of Labor Statistics series code WPUSI07003) from IHS Global, Inc (IGI). For the diagnostic radiopharmaceutical packaging threshold, we proposed to use the same methodology as that used in CY 2007 (which is discussed in more detail in the CY 2007 OPPS/ASC final rule with comment period (71 FR 68085 and 68086)) to calculate the update to the OPPS drug packaging threshold. Specifically, we proposed that starting for the CY 2026 rulemaking, we would use the most recently available four quarter moving average PPI levels to trend the final CY 2025 threshold forward from the third quarter of CY 2024 to the third quarter of CY 2025 and round the resulting dollar amount to the nearest $5 increment. We refer readers to section II.A.3.c.(4) of the CY 2025 OPPS/ASC proposed rule for information regarding our proposal to update the proposed diagnostic radiopharmaceutical packaging threshold in future years. We refer readers to section II.A.3.c of this final rule with comment period for additional details on our proposal to provide for separate payment for certain diagnostic radiopharmaceuticals, the comments we received on the proposal, and our final policy regarding separate payment for certain diagnostic radiopharmaceuticals in CY 2025.
We received no comments on our proposal to increase the drug packaging threshold to $140. We are finalizing our proposal without modification to apply a drug packaging threshold of $140 for CY 2025.
b. Packaging of Payment for HCPCS Codes That Describe Certain Drugs, Certain Biologicals, and Certain Radiopharmaceuticals Under the Cost Thresholds
To determine the proposed CY 2025 packaging status for all nonpass-through drugs, biologicals, diagnostic and therapeutic radiopharmaceuticals that are not policy packaged, we calculated, on a HCPCS code-specific basis, the per day cost of all drugs, biologicals, and therapeutic radiopharmaceuticals that had a HCPCS code in CY 2023 and were paid (via packaged or separate payment) under the OPPS. We used data from CY 2023 claims processed through December 31, 2023, for this calculation. However, we did not perform this calculation for those drugs and biologicals with multiple HCPCS codes that include different dosages, as described in section V.B.1.d of this final rule with comment period, or for the following policy-packaged items that we propose to continue to package in CY 2025: anesthesia drugs; drugs, biologicals, and contrast agents and other drugs that function as supplies when used in a diagnostic test or procedure; and drugs and biologicals that function as supplies when used in a surgical procedure. Consistent with our policy described in section V.B.5, in situations where we have no claims data and must determine if these products exceed the per-day cost threshold, we estimated the average number of units of each product that would typically be furnished to a patient during one day in the hospital outpatient setting and utilized the ASP methodology to determine whether their payment will be packaged as well as their payment status indicators.
In order to calculate the per day costs for drugs, biologicals, diagnostic radiopharmaceuticals, and therapeutic radiopharmaceuticals to determine their proposed packaging status in CY 2025, we used the methodology that was described in detail in the CY 2006 OPPS proposed rule (70 FR 42723 through 42724) and finalized in the CY 2006 OPPS final rule with comment period (70 FR 68636 through 68638). For each drug and biological HCPCS code, we used an estimated payment rate based on the ASP methodology, which is generally ASP plus 6 percent (which is the payment rate we proposed for separately payable drugs and biologicals for CY 2025, as discussed in more detail in section V.A.1 of this final rule with comment period) to calculate the CY 2025 proposed rule per day costs. We used the manufacturer-submitted ASP data from the fourth quarter of CY 2023 (data that were used for payment purposes in the physician's office setting, effective April 1, 2024) to determine the CY 2025 OPPS/ASC proposed rule per day cost.
As is our standard methodology, for CY 2025, we proposed to use payment rates based on the ASP data from the fourth quarter of CY 2023 for budget neutrality estimates, packaging determinations, impact analyses, and completion of Addenda A and B to the ( printed page 94238) CY 2025 OPPS/ASC proposed rule (which are available via the internet on the CMS website) because these are the most recent data available for use at the time of development of the CY 2025 OPPS/ASC proposed rule. These data also are the basis for drug payments in the physician's office setting, effective April 1, 2024. Exceptions to our standard methodology include:
- For therapeutic radiopharmaceuticals that do not have pass-through status as of October 1, 2024, and do not have an ASP-based payment rate, we did not use a payment rate based on WAC or AWP for those items, consistent with our policy described in section V.B.3.a of the CY 2025 OPPS/ASC proposed rule. We used their mean unit cost derived from the CY 2023 hospital claims data to determine their per day cost.
- For diagnostic radiopharmaceuticals that do not have pass-through status as of October 1, 2024, we used their mean unit cost derived from the CY 2023 hospital claims data to determine their per day cost. We did not use an ASP-based, WAC-based, or AWP-based payment rate for those items unless there was no mean unit cost reported for the product, consistent with our proposed policy described in section V.B.3.b of the CY 2025 OPPS/ASC proposed rule.
- For items other than diagnostic or therapeutic radiopharmaceuticals that did not have either an ASP-based payment rate, a payment rate based on WAC, or a payment rate based on AWP, we used mean unit cost of the items derived from the CY 2023 hospital claims data to determine their per day cost.
We proposed to package drugs, biologicals, and therapeutic radiopharmaceuticals with a per day cost less than or equal to $140 and identify items with a per day cost greater than $140 as separately payable unless they are policy-packaged. For diagnostic radiopharmaceuticals, we propose to package those items with a per day cost less than or equal to $630 and identify items with a per day cost greater than $630 as separately payable. Consistent with our past practice, we cross-walked historical OPPS claims data from the CY 2023 HCPCS codes that were reported to the CY 2023 HCPCS codes that we display in Addendum B to the CY 2025 OPPS/ASC proposed rule (which is available on the CMS website) [138] for proposed payment in CY 2025.
Our policy during previous cycles of OPPS rulemaking has been to use updated ASP and claims data to make final determinations of the packaging status of HCPCS codes for drugs, biologicals, and therapeutic radiopharmaceuticals for the OPPS/ASC final rule with comment period. We note that it is also our policy to make an annual packaging determination for a HCPCS code only when we develop the OPPS/ASC final rule with comment period for the update year. Only HCPCS codes that are identified as separately payable in the final rule with comment period are subject to quarterly updates. For our calculation of per day costs of HCPCS codes for drugs, biologicals, and radiopharmaceuticals in the CY 2025 OPPS/ASC proposed rule, we proposed to use ASP data from the fourth quarter of CY 2023, which is the basis for calculating payment rates for drugs and biologicals in the physician's office setting using the ASP methodology, effective April 1, 2024, along with updated hospital claims data from CY 2023. We note that we also proposed to use these data for budget neutrality estimates and impact analyses for the CY 2025 OPPS/ASC proposed rule.
Payment rates for HCPCS codes for separately payable drugs and biologicals included in Addenda A and B of this CY 2025 OPPS/ASC final rule with comment period are based on ASP data from the second quarter of CY 2024. These data are the basis for calculating payment rates for drugs and biologicals in the physician's office setting using the ASP methodology, effective October 1, 2024. These payment rates would then be updated in the January 2025 OPPS update, based on the most recent ASP data to be used for physicians' office and OPPS payment as of January 1, 2025. For drugs and biologicals that do not currently have a payment rate based on ASP, WAC, or AWP, for therapeutic radiopharmaceuticals that do not currently have an ASP payment rate, and for all diagnostic radiopharmaceuticals, we calculated their mean unit cost from all of the CY 2023 claims data and updated cost report information available for this final rule with comment period to determine their final per day cost.
All the comments we received regarding calculating per-day costs of drugs, biologicals, and radiopharmaceuticals pertained to our proposal to provide for separate payment for certain diagnostic radiopharmaceuticals for CY 2025. We address comments on the proposal in section II.A.3.c., where we are finalizing our proposal regarding calculating the per-day cost of diagnostic radiopharmaceuticals for purposes of determining eligibility for separate payment as proposed. We refer readers to section II.A.3.c of this final rule with comment period for a summary of the comments we received, our responses, and our final policy regarding separate payment for certain diagnostic radiopharmaceuticals in CY 2025.
After consideration of the public comments we received, we are finalizing our proposal regarding the calculation of per day costs of drugs, biologicals, therapeutic radiopharmaceuticals, and diagnostic radiopharmaceuticals without modification.
Because we use updated data to determine the packaging status of HCPCS codes for drugs, biologicals and radiopharmaceuticals for this final rule with comment period, the packaging status of some HCPCS codes for drugs, biologicals, and radiopharmaceuticals in this final rule with comment period may differ from the same HCPCS codes' packaging status determined based on the data used for proposed rule. Under such circumstances, we propose to continue to follow the established policies initially adopted for the CY 2005 OPPS (69 FR 65780) in order to more equitably pay for those drugs whose costs fluctuate relative to the proposed CY 2025 OPPS drug packaging threshold and the drug's payment status (packaged or separately payable) in CY 2024. These established policies have not changed for many years and are the same as described in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70434). Specifically, for CY 2025 and subsequent years, consistent with our historical practice, we proposed to apply the following policies to those HCPCS codes for drugs, biologicals, and therapeutic radiopharmaceuticals whose relationship to the drug packaging threshold changes based on the updated drug packaging threshold and on the final updated data:
- HCPCS codes for drugs, biologicals, and radiopharmaceuticals that were paid separately in CY 2024 and that are proposed for separate payment in CY 2025, and that then have per day costs equal to or less than the CY 2025 final rule drug packaging threshold or diagnostic radiopharmaceutical packaging threshold, based on the updated ASPs and hospital claims data used for the CY 2025 final rule, would continue to receive separate payment in CY 2025.
- HCPCS codes for drugs, biologicals, and radiopharmaceuticals that were packaged in CY 2024 and that are proposed for separate payment in CY 2025, and that then have per day costs ( printed page 94239) equal to or less than the CY 2025 final rule drug packaging threshold or diagnostic radiopharmaceutical packaging threshold, based on the updated ASPs and hospital claims data used for the CY 2025 final rule, would remain packaged in CY 2025.
- HCPCS codes for drugs, biologicals, and radiopharmaceuticals for which we proposed packaged payment in CY 2025 but that then have per-day costs greater than the CY 2025 final rule drug packaging threshold or diagnostic radiopharmaceutical packaging threshold, based on the updated ASPs and hospital claims data used for the CY 2025 final rule, would receive separate payment in CY 2025.
We did not receive any public comments on our proposal and are finalizing it without modification.
c. Policy-Packaged Drugs, Biologicals, and Radiopharmaceuticals
As mentioned earlier in this section, under the OPPS, we package several categories of nonpass-through drugs, biologicals, and radiopharmaceuticals, regardless of the cost of the products. Because the products are packaged according to the policies in 42 CFR 419.2(b), we refer to these packaged drugs, biologicals, and radiopharmaceuticals as “policy-packaged” drugs, biologicals, and radiopharmaceuticals. These policies are either longstanding or based on longstanding principles and inherent to the OPPS and are as follows:
- Anesthesia, certain drugs, biologicals, and other pharmaceuticals; medical and surgical supplies and equipment; surgical dressings; and devices used for external reduction of fractures and dislocations (§ 419.2(b)(4));
- Intraoperative items and services (§ 419.2(b)(14));
- Drugs, biologicals, and radiopharmaceuticals that function as supplies when used in a diagnostic test or procedure (including but not limited to, diagnostic radiopharmaceuticals,[139] contrast agents, and pharmacologic stress agents) (§ 419.2(b)(15)); and
- Drugs and biologicals that function as supplies when used in a surgical procedure (including, but not limited to, skin substitutes and similar products that aid wound healing and implantable biologicals) (§ 419.2(b)(16)).
The policy at § 419.2(b)(16) is broader than the policy at § 419.2(b)(14). As we stated in the CY 2015 OPPS/ASC final rule with comment period: “We consider all items related to the surgical outcome and provided during the hospital stay in which the surgery is performed, including postsurgical pain management drugs, to be part of the surgery for purposes of our drug and biological surgical supply packaging policy” (79 FR 66875). The category described by § 419.2(b)(15) is large and includes diagnostic radiopharmaceuticals that have a per day cost below the finalized diagnostic radiopharmaceutical packaging threshold that we discuss in section II.A.3 of this CY 2025 OPPS/ASC final rule,[140] contrast agents, stress agents, and some other products. The category described by § 419.2(b)(16) includes skin substitutes and some other products. We believe it is important to reiterate that cost consideration is not a factor when determining whether an item is a surgical supply (79 FR 66875).
Comment: One commenter recommended that CMS continue to apply radiolabeled product edits to the nuclear medicine procedures to ensure that all packaged costs are included on nuclear medicine claims in order to establish appropriate payment rates in the future. The commenter was concerned that many providers performing nuclear medicine procedures are not including the cost of diagnostic radiopharmaceuticals used for the procedures in their claim submissions. The commenter believes this lack of drug cost reporting could be causing the cost of nuclear medicine procedures to be underreported and therefore requested that the radiolabeled product edits be reinstated. The commenter believes that after more than ten years without the radiolabeled product edits in place providers may not be as knowledgeable about ensuring radiolabeled products are reported with nuclear medicine procedures. The commenter believes reinstating the edits will improve billing accuracy.
Response: We appreciate the commenter's feedback; however, we are not reinstating the radiolabeled product edits for nuclear medicine procedures, which required a diagnostic radiopharmaceutical to be present on the same claim as a nuclear medicine procedure for payment to be made under the OPPS. As previously discussed in the CY 2020 OPPS/ASC final rule with comment period (85 FR 86033 and 86034), the edits were in place between CY 2008 and CY 2014 (78 FR 75033). We believe the period of time in which the edits were in place was sufficient for hospitals to gain experience reporting procedures involving radiolabeled products and to become accustomed to ensuring that they code and report charges so that their claims fully and appropriately reflect the costs of those radiolabeled products. As with all other items and services recognized under the OPPS, we expect hospitals to code and report their costs appropriately, regardless of whether there are claims processing edits in place.
We welcome ongoing dialogue and engagement from interested parties regarding suggestions for payment changes for consideration in future rulemaking.
d. Packaging Determination for HCPCS Codes That Describe the Same Drug or Biological but Different Dosages
In the CY 2010 OPPS/ASC final rule with comment period (74 FR 60490 through 60491), we finalized a policy to make a single packaging determination for a drug, rather than an individual HCPCS code, when a drug has multiple HCPCS codes describing different dosages because we believe that adopting the standard HCPCS code-specific packaging determinations for these codes could lead to inappropriate payment incentives for hospitals to report certain HCPCS codes instead of others. We continue to believe that making packaging determinations on a drug-specific basis eliminates payment incentives for hospitals to report certain HCPCS codes for drugs and allows hospitals flexibility in choosing to report all HCPCS codes for different dosages of the same drug or only the lowest dosage HCPCS code. Therefore, we proposed to continue our policy to make packaging determinations on a drug-specific basis, rather than a HCPCS code-specific basis, for those HCPCS codes that describe the same drug or biological but different dosages in CY 2025.
In order to propose a packaging determination that is consistent across all HCPCS codes that describe different dosages of the same drug or biological, we aggregated both our CY 2023 claims data and our pricing information, which is based on the ASP methodology, ( printed page 94240) generally ASP plus 6 percent, across all of the HCPCS codes that describe each distinct drug or biological in order to determine the mean units per day of the drug or biological in terms of the HCPCS code with the lowest dosage descriptor. The following drugs did not have pricing information available for the ASP methodology for the CY 2025 OPPS/ASC proposed rule; and, as is our current policy for determining the packaging status of other drugs, we used the mean unit cost available from the CY 2023 claims data to make the proposed packaging determinations for them: HCPCS code C9257 (Injection, bevacizumab, 0.25 mg); HCPCS code J3471 (injection, hyaluronidase, ovine, preservative free, per 1 usp unit (up to 999 usp units)); HCPCS code J3472 (Injection, hyaluronidase, ovine, preservative free, per 1000 usp units); HCPCS code J7100 (Infusion, dextran 40,500 ml); and HCPCS code J7110 (Infusion, dextran 75,500 ml).
For all other drugs and biologicals that have HCPCS codes describing different doses, we then multiplied the proposed weighted average ASP methodology based payment rate, which is generally ASP plus 6 percent, per-unit payment amount across all dosage levels of a specific drug or biological by the estimated units per day for all HCPCS codes that describe each drug or biological from our claims data to determine if the estimated per day cost of each drug or biological is less than or equal to the proposed CY 2025 drug packaging threshold of $140 (in which case all HCPCS codes for the same drug or biological would be packaged) or greater than the proposed CY 2025 drug packaging threshold of $140 (in which case all HCPCS codes for the same drug or biological would be separately payable). The proposed packaging status of each drug and biological HCPCS code to which this methodology would apply in CY 2025 is displayed in Table 133.
We proposed that our policy to make packaging determinations on a drug-specific basis, rather than a HCPCS code-specific basis, for those HCPCS codes that describe the same drug or biological but different dosages in CY 2025 would also apply to diagnostic radiopharmaceuticals. In order to propose a packaging determination that is consistent across all HCPCS codes that describe different dosages of the same diagnostic radiopharmaceutical, we would aggregate our CY 2023 claims data across all of the HCPCS codes that describe each distinct diagnostic radiopharmaceutical in order to determine the mean units per day of the diagnostic radiopharmaceutical in terms of the HCPCS code with the lowest dosage descriptor. We would then analyze the aggregate per day cost of the ( printed page 94241) diagnostic radiopharmaceutical to determine if the per day cost is less than or equal to the proposed CY 2025 diagnostic radiopharmaceutical packaging threshold of $630 (in which case all HCPCS codes for the same diagnostic radiopharmaceutical would be packaged) or greater than the proposed CY 2025 diagnostic radiopharmaceutical packaging threshold of $630 (in which case all HCPCS codes for the same diagnostic radiopharmaceutical would be separately payable). There are currently no diagnostic radiopharmaceuticals that this policy would apply to.
We did not receive any public comments on our proposal and are finalizing it without modification.
2. Payment for Drugs and Biologicals Without Pass-Through Status That Are Not Packaged
a. Payment for Specified Covered Outpatient Drugs (SCODs) and Other Separately Payable Drugs and Biologicals
Section 1833(t)(14) of the Act defines certain separately payable radiopharmaceuticals, drugs, and biologicals and mandates specific payments for these items. Under section 1833(t)(14)(B)(i) of the Act, a “specified covered outpatient drug” (known as a SCOD) is defined as a covered outpatient drug, as defined in section 1927(k)(2) of the Act, for which a separate APC has been established and that either is a radiopharmaceutical agent or a drug or biological for which payment was made on a pass-through basis on or before December 31, 2002.
Under section 1833(t)(14)(B)(ii) of the Act, certain drugs and biologicals are designated as exceptions and are not included in the definition of SCODs. These exceptions are—
- A drug or biological for which payment is first made on or after January 1, 2003, under the transitional pass-through payment provision in section 1833(t)(6) of the Act.
- A drug or biological for which a temporary HCPCS code has not been assigned.
- During CYs 2004 and 2005, an orphan drug (as designated by the Secretary).
Section 1833(t)(14)(A)(iii) of the Act requires that payment for SCODs in CY 2006 and subsequent years be equal to the average acquisition cost for the drug for that year as determined by the Secretary, subject to any adjustment for overhead costs and considering the hospital acquisition cost survey data collected by the Government Accountability Office (GAO) in CYs 2004 and 2005, and later periodic surveys conducted by the Secretary as set forth in the statute. If hospital acquisition cost data are not available, the law requires that payment be equal to payment rates established under the methodology described in section 1842(o), section 1847A, or section 1847B of the Act, as calculated and adjusted by the Secretary as necessary for purposes of paragraph (14). We refer to this alternative methodology as the “statutory default.” Most physician Part B drugs are paid at ASP plus 6 percent in accordance with section 1842(o) and section 1847A of the Act.
Section 1833(t)(14)(E)(ii) of the Act provides for an adjustment in OPPS payment rates for SCODs to consider overhead and related expenses, such as pharmacy services and handling costs. Section 1833(t)(14)(E)(i) of the Act required MedPAC to study pharmacy overhead and related expenses and to make recommendations to the Secretary regarding whether, and if so how, a payment adjustment should be made to compensate hospitals for overhead and related expenses. Section 1833(t)(14)(E)(ii) of the Act authorizes the Secretary to adjust the weights for ambulatory procedure classifications for SCODs to consider the findings of the MedPAC study.[141]
It has been our policy since CY 2006 to apply the same treatment to all separately payable drugs and biologicals, which include SCODs, and drugs and biologicals that are not SCODs. Therefore, we apply the payment methodology in section 1833(t)(14)(A)(iii) of the Act to SCODs, as required by statute, but we also apply it to separately payable drugs and biologicals that are not SCODs, which is a policy determination rather than a statutory requirement. For CY 2023 and subsequent years, we finalized a policy to apply section 1833(t)(14)(A)(iii)(II) of the Act to all separately payable drugs and biologicals, including SCODs. Although we do not distinguish SCODs in this discussion, we note that we are required to apply section 1833(t)(14)(A)(iii)(II) of the Act to SCODs; but we also are applying this provision to other separately payable drugs and biologicals, consistent with our history of using the same payment methodology for all separately payable drugs and biologicals.
For a detailed discussion of our OPPS drug payment policies from CY 2006 to CY 2012, we refer readers to the CY 2013 OPPS/ASC final rule with comment period (77 FR 68383 through 68385). In the CY 2013 OPPS/ASC final rule with comment period (77 FR 68386 through 68389), we first adopted the statutory default policy to pay for separately payable drugs and biologicals at ASP plus 6 percent based on section 1833(t)(14)(A)(iii)(II) of the Act. We have continued this policy of paying for separately payable drugs and biologicals at the statutory default for CYs 2014 through 2024.
In the case of a drug or biological during an initial sales period in which data on the prices for sales of the drug or biological are not sufficiently available from the manufacturer, section 1847A(c)(4) of the Act permits the Secretary to make payments that are based on WAC. Under section 1833(t)(14)(A)(iii)(II) of the Act, the amount of payment for a separately payable drug equals the average price for the drug for the year established under, among other authorities, section 1847A of the Act. As explained in greater detail in the CY 2019 PFS final rule, under section 1847A(c)(4) of the Act, although payments may be based on WAC, unlike section 1847A(b) of the Act (which specifies that payments using ASP or WAC must be made with a 6 percent add-on), section 1847A(c)(4) of the Act does not require that a particular add-on amount be applied to WAC-based pricing for this initial period when ASP data are not available. Consistent with section 1847A(c)(4) of the Act, in the CY 2019 PFS final rule (83 FR 59661 to 59666), we finalized a policy that, effective January 1, 2019, WAC-based payments for Part B drugs made under section 1847A(c)(4) of the Act will utilize a 3-percent add-on in place of the 6 percent add-on that was being used according to our policy in effect as of CY 2018. For the CY 2019 OPPS, we followed the same policy finalized in the CY 2019 PFS final rule (83 FR 59661 to 59666). Since CY 2020, we have continued to utilize a 3 percent add-on instead of a 6 percent add-on for drugs that are paid based on WAC pursuant to our authority under section 1833(t)(14)(A)(iii)(II) of the Act (84 FR 61318 and 85 FR 86039), which provides, in part, that the amount of payment for a SCOD is the average price of the drug in the year established under section 1847A of the Act. We also apply this provision to non-SCOD separately payable drugs. Because we establish the average price for a drug paid based on WAC under section 1847A of the Act as ( printed page 94242) WAC plus 3 percent instead of WAC plus 6 percent, we believe it is appropriate to price separately payable drugs paid based on WAC at the same amount under the OPPS. Our policy to pay for drugs and biologicals at WAC plus 3 percent, rather than WAC plus 6 percent, applies whenever WAC-based pricing is used for a drug or biological under section 1847A(c)(4). We refer readers to the CY 2019 PFS final rule (83 FR 59661 to 59666) for additional background on this policy.
Consistent with our current policy, payments for separately payable drugs and biologicals are included in the budget neutrality adjustments, under the requirements in section 1833(t)(9)(B) of the Act. Also, the budget neutral weight scalar is not applied in determining payments for these separately payable drugs and biologicals.
Separately payable drug, biological, and radiopharmaceutical payment rates are listed in Addenda A and B to the CY 2025 OPPS/ASC proposed rule (available on the CMS website).[142] These addenda provide the proposed CY 2025 payment rates based on the ASP methodology for separately payable nonpass-through drugs, biologicals, and radiopharmaceuticals and the ASP methodology for pass-through drugs, biologicals, and radiopharmaceuticals. Except for proposed payment rates for radiopharmaceuticals, these rates are based either on ASP information that is the basis for calculating payment rates for drugs and biologicals in the physician's office setting effective April 1, 2024, or WAC, AWP, or mean unit cost from CY 2023 claims data and updated cost report information available for the CY 2025 OPPS/ASC proposed rule. For nonpass-through therapeutic radiopharmaceuticals, payment rates are based on ASP data or mean unit cost. We proposed in section II.A.3.c.(5) to pay separately at mean unit cost for diagnostic radiopharmaceuticals with per day costs above the proposed threshold; the payment rates proposed for qualifying diagnostic radiopharmaceuticals are entirely mean unit cost. In general, these published proposed payment rates are not the same as the actual January 2025 payment rates. This is because payment rates for drugs, biologicals, and therapeutic radiopharmaceuticals with ASP information for January 2025 will be determined through the standard quarterly process where ASP data submitted by manufacturers for the third quarter of CY 2024 (July 1, 2024, through September 30, 2024) will be used to set the payment rates that are released for the quarter beginning in January 2025 in December 2024. In addition, in Addenda A and B to the CY 2025 OPPS/ASC proposed rule, payment rates for drugs, biologicals, and therapeutic radiopharmaceuticals for which there was no ASP, WAC, or AWP information available for April 2024, as well as all separately payable diagnostic radiopharmaceuticals, are based on mean unit cost in the available CY 2023 claims data. If new pricing information becomes available for payment for the quarter beginning in January 2025, we will price payment for these drugs, biologicals, therapeutic radiopharmaceuticals, and diagnostic radiopharmaceuticals based on their newly available information. Finally, there may be drugs, biologicals and therapeutic radiopharmaceuticals that have ASP, WAC, or AWP information available for the proposed rule (reflecting April 2024 ASP data) that do not have ASP, WAC, or AWP information available for the quarter beginning in January 2025. These drugs, biologicals and therapeutic radiopharmaceuticals would then be paid based on mean unit cost data derived from CY 2023 hospital claims. Therefore, the proposed payment rates listed in Addenda A and B to the CY 2025 OPPS/ASC proposed rule are not for January 2025 payment purposes and are only illustrative of the CY 2025 OPPS payment methodology using the most recently available information at the time of issuance of the CY 2025 OPPS/ASC proposed rule.
We note that payment amounts for most drugs separately payable under Medicare Part B are determined using the methodology in section 1847A of the Act, and in many cases, payment is based on the average sales price (ASP) plus a statutorily mandated 6 percent add-on.
For CY 2025, we proposed to clarify that only ASP data or, if ASP data are not available, mean unit cost data, would be used to set payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals under the OPPS as described further in section V.B.3.a of the proposed rule. We proposed for CY 2025 to use mean unit cost data to set payment rates for separately payable nonpass-through diagnostic radiopharmaceuticals for which we propose separate payment because their cost exceeds the per-day threshold. Otherwise, we are not proposing any changes to our policies for payment for separately payable drugs and biologicals; and we propose to continue our payment policy that has been in effect since CY 2013 to pay for separately payable drugs and biologicals in accordance with section 1833(t)(14)(A)(iii)(II) of the Act (the statutory default).
Most of the comments we received regarding our proposed policy for separately payable drugs, biologicals, and radiopharmaceuticals pertained to our proposal to pay separately for certain diagnostic radiopharmaceuticals in CY 2025. We refer readers to section II.A.3.c of this final rule with comment period for a summary of the comments we received, our responses to those comments, and our final policy for separate payment for certain diagnostic radiopharmaceuticals in CY 2025.
Comment: Multiple commenters expressed their approval for our proposal to pay separately payable for drugs and biologicals, but not radiopharmaceuticals, at ASP plus 6 percent. One commenter supported our proposal to continue to pay for new non-pass-through Part B drugs and biologicals during an initial sales period (two quarters) for which ASP pricing data are not yet available at a rate of wholesale acquisition cost (WAC) plus 3%. The commenter also supported our proposal that if ASP and WAC data are unavailable, Medicare would pay 95% of the average wholesale price (AWP).
Response: We appreciate the commenters' support for our policies.
After consideration of the public comments we received, we are finalizing without modification our proposals regarding the payment methodology for separately payable drugs, biologicals, and radiopharmaceuticals.
b. Biosimilar Biological Products
For CY 2024, we finalized the exception of biosimilars from the OPPS threshold packaging policy when their reference products are separately paid (88 FR 81783 through 81785). This policy allows for separate payment for biosimilars even if the biosimilar's per-day cost is below the packaging threshold if the biosimilar's reference product is separately paid. This policy removes the financial incentive to use a more expensive separately payable biological and promotes biosimilar use as a lower cost alternative to higher cost reference products.
Payment rates for drugs and biologicals (including biosimilars) under Medicare Part B are determined using the methodology in section 1847A of the Act, and in many cases, payment is based on the average sales price (ASP) plus a statutorily mandated 6 percent add-on. Additionally, Section 11403 of the IRA requires that a qualifying ( printed page 94243) biosimilar be paid at ASP plus 8 percent of the reference product's ASP rather than 6 percent during the applicable 5-year period. Section 1847A(b)(8)(B)(ii) of the Act defines the applicable 5-year period for a qualifying biosimilar for which payment has been made using ASP (that is, payment under section 1847A(b)(8) of the Act) as of September 30, 2022, as the 5-year period beginning on October 1, 2022. For a qualifying biosimilar for which payment is first made using ASP during the period beginning October 1, 2022, and ending December 31, 2027, the statute defines the applicable 5-year period as the 5-year period beginning on the first day of such calendar quarter of such payment (88 FR 81783). These payment rates are published in the quarterly release of Addendum B or ASP pricing files.
c. Invoice Drug Pricing for CY 2026
In recent years there has been an increasing number of drug and biological HCPCS codes for which ASP, WAC, AWP, and mean unit cost information is not available. These are often HCPCS codes for new drugs or biologicals that have been approved for marketing, but for which the manufacturer does not have sales data, and WAC, AWP, and mean unit cost information is not available. As a result, we are unable to assign a payable status indicator to these drugs or biologicals due to a lack of payment data. The numbers of drug and biological HCPCS codes without payment rates from Addendum B for the CY 2022 through CY 2024 OPPS/ASC final rules with comment period are listed in Table 134.
To provide appropriate payment rates for these drugs and biologicals without pricing data, we proposed to adopt an invoice pricing policy beginning in CY 2026. Because this policy necessitates significant operational changes to implement, we proposed to implement it beginning in CY 2026, rather than CY 2025. For CY 2025, we proposed that the affected drugs and biologicals would continue to be assigned a non-payable status indicator until we implement our invoice pricing policy, if adopted. We believe invoice pricing is appropriate for use under the OPPS because it provides temporary drug or biological cost information to generate a representative payment rate for a drug or biological and supports the utilization of new drug or biological HCPCS codes. Otherwise, the new drug and biological HCPCS codes would not receive payment under the OPPS, which would discourage their use by providers. Currently, the Physician Fee Schedule utilizes invoice pricing for drugs and biologicals when other types of pricing information are not available.
We proposed that, for separately payable drugs or biologicals for which CMS does not provide a payment rate in Addendum B, which would indicate to MACs that CMS does not have pricing information (specifically, that ASP, WAC, AWP, and mean unit cost information is not available to determine a payment rate), MACs would calculate the payment based on provider invoices. The drug or biological invoice cost would be the net acquisition cost minus any rebates, chargebacks, or post-sale concessions. Before calculating an invoice-based payment amount, MACs would use the provider invoice to determine that: (a) the drug is not policy packaged; and (b) the per-day cost of the drug, biological, therapeutic radiopharmaceutical or diagnostic radiopharmaceutical is above the threshold packaging amount, as applicable. If both conditions are met, we proposed that MACs would use the provider invoice amount to set a payment rate for the separately payable drug, biological, or radiopharmaceutical until its payment amount becomes available to CMS. We generally would expect invoice pricing to be temporary, lasting two to three quarters, for qualified drugs required to report ASP under 1847A of the Act. For drug products that are not required to report ASP under 1847A of the Act ( i.e., diagnostic pharmaceuticals), we proposed that invoice pricing may be used longer term until a MUC can be calculated. We proposed that we would not begin using invoice pricing for drugs, biologicals, and radiopharmaceuticals without pricing information until CY 2026 because we would need to make technical updates to outpatient hospital claims to allow the hospitals to report drug invoice pricing. We noted that we intended to work with the National Uniform Billing Committee (NUBC) in order to create a value code that would allow for the reporting of invoice prices of drugs, biologicals, and radiopharmaceuticals for purposes of this policy.
Comment: Some commenters opposed our proposed drug invoice pricing policy, citing their concerns that the operational burden this policy would place on providers may lead to providers forgoing reimbursement. These commenters believe it is not feasible to provide these invoices because it would require considerable time and effort to track, document, and submit the necessary information because of the varied arrangements through which hospitals acquire drugs, as well as pricing variability and discount structures. Furthermore, commenters have concerns with using acquisition cost minus any rebates, chargebacks, or post-sale concessions because rebates are often made months after sale and commenters believed it would impede cash flow to hold claims for months until the rebate process has completed.
These commenters urged CMS to work with manufacturers to report ASP and other pricing data in a timely ( printed page 94244) manner so that CMS will have up-to-date cost information. Alternatively, commenters suggested that CMS use WAC or AWP, as it does for other drugs without ASP data. WAC data is typically available in advance of ASP data and soon after a drug is on the market.
Response: We appreciate the feedback from the commenters. We note that this policy would only apply to drugs where ASP, WAC, AWP, or mean unit cost information is not available. We will continue to examine the operational effect of this policy. We note that this policy has been in effect under the PFS and there is no evidence that healthcare professionals paid under the PFS forgo reimbursement for drugs paid at invoice prices because of administrative burden. We also note that the proposed effective date for this policy is January 1, 2026, which would allow providers a year to prepare for any operational changes.
Comment: Some commenters opposed the proposed drug invoice pricing policy, citing concerns with participating 340B providers' disclosure of their 340B drug acquisition cost, which the commenters stated is proprietary data. Some commenters believed that we should exempt 340B providers from the proposed policy if we move forward with the proposal.
Response: We understand providers' concerns regarding 340B drug acquisition cost data, and we note that it is not the intention of this policy to collect 340B drug acquisition cost data. Our policy goal is to provide temporary payment for these new drugs until their ASP, WAC, or AWP becomes available and would likely affect only a few new drugs each quarter. Invoice-based payment will result in collection of drug acquisition cost data for only a few drugs for only a short period of time, and only if the provider wants to submit the invoice in order to receive payment for the drug. We also note that CMS does not disclose proprietary data; we only disclose summarized population drug payment data in mean unit cost for ratesetting purposes.
Comment: Some commenters supported our proposal to permit invoice pricing or suggested we permit the MACs to pay at WAC or continue our current approach to make retroactive adjustments to payment status and to update quarterly payment files as new data becomes available.
Response: We thank the commenters who supported our proposal to allow invoice pricing. We believe the invoice pricing policy we are finalizing in this rule is a more appropriate and efficient way for us to pay for these drugs than our current approach of making retroactive adjustments. We note that this invoice pricing proposal only applies when data from a number of other payment methodologies, including WAC, are not available.
After consideration of the public comments we received, we are finalizing our proposal without modification for CY 2026 to allow MACs to use the provider invoice amount to set a payment rate for a separately payable drug, biological, or radiopharmaceutical until its payment amount becomes available and CMS provides a payment rate in Addendum B. HCPCS code(s) with missing payment rate in Addendum B for a separately payable drug, biological, or radiopharmaceutical will indicate to MACs that CMS does not have pricing information (specifically, that ASP, WAC, AWP, and mean unit cost information is not available to determine a payment rate) for a product, and MACs would then calculate the payment for the product based on provider invoices.
3. Payment Policy for Radiopharmaceuticals
For a complete history of the OPPS payment policy for radiopharmaceuticals, we refer readers to the CY 2005 OPPS final rule with comment period (69 FR 65811), the CY 2006 OPPS final rule with comment period (70 FR 68655), and the CY 2010 OPPS/ASC final rule with comment period (74 FR 60524).
a. Payment Policy for Therapeutic Radiopharmaceuticals
In the CY 2023 OPPS/ASC final rule with comment period, we adopted as final our proposal to continue our longstanding payment policy for therapeutic radiopharmaceuticals for CY 2023 and subsequent years. Accordingly, this payment policy for therapeutic radiopharmaceuticals will continue to apply in CY 2025.
Specifically, our policy of paying for separately payable pass-through therapeutic radiopharmaceuticals under the ASP methodology adopted for separately payable drugs and biologicals described in section V.A.1 of the proposed rule will continue to apply for CY 2025. We will pay for separately payable nonpass-through therapeutic radiopharmaceuticals through a modified ASP methodology where we pay at ASP plus 6 percent if ASP data are available. However, if ASP information is unavailable for a separately payable nonpass-through therapeutic radiopharmaceutical, we will continue to base the payment rate on mean unit cost data derived from hospital claims. Our policy not to use WAC or AWP to establish payment for separately payable nonpass-through therapeutic radiopharmaceuticals if ASP is not available will continue for CY 2025. We explained our rationale in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60524 through 60525) when we first adopted our policy to apply the principles of separately payable drug pricing to therapeutic radiopharmaceuticals.
We note that in the CY 2024 OPPS final rule with comment period (88 FR 81786), we stated that the ASP payment methodology for separately payable nonpass-through therapeutic radiopharmaceuticals did allow for using WAC or AWP to establish a payment rate for these items. This was an error and conflicted with the policy implemented in CY 2010 and continued in subsequent years. The statement also conflicted with the policy that we proposed and finalized for CY 2023 and subsequent years in the CY 2023 OPPS/ASC final rule with comment period (87 FR 71969). The policy implemented in CY 2010 regarding ASP payment for separately payable nonpass-through therapeutic radiopharmaceuticals remains our intended policy. Therefore, we will pay for all nonpass-through separately payable therapeutic radiopharmaceuticals at ASP plus 6 percent based on the statutory default described in section 1833(t)(14)(A)(iii)(II) of the Act. For a full discussion of ASP-based payment for therapeutic radiopharmaceuticals, we refer readers to the CY 2010 OPPS/ASC final rule with comment period (74 FR 60520 through 60521). We will rely on CY 2023 mean unit cost data derived from hospital claims data for payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals for which ASP data are unavailable and update the payment rates for these products according to our usual process for updating the payment rates for separately payable drugs and biologicals on a quarterly basis if updated ASP information becomes available.
The proposed CY 2025 payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals were included in Addenda A and B of the CY 2025 OPPS/ASC proposed rule (which are available on the CMS website).[143]
The final CY 2025 payment rates for separately payable nonpass-through therapeutic radiopharmaceuticals are included in Addenda A and B of this ( printed page 94245) final rule with comment period (which are available on the CMS website).[144]
b. Payment Policy for Diagnostic Radiopharmaceuticals
For CY 2025, we proposed, as described in section II.A.3 of the CY 2025 OPPS/ASC proposed rule, to pay separately for diagnostic radiopharmaceuticals with a per day cost above our proposed diagnostic radiopharmaceutical packaging threshold (proposed at $630 for CY 2025). We proposed to pay for pass-through diagnostic radiopharmaceuticals based on ASP, WAC, and AWP.
We proposed to base the payment rate for separately payable nonpass-through diagnostic radiopharmaceuticals on mean unit cost data derived from hospital claims. As discussed in section II.A.3.c.(5), we did not propose to use ASP data when mean unit cost data are available for a separately payable nonpass-through diagnostic radiopharmaceutical, but we sought comment on using ASP for setting the payment rate for nonpass-through diagnostic radiopharmaceuticals in the future. Additionally, we did not propose to use WAC or AWP as a basis for payment for nonpass-through diagnostic radiopharmaceuticals when mean unit cost data derived from hospital claims is available. We believe that paying for nonpass-through diagnostic radiopharmaceuticals using mean unit cost would appropriately pay for the average price of a nonpass-through separately payable diagnostic radiopharmaceutical. In our view, MUC is an appropriate proxy for the average price for a diagnostic radiopharmaceutical for a given year, as it is calculated based on the average costs for a particular year and is directly reflective of the actual cost data that hospitals submit to CMS. As we stated in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60523), we believe that WAC or AWP is not an appropriate proxy to provide OPPS payment for radiopharmaceuticals because these pricing methodologies do not include discounts. Specifically, the absence of appropriate ASP reporting could result in payment for a separately payable diagnostic radiopharmaceutical based on WAC or AWP indefinitely, a result which we believe would be inappropriate, as these pricing metrics do not capture all of the pricing discounts that may be reflected in the ASP.
Additionally, we proposed to base the initial payment for new diagnostic radiopharmaceuticals with HCPCS codes that do not have pass-through status or claims data on ASP, and on the WAC for these products if ASP data for these diagnostic radiopharmaceuticals are not available. If the WAC also is unavailable, we proposed to make payment for new diagnostic radiopharmaceuticals at 95 percent of the products' most recent AWP. We believe the volume of products in this category will typically be very low; however, in these rare situations, we believe it would be appropriate to use ASP until a MUC is established for new diagnostic radiopharmaceuticals with HCPCS codes that do not have passthrough status or claims data.
Please refer to section II.A.3.c of this final rule with comment period for information regarding our payment policies for diagnostic radiopharmaceuticals, including our policy to pay separately for diagnostic radiopharmaceuticals above a certain cost threshold, comments we received, and our final policies for CY 2025. The final CY 2025 payment rates for separately payable nonpass-through diagnostic radiopharmaceuticals are included in Addenda A and B of this final rule with comment period (which are available on the CMS website).[145]
4. Payment for Blood Clotting Factors
For CY 2025, we proposed to continue our established policy to provide payment for blood clotting factors using the same methodology as other separately payable drugs and biologicals under the OPPS and to continue to pay a furnishing fee. For a full discussion of our established payment policy for blood clotting factors, please refer to the CY 2023 OPPS/ASC final rule with comment period (87 FR 71969 through 71970). In accordance with our policy as finalized in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66765), we will announce the actual figure of the percent change in the applicable CPI and the updated furnishing fee calculation based on that figure through the applicable program instructions and posting on the CMS website at https://www.cms.gov/medicare/payment/fee-for-service-providers/part-b-drugs/average-drug-sales-price.
We did not receive any public comments on our proposal and are finalizing it without modification.
5. Payment for Nonpass-Through Drugs, Biologicals, and Radiopharmaceuticals With HCPCS Codes but Without OPPS Hospital Claims Data
In the CY 2023 OPPS/ASC final rule with comment period, we adopted as final our proposal to continue our longstanding payment policy for nonpass-through drugs, biologicals, and radiopharmaceuticals with HCPCS codes but without OPPS hospital claims data for CY 2023 and subsequent years. Therefore, for CY 2025, this policy will continue to apply. For a detailed discussion of the payment policy and methodology, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70442 through 70443). Consistent with our policy, because we have no claims data and must determine if these products exceed the per-day cost threshold, we estimated the average number of units of each product that would typically be furnished to a patient during one day in the hospital outpatient setting and utilized the ASP methodology to determine whether their payment will be packaged as well as their payment status indicators.
We did not receive any public comments on our proposal and are finalizing it without modification.
6. Requirement in the CY 2025 Physician Fee Schedule Proposed Rule for HOPDs and ASCs To Report Discarded Amounts of Certain Single-Dose or Single-Use Package Drugs
Section 90004 of the Infrastructure Investment and Jobs Act (Pub. L. 117-9, November 15, 2021) (“the Infrastructure Act”) amended section 1847A of the Act to re-designate subsection (h) as subsection (i) and insert a new subsection (h), which requires manufacturers to provide a refund to CMS for certain discarded amounts from a refundable single-dose container or single-use package drug. We explained in the CY 2025 OPPS/ASC proposed rule that the CY 2025 PFS proposed rule included proposals related to the discarded drug refund policy, including proposals that may impact hospital outpatient departments (HOPDs) and ambulatory surgical centers (ASCs). Similar to our CY 2023 and CY 2024 notice in the OPPS/ASC proposed rule (87 FR 71988), we wanted to ensure interested parties were aware of these proposals and knew to refer to the CY 2025 PFS proposed rule for a full description of the proposed policy. Interested parties were asked to submit comments on any proposals to implement section 90004 of the Infrastructure Act to the CY 2025 PFS ( printed page 94246) proposed rule. We noted that public comments on these proposals would be addressed in the CY 2025 PFS final rule with comment period.
7. High-Cost/Low-Cost Threshold for Packaged Skin Substitutes
a. Background
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 74938), we unconditionally packaged skin substitute products into their associated surgical procedures as part of a broader policy to package all drugs and biologicals that function as supplies when used in a surgical procedure. As part of the policy to package skin substitutes, we also finalized a methodology that divides the skin substitutes into a high-cost group and a low-cost group, to ensure adequate resource homogeneity among APC assignments for the skin substitute application procedures (78 FR 74933). In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66886), we stated that skin substitutes are best characterized as either surgical supplies or devices because of their required surgical application and because they share significant clinical similarity with other surgical devices and supplies.
Skin substitutes assigned to the high-cost group are described by HCPCS codes 15271 through 15278. Skin substitutes assigned to the low-cost group are described by HCPCS codes C5271 through C5278. Geometric mean costs for the various procedures are calculated using only claims for the skin substitutes that are assigned to each group. Specifically, claims billed with HCPCS codes 15271, 15273, 15275, or 15277 are used to calculate the geometric mean costs for procedures assigned to the high-cost group, and claims billed with HCPCS codes C5271, C5273, C5275, or C5277 are used to calculate the geometric mean costs for procedures assigned to the low-cost group (78 FR 74935).
Each of the HCPCS codes described earlier are assigned to one of the following three skin procedure APCs according to the geometric mean cost for the code: APC 5053 (Level 3 Skin Procedures): HCPCS codes C5271, C5275, and C5277; APC 5054 (Level 4 Skin Procedures): HCPCS codes C5273, 15271, 15275, and 15277; or APC 5055 (Level 5 Skin Procedures): HCPCS code 15273. In CY 2024, the payment rate for APC 5053 (Level 3 Skin Procedures) was $599.02, the payment rate for APC 5054 (Level 4 Skin Procedures) was $1,739.33, and the payment rate for APC 5055 (Level 5 Skin Procedures) was $3,421.82. This information is also available in Addenda A and B of the CY 2024 final rule with comment period (88 FR 81540) (the Addenda A and B are available on the CMS website https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
We have continued the high-cost/low-cost categories policy since CY 2014. Under the current policy, skin substitutes in the high-cost category are reported with the skin substitute application CPT codes, and skin substitutes in the low-cost category are reported with the analogous skin substitute HCPCS C-codes. For a discussion of the CY 2014 and CY 2015 methodologies for assigning skin substitutes to either the high-cost group or the low-cost group, we refer readers to the CY 2014 OPPS/ASC final rule with comment period (78 FR 74932 through 74935) and the CY 2015 OPPS/ASC final rule with comment period (79 FR 66882 through 66885).
For a discussion of the high-cost/low-cost methodology that was adopted in CY 2016 and has been in effect since then, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70434 through 70435). Beginning in CY 2016, we adopted a policy where we determined the high-cost/low-cost status for each skin substitute product based on either a product's geometric mean unit cost (MUC) exceeding the geometric MUC threshold or the product's per day cost (PDC) (the total units of a skin substitute multiplied by the mean unit cost and divided by the total number of days) exceeding the PDC threshold. We assigned each skin substitute that exceeded either the MUC threshold or the PDC threshold to the high-cost group. In addition, we assigned any skin substitute with a MUC or a PDC that did not exceed either the MUC threshold or the PDC threshold to the low-cost group (87 FR 71976).
However, some skin substitute manufacturers have raised concerns about significant fluctuation in both the MUC threshold and the PDC threshold from year to year using the methodology developed in CY 2016. The fluctuation in the thresholds may result in the reassignment of several skin substitutes from the high-cost group to the low-cost group, which, under current payment rates, can be a difference of over $1,000 in the payment amount for the same procedure. In addition, these interested parties were concerned that the inclusion of cost data from skin substitutes with pass-through payment status in the MUC and PDC calculations would artificially inflate the thresholds. Skin substitute interested parties requested that CMS consider alternatives to the current methodology used to calculate the MUC and PDC thresholds and whether it might be appropriate to establish a new cost group in between the low-cost group and the high-cost group to allow for assignment of moderately priced skin substitutes to a newly created middle group.
We share the goal of promoting payment stability for skin substitute products and their related procedures as price stability allows hospitals using such products to more easily anticipate future payments associated with these products. We have attempted to limit year-to-year shifts for skin substitute products between the high-cost and low-cost groups through multiple initiatives implemented since CY 2014, including: establishing separate skin substitute application procedure codes for low-cost skin substitutes (78 FR 74935); using a skin substitute's MUC calculated from outpatient hospital claims data instead of an average of ASP plus 6 percent as the primary methodology to assign products to the high-cost or low-cost group (79 FR 66883); and establishing the PDC threshold as an alternate methodology to assign a skin substitute to the high-cost group (80 FR 70434 through 70435).
To allow additional time to evaluate concerns and suggestions from interested parties about the volatility of the MUC and PDC thresholds, in the CY 2018 OPPS/ASC proposed rule (82 FR 33627), we proposed that a skin substitute that was assigned to the high-cost group for CY 2017 would be assigned to the high-cost group for CY 2018, even if it did not exceed the CY 2018 MUC or PDC thresholds. We finalized this policy in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59347). For more detailed information and discussion regarding the goals of this policy and the subsequent comment solicitations in CY 2019 and CY 2020 regarding possible alternative payment methodologies for graft skin substitute products, please refer to the CY 2018 OPPS/ASC final rule with comment period (82 FR 59347); the CY 2019 OPPS/ASC final rule with comment period (83 FR 58967 to 58968); and the CY 2020 OPPS/ASC final rule with comment period (84 FR 61328 to 61331).
b. Packaged Skin Substitutes for CY 2025
For CY 2025, consistent with our policy since CY 2016, we proposed to continue to determine the high-cost/low-cost status for each skin substitute product based on either a product's ( printed page 94247) geometric MUC exceeding the geometric MUC threshold or the product's PDC (the total units of a skin substitute multiplied by the MUC and divided by the total number of days) exceeding the PDC threshold. Consistent with the methodology as established in the CY 2014 OPPS/ASC through CY 2018 OPPS/ASC final rules with comment period, we analyzed CY 2023 claims data to calculate the MUC threshold (a weighted average of all skin substitutes' MUCs) and the PDC threshold (a weighted average of all skin substitutes' PDCs). The proposed CY 2025 MUC threshold is $50 per cm2 (rounded to the nearest $1) and the proposed CY 2025 PDC threshold is $840 (rounded to the nearest $1). Also, the availability of a HCPCS code for a particular human cell, tissue, or cellular or tissue-based product (HCT/P) does not mean that that product is appropriately regulated solely under section 361 of the PHS Act and the FDA regulations in 21 CFR part 1271. Manufacturers of HCT/Ps should consult with the FDA Tissue Reference Group (TRG) or obtain a determination through a Request for Designation (RFD) on whether their HCT/Ps are appropriately regulated solely under section 361 of the PHS Act and the regulations in 21 CFR part 1271.
For CY 2025, as we did for CY 2024, we proposed to assign each skin substitute that exceeds either the MUC threshold or the PDC threshold to the high-cost group. In addition, we propose to assign any skin substitute that does not exceed either the MUC threshold or the PDC threshold to the low-cost group except that we propose that any skin substitute product that is assigned to the high-cost group in CY 2024 would be assigned to the high-cost group for CY 2025, regardless of whether it exceeds or falls below the CY 2025 MUC or PDC threshold. This policy was established in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59346 through 59348).
For CY 2025, we proposed to continue to assign skin substitutes with pass-through payment status to the high-cost category. We proposed to assign skin substitutes with pricing information but without claims data to calculate a geometric MUC or PDC to either the high-cost or low-cost category based on the product's ASP plus 6 percent payment rate as compared to the MUC threshold. If ASP is not available, we proposed to use WAC plus 3 percent to assign a product to either the high-cost or low-cost category. Finally, if neither ASP nor WAC is available, we proposed to use 95 percent of AWP to assign a skin substitute to either the high-cost or low-cost category. We proposed to continue to use WAC plus 3 percent instead of WAC plus 6 percent to conform to our proposed policy described in section V.B.2.b of the proposed rule to establish a payment rate of WAC plus 3 percent for separately payable drugs and biologicals that do not have ASP data available. We proposed that any skin substitute product that is assigned a code in the HCPCS A2XXX series would be assigned to the high-cost skin substitute group including new products without pricing information. New skin substitutes without pricing information that are not assigned a code in the HCPCS A2XXX series would be assigned to the low-cost category until pricing information is available to compare to the CY 2024 MUC and PDC thresholds. For a discussion of our policy under which we assign skin substitutes without pricing information that are not assigned a code in the HCPCS A2XXX series to the low-cost category until pricing information is available, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70436).
The CY 2025 OPPS/ASC proposed rule (89 FR 59373 through 593777) includes the proposed CY 2025 cost category assignment for each skin substitute product.
Comment: The HOP Panel recommended, and several commenters supported, ending the packaging of the graft skin substitute administration add-on codes (CPT codes 15272, 15274, 15276, and 15278; HCPCS codes C5272, C5274, C5276, and C5278). The HOP Panel and the commenters requested that these codes be assigned to APCs that reflect the estimated costs of these service codes. Commenters claimed that packaging the graft skin substitute administration add-on codes eliminates the variation in payment for wound care treatments based on the size of the wound. They assert that providers are discouraged from treating wounds between 26 and 99 cm2 and over 100 cm2 in the outpatient hospital setting because of the financial losses they experience to provide such care. One of the commenters cited a recent study finding that between 2014 and 2019 treatment for larger area wounds has shifted from the outpatient hospital setting to the physician office setting where payment for individual services better reflects their cost because of the fee-for-service structure of physician payment. Commenters believe that packaging graft skin substitute administration add-on codes disrupts the methodology of how the American Medical Association (AMA), the organization that manages CPT service codes, intended graft skin substitute procedures to be paid. The commenters explained that CPT codes describe the actual amount of the graft skin substitute product that is used for an individual service when the amount of product used is 25 cm2 or more. Commenters believe that when more than 25 cm2 of product is used for a service that the cost of product used in excess of 25 cm2 should be paid at cost by making the add-on code reporting the additional product separately payable in addition to the cost to apply the graft skin substitute product. Commenters believe always packaging the cost of graft skin substitute products into the cost of the associated application procedures no matter how much product is used is an indiscriminate form of payment that, according to the commenters, does not promote payment accuracy or patient care and may raise health equity concerns because providers are not compensated for the cost of using more than 25 cm2 of graft skin substitute product for a wound. One commenter asserted that providers rarely experience a financial gain from performing a graft skin substitute application procedure and, in most cases, providers experience a financial loss when performing graft skin substitute procedures.
Response: We do not agree that we should pay separately for graft skin substitute add-on codes under the OPPS. The OPPS is a prospective payment system rather than a fee-for-service payment system. That means that we generally attempt to make one payment for all the services billed with the primary medical procedure, including add-on procedures such as the ones described by CPT codes 15272, 15274, 15276, and 15278, and HCPCS codes C5272, C5274, C5276, and C5278. More specifically, we calculate the OPPS payment rate by first calculating the geometric mean cost of the procedure. This calculation includes claims for individual services that used a lower level of resources and claims for individual services that used a higher level of resources. The resulting geometric mean cost will reflect the average service cost for a given medical procedure. Next, we group the medical procedure with other medical procedures with clinical and resource similarity in an APC and calculate the geometric mean of these related procedures to generate a base payment rate for all procedures assigned to the APC. Skin substitutes are surgical supplies and are packaged into the cost of the associated procedure. The application of graft skin substitutes ( printed page 94248) cannot occur unless a graft skin substitute is used. So, the cost of the product will be reflected in the overall cost of the application procedure.
Despite the text of the descriptors for CPT codes 15271 and 15275 and HCPCS codes C5271 and C5275, the geometric mean service cost of these procedures is likely not the cost of the procedures plus 25 cm2 of graft skin substitute product. Instead, the geometric mean service cost should reflect the geometric mean of the cost of the graft skin substitute application procedure plus the cost of graft skin substitute product for all procedures where less than 100 cm2 of product is used. The commenters claimed that most graft skin substitute procedures use substantially more than 25 cm2 of graft skin substitute product. Therefore, the geometric mean cost of these procedures would account for the average amount of product used by providers over 25 cm2 .
A prospective payment system like the OPPS is designed to pay providers the geometric mean cost of the primary service they provide, and such a system encourages efficiencies and cost-savings in the administration of health care. However, a prospective payment system is not intended to discourage providers from rendering medically necessary care to patients. For example, it is possible that a provider could experience a financial loss when they perform a service where a patient receives 85 cm2 of a graft skin substitute product, but that same provider could see a financial gain when the next patient receives a skin graft where 40 cm2 of product is used. Paying separately for add-on codes for the administration of graft skin substitutes in a prospective payment system defeats the goals of such a payment system. Therefore, we will continue to package the add-on codes for the administration of graft skin substitutes in the OPPS to encourage cost-savings and efficiencies with wound care treatment. If providers are paid at cost or nearly at cost for each individual service they render, there is no incentive for them to control costs.
Add-on codes for the administration of graft skin substitutes should be packaged with the primary medical service to be able to establish a geometric mean payment rate that gives providers incentives to keep their costs in line with typical providers throughout the Medicare program. The need for cost efficiencies in the application of graft skin substitutes to treat wounds is no different than the need for cost efficiencies in other procedures administered in the outpatient hospital setting. Packaging product costs with the costs of the associated procedures is not an indiscriminate method of paying for medical care nor is it a system where providers lose money on most of the procedures they administer and rarely receive more payment than the cost of the procedure. Prospective payment methodology helps to control the cost of hospital care, while incentivizing providers to provide quality care in an efficient manner. Packaging add-on codes to have a uniform distribution of costs for individual procedure is critical for prospective payment. Therefore, we believe that add-on codes, including the add-on codes for the administration of graft skin substitutes, should remain packaged to maintain the integrity of the OPPS.
Comment: The HOP Panel recommended, and several commenters supported, ensuring that the payment rate for graft skin substitute procedures be the same no matter where on the body the graft skin substitute product is applied to the patient. There are four graft skin substitute application procedures for high-cost skin substitute products (CPT codes 15271, 15273, 15275, and 15277) and a similar four graft skin substitute application procedures for low-cost skin substitute products (HCPCS codes C5271, C5273, C5275, and C5277). Commenters claim that the cost to apply graft skin substitute products does not depend on the location of the wound because the same amount of product is used on the wound and the same clinical resources are used to treat the wound independent of the location of the wound. Other commenters made a similar request, asking that CPT code 15277 (Application of skin substitute graft to face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits, total wound surface area greater than or equal to 100 sq cm; first 100 sq cm wound surface area, or 1 percent of body area of infants and children) that is currently assigned to APC 5054 (Level 4 Skin Procedures) be reassigned to APC 5055 (Level 5 Skin Procedures). That would mean that the two graft skin substitute application procedures for the application of high-cost skin substitute products for wounds greater than 100 cm2 (CPT codes 15273 and 15277) would be in the same APC. One commenter expressed their concern that CMS seems to not understand that provider resources and graft skin substitute product used for the procedures described by CPT codes 15273 and 15277 and for the procedures described by HCPCS codes C5273 and C5277 are the same, and that CPT codes 15273 and 15277 must be assigned to the same APC and C5273 and C5277 also must be assigned to the same APC.
Response: The reason there are four CPT codes describing graft skin substitute application services is that there are different CPT codes for applying graft skin substitutes for wounds up to 100 cm2 and for wounds that are greater than 100 cm2 ; and there are different CPT codes for applying graft skin substitutes to the trunk, arms, and legs as compared to the face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, fingers, and toes. We appreciate commenters' concerns and note that the two current codes describing the application of high-cost graft skin substitutes for wounds less than 100 cm2 (CPT codes 15271 and 15275) have been assigned to the same APC (5054), and the two current codes describing the application of low-cost graft skin substitutes for wounds less than 100 cm2 (HCPCS codes C5271 and C5275) have been assigned to the same APC (5053). Because they are currently included in the same APC, the OPPS payment for them is the same; and this payment policy is consistent with the recommendation from the HOP Panel and other commenters. This means for the application of graft skin substitute products up to 100 cm2 , the location where the graft skin substitute is applied does not affect the payment rate for the service.
We note that the code describing the application of high-cost products for wounds that are greater than 100 cm2 on the trunk, arms, and legs (CPT code 15273) has been assigned to a higher-paying APC (APC 5055) than the APC assignment for the code describing the application of high-cost graft skin substitute products for wounds greater than 100 cm2 on the face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hand, feet, fingers, and toes (CPT code 15277), which is assigned to APC 5054. Likewise, the code describing the application of low- cost products for wounds that are greater than 100 cm2 on the trunk, arms, and legs (HCPCS code C5273) has been assigned to a higher-paying APC (APC 5054) than the code for the application of low-cost graft skin substitute products for wounds greater than 100 cm2 on the face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hand, feet, fingers, and toes (HCPCS code C5277), which is assigned to APC 5054. The differences in costs that have determined APC assignments for these services for wounds greater than 100 cm2 have been supported by historical cost data. We also note that none of these service codes are in violation of the 2 times rule, which requires that the geometric mean cost of significant items ( printed page 94249) and services within an APC group to be no more than two-times the geometric mean cost of the lowest geometric mean cost for a significant item or service within the same APC group.
In response to the claim of one of the commenters that we do not understand that similar amounts of product and resources are used for the procedures described by CPT codes 15273 and 15277 or for the procedures described by HCPCS codes C5273 and C5277, we note that the cost data for CY 2023 and for many years prior show substantial cost differences between the procedures described by CPT codes 15273 and 15277 and likewise for the procedures described by HCPCS codes C5273 and C5277. We set our payment rates based on claims cost data which continue to show that these procedures have substantial cost differences and should be assigned to different APCs.
Comment: The HOP Panel recommended, and several commenters supported, that all new graft skin substitute products be assigned to the low-cost group whether they have a Q-code or an A-code until cost data become available for the product. Commenters believe it is not appropriate that products assigned Q-codes are assigned to the low-cost group while products assigned A-codes are assigned to the high-cost group. Commenters note that A-codes are being assigned to graft skin substitute products that have FDA 510(k) clearance but are not synthetic products, which conflicts with the expectation of the commenters that only graft skin substitute products that would have been described by the now-deleted HCPCS code C1849 (Skin substitute, synthetic, resorbable, per square centimeter) be assigned to the high-cost group. More broadly, commenters believed that no category of graft skin substitute products should be assigned to the high-cost group until there is cost data supporting that assignment.
Response: We appreciate the concerns of the commenters. However, we decided on an approach that would ensure that any graft skin substitute product that could potentially have been described by deleted HCPCS code C1849 be included in the high-cost group. As explained in the CY 2023 OPPS final rule (87 FR 71980 and 71981), we wanted to ensure that graft skin substitute products that were described by HCPCS code C1849 or could potentially be described by HCPCS code C1849 would be granted time to develop the cost data necessary to allow us to determine if the product should stay in the high-cost group, which provides stability for the payment of these graft skin substitute products. We wanted to avoid having products with less than two years of claims data that were originally in the high-cost group be reassigned to the low-cost group simply because of a lack of available data.
Also, as discussed in the CY 2023 OPPS final rule (87 FR 71981), the current categorization of skin substitutes as either synthetic or non-synthetic is not mutually exclusive given the expansion of skin substitute products that may contain both biological and synthetic elements. Having products with both biological and synthetic elements leads to difficulty defining which of the products assigned to the A2XXX series would be considered “synthetic” and described by HCPCS code C1849. Therefore, for CY 2023, we finalized a policy, which will continue for CY 2025, to assign to the high-cost group any skin substitute product that is assigned a code in the HCPCS A2XXX series including new products without pricing information. This policy gives the broadest definition of products that could have been described by HCPCS code C1849 and ensures that none of those graft skin substitute products would be assigned to the low-cost group until we receive cost data for them.
Comment: One commenter supported having us realign both the high-cost and low-cost application procedure codes to potentially higher-paying APC groups that reflect the current average sales prices of graft skin substitute products as manufacturers now are required to submit average sales prices for graft skin substitute products. The commenter believes combining ASP prices for graft skin substitutes and the cost of the graft skin substitute application procedures would better reflect the costs of those procedures than our current methodology of using cost data from claims to assign application procedures to APCs.
Response: We disagree with the commenter that using ASP instead of using claims cost data would be a preferrable method for estimating the graft skin substitute product cost of graft skin substitute application procedures. It is unclear from the commenter's suggestion how the product cost of the graft skin substitute would be calculated if not using the charges reported by providers. Presumably, their approach would involve extracting the units of graft skin substitute product used on a particular packaged service and then multiplying by an ASP on file to revise the cost of packaged procedure to reflect the ASP price of the graft skin substitute product units. We do not believe this is a feasible approach and it appears to be a different approach to pricing one group of packaged supplies as compared to how all other packaged supplies are priced in the OPPS. We normally use a provider's reported charges for supplies and use the appropriate cost-to-charge ratio to estimate the contribution of the supply cost to the overall cost of the procedure.
Comment: One commenter supported our current policy not to assign graft skin substitute products that are not in sheet form ( e.g., gel, powder, ointment, foam, liquid, or injected) to any APC group, because these products cannot be reported with the graft skin substitute application codes of CPT codes 15271 through 15278 (the high-cost group) or with HCPCS codes C5271 through C5278 (the low-cost group). The commenter notes that skin substitutes that are not in sheet form are used primarily for clinic visits and the debridement of chronic wounds. Also, according to the commenter, the use of skin substitutes that are not in sheet form does not conform to the AMA's directions for the application of skin substitute products.
Response: We appreciate the commenter's support of our policy.
Comment: One commenter requested that skin substitute products that receive an FDA approval through either the Premarket Approval (PMA) process, the Biological License Application (BLA) process, or the New Drug Application (NDA) process no longer be packaged in the OPPS, but instead be separately payable at a rate of ASP + 6 percent. In addition, the commenter requested that two previously approved skin substitute products, Apligraf and Dermagraft, should no longer be packaged and should be paid separately at a rate of ASP + 6 percent because these products are now regulated by the Center for Biologics Evaluation and Research (CBER) within the FDA. CBER is the center within FDA that regulates biological products for human use. The commenter notes the rigorous process that biological products must go through to confirm their safety and effectiveness to receive either a PMA, BLA, or NDA approval. The commenter states that when Apligraf and Dermagraft received their FDA approval, the products had a review process comparable to the reviews done for a PMA, BLA, or NDA approval. The commenter believes the intensive review process that skin substitute products undergo to get a PMA, BLA, or NDA approval should allow those products to be separately paid rather than packaged in the OPPS.
Response: The commenter appears to be referencing comments that were previously made for the CY 2014 OPPS/ ( printed page 94250) ASC final rule with comment period (78 FR 74931) where commenters asserted that specified covered outpatient drugs (SCODs) under section 1833(t)(14)(B) of the Act cannot be packaged in the OPPS and instead must be paid separately. Section 1833(t)(14)(B) of the Act defines a SCOD as a “covered outpatient drug (as defined in section 1927(k)(2))”. Covered outpatient drugs under section 1927(k)(2) of the Act are generally limited to products approved as drugs by the FDA, biologicals licensed under section 351 of the Public Health Service Act, and insulin. However, skin substitute products that have either a PMA, BLA, or NDA are still considered to be supplies under the policy we implemented in CY 2014 OPPS/ASC final rule with comment period (78 FR 74938). Under our policy, we unconditionally packaged skin substitute products into their associated surgical procedures as part of a broader policy to package all drugs and biologicals that function as supplies when used in a surgical procedure. Finally, as we explained in finalizing our policies of packaging diagnostic radiopharmaceuticals and contrast agents in the CY 2008 OPPS final rule (72 FR 66766), CMS has the authority to package the payment of SCODs in the OPPS and we may consider additional packaging options for SCODs and other separately payable drugs in the future (78 FR 74931).
Comment: Several commenters supported our current skin substitute payment policy to assign graft skin substitute products to either a high-cost or a low-cost group based on the product's cost. Likewise, commenters also supported our policy of keeping graft skin substitute products in the high-cost group once the cost of the product exceeds either the MUC or the PDC threshold for at least one year even if in future years the cost of the product is less than either the MUC or PDC threshold.
Response: We appreciate the commenters' support of our policies.
Comment: The manufacturer of the products described by HCPCS codes Q4122 (Dermacell, dermacell awm or dermacell awm porous, per square centimeter) and Q4150 (Allowrap dds or dry, per square centimeter) requested that these graft skin substitute products continue to be assigned to the high-cost skin substitute group for CY 2025.
Response: Based on their cost data and our policies, we are finalizing that both HCPCS codes Q4122 (Dermacell, dermacell awm or dermacell awm porous, per square centimeter) and Q4150 (Allowrap dds or dry, per square centimeter) will remain in the high-cost group for CY 2025.
Comment: The manufacturer of the products described by HCPCS codes Q4148 (Neox cord 1k, neox cord rt, or clarix cord 1k, per square centimeter) and Q4156 (Neox 100 or clarix 100, per square centimeter) requested that these graft skin substitute products continue to be assigned to the high-cost skin substitute group for CY 2025.
Response: Based on their cost data and our policies, we are finalizing that both HCPCS codes Q4148 (Neox cord 1k, neox cord rt, or clarix cord 1k, per square centimeter) and Q4156 (Neox 100 or clarix 100, per square centimeter) will remain in the high-cost group for CY 2025.
After consideration of the public comments we received, we are finalizing our proposals without modification. Our final policies are to:
- Continue to assign skin substitutes with pass-through payment status to the high-cost category.
- Assign skin substitutes with pricing information but without claims data to calculate a geometric MUC or PDC to either the high-cost or low-cost category based on the product's ASP plus 6 percent payment rate as compared to the MUC threshold. If ASP is not available for the product, we will use WAC plus 3 percent to assign a product to either the high-cost or low-cost category. Finally, if neither ASP nor WAC is available, we will use 95 percent of AWP to assign a skin substitute to either the high-cost or low-cost category.
- Continue to use WAC plus 3 percent instead of WAC plus 6 percent to conform to our policy described in section V.B.2.b of this final rule with comment period to establish a payment rate of WAC plus 3 percent for separately payable drugs and biologicals that do not have ASP data available.
- Assign any skin substitute product that is assigned a code in the HCPCS A2XXX series to the high-cost skin substitute group, including new products without pricing information. New skin substitutes without pricing information that are not assigned a code in the HCPCS A2XXX series would be assigned to the low-cost category until pricing information is available to compare to the CY 2024 MUC and PDC thresholds.
Finally, we have updated the MUC and PDC thresholds for CY 2025. The final MUC threshold will be $50 per cm2 (rounded to the nearest $1) and the final PDC threshold will be $833 (rounded to the nearest $1). Table 135 includes the final CY 2024 cost category assignment for each skin substitute product.
( printed page 94251) ( printed page 94252) ( printed page 94253) ( printed page 94254) ( printed page 94255) ( printed page 94256)8. Radioisotopes Derived From Non-Highly Enriched Uranium (non-HEU) Sources/Radioisotopes Derived From Domestically Produced Mo-99
Radioisotopes are widely used in modern medical imaging, particularly for cardiac imaging and predominantly for the Medicare population. Technetium-99m (Tc-99m), the radioisotope used in the majority of such diagnostic imaging services, is produced through the radioactive decay of molybdenum-99 (Mo-99). Historically, most of the Mo-99 used in the United States was produced in legacy reactors outside of the United States using highly enriched uranium (HEU).
The United States wanted to eliminate domestic reliance on these reactors and promoted the conversion of all medical radioisotope production to non-HEU sources. Alternative methods for producing Tc-99m without HEU are technologically and economically viable, but it was expected that this change in the supply source for the radioisotope used for modern medical imaging would introduce increased costs into the payment system that would not be fully accounted for in the historical claims data until all Tc-99m was produced from non-HEU sources.
Therefore, beginning in CY 2013, we finalized a policy to provide an additional payment of $10 for the marginal cost for radioisotopes produced by non-HEU sources (77 FR 68323). Under this policy, hospitals report HCPCS code Q9969 (Tc-99m from non-highly enriched uranium source, full cost recovery add-on per study dose) once per dose along with any diagnostic scan or scans furnished using Tc-99m as long as the Tc-99m doses used can be certified by the hospital to be at least 95 percent derived from non-HEU sources (77 FR 68323).
We stated in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68321) that our expectation was that this additional payment would be needed for the duration of the industry's conversion to alternative methods of producing Tc-99m without HEU. We also stated that we would reassess, and propose, if necessary, on an annual basis whether such an adjustment continued to be necessary and whether any changes to the adjustment were warranted (77 FR 68321). The Secretaries of Energy and Health and Human Services issued a certification regarding the supply of non-HEU-sourced Mo-99 effective January 2, 2022 (86 FR 73270). Mo-99 is the precursor material from which Tc-99m is sourced. The certification by the Secretary of Energy stated that there was a sufficient global supply of Mo-99 produced without the use of HEU available to meet the needs of patients in the United States. In the CY 2023 OPPS/ASC final rule with comment period, we stated that we believed the conversion to non-HEU sources of Tc-99m had reached a point where it was necessary to reassess our policy of providing an additional payment of $10 for the marginal cost for radioisotopes produced by non-HEU sources (87 FR 71987). ( printed page 94257)
Historically, diagnostic radiopharmaceuticals have been packaged into the cost of the associated diagnostic imaging procedure no matter the per-day cost (we are adopting a policy beginning in CY 2025 to pay separately for higher cost diagnostic radiopharmaceuticals). The cost of the radiopharmaceutical, whether it is included as a part of the cost of the diagnostic imaging procedure or is separately paid, is reported through Medicare claims data. Medicare claims data used to set payment rates under the OPPS generally are from two years prior to the payment year.
As we explained in the CY 2023 OPPS/ASC final rule with comment period (87 FR 71987), the claims data we used to set payment rates for CY 2024 (generally CY 2022 claims data) contained claims for diagnostic radiopharmaceuticals that reflect both HEU-sourced Tc-99m and non-HEU-sourced Tc99m, rather than radiopharmaceuticals sourced solely from non-HEU Tc-99m. The cost of HEU-sourced Tc-99m is substantially lower than the cost of non-HEU-sourced Tc-99m. Therefore, we explained that providers who use radiopharmaceuticals in CY 2024 that contain only non-HEU-sourced Tc-99m might not receive a payment that is reflective of the radiopharmaceutical's current cost without the add-on payment. We believed that extending the additional $10 add-on payment described by HCPCS code Q9969 for non-HEU-sourced Tc-99m through the end of CY 2024 would ensure adequate payment for non-HEU-sourced Tc-99m. Starting in CY 2025, we believed the Medicare claims data utilized to set payment rates (likely CY 2023 claims data) would only include claims for diagnostic radiopharmaceuticals that utilized non-HEU-sourced Tc-99m, meaning the data would reflect the full cost of the Tc-99m diagnostic radiopharmaceuticals that would be used by providers in CY 2025. As a result, we believed there would no longer be a need for the additional $10 add-on payment for CY 2025 or future years.
The conversion of the last major global Mo-99 producer from HEU to Low Enriched Uranium (LEU) was previously expected to be completed by December 31, 2022, but the conversion did not occur until March 2023. As a result, it is possible that some claims for diagnostic radiopharmaceuticals in CY 2023 would report the cost of HEU-sourced Tc-99m. This means that in CY 2025, as in CY 2024, there is the possibility that the payment rate for procedures using diagnostic radiopharmaceuticals could be lower than the costs providers will incur for these procedures because providers will only have access to non-HEU-sourced Tc-99m. Therefore, we adopted a policy in the CY 2024 OPPS final rule with comment period (88 FR 81803) to extend the additional $10 add-on payment described by HCPCS code Q9969 for non-HEU-sourced Tc-99m through the end of CY 2025 to continue to ensure adequate payment for non-HEU-sourced Tc-99m.
Recently, the Department of Energy and other interested parties raised another issue affecting the domestic supply chain for Mo-99 and Tc-99 that, left unaddressed, could cause payment inequity among outpatient hospital providers. Foreign Mo-99 production has historically been subsidized by foreign governments, resulting in prices below the true cost of production. These artificially low, government-subsidized prices have created a disincentive for investments in Mo-99 production infrastructure, and they also created a barrier to entry for new producers, including U.S. companies. This in turn has resulted in unreliable production and periodic shortages. In response to the 2009-2010 shortages, Congress passed the American Medical Isotopes Production Act of 2012 (AMIPA), which directs the Secretary of Energy to provide financial and technical support to U.S. companies working to build new irradiation and manufacturing facilities to produce Mo-99 without HEU.
It was expected that the transition from HEU to LEU-based production would also involve the transition to a Full Cost Recovery pricing model; however, it does not appear that this transition has occurred in practice. Foreign producers continue to rely on multipurpose nuclear research reactors for Mo-99 production, and the global Mo-99 supply chain has not established a system of verifying that all of the costs attributable to Mo-99 production are being incorporated into the price of the product.
U.S. companies have made significant progress towards establishing the infrastructure needed for large-scale Mo-99 production. Unlike many foreign producers, U.S. companies must price their products high enough to cover the full cost of operating their production facilities. Based in part on the differences in pricing models, U.S. companies have experienced challenges in competing with foreign producers for customers. Currently, there is no domestic production of Mo-99.
Once U.S. companies initiate or resume Mo-99 production, the difference in pricing models will likely create a payment inequity, as hospitals purchasing Tc-99m derived from domestically produced Mo-99 would likely pay higher prices than those purchasing Tc-99m derived from imported Mo-99. We proposed to address the payment inequity resulting from the higher cost of domestically produced Tc-99m by establishing a new add-on payment of $10 per dose for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99 starting on January 1, 2026, using our equitable adjustment authority under section 1833(t)(2)(E) of the Act. We stated that we believe the $10 add-on payment for domestically produced Tc-99m would ensure equitable payments by paying providers who use domestically produced Tc-99m radiopharmaceuticals when available an amount that reflects the anticipated higher cost of these products. We explained that the $10 add-on payment will help to ensure provider and beneficiary access to domestically produced Tc-99m radiopharmaceuticals by providing an additional payment amount that addresses the additional costs of domestically produced Tc-99m radiopharmaceuticals. The Department of Energy, National Nuclear Security Administration, (DOE/NNSA) would establish the criteria to certify whether the Tc-99m radiopharmaceutical dose is domestically produced and eligible for the add-on payment, which would be included in the CY 2026 OPPS/ASC proposed rule. The CY 2026 OPPS/ASC proposed rule would include additional details on how providers would bill for this add-on payment in CY 2026.
Comment: Multiple commenters supported our proposal to establish a new add-on payment of $10 per dose for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99.
Response: We appreciate the support of the commenters for our proposal.
Comment: Multiple commenters expressed support for an add-on payment for domestically produced Tc-99m radiopharmaceuticals, but the commenters expressed several concerns about how the add-on payment policy would work. Some commenters believe the proposed payment amount for the add-on of $10 is too low and encouraged us to work with interested parties to determine a different payment amount that would adequately cover the cost difference between domestically produced Tc-99m radiopharmaceuticals and foreign produced Tc-99m radiopharmaceuticals. One commenter asked us to provide additional analysis to support the payment amount for the add-on payment. ( printed page 94258)
Commenters had concerns about the effectiveness of the policy based on their experiences with the current add-on payment for Tc-99m radiopharmaceuticals produced from non-HEU sources. Commenters do not believe the add-on payment for Tc-99m radiopharmaceuticals produced from non-HEU sources has substantially influenced the types of Tc-99m radiopharmaceuticals purchased by outpatient hospitals or remedied cost differences between HEU and non-HEU Tc-99m for the companies who supply Tc-99m radiopharmaceuticals to hospitals. Commenters encouraged us to work with DOE/NNSA and other government agencies to ensure that domestic Mo-99 producers and Tc-99m generator manufacturers receive assistance to handle the higher costs of their products.
Commenters were also concerned about how a Tc-99m radiopharmaceutical will be defined as a domestically produced product. They asked that CMS and other government agencies show flexibility when coming up with a definition of domestically produced Tc-99m radiopharmaceuticals to avoid increasing production costs. Commenters noted that Tc-99m generators often mix Mo-99 from several sources, which may make it challenging for suppliers to adhere to strict domestic Tc-99m content requirements. Commenters want to ensure that the tracking and documentation requirements for outpatient hospital providers to prove the Tc-99m radiopharmaceuticals they use are domestically produced are easy to understand and use, and do not add an additional burden to hospitals.
Response: We will continue to monitor the differences in cost between domestically produced Tc-99m and foreign produced Tc-99m for future rulemaking. Using available information from the Organization for Economic Co-operation and Development, Nuclear Energy Agency (OECD/NEA), the methodology for a $10 add-on payment accounts for the per-dose cost of implementing full-cost recovery pricing. As such, DOE/NNSA recommends maintaining the $10 value as the add-on payment transitions from a non-HEU payment to a domestically sourced Tc-99m payment. We believe $10 is an appropriate amount for an add-on payment for domestically produced Tc-99m radiopharmaceuticals to address this expected cost difference.
We understand commenters' concerns about the ability of the add-on payment to change the purchasing behavior of providers. However, the $10 add-on payment is intended to address the expected additional cost of domestically-produced Tc-99m radiopharmaceuticals if providers choose to utilize these radiopharmaceuticals in the future. If commenters have data showing that the price differential between domestically produced and foreign produced Mo-99 will be significantly more or less than $10, they may provide that data to CMS so we can consider it for CY 2026 OPPS rulemaking.
We appreciate commenters' suggestions for how to establish requirements to determine that a Tc-99m radiopharmaceutical is domestically produced. As we stated in the CY 2025 OPPS/ASC proposed rule (89 FR 59378), DOE/NNSA is establishing the criteria to certify whether the Tc-99m radiopharmaceutical dose is domestically produced and eligible for the add-on payment, and those criteria will be included in the CY 2026 OPPS/ASC proposed rule. We will consider in CY 2026 rulemaking any additional requirements for Medicare providers to document that the Tc-99m radiopharmaceutical used in a procedure was domestically produced and can qualify to receive the add-on payment based on the criteria developed by DOE/NNSA.
Comment: One commenter was opposed to the $10 add-on payment for domestically produced Tc-99m radiopharmaceuticals. According to the commenter, the central challenge for the proposal is that the Mo-99 used to create the Tc-99m generators will come from both domestic and foreign sources, and suppliers would need to assess each generator individually to determine its domestic Tc-99 content and whether the Tc-99 meets the standard to be considered domestically produced. The Tc-99m generator suppliers would need to communicate the domestic production status of each generator to the provider receiving the generator. The provider would then have to track the Tc-99m generators and link them to the Tc-99m radiopharmaceutical administered to a patient to be able to claim the add-on payment for domestically produced Tc-99m radiopharmaceuticals. The commenter believes most providers will not be able to track or document information on the production source of the Tc-99m generators they purchase.
The commenter also does not believe that if a domestic company starts to produce Mo-99, it will price it higher than Mo-99 from foreign sources because the domestic producer will need to charge the same price for Mo-99 to stay competitive with foreign producers. Per the commenter, this would mean when the Tc-99m radiopharmaceuticals are purchased by outpatient hospital providers at the point where the add-on payment may have a policy impact, there will be no price difference between the domestically produced product and the foreign produced product, and no purpose for the add-on payment. The commenter also expressed concern that the add-on payment policy will not help the pharmacies, Tc-99m generator manufacturers, or the Mo-99 producers who supply Tc-99m radiopharmaceuticals to outpatient hospitals, and those suppliers will end up paying for the higher cost of domestically produced Tc-99m, not hospitals. The commenter asked that we not finalize our proposal or at least wait until CY 2027 before implementing it. The commenter believes that it will take until at least 2027 before a domestic producer can start producing Mo-99 for Tc-99m generators.
Response: We appreciate the concerns of the commenter. As stated in the previous comment, DOE/NNSA will be establishing the criteria to certify whether the Tc-99m radiopharmaceutical dose is domestically produced and eligible for the add-on payment, and those criteria will be included in the CY 2026 OPPS/ASC proposed rule. Regarding the challenges providers may face identifying a Tc-99m radiopharmaceutical as a domestically produced product, once requirements are established defining a domestically produced Tc-99m radiopharmaceutical, we will consider in future rulemaking any requirements for providers to document that the Tc-99m radiopharmaceutical used in a procedure was domestically produced and can qualify to receive the add-on payment. We understand commenters' concerns regarding the cost of domestically-produced Tc-99m radiopharmaceuticals for entities other than outpatient hospital departments, but note that this policy is only intended to address the additional cost to hospital outpatient departments of utilizing domestically-produced Tc-99m radiopharmaceuticals.
The commenter noted that currently there are no producers of domestically sourced Mo-99 and Tc-99, and the earliest that domestic production of Mo-99 and Tc-99 could begin is in 2027. While there may not be domestic production of Mo-99 and Tc-99in CY 2026, we believe it is better to have a regulatory framework for this policy in place when domestic production of Tc-99m radiopharmaceuticals begins. With ( printed page 94259) a regulatory framework already in place, providers will be knowledgeable about the availability of additional payments for domestically sourced Tc-99m radiopharmaceuticals. Likewise, producers of domestic Mo-99m will have certainty that the Medicare OPPS payment policy takes into account the additional costs of domestic production of Mo-99.
After consideration of the public comments we received, we are finalizing our proposal without modification to establish a new add-on payment of $10 per dose for radiopharmaceuticals that use Tc-99m derived from domestically produced Mo-99 starting on January 1, 2026. As established in the CY 2024 OPPS/ASC final rule with comment period, CY 2025 will be the final year of the $10 add-on payment for TC-99m derived from non-HEU sources.
VI. Estimate of OPPS Transitional Pass-Through Spending for Drugs, Biologicals, Radiopharmaceuticals, and Devices
A. Amount of Additional Payment and Limit on Aggregate Annual Adjustment
Section 1833(t)(6)(E) of the Act limits the total projected amount of transitional pass-through payment for drugs, biologicals, and categories of devices for a given year to an “applicable percentage,” currently not to exceed 2.0 percent of total program payments estimated to be made for all covered services under the OPPS furnished for that year. If we estimate before the beginning of the calendar year that the total amount of pass-through payments in that year would exceed the applicable percentage, section 1833(t)(6)(E)(iii) of the Act requires a uniform prospective reduction in the amount of each of the transitional pass-through payments made in that year to ensure that the limit is not exceeded. We estimate the pass-through spending to determine whether payments exceed the applicable percentage and the appropriate pro rata reduction to the conversion factor for the projected level of pass-through spending in the following year to ensure that total estimated pass-through spending for the prospective payment year is budget neutral, as required by section 1833(t)(6)(E) of the Act.
For devices, developing a proposed estimate of pass-through spending in CY 2025 entails estimating spending for two groups of items. The first group of items consists of device categories that are currently eligible for pass-through payment and that will continue to be eligible for pass-through payment in CY 2025. The CY 2008 OPPS/ASC final rule with comment period (72 FR 66778) describes the methodology we have used in previous years to develop the pass-through spending estimate for known device categories continuing into the applicable update year. The second group of items consists of devices that we know are newly eligible, or project may be newly eligible, for device pass-through payment in the remaining quarters of CY 2024 or beginning in CY 2025. The sum of the proposed CY 2025 pass-through spending estimates for these two groups of device categories equals the proposed total CY 2025 pass-through spending estimate for device categories with pass-through payment status. We determined the device pass-through estimated payments for each device category based on the amount of payment as required by section 1833(t)(6)(D)(ii) of the Act, and as outlined in previous rules, including the CY 2014 OPPS/ASC final rule with comment period (78 FR 75034 through 75036). We note that, beginning in CY 2010, the pass-through evaluation process and pass-through payment methodology for implantable biologicals newly approved for pass-through payment beginning on or after January 1, 2010, that are surgically inserted or implanted (through a surgical incision or a natural orifice) use the device pass-through process and payment methodology (74 FR 60476). As has been our past practice (76 FR 74335), in the proposed rule, we proposed to include an estimate of any implantable biologicals eligible for pass-through payment in our estimate of pass-through spending for devices. Similarly, we finalized a policy in CY 2015 that applications for pass-through payment for skin substitutes and similar products be evaluated using the medical device pass-through process and payment methodology (76 FR 66885 through 66888). Therefore, as we did beginning in CY 2015, for CY 2025, we also proposed to include an estimate of any skin substitutes and similar products in our estimate of pass-through spending for devices.
For drugs and biologicals eligible for pass-through payment, section 1833(t)(6)(D)(i) of the Act establishes the pass-through payment amount as the amount by which the amount authorized under section 1842(o) of the Act (or, if the drug or biological is covered under a competitive acquisition contract under section 1847B of the Act, an amount determined by the Secretary equal to the average price for the drug or biological for all competitive acquisition areas and year established under such section as calculated and adjusted by the Secretary) exceeds the portion of the otherwise applicable fee schedule amount that the Secretary determines is associated with the drug or biological. Consistent with current policy, we proposed to apply a rate of ASP plus 6 percent to most drugs and biologicals for CY 2025, and therefore our estimate of drug and biological pass-through payment for CY 2025 for this group of items was $10.2 million.
Payment for certain drugs,[146] specifically contrast agents without pass-through payment status, is packaged into payment for the associated procedures, and these products are not separately paid. In addition, we policy-package non-pass-through drugs and biologicals that function as supplies when used in a diagnostic test or procedure unless a high-cost diagnostic radiopharmaceutical with a per day cost greater than $630 is used for the test or procedure. We policy-package all drugs and biologicals that function as supplies when used in a surgical procedure or for anesthesia, and other categories of drugs and biologicals, as discussed in section V.B.1.c of the CY 2025 OPPS/ASC proposed rule (89 FR 59364). Consistent with current policy, we propose that all of these policy-packaged drugs and biologicals with pass-through payment status will be paid at ASP+6 percent, like other pass-through drugs and biologicals, for CY 2025, less the policy-packaged drug APC offset amount described below. Our estimate of pass-through payment for policy-packaged drugs and biologicals with pass-through payment status approved prior to CY 2025 is not $0. This is because the pass-through payment amount and the fee schedule amount associated with the drug or biological will not be the same, unlike for separately payable drugs and biologicals. In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81774 through 81776), we discussed our policy to determine if the costs of certain policy-packaged drugs or biologicals are already packaged into the existing APC structure. If we determine that a policy-packaged drug or biological approved for pass-through payment resembles predecessor drugs or biologicals already included in the costs of the APCs that are associated with the drug receiving pass-through payment, we proposed to offset the amount of pass-through payment for the policy-packaged drug or biological. For these ( printed page 94260) drugs or biologicals, the APC offset amount is the portion of the APC payment for the specific procedure performed with the pass-through drug or biological, which we refer to as the policy-packaged drug APC offset amount. Consistent with current policy, if we determine that an offset is appropriate for a specific policy-packaged drug or biological receiving pass-through payment, we proposed to reduce our estimate of pass-through payments for these drugs or biologicals by the APC offset amount.
Similar to pass-through spending estimates for devices, the first group of drugs and biologicals requiring a pass-through payment estimate consists of those products that were recently made eligible for pass-through payment and that will continue to be eligible for pass-through payment in CY 2025. The second group contains drugs and biologicals that we know are newly eligible, or project will be newly eligible, in CY 2025. The sum of the CY 2025 pass-through spending estimates for these two groups of drugs and biologicals equals the total CY 2025 pass-through spending estimate for drugs and biologicals with pass-through payment status.
B. Final Estimate of Pass-Through Spending for CY 2025
For CY 2025, we proposed to set the applicable pass-through payment percentage limit at 2.0 percent of the total projected OPPS payments for CY 2025, consistent with section 1833(t)(6)(E)(ii)(II) of the Act and our OPPS policy from CY 2004 through CY 2024 (88 FR 81805). The pass-through payment percentage limit is calculated using pass-through spending estimates for devices and for drugs and biologicals.
For the first group of devices, consisting of device categories that are currently eligible for pass-through payment and will continue to be eligible for pass-through payment in CY 2025, there are 8 active categories for CY 2025. The active categories are described by HCPCS codes C1747, C1826, C1827, C1600, C1601, C1602, C1603 and C1604. Based on the information from the device manufacturers, we estimated that HCPCS code C1747 will cost $19.5 million in pass-through expenditures in CY 2025, HCPCS code C1826 will cost $151,991 in pass-through expenditures in CY 2025, HCPCS code C1827 will cost $364,793 in pass-through expenditures in CY 2025, HCPCS code C1600 will cost $21.9 million in pass-through expenditures in CY 2025, HCPCS code C1601 will cost $14.4 million in pass-through expenditures in CY 2025, HCPCS code C1602 will cost $8.2 million in pass-through expenditures in CY 2025, HCPCS code C1603 will cost $6.6 million in pass-through expenditures in CY 2025, and HCPCS code C1604 will cost $20.0 million in pass-through expenditures in CY 2025. Therefore, we proposed an estimate for the first group of devices of $91.1 million.
We did not receive any public comments on our proposed estimate. Using our methodology for this final rule with comment period, the final estimated pass through costs based on updated data for HCPCS code C1747 will cost $18.9 million in pass-through expenditures in CY 2025, HCPCS code C1826 will cost $176,594 in pass-through expenditures in CY 2025, HCPCS code C1827 will cost $844,031 in pass-through expenditures in CY 2025, HCPCS code C1600 will cost $21.9 million in pass-through expenditures in CY 2025, HCPCS code C1601 will cost $14.4 million in pass-through expenditures in CY 2025, HCPCS code C1602 will cost $8.2 million in pass-through expenditures in CY 2025, HCPCS code C1603 will cost $6.6 million in pass-through expenditures in CY 2025, and HCPCS code C1604 will cost $20.0 million in pass-through expenditures in CY 2025. Therefore, we have finalized the CY 2025 spending estimate for the first group of devices of approximately $91.0 million.
In estimating our proposed CY 2025 pass-through spending for device categories in the second group, we included the following: (1) device categories that we assumed at the time of the development of the proposed rule would be newly eligible for pass-through payment in CY 2025; (2) additional device categories that we estimated could be approved for pass-through status after the development of the CY 2025 OPPS/ASC proposed rule (89 FR 59379) and before January 1, 2025; and (3) contingent projections for new device categories established in the second through fourth quarters of CY 2025. For CY 2025, we proposed to use the general methodology described in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66778), while also taking into account recent OPPS experience in approving new pass-through device categories. For the CY 2025 OPPS/ASC proposed rule (89 FR 59379), the proposed estimate of CY 2025 pass-through spending for this second group of device categories was $523.7 million.
We did not receive any public comments on the proposed estimate. As stated earlier in this final rule with comment period, we are approving eight devices for pass-through payment status in the CY 2025 rulemaking cycle: AGENTTM Paclitaxel-Coated Balloon Catheter, AVEIRTM DR Dual Chamber Leadless Pacemaker System (AveirTM DR System), DETOURTM System, the Ultrasound Disposable Kit—Diagnostic/Therapeutic (UDK-T) component of the EndoSound Vision SystemTM (EVSTM), iFuse Bedrock GraniteTM Implant System, Paradise® Ultrasound Renal Denervation (RDN) System, Precision GI and Symplicity SpyralTM Renal Denervation (RDN) System.[147] The manufacturers of these systems provided utilization and cost data that indicate the amount of spending for the devices would be approximately $36.0 million for AGENTTM Paclitaxel-Coated Balloon Catheter; $113.0 million for AVEIRTM DR Dual Chamber Leadless Pacemaker System; $280,000 for the UDK-T component of the EVSTM; $25.3 million for iFuse Bedrock GraniteTM Implant System; $32.8 million for Paradise® Ultrasound RDN System; $0.78 million for Precision GI; and $16.0 million for Symplicity SpyralTM RDN System. In addition, we note that HCPCS code C8000 was preliminarily approved as part of the device pass-through quarterly review process with an effective date of October 1, 2024.[148] For this final rule based on updated data, we estimate that HCPCS code C8000 will cost $2.9 million in pass-through expenditures in CY 2025. Therefore, we are finalizing an estimate of $227.1 million for this second group of devices for CY 2025.
The estimated amount of pass-through spending of $227.1 million in this final rule is substantially less that the estimated amount of pass-through spending in the CY 2025 OPPS/ASC proposed rule (89 FR 59379) of $523.7 ( printed page 94261) million because, in the proposed rule we assumed that all of the devices with pending applications to receive pass-through status would receive approval. For the final rule, we have determined the some of these devices have received pass-through status, and some of these devices have been denied pass-through status, and we adjusted our device pass-through spending estimates accordingly.
To estimate proposed CY 2025 pass-through spending for drugs and biologicals in the first group, specifically those drugs and biologicals recently made eligible for pass-through payment and continuing on pass-through payment status for at least one quarter in CY 2025, we proposed to use the CY 2023 Medicare hospital outpatient claims data regarding their utilization, information provided in their respective pass-through applications, other historical hospital claims data, pharmaceutical industry information, and clinical information regarding these drugs and biologicals to project the CY 2025 OPPS utilization of the products.
For the known drugs and biologicals (excluding policy-packaged contrast agents, drugs, biologicals, radiopharmaceuticals with per-day costs at or below the packaging threshold that function as supplies when used in a diagnostic test or procedure, and drugs and biologicals that function as supplies when used in a surgical procedure) that will be continuing on pass-through payment status in CY 2025, we estimated the pass-through payment amount as the difference between the general payment rate of ASP+6 percent and the payment rate for non-pass-through drugs and biologicals that would be separately paid. Because we proposed to utilize a payment rate of ASP plus 6 percent for most separately payable drugs and biologicals in the proposed rule, the proposed payment rate difference between the pass-through payment amount and the non-pass-through payment amount was $0 for this group of drugs.
Because payment for policy-packaged drugs and biologicals is packaged if the product is not paid separately due to its pass-through payment status, we propose to include in the CY 2025 pass-through estimate the difference between payment for the policy-packaged drug or biological at ASP+6 percent (or WAC+6 percent, or 95 percent of AWP, if ASP or WAC information is not available) and the policy-packaged drug APC offset amount, if we determine that the policy-packaged drug or biological approved for pass-through payment resembles a predecessor drug or biological already included in the costs of the APCs that are associated with the drug receiving pass-through payment. Diagnostic radiopharmaceuticals that currently have pass-through status, but would be paid separately because of the policy we are adopting described in Section II.A.3.c. to separately pay for diagnostic radiopharmaceuticals with a per day cost over $630, are not considered to be policy-packaged and are not included in this group. For this first group of policy-packaged drugs and biologicals, we estimated a pass-through spending for CY 2025 of $200,000.
We did not receive any public comments on our proposed estimate. Using our methodology for this final rule with comment period, the CY 2025 spending estimate for this first group of drugs and biologicals is around $200,000.
To estimate proposed CY 2025 pass-through spending for drugs and biologicals in the second group (that is, drugs and biologicals that we knew at the time of development of the proposed rule were newly eligible or recently became eligible for pass-through payment in CY 2024, additional drugs and biologicals that we estimated could be approved for pass-through status subsequent to the development of the proposed rule and before January 1, 2025, and projections for new drugs and biologicals that could be initially eligible for pass-through payment in the second through fourth quarters of CY 2025), we proposed to use utilization estimates from pass-through applicants, pharmaceutical industry data, clinical information, recent trends in the per unit ASPs of hospital outpatient drugs, and projected annual changes in service volume and intensity as our basis for making the CY 2025 pass-through payment estimate. We also proposed to consider the most recent OPPS experience in approving new pass-through drugs and biologicals. Using our proposed methodology for estimating CY 2025 pass-through payments for this second group of drugs, we calculated a proposed spending estimate for this second group of drugs and biologicals of approximately $10 million.
We did not receive any public comments on our proposed estimate. Using our methodology for this final rule with comment period, we are finalizing our estimate of pass-through spending for the second group of drugs and biologicals to be $10 million.
We estimated for the CY 2025 OPPS/ASC proposed rule (89 FR 59380) that the amount of pass-through spending for the device categories and the drugs and biologicals that are continuing to receive pass-through payment in CY 2025 and the amount of pass-through spending for those device categories, drugs, and biologicals that first become eligible for pass-through payment during CY 2025 would be approximately $625 million (approximately $614.8 million for device categories and approximately $10.2 million for drugs and biologicals), which represented 0.71 percent of total projected OPPS payments for CY 2025 (approximately $88.2 billion). Therefore, we estimated for the proposed rule that pass-through spending in CY 2025 would not amount to 2.0 percent of total projected OPPS CY 2025 program spending.
We estimate for this final rule with comment period that the amount of pass-through spending for the device categories and the drugs and biologicals that are continuing to receive pass-through payment in CY 2025 and the amount of pass-through spending for those device categories, drugs, and biologicals that first become eligible for pass-through payment during CY 2025 would be approximately $328.3 million (approximately $318.1 million for device categories and approximately $10.2 million for drugs and biologicals), which represents only 0.37 percent of total projected OPPS payments for CY 2025 (approximately $87.7 billion). Therefore, we estimate that pass-through spending in CY 2025 will not exceed the 2.0 percent of total projected OPPS CY 2025 program spending limit provided for in section 1833(t)(6)(E) of the Act.
VII. OPPS Payment for Hospital Outpatient Visits and Critical Care Services
For CY 2025, we proposed to continue our current clinic and emergency department (ED) hospital outpatient visits payment policies. For a description of these policies, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70448). We also proposed to continue our payment policy for critical care services for CY 2025. For a description of this policy, we refer readers to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70449), and for the history of this payment policy, we refer readers to the CY 2014 OPPS/ASC final rule with comment period (78 FR 75043).
As we stated in the CY 2022 OPPS/ASC final rule with comment period (86 FR 63663), the volume control method for clinic visits furnished by non-excepted off-campus provider-based departments (PBDs) applies for CY 2022 and subsequent years. More specifically, we finalized a policy to continue to utilize a PFS-equivalent payment rate ( printed page 94262) for the hospital outpatient clinic visit service described by HCPCS code G0463 when it is furnished by these departments for CY 2022 and subsequent years. The PFS-equivalent rate for CY 2025 is 40 percent of the proposed OPPS payment. Under this policy, these departments will be paid approximately 40 percent of the OPPS rate for the clinic visit service in CY 2025.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71748), we finalized a policy that excepted off-campus provider-based departments (PBDs) (departments that bill the modifier “PO” on claim lines) of rural Sole Community Hospitals (SCHs), as described under 42 CFR 412.92 and designated as rural for Medicare payment purposes, are exempt from the clinic visit payment policy that applies a PFS-equivalent payment rate for the clinic visit service, as described by HCPCS code G0463, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act. For the full discussion of this policy, we refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 72047 through 72051). For CY 2025, we proposed to continue to exempt excepted off-campus PBDs of rural SCHs from the clinic visit payment policy. We stated that we would continue to monitor the effect of this change in Medicare payment policy, including on the volume of these types of OPD services.
Comment: We received several comments on our overall clinic visit payment policy. Many commenters continued to express the belief that this policy undermines Congressional intent and exceeds the agency's legal authority. As they have in previous years, commenters stated that the policy is based on flawed assumptions and urged CMS to eliminate it altogether. One commenter suggested that we lack the statutory authority to implement this policy in a non-budget neutral manner. While acknowledging that American Hospital Association v. Azar, 964 F.3d 1230 (D.C. Cir. 2020), affirmed our authority to implement our policy under section 1833(t)(2)(F), the commenter stated that it has been overtaken by Loper Bright v. Raimondo, 144 S. Ct. 2244 (2024), and so should be disregarded. They requested that CMS reverse this payment adjustment or, at a minimum, budget neutralize its application consistent with statute.
Response: We continue to believe that section 1833(t)(2)(F) of the Act gives the Secretary authority to develop a method for controlling unnecessary increases in the volume of covered OPD services, including a method that controls unnecessary volume increases by removing a payment differential that is driving a site-of-service decision, and as a result, is unnecessarily increasing service volume.
As we noted in the CY 2019 OPPS/ASC proposed rule (83 FR 37138 through 37143), “[a] large source of growth in spending on services furnished in hospital outpatient departments (HOPDs) appears to be the result of the shift of services from (lower cost) physician offices to (higher cost) HOPDs.” We continue to believe that these shifts in the sites of service are unnecessary if the beneficiary can safely receive the same services in a lower cost setting but instead receives care in a higher cost setting due to payment incentives. In most cases, the difference in payment is leading to unnecessary increases in the volume of covered outpatient department services, and we remain concerned that this shift in care setting increases beneficiary cost-sharing liability because Medicare payment rates for the same or similar services are generally higher in hospital outpatient departments than in physician offices. We continue to believe that our method addresses the concerns described in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59005).
As we stated in the CY 2019 OPPS ASC final rule with comment period (83 FR 59013), we believe implementing a volume control method in a budget neutral manner would not appropriately reduce the overall unnecessary volume of covered OPD services, and instead would simply shift services within the OPPS system because of payment rather than medical necessity. We also outlined in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59013) that while section 1833(t)(9)(B) of the Act requires that certain changes made under the OPPS be made in a budget neutral manner, this section does not apply to the volume control method under section 1833(t)(2)(F) of the Act.
As noted, the D.C. Circuit previously held that our regulation was a reasonable interpretation of the statutory authority to adopt a method to control for unnecessary increases in the volume of the relevant service. AHA v. Azar, 964 F.3d at 1241-45, Loper Bright does not change the result. As an initial matter, while Loper Bright changed certain aspects of the interpretative framework the D.C. Circuit used, the Supreme Court cautioned that the “holdings of those cases that specific agency actions are lawful . . . are still subject to statutory stare decisis despite our change in interpretive methodology.” 144 S. Ct. at 2273. AHA' s holding is therefore still good law. And even under the new Loper Bright framework, the Supreme Court clarified that Congress “often” enacts statutes that “delegate[] discretionary authority to an agency.” Id. at 2263. Section 1833(t)(2)(F) is just such a statute. By instructing the Secretary to “develop a method for controlling unnecessary increases in the volume of covered OPD services” without specifying the method, the statute delegates discretionary authority to the Secretary. Because, as the D.C. Circuit explained, our policy “falls comfortably within the plain text of subparagraph (2)(F)” and is further supported by its structure, 964 F.3d at 1241, it does not fall outside the “outer statutory bounds” of the delegation, as discussed in Loper Bright. That aspect of the D.C. Circuit's holding, coupled with the considerations of stare decisis, indicate that there is no reason to doubt the continued validity of our policy. 144 S. Ct. at 2268.
Comment: We received comments supporting CMS's efforts to continue implementing its method to control for unnecessary increases in the volume of outpatient services. These commenters asked that CMS continue to consider ways to expand the current site-neutral payment policies to other services and settings. Commenters reiterated their statements from previous years suggesting that CMS apply the site-neutral payment policy to a list of 57 APCs for which MedPAC determined it would be reasonable and appropriate to align the OPPS and ASC payment rates with those set in the physician fee schedule (PFS). Other commenters recommended that CMS consider expanding the site-neutral payment policy to all services provided by excepted, off-campus PBDs. Others suggested that the site-neutral policy be extended to on-campus PBDs, ASCs, and emergency departments.
Response: We appreciate the commenters' support and we will continue to monitor this policy and take commenters' suggestions into consideration for potential future rulemaking.
After consideration of the public comments, we are continuing the volume control method under which we utilize a PFS-equivalent payment rate for the hospital outpatient clinic visit service described by HCPCS code G0463 when it is furnished by excepted off-campus PBDs in CY 2025.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71748), we finalized a policy that excepted off-campus provider-based departments (PBDs) (departments that bill the ( printed page 94263) modifier “PO” on claim lines) of rural Sole Community Hospitals (SCHs), as described under 42 CFR 412.92 and designated as rural for Medicare payment purposes, are exempt from the clinic visit payment policy that applies a Physician Fee Schedule-equivalent payment rate for the clinic visit service, as described by HCPCS code G0463, when provided at an off-campus PBD excepted from section 1833(t)(21) of the Act. For the full discussion of this policy, we refer readers to the CY 2023 OPPS/ASC final rule with comment period (87 FR 72047 through 72051). For CY 2025, we proposed to continue to exempt excepted off-campus PBDs of rural SCHs from the clinic visit payment policy. We stated that we will continue to monitor the effect of this change in Medicare payment policy, including on the volume of these types of OPD services.
The following is a summary of the comments we received and our responses to those comments.
Comment: Commenters continued to support our proposal to exempt excepted off-campus PBDs of rural SCHs from the clinic visit payment policy for CY 2025. Commenters stated that the continuation of the exemption is an important step in maintaining access to care for a segment of the population that is underserved.
Response: We thank the commenters for their support.
Comment: Several commenters reiterated their requests from previous proposed rules that CMS consider expanding the exemption to excepted off-campus PBDs of rural hospitals with fewer than 100 beds, Medicare Dependent Hospitals (MDHs), Urban SCHs, and low-volume hospitals in future rulemaking, arguing that the same reasoning that led CMS to propose to exempt SCHs also applies to these hospitals.
Response: In the CY 2006 OPPS final rule with comment period (70 FR 68556 through 68561) we uniquely identified rural SCHs as providers with demonstrated additional resource costs. We found that rural SCHs have significantly higher costs per unit than urban hospitals. We have continued to adjust payments for rural SCHs by 7.1 percent each year since 2006. Building upon that foundation, for CY 2018 we finalized a policy to exclude rural SCHs from our 340B drug payment policy and continued to do so until September 27, 2023, when the 340B drug payment policy ended and we resumed paying for 340B drugs and biologicals under the OPPS at the same rates we pay for non-340B drugs and biologicals (generally, ASP plus 6 percent)). We believe exempting rural SCHs, which have demonstrated additional resource costs, is appropriate to ensure these hospitals can remain open to serve the beneficiaries who rely on them for their care. We share commenters' concerns about the financial difficulties associated with maintaining access to care in medically vulnerable communities. However, in each of these cases, the Congress did not determine that any of these hospital types required additional payments for outpatient services. Section 1833(t)(13)(B) of the Act authorizes an appropriate adjustment for hospitals located in rural areas where the Secretary determines, based on a study, that the costs incurred by these hospitals by APC group exceed costs incurred by hospitals in urban areas. In the CY 2006 OPPS final rule with comment period (70 FR 68556 through 68561), we summarized our study of the cost of covered outpatient department services to hospitals in rural areas and found that rural SCHs were the only rural hospital type that had higher resource costs for covered outpatient department services. Rural SCHs demonstrated significantly higher cost per unit than urban hospitals after controlling for labor input prices, service-mix complexity, volume, facility size, and type of hospital. In the CY 2006 OPPS final rule with comment period (70 FR 68556 through 68561) we stated that we found no significant difference in cost between all small rural hospitals with 100 or fewer beds and urban hospitals. We found that there was insufficient evidence to conclude that rural hospitals with 100 or fewer beds have higher costs than urban hospitals. We proposed a narrow exception to our clinic visit policy largely based upon the historical treatment and documented additional resource costs of rural SCHs under the OPPS. We are only accepting rural SCHs because we continue to believe that the underlying principles of the clinic visit policy continue to justify application of the volume control method for clinic visits to the remaining hospital types, including most rural and safety-net providers. Where the difference in payment is leading to unnecessary increases in the volume of covered outpatient department services, we remain concerned that this shift in care setting increases beneficiary cost-sharing liability because Medicare payment rates for the same or similar services are generally higher in hospital outpatient departments than in physician offices. Further, we do not believe that commenters provided sufficient reasoning or data to show that the other provider types suggested (Medicare Dependent Hospitals, Urban SCHs, and low-volume hospitals) demonstrate the additional resource costs that rural SCHs do and should therefore also be exempted from this OPPS payment policy. We continue to share commenters' concerns about maintaining access to care in urban and rural settings and enhancing access to care in medically vulnerable communities. We also share commenters' concerns about profit margins. However, we must balance the concerns of providers with the concerns of beneficiaries regarding the affordability of their care. For hospitals subject to the clinic visit policy, the proposed PFS-equivalent rate for a clinic visit brings the approximate average copayment down from $26 to $10. We will continue to study access and cost to see if further exemptions to the clinic visit policy are appropriate.
Comment: One commenter noted that, while it is necessary to distinguish between urban and rural hospitals for a number of payment and policy mechanisms, they believe the Metropolitan Statistical Areas (MSAs) CMS uses to delineate between these areas are not the most precise tool. This commenter argued that CMS should extend this exemption to urban SCHs because using MSAs to determine urban and rural areas is imprecise and unfairly disadvantages urban SCHs that may be the sole source of hospital services in their communities.
Response: We acknowledge the commenters' points about the important role that urban SCHs serve in their communities. However, we have not found that urban SCHs have the additional resource costs for covered outpatient department services that rural SCHs have, and as such, we are only applying the clinic visit policy exemption to rural SCHs.
After consideration of the public comments we received, we are finalizing our proposal to continue to exempt excepted off-campus PBDs of rural SCHs from the clinic visit payment policy in CY 2025.
VIII. Payment for Partial Hospitalization and Intensive Outpatient Services
This section discusses payment for partial hospitalization services as well as intensive outpatient services. Since CY 2000, Medicare has paid for partial hospitalization services under the OPPS. Beginning in CY 2024, as authorized by section 4124 of the Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), Medicare began paying for intensive ( printed page 94264) outpatient services furnished by hospital outpatient departments, community mental health centers, federally qualified health centers, and rural health clinics in addition to opioid treatment programs. Additional background on the partial hospitalization and intensive outpatient benefits is included in the following paragraphs.
A. Background
1. Partial Hospitalization
A partial hospitalization program (PHP) is an intensive outpatient program of psychiatric services provided as an alternative to inpatient psychiatric care for individuals who have an acute mental illness, which includes, but is not limited to, conditions such as depression, schizophrenia, and substance use disorders (SUD). Section 1861(ff)(1) of the Act defines partial hospitalization services as the items and services described in paragraph (2) prescribed by a physician and provided under a program described in paragraph (3) under the supervision of a physician pursuant to an individualized, written plan of treatment established and periodically reviewed by a physician (in consultation with appropriate staff participating in such program), which sets forth the physician's diagnosis, the type, amount, frequency, and duration of the items and services provided under the plan, and the goals for treatment under the plan. Section 1861(ff)(2) of the Act describes the items and services included in partial hospitalization services. Section 1861(ff)(3)(A) of the Act specifies that a PHP is a program furnished by a hospital to its outpatients or by a community mental health center (CMHC), as a distinct and organized intensive ambulatory treatment service, offering less than 24-hour-daily care, in a location other than an individual's home or inpatient or residential setting. Section 1861(ff)(3)(B) of the Act defines a CMHC for purposes of this benefit. We refer readers to sections 1833(t)(1)(B)(i), 1833(t)(2)(B), 1833(t)(2)(C), and 1833(t)(9)(A) of the Act and 42 CFR 419.21, for additional information regarding PHP.
PHP policies and payment have been addressed under OPPS since CY 2000. In CY 2008, we began efforts to strengthen the PHP benefit through extensive data analysis, along with policy and payment changes, by implementing two refinements to the methodology for computing the PHP median. For a detailed discussion on these policies, we refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66670 through 66676). In CY 2009, we implemented several regulatory, policy, and payment changes. For a detailed discussion on these policies, we refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68688 through 68697). In CY 2010, we retained the two-tier payment approach for partial hospitalization services and used only hospital-based PHP data in computing the PHP APC per diem costs, upon which PHP APC per diem payment rates are based (74 FR 60556 through 60559). In CY 2011 (75 FR 71994), we established four separate PHP APC per diem payment rates: two for CMHCs (APC 0172 and APC 0173) and two for hospital-based PHPs (APC 0175 and APC 0176) and instituted a 2-year transition period for CMHCs to the CMHC APC per diem payment rates. For a detailed discussion, we refer readers to section X.B of the CY 2011 OPPS/ASC final rule with comment period (75 FR 71991 through 71994). In CY 2012, we determined the relative payment weights for partial hospitalization services provided by CMHCs based on data derived solely from CMHCs and the relative payment weights for partial hospitalization services provided by hospital-based PHPs based exclusively on hospital data (76 FR 74348 through 74352). In the CY 2013 OPPS/ASC final rule with comment period, we finalized our proposal to base the relative payment weights that underpin the OPPS APCs, including the four PHP APCs (APCs 0172, 0173, 0175, and 0176), on geometric mean costs rather than on the median costs. For a detailed discussion on this policy, we refer readers to the CY 2013 OPPS/ASC final rule with comment period (77 FR 68406 through 68412).
In the CY 2014 OPPS/ASC proposed rule (78 FR 43621 through 43622) and CY 2015 OPPS/ASC final rule with comment period (79 FR 66902 through 66908), we continued to apply our established policies to calculate the four PHP APC per diem payment rates based on geometric mean per diem costs using the most recent claims data for each provider type. For a detailed discussion on this policy, we refer readers to the CY 2014 OPPS/ASC final rule with comment period (78 FR 75047 through 75050). In the CY 2016 OPPS/ASC final rule with comment period (80 FR 70453 through 70467), we described our extensive analysis of the claims and cost data and ratesetting methodology, corrected a cost inversion that occurred in the final rule data with respect to hospital-based PHP providers, and renumbered the PHP APCs. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79687 through 79691), we continued to apply our established policies to calculate the PHP APC per diem payment rates based on geometric mean per diem costs and finalized a policy to combine the Level 1 and Level 2 PHP APCs for CMHCs and for hospital-based PHPs. We also implemented an eight-percent outlier cap for CMHCs to mitigate potential outlier billing vulnerabilities. For a comprehensive description of PHP payment policy, including a detailed methodology for determining PHP per diem amounts, we refer readers to the CY 2016 and CY 2017 OPPS/ASC final rules with comment period (80 FR 70453 through 70455 and 81 FR 79678 through 79680, respectively).
In the CYs 2018 and 2019 OPPS/ASC final rules with comment period (82 FR 59373 through 59381 and 83 FR 58983 through 58998, respectively), we continued to apply our established policies to calculate the PHP APC per diem payment rates based on geometric mean per diem costs, designated a portion of the estimated 1.0 percent hospital outpatient outlier threshold specifically for CMHCs, and proposed updates to the PHP allowable HCPCS codes. We finalized these proposals in the CY 2020 OPPS/ASC final rule with comment period (84 FR 61352).
In the CY 2020 OPPS/ASC final rule with comment period (84 FR 61339 through 61350), we finalized a proposal to use the calculated CY 2020 CMHC geometric mean per diem cost and the calculated CY 2020 hospital-based PHP geometric mean per diem cost, but with a cost floor equal to the CY 2019 final geometric mean per diem costs as the basis for developing the CY 2020 PHP APC per diem rates. Also, we continued to designate a portion of the estimated 1.0 percent hospital outpatient outlier threshold specifically for CMHCs, consistent with the percentage of projected payments to CMHCs under the OPPS, excluding outlier payments.
In the April 30, 2020 interim final rule with comment (85 FR 27562 through 27566), effective as of March 1, 2020 and for the duration of the COVID-19 Public Health Emergency (PHE), hospital and CMHC staff were permitted to furnish certain outpatient therapy, counseling, and educational services (including certain PHP services), incident to a physician's services, to beneficiaries in temporary expansion locations, including the beneficiary's home, as long as the location met all conditions of participation to the extent not waived. A hospital or CMHC could furnish such services using ( printed page 94265) telecommunications technology to a beneficiary in a temporary expansion location if that beneficiary was registered as an outpatient. In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72247), we confirmed that these provisions applied only for the duration of the COVID-19 PHE. On May 11, 2023, the COVID-19 PHE ended, and accordingly, these flexibilities ended as well.
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 86073 through 86080), we continued our current methodology to utilize cost floors, as needed. Since the final calculated geometric mean per diem costs for both CMHCs and hospital-based PHPs were significantly higher than each proposed cost floor, a floor was not necessary at the time, and we did not finalize the proposed cost floors in the CY 2021 OPPS/ASC final rule with comment period.
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63665 through 63666), we explained that we observed a number of changes, likely as a result of the COVID-19 PHE, in the CY 2020 OPPS claims that we would have ordinarily used for CY 2022 ratesetting, and this included changes in the claims for partial hospitalization. We explained that significant decreases in utilization and in the number of hospital-based PHP providers who submitted CY 2020 claims led us to believe that CY 2020 data were not the best overall approximation of expected PHP services in CY 2022. Therefore, we finalized our proposal to calculate the PHP per diem costs using the year of claims consistent with the calculations that would be used for other OPPS services, by using the CY 2019 claims and the cost reports that were used for CY 2021 final rulemaking to calculate the CY 2022 PHP per diem costs. In addition, for CY 2022 and subsequent years, we finalized our proposal to use cost and charge data from the Hospital Cost Report Information System (HCRIS) as the source for the CMHC cost-to-charge ratios (CCRs), instead of using the Outpatient Provider Specific File (OPSF) (86 FR 63666).
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 71995), we explained that we continued to observe a decrease in the number of hospital-based and CMHC PHP days in our trimmed dataset due to the continued effects of COVID-19; however, the Medicare outpatient service volumes appeared to be returning to more normal, pre-pandemic levels. Therefore, we finalized our proposal to use the latest available CY 2021 claims but use the cost information from prior to the COVID-19 PHE for calculating the CY 2023 CMHC and hospital-based PHP APC per diem costs. The application of the OPPS standard methodology, including the effect of budget neutralizing all other OPPS policy changes unique to CY 2023, resulted in the final calculated CMHC PHP APC payment rate being unexpectedly lower than the CY 2022 final CMHC PHP APC rate. Therefore, in the interest of accurately paying for CMHC PHP services, under the unique circumstances of budget neutralizing all other OPPS policy changes for CY 2023, and in keeping with our longstanding goal of protecting continued access to PHP services provided by CMHCs by ensuring that CMHCs remain a viable option as providers of mental health care in the beneficiary's own community, we finalized utilizing the equitable adjustment authority of section 1833(t)(2)(E) of the Act to appropriately pay for CMHC PHP services at the same payment rate as for CY 2022, that is, $142.70. In addition, we clarified the payment under the OPPS for new HCPCS codes that designate non-PHP services provided for the purposes of diagnosis, evaluation, or treatment of a mental health disorder and are furnished to beneficiaries in their homes by clinical staff of the hospital would not be recognized as PHP services; however, none of the PHP regulations would preclude a patient that is under a PHP plan of care from receiving other reasonable and medically necessary non-PHP services from a hospital (87 FR 72001 and 72002).
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81811), we revised the regulation at § 424.24(e)(1)(i) to require the physician certification for PHP services to include a certification that the patient requires such services for a minimum of 20 hours per week, as required by section 1861(ff)(1) of the Act, as amended by section 4124(a) of Division FF of the CAA, 2023. In addition, we modified the regulations for PHP at § 410.43 to include references to SUD. In the same CY 2024 OPPS/ASC final rule with comment period, we also established separate payment rates for PHP days with 3 services and days with 4 or more services. Accordingly, we established four separate PHP APC per diem payment rates: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5853 and APC 5854, respectively), and one for hospital-based PHPs for 3-service days and another for hospital-based PHPs for 4-service days (APC 5863 and APC 5864, respectively). We also finalized a policy to utilize the separate CMHC rates for 3-service and 4-service PHP days as the Medicare Physician Fee Schedule (MPFS) rates, depending upon whether a nonexcepted off-campus hospital outpatient department furnishes 3 or 4 PHP services in a day. Lastly, we finalized several changes beginning in CY 2024 to align coding, billing, and payment between PHPs and intensive outpatient programs.
2. Intensive Outpatient Program Services
Section 4124(b) of the CAA, 2023 established Medicare coverage for intensive outpatient services effective for items and services furnished on or after January 1, 2024. An intensive outpatient program (IOP) is a distinct and organized program of psychiatric services for individuals who have an acute mental illness, which includes, but is not limited to, conditions such as depression, schizophrenia, and SUD. Intensive outpatient services are not required to be provided in lieu of inpatient hospitalization. Section 1861(ff)(4) of the Act defines intensive outpatient services as the items and services described in paragraph (2) prescribed by a physician for an individual determined (not less frequently than every other month) by a physician to have a need for such services for a minimum of 9 hours per week and provided under a program described in paragraph (3) under the supervision of a physician pursuant to an individualized, written plan of treatment established and periodically reviewed by a physician (in consultation with appropriate staff participating in such program), which plan sets forth the physician's diagnosis, the type, amount, frequency, and duration of the items and services provided under the plan, and the goals for treatment under the plan. Section 1861(ff)(2) of the Act describes the items and services included in intensive outpatient services. Section 1861(ff)(4)(C) of the Act specifies that an IOP is a program furnished by a hospital to its outpatients, a CMHC, a Federally qualified health center (FQHC), or by a rural health clinic (RHC) as a distinct and organized intensive ambulatory treatment service, offering less than 24-hour-daily care, in a location other than an individual's home or inpatient or residential setting. Section 1861(ff)(3)(B) of the Act defines a CMHC for purposes of this benefit. We refer readers to sections 1833(t)(1)(B)(i), 1833(t)(2)(B), 1833(t)(2)(C), and 1833(t)(9)(A) of the Act and 42 CFR 419.21, for additional information regarding IOP. ( printed page 94266)
In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81812 through 81857), we established payment and program requirements for the IOP benefit furnished by a hospital to its outpatients, or by a CMHC, an FQHC, or an RHC. In addition, we established Medicare Part B coverage for IOP services provided by Opioid Treatment Programs (OTPs) for the treatment of opioid use disorder (OUD).
Consistent with the statutory definition of intensive outpatient services under section 1861(ff)(2) of the Act, we finalized regulations at 42 CFR 410.44 to set forth the conditions and exclusions applicable for intensive outpatient services, and at § 424.24 to set forth the content of the certification and plan of treatment requirements for intensive outpatient services. We also revised certain existing regulations at §§ 410.2, 410.3, 410.10, 410.27, 410.150, and 419.21 to add a regulatory definition of intensive outpatient services and to include intensive outpatient services in the regulations for medical and other health services paid for under Medicare Part B, and in the case of § 419.21, under the OPPS. Additionally, we created regulations at § 410.111 to establish the requirements for coverage of IOP services furnished in CMHCs, and at § 410.173 to establish conditions of payment for IOP services furnished in CMHCs. Lastly, we revised § 410.155 to exclude IOP services from the outpatient mental health treatment limitation, consistent with the statutory requirement of section 1833(c)(2) of the Act, as amended by section 4124(b)(3) of the CAA, 2023.
In addition, as discussed in greater detail in the following sections, we established coding, billing, and payment policies for IOP that align with the policies established for PHP provided in the same settings. Specifically, we established four separate IOP APC per diem payment rates at the same rates we proposed for the PHP APCs: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5851 and APC 5852, respectively), and one for hospital-based IOPs for 3-service days and another for hospital-based IOPs for 4-service days (APC 5861 and APC 5862, respectively). Similar to the policy finalized for PHP, we finalized a policy to utilize the CMHC rates for 3-service and 4-service IOP days as the MPFS rates, depending upon whether a nonexcepted hospital outpatient department furnishes 3 or 4 IOP services in a day.
We also established payment for IOP provided by an RHC or FQHC at the same rate as APC 5861, which is the 3-service hospital-based IOP rate (§ 405.2462(j)). Furthermore, we established a payment adjustment for IOP provided by an OTP based on 3 times the payment rate for APC 5861 beginning in CY 2024 (§ 410.67(d)(4)(i)(F)).
B. Coding and Billing for PHP and IOP Services Under the OPPS
In the CY 2024 OPPS/ASC final rule with comment period, we finalized a billing requirement that all providers use condition code 41 to indicate that a claim is for partial hospitalization services and use condition code 92 to identify intensive outpatient claims, effective January 1, 2024. Since the statutory definitions of both IOP and PHP generally include the same types of items and services covered, we stated in the CY 2024 OPPS/ASC final rule with comment period that we believe it is appropriate to align the programs using a consistent list of services, so that level of intensity would be the only differentiating factor between partial hospitalization services and intensive outpatient services. The use of condition codes 41 for PHP claims and 92 for IOP claims allows us to differentiate between these services for billing purposes.
We recognize that the level of intensity of mental health services that a patient requires may vary over time; therefore, we believe utilizing a consolidated list of HCPCS codes to identify services under both the IOP and PHP benefits supports a smooth transition for patients when a change in the intensity of their services is necessary to best meet their needs. For example, a patient receiving IOP services may experience an acute mental health need that necessitates more intense services through a PHP. Alternatively, an IOP patient that no longer requires the level of intensity provided by the IOP can access less intense mental health services, such as individual mental health services. The full list of HCPCs codes recognized under the PHP and IOP benefits can be found in the Medicare Claims Processing internet Only Manual, Chapter 4, sections 260.1 and 261.1, respectively, and their subsections, available at https://www.cms.gov/regulations-and-guidance/guidance/manuals/downloads/clm104c04.pdf.
To qualify for payment for the IOP APC (5851, 5852, 5861, or 5862) or the PHP APC (5853, 5854, 5863, or 5864), one service provided that day must be from the Partial Hospitalization and Intensive Outpatient Primary list. We refer readers to the CY 2024 OPPS final rule with comment period for further discussion regarding our expectation that at least one of the services on the PHP and IOP Primary list will be indicated per day for patients who need the level of care offered by a PHP or IOP program. The PHP and IOP Primary List can be found in the CY 2024 OPPS/ASC final rule with comment period at 88 FR 81821.
Beginning in CY 2024, we recognized caregiver training services and Principal Illness Navigation (PIN) services as PHP and IOP services. We explained that the reported costs associated with providing such services are included when we calculate the PHP and IOP payment rates; however, these services do not count toward the determination of whether a PHP or IOP day is paid at the 3-service or 4-service rate. We refer readers to the CY 2024 OPPS/ASC final rule with comment period for a detailed discussion of this policy (88 FR 81823 through 81825).
As finalized in the CY 2024 OPPS/ASC final rule with comment period, if new codes are established that represent the PHP and IOP services described under §§ 410.43(a)(4) and 410.44(a)(4), respectively, such codes are added to the list of codes recognized for payment for PHP or IOP through sub-regulatory guidance. We note that coding updates frequently occur outside of the standard rulemaking timeline. We adopted this sub-regulatory process in order to pay expeditiously when new codes are created that describe any of the services enumerated at §§ 410.43(a)(4) and 410.44(a)(4), which PHPs and IOPs, respectively, would provide. We explained that this policy applies to new codes that are cross walked to a previously included code, or whose code descriptor is substantially similar to a descriptor for a code on the list or describes a service on the list. We stated that any additional services not described at § 410.43(a)(4) or § 410.44(a)(4) would be added to the lists in regulation through notice and comment rulemaking. We note that for CY 2025, we did not propose to add any new services not described at § 410.43(a)(4) or § 410.44(a)(4) to the list of PHP and IOP services. However, we received comments regarding the addition of new services to the PHP and IOP benefits. A summary of these comments and our response is provided.
Comment: Several commenters advocated for caregiver training services and principal illness navigation (PIN) services to be included in the number of PHP or IOP services per day for payment. Commenters also strongly supported the inclusion of discharge support and peer services be adopted as applicable for PHP and IOP. ( printed page 94267)
Response: We thank commenters for their feedback. As discussed in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81825), PIN services are reported monthly and represent time spent throughout the month; therefore, we did not include these services in the evaluation of whether a particular PHP or IOP day receives the 3-service or 4-service day for payment. We intend to continue to analyze utilization and cost data for these services and consider any payment changes in future rulemaking to better recognize such costs. Additionally, as discussed in the CY 2024 PFS final rule with comment period (88 FR 78938 through 78949), CMS finalized to pay separately for PIN services to account for resources when clinicians involve certain types of health care support staff such as peer support specialists in furnishing medically necessary care. We believe that including these PIN HCPCS codes in the services recognized for PHP and IOP appropriately indicates that the services of peer support specialists are eligible services for inclusion in PHP and IOP.
Regarding the suggestion to count caregiver training services in the evaluation of whether a PHP or IOP day receives the 3-service or 4-service day for payment, we note that, as explained in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81822), we believe it would be appropriate to exclude these services from the determination of the number of services provided per day, but we would include such services in the calculation of cost per day for determining the PHP and IOP payment rates. We recognize that caregiver training services are an integral part of many PHPs and IOPs; however, we believe that until we have actual utilization data for the provision of caregiver training services, it would be more appropriate to base the payment for a higher-intensity PHP or IOP day on the provision of therapeutic, diagnostic, and training services that have historically been recognized for PHP or OPPS payment.
We did not propose to include caregiver training services in the determination of whether a PHP or IOP day receives the 3-service or 4-service day for payment, and we are not finalizing any changes to this policy for CY 2025. We intend to continue analyzing utilization and cost data for these services to consider whether payment changes may be appropriate in the future.
Comment: One commenter suggested CMS include the treatment of postpartum depression as applicable for PHP and IOP.
Response: We thank the commenter for their suggestion, and note that currently, the ICD-10-CM code F53.0 ( Postpartum depression ) is included in the ICD-10-CM Codes that Support Medical Necessity list for PHP.[149] A patient may receive treatment of postpartum depression under a PHP as long as all other patient eligibility requirements are met, including that PHP patients are required to be certified by a physician as needing at least 20 hours of services per week, and the patient would require inpatient hospitalization if they did not receive PHP services.
In addition, we are clarifying in this CY 2025 OPPS/ASC final rule with comment period that ICD-10-CM code F53.0 ( Postpartum depression) is included in the ICD-10-CM Codes that Support Medical Necessity list for IOP. A patient may receive treatment of postpartum depression under an IOP as long as all other patient eligibility requirements are met, including that IOP patients are required to be certified by a physician as needing at least 9 hours of services per week.
Comment: One commenter requested CMS clarify the expectation that PHP and IOP participants identified as needing medically necessary occupational therapy (OT) services should receive those services as part of the overall interdisciplinary plan of care. This commenter also requested that CMS clarify whether occupational therapists can bill for other PHP and IOP services, which the commenter stated are within the scope of practice for occupational therapists. The commenter included the following services in their letter:
- 97153 Adaptive behavior tx by tech
- 97154 Grp adapt bhv tx by tech
- 97155 Adapt behavior tx phys/qhp
- 97156 Fam adapt bhv tx gdn phy/qhp
- 97157 Mult fam adapt bhv tx gdn
- 97158 Grp adapt bhv tx by phy/qhp
- 96112 Devel tst phys/qhp 1st hr
- 96156 Hlth bhv assmt/reassessment
- 96158 Hlth bhv ivntj indiv 1st 30
- 96161 Admin of caregiver-focused hlth risk asst for ben of patient
- 96164 Hlth bhv ivntj grp 1st 30
- 96167 Hlth bhv ivntj fam 1st 30
- 97151 Bhv id assmt by phys/qhp
- 97152 Bhv id suprt assmt by 1 tech
Response: We thank the commenter for their feedback. We are clarifying in this CY 2025 OPPS/ASC final rule with comment period that occupational therapy services are an important part of PHPs and IOPs, specifically listed in the statute at section 1861(ff)(2)(B) and in the regulations at §§ 410.43(a)(4)(ii) and 410.44(a)(4). Currently, HCPCS code G0129 is the recognized code for OT services provided for PHP and IOP patients. However, occupational therapists working within their scope of practice as determined by state law can provide and bill for other services as part of a PHP or IOP, including the services mentioned by the commenter which are listed above. We note that because these codes are already included in the list of HCPCS applicable for PHP and IOP, which are identified in Table 98 in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81827 through 81828), it is not necessary to make any changes to the list to permit them to be billed as part of a PHP or IOP. We are not adding any new HCPCS codes to the list at this time. However, we remind readers that we established a sub-regulatory process in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81818 through 81822) to consider the inclusion of additional codes that represent the PHP and IOP services described under §§ 410.43(a)(4) and 410.44(a)(4), respectively. We periodically review these code lists and may add new codes to the list of codes applicable for PHP and IOP if they describe services included under the regulatory definitions of partial hospitalization services or intensive outpatient services.
Comment: A few commenters were concerned CMS did not propose to allow PHP and IOP services to be furnished remotely. Commenters noted that the availability of remote PHP services during the COVID-19 Public Health Emergency (PHE) increased access to these services, especially in rural areas. The commenters believed allowing PHP and IOP services to be furnished remotely to beneficiaries in their homes would also increase access to these benefits.
Response: As discussed in the March 31, 2020, interim final rule with comment entitled “Medicare and Medicaid Programs; Policy and Regulatory Revisions in Response to the COVID-19 Public Health Emergency” (85 FR 19232), CMS announced the expansion of telehealth services on a temporary and emergency basis pursuant to waiver authority added under section 1135(b)(8) of the Act by the Coronavirus Preparedness and Response Supplemental Appropriations Act, 2020 (Pub. L. 116-123, March 6, 2020). In addition, in the April 30, 2020, interim final rule with comment entitled ( printed page 94268) “Additional Policy and Regulatory Revisions in Response to the COVID-19 Public Health Emergency” (85 FR 27562 through 27566), we discussed that effective March 1, 2020, and for the duration of the COVID-19 PHE, a hospital or CMHC could furnish such services using telecommunications technology to a beneficiary in a temporary expansion location if that beneficiary is registered as an outpatient. The flexibility to provide PHP services remotely to a beneficiary in his or her home ended with the expiration of the PHE on May 11, 2023.
In terms of the request to allow IOP services to be furnished remotely to beneficiaries in their homes, we note that section 1861(ff) of the Act, as amended by section 4124(b)(2)(B) of the CAA, 2023 adopts much of the statutory definition of PHP for IOP. Section 1861(ff)(3)(A) provides, in part, that a PHP (and through the cross-reference in section 1861(ff)(4), an IOP) is a distinct and organized intensive ambulatory treatment offering less than 24-hour-daily care other than in an individual's home or in an inpatient or residential setting. Thus, neither PHP nor IOP services can be furnished remotely to beneficiaries in their homes.
However, we clarified in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72001) that none of the PHP regulations would preclude a patient that is under a PHP plan of care from receiving other reasonable and medically necessary non-PHP services from a hospital. This means that patients in a PHP are not precluded from receiving remote mental health services provided outside of the PHP by the same or another hospital when such services are reasonable and medically necessary.
Likewise, none of the IOP regulations would preclude a patient that is under an IOP plan of care from receiving other reasonable and medically necessary non-IOP services from a hospital. Additionally, we are reiterating in this final rule with comment period that we would expect that a physician would update the patient's PHP or IOP plan of care to appropriately reflect any change to the type, amount, duration, or frequency of the therapeutic services planned for that patient in circumstances when a PHP or IOP patient receives non-PHP/IOP remote mental health services from a hospital outpatient department. We also note that the medical documentation should continue to support the patient's eligibility for participation in a PHP or IOP.
Final Decision: After consideration of the public comments we received, we are maintaining our coding and billing policies for PHP and IOP as established in the CY 2024 OPPS/ASC final rule with comment period.
C. CY 2025 Payment Rates for PHP and IOP
1. Background
Beginning in CY 2024, we established four separate PHP APC per diem payment rates: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5853 and APC 5854, respectively), and one for hospital-based PHPs for 3-service days and another for hospital-based PHPs for 4-service days (APC 5863 and APC 5864, respectively). In addition, for hospital-based PHPs, we finalized a policy to calculate payment rates using the broader OPPS data set, instead of using hospital-based PHP data only. We explained that using the broader OPPS data set allows CMS to capture data from claims not identified as PHP, but that also include the service codes and intensity required for a PHP day. Because we established consistent coding and payment between the PHP and IOP benefits, we considered all OPPS data for PHP days and non-PHP days that include 3 or more of the same service codes. We established four separate IOP APC per diem payment rates at the same rates we proposed for the PHP APCs: one for CMHCs for 3-service days and another for CMHCs for 4-service days (APC 5851 and APC 5852, respectively), and one for hospital-based IOPs for 3-service days and another for hospital-based IOPs for 4-service days (APC 5861 and APC 5862, respectively).
In the CY 2024 OPPS/ASC final rule with comment period, we noted that the standard PHP day is typically four services or more per day. We explained that we have historically provided payment for three services a day for extenuating circumstances when a beneficiary would be unable to complete a full day of PHP treatment. As we stated in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66672), it was never our intention that days with only three units of service should represent the number of services provided in a typical PHP day. Our intention was to cover days that consisted of three units of service only in certain limited circumstances. For example, as we noted in the CY 2009 OPPS/ASC proposed rule (73 FR 41513), we believe 3-service days may be appropriate when a patient is transitioning towards discharge (or days when a patient is at the beginning of his or her PHP stay). Another example of when it may be appropriate for a program to provide only three units of service in a day is when a patient is required to leave the PHP early for the day due to an unexpected medical appointment.
We also explained that prior to CY 2024, we historically prepared the data by first applying PHP-specific trims and data exclusions and assessing CCRs. We direct the reader to the CY 2016 OPPS/ASC final rule with comment period (80 FR 70463 through 70465) for a more complete discussion of these trims, data exclusions, and CCR adjustments. In prior rules, we have typically included a discussion of PHP-specific data trims, exclusions, and CCR adjustments; we did not include that discussion in the CY 2024 OPPS/ASC proposed rule or final rule with comment period. We stated that these PHP-specific data trims and exclusions addressed limitations as well as anomalies in the PHP data. However, as noted earlier, we finalized a methodology for CY 2024 to calculate hospital-based PHP payment rates for 3 services per day and 4 services per day based on cost per day using the broader OPPS data set. Accordingly, we did not apply PHP-specific trims and data exclusions, but rather we applied the same trims and data exclusions consistent with the OPPS.
We stated in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81830) that while no IOP benefit existed prior to the CAA, 2023, the types of items and services included in IOP had been, and were, paid for by Medicare either as part of the PHP benefit or under the OPPS more generally. Additionally, we stated that prior to the CAA, 2023, CMS had begun gathering information from interested parties on IOP under Medicare. In the CY 2023 OPPS/ASC proposed rule (87 FR 44679), we issued a comment solicitation on intensive outpatient mental health treatment, including SUD treatment furnished by IOPs, to collect information regarding whether there are any gaps in coding that may be limiting access to needed levels of care for treatment of mental health disorders or SUDs for Medicare beneficiaries, and specific information about IOP services, such as the settings of care in which these programs typically furnish services, the range of services typically offered, and the range of practitioner types that typically furnish these services.
We explained that along with the requirements for IOP mandated by the CAA, 2023, we took into consideration information we received from the comment solicitation to construct an appropriate data set to develop ( printed page 94269) proposed rates for IOP. Since IOPs furnish the same types of services as PHP, just at a lower intensity, we stated that we believe it was appropriate to use the same data and methodology for calculating payment rates for both PHP and IOP for CY 2024. We explained that although PHP claims can be specifically identified, there was no specific identifier or billing code to indicate IOP services that may have been provided before CY 2024. However, we noted that hospitals have been permitted to furnish and bill for many of these services as outpatient services under the OPPS. Thus, we analyzed a broader set of data that included both PHP and non-PHP days with 3 or more services in order to calculate proposed payment for PHP services. In order to establish consistent payment between PHP and IOP, we set IOP payment rates at the same rates as PHP. We stated that the primary goal in developing the payment rate methodology for IOP and PHP services was to pay providers an appropriate amount relative to the patients' needs, and to avoid cost inversion in future years. We stated that setting the IOP payment rates equal to the PHP payment rates was appropriate because IOP was a newly established benefit, and we did not have definitive data on utilization. However, we explained that both programs utilize the same services, but furnish them at different levels of intensity, with different numbers of services furnished per day and per week, depending on the program. Therefore, we stated that we expect it would be appropriate to pay the same per diem rates for IOP and PHP services unless future data analysis supports calculating rates independently.
2. CY 2025 Payment Rate Methodology for PHP and IOP
For CY 2025, we proposed to use the latest available cost information, from cost reports beginning three fiscal years prior to the year that is the subject of the rulemaking, and CY 2023 OPPS claims to update the payment rates for the four PHP APCs and the four IOP APCs finalized in the CY 2024 OPPS/ASC final rule with comment period. The proposal is consistent with the overall proposed use of cost data for the OPPS, which is discussed in section II.A.1.a of this final rule with comment period. In accordance with the methodology finalized in the CY 2024 OPPS/ASC final rule with comment period, we proposed to base the payment rate for each PHP APC on the geometric mean per diem cost for days with three services and four or more services, calculated separately for CMHCs and hospital outpatient departments. We proposed to use the broader set of OPPS data to calculate the geometric mean costs for hospital outpatient departments, and we proposed to apply the same trims and exclusions consistent with the OPPS. We also proposed to set the payment rates for the four IOP APCs based on the geometric mean per diem cost for PHP days with three services and four or more services, calculated separately for CMHCs and hospital outpatient departments. Lastly, we proposed that if more recent data subsequently became available after the publication of the CY 2025 OPPS/ASC proposed rule, we would use such updated data, if appropriate, to determine the CY 2025 payment rates for the four PHP APCs and the four IOP APCs finalized in the CY 2024 OPPS/ASC final rule with comment period.
For beneficiaries in a PHP or IOP, we proposed to apply the four-service payment rate (that is, payment for PHP APCs 5854 for CMHCs and 5864 for hospitals, and IOP APCs 5852 for CMHCs and 5862 for hospitals) for days with 4 or more services. For days with three or fewer services, we proposed to apply the three-service payment rate (that is, payment for PHP APCs 5853 for CMHCs and 5863 for hospitals, and IOP APCs 5851 for CMHCs and 5861 for hospitals), which is consistent with the policy we established in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81833). As we noted in the CY 2024 OPPS/ASC final rule with comment period, we expect days with fewer than three services would be very infrequent, and we intend to monitor the provision of these days among providers and individual patients.
Comment: We received five comments from providers, hospital associations, and national organizations expressing concerns about the proposed PHP and IOP per diem rates. Commenters expressed that the proposed rates would not be sufficient to ensure the sustainability of PHP and IOP programs and could negatively affect access to PHP services. Some commenters noted that the proposed payment rates for PHP and IOP APCs published in Addendum A were lower than the geometric mean per diem cost figures published in the proposed rule. The commenters requested that CMS reconsider its approach to rate structures for IOPs and PHPs so that rates sufficiently cover the cost of these programs.
Response: We understand the concerns that commenters raised regarding the proposed PHP and IOP rates for CY 2025. We proposed to calculate the PHP payment rates based on our longstanding methodology, in accordance with the statutorily required relative payment weight calculations under the OPPS. Under the longstanding OPPS ratesetting methodology, CMS establishes APC payment rates by annually reviewing and revising the APC groups and relative payment weights for APCs in accordance with sections 1833(t)(2) and 1833(t)(9)(A) of the Act, as further described in section II.A.4 of this final rule with comment period. We further note that the OPPS is subject to budget neutral adjustments to the weight scaler as described in section II.A.4 of this final rule with comment period and APC payment rates are calculated using the OPPS conversion factor described in section II.B. of this final rule with comment period. As a result of the OPPS budget neutrality adjustments, the proposed and final APC payment rates may be higher or lower than their estimated APC geometric mean costs. We believe these per diem costs are in line with 2024 per diem costs and appropriately use hospital and CMHC cost data. The final payment rates for the PHP and IOP APCs can be found in Addendum A of this CY 2025 OPPS/ASC final rule with comment period.
After consideration of the public comments we received, we are finalizing the payment rate methodology for CY 2025 as proposed. Table 136 below shows the final APCs and the calculated geometric mean per diem costs for this CY 2025 OPPS/ASC final rule with comment period. Additional information about the data trims, data exclusions, and CCR adjustments applicable to the data used for this final rule can be found online at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/index.html ).[150]
( printed page 94270)D. Outlier Policy for CMHCs
For CY 2025, we proposed to maintain the calculations of the CMHC outlier percentage, cutoff point and percentage payment amount, outlier reconciliation, outlier payment cap, and fixed dollar threshold according to previously established policies to include PHP and IOP services. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81834 through 81836) for more details on CMHC outlier policies, and to section II.G.1 of this final rule with comment period for our general policies for hospital outpatient outlier payments.
1. Background
As discussed in the CY 2004 OPPS final rule with comment period (68 FR 63469 through 63470), we created a separate outlier policy specific to the estimated costs and OPPS payments provided to CMHCs. We designated a portion of the estimated OPPS outlier threshold specifically for CMHCs, consistent with the percentage of projected payments to CMHCs under the OPPS each year, excluding outlier payments, and established a separate outlier threshold for CMHCs.
2. CMHC Outlier Percentage
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59267 through 59268), we described the current outlier policy for hospital outpatient payments and CMHCs. We note that we also discussed our outlier policy for CMHCs in more detail in section VIII.C of that same final rule (82 FR 59381). We set our projected target for all OPPS aggregate outlier payments at 1.0 percent of the estimated aggregate total payments under the OPPS (82 FR 59267). This same policy was also reiterated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58996), the CY 2020 OPPS/ASC final rule with comment period (84 FR 61350), and the CY 2021 OPPS/ASC final rule with comment period (85 FR 86082). We did not propose any changes to the CMHC outlier percentage policy for CY 2025.
3. Cutoff Point and Percentage Payment Amount
Also described in the CY 2018 OPPS/ASC final rule with comment period (82 FR 59381), our policy has been to pay CMHCs for outliers if the estimated cost of the day exceeds a cutoff point. In CY 2006, we set the cutoff point for outlier payments at 3.4 times the highest CMHC PHP APC payment rate implemented for that calendar year (70 FR 68551). For CY 2018, the highest CMHC PHP APC payment rate was the payment rate for CMHC PHP APC 5853. In addition, in CY 2002, the final OPPS outlier payment percentage for costs above the multiplier threshold was set at 50 percent (66 FR 59889). In CY 2018, we continued to apply the same 50 percent outlier payment percentage that applies to hospitals to CMHCs and continued to use the existing cutoff point (82 FR 59381). Therefore, for CY 2018, we continued to pay for partial hospitalization services that exceeded 3.4 times the CMHC PHP APC payment rate at 50 percent of the amount of CMHC PHP APC geometric mean per diem costs over the cutoff point. This same policy was also reiterated in the CY 2019 OPPS/ASC final rule with comment period (83 FR 58996 through 58997), the CY 2020 OPPS/ASC final rule with comment period (84 FR 61351), the CY 2021 OPPS/ASC final rule with comment period (85 FR 86082 through 86083), the CY 2022 OPPS/ASC final rule with comment period (86 FR 63670), the CY 2023 OPPS/ASC final rule with comment period (87 FR 72004), and the CY 2024 OPPS/ASC final rule with comment period (88 FR 81835). For CY 2024, we extended this policy to intensive outpatient services. We did not propose any changes to the cutoff point and payment amount policy for CY 2025.
4. Outlier Reconciliation
In the CY 2009 OPPS/ASC final rule with comment period (73 FR 68594 through 68599), we established an outlier reconciliation policy to address charging aberrations related to OPPS outlier payments. We addressed vulnerabilities in the OPPS outlier payment system that led to differences between billed charges and charges included in the overall CCR, which are used to estimate cost and would apply to all hospitals and CMHCs paid under the OPPS. We initiated steps to ensure that outlier payments appropriately account for the financial risk when providing an extraordinarily costly and complex service but are only being made for services that legitimately qualify for the additional payment.
For a comprehensive description of outlier reconciliation, we refer readers to the CY 2023 OPPS/ASC and CY 2019 OPPS/ASC final rules with comment period (83 FR 58874 through 58875 and 81 FR 79678 through 79680, respectively). We did not propose any changes to the outlier reconciliation policy for CY 2025.
5. Outlier Payment Cap
In the CY 2017 OPPS/ASC final rule with comment period, we implemented a CMHC outlier payment cap to be applied at the provider level, such that in any given year, an individual CMHC will receive no more than a set percentage of its CMHC total per diem payments in outlier payments (81 FR 79692 through 79695). Our analysis of ( printed page 94271) CY 2014 claims data found that CMHC outlier payments began to increase similarly to the way they had prior to CY 2004. This was due to inflated cost from three CMHCs that accounted for 98 percent of all CMHC outlier payments that year and received outlier payments that ranged from 104 percent to 713 percent of their total per diem payments. To balance our concern about disadvantaging CMHCs with our interest in protecting the benefit from excessive outlier payments and to mitigate potential inappropriate outlier billing vulnerabilities, we finalized the CMHC outlier payment cap at 8 percent of the CMHC's total per diem payments (81 FR 79694 through 79695) to limit the impact of inflated CMHC charges on outlier payments. This outlier payment cap only affects CMHCs; it does not affect other provider types (that is, hospital-based PHPs), and is in addition to and separate from the current outlier policy and reconciliation policy in effect. We did not propose any changes to the outlier payment cap for CY 2025. We received comments on the outlier payment cap for CMHCs, which are summarized below and include our responses.
Comment: Several commenters urged CMS to implement a site-neutral payment for CMHCs and hospital-based providers for PHP and IOP services, stating CMS policy creates arbitrary incentives for PHP and IOP services in settings other than CMHCs; a few of these commenters went on to state that a site-neutral payment would eliminate the need for a separate outlier policy for CMHCs.
Response: We disagree with commenters who believe CMS policy creates an incentive for PHP and IOP services in settings other than CMHCs and that a site-neutral payment would eliminate the need for a separate outlier policy for CMHCs. Current data for partial hospitalization reflect significant cost structure differences between hospitals and CMHCs, and our longstanding payment policies reflect those differences as well. We have no factual basis at this time on which to assume, as many commenters suggest, that the stark difference between hospital and CMHC payment rates for PHP services indicate that such services do not reflect the actual cost structure differences between facility types. As discussed in the CY 2004 OPPS final rule with comment period (68 FR 63469 and 63470), we noted the use of historical cost-to-charge ratios applied to current charges resulted in an excessive amount of outlier payments being made to CMHCs. As a result of more in-depth analysis of the 2001 data files that were used to compute the CY 2003 PHP per diem amount, we discovered a significant difference in the amount of outlier payments made to hospitals and CMHCs for PHP. Therefore, we finalized a policy to designate a portion of the estimated 2.0 percent outlier target amount specifically for CMHCs, consistent with the percentage of projected payments to CMHCs under the OPPS in CY 2004, excluding outlier payments. Given the difference in PHP charges between hospitals and CMHCs, we did not believe it was appropriate to make outlier payments to CMHCs using the outlier percentage target amount and threshold established for hospitals. Therefore, beginning in CY 2004, we created a separate outlier policy specific to the estimated costs and OPPS payments provided to CMHCs. We designated a portion of the estimated OPPS outlier threshold specifically for CMHCs, consistent with the percentage of projected payments to CMHCs under the OPPS each year, excluding outlier payments, and established a separate outlier threshold for CMHCs. Furthermore, to balance our concern about disadvantaging CMHCs with our interest in protecting the benefit from excessive outlier payments and to mitigate potential inappropriate outlier billing vulnerabilities, we finalized the CMHC outlier payment cap at 8 percent of the CMHC's total per diem payments (81 FR 79694 and 79695) to limit the impact of inflated CMHC charges on outlier payments. In conclusion, CMS does not believe our payment methodology has any effect on outlier policy.
Final Decision: After consideration of the public comments we received, we are maintaining our existing policy to apply a cap on outlier payments as we finalized in the CY 2017 OPPS/ASC final rule with comment period,
6. Fixed-Dollar Threshold
In the CY 2018 OPPS/ASC final rule with comment period (82 FR 59267 through 59268), for the hospital outpatient outlier payment policy, we set a fixed-dollar threshold in addition to an APC multiplier threshold. Fixed-dollar thresholds are typically used to drive outlier payments for very costly items or services, such as cardiac pacemaker insertions. Currently, for CY 2024, CMHC PHP APCs (5853 or 5854) and IOP APCs (5851 or 5852) are the only APCs for which CMHCs may receive payment under the OPPS, and these APCs are for providing a defined set of services that are relatively low cost when compared to other OPPS services. Because of the relatively low cost of CMHC services that are used to comprise the structure of CMHC PHP APCs (5853 or 5854) and IOP APCs (5851 or 5852), it is not necessary to also impose a fixed-dollar threshold on CMHCs. Therefore, in the CY 2018 OPPS/ASC final rule with comment period, we did not set a fixed-dollar threshold for CMHC outlier payments (82 FR 59381). This same policy was also reiterated in the CY 2020 OPPS/ASC final rule with comment period (84 FR 61351), the CY 2021 OPPS/ASC final rule with comment period (85 FR 86083), the CY 2022 OPPS/ASC final rule with comment period (86 FR 63508), the CY 2023 OPPS/ASC final rule with comment period (87 FR 72004), and the CY 2024 OPPS/ASC final rule with comment period (88 FR 81836). We did not propose any changes to the fixed-dollar threshold policy for CY 2025, which will continue in CY 2025.
IX. Services That Will Be Paid Only as Inpatient Services
A. Background
Established in rulemaking as part of the initial implementation of the OPPS, the inpatient only (IPO) list identifies services for which Medicare will only make payment when the services are furnished in the inpatient hospital setting because of the invasive nature of the procedure, the underlying physical condition of the patient, or the need for at least 24 hours of postoperative recovery time or monitoring before the patient can be safely discharged (70 FR 68695). The IPO list was created based on the premise (rooted in the practice of medicine at that time), that Medicare should not pay for procedures furnished as outpatient services that are performed on an inpatient basis virtually all of the time for the Medicare population, for the reasons described above, because performing these procedures on an outpatient basis would not be safe or appropriate, and therefore not reasonable and necessary under Medicare rules (63 FR 47571). Services included on the IPO list were those determined to require inpatient care, such as those that are highly invasive, result in major blood loss or temporary deficits of organ systems (such as neurological impairment or respiratory insufficiency), or otherwise require intensive or extensive postoperative care (65 FR 67826). There are some services designated as inpatient only that, given their clinical intensity, would not be expected to be performed in the hospital outpatient setting. For example, we have traditionally considered certain surgically invasive ( printed page 94272) procedures on the brain, heart, and abdomen, such as craniotomies, coronary-artery bypass grafting, and laparotomies, to require inpatient care (65 FR 18456). Designation of a service as inpatient only does not preclude the service from being furnished in a hospital outpatient setting but rather means that Medicare will not make payment for the service if it is furnished to a Medicare beneficiary in the hospital outpatient setting (65 FR 18443). Conversely, the fact that a procedure is not on the IPO list should not be interpreted to mean the procedure is only appropriately performed in the hospital outpatient setting (70 FR 68696).
As part of the annual update process, we have historically worked with interested parties, including professional societies, hospitals, surgeons, hospital associations, and beneficiary advocacy groups, to evaluate the IPO list and to determine whether services should be added to or removed from the list. Interested parties are encouraged to request reviews for a particular code or group of codes; and we have asked that their requests include evidence that demonstrates that the procedure was performed on an outpatient basis in a safe and appropriate manner in a variety of different types of hospitals—including but not limited to—operative reports of actual cases, peer-reviewed medical literature, community medical standards and practice, physician comments, outcome data, and post-procedure care data (67 FR 66740).
We traditionally have used five longstanding criteria to determine whether a procedure should be removed from the IPO list. As noted in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74353), we assess whether a procedure or service meets these criteria to determine whether it should be removed from the IPO list and assigned to an APC group for payment under the OPPS when provided in the hospital outpatient setting. We have explained that while we only require a service to meet one criterion to be considered for removal, satisfying only one criterion does not guarantee that the service will be removed; instead, the case for removal is strengthened with the more criteria the service meets. The criteria for assessing procedures for removal from the IPO list are as follows:
- Most outpatient departments are equipped to provide the services to the Medicare population.
- The simplest procedure described by the code may be furnished in most outpatient departments.
- The procedure is related to codes that we have already removed from the IPO list.
- A determination is made that the procedure is being furnished in numerous hospitals on an outpatient basis.
- A determination is made that the procedure can be appropriately and safely furnished in an ASC and is on the list of approved ASC services or has been proposed by us for addition to the ASC covered procedures list.
In the past, we have requested that interested parties submit corresponding evidence in support of their claims that a code or group of codes meet the longstanding criteria for removal from the IPO list and are safe to perform on the Medicare population in the hospital outpatient setting—including, but not limited to case reports, operative reports of actual cases, peer-reviewed medical literature, medical professional analysis, clinical criteria sets, and patient selection protocols. Our clinicians then thoroughly review all information submitted within the context of the established criteria and if, following this review, we determine that there is sufficient evidence to confirm that the code could be safely and appropriately performed on an outpatient basis, we assign the service to an APC and include it as a payable procedure under the OPPS (67 FR 66740). We determine the APC assignment for services removed from the IPO list by evaluating the clinical similarity and resource costs of the service compared to other services paid under the OPPS and by reviewing the Medicare Severity Diagnosis Related Groups (MS-DRG) rate for the service under the IPPS, though we note we would generally expect the cost to provide a service in the outpatient setting to be less than the cost to provide the service in the inpatient setting.
We stated in prior rulemaking that, over time, given advances in technology and surgical technique, we would continue to evaluate services to determine whether they should be removed from the IPO list. Our goal is to ensure that inpatient only designations are consistent with the current standards of practice. We have asserted in prior rulemaking that, insofar as advances in medical practice mitigate concerns about these procedures being performed on an outpatient basis, we would be prepared to remove procedures from the IPO list and provide payment for them under the OPPS (65 FR 18443). Further, CMS has at times had to reclassify codes as inpatient only services with the emergence of new information.
We refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74352 through 74353) for a full discussion of our historic policies for identifying services that are typically provided only in an inpatient setting and that, therefore, will not be paid by Medicare under the OPPS, as well as the criteria we have used to review the IPO list to determine whether any services should be removed.
B. Changes to the Inpatient Only (IPO) List
As stated above, we encourage interested parties to request reviews for a particular code or group of codes for removal from the IPO list. For the CY 2025, we received requests from interested parties recommending that certain services be removed from the IPO list. Following our clinical review using the five criteria listed above, we did not find sufficient evidence that any of those services meet the criteria to be removed from the IPO list for CY 2025. Therefore, we did not propose to remove any services from the IPO list for CY 2025.
We proposed to add three services for which codes were newly created by the AMA CPT Editorial Panel for CY 2025 to the IPO list. These new services are described by CPT codes 0894T, 0895T, and 0896T, which will be effective on January 1, 2025. After clinical review of these services, we found that they require a hospital inpatient admission or stay and are not appropriate for payment under the OPPS. We proposed to assign these services to status indicator “C” (Inpatient Procedures) for CY 2025. The CPT codes, long descriptors, and the proposed CY 2025 payment indicators are displayed in Table 137.
Table 137 contains the proposed changes to the IPO list for CY 2025. The complete list of codes describing services that we proposed to designate as inpatient only services beginning in CY 2025 is also included as Addendum E to the CY 2025 proposed rule, which is available via the internet on the CMS website.
( printed page 94273)Comment: We received several comments in support of our proposal to add CPT codes 0894T, 0895T, and 0896T to the IPO list for CY 2025.
Response: We thank the commenters for their support.
Comment: We received one comment requesting that we remove CPT code 22848 (Pelvic fixation (attachment of caudal end of instrumentation to pelvic bony structures) other than sacrum (List separately in addition to code for primary procedure)) from the IPO list. Additionally, the commenter presented at the 2024 Hospital Outpatient Panel (HOP) on August 26, 2024, with the Panel recommending CMS remove the service from the IPO list.
The commenter stated that the service described by CPT code 22848 met the following criteria for removal: (1) most outpatient departments are equipped to provide the services to the Medicare population; (2) the simplest procedure described by the code may be furnished in most outpatient departments; (3) the procedure is related to codes that we have already removed from the IPO list; and (4) a determination is made that the procedure is being furnished in numerous hospitals on an outpatient basis.
Response: We thank the commenter for their feedback. After further in-depth review of the evidence provided, we agree with the commenter that this service meets criteria 1, 2, 3, and 4 in our regulation text at § 419.23(b)(1) through (4). For criteria 1 and 2, we found that most outpatient departments are equipped to provide this service to the Medicare population and the simplest procedure described by CPT code 22848 may be furnished in most outpatient departments. For criterion 3, the procedure is related to codes that we have already removed from the IPO list. Our review found that this procedure is the related to CPT codes 22612 (Arthrodesis, posterior or posterolateral technique, single interspace; lumbar (with lateral transverse technique, when performed)) and 22614 (Arthrodesis, posterior or posterolateral technique, single interspace; each additional interspace (list separately in addition to code for primary procedure)). Finally, for criterion 4, we found that this procedure is being furnished in numerous hospitals on an outpatient basis. Since CPT code 22848 is an add-on code and will always be packaged, we are reassigning this code to status indicator “N” (Items and Services Packaged into APC Rates).
Comment: We received one comment requesting that we add CPT code 31647 (Bronchoscopy, rigid or flexible, including fluoroscopic guidance, when performed; with balloon occlusion, when performed, assessment of air leak, airway sizing, and insertion of bronchial valve(s), initial lobe) to the IPO list. The commenter cited that the standard of care for this procedure required a stay of 3 to5 days for this procedure. The commenter was also concerned that, while a procedure on the IPO list can be performed as either outpatient or inpatient, outside payors who depend on the IPO list may refuse to approve inpatient admissions for surgeries not on the IPO list that require inpatient stays over two midnights.
Response: We thank the commenter for their feedback. CPT code 31647 was created in 2013 and is currently assigned to APC 5155 (Level 5 Airway Endoscopy) and status indicator “J1” (Hospital Part B Services Paid Through a Comprehensive APC). While our review found that the mean length of stay from the OPPS claims for CY 2020-2024 ranged from 2.10 to 3.02 days, showing that this procedure does require an overnight hospital stay for some patients, our review also found that a portion of patients are receiving ( printed page 94274) this service in the hospital outpatient setting. Because a portion of beneficiaries have been receiving this procedure in the hospital outpatient setting, we believe that it is appropriate for CPT code 31647 to remain off the IPO list and assigned to a clinical APC. As stated above, the fact that a procedure is not on the IPO list should not be interpreted to mean the procedure is only appropriately performed in the hospital outpatient setting.
Comment: We received four comments requesting that we assign services newly removed from the IPO list to New Technology APCs until sufficient data is collected to assign these services to clinical APCs.
Response: We thank the commenters for their input. As we previously stated in the CY 2021 OPPS/ASC final rule with comment period (85 FR 86093), consistent with our regulation at 42 CFR 419.31(a)(1), we classify outpatient services and procedures that are comparable clinically and in terms of resource use into APC groups. As we stated in the CY 2012 OPPS/ASC final rule with comment period (76 FR 74224), the OPPS is a prospective payment system that provides payment for groups of services that share clinical and resource use characteristics. It should be noted that for all codes newly paid under the OPPS, including codes removed from the IPO list, our policy has been to assign the service or procedure to an APC based on feedback from a variety of sources, including but not limited to, review of the clinical similarity of the service to existing procedures; advice from CMS medical advisors; information from interested specialty societies; and review of all other information available to us, including information provided to us by the public, whether through meetings with stakeholders or additional information that is mailed or otherwise communicated to us (84 FR 61229). Therefore, we believe assigning procedures removed from the IPO list to existing clinical APCs that are similar in clinical characteristics and resource costs is appropriate. We note that procedures assigned to New Technology APCs cannot be placed in clinical APCs due to insufficient clinical and cost data, unlike the procedures transitioning from the IPO list.
Comment: One commenter was concerned about the transparency of CMS's review of requests to remove or add services to the IPO list and asked that CMS publish a list of these requests in the OPPS/ASC proposed rules.
Response: We thank the commenter for their input and will consider it for future rulemaking. As stated above, we review the IPO list on a yearly basis and encourage interested parties to request reviews for a particular code or group of codes for removal or addition to the IPO list, after which our clinicians thoroughly review all information submitted within the context of the established criteria. While we have not historically published pre-proposed rule recommendations for which we did not propose any changes, we do address all codes that are recommended during the public comment period in the OPPS/ASC final rule.
Comment: We received one comment requesting that CMS preserve the treating physician's autonomy and create a formal review panel of physicians to evaluate changes to the IPO list. Another commenter also stated that the determination of the appropriate surgical site of service should be left to the judgment of surgeons.
Response: We thank the commenters for their feedback. In the CY 2022 OPPS/ASC final rule with comment period, we explained that we recognized that while physicians are able to make safety determinations for a specific beneficiary, CMS is in the position to make safety determinations for the broader population of Medicare beneficiaries. Furthermore, we explained that we want to afford physicians and hospitals the maximum flexibility in choosing the most clinically appropriate site of service for the procedure, as long as the characteristics of the procedure are consistent with the criteria listed above (86 FR 63673). While we are open to suggestions from interested parties on improving the IPO list review process, we reiterate that recommendations and changes to the IPO list are thoroughly reviewed by our clinicians.
In summary, after consideration of the public comments we received, we are finalizing our proposal with modification to add CPT codes 0894T, 0895T, and 0896T to the IPO list and assign them to status indicator “C” (Inpatient Procedures). We are also removing CPT code 22848 from the IPO list and reassigning it to status indicator “N” (Items and Services Packaged into APC Rates). Table 138 below contains the changes to the IPO list for CY 2025. The complete list of codes that are designated as inpatient only services beginning in CY 2025 is also included as Addendum E to this final rule with comment period, which is available via the internet on the CMS website.
( printed page 94275)X. Nonrecurring Policy Changes
A. Remote Services
1. Payment for Outpatient Therapy Services, Diabetes Self-Management Training, and Medical Nutrition Therapy When Furnished by Institutional Staff to Beneficiaries in Their Homes Through Communications Technology
Section 1861(p) of the Act establishes the benefit category for outpatient PT, SLP and OT services (expressly for PT services and, through section 1861(ll)(2) of the Act, for outpatient SLP services and, through section 1861(g) of the Act, for outpatient OT services).
Section 1861(p) of the Act defines outpatient therapy services in the three disciplines as those furnished by a provider of services, a clinic, rehabilitation agency, or a public health agency, or by others under an arrangement with, and under the supervision of, such provider, clinic, rehabilitation agency, or public health agency to an individual as an outpatient; and those furnished by a therapist not under arrangements with a provider of services, clinic, rehabilitation agency, or a public health agency. As such, section 1861(p) of the Act defines outpatient therapy services very broadly to include those furnished by providers and other institutional settings, as well as those furnished in office settings. Section 1834(k)(3) of the Act requires payment for outpatient therapy services to be made based on the PFS (via section 1848 of the Act), for all institutional providers listed at sections 1833(a)(8) and (9) of the Act. These providers include clinics, rehabilitation agencies, public health agencies, comprehensive outpatient rehabilitation agencies (CORFs), SNFs, home health agencies (HHAs) (to individuals who are not homebound), hospitals to outpatients or hospital inpatients who are entitled to benefits under Part A but have exhausted benefits for inpatient hospital services during a spell of illness or are not so entitled to benefits under Part A, and all other CORF services.
Section 1861(qq) of the Act defines Diabetes Self-Management Training (DSMT) services and authorizes CMS to regulate Medicare DSMT outpatient services. A “certified provider” of DSMT is further defined in section 1861(qq)(2)(A) of the Act as a physician or other individual or entity designated by the Secretary who meets certain quality requirements described in section 1861(qq)(2)(B) of the Act. In CY 2000, we finalized a standalone rule titled “Medicare Program; Expanded Coverage for Outpatient Diabetes Self-Management Training and Diabetes Outcome Measurements.” In that rule, we finalized that payment for outpatient DSMT would be made under the PFS (65 FR 83132). We further established that, in the case of payments made to other approved entities, such as hospital outpatient departments, ESRD facilities, and durable medical equipment suppliers, the payment would be equal to the amounts established under the PFS and made under the appropriate payment systems (65 FR 83142). ( printed page 94276)
Section 1861(s)(2)(V) of the Act authorizes Medicare Part B coverage of medical nutrition therapy services (MNT) for certain beneficiaries who have diabetes or a renal disease. In the CY 2002 PFS final rule, we established that payment for MNT services furnished in the institutional setting, including hospital outpatient departments (HOPDs), would be made under the PFS, not under the hospital Outpatient Prospective Payment System (OPPS) (66 FR 55279). Telehealth services may be paid under the PFS only when the services are furnished to a beneficiary at an originating site (defined at § 410.78), which prior to the PHE was not typically defined to include a beneficiary's home.
During the PHE for COVID-19, outpatient therapy services, DSMT, and MNT could be furnished via a telecommunications system to beneficiaries in their homes, and bills for these services were submitted and paid either separately or as part of a bundled payment, when either personally provided by the billing practitioner or provided by institutional staff and billed for by institutions, such as HOPDs, SNFs, and HHAs. For professionals, CMS used waiver authority provided under section 1135 of the Act to expand the range of practitioners that could serve as distant site practitioners for Medicare telehealth services as described in section 1834(m)(4)(E) of the Act and 42 CFR 410.78(b)(2), as well as to waive the originating site requirements for Medicare telehealth services described in section 1834(m)(4)(C) of the Act. This allowed for outpatient therapy services to be furnished and billed by therapists (PTs, OTs and SLPs) in private practice, and for DSMT and MNT to be furnished via Medicare telehealth to beneficiaries in urban areas, as well as rural areas, including to beneficiaries located in their homes.[151]
When CMS expanded the types of practitioners who may bill for their services when furnished as Medicare telehealth services from a distant site to include therapists using section 1135 waiver authority during the PHE for COVID-19, CMS generally took the position for services furnished in HOPDs that separate waiver authority was needed to allow hospitals to bill for services furnished by hospital staff through communication technology to beneficiaries in their homes. CMS implemented the Hospitals Without Walls (HWW) policy that relied on waiver authority, which allowed hospitals to reclassify patients' homes as part of the hospital. HWW allowed hospitals to bill two different kinds of fees for services furnished remotely to patients in their homes: (1) hospital facility payment in association with professional services billed under the PFS; and (2) single payment for a limited number of practitioner services, when statute or other applicable rules only allow the hospital to bill for services personally provided by their staff. These services are either billed by hospitals or by professionals, there would not be separate facility and professional billing. This latter category includes outpatient therapy services, DSMT, and MNT. However, while CMS relied upon PHE-specific waiver authority to allow hospital billing for these services, CMS also issued guidance instructing HOPDs to bill using modifiers consistent with those used for Medicare telehealth services. For further background, we refer readers to https://www.cms.gov/files/document/03092020-covid-19-faqs-508.pdf. In the same referenced document, CMS also issued specific guidance for other institutional providers of therapy services to use modifier 95 (indicating a Medicare telehealth service), along with the specific bill types for outpatient therapy services furnished by their staff.
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328) extended many of the flexibilities that were available for Medicare telehealth services during the PHE for COVID-19 under emergency waiver authorities, including adding PTs, OTs and SLPs as distant site practitioners through the end of CY 2024. In developing post-PHE guidance, CMS initially took the position that institutions billing for services furnished remotely by their employed practitioners (where the practitioners do not bill for their own services), would end with the PHE for COVID-19 along with the HWW waivers.[152] However, after reviewing input from interested parties, as well as relevant guidance, including applicable billing instructions, we considered whether certain institutions, as the furnishing providers, can bill for certain remotely furnished services personally performed by employed practitioners.
In the CY 2024 PFS final rule, we stated that, while we considered how we might address this topic in future rulemaking, in the interests of maintaining access to outpatient therapy, DSMT, and MNT services furnished remotely by institutional staff to beneficiaries in their homes consistent with the accessibility of these services when furnished by professionals via Medicare telehealth, we finalized that we would continue to allow institutional providers to bill for these services when furnished remotely in the same manner they have during the PHE for COVID-19 through the end of CY 2024. We sought comment on current practice for these services when billed, including how and to what degree they continue to be provided remotely to beneficiaries in their homes. We sought comment as to whether these services may fall within the scope of Medicare telehealth at section 1834(m) of the Act or if there are other relevant authorities CMS might consider in future rulemaking. For further information on this comment solicitation, please see the discussion in the CY 2024 PFS final rule (88 FR 78886 through 78888).
For DSMT specifically, we stated that the clinical staff personally delivering the service may be a type of practitioner authorized to furnish Medicare telehealth services under section 1834(m) of the Act; but we also understood that DSMT may be provided by other types of staff. Accordingly, we noted in an FAQ that we were exercising enforcement discretion in reviewing the telehealth eligibility status of the practitioner personally providing any part of a remotely furnished DSMT service, so long as the persons were otherwise qualified to provide the service until the end of 2024. For more background we refer readers to https://www.cms.gov/files/document/frequently-asked-questionscms-waivers-flexibilities-and-end-covid19-public-health-emergency.pdf.
While the amendments made by section 4113 of the CAA, 2023 to section 1834(m) of the Act have continued to expand the range of practitioners eligible to furnish telehealth services through CY 2024, without subsequent legislation these practitioners will no longer be able to bill for Medicare telehealth services beginning January 1, 2025.
In the CY 2024 PFS final rule, we articulated the importance of maintaining access to outpatient therapy, DSMT, and MNT services furnished remotely by institutional staff to beneficiaries in their homes consistent with the availability of these services when furnished by professionals via Medicare telehealth as part of our rationale for allowing institutional providers to bill for these services when furnished remotely in the ( printed page 94277) same manner they have during the PHE for COVID-19 through the end of CY 2024.
We recognize that for the past several years, through the PHE for COVID-19 and several legislative extensions of PHE-related flexibilities for Medicare telehealth services under section 1834(m) of the Act, we have generally aligned payment policies for outpatient therapy, DSMT, and MNT services furnished remotely by hospital staff to beneficiaries in their homes with policies for Medicare telehealth services. We noted in the CY 2025 OPPS/ASC proposed rule that, to the extent that therapists and DSMT and MNT practitioners continue to be distant site practitioners for purposes of Medicare telehealth services, we anticipated aligning our policy for these services with policies under the PFS and continuing to make payment to the hospital for these services when furnished by hospital staff.
Comment: Commenters supported our clarification that we would continue to align our policies for outpatient therapy, DSMT, and MNT services when furnished by hospital staff with the Medicare telehealth policies that apply when these services are billed by the same clinicians but in private practice. Commenters stated that aligning these policies would ensure clarity and consistency for providers and beneficiaries. Other commenters, while supportive, requested that CMS provide additional guidance for how outpatient therapy, DSMT, and MNT services should be billed when provided by hospitals staff versus how these services should be billed when furnished by practitioners in private practice. A few commenters suggested that CMS permanently allow payment for remote or virtual services under the OPPS.
Response: We thank commenters for their support for our clarification. Section 4113 of the CAA, 2023 expanded the range of practitioners eligible to furnish telehealth services only through CY 2024, which included PTs, OTs, and SLPs. Beginning January 1, 2025, these practitioners will no longer be able to bill for Medicare telehealth services. Consequently, beginning January 1, 2025, we likewise will no longer pay for outpatient therapy, DSMT, and MNT services when furnished remotely by hospital staff to beneficiaries in their homes. As we have stated before, and as noted by other commenters, continuing to align our policies for outpatient therapy, DSMT, and MNT services when furnished by hospital staff with the Medicare telehealth policies that apply when these services are billed by the same clinicians but in private practice ensures clarity and consistency for providers and beneficiaries.
We may consider refinements to our policies in future rulemaking.
2. Periodic In-Person Visits for Mental Health Services Furnished Remotely by Hospital Staff to Beneficiaries in Their Homes
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72017), we finalized a requirement that payment for mental health services furnished remotely to beneficiaries in their homes using telecommunications technology may only be made if the beneficiary receives an in-person service within 6 months prior to the first time the hospital clinical staff provides the mental health services remotely; and that there must be an in-person service without the use of telecommunications technology within 12 months of each mental health service furnished remotely by the hospital clinical staff. We also finalized that we would permit exceptions to the requirement that there be an in-person service without the use of communications technology within 12 months of each remotely furnished mental health service when the hospital clinical staff member and beneficiary agree that the risks and burdens of an in-person service outweigh the benefits. We stated that exceptions to the in-person visit requirement should involve a clear justification documented in the beneficiary's medical record, including the clinician's professional judgement that the patient is clinically stable and/or that an in-person visit has the risk of worsening the person's condition, creating undue hardship on the person or their family, or would otherwise result in disengaging with care that has been effective in managing the person's illness. We also finalized that hospitals must document that the patient has a regular source of general medical care and has the ability to obtain any needed point of care testing, including vital sign monitoring and laboratory studies. We finalized that these requirements would not go into effect until the 152nd day after the PHE for COVID-19 ends to maintain consistency with similar policies implemented for professional services paid under the PFS, and for RHCs/FQHCs (87 FR 72018).
Section 4113(d) of the CAA, 2023, extended the delay in implementing the in-person visit requirements until January 1, 2025, for both professionals billing for mental health services via Medicare telehealth and for RHCs/FQHCs furnishing remote mental health visits. In the CY 2024 OPPS final rule with comment period, we reiterated that we believe it is important to maintain consistent requirements for these policies across payment systems; therefore, we finalized delaying the in-person visit requirements for mental health services furnished remotely by hospital staff to beneficiaries in their homes until January 1, 2025. As such, these in-person visit requirements are currently set to take effect for services furnished on or after January 1, 2025 (88 FR 81874).
However, we noted in the CY 2025 OPPS/ASC proposed rule that, to the extent that these in-person visit requirements are delayed in the future for professionals billing for mental health telehealth services under the PFS, we anticipate that we would align the requirements for mental health services furnished remotely to beneficiaries in their homes through communications technology with mental health services furnished via Medicare telehealth in future rulemaking.
Comment: Commenters supported our clarification that we would continue to align our policies for in person visit requirements for mental health services furnished remotely to beneficiaries in their homes through communications technology with similar policies for mental health services furnished via Medicare telehealth. Of these supportive comments, many recommended that CMS permanently remove the in-person visit requirements for these services.
Response: We appreciate the supportive comments but note that the in-person visit requirement for professionals billing for mental health services via Medicare telehealth applies again beginning January 1, 2025. We continue to believe in the principle of aligning payment policy for remotely furnished mental health services across the OPPS and PFS payment systems, and accordingly, the in-person visit requirements will also apply for mental health services furnished remotely by hospital staff to beneficiaries in their homes through communications technology beginning January 1, 2025. We may consider refinements to our policy through future rulemaking.
3. HOPD Payment for Telemedicine Evaluation and Management Services
The CPT Editorial Panel created 17 new codes describing audio/video and audio-only telemedicine E/M services. For further discussion of these 17 new codes and CMS' related proposals, please see section II.E.4.18 of the CY 2025 PFS proposed rule.
In 2014, CMS established HCPCS code G0463 (Hospital outpatient clinic visit for assessment and management of ( printed page 94278) a patient) to describe the service associated with a hospital outpatient clinic visit for assessment and management of a patient. In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75042), we stated that the code is applicable for hospital use only representing any clinic visit under the OPPS. We further stated that HCPCS code G0463 replaces evaluation and management (E/M) CPT codes 99201-99205 (new patient) and 99211-99215 (established patient), thereby eliminating the distinction between new and established clinic visits.
Given the similarities between the new telemedicine E/M code set and the office/outpatient E/M code set, we believe that the telemedicine E/M codes fall within the scope of the hospital outpatient clinic visit policy because the predecessor codes (the office/outpatient E/M code set) would be reported by hospitals using HCPCS code G0463. Under the hospital outpatient clinic visit policy, the CPT codes describing office/outpatient E/M visits are not recognized under the OPPS and instead hospitals report HCPCS code G0463 (Hospital outpatient clinic visit for assessment and management of a patient) when billing for the facility costs associated with an outpatient E/M visit. Therefore, we proposed to not recognize the telemedicine E/M code set under OPPS. We invited comment on the hospital resources associated with the telemedicine E/M services, particularly any resource costs that would not be included in the payment for HCPCS code G0463. We also invited comment, should CMS finalize separate payment for these telemedicine E/M codes under the PFS, on the resource costs that would be associated with these services for hospitals and whether we should develop separate coding to describe the resource costs associated with a telemedicine E/M service.
We received public comments on the proposal. The following is a summary of the comments we received and our responses.
Comment: Some commenters supported our proposal to maintain our policy of not recognizing office/outpatient E/M visits, including those furnished via telemedicine, as payable under the OPPS and instead using HCPCS code G0463 to capture the associated facility costs. Other commenters recommended that CMS allow HCPCS code G0463 to be reported when the beneficiary is not physically in the hospital, while others encouraged CMS to provide separate payment for the telemedicine E/Ms, including recommending clinical APC assignments.
Response: We thank commenters for the additional information and may consider this issue, including any refinements to our clinic visit policy, for future rulemaking. As we are not finalizing payment for the telemedicine E/M CPT codes under the PFS, we do not believe there is a programmatic need to recognize these codes for payment under the OPPS.
After consideration of the public comments we received, we are finalizing our proposal not to recognize the telemedicine E/M code set under the OPPS as proposed.
B. Virtual Direct Supervision of Cardiac Rehabilitation (CR), Intensive Cardiac Rehabilitation (ICR), Pulmonary Rehabilitation (PR) Services and Diagnostic Services Furnished to Hospital Outpatients
1. Background
a. Virtual Direct Supervision of CR, ICR and PR Services Furnished to Hospital Outpatients (42 CFR 410.27(a)(1)(B)( 1))
In the interim final rule with comment period titled “Policy and Regulatory Provisions in Response to the COVID-19 Public Health Emergency,” published on April 6, 2020 (the April 6th COVID-19 IFC) (85 FR 19230, 19246, 19286), we changed the regulation at 42 CFR 410.27(a)(1)(iv)(D) [153] to provide that, during a Public Health Emergency as defined in 42 CFR 400.200, the presence of the physician for purposes of the direct supervision requirement for PR, CR, and ICR services includes virtual presence through audio/video real-time communications technology when use of such technology is indicated to reduce exposure risks for the beneficiary or health care provider. Specifically, the required direct physician supervision can be provided through virtual presence using audio/video real-time communications technology (excluding audio-only) subject to the clinical judgment of the supervising practitioner. We further amended § 410.27(a)(1)(iv)(B) [154] in the CY 2021 OPPS/ASC final rule with comment period to provide that this flexibility continues until the later of the end of the calendar year in which the PHE as defined in § 400.200 ends or December 31, 2021 (85 FR 86113 and 86299). In the CY 2021 OPPS/ASC final rule with comment period we also clarified that this flexibility excluded the presence of the supervising practitioner via audio-only telecommunications technology (85 FR 86113).
In the CY 2023 OPPS/ASC final rule with comment period, we finalized a policy to extend the revised definition of direct supervision of CR, ICR, and PR to include the presence of the supervising practitioner through two-way, audio/video telecommunications technology until December 31, 2023 (87 FR 72019 and 72020). In the CY 2024 OPPS/ASC final rule with comment period, we finalized a policy to further revise § 410.27(a)(1)(iv)(B)( 1 ) [155] to allow for the direct supervision requirement for CR, ICR, and PR to include the virtual presence of the physician through audio-video real-time communications technology (excluding audio-only) through December 31, 2024, and to extend this policy to the nonphysician practitioners, that is NPs, PAs, and CNSs, who were eligible to supervise these services beginning in CY 2024 (88 FR 81863 through 81867).
b. Virtual Direct Supervision of Diagnostic Services Furnished to Hospital Outpatients (42 CFR 410.28(e)(2)(iii))
In the April 6th COVID-19 IFC, for consistency with the revisions made to 42 CFR 410.27(a)(1)(iv)(D) [156] and 410.32(b)(3)(ii), we changed the regulation at 42 CFR 410.28(e) to provide that, during a Public Health Emergency as defined in 42 CFR 400.200, the presence of the physician for purposes of the direct supervision requirement for diagnostic services includes virtual presence through audio/video real-time communications technology when use of such technology is indicated to reduce exposure risks for the beneficiary or health care provider (85 FR 19245 and 19246).
In the CY 2023 OPPS/ASC final rule with comment period, to ensure consistency with additional revisions made to 42 CFR 410.27(a)(1)(iv)(B)( 1) and 410.32(b)(3)(ii) extending the end date of the flexibility allowing for the virtual supervision of the services governed by those regulations, we revised § 410.28(e) to extend the end date of the flexibility allowing for the virtual supervision of outpatient diagnostic services through audio/video real-time communications technology (excluding audio-only) from the end of the PHE to the end of the calendar year ( printed page 94279) in which the PHE ends (87 FR 72024 through 72026).
In the CY 2024 final OPPS rule with comment period, to again ensure consistency with further revisions made to 42 CFR 410.27(a)(1)(iv)(B)( 1) and 410.32(b)(3)(ii) extending the end date of the flexibility allowing for the virtual supervision of the services governed by those regulations, we revised § 410.28(e) to extend the end date of the flexibility allowing for the virtual supervision of outpatient diagnostic services through audio/video real-time communications technology (excluding audio-only) through December 31, 2024 (88 FR 81866 and 81867).
2. Extension of Virtual Direct Supervision of CR, ICR, PR Services and Diagnostic Services Furnished to Hospital Outpatients Through December 31, 2025.
In the CY 2025 PFS proposed rule, we proposed to revise the definition of direct supervision at § 410.32(b)(3)(ii) to extend the availability of virtual direct supervision of therapeutic and diagnostic services under the PFS through December 31, 2025. As described in that proposed rule, we proposed this extension based on our concern that an immediate reversion to the pre-PHE definition of direct supervision would prohibit virtual direct supervision, which may present a barrier to access to many services that have been facilitated by our PHE-related policy over the past several years; and that physicians and other practitioners need time to reorganize their practice patterns established during the PHE to reimplement the pre-PHE approach to direct supervision without the use of audio/video technology.
As we explained in the CY 2025 OPPS/ASC proposed rule, in addition to desiring uniformity under the PFS and OPPS in how regulations are applied to similarly situated providers, the beneficiary access and provider preparedness concerns that motivated us to propose extending the availability of virtual direct supervision of therapeutic and diagnostic services under the PFS through December 31, 2025, were also concerns with respect to the direct supervision of CR, ICR, PR and diagnostic services under the OPPS. Consequently, in the CY 2025 OPPS/ASC proposed rule, we proposed to revise §§ 410.27(a)(1)(iv)(B)( 1) and 410.28(e)(2)(iii) to allow for the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) through December 31, 2025.
The following is a summary of the comments we received on the proposal and our responses to those comments.
Comment: All commenters supported our proposal to make conforming revisions to §§ 410.27 and 410.28 to allow for the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) through December 31, 2025. These commenters indicated that this extension would reduce burden on providers, ensure the continuity of ongoing services, allow more time for providers to reorganize and readjust policies to meet pre-PHE requirements, improve patient access to historically underutilized services and be of particular value in rural and other underserved areas where workforce shortages remain acute.
Response: We thank commenters for their support.
Comment: Many commenters additionally requested that the availability of virtual direct supervision of these services be made permanent, arguing that virtual supervision has been in place long enough for its safety to have been established and any serious problems identified. One commenter expressed concern that if virtual supervision was not made permanent it would be more difficult to recruit and retain non-physician staff with the necessary training and experience to safely deliver services under virtual supervision and to recruit and retain physicians who can effectively provide virtual supervision because those staff and physicians would be concerned that the policy enabling remote supervision is temporary and could be revoked within a year. One commenter requested that once the virtual supervision of these services is made permanent that CMS retire the requirement for a service-level modifier to identify when direct supervision is provided via appropriate telehealth technology.
Response: We appreciate the commenters' suggestions to make the virtual direct supervision of CR, ICR, PR and diagnostic services permanent and will take them under consideration for future rulemaking as we continue to evaluate safety, quality of care, and other considerations related to virtual direct supervision.
Comment: One commenter, while supporting the proposed extension, specifically requested that the extension not be made permanent. The commenter opined that permanently allowing for the virtual direct supervision of CR, ICR, PR and diagnostic services would be undesirable because it would increase the amount of physician “incident to” billing (a Medicare billing provision that applies in the office or clinic setting and allows medical services to be performed by auxiliary personnel as an incident to the services of the billing practitioner and under their supervision) for services provided by PAs and NPs, which would obscure the extent to which PAs and NPs are actually performing the services.
Response: We appreciate the commenter's input regarding the appropriate attribution of services performed by PAs and NPs when those services are billed “incident to” a physician's service and we look forward to reviewing the information provided as we consider the most appropriate way to balance patient safety concerns with the interest of supporting access that we may address in future rulemaking. We reiterate that we did not propose to extend the availability of the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) beyond December 31, 2025.
Comment: One commenter requested that audio-only virtual supervision of CR, ICR and PR be allowed during system downtimes or situations where patients may not be comfortable or willing to participate in the video portion of the supervision of their CR, ICR, and PR. The commenter also contended that allowing this flexibility would reduce barriers to care for patients with disabilities or mobility issues who might have trouble getting into a physical location.
Response: We appreciate the commenter's suggestion that we allow for the virtual supervision of CR, ICR, and PR to be provided via audio-only, however, under the applicable definition of direct supervision, the physician/practitioner is required to be available using both audio and video. As a reminder, direct supervision means that the physician or nonphysician practitioner must be immediately available to furnish assistance and direction throughout the performance of the procedure. We have traditionally defined “immediately available” to require the physical presence of the supervising practitioner, given the importance of certain services being furnished under direct supervision to ensuring quality of care and patient safety, and in particular the ability of the supervising practitioner to intervene if complications arise. As we continue to evaluate safety, quality of care, and other considerations related to virtual direct supervision and the role of virtual direct supervision after the COVID-19 ( printed page 94280) PHE, we proposed to temporarily extend the availability of virtual direct supervision of therapeutic and diagnostic services only through December 31, 2025. To ensure quality of care and patient safety, any direct supervision furnished remotely will continue to require the use of real-time audio and visual interactive telecommunications technology. We note that does not necessarily mean that any interaction between the patient and the physician/practitioner supervising the service would require a video component.
Comment: One commenter questioned whether patient access to care is fully addressed by the virtual supervision of CR services in the absence of nationally available free or low-income access to broadband quality technology and telecommunications services. The commenter requested that CMS simultaneously adjust the facility and non-facility policies and prices under the Physician Fee Schedule (PFS) for outpatient CR services (HCPCS codes 93797 or 93798) so that Medicare pays hospital outpatient departments the PFS non-facility price at higher than current rates to allow facilities to subsidize the cost of supplying patients with broadband internet technologies for the duration of their enrollment in cardiac rehabilitation sessions.
Response: We appreciate the commenter's suggestion, but we note this comment is out of scope for this final rule.
Comment: One commenter encouraged CMS to consider additional services that could be safely supervised virtually in future years.
Response: We thank the commenter for their input and will consider taking it under consideration in future rulemaking.
Comment: Two commenters requested that CMS allow for CR and ICR to be provided virtually to patients in their homes, contending that CMS has the authority to do so under existing statute. These commenters stated that CMS has the authority under section 1861(eee) of the Act to (1) designate a patient's home as a setting for delivery of CR and ICR services and (2) allow CR and ICR services to be delivered virtually.
Response: We appreciate commenters' suggestion that section 1861(eee) of the Act provides the authority to furnish virtual CR and ICR services to outpatients in their homes and we will take their interpretation under consideration in future rulemaking.
After consideration of the public comments we received, we are finalizing, without modification, our proposal to revise §§ 410.27(a)(1)(iv)(B)( 1) and 410.28(e)(2)(iii) to allow for the direct supervision of CR, ICR, PR services and diagnostic services via audio-video real-time communications technology (excluding audio-only) through December 31, 2025.
C. All-Inclusive Rate (AIR) Add-On Payment for High-Cost Drugs Provided by Indian Health Service and Tribal Facilities
1. Background
In the CY 2000 OPPS final rule (65 FR 18434), CMS implemented the PPS for hospital outpatient services furnished to Medicare beneficiaries, as set forth in section 1833(t) of the Act. In this final rule, we noted that the OPPS applies to covered hospital outpatient services furnished by all hospitals participating in the Medicare program with a few exceptions. We identified one of these exceptions as “outpatient services provided by hospitals of the Indian Health Service (IHS).” We stated that these services would “continue to be paid under separately established rates which are published annually in the Federal Register ” and, in the CY 2002 OPPS/ASC final rule (66 FR 59856), we finalized a revision to § 419.20 (Hospitals subject to the hospital outpatient prospective payment system) by adding paragraph (b)(4), which specifies that hospitals of the IHS are excluded from the OPPS.
In the intervening years, IHS and tribal facilities have been paid under the separately established All-Inclusive Rate (AIR). On an annual basis, the IHS calculates and publishes, in the Federal Register , calendar year reimbursement rates.[157] Due to the higher cost of living in Alaska, separate rates are calculated for Alaska and the lower 48 States. For CY 2024, the Medicare Outpatient per Visit Rate is $667 for the lower 48 states and $961 for Alaska.[158]
IHS and tribal facilities have continued to expand the breadth of services that they provide to their communities. Increasingly, this has meant providing higher-cost drugs and biologics (hereinafter referred to as “drugs”) along with more complex and expensive services. While the majority of IHS and tribal facilities appear to be well served by the AIR, some IHS and tribal facilities provide specialized services for which the AIR might not adequately represent Medicare's share of costs. If providing a drug or service costs IHS and tribal facilities thousands of dollars more than the payment they receive through the AIR, it is likely not financially feasible for these facilities to routinely provide that drug or service. For example, the cost of providing a frequently used cancer drug such as Opdualag (HCPCS code 00003-7125), which has a per day cost of $28,975, greatly exceeds the $667 payment an IHS or tribal facility receives through the AIR. We are concerned that, if payments under the AIR are inadequate for high-cost drugs, this could potentially threaten the viability of the few IHS and tribal hospital outpatient specialty programs currently in operation and provide less incentive to IHS hospitals and tribal facilities not currently offering specialty services to begin doing so. This constitutes a significant equity and beneficiary access concern if IHS and tribal hospitals are not able to provide oncology services or other services that require high-cost drugs because the hospital would always receive payment for those services that is far below what it would have to pay to acquire those high-cost drugs.
Consequently, in the CY 2024 OPPS/ASC proposed rule, we sought comment on whether Medicare should pay separately for certain high-cost drugs provided by IHS and tribal facilities and, if so, how we might do so. Among other topics, we specifically requested input on which drugs it would be appropriate to pay separately for (high-cost oncology drugs or all high-cost drugs), how we might define high-cost drugs (for example, a list of named drugs versus any drugs exceeding a certain cost threshold), and what the appropriate payment amount for the separately paid drugs should be (ASP plus 6 percent, which is what hospitals are generally paid under the OPPS for separately payable drugs, or the Federal Supply Schedule (FSS), which is where IHS and tribal hospitals acquire the majority of their drugs at rates significantly lower than ASP plus 6 percent). For a full discussion of the comment solicitation, we refer readers to the CY 2024 OPPS/ASC proposed rule (88 FR 49741 through 49742).
Commenters, including a tribal facility, the CMS Tribal Technical Advisory Group (TTAG), organizations representing tribal healthcare providers, pharmaceutical companies, and other interested parties, expressed universal support for establishing a policy that would allow IHS and tribal healthcare facilities to receive separate payment outside of the AIR for high-cost drugs. The preferred approach of those ( printed page 94281) commenters who provided input on how to define a high-cost drug eligible for separate payment was to treat the amount of the Medicare Outpatient per Visit Rate for the lower 48 States' AIR (hereinafter referred to as “the lower 48 AIR”) as a payment threshold. Under this approach, if the cost of a particular drug is less than or equal to the lower 48 AIR, the provider would not receive a separate payment for the drug and if the cost of the drug was more than the lower 48 AIR, then the provider would receive a separate payment for the drug. Commenters noted that this payment approach is currently being used for all drugs (oncology and otherwise) receiving payment through Arizona Medicaid (AHCCCS) for IHS and tribal facilities located in Arizona. With respect to the payment amount, several commenters requested that separately payable drugs furnished by IHS and tribal facilities be paid at a rate of ASP plus 6 percent rather than the FSS rate. These commenters argued that the IHS is chronically underfunded and that paying ASP plus 6 percent for high-cost drugs could help with remedying those funding issues. For a full discussion of the comments we received as a result of our comment solicitation and our responses to those comments, we refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81896 through 81897).
2. AIR Add-On Payment for High-Cost Drugs Provided by Indian Health Service and Tribal Facilities
In light of the equity and beneficiary access concerns that prompted our CY 2024 comment solicitation and the input received in response to that solicitation, in the CY 2025 OPPS/ASC proposed rule we proposed, starting January 1, 2025, to separately pay IHS and tribal hospitals [159] for high-cost drugs furnished in hospital outpatient departments through an add-on payment in addition to the AIR using the authority under which the annual AIR is calculated.[160] We emphasized that the amount of the proposed add-on payment would not be carved out of the annual AIR payment amount calculation. In other words, we proposed that the add-on payment would have no effect on the calculation of the annual AIR payment amount. We invited comment on separately paying IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through the establishment of an add-on payment to the AIR using the authority under which the annual AIR is calculated.
Comment: All commenters on the proposed policy supported separately paying IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through the establishment of an add-on payment to the AIR using the authority under which the annual AIR is calculated. These commenters cited as the basis for their support the need to address beneficiary equity and access concerns resulting from the current payment policy for high-cost drugs under the AIR, historic and ongoing health disparities of American Indian and Alaska Native people compared to other Americans, including lower life expectancy, disproportionate disease burden and higher rates of colorectal, kidney, liver, lung, and stomach cancers, ensuring the continued viability of IHS and tribal hospital outpatient specialty programs currently in operation and enabling IHS and tribal facilities not currently offering specialty services to begin doing so. Several of these commenters additionally expressed support for the exclusion of the add-on payment from the calculation of the annual AIR payment, stating that it was a critically important component of the proposal because it would ensure that the add-on payment would have no effect on IHS and tribal facilities that do not seek reimbursement for high-cost drugs.
Response: We thank the commenters for their support.
After consideration of the public comments we received on the proposed policy to separately pay IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments through the establishment of an add-on payment to the AIR using the authority under which the annual AIR is calculated and to exclude the add-on payment from the calculation of the annual AIR payment, we are finalizing the policy as proposed.
In the CY 2025 OPPS/ASC proposed rule, we proposed that the drugs to which the add-on payment would apply would not be limited to high-cost oncology drugs but would include all high-cost drugs furnished in hospital outpatient departments of IHS and tribal hospitals to the extent those drugs were covered under Medicare Part B and would be paid for under the OPPS if furnished by a hospital paid under that system. In determining which drugs would be eligible for the add-on payment, we considered limiting the add-on payment to high-cost oncology drugs. However, as we discussed in the CY 2025 OPPS/ASC proposed rule, we determined that it would be appropriate to apply the add-on payment to all high-cost drugs for several reasons. First, the same equity and access concerns that supported utilizing an add-on payment for oncology drugs also supported utilizing an add-on for high-cost drugs used in other care specialties. Although this issue arose in the context of removing barriers to beneficiaries' access to high-cost oncology drugs, we concluded that there were presumably similar barriers to other specialties that used high-cost drugs that would be addressed through a broader application of the add-on payment. Second, we determined that applying the add-on payment to all high-cost drugs would eliminate the possibility of unintentionally excluding an oncology drug from separate payment due to the inherent challenge of defining a class of drugs. Third, the proposal would parallel how drugs are being paid for under Arizona Medicaid (AHCCCS) for IHS and tribal facilities. In the CY 2025 OPPS/ASC proposed rule we invited comment on applying the add-on payment to all high-cost drugs furnished in hospital outpatient departments of IHS and tribal hospitals to the extent those drugs were covered under Medicare Part B and would be paid for under the OPPS if furnished by a hospital paid under that system.
Comment: Several commenters supported applying the add-on payment to all high-cost drugs furnished in hospital outpatient departments of IHS and tribal hospitals to the extent those drugs are covered under Medicare Part B and would be paid for under the OPPS if furnished by a hospital paid under that system. These commenters observed that there are many other types of high-cost drugs that can be administered in an outpatient hospital setting that are unrelated to oncology and that the same equity and access concerns that support an add-on payment for high-cost oncology drugs applies to other high-cost drugs.
Response: We thank the commenters for their support.
After consideration of the public comments we received on the proposed policy to apply the add-on payment to all high-cost drugs furnished in hospital outpatient departments of IHS and tribal hospitals to the extent those drugs are covered under Medicare Part B and would be paid for under the OPPS if furnished by a hospital paid under that ( printed page 94282) system, we are finalizing the policy as proposed.
As to what constitutes a high-cost drug, we proposed in the CY 2025 OPPS/ASC proposed rule to define high-cost drugs for the purpose of the policy as all drugs covered under Medicare Part B and for which payment would otherwise be made under the OPPS whose per day cost exceeds two times the lower 48 AIR ($1,334 in CY 2024). We proposed a threshold greater than the lower 48 AIR to account for the fact that IHS and tribal hospitals would continue to receive the lower 48 AIR payment, in addition to the add-on payment, for encounters that include a high-cost drug. While we acknowledged that it was true that under the Arizona Medicaid program, IHS and tribal hospitals are paid the lower 48 AIR payment in addition to an add-on payment for drugs whose costs exceed the lower 48 AIR, we were concerned that providing separate payment for drugs whose costs only slightly exceed the lower 48 AIR could result in excessive payment for those drugs. For example, we pointed out that for a drug costing $700, using the CY 2024 lower 48 AIR as the threshold for our proposal would result in a payment of at least $1,367 (the $667 lower 48 AIR encounter payment plus an add-on payment for the high-cost drug as calculated under the payment methodology we proposed in the CY 2025 OPPS/ASC proposed rule) for the provision of a drug whose cost exceeds the lower 48 AIR by only $33.00. We concluded that such an outcome would be at odds with the objective of the proposed policy, which was to provide adequate payment for drugs that are high cost in relation to the lower 48 AIR. Consequently, in the CY 2025 OPPS/ASC proposed rule, we proposed two times the lower 48 AIR as the threshold triggering the add-on payment because that amount would ensure that the add-on payment would apply only to drugs whose costs significantly exceed the lower 48 AIR. The proposed cost-multiplier approach was also consistent with how CMS has implemented thresholds relating to payments to hospitals under other payment systems. For example, the OPPS outlier policy [161] requires that the cost of a service exceed 1.75 times the payment amount for the service to qualify for an additional payment. Similarly, the OPPS two-times rule requires that the highest calculated cost of an individual procedure categorized to any given Ambulatory Payment Classification (APC) not exceed two times the calculated cost of the lowest cost procedure categorized to that same APC.
In the CY 2025 OPPS/ASC proposed rule, we stated that an alternative to our proposal to set the threshold at two times the lower 48 AIR would be to set the threshold at the lower 48 AIR despite our concerns about excessive payment. We also stated that another alternative would be to set the threshold at 1.75 times the lower 48 AIR ($1,167.25 in CY 2024) to align it with the multiplier used to calculate the threshold triggering outlier payments under the OPPS. In the CY 2025 OPPS/ASC proposed rule, we invited comment on the alternatives of using the lower 48 AIR or 1.75 times the lower 48 AIR as the threshold amount for triggering the add-on payment for high-cost drugs.
Comment: Three commenters provided input on setting the threshold at the lower 48 AIR, agreeing with CMS that providing a separate payment for drugs whose costs only slightly exceed the lower 48 AIR could result in excessive payment because the facility would be receiving the lower 48 AIR payment plus the add-on payment for the drug.
Response: We thank commenters for their feedback.
Comment: Four commenters suggested that we set the threshold at 1.75 times the lower 48 AIR. Three of these commenters argued that the threshold be set at 1.75 times the lower 48 AIR because (1) doing so would align it with the multiplier used to calculate the threshold triggering outlier payments under the OPPS and (2) there were, based on the proposed rule data and example threshold of $1,334, three high-use drugs (Bendeka [Bendamustine HCL], Xembify [immune globulin] and Neulasta On-Pro [Pegfilgrastim]) that would not qualify for the add-on payment if the threshold was two times the lower 48 AIR but would qualify for the add-on payment if the threshold was 1.75 times the lower 48 AIR and one high-use drug (Vivitrol [Naltrexone]) that would qualify for the add-on payment if the threshold was two times the lower 48 AIR but was sufficiently close to the threshold that it could easily fall below the threshold and not qualify for separate payment in the future.
Response: We appreciate the commenters' preference for using 1.75 times the lower 48 AIR, but our view is that two times the lower 48 AIR is the most appropriate threshold to use as it would best ensure that only those drugs whose costs significantly exceed the amount of the annual lower 48 AIR payment would qualify for the add-on payment. It is also consistent with the two-times rule the OPPS uses to determine Ambulatory Payment Classification (APC) levels and the calculation of the diagnostic radiopharmaceutical packaging threshold that that we are finalizing in section II.A.3.c.(2) of this rule. With respect to the four high-use drugs referenced by the commenters, using final rule data, Bendeka and Xembify have per day costs that are well above $1,334 and thus qualify for the add-on payment under two times the lower 48 AIR threshold. Neulasta has a per day cost significantly below the lower 48 AIR of $667 and thus would not qualify for the add-on payment under any threshold that we are considering. Vivitrol has a per day cost that is slightly above $1,334 and therefore qualifies for the add-on payment under two times the lower 48 AIR threshold. While we acknowledge that this closeness to the threshold could cause Vivitrol to fluctuate from qualifying for separate payment to not qualifying for separate payment on an annual basis, there will always be drugs for which this is true when using any threshold-based payment criterion.
Comment: Two commenters suggested that we use non-AIR thresholds to calculate the list of drugs for which the add-on payment would be made. One of these commenters suggested that we use the standard drug packaging threshold of $140 to better recognize the costs for IHS and tribal facilities that furnish expensive drug treatment services to Native Americans with cancer. The other commenter, expressing concern that the proposed threshold was too high and might exclude some drugs that are clinically necessary and significantly costly for IHS and tribal hospitals, suggested that CMS establish a threshold based on CMS' specialty tier cost threshold criteria under Medicare Part D.[162]
Response: We appreciate the commenters' suggested alternatives to using a multiple of the AIR as the threshold; however, we do not think that these thresholds would achieve the objective of providing a separate add-on payment only for drugs that greatly exceed the lower 48 AIR. ( printed page 94283)
Comment: One commenter suggested that, regardless of which approach CMS selects to determine the high-cost threshold, that CMS “categorically apply separate payment” for biosimilar [163] products when administered in IHS and tribal facilities. The commenter observes that American Indians and Alaska Natives experience disparities in healthcare access and outcomes and argues that by ensuring that biosimilar products are financially accessible in IHS and tribal facilities, CMS would be taking a step toward reducing these disparities and promoting health equity. The commenter also stated that a “separate payment system” for biosimilar products could also incentivize their use over more expensive reference products, fostering a competitive market that may, in turn, lead to further reductions in prescription drug costs over time.
Response: It isn't entirely clear what the commenter means by “categorically apply separate payment” for biosimilar products or a “separate payment system” for biosimilar products, but it appears that the commenter is suggesting that all biosimilars be paid for separately when furnished by an IHS or tribal hospital outpatient department regardless of the payment threshold. We appreciate the commenter's suggestion; however, we do not think that paying separately for all biosimilars would achieve the objective of providing a separate add-on payment only for drugs and biologicals that greatly exceed the lower 48 AIR.
After consideration of the public comments we received on the proposed policy to define high-cost drugs as all drugs covered under Medicare Part B and for which payment would otherwise be made under the OPPS whose per day cost exceeds two times the lower 48 AIR ($1,334 in CY 2024), we are finalizing the policy as proposed.
We also sought comment in the CY 2025 OPPS/ASC proposed rule on whether we should adopt an exception to whichever AIR-based threshold we adopted that would parallel the drug packaging threshold exception for biosimilars under the OPPS. As we explained in the CY 2025 OPPS/ASC proposed rule, under the OPPS, if a drug's per-day cost is less than or equal to the drug packaging threshold, then payment for the drug is packaged. Conversely, if a drug's per-day cost exceeds the drug packaging threshold, then it is paid for separately (unless it is policy packaged). For a more detailed discussion of the drug packaging threshold, we referred readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81776 through 81778). In CY 2024, we established an exception to this threshold for biosimilars when the biosimilar's per-day cost does not exceed the threshold, but its reference product's per-day cost does. In other words, if the biosimilar's reference product is paid separately (because its per-day cost exceeds the threshold), then we also pay separately for the biosimilar even if its per-day cost does not exceed the threshold. This exception was based on our concern that packaging biosimilars when the reference product or other marketed biosimilars are separately paid might create financial incentives for providers to select more expensive, but clinically similar, products. For a more detailed discussion of the exception for biosimilars to the drug packaging threshold, we referred readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81783 through 81786). Because we proposed to use a threshold to trigger application of the add-on payment for high-cost drugs to IHS and tribal hospitals, we stated that we had the same concerns about financial incentives that motivated us to establish the exception for biosimilars to the drug packaging threshold. Consequently, in the CY 2025 OPPS/ASC proposed rule, we sought comment on whether we should pay the add-on payment to IHS and tribal hospitals for biosimilars whose per-day costs do not exceed the threshold but whose reference products per-day costs do exceed the threshold.
Comment: We received several comments on whether we should pay the add-on payment to IHS and tribal hospitals for biosimilars whose per-day costs do not exceed the threshold but whose reference products do exceed the threshold. All commenters supported doing so, indicating that they shared CMS' concerns that packaging biosimilars when the reference product is separately paid might create financial incentives for providers to select more expensive but clinically similar products.
Response: We thank the commenters for their feedback and, based on this feedback, we will adopt a drug packaging threshold exception for biosimilars in which we will pay the add-on payment to IHS and tribal hospitals for biosimilars whose per-day costs do not exceed the threshold of two times the lower 48 AIR but whose reference products do exceed the threshold.
In the CY 2025 OPPS/ASC proposed rule, we also proposed that the amount of the add-on payment for a high-cost drug would be the average sales price (ASP) for the drug with no additional payment ( i.e., ASP). We explained that this payment amount would be consistent with what hospitals receive as payment for most drugs under the OPPS (ASP plus 6 percent) but would exclude the 6 percent additional payment in recognition of the fact that IHS and tribal facilities, unlike hospitals paid under the OPPS, primarily obtain their drugs through the FSS, whose rates are significantly lower than ASP. We also emphasized that this approach would be consistent with our existing policies of paying ASP without any additional payment for certain Opioid Treatment Program drugs under 42 CFR 410.67(d)(2)(i)(A) and (B). In the event ASP pricing information was not available for a particular drug, we proposed to pay the Wholesale Acquisition Cost (WAC) plus 0 percent and if WAC pricing information was not available, we proposed to pay 89.6 percent of Average Wholesale Price (AWP).
In the CY 2025 OPPS/ASC proposed rule, we sought comment on an alternative policy of paying ASP plus 6 percent instead of ASP plus 0 percent. We also proposed that if we were to adopt this alternative policy and pricing information was not available for a particular drug, we would pay WAC plus 6 percent and if WAC pricing information was not available, we would pay 95 percent of AWP. We sought comment on our proposal to pay an add-on of ASP plus 0 percent in addition to the lower 48 AIR for drugs administered by IHS and tribal facilities with costs that exceed two times the lower 48 AIR. We also sought comment on our proposed pricing hierarchy for drugs for which ASP pricing information was not available.
Comment: Three tribal commenters supported paying ASP with no additional payment. In their view this would adequately compensate IHS and tribal hospital outpatient departments for their costs because the facilities would be receiving both the add-on payment for the drug and the lower 48 AIR encounter payment.
Response: We thank commenters for their support.
Comment: Multiple commenters suggested that the payment amount for the add-on payment should be ASP plus 6 percent. One of these commenters additionally requested that CMS pay ASP plus 8 percent for certain ( printed page 94284) qualifying biosimilar products. Many of these commenters argued that paying ASP plus 6 percent is necessary to ensure access to high-cost drugs because only ASP plus 6 percent adequately reflects the true acquisition and administration costs of high-cost drugs. Some of these commenters, while acknowledging that IHS and tribal facilities primarily obtain their drugs through the FSS, argued that not all IHS and tribal facilities might have access to the FSS and that not all drugs are necessarily available through the FSS. One commenter expressed concern that providers might perceive reimbursement without the 6 percent add-on as a “penalty” for furnishing higher-cost drugs in these settings. Finally, some of these commenters argued that CMS should pay ASP plus 6 percent for consistency with how separately payable Part B drugs are reimbursed in the OPPS setting.
Response: We thank the commenters for their input and share their concerns that the payment amount for separately paid for drugs reflect the acquisition and administration costs of those drugs and that IHS and tribal hospital outpatient departments be paid consistently with how outpatient hospital departments are paid for drugs under the OPPS. However, it is our view that paying ASP plus 0 percent would achieve these goals. We believe that ASP with no add-on payment captures the acquisition and administration costs of the drugs given that (1) IHS and tribal facilities acquire most of their drugs through the FSS at rates significantly lower than ASP plus 6 percent; and (2) IHS and tribal facilities will continue to receive the lower 48 AIR payment for the encounter in addition to the add-on payment for the drug. Given these two factors, we believe that an add-on payment equal to ASP is sufficient.
With respect to paying IHS and tribal hospital outpatient departments consistently with how we pay hospital outpatient departments under the OPPS, our view is that paying IHS and tribal hospital outpatient departments ASP with no additional add-on is consistent with how we pay hospital outpatient departments under the OPPS given the differences in how hospitals under the two payment systems primarily acquire their drugs. As we stated in our proposed rule, our rationale for paying ASP was to pay IHS and tribal hospitals the same rate we pay hospital outpatient departments under the OPPS for separately payable drugs, while excluding the 6 percent add-on in recognition of the fact that IHS and tribal hospitals, unlike hospitals under the OPPS, primarily acquire their drugs through the FSS at a significantly lower rate than hospitals under the OPPS.
Comment: Three commenters expressed support for our proposed pricing hierarchy for drugs for which ASP pricing information is not available. These commenters indicated that there may be many times when new drugs are available for which an ASP has not yet been set and the proposed hierarchy would allow IHS and tribal facilities to be reimbursed for those high-cost drugs at these other rates until an ASP is set. Without such a hierarchy in place, these commenters warned, many new drugs would not be eligible for the add-on payment.
Response: We thank commenters for their support.
After consideration of the public comments we received on the proposed policy to set the amount of the add-on payment for a high-cost drug at ASP with no additional payment (ASP plus zero percent) and the proposed pricing hierarchy for drugs for which ASP pricing information was not available, we are finalizing the policies as proposed.
To implement this policy, in the CY 2025 OPPS/ASC proposed rule we proposed that, starting with IHS's annual announcement in the Federal Register in December 2024 of the lower 48 AIR amount for CY 2025, we would multiply the lower 48 AIR amount for the payment year by two and then compare the result to the estimated per day costs of all drugs covered under Part B for which payment would otherwise be made under the OPPS. To determine the calculated per day cost for each drug and biological HCPCS code, we proposed to follow a methodology similar to our longstanding methodology used to calculate the per day cost of drugs and biologicals for OPPS payment purposes as discussed in section V.B.1.b of this final rule with comment period. Specifically, to calculate the per day cost, we proposed to use an estimated payment rate based on the ASP methodology payment rate, which for purposes of the proposal was generally ASP plus 0 percent (which is the payment rate we proposed for separately payable IHS drugs and biologicals) for CY 2025. We used the manufacturer-submitted ASP data from the fourth quarter of CY 2023 to determine the proposed rule per day cost. For drugs and biologicals that did not have either an ASP-based payment rate or a payment rate based on WAC, we used mean unit cost of the items derived from the CY 2023 hospital claims data to determine their per day cost.
In the CY 2025 OPPS/ASC proposed rule, we proposed that a list of drugs whose costs exceeded two times the lower 48 AIR would be generated using the above-described methodology and then communicated to IHS and tribal hospitals prior to January 1, 2025. During CY 2025, IHS and tribal hospitals would submit claims for drugs included on this list. The list of drugs would be updated on a quarterly basis using existing drug compendia and CMS ASP quarterly reporting only to account for newly introduced drugs. The payment rates for drugs on the list would be updated quarterly as well based on changes in drug prices. We proposed to then repeat this process on an annual basis each December when the lower 48 AIR amount for the following calendar year was announced by IHS. As an example, we included in our proposed rule a list of the drugs for which the add-on payment would have been made (drugs with a per day cost exceeding two times the CY 2024 lower 48 AIR) had our proposed policy been in place for CY 2024. We sought comment on our proposed implementation plan.
Comment: We received three comments specifically supporting our proposed implementation plan with one requested alteration. These commenters requested that when we annually create the list of drugs that qualify for the add-on payment that we use the lower 48 AIR in effect at the time of the release of the CY OPPS/ASC final rule with comment period to calculate the threshold for creating the list rather than waiting for IHS's release of the lower 48 AIR for the payment year (typically released in December). For example, to create the drug list for CY 2025, CMS would use the lower 48 AIR for 2024 rather than waiting for the release of the lower 48 AIR for 2025. These commenters indicated that while IHS has issued the lower 48 AIR in December in recent years, that has not always been the case and that in some years the lower 48 AIR was not released until months into the payment year. If that were to occur in future years, the commenters were concerned that it would delay the release of the drug list until after January 1 of the payment year. Accordingly, the commenters suggested that CMS use the lower 48 AIR in effect at the time the final OPPS rule is released and then update the list in the first quarterly update during the payment year using the new lower 48 AIR amount once it has been released by IHS.
Response: We share the commenters' concern about the release of the list being delayed beyond January 1 of the ( printed page 94285) payment year. Adopting the commenters' suggestion would allow the final list of covered drugs to be included in the OPPS/ASC final rule which must be released by November 1 of each year. In addition to ensuring that the list would not be delayed beyond January 1 of the payment year, this would also have the advantage of avoiding the operational complexity of having to finalize and release a covered drug list separately from the final OPPS rule. It would also give CMS more time to communicate the drug list to IHS and tribal hospitals and would provide those hospitals with more time to familiarize themselves with the list. With respect to the commenter's suggestion that we update the list in the next quarterly update using the new lower 48 AIR once it's released by IHS, we should explain that, as we stated in the proposed rule, the quarterly updates consist solely of adding newly introduced drugs to the list and updating the ASP for listed drugs. In other words, we will only use the lower 48 AIR once, annually, to establish the list of qualifying drugs for the payment year and we will not update that list again using the AIR during the payment year. The only quarterly updates will be to add new-to-market drugs with per-day costs that exceed two times the lower 48 AIR and to update qualifying drugs' ASPs, not to add to/change the list of qualifying drugs.
For both operational simplicity and to ensure that the list of separately paid drugs is released prior to January 1 of each year, we are finalizing using the lower 48 AIR in effect at the time of the release of the CY OPPS/ASC final rule with comment period to annually create the list of drugs qualifying for the add-on payment for the following calendar year.
Comment: Two commenters commented on CMS' proposed process to create the list of drugs qualifying for the add-on payment. One suggested that CMS clearly communicate to IHS facilities the final process to develop the list and the results of the quarterly and annual updates to that list. The other recommended that CMS maintain a transparent process that would allow manufacturers to validate the results and establish a process whereby manufacturers could seek to correct any identified errors.
Response: We agree with the commenters on the importance of employing a transparent process to develop the list of high-cost drugs for which separate payment will be made and clearly communicating that process and its results to IHS and tribal facilities on an annual and quarterly basis. The process we are using to create the list of drugs qualifying for the add-on payment is described in detail earlier in this section. The results of that process are contained in Addendum Q to this final rule, which is the list of drugs qualifying for the add-on payment for CY 2025. Quarterly updates to the list will be posted on the CMS website [164] as they occur during CY 2025. Additionally, links to Addendum Q and the quarterly updates to the list during the calendar year will be posted on the IHS website [165] and IHS will be sending “Dear Tribal Leader Letters” [166] to tribal officials informing them of the new policy and where the list can be found. With respect to allowing for the validation of the results of the process, we direct commenters to the cost statistics files [167] which can be used to validate the results of the per day cost calculations.
Comment: Another commenter encouraged CMS to monitor whether adoption of the separate payment policy will adequately facilitate equitable patient access to drugs administered in hospital outpatient facilities in tribal communities. The commenter also suggested that CMS consider reporting on access to drugs within American Indian/Alaskan Native and other racial and ethnic communities within Health and Human Services Dashboards, such as those produced by the IHS.
Response: Given the health equity and beneficiary access concerns motivating our separate payment policy, we agree with the commenter on the importance of monitoring the effects of the separate payment policy on equitable patient access to drugs administered in hospital outpatient facilities in tribal communities and we plan on assessing the impact of our separate payment policy following its implementation. We will take the commenter's suggestion to report the outcome of such monitoring within Health and Human Services Dashboards under consideration for future rulemaking.
Comment: We received a comment supporting the policy and requesting that we consider expanding the scope of the add-on payments to include other high-cost services beyond drugs, such as radiology services.
Response: We appreciate the commenter's suggestion to expand the scope of the add-on payments to include other high-cost services beyond drugs and may consider this in future rulemaking.
Comment: We received two comments supporting the policy and requesting that it be expanded to include Urban Indian Organization (UIOs) clinics. These commenters emphasized that UIOs are severely underfunded and the same equity and access concerns that support providing IHS and tribal facilities with the proposed AIR add-on payment for high-cost drugs are also relevant to the needs of UIOs.
Response: We appreciate the commenters' suggestion; however, UIO clinics are not paid the outpatient AIR for hospital outpatient services furnished to Medicare beneficiaries and therefore are outside of the scope of the AIR add-on payment policy that we are finalizing in this rule.
Finally, we proposed to implement this policy on a permanent basis but may revisit it in the future if we have any concerns about its impact once it has been implemented.
Comment: We received two comments supporting our proposal to implement the policy on a permanent basis.
Response: We thank the commenters for their input.
After consideration of the public comments we received on our proposed plan to implement the separate payment policy, we are finalizing the policy as proposed except we will use the lower 48 AIR in effect at the time of the release of the CY OPPS/ASC final rule with comment period (rather than the lower 48 AIR for the payment year) to annually create the list of drugs qualifying for the add-on payment for the following calendar year.
As discussed in the preceding sections, after consideration of the public comments we received, we are finalizing the policy to separately pay IHS and tribal hospitals for high-cost drugs furnished in hospital outpatient departments as proposed with modifications to (1) use the lower 48 AIR amount in effect at the time of the release of the CY OPPS/ASC final rule with comment period to calculate the threshold for the list of drugs qualifying for the add-on payment for the following calendar year and (2) adopt a drug packaging threshold exception for biosimilars that parallels the drug packaging threshold exception for biosimilars under the OPPS. ( printed page 94286)
D. Request for Information—Paying All IHS and Tribally Operated Clinics the IHS Medicare Outpatient All Inclusive Rate (AIR)
CMS established a Tribal Technical Advisory Group (TTAG) in 2004 to provide advice and input to CMS on policy and program issues impacting American Indians and Alaska Natives (AI/AN) populations served by CMS programs. Although not a substitute for formal consultation with tribal leaders, the TTAG enhances the government-to-government relationship between HHS and federally recognized tribes and improves understanding between CMS and tribes. The TTAG has subject specific subcommittees that meet on a regular basis in order to be more effective and perform in-depth analysis of Medicare, Medicaid, Children's Health Insurance Program (CHIP), and Health Insurance Marketplace® [168] policies that may have tribal implications. The TTAG is composed of 17 representatives. It has historically included an elected tribal leader or an appointed representative from each of the 12 geographic areas of the Indian Health Service (IHS) delivery system and a representative from each of the national Indian organizations headquartered in Washington, DC—the National Indian Health Board, the National Congress of American Indians, and the Tribal Self-Governance Advisory Group. Section 5006(e)(1) of the American Recovery and Reinvestment Act of 2009, which became effective July 1, 2009, mandates that TTAG shall be maintained within CMS and added two new representatives: a representative from a national urban Indian health organization; and a representative from the IHS.
In June 2020, the TTAG suggested that CMS amend its Medicare regulations to make all IHS and tribally operated outpatient facilities eligible for Medicare payment at the IHS Medicare outpatient per visit rate/AIR. The TTAG explained that outpatient clinics, which are otherwise similar to grandfathered tribal Federally Qualified Health Centers (FQHCs), are paid at different rates depending upon whether they meet the requirements as a provider-based facility, a grandfathered tribal FQHC, a non-grandfathered tribal FQHC, or none of the above. TTAG's position is that the rates vary based on Medicare regulatory definitions, rather than the actual costs of the outpatient clinic.[169] There are varying payment differentials among Medicare enrolled providers and suppliers under the authorities of the Social Security Administration (SSA). For example, Ambulatory Surgical Centers (ASCs) are paid differently than Hospital Outpatient Departments (HOPDs); which are paid differently depending on whether they are located in a critical access hospital.
In the CY 2022 PFS proposed rule (86 FR 39240), we acknowledged that the TTAG is concerned about ensuring appropriate Medicare payments for similar services and is also concerned about the impact on tribal Medicare beneficiaries and on ensuring equitable access to healthcare. We take these concerns seriously but noted in the CY 2022 PFS proposed rule that we had insufficient information to evaluate the costs and benefits of potential changes to these policies. Therefore, we solicited comments on the TTAG's request for CMS to amend its Medicare regulations to make all IHS- and tribally operated outpatient facilities/clinics eligible for payment at the Medicare outpatient per visit rate/AIR, regardless of whether they were owned, operated, or leased by IHS. In response to the solicitation, we did not receive specific information on costs or specific types of clinics; however, in the CY 2022 PFS final rule (86 FR 65211 through 65214), we stated we would like to continue these discussions to evaluate the impact of the commenters' proposed changes to the current Medicare payment policies and will consider these recommendations for future rulemaking. Thus, to continue these discussions, we requested this information again in the CY 2025 proposed rule (89 FR 59394).
Additionally, beginning in the Fall of 2023, CMS began participating in a workgroup related to the TTAG's Medicare priority to make the IHS Medicare outpatient AIR available to all IHS and tribally operated outpatient facilities that request it. Although we have received some information through the workgroup, we requested additional information consistent with our previous comment solicitation in the CY 2022 PFS proposed rule (86 FR 39238).
We requested information on the kinds of and number of facilities or clinics to which the Medicare outpatient IHS AIR could apply; It was unclear whether TTAG anticipated that these facilities enroll in Medicare as FQHCs going forward, or whether they are referring to FQHCs that are currently paid under the FQHC Prospective Payment System (PPS). Moreover, we requested information on whether the facilities in question were freestanding or provider-based. We asked commenters to confirm or clarify whether the clinics are physician offices, or whether they are recommending establishment of a new provider type. We solicited information regarding the relative operating costs of tribally operated outpatient clinics, as well as feedback and supporting evidence to address whether or why payment set at the IHS AIR would be more appropriate than payment rates under the FQHC PPS, the physician fee schedule, or other some other Medicare payment system. Further, we solicited comment on how the Medicare outpatient AIR, which is based upon a limited number of hospital cost reports, related to costs in tribal clinics and the kinds of services that the clinics furnish. Finally, we solicited comment on the concerns that the AI/AN community may have regarding access to or inequity of care in situations where a payment differential exists (86 FR 39240).
We noted in the CY 2025 RFI that we have information on historically excepted FQHCs, the outpatient provider-based clinics to the hospital, and some general information about the composition of IHS and tribal facilities and clinics. However, we stated there are still gaps in the data and therefore we requested responses to the following questions: If the clinic or facilities in question are not enrolled in Medicare as an FQHC or provider-based to a hospital, are they physician practices? How are these facilities organized and related? (89 FR 59394.)
Because paying the Medicare outpatient AIR to additional IHS and tribally operated facilities that are currently paid under another Medicare payment methodology or not yet enrolled in Medicare as all would potentially increase expenditures, we also solicited information on how tribally operated facilities participate in Medicare currently, which would assist us with estimating the impacts of such a policy change.
We received a few comments in response to the information we requested. Below, we provide a summary of the comments we received and our responses.
Comment: Commenters support CMS' continuing efforts to better understand the request to make all IHS and tribally operated outpatient facilities eligible for Medicare payment at the IHS Medicare outpatient per visit rate/AIR as it would provide financial stability and improve ( printed page 94287) comprehensive care delivery for tribal communities. Some of the commenters believe the current Medicare payment rates for outpatient Indian health providers are inequitable, increasingly illogical and contrary to National Indian policies, as the requested change would increase Medicare expenditures only a negligible amount. One commenter believes the most efficient, expeditious, reliable and appropriate way for CMS to gather the requested information is through the TTAG, not a Request for Information (RFI), but they provided information on the known or likely enrollment status of potentially affected programs, discussed why many small Indian Health Programs currently bill Medicare (if at all) under the Physician Fee Schedule (PFS), explained why neither the PFS nor the FQHC PPS rates come close to covering their costs, discussed why the AIR is the most appropriate payment option for all Outpatient Indian Programs, and provided some illustrative examples. Another commenter would like CMS to consider using the IHS Medicare outpatient per visit rate/AIR to pay for hospital outpatient services furnished by Urban Indian Organizations (UIOs) to Medicare beneficiaries.
Response: CMS acknowledges comments received on this important issue, and, commits to continuing close collaboration with TTAG to evaluate IHS and Tribally Operated Clinics without IHS Medicare outpatient AIR payments to identify opportunities for improving financial stability and equitable healthcare delivery in these communities, including determining whether these clinics would receive a net benefit from being paid the IHS Medicare Outpatient AIR.
D. Coverage Changes for Colorectal Cancer (CRC) Screening Services
Medicare coverage for colorectal cancer (CRC) screening tests under Part B is described in statutes (sections 1861(s)(2)(R), 1861(pp), 1862(a)(1)(H) and 1834(d) of the Social Security Act (the Act)), regulation (42 CFR 410.37), and a National Coverage Determination (NCD) (Section 210.3 of the Medicare National Coverage Determinations Manual). Section 1861(pp)(1)(D) of the Act includes in its definition of colorectal cancer screening test “[s]uch other tests or procedures, and modifications to the tests and procedures described under this subsection, with such frequency and payment limits, as the Secretary determines appropriate, in consultation with appropriate organizations.”
The regulations at 42 CFR 410.37 list and define the tests and procedures covered by Medicare as colorectal cancer screening tests. Specifically, the following tests and procedures furnished to an individual for the purpose of early detection of colorectal cancer are covered by Medicare:
- Screening fecal-occult blood tests.
- Screening flexible sigmoidoscopies.
- Screening colonoscopies, including anesthesia furnished in conjunction with the service.
- Screening barium enemas.
- Other tests or procedures established by a national coverage determination, and modifications to tests under this paragraph, with such frequency and payment limits as CMS determines appropriate, in consultation with appropriate organizations.
In recent years we have received recommendations from the public to remove Medicare coverage for the barium enema test since the test no longer meets modern clinical standards and is no longer recommended in clinical guidelines. As a replacement to the barium enema test, organizations have suggested the use of CT colonography, which is a more effective test for colorectal cancer screening. For a more extensive discussion on the background and proposal to revise the Medicare coverage for colorectal cancer screening services, we refer readers to the CY 2025 Physician Fee Schedule (PFS) proposed rule.
For CY 2025, based on public input and consultation with specialty societies, and as discussed in the CY 2025 PFS proposed rule, we proposed to exercise our authority under section 1861(pp)(1)(D) of the Act to update and expand coverage for CRC screening. As discussed in the CY 2025 PFS proposed rule, we proposed to make the following revisions to § 410.37:
- Remove coverage for the barium enema procedure.
- Add coverage for the computed tomography colonography (CTC) procedure.
- Expand the existing definition of a “complete colorectal cancer screening” to include a follow-on screening colonoscopy after a Medicare covered blood-based biomarker CRC screening test (described and authorized in NCD 210.3).
The above screening tests are currently described by existing HCPCS codes. These HCPCS codes are listed in Table 139 along with their long descriptors.
( printed page 94288)Based on the proposed coverage changes for CRC screening, we proposed to make the following changes under the OPPS for CY 2025:
- HCPCS codes G0106 and G0120 (screening barium enema): These codes were established by CMS effective January 1, 1998, to implement Medicare coverage for barium enema as a test for colorectal cancer screening. Since we proposed to remove Medicare coverage for barium enema colorectal cancer screening effective January 1, 2025, and we no longer need to keep these codes active, we proposed to delete them on December 31, 2024. Therefore, we proposed to revise the status indicator for the HCPCS codes from status indicator “S” (Procedure or Service, Not Discounted When Multiple. Paid under OPPS; separate APC payment.) to “D” (Discontinued code) to indicate that HCPCS codes G0106 and G0120 will be deleted on December 31, 2024. In addition to the deletion of these codes, we also proposed to delete HCPCS code G0122 (Colorectal cancer screening; barium enema), which is already non-covered by Medicare, on December 31, 2024.
- CPT code 74263 (screening computed tomography colonography (CTC)/virtual colonoscopy): We proposed to reassign this code from status indicator “E1” (not covered/not payable) to status indicator “S” and APC 5522 (Level 2 Imaging Without Contrast) to indicate that the code is separately payable. Based on our review, we noted that we believed the time and resources associated with performing a screening virtual colonoscopy was similar to a diagnostic virtual colonoscopy, which is described by CPT code 74261 (Computed tomographic (ct) colonography, diagnostic, including image postprocessing; without contrast material). Consequently, the proposed APC assignment for CPT code 74263 was based on its clinical and resource homogeneity to CPT code 74261, which was assigned to APC 5522.
- HCPCS code G0327 (screening blood-based biomarker): This HCPCS code is currently assigned to status indicator “A” to indicate that the test is paid separately under a different Medicare payment system than the OPPS. Since HCPCS G0327 is currently separately payable under the Clinical Laboratory Fee Schedule (CLFS), we did not propose to revise the status indicator. Specifically, with the expanded coverage to include blood-based biomarker as a screening test to detect colorectal cancer, we proposed to continue to assign HCPCS code G0327 to status indicator “A” for CY 2025.
In summary, based on the proposed coverage changes for colorectal cancer screening services, we proposed to revise the OPPS status indicator for certain HCPCS codes for CY 2025. Table 140 shows the long descriptors, current CY 2024 OPPS status indicators, and proposed CY 2025 OPPS status indicators for HCPCS codes G0106, G0120, G0122, 74263, and G0327. The proposed CY 2025 OPPS payment rates, where applicable, for these HCPCS codes can be found in Addendum B to the proposed rule. In addition, for the complete list of the proposed status indicators and their definitions, refer to Addendum D1 of the CY 2025 OPPS/ASC proposed rule.
( printed page 94289)Comment: Overall, commenters supported our proposals of the following Colorectal Cancer (CRC) Screening Services changes:
- Deleting HCPCS codes G0106 and G0120 (screening barium enema) effective December 31, 2024, and deleting HCPCS code G0122 (Colorectal cancer screening; barium enema), which is already non-covered by Medicare, on December 31, 2024.
- Reassigning the status indicator of CPT code 74263 (screening computed tomography colonography (CTC)/virtual colonoscopy) from status indicator “E1” (not covered/not payable) to status indicator “S” to indicate that the code is separately payable and assigning CPT code 74263 to APC 5522 (Level 2 Imaging Without Contrast).
- Maintaining the status indicator of HCPCS code G0327 (screening blood-based biomarker) as status indicator “A” to indicate that the test is paid separately under a different Medicare payment system than the OPPS.
Response: We thank commenters for supporting our proposals related to § 410.37 and NCD 210.3 on Colorectal Cancer (CRC) Screening Services.
Comment: A commenter requested that CMS exercise our authority in section 1861(pp)(1)(D) of the Act to expand our approach to a “complete CRC screening” to add CTC along with the Medicare covered blood-based biomarker CRC screening test and the Medicare covered non-invasive stool-based CRC screening test within the definition of a “complete colorectal cancer screening.”
Response: We disagree with commenters that requested a further expansion of a complete colorectal cancer screening to include CTC. CTC is a visualization procedure along with colonoscopy and flexible sigmoidoscopy whereas stool-based and blood-based CRC screening tests are non-visualization tests. CTC provides visualization of the contours of the whole colon and demonstrates mucosal surface abnormalities consistent with polyps and tumors. CTC tests are unlike noninvasive modalities such as stool-based and blood-based CRC screening, which present a binary positive/negative result with variable specificity and may result (in the case of a positive test) in the need for a visualization study to confirm the derived suspicion of adenoma or cancer. The follow-on colonoscopy in cases of stool-based or blood-based CRC screenings represents an extension of screening in a patient who has converted from average risk to increased risk as a result of the positive test. In the case of CTC, however, visualization of the colonic mucosal contour, as well as the remainder of the colonic wall and surrounding structures, has already been achieved and the determination of a suspicious finding has already been made. Polyps over the size threshold prompt a referral for diagnostic/therapeutic colonoscopy for the purpose of polypectomy. A follow-up colonoscopy after an abnormal finding from a CTC would be considered a diagnostic colonoscopy to biopsy or remove visualized polyps and/or cancer.
After consideration of the public comments we received, we are finalizing the proposals made in the CY 2025 OPPS/ASC proposed rule affecting Colorectal Cancer (CRC) Screening Services with one modification. Specifically, we are finalizing the deletion of HCPCS codes G0106 and G0120 (screening barium enema) effective December 31, 2024. We are finalizing the reassignment of CPT code 74263's status indicator as proposed from status indicator “E1” (not covered/not payable) to “S” to indicate that the code is separately payable. However, we are reassigning CPT code 74263 to APC 5523 (Level 3 Imaging Without Contrast) in this final rule. We refer readers to section III.E of this final rule with comment period for a detailed discussion of CPT code 74263's APC assignment. We are maintaining the status indicator of HCPCS code G0327 (screening blood-based biomarker) as status indicator “A” to indicate that the test is paid separately under a different Medicare payment system than the OPPS.
Table 141 shows the long descriptors proposed and final CY 2025 OPPS status indicators for HCPCS codes G0106, G0120, G0122, 74263, and G0327. The finalize CY 2025 OPPS payment rates, where applicable, for these HCPCS codes can be found in Addendum B to ( printed page 94290) this final rule. In addition, for the complete list of the final status indicators and their definitions, refer to Addendum D1 of this final rule with comment period.
E. Request for Comment on Payment Adjustments Under the IPPS and OPPS for Domestic Personal Protective Equipment
1. General Background
As discussed in the FY 2023 IPPS/LTCH PPS and CY 2023 OPPS/ASC rules, President Biden issued Executive Order (E.O.) 13987, “Organizing and Mobilizing the United States Government To Provide a Unified and Effective Response To Combat COVID-19 and To Provide United States Leadership on Global Health and Security,” on January 20, 2021 (86 FR 7019). This order launched a whole-of-government effort to combat the coronavirus disease 2019 (COVID-19) and prepare for future biological and pandemic threats. As the COVID-19 pandemic eased, work has continued to prepare for future pandemics. As the COVID-19 pandemic demonstrated, sufficient availability of personal protective equipment (PPE) in the health care sector is a critical component of preparedness.
The CY 2023 OPPS/ASC final rule with comment period implemented payment adjustments under the OPPS and IPPS to support a resilient and reliable supply of surgical N95 respirators—a specific type of filtering facepiece respirator that is a subset of N95 masks used in some clinical settings under conditions requiring respiratory protection from airborne pathogens and splash protection from exposure to fluids. Early on in the COVID-19 pandemic, “just-in-time” supply chains, minimal stockpiling, and overreliance on foreign imports left U.S. hospitals unable to obtain enough N95 respirators to protect health care workers. Prices for surgical N95s soared from an estimated $0.25-$0.40/unit to $5.75/unit (and up to $12.00/unit in some reported cases). Unable to obtain surgical N95s regulated by NIOSH, hospitals had to turn to KN95s—a Chinese standard respirator—and other non-NIOSH-approved respirators under Emergency Use Authorization (EUA). Skyrocketing demand during the COVID-19 pandemic also raised counterfeit respirator concerns.
Currently available payment adjustments offset the marginal costs that hospitals face in procuring domestically made NIOSH-approved and FDA-certified surgical N95 respirators. These marginal costs are due to higher per-unit acquisition prices that stem from higher costs of inputs and labor in the U.S., as compared to international suppliers, where many N95 and other respirators are made, as well as a demonstrated record of more consistent high quality for domestically made products.[170] These payment adjustments offset the additional marginal costs of hospitals that purchase domestically made NIOSH-approved surgical N95 respirators to help sustain demand for—and thus domestic production of—high-quality domestically made respirators in order to ensure quality PPE is available to health care personnel when needed.
The policy goal to maintain a baseline domestic production capacity of high-quality PPE in order to ensure that quality PPE is readily available to health care personnel when needed is emphasized in the National Strategy for a Resilient Public Health Supply Chain, published in July 2021 as directed by President Biden's Executive Order 14001 on “A Sustainable Public Health Supply Chain.” The U.S. Government has committed to purchase wholly domestically-made PPE in line with section 70953 of the Infrastructure Investment and Jobs Act (Pub. L. 117-58). ( printed page 94291)
2. Potential Modifications to Payment Adjustments for Domestic NIOSH-Approved Surgical N95 Respirators
Although the payment adjustments for domestic NIOSH-approved surgical N95 respirators under the OPPS and IPPS have applied to cost reporting periods beginning on or after January 1, 2023, use of the payment adjustments has been limited. Furthermore, market data suggests that a majority of surgical N95 respirators purchased by hospitals are not wholly domestically made. In the CY 2023 OPPS/ASC final rule with comment period, we stated that as we gain more experience with this policy and the data collected, we may also consider modifications to the reasonable cost-based payment approach we were finalizing. HHS has conducted stakeholder outreach to better understand barriers to awareness and uptake and seek feedback on potential modifications that could increase effectiveness, and continues to engage hospitals and other manufacturers on these payment adjustments. Furthermore, as discussed in more detail below, in the CY 2025 OPPS/ASC proposed rule (89 FR 59396 through 59399), we solicited feedback and comments on potential modifications to the payment adjustment in order to reduce reporting burden and achieve the policy goal to maintain a baseline domestic production capacity of PPE in order to ensure that quality PPE is readily available to health care personnel when needed.
Payment adjustment methodology: In the CY 2023 OPPS/ASC final rule with comment period, we finalized to initially base the payment adjustments on the IPPS and OPPS shares of the estimated difference in the reasonable costs. We created a new supplemental cost reporting form to enable calculation of a hospital-specific unit cost differential between domestic and non-domestic NIOSH-approved surgical N95 respirators. We noted that, based on available data, our best estimate of the difference in the average unit cost of domestic and non-domestic NIOSH-approved surgical N95 respirators was $0.20. In the CY 2023 OPPS/ASC final rule with comment period, we also noted that MedPAC, while not supportive of the proposed payment adjustments, stated that CMS should set the unit cost differential between domestic and non-domestic NIOSH-approved surgical N95 respirators at a national level (rather than on a hospital-by-hospital basis). MedPAC believed this would reduce the administrative burden on hospitals, encourage hospitals to purchase the most economical domestically made product, and reduce the ability of hospitals to increase their payments by artificially inflating reported N95 costs. In the CY 2025 OPPS/ASC proposed rule, we solicited comment on the following questions:
- Should we consider modifying the payment adjustment methodology calculation to provide a national standard unit cost differential between domestic and non-domestic NIOSH-approved surgical N95 respirators (rather than on a hospital-by-hospital basis)?
- If so, how should we calculate that standard unit cost differential between domestic and non-domestic NIOSH-approved surgical N95 respirators, and what should the current unit cost differential be?
- If we modified the payment adjustment methodology calculation to provide a national standard unit cost differential, would it be appropriate to calculate the payment adjustment by multiplying the unit cost differential by the total quantity of domestic NIOSH-approved surgical N95 respirators used by the hospital, and then multiplying by the Medicare Part A hospital inpatient cost share (to calculate the IPPS payment adjustment) or the Medicare Part B hospital outpatient cost share (to calculate the OPPS payment adjustment)?
- Do hospitals need additional support to purchase domestic-made surgical N95 respirators as opposed to non-domestic surgical N95 respirators? If so, how much support is needed, and in what form?
Comment: We received several comments supporting a modification to the payment adjustment methodology calculation that would provide a national standard unit cost differential between domestic and non-domestic NIOSH-approved surgical N95 respirators. Several commenters emphasized that this modification would minimize reporting burden for hospitals and ensure payments to hospitals are equitable. One commenter stated that CMS should provide hospitals the option of calculating their payment adjustment utilizing a national standard unit cost differential or a hospital-specific unit cost differential. A commenter stated that the national differential should be set at $0.50. Other commenters encouraged CMS to collaborate with external partners and use external resources to determine the national standard unit cost differential. Commenters suggested regularly collaborating with reliable benchmarking services, distributors, and group purchasing organizations to establish and recalibrate, if necessary, an accurate national standard unit cost differential.
Several commenters provided suggestions on additional support for hospitals to purchase domestic-made surgical N95 respirators as opposed to non-domestic surgical N95 respirators. Several commenters requested that CMS work with Congress to give CMS authority to apply this payment policy in a non-budget neutral manner under the OPPS. Other commenters requested that CMS work with Congress to give CMS authority to offset all the marginal costs incurred by the hospital in procuring domestically made surgical N95 respirators rather than just the Medicare-share of these costs. One commenter stated that CMS should pay the full cost of procuring domestically made surgical N95 respirators rather than the marginal costs of procuring domestically made surgical N95 respirators over non-domestically made surgical N95 respirators. A couple of commenters stated that CMS should reimburse hospitals 5 times the differential between the acquisition cost of domestic surgical N95 respirators and non-domestic surgical N95 respirators. One commenter stated that CMS should create a HCPCS code for surgical N95 respirators and develop a formula that reimburses hospitals that purchase domestic surgical N95s directly from manufacturers at a locked price. A few commenters encouraged CMS to increase education and communication of the payment adjustment to hospital purchasing decision-makers within the healthcare system.
Some comments addressed the issue of why use of the payment adjustments has been limited. A few commenters supportive of the payment adjustment stated that they believe providers may not be taking advantage of the adjustment because they still have stockpiles of surgical N95 respirators available to them that were purchased during the COVID-19 pandemic. A commenter stated its belief that use of the payment adjustments has been limited because the current supply and production capacity of wholly domestically made surgical N95 respirators is insufficient. One commenter stated that the payment adjustment does not result in a significant decrease in cost for providers. One commenter expressed that hospitals primarily buy surgical N95 respirators through distributors and that these distributors are not incentivized to purchase and make available to hospitals more expensive domestically produced surgical N95 respirators. ( printed page 94292)
One commenter indicated that hospitals would be more inclined to purchase domestically made surgical N95 respirators if more information was made available regarding the production capabilities of domestic suppliers. The commenter stated it is crucial for any health care facility considering incorporating new types of surgical N95 respirators into their operations to understand certain factors such as whether the manufacturer has the capability to scale up production to meet spikes in demand and what the manufacturer's lead time is for supplying new customers with surgical N95 respirators. The commenter stated that due to the fit-testing requirements, it is crucial for health care facilities to understand lead times of N95 manufacturers to ensure that respiratory protection is available when it is needed.
As reflected in the following discussion of the comments on payment adjustment eligibility, some commenters also believe that challenges in identifying surgical N95 respirators that are wholly domestically manufactured and documenting that fact for any purchased surgical N95 respirators have disincentivized hospitals from utilizing the payment adjustment. As reflected in the following discussion of the types of N95 respirators, a few commenters indicated that because non-surgical N95 respirators represent most of the N95 respirators purchased by hospitals, the fact that non-surgical N95 respirators are not eligible for the payment adjustment contributes to the lack of uptake for surgical N95 respirators.
Payment adjustment eligibility: In the CY 2023 OPPS/ASC final rule with comment period, we stated that we recognize that a hospital cannot fully independently determine if a NIOSH-approved surgical N95 respirator it purchases is domestic under our definition. Therefore, we finalized that a hospital may rely on a written statement from the manufacturer stating that the NIOSH-approved surgical N95 respirator the hospital purchased is domestic under our definition. In the CY 2025 OPPS/ASC proposed rule, we solicited comment on the following questions:
- Do hospitals have sufficient access to information on which surgical N95 models on the market are wholly domestically made?
- Have hospitals been able to obtain written statements from manufacturers stating that the NIOSH-approved surgical N95 respirator the hospital purchased is domestic under our definition?
- Would a publicly available list of products eligible for the payment adjustment (for example, if provided by CMS, NIOSH, or another government entity) make it easier for hospitals to locate products eligible for the payment adjustment?
- If we modified the payment adjustment such that hospitals that attested to purchasing wholly domestically made surgical N95 models from such a list did not need to obtain a written statement from the manufacturer, would hospitals more easily be able to utilize the payment adjustment?
Comment: In general, commenters were supportive of making publicly available a list of products eligible for the payment adjustment. Some commenters indicated that hospitals have had difficulty ascertaining which products meet the definition of domestic under this policy. Some commenters stated that hospitals have had difficulty obtaining written statements from manufacturers attesting to their products meeting the policy's definition of domestic.
Commenters stated that a publicly available list would simplify the procurement process and make it easier for hospitals to identify, locate, and purchase products eligible for the payment adjustment. Commenters also indicated that if such a list was made available, CMS should modify the payment adjustment such that hospitals that attest to purchasing wholly domestically made surgical N95 models from such a list do not need to obtain a written statement from the manufacturer. A commenter stated that removing the attestation requirement would streamline the process of purchasing eligible products and could encourage uptake of the payment adjustment. A commenter encouraged CMS to establish an ongoing process that would enable manufacturers or hospitals to submit products for certification and addition to the list. Some commenters stated that, if a list of eligible products were established, products not on the list should still be eligible for the payment adjustment if the product could be verified as domestic by some other means, such as through a written statement from the manufacturer.
One commenter stated that requiring hospitals to acquire this written attestation from manufacturers is not burdensome and believes that CMS should retain this requirement to ensure hospital accountability. One commenter stated that CMS should explore labeling requirements for manufacturers to facilitate the identification of domestic and non-domestic products. Another commenter stated that to alleviate hospital burden, CMS should work with hospital and supply chain communities to automate the reporting to CMS of domestic products purchased by hospitals.
Types of N95 respirators: In the CY 2023 OPPS/ASC proposed rule, for purposes of the payment adjustment policy, we proposed to categorize all NIOSH-approved surgical N95 respirators purchased by hospitals into two categories: (1) Domestic NIOSH-approved surgical N95 respirators; and (2) Non-domestic NIOSH-approved surgical N95 respirators. Feedback from external stakeholders has suggested that it is a challenge that the payment adjustments are limited to surgical N95 respirators, given some hospitals also procure non-surgical N95 respirators. Both surgical N95 respirators and non-surgical N95 respirators are primarily used to protect the wearer from inhaling airborne particles, including infectious agents like bacteria and viruses. They are highly efficient at filtering out at least 95% of airborne particles and are commonly used by healthcare workers during procedures that may generate aerosols, such as intubation or suctioning, or when caring for patients with infectious respiratory diseases like tuberculosis or coronavirus. Both types of N95 respirators serve as frontline defense for medical professionals. They are crucial for preventing the transmission of diseases within healthcare settings and safeguarding the health and well-being of both healthcare workers and patients. Surgical N95 respirators have the added protection against fluid penetration, and may be most useful in specialized healthcare settings ( e.g., ICU, Emergency Department, Operating Room) where the risk of fluid exposure may be greater. Additionally, during the COVID-19 pandemic, both types of N95 respirators saw issues around lack of availability and risk of counterfeit outlined in the CY 2023 OPPS/ASC final rule with comment period—issues which could compromise the safety of healthcare personnel and patients. In the CY 2025 OPPS/ASC proposed rule, we solicited comment on the following questions:
- Do hospitals procure both surgical N95 respirators and non-surgical N95 respirators?
- Has the payment adjustment's current focus on surgical N95 respirators inhibited uptake of the payment adjustments?
- Are the quality differentials between domestic and non-domestic surgical respirators also applicable to non-surgical respirators, and is a ( printed page 94293) sustained and reliable source of domestically made non-surgical N95 respirators important for strengthening hospitals' ability to protect the health and safety of personnel and patients in a public health emergency?
- Should CMS consider expanding the payment adjustments to include all domestic NIOSH-approved N95 respirators—i.e., non-surgical and surgical N95 respirators?
- If we expanded the payment adjustments to include all domestic NIOSH-approved N95 respirators, and if we modified the payment adjustment methodology calculation to provide a national standard unit cost differential between domestic and non-domestic NIOSH-approved surgical N95 respirators (rather than on a hospital-by-hospital basis), would the unit cost differential for non-surgical N95 respirators be different than the one for surgical N95 respirators?
Comment: Commenters were supportive of expanding the payment adjustment to include non-surgical N95 respirators. Commenters stated that expanding the payment adjustment to include non-surgical N95 respirators would ensure a more comprehensive and resilient domestic supply chain. A few commenters indicated that non-surgical N95 respirators represent most of the N95 respirators purchased by healthcare providers and that including them in the payment adjustment would lead to greater utilization of the payment adjustment by hospitals. A commenter stated that it would be less burdensome and more cost-effective for hospitals to procure and receive the payment adjustment for all domestic NIOSH-approved N95 respirators. A commenter stated the payment adjustment methodology could utilize the same national standard unit cost differential for surgical and non-surgical N95 respirators.
3. Potential Modifications To Include Nitrile Gloves
In addition to N95 respirators, nitrile gloves are another type of PPE for which it is particularly crucial to maintain a resilient, quality supply. Nitrile gloves protect healthcare workers and patients from the spread of micro-organisms that may potentially cause infection or illness during medical procedures and examination. They create a barrier between germs and the wearer's hands, and are generally worn anytime a healthcare worker touches blood, bodily fluids, bodily tissues, mucous membranes, or broken skin. They are disposable, enabling the use of new gloves for each patient. A resilient healthcare system needs readily available, high-quality nitrile gloves to respond efficiently and effectively to public health emergencies. During the COVID-19 pandemic, supply chain breakdowns limited the supply of quality nitrile gloves, putting U.S. healthcare workers and patients at risk. As with N95 respirators, non-domestic-sourced gloves during the COVID-19 pandemic saw counterfeit and quality challenges. The receipt of non-U.S.-made counterfeit or already-used gloves put the safety of healthcare workers and patients at risk.[171 172] Prior to 2020, over 95 percent of nitrile gloves sold in the U.S. came from other countries. As the pandemic escalated in 2020, U.S. demand for gloves outstripped available supply, leading to shortages. Around the same time, supply was also limited by coronavirus-related lockdowns in other countries that decreased production capacity, and by export restrictions of PPE. Further adding to supply pressures, forced labor violations by subsidiaries of a major glove producer led U.S. Customs and Border Protection (CBP) to issue a Withhold and Release Order, resulting in seizure of all listed products, including nitrile gloves, at CBP inspections. During the initial months of the pandemic, the cost of gloves increased, rising 18 percent from July to August 2020 (to $0.03 per glove) and then an additional 20 percent from November to December 2020 (to $0.05 per glove).[173]
During the pandemic, the U.S. government has invested in domestic glove manufacturing capabilities. U.S. glove-manufacturing projects received approximately $290 million in public funding as part of a broader $1.5-billion investment to support domestic glove manufacturing. These investments have resulted in an increase of 3.91 billion in annual production capacity for domestically manufactured nitrile gloves. The U.S. government also invested in manufacturing capacity for nitrile glove inputs such as nitrile butadiene rubber, and this manufacturing capacity is expected to become available in 2026.
However, since the pandemic began, some U.S. factories have been forced to consolidate operations or exit the industry. Further, non-U.S. nitrile glove producers have deployed cost-cutting tactics such as using lower-grade raw materials, prompting some purchasers to seek other sources out of concern for quality.[174] Producers of these lower quality products began selling gloves for the price of $0.02 each, rapidly increasing U.S. market share, going from 13% of U.S. market share in July 2020 to 19 percent in February 2021. As of 2024, only three producers of nitrile gloves are left in the United States, and they supply an estimated .05% percent of U.S. demand for nitrile gloves.
As with N95 respirators, a resilient public health industrial base requires baseline manufacturing capacity for nitrile gloves as critical PPE items, to ensure that hospitals and other institutions will be able to procure high quality gloves reliably. To help achieve this goal, certain U.S. Government departments have committed to purchase wholly domestically made nitrile gloves in line with the requirements in section 70953 of the Infrastructure Investment and Jobs Act. However, Federal demand alone cannot sustain a baseline level of nitrile glove production in the U.S. Private medical and health care users are the primary purchasers and users of medical-grade PPE, including nitrile gloves.
To ensure access to high quality products, as with N95 respirators, it is critically important to ensure that a sufficient share of nitrile gloves is wholly made in the U.S.—that is, including raw materials and components. In the CY 2023 OPPS/ASC rule, we stated our belief that the most appropriate framework for determining if a NIOSH-approved surgical N95 respirator is wholly made in the U.S. and therefore, considered domestic for purposes of the proposed adjustments, is the Berry Amendment. The Berry Amendment is a statutory requirement familiar to manufacturers that restricts the Department of Defense (DoD) from using funds appropriated or otherwise available to DoD for procurement of food, clothing, fabrics, fibers, yarns, other made-up textiles, and hand or measuring tools that are not grown, reprocessed, reused, or produced in the United States. For nitrile gloves, which are not covered by the Berry Amendment, we believe the Make PPE in America domestic content requirements outlined in section 70953 of the Infrastructure Investment and Jobs Act is the most appropriate framework for determining if a nitrile glove is wholly made in the U.S. These statutory requirements, which apply to procurement of nitrile gloves and other PPE by the U.S. Departments of Health and Human Services, and Veterans Affairs, and Homeland Security, require ( printed page 94294) the procurement PPE, including the materials and components thereof, that is grown, reprocessed, reused, or produced in the U.S. These statutory requirements have become familiar to manufacturers of nitrile gloves and other PPE. With respect to domestic manufacturing capabilities for raw materials and components, we understand that nitrile butadiene rubber (NBR), a key nitrile glove input, is currently not yet available domestically in sufficient quantity or quality to meet market needs. We understand that U.S. manufacturers do anticipate having the capability to source and manufacture all glove components domestically within the next two years.
Wholly domestically made, high quality nitrile gloves are generally more expensive than foreign-made ones, especially those of lower quality. This fact is also true for domestically made nitrile gloves that include non-domestically sourced NBR. These higher prices primarily stem from higher costs of manufacturing labor in the U.S. compared to costs in other countries, where most nitrile gloves and their inputs are made, and higher quality standards. These higher prices mean higher marginal costs for hospitals for procuring wholly domestically made nitrile gloves. Based on available data, our best estimate of the difference in the average unit cost of domestic and non-domestic nitrile gloves, is $0.13 per glove.
As outlined in this section, quality nitrile gloves are a crucial component of PPE needed to ensure the safety of health care workers and patients. The COVID-19 pandemic highlighted how overreliance on foreign imports of gloves jeopardized public health and the health and safety of healthcare workers and patients. In the CY 2025 OPPS/ASC proposed rule, we solicited comment on the following questions:
- Would modifying the payment adjustment to include nitrile gloves help offset the marginal costs that hospitals face in procuring high quality domestically made nitrile gloves?
- Would modifying the payment adjustment to include nitrile gloves help to sustain a baseline level of domestic manufacturing of nitrile gloves to ensure that hospitals and other stakeholders have ongoing, reliable access to an adequate supply of quality product?
- Would having access to a sustained and reliable source of domestically made nitrile gloves strengthen hospitals' ability to protect the health and safety of personnel and patients in a public health emergency?
- Are there other reasons why hospitals would benefit from an extension of the payment adjustment to include nitrile gloves not covered in the preceding questions?
- Do stakeholders believe a significant portion of hospitals would use domestic nitrile gloves if the payment adjustment were offered?
- If the payment adjustment was modified to include nitrile gloves, how should CMS define wholly domestically made nitrile gloves? Would it be appropriate to categorize all nitrile gloves purchased by hospitals into two categories: (1) domestic nitrile gloves that—with the exception of nitrile butadiene rubber (NBR)—comply with the Infrastructure Investment and Jobs Act's Make PPE in America Act domestic content requirements; and (2) non-domestic nitrile gloves?
- If the payment adjustment was modified to include nitrile gloves, and the categories were defined as described previously, would it be appropriate to eliminate the domestic content exception for NBR if domestic NBR production reaches a sufficient level to meet market needs?
- If the payment adjustment was modified to include nitrile gloves, should a national standard unit cost differential between domestic and non-domestic nitrile gloves be used to calculate the payment adjustment, and if so, what should the current unit cost differential be (or, what should the data source be)?
Comment: In general, commenters were supportive of modifying the payment adjustment to include nitrile gloves. Commenters explained why they believe it is important for the United States to maintain a baseline level of domestic manufacturing of nitrile gloves. For example, commenters cited quality concerns that have arisen with non-domestic nitrile gloves purchased by hospitals, especially during times of emergencies. A commenter stated that a domestic supply chain of nitrile gloves would significantly improve lead times compared to a foreign supply chain. One commenter stated that hospitals and purchasing entities desire to purchase domestically manufactured nitrile gloves to maintain stable supplies during global supply chain disruptions and to better ensure the health and safety of patients and workers.
Commenters indicated that certain domestic nitrile glove manufacturers have ceased operations. A commenter stated that the driving factor in these closures was a lack of market for domestic nitrile gloves due to their higher prices. This commenter, and other commenters, indicated that reducing or eliminating the price difference between domestic and non-domestic nitrile gloves would increase utilization of domestic nitrile gloves.
A few commenters that supported including nitrile gloves in the payment adjustment stated that it would be appropriate to apply the Make PPE in America Act requirements when defining domestic nitrile gloves for purposes of this policy. These commenters also agreed that there should be a temporary exception for NBR that could be eliminated when domestic supply of NBR is sufficient to support domestic demand. One commenter expressed concerns that neither the Berry Amendment nor the Make PPE in America Act requirements are commonly used by hospitals and health systems and therefore urged CMS to consider utilizing another standard.
A few commenters that supported including nitrile gloves in the payment adjustment also supported using a national standard unit cost differential between domestic and non-domestic nitrile gloves to calculate the payment adjustment. One commenter stated that the $0.13 per glove difference in the average unit cost of domestic and non-domestic nitrile gloves estimated by CMS is accurate and appropriate to use as the national standard unit cost differential. This commenter urged CMS to monitor the nitrile glove market and regularly update this differential to ensure hospitals are adequately reimbursed for their purchases of domestic nitrile gloves. Another commenter stated that the $0.13 per glove estimate was significantly higher than what they believe is the actual price differential between domestic and non-domestic nitrile gloves. This commenter stated that 100 percent domestically produced nitrile medical gloves sell for less than $0.13 per glove and that the actual price differential would likely be between $0.01 to $0.03 per glove. This commenter also requested that CMS work with industry experts to establish the national standard unit cost differential.
One commenter that supported including nitrile gloves in the payment adjustment stated that CMS should maintain a publicly available list of nitrile glove products that would qualify as domestic under such a policy. One commenter stated that access to a payment adjustment should be limited to hospitals that purchase at least 25% to 35% of their nitrile gloves from domestic manufacturers. A couple of commenters stated that the current production capability of domestic manufacturers is too small for the inclusion of nitrile gloves in the payment adjustment to have a meaningful impact. For this reason, a ( printed page 94295) commenter urged CMS to not include nitrile gloves in the payment adjustment.
4. Potential Modifications To Include Other PPE and Medical Devices
As noted in the CY 2023 OPPS/ASC final rule with comment period, we received many comments urging CMS to expand this policy to cover other forms of PPE and critical medical supplies. A few commenters stated that other forms of PPE suffered shortages during the pandemic similar to surgical N95 respirators and therefore investing in domestic production for these products was also important for future emergency preparedness. We stated that we will consider these comments for future rulemaking if appropriate as we gain more experience with our policy. We sought comment on other PPE types and medical devices that could be appropriate for a similar payment adjustment.
Comment: Several commenters urged CMS to expand the payment adjustment to include other PPE types and medical devices. Examples included gowns, hair nets, beard covers, bouffant caps, shoe covers, face shields, ASTM level II and III surgical masks, powered air purifying respirators, elastomeric respirators, syringes, needles, catheters, and wound care dressings. Commenters indicated that many of these products are currently being purchased from non-domestic manufacturers and have been prone to shortages and quality issues. For example, a commenter cited safety concerns regarding the quality of imported syringes and needles which they stated have had issues ranging from leaks to breakages that compromise patient safety. Some commenters indicated that domestic versions of the products they recommend including in the payment adjustment are more costly to produce than non-domestic versions. For example, one commenter stated that the higher cost of labor in the U.S. in addition to imbalances in costs of manufacturing certain inputs contributes to the cost differential between domestic and non-domestic syringes and needles. Commenters expressed that expanding the payment adjustment to more products would increase uptake of the payment adjustment by hospitals, strengthen the existing U.S. manufacturing base, incentivize other manufacturers to prioritize domestic production, and protect access to high-quality products.
Response: We greatly appreciate the many thoughtful and informative comments we received in response to our request for comment on payment adjustments under the IPPS and OPPS for domestic personal protective equipment, including potential modifications to include non-surgical N95 respirators, potential modifications to include nitrile gloves, potential modifications to include other PPE and medical devices, and potential modifications to the methodology used to calculate the payment adjustments. We thank the commenters for their feedback and agree with comments calling for improvements to and expansion of these payment adjustments. In 2026 rulemaking, we intend to propose a new payment methodology, such as one that no longer relies exclusively on hospital-specific data. In this rulemaking, we also intend to propose to expand the payment adjustments to domestic non-surgical N95 respirators and domestic nitrile gloves. We also intend to continue exploring expansion of the payment adjustments to include other types of domestically made PPE and other medical products. Hospitals are the primary purchasers and users of medical PPE, and PPE are essential for the protection of patients and hospital personnel. We believe payment modifications that account for the potentially higher cost of domestically produced critical PPE help to safeguard hospital personnel and beneficiary safety over the long term by sustaining domestic production and availability of these PPE. Finally, we will explore the feasibility of creating a list of qualifying surgical N95 respirators that are domestically made PPE given feedback from commenters. We again thank commenters for their feedback regarding these payment adjustments.
F. Payment for HIV Pre-Exposure Prophylaxis (PrEP) in Hospital Outpatient Departments
On July 12, 2023, we proposed a National Coverage Determination (NCD) to cover Pre-Exposure Prophylaxis (PrEP) to prevent Human Immunodeficiency Virus (HIV) under Medicare Part B. The proposed coverage would include coverage for the PrEP for HIV drugs, drug administration, HIV and hepatitis B screening, and individual counseling performed by either physicians or certain other health care practitioners. We explained that, if finalized as proposed, all of the components would be covered as an additional preventive service without Part B cost-sharing ( i.e., deductibles or co-pays). The final NCD was not issued before the proposed rule was issued.[175] (89 FR 59399 through 59402) The final decision memorandum for the NCD was issued and was effective on September 30, 2024. [ https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=310&fromTracking=Y&]
As explained in the proposed rule, the HCPCS codes that describe these services are outlined in Table 142.
( printed page 94296)For CY 2025, we proposed to pay for PrEP for HIV drugs and related services as additional preventive services under the OPPS, if covered in the final NCD. We stated that we believe the resource costs for HCPCS codes listed in Table 142 would be similar across different settings of care, including the HOPD and physician office, and therefore the proposed policies for determining the payment limits for drugs covered as additional preventive services (DCAPS drugs) in the CY 2025 PFS proposed rule (89 FR 61596), which would include PrEP for HIV drugs, would be appropriate for use under the OPPS as well. Therefore, we proposed to pay for the HCPCS codes listed in Table 142 that are furnished in HOPDs in a similar manner as when these codes are furnished in the physician office.
HCPCS code G0012 (Injection of pre-exposure prophylaxis (prep) drug for hiv prevention, under skin or into muscle) may be used to describe the injection of a PrEP drug for HIV prevention. For CY 2025, if covered as an additional preventive service, we proposed to assign this HCPCS code to APC 5692 (Level 2 Drug Administration) based on the crosswalk to HCPCS code 96372 (Therapeutic, prophylactic, or diagnostic injection (specify substance or drug); subcutaneous or intramuscular) based on the anticipated similarity in resource use. For the PrEP for HIV counseling services performed by hospital staff, specifically HCPCS code G0013, if covered as an additional preventive service, we proposed to assign this service to a clinical APC with a payment rate that approximates the payment rate in the physician office setting. The proposed CY 2025 payment rates can be found in Addendum B to the CY 2025 OPPS/ASC proposed rule on the CMS website. We did not propose to pay for PrEP for HIV counseling, described by HCPCS code G0011, performed by physicians under the OPPS as this is a physician-only service.
To determine the OPPS payment amount for PrEP for HIV drugs, we proposed to utilize the ASP methodology under section 1847A of the Act when ASP data is available. As discussed in the CY 2025 PFS proposed rule (89 FR 61596), we believed and continue to believe, that the use of ASP data would be preferable for determining the payment amounts for DCAPS drugs, including PrEP for HIV, for two reasons. First, this approach would determine the payment amount for these drugs in the same way as the payment amount is usually determined for most other drugs that are separately payable under Part B, when possible. This would include the application of payment limit calculations for multiple source drugs, single source drugs and biologicals, and biosimilar biological products, as is done for products under section 1847A of the Act, for each applicable billing and payment code. Second, because section 1847A(c)(3) of the Act requires that calculation of the ( printed page 94297) manufacturer's ASP for an NDC must include volume discounts, prompt pay discounts, cash discounts, free goods that are contingent on any purchase requirement, chargebacks, and rebates (other than rebates under the Medicaid drug rebate program, discounts under the 340B Program, and rebates under the Part B and Part D Medicare inflation rebate program), this would set a payment amount that would likely better reflect acquisition cost of the drug than list prices in available compendia (such as Wholesale Acquisition Cost (WAC)).
Specifically, if ASP data is not available for a particular DCAPS drug, the PFS proposal described the use of alternative pricing sources. As previously stated, we believe the resource costs should be similar regardless of whether DCAPS drugs like PrEP for HIV drugs are furnished in the HOPD or the physician office, and we propose to use the same method of utilizing alternative pricing sources for drugs paid under the OPPS as additional preventive services as was proposed under the PFS.
If ASP data for DCAPS drugs like PrEP for HIV is not available, we proposed to determine the payment amount for the applicable billing and payment code using the most recently published amount for the drug in Medicaid's National Average Drug Acquisition Cost (NADAC) survey (OMB control number 0938-1041).[176] When using NADAC data, we proposed to determine the payment amount per billing unit, which would be an average of NADAC prices for all NDCs for the drug. If a drug is available in generic and brand formulations, we proposed all NDCs will be averaged together to determine the payment amount.
As described in the CY 2025 OPPS/ASC proposed rule (89 FR 59401), regarding which NADAC survey will be used, we specified that we will use the survey “most recently published for purposes of this policy,” which means the most recently updated NADAC survey available 30 days after the close of the quarter for which ASP data would have been reported if it were available.[177] For example, if NADAC is used to determine the payment amount effective for dates of service in the third calendar quarter, CMS would use the most recent NADAC survey update available on the 30th day after the close of the first calendar quarter. This survey provides a national drug pricing benchmark for certain drugs that is adequately comprehensive to serve as the first alternative pricing source in the case that ASP data is not available. CMS conducts surveys of retail community pharmacy prices to develop the NADAC pricing benchmark in the annual NADAC pricing file. The pricing benchmark is reflective of the prices paid by retail community pharmacies to acquire prescription and over-the-counter covered outpatient drugs. NADAC data is publicly available and it can be accessed at https://data.medicaid.gov/nadac.
We stated in the CY 2025 OPPS/ASC proposed rule (89 FR 59401), that since NADAC pricing is only available for drugs typically dispensed through retail community pharmacies, there could be circumstances in which ASP and NADAC are not available for DCAPS drugs like PreP for HIV. Therefore, if both ASP and NADAC pricing data are not available for a DCAPS drug, we proposed to use the most recently published and listed prices for pharmaceutical products in the Federal Supply Schedule (FSS) to calculate the payment amount for the applicable billing and payment code. Most recently published for purposes of this policy means the most recently updated FSS survey available 30 days after the close of the quarter for which ASP data would have been reported if it were available.[178] For example, if FSS is used to determine the payment amount effective for dates of service in the third calendar quarter, CMS would use the most recent FSS update available on the 30th day after the close of the first calendar quarter. When using the FSS, we proposed that we would calculate the average price per billing unit (as described in the billing and payment code for the drug) for all NDCs listed for a drug. Drug pricing information, including FSS pricing, from the Veteran Affairs' (VA's) pharmaceutical pricing database is publicly available at the NDC level and published at https://www.va.gov/opal/nac/fss/pharmPrices.asp.
We proposed to use FSS data when ASP and NADAC data are not available because FSS data is one of the few existing options for drug pricing that includes a wide variety of drug formulations, including both self-administered drugs typically dispensed through retail community pharmacies and drugs administered incident to a physician's service. For more details on the proposed pricing methodology for the physician office setting, please see the CY 2025 PFS proposed rule (89 FR 61596).
We note that the PFS proposal included a final step of invoice pricing; however, invoice pricing is not currently available under the OPPS, so we proposed not to adopt that portion of the PFS proposal. However, please see our Invoice Drug Pricing Policy for CY 2026 in section V.B.2.d. of this final rule with comment period. Because invoice pricing is not available in the OPPS currently, we proposed that if ASP, NADAC, and FSS pricing are not available for a particular drug covered as an additional preventive service, we will use WAC plus 6 percent, or 3 percent if in an initial sales period, consistent with payment for separately payable drugs paid under the OPPS. This would result in different pricing between the OPPS and PFS if ASP, NADAC, and FSS pricing are not available, but we believe it is appropriate because invoice pricing is not currently an option under the OPPS and this pricing metric should only apply to a small subset of drugs covered as additional preventive services until one of the other pricing metrics becomes available. We proposed to treat other drugs covered as additional preventive services under this same methodology.
As we stated in the CY 2025 OPPS/ASC proposed rule (89 FR 59401), if the PreP for HIV drugs would be covered as additional preventive services, we proposed to update the payment rates determined using the methodologies previously summarized on January 1, 2025, or the date of coverage, whichever is later, which would be further updated on the same schedule as the ASP pricing file, which is updated each calendar quarter. We proposed to assign the drug products covered as additional preventive services to status indicator K (Nonpass-Through Drugs and Nonimplantable Biologicals, Including Therapeutic Radiopharmaceuticals; Paid under OPPS; separate APC payment), as this status indicator identifies drugs and biologicals that are separately paid under the OPPS and therefore would allow us to operationalize separate payment for PrEP drugs. We explained that if the PrEP for HIV drugs are covered as additional preventive services, on January 1, 2025, or the date of coverage, whichever is later, we proposed that we would assign each PrEP for HIV drug covered as an additional preventive service to its own APC, which will have a payment rate assigned according to the previously defined methodology.
As described in the CY 2025 OPPS/ASC proposed rule (89 FR 59401 through 59402), HCPCS code J0799 (HIV prep, FDA approved, noc) was created effective January 2, 2024, and may be ( printed page 94298) used to describe a PrEP for HIV drug that is FDA approved but is not otherwise classified. We proposed to pay 95 percent of AWP for HCPCS code J0799, which is consistent with how unlisted drugs and biologicals are paid under the OPPS when they are reported with HCPCS code C9399 (Unclassified drugs or biologicals). As HCPCS code J0799 and HCPCS code C9399 both describe drugs that are unclassified or not otherwise classified, we believe the payment methodologies should be similarly aligned. Section 1833(t)(15) of the Act, as added by section 621(a)(1) of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (Pub. L. 108-173), provides for payment under the OPPS for new drugs and biologicals until HCPCS codes are assigned. Under this provision, we are required to make payment for a covered outpatient drug or biological that is furnished as part of covered outpatient department services but for which a HCPCS code has not yet been assigned in an amount equal to 95 percent of AWP for the drug or biological.
In the CY 2005 OPPS/ASC final rule with comment period (69 FR 65805), we implemented section 1833(t)(15) of the Act by instructing hospitals to bill for a drug or biological that is newly approved by the FDA and that does not yet have a HCPCS code by reporting the NDC for the product along with the newly created HCPCS code C9399 (Unclassified drugs or biologicals). We explained that when HCPCS code C9399 appears on a claim, the Shared Systems suspends the claim for manual pricing by the Medicare Administrative Contractor (MAC). The MAC prices the claim at 95 percent of the drug or biological's AWP, using Red Book or an equivalent recognized compendium, and processes the claim for payment. We emphasized that this approach enables hospitals to bill and receive payment for a new drug or biological concurrent with its approval by the FDA. The hospital does not have to wait for the next quarterly release or for approval of a product-specific HCPCS code to receive payment for a newly approved drug or biological or to resubmit claims for adjustment. We instructed that hospitals would discontinue billing HCPCS code C9399 and the NDC upon implementation of a product specific HCPCS code, status indicator, and appropriate payment amount with the next quarterly update. While the statute does not require DCAPS drugs to be paid at 95 percent of AWP when not assigned to a product specific HCPCS code, we believe it would be appropriate to create a parallel policy given that HCPCS code J0799 and HCPCS code C9399 both describe drugs that are unclassified or not otherwise classified. As the payment amount for HCPCS code C9399 is statutorily mandated at 95 percent of AWP, we believe that the payment amount for HCPCS code J0799 should also be 95 percent of AWP.
Therefore, we proposed to establish an identical payment policy for HCPCS code J0799, which may be used to describe drugs that are FDA-approved for PrEP for HIV and are covered as additional preventive services. In order to effectuate payment at 95 percent of AWP, we proposed to require hospitals to bill for a drug that is newly FDA approved for PrEP for HIV, and covered as an additional preventive service, and that does not yet have a HCPCS code, by reporting the NDC for the product along with the newly created HCPCS code J0799. Similar to HCPCS code C9399, when HCPCS code J0799 appears on a claim, the Shared Systems will suspend the claim for manual pricing by the MAC. The MAC would price the claim at 95 percent of the drug or biological's AWP, using Red Book or an equivalent recognized compendium, and process the claim for payment. This approach would enable hospitals to bill and receive payment for a drug that is newly FDA approved for PrEP for HIV and covered as an additional preventive service concurrent with its approval by the FDA. The hospital would not have to wait for the next quarterly release or for approval of a product-specific HCPCS code to receive payment for a newly approved drug or to resubmit claims for adjustment. We would instruct hospitals to discontinue billing HCPCS code J0799 and the NDC upon implementation of a product specific HCPCS code, status indicator, and appropriate payment amount with the next quarterly update.
Finally, if covered as an additional preventive service, we proposed to assign all HCPCS codes describing pharmacy supplying fees for PrEP for HIV to an OPPS status indicator of “B”. This follows the longstanding OPPS practice of assigning HCPCS codes that describe a pharmacy supply or dispensing fee to status indicator “B” (Codes that are not recognized by OPPS when submitted on an outpatient hospital Part B bill type (12x and 13x); Not paid under OPPS), such as HCPCS code Q0512 (Pharmacy supply fee for oral anti-cancer, oral anti-emetic or immunosuppressive drug(s); for a subsequent prescription in a 30-day period) and HCPCS code Q0513 (Pharmacy dispensing fee for inhalation drug(s); per 30 days).
Comment: Several commenters were supportive of CMS's proposed payment policies and encouraged CMS to use its authority to pay for PrEP for HIV drugs and services as additional preventive services.
Response: We thank commenters for their support. The final decision memorandum for the NCD was issued and was effective on September 30, 2024 [ https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=310&fromTracking=Y&]. For the interim period of September 30, 2024, until December 31, 2024, before the DCAPS payment policy being finalized in this rule is effective, CMS will pay for PrEP for HIV drugs under the OPPS in a manner that closely aligns with how payment is currently made for drugs under the OPPS, which is generally based off of the ASP methodology. However, beginning January 1, 2025, as discussed in more detail below, we are finalizing a payment approach for determining a payment limit for DCAPS drugs. As PrEP for HIV is a DCAPS drug, payment will be made in accordance with the finalized payment approach.
Comment: Several commenters supported CMS's proposal to use ASP plus 6 percent as the basis of payment; however, they recommended CMS revise the proposed payment approach specifically for PrEP for HIV, or in some cases DCAPS payment in general, in situations in which ASP data is unavailable. For example, commenters recommended in situations in which ASP is unavailable, CMS should base payment on WAC plus 3 or 6 percent, rather than NADAC or FSS pricing. There were concerns from commenters that NADAC or FSS pricing were inadequate payment methodologies and could lead to underpayments that would discourage utilization.
Response: We continue to believe that NADAC and FSS pricing are appropriate payment methodologies to determine a payment limit under the OPPS for DCAPS drugs in instances in which ASP data is not available. In the CY 2020 PFS final rule (84 FR 62655), we stated that we believe NADAC survey data on invoice prices provides the closest pricing metric to ASP-based payment limits that is available. FSS data is one of the few existing options for drug pricing that includes a wide variety of drug formulations, which is why CMS chose it as an additional alternative for drugs covered as additional preventive services. Thus, our proposal explained that ASP, NADAC, and FSS are all drug pricing options that aim to estimate the accurate acquisition cost of a drug, rather than WAC, which is a list price. ( printed page 94299) Therefore, we believe WAC should be the payment methodology used under the OPPS only if ASP, NADAC, or FSS pricing are not available, as invoice pricing is not currently an option for determining a payment limit for a drug under the OPPS, unlike under the PFS.
Comment: A few commenters stated Medicare should ensure adequate payment for these services in a way that takes into account the site of service, since, in their view, hospital outpatient departments are more likely to care for patients who are more medically and socially complex than those cared for in physicians' offices. These commenters recommended that CMS calculate the payment for PrEP for HIV services furnished in an HOPD in the same manner as it does for other OPPS services, and not at the PFS rate.
Response: We thank commenters for their feedback. We continue to believe that the additional preventive services discussed in this section generally should have resource costs that align between care settings, such as the HOPD and the physician's office. Accordingly, we are adopting a similar payment approach for the drugs covered as additional preventive services as under the fee schedule for DCAPS drugs in the physician office setting.
For the PrEP for HIV counseling services performed by hospital staff, specifically HCPCS code G0013, we are finalizing our proposal to assign this service to a clinical APC with a payment rate that approximates the payment rate in the physician office setting, consistent with our belief that the resource costs would be similar across different settings of care. Based on updated data available for this final rule, as well as the expected physician office setting payment rate, this corresponds to an APC assignment of APC 5821 (Level 1 Health and Behavior Services) for HCPCS G0013. We will continue to monitor the appropriateness of these APC assignments.
We note that for the administration of the drug; however, we are finalizing our proposed assignment of HCPCS code G0012 (Injection of pre-exposure prophylaxis (prep) drug for hiv prevention, under skin or into muscle), which may be used to describe the injection of a PrEP drug for HIV prevention, to APC 5692 (Level 2 Drug Administration) based on the OPPS specific crosswalk to HCPCS code 96372 (Therapeutic, prophylactic, or diagnostic injection (specify substance or drug); subcutaneous or intramuscular) based on the anticipated similarity in resource use to other codes assigned to this APC. Since we have an existing crosswalk code under the OPPS on which to base payment for HCPCS code G0012, we believe it is appropriate to finalize the assignment of that code to APC 5692, rather than the PFS payment rate.
Comment: Several commenters shared concerns or support for the Proposed NCD for Pre-Exposure Prophylaxis (PrEP) for Human Immunodeficiency Virus (HIV) Infection Prevention, published on July 12, 2023, as the NCD was not yet finalized as of the end of the comment period for the CY 2025 PFS proposed rule on September 9, 2024. This included concerns regarding unintended consequences of NCD finalization, such as having the same drugs covered under Medicare Part B or Part D depending on the indication in which they are used, which a commenter believed would be administratively burdensome. There were also concerns regarding pharmacists being unable to enroll as prescribers under Medicare Part B. The commenters also included support for certain payment methodologies for Medicare to adopt in the interim period after the NCD was finalized, but before this rule takes effect on January 1, 2025. Commenters also expressed concerns regarding Medicare Advantage Utilization Management controls being used for products potentially covered under the NCD.
Response: We thank commenters for their feedback; however, we note comments related to the proposed and final coverage decision are out of scope for purposes of our proposed payment policies. Additionally, as the proposed and finalized policies in this rule are for payment for DCAPS drugs, including PrEP for HIV drugs, beginning January 1, 2025, comments regarding the interim payment rate for PrEP for HIV are outside the scope of this rulemaking. The public comment period on the proposed NCD for PrEP for HIV coverage under Medicare Part B was from January 12, 2023, to February 11, 2023. The final NCD was released on September 30, 2024, and is available at https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&ncaid=310&fromTracking=Y&. We direct interested parties to https://www.cms.gov/medicare/coverage/prep for more information on the final NCD and the transition of PrEP for HIV coverage and payment from Part D to Part B. This CMS PrEP web page contains and/or will contain additional guidance on implementation of PreP for HIV coverage under Part B, including coding and billing information, payments for PrEP for HIV for the period of September 30-December 31, 2024, and the implementation of the payment approach for DCAPS drugs, including PreP for HIV drugs, effective January 1, 2025.
Comment: A commenter strongly opposed use of FSS pricing to pay for PrEP for HIV drugs, but asked that we confirm that we will utilize the Other Government Agency (OGA) FSS price if we proceed with finalizing the use of the FSS price in cases where ASP and NADAC pricing data are not available. The commenter believed basing payment on the OGA FSS price would better support patient access to PrEP for HIV drugs compared to the Big 4 FSS price (the Department of Veterans Affairs (VA), DoD, the Public Health Service, and the Coast Guard), which the commenter stated reflects substantial statutory discounts that do not apply to Medicare Part B providers and they stated this could lead to significant under-payment for PrEP for HIV drugs.
Response: We confirm that the FSS pricing data used will be the OGA prices.
As previously described, the NCD was finalized on September 30th, 2024. On that date Medicare began coverage and payment for PrEP for HIV and associated services. The payment assignments for the interim period (that is, September 30, 2024, to December 31, 2024) are in Table 143.
( printed page 94300) ( printed page 94301)After consideration of the public comments we received, we are finalizing our proposal with slight modifications. For CY 2025, we are finalizing our proposal to pay for PrEP for HIV drugs and related services as additional preventive services under the OPPS. That is, we are finalizing our proposal that PrEP for HIV will be paid in accordance with the payment approach we are finalizing for all DCAPS drugs. We are finalizing the APC assignments in Table 144 generally consistent with our proposal.
We are finalizing some modifications to our proposed policies. When using NADAC pricing to determine a payment rate, and the drug is available in generic and brand formulations, we proposed all NDCs will be averaged together to determine the payment amount. Similarly, when using the FSS to determine a payment rate, we proposed that we would calculate the average price per billing unit (as described in the billing and payment code for the drug) for all NDCs listed for a drug.
In this final rule, to maintain consistency with the PFS finalized policy and comments received, we are amending our proposal, to finalize a policy to treat brand and generic drugs in a similar manner to the description in the Medicare Claims Processing Manual, Chapter 17, sections 20.1.3 and 20.4. That is, when calculating the price for multiple-source DCAPS drugs using NADAC or FSS pricing, we will use the lesser price of: (1) The median of all generic forms of the drug; or (2) The lowest brand name product.
Additionally, after consideration of public comments, we are finalizing the following payment approach for drugs covered as additional preventive services and paid under the OPPS, and note the actual rates used for payment will be published on the CMS website and updated quarterly:
- If ASP data is available for the DCAPS drug, the payment limit will be determined based on the methodology under section 1847A(b) of the Act (usually 106 percent of ASP);
- If ASP data is not available, the payment limit will be calculated using NADAC prices for the drug;
- If ASP data and NADAC prices are not available, the payment limit will be calculated using the FSS prices for the drug; and
- If ASP data, NADAC prices, and FSS prices are not available, payment will be WAC plus 6 percent, or 3 percent if in an initial sales period, consistent with payment for separately payable drugs paid under the OPPS. Because invoice pricing is not currently available in the OPPS, if ASP, NADAC, and FSS pricing are not available for a particular drug covered as an additional preventive service, we will use WAC plus 6 percent, or 3 percent if in an initial sales period.
However, as discussed in section V.B.2.d. (Invoice Drug Pricing for CY 2026) of this final rule with comment period, we are finalizing a policy to begin to price certain drugs based on their invoices starting in CY 2026. As such, we are making a conforming change such that, starting in CY 2026, if ASP data, NADAC prices, and FSS prices are not available, the payment rate would be based on the invoice price. For CY 2025 only, if ASP data, NADAC prices, and FSS prices are not available, payment will be based on WAC plus 6 percent, or 3 percent of in an initial sales period. We note this may result in different approaches to setting a payment rate between the OPPS and PFS if ASP, NADAC, and FSS pricing are not available, but we believe it is appropriate because invoice pricing is not currently an option under the OPPS and this pricing metric should only apply to a small subset of DCAPS drugs, until one of the other pricing metrics becomes available. As mentioned in CMS's responses to comments above, we are clarifying that the FSS price is the “other government agencies” price.
Finally, in the CY 2025 OPPS/ASC proposed rule, we proposed that if covered as an additional preventive service, we would assign all HCPCS ( printed page 94302) codes describing pharmacy supplying fees for PrEP for HIV to an OPPS status indicator of “B”. We stated this follows the longstanding OPPS practice of assigning HCPCS codes that describe a pharmacy supply or dispensing fee to status indicator “B” (Codes that are not recognized by OPPS when submitted on an outpatient hospital Part B bill type (12x and 13x); Not paid under OPPS), such as HCPCS code Q0512 (Pharmacy supply fee for oral anti-cancer, oral anti-emetic or immunosuppressive drug(s); for a subsequent prescription in a 30-day period) and HCPCS code Q0513 (Pharmacy dispensing fee for inhalation drug(s); per 30 days). While our longstanding OPPS practice has been to assign all HCPCS codes describing pharmacy supplying or dispensing fees to an OPPS status indicator of “B” indicating that the code is not payable under the OPPS, we believe assignment of these HCPCS codes describing pharmacy supplying or dispensing fees would be most accurately assigned to a status indicator of “M” (Items and Services Not Billable to the MAC; Not paid under OPPS), which also indicates that the codes are not paid under the OPPS. Status indicator “M” codes are not billable to the MAC. We believe status indicator “M” more clearly articulates that these codes are not billable to the MAC by hospitals and pharmacies should bill these HCPCS codes to their appropriate MAC. Whereas, status indicator “B” states the code is not recognized by OPPS, while that is true, we believe the definition for status indicator “M” is more comprehensive and better instructs proper billing as it is communicating to all hospitals that these codes are not billable to their MAC, rather than just not recognized by the OPPS. Therefore, we are finalizing this technical refinement to assign HCPCS code Q0521 (Pharmacy supplying fee for hiv pre-exposure prophylaxis fda approved prescription) to status indicator “M.” We note, HCPCS code Q0521 replaces HCPCS codes Q0516, Q0517, Q0518, Q0519, and Q0520 starting January 1, 2025.
To maintain consistency with regard to the use of status indicators under the OPPS, we are also applying this technical refinement and assigning a status indicator of “M” to other pharmacy supplying and dispensing HCPCS codes. This is consistent with our discussion and intent in the CY 2025 OPPS/ASC proposed rule, where it was discussed that pharmacy supplying and dispensing codes, such as HCPCS codes Q0512 (Pharmacy supply fee for oral anti-cancer, oral anti-emetic or immunosuppressive drug(s); for a subsequent prescription in a 30-day period) and HCPCS code Q0513 (Pharmacy dispensing fee for inhalation drug(s); per 30 days) were historically assigned to a non-payable status indicator. The pharmacy supplying and dispensing codes that we are assigning to a status indicator of “M” for CY 2025 include: Q0510 (Pharmacy supply fee for initial immunosuppressive drug(s), first month following transplant); Q0511 (Pharmacy supply fee for oral anti-cancer, oral anti-emetic or immunosuppressive drug(s); for the first prescription in a 30-day period); Q0512 (Pharmacy supply fee for oral anti-cancer, oral anti-emetic or immunosuppressive drug(s); for a subsequent prescription in a 30-day period); Q0513 (Pharmacy dispensing fee for inhalation drug(s); per 30 days); Q0514 (Pharmacy dispensing fee for inhalation drug(s); per 90 days). As described in the preceding paragraph, we believe status indicator “M” more clearly articulates that these codes are not billable to the MAC by hospitals and pharmacies should bill these HCPCS codes to their appropriate MAC. Whereas, status indicator “B” states the code is not recognized by OPPS, while that is true, we believe the definition for status indicator “M” is more comprehensive and better instructs proper billing as it is communicating to all hospitals that these codes are not billable to their MAC, rather than just not recognized by the OPPS. Similarly, as we previously summarized, we are not paying for PrEP for HIV counseling, described by HCPCS code G0011, performed by physicians under the OPPS as this is a physician-only service. Therefore, following the same logic as just discussed for the pharmacy supplying and dispensing codes, we are also finalizing assignment of this code to a status indicator of “M” to better instruct proper billing.
( printed page 94303) ( printed page 94304)Final definitions of status indicators for CY 2025 can be found in Addendum D1 to this final rule with comment period. Additionally, final CY 2025 APC payment rates can be found in Addenda A and B. Addenda to this final rule with comment period can be found on the CMS OPPS web page.
Corresponding with the finalization of this policy, we are adding conforming regulation text changes by adding new paragraph (j) to § 419.41. This new paragraph details the payment amounts for drugs covered as additional preventive services under the OPPS and includes the finalized payment methodology hierarchy as described in this section.
G. Payment Policy for Devices in Category B Investigational Device Exemption (IDE) Clinical Trials Policy and Drugs/Devices With a Medicare Coverage With Evidence Development (CED) Designation
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72027), and as authorized by section 1833(w) of the Act, we finalized a policy to make a single blended payment for devices and services in Category B IDE studies, in order to preserve the scientific validity of these studies by avoiding differences in Medicare payment methods that would otherwise reveal the group (treatment or control) to which a patient has been assigned. Specifically, we codified our process of utilizing a single packaged payment for Category B IDE studies, including the cost of the device and routine care items and services, in the regulation text for payment to hospitals in a new § 419.47. We provided in new § 419.47(a) and (b) that CMS will create a new HCPCS code, or revise an existing HCPCS code, to describe a Category B IDE study, which will include both the treatment and control arms, related device(s) of the study, as well as routine care items and services, as specified under 42 CFR 405.201, when CMS determines that the Medicare coverage IDE study criteria at § 405.212 are met, and a new or revised code is necessary to preserve the scientific validity of the IDE study, such as by preventing the unblinding of the study.
We finalized that the single blended payment rate would be dependent on the specific trial protocol and would account for the frequency with which the investigational device is used compared to the control where the investigational device is not used. For example, in a study for which CMS determines the Medicare coverage IDE study criteria in § 405.212 are met and where there is a 1:1 assignment of the device to control (no device), Medicare's payment rate would prospectively average the payment for the device with the zero payment for the control in a 1:1 ratio. Furthermore, costs for routine care items and services in the study, as specified under § 405.201, would be included in the single blended payment (87 FR 72026 through 72027).
Since implementing this policy, we have heard from interested parties that our regulation at § 419.47(a) and (b) excluded clinical trials for which there is no control arm. We appreciate the input. Category B IDE studies with no control arm would be paid normally because an alternative payment methodology would not be necessary to preserve their scientific validity. Our policy at § 419.47 applies only to IDE studies with a control arm and where a payment adjustment is necessary to preserve the scientific validity of such a study. The rule was not intended to suggest that CMS will not pay for Category B IDEs with no control arm, provided the studies meet the coverage criteria. In those circumstances, Medicare payments would be made using the usual Medicare payment methodologies.
In many instances, requests for coding and payment for devices in Category B IDE studies are submitted through our New Technology APC application process and include the submission of cost information. However, we have encountered difficulties determining accurate payment rates for Category B IDE studies in the absence of New Technology APC applications, such as ( printed page 94305) when coding for Category B IDE studies is developed through the CPT Editorial Panel process. We encourage interested parties to use the New Technology APC application process, where applicable, to submit cost information to CMS. Absent information on the resource costs associated with the services and devices in a Category B IDE study, we may assign a SI of “E2” to indicate an item, code or service for which pricing information and claims data are not available, and, therefore, the item, code or service is not paid by Medicare when submitted by an outpatient claim.
For CY 2025, we proposed to utilize a payment methodology similar to the one developed for Category B IDE clinical trials for drugs and devices covered under a national coverage determination (NCD) that uses the Coverage with Evidence Development (CED) paradigm when a payment adjustment is necessary to preserve the scientific validity of such a study. Specifically, we proposed to use our authority at section 1833(w) of the Act to develop alternative methods of payment under Medicare Part B for drugs and devices being studied in clinical trials under a CED NCD. These CED NCDs will be listed on the CMS CED website.[179]
Similar to our policy on devices in Category B IDE trials, for devices under a CED NCD, we proposed to make a single blended payment rate that would be dependent on the specific trial protocol and would account for the frequency with which the investigational device is used compared to the control where the investigational device is not used. For example, in a study for which there is a 1:1 assignment of the device to control (no device), Medicare's payment rate would prospectively average the payment for the device with the zero payment for the control in a 1:1 ratio.
As previously described, when necessary to preserve the scientific validity of the study, we proposed to make payment using an adjusted payment level representing the frequency with which the study drug and placebo, or comparator drug, is furnished. A placebo, or comparator drug, could represent what a beneficiary would typically receive in order to serve as a comparator to assess the effectiveness, or therapeutic benefit, of the study drug. These adjusted payments would protect the scientific validity of the trial by avoiding differences in Medicare payment methods that could otherwise invalidate the scientific validity of the trial, such as by revealing the group (treatment or control) to which a patient has been assigned. We proposed to base the payment amount for the study drug, or active comparator drug, on the ASP methodology, that is ASP plus 6 percent if ASP data is available. If ASP data is not available, then we proposed to pay the wholesale acquisition cost (WAC). During an initial sales period, we proposed to base the payment on WAC plus 3 percent, otherwise, we proposed to base payment on WAC plus 6 percent. If WAC is not available, then we proposed to pay 95 percent of average wholesale price (AWP). This payment hierarchy is consistent with CMS payment for non-passthrough separately payable drugs in the OPPS as discussed in section V.B. of the CY 2025 OPPS/ASC proposed rule.
These payment amounts would be used to calculate the adjusted payment level representing the frequency with which the study drug and placebo, or comparator drug, is furnished. For purposes of setting this adjusted payment level, we proposed to use a zero dollar amount for a placebo or comparator. A new, or revised, HCPCS code would be created for the drug and placebo or comparator in the CED study. We proposed that we would assign this HCPCS code to its own APC reflecting the payment amount determined appropriate based on available pricing information and the frequency with which the study drug and placebo, or comparator drug, is used.
For example, as most drugs are currently paid per dosage unit, such as per 1 mg, a payment rate, potentially priced per 1 mg of drug, placebo, or active comparator, might be based on the average sales price methodology for the drug averaged with a zero-dollar payment for the placebo, or the applicable payment rate of the comparator drug. A single averaged payment would be made regardless of whether 1 mg of study drug, 1 mg of placebo, or 1 mg of comparator drug is used. If the trial is a 1:1 (treatment: placebo) then the payment rate would be the same for every trial participant and would represent half of the total payment for the drug. In a simplified example, if the ASP plus 6 percent payment rate for Drug X was $1 per 1 mg then in this example, the payment rate for the blended code of Drug X and placebo would be $0.50 per 1 mg. If a beneficiary received 100 mg of the study drug, then a $50 payment would be made. If a beneficiary received 100 mg of the placebo, then a $50 payment would be made. The same HCPCS code would be billed in both the study drug and placebo examples. The same payment methodology would apply if the study design was 1:2 (treatment: placebos, which equals payment at 1/3 the cost of the study drug) or 1:3 (treatment: placebos, which equals payment at 1/4 the cost of the study drug).
In situations where there are multi-arm, or single-arm, cross over trials where participants receive placebo, or sham, for the first half of the trial and then the study drug for the second half of the trial, the payment would be reflective of this, and set in the same manner as a 1:1 trial, since half of the time the beneficiary would receive the placebo and the other half they would receive the study drug. No matter the trial design, CMS payment would be reflective of the expected frequency with which the study treatment, control, active comparator, or placebo is provided. We noted that we proposed to assign payment rates based on an adjusted payment level representing the frequency with which the study drug and placebo, or comparator drug, is projected to be furnished for the trial as a whole, and not necessarily the exact frequency with which the study drug and placebo, or comparator drug, is furnished to a particular hospital enrolled as a clinical trial site. Clinical trial sponsors should work with CMS to ensure timely establishment of payment and coding for drugs being studied under a CED designation requiring an adjusted level of payment.
While the items and services furnished as placebo controls may not be considered reasonable and necessary under section 1862(a)(1)(A) of the Act because they have no health benefit, these items and services can be necessary in order to conduct a scientifically valid clinical study. As such, these items can be covered under section 1862(a)(1)(E) of the Act when furnished in the context of a qualifying clinical study.[180]
CMS may cover and pay for routine costs of an approved clinical trial in both the treatment arm and the control (standard of care or placebo). Routine costs include all items and services that are otherwise generally available to Medicare beneficiaries ( i.e., there exists a benefit category for the item or service, coverage is not statutorily excluded for the item or service, and there is not a national non-coverage decision for the item or service) that are provided in either the experimental or the control ( printed page 94306) arms of a clinical trial. Although CMS may cover and pay for routine costs of an CED approved clinical trial in both the treatment arm and the control (standard of care or placebo), there may be circumstances, such as single arm studies, where no unique coding or unique payment would be required to preserve the scientific validity of such a study created for routine costs associated with clinical trials. Similarly, if the routine costs are the exact same between different arms of a trial, and routine billing and payment of those routine costs would not unblind a study, then no unique coding or payment would be required for those costs. There would be no need to include these routine costs in the HCPCS code assigned to a blended payment rate. If covered, these routine costs would be paid according to existing coding and Medicare payment mechanisms. Under the proposal, an alternate method of payment would be established only when necessary to maintain the scientific validity of the trial, such as to prevent the billing and payment of routine costs from unblinding the trial. These determinations would be made based on the clinical trial protocol communicated to CMS by the clinical trial sponsor, before CMS would establish an appropriate code with an adjusted payment level for routine costs for CED trials. CMS's determination in CED trials would differ from our policy regarding devices and procedures in Category B IDE trials, where the provision of an investigational device usually requires a combination of procedures or services to implant, or administer, the device to a patient. In contrast, the infusion of a drug is typically a more straightforward process, and associated routine costs may not be provided at the same time that the drug is administered, making it impractical to create a single code to describe the study drug and all associated routine costs.
Finally, we wanted to be sure there are no other instances where Medicare payment methodologies might interfere with the scientific validity of a trial. We sought comment on these possible alternative scenarios, such as Medicare payment interfering with clinical trial recruitment in such a way that could compromise the scientific integrity of a clinical trial and would consider adjustments to our payment policy for devices in Category B IDE clinical trials and devices/drugs in clinical trials with a CED designation in future rulemaking.
We proposed to codify our coding and payment policy to Category B IDE clinical trials with control arms through revisions to § 419.47(a) to specify that these are placebo control arms. We also proposed to codify our proposed process for developing coding and payment for devices/drugs in CED-designated clinical trials by adding new paragraphs (c) and (d) to § 419.47. Specifically, we proposed to provide in new § 419.47(c) that CMS would create a new HCPCS code, or revise an existing HCPCS code, to describe a device/drug studied in a clinical trial with the Medicare CED designation, which would include the study device/drug and control arm, when CMS determines it is necessary to establish a CED designation for a device/drug subsequent to a CED NCD. Additionally, in new § 419.47(d) we proposed that when we create a new HCPCS code or revise an existing HCPCS code under proposed paragraph (c), we would make a single payment for the HCPCS code that includes payment for the investigational device/drug and any control.
Comment: Several commenters were not supportive of our proposal because they believed the single blended payment rate would lower payments and disincentivize provider participation in clinical trials for drugs and devices with a CED designation. One commenter stated that the practical application of this policy would reduce the pool of eligible providers to those participating in settings such as academic medical centers and facilities that are already performing a high volume of procedures. The commenter further explained that if clinical trial sites were limited to academic medical centers and other facilities performing a high volume of procedures, the commenter believed that communities of color and rural communities would disproportionally face access issues, due to the use of the CED paradigm.
Response: We appreciate the commenters' input and will continue to consider the implications of a single blended payment rate to hospital outpatient departments for drugs and devices in clinical trials covered under an NCD with a CED designation, including the access and health equity impacts.
Comment: A comment signed by 18 organizations explained that it was unethical to require patients to participate in a study so that they might get access to a drug that is already FDA approved. Another commenter believed it was unethical for patients to pay for a placebo when there is an FDA approved treatment available.
Response: We appreciate the input and are sympathetic to the commenters' concerns. Although we appreciate the viewpoint of commenters expressing concern for assigning a CED designation to FDA approved drugs, we note that the comments are out of scope because they do not relate to the proposed payment methodology for CED drugs and devices. With regard to the comment stating that it was unethical for patients enrolled in clinical trials to pay for a placebo, we need additional time to consider the ethical implications of requiring a 20 percent coinsurance for all study participants, as proposed, including those that do not receive the CED drug or device. We welcome public engagement and comments on this final rule with comment period regarding the implications of requiring a coinsurance payment for beneficiaries participating in trials for CED drugs and devices, as well as the relevant statutory authorities that may allow for an adjustment of the coinsurance amount for beneficiaries participating in clinical trials for CED drugs and devices. We will continue to consider the ethical implications as part of future policymaking on payments for clinical trials.
Comment: Several commenters did not support CMS applying CED designation to drugs and biologics that are already approved by the FDA because they believe it would impede beneficiary access to medicines that FDA has already determined to be safe and effective. Two commenters stated that our proposed policy seemed to indicate that more FDA-approved drugs and biologics will be covered by Medicare under the CED pathway in the future. Commenters urged CMS to exercise caution, maintain very limited use of CED for Part B drugs, and not use CED to conduct control arm studies or to prevent access to FDA approved treatments.
Response: We appreciate hearing the public comments and concerns about the potential coverage of drugs and biologicals in future CED trials. However, these comments are outside the scope of our proposed rule because they relate to the coverage pathway for certain drugs and devices rather than the proposed payment methodology for CED drugs and devices. Because the comments relate to coverage decisions that are entirely separate from the proposed payment methodology for CED drugs and devices, we did not consider the comments submitted for this final rule with comment period. We refer readers to the “Coverage with Evidence Development: Guidance Document” issued on August 7, 2024, for more ( printed page 94307) information on CMS's policy for making NCDs using the CED paradigm.[181]
Comment: We received several comments, including from a drug manufacturer, supporting our proposal for both CED drugs and devices. Commenters stated that they believed it was necessary for CMS to finalize this policy to preserve the scientific integrity of future trials. One commenter explained that the policy's emphasis on scientific validity outweighed the small chance of lower payment for trial sites that perform more placebo or sham services versus intervention services. A couple of commenters supported our proposal and requested additional billing and coding guidance for trial sites, as well as timely, transparent, and efficient implementation of the policy.
Response: We thank the commenters for their support. While we agree that the proposed payment methodology for CED drugs and devices may have the effect of preserving the scientific integrity of CED trials, other commenters raised concerns regarding the impact of the proposed policy on provider payments, study enrollment, and beneficiary coinsurance amounts for study enrollees, that need to be further considered prior to finalizing a payment policy. Because we are not finalizing the proposed rule at this time, we will consider the comments if we undertake future rulemaking.
After consideration of the public comments we received, we are not finalizing our proposal to use our authority at section 1833(w) of the Act to develop alternative methods of payment under Medicare Part B for drugs and devices being studied in clinical trials under a CED NCD at this time. We believe that there are broader policy implications to our proposal that require further consideration prior to finalizing a payment policy for CED drugs and devices. For example, we need additional time to consider the ethical implications of requiring a coinsurance payment for all beneficiaries participating in a clinical trial for a CED drug or device, including those that do not receive the investigational treatment. We also need additional time to consider the potential impacts of a single blended payment rate on clinical trial enrollment for drugs and devices in CED trials. We will carefully consider the issues raised in the public comments received in developing a payment policy for CED drugs and devices for future rulemaking.
Accordingly, we are not revising the regulation text at § 419.47 as proposed to include a payment methodology for CED drugs and devices. We are finalizing our proposal to codify our coding and payment policy for Category B IDE clinical trials with control arms through revisions to § 419.47(a) to specify that our policy applies only to IDE studies with a control arm and where a payment adjustment is necessary to preserve the scientific validity of such a study.
XI. CY 2025 OPPS Payment Status and Comment Indicators
A. CY 2025 OPPS Payment Status Indicator Definitions
Payment status indicators (SIs) that we assign to HCPCS codes and APCs serve an important role in determining payment for services under the OPPS. They indicate whether a service represented by a HCPCS code is payable under the OPPS or another payment system and whether particular OPPS policies apply to the code.
For CY 2025 and subsequent years, we proposed to create two new status indicators, “K1” and “H1.” We proposed these two new status indicators to identify the products that qualify for separate payment under our new payment policy for non-opioid post-surgical pain management drugs, biologicals, and devices, as authorized by section 4135 of the Consolidated Appropriations Act, 2023. This policy is discussed further in section XIII.E of this final rule. The final definitions and payment status of status indicators “K1” and “H1” can be found in Table 145.
For CY 2025 and subsequent years, we proposed to modify the definition of status indicator “K” to remove the word “therapeutic” from the phrase “therapeutic radiopharmaceuticals” to indicate that both diagnostic and therapeutic radiopharmaceuticals may be assigned to status indicator “K” in accordance with our policy proposal in section II.A.3.a. of this final rule. The final definition and payment status of status indicator “K” can be found in Table 146.
( printed page 94308)We solicited public comments on the proposed definitions of the OPPS payment status indicators for 2025. We did not propose to make any other changes to the existing definitions of status indicators that were listed in Addendum D1 to this final rule with comment period, which is available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/HospitalOutpatientPPS/Hospital-Outpatient-Regulations-and-Notices.
Comment: Commenters were supportive of our proposed creation of status indicators “K1” and “H1” to identify the products that qualify for separate payment under our new payment policy for non-opioid post-surgical pain management drugs, biologicals, and devices, as authorized by section 4135 of the Consolidated Appropriations Act, 2023.
Response: We thank the commenters for their support.
Comment: We did not receive any specific comments regarding our proposal to revise status indicator “K” to remove the word “therapeutic” from the phrase “therapeutic radiopharmaceuticals” to indicate that both diagnostic and therapeutic radiopharmaceuticals may be assigned to status indicator “K” in accordance with our policy proposal in section II.A.3.a. of the CY 2025 OPPS/ASC proposed rule (89 FR 59427 through 59433). However, commenters were generally supportive of our proposals to pay separately for qualifying diagnostic radiopharmaceuticals as discussed in section II.A.3.a. of this final rule with comment period.
Response: We thank the commenters for their support.
After consideration of the public comments received on our proposal related to the definitions of the status indicators “K1,” “H1,” and “K,” we are finalizing our proposal without modification.
Additionally, in order to conform with the policy being finalized in section X.F. of this final rule with comment period, we are revising status indicator “A” to list HCPCS code J0799 (FDA approved prescription drug, only for use as HIV pre-exposure prophylaxis (not for use as treatment of HIV), not otherwise classified) in addition to HCPCS code C9399 (Unclassified drugs or biologicals) as the finalized policy treats the two unlisted drug related codes in a similar manner. Therefore, the definition for status indicator “A” will now read: “Unclassified drugs and biologicals reportable under HCPCS code C9399 and not otherwise classified FDA-approved prescription drugs for HIV PrEP reported under HCPCS code J0799.”
We are also finalizing additional policies that exclude certain items or services from C-APC packaging, impacting the definitions of status indicators for comprehensive-APCs, as described in section II.A.2.b. We are revising the “J1” and “J2” descriptors to refer to the most recent OPPS Addendum J for a list of the C-APC exclusions rather than listing them all in the status indicator definition. Therefore, status indicator “J1” will be revised to: “Paid under OPPS; all covered Part B services on the claim are packaged with the primary ‘J1’ service for the claim, except the Comprehensive APC payment policy exclusions found in the most recent Addendum J.” Similarly, status indicator “J2” will be revised to: “(1) Comprehensive APC payment based on OPPS comprehensive-specific payment criteria. Payment for all covered Part B services on the claim is packaged into a single payment for specific combinations of services, except the Comprehensive APC payment policy exclusions found in the most recent Addendum J.”
The complete list of CY 2025 payment status indicators and their definitions is displayed in Addendum D1 to this final rule with comment period, which is available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
The CY 2025 payment status indicator assignments for APCs and HCPCS codes are shown in Addendum A and Addendum B, respectively, to this final rule with comment period, which are available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
B. CY 2025 Comment Indicator Definitions
We proposed to use four comment indicators for the CY 2025 OPPS. These comment indicators, “CH,” “NC,” “NI,” and “NP,” are in effect for CY 2024; and we proposed to continue their use in CY 2025. The proposed CY 2025 OPPS comment indicators are as follows:
- “CH”—Active HCPCS code in current and next calendar year, status indicator and/or APC assignment has changed; or active HCPCS code that will be discontinued at the end of the current calendar year.
- “NC”—New code for the next calendar year or existing code with substantial revision to its code descriptor in the next calendar year, as compared to current calendar year for which we requested comments in the proposed rule; final APC assignment; comments will not be accepted on the final APC assignment for the new code.
- “NI”—New code for the next calendar year or existing code with substantial revision to its code descriptor in the next calendar year, as compared to current calendar year, interim APC assignment; comments will be accepted on the interim APC assignment for the new code.
- “NP”—New code for the next calendar year or existing code with substantial revision to its code descriptor in the next calendar year, as compared to current calendar year, proposed APC assignment; comments will be accepted on the proposed APC assignment for the new code.
The definitions of the OPPS comment indicators for CY 2025 are listed in Addendum D2 to this final rule with comment period, which is available on ( printed page 94309) the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices.
We solicited public comments on our proposed definitions of the OPPS comment indicators for 2025.
We did not receive any public comments on our proposal and therefore, we are finalizing those definitions without modification for CY 2025.
XII. MedPAC Recommendations
The Medicare Payment Advisory Commission (MedPAC) was established under section 1805 of the Act in large part to advise the U.S. Congress on issues affecting the Medicare program. As required under the statute, MedPAC submits reports to the Congress no later than March and June of each year that present its Medicare payment policy recommendations. The March report typically provides discussion of Medicare payment policy across different payment systems and the June report typically discusses selected Medicare issues. We are including this section to make stakeholders aware of certain MedPAC recommendations for the OPPS and ASC payment systems as discussed in its March 2024 report.
A. OPPS Payment Rates Update
The March 2024 MedPAC “Report to the Congress: Medicare Payment Policy,” recommended that Congress update Medicare OPPS payment rates by the amount specified in current law plus 1.5 percent. We refer readers to the March 2024 report for a complete discussion of this recommendation.[182] We appreciate MedPAC's recommendation and, as discussed further in section II.B of this final rule with comment period, we proposed to increase the OPPS payment rates by the amount specified in current law. Comments received from MedPAC on other OPPS policies are discussed in the applicable sections of this final rule with comment period.
B. Medicare Safety Net Index
In the March 2024 MedPAC “Report to the Congress: Medicare Payment Policy,” MedPAC stated that their recommended update to IPPS and OPPS payment rates of current law plus 1.5 percent may not be sufficient to ensure the financial viability of some Medicare safety-net hospitals with a poor payer mix. MedPAC recommends redistributing the current Medicare safety-net payments (disproportionate share hospital and uncompensated care payments) using the MedPAC-developed Medicare Safety-Net Index (MSNI) for hospitals. In addition, MedPAC recommends adding $4 billion to this MSNI pool of funds to help maintain the financial viability of Medicare safety-net hospitals and recommended to the Congress transitional approaches for a MSNI policy.
As discussed in the FY 2025 IPPS/LTCH PPS final rule with comment period (89 FR 70045), we look forward to working with Congress on these matters. In the FY 2024 IPPS/LTCH PPS proposed rule, we sought comments on the challenges faced by safety-net hospitals and potential approaches to help safety net hospitals meet those challenges. These comments will inform and guide our future rulemaking and other actions in this area.
C. ASC Cost Data
In the March 2024 MedPAC “Report to the Congress: Medicare Payment Policy,” MedPAC reiterated its longstanding recommendation that Congress require ASCs to report cost data to enable the Commission to examine the growth of ASCs' costs over time and analyze Medicare payments relative to the costs of efficient providers. MedPAC suggested that such cost data would enable policymakers to establish payment rates that accurately reflect ASC costs and are also necessary to determine whether an existing Medicare market basket is an appropriate proxy for ASC costs or whether an ASC-specific market basket should be developed, stating both the CPI-U and hospital market basket update likely do not reflect an ASC's cost structure. MedPAC contended that it is feasible for small facilities, such as ASCs, to provide cost information since other small facilities, such as home health agencies, hospices, and rural health clinics, currently furnish cost data to CMS. Further, ASCs in Pennsylvania submit cost and revenue data annually to a state agency to estimate margins for those ASCs, and that, as businesses, ASCs keep records of their costs for filing taxes and other purposes.[183]
While we recognize that the submission of cost data could place additional administrative burden on most ASCs, and we did not propose any cost reporting requirements for ASCs in the CY 2025 OPPS/ASC proposed rule, as in previous years, we sought public comment on methods that would mitigate the burden of reporting costs on ASCs while also collecting enough data to reliably use such data in the determination of ASC costs. Such cost data would be beneficial in establishing an ASC-specific market basket for updating payment rates under the ASC payment system.
Comment: A trade group representing ASCs suggested that a starting point on reporting ASC costs could be an effort to identify and calculate expense categories as a percentage of total expenses to help determine the appropriate weights and price proxies for the ASC setting. Further, the commenter stated that cost variability can differ greatly based on specialty, facility size, geographic location, and capital equipment of the facility. One commenter recommended that any such cost data collection should be used to inform a market basket index that is applied to both HOPDs and ASCs to ensure payments are aligned over time.
Response: We appreciate the commenters' thoughtful responses. We will take these comments into consideration in future rulemaking.
Comments received from MedPAC for other ASC payment system policies are discussed in the applicable sections of this final rule with comment period.
XIII. Updates to the Ambulatory Surgical Center (ASC) Payment System
A. Background, Legislative History, Statutory Authority, and Prior Rulemaking for the ASC Payment System
For a detailed discussion of the legislative history and statutory authority related to payments to ASCs under Medicare, we refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74377 through 74378) and the June 12, 1998 proposed rule (63 FR 32291 through 32292). For a discussion of prior rulemaking on the ASC payment system, we refer readers to the CYs 2012 to 2024 OPPS/ASC final rules with comment period (76 FR 74378 through 74379; 77 FR 68434 through 68467; 78 FR 75064 through 75090; 79 FR 66915 through 66940; 80 FR 70474 through 70502; 81 FR 79732 through 79753; 82 FR 59401 through 59424; 83 FR 59028 through 59080; 84 FR 61370 through 61410; 85 FR 86121 through 86179; 86 FR 63761 through 63815; 87 FR 72054 through 72096; and 88 FR 81900 through 81961). ( printed page 94310)
B. ASC Treatment of New and Revised Codes
1. Background on Process for New and Revised HCPCS Codes
We update the lists and payment rates for covered surgical procedures and covered ancillary services in ASCs in conjunction with the annual proposed and final rulemaking process to update the OPPS and the ASC payment systems (§ 416.173; 72 FR 42535). We base ASC payment and policies for most covered surgical procedures, drugs, biologicals, and certain other covered ancillary services on the OPPS payment policies and we use quarterly change requests (CRs) to update services paid for under the OPPS. We also provide quarterly update CRs for ASC covered surgical procedures and covered ancillary services throughout the year (January, April, July, and October). We release new and revised Level II HCPCS codes and recognize the release of new and revised CPT codes by the American Medical Association (AMA) and make these codes effective (that is, the codes are recognized on Medicare claims) via these ASC quarterly update CRs. We recognize the release of new and revised Category III CPT codes in the July and January CRs. These updates implement newly created and revised Level II HCPCS and Category III CPT codes for ASC payments and update the payment rates for separately paid drugs and biologicals based on the most recently submitted ASP data. New and revised Category I CPT codes, except vaccine codes, are released only once a year, and are implemented only through the January quarterly CR update. New and revised Category I CPT vaccine codes are released twice a year and are implemented through the January and July quarterly CR updates. We refer readers to Table 41 in the CY 2012 OPPS/ASC proposed rule for an example of how this process is used to update HCPCS and CPT codes, which we finalized in the CY 2012 OPPS/ASC final rule with comment period (76 FR 42291; 76 FR 74380 through 74384).
In our annual updates to the ASC list of, and payment rates for, covered surgical procedures and covered ancillary services, we undertake a review of excluded surgical procedures, new codes, and codes with revised descriptors, to identify any that we believe meet the criteria for designation as ASC covered surgical procedures or covered ancillary services. Updating the lists of ASC covered surgical procedures and covered ancillary services, as well as their payment rates, in association with the annual OPPS rulemaking cycle, is particularly important because the OPPS relative payment weights and, in some cases, payment rates, are used as the basis for the payment of many covered surgical procedures and covered ancillary services under the revised ASC payment system. This joint update process ensures that the ASC updates occur in a regular, predictable, and timely manner.
Payment for ASC procedures, services, and items are generally based on medical billing codes, specifically, HCPCS codes, that are reported on ASC claims. The HCPCS is divided into two principal subsystems, referred to as Level I and Level II. Level I is comprised of CPT (Current Procedural Terminology) codes, a numeric and alphanumeric coding system maintained by the AMA, and includes Category I, II, and III CPT codes. Level II of the HCPCS, which is maintained by CMS, is a standardized coding system that is used primarily to identify products, supplies, and services not included in the CPT codes. Together, Level I and II HCPCS codes are used to report procedures, services, items, and supplies under the ASC payment system. Specifically, we recognize the following codes on ASC claims:
- Category I CPT codes, which describe surgical procedures, diagnostic and therapeutic services, and vaccine codes;
- Category III CPT codes, which describe new and emerging technologies, services, and procedures; and
- Level II HCPCS codes (also known as alpha-numeric codes), which are used primarily to identify drugs, devices, supplies, temporary procedures, and services not described by CPT codes.
We finalized a policy in the August 2, 2007 final rule (72 FR 42533 through 42535) to evaluate each year all new and revised Category I and Category III CPT codes and Level II HCPCS codes that describe surgical procedures, and to make preliminary determinations during the annual OPPS/ASC rulemaking process regarding whether or not they meet the criteria for payment in the ASC setting as covered surgical procedures and, if so, whether or not they are office-based procedures. In addition, we identify new and revised codes as ASC covered ancillary services based upon the final payment policies of the revised ASC payment system. In prior rulemakings, we refer to this process as recognizing new codes. However, this process has always involved the recognition of new and revised codes. We consider revised codes to be new when they have substantial revision to their code descriptors that necessitate a change in the current ASC payment indicator. To clarify, we refer to these codes as new and revised in this CY 2025 OPPS/ASC final rule.
We have separated our discussion below based on when the codes are released and whether we propose to solicit public comments in the proposed rule (and respond to those comments in this final rule with comment period) or whether we will be soliciting public comments in this CY 2025 OPPS/ASC final rule with comment period (and responding to those comments in the CY 2026 OPPS/ASC final rule with comment period).
2. April 2024 HCPCS Codes Proposed Rule Comment Solicitation
For the April 2024 update, there were no new CPT codes; however, there were several new Level II HCPCS codes. In the April 2024 ASC quarterly update (Transmittal 12559, dated March 28, 2024, CR 13577), we added several new Level II HCPCS codes to the list of covered ancillary services. Table 147 (New Level II HCPCS Codes for Ancillary Services Effective April 1, 2024) of the CY 2025 OPPS/ASC proposed rule (89 FR 59408), displayed the new Level II HCPCS codes that were implemented April 1, 2024. These new codes that were effective April 1, 2024, were assigned to comment indicator “NP” in Addendum BB to the proposed rule to indicate that the codes were assigned to an interim APC assignment and that comments would be accepted on their interim APC assignments. In addition, we note that the entire ASC addenda, which consist of the addenda listed below, are available via the internet on the CMS website, specifically, at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices:
- ASC Addendum AA: ASC Covered Surgical Procedures (Including Surgical Procedures for Which Payment is Packaged).
- ASC Addendum BB: ASC Covered Ancillary Services Integral to Covered Surgical Procedures (Including Ancillary Services for Which Payment is Packaged).
- ASC Addendum DD1: ASC Payment Indicators (PI).
- ASC Addendum DD2: ASC Comment Indicators (CI).
- ASC Addendum EE: Surgical Procedures to be Excluded from Payment in ASCs
- ASC Addendum FF: ASC Device Offset Percentages.
- Addendum O: Long Descriptors for New Category I CPT Codes, Category III ( printed page 94311) CPT Codes, C-codes, and G-Codes Effective January 1, 2025.
We invited public comments on the proposed payment indicators for the new HCPCS codes that were recognized as ASC covered ancillary services in April 2024 through the quarterly update CRs, as listed in Table 147 (New Level II HCPCS Codes for Ancillary Services Effective April 1, 2024). The new codes that were effective April 1, 2024, were assigned to comment indicator “NP” in ASC Addendum BB to the CY 2025 OPPS/ASC proposed rule to indicate that the codes are assigned to interim payment indicators and comments would be accepted on their interim assignments. We proposed to finalize the payment indicators in this CY 2025 OPPS/ASC final rule with comment period. We did not receive any comments on the proposed ASC payment indicator assignments for the new Level II HCPCS codes implemented in April 2024 and are finalizing the proposed ASC payment indicator assignments for these codes.
We note that several of the temporary drug HCPCS C-codes have been replaced with permanent drug HCPCS J-codes. Their replacement codes are also listed in Table 147. In addition, although in prior years we included the final ASC payment indicators in the coding tables in the preamble, because we include the same information in the ASC addenda, we have not included them in Table 147. Therefore, readers are advised to refer to the ASC addenda for the final ASC payment indicators and payment rates for all codes reported under the ASC payment system. The list of ASC payment indicators and definitions used under the ASC payment system can be found in the ASC addenda. We note that the ASC addenda (AA, BB, DD1, DD2, EE, and FF) are available via the internet on the CMS website.
( printed page 94312)3. July 2024 HCPCS Codes Proposed Rule Comment Solicitation
In the July 2024 ASC quarterly update (Transmittal 12673, Change Request 13656, dated June 13, 2024), we added several separately payable CPT and Level II HCPCS codes to the list of covered surgical procedures and covered ancillary services. Table 76 (New HCPCS Codes for Covered Surgical Procedures and Covered Ancillary Services Effective July 1, 2024) of the CY 2025 OPPS/ASC proposed rule (89 FR 59409), displayed the new HCPCS codes that are effective July 1, 2024. We invited public comments on the proposed payment indicators for these Level II HCPCS codes, and indicated that the proposed comment indicators, payment indicators, and payment rates for these codes were listed in Addendum AA and Addendum BB of the proposed rule. These new codes that were effective July 1, 2024, were assigned to comment indicator “NP” in ASC Addendum AA and Addendum BB to the CY 2025 OPPS/ASC proposed rule to indicate that the codes were assigned to interim payment indicators and comments would be accepted on their interim ( printed page 94313) assignments. We further stated that we proposed to finalize the payment indicators in this CY 2025 OPPS/ASC final rule with comment period. We note that several of the temporary drug HCPCS C-codes have been replaced with HCPCS J-codes. Their replacement codes are also listed in Table 148. In addition, in prior years we included the final ASC payment indicators in the coding preamble tables, however, because the same information can be found in Addendum AA and Addendum BB, we are no longer including them in Table 148. Therefore, readers are advised to refer to the ASC addenda for the final ASC payment indicators and payment rates for all codes reported under the ASC payment system.
We did not receive any comments on the proposed ASC payment indicator assignments for the new Level II HCPCS codes that were added to the list of covered surgical procedures and ancillary services implemented in July 2024. Therefore, we are finalizing the proposed ASC payment indicator assignments for the codes.
( printed page 94314) ( printed page 94315)4. October 2024 HCPCS Codes Final Rule Comment Solicitation
For CY 2025, consistent with our established policy, we proposed in the CY 2025 OPPS/ASC proposed rule (89 FR 59410) that the Level II HCPCS codes that will be effective October 1, 2024, would be flagged with comment indicator “NI” in Addendum BB to the CY 2025 OPPS/ASC final rule with comment period to indicate that we have assigned the codes an interim ASC payment status for CY 2025. In the October 2024 ASC quarterly update (Transmittal 12824, Change Request 13800, dated September 5, 2024), we added several separately payable Level II HCPCS codes to the list of covered surgical procedures and covered ancillary services. Table 149 below lists the codes that were effective October 1, 2024. We are inviting public comments on this final rule with comment period on the interim payment indicators, which would be finalized in the CY 2026 OPPS/ASC final rule with comment period. We note these codes will be subject to comment in the CY 2026 OPPS/ASC proposed rule with comment period, which would be finalized in the CY 2026 OPPS/ASC final rule with comment period.
5. January 2025 HCPCS Codes
a. New Level II HCPCS Codes Final Rule Comment Solicitation
As has been our practice in the past, we incorporate those new Level II HCPCS codes that are effective January 1 in the final rule with comment period, thereby updating the ASC payment system for the calendar year. We note that unlike the CPT codes that are effective January 1 and are included in the OPPS/ASC proposed rules, and except for the G-codes listed in Addendum O to the CY 2025 OPPS/ASC proposed rule, most Level II HCPCS codes are not released until sometime around November to be effective January 1. Because these codes are not available until November, we are unable to include them in the OPPS/ASC proposed rule; however, the codes are flagged with comment indicator “NI” in ASC Addendum AA and Addendum BB to this final rule with comment period to indicate that we are assigning them an interim payment status, which is subject to public comment. Therefore, as we stated in the CY 2025 OPPS/ASC proposed rule, these Level II HCPCS codes that will be effective January 1, 2025, are included in this final rule with comment period and will also be released to the public through in the January 2025 ASC Update CR and the CMS HCPCS website. We are inviting public comments in this final rule with comment period on the payment indicator assignments, which would be finalized in the CY 2026 OPPS/ASC final rule with comment period. Similar to the codes effective October 1, 2024, these new Level II HCPCS codes that will be effective January 1, 2025, will be subject to comment in the CY 2026 OPPS/ASC proposed rule, which would ( printed page 94316) be finalized in the CY 2026 OPPS/ASC final rule with comment period.
b. New CY 2025 CPT Codes Proposed Rule Comment Solicitation
For the CY 2025 ASC update, we received the CPT codes that will be effective January 1, 2025, from the AMA in time to be included in the CY 2025 OPPS/ASC proposed rule. The new, revised, and deleted CPT codes can be found in ASC Addendum AA and Addendum BB to the CY 2024 OPPS/ASC proposed rule, which are available via the internet on the CMS website. We note that the new and revised CPT codes are assigned to comment indicator “NP” in ASC Addendum AA and Addendum BB of the CY 2025 OPPS/ASC proposed rule to indicate that the code is new for the next calendar year, or the code is an existing code with substantial revision to its code descriptor in the next calendar year as compared to the current calendar year with a proposed payment indicator assignment. We stated that we would accept comments and finalize the payment indicators in this CY 2025 OPPS/ASC final rule with comment period. Further, we remind readers that the CPT code descriptors that appeared in Addendum AA and Addendum BB are short descriptors and do not describe the complete procedure, service, or item described by the CPT code. Therefore, we included the 5-digit placeholder codes and their long descriptors for the new CY 2025 CPT codes in Addendum O to the CY 2025 OPPS/ASC proposed rule so that the public could comment on our proposed payment indicator assignments. The 5-digit placeholder codes were listed in Addendum O to the CY 2025 OPPS/ASC proposed rule, specifically under the column labeled “CY 2025 OPPS/ASC Proposed Rule 5-Digit Placeholder Code.” We also stated that we would include the final CPT code numbers in this CY 2025 OPPS/ASC final rule with comment period.
We did not receive any comments on the proposed ASC payment indicators for the new CPT codes effective January 1, 2025, so we are finalizing these codes as proposed.
Finally, in Table 150, we summarize our process for updating codes through our ASC quarterly update CRs, seeking public comments, and finalizing the treatment of these new codes under the ASC payment system.
6. ASC Payment and Comment Indicators
a. Background
In addition to the payment indicators that we introduced in the August 2, 2007 ASC final rule, we created final comment indicators for the ASC payment system in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66855). We created Addendum DD1 to define ASC payment indicators that we use in Addenda AA and BB to provide payment information regarding covered surgical procedures and covered ancillary services, respectively, under the revised ASC payment system. The ASC payment indicators in Addendum DD1 are intended to capture policy-relevant characteristics of HCPCS ( printed page 94317) codes that may receive packaged or separate payment in ASCs, such as whether they were on the ASC CPL prior to CY 2008; payment designation, such as device-intensive or office-based, and the corresponding ASC payment methodology; and their classification as separately payable ancillary services, including radiology services, brachytherapy sources, OPPS pass-through devices, corneal tissue acquisition services, drugs or biologicals, or NTIOLs.
We also created Addendum DD2 that lists the ASC comment indicators. The ASC comment indicators included in Addenda AA and BB to the proposed rules and final rules with comment period serve to identify, for the revised ASC payment system, the status of a specific HCPCS code and its payment indicator with respect to the timeframe when comments will be accepted. The comment indicator “NI” is used in the OPPS/ASC final rule with comment period to indicate new codes for the next calendar year for which the interim payment indicator assigned is subject to comment. The comment indicator “NI” also is assigned to existing codes with substantial revisions to their descriptors such that we consider them to be describing new services, and the interim payment indicator assigned is subject to comment, as discussed in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60622).
The comment indicator “NP” is used in the OPPS/ASC proposed rule to indicate new codes for the next calendar year for which the proposed payment indicator assigned is subject to comment. The comment indicator “NP” also is assigned to existing codes with substantial revisions to their descriptors, such that we consider them to be describing new services, and the proposed payment indicator assigned is subject to comment, as discussed in the CY 2016 OPPS/ASC final rule with comment period (80 FR 70497).
The “CH” comment indicator is used in Addenda AA and BB to the proposed rule (these addenda are available via the internet on the CMS website) to indicate that the payment indicator assignment has changed for an active HCPCS code in the current year and the next calendar year, for example, if an active HCPCS code is newly recognized as payable in ASCs or an active HCPCS code is discontinued at the end of the current calendar year. The “CH” comment indicators that are published in the final rule are provided to alert readers that a change has been made from one calendar year to the next, but do not indicate that the change is subject to comment.
In the CY 2021 OPPS/ASC final rule with comment period, we finalized the addition of ASC payment indicator “K5”—Items, Codes, and Services for which pricing information and claims data are not available. No payment made.—to ASC Addendum DD1 (which is available via the internet on the CMS website) to indicate those services and procedures that CMS anticipates will become payable when claims data or payment information becomes available.
In CY 2024 OPPS/ASC final rule with comment period, we finalized the addition of two ASC payment indicators, “D1”—“Ancillary dental service/item; no separate payment made” and “D2”—“Non office-based dental procedure added in CY 2024 or later”, for new dental codes for CY 2024 and subsequent calendar years to indicate potentially payable dental services and procedures in the ASC setting (88 FR 81907). We added these two codes to Addendum DD1 (which is available via the internet on the CMS website).
b. Final ASC Payment and Comment Indicators for CY 2025
For CY 2025, we proposed new and revised Category I and III CPT codes as well as new and revised Level II HCPCS codes. Proposed Category I and III CPT codes that are new and revised for CY 2025 and any new and existing Level II HCPCS codes with substantial revisions to the code descriptors for CY 2025, compared to the CY 2024 descriptors, are included in ASC Addenda AA and BB to the CY 2025 OPPS/ASC proposed rule and labeled with comment indicator “NP” to indicate that these CPT and Level II HCPCS codes are open for comment as part of the CY 2025 OPPS/ASC proposed rule.
We proposed to modify the descriptor of ASC payment indicator “L6”—“New Technology Intraocular Lens (NTIOL); special payment” to “Special payment; New Technology Intraocular Lens (NTIOL) or qualifying non-opioid devices”, to account for non-opioid devices paid for under the ASC payment system pursuant to section 4135 of the CAA, 2023. More information about this non-opioid policy can be found in section XIII.E of this final rule with comment period.
We refer readers to Addenda DD1 and DD2 of this final rule with comment period (these addenda are available via the internet on the CMS website) for the complete list of ASC payment and comment indicators proposed for the CY 2025 update.
We did not receive any public comments on our proposal and therefore, we are finalizing our proposal to modify the descriptor of ASC payment indicator “L6”—“New Technology Intraocular Lens (NTIOL); special payment” to “Special payment; New Technology Intraocular Lens (NTIOL) or qualifying non-opioid devices”, to account for non-opioid devices paid for under the ASC payment system pursuant to section 4135 of the CAA, 2023.
C. Payment Policies Under the ASC Payment System
1. Final ASC Payment for Covered Surgical Procedures
a. Background
Our ASC payment policies for covered surgical procedures under the revised ASC payment system are described in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66828 through 66831). Under our established policy, we use the ASC standard ratesetting methodology of multiplying the ASC relative payment weight for the procedure by the ASC conversion factor for that same year to calculate the national unadjusted payment rates for procedures with payment indicators “G2” and “A2.” Payment indicator “A2” was developed to identify procedures that were included on the list of ASC covered surgical procedures in CY 2007 and, therefore, were subject to transitional payment prior to CY 2011. Although the 4-year transitional period has ended and payment indicator “A2” is no longer required to identify surgical procedures subject to transitional payment, we have retained payment indicator “A2” because it is used to identify procedures that are exempted from the application of the office-based designation.
Payment rates for office-based procedures (payment indicators “P2,” “P3,” and “R2”) are the lower of the PFS nonfacility PE RVU-based amount or the amount calculated using the ASC standard rate setting methodology for the procedure. As detailed in section XIII.C.3.b of this final rule with comment period, we update the payment amounts for office-based procedures (payment indicators “P2,” “P3,” and “R2”) using the most recent available MPFS and OPPS data. We compare the estimated current year rate for each of the office-based procedures, calculated according to the ASC standard rate setting methodology, to the PFS nonfacility PE RVU-based amount to determine which is lower and, therefore, would be the current year payment rate for the procedure under our final policy for the revised ASC payment system (§ 416.171(d)). ( printed page 94318)
The rate calculation established for device-intensive procedures (payment indicator “J8”) is structured so only the service (non-device) portion of the rate is subject to the ASC conversion factor. We update the payment rates for device-intensive procedures to incorporate the most recent device offset percentages calculated under the ASC standard ratesetting methodology, as discussed in section XIII.C.4 of this final rule with comment period.
In the CY 2014 OPPS/ASC final rule with comment period (78 FR 75081), we finalized our proposal to calculate the CY 2014 payment rates for ASC covered surgical procedures according to our established methodologies, with the exception of device removal procedures. For CY 2014, we finalized a policy to conditionally package payment for device removal procedures under the OPPS. Under the OPPS, a conditionally packaged procedure (status indicators “Q1” and “Q2”) describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a covered surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are always packaged (payment indicator “N1”) under the ASC payment system. Under the OPPS, device removal procedures are conditionally packaged and, therefore, would be packaged under the ASC payment system. There is no Medicare payment made when a device removal procedure is performed in an ASC without another surgical procedure included on the claim; therefore, no Medicare payment would be made if a device was removed but not replaced. To ensure that the ASC payment system provides separate payment for surgical procedures that only involve device removal—conditionally packaged in the OPPS (status indicator “Q2”)—we have continued to provide separate payment since CY 2014 and assign the current ASC payment indicators associated with these procedures.
b. Update to ASC Covered Surgical Procedure Payment Rates for CY 2025
We proposed to update ASC payment rates for CY 2025 and subsequent years using the established rate calculation methodologies under § 416.171 and using our definition of device-intensive procedures, as discussed in section XIII.C.4 of this final rule with comment period. As the proposed OPPS relative payment weights are generally based on geometric mean costs, we proposed that the ASC payment system will generally use the geometric mean cost to determine proposed relative payment weights under the ASC standard methodology. We proposed to continue to use the amount calculated under the ASC standard ratesetting methodology for procedures assigned payment indicators “A2” and “G2.”
We proposed to calculate payment rates for office-based procedures (payment indicators “P2,” “P3,” and “R2”) and device-intensive procedures (payment indicator “J8”) according to our established policies and to identify device-intensive procedures using the methodology discussed in section XIII.C.4 of this final rule with comment period. Therefore, we proposed to update the payment amount for the service portion (the non-device portion) of the device-intensive procedures using the standard ASC ratesetting methodology and the payment amount for the device portion based on the proposed CY 2025 device offset percentages that have been calculated using the standard OPPS APC ratesetting methodology. We proposed that payment for office-based procedures would be at the lesser of the proposed CY 2025 MPFS nonfacility PE RVU-based amount or the proposed CY 2025 ASC payment amount calculated according to the ASC standard ratesetting methodology.
As we did for CYs 2014 through 2024, for CY 2025, we proposed to continue our policy for device removal procedures, such that device removal procedures that are conditionally packaged in the OPPS (status indicators “Q1” and “Q2”) will be assigned the current ASC payment indicators associated with those procedures and will continue to be paid separately under the ASC payment system.
Comment: One commenter expressed concern about the lack of a cap on beneficiary coinsurance in the ASC setting while there is a statutory cap on beneficiary coinsurance when a procedure is performed in the HOPD setting. The commenter believes the lack of such a cap poses a financial challenge for beneficiaries, particularly with respect to transitional pass-through devices and higher-cost procedures that are device intensive, because in such cases, the coinsurance could be higher in the ASC setting than in the HOPD setting.
Response: We thank the commenter for its input but note that comments related to statutory changes are out of scope for this final rule.
After consideration of the public comments we received, we are finalizing our proposed policies without modification to calculate the CY 2025 payment rates for ASC covered surgical procedures according to our established rate calculation methodologies under § 416.171 and our device-intensive methodology as discussed in section XIII.C.1.b. of this CY 2025 OPPS/ASC final rule with comment period. For covered office-based surgical procedures, the payment rate is the lesser of the final CY 2025 MPFS nonfacility PE RVU-based amount or the CY 2025 ASC payment amount calculated according to the ASC standard ratesetting methodology. The final payment indicators and rates set forth in this final rule with comment period are based on a comparison using the PFS PE RVUs and the conversion factor effective January 1, 2025.
c. Final Payment for ASC Add-On Procedures Eligible for Complexity Adjustments Under the OPPS
In this section, we discuss the policy to provide increased payment under the ASC payment system for combinations of certain “J1” service codes and add-on procedure codes that are eligible for a complexity adjustment under the OPPS.
(1) OPPS C-APC Complexity Adjustment Policy
Under the OPPS, complexity adjustments are utilized to provide increased payment for certain comprehensive services. As discussed in section II.A.2.b of this final rule with comment period, we apply a complexity adjustment by promoting qualifying paired “J1” service code combinations or paired code combinations of “J1” services and add-on codes from the originating Comprehensive APC (C-APC) (the C-APC to which the designated primary service is first assigned) to the next higher paying C-APC in the same clinical family of C-APCs. A “J1” status indicator refers to a hospital outpatient service paid through a C-APC. We package payment for all add-on codes, which are codes that describe a procedure or service always performed in addition to a primary service or procedure, into the payment for the C-APC. However, certain combinations of primary service codes and add-on codes may qualify for a complexity adjustment.
We apply complexity adjustments when the paired code combination represents a complex, costly form or version of the primary service when the frequency and cost thresholds are met. The frequency threshold is met when there are 25 or more claims reporting the code combination, and the cost threshold is met when there is a violation of the 2 times rule, as specified in section 1833(t)(2) of the Act and ( printed page 94319) described in section III.A.2.b of this final rule with comment period, in the originating C-APC. These paired code combinations that meet the frequency and cost threshold criteria represent those that exhibit materially greater resource requirements than the primary service. After designating a single primary service for a claim, we evaluate that service in combination with each of the other procedure codes reported on the claim that are either assigned to status indicator “J1” or add-on codes to determine if there are paired code combinations that meet the complexity adjustment criteria. Once we have determined that a particular combination of “J1” services, or combinations of a “J1” service and add-on code, represents a complex version of the primary service because it is sufficiently costly, frequent, and a subset of the primary comprehensive service overall according to the criteria described previously, we promote the claim to the next higher cost C-APC within the clinical family unless the primary service is already assigned to the highest cost APC within the C-APC clinical family or assigned to the only C-APC in a clinical family. We do not create new C-APCs with a comprehensive geometric mean cost that is higher than the highest geometric mean cost (or only) C-APC in a clinical family just to accommodate potential complexity adjustments. Therefore, the highest payment for any claim including a code combination for services assigned to a C-APC would be the highest paying C-APC in the clinical family (79 FR 66802).
As previously stated, we package payment for add-on codes into the C-APC payment rate. If any add-on code reported in conjunction with the “J1” primary service code does not qualify for a complexity adjustment, payment for the add-on service continues to be packaged into the payment for the primary service and the primary service code reported with the add-on code is not reassigned to the next higher cost C-APC. We list the final complexity adjustments for “J1” and add-on code combinations for CY 2025, along with all of the other final complexity adjustments, in Addendum J to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices).
(2) CY 2025 ASC Special Payment Policy for OPPS Complexity-Adjusted C-APCs
For CY 2025, we proposed to continue the special payment policy and methodology for OPPS complexity-adjusted C-APCs that was finalized in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72078 through 72080).
For those ASC complexity adjustment codes for which we have claims data, we proposed to use the claims data to calculate the code combination utilization and estimated payments for the ASC payment system budget neutrality calculations for CY 2025. The ASC complexity adjustment budget neutrality calculations are discussed further in section XIII.H.2.a of this final rule with comment period. The full list of the proposed ASC complexity adjustment codes for CY 2025 can be found in the CY 2025 proposed ASC addenda and the supplemental policy file, which also includes both the existing ASC complexity adjustment codes and proposed additions and was published on the CMS website at https://www.cms.gov/medicare/medicare-fee-for-service-payment/ascpayment/asc-regulations-and-notices. Since the complexity adjustment assignments change each year under the OPPS, the proposed list of ASC complexity adjustment codes eligible for the proposed payment policy changed slightly from the previous year.
Comment: A few commenters noted that there was a decline in the number of ASC complexity adjustment codes for the CY 2025 OPPS/ASC proposed rule compared to years prior. The commenters noted that there were 55 ASC complexity adjustment codes in 2023, 47 ASC complexity adjustment codes in 2024, and only 45 ASC proposed complexity adjustment codes for CY 2025. Other commenters recommended specific ASC complexity adjustment codes we did not propose.
Several commenters were concerned with how the device portion for ASC complexity adjustment codes are determined. Commenters believed we were potentially missing large device costs associated with the packaged add-on procedure that could only be realized if the device offset percentage of the ASC complexity adjustment code was calculated from the subset of “J1” primary service and add-on code claim combinations.
Response: Since the number of OPPS complexity adjustments fluctuate on a year-to-year basis, the number of ASC complexity adjustment codes will also fluctuate each calendar year. For this CY 2025 OPPS/ASC final rule with comment period, we are finalizing 49 ASC complexity adjustment codes, which is an increase from the 47 ASC complexity adjustment codes for CY 2024. Additionally, we rely on the complexity adjustment criteria under the OPPS, as described in section II.A.2.b. of this final rule with comment period, for determining eligible ASC complexity adjustment codes under the ASC payment system and do not accept specific code combination recommendations, such as those recommended by commenters, if they do not meet these criteria.
With respect to the commenters' concerns regarding the device portion of ASC complexity adjustment codes, we believe the current methodology of assigning the device portion from the primary “J1” procedure to the ASC complexity adjustment code is the most appropriate. As shown in Addendum J (which can be found on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices), the evaluation subset for a “J1” and add-on code combination is often less than 100 claims and can be significantly less than the total number of claims for that primary “J1” procedure used for ratesetting. We are concerned that using such a small subset of a procedure's available claims for determining the device portion of a device-intensive ASC complexity adjustment code would introduce unnecessary volatility. We believe that assigning a device portion for a device-intensive ASC complexity adjustment code based on the device portion of the primary “J1” procedure accurately reflects the device costs of the primary “J1” procedure—since the device costs associated with the add-on code are already packaged into the cost of the primary procedure—and provides a more stable payment rate by using the greater number of claims that are available from the primary “J1” procedure, as opposed to the subset of claims with a “J1” and add-on code combination. However, we appreciate the commenters' recommendation and may take this comment into consideration in future rulemaking.
After consideration of the public comments we received, we are finalizing the ASC special payment policy for OPPS complexity-adjusted C-APCs, as proposed. The final ASC complexity adjustment codes, based on the most recent data available for this CY 2025 OPPS/ASC final rule with comment period, can be found on the CMS website at: https://www.cms.gov/medicare/payment/prospective- ( printed page 94320) payment-systems/ambulatory-surgical-center-asc/annual-policy-files . Existing ASC complexity adjustment codes that do not meet our criteria for separate payment for CY 2025 because the code combination is not eligible for a complexity adjustment under the OPPS for CY 2025 will be non-payable and assigned a status indicator of “B5”—“Alternative code may be available; no payment made”—for CY 2025. Additionally, proposed ASC complexity adjustment codes that met our criteria based on data available for the CY 2025 OPPS/ASC proposed rule but do not meet our criteria based on claims data available for this final rule with comment period will be deleted and assigned a payment indicator of “D5”—“Deleted/discontinued code; no payment made”.
d. Final Low Volume APCs and Limit on ASC Payment Rates for Procedures Assigned to Low Volume APCs
As stated in section XIII.D.1.b of the CY 2025 OPPS/ASC proposed rule, the ASC payment system generally uses OPPS geometric mean costs under the standard methodology to determine proposed relative payment weights under the standard ASC ratesetting methodology.
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63743 through 63747), we adopted a universal Low Volume APC policy for CY 2022 and subsequent calendar years. Under our policy, we expanded the low volume adjustment policy that is applied to procedures assigned to New Technology APCs to also apply to clinical and brachytherapy APCs. Specifically, a clinical APC or brachytherapy APC with fewer than 100 claims per year would be designated as a Low Volume APC. For items or services assigned to a Low Volume APC, we use up to 4 years of claims data to establish a payment rate for the APC as we currently do for low volume services assigned to New Technology APCs. The payment rate for a Low Volume APC or a low volume New Technology procedure would be based on the highest of the median cost, arithmetic mean cost, or geometric mean cost calculated using multiple years of claims data.
Based on claims data available for the CY 2025 OPPS/ASC proposed rule, we proposed to designate six brachytherapy APCs and four clinical APCs as Low Volume APCs under the ASC payment system (89 FR 59414). The four clinical APCs and six brachytherapy APCs met our criteria of having fewer than 100 single claims in the relevant claims year (CY 2023 for the CY 2025 OPPS/ASC proposed rule) and therefore, we proposed that they would be subject to our universal Low Volume APC policy and the APC cost metric would be based on the greater of the median cost, arithmetic mean cost, or geometric mean cost using up to 4 years of claims data. Nine of the ten APCs were designated as low volume APCs in CY 2024. Based on data for the CY 2025 OPPS/ASC proposed rule, APC 2645 (Brachytx, non-stranded, hold-198) met our criteria to be designated a low volume APC; and we proposed to designate it as such for CY 2025.
Comment: Commenters supported our proposal to assign the ten APCs as Low Volume APCs.
Response: We thank the commenters for their support. Based on claims data available for this final rule with comment period, we are finalizing our proposal to designate the 4 clinical APCs and 6 brachytherapy APCs shown in Table 151 as Low Volume APCs under the ASC payment system, because they continue to meet our criteria of having fewer than 100 single claims in the relevant claims year (2023). Table 151 includes the CY 2023 claims available for ratesetting for each of the APCs we are finalizing to be designated as Low Volume APCs for CY 2025. The cost statistics for our Low Volume APCs, such as the median, arithmetic mean, and geometric mean cost are available for download with this final rule with comment period on the CMS website. We refer readers to our website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/asc-regulations-and-notices; click on the relevant regulation to download the Low Volume APC cost statistics under the standard (ASC) ratesetting methodology in the “Downloads” section of the web page.
( printed page 94321)2. Payment for Covered Ancillary Services
a. Background
Our payment policies under the ASC payment system for covered ancillary services generally vary according to the particular type of service and its payment policy under the OPPS. Our overall policy provides separate ASC payment for certain ancillary items and services integrally related to the provision of ASC covered surgical procedures that are paid separately under the OPPS and provides packaged ASC payment for other ancillary items and services that are packaged or conditionally packaged (status indicators “N,” “Q1,” and “Q2”) under the OPPS.
In the CY 2013 OPPS/ASC rulemaking (77 FR 45169 and 77 FR 68457 through 68458), we further clarified our policy regarding the payment indicator assignment for procedures that are conditionally packaged in the OPPS (status indicators “Q1” and “Q2”). Under the OPPS, a conditionally packaged procedure describes a HCPCS code where the payment is packaged when it is provided with a significant procedure but is separately paid when the service appears on the claim without a significant procedure. Because ASC services always include a surgical procedure, HCPCS codes that are conditionally packaged under the OPPS are generally packaged (payment indictor “N1”) under the ASC payment system (except for device removal procedures, as discussed in the CY 2022 OPPS/ASC proposed rule (86 FR 42083)). Thus, our policy generally aligns ASC payment bundles with those under the OPPS (72 FR 42495). In all cases, in order for ancillary items and services also to be paid, the ancillary items and services must be provided integral to the performance of ASC covered surgical procedures for which the ASC bills Medicare.
Our ASC payment policies generally provide separate payment for drugs and biologicals that are separately paid under the OPPS at the OPPS rates and package payment for drugs and biologicals for which payment is packaged under the OPPS. However, as discussed in the CY 2022 OPPS/ASC final rule with comment period, for CY 2022, we finalized a policy to unpackage and pay separately at ASP plus 6 percent for the cost of non-opioid pain management drugs and biologicals that function as a supply when used in a surgical procedure as determined by CMS under § 416.174 (86 FR 63483).
We generally pay for separately payable radiology services at the lower of the PFS nonfacility PE RVU-based (or technical component) amount or the rate calculated according to the ASC standard ratesetting methodology (72 FR 42497). However, as finalized in the CY 2011 OPPS/ASC final rule with comment period (75 FR 72050), payment indicators for all nuclear medicine procedures (defined as CPT codes in the range of 78000 through 78999) that are designated as radiology services that are paid separately when provided integral to a surgical procedure on the ASC list are set to “Z2” so that payment is made based on the ASC standard ratesetting methodology rather than the MPFS nonfacility PE RVU amount (“Z3”), regardless of which is lower (§ 416.171(d)(1)).
Similarly, we also finalized our policy to set the payment indicator to “Z2” for radiology services that use contrast agents so that payment for these procedures will be based on the OPPS relative payment weight using the ASC standard ratesetting methodology and, therefore, will include the cost for the contrast agent (§ 416.171(d)(2)).
ASC payment policy for brachytherapy sources mirrors the payment policy under the OPPS. ASCs are paid for brachytherapy sources provided integral to ASC covered surgical procedures at prospective rates adopted under the OPPS or, if OPPS rates are unavailable, at contractor-priced rates (72 FR 42499). Since December 31, 2009, ASCs have been paid for brachytherapy sources provided integral to ASC covered surgical procedures at prospective rates adopted under the OPPS.
Our ASC policies also provide separate payment for: (1) certain items and services that CMS designates as contractor-priced, including, but not limited to, the procurement of corneal tissue; and (2) certain implantable items that have pass-through payment status under the OPPS. These categories do not have prospectively established ASC payment rates according to ASC payment system policies (72 FR 42502 and 42508 through 42509; § 416.164(b)). Under the ASC payment system, we have designated corneal tissue acquisition and hepatitis B vaccines as contractor-priced. Corneal tissue acquisition is contractor-priced based on the invoiced costs for acquiring the corneal tissue for transplantation. Hepatitis B vaccines are contractor-priced based on invoiced costs for the vaccine.
Devices that are eligible for pass-through payment under the OPPS are separately paid under the ASC payment system and are contractor-priced. Under the revised ASC payment system (72 FR 42502), payment for the surgical procedure associated with the pass-through device is made according to our standard methodology for the ASC payment system, based on only the service (non-device) portion of the procedure's OPPS relative payment weight if the APC weight for the procedure includes other packaged device costs. We also refer to this methodology as applying a “device offset” to the ASC payment for the associated surgical procedure. This ensures that duplicate payment is not provided for any portion of an implanted device with OPPS pass-through payment status.
In the CY 2015 OPPS/ASC final rule with comment period (79 FR 66933 through 66934), we finalized that, beginning in CY 2015, certain diagnostic tests within the medicine range of CPT codes for which separate payment is allowed under the OPPS are covered ancillary services when they are integral to an ASC covered surgical procedure. We finalized that diagnostic tests within the medicine range of CPT codes include all Category I CPT codes in the medicine range established by CPT, from 90000 to 99999, and Category III CPT codes and Level II HCPCS codes that describe diagnostic tests that crosswalk or are clinically similar to procedures in the medicine range established by CPT. In the CY 2015 OPPS/ASC final rule with comment period, we also finalized our policy to pay for these tests at the lower of the PFS nonfacility PE RVU-based (or technical component) amount or the rate calculated according to the ASC standard ratesetting methodology (79 FR 66933 through 66934). We finalized that the diagnostic tests for which the payment is based on the ASC standard ratesetting methodology be assigned to payment indicator “Z2” and revised the definition of payment indicator “Z2” to include a reference to diagnostic services and those for which the payment is based on the PFS nonfacility PE RVU-based amount be assigned payment indicator “Z3,” and revised the definition of payment indicator “Z3” to include a reference to diagnostic services.
b. Final Payment for Covered Ancillary Services for CY 2025
We proposed to update the ASC payment rates and to make changes to ASC payment indicators, as necessary, to maintain consistency between the OPPS and ASC payment system regarding the packaged or separately payable status of services and the proposed CY 2025 OPPS and ASC ( printed page 94322) payment rates and subsequent years' payment rates. We also proposed to continue to set the CY 2025 ASC payment rates and subsequent years' payment rates for brachytherapy sources and separately payable drugs and biologicals equal to the OPPS payment rates for CY 2025 and subsequent years' payment rates.
Covered ancillary services and their final payment indicators for CY 2025 are listed in Addendum BB of this final rule with comment period (which is available via the internet on the CMS website). For those covered ancillary services where the payment rate is the lower of the rate under the ASC standard rate setting methodology and the PFS final rates (similar to our office-based payment policy), the final payment indicators and rates set forth in this final rule are based on a comparison using the final PFS rates effective January 1, 2025. For a discussion of the PFS rates, we refer readers to the CY 2025 PFS proposed rule.
Comment: One commenter stated that there is currently no CMS-level guidance on how MACs should establish separate payment under the ASC payment system for devices that are eligible for pass-through payment under the OPPS. The commenter recommends that CMS specify that payment should be at least equal to the device cost as reported by the ASC in box 19 on the ASC claim form.
Response: As previously discussed, devices that are eligible for pass-through payment under the OPPS may be separately paid under the ASC payment system and are contractor-priced. Currently, MACs have been instructed to pay for such devices in the ASC setting based on invoice or cost of the approved transitional pass-through device category. While we understand that many products with different price points may use the same approved transitional pass-through device category, we rely on the MACs to efficiently process these claims such that ASCs can be reimbursed in a timely manner for the services provided and that reimbursement for these pass-through devices is justified with supporting documentation. Therefore, we do not believe it would be appropriate to require MACs to provide payment based on the figure reported in box 19 of the ASC claim form. We believe the current guidance provided in Chapter 14, section 50 of the Medicare Claims Processing Manual (Pub. 100-04) is sufficient.
After consideration of the public comments we received, we are finalizing our proposal to update the ASC payment rates and to make changes to ASC payment indicators, as necessary, to maintain consistency between the OPPS and ASC payment system regarding the packaged or separately payable status of services and the final CY 2025 OPPS and ASC payment rates and subsequent years' payment rates.
We did not receive any public comments on and are also finalizing without modification our proposal to continue to set the CY 2025 ASC payment rates and subsequent years' payment rates for brachytherapy sources and separately payable drugs and biologicals equal to the OPPS payment rates for CY 2025 and subsequent years' payment rates.
3. Covered Surgical Procedures Designated as Office-Based Procedures
a. Background
In the August 2, 2007 ASC final rule, we finalized our policy to designate as “office-based” those procedures that are added to the ASC Covered Procedures List (CPL) in CY 2008 or later years that we determine are furnished predominantly (more than 50 percent of the time) in physicians' offices based on consideration of the most recent available volume and utilization data for each individual procedure code and/or, if appropriate, the clinical characteristics, utilization, and volume of related codes. In that final rule, we also finalized our policy to exempt all procedures on the CY 2007 ASC list from application of the office-based classification (72 FR 42512). The procedures that were added to the ASC CPL beginning in CY 2008 that we determined were office-based were identified in Addendum AA to that final rule with payment indicator “P2” (Office-based surgical procedure added to ASC list in CY 2008 or later with MPFS nonfacility PE RVUs; payment based on OPPS relative payment weight); “P3” (Office-based surgical procedures added to ASC list in CY 2008 or later with MPFS nonfacility PE RVUs; payment based on MPFS nonfacility PE RVUs); or “R2” (Office-based surgical procedure added to ASC list in CY 2008 or later without MPFS nonfacility PE RVUs; payment based on OPPS relative payment weight), depending on whether we estimated the procedure would be paid according to the ASC standard ratesetting methodology based on its OPPS relative payment weight or at the MPFS nonfacility PE RVU-based amount.
Consistent with our final policy to annually review and update the ASC CPL to include all covered surgical procedures eligible for payment in ASCs, each year we identify covered surgical procedures as either temporarily office-based (these are new procedure codes with little or no utilization data that we have determined are clinically similar to other procedures that are permanently office-based), permanently office-based, or nonoffice-based, after taking into account updated volume and utilization data.
b. CY 2025 Final Office-Based Procedures
In developing the CY 2025 OPPS/ASC proposed rule, we followed our policy to annually review and update the covered surgical procedures for which ASC payment is made and to identify new procedures that may be appropriate for ASC payment, including their potential designation as office-based. Historically, we also review the most recent claims volume and utilization data (CY 2023 claims) and the clinical characteristics for all covered surgical procedures that are currently assigned a payment indicator in CY 2024 of “G2” (Non office-based surgical procedure added in CY 2008 or later; payment based on OPPS relative payment weight) as well as for those procedures assigned one of the temporary office-based payment indicators, specifically “P2,” “P3,” or “R2” in the CY 2023 OPPS/ASC final rule with comment period (86 FR 63769 through 63773).
Our review of the CY 2023 volume and utilization data of covered surgical procedures currently assigned a payment indicator of “G2” (Non office-based surgical procedure added in CY 2008 or later; payment based on OPPS relative payment weight) resulted in the identification of two surgical procedures that we believed met the criteria for designation as permanently office-based. The data indicated that these procedures are performed more than 50 percent of the time in physicians' offices, and the services are of a level of complexity consistent with other procedures performed routinely in physicians' offices. The CPT codes that we proposed to permanently designate as office-based for CY 2025 were CPT code 0447T (Removal of implantable interstitial glucose sensor from subcutaneous pocket via incision) and CPT code 21127 (Augmentation, mandibular body or angle; with bone graft, onlay or interpositional (includes obtaining autograft)) and are listed in Table 79 of the CY 2025 OPPS/ASC proposed rule (89 FR 59416) with the associated payment indicators.
As discussed in the August 2, 2007 ASC final rule (72 FR 42533 through 42535), we finalized our policy to ( printed page 94323) designate certain new surgical procedures as temporarily office-based until adequate claims data are available to assess their predominant sites of service, whereupon if we confirm their office-based nature, the procedures are permanently assigned to the list of office-based procedures. In the absence of claims data, we use other available information, including our clinical advisors' judgment, predecessor CPT and Level II HCPCS codes, information submitted by representatives of specialty societies and professional associations, and information submitted by commenters during the public comment period.
We reviewed CY 2023 volume and utilization data for nine surgical procedures designated as temporarily office-based in the CY 2023 OPPS/ASC final rule with comment period and temporarily assigned one of the office-based payment indicators, specifically “P2,” “P3,” or “R2.” In Table 122 of the CY 2024 OPPS/ASC final rule with comment period, we finalized assigning temporary office-based designations to seven surgical procedures for CY 2024 (88 FR 81919). In Table 80 of the CY 2025 OPPS/ASC proposed rule (89 FR 59417), we showed the surgical procedure we proposed to no longer designate as temporarily office-based. This surgical procedure is CPT code 67516 (Suprachoroidal space injection of pharmacologic agent (separate procedure)) which we proposed to permanently designate as office-based.
We did not receive any public comments on our proposed permanent office-based designations and therefore, we are finalizing our proposal to designate the procedures in Table 152 as permanently office-based beginning in CY 2025.
In Table 81 of the CY 2025 OPPS/ASC proposed rule (89 FR 59418), we showed the nine surgical procedures, both new and existing, we proposed to designate as temporarily office-based for CY 2025. For CY 2025, we proposed to designate three new CY 2025 CPT codes for ASC covered surgical procedures as temporarily office-based—CPT codes 0910T (placeholder code XX34T) (Removal of integrated neurostimulation system, vagus nerve), 15013 (placeholder code 15XX3) (Preparation of skin cell suspension autograft, requiring enzymatic processing, manual mechanical disaggregation of skin cells, and filtration; first 25 sq cm or less of harvested skin), and 53866 (placeholder code 5XX06) (Catheterization with removal of temporary device for ischemic remodeling ( i.e., pressure necrosis) of bladder neck and prostate). After reviewing the clinical characteristics, utilization, and volume of related procedure codes, we determined that CPT code 0910T is most similar to 0588T, which is temporarily designated as an office-based surgical procedure. Additionally, CPT code 15013 is most similar to CPT code 11310 (Shaving of epidermal or dermal lesion, single lesion, face, ears, eyelids, nose, lips, mucous membrane; lesion diameter 0.5 cm or less), which is designated as an office-based surgical procedure. Lastly, CPT code 53866 is most similar to CPT code 51705 (Change of bladder tube) which is also designated as an office-based surgical procedure. We also proposed to continue to designate six existing procedures as temporarily office-based for CY 2025 since we did not have adequate claims data to remove the temporary designation.
Comment: One commenter recommended that we do not finalize our office-based designation for CPT code 15013 and reassign CPT code 15013 from “R2” (Office-based surgical procedure added to ASC list in CY 2008 or later without MPFS nonfacility PE RVUs; payment based on OPPS relative payment weight) to “J8” (Device-intensive procedure; paid at adjusted rate).
Response: We are not accepting this recommendation. We do not believe this procedure meets our requirements of a device-intensive procedure as described in section XIII.C.4. of this final rule with comment period because it does not use an insertable or implantable single-use device.
After consideration of the public comments we received, we are finalizing our proposal with modification. We are not finalizing our proposed temporary office-based designation to CPT code 0910T (placeholder code XX34T) (Removal of ( printed page 94324) integrated neurostimulation system, vagus nerve). As discussed in section III.E of this final rule with comment period, since the vagal nerve neurostimulation system has not yet received FDA approval, we are finalizing an OPPS status indicator “E1” to indicate that the code is not payable by Medicare when submitted on outpatient claims (any outpatient bill type). Therefore, for the ASC payment system, we are finalizing a payment indicator of “Y5”—“Nonsurgical procedure/item not valid for Medicare purposes because of coverage, regulation and/or statute; no payment made” for CY 2025.
Further, we are also not finalizing our temporary office-based designation of CPT code 65785 (Implantation of intrastromal corneal ring segments) as this code meets our criteria to be designated as device-intensive for CY 2025 and we are finalizing that designation for this CPT code. While we are finalizing the payment indicators to the other new and existing procedures which are designated as temporary office-based shown in Table 153, we are also modifying our proposal with the addition of three newly created surgical procedure codes: HCPCS codes C8002 (Preparation of skin cell suspension autograft, automated, including all enzymatic processing and device components (do not report with manual suspension preparation)), G0564 (Creation of subcutaneous pocket with insertion of 365 day implantable interstitial glucose sensor, including system activation and patient training), and G0565 (Removal of implantable interstitial glucose sensor with creation of subcutaneous pocket at different anatomic site and insertion of new 365 day implantable sensor, including system activation). We believe HCPCS C8002 is most similar to CPT code 15013 (Preparation of skin cell suspension autograft, requiring enzymatic processing, manual mechanical disaggregation of skin cells, and filtration; first 25 sq cm or less of harvested skin), a CPT code to which we are also finalizing a temporary office-based designation for CY 2025. Additionally, we believe HCPCS codes G0564 and G0565 are most similar to CPT codes 0446T (Creation of subcutaneous pocket with insertion of implantable interstitial glucose sensor, including system activation and patient training) and 0448T (Removal of implantable interstitial glucose sensor with creation of subcutaneous pocket at different anatomic site and insertion of new implantable sensor, including system activation), respectively. Both of these CPT codes are designated as office-based procedures; therefore, we are assigning a temporary office-based designation to HCPCS codes G0564 and G0545 as well. The procedures for which the final office-based designation for CY 2025 is temporary are also indicated by an asterisk in Addendum AA to this final rule with comment period (which is available via the internet on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/ASCPayment/ASC-Regulations-and-Notices).
( printed page 94325) ( printed page 94326)4. Device-Intensive ASC Covered Surgical Procedures
a. Background
We refer readers to the CY 2019 OPPS/ASC final rule with comment period (83 FR 59040 through 59041), for a summary of our existing policies regarding ASC covered surgical procedures that are designated as device-intensive.
b. CY 2025 Final Device Intensive Procedures
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59040 through 59043), for CY 2019, we modified our criteria for device-intensive procedures to better capture costs for procedures with significant device costs. We adopted a policy to allow procedures that involve surgically inserted or implanted, high-cost, single-use devices to qualify as device-intensive procedures. In addition, we modified our criteria to lower the device offset percentage threshold from 40 percent to 30 percent. The device offset percentage is the percentage of device costs within a procedure's total costs. Specifically, for CY 2019 and subsequent years, we adopted a policy that device-intensive procedures would be subject to the following criteria:
- All procedures must involve implantable or insertable devices assigned a CPT or HCPCS code;
- The required devices (including single-use devices) must be surgically inserted or implanted; and
- The device offset amount must be significant, which is defined as exceeding 30 percent of the procedure's mean cost. Corresponding to this change in the cost criterion, we adopted a policy that the default device offset for new codes that describe procedures that involve the implantation of medical devices will be 31 percent beginning in CY 2019. For new codes describing procedures that are payable when furnished in an ASC and involve the implantation of a medical device, we adopted a policy that the default device offset would be applied in the same manner as the policy we adopted in section IV.B.2 of the CY 2019 OPPS/ASC final rule with comment period (83 FR 58944 through 58948). We amended § 416.171(b)(2) of the regulations to reflect these new device criteria.
In addition, as also adopted in section IV.B.2 of the CY 2019 OPPS/ASC final rule with comment period, to further align the device-intensive policy with the criteria used for device pass-through status, we specified, for CY 2019 and subsequent years, that for purposes of satisfying the device-intensive criteria, a device-intensive procedure must involve a device that:
- Has received FDA marketing authorization, has received an FDA investigational device exemption (IDE) ( printed page 94327) and has been classified as a Category B device by FDA in accordance with 42 CFR 405.203 through 405.207 and 405.211 through 405.215, or meets another appropriate FDA exemption from premarket review;
- Is an integral part of the service furnished;
- Is used for one patient only;
- Comes in contact with human tissue;
- Is surgically implanted or inserted (either permanently or temporarily); and
- Is not any of the following:
++ Equipment, an instrument, apparatus, implement, or item of this type for which depreciation and financing expenses are recovered as depreciable assets as defined in Chapter 1 of the Medicare Provider Reimbursement Manual (CMS Pub. 15-1); or
++ A material or supply furnished incident to a service (for example, a suture, customized surgical kit, scalpel, or clip, other than a radiological site marker).
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63773 through 63775), we modified our approach to assigning device-intensive status to surgical procedures under the ASC payment system. First, we adopted a policy of assigning device-intensive status to procedures that involve surgically inserted or implanted, high-cost, single-use devices if their device offset percentage exceeds 30 percent under the ASC standard ratesetting methodology, even if the procedure is not designated as device-intensive under the OPPS. Second, we adopted a policy that if a procedure is assigned device-intensive status under the OPPS, but has a device offset percentage below the device-intensive threshold under the standard ASC ratesetting methodology, the procedure will be assigned device-intensive status under the ASC payment system with a default device offset percentage of 31 percent. The policies were adopted to provide consistency between the OPPS and ASC payment system and provide a more appropriate payment rate for surgical procedures with significant device costs under the ASC payment system.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72078 through 72080), we finalized our policy to create certain C-codes, or ASC complexity adjustment codes that describe certain combinations of a primary covered surgical procedure as well as a packaged (payment indicator = “N1”) procedure that are otherwise eligible for a complexity adjustment under the OPPS (as listed in Addendum J). Each ASC complexity adjustment code's APC assignment is based on its corresponding OPPS complexity adjustment code's APC assignment. In the CY 2023 OPPS/ASC final rule with comment period, we stated our belief that it would be appropriate for these ASC complexity adjustment codes to qualify for device-intensive status under the ASC payment system if the primary procedure of the code was also designated as device-intensive. Under our current policy, the ASC complexity adjustment code retains the device portion of the primary procedure (also called the “device offset amount”) and not the device offset percentage. Therefore, for device-intensive ASC complexity adjustment codes, we set the device portion of the combined procedure equal to the device portion of the primary procedure and calculate the device offset percentage by dividing the device portion by the ASC complexity adjustment code's APC payment rate. Further, we apply our standard ASC payment system ratesetting methodology to the non-device portion of the ASC complexity adjustment code's APC payment rate; that is, we multiply the OPPS relative weight by the ASC budget neutrality adjustment and the ASC conversion factor and sum that amount with the device portion to calculate the ASC payment rate.
As discussed in section IV.B of the CY 2025 OPPS/ASC proposed rule, the purpose of applying the default device offset percentage to new codes that describe procedures that implant or insert devices is to ensure access in the ASC setting for new procedures until claims data become available. Our ratesetting methodology sets the ASC device offset amount constant at the OPPS device offset amount. Device offset amounts under the OPPS and ASC Payment System are the device offset percentages of a procedure multiplied by the OPPS or ASC Payment System payment rate, respectively, for that procedure. While the ASC ratesetting methodology relies on the ASC conversion factor and the scaled OPPS APC relative weights to construct ASC payment rates, for device-intensive procedures, the device offset percentage of the procedure relies on the higher OPPS conversion factor while the non device portion relies on the lower ASC conversion factor. For non device-intensive procedures for which the payment is based on OPPS relative payment weight, one hundred percent of the procedure's payment rate relies on the ASC conversion factor. Therefore, the greater the device offset percentage under the ASC Payment System, the greater the ASC payment rate.
Device offset percentages, which represent the device cost portion of a procedure's total cost, are determined using the most recent claims data for that procedure. For newer procedures that describe procedures which implant or insert single-use devices that meet our definition of a device and for which the device costs are estimated to be greater than 30 percent of the total procedure cost and lack claims data, we have relied on several policies to determine an appropriate device offset percentage until such claims data becomes available. First, if the new procedure has claims data from a predecessor code, as described by CPT coding guidance, we rely on claims data from the predecessor code in assigning the device offset percentage for the new HCPCS code (88 FR 81919 through 81922). Second, in limited instances where a new device-intensive procedure does not have a predecessor code as defined by CPT, but describes a procedure that was previously described by an existing code, we may use clinical discretion to identify HCPCS codes that are clinically related or similar to the new HCPCS code but are not officially recognized as a predecessor code by CPT, and use the claims data of the clinically related or similar code(s) for purposes of determining whether to use the device offset percentage of the clinically related or similar code(s) or to apply the default device offset to the new HCPCS code (83 FR 58946). Clinically related and similar procedures for purposes of this policy are procedures that have few or no clinical differences and use the same device(s). If the new device-intensive procedure does not have claims data from a predecessor code or a clinically similar code that uses the same device, we have assigned a default device offset percentage of 31 percent. While we do allow for additional information in our consideration of a higher offset percentage than the default device offset, our payment policies under both the OPPS and ASC Payment System are meant to encourage efficiencies and promote savings to the Medicare program and we believe relying on claims data rather than external pricing data helps put downward pressure on changes in medical device prices. Therefore, it would be extremely rare that the appropriate determination of a device offset percentage would rely on pricing data or invoices from a device manufacturer rather than the default device offset percentage.
However, we are aware that there may be certain situations where newer ( printed page 94328) device-intensive procedures lack claims data from a predecessor code and a clinically similar code that uses the same device, but the default device offset percentage would not adequately reflect the existing device portion of the procedure's costs when compared to the cost of similar devices. The difference in the default device portion and the potential device cost could possibly limit access to newer, more complex, device-intensive procedures in the ASC setting if the cost of the new device does indeed reflect a cost equivalent to that of the similar existing devices. As HOPDs and ASCs perform new procedures with significant device costs, we believe it is appropriate to modify our default device offset methodology to pay HOPDs and ASCs more appropriately when we lack claims data for these newer procedures. Therefore, we proposed to modify our default device offset percentage for new device-intensive procedures. Specifically, for all new covered surgical HCPCS codes that describe procedures which implant or insert single-use devices that meet our definition of a device and for which the device costs are estimated to be greater than 30 percent of the total procedure cost and lack claims data, we would apply a default device offset percentage that is the greater of: 31 percent or the device offset percentage of the APC to which the procedure has been assigned. We proposed this methodological change for both the OPPS and ASC Payment System for CY 2025 and subsequent calendar years.
We still believe that a HCPCS code-level device offset is, in most cases, a more accurate representation of a procedure's device cost than an APC-wide average device offset based on the average device offset of all the procedures assigned to an APC. However, because newer device-intensive procedures lack claims data and therefore a HCPCS code-level device offset may not be possible, we believe the APC-wide average device offset percentage is, in most cases, a better reflection of the estimated device costs of the procedure than a default 31 percent offset. Additionally, there can be instances where the typical device costs of procedures in an APC can be significantly greater than the 31 percent default device offset. For these reasons, we propose to modify our methodology for determining the device offset percentage for new procedures that describe the implantation or insertion of a single-use device that meet our definition of a device and for which the device cost is projected to be greater than 30 percent of the total procedure cost that do not yet have associated claims data to apply a device offset percentage that is the greater of 31 percent or the device offset percentage of the APC to which the procedure has been assigned. The proposal would apply to new device-intensive procedures assigned to clinical APCs and would not apply to new procedures assigned to New Technology APCs.
Under our proposal, we would continue to first rely on the associated claims data for the new HCPCS code or any predecessor code, as described by CPT coding guidance, for the new HCPCS code. If there is no claims data from the new HCPCS or any predecessor code, we may continue to use clinical discretion to identify HCPCS codes that are clinically related or similar to the new HCPCS code but are not officially recognized as a predecessor code by CPT, and to use the claims data of the clinically related or similar code(s) for purposes of determining a device offset percentage to the new HCPCS code (83 FR 58946). Clinically related and similar procedures for purposes of this policy are procedures that have few or no clinical differences and use the same devices. For new device-intensive procedures that describe the implantation or insertion of a single-use device that meet our definition of a device and for which the device cost is significant, projected to be greater than 30 percent of the total procedure cost, and lack claims data, we would then rely on our proposed device offset policy and apply the greater of 31 percent or the device offset percentage of the APC to which the procedure has been assigned.
We solicited comments on our proposed changes to our default device offset policy for CY 2025 and subsequent calendar years under the OPPS and ASC payment system. The listing of proposed payment indicators for covered surgical procedures as well as their respective proposed device offset percentages and device offset amounts, which incorporates our proposed changes to the default device offset policy, can be found in Addendum FF to the proposed rule (which is available via the internet on the CMS website).
Comment: Commenters strongly supported our proposed change to our default device offset policy for CY 2025 and subsequent years under the ASC payment system.
Response: We thank the commenters for their support. After consideration of the public comments we received, we are finalizing our proposal without modification to apply the greater of the APC-wide device offset percentage or 31 percent for new HCPCS codes that do not have claims data or any predecessor code as described by CPT coding guidance.
Comment: Many commenters recommended that we assign device-intensive status to HCPCS code C9757 (Laminotomy (hemilaminectomy), with decompression of nerve root(s), including partial facetectomy, foraminotomy and excision of herniated intervertebral disc, and repair of annular defect with implantation of bone anchored annular closure device, including annular defect measurement, alignment and sizing assessment, and image guidance; 1 interspace, lumbar) and CPT codes 52284 (Cystourethroscopy, with mechanical urethral dilation and urethral therapeutic drug delivery by drug-coated balloon catheter for urethral stricture or stenosis, male, including fluoroscopy, when performed) and 30469 (Repair of nasal valve collapse with low energy, temperature-controlled ( i.e., radiofrequency) subcutaneous/submucosal remodeling) under the ASC payment system. The commenters argued that the proposed ASC payment rate would not adequately reimburse for the cost of the devices that are used during the procedures.
Response: As we discussed in section IV.B. of this final rule with comment period, out of concern for hospital nonreporting of device costs for procedures that require devices, we are finalizing a modification to our device edits policy which reinstates device edits for device-intensive procedures created on or after January 1, 2017. Additionally, such device edits will be permanent and will apply regardless of the procedure's device offset percentage. In conjunction with the modification to our device edits policy for CY 2025, for determining device offset percentages for procedures subject to our device edits policy for CY 2025 that cannot bypass such edit by reporting modifier “CG”, we are relying only on claims from hospitals that reported a device code for determining device offset percentages under the OPPS and ASC payment systems. Based on our revised methodology for this CY 2025 OPPS/ASC final rule with comment period, the device offset percentages for HCPCS code C9757 and CPT codes 30469 and 52284 exceed our device-intensive threshold. Therefore, we are assigning such procedures device-intensive status under the ASC payment system for CY 2025.
Comment: One commenter recommended that we assign device-intensive status to CPT codes 0686T ( printed page 94329) (Histotripsy (ie, non-thermal ablation via acoustic energy delivery) of malignant hepatocellular tissue, including image guidance) and 0888T (Histotripsy (ie, non-thermal ablation via acoustic energy delivery) of malignant renal tissue, including imaging guidance). While the commenter states the procedure is not invasive, the commenter argues the procedure does require a disposable product.
Response: We are not accepting this recommendation as this procedure does not meet our requirements for a device-intensive assignment. As previously discussed, among other requirements, a device-intensive procedure must involve a device that is surgically implanted or inserted (either permanently or temporarily).
Comment: One commenter recommended that we assign CPT code 58356 (Endometrial cryoablation with ultrasonic guidance, including endometrial curettage, when performed) device-intensive status, or, at the least, to revise the payment indicator to “A2”—Surgical procedure on ASC list in CY 2007; payment based on OPPS relative payment weight.
Response: We are not accepting this recommendation. CPT code 58356 does not have a device offset percentage that exceeds our device-intensive threshold. Additionally, CPT code 58356 has been designated as permanently office-based surgical procedure since CY 2010.
Comment: One commenter recommended that we use device cost data from CPT code 64590 (Insertion or replacement of peripheral, sacral, or gastric neurostimulator pulse generator or receiver, requiring pocket creation and connection between electrode array and pulse generator or receiver) to determine the device offset percentage for CPT code 0786T (Insertion or replacement of percutaneous electrode array, sacral, with integrated neurostimulator, including imaging guidance, when performed). The commenter argues the predecessor code for 0786T is CPT code 64590.
Response: After further review of the CPT coding guidance associated with the neurostimulator codes that were created for CY 2024, we believe applicable predecessor codes for CPT code 0786T could include both CPT code 64590 and CPT code 64561 (Percutaneous implantation of neurostimulator electrode array; sacral nerve (transforaminal placement) including image guidance, if performed). However, as discussed in section III.E of this final rule with comment period, we inadvertently listed the integrated sacral nerve stimulation system associated with CPT code 0786T as receiving FDA-approval. However, it is still pending FDA approval and therefore, we are finalizing an OPPS status indicator of “E1” until FDA approval has been received. Therefore, for the ASC payment system, we are not accepting the commenter's recommendation and we are finalizing a payment indicator of “Y5”—“Nonsurgical procedure/item not valid for Medicare purposes because of coverage, regulation and/or statute; no payment made” for CY 2025.
Comment: One commenter recommended that we use the device cost data from HCPCS code C9769 (Cystourethroscopy, with insertion of temporary prostatic implant/stent with fixation/anchor and incisional struts) to determine the device offset percentage for CPT code 53865 (Cystourethroscopy with insertion of temporary device for ischemic remodeling (ie, pressure necrosis) of bladder neck and prostate) (placeholder CPT code 5XX05). The commenter argues the predecessor code for CPT code 53865 is HCPCS code C9769.
Response: After further review of the CPT coding guidance associated with newly created CPT code 53865, we agree that the predecessor code for CPT code 53865 includes HCPCS code C9769 as well as unlisted procedures. Therefore, we are accepting the commenter's recommendation and assigning a device offset percentage for CPT code 53865 based on its predecessor code HCPCS code C9769.
Comment: One commenter supported the device offset percentage for HCPCS code C9781 (Arthroscopy, shoulder, surgical; with implantation of subacromial spacer ( e.g., balloon), includes debridement ( e.g., limited or extensive), subacromial decompression, acromioplasty, and biceps tenodesis when performed) while another commenter recommended that we not reduce the device offset percentage from 64.4% to 61.5% under the ASC payment system.
Response: We appreciate the commenters' feedback and are relying on available claims data for this final rule with comment period in our determination of device-intensive status and device offset percentages under the ASC payment system. For final CY 2025 device offset percentages based on claims data for this final rule with comment period, we refer readers to Addendum FF of this final rule with comment period.
Comment: One commenter recommended we reevaluate the device portion and device-intensive status for the following CPT codes:
- 36563 (Insertion of tunneled centrally inserted central venous access device with subcutaneous pump);
- 36568 (Insertion of peripherally inserted central venous catheter (picc), without subcutaneous port or pump, without imaging guidance; younger than 5 years of age);
- 36570 (Insertion of peripherally inserted central venous access device, with subcutaneous port; younger than 5 years of age);
- 36578 (Replacement, catheter only, of central venous access device, with subcutaneous port or pump, central or peripheral insertion site); and
- 36595 (Mechanical removal of pericatheter obstructive material (e.g, fibrin sheath) from central venous device via separate venous access).
The commenter expressed their concerned with the magnitude of the payment reduction with CPT code 36595, a permanently office-based surgical procedure, experiencing a proposed 78.3 percent reduction from CY 2024 as a result of losing device-intensive status and a proposed payment rate based on the proposed CY 2025 PFS rate.
Response: Based on claims data available for this final rule with comment period, CPT codes 36563, 36568, 36570, and 36595 do not have device offset percentages that exceed the device-intensive threshold of greater than 30 percent. Therefore, we are not assigning device-intensive status to these procedures for CY 2025. However, the device offset percentage for CPT code 36578 exceeds our device-intensive threshold of 30 percent. Therefore, we are maintaining the CY 2024 device-intensive designation of CPT code 36578 for CY 2025.
Regarding the proposed payment rate for CPT code 36595, we believe the stakeholder brings up an important issue regarding the volatility in our payment rates for surgical procedures that are permanently designated as office-based but contain device costs that fluctuate above and below our threshold for device-intensive status. In previous rulemaking, we have stated that office-based surgical procedures that meet our requirements for device-intensive status will be assigned device-intensive status rather than office-based status for the applicable calendar year. However, since this procedure has been assigned office-based status and, based on the most recent claims data available, does not have significant device costs, we believe reassigning the procedure its office-based status for CY 2025 is appropriate. While we believe the number of office-based procedures that may gain or lose device-intensive status ( printed page 94330) each year is very low, we will monitor payment fluctuations for these procedures going forward and may address these concerns in future rulemaking.
Comment: Many commenters supported the proposed device offset percentages for the following procedures:
- CPT code 0671T (Insertion of anterior segment aqueous drainage device into the trabecular meshwork, without external reservoir, and without concomitant cataract removal, one or more);
- CPT code 0816T (Open insertion or replacement of integrated neurostimulation system for bladder dysfunction including electrode(s) (e.g., array or leadless), and pulse generator or receiver, including analysis, programming, and imaging guidance, when performed, posterior tibial nerve; subcutaneous);
- CPT code 0817T (Open insertion or replacement of integrated neurostimulation system for bladder dysfunction including electrode(s) (e.g., array or leadless), and pulse generator or receiver, including analysis, programming, and imaging guidance, when performed, posterior tibial nerve; subfascial);
- CPT code 31242 (Nasal/sinus endoscopy, surgical; with destruction by radiofrequency ablation, posterior nasal nerve);
- CPT code 31298 (Nasal/sinus endoscopy, surgical, with dilation (e.g., balloon dilation); frontal and sphenoid sinus ostia);
- CPT code 66174 (Transluminal dilation of aqueous outflow canal (e.g., canaloplasty); without retention of device or stent);
- CPT code 66989 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1-stage procedure), manual or mechanical technique (e.g., irrigation and aspiration or phacoemulsification), complex, requiring devices or techniques not generally used in routine cataract surgery ( e.g., iris expansion device, suture support for intraocular lens, or primary posterior capsulorrhexis) or performed on patients in the amblyogenic developmental stage; with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more); and
- CPT code 66991 (Extracapsular cataract removal with insertion of intraocular lens prosthesis (1 stage procedure), manual or mechanical technique (e.g., irrigation and aspiration or phacoemulsification); with insertion of intraocular ( e.g., trabecular meshwork, supraciliary, suprachoroidal) anterior segment aqueous drainage device, without extraocular reservoir, internal approach, one or more).
Response: We appreciate the commenters' support of the proposed device offset percentages. For these CPT codes, we are finalizing device offset percentages based on claims data available for this final rule with comment period. CPT codes that were designated as device-intensive based on claims data available for the proposed rule may or may not be designated as device-intensive based on claims data available for this final rule with comment period.
For final CY 2025 device offset percentages based on claims data for this final rule with comment period, we refer readers to Addendum FF of this final rule with comment period.
c. Adjustment to ASC Payments for No Cost/Full Credit and Partial Credit Devices
Our ASC payment policy for costly devices implanted or inserted in ASCs at no cost/full credit or partial credit is set forth in § 416.179 of our regulations and is consistent with the OPPS policy that was in effect until CY 2014. We refer readers to the CY 2008 OPPS/ASC final rule with comment period (72 FR 66845 through 66848) for a full discussion of the ASC payment adjustment policy for no cost/full credit and partial credit devices. ASC payment is reduced by 100 percent of the device offset amount when a hospital furnishes a specified device without cost or with a full credit and by 50 percent of the device offset amount when the hospital receives partial credit in the amount of 50 percent or more of the cost for the specified device.
Effective CY 2014, under the OPPS, we finalized our proposal to reduce OPPS payment for applicable APCs by the full or partial credit a provider receives for a device, capped at the device offset amount. Although we finalized our proposal to modify the policy of reducing payments when a hospital furnishes a specified device without cost or with full or partial credit under the OPPS, in the CY 2014 OPPS/ASC final rule with comment period (78 FR 75076 through 75080), we finalized our proposal to maintain our ASC policy for reducing payments to ASCs for specified device-intensive procedures when the ASC furnishes a device without cost or with full or partial credit. Unlike the OPPS, there is currently no mechanism within the ASC claims processing system for ASCs to submit to CMS the amount of the actual credit received when furnishing a specified device at full or partial credit. Therefore, under the ASC payment system, we finalized our proposal for CY 2014 to continue to reduce ASC payments by 100 percent or 50 percent of the device offset amount when an ASC furnishes a device without cost or with full or partial credit, respectively.
Under current ASC policy, all ASC device-intensive covered surgical procedures are subject to the no cost/full credit and partial credit device adjustment policy. Specifically, when a device-intensive procedure is performed to implant or insert a device that is furnished at no cost or with full credit from the manufacturer, the ASC appends the HCPCS “FB” modifier on the line in the claim with the procedure to implant or insert the device. The contractor reduces payment to the ASC by the device offset amount that we estimate represents the cost of the device when the necessary device is furnished without cost or with full credit to the ASC. We continue to believe that the reduction of ASC payment in these circumstances is necessary to pay appropriately for the covered surgical procedure furnished by the ASC.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59043 through 59044) we adopted a policy to reduce the payment for a device-intensive procedure for which the ASC receives partial credit by one-half of the device offset amount that would be applied if a device was provided at no cost or with full credit if the credit to the ASC is 50 percent or more (but less than 100 percent) of the cost of the new device. The ASC will append the HCPCS “FC” modifier to the HCPCS code for the device-intensive surgical procedure when the facility receives a partial credit of 50 percent or more (but less than 100 percent) of the cost of a device. To report that the ASC received a partial credit of 50 percent or more (but less than 100 percent) of the cost of a new device, ASCs have the option of either: (1) submitting the claim for the device-intensive procedure to their Medicare contractor after the procedure's performance, but prior to manufacturer acknowledgment of credit for the device, and subsequently contacting the contractor regarding a claim adjustment, once the credit determination is made; or (2) holding the claim for the device implantation or insertion procedure until a determination is made by the manufacturer on the partial credit and submitting the claim with the “FC” modifier appended to the implantation procedure HCPCS code if the partial ( printed page 94331) credit is 50 percent or more (but less than 100 percent) of the cost of the device. Beneficiary coinsurance would be based on the reduced payment amount. As finalized in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66926), to ensure our policy covers any situation involving a device-intensive procedure where an ASC may receive a device at no cost or receive full credit or partial credit for the device, we apply our “FB”/“FC” modifier policy to all device-intensive procedures.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59043 through 59044) we stated we would reduce the payment for a device-intensive procedure for which the ASC receives partial credit by one-half of the device offset amount that would be applied if a device was provided at no cost or with full credit, if the credit to the ASC is 50 percent or more (but less than 100 percent) of the cost of the device. In the CY 2020 OPPS/ASC final rule with comment period, we finalized continuing our existing policies for CY 2020. We note that we inadvertently omitted language that this policy would apply not just in CY 2019 but also in subsequent calendar years. We intended to apply this policy in CY 2019 and subsequent calendar years. Therefore, we finalized our proposal to apply our policy for partial credits specified in the CY 2019 OPPS/ASC final rule with comment period (83 FR 59043 through 59044) in CY 2022 and subsequent calendar years (86 FR 63775 through 63776). Specifically, for CY 2022 and subsequent calendar years, we would reduce the payment for a device-intensive procedure for which the ASC receives partial credit by one-half of the device offset amount that would be applied if a device was provided at no cost or with full credit, if the credit to the ASC is 50 percent or more (but less than 100 percent) of the cost of the device. To report that the ASC received a partial credit of 50 percent or more (but less than 100 percent) of the cost of a device, ASCs have the option of either: (1) submitting the claim for the device intensive procedure to their Medicare contractor after the procedure's performance, but prior to manufacturer acknowledgment of credit for the device, and subsequently contacting the contractor regarding a claim adjustment, once the credit determination is made; or (2) holding the claim for the device implantation or insertion procedure until a determination is made by the manufacturer on the partial credit and submitting the claim with the “FC” modifier appended to the implantation procedure HCPCS code if the partial credit is 50 percent or more (but less than 100 percent) of the cost of the device. Beneficiary coinsurance would be based on the reduced payment amount.
We did not receive any comments on our policies related to no cost/full credit or partial credit devices, and we are finalizing the continuation of our existing policies for CY 2025 without modification.
5. Requirement in the Physician Fee Schedule CY 2025 Proposed Rule for HOPDs and ASCs To Report Discarded Amounts of Certain Single-Dose or Single-Use Package Drugs
Section 90004 of the Infrastructure Investment and Jobs Act (Pub. L. 117-9, November 15, 2021) (“the Infrastructure Act”) amended section 1847A of the Act to re-designate subsection (h) as subsection (i) and insert a new subsection (h), which requires manufacturers to provide a refund to CMS for certain discarded amounts from a refundable single-dose container or single-use package drug.
The CY 2025 PFS proposed rule included proposals related to the discarded drug refund policy, including proposals that may impact hospital outpatient departments (HOPDs) and ambulatory surgical centers (ASCs). Similar to our CY 2023 and CY 2024 notice in the OPPS/ASC proposed rules (87 FR 71988 and 88 FR 49760), we included a notice in the CY 2025 proposed rule (89 FR 59421) to ensure interested parties were aware of these proposals and knew to refer to the CY 2025 Physician Fee Schedule proposed rule for a full description of the proposed policy. Interested parties were asked to submit comments on any proposals to implement section 90004 of the Infrastructure Act to the CY 2025 PFS proposed rule. We stated that public comments on these proposals would be addressed in the CY 2025 PFS final rule with comment period. We note that this same notice appeared in section V.B.6 of the CY 2025 OPPS/ASC proposed rule (89 FR 59370).
Please see a full description of the finalized policy in section V.B.6 of this CY 2025 OPPS/ASC final rule with comment period.
D. Additions to ASC Covered Surgical Procedures and Covered Ancillary Services Lists
1. Additions to the List of ASC Covered Surgical Procedures
Section 1833(i)(1) of the Act requires us, in part, to specify, in consultation with appropriate medical organizations, surgical procedures that are appropriately performed on an inpatient basis in a hospital but that can also be safely performed in an ASC, a CAH, or an HOPD, and to review and update the list of ASC covered surgical procedures at least every 2 years. We evaluate the ASC covered procedures list (ASC CPL) each year to determine whether procedures should be added to or removed from the list, and changes to the list are often made in response to specific concerns raised by stakeholders.
Under our regulations at §§ 416.2 and 416.166, covered surgical procedures furnished on or after January 1, 2022, are surgical procedures that meet the general standards specified in § 416.166(b) and are not excluded under the general exclusion criteria specified in § 416.166(c). Specifically, under § 416.166(b), the general standards provide that covered surgical procedures are surgical procedures specified by the Secretary and published in the Federal Register and/or via the internet on the CMS website that are separately paid under the OPPS, that would not be expected to pose a significant safety risk to a Medicare beneficiary when performed in an ASC, and for which standard medical practice dictates that the beneficiary would not typically be expected to require active medical monitoring and care at midnight following the procedure.
Section 416.166(c) sets out the general exclusion criteria used under the ASC payment system to evaluate the safety of procedures for performance in an ASC. The general exclusion criteria provide that covered surgical procedures do not include those surgical procedures that: (1) generally result in extensive blood loss; (2) require major or prolonged invasion of body cavities; (3) directly involve major blood vessels; (4) are generally emergent or life-threatening in nature; (5) commonly require systemic thrombolytic therapy; (6) are designated as requiring inpatient care under § 419.22(n); (7) can only be reported using a CPT unlisted surgical procedure code; or (8) are otherwise excluded under § 411.15.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59029 through 59030), we defined a surgical procedure under the ASC payment system as any procedure described within the range of Category I CPT codes that the CPT Editorial Panel of the AMA defines as “surgery” (CPT codes 10000 through 69999) (72 FR 42476), as well as procedures that are described by Level II HCPCS codes or by Category I CPT codes or by Category III CPT codes that directly crosswalk or are clinically ( printed page 94332) similar to procedures in the CPT surgical range that we determined met the general standards established in previous years for addition to the ASC CPL.
In the CY 2024 OPPS/ASC final rule with comment period, we finalized adding several dental surgical procedures to the ASC CPL that met our regulatory criteria at §§ 416.166. We noted that there are statutory and regulatory limitations regarding Medicare coverage and payment for dental services. Section 1862(a)(12) of the Act generally precludes Medicare Part A or Part B payment for services in connection with the care, treatment, filling, removal, or replacement of teeth or structures directly supporting teeth (collectively referred to in this section as “dental services”). The regulation at § 411.15(i) similarly prohibits payment for dental services. In the CY 2023 PFS final rule (87 FR 69663), we explained that there are certain instances where dental services are so integral to other medically necessary services that they are not in connection with dental services within the meaning of section 1862(a)(12) of the Act. Rather, such dental services are inextricably linked to, and substantially related to the clinical success of, other covered services (hereafter in this section, “inextricably linked”). To provide greater clarity to current policies, the CY 2023 PFS final rule finalized: (1) a clarification of our interpretation of section 1862(a)(12) of the Act to permit payment for dental services that are inextricably linked to other covered services; (2) clarification and codification of certain longstanding Medicare FFS payment policies for dental services that are inextricably linked to other covered services; (3) that, beginning for CY 2023, Medicare Parts A and B payment can be made for certain dental services inextricably linked to Medicare-covered organ transplant, cardiac valve replacement, or valvuloplasty procedures; and, (4) beginning for CY 2024, that Medicare Parts A and B payment can be made for certain dental services inextricably linked to Medicare-covered services for treatment of head and neck cancers (87 FR 69670 through 69671).
For the ASC setting, services must meet all applicable Medicare conditions for coverage and payment to be paid by Medicare, including those as specified under the CY 2023 PFS final rule (87 FR 69687 through 69688) and § 411.15(i)(3). Medicare payment may be made in the ASC setting for dental services for which payment may be made under Medicare Part B, paid under the OPPS, and that meet the ASC CPL criteria. The fact that a drug, device, procedure, or service is assigned a HCPCS code and a payment rate under the ASC payment system indicates only how the product, procedure, or service may be paid if covered by the program. MACs will be involved in the final decision regarding whether a drug, device, procedure, or other service meets all program requirements and conditions for coverage and payment. Therefore, even if a code describing a dental service has an associated payment rate on the ASC CPL, Medicare will only make payment for the service if it meets applicable requirements. We also clarify that adding dental procedures to the ASC CPL does not serve as a coverage determination for dental services under general anesthesia. We direct readers to the CY 2025 PFS proposed rule for additional discussion of Medicare coverage and payment for dental services.
For a detailed discussion of the history of our policies for adding surgical procedures to the ASC CPL, we refer readers to the CY 2021 through CY 2024 OPPS/ASC final rules with comment period (85 FR 86143 through 86145; 86 FR 63777 through 63805; 87 FR 72068 through 72076; and 88 FR 81923 through 81945).
2. Final Changes to the List of ASC Covered Surgical Procedures for CY 2025
Our current policy, which includes consideration of the general standards and exclusion criteria we have historically used to determine whether a surgical procedure should be added to the ASC CPL, is intended to ensure that surgical procedures added to the ASC CPL can be performed safely in the ASC setting on the typical Medicare beneficiary.
As part of our evaluation process to add procedures to the CPL, we assess potential procedures against the specific list of ASC CPL criteria at § 416.166. We also examine clinical data on these procedures from multiple sites of services, review literature and experiential data, and analyze claims data trends to ensure that these procedures meet all our criteria and are not expected to pose a significant risk to beneficiary safety when performed in an ASC. For CY 2025, we also reviewed supporting evidence received in the pre-proposed rule nominations process to inform our procedure evaluations. Based upon this review, we proposed to update the ASC CPL by adding 20 medical and dental surgical procedures to the list for CY 2025.
After reviewing the clinical characteristics of these twenty procedures and consulting with stakeholders and multiple clinical advisors, we determined that these procedures are separately paid under the OPPS, would not be expected to pose a significant risk to beneficiary safety when performed in an ASC, and would not be expected to require active medical monitoring and care of the beneficiary at midnight following the procedure. These procedures are surgical or surgery-like, clinically similar to procedures in the CPT surgical range that we determined met the general standards for addition to the ASC CPL. These procedures are not excluded from being included on the ASC CPL because they do not generally result in extensive blood loss, require major or prolonged invasion of body cavities, commonly require systemic thrombolytic therapy, or directly involve major blood vessels; are not generally emergent or life-threatening in nature or designated as requiring inpatient care; or can only be reported using a CPT unlisted surgical procedure code or are otherwise excluded under Medicare. Therefore, we believed these procedures may all be appropriately performed in an ASC and proposed to include them on the ASC CPL for CY 2025.
We continue to focus on maximizing patient access to care by adding procedures to the ASC CPL when appropriate. While expanding the ASC CPL offers benefits, such as preserving the capacity of hospitals to treat more acute patients and promoting site neutrality, we also believe that any additions to the CPL should be added in a carefully calibrated fashion to ensure that the procedure is safe to be performed in the ASC setting. We encourage interested parties to submit procedure recommendations to be added to the ASC CPL, particularly if there is evidence that these procedures meet our criteria and can be safely performed in the ASC setting. We expect to continue to gradually expand the ASC CPL, as medical practice and technology continue to evolve and advance in future years.
Comment: Multiple commenters supported the proposed addition of 20 procedures, including 16 dental procedures, to the ASC CPL for CY 2025.
Response: We thank the commenters for their support.
Comment: A few commenters provided feedback on different approaches for CMS to consider when approaching the ASC CPL. These commenters felt there was a lack of transparency in the pre-proposed rule recommendation process and requested ( printed page 94333) that CMS provide the full list of recommended procedures and rationales for all procedures recommended by the public, even if CMS did not propose to add those procedures to the ASC CPL. Additionally, one commenter requested greater clarification on the definition of the “typical Medicare beneficiary.”
Response: We thank the commenters for their input and will consider it for future rulemaking. As part of our evaluation process, we assess recommended procedures against the specific list of ASC CPL criteria at 42 CFR 416.166, examining clinical data on these procedures from multiple sites of services, reviewing the literature and experiential data provided in public comments, and examining claims volume to ensure that procedures are not expected to pose a significant risk to beneficiary safety when performed in an ASC.
While we have not historically published pre-proposed rule recommendations for which we did not propose any changes, we do address and publish a list of all codes that are recommended during the public comment period in the OPPS/ASC final rule. Additionally, in the CY 2022 OPPS/ASC final rule, we provided additional context on the typical Medicare beneficiary, whose health status is representative of the broader Medicare population, and we believe this information is sufficient to understand the typical Medicare beneficiary terminology without additional clarification at this time (86 FR 63777-63779).
Comment: One commenter requested that CMS not finalize its proposal to add two leadless pacemaker procedures, CPT codes 0795T ( Transcatheter insertion of permanent dual-chamber leadless pacemaker, including imaging guidance (e.g., fluoroscopy, venous ultrasound, right atrial angiography, right ventriculography, femoral venography) and device evaluation (e.g., interrogation or programming), when performed; complete system (i.e., right atrial and right ventricular pacemaker components)) and 0801T ( Transcatheter removal and replacement of permanent dual-chamber leadless pacemaker, including imaging guidance (e.g., fluoroscopy, venous ultrasound, right atrial angiography, right ventriculography, femoral venography) and device evaluation (e.g., interrogation or programming), when performed; dual-chamber system (i.e., right atrial and right ventricular pacemaker components)), citing previous CMS guidance that excludes contractors from paying claims for leadless pacemakers when the service is provided in the ASC setting.
Response: We thank the commenter for their feedback. As of January 18, 2017, CMS covers leadless pacemakers through Coverage with Evidence Development (CED) when procedures are performed in CMS-approved CED studies and are provided in one of the following places of service: Indian Health Service Provider Based Facility, Inpatient Hospital, On Campus-Outpatient Hospital, and Military Treatment Facility (Medicare Claims Processing Manual (Pub. L. 100-04), Chapter 32, Section 380.1.1). We agree with the commenter that ASCs are excluded from payment for leadless pacemaker procedures under this policy, therefore, we are not finalizing our proposal to add CPT codes 0795T and 0801T to the ASC CPL.
Comment: Most commenters on this policy recommended specific codes to be added to the ASC CPL including arthrodesis, cardiac ablations, echocardiography, electrophysiological studies, vascular injections, and electroconvulsive therapy. We received 74 procedure recommendations for the CPL, listed in Table 155, below.
Response: We thank commenters for their recommendations. We individually assessed each of the recommended procedures, evaluating clinical data on these procedures from multiple sites of service, reviewing the literature and experiential data provided in public comments, and examining claims volume to determine whether these procedures meet each of the regulatory criteria at 42 CFR 416.166.
Based on our review of the clinical characteristics of the procedures and their similarity to other procedures that are currently on the ASC CPL, we believe that 3 procedures (CDT codes D7320, D7321, and D7471) out of the 74 procedure recommendations we received can be safely performed in the ASC setting and meet the general standards and exclusion criteria for the ASC CPL as set forth in 42 CFR 416.166(b) and (c), respectively. We agree with the commenter who provided support and evidence stating that these procedures can be safely performed in an ASC setting. We will continue to monitor clinical data on these services in the ASC setting and address any new trends in future rulemaking. These procedures, listed in Table 155 below, are:
- D7320 (Alveoloplasty not in conjunction with extractions—four or more teeth or tooth spaces, per quadrant).
- D7321 (Alveoloplasty not in conjunction with extractions—one to three teeth or tooth spaces, per quadrant).
- D7471 (Removal of lateral exostosis (maxilla or mandible)).
Below, we explain our rationale for not including the 71 recommended procedures, organized by category.
- 1 anesthesia code for total knee arthroplasty. Payment for anesthesia services is packaged into the ASC payment for covered surgical procedures.
- 2 integumentary system, including placement of breast localization devices. These codes are already on the ASC CPL as packaged procedures. We believe this placement is appropriate, given that these procedures are performed with a primary procedure.
- 18 cardiovascular codes, including cardiac ablations, cardioversion, electrophysiological studies and procedures, and echocardiography. Many of these codes have associated inpatient admissions, where the beneficiary requires active medical monitoring and care at midnight following the procedure. The cardioversion and echocardiography codes are non-surgical procedures, which means they would not qualify for addition to the ASC CPL, and most of the ancillary codes are not integral to a covered surgical procedure on the ASC CPL.
- 2 musculoskeletal code, which are arthrodesis procedures. While these procedures have some claims volume in the outpatient setting, the claims also showed multiple post-operative inpatient days, indicating that the beneficiary would require active monitoring and care past midnight following the procedure. These procedures can also involve prolonged invasion of body cavities. In addition, we acknowledge the findings of studies that commenters provided related to these procedures. However, the studies we received had significant limitations including selection bias and an absence of age groups representative of the Medicare population.
- 1 psychiatry code for electroconvulsive therapy. As this procedure is non-surgical, it would not qualify as a separately payable surgical procedure on the ASC CPL.
- 18 radiology codes, including aortography, angiography, venography, and transvascular biopsy. Most of these codes are currently on the covered ancillary services list. As they are non-surgical, they would not qualify as separately payable surgical procedures on the ASC CPL.
- 29 vascular codes, including catheter placements. Nearly all the catheter placement codes recommended ( printed page 94334) are already on the ASC CPL as packaged procedures. We believe this placement is appropriate, given that these procedures are performed with a primary service. The remaining vascular codes related to revascularization directly involve major blood vessels and there are concerns for potential extensive blood loss.
Given these considerations, we believe that these 71 codes do not meet the criteria for inclusion on the ASC CPL due to the following factors: likelihood of inpatient admissions, the need for multiple-day stays past midnight, safety risks posed to the typical beneficiary without active postoperative monitoring, involvement of major blood vessels, prolonged invasion of a body cavity, or are non-surgical.
Therefore, in this CY 2025 OPPS/ASC final rule with comment period, we are finalizing our proposal, with modification, to add 21 procedures, including 19 dental procedures, to the ASC CPL. These procedures are listed below in Table 154 of this CY 2025 OPPS/ASC final rule with comment period.
( printed page 94335) ( printed page 94336) ( printed page 94337) ( printed page 94338) ( printed page 94339) ( printed page 94340) ( printed page 94341)3. Covered Ancillary Services
Covered ancillary services are specified in § 416.164(b) and, as stated previously, are eligible for separate ASC payment. As provided at § 416.164(b), we make separate ASC payments for ancillary items and services when they are provided integral to ASC covered surgical procedures that include the following: (1) brachytherapy sources; (2) certain implantable items that have pass-through payment status under the OPPS; (3) certain items and services that we designate as contractor-priced, including, but not limited to, procurement of corneal tissue; (4) certain drugs and biologicals for which separate payment is allowed under the OPPS; (5) certain radiology services for which separate payment is allowed under the OPPS; and (6) non-opioid pain management drugs that function as a supply when used in a surgical procedure. Payment for ancillary items and services that are not paid separately under the ASC payment system is packaged into the ASC payment for the covered surgical procedure.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59062 through 59063), consistent with the established ASC payment system policy (72 FR 42497), we finalized the policy to update the ASC list of covered ancillary services to reflect the payment status for the services under the OPPS and to continue this reconciliation of packaged status for subsequent calendar years. As discussed in prior rulemaking, maintaining consistency with the OPPS may result in changes to ASC payment indicators for some covered ancillary services. For example, if a covered ancillary service was separately paid under the ASC payment system in CY 2024, but will be packaged under the CY 2025 OPPS, we would also package the ancillary service under the ASC payment system for CY 2025 to maintain consistency with the OPPS. Comment indicator “CH” is used in Addendum BB (which is available via the internet on the CMS website) to indicate covered ancillary services for which we proposed a change in the ASC payment indicator to reflect a proposed change in the OPPS treatment of the service for CY 2025.
In the CY 2022 OPPS/ASC final rule with comment period, we finalized our proposal to revise 42 CFR 416.164(b)(6) to include, as ancillary items that are integral to a covered surgical procedure and for which separate payment is allowed, non-opioid pain management drugs and biologicals that function as a supply when used in a surgical procedure as determined by CMS (86 FR 63490).
New CPT and HCPCS codes for covered ancillary services for CY 2025 can be found in section XIII.B of this final rule with comment period. All ASC covered ancillary services and their final payment indicators for CY 2025 are also included in Addendum BB to this final rule with comment period (which is available via the internet on the CMS website).
Comment: We received one comment requesting CMS not finalize its proposal to add HCPCS code C1605 to the list of covered ancillary services, due to CMS's policy that excludes contractors from paying claims for leadless pacemakers when the service is provided in the ASC setting.
Response: We thank the commenter for their feedback. As discussed in section XIII.D.2 above, procedures for leadless pacemakers are not covered when provided in the ASC setting (Medicare Claims Processing Manual (Pub. L. 100-04), Chapter 32, section 380.1.1). Therefore, we agree with the commenter and are not finalizing our proposal to add HCPCS code C1605 to the list of covered ancillary services.
Comment: We received several comments from physician societies requesting that CMS not finalize its proposal to add CPT code 93355 ( printed page 94342) (Echocardiography, transesophageal (tee) for guidance of a transcatheter intracardiac or great vessel(s) structural intervention(s) ( e.g., tavr, transcatheter pulmonary valve replacement, mitral valve repair, paravalvular regurgitation repair, left atrial appendage occlusion/closure, ventricular septal defect closure) (peri-and intra-procedural), real-time image acquisition and documentation, guidance with quantitative measurements, probe manipulation, interpretation, and report, including diagnostic transesophageal echocardiography and, when performed, administration of ultrasound contrast, doppler, color flow, and 3d) to the covered ancillary procedures list. The commenters expressed safety concerns with adding CPT code 93355 to the covered ancillary services list, as the primary procedures that would be billed with this service are not performed in ASCs, with some of the procedures also being on the inpatient only list.
Response: We thank the commenters for their input. After further review, we agree with the commenters that the majority of primary procedures associated with CPT code 93355 are not on the ASC CPL and/or are performed in the inpatient setting. Therefore, we are not finalizing our proposal to add 93355 to the covered ancillary services list.
Claims Processing Limitations for Covered Ancillary Procedures Performed With G0330
We finalized adding HCPCS code G0330 ( Facility services for dental rehabilitation procedure(s) performed on a patient who requires monitored anesthesia (e.g., general, intravenous sedation (monitored anesthesia care) and use of an operating room)) to the ASC CPL in the CY 2024 OPPS/ASC final rule (88 FR 81924). In ASC Addendum BB, there is a specific and definitive list of covered ancillary dental services with payment indicator of “D1,” indicating an ancillary dental service or item with no separate payment made. In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81945 and 81946), we finalized that code G0330 could only be billed when accompanied by a covered ancillary procedure that has the payment indicator of “D1.” Performance of at least one of these covered ancillary services is integral to each of the surgical procedures that correspond to G0330. This limitation ensures that only covered ancillary services we evaluated for safety in the ASC setting could be performed with code G0330.
While HCPCS code G0330 must be billed with a covered ancillary procedure with a payment indicator of “D1,” these covered ancillary procedures with a payment indicator of “D1” can be billed with surgical procedures other than G0330. When billed with procedures other than code G0330, these ancillary procedures would be packaged in accordance with our policy for covered ancillary procedures. Additionally, other than HCPCS code G0330, procedures assigned to payment indicator “D2”, indicating non office-based dental procedure added in CY 2024 or later, are not required to be billed with a covered ancillary procedure assigned to payment indicator “D1” to receive payment for the procedure.
The fact that a drug, device, procedure, or service is assigned a HCPCS code and a payment rate under the ASC payment system indicates only how the product, procedure, or service may be paid if covered by the program. MACs will be involved in the final decision regarding whether a drug, device, procedure, or other service meets all program requirements and conditions for coverage and payment. Therefore, even if a code describing a dental service has an associated payment rate on the ASC CPL, Medicare will only make payment for the service if it meets applicable requirements.
Comment: One commenter requested that CMS remove the requirement that HCPCS code G0330 must be billed along with a covered but non-payable dental ancillary service. The commenters stated that dental rehabilitation meet the regulatory requirements for inclusion on the ASC CPL.
Response: We thank the commenters for their input. The claims processing limitations around code G0330, for example, the requirement that code G0330 must be billed with a covered dental ancillary procedure with payment indicator “D1,” in the ASC setting, allows us to ensure that only covered ancillary services we have evaluated for safety in the ASC setting can be performed with code G0330.
After consideration of the public comments we received, we are finalizing this policy as proposed.
E. ASC Payment Policy for Non-Opioid Post-Surgery Pain Management Drugs, Biologicals, and Devices
1. Background on OPPS/ASC Non-Opioid Pain Management Packaging Policies
On October 24, 2018, the Substance Use-Disorder Prevention that Promotes Opioid Recovery and Treatment for Patients and Communities (SUPPORT) Act (Pub. L. 115-271) was enacted. Section 1833(t)(22)(A)(i) of the Act, as added by section 6082(a) of the SUPPORT Act, states that the Secretary must review payments under the OPPS for opioids and evidence based non-opioid alternatives for pain management (including drugs and devices, nerve blocks, surgical injections, and neuromodulation) with a goal of ensuring that there are not financial incentives to use opioids instead of non-opioid alternatives. As part of this review, under section 1833(t)(22)(A)(iii) of the Act, the Secretary must consider the extent to which revisions to such payments (such as the creation of additional groups of covered outpatient department (OPD) services to separately classify those procedures that utilize opioids and non-opioid alternatives for pain management) would reduce the payment incentives for using opioids instead of non-opioid alternatives for pain management. In conducting this review and considering any revisions, the Secretary must focus on covered OPD services (or groups of services) assigned to C-APCs, APCs that include surgical services, or services determined by the Secretary that generally involve treatment for pain management. If the Secretary identifies revisions to payments pursuant to section 1833(t)(22)(A)(iii) of the Act, section 1833(t)(22)(C) of the Act requires the Secretary to, as determined appropriate, begin making revisions for services furnished on or after January 1, 2020. Revisions under this paragraph are required to be treated as adjustments for purposes of paragraph (9)(B) of the Act, which requires any adjustments to be made in a budget neutral manner. Section 1833(i)(8) of the Act, as added by section 6082(b) of the SUPPORT Act, requires the Secretary to conduct a similar type of review as required for the OPPS and to make revisions to the ASC payment system in an appropriate manner, as determined by the Secretary.
For a detailed discussion of rulemaking on non-opioid alternatives prior to CY 2020, we refer readers to the CYs 2018 and 2019 OPPS/ASC final rules with comment period (82 FR 59345; 83 FR 58855 through 58860).
For the CY 2020 OPPS/ASC proposed rule (84 FR 39423 through 39427), as required by section 1833(t)(22)(A)(i) of the Act, we reviewed payments under the OPPS for opioids and evidence- based non-opioid alternatives for pain management (including drugs and devices, nerve blocks, surgical injections, and neuromodulation) with a goal of ensuring that there are not financial incentives to use opioids ( printed page 94343) instead of non-opioid alternatives. For the CY 2020 OPPS/ASC proposed rule (84 FR 39423 through 39427), we proposed to continue our policy to pay separately at ASP plus 6 percent for non-opioid pain management drugs that function as surgical supplies in the performance of surgical procedures when they are furnished in the ASC setting and to continue to package payment for non-opioid pain management drugs that function as surgical supplies in the performance of surgical procedures in the hospital outpatient department setting.
In the CY 2020 OPPS/ASC final rule with comment period (84 FR 61173 through 61180), after reviewing data from stakeholders and Medicare claims data, we did not find compelling evidence to suggest that revisions to our OPPS payment policies for non-opioid pain management alternatives were necessary for CY 2020. We finalized our proposal to continue to unpackage and pay separately at ASP plus 6 percent for non-opioid pain management drugs that function as surgical supplies when furnished in the ASC setting for CY 2020. Under this policy, for CY 2020, the only drug that qualified for separate payment in the ASC setting as a non- opioid pain management drug that functions as a surgical supply was Exparel.
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 85896 through 85899), we continued the policy to pay separately at ASP plus 6 percent for non-opioid pain management drugs that function as surgical supplies in the performance of surgical procedures when they were furnished in the ASC setting and to continue to package payment for non-opioid pain management drugs that function as surgical supplies in the performance of surgical procedures in the hospital outpatient department setting for CY 2021. For CY 2021, only Exparel and Omidria met the criteria as non-opioid pain management drugs that function as surgical supplies in the ASC setting and received separate payment under the ASC payment system.
In the CY 2022 OPPS/ASC final rule with comment period (86 FR 63483), we finalized a policy to unpackage and pay separately at ASP plus 6 percent for non-opioid pain management drugs that function as surgical supplies when they are furnished in the ASC setting, are FDA-approved, have an FDA-approved indication for pain management or as an analgesic, and have a per-day cost above the OPPS/ASC drug packaging threshold; and we finalized our proposed regulation text changes at 42 CFR 416.164(a)(4) and (b)(6), 416.171(b)(1), and 416.174 as proposed.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72089), we determined that five products were eligible for separate payment in the ASC setting under our final policy for CY 2022. We noted that future products, or products not discussed in that rulemaking that may be eligible for separate payment under this policy, would be evaluated in future rulemaking (86 FR 63496). In the CY 2023 final rule with comment period, we finalized that five drugs would receive separate payment in the ASC setting for CY 2023 under the policy for non-opioid pain management drugs and biologicals that function as surgical supplies (86 FR 63496).
In the CY 2023 OPPS/ASC final rule with comment period, we finalized a clarification of our policy by codifying the two additional criteria for separate payment for non-opioid pain management drugs and biologicals that function as surgical supplies in the regulatory text at § 416.174 as a technical change. First, we finalized at new § 416.174(a)(3) that non-opioid pain management drugs or biologicals that function as a supply in a surgical procedure are eligible for separate payment if the drug or biological does not have transitional pass-through payment status under § 419.64. In the case where a drug or biological otherwise meets the requirements under § 416.174 and has transitional pass-through payment status that will expire during the calendar year, the drug or biological would qualify for separate payment under § 416.174 during such calendar year on the first day of the next calendar year quarter after its pass-through status expires. Second, we finalized that new § 416.174(a)(4) would reflect that the drug or biological must not already be separately payable in the OPPS or ASC payment system under a policy other than the one specified in § 416.174.
In the CY 2024 OPPS/ASC final rule with comment period, we finalized four drugs as eligible to receive separate payment as a non-opioid pain management drug that functions as a supply in a surgical procedure under the ASC payment system and which met the criteria at § 416.174(a) for CY 2024.
F. Final CY 2025 Non-Opioid Policy for Pain Relief Under the OPPS and ASC Payment System
1. Background on Access to Non-Opioid Treatments for Pain Relief
The Consolidated Appropriations Act (CAA), 2023 (Pub. L. 117-328), was signed into law on December 29, 2022. Section 4135(a) and (b) of the CAA, 2023, titled Access to Non-Opioid Treatments for Pain Relief, amended section 1833(t)(16) and section 1833(i) of the Social Security Act, respectively, to provide for temporary additional payments for non-opioid treatments for pain relief (as that term is defined in section 1833(t)(16)(G)(i) of the Act). In particular, section 1833(t)(16)(G) provides that with respect to a non-opioid treatment for pain relief furnished on or after January 1, 2025, and before January 1, 2028, the Secretary shall not package payment for the non-opioid treatment for pain relief into payment for a covered OPD service (or group of services) and shall make an additional payment for the non-opioid treatment for pain relief as specified in clause (ii) of that section. Clauses (ii) and (iii) of section 1833(t)(16)(G) of the Act provide for the amount of additional payment and set a limitation on that amount.
Paragraph (10) of section 1833(i) of the Act cross-references the OPPS provisions about the additional payment amount and payment limitation for non-opioid treatments for pain relief and applies them to payment under the ASC payment system. In particular, subparagraph (A) of paragraph (10) of section 1833(i) of the Act, as added by section 4135(b) of the CAA, 2023, provides that in the case of surgical services furnished on or after January 1, 2025, and before January 1, 2028, additional payments shall be made under the ASC payment system for non-opioid treatments for pain relief in the same amount provided in clause (ii) and subject to the limitation in clause (iii) of section 1833(t)(16)(G) of the Act for the OPPS. Subparagraph (B) of section 1833(i)(10) of the Act provides that a drug or biological that meets the requirements of 42 CFR 416.174 and is a non-opioid treatment for pain relief shall also receive additional payment in the amount provided in clause (ii) and subject to the limitation in clause (iii) of section 1833(t)(16)(G) of the Act.
Additional payments are required to begin on January 1, 2025. The statute directs CMS to provide “additional payment”, and for purposes of the proposal, we interpret this language to be equivalent to “separate payment,” since CMS provides an additional payment by unpackaging the product and then making a separate payment. “Separate payment” is the more commonly used terminology in the OPPS rule and likely more familiar to readers. To avoid confusion, we will be using “separate payment” throughout the rest of this section, which we ( printed page 94344) believe to be synonymous with “additional payment.”
Our CY 2025 proposals to implement the amendments to sections 1833(t)(16) and section 1833(i) of the Act required by section 4135 of the CAA, 2023 are discussed below, followed by a summary of comments, our responses to those comments, and our finalized policy for CY 2025.
2. Final CY 2025 Non-Opioid Policy Implementation of Section 4135 of the CAA, 2023
a. Statutory Authority for OPPS/ASC Non-Opioid Policy
Prior to CY 2025, the statutory authority for the ASC non-opioid policy has been section 1833(i)(8) of the Act, as added by section 6082(b) of the SUPPORT Act. Section 1833(i)(8) of the Act refers to paragraph (t)(22), which states that the Secretary shall conduct a similar type of review as the one required for the OPPS for opioids and evidence based non-opioid alternatives for pain management (including drugs and devices, nerve blocks, surgical injections, and neuromodulation) with a goal of ensuring that there are not financial incentives to use opioids instead of non-opioid alternatives and make such revisions as the Secretary determines appropriate. As discussed in the previous section, CMS's policy for CY 2024 is to provide separate payment in the ASC setting for certain qualifying non-opioid pain management drugs that function as a supply in a surgical procedure.
As noted previously, section 4135 of the CAA, 2023, provides for temporary separate payments for certain non-opioid treatments for pain relief in both the hospital outpatient department and ambulatory surgical center settings from January 1, 2025, through December 31, 2027. Specifically, these separate payments are for qualifying drugs, biologicals, and devices that, among other requirements, have their payment packaged into payment for a covered OPD service (or group of services). Pursuant to section 1833(t)(2)(E) of the Act, the temporary separate payments must be made in a budget neutral manner.
(1) Drugs and Biologicals Subject to the ASC Non-Opioid Policy (42 CFR 416.174)
Section 1833(i)(10)(B), titled “Transition,” provides that a drug or biological that meets the requirements of the regulation at 42 CFR 416.174, the current ASC non-opioid policy, and also meets the definition of a non-opioid treatment for pain relief at section 1833(t)(16)(G)(iv) shall receive separate payments under section 4135 of the CAA, 2023, subject to the payment limitation. In light of this requirement, we proposed that drugs and biologicals that meet the definition of a non-opioid treatment for pain relief for purposes of section 4135 that are currently subject to the ASC policy for non-opioid treatments authorized by section 6082 of the SUPPORT Act, would instead receive separate payments, subject to the limitation, for the duration of the payment period for section 4135. These drugs and biologicals are described in the discussions that follow.
Comment: Commenters were generally supportive of CMS paying for drugs paid under the ASC non-opioid treatment policy authorized by section 6082 of the SUPPORT Act under the policy authorized by section 4135 of the CAA, 2023. Commenters recommended that CMS ensure payment did not decrease, to the extent possible, when the transition between policies occurs. Finally, commenters were looking ahead and recommended that CMS begin to assess its authority for continuing a policy for the payment of non-opioid treatments for pain relief, starting in CY 2028 and beyond. Some commenters recommended CMS assess their waiver authority and look to extend the policy in a non-budget neutral manner.
Response: We thank commenters for their support. As described in the following sections, we must comply with the requirements of the statute in our policy implementation. We will continue to monitor the effects of our payment policies, explore our authorities, and consider what policies may be appropriate in future rulemaking.
Based on the comments received, we are finalizing our proposal that drugs and biologicals that meet the definition of a non-opioid treatment for pain relief for purposes of section 4135 that are currently subject to the ASC policy for non-opioid treatments authorized by section 6082 of the SUPPORT Act, would instead receive separate payments, subject to the payment limitation, for the duration of the payment period for section 4135.
(2) Definition of Non-Opioid Treatment for Pain Relief
Section 1833(t)(16)(G)(iv) of the Act defines a non-opioid treatment for pain relief. In order for a drug or biological product to qualify as a non-opioid treatment for pain relief, pursuant to section 1833(t)(16)(G)(iv)(I), the product must have “a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors.” In order for a medical device to qualify as a non-opioid treatment for pain relief, pursuant to section 1833(t)(16)(G)(iv)(II)(bb), the medical devices must be “used to deliver a therapy to reduce postoperative pain, or produce post-surgical or regional analgesia.” This subparagraph also defines such a device as having “an application under section 515 of the Federal Food, Drug, and Cosmetic Act that has been approved with respect to the device, been cleared for market under section 510(k) of such Act, or is exempt from the requirements of section 510(k) of such Act pursuant to subsection (l) or (m) or section 510 of such Act or section 520(g) of such Act” and “demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal.”
Comment: Commenters were very supportive of CMS using the exact language in the statue in the definition of a non-opioid treatment for pain relief. Commenters recommended CMS codify this definition.
Response: We thank commenters for their support.
Comment: One commenter recommended CMS adopt a definition that allows for the combined use of devices and drugs together, rather than just the individual components, for the purposes of separate payment for a non-opioid treatment for pain relief.
Response: We thank the commenter for their suggestion. Section 4135 does not contemplate a situation where a combination of products may qualify. However, if the individual components, either a drug, biological, or device, meet the statutory criteria, it would qualify for separate payment under this provision.
Comment: A couple commenters recommended that CMS include orally self-administered drugs as potentially qualifying drugs for separate payment under this policy. One comment requested CMS affirm that self-administered non-opioid pain relievers will qualify for separate payment and requested CMS affirm that such drugs that function as a supply when used in conjunction with a surgical procedure qualify for separate payment under the proposed policy. Similarly, one drug manufacturer of an investigational oral drug product recommended that self -administered non-opioid treatments for pain relief should qualify for separate payment in certain circumstances. For their specific product, they stated that ( printed page 94345) they anticipated if the drug is FDA-approved, providers may administer the oral product to beneficiaries before or after a surgical procedure. This commenter stated that the statute makes no distinction between the method of administration of the qualifying drug and that current OPPS regulations indicate that, in certain circumstances, drugs that may be considered usually self-administered by the patient may be paid when they function as supplies.
Response: We thank the commenters for raising these concerns through their comments. We believe it is premature to evaluate a product that is not FDA-approved for inclusion in this policy, as FDA approval is a requirement for drugs and biological products. We will evaluate all products, including newly FDA-approved products, to determine whether they meet the criteria in the statute and the regulations at 42 CFR 419.43(k).
(3) Evidence Requirement for Medical Devices
For CY 2025, to determine whether a medical device fulfills the statutory requirement that it has demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal, we proposed to review all data submitted during the public comment period to determine if the device demonstrates the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids. We encouraged interested parties submitting non-opioid device recommendations for CY 2025 to also submit any relevant literature that demonstrates that the named medical device replaces, reduces, or avoids opioid use per this statutory provision with their public comments. We proposed that CMS would review any literature submitted and determine whether it meets this evidence criterion. We did not require that commenters submit any data or literature with their device recommendations. If there was no data or literature submitted for a medical device, or if the materials submitted do not demonstrate any ability of the medical device to replace, reduce, or avoid opioids, the medical device will not meet this evidence criterion and will therefore not qualify for separate payment under section 4135.
Comment: Most commenters recommended that CMS evaluate any literature submitted and not implement additional or burdensome requirements for peer reviewed literature. For example, one commenter stated that given the unique challenges of testing new non-opioid pain treatments, they believed the need for evidence must be balanced against the costs and time involved in collecting the data and how this process could delay the availability of novel technologies to treat unmet clinical needs. This commenter asked CMS to confirm that well-designed prospective, retrospective, and real-world evidence clinical trials that are published in peer-reviewed journals provide a means of satisfying the requirement for medical devices.
Response: We are adhering to the statutory language of the Act and are requiring that a medical device has demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. To the extent that literature submitted by interested parties satisfies that requirement, the medical device would qualify for separate payment. We cannot say with certainty that a specific study design would always or would never satisfy this criterion.
After consideration of the public comments we received, we are finalizing our proposal that we will review all data submitted during the public comment period to determine if the device demonstrates the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids.
b. Non-Opioid Product Indications
(1) FDA-Approved Indications for Drugs and Biologicals
Section 1833(t)(16)(G)(iv)(I) of the Act specifies that to meet the definition of a non-opioid treatment for pain relief and to be eligible for separate payment, a drug or biological product must have a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors.
Given these statutory requirements, we proposed only to approve separate payment for drug or biological products with an FDA-approved indication that closely aligns with the statutorily required indication language to reduce post-operative pain or produce post-surgical or regional analgesia for CY 2025. We noted that products with an indication that does not meet the statutory requirement would not qualify. Table 84 from the CY 2025 OPPS/ASC proposed rule (89 FR 59431), and Table 156 in this final rule with comment period, includes citations to the indications of the drugs and biologicals proposed to have met the statutory requirements and qualify for separate payment in the CY 2025 OPPS/ASC proposed rule.
Comment: The majority of commenters supported CMS using the language in the Act as the basis for determining whether a drug or biologic had the appropriate FDA-approved indication, in order to qualify for separate payment under this policy. A few commenters indicated that the specific indication requirements for drugs and biologicals was an intentional choice, as Congress did not require a specific labeled indication for medical devices.
Response: We thank commenters for their feedback and support.
Comment: Some commenters recommended CMS interpret more broadly the statutory language requiring drugs and biologicals to have an FDA-approved indication to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors. For example, one commenter recommended CMS consider drugs that are used off-label for post-surgical pain, meaning a drug that does not have an FDA-approved indication for reducing post-operative pain, but is used for that purpose, based on existing evidence. One example provided was the drug Dexycu, which the commenter states reduces post-operative inflammation, and therefore pain, following cataract surgery. Another commenter provided the example of IV acetaminophen and stated that the product has been used in pain management but is not specifically indicated in FDA-approved labeling for post-surgical pain management. These commenters contend that these products have been used off-label in pain management for decades and they are concerned that the statute inappropriately excludes those types of products from qualifying for separate payment. They also recommend CMS address the packaging of these products, by exempting non-opioid drugs and regional anesthesia procedures to manage pain in a surgical procedure from comprehensive APC (C-APC) packaging policies.
The manufacturer of a similar product, Caldolor, an intravenous formulation of ibuprofen, which has a general acute pain indication for the management of mild to moderate pain and the management of moderate to severe pain as an adjunct to opioid analgesics, also made similar recommendations. This commenter ( printed page 94346) stated this product is similar to several other non-opioid pain management products listed in the proposed rule, such as ketorolac tromethamine injection. This commenter cited FDA guidance where a product may be granted a general acute pain indication when supported by at least two clinical trials, each in a different pain population. This commenter believed that their more general pain indication should satisfy the statutory requirement that a qualifying drug or biological must have a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors. They do not believe that the statute requires that the FDA-approved indication expressly refer to use for postoperative or postsurgical pain. They noted that their product was studied in a post-surgical population. Additionally, this commenter does not believe there is evidence that Congress intended section 4135 to exclude a non-opioid product that has a general pain indication or only include a product if the indication statement expressly refers to use for postoperative or postsurgical pain (for example, by stating that a product is used usually in a postoperative setting).
Response: Congress specifically included language requiring that drugs or biologicals have “a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors.” We also note that many patients who receive services paid under the OPPS and ASC payment system are often in a post-surgical environment, given the nature of the procedure typically performed in an ASC or HOPD.
As there is no mention of post-operative or post-surgical use in the FDA-approved indications for the products Caldolor,[184] Dexycu,[185] or Ofirmev [186] (IV acetaminophen), we do not believe these products meet the statutory criteria and therefore do not qualify for separate payment under this policy. Additionally, we do not believe other modifications to longstanding OPPS payment policies are warranted at this time to provide separate payment for these products. We note that all other drug products proposed and finalized for separate payment have an FDA indication for reduction of postoperative pain, or production of postsurgical or regional analgesia, without acting upon the body's opioid receptors. This includes a similar product, intravenous ketorolac, that was mentioned in one comment letter, which is indicated for the short-term management of moderately severe acute pain that requires analgesia at the opioid level, usually in the postoperative setting.[187] We do not believe that products mentioned by commenters that lack an FDA-approved indication to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors meet the statutory requirement. We also note that those products that do meet the statutory requirements will be exempt from C-APC packaging. We are not finalizing any additional changes to our C-APC policy at this time, as we continue to believe that generally the C-APC packaging policy maintains the important packaging principles of the OPPS.
After a consideration of comments received, we are finalizing our proposal without modification, to only approve separate payment for drug or biological products with an FDA-approved indication to reduce post-operative pain or produce post-surgical or regional analgesia. Products such as those with only a general pain indication will not qualify.
(2) Indications for Medical Devices
With respect to medical devices, section 1833(t)(16)(G)(iv)(II) of the Act specifies that such a device must be used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia to qualify for separate payment under section 4135. It also must have an application approved under section 515 of the Federal Food, Drug, and Cosmetic Act (FDCA), have been cleared for market under section 510(k) of the FDCA, or be exempt from the requirements of section 510(k) of the FDCA pursuant to section 510(l) or (m) or 520(g) of the FDCA. For CY 2025, for medical devices, we proposed to only approve medical devices with an indication that specifies that the device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia and which also have FDA approval, market clearance, or an appropriate exemption from the requirements of section 510(k).
Comment: Commenters recommended CMS adhere closely to the statutory language when creating its policy. Several commenters recommended CMS clarify their proposal and the regulation text at 42 CFR 419.83(k)(2)(i) through (iv). Specifically, commenters were concerned that CMS was deviating from the statute and requiring an additional FDA-approved indication for use for medical devices that was not in statute. Commenters believed that CMS should not require medical devices to have an indication that specifies that the device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. Commenters believed Congress had clear and distinct intentions regarding the statutory criteria for medical devices compared to drugs and biologicals and chose not to subject medical devices to the same indications for use standard that applies to drugs and biologicals.
Response: We thank commenters for raising this issue and requesting clarification from CMS. We acknowledge the proposed rule may not have been sufficiently clear regarding the FDA-approved indications for medical devices. However, we confirm that while medical devices must be used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia, they are not required by statute to have specific language to this effect in a FDA-approved indication. Rather, the medical device must have an application approved under section 515 of the Federal Food, Drug, and Cosmetic Act (FDCA), have been cleared for market under section 510(k) of the FDCA, or be exempt from the requirements of section 510(k) of the FDCA pursuant to section 510(l) or (m) or 520(g) of the FDCA. We note that the regulation text that we proposed, and are finalizing without modification at 42 CFR 419.43(k)(2)(i) through (iv), does not require a specific FDA-approved indication for medical devices. However, one way to show that a medical device is used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia, could be through language indicating that in an FDA-approved indication.
After consideration of the public comments we received, we are finalizing without modification our proposal that a device must be used to deliver a therapy to reduce postoperative pain or produce post- ( printed page 94347) surgical or regional analgesia to qualify for separate payment under section 4135 for CY 2025. It also must have an application approved under section 515 of the Federal Food, Drug, and Cosmetic Act (FDCA), have been cleared for market under section 510(k) of the FDCA, or be exempt from the requirements of section 510(k) of the FDCA pursuant to section 510(l) or (m) or 520(g) of the FDCA. This is consistent with the regulation text that we are finalizing without modification at 42 CFR 419.43(k)(2)(i) through (iv). We are clarifying that medical devices are not required to have specific language in their FDA approved indication to have a specific FDA-approved indication like drugs and biologicals. However, one way to show that a medical device is used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia, could be through language indicating that it has an FDA-approved indication for one or more of those uses.
c. Amount of Payment
Section 1833(t)(16)(G)(ii)(I) of the Act provides that, for a non-opioid treatment for pain relief that is a drug or biological product, the amount of separate payment is the amount of payment for such product determined under section 1847A of the Act that exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the drug or biological, subject to a limitation, as described in the next section. Section 1833(t)(16)(G)(ii)(II) of the Act provides that, for a non-opioid treatment for pain relief that is a medical device, the amount of separate payment is the amount of the hospital's charges for the device, adjusted to cost, that exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the device, subject to a limitation, as described in the next section. As the language in section 1833(t)(16)(G)(ii)(II) of the Act is very similar to the transitional pass-through language at section 1833(t)(6)(D)(i) and (ii) of the Act, we proposed implementing a similar payment methodology for non-opioid products. A payment offset is the amount reflecting the portion of the non-opioid product in the procedure payment rate.
We proposed to assign a payment offset of zero dollars for the qualifying drugs, biologicals, and devices for CY 2025. A zero offset means that we would not offset or remove the amount that the non-opioid product represents from the procedure payment rate when setting payment rates. We proposed this would apply for CY 2025 for all non-opioid drugs, biologicals, and devices that qualify for separate payment. We proposed a zero dollar offset for the initial year of the policy as some of these products are new products or newly separately paid in the OPPS setting and their costs may not be fully reflected yet in the cost of procedures in which they may be used. Therefore, the separate payment for a drug or biological will be determined by subtracting from the amount calculated using the methodology outlined in section 1847A of the Act the portion of the otherwise applicable Medicare OPD fee schedule associated with the drug or biological, which as previously discussed, we proposed to be zero dollars for CY 2025. For the amount of payment for a medical device, the separate payment amount will be determined by subtracting from the hospital's charges for the device, adjusted to cost, the portion of the otherwise applicable Medicare OPD fee schedule amount associated with the medical device, which as previously discussed, we proposed to be zero dollars for CY 2025. These separate payment amounts will all be subject to the payment limitation, described in the subsequent section.
Section 1833(i)(10) of the Act establishes the same separate payment for the ASC setting as for hospital outpatient departments, as described in section 1833(t)(16)(G)(ii) of the Act. Both separate payments are subject to the limitation in section 1833(t)(16)(G)(iii) of the Act, which specifies that the separate payment amount shall not exceed the estimated average of 18 percent of the OPD fee schedule amount for the OPD service (or group of services) with which the non-opioid treatment for pain relief is furnished. Our final implementation of this payment limitation is discussed in further detail below. Given this statutory requirement, we proposed to pay the same separate payment amount for qualifying non-opioid products in both the HOPD and ASC settings.
As the statute requires separate payment for these non-opioid treatments for pain relief, these products cannot be packaged into the procedure payment. Under our current threshold packaging policy, if the estimated per day cost for a drug or biological is less than or equal to the applicable OPPS drug packaging threshold, we package payment for the drug or biological into the payment for the associated procedure. Similarly, under our comprehensive APC (C-APC) policy, we package all payments for services integral, ancillary, supportive, dependent, and adjunctive to the primary service into a single payment for the primary comprehensive service. For CY 2025, we proposed that non-opioid treatments for pain relief would not be subject to the threshold packaging policy in section V.B.1.a. of the CY 2025 OPPS/ASC proposed rule (89 FR 59363) and would also be separately paid when used during a comprehensive APC (C-APC) procedure in the HOPD setting. For more information on the drug packaging threshold, see section V.B.1.a of this final rule with comment period, and section II.A.b of this final rule with comment period for further information on C-APC packaging.
Comment: Most commenters strongly agreed with our proposals to provide separate payment for qualifying products. Commenters agreed that it was appropriate to assign a zero dollar offset for the initial year, CY 2025. Some commenters recommended CMS continue to assign a zero dollar offset for the duration of the 3-year separate payment policy.
Response: We thank commenters for their support. We will evaluate the need to continue to assign a zero dollar offset for future years in future rulemaking.
Comment: Some commenters recommended specific payment and payment limitation amounts be assigned for payment for certain medical devices.
Response: We note that the statute requires the additional payment for qualifying medical devices to be based on the hospital's charges for the device, adjusted to cost; therefore, we do not find it appropriate to assign a specific payment amount for a qualifying medical device.
Comment: A small number of commenters did not agree with our proposal to pay separately and unpackage qualifying non-opioid treatments for pain relief. Specifically, one commenter upheld the value and importance of the OPPS packaging policies, though they recognized that the agency was required to implement the statute and believed our proposed implementation was reasonable. Another commenter cautioned over-incentivizing non-opioid products, noting that opioids used with appropriate prescribing oversight and medical supervision remain an effective tool for pain.
Response: We thank commenters for their input.
After consideration of the public comments we received, we are finalizing our policy as proposed for CY 2025. Specifically, we are finalizing that the separate payment amount for a drug ( printed page 94348) or biological will be determined by subtracting from the amount calculated using the methodology outlined in section 1847A of the Act the portion of the otherwise applicable Medicare OPD fee schedule associated with the drug or biological, which we are finalizing to be zero dollars for CY 2025. For the amount of payment for a medical device, the separate payment amount will be determined by subtracting from the hospital's charges for the device, adjusted to cost, the portion of the otherwise applicable Medicare OPD fee schedule amount associated with the medical device, which we are finalizing to be zero dollars for CY 2025. Additionally, qualifying non-opioid treatments for pain relief will not be subject to the drug threshold packaging policy, described in section V.B.1.a. of this final rule with comment period, and will also be separately paid when used during a comprehensive APC (C-APC) procedure in the HOPD setting.
d. Payment Limitation
Section 1833(t)(16)(G)(iii) of the Act states that the separate payment amount specified in clause (ii), (which is described in the previous section) shall not exceed the estimated average of 18 percent of the OPD fee schedule amount for the OPD service (or group of services) with which the non-opioid treatment for pain relief is furnished, as determined by the Secretary.
For the non-opioid products that are currently billed under the OPPS, we conducted a claims analysis of CY 2023 OPPS claims, which are the claims available for CY 2025 rulemaking, and found that approximately 90 percent of the utilization, on average, for these non-opioid products is focused in the top five most frequently performed services for each product. Given this, we believed that using the top five services would provide a representative estimate for purposes of the payment limitation. As illustrated in Table 85 in the CY 2025 OPPS/ASC proposed rule (89 FR 59432), we proposed to use the top five services by volume associated with a drug, biological, or medical device, to determine the volume-weighted payment rate per claim and the 18 percent payment limitation specified by statute, based on the most recent claims data available for CY 2025. This payment limitation approach was also generally consistent with the comments received in response to the comment solicitation in the CY 2024 OPPS/ASC proposed rule (88 FR 49767 through 49769). For example, in response to the CY 2024 comment solicitation, several commenters supported CMS establishing a payment limitation for each non-opioid treatment item, based on a volume-weighted OPPS payment rate for the top five services that package the item into their payment rate.
We proposed to apply the 18 percent payment limitation per date of service billed, rather than per HCPCS dosage unit. This is due to the fact that there are typically multiple HCPCS dosage units, also called billing units, of each drug or biological billed per claim. Thus, the total units of a drug billed on a date of service is more reflective of the cost of the drug in that encounter. The amount of drug or biological used during an encounter, represented by a date of service for purposes of the proposal, will impact whether the separate payment for the drug or biological exceeds the payment limitation required by statute. Meaning, the same drug or biological may or may not be subject to the payment limitation, depending on the amount of drug used. For example, a drug is paid $1 per 1 mg (per billing unit) and has a payment limitation set at $100, based on 18 percent of the volume weighted average of the payment of the top 5 services associated with the use of the drug. If 50 mg (50 billing units) of this drug were to be billed during one patient encounter or one date of service, then $50 would be paid. The payment limitation would not apply as the payment for the drug did not exceed the payment limitation of $100. If 200 mg (200 billing units) of that same drug were to be billed during one patient encounter or one date of service, then the $200 payment would be limited to $100. In this case, the payment limitation would apply as the payment for the drug exceeded the payment limitation of $100. We proposed to apply this payment limitation to the date of service billed as the payment limitation applies to the total amount of separate payment, rather than the HCPCS dosage unit payment, which may only represent a small fraction of the total amount of payment.
For CY 2025, we proposed to create new status indicators for non-opioid drugs and devices to implement this payment limitation. Under the OPPS, non-opioid drugs and biologicals under this policy would be assigned a status indicator of “K1”—Non-Opioid Drugs and Biologicals For Post-Surgical Pain Relief, while non-opioid devices would be assigned a status indicator of “H1”—Non-opioid Medical Devices For Post-Surgical Pain Relief. Further discussion of the proposed new status indicators can be found in section X1.A of the CY 2025 OPPS/ASC proposed rule (89 FR 59404).
As discussed in section XIII.B.6.b. of the CY 2025 OPPS/ASC proposed rule (89 FR 59412), we proposed to modify the descriptor of ASC payment indicator “L6”—“New Technology Intraocular Lens (NTIOL); special payment” to “Special payment; New Technology Intraocular Lens (NTIOL) or qualifying non-opioid devices” and proposed to assign qualifying non-opioid medical devices to this payment indicator to operationalize payment of these devices. We proposed to assign qualifying drugs and biologicals to existing payment indicator “K2”—Drugs, biologicals, and radiopharmaceuticals paid separately when provided integral to a surgical procedure on ASC list; payment based on OPPS rate.
We welcomed public comment on the methodology used to determine the payment limitation.
Comment: Most commenters agreed with CMS's rationale and methodology that using a volume weighted average of the top five procedures based on available claims data and applied per date of service was a reasonable approach in order to implement the 18 percent payment limitation requirement.
Response: We thank commenters for their support.
Comment: Commenters were supportive of utilizing new OPPS status indicators “H1” and “K1” as well as existing ASC payment indicators “L6” and “K2” in order to operationalize payment for qualifying products.
Response: We thank commenters for their support.
Comment: A few commenters recommended CMS modify its proposed payment limitation approach. For example, one commenter recommended CMS explore its discretion to provide higher payment limitations, such as by using the top 10 procedures by volume to determine a payment limitation. It believed this would increase the payment limitation for its product, Dextenza, as evidenced by independent analysis provided with its comment.
Response: We appreciate the feedback, information, and analysis provided by commenters. Based on our analysis, the vast majority of the utilization of a product occurs within the top 5 procedures in which it is used. For example, for the drug Dextenza, approximately 96 percent of the product's utilization is concentrated in the top five procedures. This is also supported by the data presented in the comment letter. We are concerned that using the top ten procedures would include unlikely procedures in the calculation of the payment limitation, which may inappropriately skew the ( printed page 94349) payment limitation. Additionally, given the overall support for using the top 5 procedures we are finalizing that methodology.
Comment: Many commenters recommended CMS update the data it used for its payment limitation calculation in the final rule. Many commenters supported CMS identifying the services with which a product would be expected to be furnished and would typically be packaged absent this policy. However, they did not believe that the procedures listed in CY 2025 OPPS/ASC proposed rule Table 85 were primary procedures. Commenters recommended that CMS use updated data for CY 2025 and only include primary procedures as the basis for calculating the payment limitation. Commenters also recommended CMS not use procedures described by unlisted codes in the payment limitation calculation. They also recommended CMS use Revenue Center Payment Method Indicators to filter the data for primary procedures only, including those HCPCS codes with a Revenue Center Payment Method Indicator of “1” and excluding those HCPCS codes with a Revenue Center Payment Method Indicator value of “9”.
Response: We thank commenters for their feedback and review of our proposed methodology. Our intent is to only use primary procedures with which the qualifying non-opioid treatment for pain relief drug or device would typically be packaged. We acknowledge that in the proposed rule, some non-primary procedures were inadvertently included because they were among the most common procedures furnished with the non-opioid treatment for pain relief by volume; however, payment for the non-opioid treatment for pain relief was not always packaged into those common procedures. Therefore, consistent with the intent of our proposal and the comments received, we have updated the payment limitations accordingly with the most recent data available for this final rule and have excluded any procedures with which the payment for the non-opioid treatment was not packaged.
Using the updated final rule data and excluding non-primary procedures, the HCPCS codes identified are the primary procedure codes with which each non-opioid treatment for pain relief had its payment packaged. None of these updated top five procedures based on volume were unlisted HCPCS codes, so we are not using unlisted HCPCS codes to determine the payment limitation. We are using the CY 2025 OPPS/ASC proposed rule procedure payment rates to determine the payment limitations because we believe they are the best proxy for determining the estimated payment limitations for each product. The proposed rule procedure payment rates were the most recently available data at the time of calculation, and we expect those rates to be relatively unchanged from the final rule rates. We refer commenters to Table 157 to see the final list of primary procedures for CY 2025.
Comment: A few commenters asked for clarification regarding the payment limitation. For example, one commenter noted the payment limitation may pose challenges for use of medical devices under this policy, as the underlying procedures are also likely to be device intensive. Commenters also requested greater clarity regarding which top five services will be selected for the payment calculation process, including whether device offset costs will be included in those procedures. For ASC payments, it is also unclear whether the zero percent device offset would result in the payment being deflated by the ASC scaler, and therefore result in a lower ASC payment for procedures using these technologies. The commenter stated that this is particularly important because many non-opioid pain treatment devices are used in the ASC setting, and the decreased ASC payments may result in unintended consequences around use of these technologies.
Response: Section 1833(t)(16)(G)(ii) requires that we provide temporary additional payment for non-opioid drugs, biologicals, and medical devices, subject to a payment limitation for which the drug, biological, or medical device cost exceeds the portion of the OPPS APC payment rate that is associated with that drug, biological, or medical device. As we discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59429) we proposed to assign a payment offset of zero dollars for qualifying drugs, biologicals, and devices for CY 2025. A zero offset means that we would not offset or remove the amount that the non-opioid product represents from the procedure payment rate when setting payment rates.
We believe it is appropriate to finalize a zero dollar offset for the initial year of the policy as some of these procedures are new products or newly separately paid in the OPPS or ASC settings. For the payment limit determination, the top five services by volume into which a qualifying non-opioid treatment for pain relief has its payment packaged will be used to determine the volume-weighted 18 percent payment limitation. The existing device portions of device-intensive procedures do not contain cost and utilization data for qualifying separately payable non-opioid devices, nor can we currently identify other device costs associated with these medical devices. We anticipate we would only have cost and utilization data from claims of non-opioid medical devices or device costs associated with non-opioid medical devices when CY 2025 data becomes available. Because this data is not yet available, we believe it is appropriate to finalize a zero dollar offset for CY 2025.
Payment rates and the payment limitation for non-opioid drugs, biologicals, and medical devices are not affected by the ASC weight scalar. However, section 1833(i)(10)(A) of the Act requires that the temporary additional payments for non-opioid drugs, biologicals, and medical devices be provided in a budget neutral manner. Therefore, the ASC weight scalar calculation incorporates current and prospective ASC expenditures related to non-opioid treatments. Increases in prospective expenditures related to these non-opioid treatments will decrease the ASC weight scalar, which is applied to the payment rates for surgical procedures and ancillary procedures paid based on the OPPS relative weights and the non-device portion of device-intensive procedures. As discussed in section XIII.H.2. of this final rule with comment period, we project a decrease in prospective expenditures since the non-opioid drugs and biologicals are currently separately payable under the ASC payment system without payment limitation, and because we are unable to estimate prospective utilization and expenditures for the non-opioid medical devices for CY 2025.
After consideration of the public comments we received, we are finalizing our policy to base the 18 percent payment limitation on the volume weighted average of the CY 2025 payment rates of the top five primary procedures by volume into which a non-opioid treatment for pain relief would have their payment packaged, absent this policy. We are finalizing that we will apply the 18 percent payment limitation per date of service billed. We have updated the payment limitations for each qualifying product for CY 2025 with the 2023 claims data available for this final rule and discuss each in the following sections for each specific product. We are also finalizing our policy to create new OPPS status indicators for non-opioid drugs and devices to implement this payment limitation for CY 2025. ( printed page 94350)
e. Payment Limitation With No Claims Data
For drugs, biologicals, and devices with no claims data, such as for newly FDA-approved and marketed products or products that did not previously have their own product-specific HCPCS code by which to track payment and utilization data, we solicited comment on the best approach for determining a payment limitation, as required by section 1833(t)(16)(G)(iii) of the Act. As discussed in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81953), we proposed CMS could utilize the services with which a product would be expected to be furnished and would typically be packaged absent this policy, based on expected clinical use patterns. Determining the service, or group of services, to use to calculate the payment limitation could be accomplished through engagement with interested parties and a review by CMS Medical Officers and clinical staff. Absent engagement from interested parties, we proposed CMS could determine the service, or group of services, to use to calculate the payment limitation based on expected clinical use patterns. CMS could then adjust the services that are used to calculate the payment limitation as claims data becomes available in subsequent years. We welcomed comments on how to set a payment limitation for a product for which we do not have claims data on which to base a payment limitation. The proposed product was described by placeholder HCPCS code C98X4 (ON-Q Pump). We solicited comment on the top five procedures performed with this product, and the HCPCS code that describes the procedure, in order to calculate a volume weighted payment limitation for this device for CY 2025.
We noted that we may update the payment limitation amount in future rulemaking as we gather additional claims data on the utilization of and payment for this product.
Comment: The manufacturer of the ON-Q Pump responded to our comment solicitation on the procedures to use as the basis for the payment limitation for this product, given the absence specific claims data associated with that device. The commenter provided the top 5 primary procedures with which HCPCS code A4306 (Disposable drug delivery system, flow rate of less than 50 ml per hour) was packaged and recommended CMS use these top five procedures to set the payment limitation for this product. However, they noted that HCPCS code A4306 is only a proxy for their product, as this HCPCS code is not specific to the ON-Q pump system, and they also believe that many HOPDs and ASCs are likely not to report the code currently due to the C-APC packaging policy.
Response: We thank the commenter for this information. We have reviewed the commenter's recommended codes and agree with the recommendations because they align with the procedures into which HCPCS code A4306 is currently packaged in our claims data. We also agree that HCPCS code A4306 is a reasonable proxy for the utilization of the ON-Q pump, absent a product specific code with claims data. We note also that there is no available claims data for code C98X4, the proposed placeholder code for the ON-Q pump system, so we are unable to identify procedures with which this code is packaged. Based on our review, we will use the HCPCS codes recommended by the commenter, as shown in Table 157 at the end of this section, as the basis for the calculation of the payment limitation for the ON-Q pump system for CY 2025 and will use the utilization data provided by the commenter for HCPCS code A4306.
Comment: Commenters supported CMS's proposed methodology for determining the procedures used to calculate a new product's specific payment limitation when there is no claims data and encouraged engagement with manufacturers and the use of manufacturer-specific data. Commenters provided specific procedure codes and data supporting the likely use of their products in the HOPD and ASC settings.
Response: We thank commenters for their support. The specific recommendations made by commenters and our assessment of each of those recommendations are explained in the following section f. Qualifying Products.
After consideration of the public comments we received, we are finalizing a policy where CMS will utilize the services with which a product would be expected to be furnished and would typically be packaged absent this policy, to calculate the payment limitation based on expected clinical use patterns for CY 2025. CMS will determine the service, or group of services, to use to calculate the payment limitation through engagement with interested parties and a review by CMS Medical Officers and clinical staff during annual rulemaking. In the absence of engagement from interested parties, we will determine clinically appropriate procedures with which we would expect the drug or device to be frequently used in order to determine the payment limitation, including review of FDA approval materials, procedures identified in literature available to CMS, and other relevant materials. We will consider this topic further in future rulemaking if needed.
f. Qualifying Products
The following table, Table 156, lists the non-opioid alternatives of which we were aware and that we proposed would receive separate payment as a non-opioid pain management drug or device under section 4135 criteria for CY 2025.
As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59427 through 59433), in general, CMS routinely receives comments from readers of the proposed rule with detailed rationales as to why and how they believe a particular drug, biological, medical device, or other item or service should be paid. As such, we solicited comment on whether there are any additional drugs, biologicals, or medical devices that meet the statutory requirements outlined in section 1833(t)(16)(G) and 1833(i)(10).
As discussed previously in this section, there are specific requirements with respect to FDA approval that must be met in order for the product to qualify for separate payment. For medical devices, the statute also requires that the device has demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. Interested parties that believe that a product not addressed in the proposed rule meets the statutory requirements were encouraged to submit information during the comment period that indicated that such product meets the statutory eligibility requirements. We stated that if CMS determines that such product(s) do in fact meet the statutory eligibility requirements, we would finalize separate payment for that product in the CY 2025 OPPS/ASC final rule with comment period. We stated that for drugs and biological products not addressed in the proposed rule, if no comment is submitted that outlines how that drug or biological meets the statutory criteria, then CMS would not finalize separate payment for such product for CY 2025. Additionally, we stated that for medical devices not addressed in the proposed rule, unless a comment was submitted that both outlines how that device meets the statutory criteria and included literature that demonstrates that the device has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal, ( printed page 94351) then CMS would not finalize separate payment for such device for CY 2025.
Comment: Commenters were supportive of our proposals on the qualifying products in Table 156 for CY 2025 and they recommended that we finalize those products for separate payment for CY 2025.
Response: We thank the commenters for their support, and we are finalizing those products for separate payment, with updates to their product specific payment limitations as listed in Table 157.
Comment: The manufacturer of the medical device, ON-Q pump, recommended that CMS include the brand name “ON-Q Pain Relief System” in the HCPCS code descriptor to help decrease the probability that non-qualifying elastomeric infusion pumps would be reported by HOPDs and ASCs. Additionally, this commenter recommended that CMS include in the descriptor “non-opioid anesthetic” so that hospitals and ASCs may also include charges for the drug used with the medical device.
Response: We thank the commenter for their feedback on our proposals. We agree that the C-code descriptor should include the brand of product for better tracking purposes and the descriptors should ensure only products meeting the statutory criteria qualify for separate payment under this provision use the specific HCPCS code. Therefore, we are updating the HCPCS code descriptor accordingly for HCPCS code C98X4, replaced by HCPCS code C9804, to include the brand of the device. However, we do not agree that “non-opioid anesthetic” should be included in the descriptor, as the anesthetic is a separate therapeutic agent that has its own FDA approval.
Several commenters suggested additional non-opioid products that they believed qualified for separate payment under this provision. We have summarized these comments and provided our responses in the following sections.
( printed page 94352)SPRINT Peripheral Nerve Stimulator System (PNS)
Comment: The SPRINT PNS System was suggested by commenters as an alternative to opioids. Commenters stated that this device provides a temporary, minimally invasive solution that utilizes electrical stimulation to target peripheral nerves, offering significant pain relief while avoiding the risks associated with opioid medications. Commenters provided that this product is FDA cleared [196] and has supporting literature.[197] The manufacturer also provided the 5 procedure HCPCS codes with which they believed the SPRINT PNS device would most likely be used for post-operative pain management.
Response: Based on comments received, we believe that this medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared [198] and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal.[199] The medical device does not currently receive transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service, absent this policy.
Therefore, we believe that the SPRINT PNS System meets the statutory requirements and are finalizing that it will be paid separately under this provision. We agree that the top 5 procedures described in the comment letter are reasonable to base the payment limitation on and we will assume an equal utilization of the device with these 5 procedures for the payment limitation calculation, as shown in Table 157 at the end of this section. We note that in the absence of utilization data for the top 5 procedures, we believe it is reasonable to assume an equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 157 for the payment limitation calculation and the new C-code used to describe the device.
IceMan Motorized Cold Therapy Device
Comment: Commenters recommended CMS evaluate and approve motorized cold therapy devices, such as the IceMan, manufactured by Enovis, under our non-opioid treatment for pain relief separate payment policy. They noted that the device is used to reduce pain after surgery. Additionally, they stated that the IceMan product is recognized by FDA as a Class 2, 510(k)-exempt device (product category “ILO”) [200 201 202] and has supporting literature.[203 204 205] The manufacturer stated that the key studies provided on the use of continuous cold therapy include cold therapy delivered by the IceMan device.
The manufacturer also requested that CMS establish a new C-code to report use of their device and assign that code to status indicator “H1,” and they provided 5 HCPCS codes with which they requested CMS establish the payment limitation. These HCPCS codes are 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 29888 (Arthroscopically aided anterior cruciate ligament repair/augmentation or reconstruction), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), 23470 (Arthroplasty, glenohumeral joint; hemiarthroplasty), and 23410 (Repair of ruptured musculotendinous cuff ( e.g., rotator cuff) open; acute). The manufacturer recommended CMS establish a payment limit based on the Medicare payment rate for APC 5114.
Response: Based on the comments received, we believe that the Iceman medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is exempt from 510(k) premarket clearance by the FDA. However, the supporting literature does not demonstrate that the IceMan device specifically has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. The studies provided only broadly reference continuous cold therapy devices. We believe that the device demonstrating the ability to reduce, replace, or avoid opioid use in a clinical trial or peer-reviewed journal must be the same device as the one being studied in order to qualify for separate payment under section 4135. Therefore, we believe that the IceMan Motorized Cold Therapy Device does not meet the statutory requirements and are not finalizing separate payment for it under this provision.
CryoCuff Motorized Cold Therapy Device
Comment: Commenters recommended CMS evaluate and approve motorized cold therapy devices, such as the CryoCuff, manufactured by Enovis, under our non-opioid treatment for pain relief separate payment policy. They noted that the CryoCuff device is used to reduce pain after surgery. Additionally, they stated that the CryoCuff product is recognized by FDA as a Class 2, 510(k)-exempt devices
( printed page 94353)(product category “ILO”) [206 207 208] and has supporting literature.[209 210 211] The manufacturer stated that the key studies provided on the use of continuous cold therapy include cold therapy delivered by the Cryocuff device.
The manufacturer also requested that CMS establish a new C-code to report use of their device and assign that code to status indicator “H1,” and they provided 5 HCPCS codes with which they requested CMS establish the payment limitation. These HCPCS codes are 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 29888 (Arthroscopically aided anterior cruciate ligament repair/augmentation or reconstruction), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g. total shoulder))), 23470 (Arthroplasty, glenohumeral joint; hemiarthroplasty), and 23410 (Repair of ruptured musculotendinous cuff ( e.g., rotator cuff) open; acute). The manufacturer recommended CMS establish a payment limit based on the Medicare payment rate for APC 5114.
Response: Based on comments received, we believe that the CryoCuff medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is exempt from 510(k) premarket clearance by the FDA. However, the supporting literature does not demonstrate that the CryoCuff device specifically has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. The studies provided only broadly reference continuous cold therapy devices. We believe that the device demonstrating the ability to reduce, replace, or avoid opioid use in a clinical trial or peer-reviewed journal must be the same device as the one being studied in order to qualify for separate payment under section 4135.
Therefore, we believe that the CryoCuff Motorized Cold Therapy Device does not meet the statutory requirements and are not finalizing separate payment for it under this provision.
ThermaZone Thermal Therapy Device
Comment: Commenters recommended CMS evaluate and approve thermal therapy devices, such as the ThermaZone manufactured by Innovative Medical Equipment, under our non-opioid treatment for pain relief separate payment policy. They noted that the device is used to reduce pain after surgery. Additionally, they stated that the product is recognized by the FDA as a Class II, 510(k)-exempt device under Product Code “ILO” (Pack, hot or cold, water circulating) and Regulation Number 890.5720 and has supporting literature.[212 213 214 215] Data from an unpublished retrospective cohort study was also provided. The manufacturer stated that numerous studies were published in peer-reviewed journals that show that continuous cold therapy devices can replace and reduce opioids prescribed in the post-surgical setting, which applies to thermal therapy delivered by the ThermaZone device. The manufacturer also provided the top 5 procedures for which the ThermaZone is typically used, weighted by use, and calculated an approximate payment limitation of $1,924.72 based on the HPCCS codes 27447 (Arthroplasty, knee, condyle and plateau; medial and lateral compartments with or without patella resurfacing (total knee arthroplasty)), 27130 (Arthroplasty, acetabular and proximal femoral prosthetic replacement (total hip arthroplasty), with or without autograft or allograft), 29888 (Arthroscopically aided anterior cruciate ligament repair/augmentation or reconstruction), 23472 (Arthroplasty, glenohumeral joint; total shoulder (glenoid and proximal humeral replacement ( e.g., total shoulder))), and 23410 (Repair of ruptured musculotendinous cuff ( e.g., rotator cuff) open; acute)). The manufacturer of the device stated that the device is different from traditional motorized cold therapy devices in that it uses ice-less technology, offers heating and cooling technologies and provides precise temperature control for a variety of temperature settings among other benefits in the commenter's view. Many users of the ThermaZone device commented on the success they had with the device alleviating their pain and reducing their opioid use.
Response: Based on comments received, we believe that the ThermaZone medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is recognized by the FDA as a Class II, 510(k)-exempt device. However, the supporting literature does not demonstrate that the ThermaZone device specifically has the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. The studies provided only broadly reference continuous cold therapy devices. We believe that the device demonstrating the ability to reduce, replace, or avoid opioid use in a clinical trial or peer-reviewed journal must be the same device as the one being studied in order to qualify for separate payment under section 4135. Therefore, we do not believe that the ThermaZone Device meets the statutory requirements and are not finalizing separate payment for it under this provision.
Cyro Nerve Block Therapy (CryoNB)
Comment: Commenters suggested Cryo Nerve Block Therapy (CryoNB) as an alternative to opioids. Atricure, the manufacturer, stated that this therapy is ( printed page 94354) used for postoperative pain management for patients who undergo certain cardiac, thoracic, and other surgical procedures and is administered through cryoablation probes. The manufacturer stated that this device allows a surgeon to ablate nerve tips near the primary surgery incision site to block the sensory nerve from propagating a pain signal after surgery. Commenters provided that this product is FDA cleared with a 510(k) clearance (K182565,[216] K200697,[217] and K233170 [218] ) and has supporting literature.[219 220 221 222] The manufacturer also provided the 5 procedure HCPCS codes with which they believed the CryoNB device would most likely be used for post-operative pain management. The manufacturer also stated that the device does not receive transitional pass-through payment and currently payment is packaged within the covered hospital outpatient department service. The manufacturer also recommended that CMS establish unique C-codes for each of their commercial products and a G-code to describe the additional intraoperative time required.
Response: Based on comments received, we believe that the CryoNB medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. The medical device is not currently receiving transitional passthrough status and we believe it would have payment that is otherwise packaged into a payment for a covered OPD service.
Therefore, we believe that the CryoNB System meets the statutory requirements and should be paid separately under this provision. We agree that the top 5 procedures described in the comment letter are reasonable to base the payment limitation on and will assume an equal utilization of the device with these 5 procedures for the payment limitation calculation. In the absence of utilization data for the top 5 procedures, we believe it is reasonable to assume an equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 157 for the payment limitation calculation and new C-code to describe the device. Section 4135 of the CAA, 2023 statute does not address separate payment for the services associated with the provision of a medical device, and therefore we are not creating a G-code to describe the additional intraoperative time required at this time.
ambIT Electronic Pain Control System
Comment: The ambIT Electronic Pain Control System, a medical device manufactured by Avanos, was suggested by commenters as an alternative to opioids. Commenters stated that this system provides a similar benefit as the ON-Q pump, as it is a battery powered pump that delivers a local non-opioid anesthetic to a patient's surgical site or nerves to provide target pain relief while avoid systemic effects. Commenters clarified that this version of the pump is disposable. Commenters provided that this product is FDA cleared under a 510(k) [223] and has supporting literature.[224] The manufacturer also provided the 5 procedure HCPCS codes in which they believed the ambIT device would most likely be used for post-operative pain management. The manufacturer also stated that the product does not currently have pass-through payment status and currently would have its payment packaged into the payment for a covered OPD service. The manufacturer also provided suggested descriptor language for the creation of a product specific C-code.
Response: Based on comments received, we believe that the ambIT medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. The medical device is not currently receiving transitional passthrough status and we believe would have payment that is otherwise packaged into a payment for a covered OPD service.
Therefore, we believe that the ambIT System meets the statutory requirements and should be paid separately under this provision. We agree that the top 5 procedures described in the comment letter are reasonable to base the payment limitation. Please see Table 157 for the payment limitation calculation and new C-code to describe the device.
SimplFusor Elastomeric Pump
Comment: Commenters recommended CMS consider SimplFusor, a device manufactured by Baxter, as an effective alternative to opioids. Commenters stated that this device is functionally equivalent to and interchangeable with the ON-Q elastomeric pump; therefore, the SimplFusor pump should also qualify. Commenters provided that this product is FDA cleared through a 510(k) (K151650). The commenter stated that the SimplFusor device has not specifically been studied, but that literature, specifically the literature on the ON-Q pump, demonstrates that the use of elastomeric infusion pumps to deliver pain management therapies reduces post-operative pain and opioid use. The manufacturer stated that the product is not on pass-through status and would usually have its cost packaged into the APC associated with its use.
Response: Based on the comments received, we believe that the SimplFusor medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared. The medical device is not currently receiving transitional passthrough status and we believe would have payment that is otherwise packaged into a payment for a covered OPD service.
However, this product does not have literature supporting that its specific use reduced opioid use. We acknowledge that commenters provided FDA documentation that their device is substantially equivalent to a device that ( printed page 94355) does demonstrate the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. We believe that the device demonstrating the ability to reduce, replace, or avoid opioid use in a clinical trial or peer-reviewed journal must be the same device as the one being studied in order to qualify for separate payment under section 4135. Therefore, we believe that the SimplFusor System does not meet the statutory requirements and are not finalizing separate payment for it under this provision.
Iovera System
Comment: The Iovera System manufacturer, Pacira Biosciences, Inc., suggested that this medical device should qualify for separate payment under this provision. The manufacturer stated that this device is used to apply freezing cold to the peripheral nervous system for the treatment of pain, referred to as cyroneurolysis or cryoanalgesia. Commenters provided that this product is used to deliver a therapy to reduce postoperative pain, or produce post-surgical or reginal analgesia, is FDA cleared under a 510(k) [225] and has supporting literature.[226 227 228 229] The manufacturer also provided two HCPCS codes in which the device would have its payment packaged.
Response: Based on comments received, we believe that the Iovera medical device is used to deliver a therapy to reduce postoperative pain or produce post-surgical or regional analgesia. We confirmed that this product is FDA cleared and has supporting literature demonstrating the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal. The medical device is not currently receiving transitional passthrough status and we believe would have payment that is otherwise packaged into a payment for a covered OPD service.
Therefore, we believe that the Iovera System meets the statutory requirements and should be paid separately under this provision. The manufacturer only provided two procedures in which the device is used, and we agree with those two procedures are likely the most common procedures in which the device is used, given the clinical nature of the device. Therefore, for this product only, we are only using these top 2 procedures as the basis for calculating the payment limitation for the device. We will assume an equal utilization of the device with these 2 procedures for the payment limitation calculation as shown in Table 157 at the end of this section. In the absence of utilization data for the top 2 procedures, we believe it is reasonable to assume an equal utilization for purposes of calculating the payment limitation until claims data are available. Please see Table 157 for the payment limitation calculation and new C-code to describe the device.
Additional Non-Qualifying Products
Comment: Commenters suggested additional products for CMS to consider under this policy. One example is the MAKO System, an FDA-cleared medical device used in hip and knee joint procedures to enable a more predictable implant placement to plan surgical experience. The system is intended to assist the surgeon in providing software-defined spatial boundaries for orientation and reference information to anatomical structures during orthopedic procedures. Another example is the PMD-200, which is a device that uses a finger probe to measure physiological signals that are elevated during surgical stimulation, and then a machine learning algorithm uses these variables to provide a clinician a continuous numerical value for pain to assist the clinician with opioid dosing.
Response: We appreciate these comments, but we do not believe these medical devices need to be evaluated under this policy because they are not directly delivering pain management therapies. The statute specifies that any device under this policy should deliver a therapy to reduce postoperative pain or produce postsurgical or regional analgesia. Both the MAKO and the PMD-200 function as supplemental tools that can assist the clinician, such as robotic assistance or machine learning assistance, in assessing the patient and planning treatment. However, the devices themselves are not medical devices that are used to deliver a therapy to reduce postoperative pain or produce postsurgical or regional analgesia. As these devices do not meet the basic definitions to be considered under this policy, we are not evaluating them further against the qualifying criteria.
Comment: Commenters also recommended other products to CMS but did not provide any supporting documentation or information on the products.
Response: For those products suggested by commenters that did not provide supplemental information, we are unable to fully evaluate the product against the statutory criteria for inclusion in this policy and therefore are not finalizing separate payment for any of these products. The final list of qualifying products can be found in Table 158.
( printed page 94356) ( printed page 94357) ( printed page 94358)( printed page 94359)
( printed page 94360)
The implementation of section 4135 of the CAA, 2023, which establishes the eligibility criteria for temporary additional payments for certain non-opioid treatments for pain relief, has been finalized in this section. We note that the HCPCS codes describing the qualifying devices and drugs in Table 158 have been placed on the ASC covered ancillary procedures list. While we acknowledge HCPCS code A4306 (Disposable drug delivery system, flow rate of less than 50 ml per hour) was previously discussed as a code that could describe a general category of medical pumps in which the ON-Q pump system could have been categorized, it did not previously appear on the ASC covered ancillary procedures list. However, we also note that Medicare Administrative Contractors (MACs) determine whether a drug, device, procedure, or other service meets all program requirements and conditions for coverage and payment. HOPDs and ASC only receive payment for qualifying drugs, biologicals, and medical devices when the appropriate MAC determines that the service meets the relevant conditions for coverage and payment. As we have consistently stated in past OPPS/ASC final rules (see, e.g.,87 FR 71879 and 88 FR 81660 through 81661), the fact that a drug, device, procedure or service is assigned a HCPCS code and a payment rate under the OPPS does not imply coverage by the Medicare program, but indicates only how the product, procedure, or service may be paid if covered by the program (see, e.g., Pub 100-04 Medicare Claims Processing, Transmittal 11937).
We proposed revisions to 42 CFR 416.174 for the ASC payment system and § 419.43 for the OPPS to codify these policies. Specifically, we proposed revisions to § 419.174(a) to establish the eligibility for non-opioid pain management drugs and biologicals, and by adding modifications to paragraphs (a)(1) through (3) to outline drug and biological FDA approval requirements, the exclusion of drugs and biologicals with pass-through status, and the requirement that the drug or biological has payment that is packaged. We proposed new § 419.174(b) to establish the eligibility for non-opioid pain management medical devices, which includes new paragraphs (b)(1) through (4). These new paragraphs describe medical device FDA requirements, medical device clinical trial or peer-reviewed journal requirements, the exclusion of medical devices with pass-through status, and the requirement that the medical device has payment that is packaged. New paragraph (c) describes the payment amounts for qualifying drugs and biologicals in paragraph (c)(1) and medical devices in paragraph (c)(2), as well as the payment limitation for drugs, biologicals, and medical devices in paragraph (c)(3). Similarly, we also propose new § 419.43(k), which contains payment for non-opioid pain management drugs and biologicals. Specifically, new paragraph (k)(1) outlines the eligibility for separate payment for non-opioid pain management drugs and biologicals, with new paragraphs outlining (i) the drug or biological's required FDA status, (ii) the drug or biological's pass-through status, and (iii) the drug or biological's packaged status.
We also proposed to add new § 419.43(k)(2), which contains payment for non-opioid pain management medical devices. Specifically, new paragraph (k)(2) outlines the eligibility for separate payment for non-opioid pain management medical devices, with new paragraphs outlining (i) the medical device's required FDA status, (ii) the medical device clinical trial or peer-reviewed journal requirements, (iii) the medical device's pass-through status, and (iv) the medical device's packaged status. New § 419.43(k)(3) describes the separate payment amount for qualifying non-opioid treatments for pain relief. Specifically, paragraph ( printed page 94361) (k)(3)(i) sets the separate payment amount for a qualifying drug or biological, paragraph (k)(3)(ii) sets the separate payment amount for a qualifying medical device, and paragraph (k)(3)(iii) sets the payment limitation for drugs, biologicals, and medical devices.
We did not receive any public comments on the proposed regulation text and are finalizing as proposed with slight changes to one provision. In particular, we are clarifying the description of the payment limitation at §§ 416.174(c)(3) and 419.43(k)(3)(iii) to state that the volume weighted average for the payment limitation will be based on the most frequent 5 OPD primary procedures into which a non-opioid treatment for pain relief would be packaged. We believe the “packaged” language is more accurate and consistent with our proposed policy than the previous language “furnished with.” As previously discussed, for the payment limitation calculation, non-primary procedures will not be included in the top five procedures list, even if they are the most common procedures furnished with that non-opioid treatment for pain relief by volume. Therefore, we are modifying the proposed regulatory text to specifically state that the payment limitation will be based on the most frequent five OPD primary procedures into which payment for the non-opioid treatment for pain relief is packaged. Except for this modification, we are finalizing the regulation text as proposed. Additionally, we are revising § 416.164(a)(4) to remove “with the exception of non-opioid pain management drugs and biologicals that function as a supply when used in a surgical procedure as determined by CMS under § 416.174,” as these drugs will be separately payable in both the OPPS and ASC under this finalized policy. Similarly, we are revising § 416.164(b)(6) to read “Non-opioid pain management drugs, biologicals, and medical devices as determined by CMS under § 416.174,” as now qualifying medical devices will be on the ASC covered ancillary services list under this finalized policy.
G. New Technology Intraocular Lenses (NTIOLs)
New Technology Intraocular Lenses (NTIOLs) are intraocular lenses that replace a patient's natural lens that has been removed in cataract surgery and that also meet the requirements listed in § 416.195.
1. NTIOL Application Cycle
Our process for reviewing applications to establish new classes of NTIOLs is as follows:
- Applicants submit their NTIOL requests for review to CMS by the annual deadline which is announced in the annual OPPS/ASC final rule with comment period. For a request to be considered complete, we require submission of the information requested in the guidance document titled “Application Process and Information Requirements for Requests for a New Class of New Technology Intraocular Lenses (NTIOLs) or Inclusion of an IOL in an Existing NTIOL Class” posted on the CMS website at:https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/new-technology-intraocular-lenses-ntiols.
- We announce annually, in the proposed rule updating the ASC and OPPS payment rates for the following calendar year, a list of all requests to establish new NTIOL classes accepted for review during the calendar year in which the proposal is published. In accordance with section 141(b)(3) of Public Law 103-432 and our regulations at § 416.185(b), the deadline for receipt of public comments is 30 days following publication of the list of requests in the proposed rule.
- In the final rule with comment period updating the ASC and OPPS payment rates for the following calendar year, we—
++ Provide a list of determinations made as a result of our review of all new NTIOL class requests and public comments.
++ When a new NTIOL class is created, identify the predominant characteristic of NTIOLs in that class that sets them apart from other IOLs (including those previously approved as members of other expired or active NTIOL classes) and that is associated with an improved clinical outcome.
++ Set the date of implementation of a payment adjustment in the case of approval of an IOL as a member of a new NTIOL class prospectively as of 30 days after publication of the ASC payment update final rule, consistent with the statutory requirement.
++ Announce the deadline for submitting requests for review of an application for a new NTIOL class for the following calendar year.
2. Requests To Establish New NTIOL Classes for CY 2025
We did not receive any requests for review to establish a new NTIOL class for CY 2025 by March 1, 2024, the due date published in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81956).
3. Payment Adjustment
The current payment adjustment for a 5-year period from the implementation date of a new NTIOL class is $50 per lens. Since implementation of the process for adjustment of payment amounts for NTIOLs in 1999, we have not revised the payment adjustment amount, and we did not propose to revise the payment adjustment amount for CY 2025.
Comment: Commenters recommended that we increase the NTIOL payment adjustment. Commenters noted that the payment adjustment has not been adjusted since CY 1999 and has significantly lagged the overall inflation rate and does not reflect the technological advancements to support continued innovation. Some commenters recommended that we establish the NTIOL payment adjustment at $91.04 while others recommended we establish the payment adjustment at $94.63 and that we increase such payment adjustment annually.
Response: We responded to this comment in last year's final rule with comment period. We refer readers to the CY 2024 OPPS/ASC final rule with comment period for our response on an increase to the NTIOL $50 per lens payment adjustment (88 FR 81955 through 81956).
4. Announcement of CY 2026 Deadline for Submitting Requests for CMS Review of Applications for a New Class of NTIOLs
In accordance with 42 CFR 416.185(a) of our regulations, CMS announces that in order to be considered for payment effective beginning in CY 2025, requests for review of applications for a new class of new technology IOLs must be received by 5 p.m. Eastern Standard Time, on March 1, 2025. Send requests via email to outpatientpps@cms.hhs.gov or by mail to ASC/NTIOL, Division of Outpatient Care, Mailstop C4-05-17, Centers for Medicare and Medicaid Services, 7500 Security Boulevard, Baltimore, MD 21244-1850. To be considered, requests for NTIOL reviews must include information requested on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/new-technology-intraocular-lenses-ntiols. ( printed page 94362)
H. Calculation of the ASC Payment Rates and the ASC Conversion Factor
1. Background
In the August 2, 2007, ASC final rule (72 FR 42493), we established our policy to base ASC relative payment weights and payment rates under the revised ASC payment system on APC groups and the OPPS relative payment weights. Consistent with that policy and the requirement at section 1833(i)(2)(D)(ii) of the Act that the revised payment system be implemented so that it would be budget neutral, the initial ASC conversion factor (CY 2008) was calculated so that estimated total Medicare payments under the revised ASC payment system in the first year would be budget neutral to estimated total Medicare payments under the prior (CY 2007) ASC payment system (the ASC conversion factor is multiplied by the relative payment weights calculated for many ASC services in order to establish payment rates). That is, application of the ASC conversion factor was designed to result in aggregate Medicare expenditures under the revised ASC payment system in CY 2008 being equal to aggregate Medicare expenditures that would have occurred in CY 2008 in the absence of the revised system, taking into consideration the cap on ASC payments in CY 2007, as required under section 1833(i)(2)(E) of the Act (72 FR 42522). We adopted a policy to make the system budget neutral in subsequent calendar years (72 FR 42532 through 42533; § 416.171(e)).
We note that we consider the term “expenditures” in the context of the budget neutrality requirement under section 1833(i)(2)(D)(ii) of the Act to mean expenditures from the Medicare Part B Trust Fund. We do not consider expenditures to include beneficiary coinsurance and copayments. This distinction was important for the CY 2008 ASC budget neutrality model that considered payments across the OPPS, ASC, and MPFS payment systems. However, because coinsurance is almost always 20 percent for ASC services, this interpretation of expenditures has minimal impact for subsequent budget neutrality adjustments calculated within the revised ASC payment system.
In the CY 2008 OPPS/ASC final rule with comment period (72 FR 66857 through 66858), we set out a step-by-step illustration of the final budget neutrality adjustment calculation based on the methodology finalized in the August 2, 2007, ASC final rule (72 FR 42521 through 42531) and as applied to updated data available for the CY 2008 OPPS/ASC final rule with comment period. The application of that methodology to the data available for the CY 2008 OPPS/ASC final rule with comment period resulted in a budget neutrality adjustment of 0.65.
For CY 2008, we adopted the OPPS relative payment weights as the ASC relative payment weights for most services and, consistent with the final policy, we calculated the CY 2008 ASC payment rates by multiplying the ASC relative payment weights by the final CY 2008 ASC conversion factor of $41.401. For covered office-based surgical procedures, covered ancillary radiology services (excluding covered ancillary radiology services involving certain nuclear medicine procedures or involving the use of contrast agents, as discussed in section XIII.D.2 of the CY 2023 OPPS/ASC proposed rule (87 FR 44715 through 44716)), and certain diagnostic tests within the medicine range that are covered ancillary services, the established policy is to set the payment rate at the lower of the MPFS unadjusted nonfacility PE RVU-based amount or the amount calculated using the ASC standard ratesetting methodology. Further, as discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66841 through 66843), we also adopted alternative ratesetting methodologies for specific types of services (for example, device-intensive procedures).
As discussed in the August 2, 2007 ASC final rule (72 FR 42517 through 42518) and as codified at § 416.172(c) of the regulations, the revised ASC payment system accounts for geographic wage variation when calculating individual ASC payments by applying the pre-floor and pre-reclassified IPPS hospital wage indexes to the labor-related share, which is 50 percent of the ASC payment amount based on a GAO report of ASC costs using 2004 survey data. Beginning in CY 2008, CMS accounted for geographic wage variation in labor costs when calculating individual ASC payments by applying the pre-floor and pre-reclassified hospital wage index values that CMS calculates for payment under the IPPS, using updated Core Based Statistical Areas (CBSAs) issued by OMB in June 2003.
The reclassification provision in section 1886(d)(10) of the Act is specific to acute care hospitals. We believe that using the most recently available pre-floor and pre-reclassified IPPS hospital wage indexes result in the most appropriate adjustment to the labor portion of ASC costs. We continue to believe that the pre-floor, pre-reclassified hospital wage indexes, which are updated yearly and are used by several other Medicare payment systems, appropriately account for geographic variation in labor costs for ASCs (89 FR 23424). Therefore, the wage index for an ASC is the pre-floor and pre-reclassified hospital wage index for the fiscal year under the IPPS of the CBSA that maps to the CBSA where the ASC is located.
On July 21, 2023, OMB issued OMB Bulletin No. 23-01, which provides the delineations of all Metropolitan Statistical Areas, Metropolitan Divisions, Micropolitan Statistical Areas, Combined Statistical Areas, and New England City and Town Areas in the United States and Puerto Rico based on the standards published on July 16, 2021, in the Federal Register (86 FR 37770) and 2020 Census Bureau data. (A copy of this bulletin may be obtained at: https://www.whitehouse.gov/wp-content/uploads/2023/07/OMB-Bulletin-23-01.pdf.) The pre-floor pre-reclassified IPPS hospital wage indexes for CY 2024 do not reflect OMB's new area delineations and, because the ASC wage indexes are the pre-floor and pre-reclassified IPPS hospital wages indexes, the CY 2024 ASC wage indexes do not reflect the most recent OMB changes. As discussed in the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36139 through 36150), we proposed to use the new CBSAs delineations issued by OMB in OMB Bulletin 23-01 for the IPPS hospital wage index beginning in CY 2025. Therefore, because the ASC wage indexes for the calendar year are the pre-floor and pre-reclassified IPPS hospital wage indexes for the fiscal year, we proposed to incorporate the new OMB delineations into CY 2025 ASC wage indexes. We believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the ASC wage index system by creating a more accurate representation of current geographic variations in wage levels.
In adopting the revised CBSA delineations from the 2010 Census data which were issued by OMB on July 15, 2015, through OMB Bulletin No. 15-01, for ASCs in counties that would see a decline in their ASC wage index for CY 2015, we adopted a blended wage index of 50 percent of the CY 2014 wage index value and 50 percent of the CY 2015 wage index value (79 FR 66937). However, we note that other Medicare payment systems incorporate a policy of capping year-to-year wage index decreases for each facility at 5 percent of the previous year's wage index value (89 FR 23431 through 23433). We believe such a policy would also be appropriate for the ASC payment system as we transition to the CBSA ( printed page 94363) delineations based on the 2020 Census data. As discussed in the IPPS/LTCH FY 2025 proposed rule, the 5-percent cap mitigates any large negative impacts of adopting the new delineations and prevents large year-to-year declines in wage index values as a means to reduce volatility (89 FR 36150). Therefore, for CY 2025, we proposed to incorporate the new OMB delineations into the CY 2025 ASC wage indexes and propose to apply a 5-percent cap on wage index decreases at the county level (or county-equivalent level) and the ASC wage index of that county would apply to all ASCs physically located in that county. We note that this 5-percent cap is applied in a budget neutral manner. The 5-percent cap reduces the wage index scalar for a calendar year which, in turn, will reduce the ASC conversion factor and the payment rates for covered ASC services in counties that are not affected by the 5-percent cap on wage index decreases. Further, we solicited comments on whether we should extend this policy after CY 2025 and permanently adopt a budget-neutral 5-percent cap on year-to-year wage index decreases.
Comment: Commenters supported our proposal to implement a 5-percent cap on year-to-year wage index decreases. However, two commenters requested that we should apply the OPPS geographic reclassifications and wage index floor policies to further align payment between the ASC payment system and the OPPS.
Response: As previously discussed in this section, the reclassification provision in section 1886(d)(10) of the Act is specific to acute care hospitals. We believe that using the most recently available pre-floor and pre-reclassified IPPS hospital wage indexes result in the most appropriate adjustment to the labor portion of ASC costs.
After consideration of the public comments we received, we are finalizing our proposal to implement a 5-percent cap on wage index decrease for CY 2025.
The final CY 2025 ASC wage indexes fully reflect the OMB labor market area delineations (including the revisions to the OMB labor market delineations discussed previously, as set forth in OMB Bulletin No. 23-01) including replacing the eight counties with the county-equivalent planning regions of Connecticut. We note that, in certain instances, there might be urban or rural areas for which there is no IPPS hospital that has wage index data that could be used to set the wage index for that area. When all of the areas contiguous to the urban CBSA of interest are rural and there is no IPPS hospital that has wage index data that could be used to set the wage index for that area, our policy has been to determine the ASC wage index by calculating the average of all wage indexes for urban areas in the State (75 FR 72058 through 72059). In other situations, where there are no IPPS hospitals located in a relevant labor market area, we apply our current policy of calculating an urban or rural area's wage index by calculating the average of the wage indexes for CBSAs (or metropolitan divisions where applicable) that are contiguous to the area with no wage index. For example, for CY 2025, we are applying a proxy wage index based on this methodology to ASCs located in CBSA 25980 (Hinesville, GA) and in CBSA 35 (Rural North Dakota).
2. Calculation of the ASC Payment Rates
a. Updating the ASC Relative Payment Weights for CY 2025 and Future Years
We update the ASC relative payment weights each year using the national OPPS relative payment weights (and PFS nonfacility PE RVU-based amounts, as applicable) for that same calendar year and uniformly scale the ASC relative payment weights for each update year to make them budget neutral (72 FR 42533). The OPPS relative payment weights are scaled to maintain budget neutrality for the OPPS. We then scale the OPPS relative payment weights again to establish the ASC relative payment weights. To accomplish this, we hold estimated total ASC payment levels constant between calendar years for purposes of maintaining budget neutrality in the ASC payment system. That is, we apply the weight scalar to ensure that projected expenditures from the updated ASC payment weights in the ASC payment system are equal to what would be the current expenditures based on the scaled ASC payment weights. In this way, we ensure budget neutrality and that the only changes to total payments to ASCs result from increases or decreases in the ASC payment update factor.
As discussed in section II.A.1.a of this final rule with comment period, we are using the CY 2023 claims data to be consistent with the OPPS claims data for the proposed rule. Consistent with our established policy, we proposed to scale the CY 2025 relative payment weights for ASCs according to the following method. Holding ASC utilization, the ASC conversion factor, and the mix of services constant from CY 2023, we proposed to compare the estimated total payment using the CY 2024 ASC relative payment weights with the estimated total payment using the CY 2025 ASC relative payment weights to take into account the changes in the OPPS relative payment weights between CY 2024 and CY 2025.
In consideration of our policy to provide a higher ASC payment rate with ASC complexity adjustment codes for certain primary procedures when performed with add-on packaged services, we incorporated estimated total spending and estimated utilization for these codes in our budget neutrality calculation for CYs 2023 and 2024. We estimated in the CY 2023 OPPS/ASC final rule with comment period (87 FR 72094) that the impact on CY 2023 estimated total payments from our finalized CY 2023 ASC complexity adjustment codes would be $5 million in spending and we finalized our proposal to incorporate this $5 million in estimated CY 2023 total payments for the budget neutrality calculation. Based on CY 2023 utilization data, we estimated that the actual amount of spending on the new CY 2023 ASC complexity adjustment codes for CY 2023 was $24 million. We estimated that there will not be an additional increase in ASC spending related to our proposed ASC complexity adjustment codes for CY 2025.
Additionally, as discussed in section XIII.E of the CY 2025 OPPS/ASC proposed rule, section 4135(a) and (b) of the CAA, 2023, titled “Access to Non-Opioid Treatments for Pain Relief,” amended section 1833(t)(16) and section 1833(i) of the Act, respectively, to provides for temporary separate payments for non-opioid treatments for pain relief. As discussed in further detail in section XIII.E of the CY 2025 OPPS/ASC proposed rule, for qualifying nonopioid products, we proposed to apply an 18 percent payment limitation on the volume weighted payment average of the top 5 services associated with the use of the qualifying non-opioid product. In CY 2024, four of these qualifying nonopioid products are separately payable without the 18 percent payment limitation—HCPCS Codes C9089 (Bupivacaine implant, 1 mg), C9290 (Inj, bupivacaine liposome), J1096 (Dexametha opth insert 0.1 mg), and J1097 (Phenylep ketorolac opth soln). Therefore, to maintain budget neutrality, we estimated the total anticipated reduction as a result of the 18 percent payment limitation required by section 4135 of the CAA, 2023. Using CY 2023 utilization for these four drugs and CY 2024 ASC payment rates, we anticipated that the 18 percent payment limitation will reduce CY 2025 ASC expenditures by approximately $9 million. Therefore, we proposed to ( printed page 94364) reduce estimated CY 2025 total payments by $9 million in our weight scalar calculation as a result of section 4135 of the CAA, 2023.
We proposed to use the ratio of estimated CY 2024 to estimate CY 2025 total payments (the weight scalar) to scale the ASC relative payment weights for CY 2025. The proposed CY 2025 ASC weight scalar was 0.876. We note that we have historically displayed this figure rounded to the nearest ten thousandth; however, we believe this level of specificity is unnecessarily burdensome for an ASC payment system that is less than one-tenth the size of the OPPS (in which the weight scalar is rounded to the nearest ten-thousandth). An ASC weight scalar rounded to the nearest ten thousandth is highly sensitive to spending changes and can require the costly reissuance of new ASC payment rates from only very minor payment rate changes within the ASC Payment System, such as a revised PFS conversion factor as a result of Congressional action. Therefore, for CY 2025 and subsequent calendar years, we proposed to set the ASC weight scalar rounded to the nearest thousandth. Consistent with historical practice, we proposed to scale, using this method, the ASC relative payment weights of covered surgical procedures, covered ancillary radiology services, and certain diagnostic tests within the medicine range of CPT codes, which are covered ancillary services for which the ASC payment rates are based on OPPS relative payment weights.
We proposed that we would not scale ASC payment for separately payable covered ancillary services that have a predetermined national payment amount (that is, their national ASC payment amounts are not based on OPPS relative payment weights), such as drugs and biologicals that are separately paid or services that are contractor-priced or paid at reasonable cost in ASCs. Any service with a predetermined national payment amount would be included in the ASC budget neutrality comparison, but scaling of the ASC relative payment weights would not apply to those services. The ASC payment weights for those services without predetermined national payment amounts (that is, those services with national payment amounts that would be based on OPPS relative payment weights) would be scaled to eliminate any difference in the total payment between the current year and the update year.
For any given year's ratesetting, we typically use the most recent full calendar year of claims data to model budget neutrality adjustments. We proposed to use the CY 2023 claims data to model our budget neutrality adjustment for CY 2025.
Comment: Many commenters reiterated a longstanding recommendation that we discontinue our budget neutrality adjustment under the ASC payment system. The commenters believe this to be antiquated and preventing the migration of procedures from the more expensive hospital setting to the ASC setting. Commenters noted that the OPPS relative weights used for the ASC payment system have already been scaled for budget neutrality under the OPPS. They argued the budget neutrality adjustment under the ASC payment system could be combined with the OPPS budget neutrality adjustment thereby establishing a single OPPS and ASC weight scalar. One commenter requested more transparency in the ASC weight scalar calculation and the driving factors behind the change in the scalar.
Response: We are not accepting the commenters' recommendation to discontinue applying the ASC weight scalar. As we have stated in past rulemaking (82 FR 59421), applying the ASC weight scalar ensures that the ASC payment system remains budget neutral. We apply the ASC weight scalar to scaled OPPS relative weights to ensure that current Medicare payments under the ASC payment system do not increase as a result of newer data to determine the cost relativity between surgical procedures. The scaled prospective OPPS relative weights that are used to determine scaled prospective ASC relative weights have not, as commenters suggest, been adjusted to achieve budget neutrality within the ASC payment system prior to the application of the ASC weight scalar. We also note that no stakeholder presented empirical evidence that the budget neutrality adjustment under the ASC payment system has impacted beneficiary access to surgical procedures in the ASC setting.
In regard to the commenter's request for more transparency in the ASC weight scalar calculation, we are unclear what additional information is needed. We provide Limited Data Set files on the CMS website such that the public can use the same data the agency used in determining the ASC weight scalar for this final rule with comment period and we describe our budget neutrality adjustment process in this section of the final rule with comment period. However, to better assist the public in understanding changes in the ASC weight scalar, we have provided more context and information in the paragraph below regarding the impact of CY 2025 changes in our inputs and how they impact the ASC weight scalar. We will continue to provide this information in our OPPS/ASC proposed and final rules to enable interested parties to better understand the changes in the ASC weight scalar.
After consideration of the public comments we received, we are finalizing our proposal to use the ratio of CY 2024 to CY 2025 total payments (the weight scalar) to scale the ASC relative payment weights for CY 2025. We estimate that there will not be an additional increase in ASC spending related to our final ASC complexity adjustment codes for CY 2025. Based on the revised payment limitations from data available for this final rule with comment period, we estimate the implementation of section 4135 of the CAA, 2023 to decrease ASC spending by $5.5 million for CY 2025 as a result of the payment limitation to currently separately payable drugs qualifying nonopioid products. Therefore, we reduced estimated CY 2025 total payments by $5.5 million in our weight scalar calculation. We will monitor the utilization and will adjust our payments to maintain budget neutrality accordingly, if appropriate, through future rulemaking.
The final CY 2025 ASC weight scalar is 0.872. The final CY 2025 ASC weight scalar represents a 1.9 percent decrease from the final CY 2024 ASC weight scalar of 0.8889. We estimate that increases in the scaled OPPS relative weights as a result of relatively greater increases in the geometric mean cost of commonly-performed surgical procedures are the most significant factor causing the decline in the ASC weight scalar.
Consistent with historical practice, we are finalizing our proposal to scale the ASC relative payment weights of covered surgical procedures, covered ancillary radiology services, and certain diagnostic tests within the medicine range of CPT codes, which are covered ancillary services for which the ASC payment rates are based on OPPS relative payment weights. Additionally, we do not anticipate any budgetary impact related to our final list of ASC complexity adjustment codes for CY 2025.
b. Updating the ASC Conversion Factor
Under the OPPS, we typically apply a budget neutrality adjustment for provider-level changes, most notably a change in the wage index values for the upcoming year, to the conversion factor. Consistent with our final ASC payment policy, for the CY 2017 ASC payment ( printed page 94365) system and subsequent years, in the CY 2017 OPPS/ASC final rule with comment period (81 FR 79751 through 79753), we finalized our policy to calculate and apply a budget neutrality adjustment to the ASC conversion factor for supplier-level changes in wage index values for the upcoming year, just as the OPPS wage index budget neutrality adjustment is calculated and applied to the OPPS conversion factor.
For CY 2025, we calculated the proposed adjustment for the ASC payment system by using the most recent CY 2023 claims data available and estimating the difference in total payment that would be created by introducing the proposed CY 2025 ASC wage indexes. Specifically, holding CY 2023 ASC utilization, service-mix, and the proposed CY 2025 national payment rates after application of the weight scalar constant, we calculated the total adjusted payment using the CY 2024 ASC wage indexes and the total adjusted payment using the proposed CY 2025 ASC wage indexes which included our proposed 5-percent cap on wage index declines. We used the 50 percent labor-related share for both total adjusted payment calculations. We then compared the total adjusted payment calculated with the CY 2024 ASC wage indexes to the total adjusted payment calculated with the proposed CY 2025 ASC wage indexes and applied the resulting ratio of 0.9958 (the proposed CY 2025 ASC wage index budget neutrality adjustment) to the CY 2024 ASC conversion factor to calculate the proposed CY 2025 ASC conversion factor.
Section 1833(i)(2)(D)(v) of the Act requires that the ASC conversion factor be reduced by a productivity adjustment in each calendar year. Section 1886(b)(3)(B)(xi)(II) of the Act defines the productivity adjustment to be equal to the 10-year moving average of changes in annual economy-wide private nonfarm business multifactor productivity (MFP). We finalized the methodology for calculating the productivity adjustment in the CY 2011 PFS final rule with comment period (75 FR 73394 through 73396) and revised it in the CY 2012 PFS final rule with comment period (76 FR 73300 through 73301) and the CY 2016 OPPS/ASC final rule with comment period (80 FR 70500 through 70501). The proposed productivity adjustment for CY 2025 was projected to be 0.4 percentage point, as published in the FY 2025 IPPS/LTCH PPS proposed rule (89 FR 36204) based on IGI's 2023 fourth quarter forecast.
Section 1833(i)(2)(C)(i) of the Act requires that, if the Secretary has not updated amounts established under the revised ASC payment system in a calendar year, the payment amounts shall be increased by the percentage increase in the Consumer Price Index for all urban consumers (CPI-U), U.S. city average, as estimated by the Secretary for the 12-month period ending with the midpoint of the year involved. The statute does not mandate the adoption of any particular update mechanism, but it requires the payment amounts to be increased by the CPI-U in the absence of any update. Because the Secretary updates the ASC payment amounts annually, we adopted a policy, which we codified at § 416.171(a)(2)(ii)), to update the ASC conversion factor using the CPI-U for CY 2010 and subsequent calendar years.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59075 through 59080), we finalized a policy to apply the productivity-adjusted hospital market basket update to ASC payment system rates for an interim period of 5 years (CY 2019 through CY 2023), during which we would assess whether there is a migration of the performance of procedures from the hospital setting to the ASC setting as a result of the use of a productivity-adjusted hospital market basket update, as well as whether there are any unintended consequences, such as less than expected migration of the performance of procedures from the hospital setting to the ASC setting. The most recent available full year of claims data to assess the expected migration applying the productivity-adjusted hospital market basket update during the interim period would fall within the period from CY 2019 through CY 2022. However, the impact of the COVID-19 PHE on health care utilization, in particular in CY 2020, was tremendously profound, particularly for elective surgeries, because many beneficiaries avoided healthcare settings, when possible, to avoid possible infection from the SARS-CoV-2 virus. As a result, it is nearly impossible to disentangle the effects from the COVID-19 PHE in our analysis of whether the higher update factor for the ASC payment system caused increased migration to the ASC setting. To analyze whether procedures migrated from the hospital setting to the ASC setting, we need to use claims data from a period during which the COVID-19 PHE had less of an impact on health care utilization. Therefore, for CY 2024, we finalized our proposal to extend the 5-year interim period an additional 2 years, that is, through CY 2024 and CY 2025. We believed hospital outpatient and ASC utilization data from CYs 2023 and 2024 will enable us to more accurately analyze whether the application of the productivity-adjusted hospital market basket update to the ASC payment system had an effect on the migration of services from the hospital setting to the ASC setting. We revised our regulations at 42 CFR 416.171(a)(2)(iii) and (iv), which establish the annual update to the ASC conversion factor, to reflect this 2-year extension. We also revised our regulations at § 416.171(a)(2)(vi) and (vii), which establish the 2.0 percentage point reduction for ASCs that fail to meet the standards for reporting ASC quality measures, and § 416.171(a)(2)(viii)(B) and (C), which establish the productivity adjustment, to reflect this 2-year extension.
For CY 2025, we proposed to utilize the proposed hospital market basket percentage increase of 3.0 percent reduced by the proposed productivity adjustment of 0.4 percentage point, resulting in a proposed productivity-adjusted hospital market basket update of 2.6 percent for ASCs meeting the quality reporting requirements. Therefore, we proposed to apply a proposed 2.6 percent productivity-adjusted hospital market basket update factor to the CY 2024 ASC conversion factor for ASCs meeting the quality reporting requirements to determine the CY 2025 ASC payment amounts. The ASCQR Program affected payment rates beginning in CY 2014 and, under this program, there is a 2.0 percentage points reduction to the productivity-adjusted hospital market basket update factor for ASCs that fail to meet the ASCQR Program requirements. We refer readers to section XIV.E of the CY 2019 OPPS/ASC final rule with comment period (83 FR 59138 through 59139) and section XIV.E of this final rule with comment period for a detailed discussion of our policies regarding payment reduction for ASCs that fail to meet ASCQR Program requirements. We proposed to utilize the proposed inpatient hospital market basket percentage increase of 3.0 percent reduced by 2.0 percentage points for ASCs that do not meet the quality reporting requirements and then reduced by the proposed 0.4 percentage point productivity adjustment. Therefore, we proposed to apply a 0.6 percent productivity-adjusted hospital market basket update factor to the CY 2024 ASC conversion factor for ASCs not meeting the quality reporting requirements. We also proposed that if more recent data are subsequently available (for example, a more recent estimate of the inpatient hospital market ( printed page 94366) basket percentage increase or productivity adjustment), we would use such data, if appropriate, to determine the CY 2025 ASC update for the CY 2025 OPPS/ASC final rule with comment period.
For CY 2025, we proposed to adjust the CY 2024 ASC conversion factor ($53.514) by the proposed wage index budget neutrality factor of 0.9958 in addition to the proposed productivity-adjusted hospital market basket update of 2.6 percent discussed previously, which results in a proposed CY 2025 ASC conversion factor of $54.675 (a 2.2 percent increase) for ASCs meeting the quality reporting requirements. For ASCs not meeting the quality reporting requirements, we proposed to adjust the CY 2024 ASC conversion factor ($53.514) by the proposed wage index budget neutrality factor of 0.9958 in addition to the proposed quality reporting/productivity-adjusted hospital market basket update of 0.2 percent discussed previously, which resulted in a proposed CY 2025 ASC conversion factor of $53.609 for ASCs not meeting the quality reporting requirements.
Comment: Many commenters supported our proposal to increase the ASC conversion factor by the productivity-adjusted hospital market basket update and recommended that we continue applying the same updated factor for both the OPPS and ASC payment system in subsequent years to align both payment systems and mitigate growing payment disparities between ASCs and HOPDs. Some commenters argued that the proposed payment rate update was insufficient when compared to the broader increases in healthcare costs. However, one commenter representing hospitals opposed our proposal and recommended that the agency collect ASC costs such that it can develop an appropriate update factor for ASCs.
Response: We appreciate the commenters' support. In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72094 through 72096), we finalized our proposal to increase the ASC conversion factor by the productivity-adjusted hospital market basket update for CYs 2024 and 2025. In the CY 2025 OPPS/ASC proposed rule (89 FR 59436 through 59437), we proposed to continue this policy and increase the proposed ASC conversion factor by the productivity-adjusted hospital market basket update for CY 2025. We intend to address commenters' recommendations regarding the most applicable and appropriate update factor for the ASC payment system in our CY 2026 OPPS/ASC proposed rule. As discussed in section XII.C of this final rule with comment period, we appreciate the thoughtful comments and responses regarding our solicitation on methods that would mitigate the burden of reporting costs on ASCs while also collecting enough data to reliably use in the determination of ASC costs. We will take these comments into consideration or future rulemaking.
With respect to commenter's concern that the hospital market basket update would insufficiently increase ASC payments, as discussed in section II.B of this final rule with comment period, section 1833(t)(3)(C)(iv) of the Act requires the OPPS OPD fee schedule increase factor for a year to equal the IPPS market basket percentage increase factor (hospital market basket update) applicable under section 1886(b)(3)(B)(iii) to hospital discharges in the fiscal year ending in such year. Accordingly, we are unable to adopt a final OPPS OPD fee schedule increase factor different than the hospital market basket update finalized in the FY 2025 IPPS/LTCH PPS final rule.
After consideration of the public comments we received, for CY 2025, we are finalizing using the hospital market basket update of 3.4 percent reduced by the productivity adjustment of 0.5 percentage point, resulting in a productivity-adjusted hospital market basket update factor of 2.9 percent for ASCs meeting the quality reporting requirements. Therefore, we apply a 2.9 percent productivity-adjusted hospital market basket update to the CY 2024 ASC conversion factor for ASCs meeting the quality reporting requirements to determine the C Y 2025 ASC payments. We are finalizing the hospital market basket update of 3.4 percent reduced by 2.0 percentage points for ASCs that do not meet the quality reporting requirements and then reduced by the productivity adjustment of 0.5 percentage point. Therefore, we apply a 0.9 percent productivity-adjusted hospital market basket update factor to the CY 2024 ASC conversion factor for ASCs not meeting the quality reporting requirements.
For CY 2025, we are adjusting the CY 2024 ASC conversion factor ($53.514) by a wage index budget neutrality factor of 0.9969 in addition to the productivity-adjusted hospital market basket update of 2.9 percent, discussed above, which results in a final CY 2025 ASC conversion factor of $54.895 for ASCs meeting quality reporting requirements. For ASCs not meeting quality reporting requirements, we are adjusting the CY 2024 ASC conversion factor ($53.514) by the wage index budget neutrality factor of 0.9969 in addition to the reduced productivity-adjusted hospital market basket update of 0.9 percent, discussed above, which results in a final CY 2025 ASC conversion factor of $53.828 for ASCs not meeting the quality reporting requirements.
3. Display of the Final CY 2025 ASC Payment Rates
Addenda AA and BB to this final with comment period (which are available on the CMS website) display the final ASC payment rates for CY 2025 for covered surgical procedures and covered ancillary services, respectively. The final payment rates included in Addenda AA and BB to this final rule with comment period reflect the full ASC payment update and not the reduced payment update used to calculate payment rates for ASCs not meeting the quality reporting requirements under the ASCQR Program.
These Addenda contain several types of information related to the final CY 2025 payment rates. Specifically, in Addendum AA, a “Y” in the column titled “To be Subject to Multiple Procedure Discounting” indicates that the surgical procedure would be subject to the multiple procedure payment reduction policy. As discussed in the CY 2008 OPPS/ASC final rule with comment period (72 FR 66829 through 66830), most covered surgical procedures are subject to a 50 percent reduction in the ASC payment for the lower-paying procedure when more than one procedure is performed in a single operative session.
The values displayed in the column titled “Final CY 2025 Payment Weight” are the final relative payment weights for each of the listed services for CY 2025. The final relative payment weights for all covered surgical procedures and covered ancillary services where the ASC payment rates are based on OPPS relative payment weights were scaled for budget neutrality. Therefore, scaling was not applied to the device portion of the device-intensive procedures; services that are paid at the MPFS nonfacility PE RVU-based amount; separately payable covered ancillary services that have a predetermined national payment amount, such as drugs and biologicals and brachytherapy sources that are separately paid under the OPPS; or services that are contractor-priced or paid at reasonable cost in ASCs. This includes separate payment for non-opioid pain management drugs.
To derive the final CY 2025 payment rate displayed in the “Final CY 2025 Payment Rate” column, each ASC payment weight in the “Final CY 2025 ( printed page 94367) Payment Weight” column was multiplied by the final CY 2025 conversion factor. The conversion factor includes a budget neutrality adjustment for changes in the wage index values and the annual update as reduced by the productivity adjustment. The final CY 2025 ASC conversion factor uses the final CY 2025 productivity-adjusted hospital market basket update factor of 2.9 percent (which is equal to the inpatient hospital market basket update of 3.4 percent reduced by the productivity adjustment of 0.5 percentage point).
In Addendum BB, there are no relative payment weights displayed in the “Final CY 2025 Payment Weight” column for items and services with predetermined national payment amounts, such as separately payable drugs and biologicals. The “Final CY 2025 Payment” column displays the final CY 2025 national unadjusted ASC payment rates for all items and services. The final CY 2025 ASC payment rates listed in Addendum BB for separately payable drugs and biologicals are based on the most recently available data used for payment in physicians' offices.
For CY 2021, we finalized adding a new column to ASC Addendum BB titled “Drug Pass-Through Expiration during Calendar Year” where we flag through the use of an asterisk each drug for which pass-through payment is expiring during the calendar year (that is, on a date other than December 31st).
Addendum EE to this final rule with comment period provides the HCPCS codes and short descriptors for surgical procedures that are to be excluded from payment in ASCs for CY 2025.
Addendum FF to this final rule with comment period displays the OPPS payment rate (based on the standard ratesetting methodology), the APC device offset percentage, the device offset percentage for determining device-intensive status (based on the standard ratesetting methodology), and the device portion of the ASC payment rate for CY 2025 for covered surgical procedures.
XIV. Cross-Program Measures for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs
A. Background
We refer readers to sections XV, XVI, and XVII of this final rule with comment period for program specific background information, including the statutory authorities and previously finalized and newly finalized measure sets, for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs, respectively.
B. CMS Commitment To Advancing Health Equity Using Quality Measurement
We are committed to advancing health equity and improving health outcomes through our quality reporting programs. The CMS Framework for Health Equity acknowledges that “addressing health and healthcare disparities and achieving health equity should underpin efforts to focus attention and drive action on our nation's top health priorities.” CMS defines health equity as “the attainment of the highest level of health for all people, where everyone has a fair and just opportunity to attain their optimal health regardless of race, ethnicity, disability, sexual orientation, gender identity, socioeconomic status, geography, preferred language, or other factors that affect access to care and health outcomes.” [255]
Significant and persistent disparities in healthcare outcomes exist in the United States (U.S.). Belonging to a racial or ethnic minority group, living with a disability, being a member of the lesbian, gay, bisexual, transgender, and queer (LGBTQ+) community, living in a rural area, or being near or below the poverty level are often associated with worse health outcomes.[256 257 258] Health disparities manifest primarily as worse health outcomes in populations where access to care is inequitable.[259 260] Such differences persist across geography and healthcare settings irrespective of improvements in quality of care over time.[261 262] Inequities in the social determinants of health affecting these groups are interrelated and influence a wide range of health and quality of life outcomes and risks.[263]
Inequities related to the social determinants of health may affect health-related social needs (HRSNs). HRSNs are individual-level, adverse social conditions that negatively impact an individual's health or healthcare and are associated with worse health outcomes and increased healthcare utilization.[264] While HRSNs account for 50 to 70 percent of health outcomes, the mechanisms by which this connection emerges are complex and multifaceted.[265 266] Growing evidence demonstrates that specific HRSNs are directly associated with patient health outcomes as well as healthcare utilization, costs, and performance in quality-based payment programs.[267 268] The persistent interactions among individuals' HRSNs, medical providers' practices and behaviors, and community resources significantly impact ( printed page 94368) healthcare access, quality, and costs, as described in the CMS Equity Plan for Improving Quality in Medicare.[269 270] Assessment of HRSNs is an essential mechanism for capturing the interaction between social, community, and environmental factors associated with health status and health outcomes.[271 272] Studies indicate that healthcare facility leadership can positively influence culture for better quality, patient outcomes, and experience of care.[273 274 275]
We are committed to supporting healthcare facility leadership in building a culture of equity that focuses on eliminating health disparities to provide patients with high quality healthcare through the collection and public reporting of health equity focused measures, including in outpatient care settings.[276]
Health equity quality measurement supports the Meaningful Measures 2.0 goal to “Leverage Quality Measures to Promote Equity and Close Gaps in Care” as well as the objective to “commit to a patient-centered approach in quality measure and value-based incentives programs.” Additionally, under the CMS National Quality Strategy, adoption of health equity quality measures would support addressing the quality priority to “advance health equity and whole-person care” by employing a uniform approach for gathering, reporting, and analyzing health equity data across CMS quality programs.[277]
1. Adoption of the Hospital Commitment to Health Equity (HCHE) Measure for the Hospital Outpatient Quality Reporting (OQR) and Rural Emergency Hospital Quality Reporting (REHQR) Programs and the Facility Commitment to Health Equity (FCHE) Measure for the Ambulatory Surgical Center Quality Reporting (ASCQR) Program Beginning with the CY 2025 Reporting Period/CY 2027 Payment Determination or Program Determination.[278]
a. Background
Strong and committed leadership from healthcare facility management is essential in shifting organizational culture to reduce health disparities and reach health equity goals.[279] The Agency for Healthcare Research and Quality and the Joint Commission identified that healthcare facility leadership plays an important role in promoting a culture of quality and safety.[280 281] The Institute of Healthcare Improvement's research shows that health equity must be a priority championed by leadership teams to improve both patient access to needed healthcare services and outcomes among disadvantaged populations.[282] Based upon these findings, we believe that healthcare facility leadership is instrumental in setting specific, measurable, attainable, realistic, and time-based (SMART) goals to assess progress towards achieving equity priorities and ensuring high-quality care is equally accessible to all individuals.
In the FY 2022 IPPS/LTCH PPS proposed rule (86 FR 25592 and 25593), we sought public comment on potential future efforts to address health equity in the Hospital Inpatient Quality Reporting (IQR) Program, particularly the inclusion of a structural measure to assess the degree of hospital leadership commitment to collecting and monitoring health equity performance data. We specifically sought feedback on (1) conceptual and measurement priorities to facilitate organizational efforts to improve health equity; and (2) an appropriate measure regarding organizational commitment to health equity and accessibility for individuals with intellectual and developmental disabilities. In response, we received support for the development and implementation of a health equity structural measure. We also received comments expressing concerns about such a health equity structural measure. We refer readers to the FY 2022 IPPS/LTCH PPS final rule (86 FR 45414 through 45416) for summaries of the comments we received related to this solicitation.
We considered this feedback with the intent that future health equity measures would align across the Medicare quality reporting programs, including the Hospital OQR, REHQR, and ASCQR Programs, to ensure equitable care across both inpatient and outpatient settings to the greatest extent possible within facilities and hospitals participating in Medicare. In addition, we believe that measuring leadership commitment to health equity should not be limited to the inpatient hospital setting but should cover the continuum of care as patients seek and receive care at various care settings.
We initially developed the HCHE and FCHE measures for use in the Hospital IQR and Inpatient Psychiatric Facility Quality Reporting (IPFQR) Programs, respectively, with the expectation of expansion into other Medicare quality reporting programs. The HCHE and FCHE measures are attestation-based structural measures that assess hospitals' and facilities' commitment to ( printed page 94369) health equity across the following five domains adapted from the CMS Office of Minority Health's (OMH) “Building an Organizational Response to Health Disparities” framework: equity as a strategic priority, data calculation, data analysis, quality improvement, and leadership engagement.[283] These measures are intended to encourage hospitals and facilities to analyze their data to understand how factors, including race, ethnicity, and the social determinants of health can contribute to the delivery of more equitable care.[284]
We believe these domains provide actionable focus areas for the assessment of healthcare facility leadership commitment because they are foundational to incentivizing hospitals and facilities to collect and utilize data to identify critical equity gaps, implement plans to address those gaps, and ensure that resources are dedicated toward healthcare equity initiatives. We also believe these measures support hospitals and facilities in quality improvement, promote efficient and effective use of resources, and leverage available data.
Adoption of these measures in the Hospital OQR, REHQR, and ASCQR Programs would support our efforts to align measures across CMS quality reporting programs, including the Hospital IQR Program (87 FR 49191 through 49201), IPFQR Program (88 FR 51100 through 51107), PPS-Exempt Cancer Hospital Quality Reporting (PCHQR) Program (88 FR 59204 through 59210), and End-Stage Renal Disease Quality Incentive Program (ESRD QIP) (88 FR 76437 through 76446). We believe that alignment across the quality reporting programs is important to ensure that health equity, which impacts patients regardless of where they receive their care, is addressed in every healthcare delivery setting. Adopting these measures across quality reporting programs would incentivize quality reporting entities to collect and utilize data to identify critical equity gaps, implement plans to address said gaps, and ensure that resources are dedicated toward addressing health equity initiatives.
b. Overview of the Measures
The HCHE and FCHE measures assess a hospital's or facility's commitment to health equity by using equity-focused organizational domains aimed at advancing health equity for all patients, including but not limited to those in racial and ethnic minority groups, people with disabilities, members of the LGBTQ+ community, individuals with limited English proficiency, rural populations, religious minorities, and people facing socioeconomic challenges. Table 159 and Table 160 describe the five attestation domains and their elements for the HCHE and FCHE measures, respectively.
( printed page 94370) ( printed page 94371)The HCHE measure is currently used in the Hospital IQR and PCHQR Programs. As further discussed below, we proposed to adopt the HCHE measure for the Hospital OQR and REHQR Programs. The FCHE measure is currently used in the IPFQR Program and ESRD QIP. As further discussed below, we proposed to adopt the FCHE measure for the ASCQR Program.
We note that there are two measure specification variations between the HCHE and FCHE measures, as reflected in Tables 159 and 160. First, Table 159 references hospitals (such as HOPDs and REHs) in connection with HCHE; Table 160 references facilities (such as ASCs, which are not hospitals) in ( printed page 94372) connection with FCHE. Second, Domain 2C of the HCHE measure requires hospitals to use a certified electronic health record (EHR) technology (CEHRT) [285] in order to attest “yes”; Domain 2C of the FCHE measure requires facilities to use EHR technology, but does not require the use of CEHRT, in order to attest “yes.” We recognize that ASCs have governance structures and operational circumstances that are distinct from hospitals. We also recognize that many non-hospital facilities, including ASCs, have not adopted CEHRT, but may use some EHR technology,[286] justifying this variation in Domain 2C between the HCHE and FCHE measures.[287]
c. Pre-Rulemaking Measure Review
As required under section 1890A of the Act, the Consensus-Based Entity (CBE), currently Battelle, established the Partnership for Quality Measurement (PQM), comprised of clinicians, patients, measure experts, and health information technology specialists, to participate in the pre-rulemaking process and the measure endorsement process and provide input on the selection of quality and efficiency measures. The pre-rulemaking process, which we refer to as the Pre-Rulemaking Measure Review (PRMR), includes a review of measures published on the publicly available list of Measures Under Consideration (MUC List) by one of several committees convened by the PQM for the purpose of providing multi-stakeholder input to the Secretary on the selection of quality and efficiency measures under consideration for use in certain Medicare quality programs, including the Hospital OQR, REHQR, and ASCQR Programs. More details regarding the PRMR process may be found in the PQM Guidebook of Policies and Procedures for Pre-Rulemaking Measure Review and Measure Set Review, including details of the measure review processes in Chapter 3.[288]
As part of the PRMR process, the Hospital Recommendation Group reviewed and voted on the HCHE and FCHE measures during their meeting on January 18 and 19, 2024.[289 290] The voting results of the HCHE measure for the Hospital OQR and REHQR Programs were “recommend with conditions,” and the voting results of the FCHE measure were “recommended without conditions” for the ASCQR Program. The conditions for the HCHE measure for the Hospital OQR and REHQR Programs were: (1) obtaining CBE endorsement; (2) additional specificity around attestation requirements; and (3) ongoing data collection for further measure testing, particularly with regard to smaller entities.[291] We have taken these conditions into account, as follows, and proposed both of these measures for adoption. We discuss CBE endorsement in section XIV.B.1.d of this final rule with comment period below.
Regarding the condition to provide additional specificity around attestation requirements, we note that these domains were developed based on the recommendations from a technical expert panel (TEP) that informed our initial selection and development of this measure.[292] We also addressed this concern during the January 18-19, 2024 PRMR meeting by sharing that there are accompanying guidance documents available to provide information and examples of qualifying activities for the HCHE measure (which can also be applied to the FCHE measure).[293 294]
With respect to the condition related to ongoing data collection for further measure testing due to concerns that smaller entities may face challenges regarding data collection and analysis, we reiterate that HCHE is an attestation measure only in Hospital OQR, a pay-for-reporting program, and REHQR, a program with no associated payment adjustment.[295] While we acknowledge the limitations in testing structural measures, we believe this measure captures useful information regarding providers' commitment to promoting health equity to inform patient choice. We have therefore considered the Hospital Recommendation Group's concerns and determined that they are adequately addressed.
d. CBE Endorsement
Section 1833 (t)(17)(C)(i) of the Act provides that the Hospital OQR Program, to the extent feasible and practicable, shall include measures set forth by one or more national consensus building entities (not necessarily the CBE). Under section 1833(i)(7)(B) of the Act, this requirement at section 1833(t)(17)(C) applies to the ASCQR Program except as the Secretary may otherwise provide. For the Hospital OQR Program and ASCQR Program, we note that 1833(t)(17) of the Act does not require that each measure we adopt for these programs be CBE-endorsed (75 FR 72064 and 72065 for the Hospital OQR Program and 76 FR 74494 for the ASCQR Program).
Section 1833(t)(17)(C)(i) of the Act provides that the Hospital OQR Program, to the extent feasible and practicable, shall include measures set forth by one or more national consensus building entities (not necessarily the CBE). Under section 1833(i)(7)(B) of the Act, this requirement at section 1833(t)(17)(C) applies to the ASCQR Program except as the Secretary may otherwise provide. For the Hospital OQR Program and ASCQR Program, we note that section 1833(t)(17) of the Act ( printed page 94373) does not require that each measure we adopt for these programs be CBE-endorsed (75 FR 72064 and 72065 for the Hospital OQR Program and 76 FR 74494 for the ASCQR Program).
Section 1833(t)(17)(C)(i) of the Act also requires measures developed for the Hospital OQR Program to reflect consensus among affected parties. Under section 1833(i)(7)(B) of the Act, this requirement also applies to the ASCQR Program except as the Secretary may otherwise provide. As we have noted in previous rulemaking, consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment (75 FR 72064 and 72065 for the Hospital OQR Program and 76 FR 74494 for the ASCQR Program).
For the REHQR Program, section 1861(kkk)(7)(C)(i) of the Act generally requires that quality measures specified by the Secretary for the REHQR Program be endorsed by a CBE; however, section 1861(kkk)(7)(C)(ii) of the Act provides an exception to the general CBE-endorsement requirement, stating that in the case of a specified area or medical topic determined appropriate by the Secretary for which a measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed CBE-endorsed measures and were unable to identify any other CBE-endorsed measures on this topic, and therefore we believe the exception in section 1861(kkk)(7)(C)(ii) of the Act applies for purposes of this measure for the REHQR Program.
At this time, we find no other feasible and practicable measures set forth by a national consensus building entity on the topic of a hospital's or facility's leadership commitment to health equity. While we recognize the value of measures undergoing CBE endorsement review and prefer to use endorsed measures, there are currently no CBE-endorsed measures that address hospital or facility commitment to health equity. Given the urgency of achieving health equity, it is important to implement this measure as soon as possible. As previously noted, the HCHE measure was developed based on the consensus of a TEP whose recommendations informed the initial selection, development, and emphasis of the importance of this measure and subsequently the FCHE measure, which, as noted in section XIV.B.1.b of this final rule with comment period above, is a similar measure with only two measure specification variations to accommodate setting-specific realities with regard to CEHRT adoption.[296] We will consider submitting the HCHE and FCHE measures to the CBE for endorsement in the future.
e. Measure Calculation
The proposed HCHE and FCHE measures each consist of the same five attestation-based domains as shown in Table 159 and Table 160 respectively, subject to variations noted above.
The numerator of both the HCHE and FCHE measures would capture the total number of domains to which the hospital or facility is able to attest affirmatively, up to a maximum of five domains. We proposed that a hospital or facility would only receive a point for a domain if it attested “yes” to all the elements within that domain. We would not accept an attestation whereby a hospital or facility attests “yes” to some, but not all, of the elements; in the event a hospital or facility would not be able to attest “yes” to one or more elements within a domain, or the entirety of a domain, they would respond “no.” For example, for Domain 1, if the hospital or facility's strategic plan meets elements (A) and (B), but not (C) and (D) of Domain 1, then the hospital or facility would not be able to affirmatively attest “yes” and would receive zero points for Domain 1.
The denominator of both the HCHE and FCHE measures would constitute a total of five points (that is, one point per domain).
We also refer readers to the measure specifications, available on our QualityNet website.[297]
As noted above, Domain 2C of the HCHE measure requires the use of CEHRT, while Domain 2C of the FCHE measure requires the use of EHR technology, which is not required to be certified by ONC in accordance with ONC's requirements. We made this distinction because we recognize that many non-hospital facilities, including ASCs, currently have not adopted CEHRT and instead use non-certified EHR technology,[298] while a majority of hospitals have adopted CEHRT.[299] Although REHs are a new Medicare provider type, the majority of REH-eligible facilities, as noted in the CY 2024 OPPS/ASC final rule (88 FR 82069), have met requirements for the reporting of electronic clinical quality measures (eCQMs), which require CEHRT, under the Medicare Promoting Interoperability Program.
f. Data Submission Requirements
In the CY 2025 OPPS/ASC proposed rule, we proposed to require hospitals, REHs, and ASCs to submit their yes/no attestation responses on these structural measures in all three programs by an annual deadline using the CMS-designated information system (currently, the Hospital Quality Reporting (HQR) system) consistent with the data submission requirements of these measures in the Hospital IQR, IPFQR and PCHQR Programs. We refer readers to sections XV.E.2.a, XVI.E.3.b, and XVII.E.2.a of this final rule with comment period for additional details regarding data submission deadlines for web-based measure reporting such as the HCHE and FCHE measures for the Hospital OQR, REHQR, and ASCQR Programs, respectively.
We invited public comment on our proposal to adopt the HCHE measure for the Hospital OQR Program beginning with the CY 2025 reporting period/CY 2027 payment determination, to adopt the HCHE measure for the REHQR Program beginning with the CY 2025 reporting period/CY 2027 program determination, and to adopt the FCHE measure for the ASCQR Program beginning with the CY 2025 reporting period/CY 2027 payment determination.
The following comments and responses are applicable to both the HCHE and FCHE measures for the Hospital OQR, REHQR, and ASCQR Programs. Program-specific comments and responses are addressed subsequently.
Comment: Many commenters expressed support for our proposal to adopt the HCHE and FCHE measures. Many commenters stated that these measures will align quality ( printed page 94374) measurement across quality reporting programs as well as across hospitals and systems. Commenters expressed that this alignment would provide important measurable data to reduce health disparities and work towards a more equitable healthcare system. A few commenters stated that systematic data collection is an important first step hospitals must take to identify and work to prevent health inequities. A commenter specifically stated that these measures can serve as a foundation to address inequities in HIV care and outcomes across racial/ethnic minorities, sexual orientation and gender identity, and other social factors.
Response: We thank commenters for their support.
Comment: Several commenters recommended that CMS include voluntary reporting or delay mandatory reporting for these measures to allow hospitals and facilities adequate time to expand health equity improvement initiatives to the outpatient areas, as well as create the appropriate structure for consistent communication and build and deploy processes, such as utilizing EHR technology. A few commenters recommended phasing in measure domains incrementally to allow hospitals and facilities time to familiarize themselves with the measure. Commenters also suggested that delaying mandatory reporting would provide CMS with more time to work with stakeholders to better define each domain, and to continue to evolve and potentially respecify the measures to reduce burden on providers.
Response: We appreciate commenters' concerns about the timing of mandatory reporting; however, achieving health equity is a pressing issue which deserves serious focus and rapid action. We emphasize that this is an annual measure that was proposed for adoption beginning with the CY 2025 reporting period/CY 2027 payment or program determination, which will allow hospitals, REHs, and ASCs more than a year before the submission deadline of May 15, 2026, at which time these healthcare facilities would submit attestations regarding the identified domains during the CY 2025 reporting period. Hospitals, REHs, and ASCs can spend the remainder of CY 2024 and CY 2025 addressing potential barriers to positive attestations for the HCHE/FCHE measure before December 31, 2025. We additionally consider this measure to be a building block for a more comprehensive suite of measures that would assess progress in providing high-quality healthcare for all patients regardless of social risk factors or demographic characteristics. We also reiterate that this is an attestation measure intended to assess the capacity and commitments of the HOPD, REH, or ASC in regard to health equity and help these healthcare facilities identify potential gap areas on this topic to improve quality of care.
We also note that hospitals that participate in the Hospital IQR Program will already have 1 year of experience reporting data on the HCHE measure for the FY 2025 payment determination (that is, data submitted in FY 2024 representing the FY 2023 performance period) (87 FR 49191 through 49201). Given the timing of this measure's implementation in the Hospital IQR Program, hospitals, REHs, and ASCs will have had the opportunity to learn from the experiences of hospitals participating in the Hospital IQR Program, including best practices for minimally burdensome assessment of performance on the required domains. We will monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if we identify implementation strategies to reduce burden.
Comment: A few commenters recommended CMS develop an audit function to ensure the accuracy of self-reported data and to ensure that a facility's or hospital's stated commitments to health equity are being implemented in a way that creates meaningful change.
Response: We understand commenters' concerns regarding the accuracy of self-reported data. While we do not have a specific means to validate hospital and facility attestation to these measures at this time, we have provided an Attestation Guidance document and a FAQ document, which clearly define what constitutes an affirmative attestation and provide answers to frequently asked questions, for the HCHE measure in Hospital IQR Program.[300 301] We intend to provide specific guidance for the Hospital OQR, REHQR, and ASCQR Programs, consistent with the specifications described in Tables 159 and 160 and requirements being finalized in this section of the final rule, in the future. Hospitals, REHs, and ASCs may also refer to the measure specifications for additional attestation guidance.[302]
Comment: A few commenters recommended that CMS communicate with stakeholders to refine its portfolio of health-equity related measures while creating a streamlined measure set that is applicable and standardized across settings. A commenter recommended collaborating with stakeholders toward the development of a measure that is meaningful for hospitals as they design interventions to address HRSNs in partnership with their communities. Another commenter recommended that CMS evaluate the results of these measures in conjunction with stakeholder communication before expanding measure requirements.
Response: We thank the commenters for this feedback. We reiterate that stakeholders convened by the PRMR entity previously reviewed the HCHE and FCHE measures and provided feedback during the PRMR process. We refer readers to section XIV.B.1.c of this final rule with comment period for a discussion of the PRMR process. The purpose of the PRMR process is to solicit and provide multi-stakeholder input to the Secretary on the selection of quality and efficiency measures under consideration for use in CMS quality programs, including the Hospital OQR, REHQR, and ASCQR Programs. We will continue identifying opportunities for collaboration with all stakeholders to align assessment of social health drivers across CMS programs. The HCHE and FCHE measures are foundational additions to our programs, and we plan to identify additional health equity measures for development and potential subsequent adoption. We will continue to seek stakeholder input through the pre-rulemaking process, as required under section 1890A of the Act, as well as through notice-and-comment rulemaking, before modifying the HCHE and FCHE measures or adding new measures to ensure meaningful outcomes.
Comment: Several commenters recommended strategies to best implement the HCHE and FCHE measures. Commenters recommended additional factors to include in these measures to make them more meaningful, including the collection of demographics other than race and ethnicity (such as age, gender, and ( printed page 94375) health literacy). A commenter recommended using a community needs assessment and implementing regulatory flexibilities to explore additional ways to support hospitals in addressing health equity. Another commenter recommended that CMS create a plan of action prior to measure implementation to meet the equity intention behind these measures.
Several commenters recommended the adoption of quality reporting measures to supplement the HCHE and FCHE measures. A commenter specifically recommended the adaptation and inclusion of the Age Friendly Hospital measure in the outpatient setting, as it is currently included in the Hospital IQR measure set.
Several commenters recommended that CMS consider alternative approaches to address health equity. A few commenters recommended that CMS further refine or develop alternative measure concepts, while incorporating a more holistic view of the hospital or facility, to accurately evaluate leadership commitment to health equity. A commenter suggested the adoption of measures with numerical results instead of yes or no answers. This commenter stated that structural measures do not always drive action since they are not linked to direct quality outcomes and do not have a sound validation process. The commenter further recommended that it would be more impactful for CMS to develop new outcome measures related to health equity that drive action and measurable change. Another commenter suggested that CMS revisit its current definition of “health equity” to instead focus on the absence of disparities in the processes or outcomes of hospital care, leading to a more narrow and actionable definition. Additionally, a commenter recommended that CMS explore developing measures using data that CMS already collects through claims or could be collected through other digital measurement sources.
Response: We thank commenters for their recommendations. Adoption of the HCHE and FCHE measures in the Hospital OQR, REHQR, and ASCQR programs would lay the groundwork for a more comprehensive suite of measures that would assess progress in providing high-quality healthcare for all patients regardless of social risk factors or demographic characteristics. We will consider this feedback in future rulemaking and measure development.
Comment: A few commenters expressed concern about hospital and facility penalization. A commenter cautioned that these measures penalize providers rather than incentivize them to comply with data collection and implementation that advances health equity. Another commenter recommended sensitivity and a formulaic coefficient when evaluating under-resourced facilities to ensure congruency on quality performance relative to facilities with more resources. This commenter also recommended adjusting programmatic requirements to ensure that reporting on quality measures is feasible for all facilities and that under-resourced facilities do not face undue difficulty or burdensome penalties that could affect access to care for vulnerable populations.
Response: We acknowledge commenters' concerns regarding resources, including increased data collection burden. The benefits of encouraging hospital and facility commitment to health equity outweigh the burden of attestation under this measure. We will consider the concept of a single streamlined health equity measure at the organizational level in future rulemaking. We remind commenters that the Hospital OQR and ASCQR Programs are pay-for-reporting programs. Participants are only penalized for failure to submit required data on quality measures as specified; their payments are not affected based on their performance on measures. Additionally, the REHQR Program does not include a financial incentive or penalty for REHs.
As described in the CY 2025 OPPS/ASC proposed rule (89 FR 59516, 59522, 59526), the estimated burden for HOPDs and REHs is 10 minutes annually for the HCHE measure, and the estimated burden for ASCs is 10 minutes annually for the FCHE measure, which is a reasonable expectation for quality reporting data submission. Finally, we would also like to note that the HCHE and FCHE measures are not meant to compare facilities with one another, but rather for facilities to identify and address structural gaps within their strategic planning, data collection and analysis, quality improvement, and leadership engagement.
Comment: Several commenters opposed to the adoption of the HCHE and FCHE measures expressed concern that, without additional requirements for facilities to make changes based on identified health equity gaps, the HCHE and FCHE measures may only serve as a checklist measure rather than incentivizing actionable change at the systemic level. A few commenters recommended CMS provide more timely and actionable data to hospitals to yield more prompt advancements in safety and quality. Another commenter expressed concern that there is no published clinical evidence to link hospital measurement of social determinants of health to improvements in quality of care or outcomes.
Response: We thank commenters for their feedback and acknowledge their concerns. Hospitals and facilities are responsible for ensuring the best outcomes possible for the patients that they serve, including identifying potential barriers to optimal health and helping patients identify resources to address those barriers. These measures are important, foundational measures for improving health equity among those that have been disadvantaged or underserved by the healthcare system. Adoption of the HCHE and FCHE measures will incentivize hospitals and facilities to collect and utilize data across the five domains to identify critical equity gaps, implement plans to address these gaps, and ensure that resources are dedicated toward addressing health equity initiatives, therefore resulting in actionable initiatives and change. These measures aim to support hospitals and facilities in leveraging available data, pursuing focused quality improvement activities, and promoting efficient and effective use of resources. Leadership commitment to health equity can foster organizational competencies aimed at achieving health equity for the facility's patients.
Regarding the commenter's concern regarding published clinical evidence to link hospital measurement of social determinants of health to improvements in quality of care or outcomes, as we discussed in section XIV.B of the CY 2025 OPPS/ASC proposed rule, there is substantial research showing differences in care and experiences among these populations (89 FR 59437 and 59438). We also refer readers to HHS' Healthy People 2030 Framework's website for social determinants of health, which includes literature summaries of the latest research related to social determinants of health and how it affects health outcomes and health disparities: https://health.gov/healthypeople/priority-areas/social-determinants-health/literature-summaries. Although the HCHE and FCHE measures are not clinical measures, they aim to improve hospital and facility awareness of the tie between its structural practices and patient outcomes, which will lead to improved clinical outcomes for patients. Advancing health equity helps foster a healthcare system that benefits all and ( printed page 94376) the delivery of more equitable care will, in turn, improve patient outcomes.[303]
We appreciate the commenter's recommendation to provide more timely and actionable data to hospitals to yield more prompt advancements in safety and quality and will consider this input throughout these measures' implementation.
Comment: A few commenters that did not support adoption of the HCHE and FCHE measures expressed concern about the “one size fits all” approach to implementing these measures and stated that the measures are not appropriately tailored to the hospital outpatient, REH, or ASC settings.
Response: We acknowledge that this measure was initially developed for the general acute care setting. The inclusion of the HCHE and FCHE measures in other care settings, including inpatient hospitals, PPS-Exempt cancer hospitals, and dialysis facilities, demonstrate broad applicability of the measure concept and specifications for different healthcare facility types, including in HOPDs, REHs, and ASCs. Hospitals and facilities of every type could benefit from these commitment to health equity measures, as they serve to incentivize the collection and utilization of data across the five domains to identify critical equity gaps, implement plans to address these gaps, and ensure that resources are dedicated toward addressing health equity initiatives, therefore resulting in actionable initiatives and change. These measures aim to support hospitals and facilities in leveraging available data, pursuing focused quality improvement activities, and promoting efficient and effective use of resources. We also reiterate the modifications to Domains 2 and 5 of the FCHE measure (compared to the HCHE measure), which were constructed to address nuances within non-hospital settings. Strong and consistent facility leadership can be instrumental in establishing specific, measurable, and attainable goals to advance equity priorities and improve care for all patients in any care setting, including patients who receive care in HOPDs, REHs, or ASCs. Leaders of health services organizations across the healthcare system are likely to encounter the same challenges and use the same types of strategies to achieve organizational goals related to improving health equity within their respective organizations and patient populations. We note that health equity is a critical focus area for all hospitals and facilities, including those that treat a disproportionate share of patients with health disparities. We will monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if improvements are identified through this process.
Comment: A few commenters that did not support adoption of the HCHE and FCHE measures stated concerns that collecting and publicly reporting the attestation domains for assessing organizational-level leadership involvement in healthcare equity could be misleading to the public and may further underscore disparity without the necessary context. This commenter stated that, without a holistic view of organizational culture, internal policies, specialties, and community demographics, it may not be possible to accurately and fairly evaluate organizations under these measures.
A few commenters further recommended the public display of attestations for each element in a domain as well as additional language stating what hospitals and facilities are doing to achieve health equity. A few commenters recommended that CMS allow for “partial credit,” if hospitals and facilities meet the requirements of only a few elements in a specific domain.
Response: We acknowledge commenters' concern about public reporting of this measure and interpretation by the public. Public reporting of these measures will provide insightful information to healthcare providers and the public on the number of hospitals and facilities currently participating in health equity strategic planning, collecting data, using these data to identify equity gaps, establishing key performance indicators, and reviewing indicators with hospital and facility senior leaders. We intend to provide educational materials as part of our outreach and public reporting of this measure to ensure understanding and interpretation of publicly reported data.
Regarding commenter request for partial credit, the five domains of this measure are actionable focus areas, and assessment of hospital and facility leadership commitment to them is foundational. As stated in the CY 2025 OPPS/ASC proposed rule, the five domains of this measure were adapted from the CMS Office of Minority Health's Building an Organizational Response to Health Disparities framework, which focuses on data collection, data analysis, culture of equity, and quality improvement, and we encourage its use for data analysis to further understand the factors we have highlighted (89 FR 59439). All elements within each domain are necessary for a hospital or facility to indicate that they are committed to advancing health equity for their patients. Each domain represents a core principle in a hospital or facility's commitment to health equity. Achieving credit for each overarching domain, as this data will be publicly displayed by overall score and by domain, rather than by element, should be an incentive for each hospital or facility to work towards and can be used to track a facility or hospital's progress within a particular focus area from year to year. We recognize that hospitals, REHs, and ASCs may be unable to positively attest to every element and domain in this measure as reporting begins. With time, we anticipate hospitals and facilities will be able to positively attest to more domains as they continue to establish their compliance with each element within each domain. This measure will incentivize providers to collect and utilize data to identify critical equity gaps, implement plans to address said gaps, and ensure that resources are dedicated toward addressing healthcare equity initiatives. We encourage providers to analyze their own data to understand the many factors that may influence health outcomes, including various drivers of health, such as housing stability and food security, to deliver more equitable care in their communities and in turn improve patient outcomes for all patients.
Additionally, we note that hospitals and facilities will receive credit for the reporting of their measure results regardless of their responses to the attestation questions. The Hospital OQR Program and the ASCQR Programs are pay-for-reporting programs, and hospitals and ASCs are not scored based on their performance on measures. The REHQR Program does not include a financial incentive or penalty for REHs.
The following comment and response section is applicable to only the HCHE measure for the Hospital OQR and REHQR Programs.
Comment: Several commenters that did not support adoption of the HCHE measure in the Hospital OQR and REHQR Programs expressed concern that the measure has not been endorsed by a consensus-based entity (CBE) and that it has not been reviewed or validated specifically for the outpatient rural emergency setting.
Response: While we recognize the value of measures undergoing review for potential CBE endorsement, measures of health equity are a priority for CMS, and ( printed page 94377) it is important to implement this measure as soon as possible.
For the Hospital OQR Program, we note that section 1833(t)(17) of the Act does not require that each measure we adopt is CBE-endorsed, but states that the Hospital OQR Program, to the extent feasible and practicable, shall include measures set forth by one or more national consensus building entities. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures on this topic endorsed by a consensus organization, so the inclusion of such a measure is not feasible or practicable. Section 1833(t)(17)(C)(i) also requires measures included in the Hospital OQR Program to reflect consensus among affected parties. As we have stated in previous rulemaking (75 FR 72064), consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment. As stated previously, the HCHE measure was developed based on the consensus of a TEP whose recommendations informed the initial selection, development, and emphasis of the importance of this measure and subsequently the FCHE measure.[304] Additionally, these measures have been adopted through rulemaking with public comment periods in other care settings, including inpatient hospitals, PPS-Exempt cancer hospitals, and dialysis facilities, which demonstrate broad applicability and acceptability of the measure concept and specifications for different healthcare facility types, including in HOPDs, REHs, and ASCs.
For the REHQR Program, we note that under section 1861(kkk)(7)(C)(ii) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures on this topic endorsed by a consensus organization, and therefore, the exception in section 1861(kkk)(7)(C)(ii) of the Act applies.
Comment: Several commenters expressed concerns regarding duplicative reporting of attestations for the HCHE measure in the Hospital OQR Program, noting that this measure has already been adopted into the Hospital IQR Program. These commenters requested the ability to submit one single submission jointly for both programs.
Response: We appreciate commenter input on mechanisms to reduce burden and reporting duplication and will incorporate this input into future rulemaking. We acknowledge that the HCHE measure has been adopted into the Hospital IQR Program, and the importance of prioritizing resources and streamlining hospital-wide system processes to promote administrative efficiency. However, because hospital inpatient departments and hospital outpatient departments treat different patient populations and employ different staff, we are requiring that hospitals participating in both the Hospital IQR and Hospital OQR Program submit their HCHE measure data separately. We will monitor implementation of the HCHE measure and will address any modifications in future rulemaking.
Comment: Another commenter stated that many of the priorities included in this measure are currently addressed by hospitals and health systems and urged CMS to catalogue what hospitals are already doing before establishing new measures or requirements to reduce burden and redundancy. A commenter recommended conducting an environmental scan, listening sessions, focus groups, and a technical expert panel (TEP) to reduce instances of redundant measures.
Response: We would like to clarify to commenters that we review environmental scans, which include a review of current hospital and health system activities, and frequently consult with stakeholders before adopting measures. We refer readers to section XIV.B.1.c of this final rule with comment period for a discussion of PRMR process, which holds the purpose of providing multi-stakeholder input to the Secretary on the selection of quality and efficiency measures under consideration for use in quality reporting programs. We will continue to adhere to these practices and will continue to evaluate current practices to reduce redundancy.
Comment: A commenter cited a concern that it will be difficult to implement a performance improvement project that can provide meaningful change as described in Domain 1, given the patient's limited contact with an outpatient department.
Response: We acknowledge the commenters' concerns. We recognize that hospitals are responsible for ensuring the best outcomes possible for the patients that they serve, even within short patient interactions. Ensuring the best possible outcomes requires treating the whole patient, which includes identifying potential barriers to optimal health, and helping patients identify resources to address those barriers. We encourage hospitals to refer vulnerable patients to appropriate resources.
Comment: A commenter recommended the inclusion of a patient experience measure(s) as a means of ensuring quality of care and equitable care in the REHQR program. Additionally, specifically related to the Hospital OQR Program, a commenter recommended that CMS build upon these equity measures with additional equitable care practices such as the Health Equity Report Card (HERC), a useful tool for hospitals seeking to meet the requirements of the HCHE measure, to provide best practice recommendations and ensure accountability for health systems.
Response: We thank these commenters for their recommendations and will consider this input in future rulemaking.
The following comment and response section is applicable to only the FCHE measure for the ASCQR Program.
Comment: Several commenters that did not support adoption of the FCHE measure expressed concern about the lack of testing of the FCHE measure in the ASC setting, and their belief that this measure will not be feasible to implement in ASCs due to differences between ASCs and inpatient hospitals. These commenters suggested that this measure not be adopted until it has been tested for validity and reliability in the ASC setting. A commenter stated that the technical expert panel (TEP) which informed the selection and development of the measure did not include any ASC representatives. The commenter further stated that the TEP only reviewed the hospital version of the measure. The commenter mentioned that there is a difference in ASCs' operational infrastructure compared to hospitals, stating that ASCs do not have the same data collection and analysis capabilities, nor the same technological tools as hospitals. The commenter further added that ASCs do not have immediate access to subject matter experts in culturally sensitive collection of demographic or social determinant of health information and do not have the same resources to properly outsource patients to local and national resources. Commenters stated their concerns about ASCs' lack of long-term relationships with patients, inability to follow-up on interventions, ( printed page 94378) and limited staff. These commenters added that ASCs do not have the same extensive leadership organization as hospitals, which they believe will make implementation of this measure difficult for ASCs.
Response: We acknowledge that this measure was initially developed for the general acute care setting. We understand commenter concern regarding a lack of input from ASC representatives within the TEP. The inclusion of the FCHE measures in other care settings, including inpatient psychiatric facilities, PPS-Exempt cancer hospitals, and dialysis facilities, demonstrates the broad applicability of the measure concept and specifications for different healthcare facility types, including ASCs. Healthcare facilities of every type can benefit from this measure, as it serves to incentivize facilities to collect and utilize data across the five domains to identify critical equity gaps, implement plans to address these gaps, and ensure that resources are dedicated toward addressing health equity initiatives, therefore resulting in actionable initiatives and change. This measure aims to support healthcare facilities in leveraging available data, pursuing focused quality improvement activities, and promoting efficient and effective use of resources, which applies to all settings of care.
Strong and consistent facility leadership can be instrumental in establishing specific, measurable, and attainable goals to advance equity priorities and improve care for all patients in any care setting, including patients who receive care in ASC facilities. Leaders of health services organizations across the healthcare system, including both ASCs and acute care hospitals, are likely to encounter the same challenges and use the same types of strategies to achieve organizational goals related to improving health equity within their respective organizations and patient populations.
Relating to commenter concerns regarding the difference in ASC resources, the FCHE measure accounts for differences in EHR technology in ASCs as compared to hospital settings, as we are not requiring the use of CEHRT. Additionally, if all ASCs have similar issues with attesting “yes” to specific elements within a domain, that will be reflected in public reporting.
Leadership commitment to health equity in all healthcare facilities, including ASCs, is important to addressing health disparities. We will monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if improvements are identified through this process. We have provided an Attestation Guidance document and a FAQ document, which clearly define what constitutes an affirmative attestation and provide answers to frequently asked questions, for the HCHE measure in Hospital IQR Program.[305 306] We intend to provide specific guidance for the ASCQR Program, consistent with the specifications described in Table 160 and requirements being finalized in this section of the final rule, in the future. Facilities may also refer to the measure specifications for additional attestation guidance.[307]
Comment: A few commenters that did not support adoption of the FCHE measure stated that this measure is not actionable as it is not linked to clinical outcomes and there is not sufficient evidence proving that this measure will impact quality of care. A commenter stated that evidence of health equity gaps in ASCs has not been clearly conveyed as previous analyses performed on ASCQR program's claims-based measures did not show racial or socioeconomic disparities.
Response: We thank commenters for their concerns. ASCs are responsible for ensuring the best outcomes possible for the patients that they serve, including identifying potential barriers to optimal health, and helping patients identify resources to address those barriers. This measure is an important foundational measure for improving health equity among those that have been disadvantaged or underserved by the healthcare system.
As discussed in section XIV.B of CY 2025 OPPS/ASC proposed rule, there is substantial research showing differences in care and experiences among these populations (89 FR 59437 and 59438). Regarding the commenter's concern about the lack of evidence of equity gaps among ASCs, research indicate disparities in access to ASCs and a need for more data collection to ensure patient appropriateness and to be able to track quality outcomes.[308 309] The FCHE measure ensures facilities are collecting and evaluating patient data in light of demographic considerations and challenges, which is the first step in identifying equity gaps at the facility level, justifying its use in the ASC setting.
Adoption of the FCHE measure will incentivize ASCs to collect and utilize data across the five domains to identify critical equity gaps, implement plans to address these gaps, and ensure that resources are dedicated toward addressing health equity initiatives, therefore resulting in actionable initiatives and change. This measure aims to support ASCs in leveraging available data, pursuing focused quality improvement activities, and promoting efficient and effective use of resources. A commitment to health equity by facility leadership can foster organizational competencies aimed at achieving health equity for the facility's patients. Although the FCHE measure is not a clinical measure, the measure aims to improve facility awareness of the tie between its structural practices and its patient outcomes, which could lead to improved clinical outcomes for patients.
Comment: A few commenters that did not support adoption of the FCHE measure raised concern that this measure is not CBE endorsed.
Response: While we recognize the value of measures undergoing review for potential CBE endorsement, measures of health equity are a CMS priority. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures on this topic endorsed by a consensus organization, so the inclusion of such a measure is not feasible or practicable. For the ASCQR Program, under section 1833(i)(7)(B) of the Act, the requirement that measures be endorsed at section 1833(t)(17)(C) of the Act applies to the ASCQR Program except as the Secretary may otherwise provide. We note that section 1833(t)(17) of the Act does not require that each measure we adopt for the program is CBE-endorsed (76 FR 74494). Section 1833(t)(17)(C)(i) states that the ASCQR Program, to the extent feasible and practicable, shall include measures set forth by one or more ( printed page 94379) national consensus building entities. Section 1833(t)(17)(C)(i) also requires measures included in the ASCQR Program to reflect consensus among affected parties. As we have stated in previous rulemaking (76 FR 74494), consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment. The FCHE measure achieved the aim of seeking consensus among affected parties by garnering stakeholder input during measure development. We refer readers to section XIV.B.1.c of this final rule with comment period for a discussion of PRMR process, which holds the purpose of providing multi-stakeholder input to the Secretary on the selection of quality and efficiency measures under consideration for use in quality reporting programs.
Comment: Several commenters that did not support adoption of the FCHE measure raised concern that many ASCs do not have an EHR, and therefore, would not be able to attest ‘yes’ to Domain 2. The commenters stated that EHRs are costly to implement and maintain. A commenter stated that a more realistic estimate of ASCs owning and operating an EHR is around 20 percent. The commenter further stated that ASCs do not have the technology or personnel to develop patient-level data, stratify this data, identify equity gaps, and produce performance dashboards. The commenter stated that most ASCs do not have the technological tools and data analysis skills needed to “identify priority populations who experience health disparities” within the communities they serve.
Response: We acknowledge that some ASCs may face challenges adopting EHR technology. We reiterate that the ASCQR Program is a pay-for-reporting program, not a pay-for-performance program. ASCs that do not have EHR technology can attest that they satisfy the other domains, as applicable, and receive a score of 0-4 out of 5 without any financial penalties. We understand the commenters' concern that the public may misinterpret ASCs' reported results as a lack of commitment to health equity. To reduce the likelihood of misinterpretation, we intend to provide educational materials as part of our outreach and public reporting of this measure to ensure understanding and appropriate interpretation of publicly reported data.
We encourage ASCs to allocate resources towards EHR adoption if they are able. Adoption of EHR technology within ASCs will lead to more positive outcomes, as EHR usage has been shown to have positive impacts on care quality, coordination, efficiency, patient care safety, documentation accuracy, and cost reduction.[310] Although initial EHR adoption has associated costs, it is expected that, over time, ASCs will experience a decrease in financial and operational burden. By purchasing an EHR, an ASC will save costs previously spent on maintaining and managing patient charts.[311] EHRs additionally decrease duplicative testing and interventions. Furthermore, EHRs ensure all required patient information is included in the operative note, leading to accurate reimbursements.[312] We continue to encourage ASCs to consider EHR adoption to streamline documentation of patient health information, including demographic characteristics and social determinants of health, which will support ASCs in collecting and utilizing data to identify critical equity gaps, implementing plans to address these gaps, and ensuring that resources are dedicated toward addressing health equity initiatives.
Comment: A few commenters recommended the development of a new health equity measure or the adaptation of the FCHE measure to be better suited to address the differences between ASCs and hospitals. A commenter advocated for a measure to be developed which assesses whether an ASC's quality program addresses equitable care and outcomes for the surgical services provided at the ASC.
A commenter stated that the measure must be adapted for small facilities to prevent biased scores that favor large facilities with greater resources. The commenter recommended that CMS assess current capabilities of the ASC industry through an environmental scan before additional rulemaking action around this measure. The commenter stated that the results of the scan could inform a restructuring of the measure and allow for a reasonable time frame for implementation. The commenter recommended delaying this measure's implementation by 5 years if an environmental scan is not conducted.
Response: We appreciate commenter recommendations surrounding the development of new equity measures tailored to ASC specific needs. We reiterate our awareness that this measure was initially developed for the general acute care setting. Our environmental assessments indicate that there are currently no other existing measures that address facility commitment to health equity. Therefore, it is important to implement this measure as soon as feasible. We would like to remind commenters that this measure addition serves as a foundation for a suite of future equity-focused measures. We see inclusion of such measures in the ASCQR Program as informing efforts to advance and achieve health equity among ASCs by allowing for the recognition and tracking of disparities for the population they serve. Our approach to developing equity-focused measures is incremental and will evolve over time to capture healthcare equity outcomes, as well as align as closely as possible with the measure's respective healthcare setting.
We understand commenter concern regarding the compatibility of the FCHE measure with small ASCs. We would like to note that the FCHE measure is not meant to compare facilities with one another, but rather for facilities to identify structural gaps within their strategic planning, data collection and analysis, quality improvement, and leadership engagement. We remind facilities that the ASCQR Program is a pay-for-reporting program, not a pay-for-performance program. Upon reporting this attestation-based measure, ASCs that do not meet all domain criteria will not be financially penalized.
We appreciate the commenter recommendation to use environmental scans to inform measure development. We include evidence extracted from environmental scans to inform our measure development and will continue to incorporate this critical step in our quality measure development processes for future measures.
Comment: Commenters raised concerns that the rule does not explain how or if CMS will provide financial or administrative support to implement these measures. A commenter recommended that reimbursements should be provided for time spent implementing this measure. A commenter further recommended that CMS engage with hospitals and facilities to better understand the reporting burden of quality measures before their finalization. A few commenters raised concern that ASCs have a lower reimbursement rate than hospitals, making this measure more difficult to implement.
Response: We have provided an Attestation Guidance document and an FAQ document, which clearly define what constitutes an affirmative attestation and provide answers to frequently asked questions, for the HCHE measure in Hospital IQR ( printed page 94380) Program.[313 314] We intend to provide specific guidance for the ASCQR Program, consistent with the specifications described in Table 160 and requirements being finalized in this section of the final rule, in the future. ASCs may also refer to the measure specifications for additional attestation guidance.[315]
We reiterate that the ASCQR program is statutorily limited to reduce the ASC fee schedule increase factor by 2-percentage points in the event the ASC fails to meet program requirements as established by the Secretary (section 1833(i)(7) of the Act). The ASCQR program statute does not provide authority for CMS to provide financial incentives. Regarding commenters' concern that ASCs have a lower reimbursement rate than hospitals, we reiterate that this is an attestation-based measure of a facility's commitment to addressing health equity. Additionally, medical procedures can cost up to 58 percent more when performed in an HOPD, rather than an ASC, justifying the disparity in Medicare reimbursement rates.[316] We disagree that this attestation measure would be more difficult to implement due to differing reimbursement rates.
Comment: A commenter that did not support adoption of the FCHE measure expressed concern that, when this measure was reviewed during the 2021-2022 Measures Application Partnership (MAP) process for the Hospital IQR Program, the measure received a “do not support” recommendation, as the group believed there was insufficient evidence proving that this measure will influence patient outcomes.
Response: We understand the commenter's concern. We note that during the more recent 2023 MAP process as previously discussed, 78.95 percent of committee voters recommended the FCHE measure for inclusion in the ASCQR Program without conditions.[317] The committee expressed support for a measure that captures commitment to health equity consistently across settings.
Comment: Several commenters that did not support adoption of the FCHE measure expressed concern over reporting burden, as this timeline coincides with OAS CAHPS and THA/TKA PRO-PM measure compliance. A few commenters expressed that there would not be sufficient time to implement processes, technology, and training needed to successfully report this measure.
A commenter recommended that CMS weigh the burden of a finalized measure with the value it provides. A few commenters suggested delaying the implementation of this measure or allowing for voluntary reporting.
Response: We acknowledge commenters' concerns regarding resources, including increased data collection burden; however, achieving health equity is an issue which deserves serious focus and rapid action for improvement. Therefore, we do not find it appropriate to delay the implementation of this measure. The benefits of encouraging ASC commitment to health equity outweigh the burden of attestation under this measure.
We understand commenter concerns related to timing of mandatory reporting of the FCHE measure. We emphasize that this measure was proposed for adoption beginning with the CY 2025 reporting period/CY 2027 payment or program determination, which will allow ASCs more than a year before the submission deadline of May 15, 2026, at which time ASCs would submit attestations regarding the identified domains during the CY 2025 reporting period. ASCs can spend the remainder of CY 2024 and CY 2025 establishing processes and addressing potential barriers to positive attestations for the FCHE measure before December 31, 2025, enabling the possibility to report “yes” on each domain by the end of CY 2025.
We note that hospitals participating in the Hospital IQR Program will have already reported data on the similar Hospital Commitment to Health Equity measure for the FY 2025 payment determination (that is, data submitted in FY 2024 representing the FY 2023 performance period) (87 FR 49191 through 49201) before required reporting of the Facility Commitment to Health Equity measure begins within the ASCQR Program, as reporting for this measure would begin with the CY 2027 payment determination. Given the timing of this similar measure's implementation in the Hospital IQR Program, ASCs will have had the opportunity to learn from the experiences of acute care hospitals, including best practices for minimally burdensome assessment of performance on the required domains.
We will also monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if we identify implementation strategies to reduce burden.
Comment: A few commenters that did not support adoption of the FCHE measure expressed concern about potential unintended consequences that may result from the adoption of this measure, including a reduction in patient safety investments due to funding being instead allocated towards FCHE measure reporting.
Response: We appreciate the commenter's concern and will monitor this measure, as we do all ASCQR measures, for any unintended or adverse outcomes associated with implementation. The ability of medical providers to contextualize a facility's commitment to health equity through the five attestation domains is important and could strengthen referrals to and partnerships with community-based service providers for patients with the most complex needs.
Comment: A commenter recommended that ASCs should be able to rely on the inpatient HCHE measure to meet the ASCQR FCHE reporting requirement in cases where hospital-based ASCs have the same leadership and board members as the inpatient hospital.
Response: We appreciate commenter concerns regarding duplicative reporting. We would like to note that a sizeable portion of ASCs are specialized facilities and may have different health equity goals than hospitals.[318] Therefore, it is important that the FCHE measure is reported independently from the HCHE measure.
( printed page 94381)After consideration of the public comments we received, we are finalizing adoption of the HCHE measure for the Hospital OQR Program and the REHQR Program as proposed. Additionally, we are finalizing the FCHE measure for the ASCQR Program, as proposed.
2. Adoption of the Screening for Social Drivers of Health (SDOH) Measure for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs Beginning with Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting for the CY 2026 Reporting Period/CY 2028 Payment or Program Determination
a. Background
SDOH is an umbrella term that refers to community-level factors that impact health and well-being, while HRSNs are social and economic needs that individuals experience that affect their ability to maintain their health and well-being.[319] Consistent screening of patients for potential HRSNs helps healthcare facilities identify individuals who have historically been underserved by the healthcare system and could support ongoing quality improvement initiatives at the population level by providing data to stratify patient risk and organizational performance to address SDOH.[320 321] While widespread interest exists in addressing SDOH at community, State, and national levels and in supporting HRSNs for patients who experience one or more HRSNs, action is inconsistent, with 92 percent of hospitals screening for one or more of the five HRSNs listed in Table 161 but only 24 percent of hospitals screening for all five of these HRSNs.[322] Additionally, pilot studies screening for HRSNs have been conducted in the HOPD and ASC settings, with clinicians and staff agreeing that HRSN data are important and relevant to collect in these settings to improve patient care and communication as well as to connect patients with social-related services.[323 324] We believe that it is essential for healthcare facilities to screen for patient-level HRSN data to support the improvement of patient outcomes and their identified social needs.
In 2017, the CMS Center for Medicare and Medicaid Innovation (CMMI) launched the Accountable Health Communities (AHC) Model, which tested whether systematically identifying and addressing the HRSNs of Medicare and Medicaid beneficiaries through screening, referral, and community navigation services impacted their health outcomes and related healthcare utilization and costs.[325 326] Evaluation of the AHC Model's standard 10-item AHC Health-Related Social Needs Screening Tool (AHC HRSN Screening Tool) found a reduction in emergency department (ED) visits among Medicaid and Medicare fee-for-service (FFS) beneficiaries.[327]
Under the AHC Model, the following five core domains were selected to screen for HRSNs among Medicare and Medicaid beneficiaries: (1) food insecurity; (2) housing instability; (3) transportation needs; (4) utility difficulties; and (5) interpersonal safety. These domains were chosen based upon literature review and expert consensus utilizing the following criteria: (1) availability of high-quality scientific evidence linking a given HRSN to adverse health outcomes and increased healthcare utilization, including hospitalizations and associated costs; (2) ability for a given HRSN to be screened and identified prior to discharge, be addressed by community-based services, and potentially improve healthcare outcomes, including reduced readmissions; and (3) evidence that a given HRSN is not systematically addressed by healthcare providers.328 In addition to established evidence of their association with health status, risk, and outcomes, these five domains were selected for the AHC Model because they can be assessed across the broadest spectrum of individuals in a variety of settings.329
( printed page 94382)( printed page 94383)
These five evidence-based HRSN domains described in Table 161 informed our development of the Screening for SDOH and Screen Positive Rate for SDOH measures. We used these five HRSN domains to inform the development of the SDOH measure we proposed to adopt in this final rule because the AHC Model's HRSN Screening Tool allows healthcare facilities to quickly screen for patients' core health-related social needs and was designed to work in a variety of clinical settings, making it ideal for implementing across quality reporting programs, including the Hospital OQR, REHQR, and ASCQR Programs, with minimal burden to healthcare facilities.[346]
We recognize that patient interaction with the healthcare system may be limited by setting. For example, a patient receiving care in an HOPD, REH, or ASC may not have recently received care in an acute care hospital paid under IPPS, inpatient psychiatric facility cancer hospital, or dialysis facility, and therefore would not have the opportunity to benefit from being screened for SDOHs despite this measure's prior adoption in other quality programs. By adopting aligned Screening for SDOH measures within the Hospital OQR, REHQR, and ASCQR Programs, we expect to increase the likelihood that these settings will screen patients and provide contextualized care and any necessary relevant referrals to address their patient's needs.
Screening for food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety in HOPDs, REHs, and ASCs can help identify and provide appropriate referrals for patients who may benefit from greater support in one or more of those areas. Adoption of the Screening for SDOH measure in the Hospital OQR, REHQR, and ASCQR Programs would continue to support our priority of identifying risk factors for inadequate healthcare access and adverse health outcomes among patients.
b. Measure Overview
The Screening for SDOH measure is a process measure that assesses the total number of patients, who were 18 years or older on the date of service, screened for social risk factors (specifically, the five HRSNs of food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety) as they receive care from a HOPD, REH, or ASC.
c. Pre-Rulemaking Measure Review
As part of the PRMR process, the Hospital Recommendation Group reviewed and voted on the Screening for SDOH measure during their meeting on January 18 and 19, 2024.[347] The Hospital Recommendation Group “recommended with conditions” the Screening for SDOH measure for all three programs (that is, the Hospital OQR, REHQR, and ASCQR Programs).
The committee recommended a condition specific to the Hospital OQR Program, which was to allow hospitals to report this measure one time each year for both the Hospital IQR Program and Hospital OQR Program if applicable.[348] We note that we considered allowing hospitals to report this measure jointly for the Hospital IQR and Hospital OQR Programs (if applicable); however, as the patient populations represented by the programs are different, as is the measure calculation due to this difference in the denominator, we proposed to require a separate data submission for each program. More importantly, patients and consumers would likely find useful Compare tool information on screening rates separated for inpatient and outpatient departments of the same hospital.
d. CBE Endorsement
Section 1833(t)(17)(C)(i) of the Act provides that the Hospital OQR Program, to the extent feasible and practicable, shall include measures set forth by one or more national consensus building entities (not necessarily the CBE). Under section 1833(i)(7)(B) of the Act, this requirement at section 1833(t)(17)(C) applies to the ASCQR Program except as the Secretary may otherwise provide. For the Hospital OQR Program and ASCQR Program, we note that section 1833(t)(17) of the Act does not require that each measure we adopt for these programs be CBE-endorsed (75 FR 72064 and 72065 for the Hospital OQR Program and 76 FR 74494 for the ASCQR Program).
Section 1833(t)(17)(C)(i) of the Act also requires measures developed for the Hospital OQR Program to reflect consensus among affected parties. Under section 1833(i)(7)(B), this requirement also applies to the ASCQR Program except as the Secretary may otherwise provide. As we have stated in previous rulemaking, consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment (75 FR 72064 and 72065 for the Hospital OQR Program and 76 FR 74494 for the ASCQR Program).
For the REHQR Program, section 1861(kkk)(7)(C)(i) of the Act generally requires that quality measures specified by the Secretary for the REHQR Program be endorsed by a CBE; however, section 1861(kkk)(7)(C)(ii) of the Act provides an exception to the general CBE-endorsement requirement, stating that in the case of a specified area or medical topic determined appropriate by the Secretary for which a measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed CBE endorsed measures and were unable to identify any other CBE-endorsed measures on this topic, and therefore we believe the exception in section 1861(kkk)(7)(C)(ii) of the Act applies for purposes of this measure for the REHQR Program. ( printed page 94384)
At this time, we find no other feasible and practicable measures set forth by a national consensus building entity on the topic of screening for SDOH. While we recognize the value of measures undergoing CBE endorsement review and prefer to use endorsed measures, there are currently no CBE-endorsed measures that address screening for SDOH in the outpatient setting. Given the urgency of achieving health equity, it is important to implement this measure as soon as possible. We note that the five domains for which patients would be screened were chosen based upon literature review and expert consensus, and that these five domains informed development of the Screening for SDOH measure. We will consider submitting the Screening for SDOH measure to the CBE for endorsement in the future.
e. Data Sources
For data collection of the Screening for SDOH measure, we proposed that healthcare facilities would use a self-selected screening tool to collect these data. We proposed to allow healthcare facilities to select their screening tool to reduce burden and in recognition of the fact that some healthcare facilities may already be screening their patients for HRSNs. If a healthcare facility is not already doing so, many screening tools for HRSNs already exist. While we acknowledge the potential benefits of requiring all healthcare facilities to use the same screening instrument or a prescribed set of standards around the number or types of screening questions used, we also recognize the benefits of providing healthcare facilities with flexibility to customize screening and data collection to their patient populations and individual needs.
One example of a screening tool that healthcare facilities could consider using is the AHC HRSN Screening Tool, which providers used in the AHC Model to screen for HRSNs in their Medicare, Medicaid, and dually eligible beneficiary populations.[349] We have tested the AHC HRSN Screening Tool across many care delivery sites in diverse geographic locations and determined that it demonstrates evidence of both reliability and validity.[350] The AHC HRSN Screening Tool can be implemented in a variety of healthcare settings, including HOPDs, REHs, and ASCs. While the AHC Model focused on HRSNs among community-dwelling Medicare and Medicaid beneficiaries, the AHC HRSN Screening Tool can be used to screen patients with any insurance status or type, including commercially insured and uninsured individuals. The AHC HRSN Screening Tool has broad applicability in settings outside of the AHC Model as it screens for a range of five HRSN domains while also being concise, limited to only ten questions. We believe this promotes manageable integration into clinical workflow settings and provides greater accessibility and application to diverse patient populations.[351 352]
For additional screening tools for healthcare facilities to consider using to collect data for this Screening for SDOH measure, we refer readers to evidence-based resources like the Social Interventions Research and Evaluation Network (SIREN) website, for example, which provides comprehensive information about the most widely used HRSN screening tools.[353 354] SIREN contains descriptions of the content and characteristics of various tools, including information about intended populations, completion time, and number of questions.
We also encourage healthcare facilities to consider digital standardized screening tools. We refer readers to the FY 2023 IPPS/LTCH PPS final rule (87 FR 49207 through 49208), where we discuss how the use of certified health information technology (IT), including but not limited to CEHRT,[355] can support capture of HRSN information in a standardized, interoperable fashion. We also encourage healthcare facilities to learn about the United States Core Data for Interoperability (USCDI) standard used in certified health IT and how this standard can support interoperable exchange of health and HRSN assessment data.[356]
In alignment with the Hospital IQR Program, we proposed that HOPDs, REHs, and ASCs could confirm the current status of any previously reported HRSNs in another care setting and inquire about others not previously reported, in lieu of re-screening a patient within the reporting period. In addition, if this information has been captured in the EHR in another outpatient setting or the inpatient setting during the same reporting period, we proposed that the HOPD, REH, and ASC could use that information for purposes of reporting the measure in lieu of screening the patient. We intend to monitor and evaluate the measure screening requirements, including frequency, in these outpatient settings to ensure balance between quality of care for patients and facility burden.
f. Measure Calculation
The Screening for SDOH measure is calculated as a percentage equal to the numerator over the denominator. The numerator is defined as the number of patients admitted to an HOPD, REH, or ASC, who are 18 years or older on the date of admission and are screened for all five HRSNs described in Table 161 during their receipt of services in the HOPD, REH, or ASC, as applicable.[357] The denominator is defined as the number of patients who are admitted to a HOPD, REH, or ASC, as applicable, and who are 18 years or older.
The measure excludes patients who: (1) opt-out of screening; or (2) are themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf.
g. Data Submission and Reporting
In the CY 2025 OPPS/ASC proposed rule, we proposed to allow healthcare facilities to voluntarily submit to CMS aggregate data for this measure for the CY 2025 reporting period and then to require mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determination. Specifically, we ( printed page 94385) proposed that healthcare facilities would aggregate data they collect for the numerator and the denominator to CMS (as described in section XIV.B.2.f of this final rule with comment period), and that they would not be required to submit patient-level data. We proposed to require aggregate data because we believe patient-level reporting is unnecessary and would cause undue burden due to the transfer of large quantities of data. However, in the future, we may consider requiring the reporting of patient-level information. This measure aims to encourage healthcare facilities to screen for and identify HRSNs in order to identify and address social needs among their patient populations.
We also proposed that healthcare facilities would be required to submit data on this measure annually using the CMS-designated information system (currently, the HQR system) consistent with the data submission requirements for this measure in the Hospital IQR, IPFQR and PCHQR Programs. We refer readers to sections XV.E.2.a, XVI.E.3.b, and XVII.E.2.a of this final rule with comment period for additional details regarding data submission using the CMS-designated information system in the Hospital OQR, REHQR, and ASC Programs, respectively.
We proposed to adopt this measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determination. We proposed to begin with 1 year of voluntary reporting to provide a transition period for healthcare facilities to select and integrate screening tools into their clinical workflow processes.
We invited public comment on our proposal to adopt the Screening for SDOH measure for the Hospital OQR, REHQR, and ASCQR Programs beginning with voluntary reporting for the CY 2025 reporting period, and to require mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determination, as described above.
The following comments and responses are applicable for the Hospital OQR, REHQR, and ASCQR Programs unless noted otherwise.
Comment: Many commenters supported the Screening for SDOH measure as well as CMS's efforts to align measures across programs and increase screening for SDOH in outpatient, rural, and ambulatory settings. Several commenters expressed support for adopting the Screening for SDOH measure in the outpatient programs because they believe the measure helps better identify patients with food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety concerns, and they appreciate CMS's commitment to improving health outcomes for all patients with higher health-related needs. A few commenters expressed support for screening for food insecurity specifically, because it can help provide appropriate referrals for patients who may benefit from additional nutrition, identify malnutrition risk across outpatient settings, facilitate access to appropriate and timely nutrition interventions, and connect patients to community-based nutrition programs and services that address ongoing needs. A commenter supported the adoption of the Screening for SDOH measure across the Hospital OQR, ASCQR, and REHQR programs because the measure provides structured support that healthcare providers need to better support patients who have SDOH needs and help providers acknowledge how SDOH impact patient outcomes. Another commenter expressed appreciation for CMS's indication that the patient screening in the outpatient setting can leverage information that the patient has already provided in a prior encounter in the performance year.
Response: We thank commenters for their support of the Screening for SDOH measure. We agree with commenters that availability of SDOH data will potentially identify innovative opportunities to support enhanced availability of community resources to meet the needs identified by the Screening for SDOH measure. We agree with the commenters' statement that this measure could support efforts to connect patients in need with community resources.
Comment: Many commenters supported the adoption of the Screening for SDOH measure but offered specific recommendations, including that CMS provide a mechanism to confirm that referrals to appropriate services resulted in actual service delivery since the actual services, not the referrals, are the goal. These commenters recommended that CMS continue to take steps towards measuring prompt action to connect patients to a community-based organization, interventions to address the HRSN, the resolution of the need, and efforts to ensure that providers are able and incentivized to connect patients with necessary clinical, mental, behavioral, and social services as seamlessly as possible.
Response: We thank the commenters for their broad support and recommendations. We will take into consideration as appropriate the recommendations to ensure that referrals of patients to various support services are addressing needs and resulting in actual services.
Comment: Many commenters supported the adoption of the Screening for SDOH measure in the Hospital OQR Program because it affirms that any patient entering the care of the hospital undergoes an HRSN screen. Several commenters expressed support for the Screening for SDOH measure specifically in the Hospital OQR program noting that robust data collection on patient demographics and SDOH enables more accurate analysis of health equity and improves the quality of care for all beneficiaries in the Medicare program, and that the addition of the health equity measures HOPDs will allow for more consistency between the outpatient and inpatient space and will aid in identifying and assisting patients who are struggling with one of the five domains. A commenter supported the measure because it will allow for 1 year of voluntary reporting to support implementation in the outpatient setting and appreciated that HOPD staff could confirm the current status of any previously reported HRSNs in another care setting and inquire about others not previously reported. The commenter also supported that CMS would allow hospitals to use SDOH screening information that is recorded in the EHR in another health setting during the same reporting period to report data on the measures. A commenter supported that CMS emphasized a limited number of universal health-related domains and strongly supported CMS allowing health systems flexibility to use their preferred screening tool. Another commenter supported the measure because collecting SDOH data allows HOPDs to refer patients to care that can address their SDOH and connect patients to community-based resources based on their needs. Additionally, a commenter supported CMS's commitment to advancing health equity and improving health outcomes because occupational therapy (OT) practitioners can address SDOH at the person, group, and population level by assessing and addressing both the social needs of the individual clients and the structural challenges that impact the ability to meet these social needs. This commenter further stated that addressing SDOH within OT interventions can improve health outcomes, increase health equity, expand occupational engagement, and contribute to wellness. A commenter appreciated the decision to allow ( printed page 94386) inpatient screenings in the same reporting period to also be considered for measuring screening rates in the outpatient setting but requested clarification on whether non-office, procedural visits such as imaging or lab services in the outpatient department would be considered “admissions” to an HOPD subject to screening in the measure calculation. A commenter recommended that outpatient lab tests and X-Rays be included in the proposed outpatient requirements, and that clarification be provided on qualifying outpatient hospital encounters and on the types of HOPD environments SDOH screening requirements will cover.
Response: We thank the commenters for their support of the Screening for SDOH measure in the Hospital OQR Program. We agree that HRSNs are critical factors that impact patient outcomes, and increased knowledge about patients' HRSNs will help hospitals shape goals associated with health equity. We agree with commenters that availability of SDOH quality data will support the identification of innovative opportunities to enhance availability of community resources to meet the needs identified by the Screening for SDOH measure. Further, we share the commenters' belief that this measure could support efforts to connect patients in need with community resources. We thank commenters for their questions regarding the technical specifications of the measure. We agree that screening for SDOH is most appropriate when administered so that the patient's information can be used by the patient's care team. As we stated in the proposed rule, screening for food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety can help identify and provide appropriate referrals for patients who may benefit from greater support in one or more of those areas. As such, we are clarifying that while outpatient care can refer to numerous types of health services, such as emergency department (ED) services, observation services, outpatient surgical services, lab tests, and X-rays, for purposes of this measure in the Hospital OQR Program, REHQR, and ASCQR programs, patients receiving services that are limited to specific medical tests are not included in the denominator cohort. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
Comment: Several commenters supported the adoption of the Screening for SDOH measure and offered recommendations about the data in the Screening for SDOH measure, including that CMS should publish the standards on how SDOH data will be protected, and that CMS should require disaggregated collection of the measures at the individual level to allow providers to assess the factors impacting an individual patient's health to better meet their needs. These commenters also supported the expansion of the SDOH reporting and the changes to the transportation element, but recommended CMS focus on ensuring the data are collected rather than on the specific vendor or tool used for the collection.
Response: We thank the commenters for their support and recommendations. We have given this significant consideration to ensuring data is protected in development of the SDOH measures. The combination of existing privacy laws and the measure specifications (requiring submission of aggregated data) address the concerns relative to patient rights and transparency. HOPDs, REHs, and ASCs generally must comply with applicable laws governing the confidentiality, privacy, and security of patient information. We have also specified the measure to allow patients to opt-out of screening. We thank the commenter for their recommendation to ensure that data are collected rather than focusing on a vendor or tool. We note that we do not require HOPDs, REHs or ASCs to report this measure using a specific vendor or tool. As previously discussed, outpatient settings are provided the flexibility to choose their own screening tool to eliminate redundancy, utilize interoperable systems, and reduce burden. We have provided suggestions for validated screening tools, such as the AHC HRSN Screening Tool, that HOPDs, REHs and ASCs may use if they choose, but we do not require outpatient settings to use these tools to maintain this flexibility.
Comment: A few commenters supported the Screening for SDOH measure and recommended that CMS continue engaging with stakeholders to ensure that the measure remain relevant. A few commenters supported the Screening for SDOH measure but recommended additional efforts, including that CMS consider opportunities to implement higher level holistic SDOH screening approaches and accompany the SDOH screening with responsive care models that provide effective interventions to address patients' unmet needs, that CMS consider adopting the Social Need Screening and Intervention measure in its quality reporting programs as an alternative, and that CMS make the question related to interpersonal violence (IPV) optional.
A commenter agreed with HOPDs having the flexibility to choose their own screening tool to measure HRSNs and recommended that we incorporate this flexibility across existing and future quality measures to eliminate redundancy, utilize interoperable systems, and reduce burden. A commenter appreciated that CMS allows HOPDs to utilize previous screens to avoid the burden of re-screening but recommended that CMS work with measure developers to develop a measure that focuses on the actions that were taken following a positive screen and evaluate the impact of SDOH on health outcomes to adjust the domains for social drivers. The commenter also recommended that CMS share the roadmap for moving the healthcare system beyond screening towards effectively closing gaps for social needs of patients and a plan to evaluate the link between identifying a social need and utilization of services to address the patients' needs. They also recommended expanding the measure and metric to include additional SDOH such as access to education, employment opportunities, digital connectivity, and environmental factors which can have significant impacts on health outcomes.
A commenter recommended finalizing the Screening for SDOH measure with flexibility to allow HOPDs to use different screening tools and increasing support for safety net hospitals' ability to respond to SDOH needs, and another commenter recommended enhanced transparency because consumers should be able to identify the screening rate a facility attains for each domain and the reporting of more granular level results for each measure will contribute to driving improvement where the largest deficits are identified. A commenter recommended that CMS require disaggregated collection of the measures at the individual level to allow providers to assess the factors impacting an individual patient's health to better meet their needs. A commenter supported the adoption of the Screening for SDOH measure as an important first step but recommended that CMS increase funding to ensure that patients who screen positive for SDOH are able to receive resources including things like transportation, housing, and food security. The commenter also recommended that CMS align with ( printed page 94387) other organizations and national initiatives that are establishing coding and documentation standards in this space, simplifying the process to remove ambiguity, and supporting easy integration into providers' current workflow. Another commenter recommended that efforts to address SDOH ensure that providers are able and incentivized to connect patients with necessary clinical, mental, behavioral, and social services as seamlessly as possible.
Response: We thank the commenters for the recommendations to engage in further efforts to implement holistic screening approaches, alternative measures, and expansion of additional social drivers of health and we will consider these recommendations in future rulemaking. However, in response to the recommendation to make the interpersonal safety domain optional, we note that we have prioritized selection of the proposed five HRSN domains based on existing evidence from both the AHC Model that informed the initial selection, and emerging evidence of correlations between given social drivers of health and worse health outcomes and SDOH for which interventions have shown marked improvements in health outcomes and healthcare utilization.
It is imperative that HOPDs, REHs, and ASCs screen for all five domains established in this measure. We understand the sensitive nature of screening for risk of interpersonal violence and that patient safety must remain the hospital's, REH's, and ASC's principal concern. We recommend that hospitals, REHs, and ASCs ensure that patients feel that they are safe answering questions and remind patients that they may opt out of the screening for any reason. We note that the measure specifications exclude patients: (1) who opt-out of screening; and (2) are themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf (89 FR 59447). We also note that while individual responses to the SDOH survey may constitute protected health information (PHI) under the HIPAA Privacy Rule (45 CFR parts 160 and 164, subparts A and E), the information HOPDs, REHs, and ASCs must provide CMS to meet the requirements of the SDOH measure consist of aggregated, de-identified data only. Information that does not identify the individual that is the subject of the health information and for which there is no reasonable basis to believe it could be used to identify an individual, is not individually identifiable health information (IIHI) and is therefore not PHI (45 CFR 160.103). HIPAA covered entities must comply with requirements to use and disclose PHI only as permitted or required by the HIPAA Privacy Rule, but the HIPAA Privacy Rule does not place restrictions on the use or disclosure of information that is not PHI (45 CFR 164.502). While HOPDs, REHs, and ASCs that begin to collect SDOH information will be creating additional PHI that they did not previously create, the SDOH quality measure will not require them to disclose any PHI or IIHI to CMS. We remind HOPDs, REHs, and ASCs that as covered entities, they must ensure the confidentiality, integrity, and availability of all electronic PHI, including any electronic PHI generated in the course of collecting responses to any SDOH survey (45 CFR 164.306).
In response to the recommendation to align with other organizations and national initiatives, this data collection will inform meaningful and sustainable solutions for other provider types through similar collections in other quality reporting programs and note that we will continue identifying opportunities for collaboration with other stakeholders to align SDOH assessment across CMS programs. We commend additional stakeholder efforts currently underway to expand capabilities to capture additional drivers of health data elements. We will continue building the overarching strategy for integrating social drivers of health screening into quality improvement and future rulemaking. We also note that we will continue identifying opportunities for collaboration with other stakeholders to ensure the measure remains relevant in future years.
As to commenters' request for additional information on CMS's work on health equity, we refer commenters to the CMS Framework for Health Equity 2022-2032 available at: https://www.cms.gov/priorities/health-equity/minority-health/equity-programs/framework.
Comment: A few commenters expressed their support for the Screening for SDOH measure but requested clarifications including that CMS clarify any restrictions on what tools, modified or not, a hospital or facility can use to screen patients for the HRSNs. A commenter also requested clarification on the outpatient population in the denominator of this measure for use in the Hospital OQR program because not all outpatient interactions involve an office visit where screening would be appropriate. A commenter requested clarification on qualifying outpatient hospital encounters and further defining of the types of HOPD environments SDOH screening requirements will cover.
Response: We appreciate the commenters' support and requests for clarification. We note that we are not mandating use of a specific screening tool, and we therefore defer to providers to make the appropriate determination regarding how to collect the data. We also note that, while outpatient care can refer to numerous types of health services, such as emergency department (ED) services, observation services, outpatient surgical services, lab tests, and X-rays, for purposes of this measure in the Hospital OQR, REHQR, and ASCQR Programs, patients receiving services that are limited to specific medical tests are not included in the denominator cohort. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
Comment: A few commenters fundamentally supported the SDOH screening measure but expressed concerns that the data required for the measure may not currently be reliably documented using discrete data fields that can be captured electronically. A commenter recommended adding economic insecurity as a social risk factor for screening as it can lead to many other social determinants of health.
Response: We thank the commenters for their broad support of the Screening for SDOH measure. We appreciate the commenter's concerns about the limits of the discrete data fields. The data collected by the measure captures critical information about SDOH, and we note that this measure does not restrict hospitals or facilities from documenting more qualitative data about SDOH. Hospitals and facilities are not restricted to the data fields provided if they wish to gather more qualitative data on their patients. We appreciate the recommendation to include economic insecurity and will consider adding it to the measure in future rulemaking.
Comment: A few commenters recommended that CMS work with stakeholders on the best ways to gather more granular data while maintaining strong privacy and patient safeguards so researchers and other stakeholders can evaluate how hospitals are evaluating HRSNs as well as whether hospitals and large hospital systems are adequately evaluating HRSNs because it is important to understand how HRSNs contribute to healthcare spending, quality, and outcomes at the individual ( printed page 94388) and community levels. A commenter supported the use of Z-codes to inform social risk adjustment and recommended that CMS continue to use Z-codes as a proxy for social risk and to maintain a broad viewpoint on how social risk is identified. The commenter also recommended that CMS avoid leveraging SDOH screening data as if it were epidemiologic data rather than data intended to understand patients' needs and optimize their health and well-being. Another commenter recommended that CMS include the Distress Thermometer as a useful tool for providers seeking free resources to meet these measures. Another commenter noted that the measure is a welcome addition to the measure set but recommended that CMS develop a stronger audit function because without an adequate auditing function, the measures remain highly susceptible to subjectivity.
The commenter also recommended keeping the Hospital IQR Program reporting separate from the Hospital OQR Program reporting because it will ensure a more accurate picture allowing for improved consistency in data collection over time. Another commenter recommended that CMS continue to make important changes to the Hospital OQR program by strengthening requirements for hospitals to report on a more comprehensive set of quality and equity measures. A commenter supported the inclusion of health equity and also recommended consolidating the reporting of HRSNs at the organizational level rather than separately for inpatient and outpatients to align human and technological resources. Another commenter supported CMS's work to identify and address social drivers of health in all patient care settings but recommended additional guidance from CMS and additional time to operationalize the screenings during outpatient visits.
Response: We thank the commenters for the recommendation to engage with stakeholders and we will continue identifying opportunities for collaboration with stakeholders to ensure the measure is gathering granular data and adequately evaluating HRSNs. We agree with commenters that the intention of the measure is to understand patients' needs, rather than to provide epidemiologic information. We will also consider including references to the Distress Thermometer in our resources for this measure. Additionally, in response to the recommendation to keep the Hospital IQR Program reporting and the Hospital OQR Program reporting separate, we note that, as previously stated in the CY 2025 OPPS/ASC proposed rule, the populations represented by the Hospital IQR and Hospital OQR programs are distinct, resulting in a different denominator calculation of the measure for each program (89 FR 59446). We will continue to require a separate data submission for each program in the HQR system.
We appreciate the recommendations to consolidate the reporting of HRSNs at the organizational level, and to strengthen the requirements for hospitals to report on a more comprehensive set of equity measures. Additionally, regarding the recommendation to evaluate the initial metrics from the measure, we note that we will monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if improvements are identified through this process. Regarding the use of Z codes, we appreciate commenters' recommendations; however, adoption of Z codes across healthcare systems has been slow and unstandardized, and the Screening for SDOH measure will be beneficial to clinicians in addressing patient social needs while provider uptake of Z codes improves.
Comment: A few commenters supported the inclusion of the Screening for SDOH measure in the REHQR Program noting that they support the broad effort to evaluate and promote health equity and SDOH within the REH model and commended CMS for focusing on measures related to health equity for REHs because they play a vital role in ensuring healthcare access in rural and remote communities. They stated that inclusion of the measure will help ensure a focus on health-related social needs and address health equity across facilities. A commenter recommended that CMS prioritize inclusion of a patient experience measure in future rulemaking because they believe such measurement is currently missing from the REHQR program and is an important reflection of providing culturally relevant care.
Response: We thank the commenters for their support of the inclusion of the Screening for SDOH measure in the REHQR Program, and we agree that HRSNs are critical factors that impact patient outcomes, and increased knowledge about patients' HRSNs will help REHs shape goals associated with health equity. Further, we agree that collecting these data will help REHs improve coordination with outpatient and community resources to better deliver patient-centered care. We thank the commenter for the recommendation and will consider inclusion of a patient experience measure in the REHQR Program in future rulemaking.
Comment: A few commenters expressed specific support for the adoption of the Screening for SDOH measure in the ASCQR Program because it aligns with the measure as adopted in inpatient settings, reflects important priorities in safety and health equity, and will ensure that quality reporting is consistent across different healthcare settings. A commenter expressed support for CMS's efforts to improve health equity with the Screening of SDOH measure in the Hospital OQR and ASCQR Programs but recommended that CMS begin with voluntary reporting to allow HOPDs and ASCs time to implement the screening tools and workflows needed. The commenter also requested guidance on the cadence of required screenings such as if a patient is screened every visit or once every 6 months, or if a patient receives a HRSN screening in the emergency department and is then admitted, if they would need an additional screening to satisfy Hospital IQR Program reporting.
Response: We appreciate the commenters' support and recommendation. We agree that the measure ensures alignment across quality programs and will help shape our goals associated with health equity. We thank the commenter for their recommendation to implement this measure with a voluntary reporting period. We remind readers that we proposed this measure with an initial voluntary reporting period beginning with the CY 2025 reporting period and proposed to require mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determination.
Regarding the commenter's request to clarify the cadence of screening we note that outpatient settings would not be required to re-screen patients in the same reporting period if the data are already captured in the EHR. HOPDs, REHs and ASCs could confirm the current status of any previously reported HRSNs in another care setting and inquire about others not previously reported in lieu of re-screening a patient for all HRSN domains within the reporting period.
Additionally, as previously discussed in the CY 2025 OPPS/ASC proposed rule, the populations represented by the Hospital IQR and other outpatient quality reporting programs are distinct, resulting in a different denominator calculation of the measure for each program (89 FR 59446). Therefore, inpatient and outpatient settings will be unable to accurately report this measure ( printed page 94389) if data are combined for inpatient and outpatient programs. We will therefore continue to require a separate data submission for each program in the HQR system.
In addition, as we stated in the proposed rule, screening for food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety can help identify and provide appropriate referrals for patients who may benefit from greater support in one or more of those areas. As such, we are clarifying that while outpatient care can refer to numerous types of health services, such as emergency department (ED) services, observation services, outpatient surgical services, lab tests, and X-rays, for purposes of this measure in the Hospital OQR, REHQR, and ASCQR Programs, patients receiving services that are limited to specific medical tests are not included in the denominator cohort. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
The following comments and responses are broadly applicable to the Hospital OQR, REHQR, and ASCQR Programs, unless noted otherwise.
Comment: Many commenters expressed concerns about the administrative burden and insufficient reimbursement rate associated with the work required to collect data and report this measure for outpatient settings that are already under-resourced. These commenters recommended that CMS consider the administrative burden of the measure to ensure that new reporting processes do not place undue strain on entities. A few commenters recommended working with stakeholders to develop an electronic clinical quality measure to reduce burden. A few commenters recommended increased reimbursement due to the reporting burden of this measure. A commenter recommended removing other quality reporting requirements if there are no additional financial incentives.
Response: We acknowledge commenters' concerns regarding resource constraints, including increased administrative burden; however, achieving health equity is an issue that deserves serious focus and rapid action for improvement. The benefits of screening patients for SDOH outweigh the minimal burden under this measure. As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59516 and 59517, 89 FR 59522 and 59523, and 89 FR 59526, respectively) and in sections XXVI.A.c, XXVI.B.c, and XXVI.C.c of this final rule with comment period, we estimated the collection burden for this measure to be 2 minutes per patient screening and 10 minutes annually per HOPD, REH, and ASC to report the measure data. We note that there are ongoing reevaluation efforts that aim to improve the Screening for SDOH measure through the development of the Addressing Social Needs (ASN) eCQM for introduction to the Hospital IQR Program and eventually the Merit-based Incentive Payment System (MIPS) and Hospital OQR Programs.[358] As we move toward interoperability, we will consider this recommendation in future rulemaking. We will continue to identify opportunities for collaboration with stakeholders to align screening for SDOH assessment across CMS programs.
We seek to reduce reporting burden in all our quality reporting programs and value-based purchasing programs. As an example of our commitment to reducing burden in the ASCQR Program, we refer readers to our RFI on the Specialty-Select and the Specialty-Threshold frameworks as detailed in section XVII.G of this final rule with comment period, in an effort to decrease reporting burden by ensuring that ASCs are reporting on measures most meaningful to their facilities.
Regarding commenters' recommendations for increased reimbursement, we reiterate that the Hospital OQR and ASCQR Programs are statutorily limited to reduce the fee schedule increase factor by 2-percentage points in the event the HOPD or ASC fails to meet program requirements as established by the Secretary (section 1833(i)(7) of the Act). The Hospital OQR, REHQR and ASCQR Program statutes do not provide authority for CMS to provide financial incentives. For the REHQR Program, there is no financial penalty or incentive for quality reporting (section 1861(kkk)(7)); however, REHs will be paid for furnishing covered REH services at a rate that includes an additional five percent increase over the OPPS payment rate.
To support implementation of the Screening for SDOH measure within HOPDs, REHs and ASC, we refer readers to the Hospital IQR Program's FAQ document regarding this measure in the Hospital IQR Program, available at: https://www.qualityreportingcenter.com/globalassets/2024/04/iqr/17.-sdoh-measure--faqs_april-2024_vfinal508.pdf.
We will develop a similar Frequently Asked Questions document for the Hospital OQR, REHQR, and ASCQR Programs as part of providing educational and outreach materials; this document will be conveyed through routine communication channels.
Comment: Many commenters expressed concern about this Screening for SDOH measure under the ASCQR Program, stating that ASCs have insufficient resources, are likely understaffed, and have a lower reimbursement rate than hospitals, making this measure more difficult to implement given the increased reporting and operational burden required. A commenter stated that this measure expands the role of ASCs beyond their scope, without additional funding or resources. A commenter stated that “we recently finalized a new standalone HCPCS G code describing a SDOH Risk Assessment” and stated that providers can be paid for performing this service. This commenter further stated that this measure requires the “same resource use in ASCs” and recommended that CMS allow ASCs to receive reimbursement for the SDOH Risk Assessment. A commenter recommended implementing measures that only provide the greatest value to patient safety and quality of care in ASCs, as additional mandatory measures may divert revenue away from other patient safety investments.
A few commenters expressed concern about the burden associated with aggregating and reporting results of existing screening for SDOH among HOPDs. A commenter expressed concern that the addition of this measure in the Hospital OQR Program may significantly increase burden on Emergency Departments (EDs), which are often resource strained.
Response: We thank commenters for their concern that this measure is out of scope for ASCs; however, we respectfully disagree as ASCs are responsible for ensuring the best outcomes possible for the patients that they serve. Ensuring the best possible outcomes requires treating the whole patient, which includes identifying potential barriers to optimal health, and helping patients identify resources to address those barriers. Under the AHC Model, the five core domains selected to screen HRSNs (food insecurity, housing instability, transportation needs, utility difficulties and interpersonal safety) were chosen based upon literature ( printed page 94390) review and expert consensus utilizing the availability of scientific evidence linking a given HRSN to adverse health outcomes and increased healthcare utilization, including hospitalizations and associated costs (89 FR 59444). ASCs that have successfully connected patients to community resources may show a decrease in readmission rates and in cost savings associated with healthcare delivery over time.[359 360] This may be beneficial for the health of the communities ASCs serve. We encourage ASCs to refer vulnerable patients to appropriate resources wherever possible or feasible.[361]
Additionally, we appreciate the commenter's concern that this measure may divert revenue away from other patient safety investments among facilities and note the minimal burden of collecting data (estimated at 2 minutes per patient screening and 10 minutes annually per HOPD to report the measure data). We will monitor this measure, as we do all ASCQR measures, for any unintended or adverse outcomes associated with potential future implementation. The ability for medical providers to screen for social drivers of health through the five HRSN domains is important and will help patients with the most complex needs.
We acknowledge commenters' concerns that this measure may place increased burden on HOPDs and reiterate that the benefits of screening patients for SDOH outweigh the minimal burden of collecting data (estimated at 2 minutes per patient screening and 10 minutes annually per HOPD to report the measure data) under this measure as achieving health equity requires swift action for improvement. We also note that hospitals participating in the Hospital IQR Program that choose to voluntarily report this measure will have experience screening patients for SDOH under the Hospital IQR Program, and HOPDs would not be required to re-screen the same patients for the Hospital OQR Program if the data are already captured in the EHR.
Comment: Many commenters that did not support the Screening for SDOH measure expressed concern about the additional burden that would be created by implementing EHR systems to collect SDOH data. These commenters recommended additional time for measure implementation.
Several commenters expressed concern that implementation of this measure under the ASCQR Program lacks feasibility and creates additional burden as a large portion of ASCs have not integrated EHRs into current workflow processes and would have limited resources to finance this technology due to the investments required for this quality reporting measure. A commenter recommended conducting additional research on how this measure can be applied to the ASC setting, especially for ASCs that are under-resourced and unable to implement EHR systems.
Response: We thank the commenters for their feedback and note that the Screening for SDOH measure does not require EHRs for healthcare facilities that have not yet acquired one. We disagree that this concern is applicable to the adoption of the Screening for SDOH measure in the Hospital OQR and REHQR Programs. We note that, as of 2021, 96 percent of hospitals have already integrated EHRs into their workflow processes.[362] Further, hospitals that participate in the Hospital OQR Program are required to utilize health information technology modules certified to the 2015 Edition Cures Update, which was a specific edition of ONC health IT certification criteria, beginning with the CY 2023 reporting period/CY 2025 payment determination to report eCQMs (86 FR 63868 through 63869).[363] We note that this rule with comment period finalizes a requirement at § 419.46(j)(1) that hospitals participating in the Hospital OQR Program “utilize certified technology updated to be consistent with the Office of the National Coordinator for Health Information Technology's health IT certification criteria, as adopted and updated in 45 CFR 170.315,” which is consistent with ONC's current “edition-less” approach to the ONC health IT certification criteria (89 FR 1207). We also note that, for clarity, this finalized requirement differs from our proposal by the removal of an acronym and its replacement by the full agency name. Additionally, many REHs reported under the Medicare Promoting Interoperability and Hospital IQR Programs prior to conversion, and thus are likely to also have EHR systems already embedded in their workflows.
Adoption of EHR technology will lead to more positive outcomes in ASC settings, as EHR usage has been shown to have positive impacts on care quality, coordination, efficiency, patient care safety, documentation accuracy, and cost reduction.[364] Although initial EHR adoption has associated costs, we expect that, over time, ASCs will experience less financial and operational burden. By purchasing an EHR, an ASC will save costs previously spent on maintaining and managing patient charts.[365] EHRs additionally decrease duplicative testing and interventions and ensure all required patient information is included in the operative note, leading to accurate reimbursements.[366] We continue to encourage ASCs to consider EHR adoption to streamline documentation of patient health information, including demographic characteristics and SDOH, which will incentivize ASCs to collect and utilize data to identify critical equity gaps, implement plans to address these gaps, and ensure that resources are dedicated toward addressing health equity initiatives.
Comment: Many commenters recommended delaying voluntary implementation of the measure by at least 1 year and extending voluntary reporting to provide outpatient settings enough time to prepare and build the needed infrastructure and community networks or add a phased approach to mandatory reporting that allows flexibility for organizations to not burden their administrative tasks in ways that may hinder day-to-day operations. These commenters, across all programs, stated that the measure requires significant time and financial resources to integrate into workflows.
A commenter recommended delaying implementation in the Hospital OQR Program until a comprehensive measure review has been performed. Another commenter specifically recommended that CMS delay voluntary reporting for the Hospital OQR Program until CY ( printed page 94391) 2027 and begin mandatory reporting in CY 2029.
Response: We thank the commenters for their feedback, and we acknowledge their concerns about the operational complexity of introducing new quality measures into existing clinical workflows and EHR systems. Beginning data collection remains imperative as we continue to build on our strategic pillar to advance health equity by addressing the health disparities that underlie our health system, and the proposed voluntary and mandatory reporting periods prioritize the urgency of capturing SDOH data and taking actionable steps towards closing health equity gaps.
We acknowledge commenters' concerns regarding the financial resources needed to integrate this measure into workflows; however as previously noted, achieving health equity is an issue that deserves serious focus and rapid action for improvement. The benefits of screening patients for SDOH outweigh the minimal burden under this measure (estimated at 2 minutes per patient screening and 10 minutes annually per HOPD, REH, and ASC to report the measure data).
Additionally, hospitals participating in the Hospital IQR Program that choose to voluntarily report this measure will have already reported data in FY 2024 (87 FR 49207 through 49208), and the Hospital IQR Program finalized mandatory reporting of this measure for the FY 2026 payment determination (that is data submitted in FY 2025 representing the FY 2024 performance period) (87 FR 49207 through 49208). Given the timing of reporting this measure in the Hospital IQR Program, outpatient settings will have the opportunity to learn from the experiences of hospitals participating in the Hospital IQR Program, including best practices for collecting HRSN data, prior to mandatory reporting for the Hospital OQR, REHQR, and ASCQR Programs for the CY 2026 Reporting Period/CY 2028 Payment or Program Determination.
Comment: Many commenters that did not support adoption of the Screening for SDOH measure expressed concern that it has not been tested in the HOPD, REH or ASC settings. Many commenters urged CMS to continue testing the measure in these outpatient settings to demonstrate feasibility, reliability, validity, and to address challenges endured in the inpatient setting prior to expanding it as mandatory in the outpatient setting.
Several commenters did not support adoption of the Screening for SDOH measure in the ASCQR Program based on their belief that there is insufficient evidence that this measure leads to improved patient outcomes among ASCs. A commenter expressed concern that the burden of implementation of this measure in the ASCQR Program and the potential for skewed results due to lack of testing outweighs the benefit of screening for SDOH due to the lack of evidence-based research and inaccuracy of non-risk adjusted data. A commenter expressed concern that without testing in the ASC setting, there are many unknowns regarding requirements for implementation such as determining the full-time equivalent (FTE) impact of staffing the screening and referral processes. A commenter recommended first conducting screenings for SDOH in a smaller outpatient population to provide lessons learned.
Response: We acknowledge that this measure was initially developed for the acute care setting. We recognize the value of measures undergoing testing and evaluation of validity and feasibility in the setting for which they are being adopted. As part of the PRMR process, the measure underwent an assessment of the measure's testing data and appropriateness for the outpatient setting, including a review of the measure's scientific acceptability, feasibility, and usability. Additionally, we will continue to test the performance of this measure in the HOPD, REH, and ASC settings throughout the voluntary reporting period. Given the urgency of achieving health equity, and as there are currently no other existing measures that address screening of HRSNs, it is important to implement this measure as soon as feasible. This measure is not specific to any particular setting of care and is equally applicable to the outpatient setting as to general acute care settings. Identifying the HRSNs of HOPD, REH, and ASC patients will be equally valuable in understanding patients' needs, improving care coordination with outpatient and community resources, increasing the dignity and respect with which patients are treated, and supporting development of patient-centered treatment plans. Additionally, outpatient settings are encouraged to prioritize screening tools that have undergone adequate testing to ensure they are accurate and reliable. For example, the AHC HRSN Screening Tool has been tested across many care delivery settings in diverse geographic locations and has demonstrated evidence of both reliability and validity.[367] We thank the commenters for their recommendations and will monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if improvements are identified through this process.
Comment: Many commenters did not support the Screening of SDOH measure due to their beliefs that the lack of standardized screening approach could introduce inaccuracies in data collection. These commenters recommended that CMS require outpatient settings to take a standardized screening approach for implementing this measure, including providing set screening tools, as this would allow for more valid comparisons between HOPDs, REHs, and ASCs and would reduce ambiguity.
A few commenters recommended working with stakeholders to develop consistent survey questions while maintaining the flexibility for how and when facilities screen patients. A commenter expressed concern that not all tools compiled on the SIREN website have been validated or “vetted through all steps of gold standard measure development.” This commenter recommended that CMS provide a list of screening tools that are adequately tested and validated to reduce potential burden that may occur if CMS were to mandate a standardized tool at a later time. A commenter expressed their belief that standardized measure definitions within and across programs, coupled with more timely performance data provided to facilities will greatly support the advancement of patient safety and quality improvement by creating consistent and actionable goals and results. The commenter recommended providing more timely actionable data to promote immediate advancements in patient safety and quality.
Response: We thank the commenters for their feedback. We agree that allowing outpatient settings flexibility with tool selection is a tradeoff, but, as we discussed previously, this flexibility aims to reduce burden for HOPDs, REHs and ASCs, by allowing these settings to continue to use screening tools that they may already use for greater customization of screening and data collection for each settings' patient population and individual needs. This is the first time we will be collecting SDOH data in these settings as part of quality performance measurement and ( printed page 94392) we want to ensure that all HOPDs, REHs, and ASCs are working towards initial screening, in a form and manner that works for them. We anticipate that for HOPDs, REHs, and ASCs associated with other healthcare settings where this measure has been adopted, such as the Hospital IQR Program, an organic alignment of screening tools will emerge and generate a degree of industry-led standardization. We also anticipate additional emphasis on standardized and validated screening instruments in future versions of these measure. Outpatient settings are encouraged to prioritize screening tools that have undergone adequate testing to ensure they are accurate and reliable. As we indicated previously, health equity is a key priority, and we intend to continue to develop relevant measures. We are taking commenters' recommendations under consideration as appropriate to inform potential future notice-and-comment rulemaking.
In response to the commenter's recommendation to provide more timely actionable data, we intend to publicly display data for the Screening for SDOH measure on the first available refresh of Care Compare. Typically, this would occur in October of the year following data submission. For example, for the CY 2026 Reporting Period/2028 Payment Determination or Program year, the data would be displayed on Care Compare in October 2027. This is as soon as technically feasible for data collected during the CY 2026 performance period and submitted between January 1 and May 15, 2027.
Comment: Many commenters that did not support adoption of the Screening for SDOH measure stated that the measure is not appropriate for the outpatient setting where there is little to no patient follow-up. A few of these commenters noted that outpatient settings would need to hire staff such as case managers and social workers to conduct screening in advance of a patient visit and make any needed workflow changes.
For the ASCQR Program specifically, many commenters stated their belief that the measure does not capture the essence of the work currently done by ASCs, the measure falls outside of the primary ASC function, the questions asked are inappropriate for the ASC setting, and the data should be collected by primary care physicians or hospitals.
For the Hospital OQR Program, a commenter expressed concern that HOPDs that focus on diagnostic testing are not suited to conduct screening for SDOH due to the absence of specialized staff to conduct this screening. A commenter recommended a more targeted approach to screening that only requires screening under the Hospital OQR Program in HOPDs with long-term patient and provider relationships, adequate staff, and resources to respond to results from screening. This commenter recommended convening a TEP with representation from the hospital community to determine how to identify HOPDs that have the resources to implement this measure.
Response: We thank the commenters for their concerns. However, we disagree with the commenters' assertion that the measure is not appropriate for outpatient settings. HOPDs, ASCs, and REHs are responsible for treating the whole patient and ensuring the best outcomes possible for the patients that they serve, including identifying potential barriers to optimal health, and helping patients identify resources to address those barriers. One of the benefits of collecting data from screening for HRSNs will be identification of opportunities to enable meaningful action, including prioritizing and investing in such resources. As part of the PRMR process, the measure underwent an assessment of the measure's testing data and appropriateness for the outpatient setting, including a review of the measure's scientific acceptability, feasibility, and usability. We are taking commenters' recommendations under consideration as appropriate to inform future potential rulemaking.
Additionally, as previously noted, while outpatient care can refer to numerous types of health services, such as emergency department (ED) services, observation services, outpatient surgical services, lab tests, and X-rays, for purposes of this measure in the Hospital OQR, REHQR, and ASCQR Programs, patients receiving services that are limited to specific medical tests are not included in the screening cadence of this measure. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
Comment: Many commenters that did not support adoption of the Screening for SDOH measure cited concerns about whether the data would be meaningful, accurately and fairly evaluate organizations, and lead to progress or change. A commenter questioned the efficacy and validity of the AHC Model and whether the testing of universal HRSN screening demonstrated meaningful impact on health outcomes and health equity. A commenter expressed concern about the required author permissions when using the AHC tool and noted that healthcare facilities would need to “reach out to the screening question author to notify them of their plan to use it.”
Several commenters noted other factors to include in the measure to make it more meaningful, including demographics across communities other than race and ethnicity (such as age, gender, and health literacy, accessibility of services, etc.). A commenter recommended using a community needs assessment and implementing regulatory flexibilities to help providers effectively act on social determinants of health and gaps in access to care.
A commenter urged CMS to develop a more meaningful measure for Hospital OQR Program that captures a granular scale of data and differentiates patients with the greatest need for follow-up.
Response: We thank the commenters for their input. The intent of this measure is to promote adoption of screening patients for HRSNs by healthcare providers as well as taking action to connect patients who identify one or more HRSNs with available resources. Evaluation of the AHC Model concluded that universal screening may identify needs that would otherwise remain undetected.[368] While broad availability of community-based resources that address patients' HRSNs would be ideal, one of the benefits of collecting data from screening for HRSNs will be identification of opportunities to enable meaningful action, including prioritizing and investing in such resources. Beginning to collect the data remains imperative and such data collection has already allowed some entities to reallocate resources to address HRSNs that disproportionately affect a given patient population or geographic region, as noted in the FY 2023 IPPS/LTCH PPS final rule, in which the Hospital IQR Program adopted this measure (87 FR 49213). This input is valuable in the continuing development of the CMS health equity quality measurement efforts and supports our aims to acknowledge the impact of HRSNs on healthcare access, utilization, outcomes, and costs. We will continue to take all concerns, comments, and suggestions into account for any potential future development and expansion of our health equity quality measurement efforts.
Regarding the commenter's concern about using the AHC Screening tool, we ( printed page 94393) note that use of this tool is not required, but if healthcare facilities would like to use it, they can follow the guidance provided in the “Accountable Health Communities Health-Related Social Needs Screening Tool Citation and Notification Information” document, which requires citing the authors and in some instances, notifying the authors of intended use.[369] Beyond notification and citation, there are no restrictions on use of the AHC screening tool questions. Furthermore, healthcare facilities may create or choose other tools to use for implementing this measure.
Comment: Several commenters that did not support adoption of the Screening for SDOH measure expressed concern that this measure has not received endorsement by the CBE. Some of these commenters noted that this was a previous condition placed by the MAP when the Screening for SDOH measure was adopted in other quality reporting programs. Several commenters recommended submitting the measure for endorsement by a CBE. A commenter also suggested using a different measure for achieving health equity that is endorsed by the National Quality Forum (NQF).
Response: While we recognize the value of measures undergoing review for potential CBE endorsement, measures of health equity are a priority for CMS, and it is important to implement this measure as soon as possible, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting for the CY 2026 reporting period. We note that section 1833(t)(17) of the Act requires measures developed for the Hospital OQR Program to reflect consensus among affected parties. Under section 1833(i)(7)(B), this requirement also applies to the ASCQR Program except as the Secretary may otherwise provide. Under section 1833(t)(17)(C)(i), the Hospital OQR Program is only required to include measures set forth by a national consensus building entity “to the extent feasible and practicable.” We have been unable to identify any measures on this topic set forth by a consensus building entity and it is not feasible and practicable to include such a measure at this time. As we have noted in previous rulemaking, consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment (75 FR 72064 and 72065 for the Hospital OQR Program and 76 FR 74494 for the ASCQR Program). As stated previously, this measure has been adopted through rulemaking with public comment periods in other care settings, including inpatient hospitals, PPS-Exempt cancer hospitals, and dialysis facilities, which evidence broad applicability and acceptability of the measure concept and specifications for different healthcare facility types, both inpatient and outpatient.
For the REHQR Program, we note that under section 1861(kkk)(7)(C)(ii) of the Act, the Secretary may specify a measure that is not endorsed if due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures that assess screening for SDOH and are endorsed by a consensus organization, and therefore, the exception in section 1861(kkk)(7)(C)(ii) of the Act applies.
We note further that the Screening for SDOH measure has been adopted in four other CMS quality reporting programs (Hospital IQR Program, IPFQR Program, PCHQR Program, and ESRD QIP), and that this measure establishes an important foundation to prioritize the achievement of health equity among outpatient settings.
Regarding the commenter's recommendation that we implement measures endorsed by the NQF, we wish to clarify that the CBE is currently Battelle. Battelle administers the PQM in the pre-rulemaking and measure endorsement processes. As previously discussed in section XIV.B.1.c of this final rule with comment period, this measure has been reviewed during the Pre-Rulemaking Measure Review (PRMR) process, where the committee recommended this measure with conditions. We will consider submitting the Screening for SDOH measure to the CBE for endorsement in the future.
Additionally, we note that our approach to developing health equity measures is incremental and intended to evolve over time to capture social drivers of health in the Hospital OQR, REHQR and ASCQR Programs. We will continue to take all concerns, comments, and suggestions into account for any potential future development and expansion of our health equity quality measurement efforts. As with all Hospital OQR, REHQR and ASCQR measures, we will monitor the data and any unintended consequences of the measure as part of standard measure maintenance.
Comment: Several commenters recommended CMS use consistent terminology when describing social risk factors related to health outcomes to ensure consistency with other quality reporting programs and prevent unintended consequences that may negatively impact patients. A few commenters recommended that CMS work with stakeholders to clearly define terms and domains related to this measure to improve processes and data collection.
Response: We thank the commenters for their feedback. HRSNs, which we have previously defined as individual-level, adverse social conditions that negatively impact a person's health or healthcare, are significant risk factors associated with worse health outcomes as well as increased healthcare utilization.[370] As previously mentioned, SDOH is an umbrella term that refers to community-level factors that impact health and well-being, while HRSNs are social and economic needs that individuals experience that affect their ability to maintain their health and well-being.[371] Conceptually, HRSNs exist along a continuum with other equity-related terms—such as “social determinants of health” and “social risk factors”—used to describe upstream factors that can adversely affect the health of individuals and communities. We agree these terms are often conflated and even used interchangeably, and the variety of terms has created both confusion as well as concern, prompting leaders in the field to adopt “social drivers of health” instead. In the future, we intend to utilize “social drivers of health” terminology to more holistically capture aforementioned and related concepts, while minimizing potential misinterpretation or negative connotation. We thank commenters for their recommendations and will continue identifying opportunities for collaboration with stakeholders to determine the most appropriate terminology for screening for social drivers of health.
( printed page 94394)Comment: Several commenters expressed concerns regarding the measure specifications for the Screening for SDOH measure and asked for further clarification on the target population (or denominator), frequency of screening, and screening across multiple outpatient care settings.
A commenter was further concerned that some domains will have small denominators that could produce inaccurate or biased results. Another commenter noted that the measure specifications were vague and needed additional clarification. A commenter requested clarification on how entities will verify and confirm that screenings were previously conducted within the reporting period. The commenter recommended allowing screening results to remain valid for 12 months to reduce risk of over-screening.
A few commenters requested clarification on the measure specifications under the Hospital OQR Program. A commenter requested additional clarification on the denominator of the measure as the word “admitted” is vague. Another commenter requested clarification on the frequency of screening required under the Hospital OQR Program. A commenter expressed concern that the broad screening policy fails to consider the nuances of screening pediatric patients for the Hospital OQR program. The commenter asked for clarification on the screening and reporting requirements for screening pediatric patients along with members of the household.
Response: We acknowledge commenters' concerns regarding the measure specifications. The target population, or denominator, for the Screening for SDOH measure includes patients who are 18 years or older on the date of service and who are admitted to an HOPD, REH, or ASC. Patients can be excluded from the denominator if they opt-out of screening or are unable to complete the screening and have no legal guardian or caregiver to do so on their behalf during the visit. Additionally, as previously noted, patients receiving services that are limited to specific medical tests are not included in the denominator of this measure. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
In addition, we acknowledge that certain patients will have frequent outpatient visits, such as patients receiving outpatient hospital-based chemotherapy or patients regularly receiving outpatient hospital-based infusions or injections. These patients and other patients frequently admitted to the hospital do not need to be re-screened at every outpatient admission. Rather, after receiving an initial screening, during subsequent visits within the calendar year the patient's provider could confirm the accuracy of their previously reported screening result and provide an opportunity for the patient to update that information as needed. In addition, if a patient has documented SDOH screening results available for review within the patient's medical record or EHR at the time of the outpatient admission, even if the screening occurred outside of the HOPD, REH, or ASC, and these SDOH screening results fall within the same measure performance period year, then these previously captured SDOH Screening results could be included in the outpatient settings' reporting of numerator and denominator data, during the measure's reporting period. In addition, if a patient has multiple admissions during the same performance period, and receives multiple SDOH screenings and has results of each SDOH screening documented, then the most recent result should be submitted.[372 373] For example, if the patient was screened in May 2025 and then screened again in December 2025, the results of the December 2025 admission would be used for the CY 2025 reporting period.[374] We intend to monitor and evaluate the measure screening requirements, including frequency, in these outpatient settings to ensure balance between quality of care for patients and facility burden.
We thank the commenter for their concern regarding screening pediatric patients in the Hospital OQR Program and note that as outlined in the measure specifications, screening is only required for patients who are admitted to an outpatient facility and who are 18 years or older on the date of admission. If the patient admitted to the HOPD is over 18 years of age and other members of the household are present, only the admitted patient is required to be screened.[375] We note that the measure specifications do not prevent hospitals from screening additional patients at their discretion.
With respect to the commenter's question regarding the use of the word “admitted” in the HOPD setting, the term admitted patients refers to a person who receives ambulatory care in these designated settings. We agree that screening for SDOH is most appropriate when administered so that the patient's information can be used by the patient's care team. As we stated in the proposed rule, screening for food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety can help identify and provide appropriate referrals for patients who may benefit from greater support in one or more of those areas. As such, we are clarifying that while outpatient care can refer to numerous types of health services, such as emergency department (ED) services, observation services, outpatient surgical services, lab tests, and X-rays, for purposes of this measure in the Hospital OQR Program, REHQR, and ASCQR programs, patients receiving services that are limited to specific medical tests are not included in the denominator cohort. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
For additional information on how to apply and report these screenings, we refer readers to the Hospital IQR Program's FAQ document regarding this measure in the Hospital IQR Program, available at: https://www.qualityreportingcenter.com/globalassets/2024/04/iqr/17.-sdoh-measure--faqs_april-2024_vfinal508.pdf. We will develop a similar Frequently Asked Questions document for the Hospital OQR, REHQR and ASCQR Programs as part of providing educational and training materials; this document will be conveyed through routine communication channels.
Comment: A few commenters asked for clarification as to why the five HRSN domains were chosen for screening.
Response: We have prioritized selection of the proposed five HRSN domains based on existing evidence ( printed page 94395) from the AHC Model that informed the initial selection, and emerging evidence of correlations between given SDOH and worse health outcomes and SDOH for which interventions have shown marked improvements in health outcomes and healthcare utilization (89 FR 59444).
Comment: A few commenters raised concerns about penalization among healthcare facilities for not complying with the measure requirements. A commenter recommended sensitivity and a formulaic coefficient when evaluating under-resourced ASCs to ensure congruency on quality performance relative to facilities with more resources and adjusting programmatic requirements to ensure that reporting on quality measures is feasible for all facilities and that under-resourced facilities do not face undue difficulty or burdensome penalties that could affect access to care for vulnerable populations.
A commenter expressed concern that mandatory reporting of SDOH data would place undue burden on REHs that could penalize or reduce reimbursement. This commenter recommended to continue with voluntary reporting and avoid linking mandatory reporting to financial penalties or reimbursement reductions.
Response: We thank the commenters for their feedback and acknowledge their concerns. We note that the Hospital OQR and ASCQR Programs are pay-for-reporting programs, and participants are not scored, and do not have their payments affected, based on their performance on measures. The REHQR Program does not include a financial incentive or penalty for REHs. Additionally, we note that as discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59516 and 59517, 89 FR 59522 and 59523, and 89 FR 59526, respectively) and in sections XXVI.A.c, XXVI.B.c, and XXVI.C.c of this final rule with comment period, we estimate minimal burden of 2 minutes per patient screening and 10 minutes per hospital/facility to report the measure annually.
Comment: A few commenters recommended we ensure alignment with Project Gravity standards and promote interoperability standards for data collection. A commenter recommended we harmonize drivers of health assessment approaches with the National Committee for Quality Assurance (NCQA) to ensure these initiatives that address SDOH in patient assessments are not active performance measures, cause confusion, or waste resources. This commenter also recommended we ensure screening tools are approved by the Social Innovation and Research Evaluation Network (SIREN) and with the Core Quality Measures Collaborative (CQMC) to promote alignment across public and private payers.
Response: We thank the commenters for this feedback. This data collection will inform meaningful and sustainable solutions for other provider types through similar collections in other quality reporting programs. We will continue identifying opportunities for collaboration with other stakeholders to align drivers of health assessment across CMS programs. We commend additional stakeholder efforts currently underway to expand capabilities to capture additional drivers of health data elements, including the Gravity Project.[376] We support harmonization of social risk factor data for interoperable electronic health information exchange that will meet information exchange standards.
We note that this rule finalizes a requirement at § 419.46(j)(1) that hospitals participating in the Hospital OQR Program “utilize certified technology updated to be consistent with the Office of the National Coordinator for Health Information Technology's health IT certification criteria, as adopted and updated in 45 CFR 170.315,” which is consistent with ONC's current “edition-less” approach to the ONC health IT certification criteria (89 FR 1207). We also note that, for clarity, this finalized requirement differs from our proposal by the removal of an acronym and its replacement by the full agency name. Additionally, many REHs reported under the Medicare Promoting Interoperability and Hospital IQR Programs prior to conversion, and thus are likely to also have EHR systems already embedded in their workflows.
Extraction of structured data from a certified EHR can make the data more accessible for utilization and submission for quality measurement reporting.[377] As previously stated, this rule finalizes a requirement at § 419.46(j)(1) that hospitals participating in the Hospital OQR Program “utilize certified technology updated to be consistent with the Office of the National Coordinator for Health Information Technology's health IT certification criteria, as adopted and updated in 45 CFR 170.315.” We encourage use of tools that will meet information exchange standards and facility interoperability. We also encourage providers to identify and utilize tools that rely on standards-based approaches to data collection and utilization to support interoperability of these data.
We agree with the commenter that SIREN is a good resource when selecting a screening tool as it contains descriptions of the content and characteristics of various tools, including information about intended populations, completion time, and number of questions. Another example is the AHC HRSN Screening Tool. The AHC HRSN Screening Tool is a 10-item screening tool, with 16 supplemental questions, to identify patient needs that can be addressed through community services in four domains (economic stability, social and community context, neighborhood and physical environment, and food). Outpatient settings are encouraged to prioritize screening tools that have undergone adequate testing to ensure they are accurate and reliable.
Comment: A few commenters recommended ensuring that screenings are performed in a person-centered way due to the sensitive nature of the screening domains and encouraged educating and building trust with patients as to why this SDOH data is being used and collected. A few commenters encouraged working with stakeholders to ensure different approaches to social needs screening are considered. These commenters recommended developing a database that provides information on the methods and processes that are most effective for HRSN screening.
Response: We thank commenters for their recommendations on screening methods and agree with the importance of conducting person-centered screenings to maintain trust between patients and their providers. We reiterate that healthcare facilities would have the flexibility to customize screening and data collection to their patient populations and individual needs. We encourage HOPDs, REHs, and ASCs to train staff on culturally sensitive engagement and trauma-centered care. We also encourage HOPDs, REHs, and ASCs to ensure that patients feel safe when answering questions and remind patients that they may opt out of the screening for any reason. We note that the measure specifications exclude patients: (1) who opt-out of screening; and (2) are ( printed page 94396) themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf (89 FR 59447). Providers are also encouraged to use tools and screening protocols that consider the sensitive nature of the screening domains. We will continue identifying opportunities for collaboration with interested parties to align screening for social drivers of health assessment across CMS programs.
Comment: Many commenters expressed concerns about the risk of over-screening, as patients may receive care in multiple settings and that interoperability challenges prevent access to data collected by other healthcare facilities. These commenters noted that repeated screenings could be burdensome for both staff and patients and distressing for patients who would expect the healthcare facility to address their needs after the screening.
A commenter expressed concern that ASCs are already engaging in facility-appropriate screening efforts to support patients with the social determinants of health and additional screening would be duplicative.
Response: We acknowledge commenter's concerns regarding the risk of over screening and reiterate that HOPD, ASCs, and REHs could confirm the current status of any previously reported HRSNs in another care setting and inquire about others not previously reported in lieu of re-screening a patient for all HRSN domains within the reporting period. This will reduce the burden and time necessary for re-screening a patient for all five HRSN domains. If this information has been captured in the EHR in another outpatient setting or the inpatient setting during the same reporting period, the HOPD, REH, and ASC could include that information in the reporting of numerator and denominator data during the measure's reporting period. Patients should be screened during every admission, but only unique patients should be included in each reporting period (year).[378]
We thank commenters for their questions regarding the technical specifications of the measure. In addition, as we stated in the proposed rule, screening for food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety can help identify and provide appropriate referrals for patients who may benefit from greater support in one or more of those areas. As such, we are clarifying that while outpatient care can refer to numerous types of health services, such as emergency department (ED) services, observation services, outpatient surgical services, lab tests, and X-rays, for purposes of this measure in the Hospital OQR Program, REHQR, and ASCQR programs, patients receiving services that are limited to specific medical tests are not included in the denominator cohort. These services include imaging, laboratory, and pharmacy services, which are typically specific types of auxiliary services to a patient's more comprehensive care, where such screenings and referrals should be provided.
We commend ASC efforts currently underway to capture additional drivers of health data elements. As previously stated in the proposed rule, we are allowing healthcare facilities to select their screening tool to reduce burden and in recognition of the fact that some healthcare facilities may already be screening their patients for HRSNs as we recognize the benefits of providing healthcare facilities with flexibility to customize screening and data collection to their patient populations and individual needs (89 FR 59447). We note that while the Screening for SDOH measure requires screening for the five identified HRSNs, ASCs may screen for additional HRSNs that they determine are relevant for their patient population and the community in which they serve. For example, the AHC HRSN Screening Tool includes questions for eight supplemental domains, including financial strain. Furthermore, we note that this measure is a first step towards development of a long-term strategy to integrate social drivers of health and HRSN data into quality performance measurement and is part of our broader commitment to health equity.
Comment: A few commenters that did not support the Screening for SDOH measure expressed concerns regarding patient safety and privacy when conducting screening in small or rural communities. A commenter expressed concern about the increased reliance on quality measures for measuring safety. The commenter noted their belief that safety measures are surrogate measures of harm, subject to variation based on patient populations.
Response: We acknowledge the commenters' concerns and recognize that facilitating quality improvement for rural healthcare facilities can present unique challenges and is a high priority under the Meaningful Measures Framework. One of the potential benefits of screening data will be identification of opportunities to enable meaningful action, including prioritizing and investing in community-based resources that address patients' health-related social needs. We believe that each hospital best understands the patient population they serve and hope that hospitals can discern whether they have existing resources to meet their populations' unmet needs or dedicate further resources for which they know a need exists and now have evidence of the extent that resource allocation is necessary. It is important to use quality measures as a benchmark for identifying best practices in care and to accurately track quality improvement progress.[379] We emphasize that quality measures help to improve the quality of healthcare through an approach that is consistent and holds healthcare facilities accountable.[380]
We also understand the sensitive nature of screening, and that patient safety must remain the HOPD's, REH's, and ASC's principal concern. We encourage all outpatient settings to train staff on culturally sensitive engagement and trauma-centered care and note that there are a variety of methods to screen for HRSNs that increase privacy, including written or digital methods, such electronically through a patient portal or on tablets. We advise HOPDs, REHs, and ASCs to ensure that patients feel safe answering questions and to further remind patients that they may opt out of the screening for any reason. We note that the measure specifications exclude patients: (1) who opt-out of screening; and (2) are themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf (89 FR 59447). We also note that, because these healthcare facilities are covered entities under the HIPAA Privacy and Security Rule (codified at 45 CFR parts 160 and 164),[381] information provided by patients in response to screening for this ( printed page 94397) measure likely would be PHI.[382] Therefore, these healthcare facilities are responsible for complying with applicable privacy and security requirements under HIPAA and other laws to ensure that patients' PHI is protected and not impermissibly disclosed.
In response to the commenter's concern about the increased reliance on quality measures for measuring safety, it is important to use quality measures as a benchmark for identifying best practices in care and to accurately track quality improvement progress.[383] We emphasize that quality measures help to improve the quality of healthcare through an approach that is consistent and holds healthcare facilities accountable.[384]
Comment: A few commenters that did not support the Screening for SDOH measure expressed concern that the measure does not account for geographic variation in screening. A commenter noted that there exists significant variation in community needs across geographic areas and not accounting for geographic variation of social drivers could be misleading. The commenter recommended using the Vizient Vulnerability Index for benchmarking to address these concerns.
Response: Due to variability across healthcare facilities and the populations they serve, and in alignment with the Hospital IQR Program, we would allow healthcare facilities flexibility with the selection of tools to screen for food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety. As we noted in the CY 2025 OPPS/ASC proposed rule (89 FR 59447), one example of a screening tool that healthcare facilities could consider using is the AHC HRSN Screening Tool, which providers used in the AHC Model to screen for HRSNs in their Medicare, Medicaid, and dually eligible beneficiary populations. The AHC HRSN Screening Tool has been tested across many care delivery sites in diverse geographic locations across the United States and demonstrates evidence of both reliability and validity across geographic regions. Moreover, the AHC HRSN Screening Tool can be implemented in a variety of healthcare settings, including HOPDs, REHs, and ASCs. We thank the commenter for their recommendation to utilize the Vizient Vulnerability Index and will take this into consideration as appropriate as we monitor implementation of the Screening for SDOH measure in our quality reporting programs.
Comment: A few commenters recommended combining inpatient and outpatient reporting and noted that this would reduce duplicative efforts.
Response: We thank commenters for their recommendation. As previously stated in the CY 2025 OPPS/ASC proposed rule, the populations represented by the Hospital IQR and Hospital OQR programs are distinct, resulting in a different denominator calculation of the measure for each program (89 FR 59446). Therefore, inpatient and outpatient settings will be unable to accurately report this measure if data are combined for the Hospital IQR and Hospital OQR Programs. We will continue to require a separate data submission for each program in the HQR system.
Comment: A few commenters did not support the Screening for SDOH measure in the ASCQR Program due to the sensitive nature of screening for risk of interpersonal violence. These commenters recommended changes that included removing this domain from the measure specifications and updating the measure to ensure patient privacy when responding to this screening question, either by excluding patients who could not respond to the question confidentially, or by ensuring responses remain hidden in all records and handouts accessible to patients. Additionally, a commenter expressed concerns regarding the cited literature, stating their belief that the discussed pilot study was not representative of the ASC setting. A commenter also stated that CMS's assertion that ASCs already collect data regarding HRSNs is inaccurate.
Response: We have prioritized selection of the proposed five HRSN domains based on existing evidence from the AHC Model that informed the initial selection, and emerging evidence of correlations between given social drivers of health and worse health outcomes and social drivers of health for which interventions have shown marked improvements in health outcomes and healthcare utilization. However, we understand commenters' concerns regarding the sensitive nature of screening for risk of interpersonal violence within the ASC environment and agree that patient safety must remain the ASC's principal concern.
Regarding the interpersonal safety domain and appropriateness of screening in the ASC environment, we note that with a year of voluntary reporting, there is time for ASCs to train staff in the collection of data for HRSNs. Additionally, there are a variety of methods, other than audible methods, to screen for HRSNs that increase privacy, including written or digital methods, including electronically through a patient portal or on tablets. We advise ASCs to ensure that patients feel safe answering questions and to further remind patients that they may opt out of the screening for any reason. We note that the measure specifications as proposed excludes patients: (1) who opt-out of screening; and (2) are themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf (89 FR 59447).
Comment: A commenter expressed concern that the lack of standardization for this measure is not in alignment with the Medicaid and Children's Health Insurance Program (CHIP) data collection standards which require that data collection on race, ethnicity, sex, primary language, and disability status must be standardized. This commenter requested clarification on which aspects of SDOH would be exempt from necessary aggregation due to intrinsic intersection with race, ethnicity, sex, primary language, and disability status. The commenter further noted that if the data collected by a tool developed for this new proposed measure are not applicable or in line with Medicaid and CHIP data standards, it does not align with the first priority of CMS's Health Equity Framework of the “collection . . . of standardized data . . . across CMS programs”.
Response: We acknowledge the commenter's concern. We will explore opportunities to align with Medicaid and CHIP data collection standards; however, we currently collect race and ethnicity data for chart-abstracted measures, CAHPS surveys, and eCQMs.
After consideration of the public comments we received, we are finalizing the adoption of the Screening for SDOH measure as proposed in the Hospital OQR, REHQR, and ASCQR Programs beginning with voluntary reporting for the CY 2025 reporting period/CY 2027 payment or program determination followed by mandatory reporting beginning with the CY 2026 ( printed page 94398) reporting period/CY 2028 payment or program determination.
3. Adoption of the Screen Positive Rate for Social Drivers of Health (SDOH) Measure for the Hospital Outpatient Quality Reporting (OQR), Rural Emergency Hospital Quality Reporting (REHQR), and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Payment or Program Determination
a. Background
In the outpatient setting, we encourage systematic screening of patients' HRSNs to identify patient needs and support improvements in health outcomes. While the Screening for SDOH measure (discussed previously in section XIV.B.2 of this final rule with comment period) identifies individuals with HRSNs, the Screen Positive Rate for SDOH measure estimates the magnitude of these needs for a healthcare facility's patient population served. We believe the adoption of the Screen Positive Rate for SDOH measure would encourage healthcare facilities to track the prevalence of specific HRSNs among patients over time and use the data to stratify risk as part of quality performance improvement efforts.
We proposed that healthcare facilities would be required to report the Screen Positive Rate for SDOH measure as the rate of patients who screened positive for each of the five core HRSNs domains discussed in Table 161: food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety.[385]
b. Measure Overview
While the Screening for SDOH measure (discussed in section XIV.B.2 of this final rule with comment period) enables identification of individuals with HRSNs, the Screen Positive Rate for SDOH measure would allow healthcare facilities to capture the magnitude of these needs by requiring healthcare facilities to report the rates of patients who screened positive for each of the five core HRSNs. The Screen Positive Rate for SDOH is a process measure that provides information on the percent of patients receiving care at an HOPD, REH, or ASC, who were 18 years or older on the date of service, who were screened for all five HRSNs described in Table 161, and who screened positive for one or more of those HRSNs. Healthcare facilities would report this measure as five separate rates, one for each of the HRSNs: food insecurity, housing instability, transportation needs, utility difficulties, and interpersonal safety. This measure is not intended for comparison of screen positive rates of HRSNs between healthcare facilities but is rather to provide transparency in the delivery of care and actionable information to healthcare facilities on the unmet needs among their patients.
c. Pre-Rulemaking Measure Review
As part of the PRMR process, the Hospital Recommendation Group reviewed and voted on the Screen Positive Rate for SDOH measure [386] during their meeting on January 18 and 19, 2024.[387] [388] The committee did not reach the 75 percent vote required to reach a consensus as to its recommendation for the Screen Positive Rate for SDOH measure for the Hospital OQR, REHQR, or ASCQR Programs. The committee expressed a concern about ambiguity in the interpretation of data from the Screen Positive Rate for SDOH measure as well as expectations regarding healthcare facilities. We acknowledge that a high score could be interpreted in different ways but that the objective of this measure is to incentivize collection of these data to help identify patient needs and where resources constraints exist. The committee also discussed a condition specific to the Hospital OQR Program, which was to allow hospitals to report this measure one time each year for both the Hospital IQR Program and Hospital OQR Program.[389] We note that we considered allowing hospitals to report this measure jointly for the Hospital IQR and Hospital OQR Programs (if applicable); however, as the patient populations represented by the programs are different, as is the measure calculation due to this difference in the denominator, we proposed to require a separate data submission for each program.
Further, we have identified the implementation of this measure in the Hospital OQR, REHQR, and ASCQR Programs as an important way to address the health equity measurement gap. We also believe that the information collected from this measure can help HOPDs, REHs, and ASCs understand SDOH needs in their patient population and to devise appropriate interventions. On this basis, we proposed this measure for adoption for all three of our programs.
d. CBE Endorsement
As discussed in section XIV.B.2.d of this final rule with comment period, we find no other feasible and practicable measures set forth by a national consensus building entity on the topic of screening for SDOH. While we recognize the value of measures undergoing CBE endorsement review and prefer to use endorsed measures, there are currently no CBE-endorsed measures that address screening for SDOH in the outpatient setting. Given the urgency of achieving health equity, it is important to implement this measure as soon as possible. We note that the five domains for which patients would be screened were chosen based upon literature review and expert consensus, and that these five domains informed development of the Screen Positive Rate for SDOH measure. We will consider submitting the Screen Positive Rate for SDOH measure to the CBE for endorsement in the future.
e. Data Sources
The data sources for this measure are as described for the Screening for SDOH measure found in section XIV.B.2.e of this final rule with comment period.
f. Measure Calculation
The Screen Positive Rate for SDOH measure is calculated with a numerator and denominator. The numerator is defined as the number of patients receiving care at an HOPD, REH, or ASC who are 18 years or older on the date of admission, who were screened for all five HRSNs described in Table 161, and who screen positive for having a need in one or more of those HRSNs (calculated separately). The denominator is defined as the number of patients receiving care at the HOPD, REH, or ASC who are 18 years or older on the date of admission and are screened for all five HRSNs during their care. ( printed page 94399)
The results of this measure are calculated and reported as five separate rates—one for each HRSN, each calculated with the same denominator. The measure excludes patients who: (1) opt-out of screening; or (2) are themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf.
g. Data Submission and Reporting
While this measure would require healthcare facilities to collect patient-level data on their patients' SDOH screening results, consistent with the Screening for SDOH measure, we proposed to adopt this measure as an aggregate measure in the CY 2025 OPPS/ASC proposed rule. Specifically, we proposed that healthcare facilities would be required to submit aggregated data representing the total numerator results for each of the five screening areas and the total number of patients screened for all five of the HRSNs. We proposed to require aggregate data because we believe it is unnecessary for healthcare facilities to submit data collected at the patient level as this would cause undue burden due to the transfer of large quantities of data. However, in the future, we may consider the reporting of patient-level information. This measure aims to encourage healthcare facilities to screen for and identify HRSNs as it is most important for healthcare facilities to collect this HRSN data to address social needs among their patient populations.
Healthcare facilities would be required to submit information via a CMS-designated information system (currently the HQR system) consistent with the prior adoption of this measure in the Hospital IQR, IPFQR and PCHQR Programs. We refer readers to sections XV.E.2.a, XVI.E.3.b, and XVII.E.2.a of this final rule with comment period for additional details regarding data submission using the CMS-designated information system in the Hospital OQR, REHQR, and ASC Programs, respectively.
As we noted in section XIV.B.3.c of this final rule with comment period, we considered allowing hospitals to report this measure jointly for the Hospital IQR and Hospital OQR Programs; that is, allowing hospitals to submit once under both programs rather than submitting data twice in the HQR system. However, as the populations represented by the programs are different, resulting in different calculations of the measure denominator under each program, we proposed to require a separate data submission for each program.
We proposed to adopt this Screen Positive Rate for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period, followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determination to be consistent with the Screening for SDOH measure. Similar to the Screening for SDOH measure, a voluntary period would allow time for healthcare facilities to select and integrate screening tools into their clinical workflow processes and gain experience with both measures before measure results are publicly displayed on the Compare tool.
We invited public comment on our proposal to adopt the Screen Positive Rate for SDOH measure for the Hospital OQR, REHQR, and ASCQR Programs beginning with voluntary reporting on this measure for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment or program determination, as described above.
Comment: Many commenters supported adoption of the Screen Positive Rate for SDOH measure and CMS's continuing efforts towards measure alignment across the CMS quality reporting programs. Commenters expressed support for hospitals not only tracking who gets screened but also who screens positive as a step toward hospitals developing action plans to address the risk factors identified. Commenters additionally supported the five domains identified as they support a stronger safety net and focus on unmet needs, noting that all five domains affect health and quality of patient lives. Some commenters noted that adoption of the measure will ensure that the practitioners' time conducting the screening is properly reimbursed.
Response: We thank commenters for their support of the Screen Positive Rate for SDOH measure. We agree that HRSNs are critical factors that impact patient outcomes and health equity. By systematically screening for these factors and enabling early interventions, healthcare providers can address the root causes of disparities and encourage a shift toward a healthcare culture that prioritizes proactive intervention and data-driven policy changes to close the gap in health outcomes between different populations.
Comment: A few commenters did not support the Screen Positive Rate for SDOH measure because it was not endorsed by the CBE or because the PQM did not reach consensus for recommending the measure. A few commenters recommended additional testing in the outpatient setting. A few commenters recommended that CMS continue to test these measures through voluntary reporting in the programs where they have already been adopted prior to expanding them into additional quality reporting programs. For ASCs, a commenter noted that the measure cannot be said to meet the requirement for consensus through broad acceptance of the measure by ASCs or through use of the measure by ASCs as ASCs were not involved in the development of the measure.
Response: While we recognize the value of measures undergoing review for potential CBE endorsement, measures of health equity are a priority for CMS, and it is important to implement this measure as soon as possible. For the Hospital OQR and ASCQR Programs, we note that section 1833(t)(17) of the Act does not require that each measure we adopt be CBE-endorsed (76 FR 74494), but states that the Hospital OQR and ASCQR Programs, to the extent feasible and practicable, shall include measures set forth by one or more national consensus building entities. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures on this topic endorsed by a consensus organization, so the inclusion of such a measure is not feasible or practicable. Section 1833(t)(17)(C)(i) also requires measures included in the Hospital OQR and ASCQR Programs to reflect consensus among affected parties. As we have stated in previous rulemaking (75 FR 72064 and 76 FR 74494), consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment. The Screen Positive Rate for SDOH measure achieved the aim of consensus among affected parties through its broad use in a number of quality reporting programs, including the Hospital IQR Program (87 FR 49215 through 49220), IPFQR Program (88 FR 51117 through 51121), PCHQR Program (88 FR 59219 through 59222), and ESRD QIP (88 FR 76476 through 76480).
For the REHQR Program, we note that under section 1861(kkk)(7)(C)(ii) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We reviewed measures endorsed by consensus organizations and were unable to identify any other measures on this topic endorsed by a consensus organization, and therefore, the ( printed page 94400) exception in section 1861(kkk)(7)(C)(ii) of the Act applies.
As noted above, the Screen Positive Rate for SDOH measure has been adopted in four other CMS quality reporting programs (Hospital IQR Program, IPFQR Program, PCHQR Program, and ESRD QIP), and this measure establishes an important foundation to prioritize the achievement of health equity among outpatient settings. Additionally, we intend to conduct outreach and education to share information about the Screen Positive Rate for SDOH measure rates in conjunction with public reporting to prevent potential misinterpretation of the measure.
Comment: Several commenters that did not support adoption of the Screen Positive Rate for SDOH measure raised concern with the burden associated with data collection and operational challenges associated with addressing needs identified through screening. A few commenters urged CMS to consider the staff burden and challenges experienced by different provider and facility types. A few commenters expressed concern about the administrative burden this may place on outpatient settings, particularly those that are under resourced, without accompanying reimbursement. A commenter expressed concern about the lag time between reporting or performance periods and reimbursement adjustments. A commenter suggested that CMS adjust programmatic requirements for under-resourced providers to ensure that health equity gaps could feasibly be addressed.
For the ASCQR Program, a few commenters did not support the Screen Positive Rate for SDOH measure and raised concerns about interoperability challenges, noting that a large percentage of ASCs do not have an EHR. Therefore, they noted that those healthcare facilities would have to acquire EHR technology to improve patient care coordination and then turn their attention to SDOH data collection.
Response: We thank commenters for their feedback. We acknowledge commenters' concerns regarding burden; however, achieving health equity is an issue that deserves serious focus and rapid action development. Additionally, as outlined in the CY 2025 OPPS/ASC proposed rule (89 FR 59517, 89 FR 59523, and 89 FR 59527), the estimated burden for HOPDs, REHs, and ASCs to report this measure is estimated to be about 10 minutes annually. Considering this burden estimate is a reasonable expectation for quality data submission, we do not agree it is necessary to provide reimbursements for the time taken to report this measure. We view this measure as a first step towards development of a long-term strategy to integrate social drivers of health data into healthcare facility quality performance measurement as part of our broader commitment to health equity. We will continue to take comments, concerns, and suggestions into consideration as appropriate in any future rulemaking.
Regarding the ASCQR Program, we acknowledge that some ASCs may face challenges adopting EHR technology. We note that the ASCQR Program is a pay-for-reporting program, not a pay-for-performance program, and ASCs that do not have EHR technology can still satisfy reporting of both SDOH measures as the measures does not require an EHR for screening. As noted in the CY 2025 OPPS/ASC proposed rule (89 FR 59447), in alignment with the Hospital IQR Program, we proposed that HOPDs, REHs, and ASCs could confirm the current status of any previously reported HRSNs in another care setting and inquire about others not previously reported, in lieu of re-screening a patient within the reporting period.
We note that this measure was designed to be implemented with or without an EHR. However, we also encourage ASCs to allocate resources towards EHR adoption if they are able. Adoption of EHR technology within ASCs will lead to more positive outcomes, as EHR usage has been shown to have positive impacts on care quality, coordination, efficiency, patient care safety, documentation accuracy, and cost reduction.[390] Although initial EHR adoption has associated costs, it is expected that, over time, ASCs will experience a decrease in financial and operational burden. By purchasing an EHR, an ASC will save costs previously spent on maintaining and managing patient charts.[391] EHRs additionally decrease duplicative testing and interventions. Furthermore, EHRs ensure all required patient information is included in the operative note, leading to accurate reimbursements.[392] We continue to encourage ASCs to consider EHR adoption to streamline documentation of patient health information, including demographic characteristics and social determinants of health, which will support ASCs in collecting and utilizing data to identify critical equity gaps, implementing plans to address these gaps, and ensuring that resources are dedicated toward addressing health equity initiatives.
Comment: A few commenters that did not support adoption of the Screen Positive Rate for SDOH measure expressed the concern that implementation of the measure raised expectations for providers to not only collect SDOH data but also be equipped to aid individuals in finding available community services. Some commenters expressed concern that this measure was not actionable and does not adequately match patients with resources. A few commenters argued the measure was inappropriate for the outpatient setting due to the lack of follow-up with patients. Several commenters expressed concern that small or rural outpatient settings may not have the community resources to assist patients.
For the ASCQR Program, some commenters expressed concern that screenings could erode the provider-patient relationship if SDOHs are assessed, but the provider does not have a robust system in place to address the patient's needs once identified. A commenter expressed concerns with the requirement to screen for interpersonal safety in the context of outpatient surgical care, noting that this could jeopardize patient privacy and carry risk of patient harm.
Response: We thank the commenters for their input. We appreciate the concerns noted and we have given them significant consideration in development of the proposal to adopt the Screen Positive Rate for SDOH measure. Regarding commenters' concerns with regard to potential erosion of the provider-patient relationship in the ASCQR Program, we first note that patients may decline participation, and second, reiterate that the intent of the measure is to promote adoption of HRSN screening by providers as part of a larger long-term strategy to improve patient outcomes and eliminate health equity gaps. As we noted in the CY 2025 OPPS/ASC proposed rule (89 FR 59448), the measure is intended to provide information to providers on the level of unmet social needs among patients served. Initiating data collection remains imperative and such data collection has already allowed some entities to reallocate resources to address HRSNs in disproportionately affected populations. We note this data collection is valuable in the continuing development of the CMS health equity quality measurement efforts and our aims to acknowledge the impact of HRSNs on healthcare access, utilization, ( printed page 94401) outcomes, and costs. The ability of medical providers to contextualize the interaction between HRSNs and poor health outcomes could strengthen referrals to and partnerships with community-based service providers for patients with the most complex needs.
Regarding the interpersonal safety domain and appropriateness of screening in the ASC environment, we note that with a year of voluntary reporting, there is time for ASCs to train staff in the collection of data for HRSNs. Additionally, there are a variety of methods, other than audible methods, to screen for HRSNs that increase privacy, including written or digital methods, and electronically through a patient portal or on tablets. We advise ASCs to ensure that patients feel safe answering questions and to further remind patients that they may opt out of the screening for any reason. We note that the measure specifications as proposed excludes patients: (1) who opt-out of screening; and (2) are themselves unable to complete the screening and have no legal guardian or caregiver able to do so on the patient's behalf (89 FR 59447).
Comment: A few commenters that did not support adoption of the Screen Positive Rate for SDOH measure expressed concern regarding interpretation and potential penalization of measure reporting, especially for REHs. A few commenters expressed concern that the measure is not standardized or validated, potentially leading to inappropriate comparisons among outpatient settings. A few commenters noted that the measure specifications were vague and needed additional clarification. A commenter was also concerned that facilities with higher screen positive rates would be penalized.
For the ASCQR Program, a commenter requested that CMS maintain the measure as voluntary for the ASCQR Program as CMS continues to work through technical reporting challenges, such as publishing guidance on what constitutes a “positive” screen, with providers already reporting the measure in other quality reporting programs.
Response: We appreciate the commenters' concerns. The measure is intended to provide information to providers on the level of unmet need among their patients and potentially in the community. To prevent potential misinterpretation of the measure rates, explanatory information will be included for the Screen Positive Rate for SDOH measure rates in conjunction with public reporting. We reiterate that the objective of this measure is to incentivize collection of these data to help identify patient needs and where resources constraints exist and is not intended for comparison between providers. Public reporting of healthcare quality data promotes transparency in the delivery of care by increasing the involvement of leadership in healthcare quality improvement, creating a sense of accountability, helping to focus organizational priorities, and providing a means of delivering important healthcare information to consumers and patient advocates. When the measure rates are publicly reported, the data will be accompanied by detailed explanations to understand what the measure means and how to interpret the rates in the appropriate context.
Reporting a separate screen positive rate for each of the five HRSNs will provide important information to healthcare facilities, the communities that they serve, and policy makers. Because different community-based resources are appropriate to address each of the five HRSNs, reporting each of these rates separately will provide reliable and valid information to identify which communities are most in need of which resources to better enable support in addressing the most prevalent HRSNs. Additionally, as we stated in the CY 2025 OPPS/ASC proposed rule (89 FR 59448), this measure is not for comparison between healthcare facilities. Rather, the objective of this measure is to incentivize collection of these data to help identify patient needs and where resources constraints exist. Moreover, when the measure rates are publicly reported, the data will be accompanied by detailed explanations to understand what the measure means and how to interpret the rates in the appropriate context.
Regarding commenters' concerns that these measures would penalize providers, we remind readers that the Hospital OQR and ASCQR Programs are pay-for-reporting programs. Participants are not scored, and their payments are not affected, based on their performance on measures; participants are only penalized for failure to submit required data on quality measures as specified. Additionally, the REHQR Program does not include a financial incentive or penalty for REHs.
As we noted in the CY 2025 OPPS/ASC proposed rule (89 FR 59447), one example of a screening tool that healthcare facilities could consider using is the AHC HRSN Screening Tool, which providers used in the AHC Model to screen for HRSNs in their Medicare, Medicaid, and dually eligible beneficiary populations. We have tested the AHC HRSN Screening Tool across many care delivery sites in diverse geographic locations and determined that it demonstrates evidence of both reliability and validity. The AHC HRSN Screening Tool can be implemented in a variety of healthcare settings, including HOPDs, REHs, and ASCs. While the AHC Model focused on HRSNs among community-dwelling Medicare and Medicaid beneficiaries, the AHC HRSN Screening Tool can be used to screen patients with any insurance status or type, including commercially insured and uninsured individuals.
We acknowledge that this measure was initially developed for the general acute care setting. While we recognize the value of measures undergoing testing and evaluation of validity and feasibility in the setting for which they are being adopted, given the urgency of achieving health equity, it is important to implement this measure as soon as feasible. We note that health equity is a critical topic for patients and that there are high levels of health disparities experienced by the ASC patient population, for example. Measure specifications are posted at: https://qualitynet.cms.gov/outpatient/oqr/proposedmeasures2#tab2 and https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 and may be moved to other pages on QualityNet upon publication of the final rule. We will monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if improvements are identified through this process.
Regarding comments about the ASCQR Program, for additional information on what constitutes a “positive” screen, we refer readers to discussion of this measure in section XIV.B.3 of this final rule with comment period and the CY 2025 OPPS/ASC proposed rule (89 FR 59448 through 59449). We also refer readers to a Frequently Asked Questions document for the SDOH measures developed for the Hospital IQR Program, which can be found at: https://www.qualityreportingcenter.com/globalassets/2024/04/iqr/17.-sdoh-measure--faqs_april-2024_vfinal508.pdf. We intend to conduct outreach and education to share information about the Screen Positive Rate for SDOH measure rates in conjunction with public reporting. We reiterate that with a year of voluntary reporting, there is time for ASCs to train staff in the collection of data for HRSNs and that there are a variety of methods to screen for HRSNs including electronically through a patient portal or via tablets. ( printed page 94402)
Comment: Several commenters expressed that publicly reporting these data may lead to inaccurate perceptions of the quality of care in outpatient settings, particularly in the absence of any context. A commenter added that interpretation of this measure is difficult. Therefore, these commenters recommend that the measure not be publicly reported and instead be used for organizational quality improvement.
Response: We appreciate the commenters' concerns. The measure is intended to provide information on the level of unmet need among patients and potentially in the community, and not for comparison between healthcare facilities (89 FR 59448). The objective of this measure is to incentivize collection of these data to help identify patient needs and determine where resource constraints exist. Public reporting of healthcare quality data promotes transparency in the delivery of care by increasing the involvement of leadership in healthcare quality improvement, creating a sense of accountability, helping to focus organizational priorities, and providing a means of delivering important healthcare information to consumers and patient advocates. When the measure rates for the Screen Positive Rate for SDOH measure are publicly reported, the data will be accompanied by detailed explanations to understand what the measure means and how to interpret the rates in the appropriate context. We also intend to conduct outreach and education to share information about the Screen Positive Rate for SDOH measure in conjunction with public reporting.
Comment: Several commenters requested that CMS delay mandatory implementation of the measure until the CY 2027 reporting period, with mandatory reporting beginning in CY 2029, which would give providers at least 2 years to build needed infrastructure and community networks, and effectively collect and analyze necessary information without compromising care quality.
For the Hospital OQR Program, a commenter noted that the measure would require significant time for participating hospitals to integrate the data collection and create clinical workflows to capture and improve patient care, particularly if they intend to integrate the measure into the HOPD's EHR system, which is often not the same vendor as what is used in the inpatient setting.
For the ASCQR Program, a commenter noted challenges with the implementation timeline for the Screen Positive Rate for SDOH measure because it does not account for mandatory reporting of the OAS CAHPS Survey measure in the ASCQR Program, which begins in 2025, and would thus not allow sufficient time for work that would need to be done for the Screening for SDOH measure, if it were to include screening for interpersonal safety.
Response: We thank the commenters for their recommendation. Collecting these data will help healthcare facilities improve care coordination and use of community resources to better deliver patient-centered care. The proposed voluntary reporting period provides a sufficient transition period for healthcare facilities to select and integrate screening tools into their clinical workflow processes (89 FR 59448), as we have permitted flexibility in tool selection and have already adopted this measure in other care settings to which these settings may utilize as resources for best practices. Regarding interoperability, we encourage healthcare facilities to configure their health IT in a manner that uses health IT vocabulary standards that enable interoperability of this data across systems. We note that the use of certified health IT that uses standardized exchange protocols and vocabularies can support the capture and exchange of HRSN information in an interoperable fashion, because standards increase the likelihood that facilities use the same encoding and exchange protocols for data transfer. These data can then be shared across the care continuum to support coordinated care.
Regarding the ASCQR Program, we reiterate that with a year of voluntary reporting, there is time for ASCs to train staff in the collection of data for HRSNs and that there are a variety of methods to screen for HRSNs including electronically through a patient portal or via tablets. We will consider feedback about timeline, OAS CAHPS Survey overlap, and reporting as appropriate, in future potential rulemaking, should issues arise as we monitor implementation of this measure in the ASCQR Program.
Comment: A few commenters recommended we facilitate the development of partnerships as well as sharing HRSN information with community health workers and community-based organizations to help patients with basic resource needs. A commenter recommended we ensure alignment with Project Gravity standards and promote interoperability standards for data collection. A commenter recommended harmonization of drivers of health assessment approaches with the National Committee for Quality Assurance (NCQA). The commenter also recommended we ensure screening tools are approved by the Social Innovation and Research Evaluation Network (SIREN) and with the Core Quality Measures Collaborative (CQMC) to promote alignment across public and private payers. A commenter recommended improving the measure's interoperability to avoid administrative burden and duplicative efforts.
Response: We thank the commenters for their recommendations. We are working with our partners to ensure standardization and alignment where possible. The measure aligns with NCQA's Healthcare Effectiveness Data and Information Set (HEDIS®) measure, Social Need Screening and Intervention (SNS-E), by focusing on food insecurity, housing instability, and transportation needs. We also note that the five domains covered by this measure are included within the “social risk domains” of the Gravity Project. We also note ongoing reevaluation efforts that aim to improve the Screening for SDOH and Screen Positive Rate for SDOH measures through the development of the Addressing Social Needs (ASN) electronic clinical quality measure (eCQM) for introduction into the Hospital IQR Program and eventually the MIPS and Hospital OQR Programs.[393] As noted in the CY 2025 OPPS/ASC proposed rule (89 FR 59447), we refer readers to evidence based resources like the SIREN website which includes comprehensive information about the most widely used HRSN screening tools. We encourage healthcare facilities to consider digital standardized screening tools. We refer readers to the FY 2023 IPPS/LTCH PPS final rule (87 FR 49207 through 49208), where we discuss how the use of certified health information technology (IT), including but not limited to CEHRT, can support capture of HRSN information in a standardized, interoperable fashion. We also encourage healthcare facilities to learn about the United States Core Data for Interoperability (USCDI) standard used in certified health IT and how this standard can support interoperable exchange of health and HRSN assessment data. We wish to reiterate that our approach to developing health equity measures is incremental and will evolve over time to capture health ( printed page 94403) equity outcomes and ensure alignment with our partners.
Comment: A few commenters recommended other factors to include in the measure to make it more meaningful, including demographics across communities other than race and ethnicity (such as age, gender, health literacy, etc.), accessibility of services, as well as more granularity in reporting to identify the patients with the greatest need for follow-up referrals or to vary interventions based on patient need. A few commenters urged CMS to evaluate ways to better support providers and community-based organizations in addressing HRSNs, including through Innovation Center model tests and flexibilities in the Medicare statute.
For the ASCQR Program, a few commenters suggested that a measure be developed to assess whether an ASC's quality program addresses equitable care and outcomes for the surgical services provided at the center. A commenter suggested that CMS should develop claims-based measures that evaluate surgical care outcomes stratified by patient characteristics such as race and ethnicity, SES, dual eligibility and other related factors.
Response: We thank the commenters for their recommendations and will take them into consideration as appropriate as we monitor implementation of the Screen Positive Rate for SDOH measure in our quality reporting programs.
After consideration of the public comments we received, we are finalizing the adoption of the Screen Positive Rate for SDOH measure as proposed in the Hospital OQR, REHQR and ASCQR Programs.
C. Modification of the Immediate Measure Removal Policy for the Hospital Outpatient Quality Reporting (OQR) and Ambulatory Surgical Center Quality Reporting (ASCQR) Programs Beginning With CY 2025
In the CY 2010 OPPS/ASC final rule with comment period (74 FR 60634 through 60635) and the CY 2015 OPPS/ASC final rule with comment period (79 FR 66967 through 66968), we finalized a process for immediate removal of Hospital OQR and ASCQR Program measures, respectively, based on evidence that the continued use of a measure as specified raises patient safety concerns. We refer readers to our regulations at 42 CFR 419.46(i)(2) for the Hospital OQR Program and 42 CFR 416.320(b) for the ACSQR Program for the codification of these immediate measure removal policies.
When there is evidence that continued use of a measure potentially raises patient safety concerns, we believe that immediate action should be taken to discontinue collection of the measure to not encourage potentially harmful practices. We also believe that seeking public input on the removal of such measures increases the public's voice in decision-making and increases transparency. We noted this in the CY 2024 OPPS/ASC final rule (88 FR 82052), where we finalized an immediate measure suspension policy for the REHQR Program in lieu of an immediate measure removal policy. The REHQR Program's immediate measure suspension policy more appropriately provides that, in cases where we believe that a measure raises patient safety concerns, we will suspend the measure's use in the program, instead of immediately removing the measure, until its potential removal undergoes the standard rulemaking process (88 FR 82052).
We believe that our rationale for finalizing the immediate measure suspension policy in the REHQR Program (88 FR 82052) also applies to the Hospital OQR and ASCQR Programs. On this basis, in the CY 2025 OPPS/ASC proposed rule, we proposed to modify the immediate measure removal policies in the Hospital OQR and ASCQR Programs so that they are more appropriately referred to as immediate measure suspension policies beginning with CY 2025.
Under the proposed immediate measure suspension policy in the Hospital OQR or ASCQR Programs, in cases where we determine there is evidence that the collection and reporting of a measure raises potential patient safety concerns, we would suspend the measure from the program (as applicable) until potential removal can be proposed through the rulemaking process. We will notify the healthcare facility (HOPDs or ASCs, as applicable) and the public of the decision to suspend the measure through standard communication channels, including, but not limited to, program-specific listservs and program guidance currently housed on a CMS-designated website. We would then address the suspension and propose policies regarding any such suspended measure in the next feasible rulemaking cycle.
In the CY 2025 OPPS/ASC proposed rule, we also proposed to revise the Hospital OQR Program regulatory text at § 419.46(i)(2) and the ASCQR Program regulatory text at § 416.320(b) to codify the immediate measure suspension policy. We further proposed to clarify the standard for immediate measure suspension in these regulatory texts by revising references to patient safety concerns raised by “continued use of a measure as specified” to patient safety concerns raised by “collection and reporting activities related to a quality measure”.
We invited public comment on these proposals.
Comment: Several commenters expressed broad support of CMS's proposal to modify the immediate measure removal policy to an immediate measure suspension policy for the Hospital OQR and ASCQR Programs, stating that the revised policy promotes transparency and allows for thorough assessment of a measure prior to its potential removal. A few commenters supported soliciting public input through rulemaking prior to removal, and one commenter specifically noted that this could result in modifications to a measure rather than permanent removal.
Response: We thank commenters for their support of our proposal to immediately suspend a measure, instead of removing it, when potential patient safety concerns arise. As previously noted, we would suspend the use of such a measure until removal, modification, or retention could be proposed and finalized through the rulemaking process. We agree with the commenters that this revised policy would enhance transparency and increase public involvement in decision-making regarding permanent action for suspended measures.
Comment: A commenter supported finalizing the immediate measure suspension policy for the ASCQR Program, stating their belief that the proposed policy will help address potential safety concerns associated with screening for interpersonal safety as part of the Screening for SDOH measure.
Response: We thank the commenter for their support of our proposal, and we acknowledge their concern regarding possible patient safety issues with the Screening for SDOH measure. We will monitor the impacts of this measure on patient safety, as we do with all measures, and in cases where we determine there is evidence that the collection and reporting of a measure raises patient safety concerns, we will suspend the measure from the program (as applicable) until potential removal can be proposed through the rulemaking process.
Comment: A commenter did not support the proposal and urged CMS to utilize the rulemaking process prior to suspending a measure. If the proposal is finalized, the commenter urged CMS to limit suspension to public reporting while continuing data collection and measure calculation to assess whether a ( printed page 94404) lack of transparency results in diminished performance for the measure. Additionally, the commenter suggested that CMS focus on improving and developing measures rather than removing them.
Response: We thank the commenter for their feedback. However, we note that patient safety is a high priority area, as underscored by our National Quality Strategy goal to achieve zero preventable harm. When a measure raises potential patient safety concerns, immediate action must be taken to halt data collection and reporting activities. Limiting the suspension to public reporting alone would not accomplish our objective of preventing patient harm. Public reporting does not occur until after the performance period, whereas data collection is continuous. Immediate suspension of both data collection and reporting ensures that potentially harmful actions do not continue in between rulemaking periods and that appropriate modification or removal can then be proposed and finalized in a subsequent rulemaking cycle. We acknowledge the commenter's recommendation to assess whether a lack of transparency results in diminished performance. However, when a measure presents a potential safety risk, the priority is eliminating any risks to patient harm.
While we agree that the Hospital OQR and ASCQR Programs should prioritize retaining and adopting meaningful, high-quality measures, patient safety is an urgent issue and measures that result in potential safety risks should be thoroughly evaluated and considered for potential removal or modification from the programs. This approach allows us to safeguard patients while still ensuring transparency and public participation in decision-making.
We also reiterate that this is an amendment to a currently adopted policy in which a measure is immediately removed if there are patient safety concerns. This revised policy would allow for appropriate modification or retention of a measure over removal, thus supporting the preservation of a measure set that ensures safe, quality care.
Comment: A commenter recommended that once a measure is suspended, hospitals should not be penalized for non-compliance with the requirements of the suspended measure in the subsequent payment determination year. Another commenter suggested expanding the immediate measure suspension policy to the REHQR Program.
Response: We appreciate the commenters' feedback, and we wish to clarify that, under the immediate measure suspension policy we proposed, once a measure is suspended, data collection and reporting would cease until permanent action could be determined in a subsequent rulemaking cycle. Entities would not be penalized for non-compliance with the suspended measure as the requirement to collect and report data would not be in effect. This approach ensures that any potentially harmful actions related to the measure are halted until permanent action regarding the suspended measure is determined. We also wish to clarify that this policy was previously finalized for the REHQR Program in the CY 2024 OPPS/ASC final rule (88 FR 82052).
After consideration of the public comments we received, we are finalizing immediate measure suspension policies for the Hospital OQR and ASCQR Programs, as proposed. We are codifying these policies at § 419.46(i)(2) for the Hospital OQR Program and at § 416.320(b) for the ASCQR Program, as proposed.
XV. Hospital Outpatient Quality Reporting (OQR) Program
A. Background and Statutory Authority
The Hospital Outpatient Quality Reporting (OQR) Program is a pay-for-reporting program intended to improve the quality of care provided to Medicare beneficiaries, facilitate public transparency, and ensure accountability of hospital outpatient departments (HOPDs). Section 1833(t)(17)(A) of the Social Security Act (the Act) states that subsection (d) hospitals (as defined under section 1886(d)(1)(B) of the Act) that do not submit data required for measures selected with respect to such a year, in the form and manner required by the Secretary, will incur a 2.0 percentage point reduction to their annual Outpatient Department (OPD) fee schedule increase factor.
We refer readers to the CY 2011 OPPS/ASC final rule (75 FR 72064 through 72065) for a detailed discussion of the statutory history of the Hospital OQR Program, as well as program requirements codified at 42 CFR 419.46, and to the CY 2024 OPPS/ASC final rule for information regarding the program's regulatory history (88 FR 81961 through 82012).
1. Previously Finalized Program Measure Set Beginning With the CY 2027 Payment Determination
Table 162 summarizes the previously finalized Hospital OQR Program measures beginning with the CY 2027 payment determination:
( printed page 94405)B. Program Measure Set Policies
1. Measure Retention
We refer readers to § 419.46(i)(1) and the CY 2013 OPPS/ASC final rule (77 FR 68471) for our policies regarding measure retention. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
2. Measure Suspension or Removal
We refer readers to § 419.46(i)(2) and (3) and the CY 2013 OPPS/ASC final rule (77 FR 68472 and 68473) for our program policies regarding: (1) general measure removal, suspension, and replacement; and (2) immediate measure removal.
We refer readers to section XIV.C of this final rule with comment period for the discussion of public comments received and our subsequent decision regarding our cross-program proposal to modify the immediate removal policy for adopted Hospital OQR Program measures.
3. Measure Adoption
We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81973) for a discussion of the statutory requirements and our considerations for adopting quality measures under the Hospital OQR Program. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
C. Program Measure Updates
1. Adoption of New Measures for the Hospital OQR Program Measure Set
a. Adoption of the Health Equity Measures in the Hospital OQR Program
We refer readers to sections XIV.B.1, XIV.B.2, and XIV.B.3 of the final rule with comment period, respectively, for our cross-program proposals and discussion of public comments received to adopt the following measures in the Hospital OQR Program: (1) the Hospital Commitment to Health Equity (HCHE) measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting ( printed page 94406) period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period and mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination.
b. Adoption of the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM) Beginning With Voluntary Reporting for the CY 2026 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2027 Reporting Period/CY 2029 Payment Determination
(1) Background
Recent studies have shown that compared to inpatient settings, outpatient settings are associated with worse patient understanding and lower patient activation (that is, an individual's understanding, competence, and willingness to participate in care decisions during their recovery), indicating an area for quality of care improvement.[394 395 396] One study found that providers in the inpatient setting provided more complete discharge instructions and end-of-visit summaries to patients when compared to providers in the ambulatory setting, including continuing medication names and instructions (96 percent vs. 40 percent), new medication names and instructions (99 percent vs. 29 percent), and pending diagnostic test names and instructions (90 percent vs. 61 percent).[397] A lack of understanding of recovery information [398] and other aspects of health literacy have been linked to poor adherence to treatment, decreased patient safety, increased return to the emergency department (ED), lower levels of patient satisfaction, and disproportionate effects on patients with limited English proficiency and patients over age 65, who face additional barriers and recovery issues after their receipt of a hospital outpatient service.[399 400] Reduced patient engagement and a deficiency in detailed discharge information in the inpatient setting were also associated with a higher risk of readmissions to an inpatient setting.[401] Research indicates that information that is simpler to read and more complete has been associated with fewer follow-up calls to providers as well as less frequent hospital readmissions.[402 403]
(2) Measure Overview
The Information Transfer PRO-PM aims to assess the level of clear, personalized recovery information provided to patients aged 18-years or older who had surgery or a procedure at an HOPD. The measure reports the average score of a patient's ratings on a three-domain, 9-item survey [404] to evaluate the clarity of the clinical information patients are given before, during, and after an outpatient surgery or procedure. The survey covers three domains for patients or their caregivers to rate the clarity of information received regarding their post-discharge [405] recovery: applicability to patient needs, medication, and daily activities. The applicability to patient needs domain assesses whether the recovery information considered a patient's health needs and personal circumstances. The medications domain examines the clarity of medication information provided, specifically guidance on taking new medications, potential side effects, and discontinuing medication. The daily activities domain assesses the clarity of guidelines around diet, physical activity, returning to work, and driving. Results from the survey provide hospitals with patient reported outcome (PRO) data designed to assess communication efforts and enable hospitals to reduce the risk of patient harm that may occur if the patient does not fully understand the recovery information.
This measure addresses the priority area stated in our Meaningful Measures Framework of adopting high-quality measures that focus on person-centered care.[406] Additionally, the Information Transfer PRO-PM supports the National Quality Strategy goal of equity and engagement by engaging individuals to become partners in their care and ensuring that individuals and caregivers have the information needed to make the best choices for their health.[407]
Pilot testing conducted by the measure developer in 26 HOPDs in five states demonstrated that the measure is reliable and meaningful.[408 409] The measure developer assessed reliability of the measure using the Cronbach alpha score [410] to determine whether the nine survey questions reliably measured the same underlying characteristic; that is, the clarity and applicability of recovery instructions. The Cronbach alpha score, which compares the amount of shared variance, or covariance, among the instrument items ( printed page 94407) to the total variance, indicated that the survey items are reliable because they reflect a high level of covariance relative to the total variance.[411] Additionally, the measure developer found the performance scores among facilities in the pilot study to be moderately reliable using a signal-to-noise ratio, which estimated variance among facilities and facility specific errors to determine the extent to which variance in facility scores can be attributed to variance in actual performance.[412]
To assess meaningfulness, the measure developer asked members of a Patient and Family Engagement (PFE) Work Group and a Technical Expert Panel (TEP) to vote on the measure's ability to distinguish between good and poor quality of care at measured facilities.[413 414] All of the patients from the PFE Work Group and 80 percent of the TEP panel members who participated in the vote agreed that the measure could distinguish between good and poor quality of care.[415] We refer readers to https://p4qm.org/measures/4210 for more information about the feasibility, scientific acceptability, meaningfulness, and validity of the Information Transfer PRO-PM.
As previously stated in the CY 2024 OPPS/ASC final rule (88 FR 81985), while we acknowledge that PRO-PMs require providers to integrate data collection into clinical information systems, this integration provides an important opportunity for patient-reported outcomes to inform clinical decision-making and benefits patients by engaging them in discussions about potential outcomes. The testing of this measure by the measure developer, which included interviews with clinicians, nurses, quality improvement officers, and data administrators in HOPDs, indicated that the increased burden on HOPDs would be minimal because the data would be collected and reported electronically by administrative staff and quality officers engaged in data sharing activities, outside of the clinical workflow, before being integrated into a clinical information system. Additionally, testing indicated that the increased burden on respondents would be minimal and contribute minimally to patient survey fatigue because the survey is easily understood and consists of only nine questions administered electronically,[416] presenting a low burden for completion.
(3) Pre-Rulemaking Measure Review
Under the PRMR process, the Hospital Recommendation Group reviewed and voted on the Information Transfer PRO-PM [417] during their meeting on January 18-19, 2024.[418] The voting results for the Information Transfer PRO-PM measure for the Hospital OQR Program were “recommend with conditions”. The condition was that the survey be administered at the time of the surgery or procedure so there is no conflict with other measured pain and function outcomes to improve response rates.[419] We have taken into account the condition to administer the survey at the time of the surgery or procedure; however, we have determined that allowing time after the surgery or procedure before administration of the survey is important to limit the possibility that the patient's responses are influenced by time-dependent variables related to proximity to the surgery or procedure, such as medications that could affect comprehension, fatigue, or acute pain. In addition, administering the survey more than 1 day but less than 7 days post-procedure mitigates overlap of the initial administration and survey reminder of the OAS CAHPS, which is administered on the first day post-procedure and then followed up at 14 days.[420]
(4) CBE Endorsement
We submitted the Information Transfer PRO-PM to the CBE for endorsement review in the Fall 2023 cycle (CBE #4210), and the CBE endorsed the measure on March 18, 2024.[421]
(5) Data Collection, Submission, and Reporting
(a) Data Collection
(i) Data Sources
In the CY 2025 OPPS/ASC proposed rule (89 FR 59453 through 59454), we proposed that the Information Transfer PRO-PM would be calculated based on PRO data collected by HOPDs directly or through their authorized third-party vendors through a web-based survey instrument distributed to patients or their caregivers.
( printed page 94408)We also proposed that the survey would be administered 2-to-7 days post-procedure or surgery, based on evidence that the most common time period for patients to be delivered a survey is within 0-48 hours post-procedure or surgery at a HOPD (n=6),[422 423 424 425 426 427] while other time periods include within 2 weeks post-procedure or surgery (n=4),[428 429 430 431] 1 week post-procedure or surgery, (n=3) [432 433 434] or 90 days post-procedure or surgery (n=1).[435] We proposed that the survey would be administered not less than 2 days post-procedure or surgery because we have determined, as discussed above, that allowing time after the surgery or procedure before administration of the survey will limit the possibility that the patient's responses are influenced by time-dependent variables related to proximity to the surgery or procedure, such as medications that could affect comprehension, fatigue, or acute pain. We proposed that the survey would be administered no later than 7 days post-procedure or surgery because this timeframe may be more appropriate for patient reporting of specific events than longer time periods.[436 437] In pilot testing, patients were sent a reminder to complete the survey 7 days after receipt. The survey remained open until pilot testing was completed, with the mean length of time between the procedure date to the survey response date being 65 days, or approximately 2 months. We therefore proposed a 65-day window for patient response.
The survey has been tested and reliability determined in English and Spanish, and the survey can be completed using a translator, proxy, or caregiver.
(ii) Measure Specifications
The measure numerator is the sum of all individual scores a HOPD receives from eligible respondents, which could be patients or caregivers. Individual scores are calculated using a top-box approach; each individual score is calculated for each respondent by taking the sum of items for which the respondent gave the most positive response (“Yes” or “Very Clear”) and dividing by the number of items the respondent deemed applicable to their procedure or surgery. Applicable items are calculated by subtracting the sum of items for which the respondent selected “Does not apply” from the total number of survey items (nine).[438]
The measure denominator is the total number of patients 18 years or older who had a procedure or surgery in an HOPD, left the HOPD alive, and responded to the survey.[439 440] Only fully completed surveys are included in the measure calculation.
The intent of the measure is to encourage HOPDs to provide individualized recovery instructions regardless of the patient's unique characteristics; therefore, there is no need for risk-adjustment. For additional details regarding the measure specifications, we refer readers to our QualityNet website.[441]
(b) Data Submission and Reporting
In the CY 2025 OPPS/ASC proposed rule (89 FR 59492 through 59454), we proposed to adopt the Information Transfer PRO-PM as a voluntary measure for the CY 2026 reporting period followed by mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination. We would utilize the voluntary period to monitor the implementation and operationalization of the measure.
We refer readers to section XV.E.2.c of this final rule with comment period for a discussion of the Information Transfer PRO-PM form, manner, and timing of data submission and reporting requirements.
We invited public comment on the proposal.
Comment: Many commenters supported adopting the Information Transfer PRO-PM into the Hospital OQR Program. Commenters noted that this measure addresses an important measurement area (care coordination) and is an effective means of collecting patient experience data. A few of these commenters noted the importance of patients receiving clear, personalized recovery information and engaging in a shared decision-making process with their healthcare provider. A few commenters further emphasized that this measure may increase patient satisfaction and trust in their healthcare providers, as patients feel more informed and supported during their recovery. Commenters also stated that this measure can provide valuable feedback to clinicians about their communication practices, highlighting areas for improvement. A commenter applauded CMS for including caregivers as eligible respondents for providing survey feedback, stating that caregivers play a vital role in a patient's post-procedure care. Another commenter expressed gratitude for the time CMS has allotted for hospitals to implement this measure.
Response: We thank commenters for their support. We agree that this measure addresses an important measurement area (care coordination) and is an effective means of collecting patient experience data. We agree with ( printed page 94409) commenters that this measure can provide valuable feedback to clinicians about their communication practices and highlight areas of needed improvement.
Comment: A few commenters supported CMS' proposal and noted that the measure is CBE-endorsed and tested, showing moderate reliability and a reliable Cronbach alpha score.
Response: We thank commenters for their support.
Comment: Several commenters supported CMS' proposal to adopt the Information Transfer PRO-PM citing evidence that poor recovery instructions result in poor outcomes, such as mortality and readmissions. A few of these commenters noted that clear communication between healthcare providers and patients regarding post-procedure recovery is crucial to ensure positive health outcomes and fewer readmissions. A commenter noted that many older patients being discharged from surgery experience post-operative cognitive dysfunction (POCD), which, when added to possible limited English proficiency, literacy, or pre-existing vision or cognitive concerns, can significantly impact a patient's ability to understand and follow post-operative recommendations without additional intervention and instruction. Further, this commenter noted that post-surgical instructions received by occupational therapy (OT) clinics are often incomplete, missing, or confusing to the patient and the OT practitioner, forcing the practitioner to seek additional clarification resulting in delay in care initiation and increased risk of re-injury.
Response: We thank commenters for their support. We agree that clear communication between healthcare providers and patients is crucial to ensure positive health outcomes and fewer readmissions. We further thank commenters for their feedback of how poor post-procedure instructions to patients could adversely impact their ability to receive proper and timely post-procedure care such as physical or occupational therapy.
Comment: A commenter supported CMS' use of PRO-PMs in quality reporting programs because PRO-PMs provide valuable insights into patients' perspectives on their health, quality of life, and functional status. This commenter further noted that PRO-PMs are especially important in the outpatient setting where procedures and patients are typically less complex compared to the inpatient setting. The commenter noted that PRO-PMs offer every patient an opportunity to provide direct feedback about their care, which is valuable when this feedback is typically collected only when measuring a rare or adverse event.
Response: We thank the commenter for their support of the use of PRO-PMs.
Comment: Several commenters expressed concerns with CMS' proposal to adopt the Information Transfer PRO-PM due to the burden on HOPDs to implement the PRO-PM. Commenters expressed concern regarding the challenge of integrating surveys into provider workflows. Commenters expressed concern that HOPDs are currently facing staffing challenges and resource constraints. Commenters stated that the burden of implementing this measure is greater than CMS indicated in the proposed rule and does not account for a facility's time spent strategizing, reviewing and updating policies, building the technology, training staff, and implementing change management processes required for new measures. A commenter expressed concern that the cost of programming the measure into health information technology may not be minimal because the survey would have to be set up as a stand-alone survey instead of being grafted onto an already existing survey, such as OAS CAHPS, and the potential cost of the initial programming and implementation is not acknowledged in the proposed rule. Another commenter expressed concern that ensuring consistency in communication, so all patients receive the same quality and clarity of recovery information, can also be challenging, especially in busy hospital outpatient departments.
Response: We acknowledge that collecting PRO-PM data may involve more burden and initial implementation resources compared to some other types of quality measures. However, we believe the benefit of collecting direct functional improvement information from the patients outweighs the burden. Measuring patient-reported outcomes is an important aspect of patient-centered healthcare and we continue to emphasize, as highlighted in our Meaningful Measures 2.0 Framework, that the patient voice should be elevated across the healthcare system. As also discussed in a recent request for information related to guiding principles for patient-reported outcome measures in Federal models and quality reporting and payment programs, patient-reported outcomes, as captured using PRO-PMs, are critical for the support of person-centered care, as they provide information from the patient or caregiver perspective and offer important information to improve patient-provider communications (89 FR 62043 through 62044).
As we stated in the CY 2024 OPPS/ASC proposed rule (89 FR 59518), we estimate that annually each HOPD would spend 10 minutes (0.167 hours) per response to report the Information Transfer PRO-PM data to CMS, and that annually 498,843,518 patients would be screened if the Information Transfer PRO-PM became mandatory. While PRO-PMs may require providers to integrate data collection into clinical workflows, this integration provides an important opportunity for patient-reported outcomes to inform clinical decision-making and benefits patients by engaging them in discussions about potential outcomes. Specifically, regarding the Information Transfer PRO-PM, as stated in the CY 2025 OPPS/ASC proposed rule, data would be collected and reported electronically by administrative staff and quality officers engaged in data sharing activities, outside of the clinical workflow, before being integrated into a clinical information system (89 FR 59453). Additionally, to provide more flexibility, we are not requiring HOPDs to collect data in a standardized way. HOPDs may use a variety of data collection, storage, and submission approaches, and we encourage HOPDs to use processes best suited to them. Further, a study in the Journal of the American Medical Association found that, when reviewing the annual person-hours and personnel costs across different elements of quality reporting (that is, entering information, reviewing reports, tracking specifications, developing processes, and collecting and validating data) for surveys, the majority of annual person-hours and personnel costs were allocated to reviewing reports.[442] Therefore, the burden of developing processes (such as a facilities' time strategizing, reviewing and updating policies, building technology, training staff, and implementing change management processes) for the Information Transfer PRO-PM should be minimal. Finally, we note that additional actions an HOPD may take in the process of implementing this measure will vary among HOPDs due to, for example, an HOPD's size, services, and current processes and policies in place.
As to commenters' concern regarding ensuring consistency in communication so all patients receive the same quality and clarity of recovery information, we note that the goal of this measure is to assess how well facilities provide clear, ( printed page 94410) personalized recovery information. While we agree that HOPDs are busy, it is important for patients to understand their recovery information and for HOPDs to communicate to each patient in a clear, personalized way.
Comment: Several commenters expressed concerns with CMS' proposal to adopt the Information Transfer PRO-PM due to patient survey burden. Commenters stated that the addition of this patient survey to the Hospital OQR Program would result in patient survey fatigue and decreased patient responses to surveys across the board. A commenter noted that if a patient received a hospital outpatient total knee replacement, the surveys the patient would receive would include: (1) screening for five SDOH domains, (2) pre-operative data collection for the THA/TKA PRO-PM, (3) OAS CAHPS, (4) Information Transfer PRO-PM, and (5) post-operative data collection for the THA/TKA PRO-PM. The commenter further noted that patients would likely also receive additional questionnaires (such as the CAHPS for MIPS survey) from other provider types. A few commenters expressed concern regarding low participation rates due to language barriers, lack of internet access, lack of understanding of or access to technology, and the current timeframe in which the survey is administered.
Response: Our aim is to promote better collection and integration of patients' voices by incorporating Patient Reported Outcome Measures (PROMs) that are embedded into clinical workflow, easy to use, and as minimally burdensome to patients and providers as possible. We are dedicated to improving the quality of care provided to patients, and believe patients are a vital source of information in assessing quality of care provided at a HOPD. Patient feedback from a Technical Expert Panel (TEP), which informed our initial selection and development of this measure, indicated that patients are experiencing survey fatigue in all facets of their lives, and as such preferred shorter surveys in an electronic format.[443] Several TEP members also noted the importance of a quick and easy survey built into the care process as part of a positive patient experience following outpatient surgery.[444]
Regarding screening for five SDOH domains, as finalized in section XIV.B of this final rule with comment period, we recognize that some healthcare facilities may already be screening their patients for HRSNs, and in alignment with the Hospital IQR Program, we proposed that HOPDs could confirm the status of any previously reported HRSNs in another care setting and inquire about others not previously reported, in lieu of re-screening a patient within the reporting period. In addition, if this information has been captured in the EHR in another outpatient setting or the inpatient setting during the same reporting period, we proposed that the HOPD could use that information for purposes of reporting the measure in lieu of screening the patient.
Regarding pre-operative and post-operative data collection for the THA/TKA PRO-PM, the Information Transfer PRO-PM is a nine-item survey offered once to patients who meet the measure denominator specifications and, unlike the THA/TKA PRO-PM, does not require pre-operative and post-operative surveys to evaluate functional improvement following the surgical procedure. Regarding OAS CAHPS, we selected a timeframe for administration of 1 to 7 days post-procedure for the Information Transfer PRO-PM, both to limit the possibility that the patient's responses are influenced by time-dependent variables related to proximity to the surgery or procedure (such as medications that could affect comprehension, fatigue, or acute pain), and to mitigate overlap of the initial administration and survey reminder of the OAS CAHPS, which is administered on the first day post-procedure and then followed up at 14 days.
Regarding concerns about low participation rates due to language barriers, lack of internet access, lack of understanding of or access to technology, and the current timeframe in which the survey is administered, as we note later in this final rule, the Information Transfer PRO-PM is available in English and Spanish. Additionally, in order to ensure that hospitals are administering the survey in the patient's preferred language, hospitals are permitted to use interpretation and translation services. For patients that lack internet access or lack understanding of or access to technology, we note that the Information Transfer PRO-PM can be completed using a translator, proxy, or caregiver.
We anticipate data collection for the Information Transfer PRO-PM to present a low burden to patients thereby fostering receptiveness to survey participation. As we stated in the CY 2024 OPPS/ASC proposed rule (89 FR 59518), we estimate that each patient would require an average of 6 minutes to complete the nine-item survey. We intend to evaluate response rates associated with the Information Transfer PRO-PM and continue to consider ways to reduce patient survey fatigue.
Comment: A few commenters did not support adoption of any additional PROMs, including the Information Transfer PRO-PM, because of the challenges they have recently experienced while implementing the THA/TKA PRO-PM measure, such as the financial burden of implementing that measure and challenges in meeting the current 50 percent reporting threshold for the THA/TKA measure.
Response: We do not believe that the challenges providers may face in implementing the THA/TKA PRO-PM warrant declining to adopt other important PRO-PMs in the Hospital OQR Program. We addressed commenters' concerns regarding implementation of the THA/TKA PRO-PM in the CY 2024 OPPS/ASC final rule (88 FR 81984 through 81986) and maintain that this measure is an important addition to the Hospital OQR Program as the volume of THA and TKA procedures for Medicare beneficiaries aged 65 years and older have been increasing in outpatient settings.
We note that, in comparison to the THA/TKA PRO-PM, the Information Transfer PRO-PM does not have a reporting threshold and thus providers would not be subject to a similar reporting threshold. Additionally, we note that the Information Transfer PRO-PM is a nine-item survey offered to patients who meet the measure denominator specifications as a one-time survey, and that, unlike the THA/TKA PRO-PM, it does not require pre-operative and post-operative surveys to evaluate functional improvement following the surgical procedure. We therefore believe that the financial burden would be reduced in comparison to the THA/TKA PRO-PM which has multiple surveys within the measure.
Comment: A few commenters expressed concerns around the measure testing of the Information Transfer PRO-PM and recommended more testing be done before inclusion into the Hospital OQR Program. A commenter stated that the limited geographic scope of testing might not have captured the diversity of patient experiences across the country. A commenter expressed concern regarding the lack of risk adjustment in the measure because it prevents understanding of which vulnerable ( printed page 94411) patients/populations could benefit from additional education.
Response: We thank the commenters for their feedback on this measure. Testing of the measure results including for risk adjustment need, response rates, and data missingness was conducted and is described in the methodology report.[445] As the outcome for this measure is the patient's rating of the clarity of information about their recovery process, it is important to test for certain patient characteristics that may bias results. Analysis of factors that are and are not correlated with the measure is critical in assessing whether the measure should be risk adjusted. Evaluation of risk adjustment in pilot testing survey results found no statistically significant differences between adjusted and unadjusted result scores. Additionally, non-response bias, which is the likelihood of a patient responding to a survey based on patient characteristics, was also analyzed during pilot testing. Non-responders were significantly younger and more likely to be male, but these factors were not significant toward measure results. We reiterate that this measure was submitted to the CBE by the measure developer for endorsement review (CBE #4210), which includes a review of testing data, measure importance, feasibility, and usability. When submitted for endorsement, a measure is reviewed on its reliability and validity within the context of its intended application, justifying a measure's use in the HOPD setting.[446] This measure was endorsed on March 18, 2024. Furthermore, during measure testing, the measure developer engaged in extensive discussions with clinicians and patient stakeholders regarding the need to risk-adjust. After discussion and analysis of pilot data, risk-adjustment was not added to the measure because it could unintentionally encourage disparate outcomes between different patient populations. The performance differences between the adjusted and unadjusted measures in testing were also minimal.[447]
Comment: Several commenters expressed concern regarding the measure's feasibility as CMS requires a minimum random sample size of 300 completed surveys, which is inconsistent with the minimum responses outlined in the measure specifications. A commenter requested clarification on how to count partially completed surveys, and whether 100 percent of questions need to be completed to meet the 300-survey minimum.
Response: We acknowledge the commenters' concerns regarding the inconsistency of the minimum sample size as stated in the proposed rule (89 FR 59459) and the measure specifications. We intend to update the measure specification manual to reflect the minimum random sample size of 300 completed surveys. A minimum random sample size of 300 completed surveys is recommended for a population of 1,500 to provide a 95 percent confidence interval and a 90 percent confidence interval for a population of over 10,000 by reducing the standard error.[448 449] We also believe it is important for the Information Transfer PRO-PM to be administered to every patient following an outpatient surgery or procedure, as recent studies show outpatient settings are associated with worse patient understanding,[450 451 452] and that a lack of understanding of patient recovery information is associated with worse outcomes, including poor adherence to treatment, decreased patient safety, increased return to the ED, lower levels of patient satisfaction, and disproportionate effects on patients with limited English proficiency and patients over age 65.[453 454] We note that all questions on the survey need to be completed to meet the 300-survey minimum. Partially completed surveys should not be counted towards the 300-survey minimum.
Comment: A commenter expressed concern that safety net hospitals might face difficulty adopting the Information Transfer PRO-PM due to a large, underserved population with low health literacy, and language and cultural differences contributing to lower response rates. The commenter recommended CMS investigate how disadvantaged populations may impact PRO data collection before adopting PRO-PMs in the Hospital OQR Program.
Response: We believe that the measurement of patient-reported outcomes is vital for advancing patient-centered care and improving overall healthcare quality for all patients. We note that one of our CMS National Quality Strategy goals, “Advance Equity and Engagement for All Individuals” seeks to ensure engagement to improve healthcare quality by incorporating individual and community input into strategy and policy. PRO-PMs are unique because they are based on patients' input on their health and the effectiveness of the care they receive, providing a crucial dimension to quality improvement that other measures may not offer. By integrating the Information Transfer PRO-PM into the Hospital OQR Program as a complement to the other outcome measures, CMS can better support patient-centered quality improvement efforts.
While we acknowledge the commenter's concern that safety net hospitals may face lower response rates due to a large, underserved population with low health literacy, in addition to language and cultural differences, the Information Transfer PRO-PM has mitigated these challenges by limiting the survey to nine items and offering the survey in English and Spanish. Additionally, to ensure that hospitals are administering the survey in the patient's preferred language, hospitals ( printed page 94412) are permitted to use interpretation and translation services. Minimizing language barriers and survey duration can improve response rates.[455] As previously noted, testing of the measure results including for risk adjustment need, response rates, and data missingness was conducted and is described in the methodology report.[456] As the outcome for this measure is the patient's rating of the clarity of information about their recovery process, it is important to test for certain patient characteristics that may bias results. Analysis of factors that are and are not correlated with the measure is critical in assessing whether the measure should be risk adjusted. Evaluation of risk adjustment in pilot testing survey results found no statistically significant differences between adjusted and unadjusted result scores. Additionally, non-response bias, the likelihood of a patient responding to a survey based on patient characteristics, was also analyzed during pilot testing. Non-responders were significantly younger and more likely to be male, but these factors were not significant toward measure results. We will continue to monitor trends and evaluate feedback on data collection for the Information Transfer PRO-PM.
Comment: A commenter expressed concerns about CMS' ability to correlate patient understanding with overall patient outcomes, stating that recovery can be influenced by numerous external factors beyond comprehension, and that using data from this measure could lead to misguided conclusions about effectiveness of care.
Response: Usability of the measure was tested during measure development. The measure developer interviewed quality officers of the organizations that participated in pilot testing about the meaningfulness of the performance score. The quality officers indicated that the scores aligned with known issues raised by patients in an open-ended survey and accurately quantified known issues with the post-operative process.[457] Further, as previously noted, the measure developer asked members of a PFE Work Group and a TEP panel to vote on the measure's ability to distinguish between good and poor quality of care at measured facilities.[458 459] All of the patients from the PFE Work Group and 80 percent of the TEP panel members who participated in the vote agreed that the measure could distinguish between good and poor quality of care.[460] Therefore, we do not expect data from this measure to lead to misguided conclusions about effectiveness of care. Additionally, we are not using PRO-PMs such as the Information Transfer PRO-PM to replace outcome measures, but to serve as complementary measures to support patient-centered care.
Comment: Many commenters recommended integrating the Information Transfer PRO-PM into the OAS CAHPS survey, expressing concern regarding the potential for patient confusion, low response rates, and survey fatigue. A commenter stated that OAS CAHPS is experiencing declining response rates over time, citing an 18 percent drop in survey response rates in a seven-year period. A few commenters stated that the Information Transfer PRO-PM survey administration overlaps with the OAS CAHPS survey administration, expressing concern that response rates for both surveys may be affected as patients may experience survey fatigue. A commenter noted that sending multiple surveys would result in survey fatigue and decrease the likelihood of response. A few commenters recommended expanding the survey beyond English and Spanish, stating that it should be made available in the nine languages that are offered for OAS CAHPS.
Response: We considered integrating questions from the Information Transfer PRO-PM survey into the OAS CAHPS survey to reduce the number of surveys a patient could receive regarding their episode of care. However, we decided against doing so as the specifications for administering the OAS CAHPS (such as the survey timing, allowing multiple submission modes, and requiring a CMS-approved vendor) do not align with the Information Transfer PRO-PM. We note that the voting results of the Hospital Recommendation Group under the PRMR process were “recommend with conditions” for the Information Transfer PRO-PM measure for the Hospital OQR Program, and that the condition was to administer the survey at the time of the surgery or procedure so there is no conflict with other measured pain and function outcomes to improve response rates.[461] We considered this condition and decided on the current timeframe for survey administration of more than 1 day but less than 7 days post-procedure to strike a balance between patient recovery and comprehension of the Information Transfer PRO-PM and mitigating overlap with the initial administration of the OAS CAHPS on the first day post-procedure and the survey reminder at 14 days.
We are currently working to expand the survey to additional languages beyond English and Spanish. Additionally, in order to ensure that hospitals are administering the survey in the patient's preferred language, hospitals are permitted to use interpretation and translation services.
Comment: A few commenters recommended CMS build on this measure to better align with patient goal identification and attainment across the episode of care. A commenter recommended broadening PRO-PMs to assess the degree to which the entire care journey is understood and accepted. A commenter recommended adding questions to the medication question category of the Information Transfer PRO-PM to ensure proper measurement regarding the completeness of recovery information, and to account for a patient's understanding of the care plan. A commenter recommended extending patient response periods for the Information Transfer PRO-PM beyond 65 days. Another commenter recommended measurement on pre-procedure screening for patients' communication disorders to help ensure post-operative instructions are understood. A commenter recommended that CMS explore ways to leverage technology to reduce the effort required to participate in the survey, such as administering it online rather than on paper. Another commenter recommended that CMS consider how it can support hospitals in efforts to increase responsiveness. Another commenter recommended that CMS consider implementing a bonus to ( printed page 94413) encourage investment in reporting PRO-PMs.
Response: We thank commenters for their support and recommendations and will take them into consideration as we begin implementing the Information Transfer PRO-PM in the Hospital OQR Program. As previously noted, the Information Transfer PRO-PM can be distributed electronically via text or email by a vendor or individual facility. Regarding the implementation of a bonus to encourage investment in reporting PRO-PMs, we note that the statutory payment provisions for the Hospital OQR Program do not provide for the payment of any bonus.
Comment: Many commenters expressed concern regarding the implementation timeline of the Information Transfer PRO-PM and recommended that CMS delay voluntary reporting or remove the mandatory reporting requirement. A commenter recommended that CMS provide feedback reports during the voluntary period.
Response: Given the importance of patient understanding of recovery instructions, we believe it is important to adopt this PRO-PM in the Hospital OQR Program and begin reporting as soon as possible while also allowing HOPDs time to implement processes for data collection on this measure, which is why we proposed to begin with a voluntary reporting period. Because the measure only uses a nine-item survey with flexibility to HOPDs on the modes of data collection, as well as sampling when reporting the PRO-PM data to CMS, we consider a one-year voluntary reporting period to be sufficient. We will monitor implementation of the measure and carefully consider feedback received during the voluntary reporting period.
Comment: A few commenters requested additional information regarding measure specifications. A commenter requested that CMS disclose the copyright protections on surveys when adopting new PRO-PMs in the future.
Response: We thank commenters for their questions and point them to the technical specification manuals that can be found on the CMS website at: https://qualitynet.cms.gov/outpatient/specifications-manuals. We note that the Information Transfer PRO-PM survey, as well as the THA/TKA PRO-PM survey, is nonproprietary and free to use. We will disclose copyright protections on surveys as necessary.
After consideration of the public comments we received, we are finalizing our proposal to adopt the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery Patient Reported Outcome-Based Performance Measure beginning with voluntary reporting for the CY 2026 reporting period followed by mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination.
2. Measure Removals From the Hospital OQR Program Measure Set
a. Removal of the MRI Lumbar Spine for Low Back Pain Measure Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
In the CY 2009 OPPS/ASC final rule (73 FR 68766), we adopted the MRI Lumbar Spine for Low Back Pain measure beginning with the CY 2010 payment determination. This claims-based measure evaluates the percentage of magnetic resonance imaging (MRI) of the lumbar spine studies for low back pain performed in the outpatient setting where conservative therapy was not attempted prior to the MRI. The MRI Lumbar Spine for Low Back Pain measure was initially endorsed by a consensus-based entity (CBE) in 2008, but endorsement of this measure was removed in 2017 because the measure developer did not submit the measure for review during its designated measure endorsement maintenance cycle.
When we adopted this measure for the Hospital OQR Program, we cited growing concerns about the overuse of imaging services and evidence that a substantial portion of MRIs for low back pain does not lead to any modification of therapy based on MRI results, especially when performed on the first visit prior to any attempt to diagnose or treat the patient through more conservative means (73 FR 68764). Since then, our internal analyses have shown that the measure has maintained stable national performance (excluding the CY 2022 performance period impacted by our COVID-19 exception policies) and low average volumes, indicating limited reliability and capacity to improve the quality of care for patients with reported low back pain. A study in the Journal of the American College of Radiology found that documentation of conditions that fall into the exclusion criteria of the measure increased after implementation, resulting in smaller patient populations and indicating that the measure may not translate to improvement of imaging appropriateness.[462] Other studies have shown that the MRI Lumbar Spine for Low Back Pain measure has not correlated with improved outcomes.[463 464 465] The latest findings are consistent with responses to a 2020 request for public comment where commenters expressed concerns regarding measure exclusion conditions, imaging modalities, measure validity, and measure usability. In response to that request for public comments, commenters also stated that an unintended consequence of using this measure may be delayed diagnoses.[466]
Based on these findings, this measure meets the criteria that we have adopted for measure removal Factor 2 (that is, performance or improvement on a measure does not result in better patient outcomes), as codified under § 419.46(i)(3)(i)(B). Therefore, in the CY 2025 OPPS/ASC proposed rule (89 FR 59454 through 59455), we proposed to remove the MRI Lumbar Spine for Low Back Pain measure from the Hospital OQR Program beginning with the CY 2025 reporting period/CY 2027 payment determination.
We invited public comment on the proposal, including feedback on other potential measures that may better address unnecessary imaging, which we will consider for adoption into the Hospital OQR Program in future rulemaking.
Comment: Many commenters supported CMS' proposal to remove the MRI Lumbar Spine for Low Back Pain measure beginning with the CY 2025 reporting period/CY 2026 payment determination. Several of these commenters concurred with the rationale in the CY 2025 OPPS/ASC proposed rule to remove the measure based on measure removal Factor 2, stating that the measure does not provide actionable data toward quality improvement and lacks sufficient ( printed page 94414) evidence that the measure promotes quality of care and improved patient outcomes. A few of these commenters supported CMS' proposal to remove the MRI Lumbar Spine for Low Back Pain measure because the measure is no longer CBE endorsed. Commenters also supported CMS' proposal because it would allow HOPDs to focus improvement initiatives on other areas of quality.
Response: We thank commenters for their support.
Comment: A commenter recommended that CMS continue to prioritize overutilization measures and to work with stakeholders to develop clinically meaningful measures that promote value for all stakeholders and protect patients from harm from unnecessary services.
Response: We thank the commenter for their recommendation to utilize stakeholders in developing clinically meaningful measures. We note that one of our CMS National Quality Strategy goals, “Advance Equity and Engagement for All Individuals,” seeks to ensure engagement to improve healthcare quality by incorporating individual and community input into strategy and policy. We will consider the commenter's recommendation to prioritize imaging efficiency and overutilization measures in future potential rulemaking as appropriate.
Comment: A commenter did not support removing the MRI Lumbar Spine for Low Back Pain measure from the measure set, noting that four additional ICD-10-CM codes that describe “discogenic back pain” were issued.
Response: We recognize that there are now four additional ICD-10-CM codes that describe “discogenic back pain.” These additional ICD-10-CM codes do not reflect a new condition but allow practitioners more specificity in coding conditions already captured by the measure. We therefore do not believe the additional ICD-10 codes would substantively expand the measure cohort or impact our analysis that the measure has maintained stable national performance (excluding the CY 2022 performance period impacted by our COVID-19 exception policies) and low average volumes, indicating limited reliability and capacity to improve the quality of care for patients with reported low back pain.
Comment: A few commenters did not support removing the MRI Lumbar Spine for Low Back Pain measure and expressed criticism of the studies cited by CMS supporting the assertion that the measure does not correlate with improved outcomes. The commenters stated that none of the studies analyzed the relationship between the measure and outcomes, and that one study provides a rationale to retain the measure as it underscores disparities in care. These commenters also expressed concern that the rate of inappropriate use of MRIs is high, based on a recent publication of the CMS Provider Data Catalog showing an average rate of 38 percent. Finally, the commenters recommended that CMS make public the denominator for results across all measures.
Response: We appreciate commenters' feedback regarding the removal of the MRI Lumbar Spine for Low Back Pain measure. The studies cited by CMS in the proposed rule (89 FR 59455) analyzed and found limited correlation between the measure and utilization of lumbar MRIs, utilization of conservative therapy, and usage of MRI lumbar imaging in hospitals, respectively.[467 468 469] Further, one study found that less than five percent of all MRIs of the lumbar spine for low back pain were included in the MRI Lumbar Spine for Low Back Pain measure because of documented conditions that fell within the measure exclusion criteria.[470] The study also noted that compliance rates may be related to more complete documentation and coding, rather than actual changes in patient care.[471] These findings indicate that improvement on this measure is a result of the continued exclusion of conditions, and that improvement does not drive better patient outcomes.
Regarding recent measure performance rates, this measure calculates the percentage of MRI of the lumbar spine studies with a diagnosis of low back pain on the imaging claim for which the beneficiary did not have prior claims-based evidence of antecedent conservative therapy with lower scores indicating higher performance. The most recent mean score as published in the July 2024 refresh of the CMS Provider Data Catalog for national data was 36.2 percent for data collected from July 2022 to June 2023.[472] As we discussed in the CY 2025 OPPS/ASC proposed rule, the measure has maintained stable national performance and low average volumes, indicating limited reliability and capacity that the measure improves the quality of care for patients with reported low back pain. We will take into consideration commenters' recommendation that CMS make public the denominator for results across all measures in the Provider Data Catalog as appropriate in future potential rulemaking.
Comment: A commenter recommended that CMS review alternative measure concepts that could improve care for patients with low back pain.
Response: We thank the commenter for their recommendation. We will continue engaging with interested parties through education and outreach opportunities for any feedback about potential new measures in the future.
After consideration of the public comments received, we are finalizing our proposal to remove the MRI Lumbar Spine for Low Back Pain measure beginning with the CY 2025 reporting period/CY 2027 payment determination.
b. Removal of the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery Measure Beginning with the CY 2025 Reporting Period/CY 2027 Payment Determination
In the CY 2011 OPPS/ASC final rule (75 FR 72079 and 72080), we adopted the claims-based Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure beginning with the CY 2012 payment determination. This measure calculates the percentage of stress echocardiography, single photon emission computed tomography myocardial perfusion imaging (SPECT MPI), stress magnetic resonance imaging (MRI), or computed coronary tomography angiography (CCTA) performed at each facility in the 30 days prior to an ambulatory non-cardiac, low-risk surgery performed at any location, and was endorsed by a CBE in 2011. Endorsement was removed in 2021 after the measure developer did not submit the measure for review during its designated measure endorsement maintenance cycle.
We adopted the measure for the Hospital OQR measure set, in part, to ( printed page 94415) address an area of patient safety related to one of the most common imaging services in the Medicare population at the time, as we believed inappropriate use could increase the patient's risk of cancer, contribute no benefit to the quality of care, and result in the unnecessary waste of services (75 FR 72076). In response to commenter concerns regarding the infrequent occurrence of low-risk non-cardiac surgeries, and whether this measure may assess significant differences in the provision of imaging tests and their impact on the quality of care provided, we stated at the time that the measure could identify outlier practice patterns and encourage HOPDs to improve their quality of care.
Our routine monitoring and evaluation shows that the range of cases per HOPD varies greatly (that is, from one to over 1,300 cases), posing limitations when assessing and interpreting comparative performance trends over time. In addition, while there was a slight average performance score improvement from payment determination years CY 2020 to 2024 (despite the COVID-19 pandemic and the larger pool of reporters) of about one percent (4.7 percent and 3.6, respectively), the variation between the 10th and 25th percentiles of performance is not statistically distinguishable, indicating the measure may not provide meaningful data for informing consumers about quality of care for this service in HOPDs. Furthermore, at a 3.5 percent average overall rate for this measure for the CY 2024 payment determination year, there is little room for national performance on this measure to show significant improvement as lower rates are better for this measure.
Based on these findings, this measure meets the criteria for measure removal Factor 2 (that is, performance or improvement on a measure does not result in better patient outcomes), as codified under § 419.46(i)(3)(i)(B). Therefore, in the CY 2025 OPPS/ASC proposed rule (89 FR 59455), we proposed to remove the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure beginning with the CY 2025 reporting period/CY 2027 payment determination.
We invited public comment on the proposal, including feedback on other potential measures that may better address unnecessary imaging, which we will consider for adoption into the Hospital OQR Program in future rulemaking.
Comment: Many commenters supported CMS' proposal to remove the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure beginning with the CY 2025 reporting period/CY 2027 payment determination. Several of these commenters concurred with the rationale in the CY 2025 OPPS/ASC proposed rule to remove the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure based on measure removal Factor 2, stating the measure does not provide actionable data toward quality improvement and lacks sufficient evidence that the measure promotes quality of care and improved patient outcomes. A few of these commenters supported the removal of the measure as it is no longer CBE endorsed. A commenter stated that removing this measure would allow HOPDs to focus improvement initiatives on other areas of quality. Another commenter stated that the removal of this measure can help cardiovascular specialists focus on more meaningful and high-value care practices, aligning with initiatives like Choosing Wisely, a campaign by the American Board of Internal Medicine that advocates for avoiding unnecessary medical tests and procedures. This commenter further noted that studies have shown that such imaging often does not change patient management or outcomes and can lead to unnecessary costs.
Response: We thank commenters for their support.
Comment: A commenter recommended CMS continue to prioritize overutilization measures and to work with stakeholders to develop clinically meaningful measures that promote value for all stakeholders and protect patients from harm from unnecessary services.
Response: We thank the commenter for their recommendation to utilize stakeholders in developing clinically meaningful measures and to prioritize overutilization measures. We note that one of our CMS National Quality Strategy goals, “Advance Equity and Engagement for All Individuals,” seeks to ensure engagement to improve healthcare quality by incorporating individual and community input into strategy and policy. We will consider the commenter's recommendation to prioritize imaging efficiency and overutilization measures in future rulemaking.
Comment: A few commenters did not support removal of the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure, stating that measure removal Factor 2 (that is, performance or improvement does not result in better outcomes) is inapplicable because inappropriate testing causes adverse reactions. Commenters expressed concern about categorizing the performance score improvement (4.7 percent to 3.6 percent) from 2020 to 2024 as `slight,' stating that it was a significant reduction in unnecessary testing.
Response: We disagree with commenters' feedback that removal Factor 2 is inapplicable to this measure. We cannot solely evaluate the 1.1 percentage point improvement from the 4.7 percent 2020 performance score in determining clinical significance for quality measure performance. To adequately determine whether removal Factor 2 applies, we review the performance score data in its entirety, including the number of HOPDs meeting case minimums and the variation between the 10th and 25th percentiles of performance. As previously discussed, this variation is not statistically distinguishable, indicating that the measure may not provide meaningful data for informing consumers about quality of care for this service in HOPDs. Additionally, while inappropriate testing causes adverse reactions, our routine monitoring and evaluation shows that the range of cases per HOPD varies from one to over 1,300 cases, posing limitations when assessing and interpreting comparative performance trends over time. We therefore cannot extrapolate patient outcomes from the measure results.
After consideration of the public comments received, we are finalizing our proposal to remove the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure beginning with the CY 2025 reporting period/CY 2027 payment determination.
3. Summary of Finalized Program Measure Set Updates
a. Newly Finalized Program Measure Set Beginning With the CY 2027 Payment Determination
Table 163 summarizes the newly finalized Hospital OQR Program measure set beginning with the CY 2027 payment determination, which removes the two imaging efficiency measures discussed in sections XV.C.2.a and XV.C.2.b of this final rule with comment period and adds three cross-program health equity measure discussed in sections XV.C.1.a and XIV.B of this final rule with comment period.
( printed page 94416)b. Newly Finalized Program Measure Set Beginning With the CY 2031 Payment Determination
Table 164 summarizes the newly finalized Hospital OQR Program measure set for the CY 2031 payment determination, which removes the two imaging efficiency measures, discussed in sections XV.C.2.a and XV.C.2.b of this final rule with comment period; adds the Information Transfer PRO-PM, discussed in section XV.C.1.b of this final rule with comment period; and adds three cross-program health equity measures, discussed in section XIV.B of this final rule with comment period.
( printed page 94417)D. Administrative Requirements
We refer readers to § 419.46(b) and (c) and the CYs 2014, 2016, and 2019 OPPS/ASC final rules (78 FR 75108 through 75109, 80 FR 70519, and 83 FR 59103 through 59104, respectively) for our policies regarding program participation requirements and withdrawal from the program. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
E. Form, Manner, and Timing of Data Submission
1. General Data Submission Policy
We refer readers to § 419.46(d) and the CY 2023 OPPS/ASC final rule with comment period (87 FR 72110 through 72112) for our general program policies regarding: (1) submission of data under the Hospital OQR Program generally; (2) review and correction of submitted data; and (3) extraordinary circumstance exception requests (ECE) for data submission.
We also refer readers to the CYs 2019 and 2022 OPPS/ASC final rules (83 FR 59104 through 59105 and 86 FR 63861, respectively) for details regarding our maintenance of technical specifications. We maintain measure technical specification manuals (referred to as Specifications Manuals) that can be found on the CMS website at: https://qualitynet.cms.gov/outpatient/specifications-manuals.
We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
2. Measure Specific Data Submission and Reporting Requirements
We refer readers to the CYs 2014, 2016, 2022, 2023, and 2024 OPPS/ASC final rules (77 FR 68484; 80 FR 70521, 87 FR 72110 through 72112; 78 FR 75097 through 75100; and 88 FR 82004 through 82006, respectively) for information regarding our claims-based, web-based, eCQM, chart-abstracted, PRO-PM, and survey-based data submission and reporting requirements. ( printed page 94418)
a. Web-Based Measures
(1) CMS-Designated Information System and Data Submission for the Hospital Commitment to Health Equity (HCHE), Screening for Social Drivers of Health (SDOH), and Screen Positive Rate for SDOH Measures
We refer readers to the CY2014 OPPS/ASC final rule (78 FR 75112 through 75115), the CY2016 OPPS/ASC final rule (80 FR 70521), and the CMS website, currently available at https://qualitynet.cms.gov, for a discussion of the requirements for measure data submitted via the Hospital Quality Reporting (HQR) System (formerly referred to as the QualityNet Secure Portal). The HQR System safeguards protected health information in compliance with the HIPAA Privacy and Security Rules (45 CFR parts 160 and 164, subparts A, C, and E).
In sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period, we are adopting: (1) the Hospital Commitment to Health Equity measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination.
Consistent with our established data submission requirements (80 FR 70521 and 70522), we proposed in the CY 2025 OPPS/ASC proposed rule (89 FR 59458) that HOPDs would be required to submit all of the data required to calculate each of these three measures annually using a CMS-approved, web-based, data collection tool available within the HQR System starting January 1 through and including May 15 in the year prior to the applicable payment determination year. For the Hospital OQR Program, the performance period (which we refer to as the CY reporting period) for each of these measures on which data is submitted using a web-based tool would be January 1 through and including December 31 of the year that is 2 years prior to the applicable payment determination year; and the data submission period would be January 1 through and including May 15 in the calendar year immediately following the CY reporting period and immediately prior to the applicable payment determination year. For example, for the CY 2025 reporting period/2027 payment determination, the data submission period would be January 1, 2026, through and including May 15, 2026, covering the performance period of January 1, 2025, through and including December 31, 2025. Pursuant to § 419.46(d)(4), a review and corrections period runs concurrently with the data submission period. During this timeframe, HOPDs would be able to enter, review, and correct data submitted for these measures.
We invited public comment on the proposal.
Comment: Several commenters expressed concerns regarding duplicative reporting of attestations between hospital inpatient departments and hospital outpatient departments for the HCHE measure in the Hospital OQR Program, noting that this measure has already been adopted into the Hospital IQR Program. These commenters requested the ability to submit one single submission jointly for both programs. Another commenter noted that many of the priorities included in this measure are currently addressed by hospitals and health systems and urged CMS to catalogue what hospitals are already doing before establishing new measures or requirements to reduce burden and redundancy. A commenter recommended conducting an environmental scan, listening sessions, focus groups, and a TEP to reduce instances of redundant measures.
Response: We appreciate commenters' input on mechanisms to reduce burden and reporting duplication. We acknowledge that the HCHE measure has been adopted into the Hospital IQR Program, and the importance of prioritizing resources and streamlining hospital-wide system processes to promote administrative efficiency. However, because hospital inpatient departments and hospital outpatient departments treat different patient populations and employ different staff, we are requiring that hospitals participating in both the Hospital IQR and Hospital OQR Programs submit their HCHE measure data separately. We will monitor implementation of the HCHE measure and consider modifications to the manner of data submission for this measure in future rulemaking.
We refer readers to sections XIV.B.1, XIV.B.2, XIV.B.3 of this final rule with comment period for our cross-program proposals and full discussion of public comments received. We are finalizing our proposal that HOPDs would be required to submit all of the data required to calculate the HCHE, Screening for SDOH, and Screen Positive Rate for SDOH measures annually using a CMS-approved, web-based, data collection tool available within the HQR System starting January 1 through and including May 15 in the year prior to the applicable payment determination year.
(2) National Healthcare Safety Network (NHSN)
We refer readers to the CY 2014 OPPS/ASC final rule (78 FR 75097 through 75100) for a discussion of the previously finalized requirements for measure data submitted via the Centers for Disease Control and Prevention NHSN website. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
b. Electronic Clinical Quality Measures (eCQMs) and the Requirement That Electronic Health Record (EHR) Technology Be Certified to All eCQMs Available To Report Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
In the CY 2024 Medicare Physician Fee Schedule (PFS) final rule (88 FR 79307 through 79312), we finalized revisions to the definition of certified electronic health record technology (CEHRT) for the Medicare Promoting Interoperability Program at 42 CFR 495.4 and for the Quality Payment Program at 42 CFR 414.1305. Specifically, we added a reference to the “Base EHR definition,” which ONC proposed in the Health Data, Technology, and Interoperability: Certification Program Updates, Algorithm Transparency, and Information Sharing (HTI-1) proposed rule (88 FR 23759, 23905). We finalized these revisions to ensure, if the HTI-1 proposals were finalized, the “Base EHR definition” would be applicable for the CEHRT definitions going forward (88 FR 79309 through 79312).[473] ONC subsequently finalized a definition of “Base EHR” in the Health Data, Technology, and Interoperability: Certification Program Updates, Algorithm Transparency, and Information Sharing final rule (89 FR 1192, 1298).
We also finalized the replacement of references to the “2015 Edition health IT certification criteria” with “ONC health IT certification criteria,” and the addition of the regulatory citation for ONC health IT certification criteria in 45 CFR 170.315. We finalized the proposal to specify that EHR technology must meet ONC's health IT certification criteria “as adopted and updated in 45 ( printed page 94419) CFR 170.315” to qualify as CEHRT (88 FR 79553). These revisions, finalized in the CY 2024 PFS final rule, are consistent with the policy subsequently finalized in ONC's HTI-1 final rule, which appeared in the Federal Register on January 9, 2024 (89 FR 1205 through 1210). For additional background and information on this update, we refer readers to the discussion in the CY 2024 PFS final rule on this topic (88 FR 79307 through 79312).
In the CY 2022 OPPS/ASC final rule (86 FR 63868 and 63869), we adopted a requirement for hospitals to utilize certified technology updated to be consistent with the 2015 Edition Cures Update (now referred to as the ONC health IT certification criteria) for reporting eCQMs under the Hospital OQR Program, beginning with the CY 2023 reporting period/CY 2025 payment determination. However, we did not propose or finalize a requirement that the EHR technology used for eCQM reporting must be certified to all eCQMs (that is, tested and validated on each individual eCQM) in the Hospital OQR Program.
The Hospital IQR Program and the Medicare Promoting Interoperability Program require EHRs to be certified to all available eCQMs in the programs. We finalized this policy for the Hospital IQR Program in the FY 2018 IPPS/LTCH PPS final rule (82 FR 38393) for the FY 2019 and FY 2020 payment determination years, and we finalized in the FY 2020 IPPS/LTCH PPS final rule (84 FR 42505 through 42506) that this policy would continue beginning with the CY 2020 reporting period/FY 2022 payment determination. For the Medicare Promoting Interoperability Program, we finalized this policy in the FY 2018 and FY 2019 IPPS/LTCH PPS final rules for CYs 2018 and 2019, respectively (82 FR 38483 through 38485 and 83 FR 41671 through 41672, respectively). We also finalized the continuation of this requirement in the FY 2020 IPPS/LTCH PPS final rule (84 FR 42600 and 42601) for CY 2020 and subsequent years. When EHRs are certified to all available eCQMs in a program measure set, hospitals are able to accurately capture and report data for these measures. For this reason, and to align the Hospital OQR Program's eCQM certification requirements with the Hospital IQR Program and Medicare Promoting Interoperability Program clinical quality measure electronic submission requirements for eligible hospitals, we proposed in the CY 2025 OPPS/ASC proposed rule (89 FR 59458 through 59459) that beginning with the CY 2025 reporting period/CY 2027 payment determination, a HOPD using EHR technology certified to the ONC health IT certification criteria would be required to have its EHR technology certified to all eCQMs that are available to report under the Hospital OQR Program to meet reporting requirements for the Hospital OQR Program.
We further proposed that for the CY 2025 reporting period/CY 2027 payment determination and subsequent years, HOPDs would additionally be required to use the most recent version of the eCQM electronic measure specifications for the designated reporting period available on the Electronic Clinical Quality Improvement (eCQI) Resource Center website at: https://ecqi.healthit.gov/. We noted in the CY 2021 OPPS/ASC final rule (86 FR 63861) that we would generally update the measure specifications on an annual basis to align with current clinical guidelines and code systems.
Our proposal to require that EHRs be certified to all available eCQMs would promote more accurate electronic quality reporting by incentivizing HOPDs to have their EHR and other health information technology (IT) vendors test all available eCQMs and offer reporting modules with certified eCQMs. Through this requirement, we expect greater certainty for hospitals that their EHR systems are capable of accurately calculating the eCQMs reported to CMS under the Hospital OQR Program because the EHR technology would be up to date and tested on each eCQM. Additionally, we anticipate this requirement would help reduce burden for hospitals by potentially reducing the frequency of needing to consult with their EHR and other health IT vendors to troubleshoot implementation or reporting issues.
Finally, in the CY 2025 OPPS/ASC proposed rule (89 FR 59459) we proposed to revise regulatory text at § 419.46 to add a new paragraph (j) to codify submission requirements for eCQMs under the Hospital OQR Program. Under the proposal, we would codify in § 419.46(j)(1) the requirement for hospitals to utilize certified technology updated to be consistent with ONC's health IT certification criteria, as adopted and updated in 45 CFR 170.315, for reporting eCQMs under the Hospital OQR Program. We proposed to codify in § 419.46(j)(2) the requirement that the EHR technology used for eCQM reporting must be certified to all eCQMs (that is, tested and validated on each individual eCQM) available to report under the Hospital OQR Program. We also proposed to codify in § 419.46(j)(3) the requirement that hospitals use the most recent version of the eCQM electronic measure specifications for the applicable reporting period available on the Electronic Clinical Quality Improvement (eCQI) Resource Center website at: https://ecqi.healthit.gov/ or another website as designated by CMS.
We invited public comment on these proposals.
Comment: Several commenters supported the proposal requiring EHRs to be certified to all available eCQMs in the Hospital OQR Program, citing that this policy is an important step to promoting interoperability and will incentivize EHR vendors to take responsibility for eCQMs. A commenter applauded CMS for promoting alignment with other programs.
Response: We thank commenters for their support.
Comment: A few commenters expressed concern about the potential financial and operational burden for hospitals to meet the requirements, such as the need to invest in new technology, train staff, coordinate with EHR vendors, implement new workflows, and the risk of incurring potential penalties due to loss of quality reporting reimbursement. A commenter noted that hospitals continue to grapple with eCQM calculation, data analysis, and performance improvement. The commenter further stated that requiring EHR certification for all eCQMs without adequate time for system updates and testing could create undue burden and hinder quality improvement efforts. A few commenters requested that CMS delay implementation of the policy to provide additional time to meet requirements and for smoother implementation.
Response: Requiring EHR technology to be certified to all available eCQMs is important in collecting the most relevant electronic data and would help streamline the electronic data extrapolation component of HOPD workflow in the future. We recognize that requiring EHRs to be certified to all available eCQMs initially creates some costs for both HOPDs and health IT vendors. However, once the initial process of certifying the EHR to all available eCQMs has been completed, hospitals would not have to certify measures individually, and the EHR would accurately capture and calculate all eCQMs in the Hospital OQR Program. Given the benefits of certifying EHRs to all eCQMs and the small number of eCQMs in the Hospital OQR Program, we will not be delaying the policy.
Comment: A few commenters expressed concerns over accessing timely and reliable eCQM data through their EHR. A commenter specifically ( printed page 94420) noted that the ability to leverage CEHRT and the most recent eCQM specifications is dependent on updates incorporated by EHR vendors. Commenters urged CMS to work with the Office of the National Coordinator for Health Information Technology (ONC) and health IT vendors to include commensurate requirements on vendors, including making eCQM performance data available within 1 month of the start of the performance period and encouraging EHR vendors to provide functionality with sufficient lead time.[474]
Response: Utilizing certified technology updated consistent with ONC's health IT certification criteria and certified to all eCQMs would streamline and mitigate challenges related to accessing and exporting eCQM data. As specified in 45 CFR 170.315(c)(1) through (4), health IT systems that present for certification to these criteria under the ONC Health IT Certification Program must support the recording, exporting, importing, calculating, reporting, and filtering of clinical quality measures selected for certification by developers and CMS. Specifically, certified health IT certified to 45 CFR 170.315(c)(1) and (3) must be able to record all data necessary to calculate each clinical quality measure and enable users to export a data file that is formatted in accordance with Quality Reporting Document Architecture (QRDA) Category 1 and do so without subsequent developer assistance. For additional information on the ONC certification criteria for health IT, we refer readers to the web page available at: https://www.healthit.gov/topic/certification-ehrs/certification-criteria.
We will continue to seek input from health IT vendors and collaborate with ASTP/ONC to define standards that influence eCQM data collection and reporting for our quality reporting programs.
Comment: A commenter stated their belief that CEHRT would not enable CMS to meet its goal of improving data standardization and standardized data collection for eCQMs and encouraged CMS to take these factors into consideration.
Response: We disagree that CEHRT updated consistent with ONC's health IT certification criteria would not support our goal of standardization. Key elements of a number of ONC certification criteria for health IT, such as adopted versions of the United States Core Data for Interoperability (USCDI), are designed to promote the standardization of data collection and exchange, including standards for structured data capture and the use of relevant health data classes and elements.
After consideration of the public comments we received, we are finalizing our proposal requiring use of EHR technology certified to all eCQMs that are available to report under the Hospital OQR Program and codifying this policy in § 419.46(j)(2). We are also codifying in § 419.46(j)(1) the requirement for hospitals to utilize certified technology updated consistent with ONC's health IT certification criteria and codifying in § 419.46(j)(3) the requirement that hospitals use the most recent version of the eCQM electronic measure specifications.
c. Patient-Reported Outcome-Based Performance Measures (PRO-PMs)
(1) Data Submission of PRO-PM Data
In the CY 2024 OPPS/ASC final rule (88 FR 82006) we finalized that for the Total Hip Arthroplasty and/or Total Knee Arthroplasty (THA/TKA) PRO-PM, hospitals must use the HQR system for data submission for a PRO-PM. In the CY 2025 OPPS/ASC proposed rule (89 FR 59459), we proposed to apply this submission method to PRO-PMs generally, including the Information Transfer PRO-PM. We proposed that hospitals must use the HQR system for data submission for any PRO-PM that we adopt for the Hospital OQR Program measure set. HOPDs may choose to: (1) directly submit their PRO-PM data to CMS using the HQR system; or (2) utilize a third-party entity, such as a vendor or registry, to submit their data using the HQR system. The HQR system allows for data submission using multiple file formats (such as CSV, XML) and a manual data entry option, allowing HOPDs additional flexibility in data submission.
We invited public comment on the proposal.
We did not receive any public comments on our proposal and therefore, we are finalizing our proposal that hospitals must use the HQR system for data submission for any PRO-PM that we adopt for the Hospital OQR Program measure set.
(2) Data Submission and Reporting Requirements for the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM)
As discussed in section XV.C.1.b of this final rule with comment period, we are adopting the Information Transfer PRO-PM beginning with voluntary reporting for the CY 2026 reporting period followed by mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59459), we proposed that the performance period on which data is submitted would be January 1 through and including December 31 of the year that is two years prior to the applicable payment determination year. We proposed to require HOPDs to submit their Information Transfer PRO-PM data between the period starting January 1st through and including May 15 of the year prior to the applicable payment determination year. All deadlines occurring on a Saturday, Sunday, or legal holiday, or on any other day all or part of which is declared to be a non-workday for Federal employees by statute or Executive order would be extended to the first day thereafter.
We proposed to require HOPDs to offer all patients meeting the measure's denominator specifications the opportunity to complete the survey. Additionally, we proposed a minimum random sample size of 300 completed surveys to ensure the reliability of the measure, as this is a recommended minimum sample size for a population of 1,500 to provide a 95 percent confidence interval and a 90 percent confidence interval for a population of over 10,000; this is also generally accepted as a minimum sample size for stable population estimates.[475 476]
( printed page 94421)HOPDs that are unable to collect 300 completed surveys will not be able to perform random sampling, and would instead be required to submit data on survey responses from all completed surveys received.
We invited public comment on these proposals.
We did not receive any public comments on our proposal and therefore, we are finalizing our proposal for data submission and reporting requirements for the Information Transfer PRO-PM as proposed.
F. Public Reporting of Measure Data
1. General Policy
We refer readers to the CY 2024 OPPS/ASC final rules (88 FR 81995 and 81996) for our previously finalized policies regarding public display of quality measures.
2. Public Reporting of Median Time From Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients—Psychiatric/Mental Health Patients Strata on Care Compare
We refer readers to the CY 2011 OPPS/ASC final rule (75 FR 72086) where we adopted the Median Time from ED Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients) measure beginning with CY 2013 payment determination. The Median Time for Discharged ED Patients measure is a chart-abstracted measure that evaluates the time from ED arrival to departure, also known as ED throughput time. The measure data are stratified into four separate calculations: (a) Median Time for Discharged ED Patients—Overall Rate; (b) Median Time for Discharged ED Patients—Reporting Measure, which excludes psychiatric/mental health and transfer patients; (c) Median Time for Discharged ED Patients—Psychiatric/Mental Health Patients, which includes information only for psychiatric/mental health patients; and (d) Median Time for Discharged ED Patients—Transfer Patients, which includes information only for patients transferred from the ED.
In the CY 2024 OPPS/ASC final rule (88 FR 81995 and 81996), we finalized that data for three measure strata (that is, the Overall Rate, Reporting Measure, and Transfer Patients strata) would be publicly reported both on data.medicare.gov in downloadable data files and on Care Compare (or subsequent CMS-designated websites). Data for the Psychiatric/Mental Health Patients stratum are not currently publicly reported on Care Compare, though these data are published on data.medicare.gov in downloadable data files (82 FR 59438). In the CY 2018 OPPS/ASC final rule (82 FR 52576 through 52578), we summarized commenters' concerns that delays in ED discharge of mental health patients may be influenced, in part, by the availability of community resources. In response, we stated that we would take additional time for further consideration prior to displaying this subset of data on Care Compare. We have considered commenters' concern that factors outside of an HOPD's control may influence ED throughput for psychiatric/mental health patients; however, it is our understanding that many hospitals face such concerns, and that timely care is a critical aspect of quality of care. We also stated in the CY 2024 OPPS/ASC final rule (88 FR 82061) in the context of adopting this measure for the REHQR Program that the public reporting of these data on Care Compare could help patients and their caregivers identify which facilities are performing better than others despite potential challenges, and drive quality improvement efforts.
Our routine monitoring and evaluation of the CY 2024 performance period for this measure has shown a median ED throughput time of 4.7 hours for psychiatric/mental health patients compared to 2.6 hours for non-psychiatric/mental health patients, suggesting this is an area that may benefit from additional quality improvement efforts. Data from the Median Time for Discharged ED Patients—Psychiatric/Mental Health Patients will be useful for patients choosing a care location, as well as researchers and hospital staff as they attempt to address health disparities and improve the timeliness of care for mental health patients. Since the data required for public reporting are already collected and submitted by participating HOPDs, publicly reporting this stratification would not create additional hospital burden.
For these reasons, in the CY 2025 OPPS/ASC proposed rule (89 FR 59459 through 59460), we proposed to make data for the Psychiatric/Mental Health Patients stratification available on Care Compare, including data that were previously published on data.medicare.gov but not displayed on Care Compare, beginning in CY 2025.
We invited public comment on the proposal.
Comment: Several commenters supported CMS' proposal to publicly report data for the Psychiatric/Mental Health patient stratum on Care Compare, noting that reporting these data would help consumers make educated decisions for timely, appropriate care and promote accountability for throughput time. A commenter noted that these data will provide policymakers the necessary information to address challenges with ED boarding for this patient population.
Response: We thank commenters for their support of our proposal.
Comment: A commenter supported making data for the Median Time for Discharged ED Patients—Psychiatric/Mental Health stratum more accessible but did not support imposing additional reporting burdens on hospitals.
Response: We appreciate the commenter's support for increasing data accessibility. We wish to clarify that the proposed policy would not increase reporting burden for HOPDs. Data for the Median Time for Discharged ED Patients measure are already collected and submitted by participating HOPDs and are currently accessible in downloadable files from CMS' Provider Data Catalog available at data.cms.gov. The public reporting of the Psychiatric/Mental Health Patients stratum on Care Compare would not require hospitals to collect or submit any additional data beyond what is already required.
Comment: Many commenters opposed the proposal to publicly report the Median Time for Discharged ED Patients—Psychiatric/Mental Health stratum on Care Compare, noting that ED throughput times for this population are influenced by factors outside of a provider's control, including lack of psychiatric resources in a community, barriers to efficient transition, and challenges with screening this population. Several commenters noted their beliefs that the measure does not reflect the efficacy and quality of care provided in an ED. A few commenters stated concerns regarding a lack of evidence demonstrating that ED throughput time influences patient outcomes.
Response: We understand commenters' concerns that there are many factors outside a hospital's control that could affect ED throughput, such as limited psychiatric resources; however, the Compare tool data indicate that hospitals with 50 beds or less performed better on this measure overall and had lower median ED throughput times for discharged psychiatric/mental health patients than larger hospitals (88 FR 82049), despite evidence indicating that smaller hospitals often lag behind in their quality performance and maintain limited scale and resources compared to ( printed page 94422) large systems.[477 478 479] Therefore, while external factors can play a role in Median Time from ED Arrival to ED Departure, resource constraints alone do not fully account for variations in performance, as evidenced by the better performance of smaller hospitals on this measure.
We respectfully disagree that the measure does not represent the efficacy or quality of care provided in an ED. While ED throughput alone may not reflect all aspects of care in an ED, it is an important indicator of operational efficiency [480] and timely care, which have direct influence on the quality of care provided to patients.[481] Prolonged ED throughput times can lead to overcrowding, and overcrowding is associated with avoidable problems in the ED, such as poor outcomes resulting from delays in treatment and ambulance diversion.[482] Additionally, ED throughput has a direct influence on patient experience [483] in the ED setting, and studies demonstrate that higher patient satisfaction is associated with improved patient outcomes, including decreased mortality [484] and lower readmission rates.[485]
We reiterate that timely care is a critical aspect of quality of care and directly impacts patient outcomes, particularly for an ED episode of care. The public reporting of these data can help patients and their caregivers identify which hospitals are performing better than others, despite the challenges many hospitals face, and drive quality improvement efforts. Additionally, aligning the reporting of ED throughput data across the Hospital OQR and REHQR Programs will allow consumers to make informed comparisons across settings.
Comment: A few commenters expressed their belief that there is no added benefit to reporting the Median Time for Discharged ED Patients—Psychiatric/Mental Health data on Care Compare since these data are already available in downloadable files on cms.data.gov. Another commenter noted that reporting the data would be duplicative of other strata listed on the Compare tool, and a few commenters expressed concern that publicly reporting the stratum may not help patients when making care decisions.
Response: The inclusion of the Median Time for Discharged ED Patients—Psychiatric/Mental Health stratum on the Compare tool addresses a critical behavioral health gap in the publicly reported Hospital OQR Program measure set and is not duplicative of other data listed on the Compare tool. By publicly reporting these data, we aim to increase transparency around the care provided to this vulnerable patient population and highlight areas where resources and processes may need to be strengthened. We reiterate that publicly reporting these data on the Compare tool will be useful for patients choosing a care location, as well as researchers, policymakers, and hospital staff as they attempt to address health disparities and improve the timeliness of care for mental health patients.
Comment: A few commenters expressed concerns regarding potential unintended consequences of publicly reporting the data. A commenter expressed their belief that publicly displaying the data could discourage facilities from taking the necessary time to ensure patients have adequate resources in place before discharge. Several commenters stated their belief that reporting the data may deter individuals from seeking care during mental health crises.
Response: We appreciate the commenter's input regarding potential unintended consequences of displaying these data on the Compare tool. We remain confident that rather than deterring individuals from seeking care, publicly reporting these data on the Compare tool would help patients and their caregivers identify the hospitals that are performing better on this metric and empower consumers to make informed decisions. We also respectfully disagree that publicly reporting ED throughput data on the Compare tool for the Psychiatric/Mental Health stratum would incentivize HOPDs to inappropriately limit their care to quickly discharge one of their most vulnerable patient populations from the ED. Rather, we remain confident that HOPDs will continue to provide high quality care for psychiatric/mental health patients and submit data as part of their commitment to the patient experience and ongoing quality improvement efforts. We further note that these data have been available since 2018 in the Provider Data Catalog located at https://data.cms.gov (82 FR 59437), as part of CMS' assessment of timely and effective care, and that median time for ED discharge continued to increase for psychiatric/mental health patients after 2018, indicating that HOPDs were not incentivized to limit their care after these data became publicly available.
Comment: A commenter raised concerns that limited pediatric inpatient psychiatric beds contribute to very long boarding times in the ED and recommended further stratifying the data by pediatric and adult populations.
Response: We acknowledge the commenter's concerns that limited pediatric psychiatric resources may contribute to longer ED throughput times for the pediatric patient population. However, we believe it is important to not separate the pediatric and adult populations when calculating this measure. The current measure aims to capture performance for all Psychiatric/Mental Health patients. Stratifying by age does not align with our broader goals of improving emergency care across all age groups, as these patient groups should be provided the same level of care in the ED setting. We will continue to monitor feedback on this measure and evaluate potential future refinements based on trends and stakeholder input.
Comment: A commenter stated that the current data, presented without additional context or risk adjustment may lead to misinterpretation by consumers. The commenter recommended risk-adjusting the data and establishing a cap on the maximum time from ED arrival to ED departure, ( printed page 94423) noting that a ceiling on this metric would help level the playing field for hospitals in areas with bed shortages.
Response: We thank the commenter for their input. We believe that publicly displaying the full range of time from ED arrival to ED departure is necessary to improve transparency around wait times for all hospitals and allow for more informed decision-making for patients. Establishing a cap or ceiling for this metric could result in extremely long or outlier wait times not being shown on Care Compare, and we believe that information about outlier wait times is important to display on Care Compare for patient awareness. Similarly, we disagree that risk-adjustment is appropriate for this measure because a patient's ED wait time should not be related to any patient-specific factors.
Comment: A few commenters recommended that the publicly displayed data for the Median Time for Discharged ED Patients—Psychiatric/Mental Health population also include data on the mental health resources in a HOPD's community. Commenters also encouraged partnership with stakeholders to provide the needed resources for this patient population.
Response: We appreciate the recommendation to provide context for the data publicly displayed for the Median Time for Discharged ED Patients—Psychiatric/Mental Health population. Dashboards on resource availability do exist, such as the Health Resources and Services Administration's (HRSA) tool for Health Professional Shortage Areas and the Medically Underserved Areas/Populations dashboard. We will consider using HRSA's dashboards, or other tools that provide context on the availability of resources in a community, to potentially supplement data for the Psychiatric/Mental Health Patients population on Care Compare as appropriate in the future.
We thank commenters for their feedback to work with interested parties and will take these recommendations into consideration as appropriate as we continue to evaluate all elements of the Hospital OQR Program to ensure a relevant and meaningful measure set.
Comment: A few commenters recommended removing the Median Time for Discharged ED Patients measure from the Hospital OQR Program measure set. A commenter specifically noted that the measure currently lacks CBE endorsement and raised concerns regarding the strength of evidence supporting the measure's use in quality reporting programs.
Response: One of the Meaningful Measures 2.0 goals is to address measurement gaps, reduce burden, and increase efficiency by using high-value quality measures impacting key quality domains. ED performance and care continues to be a key quality domain of the Hospital OQR Program. Removal of the Median Time for Discharged ED Patients measure would result in an incomplete measure set with no measures that review ED throughput. We continue to believe that the Median Time for Discharged ED Patients measure supports our Meaningful Measures 2.0 goals. Additionally, behavioral health is a high-priority outcome area and one of the CMS National Quality Strategy Goals is to improve quality and health outcomes across the care journey by driving improvements in high-priority areas, such as behavioral health. We acknowledge that the measure is no longer CBE endorsed because the measure was withdrawn by the developer,[486] however, we continue to assert that the Median Time for Discharged ED Patients continues to provide valuable information regarding timely care. Furthermore, quality improvement efforts aimed at reducing ED overcrowding and length of stay have been associated with an increase in ED patient volume, decrease in number of patients who leave without being seen, reduction in costs, and increase in patient satisfaction.[487 488 489 490] Section 1833(t)(17)(C)(i) of the Act provides that the Hospital OQR Program, to the extent feasible and practicable, shall include measures that reflect consensus among affected parties and, to the extent feasible and practicable, measures set forth by one or more national consensus building entities. As we have noted in previous rulemaking, consensus among affected parties can be reflected in ways other than CBE endorsement, including through the measure development process, through broad acceptance and use of the measure(s), and through public comment (75 FR 72064 and 72065). The CBE's continued endorsement until the measure developer withdrew the measure, and the continuous use of the measure in both the Hospital OQR Program and in academic studies, such as those cited in a previous endorsement cycle,[491 492 493 494 495] demonstrate ongoing acceptance of its value.
After consideration of the public comments we received, we are finalizing our proposal to publicly display the Median Time for Discharged ED Patients—Psychiatric/Mental Health stratum on the Compare tool, beginning with measure data for the CY 2025 reporting period.
G. Payment Reduction for Hospitals That Fail To Meet the Hospital OQR Program Requirements for the CY 2025 Payment Determination
1. Background
Section 1833(t)(17) of the Act, which applies to subsection (d) hospitals (as defined under section 1886(d)(1)(B) of the Act), states that hospitals that fail to report data required to be submitted on measures selected by the Secretary, in the form and manner, and at a time, specified by the Secretary will incur a 2.0 percentage point reduction to their Outpatient Department (OPD) fee schedule increase factor; that is, the annual payment update factor. Section 1833(t)(17)(A)(ii) of the Act specifies that any reduction applies only to the payment year involved and will not be taken into account in computing the applicable OPD fee schedule increase factor for a subsequent year. ( printed page 94424)
The application of a reduced OPD fee schedule increase factor results in reduced national unadjusted payment rates that apply to certain outpatient items and services provided by hospitals that are required to report outpatient quality data in order to receive the full payment update factor and that fail to meet the Hospital OQR Program requirements. Hospitals that meet the reporting requirements receive the full OPPS payment update without the reduction. For a more detailed discussion of how this payment reduction was initially implemented, we refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68769 through 68772).
The national unadjusted payment rates for many services paid under the OPPS equal the product of the OPPS conversion factor and the scaled relative payment weight for the APC to which the service is assigned. The OPPS conversion factor, which is updated annually by the OPD fee schedule increase factor, is used to calculate the OPPS payment rate for services with the following status indicators (listed in Addendum B to the proposed rule, which is available via the internet on the CMS website): “J1,” “J2,” “P,” “Q1,” “Q2,” “Q3,” “R,” “S,” “T,” “V,” or “U.” Payment for all services assigned to these status indicators will be subject to the reduction of the national unadjusted payment rates for hospitals that fail to meet Hospital OQR Program requirements, with the exception of services assigned to New Technology APCs with assigned status indicator “S” or “T.” We refer readers to the CY 2009 OPPS/ASC final rule with comment period (73 FR 68770 through 68771) for a discussion of this policy. In the CY 2017 OPPS/ASC final rule with comment period (81 FR 79796), we clarified that the reporting ratio does not apply to codes with status indicator “Q4” because services and procedures coded with status indicator “Q4” are either packaged or paid through the Clinical Laboratory Fee Schedule and are never paid separately through the OPPS.
The OPD fee schedule increase factor is an input into the OPPS conversion factor, which is used to calculate OPPS payment rates. To reduce the OPD fee schedule increase factor for hospitals that fail to meet reporting requirements, we calculate two conversion factors—a full market basket conversion factor (that is, the full conversion factor), and a reduced market basket conversion factor (that is, the reduced conversion factor). We then calculate a reduction ratio by dividing the reduced conversion factor by the full conversion factor. We refer to this reduction ratio as the “reporting ratio” to indicate that it applies to payment for hospitals that fail to meet their reporting requirements. Applying this reporting ratio to the OPPS payment amounts results in reduced national unadjusted payment rates that are mathematically equivalent to the reduced national unadjusted payment rates that would result if we multiplied the scaled OPPS relative payment weights by the reduced conversion factor. For example, to determine the reduced national unadjusted payment rates that applied to hospitals that failed to meet their quality reporting requirements for the CY 2010 OPPS/ASC final rule with comment period, we multiplied the final full national unadjusted payment rate found in Addendum B of the CY 2010 OPPS/ASC final rule with comment period by the CY 2010 OPPS final rule with comment period reporting ratio of 0.980 (74 FR 60642).
We note that the only difference in the calculation for the full conversion factor and the calculation for the reduced conversion factor is that the full conversion factor uses the full OPD update, and the reduced conversion factor uses the reduced OPD update. The baseline OPPS conversion factor calculation is the same since all other adjustments would be applied to both conversion factor calculations. Therefore, our standard approach of calculating the reporting ratio as described earlier in this section is equivalent to dividing the reduced OPD update factor by that of the full OPD update factor. In other words:
Full Conversion Factor = Baseline OPPS conversion factor * (1 + OPD update factor)
Reduced Conversion Factor = Baseline OPPS conversion factor * (1 + OPD update factor −0.02)
Reporting Ratio = Reduced Conversion Factor/Full Conversion Factor
Which is equivalent to:
Reporting Ratio = (1 + OPD Update factor −0.02)/(1 + OPD update factor)
In the CY 2009 OPPS/ASC final rule with comment period (73 FR 68771 through 68772), we established a policy that the Medicare beneficiary's minimum unadjusted copayment and national unadjusted copayment for a service to which a reduced national unadjusted payment rate applies would each equal the product of the reporting ratio and the national unadjusted copayment or the minimum unadjusted copayment, as applicable, for the service. Under this policy, we apply the reporting ratio to both the minimum unadjusted copayment and national unadjusted copayment for services provided by hospitals that receive the payment reduction for failure to meet the Hospital OQR Program reporting requirements. This application of the reporting ratio to the national unadjusted and minimum unadjusted copayments is calculated according to § 419.41 of our regulations, prior to any adjustment for a hospital's failure to meet the quality reporting standards according to § 419.43(h). Beneficiaries and secondary payers thereby share in the reduction of payments to these hospitals.
In the CY 2009 OPPS/ASC final rule with comment period (73 FR 68772), we established the policy that all other applicable adjustments to the OPPS national unadjusted payment rates apply when the OPD fee schedule increase factor is reduced for hospitals that fail to meet the requirements of the Hospital OQR Program. For example, the following standard adjustments apply to the reduced national unadjusted payment rates: the wage index adjustment, the multiple procedure adjustment, the interrupted procedure adjustment, the rural sole community hospital adjustment, and the adjustment for devices furnished with full or partial credit or without cost. Similarly, OPPS outlier payments made for high cost and complex procedures will continue to be made when outlier criteria are met. For hospitals that fail to meet the quality data reporting requirements, the hospitals' costs are compared to the reduced payments for purposes of outlier eligibility and payment calculation. We established this policy in the OPPS beginning in the CY 2010 OPPS/ASC final rule with comment period (74 FR 60642). For a complete discussion of the OPPS outlier calculation and eligibility criteria, we refer readers to section II.G of the CY 2023 OPPS/ASC proposed rule (87 FR 44533 through 44534).
2. Reporting Ratio Application and Associated Adjustment Policy for CY 2025
We proposed to continue our established policy of applying the reduction of the OPD fee schedule increase factor through the use of a reporting ratio for those hospitals that fail to meet the Hospital OQR Program requirements for the full CY 2025 annual payment update factor. For the CY 2025 OPPS/ASC proposed rule, the proposed reporting ratio was 0.9805, which, when multiplied by the proposed full conversion factor of $89.379, equaled a proposed conversion factor for hospitals that fail to meet the ( printed page 94425) requirements of the Hospital OQR Program (that is, the reduced conversion factor) of $87.636. We proposed to continue to apply the reporting ratio to all services calculated using the OPPS conversion factor. We proposed to continue to apply the reporting ratio, when applicable, to all HCPCS codes to which we have proposed status indicator assignments of “J1,” “J2,” “P,” “Q1,” “Q2,” “Q3,” “R,” “S,” “T,” “V,” and “U” (other than New Technology APCs to which we have proposed status indicator assignments of “S” and “T”). We proposed to continue to exclude services paid under New Technology APCs. We proposed to continue to apply the reporting ratio to the national unadjusted payment rates and the minimum unadjusted and national unadjusted copayment rates of all applicable services for those hospitals that fail to meet the Hospital OQR Program reporting requirements. We also proposed to continue to apply all other applicable standard adjustments to the OPPS national unadjusted payment rates for hospitals that fail to meet the requirements of the Hospital OQR Program. Similarly, we proposed to continue to calculate OPPS outlier eligibility and outlier payment based on the reduced payment rates for those hospitals that fail to meet the reporting requirements. In addition to our proposal to implement the policy through the use of a reporting ratio, we also proposed to calculate the reporting ratio to four decimals (rather than the previously used three decimals) to more precisely calculate the reduced adjusted payment and copayment rates.
For CY 2025, the proposed reporting ratio was 0.9805, which, when multiplied by the proposed full conversion factor of $89.379, equaled a proposed conversion factor for hospitals that fail to meet the requirements of the Hospital OQR Program (that is, the reduced conversion factor) of $87.636.
We did not receive any public comments on our proposal. For this final rule with comment period, the final reporting ratio is 0.9806, which, when multiplied by the final full conversion factor of $89.169, equals a final conversion factor for hospitals that fail to meet the requirements of the Hospital OQR Program (that is, the reduced conversion factor) of $87.439. We are finalizing our proposal to continue to calculate OPPS outlier eligibility and outlier payment based on the reduced payment rates for those hospitals that fail to meet the reporting requirements. We are also finalizing our proposals to implement the policy through the use of a reporting ratio, and to calculate the reporting ratio to four decimals (rather than the previously used three decimals) to more precisely calculate the reduced adjusted payment and copayment rates for hospitals that fail to meet the Hospital OQR Program requirements for CY 2025 payment.
XVI. Rural Emergency Hospital Quality Reporting (REHQR) Program
A. Background and Statutory Authority
The Rural Emergency Hospital Quality Reporting (REHQR) Program's overarching goals are to improve the quality of care provided to Medicare beneficiaries, facilitate public transparency, ensure accountability, and safeguard the accessibility of hospitals in rural settings.
Section 1861(kkk)(7)(A) of the Social Security Act (the Act) provides that the Secretary shall establish quality measurement reporting requirements for Rural Emergency Hospitals (REHs), which may include the use of a small number of claims-based outcomes measures or surveys of patients with respect to their experience in the REH. In selecting measures for quality reporting, section 1861(kkk)(7)(C)(iii) provides that the Secretary shall take into consideration ways to account for REHs that lack sufficient case volume to ensure that the performance rates for such measures are reliable. Although section 1861(kkk)(7)(C)(i) of the Act requires that measures specified by the Secretary for use in the REHQR Program be endorsed by the entity with a contract under section 1890(a) of the Act, section 1861(kkk)(7)(C)(ii) of the Act states that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary.
In addition, section 1861(kkk)(7)(D) of the Act provides that the Secretary shall establish procedures for making data submitted by REHs for the REHQR Program available to the public, following the opportunity for the REH to review and submit corrections on such data, with such data to be posted on a CMS website as determined appropriate by the Secretary. Beginning with 2023 (or each year beginning on or after the date that is 1 year after one or more measures are first specified), section 1861(kkk)(7)(B)(ii) of the Act requires REHs to submit quality measure data to the Secretary “in a form and manner, and at a time, specified by the Secretary.”
We refer readers to section XVI of the CY 2024 OPPS/ASC final rule (88 FR 82046 through 82076) for an overview of the REHQR Program, which includes a more detailed discussion of the statutory history and program requirements codified at 42 CFR 419.95.
1. Previously Finalized Program Measure Sets
We refer readers to the CY 2024 OPPS/ASC final rule (88 FR 82066 through 82067) for more information regarding the previously finalized REHQR Program measure set beginning with the CY 2024 reporting period.
Table 165 below summarizes the previously finalized REHQR Program measure set and initial reporting periods with program determinations beginning with the CY 2026 program determination.[496]
( printed page 94426)B. Program Measure Set Policies
We refer readers to § 419.95(e) and the CY 2024 OPPS/ASC final rule (88 FR 82051 through 82053) for our program policies regarding measure retention, and immediate and general measure suspension and removal, and to § 419.95(d) and the CY 2024 OPPS/ASC final rule (88 FR 82054) for our program policies regarding modifications to previously adopted measures.
We further refer readers to the CY 2024 OPPS/ASC final rule (88 FR 82047 through 82051) for a discussion of our considerations for adopting quality measures under the REHQR Program, and to section XIV.B.1.c of this final rule with comment period for information regarding the pre-rulemaking process.
We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
C. Program Measure Updates
1. Adoption of Health Equity Quality Measures in the REHQR Program
We refer readers to sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period for a discussion of our cross-program proposals to adopt the following measures in the REHQR Program: (1) the Hospital Commitment to Health Equity (HCHE) measure, beginning with the CY 2025 reporting period/CY 2027 program determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting period, followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period, followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination. In these sections, we also discuss the public comments received, our responses thereto, and our final decisions regarding these proposals for the REHQR Program.
As noted in the CY 2025 OPPS/ASC proposed rule (89 FR 59462 through 59463), in selecting measures for the REHQR Program, CMS takes into consideration ways to account for REHs that lack sufficient case volume to ensure that performance rates for such measures are reliable.[497] Once mandatory reporting begins, the measure specifications require all patients to be screened so we do not believe the Screening for SDOH measure or the Screen Positive Rate for SDOH measure would suffer from low case volumes. In addition, we noted that, as stated in the CY 2024 OPPS/ASC final rule (88 FR 82066), CMS does not report measures publicly unless it achieves sufficient case volumes for reliability purposes to allow for public reporting of the collected data.
2. Modification to the Reporting Period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery Measure Beginning With the CY 2027 Program Determination
In the CY 2024 OPPS/ASC final rule, we adopted the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure in the REHQR Program with a 1-year reporting period beginning with the CY 2024 reporting period (88 FR 82064 through 82066).
This measure is calculated from Part A and Part B Medicare administrative claims data for Medicare Fee-for-Service (FFS) beneficiaries with an outpatient same-day surgical procedure excluding eye surgeries and colonoscopies (except colonoscopy with biopsy). Colonoscopies are excluded from this measure as these procedures are examined separately on their own. Eye surgeries are excluded because they are performed in high volume and are generally perceived as being “low risk.” As stated in the CY 2024 OPPS/ASC final rule (88 FR 82064), this measure makes unplanned patient hospital visits (ED visits, observation stays, or unplanned inpatient admissions) after surgery more visible to providers and patients through publicly reporting scores.
As noted in the CY 2024 OPPS/ASC final rule (88 FR 82064), we believe this measure could also encourage providers to engage in quality improvement activities to reduce these visits by providing feedback to hospitals and providers. This measure meets the National Quality Strategy goals of embedding quality into the care journey and promoting safety,[498] and we expect that the measure would promote improvement in patient care over time (88 FR 82064 through 82065).
We have monitored and evaluated the reporting patterns of hospitals that have converted to REH status and have found that under the Hospital Outpatient Quality Reporting (OQR) Program, a ( printed page 94427) limited number of current REHs were able to publicly report on this measure as specified based on case threshold minimums. Therefore, in consideration of our statutory obligation to consider ways to account for low case volumes and to publicly report on quality-of-care metrics for REHs, in the CY 2025 OPPS/ASC proposed rule (89 FR 59463 through 59464), we proposed to increase the reporting period from 1 year to 2 years beginning with the CY 2027 program determination.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59463), we explained that under the proposal, the previously finalized 1-year data collection period for the CY 2026 program determination would remain (that is, encounters from January 1, 2024, through December 31, 2024), and then beginning with the CY 2027 program determination, the reporting period would be supplemented with data from the prior calendar year. For example, for the CY 2027 program determination, the reporting period would comprise data from CYs 2024 and 2025 (that is, encounters from January 1, 2024, through December 31, 2025). We noted that, as stated in the CY 2024 OPPS/ASC final rule (88 FR 82066), CMS does not report measures publicly unless it achieves sufficient case volumes to allow for public reporting based on reliability or privacy concerns of the collected data.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59463), we explained that as seen in Table 166, the longer reporting period of 2 years would facilitate greater case volumes for this measure and, subsequently, a larger portion of REHs would have data that could be reported publicly as more REHs attain the Risk-Standardized Hospital Visits Within 7 Days After Outpatient Surgery measure's minimum case threshold for reliability of 30 surgical cases. In addition, REHs reporting on the measure with 2 years of data would have more eligible patients to assess; this increase in eligible cases would also reduce the error estimate, making the confidence interval narrower, that is, increasing the reliability of the calculated measure. We refer readers to the CY 2019 OPPS/ASC final rule (83 FR 59106 through 59107) where we finalized a similar policy to extend the reporting period of the Facility 7-Day Risk-Standardized Hospital Visit Rate After Outpatient Colonoscopy measure in the Hospital OQR Program from 2 to 3 years.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59464), we explained that under the proposal, there would be no gap in public reporting nor delay in providing REHs with data for quality improvement efforts. As this measure is calculated solely from administrative Medicare information, REHs would not have any additional reporting burden associated with a longer reporting period.
We invited public comment on the proposal.
Comment: Several commenters supported the proposal to extend the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure, stating that finalizing the policy would allow a broader range of hospitals to reliably and validly report the measure, provide consumers with ratings for a greater number of facilities, enhance the reliability of the measure results due to the larger sample size, provide hospitals with a more accurate and comprehensive understanding of performance trends over time, allow for improved data analysis and better-informed decisions regarding patient care, and reduce the pressure on hospitals to gather and report data within a shorter window thereby supporting more sustainable and effective reporting practices.
Response: We thank the commenters for their support.
After consideration of the public comments we received, we are finalizing the extension of the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure ( printed page 94428) from 1 year to 2 years, beginning with the CY 2027 program determination, as proposed.
3. Summary of Finalized Program Measure Set Updates
a. Finalized Program Measure Set Beginning With the CY 2027 Program Determination
Table 167 summarizes the finalized REHQR Program measure set and reporting periods beginning with the CY 2027 program determination.
Table 167 includes the previously finalized measure set with updates to reflect the finalized extension of the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure beginning with the CY 2027 program determination, and the three new cross-program health equity measures as detailed in sections XIV.B.1, XIV.B.2, and XIV.B.3, respectively, of this final rule with comment period:
b. Finalized Program Measure Set Beginning With the CY 2028 Program Determination
Table 168 summarizes the finalized REHQR Program measure set and reporting periods beginning with the CY 2028 program determination.
Table 168 includes the previously finalized measure set with updates to reflect the finalized extension of the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure beginning with the CY 2028 program determination, and the three new cross-program health equity measures as detailed in sections XIV.B.1, XIV.B.2, and XIV.B.3, respectively, of this final rule with comment period.
( printed page 94429)D. Administrative Requirements
We refer readers to § 419.95(b) and the CY 2024 OPPS/ASC final rule (88 FR 82074) for our policies regarding administrative requirements previously finalized for the REHQR Program. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
E. Form, Manner, and Timing of Data Submission
1. General Policy
We refer readers to § 419.95(c) and (g) and the CY 2024 OPPS/ASC final rule (88 FR 82074 through 82076) for our general policies regarding: (1) submission of data under the REHQR Program generally; (2) review and correction of submitted data; and (3) extraordinary circumstance exception (ECE) requests for data submission. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
2. Data Submission Policy Following Conversion to REH Status
In the CY 2025 OPPS/ASC proposed rule (89 FR 59465), we noted that with implementation of these general data reporting policies and hospitals continuing to convert to REH status throughout the year, we believed that it was necessary to specify when a hospital that converts to REH status is required to begin reporting data to the REHQR Program. Thus, we proposed that an REH must begin submitting data to the REHQR Program on the first day of the quarter following the date that a hospital has been designated as converted to an REH in accordance with the process outlined in section 1861(kkk) of the Act.
We invited public comment on the proposal.
Comment: Several commenters supported the proposal to require REHs to begin submitting data to the REHQR Program on the first day of the quarter following the date that a hospital has been designated as converted to an REH, stating that this aligns with regulatory requirements and ensures a structured transition for newly designated REHs.
Response: We thank the commenters for their support.
Comment: A commenter recommended that CMS provide additional technical assistance to hospitals during the conversion period to help them prepare for reporting and suggested that CMS develop strategies to mitigate the financial impacts associated with reporting requirements in the first fiscal year following the REH conversion, to support a smoother transition and reduce the burden on these facilities.
Response: Technical assistance for reporting information is available on the QualityNet website ( https://qualitynet.cms.gov/reh) and questions can be submitted via the Quality Question and Answer Tool ( https://cmsqualitysupport.servicenowservices.com/qnet_qa). We have limited the burden associated with reporting by limiting the number and types of measures in the REHQR Program (for example, by primarily implementing measures calculated using Medicare administrative data and measures submitted using aggregate counts).
After consideration of the public comments we received, we are finalizing the data submission policy following conversion to REH status as proposed.
3. Measure-Specific Data Submission and Reporting Requirements
a. Data Submission Requirements for Chart-Abstracted Measures
We refer readers to the CY 2024 OPPS/ASC final rule (88 FR 82074 through 82075) for information regarding chart-abstracted data submission and reporting requirements. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule. ( printed page 94430)
b. HCHE, Screening for SDOH, and Screen Positive Rate for SDOH Measures' Data Submission Requirements and Reporting Requirements
In the CY 2025 OPPS/ASC proposed rule (89 FR 59465 through 59466), to align with the Hospital OQR (80 FR 70521 through 70522) and ASCQR (81 FR 79821 through 79822) Programs, we proposed a web-based submission policy where REHs would submit data for applicable measures once annually using a CMS-approved, web-based, data collection tool available within the Hospital Quality Reporting system.[499] We proposed that, in alignment with the Hospital OQR and ASCQR Programs, REHs would submit data during the period of January 1 to May 15 in the year prior to the affected program determination year. For example, for the CY 2025 reporting period/CY 2027 program determination, the data submission period would be January 1, 2026, to May 15, 2026, covering the performance period of January 1, 2025, to December 31, 2025. Under the review and corrections period provided at § 419.95(c)(3), REHs would be able to enter, review, and correct data submitted during the data submission period.
In the CY 2025 OPPS/ASC proposed rule (89 FR 59465), we proposed that these policies would apply to web-based measures adopted by the REHQR Program, including the following three measures:
- The HCHE measure, beginning with the CY 2025 reporting period/CY 2027 program determination;
- The Screening for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period, followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination; and
- The Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period, followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination.
As discussed in sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period, we are finalizing adoption of the HCHE, Screening for SDOH, and Screen Positive Rate for SDOH measures into the REHQR Program.
We invited public comment on the proposal.
Comment: A commenter supported the proposed policy to align the web-based measure submission policy for the REHQR Program with the Hospital OQR and ASCQR Programs.
Response: We thank the commenter for its support.
After consideration of the public comments we received, we are finalizing the data submission requirements related to the HCHE, Screening for SDOH, and Screen Positive Rate for SDOH measures as proposed.
c. Data Submission Requirements for Claims-Based Measure Data
In addition, we refer readers to section XVI.C.2 of this final rule with comment period where we discuss finalizing a modification to the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery Measure beginning with the CY 2027 program determination. This claims-based measure would continue to be reported in accordance with other claims-based measures, as previously finalized in the CY 2024 OPPS/ASC final rule (88 FR 85075). We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
F. Public Reporting of Measure Data
We refer readers to § 419.95(f) and the CY 2024 OPPS/ASC final rule with comment period (88 FR 82071 through 82074) for our program policy regarding the public reporting of quality data. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
XVII. Ambulatory Surgical Center Quality Reporting (ASCQR) Program
A. Background and Statutory Authority
The Ambulatory Surgical Center Quality Reporting (ASCQR) Program is a pay-for-reporting program intended to improve the quality of care provided to Medicare beneficiaries, facilitate public transparency, and ensure accountability of ambulatory surgical centers (ASCs). Section 1833(i)(7)(A) of the Act authorizes the Secretary to reduce any annual increase under the revised ambulatory surgical center (ASC) payment system by 2.0 percentage points for such year that an ASC that fails to submit required data on quality measures specified by the Secretary in accordance with section 1833(i)(7)(B) of the Act. Section 1833(i)(7)(B) of the Act states that, except as the Secretary may otherwise provide, several of the statutory provisions governing the Hospital Outpatient Quality Reporting (OQR) Program, specifically sections 1833(t)(17)(B) through (E) of the Act, also apply to the services of ASCs under the ASCQR Program in a similar manner to the manner in which they apply to the services of hospital outpatient departments under the Hospital OQR Program. Sections 1833(t)(17)(B) through (E) of the Act generally govern the adoption and replacement of quality measures, the form and manner of submission of data to CMS, and procedures for making the data submitted to CMS available to the public.
We refer readers to the CY 2012 OPPS/ASC final rule (76 FR 74492 through 74494) for a detailed discussion of the program's statutory authority, as well as program requirements codified at 42 CFR part 416, subpart H (§§ 416.300 through 416.330), and the CY 2024 OPPS/ASC final rule (88 FR 82012) for information regarding the program's regulatory history.
1. Previously Finalized Program Measure Sets
We refer readers to the CY 2024 OPPS/ASC final rule (88 FR 82038) for additional information regarding the previously finalized ASCQR Program measure set beginning with the CY 2027 payment determination.
a. Previously Finalized Measure Set Beginning With the CY 2027 Payment Determination
Table 169 summarizes the previously finalized ASCQR Program measures beginning with the CY 2027 payment determination.
( printed page 94431)b. Previously Finalized Measure Set Beginning With the CY 2031 Payment Determination
Table 170 summarizes the previously finalized ASCQR Program measures beginning with the CY 2031 payment determination.
( printed page 94432)B. Program Measure Set Policies
1. Measure Retention
We refer readers to § 416.320 and the CY 2012 OPPS/ASC final rule (76 FR 74504) for our policies regarding measure retention. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
2. Measure Suspension or Removal
We refer readers to § 416.320 and the CY 2019 OPPS/ASC final rule (83 FR 59111 through 59115) for our program policies regarding: (1) general measure removal, suspension, or replacement; and (2) immediate measure removal.
We refer readers to section XIV.C of this final rule with comment period for the discussion of public comments received and our subsequent decision regarding our cross-program proposal to modify the immediate measure removal policy for quality measures adopted for the ASCQR Program.
3. Measure Modification
We refer readers to § 416.325 and the CY 2016 OPPS/ASC final rule (80 FR 70531) for our program policies regarding modifications to previously adopted measures. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
4. Measure Adoption
We refer readers to the CY 2013 OPPS/ASC final rule (77 FR 68493 and 68494) for details regarding program priorities we consider for quality measure selection. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
C. Program Measure Updates
1. Adoption of the Health Equity Quality Measures in the ASCQR Program
We refer readers to sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period for our discussion of our cross-program proposals to adopt the following measures in the ASCQR Program: (1) the Facility Commitment to Health Equity (FCHE) measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination. In these sections, we also discuss the public comments received, our responses thereto, and our final decisions regarding these proposals for the ASCQR Program. As discussed in sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period, ( printed page 94433) we are finalizing these three measures for the ASCQR Program, as proposed.
2. Summary of Finalized Program Measure Set Updates
a. Newly Finalized ASCQR Program Measure Set Beginning With the CY 2027 Payment Determination
Table 171 summarizes the newly finalized ASCQR Program measure set beginning with the CY 2027 payment determination.
( printed page 94434)b. Newly Finalized ASCQR Program Measure Set Beginning With the CY 2031 Payment Determination
Table 172 summarizes the newly finalized ASCQR Program measure set beginning with the CY 2031 payment determination.
3. Comments Regarding the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) Measure
We received many comments requesting that we remove the COVID-19 Vaccination Coverage Among Healthcare Personnel (HCP) from the ASCQR Program. A few commenters recommended, in the absence of removal, that we revise the measure specifications to have data submitted annually. While we appreciate these comments, we note that they fall outside the scope of this rulemaking. We will consider these comments as we continue to evolve the ASCQR Program in the future.
D. Administrative Requirements
We refer readers to § 416.305 and the CY 2016 OPPS/ASC final rule (80 FR 70533 and 70534) for our program policies regarding participation and withdrawal requirements. We did not ( printed page 94435) propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
E. Form, Manner, and Timing of Data Submission
1. General Policy
We refer readers to § 416.310 and the CYs 2017, 2018, and 2021 OPPS/ASC final rules (81 FR 79824 and 79825; 82 FR 59472 through 59475; and 85 FR 86191 and 86192, respectively) for our general program policies regarding: (1) submission of data under the ASCQR Program generally; (2) review and correction of submitted data; and (3) extraordinary circumstance exception (ECE) requests for data submission. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
We also refer readers to the CY 2016 OPPS/ASC final rule (80 FR 70531) for details regarding submission requirements for previously adopted ASCQR Program measures in the ASCQR Program Specifications Manual.
2. Measure-Specific Data Submission and Reporting Requirements
We refer readers to § 416.310 and the CYs 2016, 2022, and 2024 OPPS/ASC final rules (80 FR 70534 through 70536; 86 FR 63905 through 63909; and 88 FR 82041 through 82045, respectively) for information regarding our claims-based, survey-based, and PRO-PM data submission and reporting requirements.
a. Web-Based Measures
(1) CMS-Designated Information System and Proposal for Data Submission for the Facility Commitment to Health Equity (FCHE), Screening for Social Drivers of Health (SDOH), and Screen Positive Rate for SDOH Measures
We refer readers to § 416.310(c)(1), the CY 2017 OPPS/ASC final rule (81 FR 79821 and 79822), the CY 2018 OPPS/ASC final rule (82 FR 59473), and the CY 2024 final rule (88 FR 82039 and 82040) for details regarding submission of web-based data via a CMS-designated information system (currently the Hospital Quality Reporting (HQR) system).
In sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period, we discuss the proposals and public comments received regarding the adoption of: (1) the FCHE measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination.
Consistent with our established data submission requirements (81 FR 79821 and 79822; 82 FR 59473; 88 FR 82039 and 82040), we proposed in the CY 2025 OPPS/ASC proposed rule (89 FR 59471) that ASCs would be required to submit all of the data required to calculate each of these three measures annually using a CMS-approved, web-based, data collection tool available within the HQR System starting January 1 through and including May 15 in the year prior to the applicable payment determination year. As discussed in sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period, we are finalizing the FCHE measure, the Screening for SDOH measure, and the Screen Positive Rate for SDOH measure as proposed.
For the ASCQR Program, the performance period (which we refer to as the CY reporting period) for each measure on which data is submitted using a web-based tool would be January 1 through and including December 31 of the year that is 2 years prior to the applicable payment determination year; and the data submission period would be January 1 through and including May 15 in the calendar year immediately following the CY reporting period and immediately prior to applicable payment determination year. For example, for the CY 2025 reporting period/CY 2027 payment determination, the data submission period would be January 1, 2026, through and including May 15, 2026, covering the performance period of January 1, 2025, through and including December 31, 2025. Pursuant to § 416.310(c)(1)(iii), a review and corrections period runs concurrently with the data submission period. During this timeframe, ASCs would be able to enter, review, and correct data submitted during the data submission period.
We invited public comment on the proposal.
Comment: Several commenters that did not support adoption of the FCHE measure expressed concern over reporting burden, as this timeline coincides with OAS CAHPS and THA/TKA PRO-PM measure compliance. A few commenters expressed that there would not be sufficient time to implement processes, technology, and training needed to successfully report this measure. A few commenters suggested delaying the implementation of this measure or allowing for voluntary reporting.
Response: We acknowledge commenters' concerns regarding resources, including increased data collection burden; however, achieving health equity is an issue which deserves serious focus and rapid action for improvement. Therefore, we do not find it appropriate to delay the implementation of this measure. We believe the benefits of encouraging ASC commitment to health equity outweigh the burden of attestation under this measure.
We note that hospitals participating in the Hospital IQR Program will have already reported data on the similar Hospital Commitment to Health Equity measure for the FY 2025 payment determination (that is, data submitted in FY 2024 representing the FY 2023 performance period) (87 FR 49191 through 49201) before required reporting of the Facility Commitment to Health Equity measure begins within the ASCQR Program, as reporting for this measure would begin with the CY 2027 payment determination. Given the timing of this similar measure's implementation in the Hospital IQR Program, ASCs will have had the opportunity to learn from the experiences of acute care hospitals, including best practices for minimally burdensome assessment of performance on the required domains. We will also monitor measure implementation and data reporting as part of standard program and measure review and will consider updates to the measure if we identify implementation strategies to reduce burden.
Comment: A commenter requested that CMS maintain the Screening for SDOH and the Screen Positive Rate for SDOH measures as voluntary for the ASCQR Program as CMS continues to work through technical reporting challenges, such as publishing guidance on what constitutes a “positive” screen, with providers already reporting the measure in other quality reporting programs. Another commenter noted challenges with the implementation timeline in the ASCQR Program because it does not account for mandatory reporting of the OAS CAHPS Survey measure, which begins in 2025, and does not allow sufficient time for the amount of work that would need to be done for the Screening for SDOH measure, if it were to include screening for interpersonal safety.
Response: We thank the commenters for their feedback and recommendations. As previously discussed in sections XIV.B.2 and XIV.B.3 of this final rule with comment ( printed page 94436) period, the Screening for SDOH and Screen Positive Rate for SDOH measures provide information on the percent of patients receiving care at an ASC who were 18 years or older on the date of service, who were screened for all five HRSNs described in Table 171, and who screened positive for one or more of those HRSNs. For additional information on what constitutes a “positive” screen we refer readers to: https://www.qualityreportingcenter.com/globalassets/2024/04/iqr/17.-sdoh-measure--faqs_april-2024_vfinal508.pdf. We will develop a similar Frequently Asked Questions document for the ASCQR Program as part of providing educational and training materials. This document will be conveyed through routine communication channels, including, but not limited to, issuing memos, emails, and notices on a CMS website.
We will consider feedback about timeline, OAS CAHPS Survey overlap, and reporting in future rulemaking and measure development. We may address related proposals, including the form, manner, and timing of data reporting, in future rulemaking. We reiterate that, with a year of voluntary reporting for these measures, there is time for ASCs to train staff in the collection of data for HRSNs. Additionally, there are a variety of methods, other than audible methods, to screen for HRSNs that increase privacy, such as written or digital methods, including electronically through a patient portal or on tablets. We advise ASCs to ensure that patients feel safe answering questions and to further remind patients that they may opt out of the screening for any reason.
After consideration of the public comments we received, we are finalizing the data submission requirements related to the adoption of the FCHE, Screening for SDOH, and the Screen Positive Rate for SDOH measures in the ASCQR Program as proposed.
We refer readers to sections XIV.B.1, XIV.B.2, and XIV.B.3 of this final rule with comment period for our cross-program proposals and discussion of all public comments received regarding the FCHE, Screening for SDOH, and Screen Positive Rate for SDOH measures.
(2) National Healthcare Safety Network (NHSN)
We refer readers to § 416.310(c)(2) and the CY 2014 OPPS/ASC final rule (78 FR 75139 and 75140) for our policies regarding submission of web-based data via the Centers for Disease Control and Prevention's NHSN. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
F. Public Reporting of Measure Data
We refer readers to § 416.315 and the CY 2018 OPPS/ASC final rule (82 FR 59472) for our program policies regarding public reporting of quality data. We did not propose any changes to these policies in the CY 2025 OPPS/ASC proposed rule.
G. Request for Information (RFI)—Development of Frameworks for Specialty Focused Reporting and Minimum Case Number for Required Reporting
The ASCQR Program promotes informed patient decision-making regarding clinical care across ASC procedures through a robust set of quality measures, data on which the ASCQR Program publicly reports as discussed in section XVII.F of this final rule with comment period. The ASCQR Program's current measure set captures clinical quality across all ASCs, including specialty clinical procedures performed only by a subset of ASCs. Thus, a portion of the ASCQR Program measure set only applies to an ASC if it performs those specialty procedures. Currently, ASCs are required to attest if they do not have cases for a given measure, increasing reporting burden.
We seek to ensure the most meaningful measures apply to each facility, as requiring an ASC to report on measures minimally relevant to their patient population increases burden with minimal benefit. Therefore, we sought comment on two potential future frameworks which would achieve the following outcomes: (1) the addition of case minimums for specialty measure reporting; (2) the removal of the zero case attestation requirement for specialty measures to decrease reporting burden; and (3) the verification of individual measure case counts using claims data to determine which specialty measures would potentially be required for reporting for individual ASCs. Verifying case counts using claims data would allow us to confirm that individual ASCs are reporting on measures meeting or surpassing case minimums.
Under these potential frameworks, we are considering revising the data reporting requirements for the ASCQR Program to only require that ASCs report data to CMS on quality measures that are related to their medical interventions, policies, processes, and procedures, or can be abstracted from claims. These potential frameworks would require ASCs to report measures generally applicable to all ASCQR Program participants and relevant specialty-specific measures, defined as those which evaluate performance on certain specialty clinical procedures performed only by a subset of ASCs.
The current ASCQR Program measure set has seven generally applicable measures for which reporting would be required in both frameworks for all ASCs: four patient safety measures (Patient Burn; Patient Fall; Wrong Site, Wrong Patient, Wrong Procedure, Wrong Implant; All-Cause Hospital Transfer Admission), one general surgery measure (Facility-Level 7-Day Hospital Visits After General Surgery Procedures Performed at Ambulatory Surgical Centers), one vaccination measure (COVID-19 Vaccination Coverage Among Healthcare Personnel), and one patient experience of care survey measure (OAS CAHPS). In addition, in the CY 2025 OPPS/ASC proposed rule (89 FR 59439 through 59449), we proposed the adoption of three new generally applicable measures (FCHE, Screening for SDOH, and Screen Positive Rate for SDOH), which ASCs would also be required to report. As discussed in section XVII.C.1 of this final rule with comment period, we are finalizing these three measures for the ASCQR Program, as proposed.
The specialties addressed by the current ASCQR Program measure set, and the related specialty-specific measures, are described in Table 173. Under our first potential framework, the “Specialty-Select” framework, all ASCs would be required to report all specialty-specific, claims-based measures (currently, three) [500] because these measures are not administratively burdensome to ASCs. Additionally, ASCs would also be required to select a specified number of the remaining non-claims-based specialty-specific measures (currently, five) [501] to report if those measures are applicable to that ASC. We would define the number of ( printed page 94437) non-claims-based specialty-specific measures that ASCs would be required to report in future rulemaking.
To determine if a non-claims-based specialty-specific measure is applicable to an ASC, we are considering the implementation of a case threshold minimum, which we would specify in future rulemaking, for each measure. We would determine if case threshold minimums, defined as the number of cases for a specific measure that must be met or exceeded to potentially require reporting, have been met using claims data. Once an ASC met the measure's case threshold minimum, that measure would become available for that ASC to select to meet reporting requirements. We note that reporting claims-based specialty-specific measures would be required regardless of whether the case threshold minimum is met. In the CY 2025 OPPS/ASC proposed rule (89 FR 59472), we sought comment on the number of non-claims-based specialty specific measures that ASCs should be required to report and what the appropriate threshold for the case threshold minimum should be.
We are considering the use of Medicare Fee-for-Service (FFS) and Medicare Advantage claim volume data to determine which non-claims-based specialty-specific measures have met the specified case threshold minimum (that is, claims information would indicate whether an ASC was performing sufficient case volumes in a specialty area). We note that this threshold would be independent from our “Minimum case volume for program participation” policy, which exempts ASCs with fewer than 240 total Medicare claims per year from participating in the ASCQR Program, as specified at § 416.305(c). The case threshold minimum discussed in this RFI would be applied to individual non-claims-based specialty-specific measures for ASCs required to participate in the ASCQR Program.
For example, if we decide that each ASC must select three out of the five available non-claims-based specialty-specific measures to report, and an ASC surpasses the specified case threshold minimum for all five non-claims-based specialty-specific measures, the ASC would then choose three out of the five non-claims-based specialty-specific measures to report. If an ASC surpasses the specified case threshold minimum for only one or two non-claims-based specialty-specific measures, the ASC will no longer have a choice, and must report all measures meeting the case threshold minimum. If an ASC does not meet the case threshold minimum for any non-claims-based specialty-specific measures, reporting for any of these measures would be voluntary. Under such a framework, ASCs could not utilize the claims-based measures to meet the Specialty-Select reporting requirements nor could ASCs opt-out of reporting these measures. ASCs which do not have one or more cases for a given measure would no longer be required to provide an attestation of having zero cases.
( printed page 94438)Five of these measures are not claims-based and, under this potential framework, would not be applicable or required for all ASCs to report, but would rather be available for selection upon meeting a specified case threshold minimum:
- Unplanned Anterior Vitrectomy;
- Cataracts: Improvement in Patient's Visual Function within 90 Days Following Cataract Surgery (voluntary);
- Normothermia Outcome;
- Risk-Standardized Patient-Reported Outcome-Based Performance Measure (PRO-PM) Following Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) in the ASC Setting (THA/TKA PRO-PM); and
- Endoscopy/Polyp Surveillance: Appropriate Follow-Up Interval for Normal Colonoscopy in Average Risk Patients.
We acknowledge that currently there are few non-claims-based specialty-specific measures from which to choose to report. However, we are interested to learn if such a framework with both mandatory measures applicable to all ASCs and selectable, specialty-specific measures could lay the groundwork for providing higher quality data to patients while ensuring ASCs are not reporting data on measures that are minimally relevant, if not irrelevant, to their patient population.
Regarding this Specialty-Select framework, we requested comment on the following questions:
- Given that ASCs would still be required to report claims-based specialty-specific measures, as these measures are not administratively burdensome to ASCs, and there are currently only five non-claims-based specialty-specific quality measures in the ASCQR Program data set, how many non-claims-based specialty-specific measures should we require ASCs select to report?
- Are there specialty-specific measures that commenters would recommend for development and adoption in the ASCQR Program measure set to create a more robust selection?
- How should we determine what non-claims-based specialty-specific measures would be eligible for a given ASC to select toward meeting reporting requirements? In other words, how can we determine if an ASC meets the minimum case number for a given measure, which would allow the ASC to choose that measure to meet reporting requirements?
As an alternative to the Specialty-Select framework discussed previously, we are considering requiring reporting for all non-claims-based specialty-specific measures for which case counts reach a specified case threshold minimum. This case threshold minimum would not apply to claims-based specialty-specific measures, as their reporting would be mandatory since these measures are not administratively burdensome to ASCs. Under this alternative framework, mandatory data reporting for non-claims-based specialty-specific measures would occur only if an ASC met established case threshold minimums. For example, if an ASC has 30 or more qualifying patients for the measure during the applicable reporting period, which is the current minimum case threshold required for public reporting for some measures, the ASC would be required to submit data for these measures. Likewise, if an ASC has fewer than 30 patients for the measure, data reporting on the measure would be voluntary. This framework could be termed a Specialty Threshold framework and would differ from the previously discussed Specialty-Select framework as an ASC would be required to report on all non-claims-based specialty-specific measures for which the ASC reaches the case threshold minimum.
Regarding both the Specialty Threshold framework and the Specialty-Select framework, we requested comment on the following questions:
- Would use of Medicare Fee-for-Service (FFS) claim volume be sufficient for determining minimum case volumes?
- Should Medicare Advantage claim volume or service data be included when determining case volume thresholds for reporting a measure?
- Do commenters recommend any processes that could be followed or analyses we could conduct to determine case minimums?
We invited public comment on both the Specialty-Select framework and the alternative Specialty Threshold framework for potential inclusion in the ASCQR Program.
Comment: Several commenters supported the implementation of either the Specialty-Select framework or the Specialty-Threshold framework, as they would decrease reporting burden on ASCs to ensure the most meaningful measures apply to each facility. A few commenters specifically supported a Specialty-Threshold framework instead of the Specialty-Select framework to reduce the complexity that may result from measure selection. A commenter specifically supported the use of case minimums for specialty reporting.
Response: We thank commenters for their support of the specialty-focused frameworks. We also believe that these frameworks would reduce burden while ensuring ASCs are reporting on measures most applicable to their performed procedures.
Comment: A few commenters provided feedback on the inclusion of Medicare Advantage data when determining case volume thresholds. A few commenters recommended that CMS only use FFS data and exclude Medicare Advantage service or volume data when determining the applicable case volume thresholds. A commenter further added that ASCs offer a diverse range of services, and the patient population can vary significantly from one center to another. This commenter expressed concern that including Medicare Advantage claim volume in the threshold determination could lead to a misrepresentation of the services most commonly provided by an ASC.
Response: We thank the commenters for their input and appreciate the many thoughtful responses on the use of administrative data that CMS already receives when determining case volume thresholds. We will consider these comments in any future rulemaking on this topic.
Comment: Several commenters offered recommendations for the specialty-focused frameworks. A few commenters recommended measure additions and modifications to the ASCQR Program measure set to support a specialty-focused framework. A few commenters recommended the inclusion of the Toxic Anterior Segment Syndrome (TASS) measure in the ASCQR Program. Another commenter recommended areas for measure development to support a specialty framework, including ASC discharges with subsequent unplanned hospital visits, pain management, and surgical site infections (SSIs). This commenter added that the specialty-specific suitability of targeted services performed in ASCs should be evaluated, as there are currently procedures and treatments that practitioners widely agree are inappropriately used and should be eliminated. A commenter recommended the development of condition or procedure specific PROMs or PRO-PMs for patient goal attainment and patient experience. Another commenter expressed that a number of currently included ASCQR Program measures were not developed for measuring facility-level performance. This commenter recommended that all measures considered for inclusion in the ASCQR Program should first be tested at the facility level for analysis.
Regarding how CMS would determine what non-claims-based, specialty- ( printed page 94439) specific measures would be eligible for selection, a commenter recommended that there should be a process and analysis managed by CMS to determine an appropriate case threshold for reporting non-claims-based, specialty-specific measures with adequate reliability. This commenter further recommended that reporting of non-claims-based, specialty-specific measures should be mandatory only if a facility meets the minimum threshold requirement during the previous and current performance year. The commenter suggested that if the facility only meets the requirement in the current year, then voluntary reporting should be permitted.
Response: We thank the commenters for their input and appreciate the many recommendations received regarding the specialty-focused frameworks. We will take these comments into consideration as appropriate in any future rulemaking on this topic.
Comment: A few commenters requested clarification regarding the Specialty-Select and Specialty-Threshold frameworks. A commenter requested clarification on when the case count for each specialty measure would be calculated. The commenter further requested clarification on the potential consequences of not meeting the specified case threshold for the specified number of specialty measures ASCs would be required to report. Another commenter raised concern that ASCs may not know definitively which measures they would be required to report on until after the end of the reporting period. This commenter requested clarification on when CMS plans to release this information. Furthermore, a commenter requested clarification on whether measure case counts would include Medicare Advantage data and whether the counts would only include paid claims.
Response: We thank the commenters for highlighting aspects of these frameworks we could clarify in any future rulemaking. We will consider these comments moving forward.
Comment: A few commenters raised concern over the specialty-focused frameworks. A few commenters stated that the ASCQR Program has a small measure set, and raised concerns that these frameworks may inhibit a patient's ability to compare ASCs, or an ASC's ability to compare their performance to other ASCs, as different ASCs may report different measures. These commenters noted that this disparity in quality information may result in the unintended consequence of people electing to receive care in hospital settings for procedures that could be performed at more cost-efficient ASCs. Additionally, a commenter noted that selecting non-claims-based specialty-specific measures may be challenging, as there are few in the measure set. Furthermore, a few commenters raised concerns that these frameworks could increase burden for multi-specialty ASCs, as they would be required to report on more specialty measures than single-specialty ASCs. A few commenters raised concerns that if an ASC has a choice of which measures to report, they will choose the measures which will yield the most favorable results, creating an inaccurate representation of quality.
Furthermore, a few commenters raised concerns about the minimum case threshold, stating that volume and quality are positively correlated. These commenters stated that the exclusion of data from low-volume facilities may result in reduced reporting in clinical areas where performance is worse. A commenter raised concern over the burden of having to collect data regardless of if the case threshold minimum is met, as ASCs would only find out if reporting was mandatory after the relevant reporting period. Another commenter raised concern that the implementation of a specialty framework will be inconsistent with other programs. This commenter further stated that a specialty approach would lead to redundant reporting since Qualified Clinical Data Registries already report specialty data to the Merit-based Incentive Payment System for eligible clinicians.
Response: We thank commenters for raising concerns about the specialty-focused frameworks. We appreciate commenters' identification of areas that may need further consideration and will review these comments during future rulemaking.
Comment: Regarding the current zero-case attestation requirement, a commenter recommended the implementation of automated data validation checks to identify incomplete fields, to alert an ASC of missing information during the completion of the zero-case attestation requirement.
Response: We thank commenters for their feedback and will consider this feedback for any future rulemaking. We would like to note that our HQR system already has mechanisms in place which ensure facilities are aware of missing data entry information. These mechanisms include an online lookup tool where facilities can download data entry reports, as well as phone call and fax reminders.
Comment: A commenter added that public reporting of ASC data should be available at the facility level, rather than the system level.
Response: We thank commenters for their feedback and will consider this feedback for any future rulemaking. We would like to remind commenters that public reporting of ASC data is already captured at the facility level, as opposed to the system level.
Data that an ASC submitted for the ASCQR Program are displayed publicly on a CMS website. Before these data are published, ASCs can review the data. ASC data are publicly displayed by the National Provider Identifier (NPI) when data are submitted by the NPI. When data are submitted by the CCNS, we will publicly display ASC data by the CMS Certification Number (CCN).
H. Payment Reduction for ASCs That Fail To Meet the ASCQR Program Requirements
1. Statutory Background
We refer readers to the CY 2012 OPPS/ASC final rule with comment period (76 FR 74492 through 74493) for a detailed discussion of the statutory background regarding payment reductions for ASCs that fail to meet the ASCQR Program requirements.
2. Policy Regarding Reduction to the ASC Payment Rates for ASCs That Fail To Meet the ASCQR Program Requirements for a Payment Determination Year
The national unadjusted payment rates for many services paid under the ASC payment system are equal to the product of the ASC conversion factor and the scaled relative payment weight for the APC to which the service is assigned. For CY 2025, the ASC conversion factor is equal to the conversion factor calculated for the previous year updated by the productivity-adjusted hospital market basket update factor. The productivity adjustment is set forth in section 1833(i)(2)(D)(v) of the Act. The productivity-adjusted hospital market basket update is the annual update for the ASC payment system for a 5-year period (CY 2019 through CY 2023), which was extended an additional 2 years (through CY 2025) in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81960). Under the ASCQR Program, in accordance with section 1833(i)(7)(A) of the Act and as discussed in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68499), any annual increase in certain payment rates under the ASC payment system shall be reduced by 2.0 percentage points for ASCs that fail to meet the reporting requirements of the ASCQR ( printed page 94440) Program. This reduction applied beginning with the CY 2014 payment rates (77 FR 68500). For a complete discussion of the calculation of the ASC conversion factor and our finalized proposal to update the ASC payment rates using the inpatient hospital market basket update for CYs 2019 through 2023, we refer readers to the CY 2019 OPPS/ASC final rule with comment period (83 FR 59073 through 59080).
In the CY 2013 OPPS/ASC final rule with comment period (77 FR 68499 through 68500), in order to implement the requirement to reduce the annual update for ASCs that fail to meet the ASCQR Program requirements, we finalized the following policies: (1) to calculate a full update conversion factor and an ASCQR Program reduced update conversion factor; (2) to calculate reduced national unadjusted payment rates using the ASCQR Program reduced update conversion factor that would apply to ASCs that fail to meet their quality reporting requirements for that calendar year payment determination; and (3) that application of the 2.0 percentage point reduction to the annual update may result in the update to the ASC payment system being less than zero prior to the application of the productivity adjustment. The ASC conversion factor is used to calculate the ASC payment rate for services with the following payment indicators (listed in Addenda AA and BB to the proposed rule, which are available via the internet on the CMS website): “A2,” “G2,” “P2,” “R2” and “Z2,” as well as the service portion of device-intensive procedures identified by “J8” (77 FR 68500). We finalized our proposal that payment for all services assigned the payment indicators listed would be subject to the reduction of the national unadjusted payment rates for applicable ASCs using the ASCQR Program reduced update conversion factor (77 FR 68500).
The conversion factor is not used to calculate the ASC payment rates for separately payable services that are assigned status indicators other than payment indicators “A2,” “D2”, “G2,” “J8,” “P2,” “R2” and “Z2.” These services include separately payable drugs and biologicals, pass-through devices that are contractor-priced, brachytherapy sources that are paid based on the OPPS payment rates, and certain office-based procedures, radiology services and diagnostic tests where payment is based on the PFS nonfacility PE RVU-based amount, and a few other specific services that receive cost-based payment (77 FR 68500). As a result, we also finalized our proposal that the ASC payment rates for these services would not be reduced for failure to meet the ASCQR Program requirements because the payment rates for these services are not calculated using the ASC conversion factor and, therefore, are not affected by reductions to the annual update (77 FR 68500).
Office-based surgical procedures (generally those performed more than 50 percent of the time in physicians' offices) and separately paid radiology services (excluding covered ancillary radiology services involving certain nuclear medicine procedures or involving the use of contrast agents) are paid at the lesser of the PFS nonfacility PE RVU-based amounts or the amount calculated under the standard ASC ratesetting methodology. Similarly, in the CY 2015 OPPS/ASC final rule with comment period (79 FR 66933 through 66934), we finalized our proposal that payment for certain diagnostic test codes within the medical range of CPT codes for which separate payment is allowed under the OPPS will be at the lower of the PFS nonfacility PE RVU-based (or technical component) amount or the rate calculated according to the standard ASC ratesetting methodology when provided integral to covered ASC surgical procedures. In the CY 2013 OPPS/ASC final rule with comment period (77 FR 68500), we finalized our proposal that the standard ASC ratesetting methodology for this type of comparison would use the ASC conversion factor that has been calculated using the full ASC update adjusted for productivity. This is necessary so that the resulting ASC payment indicator, based on the comparison, assigned to these procedures or services is consistent for each HCPCS code, regardless of whether payment is based on the full update conversion factor or the reduced update conversion factor.
For ASCs that receive the reduced ASC payment for failure to meet the ASCQR Program requirements, we have noted our belief that it is both equitable and appropriate that a reduction in the payment for a service should result in proportionately reduced coinsurance liability for beneficiaries (77 FR 68500). Therefore, in the CY 2013 OPPS/ASC final rule with comment period (77 FR 68500), we finalized our proposal that the Medicare beneficiary's national unadjusted coinsurance for a service to which a reduced national unadjusted payment rate applies will be based on the reduced national unadjusted payment rate.
In the CY 2013 OPPS/ASC final rule with comment period, we finalized our proposal that all other applicable adjustments to the ASC national unadjusted payment rates would apply in those cases when the annual update is reduced for ASCs that fail to meet the requirements of the ASCQR Program (77 FR 68500). For example, the following standard adjustments would apply to the reduced national unadjusted payment rates: the wage index adjustment; the multiple procedure adjustment; the interrupted procedure adjustment; and the adjustment for devices furnished with full or partial credit or without cost (77 FR 68500). We believe that these adjustments continue to be equally applicable to payment for ASCs that do not meet the ASCQR Program requirements (77 FR 68500).
In the CY 2015 through CY 2024 OPPS/ASC final rules with comment period, we did not make any other changes to these policies. We proposed to continue applying these policies for the CY 2025 reporting period/CY 2027 payment determination and for subsequent years.
We did not receive any public comments on our proposal. We are finalizing as proposed the continuation of these policies for the CY 2025 reporting period/CY 2027 payment determination and for subsequent years.
XVIII. Medicaid Clinic Services Four Walls Exceptions
A. Background
Under section 1902(a)(10) of the Act, States may offer certain Medicaid benefits, at State option, to categorically needy and medically needy Medicaid beneficiaries, as described in that section of the statute. Clinic services are one of these optional benefit categories. Section 1905(a)(9) of the Act, as amended by section 4105 of part 1 of subtitle B of title IV of the Omnibus Budget Reconciliation Act of 1987 (OBRA '87, Pub. L. 100-203), defines clinic services as services furnished by or under the direction of a physician, without regard to whether the clinic itself is administered by a physician, including such services furnished outside the clinic by clinic personnel to an eligible individual who does not reside in a permanent dwelling or does not have a fixed home or mailing address (hereinafter referred to as “individuals who are unhoused”).[502 503]
( printed page 94441)The regulation implementing section 1905(a)(9) of the Act, 42 CFR 440.90, includes certain conditions and limitations on Medicaid coverage of clinic services. Specifically, § 440.90 defines clinic services as preventive, diagnostic, therapeutic, rehabilitative, or palliative services that are furnished by a facility that is not part of a hospital but is organized and operated to provide medical care to outpatients.[504] Section 440.90 further provides that clinic services include two types of services furnished to outpatients, listed at § 440.90(a) and (b). The first type of services included in the benefit, under § 440.90(a), is services furnished at the clinic (hereinafter referred to as the “four walls” requirement) by or under the direction of a physician or dentist. Section 440.90(b) implements the statutory language providing that clinic services also include services furnished outside the clinic, by clinic personnel under the direction of a physician, to an eligible individual who is unhoused. In section 4320 of the State Medicaid Manual, we explained that if a State elects to cover clinic services, the State may choose the type of clinics or clinic services that are covered.[505]
We added § 440.90(b) in 1991, after Congress added the language about services furnished outside the clinic to individuals who are unhoused to section 1905(a)(9) of the Act in OBRA ’87.[506] In the preamble to that rule, we explained that clinic services have always been limited to people who go to the clinic (or a satellite location) and get the services onsite, and that the exception added by OBRA ’87 represents an exception to the general coverage requirement for services to be furnished on the premises of the clinic. Further, we explained our view that Congress ratified the requirement that other clinic services must be furnished onsite by establishing an explicit exception to the requirement that clinic services be furnished onsite in order to be covered. CMS has long interpreted the exception to the four walls requirement at § 440.90(b) to be mandatory for States that opt to cover the clinic services benefit. We reiterated CMS's longstanding interpretation that section 1905(a)(9) of the Act and § 440.90 establish a four walls requirement in a frequently asked questions document that we published on January 18, 2017 (hereinafter referred to as “the January 18, 2017 FAQ”), to supplement State Health Official letter number 16-002, Federal Funding for Services “Received Through” an IHS/Tribal Facility and Furnished to Medicaid-Eligible American Indians and Alaska Natives.[507 508]
The Medicaid clinic services benefit is distinct from the Medicaid federally qualified health center (FQHC) services benefit and the Medicaid rural health clinic (RHC) services benefit. The Medicaid FQHC services benefit is defined at section 1905(a)(2)(C) of the Act, and FQHCs and FQHC services are further defined at section 1905( l)(2) of the Act. The Medicaid RHC services benefit is defined at section 1905(a)(2)(B) of the Act, and RHCs and RHC services are further defined at section 1905( l )(1) of the Act. Unlike the clinic services benefit, which is an optional benefit for States, the FQHC and RHC benefits are mandatory for categorically needy Medicaid beneficiaries under section 1902(a)(10) of the Act. In addition, there is no Federal four walls requirement under the Medicaid FQHC or RHC services benefits, unlike the clinic services benefit. Federal Medicaid law does not prevent States from covering Medicaid FQHC and RHC services provided outside of the four walls of an FQHC or RHC.[509]
On January 28, 2021, President Biden signed Executive Order (E.O.) 14009, “Strengthening Medicaid and the Affordable Care Act,” which established the policy objective to protect and strengthen Medicaid and the Affordable Care Act and to make high-quality health care accessible and affordable for every American, and directed executive departments and agencies to review existing regulations, orders, guidance documents, and policies to determine whether such agency actions are inconsistent with this policy.[510] As part of this review of existing policies, E.O. 14009 directed Federal agencies to consider whether to suspend, revise, or rescind agency actions considered inconsistent with this objective. On April 5, 2022, E.O. 14070, “Continuing to Strengthen Americans' Access to Affordable, Quality Health Coverage,” directed Federal agencies with responsibilities related to Americans' access to health coverage to review agency actions to identify ways to continue to expand the availability of affordable health coverage, to improve the quality of coverage, to strengthen benefits, and to help more Americans enroll in quality health coverage.[511]
On November 6, 2000, President Clinton signed E.O. 13175, “Consultation and Coordination With Indian Tribal Governments,” which recognizes the unique legal relationship between the United States and Indian Tribal governments and, to strengthen this government-to-government relationship and support Tribal sovereignty and self-determination, charges all executive departments and agencies with engaging in meaningful and timely consultation with Tribal officials in the development of Federal policies that have Tribal implications.[512] On January 26, 2021, President Biden issued a “Memorandum on Tribal Consultation and Strengthening Nation-to-Nation Relationships,” which reaffirms E.O. 13175's directive to engage in regular, meaningful, and robust consultation with Tribal officials in the development ( printed page 94442) of Federal policies that have Tribal implications.[513]
Consistent with E.O. 13175, CMS issued a Tribal Consultation policy in 2011 and updated it in 2015 for the purpose of building meaningful relationships with Indian Tribes and to establish a clear, concise and mutually acceptable process through which consultation can take place between CMS and Tribes.[514] [515] As one of its core principles, the policy provides that, because Congress amended titles XVIII and XIX of the Act to authorize Indian Health Service (IHS) and Tribal health programs to bill Medicare and Medicaid, “[t]he involvement of Indian tribes in the development of CMS policy is crucial for mutual understanding and development of culturally appropriate approaches to improve greater access to CMS programs for American Indians and Alaska Natives (AI/ANs), to enhance health care resources to IHS and tribal health programs, and to contribute to overall improved health outcomes for American Indians.” As part of its government-to-government relationship with the Tribes, CMS has engaged in meaningful consultation with Tribes and Tribal leaders, the CMS Tribal Technical Advisory Group (TTAG), and the HHS Secretary's Tribal Advisory Committee (STAC) regarding concerns about the impact that the four walls requirement could have on IHS/Tribal clinics and AI/AN beneficiaries' access to health care when a grace period currently in place for IHS/Tribal clinics (as discussed later in this section of the preamble) ends. As part of this consultation, Tribes requested a permanent exemption from the four walls requirement for IHS/Tribal clinics.
IHS, a Federal agency within the Department of Health and Human Services, is responsible for furnishing comprehensive, culturally appropriate health services to, as of April 2024, almost $2.8 million AI/ANs who are eligible for services from IHS, per regulations at 42 CFR part 136, as well as other individuals whom IHS or Tribes are authorized to serve under 25 U.S.C. 1680c.[516] IHS's provision of health services to its beneficiaries stems from the special government-to-government relationship between the Federal Government and Indian Tribes. The Federal Government's relationship with Tribes is based on Article I, section 8 of the Constitution, and has been given form and substance by numerous treaties, statutes, Supreme Court decisions, and Executive orders. The IHS delivery system includes hospitals and clinics that are owned and operated by IHS, owned by IHS and tribally-operated as authorized by the Indian Self-Determination and Education Assistance Act (ISDEAA, Pub. L. 93-638 (as amended)), or owned and operated by Tribes and Tribal organizations as authorized by the ISDEAA.[517] We refer to these three kinds of facilities in our discussions of the proposed amendments to § 440.90 as “IHS/Tribal facilities” or, when referring to circumstances where these facilities operate as Medicaid clinic services providers, “IHS/Tribal clinics.” [518] Section 1911 of the Act and implementing regulations at § 431.110 provide that a facility of IHS, whether operated by IHS or by a Tribe or Tribal organization (CMS has interpreted similar language in section 1905(b) of the Act to refer to all three kinds of IHS/Tribal facilities described previously),[519] may participate in the Medicaid program subject to the conditions and requirements generally applicable under Title XIX of the Act. Many IHS/Tribal facilities are covered and paid as clinic services providers in the Medicaid program. Under section 1903(a)(1) of the Act, the Federal Government is required to match State expenditures for medical assistance at the Federal Medical Assistance Percentage (FMAP), which is defined at section 1905(b) of the Act to be 100 percent for State expenditures for Medicaid-covered services received through an IHS facility whether operated by IHS or by a Tribe or Tribal organization (which, again, CMS has interpreted to refer to all three kinds of IHS/Tribal facilities described previously). Under CMS's longstanding interpretation of section 1905(b) of the Act, this 100 percent FMAP is available only for State expenditures on services received through an IHS/Tribal facility (such as a clinic) by AI/AN Medicaid beneficiaries. State expenditures on services furnished by an IHS/Tribal facility to other individuals are not matched by the Federal Government at 100 percent, but rather at the State's regularly applicable FMAP rate.
As part of our Center for Medicaid & CHIP Services (CMCS) Mental Health and Substance Use Disorder Action Plan published in July 2023, we are pursuing strategies to increase access to prevention and treatment, engagement in care, and improve quality of care for beneficiaries with behavioral health disorders.[520] Behavioral health disorders include both substance use disorders and mental health disorders. Medicaid plays a crucial role in financing health care for individuals with behavioral health disorders and is the largest payer of behavioral health services.[521] There are no Federal requirements for States to cover services furnished by behavioral health clinics or any specific types of behavioral health clinics under the clinic services benefit. However, we are aware that approximately 16 States cover services provided by behavioral health clinics of varying types under the clinic services benefit, such as Community Mental Health Centers certified under the Medicare Conditions of Participation at 42 CFR part 485, subpart J, substance use disorder clinics, or mental health clinics.
We released a framework for advancing health care in rural, Tribal, and geographically isolated communities in November 2022.[522] Our framework focuses on six priorities, ( printed page 94443) including expanding access to comprehensive health care coverage, benefits, and services and supports to individuals who live in these communities. Medicaid plays an important role in financing health care in rural areas, as nearly a quarter of individuals under age 65 who live in rural areas are covered by Medicaid. Importantly, Medicaid also provides critical access to care for individuals in rural areas who are older or disabled, as more than one in five residents of rural areas (approximately 22 percent) are dually enrolled in Medicaid and Medicare.[523] There are no Federal requirements under the clinic services benefit governing how States should provide coverage of services furnished specifically by clinics located in rural areas under that benefit—the Federal requirements that apply generally to that benefit, including the four walls requirement, also apply to services furnished by clinics in rural areas. A State may cover Medicaid clinic services provided by various types of clinics located in rural areas, such as primary care clinics, behavioral health clinics, surgical clinics, and other types of clinics. As noted earlier in this section, the Medicaid RHC services benefit is different from the Medicaid clinic services benefit and does not include a four walls requirement under Federal Medicaid law; thus, facilities that qualify as RHCs under Federal Medicaid law could provide Medicaid services under the RHC services benefit, including outside of the four walls.
Section 1902(a)(30)(A) of the Act requires that Medicaid payments for services be consistent with efficiency, economy, and quality of care, and be sufficient to enlist enough providers so that care and services are available under the plan at least to the extent that such care and services are available to the general population in the geographic area. Under this requirement, States generally have significant latitude in setting payment methodologies and rates for covered services, and there is no specific payment methodology required for clinic services, although regulations at § 447.321 require the application of upper payment limits for clinics that are not IHS/Tribal clinics. States generally pay for clinic services via a facility rate. They typically adopt, as the payment rate for Medicaid clinic services furnished by IHS/Tribal clinics, the Outpatient per Visit Rate (excluding Medicare) that IHS establishes for services provided by IHS facilities to Medicaid beneficiaries and for certain other Federal programs. This rate, and a set of three other rates for Medicare outpatient visits and certain inpatient services, are frequently referred to collectively as the IHS all-inclusive rates (AIRs), and therefore this IHS Outpatient per Visit Rate (excluding Medicare) is hereinafter referred to as the “AIR.” [524] In contrast, States generally pay for Medicaid benefits provided by individual practitioners, such as the physician services benefit, at a professional fee schedule rate under the Medicaid State plan.
As we noted in the January 18, 2017 FAQ, CMS recognized in 2017 that IHS/Tribal clinics were providing services outside of the four walls, including to individuals to whom the existing statutory and regulatory exception does not apply, and that States were paying for these services at the clinic services rate (which in all or nearly all cases is the AIR). In the January 18, 2017 FAQ, we announced a 4-year grace period to January 30, 2021, to allow States time to come into compliance with the four walls requirement for IHS/Tribal clinics. On January 15, 2021, due to the COVID-19 Public Health Emergency (PHE), CMS issued a CMCS Informational Bulletin (CIB) announcing an extension of the four walls grace period to October 31, 2021. CMS issued subsequent CIBs on October 4, 2021, and September 8, 2023, announcing further extensions of the grace period to nine months from when the COVID-19 PHE ended, and February 11, 2025, respectively.[525]
Since the release of the January 18, 2017 FAQ and throughout the grace period, we have heard from Tribes, the CMS TTAG, and the HHS STAC, that the four walls requirement will create barriers in access to care for Medicaid beneficiaries who receive care from IHS/Tribal clinics after the grace period expires. Tribes, the TTAG, and the STAC have requested that CMS eliminate the four walls requirement for IHS/Tribal clinics. In addition to these requests, CMS has received a handful of other requests from States to allow exceptions to the four walls requirements for clinics that serve vulnerable populations. For example, we received one section 1115 demonstration request to cover clinic services outside of the four walls for behavioral health clinics, under which the State sought to use the requested section 1115 demonstration authority to improve access to and retention in behavioral health treatment. In addition, we received inquiries from States seeking to cover, under the clinic services benefit, mobile crisis services provided by behavioral health clinics to individuals experiencing a behavioral health crisis, but we advised those States that we could not approve coverage of mobile crisis services under the clinic services benefit due to the four walls requirement.
The proposed rule aimed to address the concerns we have heard from Tribes, the TTAG, the STAC, States, and other interested parties. It aimed to fulfill E.O.s 14009 and 14070 by helping States to strengthen and improve access to clinic services. It also helped to fulfill E.O. 13175 by recognizing the United States' unique legal relationship with Tribes and by responding to advice and input received from Tribes through consultation. In addition, we believe the proposed rule is consistent with our strategies, goals, and objectives to advance health equity and improve health care access for Tribal, behavioral health, and rural populations as described in our CMCS Mental Health and Substance Use Disorder Action Plan and CMS Framework for Advancing Health Care in Rural, Tribal, and Geographically Isolated Communities.
Consistent with our statutory authority at section 1905(a)(9) of the Act, we proposed to add three exceptions to the four walls requirement at § 440.90, for the reasons set forth in section XVIII.B of the proposed rule. First, we proposed to add an exception for clinic services furnished by IHS/Tribal clinics. Second, we proposed to add an exception for clinic services furnished by a clinic that is primarily organized for the care and treatment of outpatients with behavioral health disorders, including mental health and substance use disorders. Third, we ( printed page 94444) proposed to add an exception for clinic services furnished by a clinic located in a rural area (and that is not an RHC, which could already provide services covered under a separate Medicaid benefit). We proposed to make the exception for clinic services furnished by IHS/Tribal clinics a mandatory component of the clinic benefit and to make the exceptions for clinic services furnished by behavioral health clinics and clinics located in rural areas optional for States.
B. Summary of the Medicaid Clinic Services Four Walls Exceptions Proposed Provisions, Public Comments and Responses to Comments
On July 22, 2024, the CY 2025 OPPS/ASC proposed rule (89 FR 59186) was published in the Federal Register . In response to the proposed exceptions, we received 96 public comments. Commenters included individuals, State Medicaid agencies, health care provider associations, individual health care providers, Tribes, Tribal organizations, urban Indian organizations (UIOs), health plans and associations, advocacy groups, academic and research organizations, and other health organizations. In this section, we provide a summary of the proposals, a summary of the public comments received, our responses to the public comments, and the policies we are finalizing for the Medicaid clinic services benefit.
1. General Comments
Comment: One commenter recommended that the final rule include a severability provision to clarify that the Medicaid clinic services benefit four walls exceptions at § 440.90 are not connected to other provisions contained in the OPPS/ASC final rule, so that if any other provision in the OPPS/ASC final rule is held to be invalid or unenforceable it would not apply to the Medicaid clinic services benefit four walls exceptions.
Response: We thank the commenter for their recommendation that the final rule include a severability provision to clarify that the Medicaid clinic services benefit four walls exceptions at § 440.90 are not connected to other provisions contained in the OPPS/ASC final rule. We agree with the commenter and have included language in the supplementary information section of the final rule explaining our intent that if any provision in the OPPS/ASC final rule is held to be invalid or unenforceable, the remaining provisions that could function independently should take effect and be given the maximum effect permitted by law.
Comment: Many commenters expressed gratitude and support for CMS's proposed efforts to mitigate operational burdens under the proposed rule, such as not proposing to require behavioral health clinics to verify that an individual has a behavioral health diagnosis or that clinics located in rural areas verify that an individual lives in a rural area.
Response: We appreciate the comments in support of our proposed efforts to mitigate operational burdens.
Comment: Many commenters thanked CMS for including a clarifying footnote that the Consolidated Appropriations Act, 2024, Division G, Title I, section 209 (Pub. L. 118-42) amended section 1905(a) of the Act to establish a certified community behavioral health clinic (CCBHC) services benefit effective March 9, 2024; the footnote indicated that the CCBHC services benefit is distinct from the clinic services benefit and that there is no four walls requirement for the CCBHC services benefit under Federal Medicaid law.
Response: We appreciate the comments thanking us for clarifying that the Medicaid CCBHC services benefit is distinct from the Medicaid clinic services benefit and that there is no four walls requirement for the CCBHC services benefit under Federal Medicaid law.
Comment: Three commenters expressed concerns with the physician direction requirement, with two of the comments expressing that the physician direction requirement may limit access to care for individuals who receive services from IHS/Tribal clinics. One commenter recommended that CMS defer to States and providers in determining the meaning of the physician direction requirement for the Medicaid clinic services benefit and indicated that the current definition is overly stringent and reduces access to care.
Another commenter wanted to know if CMS defined “under the direction of a physician” in guidance and recommended that CMS reinterpret the physician direction requirement to include non-physician licensed practitioners; the commenter explained that they believed CMS has the statutory authority to allow clinic services to be furnished under the direction of non-physician licensed practitioners because the current regulation at § 440.90 allows clinic services to be under the direction of a dentist despite not being included in section 1905(a)(9) of the Act.
Finally, the third commenter recommended CMS amend the final rule to not apply the physician direction requirement or other clinic services benefit requirements to IHS/Tribal clinics.
Response: We thank the commenters for sharing their concerns about the physician direction requirement. As we noted in section XVIII of the proposed rule (89 FR 59186), section 1905(a)(9) of the Act, as amended by section 4105 of part 1 of subtitle B of title IV of the Omnibus Budget Reconciliation Act of 1987 (OBRA '87, Pub. L. 100-203), defines clinic services as “services furnished by or under the direction of a physician, without regard to whether the clinic itself is administered by a physician, including such services furnished outside the clinic by clinic personnel to an eligible individual who does not reside in a permanent dwelling or does not have a fixed home or mailing address.” Because all services covered under the clinic services benefit are required by statute to be furnished by or under the direction of a physician, we proposed to include language in each of the proposed exceptions specifying that services subject to the exception would have to be furnished under the direction of a physician, to make that requirement clear.
Physician direction has been a longstanding component of the clinic benefit. We do not have the statutory authority to eliminate the requirement that clinic services must be furnished by or under the direction of a physician. In addition, we did not propose changes to that requirement, so we are finalizing this rulemaking without any changes to the meaning of “under the direction of a physician.”
Comment: Two commenters supported CMS's goal of expanding access but expressed concern about the Federal and State budgetary impacts of the proposed policies, given that the policies may increase provider payment rates. The commenters recommended that CMS reinforce that it is important for States to meet the statutory requirement that capitated rates to Medicaid managed care plans be made on an actuarially sound basis during the annual rate setting process or as a mid-year rate update if warranted when payments for clinic services are made through managed care.
Response: We thank the commenters for their comments. We acknowledge that adding additional exceptions to the four walls requirement for IHS/Tribal clinics, and (at State option) behavioral health clinics and clinics located in rural areas is likely to have a Federal and State budgetary impact. As we noted in our economic analysis in section XXVI of the proposed rule, the total estimated impact of the proposed regulation over five years is $1.18 ( printed page 94445) billion, including Federal impact of $1.15 billion and State impact of $30 million. However, as we also described in section XXVI of the proposed rule, the changes to the Medicaid clinic services benefit are expected to benefit Medicaid beneficiaries, Tribes, and States by improving access to care for the populations served by IHS/Tribal clinics, behavioral health clinics, and clinics in rural areas.
When benefits are provided through a managed care delivery system, States must meet statutory and regulatory requirements for setting actuarially sound capitation rates that are projected to provide for all reasonable, appropriate, and attainable costs that are required under the terms of the contract and for the operation of the managed care plan for the time period and the population(s) covered under the terms of the contract. We remind States of the importance of meeting these requirements. States and their actuaries should assess if the programmatic changes under this final rule would materially impact actuarially sound capitation rates, and if so the State and its actuary should account for this programmatic change appropriately in developing capitation rates in accordance with 42 CFR 438.4 and 438.5.
Comment: Two commenters recommended that CMS ensure that all populations that are exempt from the four walls requirement receive the full spectrum of physical and behavioral health care.
Response: We appreciate the commenters' recommendation that we ensure that all populations that are exempt from the four walls requirement receive the full spectrum of physical and behavioral health care. We note that for most Medicaid beneficiaries, the clinic benefit would generally be one component of a broader Medicaid benefit package. Per section 1902(a)(10)(A) and (C) of the Act and implementing regulations at 42 CFR part 440, most beneficiaries in the categorically and medically needy eligibility groups are eligible for a benefit package consisting of an array of mandatory services that address a broad range of healthcare needs, and States can choose to include optional services, such as the clinic services benefit, in that coverage. Other beneficiaries may have different or more limited coverage under the governing statutes and regulations. Nothing about our proposed exceptions to the four walls requirement under the clinic benefit affects States' obligations and options regarding beneficiaries' benefit packages. We are not including any new requirements in response to these comments.
Comment: Several commenters urged CMS to work closely with States and Tribes in implementing the additional exceptions to the four walls requirement if the rule is finalized. One commenter recommended that CMS issue detailed implementation guidance if the proposed rule is finalized.
Response: We appreciate the commenters' suggestions and we intend to work closely with States and Tribes in implementing these exceptions. We have no current plans to issue detailed implementation guidance, but we might revisit those plans based on States' and Tribes' needs for assistance.
Comment: One commenter urged CMS to ensure that FDA-cleared medical devices that help prevent overdose and significantly reduce opioid withdrawal symptoms be adequately covered and reimbursed when furnished outside of the four walls by IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas if the proposal is finalized.
Response: We appreciate the commenter's recommendation; however, we are not including any new requirements related to this recommendation. We note that nothing about the proposed exceptions to the four walls requirement reduces the scope of items and services that may be paid for under the clinic benefit.
2. Justification and Criteria for Additional Medicaid Clinic Services Four Walls Exceptions
As explained in section XVIII.A of the proposed rule, we previously interpreted section 1905(a)(9) of the Act to limit Medicaid clinic services to services furnished within the four walls of the clinic, except only for services furnished by clinic personnel to individuals who are unhoused. We continue to believe that because Congress added only one specific reference to services furnished outside the clinic to the statute in OBRA `87, it generally ratified our prior interpretation of the four walls requirement. Thus, we continue to believe that the statute authorizes neither broad exceptions to the four walls requirement that have no relationship to the current exception nor a complete elimination of the four walls requirement. However, we now interpret section 1905(a)(9) of the Act as permitting additional exceptions to the four walls requirements for populations served by clinics if those populations have similar health care access issues to individuals who are unhoused. When Congress added the exception to the statute, it introduced the exception with the word “including” (OBRA '87). We interpret the word “including” in the statute as not precluding additional exceptions to the four walls requirement, so long as any additional exception is similar to the exception for individuals who are unhoused. Had Congress wanted to limit the clinic benefit to only services provided within the four walls and services provided outside the four walls to the unhoused, it could have written a narrower exception instead of using “including” as it did when adding the exception to section 1905(a)(9) of the Act. As discussed in the Congressional Record for OBRA '87 in H.R. Rep. 100-391, Congress amended section 1905(a)(9) of the Act to create an exception to the four walls requirement for individuals who are unhoused to address access concerns for a population that has unmet health needs, distrusts mainstream providers, and has difficulty accessing care when providers are unable to meet them where they are located.[526] We believe that adding exceptions to the four walls requirement for populations with similar needs and barriers to access as individuals who are unhoused is consistent with the statutory text and purpose of the initial exception.
In developing the proposed exceptions, we considered the characteristics of the unhoused population that is targeted by the current statutory and regulatory exception. According to data from the Department of Housing and Urban Development (HUD), 21 percent of individuals who are unhoused reported having a serious mental illness while 16 percent reported having a substance use disorder.[527] Individuals who are unhoused often lack transportation to access health care and cite this lack of transportation as a barrier to managing their health.[528 529] In many cases, individuals who are unhoused distrust providers due to perceptions of ( printed page 94446) disrespect and discrimination.[530] Individuals who are unhoused also experience much poorer health outcomes than those who are housed; for example, nearly two thirds of individuals who are unhoused experience clinically significant dental problems and are four times as likely to visit an emergency department.[531 532] A recent study found that when controlling for demographic and geographic differences, an individual who is unhoused is three and one half times more likely to experience early mortality than an individual who is housed.[533] As indicated in the proposed rule, we believe that providing additional exceptions to the clinic services four walls requirement for populations with similar needs and barriers to access as individuals who are unhoused is consistent with the statute.
The exceptions outlined in the proposed rule followed four criteria that mirror the needs and barriers to access experienced by individuals who are unhoused:
- The population experiences high rates of behavioral health diagnoses or difficulty accessing behavioral health services;
- The population experiences issues accessing services due to lack of transportation;
- The population experiences a historical mistrust of the health care system; and
- The population experiences high rates of poor health outcomes and mortality.
By authorizing additional clinic services to be furnished outside of the four walls, the proposed exceptions are expected to improve access to care for the populations targeted by the exceptions. The exceptions would authorize States to pay the facility-based clinic services payment rates (such as the AIR for IHS/Tribal clinics) for the excepted services. Currently, due to the four walls requirement, States can cover and pay for services that are provided by clinic personnel outside the four walls—but that do not fit within the exception at § 440.90(b)—only under Medicaid practitioner services benefits, such as physician services, rehabilitative services, or other licensed practitioner services—not under the clinic services benefit.
It is CMS's understanding that State payment rates for these Medicaid practitioner services benefits are generally lower than the facility-based payment rates that States establish or adopt for Medicaid clinic services (such as the facility-based payment rate under the AIR, in the case of IHS/Tribal clinics), because the facility-based payment rates typically account for more overhead costs. While it is CMS's understanding that States generally pay lower rates for Medicaid practitioner services than they do for Medicaid facility-based services, it should be noted that States generally have the flexibility to increase practitioner services payment rates. States must also comply with section 1902(a)(30)(A) of the Act, which requires States to assure that payments are consistent with efficiency, economy, and quality of care, and are sufficient to enlist enough providers so that care and services are available under the Medicaid State plan at least to the extent that such care and services are available to the general population in the geographic area.
Creating the exceptions could thus result in higher payments to providers for the excepted services. Studies of Medicaid payment rates have found that provider willingness to furnish services may be greater in States that pay providers at higher rates.[534 535] Further, practitioners may be reluctant to provide home-based care when paid under a professional fee schedule rate, since travel expenses and time are often not factored into the payment rate.[536] As this evidence suggests, higher payment rates for services are more likely to incentivize providers to furnish those services. The proposal would authorize payment at the generally higher facility-based clinic services payment rates for the excepted services, therefore we believe that it would incentivize providers to provide these services, and thereby meet these beneficiaries where they are located, which for reasons further discussed later in this section of this preamble, will help to ensure access to necessary care.
We considered whether this change in interpretation could burden States, beneficiaries, providers, or others who have relied on our current interpretation. Based on our current awareness of how States implement the Medicaid clinic services benefit, we do not anticipate that our proposal would create burdens for Medicaid clinic services providers or Medicaid beneficiaries, and we have considered the possible burden for State Medicaid programs in developing the proposal. We invited comments on whether our proposal might create any burdens for States, beneficiaries, providers, or other interested parties.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Many commenters agreed with CMS that authorizing payment at the generally higher facility-based clinic services payment rates for services furnished outside of the four walls would help incentivize providers to furnish these services, and thereby meet beneficiaries where they are located and help ensure access to care.
Response: We appreciate the comments that agreed that that authorizing payment at the generally higher facility-based clinic services payment rates for the expected services would help incentivize providers to furnish these services, and thereby meet beneficiaries where they are located and help ensure access to care.
Comment: Many commenters agreed with CMS's interpretation that it is within CMS's authority under section 1905(a)(9) of the Act to include additional exceptions to the four walls requirement for populations with similar needs and barriers to access as individuals who are unhoused.
Response: We appreciate the comments in support of the explanation in the proposed rule for why it is within our authority under section 1905(a)(9) of the Act to include additional exceptions ( printed page 94447) to the four walls requirement for populations that meet more of the four criteria we described in the proposed rule.
Comment: Three commenters recommended CMS consider eliminating the four walls requirement completely as they believed the four walls requirement creates barriers to the delivery of Medicaid clinic services via telehealth. One commenter urged CMS to work with Congress to eliminate the four walls requirement through legislation; while another commenter indicated that CMS is misinterpreting section 1905(a)(9) of the Act and that the statute does not require clinic services to be furnished within the four walls of a clinic.
Response: We thank the commenters for their recommendations, but we do not interpret the statute as authorizing us to completely eliminate the four walls requirement in § 440.90 through rulemaking. As we discussed in section XVIII.B of the proposed rule (89 FR 59186), we continue to believe that the statute authorizes neither broad exceptions to the four walls requirement that have no relationship to the current exception nor a complete elimination of the four walls requirement. However, in this rule, we now interpret section 1905(a)(9) of the Act as permitting additional exceptions to the four walls requirements for populations served by clinics if those populations have similar health care access issues to individuals who are unhoused. We will take the commenters' concerns about telehealth into consideration as we contemplate issuing sub-regulatory guidance regarding our interpretation of how the four walls requirement applies when Medicaid clinic services are delivered via telehealth.
3. IHS/Tribal Clinics
In response to advice and input received through Tribal consultation, we proposed to add a new paragraph (c) to § 440.90 to add an exception to the four walls requirement for IHS/Tribal clinics, to authorize payment for clinic services provided outside the four walls by IHS/Tribal clinic personnel. This exception would be mandatory for all States that opt to cover the Medicaid clinic services benefit. We referred in the proposed regulation text to clinics that are facilities of the IHS, whether operated by IHS or by a Tribe or Tribal organization as authorized by the ISDEAA, to make clear that this exception applies only to IHS/Tribal clinics. The proposed regulatory language identifying the facilities that would be subject to the exception is consistent with our longstanding interpretation of the language used in sections 1905(b) and 1911 of the Act, and would mean clinics that are owned and operated by IHS, clinics that are owned by IHS and tribally-operated as authorized by the ISDEAA, or clinics that are owned and operated by Tribes and Tribal organizations as authorized by the ISDEAA.[537]
Under section 1903(a)(1) of the Act, as discussed earlier, the Federal Government is required to match State expenditures for medical assistance at the FMAP, which is defined at section 1905(b) of the Act to be 100 percent for State expenditures for Medicaid-covered services received through an IHS facility whether operated by IHS or by a Tribe or Tribal organization (which, again, CMS has interpreted to refer to all three kinds of IHS/Tribal facilities described previously). Under CMS's longstanding interpretation of section 1905(b) of the Act, this 100 percent FMAP is available only for State expenditures on services received through an IHS/Tribal facility (such as a clinic) by an AI/AN Medicaid beneficiary. State expenditures on services furnished by an IHS/Tribal facility to other Medicaid beneficiaries are not matched by the Federal Government at 100 percent, but rather at the otherwise applicable FMAP, and this would continue to apply for services provided outside the four walls of a clinic.
We did not propose to include facilities operated by UIOs in the proposed exception, because it is our understanding that many of those facilities currently participate in Medicaid as providers of the Medicaid FQHC services benefit, not as providers of the clinic services benefit. Because Medicaid FQHC services are not subject to a four walls requirement under Federal Medicaid law, we believe that UIOs are unlikely to need the proposed exception. UIO facilities that provide Medicaid clinic services might qualify as behavioral health clinics or clinics in rural areas and be exempt from the four walls requirement under one of the two optional exceptions discussed later in this section of the preamble.
This exception would apply to any Medicaid beneficiary who receives services from the IHS/Tribal clinic. Under IHS authorities, these clinics serve Medicaid beneficiaries who are eligible to receive services from the IHS/Tribal clinic under IHS regulations at 42 CFR part 136, and also may serve other Medicaid beneficiaries under 25 U.S.C. 1680c. As mentioned in section XVIII.A of the proposed rule, all services covered under the clinic services benefit must be furnished by or under the direction of a physician, so we proposed to include language in this exception specifying that services subject to the exception would have to be furnished under the direction of a physician to make that requirement clear.
We proposed this exception based on advice and input received through Tribal consultation and because the population served by IHS/Tribal clinics, which is predominately AI/AN, tends to meet the criteria CMS has identified that warrant an exception from the four walls requirement (for example, high rates of behavioral health needs, lack of accessible transportation, mistrust of the health care system, and high rates of morbidity and poor health outcomes).
AI/ANs experience high rates of behavioral health diagnoses. In particular, the opioid crisis plaguing many communities is especially acute in Tribal communities.[538] As reported by the Centers for Disease Control and Prevention (CDC), AI/ANs have the highest rate of drug overdose compared to other U.S. populations, and they experienced a 39 percent increase in overdoses between 2019 and 2020.[539]
Many AI/ANs also experience difficulties accessing services due to lack of transportation. Tribal lands encompass about 56 million acres nationwide, including 145,000 miles of roads.[540] Roads in Tribal communities are typically rudimentary and in poor condition. For example, about 70 percent of Tribal roads across the country are unpaved compared to 45 percent of all rural roads.[541 542] Because Tribal communities are often located in rural or remote areas covering vast distances, providers can be extremely far away from their patients. For example, it is common for AI/ANs to have to travel between 60 and 90 miles one-way for health care ( printed page 94448) appointments.[543] Many AI/ANs also do not have reliable personal transportation. The rate of AI/ANs without a personal vehicle is more than double that of individuals in other rural areas.[544] Per a recent CDC report, approximately 17.1 percent of AI/ANs lack reliable transportation, the highest rate compared to other U.S. populations, and this is a barrier to accessing health care.[545] Many AI/ANs have a profound mistrust of the Federal Government and mainstream providers based on trauma from a long history of harmful U.S. Tribal policies, such as removal of AI/ANs from homelands and Tribal community structures, bans on cultural practices and language, forced relocation to reservations, abusive boarding school practices, and other destructive policies. AI/AN health disparities are the visible, lingering result of these harmful policies.[546]
Furthermore, AI/ANs face poorer health outcomes than all other adults on average and have the lowest life expectancy compared to other U.S. populations. For example, AI/ANs have higher rates of obesity, heart disease, and diabetes than other adults in the U.S. population on average.[547] The CDC's Provisional Life Expectancy Estimates for 2021 found a severe drop in life expectancy for AI/ANs—decreasing by 6.6 years from 2019 to 2021.[548] Not only do AI/ANs, on average, die younger than all other Americans, but this disparity is worsening at an alarming rate. AI/AN life expectancy today is the same as it was for the average American in 1944.[549]
This evidence indicates that an exception to the four walls requirement is warranted for IHS/Tribal clinics because the individuals served by these clinics are more likely than those in other groups to meet a higher number of the four criteria we described in the proposed rule. Through Tribal Consultation, Tribal leaders indicated that IHS/Tribal clinics need the flexibility to provide services to AI/ANs where they are located due to their high levels of behavioral health diagnoses, challenges accessing services due to lack of transportation and appropriate infrastructure, historic mistrust of the Federal Government and the health care system, and poor health outcomes.
As explained later in this section of the preamble, we proposed that behavioral health clinics and clinics in rural areas would serve as a proxy for their patient populations, instead of limiting the exception for behavioral health clinics to patients with behavioral health disorders or limiting the exception for clinics in rural areas to patients residing in rural areas. We proposed this approach because we believe that these clinics serve predominantly patients with behavioral health disorders or who live in rural areas (as applicable), and to reduce the operational burden of implementing these exceptions. Similar to the proposed exceptions for behavioral health clinics and clinics in rural areas, we also proposed that the IHS/Tribal clinics would be a proxy for their patient population, but for somewhat different reasons. The operational burden that the proposed proxy approach would address for behavioral health clinics and clinics in rural areas would not be as much of an issue for IHS/Tribal clinics, because the entire patient population of an IHS/Tribal clinic is likely to meet some or all of the four criteria described in the proposed rule. For that same reason, a proxy approach would be appropriate for these clinics. These clinics serve a clearly identifiable group of Medicaid beneficiaries under IHS statutes and regulations: Medicaid beneficiaries whom IHS/Tribal clinics serve under 42 CFR part 136 or other Medicaid beneficiaries whom these clinics may serve under 25 U.S.C. 1680c. As discussed previously, the population served by IHS/Tribal clinics, which is predominately AI/AN, is more likely than other groups to meet a higher number of the criteria identified in the proposed rule as warranting an exception.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Most commenters expressed support for our proposed exception to the Medicaid clinic services benefit four walls requirement for IHS/Tribal clinics at § 440.90(c). Many commenters indicated that Tribal communities face significant barriers in accessing healthcare facilities and could benefit from an exception to the four walls requirement. Several commenters noted that IHS/Tribal clinics are experiencing substantial practitioner shortages that create long wait times for in-person services, and that without a permanent exception to the Medicaid four walls requirement the wait times for in-person services will worsen. One commenter explained that continued access to Medicaid clinic services outside the four walls of IHS/Tribal clinics is vital for isolated Tribal communities that rely upon rely upon traveling practitioners from these clinics, as there are great distances between the communities and the closest IHS/Tribal clinics. Finally, commenters also noted that allowing an exception to the four walls requirement for IHS/Tribal clinics will ensure that services continue to be provided in the location that is best for the Tribal member after the end of the grace period.
Response: We appreciate the comments in support of our proposed exception to the Medicaid clinic services benefit four walls requirement for IHS/Tribal clinics. We agree that the population served by IHS/Tribal clinics faces significant barriers in accessing services and could benefit from an exception to the four walls requirement. We also agree that this exception will allow this population to continue to receive services in the location that best meets the individual's needs after the grace period.
Comment: Many commenters agreed with CMS that the population that receives services from IHS/Tribal clinics meets more of the four criteria we described in the proposed rule than other groups and that an exception to the four walls requirement is justified for IHS/Tribal clinics. Several commenters noted that AI/ANs experience some of the highest rates of chronic illness of any population in the U.S., and a few commenters noted that AI/ANs are more than twice as likely to miss appointment times than other U.S. populations. ( printed page 94449)
Response: We appreciate the comments in support of the evidence we presented that the population that receives services from IHS/Tribal clinics meets more of the four criteria we described in the proposed rule than other groups and that an exception to the four walls requirement is justified for IHS/Tribal clinics.
Comment: A few commenters inquired whether the proposed IHS/Tribal clinic exception at § 440.90(c) would apply to services furnished to individuals who are not AI/AN.
Response: As we indicated in section XVIII.B of the proposed rule (89 FR 59186), the exception would apply to any Medicaid beneficiary who receives services from the IHS/Tribal clinic, not just individuals who are AI/AN.
Comment: One commenter inquired whether the proposed IHS/Tribal clinic exception at § 440.90(c) would apply to Tribal facilities that participate in Medicaid as FQHCs.
Response: We noted in section XVIII.B of the proposed rule (89 FR 59186) that the Medicaid clinic services benefit is distinct from the Medicaid FQHC services benefit and there is no Federal four walls requirement under the Medicaid FQHC benefit, unlike the Medicaid clinic services benefit.
Comment: One commenter inquired whether the proposed IHS/Tribal clinic exception at § 440.90(c) would apply to traditional healing services.
Response: CMS recently approved several States' section 1115 demonstration proposals to cover traditional health care practices, which cannot currently be covered under Medicaid State plan benefits, such as the clinic services benefit.[550] The traditional health care practices that can be covered under these demonstrations are not clinic services within the meaning of section 1905(a)(9) of the Act, 42 CFR 440.90, or this rulemaking. As such, the clinic services benefit four walls requirement does not apply to that coverage.
Comment: One commenter recommended that CMS expand the IHS/Tribal clinic exception at § 440.90(c) in the final rule to include non-Tribal clinics that operate on Tribal lands.
Response: We appreciate the commenter's recommendation and will take it into consideration for future rulemaking; however, we decline to expand the IHS/Tribal clinic exception at § 440.90(c) in this final rule to include non-Tribal clinics that operate on Tribal lands. We did not propose including non-Tribal clinics that operate on Tribal lands within the exception for IHS/Tribal clinics and believe that such a change would require additional consideration and discussion with the public beyond what could be finalized in this current rule. However, we would also note that we included an exception at § 440.90(e) for clinics located in rural areas and non-Tribal clinics that operate on Tribal lands might fall within that exception in States that choose to adopt such exception.
After consideration of the public comments we received, we are finalizing, without modification, an exception to the Medicaid clinic services four walls requirement at § 440.90(c) for IHS/Tribal clinics.
4. Behavioral Health Clinics
We proposed to add a new paragraph (d) to § 440.90 to authorize an exception to the four walls requirement for clinic services provided outside the four walls by personnel of behavioral health clinics. This exception would not be mandatory in States that opt to cover the clinic services benefit but could be implemented as a State option. Specifically, we proposed an exception for clinics that are primarily organized for the care and treatment of outpatients with behavioral health disorders, including mental health disorders and substance use disorders. We noted that the proposed exception would include any clinic services furnished outside of the four walls by a behavioral health clinic, including non-behavioral clinic services such as physical health services.
The proposed exception would include behavioral clinic types that are recognized nationally, such as Community Mental Health Centers, and other behavioral health clinics organized in a State. We recognize that the types of behavioral health clinics within a State may vary, so we did not propose to limit this exception to specific types of behavioral health clinics. However, to be considered a behavioral health clinic under the proposed exception, the clinic would have to be primarily organized to treat outpatients with behavioral health disorders regardless of the patient mix of the clinic. For example, if a State has established separate licensure or certification requirements for mental health clinics and primary care clinics, under which primary care clinics are licensed to treat outpatients for a range of services beyond the treatment of behavioral health disorders, then we would consider a mental health clinic in that State to be primarily organized to treat outpatients with behavioral health disorders but would not consider a primary care clinic in that State to be primarily organized to treat such outpatients. We recognize that there may be other means by which a State determines that a clinic is primarily organized to treat outpatients with behavioral health disorders (that is, other than through licensure or certification), including behavioral health accreditation by accrediting organizations, such as the Joint Commission, or based on the organizing documents of the clinic, such as a business charter. If the proposal were finalized as described, States that choose to adopt this exception would describe the types of behavioral health clinics such exception applies to in their Medicaid State plan. Just like our proposed exception for IHS/Tribal clinics, we proposed to include language in this exception specifying that services subject to the exception would have to be furnished under the direction of a physician.
Per 2022 data from the Substance Abuse and Mental Health Services Administration (SAMHSA), approximately 94.7 percent of adults nationwide with a substance use disorder did not seek substance use treatment and nearly half of adults nationwide with a mental health disorder did not receive mental health treatment, which suggests that this population may have difficulty accessing behavioral health services.[551] Lack of transportation and geographic distance from behavioral health services are often cited in research as barriers to behavioral health treatment.[552] One study of transportation-disadvantaged adults found that nearly half of adults nationwide who lacked medical transportation were diagnosed with depression or another mental health disorder.[553] Studies have found that ( printed page 94450) individuals with behavioral health disorders often report negative experiences with providers and stigmatizing attitudes from providers are common, which can lead to a mistrust of the health care system and forgone care.[554] Finally, research has found that individuals with a severe mental illness or substance use disorder experience worse health outcomes and increased risk of premature mortality, with one recent study finding individuals with a severe mental illness or substance use disorder experiencing a shorter life span than comparable individuals by an average of 6 years.[555]
State-specific circumstances may affect the degree to which a State's population of individuals with behavioral health disorders meets the four criteria described in the proposed rule. The Health Resources and Services Administration (HRSA), in coordination with State primary care offices, designates as Health Professional Shortage Areas (HPSAs) [556] areas experiencing a shortage in primary care, dental care, or mental health care providers for a whole geographic area, a specific population within a geographic area, and facilities that serve these areas. HRSA publishes data for each State on the percent of need met for primary care, dental care, and mental health providers, with a lower percentage indicating a lower availability of providers. It should be noted that the types of mental health providers counted in HPSAs are set in regulation, and based on the regulations, HRSA allows State Primary Care Offices to choose whether to count: psychiatrists only, core mental health professionals (psychiatrists, clinical psychologists, clinical social workers, psychiatric nurse specialists, and marriage and family therapists), or a combination of all types. As of December 31, 2023, there is significant variation among States in the percent of need met for mental health care, with a low of 9 percent and a high of 63 percent.[557] This variation in availability of mental health care providers may suggest that populations of individuals with behavioral health disorders in some States may have greater difficulty accessing behavioral health services or accessing transportation to a behavioral health provider than those populations in other States. There may also be significant variability between States with regard to behavioral health outcomes and mortality. For example, in 2021 the age-adjusted drug overdose mortality rate by State had significant variation, from a low of 11 per 100,000 to a high of 90 per 100,000.[558] These differences between populations of individuals with behavioral health disorders in different States may suggest that the degree to which a State's population of individuals with behavioral health disorders meets the four criteria may be variable.
This evidence indicates that an exception to the clinic services four walls requirement could be warranted, based on State-specific circumstances, for clinics that are primarily organized for the care and treatment of outpatients with a behavioral health disorder, as these clinics might primarily serve a patient population that may be more likely than other groups to meet more of the four criteria we described in the proposed rule. The evidence also suggests that this patient population is less likely to meet as many of the criteria as consistently nationwide as patients served by IHS/Tribal clinics. Under the proposal, a State could determine that individuals with a behavioral health disorder in that State should be engaged by behavioral health clinic personnel where they are located due to their challenges accessing services, including lack of transportation and geographic distance from services, historic mistrust and stigmatization in the health care system, and poor health outcomes.
We considered proposing that, to qualify for the proposed exception, clinic services would have to be provided specifically to individuals with a behavioral health disorder, in addition to being provided by personnel of a behavioral health clinic. However, we believe that such a requirement would be too operationally burdensome and that instead behavioral health clinics can serve as a proxy for a population that generally consists of individuals with a behavioral health disorder. We recognize there may be circumstances in which a behavioral health clinic furnishes services to an individual who does not have a behavioral health disorder, but it is our understanding that behavioral health clinics generally serve a patient population that consists primarily of individuals with behavioral health disorders (including individuals with a formal behavioral health disorder diagnosis and those with an undiagnosed behavioral health disorder). Thus, these clinics can serve as a proxy for a patient population that is more likely to have such a disorder—and thus, that includes people who are more likely to meet more of the four criteria. In addition, we believe that requiring clinics or States to verify that a clinic patient has a behavioral health disorder and to deny coverage of Medicaid clinic services provided outside the four walls if the patient does not, would be too operationally burdensome. For example, an individual might experience or present their behavioral health symptoms in an uncommon way, an individual might be misdiagnosed, or an individual might be experiencing a crisis where services are needed urgently and verifying that they have a behavioral health disorder might delay needed care. Because we believe that behavioral health clinics can serve as a proxy for individuals with behavioral health disorders, and because we do not want to make this exception too operationally burdensome, we did not propose that to qualify for the proposed exception, clinic services must be provided by a behavioral health clinic specifically to an individual with a behavioral health disorder.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Most commenters expressed support for our proposed exception to the Medicaid clinic services benefit four walls requirement for behavioral health clinics at § 440.90(d). Commenters indicated that individuals with behavioral health needs would benefit from receiving care in environments where they feel most comfortable rather than solely within the four walls of a clinic. One commenter noted that an exception to the four walls requirement for behavioral health clinics might lead to earlier diagnosis, better adherence to ( printed page 94451) treatment plans, and improved outcomes for beneficiaries with behavioral health needs. Many commenters explained that beneficiaries that receive services from behavioral health clinics often experience transportation, financial, safety, behavioral health, or physical health challenges that impede their ability to travel to behavioral health clinics.
Several commenters expressed that telehealth flexibilities adopted by States under section 1135 waivers during the COVID-19 public health emergency (PHE) to allow coverage of Medicaid clinic services outside of the four walls (when neither the clinic practitioner nor beneficiary was present at the clinic) increased access to services furnished by behavioral health clinics, and that a permanent exception to the four walls requirement for behavioral health clinics would increase access to care. Many commenters indicated that CMS's current interpretation of how the four walls requirement applies when Medicaid clinic services are furnished via telehealth, under which clinic services furnished via telehealth are covered only when either the clinic practitioner or the beneficiary is physically onsite at the clinic facility, is burdensome for behavioral health clinics.
In addition, several commenters noted that there are national behavioral health workforce shortages and allowing an exception to the four walls requirement for behavioral health clinics would increase clinic practitioner recruitment and retention rates.
Response: We appreciate the comments in support of our proposed exception to the Medicaid clinic services benefit four walls requirement for behavioral health clinics. We thank the commenters for sharing additional information on how the population served by behavioral health clinics may benefit from an exception to the four walls requirement and how an exception could lead to better workforce recruitment and retention rates. We also agree that an exception to the four walls requirement for behavioral health clinics could increase access to care. Finally, we agree with the commenters that noted that an exception to the four walls requirement for behavioral health clinics might increase clinic practitioner recruitment and retention rates.
We acknowledge the commenters' concerns regarding our current interpretation of how the four walls requirement applies when Medicaid clinic services are furnished via telehealth. We note that upon the effective date of this rule, if a State adopts exceptions to the four walls requirement for behavioral health clinics and/or clinics located in rural areas, then neither the beneficiary nor the clinic practitioner would need to be present in such a clinic for services to be delivered via telehealth. In addition, IHS/Tribal clinic services can be delivered via telehealth without the beneficiary or clinic practitioner present in the clinic upon the effective date of this final rule with comment period. Finally, we will take these comments into consideration as we contemplate issuing sub-regulatory guidance regarding our interpretation of how the four walls requirement applies when Medicaid clinic services are delivered via telehealth.
Comment: One commenter inquired whether psychiatric outpatient clinics are considered behavioral health clinics, for purposes of the proposed exception for behavioral health clinics at § 440.90(d).
Response: The proposed exception for behavioral health clinics applies to services furnished outside of a clinic that is primarily organized for the care and treatment of outpatients with behavioral health disorders, including mental health and substance use disorders, by clinic personnel under the direction of a physician. As we discussed in the proposed rule preamble, the types of behavioral health clinics within a State may vary, so we are not proposing to limit this exception to specific types of behavioral health clinics, and States that choose to adopt this exception would describe the types of behavioral health clinics to which the exception applies in their Medicaid State plans. All such clinics would have to be primarily organized to treat outpatients with behavioral health disorders regardless of the patient mix of the clinic. Additionally, per § 440.90, Medicaid clinic services must be furnished by a facility that is not part of a hospital but that is organized and operated to provide medical care to outpatients.
Comment: Many commenters expressed support for including behavioral health clinic types that are recognized nationally, such as Community Mental Health Centers (CMHCs), and other behavioral health clinics organized in a State as behavioral health clinics at § 440.90(d).
Response: We remind the commenters that the States implementing this exception will determine which clinics are subject to it. As we discussed in the proposed rule preamble, States that choose to adopt this exception would describe the types of behavioral health clinics to which the exception applies in their Medicaid State plans, provided that such clinics are primarily organized to treat outpatients with behavioral health disorders. However, as we indicated in the proposed rule preamble, we expect all States that opt to implement this exception to include behavioral health clinic types that are recognized nationally.
Comment: Many commenters expressed support for CMS's proposal to allow the behavioral health clinic exception at § 440.90(d) to apply to any services furnished by behavioral health clinics and not just behavioral health services.
Response: We appreciate the comments in support of our proposal to allow the exception to the four walls for behavioral health clinics to apply to any services furnished by behavioral health clinics.
Comment: Many commenters agreed with CMS that the behavioral health population meets more of the four criteria we described in the proposed rule than other groups that are not the focus of an exception in this rulemaking and that an exception to the four walls requirement is justified for behavioral health clinics. One commenter noted that there is evidence that individuals with behavioral health conditions have increased difficulties establishing trust relationships with practitioners, which leads to a mistrust of the health care system. In addition, the commenter explained that behavioral health conditions can make navigating public transit more difficult, which creates transportation barriers for individuals with behavioral health conditions.
Response: We appreciate the comments in support of the evidence we presented that the behavioral health population meets more of the four criteria we described in the proposed rule than other groups that are not the focus of an exception in this rulemaking and that an exception to the four walls requirement is justified for behavioral health clinics.
Comment: Two commenters recommended that CMS not limit the exception for behavioral health clinics at § 440.90(d) to only include clinics that are primarily organized for the treatment of outpatients with behavioral health disorders. One commenter recommended that CMS consider an application or appeals process to allow clinics which serve a behavioral health population that meets the four criteria we described in the proposed rule (but do not fall under the proposed exception) to also furnish services outside of the four walls.
Another commenter expressed that in rural and frontier areas, individuals with behavioral health disorders may ( printed page 94452) have access to behavioral health services only through primary care clinics or other clinics that are not primarily organized for the care and treatment of outpatients with behavioral health disorders. The commenter recommended that the proposed exception at § 440.90(d) include any clinic that furnishes behavioral health services.
Response: We decline to expand the exception to include non-behavioral health clinics that furnish behavioral health services. As we noted in section XVIII.B of the proposed rule (89 FR 59186), behavioral health clinics can serve as a proxy for a patient population that is more likely to have a behavioral health disorder—and thus, that includes people who are more likely to meet more of the four criteria we described in the proposed rule. It is our understanding that non-behavioral health clinics do not generally serve a patient population that consists primarily of individuals with behavioral health disorders. We do not believe that non-behavioral health clinics that furnish behavioral health services can serve as a proxy for such a population. We would also note that we included an exception at § 440.90(e) for clinics located in rural areas and are not limiting the types of services that those clinics may furnish outside of the four walls under that exception.
Comment: One commenter encouraged CMS to require States to establish a robust referral system, including ongoing transportation support, to ensure that individuals who receive services from behavioral health clinics can get connected to and receive care for any additional health needs outside of the clinic.
Response: We decline to amend the regulation to require States to establish a referral system. Federal Medicaid regulations governing transportation and case management services are beyond the scope of this rulemaking. However, we understand the critical role that transportation to services can play in improving the health outcomes of Medicaid beneficiaries, and note that per section 1902(a)(4)(A) of the Act and 42 CFR 431.53, State Medicaid plans must specify that the Medicaid agency will ensure necessary transportation for beneficiaries to and from providers. CMS issued guidance on the Medicaid transportation assurance in the form of a State Medicaid Director letter (SMD) 23-006 that introduces the Medicaid Transportation Coverage Guide. Case management services are optional in the Medicaid program under sections 1905(a)(19) and 1915(g) of the Act and 42 CFR 440.169, and States may choose to implement case management services under a variety of authorities. We acknowledge the role that case management can play in linking individuals to needed medical, behavioral health, and other services.
Comment: Two commenters recommended that CMS implement the proposed exception at § 440.90(d) at the beneficiary level (in addition to the clinic level) to ensure that the entire behavioral population that meets the four criteria we described in the proposed rule is able to access clinic services outside the four walls.
Response: We understand the commenters to be recommending application of this exception to any Medicaid beneficiary with a behavioral health condition, regardless of the type of clinic. We thank the commenters for their recommendation that we implement the proposed exception at § 440.90(d) at the beneficiary level (in addition to the clinic level) to ensure that the entire behavioral health population that meets the four criteria we described in the proposed rule is able to access clinic services outside the four walls; however, we will not be making changes in the final rule to allow States to apply the exception at § 440.90(d) at the beneficiary level. As we noted in section XVIII.B of the proposed rule (89 FR 59186), behavioral health clinics can serve as a proxy for a patient population that is more likely to have a behavioral health disorder—and thus, that includes people who are more likely to meet more of the four criteria we described in the proposed rule. In addition, we believe that requiring clinics or States to verify that a clinic patient has a behavioral health disorder and to deny coverage of Medicaid clinic services provided outside the four walls if the patient does not, would be too operationally burdensome.
After consideration of the public comments we received, we are finalizing, without modification, an exception to the Medicaid clinic services four walls requirement at § 440.90(d) for clinics that are primarily organized for the care and treatment of outpatients with behavioral health disorders, including mental health and substance use disorders.
5. Clinics Located in Rural Areas
We proposed to add a new paragraph (e) to § 440.90 to authorize an exception to the four walls requirement for clinic services provided outside the four walls by personnel of clinics located in rural areas, but that are not RHCs as referenced in section 1905(a)(2)(B) of the Act and § 440.20(b). This exception would not be mandatory in States that opt to cover the clinic services benefit, but could be implemented at State option. Just like our proposed exception for IHS/Tribal clinics and behavioral health clinics, we proposed to include language in this exception specifying that services subject to the exception would have to be under the direction of a physician.
Per SAMHSA data, rates of mental illness and substance use disorders are similar in rural and urban areas.[559] However, individuals in rural areas with a mental illness or substance use disorder are less likely to receive treatment than individuals in urban areas due to more limited access to providers, as rural areas are more likely to lack trained and specialized behavioral health providers.[560] For example, in 2021 the number of psychologists per 100,000 people in rural counties was less than half of the number in urban counties.[561] A recent study found that rural individuals on average are 22 percent less likely than urban individuals to utilize primary and specialty clinic services.[562] Studies have found that lack of transportation and distance to providers is a common barrier to rural individuals accessing health care services.[563] Furthermore, a recent Government Accountability Office (GAO) report found that rural individuals need to travel 40 miles on average to access specialty services.[564] With regards to mistrust of the health care system, research has found that rural individuals have historically mistrusted the health care at higher ( printed page 94453) rates, and that some of this mistrust comes from a perception that the health care system prioritizes urban communities over rural communities.[565] Per the CDC, rural individuals are at greater risk of poor health outcomes as they tend to be older and sicker than urban individuals.[566] Finally, according to a CDC National Center for Health Statistics (NCHS) study, age-adjusted mortality rates are higher for rural individuals, with the mortality gap increasing since 1999 between rural and urban individuals.[567]
State-specific circumstances may affect the degree to which a State's population of individuals in rural areas meets the four criteria described in the proposed rule. A study found that 21 percent of adults without access to a vehicle or public transit reported skipping needed medical care compared to only 9 percent who did not own a vehicle but had access to public transit.[568] According to a Federal Highway Administration publication, just under 90 percent of passenger trips in rural areas occur in personal vehicles.[569] For the rural individuals who lack access to a personal vehicle, public transit is generally less available, with an approximate 40 percent of rural individuals living in an area without public transit.[570] However, some States establish rural public transit systems that guarantee service coverage to all residents.[571] This variation between States in their rural populations' access to public transit may suggest that the degree to which a State's rural population is able to access transportation to medical services may differ from State to State. There may also be significant variability between States with regards to health outcomes and mortality. For example, a CDC report found that the percentage of excess mortality from heart disease in rural counties varied significantly between States in the northeast and the south with a 13 percent excess rate for the northeast States and 56 percent for the southern States.[572] These differences between State populations of individuals in rural areas may suggest that the degree to which a State's rural population meets the four criteria may be variable.
This evidence indicates that an exception to the clinic services four walls requirement could be warranted, based on State-specific circumstances, for services furnished by clinics located in rural areas that are not RHCs, as these clinics might primarily serve a patient population that may be more likely than other groups to meet more of the four criteria we identified in the proposed rule. The evidence also suggests that this patient population is less likely to meet as many of the criteria as consistently nationwide as patients served by IHS/Tribal clinics. Under the proposal, a State could determine that individuals who reside in rural areas in that State should be engaged where they are located by personnel of a clinic located in a rural area, due to their challenges accessing behavioral health services, overall health care access challenges stemming from lack of transportation and distance from providers, historic mistrust of the health care system, and poor health outcomes. We note that clinics located in rural areas providing optional services as authorized under sections 1902(a)(10) and 1905(a)(9) of the Act and 42 CFR 440.90 are distinct from RHCs providing mandatory services as authorized under sections 1902(a)(10) and 1905(a)(2)(B) of the Act and 42 CFR 440.20(b). RHC services are a separate Medicaid benefit provided by a type of facility that is referenced in section 1905(a)(2)(B) of the Act and 42 CFR 440.20(b), and a four walls requirement does not apply to that benefit under Federal Medicaid law.
We considered proposing that, to qualify for the proposed exception, clinic services would have to be provided specifically to individuals who reside in rural areas, in addition to being provided by personnel of a clinic located in a rural area. However, we believe clinics located in rural areas can serve as a proxy for a population that generally consists of individuals who reside in rural areas, and that such a requirement would be too operationally burdensome. We recognize there may be circumstances in which a clinic located in a rural area furnishes services to an individual who does not reside in a rural area, but it is our understanding that clinics located in rural areas generally serve a patient population that consists primarily of individuals who reside in rural areas. Thus, these clinics can serve as a proxy for a patient population that is more likely to reside in a rural area—and thus, that includes people who are more likely to meet more of the four criteria. In addition, we believe that requiring clinics or States to verify that a clinic patient lives in a rural area, and to deny coverage of Medicaid clinic services provided outside the four walls if the patient does not, would be too operationally burdensome. For example, an individual's address might change frequently, an individual might refuse to provide their address, or the clinic might be located in a rural area that borders a non-rural area. Because we believe that clinics located in rural areas can serve as a proxy for individuals who reside in rural areas, and because we do not want to make this exception too operationally burdensome, we did not propose that to qualify for the proposed exception, clinic services must be provided by a clinic located in a rural area specifically to an individual who resides in a rural area.
We did not include a definition of “rural” in our proposed rule text, but we considered defining that term in the final rule and we considered various approaches to doing so, on which we sought comment. There are many Federal and State definitions of rural for various programs, and no single definition precisely identifies all rural areas. The Rural Health Information Hub provides a non-official tool that could be used to help identify if a specific location is considered a rural location based on various definitions.[573] Some rural definitions may categorize areas that are generally recognized as suburban as rural, while other ( printed page 94454) definitions may classify sparsely populated remote areas as urban. For example, the population residing in rural areas identified by a more limited rural definition may more closely meet more of the four criteria identified in the proposed rule than the population residing in rural areas identified under a broader definition. Definitions of rural adopted and used by Federal governmental agencies for programmatic purposes include the definition used by the Census Bureau, the definition used by the Office of Management and Budget (OMB), and the definition used by HRSA's Federal Office of Rural Health Policy (FORHP).[574 575] In addition, we believe that State-level variations may also affect whether certain ways of defining rural are appropriate in specific States. States may have their own definitions of rural under State law or regulation for various programmatic purposes, such as definitions adopted by State primary care offices or State Offices of Rural Health.
Under any definition of rural, the specific areas identified as rural may change over time and that would have a direct impact on the scope of clinics eligible for this four walls exception. For example, areas identified as rural under the Census definition may change after the decennial census, which may result in some clinics no longer being located in rural areas under that updated definition.
We considered the following approaches to defining rural: adopting one of the commonly used definitions of rural adopted by the Federal governmental agencies referenced previously, permitting a State to adopt a definition of rural that is adopted and used by a Federal governmental agency for programmatic purposes, permitting a State to adopt a definition of rural that is adopted and used by a State governmental agency with a role in setting State rural health policy, or not adopting any definition of rural.
We note that the research, data, and reports cited earlier in this section do not all use the same definition of rural, and for four of the citations it is unclear what definition of rural was used. The SAMHSA data, the study on primary and specialty care utilization, and NCHS study use the OMB definition while the CDC health outcomes research uses the Census Bureau definition and the GAO report uses the FORHP definition of rural.
We explained that if we were to adopt a Federal definition, we would finalize in rulemaking that for the purposes of this exception rural is defined as the definition of rural adopted or used by the Census Bureau, OMB, or FORHP (we would adopt only one of these definitions). The benefits to adopting a Federal definition include that the definition would be consistent for all States electing to implement the exception and all clinics located in rural areas in such States. However, if we adopted a specific Federal definition of rural then States could not consider the variation in which their rural populations under different rural definitions meet the four criteria we describe in the proposed rule. In addition, CMS does not directly control any of these Federal definitions, so if we adopted a specific Federal definition then future rulemaking might be necessary to align our rule with another Federal agency's changes to that Federal definition.
The Census Bureau does not specifically define rural but considers any area that is not urban as rural. An urban area must meet certain density standards and contain at least 2,000 housing units or at least 5,000 people. There are 2,644 urban areas defined by the Census Bureau following the 2020 Census. Over 80 percent of the Census-defined urban areas (2,134 urban areas) have populations of less than 50,000 people while the remaining 19 percent (510 urban areas) have populations of 50,000 people or more.[576] Following the 2020 Census, the Census Bureau does not sub-categorize urban areas as Urbanized Areas or Urban Clusters.[577] If we adopted the Census Bureau definition, then we would finalize in rulemaking that a rural area is defined as an area identified by the Census Bureau in “Urban Area Criteria for the 2020 Census-Final Criteria,” 87 FR 16706, as not being an urban area. The advantage to the Census Bureau definition of rural is that it is a widely recognized definition, which may make it an easier definition to implement for purposes of an exception to the clinic services four walls requirement. A disadvantage to the Census Bureau definition is that the Census Bureau's urban area boundaries do not follow other administrative units, such as county or municipality borders, and may be complex to operationalize. The Census Bureau provides TIGERweb Decennial online mapping tools for urban area boundaries at https://tigerweb.geo.census.gov/ that may be helpful for interested parties considering what it would have meant for CMS to finalize a rule that defines rural according to the U.S. Census Bureau's definition. The CDC research on health outcomes we cite elsewhere in this section used the Census Bureau definition, which demonstrates that this definition can be linked to the four criteria described in the proposed rule. However, the Census Bureau definition is broad, and some policy experts point out that the definition classifies many suburban areas as rural while also classifying towns and small cities with populations of less than 50,000 people as non-rural.[578]
OMB also does not specifically define rural, but designates areas as metropolitan, micropolitan, or neither (also known as noncore).[579] A metropolitan area consists of an urban core of 50,000 or more individuals, a micropolitan area consists of an urban core of 10,000 to 49,999 individuals, and all other areas are considered neither. Areas that are micropolitan or neither are considered rural while metropolitan areas are considered urban.[580] If we adopted the OMB definition, then we would finalize in regulation text that a rural area is defined as an area not identified as metropolitan by OMB, as described in “2020 Standards for Delineating Core Based Statistical Areas,” 86 FR 37770. Like the Census Bureau definition of rural, the OMB definition is a widely recognized definition that may be an easier definition to implement for purposes of an exception to the clinic services four walls requirement. For example, the study on primary and specialty care utilization and the NCHS study on mortality we cite elsewhere in this section use the OMB definition, which demonstrates that this definition ( printed page 94455) can be linked to the four criteria described in the proposed rule. In addition, the NCHS Urban-Rural Classification Scheme for Counties follows the OMB definition of rural and is widely used in health research.[581] However, the OMB definition is considered by some policy experts to be too narrow as areas OMB defines as metropolitan include areas that are often considered to be rural, like for example the Grand Canyon.[582]
The last Federal definition of rural we considered was the FORHP definition, which consists of all non-metropolitan counties, all metropolitan census tracts with Rural-Urban Commuting Area (RUCA) codes four through ten, large area census tracts of at least 400 square miles in area with population density of 35 or less per square mile with RUCA codes two to three, and all outlying metropolitan counties without an Urbanized Area.[583 584] If we adopted the FORHP definition, then we would finalize in regulation text that a rural area is defined as an area identified as rural by FORHP, as described in “Response to Comments on Revised Geographic Eligibility for Federal Office of Rural Health Policy Grants,” 86 FR 2418. We recognized that the FORHP definition uses terminology that has not yet been updated to align with the latest Census Bureau terminology, that is, FORHP currently refers to urbanized area, but we still considered the FORHP definition, as is, based on its wide use and the benefits described in this paragraph. We noted that FORHP is proposing to update the FORHP definition to incorporate the U.S. Department of Agriculture's Economic Research Service (ERS) Road Ruggedness Scale (RRS) measure of rugged terrain into the existing definition, specifically for census tracts of at least 20 square miles in area in metro counties with RRS 5 and RUCA code 2 or 3. In addition, if finalized, the update would have aligned the FORHP definition's use of Census Bureau terminology with the current Census Bureau definition.[585 586] We explained that if the proposed update to the FORHP definition were finalized, we would then consider the updated FORHP definition for the final rule over the existing FORHP definition. The advantage to the FORHP definition is that it is more precise, as it is narrower than the Census Bureau definition and broader than the OMB definition. In addition, as described elsewhere in this section, the GAO report that identified rural individuals needing to travel 40 miles on average to access specialty care used the FORHP definition in effect at the time of the report, which demonstrates that this definition can be linked to the four criteria we describe in the proposed rule. However, some have criticized the FORHP definition for excluding some areas that used to be considered rural while others consider the definition to be too expansive.[587]
Instead of specifying a uniform definition of rural nationwide for this exception, we also considered allowing States to adopt a definition of rural that has been adopted by a Federal governmental agency. If we permitted States to adopt a definition of rural that is adopted by a Federal governmental agency for programmatic purposes, then we would finalize in regulation text that a rural area is defined by the State based upon a reasonable definition adopted by a Federal governmental agency for programmatic purposes. We would not specifically list out the Federal definitions of rural that we consider reasonable in the regulation text. In addition to the Census, OMB, and FORHP definitions, we would consider rural definitions developed by the U.S. Department of Agriculture's Economic Research Service (ERS) to be reasonable definitions for a State to select if we adopted this option in the final rule (this would include RUCAs, Rural-Urban Continuum Codes, Urban-Influence Codes, and Frontier and Remote Area Codes).[588] We did not consider adopting any of the ERS definitions as one of the Federal definitions we considered (as described previously) because it is our understanding that the ERS definitions are less commonly used on their own (that is, not in conjunction with other Federal definitions) in identifying rural areas in health care. However, the ERS definitions could be used by States if we opted to permit States to identify a Federal definition. While we do not believe that any of the ERS definitions should be adopted as one definition for all States to follow, if we provided States with the flexibility to adopt a Federal definition, then we wanted to ensure that we are not too prescriptive in the definitions they may choose from. It is possible that a State could determine that one of the ERS definitions better captures the population of rural individuals that meets the four criteria described in the proposed rule. Under such an approach, States that elect this exception would identify the specific Federal definition of rural (that is, Census Bureau, OMB, FORHP definition, or one of the ERS definitions) they are adopting in their State plan and attest that the selected definition best captures the population of rural individuals that meets more of the four criteria described in the proposal. The benefits to this approach include that each State can consider which Federal definition of rural best captures the population of rural individuals that meet more of the four criteria described in the proposed rule for that State (and States would attest to this in their State plan), while also being required to adopt a rural definition commonly accepted as a legitimate definition for programmatic purposes at the national level. Requiring the State to attest that the selected Federal definition best captures the population of rural individuals that meets more of the four criteria would help to ensure that there is an explanation for any variations in the definitions selected by different States. However, even if the variations in the definitions chosen by different States can be explained, it might burden or cause confusion for some beneficiaries if the States that elect this exception have different definitions of rural. For example, a beneficiary that moves from a State that has adopted this exception with a broader definition of rural to another State that has adopted the exception but has a narrower definition of rural might lose access to clinic services provided outside of the four walls. In addition, if we finalized the proposal, clinics that operate in different States that have adopted this exception might find it confusing or burdensome to track each such State's definition of rural.
If we permitted States to adopt a definition of rural that is adopted by a State governmental agency with a role ( printed page 94456) in setting State rural health policy, then we would finalize in regulation text that a rural area is defined by the State based upon a rural definition adopted by a State governmental agency with a role in setting State rural health policy. Under such an approach, a State that elects this exception would describe in its State plan the specific definition of rural that it is adopting, attest that this definition has been adopted by a State governmental agency with a role in setting State rural health policy (such as a State primary care office or State Office of Rural Health), and attest that the selected definition best captures the population of rural individuals that meets more of the four criteria described in the proposal. The benefits to this approach include that States may consider a State definition of rural that best identifies the population of rural individuals that meet more of the four criteria described in the proposed rule, and attest to in their State plan that the definition does so. Requiring the State to attest that the selected definition best captures the population of rural individuals that meets more of the four criteria would help to ensure that there is an explanation for any variations in the definitions selected by different States. In addition, under this approach to defining rural, the State would adopt a rural definition commonly accepted and used to manage State programs, which thus may be a more familiar definition to providers and be easier for a State to implement since that definition is also used for other health policy purposes in that State. However, even if the variations in the definitions chosen by different States can be explained, it might burden or cause confusion for some beneficiaries if the States that elect this exception have different definitions of rural. For example, a beneficiary that moves from a State that has adopted this exception with a broader definition of rural to another State that has adopted the exception but has a narrower definition of rural may lose access to clinic services provided outside of the four walls. In addition, if we finalized the proposal, clinics that operate in different States that have adopted this exception might find it confusing or burdensome to track each such State's definition of rural.
Finally, we explained that if we chose not to define rural in the final rule, then we would finalize proposed regulation text with no definition of rural. Under this approach, a State that elects this exception would choose any definition of rural that can be linked to the four criteria we describe in the proposed rule and meets its program needs, but would not identify the definition in the State plan or submit it to CMS for review and approval. We would require and finalize in rule text that the State would publish its rural definition on a website maintained by the State that is accessible to the public. The benefits to not adopting a definition of rural under the final rule would include that States can consider which definition of rural best captures the population of rural individuals that meets more of the four criteria described in the proposed rule. This approach also recognizes that States may have the best information and data to determine the definition of rural that best meets their operational needs. However, under this approach CMS would not be reviewing State definitions of rural, and a State might adopt a definition of rural that could be considered to be overly broad or overly narrow. For example, a State might adopt a definition of rural that encompasses large urban areas, such as a populous city. As we stated in the proposed rule, we are aware that there are many definitions of rural, so the other approaches we considered could potentially leave out reasonable definitions of rural, although we are not currently aware of any such reasonable definitions. In our proposed rule, we invited comment on which approach to defining rural we should adopt if the rule were finalized.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Most commenters expressed support for our proposed exception to the Medicaid clinic services benefit four walls requirement for clinics located in rural areas at § 440.90(e). Many commenters indicated that rural areas are experiencing provider shortages and that an exception to the four walls requirement for clinics located in rural areas would improve access to care for individuals in rural areas. Several commenters noted that in rural areas experiencing provider shortages individuals often have to travel long distances to receive services, and that initiatives aimed at meeting individuals in rural areas where they are located have found success in improving access to care. In addition, a few commenters noted that an exception to the four walls requirement for clinics located in rural areas could alleviate transportation barriers, reduce missed appointments, and lead to more timely care.
Response: We appreciate the comments in support of our proposed exception to the Medicaid clinic services benefit four walls requirement for clinics located in rural areas. We agree that this exception may improve access to care for the population served by clinics located in rural areas.
Comment: Many commenters agreed with CMS that the rural population meets more of the four criteria we described in the proposed rule than other groups that are not the focus of an exception in this rulemaking and that an exception to the four walls requirement is justified for clinics located in rural areas.
Response: We appreciate the comments in support of the evidence we presented that the rural population meets more of the four criteria we described in the proposed rule than other groups that are not the focus of an exception in this rulemaking and that an exception to the four walls requirement is justified for clinics located in rural areas.
Comment: Two commenters recommended that CMS implement the proposed exception at § 440.90(e) at the beneficiary level (in addition to the clinic level) to ensure that the entire rural population that meets the four criteria we described in the proposed rule is able to access clinic services outside the four walls.
Response: We understand the commenters to be recommending application of this exception to any Medicaid beneficiary who resides in a rural area, regardless of the type of clinic. We thank the commenters for their recommendation that we implement the proposed exception at § 440.90(e) at the beneficiary level (in addition to the clinic level) to ensure that the entire rural population that meets the four criteria we described in the proposed rule is able to access clinic services outside the four walls; however, we will not be making changes in the final rule to allow States to apply the exception at § 440.90(e) at the beneficiary level. As we noted in section XVIII.B of the proposed rule (89 FR 59186), clinics located in rural areas can serve as a proxy for a patient population that is more likely to live in a rural area—and thus, that includes people who are more likely to meet more of the four criteria we described in the proposed rule. In addition, we believe that requiring clinics or States to verify that a clinic patient lives in a rural area and to deny coverage of Medicaid clinic services provided outside the four walls if the patient does not, would be too operationally burdensome.
Comment: Many commenters recommended that CMS not adopt a definition of rural at § 440.90(e) in the final rule in response to CMS's ( printed page 94457) solicitation of comments on the best approach to defining rural in the final rule. However, many other commenters recommended that CMS allow States to choose any State or Federal definition of rural. These commenters believed States were best equipped to determine the extent to which a specific definition best captures the rural population that meet the four criteria we described in the proposed rule and that this would recognize each States' unique geographic needs. Several of these commenters expressed concerns that if CMS were to adopt a Federal definition of rural, that this would create a one-size-fits-all approach that would create barriers for beneficiaries and providers. A few of the commenters indicated that in other instances where a Federal program adopted a single definition of rural that it created barriers and hardships for them by not recognizing their States' unique geographic differences. Finally, one commenter explained that under some of the Federal definitions of rural a small city with no access to roads and that is surrounded by rural areas was classified as urban.
Three commenters recommended that CMS define rural with a definition that is adopted and used by a Federal governmental agency for programmatic purposes. One of the two commenters recommended CMS adopt the Census definition of rural in the final rule, as they believed the Census definition is the broadest definition, and that CMS should adopt a definition of rural that is applied consistently. Another commenter recommended that CMS adopt the FORHP definition of rural, as they believed this definition best captures the rural populations that meet the four criteria we described in the proposed rule. Finally, the third commenter recommended that CMS adopt a Federal definition of rural, although they did not specify which definition, as they were concerned that if a State adopted a definition of rural that it might adopt a definition of rural that arbitrarily excludes certain clinics.
Another commenter recommended CMS adopt a definition of rural that is adopted by a State governmental agency with a role in setting rural health policy.
Finally, one commenter, in addition to recommending that CMS adopt a broad definition of rural, recommended that any clinic that furnishes services to individuals in rural areas under a range of definitions be allowed to qualify for the exception.
Response: We thank the commenters for sharing their recommendations on how we should approach defining rural in the final rule. As we noted in section XVIII.B of the proposed rule (89 FR 59186), there are many Federal and State definitions of rural for various programs, no single definition precisely identifies all rural areas, and different definitions might more accurately target a population meeting more of the criteria described in the proposed rule in different States. Some rural definitions may categorize areas that are generally recognized as suburban as rural, while other definitions may classify sparsely populated remote areas as urban. We believe that State-level variations may also affect whether certain ways of defining rural are appropriate in specific States. Further, under any definition of rural, the specific areas identified as rural may change over time and that would have a direct impact on the scope of clinics eligible for our proposed four walls exception.
We solicited comments on the following approaches to defining rural in the final rule: adopting one of the commonly used definitions of rural adopted by the Federal governmental agencies referenced previously, permitting a State to adopt a definition of rural that is adopted and used by a Federal governmental agency for programmatic purposes, permitting a State to adopt a definition of rural that is adopted and used by a State governmental agency with a role in setting State rural health policy, or not adopting any definition of rural. We appreciate all the different perspectives that commenters shared with us. We note, however, that the overwhelming majority of commenters recommended that CMS either not adopt a definition of rural or allow States to choose either a State or Federal definition of rural that best captures the rural population that meets the four criteria we described in the proposed rule.
Taking these comments into account, we believe that the best approach is to permit States to choose either a State or Federal definition of rural that best captures the State's rural population that meets the four criteria. Specifically, under the final rule, States implementing the exception must include in their State plans a definition of rural area. This definition must be either a definition adopted and used by a Federal governmental agency for programmatic purposes, or a definition adopted by a State governmental agency with a role in setting State rural health policy. As part of their State plan submission, States will need to attest that the selected definition falls into one of the two described categories and that it best captures the population of rural individuals that meets more of the four criteria described in this final rule. This approach gives States the flexibility to choose a definition that best targets the exception to a rural population that meets more of the four criteria we describe in this final rule. It also helps to ensure that States are using a definition that is commonly accepted and used in relevant contexts and generally accepted as a legitimate definition. States will be more likely to adopt a definition that may be more familiar to providers and be easier for a State to implement, since these definitions are also used for other purposes. We also agree with the concerns that commenters raised about how adopting a single Federal definition of rural could create barriers for beneficiaries and providers. This approach also recognizes that States may have the best information and data to determine the definition of rural that best meets the goals identified in the final rule, as well as the definition that best meets their operational needs. If, as some commenters requested, we instead finalized a Federal definition of rural that would apply in every State opting to implement this exception, that would not have accounted for State-specific variations in the degree to which different definitions might be better at targeting rural populations that meet more of the four criteria identified in the proposed rule in different States. Finally, because the definition will be identified in the State plan, it will be changed through the State plan amendment process, which gives CMS and the public notice of the change.
In the proposed rule, we did not include among the discussed options for defining rural an option under which any clinic that furnishes services to individuals in rural areas under a range of definitions would qualify for the exception, and are thus not including such an approach in this final rule. It is not clear to us how such an approach would effectively target a population that meets more of the criteria described in the proposed rule. Under the approach we are finalizing, in contrast, States will attest that the Federal or State definition of rural area that they are adopting best captures the population of rural individuals that meets more of the four criteria we described in the proposed rule.
After consideration of the public comments we received, we are finalizing an exception to the Medicaid clinic services four walls requirement at § 440.90(e) for clinics located in rural areas, with a modification that a State must include a definition of rural area in its State plan that must be either a definition adopted and used by a ( printed page 94458) Federal governmental agency for programmatic purposes, or a definition adopted by a State governmental agency with a role in setting State rural health policy.
6. Additional Four Walls Considerations
We proposed that the proposed exception to the four walls requirement for IHS/Tribal clinics would be a mandatory component of the clinic services benefit for States electing to cover that benefit. We proposed that the proposed exceptions for behavioral health clinics and clinics located in rural areas would be optional for States covering that benefit. In addition, we proposed to codify in regulation text our longstanding interpretation (discussed in section XVIII.A of the proposed rule) that existing § 440.90(a) and (b) are mandatory components of the clinic services benefit for States that elect to cover that benefit. Finally, we proposed to delete the word “eligible” from existing regulation text at § 440.90(b) because there is no Federal authority for States to provide Medicaid-covered services to individuals who are ineligible for Medicaid, so we believe it is unnecessary to specify that the individuals who would receive services under this exception are eligible.
We proposed to make the exception for IHS/Tribal clinics mandatory because the population served by IHS/Tribal clinics more consistently meets the four criteria described previously, both within and across States, than the populations targeted by the optional exceptions, especially given the degree of State variability in whether the populations targeted by the optional exceptions meet those criteria. Further, Medicaid is the largest source of third-party payment for services billed by IHS facilities, accounting for nearly two-thirds of health coverage payments to these facilities.[589] Given the significant role of Medicaid as a payer for IHS/Tribal clinic services, any reduction in the Medicaid payments IHS/Tribal clinics receive for services (such as a reduction in payment from the AIR to a professional services rate for services furnished outside the four walls by the clinic) might uniquely burden IHS/Tribal clinics. These clinics might need to curtail their available services, or no longer provide services outside the four walls, which could significantly impede their ability to serve their patients. For these reasons, we proposed a mandatory exception to the clinic services four walls requirement for IHS/Tribal clinics.
In contrast to the exception for IHS/Tribal clinics, we believe that the exceptions for behavioral health clinics and clinics located in rural areas should be optional because there may be geographic variability in the degree to which the populations served by these clinics meet the four criteria we described previously, and thus there may be State-specific variation in the degree to which these populations have the four characteristics described in the proposed rule. For example, the populations served by behavioral health clinics and clinics located in rural areas may not as consistently face transportation challenges nationwide, to the extent that Tribal populations do. In addition, it is our understanding that Medicaid funding is less often the largest source of payment for behavioral health clinics and clinics located in rural areas, compared to IHS/Tribal clinics. We believe it best to let each State assess the degree to which these two exceptions might be warranted based on the State's specific circumstances. In making this assessment, each State should consider the degree to which individuals located in rural areas of the State and/or individuals with behavioral health disorders in the State meet the four criteria described in the proposed rule. We solicited comment on the arguments made in the CY 2025 OPPS/ASC proposed rule in support of the mandatory and optional exceptions, and on whether the optional exceptions should also be mandatory for States opting to cover the clinic services benefit.
We explained that if we finalized the exceptions as proposed, then upon the effective date of the final rule, services qualifying for the exception for IHS/Tribal clinics must be paid for as Medicaid clinic services in States that opt to cover that benefit. Accordingly, we would require States that cover the clinic services benefit to submit a State plan amendment (SPA), as applicable, to attest to coverage of IHS/Tribal clinic services under the exception. Similarly, we explained that if we finalized the proposed rule as proposed, then no earlier than the effective date of a SPA or SPAs implementing one or both of the optional exceptions, services provided outside the four walls under the exceptions may be paid for as Medicaid clinic services. Under any of the exceptions, the excepted services could be paid for using a facility-based Medicaid clinic services payment methodology, which for most IHS/Tribal clinics is the AIR.
We did not propose any additional exceptions to the clinic services four walls requirement. It is our understanding that other populations are better able than those targeted by the proposed exceptions to access services through Medicaid benefits to which a four walls requirement does not apply under Federal Medicaid law (for example, FQHC services, RHC services, outpatient hospital services, etc.). As described in the proposed rule, States have considerable discretion regarding the types of clinics they opt to cover under the clinic services benefit. There are no specific Federal Medicaid credentialling requirements, such as licensure or certification, for providers of the Medicaid clinic services benefit like there are for other Medicaid facility State plan benefits, such as hospitals and nursing facilities. This leads to considerable variability in the types of clinics providing services that a State may cover under the clinic services benefit. We invited comment on whether there are additional populations that are likely to meet the four criteria described in the CY 2025 OPPS/ASC proposed rule and that have no alternative access to services through Medicaid benefits not subject to a four walls requirement under Federal Medicaid law, and on whether there are additional types of clinics that might serve as a proxy for such a population.
We did not receive any comments on our proposal to codify in regulation text our longstanding interpretation that existing § 440.90(a) and (b) are mandatory components of the clinic services benefit for States that elect to cover that benefit or on our proposal to delete the word “eligible” from existing regulation text at § 440.90(b). We received public comments on the following proposals: that the exception to the four walls requirement for IHS/Tribal clinics would be a mandatory component of the clinic services benefit for States electing to cover that benefit; that the exceptions for behavioral health clinics and clinics located in rural areas would be optional for States covering that benefit; and that we would not propose any additional exceptions to the clinic services four walls requirement. The following is a summary of the comments we received and our responses.
Comment: Most commenters expressed support for CMS's proposal at § 440.90 to make the IHS/Tribal clinic four walls exception mandatory for States that cover the clinic services benefit. In addition, many commenters supported CMS's proposal at § 440.90 ( printed page 94459) for the exceptions to the four walls requirement for behavioral health clinics at § 440.90(d) and clinics located in rural areas at § 440.90(e) to be at State option. Commenters noted that this would allow States to meet each State's unique populations, provider landscape, and program needs. A few commenters noted that States are in the best position to assess the extent to which their behavioral health and rural populations meet the four criteria we described in the proposed rule. In addition, one commenter noted that behavioral health services can be covered under several benefit categories under section 1905(a) of the Act; and that CMS's proposal that an exception for behavioral health clinics at § 440.90(d) be at State option would allow States to cover behavioral health services in a tailored way to recognize the unique needs of each States' behavioral health population.
However, many commenters also urged CMS to make the exception for behavioral health clinics at § 440.90(d) mandatory rather than optional for States that cover the clinic services benefit. The commenters explained that making the exception to the four walls requirement for behavioral health clinics mandatory would make services more accessible, strengthen States' behavioral health workforces, and more effectively enable providers to meet individuals where they are in need. Several commenters noted that the behavioral health provider shortage is at a crisis point nationally and therefore, an exception to the four walls requirement for behavioral health clinics should be mandatory.
Finally, two commenters recommended that CMS amend § 440.90 to make the exception for clinics located in rural areas at § 440.90(e) mandatory for States that cover the clinic services benefit. The commenters indicated that individuals in rural areas experience barriers in accessing health care within and across States, and they disagreed that there was State variation.
Response: We thank the commenters for their support for our proposal at § 440.90 to make the IHS/Tribal clinic four walls exception mandatory for States that cover the clinic services benefit. In addition, we thank the commenters who shared their recommendations on whether the exceptions for behavioral health clinics and clinics located in rural areas should be at State option or mandatory.
As we noted in section XVIII.B of the proposed rule (89 FR 59186), our proposal to make the IHS/Tribal clinic four walls exception mandatory and the exceptions for behavioral health clinics and clinics located in rural areas optional was based on the population served by IHS/Tribal clinics more consistently meeting the four criteria described in the proposed rule, both within and across States, than the populations served by behavioral health clinics or clinics located in rural areas. We indicated that this is especially true given the degree of State variability in whether the populations served by behavioral health clinics or clinics located in rural areas meet those criteria. Medicaid is the largest source of third-party payment for services billed by IHS/Tribal facilities; it is our understanding that Medicaid funding is less often the largest source of payment for behavioral health clinics and clinics located in rural areas compared to IHS/Tribal clinics.
While we appreciate the evidence and information that some commenters shared that there is a national behavioral health provider shortage and that a mandatory exception to the four walls requirement for behavioral health clinics could make services more accessible, strengthen States' behavioral health workforces, and more effectively enable providers to meet individuals where they are in need, we do not believe that the evidence or information included in some public comments contradicts our assumptions described in the preceding paragraph. The evidence and information shared by the commenters did not contradict our understanding that the population served by IHS/Tribal clinics more consistently meets the four criteria described in the proposed rule, that there is considerable State variability in the degree to which the behavioral health population meets the four criteria, and that Medicaid is less often the largest source of payment for behavioral health clinics than for IHS/Tribal clinics. The commenters supporting a mandatory exception to the four walls requirement for clinics located in rural areas did not share any data or evidence in support of their recommendation. We continue to believe, based on the information available to us, that the assumptions we describe in the preceding paragraph are accurate. We are therefore finalizing our proposal at § 440.90 to make the IHS/Tribal clinic four walls exception mandatory and the exceptions for behavioral health clinics and clinics located in rural areas at State option for States that cover the clinic services benefit.
Comment: Many commenters recommended CMS consider additional exceptions to the four walls requirement at § 440.90 in response to CMS's solicitation of comments on whether there are additional populations that are likely to meet the four criteria described in the proposed rule and that have no alternative access to services through Medicaid benefits not subject to a four walls requirement under Federal Medicaid law, and on whether there are additional types of clinics that might serve as a proxy for such a population. Most of the commenters that recommended additional exceptions to the four walls requirement at § 440.90 indicated that CMS's current interpretation of how the four walls requirement applies to Medicaid clinic services delivered via telehealth creates access to care issues for these populations.
Most of the commenters recommending that CMS include additional exceptions to the four walls requirement at § 440.90 advocated for exceptions for underserved and high-risk populations including racial and ethnic minorities, individuals with disabilities, LGBTQ+ individuals, low-income individuals, women, and individuals who are elderly; the commenters indicated they believed that these populations also meet the four criteria described in the proposed rule. A few commenters noted that transportation and access to care issues are also experienced in suburban and urban areas and that the populations served by clinics in these areas may also benefit from an exception from the four walls requirement.
One commenter recommended additional exceptions for specific types of services including maternity care, behavioral health care, urgent care, and emergency care, regardless of the type of clinic or population.
A few commenters requested that CMS include additional exceptions in the final rule for populations served by specific types of clinics. One commenter requested that CMS include an exception for school-based services that are covered in Medicaid as clinic services. Another commenter requested that CMS include a mandatory exception for essential community providers that operate as clinics and furnish services to underserved populations; the commenter indicated that they believe these underserved populations meet the four criteria described in the proposed rule, are more likely to meet the four criteria than other populations, and that Medicaid is a significant payer for essential community providers. A commenter requested that CMS include an additional exception to the four walls for clinics that are grandfathered by section 6411(g) of the Omnibus Budget Reconciliation Act of 1989 to provide ( printed page 94460) habilitation services under the clinic services benefit, to help individuals in hard-to-serve areas.
Finally, two commenters recommended an additional exception to the four walls requirement for UIOs and indicated they believe the population served by UIOs meets the four criteria described in the proposed rule. One of the commenters agreed with CMS's understanding that most UIOs participate in Medicaid as FQHCs while the other commenter disagreed with CMS and indicated that most UIOs do not participate in Medicaid as FQHCs.
Response: We thank the commenters that shared their recommendations for additional exceptions to the clinic services benefit four walls requirement at § 440.90 and acknowledge the concerns commenters raised regarding our current interpretation of how the four walls requirement applies to Medicaid clinic services delivered via telehealth. We also thank the commenters that took the time to describe why they believe additional exceptions may be warranted based on the four criteria described in the proposed rule.
As we noted in section XVIII.B of the proposed rule (89 FR 59186), it was our understanding that other populations are better able than those targeted by the proposed exceptions to access services through Medicaid benefits to which a four walls requirement does not apply under Federal Medicaid law (for example, FQHC services, RHC services, outpatient hospital services, etc.). We invited comment on whether there are additional populations that are likely to meet the four criteria described in the proposed rule and that have no alternative access to services through Medicaid benefits not subject to a four walls requirement under Federal Medicaid law, and on whether there are additional types of clinics that might serve as a proxy for such a population.
At this time, we believe that including additional exceptions to the four walls requirement would require additional consideration and discussion with the public beyond what could be finalized in this current rule. We will take these recommendations into consideration when determining whether possible future rulemaking on additional exceptions to the four walls requirement is warranted. We continue to believe that most UIOs participate in Medicaid as FQHCs, based on the information available to us, including the information shared by the two commenters. Finally, we will take these comments into consideration as we contemplate issuing sub-regulatory guidance regarding our interpretation of how the four walls requirement applies when Medicaid clinic services are delivered via telehealth.
After consideration of the public comments we received, we are finalizing, without modification, our proposals at § 440.90 that the exception to the four walls requirement for IHS/Tribal clinics would be a mandatory component of the clinic services benefit for States electing to cover that benefit, and that the exceptions for behavioral health clinics and clinics located in rural areas would be optional for States covering that benefit. In addition, we are finalizing that the existing regulation text at § 440.90(a) and (b) are mandatory components of the clinic services benefit for States that elect to cover that benefit and the deletion of the word “eligible” from existing regulation text at § 440.90(b).
XIX. Changes to the Review Timeframes for the Hospital Outpatient Department (OPD) Prior Authorization Process
The CMS Interoperability and Prior Authorization final rule (89 FR 8758) (Medicare and Medicaid Programs; Patient Protection and Affordable Care Act; Advancing Interoperability and Improving Prior Authorization Processes for Medicare Advantage Organizations, Medicaid Managed Care Plans, State Medicaid Agencies, Children's Health Insurance Program (CHIP) Agencies and CHIP Managed Care Entities, Issuers of Qualified Health Plans on the Federally-Facilitated Exchanges, Merit-Based Incentive Payment System (MIPS) Eligible Clinicians, and Eligible Hospitals and Critical Access Hospitals in the Medicare Promoting Interoperability Program) creates, improves, or shortens prior authorization timeframes for certain payers such as Medicare Advantage organizations and applicable integrated plans, CHIP FFS programs, Medicaid managed care plans, and CHIP managed care entities to respond to prior authorization requests for covered items and services, excluding drugs (89 FR 8878). The final rule requires impacted payers (excluding Qualified Health Plan issuers on the Federally-Facilitated Exchanges) to send prior authorization decisions as expeditiously as the enrollee's health condition requires or as the beneficiary's health condition requires but no later than 72 hours for expedited (that is, urgent) requests and 7-calendar days for standard (that is, non-urgent) requests.
As part of the CY 2020 OPPS/ASC final rule with comment period (84 FR 61446 through 61456), CMS established a nationwide prior authorization process and requirements for certain OPD services. OPD providers must submit to the Medicare Administrative Contractor (MAC) a prior authorization request for any service on the list of outpatient department services that require prior authorization. CMS currently requires prior authorization for the following services: blepharoplasty, rhinoplasty, botulinum toxin injections, panniculectomy, vein ablation, cervical fusion with disc removal, implanted spinal neurostimulators, and facet joint interventions. Upon receipt of the prior authorization request, the MAC should review it and issue a decision within specific timeframes, which are listed in the regulation text at § 419.82(d)(1)(iii) and (d)(2). These timeframes ensure providers receive timely responses and beneficiaries get appropriate care.
While Medicare FFS is not an impacted payer under the CMS Interoperability and Prior Authorization final rule, we proposed to align our Medicare FFS prior authorization review timeframe for standard review requests for hospital outpatient department services with the timeframe in this final rule. This change would not only streamline the prior authorization processes so that they are the same across payers but would also help to reduce provider burden by having the same timeframe and reducing the potential for delays in care by decreasing the time beneficiaries and providers wait for prior authorization decisions on standard requests in FFS Medicare. We proposed to change the current review timeframe for provisionally affirmed or non-affirmed standard review requests for these services from 10-business days to 7-calendar days in § 419.82(d)(1)(iii). For example, if a standard request is submitted on a Tuesday, June 2, under the new timeframe, a decision must be rendered by the next Monday, June 8, whereas under the old timeframe, the decision must be rendered by Monday, June 15.
We are still considering the impact of aligning our expedited review decision timeframe with the expedited review decision timeframe in the CMS Interoperability and Prior Authorization final rule because, depending on when the expedited request is submitted, it may take longer for OPD provider to receive a decision using the 72-hour timeframe than our current expedited timeframe of 2-business days. The goal of changing the standard review timeframe is not only to align the timeframe across the prior authorization programs but also to reduce the time ( printed page 94461) beneficiaries wait to access the care they need. Since changing the expedited review decision timeframe from 2-business days to 72 hours would not reduce beneficiaries' wait time in all circumstances, we did not propose to conform that timeframe with the one in the CMS Interoperability and Prior Authorization final rule at this time, but we may address this issue in future rulemaking.
We received 94 comments on the proposal, including comments from healthcare providers, professional, and trade organizations. The following is a summary of the comments we received and our responses.
Comment: We received comments in support of aligning the prior authorization review timeframe for the standard review requests with the timeframe in the Interoperability and Prior Authorization final rule.
Response: We appreciate the positive responses. This change would not only streamline the prior authorization standard timeframe so that it is the same across payers but would also help to reduce provider burden and beneficiaries' waiting time to receive care in FFS Medicare.
Comment: We received comments requesting that the standard timeframe and expedited timeframe be shorter than what we proposed. Another commenter asked CMS to clarify specific scenarios where it may take longer for an HOPD provider to receive an expedited decision using the CMS Interoperability and Prior Authorization final rule's 72-hour timeframe versus the current two-business day timeframe. Additionally, a commenter requested that CMS address prior authorization requests that occur immediately preceding a holiday or weekend.
Response: We thank the commenters for the feedback. As noted above, our proposed change to the standard timeframe aligns with the timeframe regulated in the CMS Interoperability and Prior Authorization final rule, and we believe that the 7-day timeframe for obtaining a decision on a prior authorization request is reasonable, considering that these are non-emergency procedures that typically require the beneficiary to undergo conservative treatment prior to the procedure. Changing the expedited review decision timeframe from 2-business days to 72 hours would not reduce beneficiaries' wait time in all circumstances. If an expedited request is submitted on Monday, August 5, at 8 a.m., under a 72-hour timeframe, a decision must be rendered by 8 a.m. on Thursday, August 8, whereas under the current timeframe of two business days, the decision must be rendered by the end of the business day on Tuesday, August 6. In contrast, when Monday is a Federal holiday and an expedited request is submitted on Saturday, August 10, at 8 a.m., under a 72-hour timeframe, the decision must be rendered by 8 a.m. on Tuesday, August 13, whereas under the current timeframe of 2-business days, the decision must be rendered by Wednesday, August 14. Our decision not to propose changes to the current timeframe for expedited requests was based on our recognition that a change to 72-hours will not always benefit beneficiaries.
Comment: Several commenters suggested that “gold carding” be approved for providers that have a history of prior authorization approval. We also received comments from mental health organizations concerning prior authorization requirements for mental health and substance use services, specifically that their comprehensive treatment plan is more restrictive and complex compared to the assessment for medical-surgical healthcare services. These commenters suggested updating comprehensive treatment plans that would help to improve beneficiary access to services.
Response: Thank you for the comment. We note that under the regulation at 42 CFR 419.83(c), we may elect to exempt a provider from the prior authorization process upon the providers' demonstration of compliance with Medicare coverage, coding, and payment rules. This exemption would remain in effect until we elect to withdraw the exemption. Providers who are exempt do not need to submit prior authorization requests. Additionally, mental health and substance use services do not require prior authorization under Medicare FFS. Also, and as we noted above, our prior authorization policy does not create any new documentation requirements.
Comment: Some commenters suggested that we remove implanted spinal neurostimulators and facet joint interventions from OPD prior authorization as they are contrary to efforts to promote non-opioid alternatives to pain management. One commenter stated that trends showed that utilization for facet joint interventions was decreasing without need for prior authorization requirements.
Response: We thank the commenters for their comments. We are committed to evaluating and adjusting our current guidelines and payment rules, if necessary, in order to ensure appropriate care for beneficiaries amid the current opioid epidemic. We acknowledge the benefits that implanted spinal neurostimulators and facet joint intervention services offer for chronic pain. At the same time, we reiterate the fact that these are non-emergency procedures that require the beneficiary to undergo conservative treatment prior to the procedure. Providers may also request expedited review of a prior authorization request, where the processing of the request must be expedited due to the beneficiary's life, health, or ability to regain maximum function being in jeopardy. To show the changes in the utilization of the services on the prior authorization list, we have posted statistics on the CMS OPD Prior Authorization and Pre-claim Review Initiatives website.[590] The statistics show the total number of prior authorization requests received and reviewed, the number of requests affirmed or non-affirmed, appeals and accuracy rates. We continue to analyze data associated with the services that require prior authorization and hopes to share additional data in the future.
We refer the commenters to the CY 2023 OPPS/ASC [591] and CY 2021 OPPS/ASC [592] final rules with comment period for our rationale on requiring prior authorization for facet joint interventions and implanted spinal neurostimulators.
Comment: We received comments requesting that CMS do more to streamline and automate the prior authorization process.
Response: Thank you for your comment. We recognize the value of automation in shortening the time between receipt of prior authorization requests and our responses. Finalizing the proposal to reduce the timeframe that beneficiaries wait to receive prior authorization decisions is one way to streamline the overall prior authorization process. We are aware that not all providers have the same level of technology and thus we allow various methods of submission of a prior authorization request. With regard to the hospital OPD prior authorization process, most providers submit requests and medical documentation to the MACs via individual MAC portals. Other providers submit the requests through the United States (U.S.) postal service, facsimile, or medical documentation (esMD) system. We continue to monitor and implement ( printed page 94462) other Federal and industry initiatives in order to improve the efficiency of our prior authorization processes, including increasing the number of requests submitted electronically in order to reduce provider burden, decrease delays in patient care, and promote high-quality, affordable health care.
Comment: Some commenters suggested that we eliminate outpatient hospital services prior authorization requirements as a way to improve patient access and reduce physician administrative burden. There were also concerns about prior authorization creating delays in patient care.
Response: Thank you for your comment. We remain fully committed to the agency's initiative to reduce unnecessary burdens while protecting our programs' sustainability by serving as a responsible steward of public funds. We believe that we have structured the prior authorization processes to effectively account for concerns associated with processing timeframes, patient care, and other administrative concerns. Finalizing the new timeframe should further alleviate the providers' burden and enable hospitals to receive timely provisional affirmations. Additionally, our prior authorization policy does not create any new documentation or administrative requirements. Instead, it just requires the same documents that are currently required to be submitted earlier in the process. Therefore, hospital OPDs should not need to divert resources from patient care. We note that prior authorization has the added benefit of giving hospitals some assurance of payment for services for which they received a provisional affirmation. In addition, beneficiaries have information regarding coverage prior to receiving the service and benefit by knowing in advance of receiving the service if they will incur financial liability for non-covered services. CMS will continue tracking MAC timeliness metrics and is confident that the MACs will continue to meet the required review and decision timeframes so as not to cause an additional burden for OPD providers or delay medically necessary services.
Comment: One commenter posed concerns about the time it takes to obtain prior authorization with Medicare Advantage plans, which often leads to delayed access to care.
Response: Thank you for the comment. While Medicare Advantage plans are outside the scope of the proposal, under the CMS Interoperability and Prior Authorization final rule, beginning January 1, 2026, Medicare Advantage organizations and applicable integrated plans must provide notice of prior authorization decisions as expeditiously as a patient's health condition requires, but no later than 7-calendar days for standard requests.
Comment: We received comments asking whether section 1833(t)(2)(F) of the Act grants CMS the authority to establish a prior authorization process.
Response: As we conveyed in the CY 2020 OPPS/ASC, CY 2021 OPPS/ASC, and CY 2023 OPPS/ASC final rules with comment periods, section 1833(t)(2)(F) of the Act gives us the discretion to determine the appropriate methods to control unnecessary increases in the volume of covered OPD services.
Comment: We received comments with concerns about unclear reasons for denial, challenges in understanding the prior authorization process, and that prior authorization could be discriminatory and worsen health disparities.
Response: CMS maintains a robust oversight process to ensure the accuracy and consistency of MACs' review decisions. In all Medicare Fee-for-Service medical review programs, we require that MACs utilize clinicians, specifically registered nurses when reviewing medical documentation. We also require the oversight of a medical director and additional clinician engagement if necessary to ensure that review decisions are accurate and written in plain language. We are committed to ensuring transparency of the prior authorization process. CMS publishes any changes to the prior authorization process, a list of services requiring prior authorization, an operational guide, frequently asked questions, and, as we already noted, program statistics on the CMS OPD Prior Authorization and Pre-Claim Review Initiatives website. Although specific data is not available to determine the actual impacts on underserved communities, CMS believes that finalizing the proposal to reduce the timeframe will help beneficiaries who meet Medicare requirements for services under prior authorization receive those services in a timelier manner. Also, CMS allows multiple methods to submit prior authorization requests to the MACs (for example, electronic, fax, mail) to account for differences in the utilization of technology among different providers. CMS continues to monitor to ensure that this OPD prior authorization program does not disproportionately impact underserved populations and works to advance health equity by operationalizing policies and programs that support health for all beneficiaries.
After consideration of the public comments we received, we are finalizing our proposal to change the current review timeframe for provisionally affirmed or non-affirmed standard review requests for covered OPD services subject to prior authorization from 10-business days to 7-calendar days and making this change in regulation text at § 419.82(d)(1)(iii).
XX. Provisions Related to Medicaid and the Children's Health Insurance Program (CHIP)
A. Continuous Eligibility in Medicaid and CHIP (42 CFR 435.926 and 457.342)
Prior to January 1, 2024, States had the option to provide up to 12 months of continuous coverage to children under age 19 enrolled in Medicaid or CHIP, regardless of changes in circumstances that otherwise would impact their eligibility for these programs. This option has been available to State Medicaid programs under section 1902(e)(12) of the Act and Federal regulations at § 435.926; and to States' separate CHIP programs through Federal regulations at § 457.342. Under this option, States had the option to elect an age limit under age 19 and/or continuous eligibility (CE) periods shorter than 12 months. However, except for the limited exceptions defined in the regulations, states could not terminate the coverage of children during a CE period.
Section 5112 of Title V, subtitle B of the Consolidated Appropriations Act, 2023 (CAA, 2023) amended section 1902(e)(12) of the Act to make the previously optional CE policy a requirement under the state plan or waiver of the state plan for children enrolled in Medicaid. The CAA, 2023 also added a new paragraph (K) to section 2107(e)(1) of the Act, adopting by reference Medicaid's CE policy into CHIP. Thus, effective January 1, 2024, States are required to provide a 12-month period of CE that offers continuous coverage to children under the age of 19 in Medicaid and CHIP, with limited exceptions.
B. Summary of the Proposed Provisions, Public Comments and Responses to Comments on Medicaid and CHIP continuous eligibility
We proposed to update the Medicaid regulations at § 435.926 to conform to changes to the CE policy effectuated by the CAA, 2023 amendments to section 1902(e)(12) of the Act, which are adopted by reference into the CHIP regulations at § 457.342(a). Specifically, we proposed to revise § 435.926(b) to specify that a State must provide CE for ( printed page 94463) the period specified in paragraph (c) (12 months). We also proposed to revise § 435.926(b)(1) to remove the option to limit CE to an age younger than 19. We further proposed to revise § 435.926(c)(1) to remove the option to limit CE to a period of time of less than 12 months. Finally, we proposed to revise § 435.926(d)(1) to remove the option of ending a CE period for a person when they reach the State-specified maximum age, as now all States must provide CE to children until they reach age 19. We also proposed to remove the option in § 457.342(b) to disenroll children from separate CHIP coverage for failure to pay required premiums or enrollment fees during a continuous eligibility period.
The existing Medicaid continuous eligibility regulation includes three exceptions that were unaffected by the CAA, 2023, and we did not propose changes to those exceptions. These exceptions permit States to terminate coverage for children during a CE period in specific circumstances that do not involve a re-determination of eligibility based on a change in the beneficiary's circumstances during the CE period. Those situations arise if the child or child's representative requests a voluntary termination of eligibility; the agency determines that eligibility was erroneously granted at the most recent determination, redetermination, or renewal of eligibility because of agency error or fraud, abuse, or perjury attributed to the child or the child's representative; or the child is deceased. The CAA, 2023 amended section 1902(e)(12) of the Act to make the CE option mandatory for state Medicaid programs, but it did not foreclose termination of coverage in these situations, which are important to maintain program integrity. The death or voluntary termination of a child does not involve the State's evaluation of eligibility at all, and therefore is not barred by the prohibition on reassessing eligibility based on changed circumstances during a CE period. Indeed, it is unclear how coverage could continue when the child has died or is voluntarily removed from the program. Similarly, a decision that coverage was improperly provided to begin with (based on mistake, fraud, abuse, or perjury) is a decision about eligibility before the CE period began, not a re-determination about eligibility based on changed circumstances during the CE period. We described our intention to retain these exceptions in CMS State Health Official (SHO) Letter #23-004, Section 5112 Requirement for all States to Provide Continuous Eligibility to Children in Medicaid and CHIP under the Consolidated Appropriations Act, 2023, which was issued on September 29, 2023.
For a full discussion of these proposals, please see the CY 2025 OPPS/ASC proposed rule (89 FR 59487).
We received over 70 timely comments on the proposals to require 12-months of continuous eligibility in Medicaid and CHIP and to eliminate nonpayment of premiums as an optional exception to continuous eligibility in CHIP, in accordance with sections 1902(e)(12) and 2107(e)(1)(K) of the Act as amended by CAA, 2023. A summary of the public comments and our responses is below.
1. Requiring 12 Months of Continuous Eligibility in Medicaid and CHIP
Comment: The vast majority of commenters were supportive of the Medicaid and CHIP Continuous Eligibility proposal published in the CY 2025 OPPS/ASC proposed rule. Commenters noted that individuals with continuous coverage experience fewer unmet health care needs and better health outcomes than individuals with gaps in coverage. In addition, commenters noted that uninterrupted health coverage may promote access to preventive care, necessary treatments for acute health needs, and continuity of care. Other commenters noted that individuals with chronic or serious health needs require consistent access to health care and that any gap in coverage, even for as little as 1 month, may put individuals at risk of delays in care and declining health. Commenters also noted that these provisions will reduce coverage gaps that may occur when individuals transition from Medicaid or CHIP to Marketplace coverage and reduce the rates of churning on and off coverage.
Response: We appreciate the support from these commenters. We are finalizing the Medicaid and CHIP continuous eligibility provisions as proposed.
Comment: One commenter suggested that CMS track rates of churn for children to help evaluate the effectiveness of the continuous eligibility policy.
Response: We appreciate the commenter's suggestion. We currently utilize data from the Transformed Medicaid Statistical Information System (T-MSIS) Analytic Files (TAF) to monitor churn across several eligibility groups. We will continue to monitor churn to inform the impact of this continuous eligibility policy.
Comment: A few commenters suggested that CMS extend the continuous eligibility requirements to populations not explicitly addressed by the CAA, 2023. Specifically, one commenter discussed the importance of continuous eligibility in ensuring that patients will not lose coverage mid-treatment or immediately following treatment. This commenter suggested that CMS finalize its proposal and consider future extensions of this policy for patients undergoing active treatment for serious illnesses. Another commenter suggested expanding continuous eligibility to vulnerable adults, including pregnant women or adults with disabilities. Another commenter suggested expanding continuous eligibility to all adults and children covered under any Medicaid eligibility category.
Response: We thank the commenters for these suggestions. At this time, we are only codifying the requirements of the CAA, 2023 related to providing continuous eligibility to children up to age 19 in Medicaid and CHIP.
We note that, with respect to continuing eligibility during acute care treatment, section 1902(e)(7) of the Act and § 435.172 currently protect children enrolled in the mandatory Medicaid eligibility group for infants and children under age 19 (described at § 435.118) who are receiving inpatient services when they age out of coverage in their eligibility group and would otherwise remain eligible for coverage. This provision requires that states provide these children with continued eligibility, despite exceeding the maximum age for the group, until the end of their inpatient stay.
For pregnant women, under section 1902(e)(6) of the Act and § 435.170(c), most pregnant Medicaid beneficiaries are eligible for coverage under the state plan through the end of the month in which a 60-day period, beginning on the last day of pregnancy, ends (referred to as the “60-day postpartum period”), regardless of changes in household income. States also have the option to extend CHIP eligibility to “targeted low-income pregnant women” through the end of the 60-day postpartum period, similar to Medicaid. States have the option under the state plan to provide 12 months of continuous postpartum coverage, regardless of any changes in circumstances, to all pregnant women in Medicaid and CHIP. Section 1902(e)(16) of the Act provides this authority in Medicaid, which is extended to CHIP through cross-reference at section 2107(e)(1)(J) of the Act. When the State elects this option in Medicaid, it must also make the same election in a separate CHIP, as required by section 1902(e)(16)(C) of the Act and cross-referenced in section 2107(e)(1)(J) of the ( printed page 94464) Act. As of September 2024, 46 States, the District of Columbia, and the U.S. Virgin Islands have elected to extend continuous postpartum coverage to 12 months. For more information on coverage options for pregnant women, please see the December 7, 2021 CMS State Health Official Letter #21-0007, “Re: Improving Maternal Health and Expanding Postpartum Coverage in Medicaid and the Children's Health Insurance Program (CHIP).” [593]
We encourage States that may be interested in extending continuous eligibility to other populations in Medicaid and CHIP or for children for longer than periods of 12 months to contact their section 1115 demonstration project officers to discuss options for doing so.
Comment: One commenter requested clarification regarding the start date of a child's continuous eligibility period and how the continuous eligibility period aligns with their renewal cycle. The commenter expressed concern that States may be setting renewal dates based on the date of the child's first approved application, rather than the application that led to the child's current coverage, which could result in a child losing coverage in under 12 months.
Response: As discussed in the September 29, 2023, State Health Official Letter #23-004, under 42 CFR 435.926 and 457.342, the 12-month continuous eligibility period begins on the effective date of the child's eligibility or most recent renewal of eligibility.
Because almost all children have 12-month eligibility periods and the 12-month CE period begins on the effective date of the child's most recent determination or redetermination of eligibility, a child's CE period generally will align with their renewal cycle, which is based on the child's current coverage period. Each time a child's eligibility is renewed in Medicaid or CHIP, the state must provide a new 12-month CE period. A child's coverage may not be terminated during the CE period, unless the child meets one of the exceptions to continuous eligibility enumerated at 42 CFR 435.926(d).
2. Removing Nonpayment of Premiums as an Optional Exception to Continuous Eligibility in CHIP
Comment: The majority of commenters supported removing nonpayment of premiums as an optional exception to continuous eligibility in CHIP. Commenters noted that requiring financial contributions from individuals may be a barrier to care for lower-income populations. Other commenters noted that removing this exception to continuous eligibility will allow children to have access to health care when they need it, regardless of the financial hardships families may face month to month.
Response: We appreciate the support from these commenters. We are finalizing this policy as proposed.
Comment: Several commenters suggested that CMS monitor States' responses to the elimination of the option to disenroll individuals due to nonpayment of premiums, which could create incentives for States or health plans to increase CHIP premiums to make up for lost revenue due to nonpayment. Another commenter expressed concern that CHIP managed care organizations could potentially place families in collections for the outstanding premiums. Another commenter requested strong monitoring and enforcement mechanisms to prevent unauthorized disenrollment practices and to ensure state compliance with this requirement.
Response: We appreciate the commenters' suggestions. We note that under section 2105(d)(3) of the Act, States shall not have in effect eligibility standards, methodologies, or procedures under a CHIP State plan or waiver of a State plan that are more restrictive than the eligibility standards, methodologies, or procedures under such plan (or waiver) as in effect on March 23, 2010. This requirement applies to children in families whose income does not exceed 300 percent of the Federal poverty level (FPL) and remains in effect through September 30, 2029.[594] These requirements are referred to as the “maintenance of effort” or MOE provisions. In the February 25, 2011, State Medicaid Director Letter #11-001,[595] CMS clarified that for CHIP, increases to existing premiums or the imposition of new premiums to existing eligibility groups are not consistent with MOE. States may only adopt, through State plan or demonstration amendments, certain inflation-related adjustments to premium levels. These adjustments must be based on (and no more than) the percentage increase in the Consumer Price Index (CPI) trended forward using the applicable CPI for Medical Care (or another State specific index submitted by the State and approved by CMS). Therefore, States may not increase their premiums to account for nonpayment of premiums, beyond the amounts charged on March 23, 2010, other than by an inflation-related adjustment, at least through September 30, 2029.
We also note that the CHIP statute and these regulations do not prohibit States or health plans from pursuing the collection of past due premiums. However, States can prohibit the CHIP managed care organizations from placing families in collections for the outstanding premiums through their contractual arrangements with the plans, and attempt to collect any past due premiums through other means.
With regards to enforcement, all States will be required to submit a CHIP State plan amendment (SPA) to demonstrate compliance with the provision of this final rule regarding removing nonpayment of premiums as an exception to continuous eligibility in CHIP. Most States are already in compliance with this provision and have submitted SPAs to demonstrate compliance. CMS will provide technical assistance to any State that needs assistance coming into compliance with the requirements of this provision. As noted above, we will also be using TAF data to monitor churn and expect to see modest drops in churn for states newly adopting continuous eligibility.
Comment: One commenter suggested that CMS further reduce barriers to coverage by eliminating premiums or the ability to disenroll children due to nonpayment of premiums in Medicaid and CHIP.
Response: For CHIP, Section 2103(e) of the Act allows for the imposition of premiums, subject to the limitations described in section 2103(e)(3). For children with family income below 150 percent of the FPL, premiums may not exceed the maximum monthly charge permitted consistent with standards established to carry out section 1916(b)(1) of the Act (with respect to individuals described in such section). For children with family income above 150 percent of the FPL, total aggregate cost-sharing (including premiums) may not exceed 5 percent of the family's income for the year. In Medicaid, sections 1902(a)(14), 1916, and 1916A of the Act permit States to require certain beneficiaries to share in the costs of providing medical assistance through premiums and cost sharing. Most premiums for children assessed in Medicaid are subject to the aggregate cost sharing limit of 5 percent of family income.
In the absence of a statutory requirement preventing disenrollment, ( printed page 94465) like the continuous eligibility requirement under section 1902(e)(12) and 2107(e)(1)(K) of the Act, we do not believe the Medicaid and CHIP statutes limit states' ability to disenroll children due to nonpayment of premiums at the end of a CE period provided required procedures are followed.
Comment: A few commenters requested specific technical assistance from CMS regarding these policies. One commenter noted that CHIP health plans may collect premiums from beneficiaries and that CMS should release guidance for States on working with CHIP health plans that are collecting premiums, if there is an instance of non-payment during a continuous eligibility period. The commenter emphasized the importance of standardizing implementation across states and reducing administrative and financial burden for CHIP health plans. Another commenter suggested CMS provide States with template notices to send to beneficiaries in order to alleviate enrollee confusion and administrative burden for States. Another commenter requested that CMS provide outreach and education efforts to inform beneficiaries of these policy changes and provide technical assistance to states in implementing these changes.
Response: We appreciate the suggestions and are working on additional guidance regarding implementation of the policies in this final rule. States with technical assistance needs regarding notices or other implementation areas should reach out to their Medicaid State leads or CHIP project officers for assistance.
Comment: Two commenters were unsupportive of removing nonpayment of premiums as an optional exception to continuous eligibility in CHIP. Specifically, these commenters requested clarity regarding the authority under which CMS can remove this previously optional exception and cited concerns regarding the potential increased costs to states or health plans if premiums are unpaid. One commenter stated the CAA, 2023 does not require CMS to remove nonpayment of premiums as an exception to continuous eligibility in CHIP. One commenter was concerned that removing nonpayment of premiums as an optional exception to continuous eligibility in CHIP would render CHIP an entitlement program.
Response: Under prior law, the Medicaid statute gave States the option to include in their State plans a “continuous eligibility” period of up to 12 months for children under 19. During this period, a child “determined to be eligible for benefits under” a State plan generally had to remain “eligible for those benefits.” In 2016, CMS promulgated a regulation implementing this section of the Medicaid statute. The regulation specified that “[a] child's [Medicaid] eligibility may not be terminated during a continuous eligibility period, regardless of any changes in circumstances, unless” one of five exceptions was met: (1) the child attains the maximum age specified in accordance with § 435.926(b)(1); (2) the child or child's representative requests a voluntary termination of eligibility; (3) the child ceases to be a resident of the State; (4) the agency determines that eligibility was erroneously granted at the most recent determination, redetermination or renewal of eligibility because of agency error or fraud, abuse, or perjury attributed to the child or the child's representative; or (5) the child dies. Notably, this regulation did not permit termination of Medicaid coverage for nonpayment of premiums during periods of continuous eligibility, consistent with then-applicable (and the amended) version of section 1902(e)(12) of the Act and preexisting Medicaid regulations in 42 CFR 435.930(b), which provides that states “must . . . [c]ontinue to furnish Medicaid regularly to all eligible individuals until they are found to be ineligible.”
At the same time, CMS also issued a regulation permitting states to include a similar period of continuous eligibility in their CHIP plans, under 42 CFR 457.342. This regulation stated that, “[i]n addition to the reasons provided at § 435.926(d) of this chapter”—that is, the five exceptions set forth above permitting termination of Medicaid eligibility—an additional exception applied in the CHIP context, where “a child may be terminated during the continuous eligibility period for failure to pay required premiums or enrollment fees required under the State plan,” id. § 457.342(b). This exception did not apply to Medicaid—it was unique to CHIP. Notably, prior to the amendments made by the CAA, 2023, there was no statutory provision specifically addressing provision of continuous eligibility under CHIP; this option was implemented through regulations only.
The CAA, 2023 made two fundamental changes relevant to continuous eligibility. First, it amended section 1902(e)(12) of the Medicaid statute to provide that State plans “shall” include a 12-month continuous eligibility period, which had previously been optional. Second, it amended the CHIP statute to adopt this Medicaid continuous eligibility requirement by reference. As a result, the CHIP statute now reads that section 1902(e)(12) of the Medicaid statute, as amended by section 5112(a) of the CAA, “shall apply to States under this subchapter [ i.e., CHIP] in the same manner as [it] appl[ies] to a State under subchapter XIX [ i.e., Medicaid].” The sole exception to this “in the same manner” directive is that CHIP coverage may end if a child “becomes eligible for full benefits” and is “transferred to the [state's] Medicaid program . . . for the remaining duration of the 12-month continuous eligibility period.”
Accordingly, Congress made clear that the 12-month period of continuous eligibility under Medicaid must apply “in the same manner” with respect to CHIP coverage. And termination of coverage for nonpayment of premiums is not permitted during periods of continuous eligibility under Medicaid. Thus, termination for nonpayment of premiums is not permitted during a 12-month period of continuous eligibility under CHIP either. When Congress required that continuous eligibility be applied “in the same manner” in the two programs, Congress necessarily understood that CHIP beneficiaries would now receive uninterrupted coverage during the continuous eligibility period, as Medicaid beneficiaries already did. Therefore, the CAA, 2023 compels CMS to remove nonpayment of premiums as an exception to continuous eligibility in CHIP, just as nonpayment of premiums previously did not serve as an exception to the continuous eligibility option provided under the Medicaid statute. We also note that continuous eligibility can lead to more efficient care and better health outcomes, by stabilizing health coverage. In order to achieve this, unnecessary exceptions to continuous eligibility that are not provided for by statute, such as the exception for nonpayment of premiums, must be eliminated.
Regarding costs to States or managed care organizations (MCOs), States are not prohibited from entering or renegotiating contract `per-member, per-month' (PMPM) amounts that take into consideration that supplemental MCO revenue could be reduced if the MCO is not able to collect 100 percent of premiums, which could result in an increase in state expenditures to cover the MCO's PMPM cost of care. However, the state may only claim FFP for the expenses set out in section 2105(a)(1)(A) through (D) of the Act. Uncollected premiums are not themselves considered an `expense' within the meaning of section 2105. Although States may have decreased revenue due to unpaid premiums, States may find ( printed page 94466) administrative costs savings or reduced administrative burden, as a result of fewer children churning on and off coverage. Continuous eligibility, including eliminating disenrollment for failure to pay premiums, can reduce the need for repeatedly reviewing eligibility, disenrolling children, and then re-enrolling them.[596]
Removing nonpayment of premiums as an exception to continuous eligibility in CHIP does not render CHIP an entitlement program. States are not prohibited from charging and pursuing the collection of past-due premiums or from requiring an initial premium or enrollment fee to be paid prior to an individual enrolling in coverage. Rather, States simply may not disenroll individuals due to nonpayment of premiums during the continuous eligibility period.
Comment: One commenter questioned if removing this optional exception conflicted with the CHIP premium grace period requirements. Another commenter asked if individuals can be disenrolled after nonpayment of one premium or multiple premiums.
Response: Removing nonpayment of premiums as an exception to continuous eligibility does not conflict with the CHIP premium grace period requirements addressed in section 2103(e)(3)(C) of the Act. Under section 2103(e)(3)(C)(ii) of the Act, the State must provide the child with a grace period of “at least 30 days from the beginning of a new coverage period to make premium payments before the individual's coverage” may be terminated. Section 2103(e)(3)(C)(ii)(II) of the Act defines “new coverage period” as “the month immediately following the last month for which the premium has been paid.” Section 2103(e)(3)(C)(ii) of the Act also requires the State to provide notice no later than 7 days after the first day of the grace period (typically 7 days after the premium payment was due) that failure to make a premium payment within the grace period will result in termination of coverage and when such termination will be effective.
Disenrollment may occur at the end of the continuous eligibility period after nonpayment of one or multiple premiums, depending on how the State has structured their premium policies. States may disenroll individuals at the end of the continuous eligibility period for nonpayment of premiums during the continuous eligibility period, provided the State has afforded the enrollee a minimum of 30 days to pay the past-due premiums. If a State requires monthly premiums that are due in advance of the month of coverage, it is possible that an individual may be disenrolled at the end of the continuous eligibility period after nonpayment of only one premium payment.
Example: Jose is enrolled in CHIP with a 12-month continuous eligibility period of January 1, 2024, through December 31, 2024. The State requires that any unpaid premiums owed for one or more months in a child's current CE period must be paid prior to the end of the CE period in order to avoid termination at the end of the CE period. Jose's family paid the monthly premiums from January through November, due on the 1st of each respective month. However, the family did not pay the required premiums due for December.
Consistent with its renewal process, the State initiates the ex parte process for Jose's renewal on October 1, 2024. The State is unable to determine Jose's eligibility based on available information. The State therefore sends the family a pre-populated renewal form on November 1 and provides the family with 30 days to respond. Jose's family returns the renewal form (along with any requested documentation) later that month, and the State begins to process the information received. On December 7, the State sends Jose's family the notice required under section 2103(e)(3)(C)(ii) of the Act that Jose's coverage will end on December 31, 2024, if the family does not pay the December premium by that date. On December 10, the State completes processing Jose's renewal and determines that Jose continues to meet the other eligibility requirements. However, because Jose's family failed to pay the December premium, the State will not send Jose's family a notice that his eligibility is being renewed. Rather, the State would send Jose's family a notice explaining that: (1) it has determined that Jose meets the other eligibility requirements to have his coverage renewed but; (2) in order for Jose's coverage to be renewed for another 12 months effective January 1, 2025, the family must pay the December premium by December 31. If the State requires that the premium for January 2025 be paid prior to renewing Jose's coverage, the notice must include this information as well.
If the family pays the outstanding premium by December 31, 2024, Jose cannot be terminated due to failure to pay the premium. If Jose's coverage is erroneously terminated (for example, because the family did not submit the premium until December 31 and the State was unable to reverse the termination), the State must reinstate his coverage effective January 1, 2025. Conversely, if the family does not pay the outstanding premium by December 31, Jose can be terminated effective January 1, 2025, due to failure to pay the premium.
Comment: One commenter questioned if States could require CHIP enrollees to pay an initial premium or enrollment fee prior to enrolling in coverage, given this is prohibited in Medicaid. This commenter suggested that if continuous eligibility is to apply in the same manner in CHIP as it does in Medicaid, this practice may be prohibited.
Response: States have long had the option to require individuals who are determined eligible for separate CHIP coverage to pay an initial premium or enrollment fee prior to enrolling the individual in coverage. This rule does not preclude States from continuing this practice. Rather, this rule requires that once enrolled in coverage and the continuous eligibility period begins, States may not disenroll individuals from coverage due to nonpayment of premiums during the continuous eligibility period.
In Medicaid, section 1916(c)(3) of the Act specifies that a state shall not require prepayment of a premium imposed. Additionally, section 1902(a)(8) of the Act specifies that the State must provide that all individuals wishing to make application for medical assistance under the plan shall have opportunity to do so, and that such assistance shall be furnished with reasonable promptness to all eligible individuals. These statutory requirements do not apply to CHIP, and thus, States may be permitted to require the payment of an initial premium or enrollment fee prior to enrolling a child in CHIP. Once the period of continuous eligibility begins, however, that period applies “in the same manner” as in Medicaid, and therefore coverage cannot be terminated for nonpayment of premiums.
Comment: One commenter stated their State law requires disenrollment for failure to pay premiums. The commenter questioned if these regulations could apply to State programs that were grandfathered into CHIP, when it was created in 1997.
( printed page 94467)Response: We recognize that certain changes in this final rule, including eliminating failure to pay premiums as an optional exception to continuous eligibility, may require updates to information technology systems or changes in State laws. However, the vast majority of States have already implemented the Medicaid and CHIP continuous eligibility provisions in this final rule as of January 1, 2024, the effective date of the provisions under the CAA, 2023. Moreover, these regulations will be effective January 1, 2025, which provides additional time for States to make necessary changes. As previously mentioned, CMS is available to assist States with any technical assistance needs related to implementation of these provisions.
These regulations do apply to States whose programs were grandfathered into CHIP when it was created in 1997. CHIP regulations in § 457.410(a)(3) specify that States may offer health benefits coverage in the form of an existing comprehensive State-based coverage, in accordance with § 457.440. These two regulations describe the requirements regarding the benefits grandfathered health plans must provide but do not relieve States from complying with other programmatic requirements, such as requirements related to eligibility for coverage. As described above, CMS is available to provide technical assistance to States on these requirements.
After consideration of the public comments we received, we are finalizing our proposed revisions to Medicaid and CHIP regulations to codify the requirement within the CAA, 2023 to require States to provide 12 months of continuous eligibility to children under the age of 19 in Medicaid and CHIP, with limited exceptions. We are also finalizing our proposal to remove nonpayment of premiums as an optional exception to continuous eligibility in CHIP.
XXI. Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals
A. Background and Statutory Authority
CMS has broad statutory authority to establish health and safety regulations, which includes the authority to establish requirements that protect the health and safety of pregnant, postpartum, and birthing patients. Several statutes applicable to specific provider and supplier types explicitly give CMS the authority to enact regulations that the Secretary finds necessary in the interest of the health and safety of individuals who are furnished services in an institution, while others give CMS the authority to prescribe regulations as may be necessary to carry out the administration of the program.
Sections 1861(e)(1) through (8) of the Social Security Act (the Act) provide that a hospital participating in the Medicare program must meet certain specified requirements. Section 1861(e)(9) of the Act specifies that a hospital also must meet such other requirements as the Secretary finds necessary in the interest of the health and safety of individuals furnished services in the institution. Under this authority, the Secretary has established regulatory requirements that a hospital must meet to participate in Medicare at 42 CFR part 482, “Conditions of Participation [CoPs] for Hospitals.” Section 1905(a) of the Act provides that Medicaid payments from States may be applied to hospital services. Under regulations at 42 CFR 440.10(a)(3)(iii) and 440.20(a)(3)(ii), Medicaid-participating hospitals are required to meet the Medicare CoPs in order to participate in Medicaid.
Sections 1820 and 1861(mm) of the Act, as amended by section 4201 of the Balanced Budget Act (BBA) of 1997, replaced the EACH/RPCH program with the Medicare Rural Hospital Flexibility Program (MRHFP), under which a qualifying facility can be designated and certified as a critical access hospital (CAH). CAHs participating in the MRHFP must meet the conditions for designation specified in the statute under section 1820(c)(2)(B) of the Act, and to be certified must also meet other criteria the Secretary may require, under section 1820(e)(3) of the Act. Under this authority, the Secretary has established regulatory requirements that a CAH must meet to participate in Medicare at 42 CFR part 485, subpart F.
The CoPs for hospitals and CAHs are organized according to the categories of services a hospital or CAH may offer, and include specific, process-oriented requirements for each hospital or CAH service or department. The purposes of these CoPs are to protect patient health and safety and to ensure that quality care is furnished to all patients in Medicare-participating hospitals and CAHs. In accordance with section 1864 of the Act, State surveyors assess hospital and CAH compliance with the conditions as part of the process of determining whether a hospital qualifies for a provider agreement under Medicare. However, under section 1865 of the Act, hospitals and CAHs can elect to be reviewed instead by private accrediting organizations approved by CMS as having standards that meet or exceed the applicable Medicare standards and survey procedures comparable to those CMS requires for State survey agencies.
B. The U.S. Maternal Health Crisis
The U.S. is currently facing a maternal health crisis which has not only led to a maternal mortality rate that is among the highest in high-income countries, but also disproportionately affects racial and ethnic minorities. In 2022, the most recent year for which there is data, there were 22 maternal deaths for every 100,000 live births in the U.S. which is more than double the rate for most other high-income countries. For example, in 2022, Canada, France, the United Kingdom, Germany, and Japan had maternal death rates of 8.6 deaths per 100,000 live births or lower.[597] Over 80 percent of pregnancy-related deaths are considered preventable.[598] Approximately 13 percent of all pregnancy-related deaths (a death during pregnancy or within 1 year of the end of pregnancy from a pregnancy complication, a chain of events initiated by pregnancy, or the aggravation of an unrelated condition by the physiologic effects of pregnancy) occur at the time of delivery, and nearly 12 percent occur between 1 and 6 days after the end of pregnancy.[599]
Native Hawaiian and Pacific Islander women, Black women, and American Indian/Alaska Native (AI/AN) women are two to four times more likely to suffer a pregnancy-related death than non-Hispanic White women.[600] Black and AI/AN women experience severe maternal morbidity rates that are more than two times higher than their White counterparts.[601 602] Systemic societal barriers, including a patient's social determinants of health, have meant that these individuals experience a greater ( printed page 94468) share of these poor maternal health outcomes.[603 604 605]
Pregnant women who live in rural communities are about 60 percent more likely to die before, during, or following birth than women in urban communities.[606] Pregnant women with disabilities receive lower quality maternity care, experience a higher risk of pregnancy and birth-related complications, and are eleven times more likely to experience maternal death than people without disabilities.[607 608]
Based on the issues regarding the delivery of maternity care referenced, we proposed a new OB services CoP, including proposed requirements for the organization, staffing, and delivery of OB services and staff training. We also proposed revisions to the current hospital and CAH QAPI requirements, hospital and CAH emergency services requirements, and hospital discharge planning requirements. We also solicited comments on whether these proposed requirements should also apply to REHs.
C. Summary of the Proposed Provisions, Public Comments and Responses to Comments on Health and Safety Standards for Obstetrical Services in Hospitals and Critical Access Hospitals
In response to these proposals, we received 153 total comments. A broad range of individuals, groups, and organizations responded to the proposed rule. Commenters included hospitals, healthcare systems, hospital associations, medical associations and societies, professional trade organizations, patient advocacy organizations, and individuals, among others.
Our goal is to establish new requirements for the provision of obstetrical services to protect the health and safety of pregnant, birthing, and post-partum patients. These requirements are designed to establish baseline standards for obstetrical care and, in many cases, mirror existing optional services in other areas of the Conditions of Participation for hospitals, which helps to minimize burden. These requirements do not dictate standards of care or otherwise require hospitals to offer any specific type of care to patients. We believe that these requirements are balanced and will drive improved care for pregnant, birthing, and post-partum patients.
In this final rule, we provide a summary of each proposed provision, a summary of the public comments received and our responses to them, and an explanation for changes in the policies that we are finalizing.
1. General Comments
Comment: Many commenters expressed strong support for the proposed CoPs and CMS' efforts to improve maternal health with many commenters acknowledging the importance of addressing health disparities and social determinants of health. These commenters largely supported the overall goal of the proposed requirements and applauded these requirements as a major and historic step to improving maternal health outcomes. Some commenters also stated that these requirements were essential in addressing maternal health disparities. A few commenters also observed that these requirements will improve health equity by establishing standards and procedures across all hospitals, ensuring that facilities are held to a consistent standard of high-quality care. Additionally, a commenter noted that these requirements are a crucial step to transforming the health care system to be more responsive to the peri-natal needs of high-risk patients and communities. A few commenters also expressed support for the use of standardized practices for health care, quality measures, training, and assessments. Further, a commenter recommended future continuous evaluation of the standards to further positively impact patient care.
Response: We thank commenters for their support. We believe these requirements are an important component of the agency's efforts to improve maternal health and advance health equity. CMS routinely reviews new evidence, research, and information and may consider the possibility of revising the standards in the future if additional updates would be appropriate to improve quality and further protect the health and safety of patients.
Comment: Some commenters expressed support for these proposals, indicating their beliefs that the new requirements would promote consistent, high-quality maternal health care across the country, especially following the Dobbs v. Jackson Women's Health Organization (597 U.S. 215 (2022)) decision and related State policy changes restricting access to reproductive health care and its impact on the provision of emergency services. With this, many commenters recommended that CMS strengthen or reinforce the Emergency Medical Treatment and Labor Act (EMTALA) protections for patients experiencing pregnancy-related emergencies to reduce maternal morbidity and mortality. A commenter expressed concern that there is lack of clarity between the Federal requirements under EMTALA and State abortion restrictions. This commenter expressed concern that without EMTALA protections patients may face delays in accessing medically necessary emergency care. In addition, other commenters recommended that CMS remind hospitals of their legal responsibility to provide stabilizing treatment to pregnant women and requested that CMS work with policymakers and stakeholders to uphold EMTALA protections related to maternity care, especially in States with laws restricting women's health care. Further, commenters stated that these requirements were urgently needed given the nationwide shortage in OB providers, particularly in rural areas and southern States and would ensure timely and effective treatment for pregnant women regardless of their geographic location.
Response: We appreciate support for these requirements. With these requirements, we aim to support the provision of timely, effective, and high-quality maternal care. Of note, these requirements do not dictate standards of care or otherwise require hospitals to offer any specific type of care to patients. Rather, we proposed to require hospitals to have appropriate protocols and adequate provisions in place to meet the needs of patients with emergency conditions, including but not limited to patients who are experiencing obstetrical emergencies, complications, and immediate post-delivery care in alignment with the facility's complexity ( printed page 94469) and scope of services. Hospitals will have the flexibility to develop their own protocols consistent with evidence-based, nationally recognized guidelines.
Other laws, such as the Emergency Medical Treatment and Labor Act (EMTALA), require certain facilities with emergency departments to provide an appropriate medical screening exam and, if an emergency medical condition exists, offer stabilizing treatment or, under certain circumstances, appropriate transfer for a patient to receive stabilizing care. HHS reaffirmed its commitment supporting hospitals in complying with Federal requirements under EMTALA, consistent with binding court orders, and enforcing these requirements as recently as July 2, 2024.[609] Following the Supreme Court's decision in Moyle v. United States, HHS and CMS leadership jointly sent a letter to hospitals and provider associations reminding them of relevant facilities' legal responsibilities under EMTALA.[610] HHS will continue to partner with hospital and provider associations to disseminate training on facilities' obligations under EMTALA and maintain a team of HHS experts to support hospitals in complying with EMTALA.[611] Further, HHS and CMS also introduced a new EMTALA complaint form in both English and Spanish and issued updated model signage for hospitals, to help further educate patients about their rights under EMTALA.[612 613] The electronic EMTALA form is also now available on the CMS website.[614] We emphasize that EMTALA requirements are separate and distinct from these requirements. Rather, these rules will require hospitals to adopt protocols and procedures so as to be adequately prepared to meet both the emergency and non-emergency needs of all patients, including those with obstetrical emergencies.
Comment: Many commenters recommended that CMS withdraw the proposed CoPs for OB services and the proposed revisions to the current CoPs. Some stated that CMS should engage in additional public outreach, such as working with hospitals and other interested parties to adopt guidelines, prior to developing policies for improving maternal health. Many commenters did not support the adoption of CoPs related to maternal health because of the belief that CoPs are not the appropriate mechanism to achieve change, and their view that the potential risk of losing Medicare certification for not meeting the proposed CoPs is excessively punitive for hospitals. These commenters stated that adoption of CoPs may limit flexibility for hospitals to address the needs of their patient populations. Some of these commenters suggested alternative mechanisms to achieve change including technical assistance programs, learning collaboratives, and well-tested quality metrics within the CMS Quality Reporting programs. Commenters stated that by engaging interested parties, CMS will have an opportunity to better understand the positive and negative impacts of potential policies. Lastly, a commenter stated that because the number of Medicare patients seeking maternity care is low, a more targeted approach through Medicaid would be more appropriate to implement these changes.
Response: As we previously noted, the U.S. is currently facing a maternal health crisis which has not only led to a maternal mortality rate that is amongst the highest in high-income countries, but also disproportionately affects racial and ethnic minorities. After analyzing the issue of high rates of maternal mortality and morbidity in the U.S., receiving feedback from various stakeholders on improving maternal health care, and reviewing available resources and current requirements, we believe that it is necessary to establish new baseline requirements for the provision of obstetrical services to protect the health and safety of pregnant, birthing, and postpartum patients. Doing so would be part of a multi-pronged HHS-wide approach to address the maternal health crisis and would hold hospitals and CAHs accountable to a consistent set of high-quality requirements that would improve outcomes for pregnant, birthing, and postpartum patients. We therefore are finalizing our proposals, with the modifications discussed below.
We also note that the role of the CoPs is to establish health and safety standards that health care providers and suppliers (in this case hospitals and CAHs) must meet to participate in the Medicare and Medicaid programs. These standards are foundational for improving quality and protecting the health and safety of all patients seeking care from these facilities including pregnant, postpartum, and birthing patients. As noted above, these requirements do not dictate standards of care or otherwise require hospitals to offer any specific type of care to patients. As discussed in the CY 2025 OPPS/ASC proposed rule (89 FR 59488) some of these patients are currently not receiving consistent high quality, safe care. Therefore, it is appropriate to expand the CoPs to ensure that the care provided to these patients meets minimum quality standards, in alignment with the facility's complexity and scope of services. We note that the CoPs apply to all patients in facilities, not only those covered by Medicare and Medicaid.
We appreciate commenters' suggestions regarding other channels for improving the quality and safety of obstetric services. We recognize that updating the CoPs for hospitals and CAHs is not a complete solution to the maternal health crisis and that updating the CoPs for hospitals and CAHs is one element of the HHS-wide approach to strengthening maternal health, among other activities.[615] Across the Department, other relevant activities include developing and implementing a strategy to improve maternal mental health care,[616] to extend postpartum coverage,[617] and to support innovative programs to address racial equity for postpartum patients enrolled in Medicaid.[618] Within CMS, as part of our ( printed page 94470) Maternity Care Action Plan,[619] we have established the “birthing friendly” designation [620] for hospitals and have established funding through CMS' Innovation Center model (the Transforming Maternal Health Model) focused developing whole-person approaches to pregnancy, childbirth, and postpartum care.[621] In addition, the Maternal Morbidity Structural Measure requires hospitals participating in the Hospital Inpatient Quality Reporting Program (IQR) to assess whether or not a hospital participates in a statewide or national Perinatal Quality Improvement (QI) Collaborative initiative, and implements patient safety practices and/or bundles related to maternal morbidity from that QI Collaborative.[622] We note in our current QAPI proposal that participating in Perinatal QI Collaborative would be consistent with meeting this new requirement. Other specific hospital IQR maternal health quality measures include: (1) measurement of severe obstetric complications (which describes the number of inpatient hospitalizations for patients with severe complications occurring during the delivery hospitalization, such as hemorrhage), and (2) measurement of low-risk Cesarean section rates, which describes the share of patients with low-risk pregnancies who give birth via a Cesarean section.[623] Given that a significant portion (42 percent) of U.S. births are covered by the Medicaid, the Medicaid program has also prioritized improving maternal and infant health care quality. This includes the Maternal and Infant Health Initiative (MIHI), which has supportive tools such as a webinar series, affinity groups, and a quality improvement resource list.[624]
While we understand commenters' concern that failure to comply with the Medicare CoPs may result in loss of certification, we note that if a surveyor identifies deficiencies during their survey, the initial step is for the hospital or CAH to develop a Plan of Correction (PoC) to remedy the deficiency. This activity is intended to ensure that the hospital or CAH becomes compliant with the CoPs instead of requiring that CMS terminate the hospital from its programs. CMS remains committed to collaborating with hospitals and CAHs, including by providing technical assistance and issuing interpretive guidance to help hospitals and CAHs come into compliance with the CoPs.
With regards to comments on public outreach, we note that public outreach is a vital element of understanding the potential impacts of policies on facilities and their patients, and we did so when developing these proposed updates to the CoPs. As part of that outreach, in 2022 we solicited public input on a wide range of maternal health issues (86 FR 44774) and published the responses in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49290 through 49292). We then conducted a systematic literature review to inform development of the proposed CoPs. We also held listening sessions with industry groups, patient advocacy groups, and health care professionals to further understand the potential impacts of possible updates to the CoPs, including an obstetrical services CoP. Finally, we issued an RFI in the FY 2025 IPPS proposed rule (89 FR 35934) and included a summary of these comments in the CY 2025 OPPS/ASC proposed rule (89 FR 59186, 59490). We evaluated input from a wide range of interested parties including advocacy groups, industry associations, State health departments, labor unions, professional organizations, among others who responded to this RFI in developing our current requirements. Commenters who provided feedback in prior rulemaking were supportive of CoPs for obstetrical care services noted that establishing CoPs for obstetrical care would enhance the quality and safety of maternal care. These commenters also stated that obstetrical services CoPs regarding organization and staffing would promote multi-disciplinary, team-based care with specialists, among others providing care to pregnant women. Other commenters stated that establishing obstetrical training standards for hospitals and CAHs could improve maternal health outcomes and also supported staff training on equitable-person centered care, cultural competency, and trauma-informed care. Some commenters supported requiring facilities to report directly to the Maternal Mortality Review Committee (MMRC) and others supported specific transfer protocol requirements. Because we have engaged in extensive public outreach as part of developing our plan for improving maternal health, we do not believe that it would be appropriate to further delay these important updates. However, we continually monitor the effectiveness of the CoPs and we will continue to collaborate with interested parties and make any necessary updates in the future if new information warrants revisions or additional requirements.
Comment: Several commenters recommended monitoring the impact of these CoPs to ensure that hospitals can effectively implement the new requirements while maintaining high-quality care and essential services. A commenter recommended establishing minimum baseline standards, in accordance with State regulations, to reduce hardships small or under-resourced facilities experience in meeting these requirements.
Response: We continually monitor the effectiveness of the CoPs on advancing patient health and safety. We understand the commenters' concerns regarding small and under-resourced facilities and have opted to provide additional time for facilities to come into compliance with these requirements to reduce any potential implementation burden or unintended consequences (see section XXI.A.7 of this final rule with comment period). We acknowledge that these requirements will require a robust review of existing programs and would necessitate changes to ensure full compliance with the new requirements. We anticipate that additional time will be needed to develop revised interpretive guidance for surveyors and survey processes, conduct surveyor training on the changes, and implement the software changes in the Quality Indicator Survey (QIS) system. We believe that these updates to the CoPs will ensure that providers deliver care that is consistent with evidence-based standards, which is a vital part of providing high quality, safe care.
Comment: A few commenters recommended that CMS collaborate with affected parties, including providers and communities, in the development of subregulatory guidance for compliance with the CoPs.
Response: As discussed in the CY 2025 OPPS/ASC proposed rule, we engaged in public collaboration and outreach while developing these ( printed page 94471) policies (89 FR 59489). As with all regulations regarding the CoPs, the interpretive guidance will be updated once this final rule is published. The development of the interpretive guidance is a subregulatory process intended to clarify the regulatory requirement as described in the preamble of the final rule. However, CMS will consult with clinical subject matter experts in developing the subregulatory process and notify the public as we move forward with the guidance process.
Comment: A few commenters expressed concern that there has been insufficient time and analysis to adopt what they characterize as far-reaching CoPs. These commenters stated that there was insufficient time to properly analyze public input on the RFI published in the FY 2025 IPPS final rule. Several commenters stated that there is a lack of evidence that the specific structures and associated processes in the proposed CoPs will contribute to improved patient outcomes.
Response: As discussed previously, we engaged in extensive public outreach and information collection and worked with the public as we developed these updated requirements; This research and outreach included analyzing and evaluating the evidence base for each proposal. We also conducted a thorough analysis of the comments on the most recent RFI on maternal health to determine whether the concerns expressed by commenters offset the benefits of adopting CoPs to reduce maternal morbidity and mortality and improve care for patients. As discussed in the CY 2025 OPPS/ASC proposed rule, commenters supportive of CoPs for obstetrical care services noted that establishing CoPs for obstetrical care would enhance the quality and safety of maternal care and provide the opportunity to standardize services across various health care settings. On prior rulemaking, these commenters also stated that obstetrical services CoPs regarding organization and staffing would promote multi-disciplinary, team-based care with specialists, among others providing care to pregnant women. Other commenters stated that establishing obstetrical training standards for hospital and CAHs can help improve maternal health outcomes and also supported staff training on equitable person-centered care, cultural competency, and trauma-informed care. Some commenters supported requiring facilities to report directly to the MMRC and others supported specific transfer protocol requirements. Following that evaluation and discussed throughout this final rule, we determined that there was sufficient information to develop and support finalization of these requirements. To the extent possible, we allow hospitals and CAHs flexibility to adopt evidence-based structures and processes consistent with nationally recognized standards. We address more specific concerns related to the evidence base for individual requirements in the respective section of these comment responses. The obstetrical services CoPs are just one component of a multipronged approach to addressing the maternal health crisis and are complementary to other HHS maternal health initiatives.
Comment: Many commenters expressed concern that the burden of complying with new CoPs could potentially cause hospitals to choose to no longer offer OB services which would exacerbate disparities and increase access challenges as hospitals do not have sufficient resources to implement some best practices. Commenters cited ongoing OB staff shortages and provider burnout, particularly in rural areas, for these reasons, the commenter urged CMS to delay implementing these requirements. Additionally, a commenter stated that there would be additional burden for hospitals to meet these proposed standards even if the hospitals were already meeting the accreditation standards set by organizations such as the Joint Commission (TJC). These commenters recommended that CMS provide monetary assistance to hospitals to meet the proposed CoPs or implement other financial incentives or supports. Many commenters also recommended that CMS offer technical assistance, resource guides, and a FAQ document to help hospitals improve OB care explaining that these resources would help hospitals comply with the proposed CoPs. Conversely, other commenters recommended offering these resources as a more supportive way to improve OB care without updating the CoPs.
Some commenters stated that because these services are optional, and some of the proposed new CoPs only apply to hospitals with OB services, hospitals may choose to close these units as opposed to complying with new regulations. Other commenters stated that even for hospitals that do not close their OB units, there is the potential for resources to be drawn away from providing care to focus on complying with regulations. These commenters stated that this would worsen outcomes for patients. Some commenters stated that these closures could have a disproportionate effect on rural communities or communities of color. Some commenters recommended that CMS exempt rural hospitals and CAHs to ensure that access is not limited due to potential closures. A few commenters recommended that CMS conduct a study of OB unit closures and their impact on equity and revise the proposed CoPs based on the results of that study to ensure CoPs do not strain OB units.
Response: We thank commenters for expressing their concerns and we understand the challenges that new requirements may present for some facilities. We are implementing these requirements in response to the ongoing maternal health crisis, which has led to a U.S. maternal mortality rate that is among the highest across high-income countries and disproportionately affects racial and ethnic minorities as well as those living in rural areas and those with disabilities. Ensuring that all hospitals and CAHs participating in the Medicare and Medicaid programs meet or exceed evidence-based standards of care will lead to an overall improvement in care for all patients. We also note that we proposed these CoPs to provide flexibility to hospitals and CAHs, recognizing that hospitals and CAHs already adhere to complementary accrediting organization requirements and may also utilize guidelines from expert organizations. We believe that these requirements are balanced and would drive improved care for pregnant and post-partum women. Moreover, these proposed CoPs are only one component of an HHS-wide strategy to address the maternal health crisis. To alleviate burden, we have revised some of our requirements and will also allow for a phased-in implementation. Please refer to section XXI.A.7 of this final rule with comment period for more information on the implementation delay.
HHS has taken a comprehensive approach to addressing health care worker staffing shortages. For example, HRSA has several programs that support women's health, including the National Health Service Corps.[625] The National Health Service Corps uses both Health Professional Shortage Areas (HPSAs) and designated Maternity Care Target Area (MCTA) scoring to distribute maternity care health professionals.[626] In addition, HRSA recently announced nearly $100 million in awards to grow, support, and strengthen the health ( printed page 94472) workforce and improve access to quality care in high-need areas across the country. These awards will enhance and expand the number of nurses and primary care physicians.[627] In 2023, HHS launched the HHS Health Workforce Initiative with the goal of supporting, strengthening, and growing the health workforce by leveraging programs across the Department. The Initiative includes a coordinated Department-wide effort to identify opportunities to improve health workforce recruitment and retention, and career advancement, with equity at the center of its work.[628] We discuss organization, staffing, and delivery of services requirements further in section XXI.A.2 of this final rule with comment period.
While payment policy is not within the scope of the CoPs we agree with commenters regarding the importance of providing resources to support hospitals in improving OB care. We note that HHS generally, and CMS specifically, are engaged in a long-term, multi-agency effort to improve the quality of maternal health care across the full continuum of care. For example, some specific resources we refer readers to include the HHS resources on Strengthening Maternal Health,[629] the CMS Maternity Care Action Plan,[630] and the Medicaid Maternal and Infant Health Initiative resources.[631] Additionally as we implement these new requirements, we will continue to provide resources for hospitals, State survey agencies, and accreditation organizations, including subregulatory guidance. We note that accreditation organizations (such as the Joint Commission) [632] and expert organizations (such as ACOG) [633] have resources available to support maternal health quality improvement efforts. Further, CMS will collaborate with HHS partners to monitor the impact of OB unit closures and consider further analysis and evaluation of this issue.
Comment: Many commenters stated that the CoPs do not address the underlying issues with high quality maternity and neonatal care. Some commenters stated that many of the reasons for poor maternal outcomes are not associated with obstetrical care that patients receive in the hospital but are associated with social determinants of health (SDOH) or non-obstetric health concerns such as substance use disorder (SUD) or mental health conditions. These commenters recommended that CMS consider solutions that address the continuum of maternal health care rather than focus on inpatient OB care.
Response: We agree with commenters that improving maternal health outcomes is a complex, multifaceted issue. While the proposed updates to the CoPs for hospitals and CAHs are only one element of our multi-agency effort to improve the quality of maternal health care across the health care continuum, we believe these new standards will advance patient health and safety and compliment the activities of other agencies. There are currently no minimum care standards that hospitals and CAHs must comply with pertaining to emergency readiness, transfer protocols, and organization, staffing, and delivery of services. These requirements hold hospitals and CAHs to minimum, baseline standards to ensure facilities are held accountable for delivering care that meets or exceeds evidence-based standards of care. Examples of specific resources include HHS' resources on Strengthening Maternal Health,[634] the CMS Maternity Care Action Plan,[635] the Medicaid Maternal and Infant Health Initiative resources,[636] and the Alliance for Innovation on Maternal Health (AIM) bundles.[637] Some of these initiatives are focused on maternal mental health [638] and others involve financial support to address maternal health equity.[639] We appreciate the comments on funding resources and payment policy, though they are beyond the scope of the CoPs.
Comment: Many commenters stated that the proposed CoPs would duplicate existing regulations and guidelines including existing CoPs (specifically the QAPI requirements), State regulations, and clinical guidelines produced by expert organizations. A commenter recommended allowing States to regulate OB care.
Response: We disagree that the proposed requirements duplicate existing CoPs. While the updates have similarities to the existing CoPs, there are currently no CoPs specific to the organization, staffing, and delivery of OB services; to staff training on evidence-based best practices to improve the delivery of maternal care within a facility; emergency services readiness; or to OB transfer protocols. We also note that we proposed these CoPs to provide flexibility to hospitals and CAHs, recognizing that hospitals and CAHs already adhere to complementary State regulations and may also utilize guidelines from expert organizations. By establishing a baseline through the CoPs, we will ensure that all hospitals and CAHs meet a minimum standard of quality and safety.
We acknowledge that these CoPs may include new requirements beyond those currently adopted by the Joint Commission (TJC) and other CMS-approved accrediting organizations. In the CY 2025 OPPS/ASC proposed rule, we also stated that we are aware that accrediting organizations may also have additional requirements that exceed the Medicare CoP requirements as part of their CMS-approved deeming program (89 FR 59533), so there would not be a significant burden for hospitals that are accredited by these other organizations. In our cost estimates, we did not assume that TJC-accredited facilities would already be meeting any of the proposed requirements beyond the requirement ( printed page 94473) that if a hospital or Critical Access Hospital (CAH) provides OB services, such services must be well-organized and in accordance with nationally recognized acceptable standards of practices for physical and behavioral health of pregnant, birthing, and postpartum patients and included the costs for TJC-accredited facilities to meet the other requirements for the purposes of our impact analysis.
It is the role of CMS to ensure access to high quality, safe care for patients seeking care in facilities that participate in our programs across the nation. We note that rates of maternal mortality vary significantly across States by as much as four times as many maternal deaths per 100,000 live births as the best performing State.[640] By establishing uniform baseline requirements we aim to improve access to high quality care for all patients, regardless of State residence.
Comment: Several commenters expressed concern about the potential unintended consequences of new OB CoPs following the Supreme Court's decision in Dobbs v. Jackson Women's Health Organization (597 U.S. 215 (2022)), stating that State restrictions on abortion may potentially compound the existing maternal health crisis. For example, a commenter noted that training and educational curricula will suffer due to the limited scope of patient care being administered. In addition, a commenter expressed concern that the CoPs related to OB services, may enable inappropriate monitoring and scrutiny of hospitals' reproductive and obstetrical care standards and activities as well as additional undue oversight of OB providers and patients.
Response: As previously noted, we believe that the proposed CoPs are a critical step in addressing the maternal health crisis and part of HHS's comprehensive strategy to improve the quality of maternal health services. The CoPs require hospitals and CAHs to have appropriate protocols and adequate provisions and structures in place to meet the needs of obstetrical and emergency services patients. Hospitals have the flexibility to develop their own protocols consistent with evidence-based, nationally recognized guidelines and with the complexity and scope of services offered by the facility. The CoPs do not dictate specific standards of care or otherwise require hospitals to offer any specific type of care to patients or stock particular medications or supplies. HHS has no reason to expect inappropriate monitoring and scrutiny because of these CoPs. HHS will monitor for and investigate any concerns about inappropriate monitoring and scrutiny of hospitals' reproductive and obstetrical care standards and activities. We note that the vast majority of hospitals participate in the Medicare program through deemed status with an accrediting organization [641] and are therefore surveyed by their accrediting organization rather than a State agency, which in any case would survey for Federal, not State standards. We further note that patients' reproductive care protected health information (PHI) is protected under the recently finalized Privacy Rule (89 FR 32976).
Comment: Several commenters expressed that, in trying to maintain flexibility, the CoPs are vague which may contribute to implementation and compliance burden. A few commenters specifically noted that words including “protocol,” “practice,” “staff,” “training” and “equipment” are all subject to interpretation. A few commenters recommended that CMS define what services fall under the term “obstetric services.”
Response: As commenters noted, we have intentionally not defined some terms or requirements to allow hospitals and CAHs to meet the requirements while addressing the needs of their patient population and adhering to State regulations or other requirements. To maintain this flexibility, we will provide interpretive guidance through subregulatory channels, including updates to the State Operations Manual (SOM), with respect to these new requirements and how facilities can achieve compliance. As noted in the CY 2025 OPPS/ASC proposed rule, CMS considers a facility to “offer obstetrical services” when the facility “[holds itself] out to the public (by name, posted signs, advertising, or other means) as a place that provides care for obstetrical medical conditions.” (89 FR 59496) This is similar to how emergency departments are defined in EMTALA (42 CFR 489.24(b), definition of “dedicated emergency department”, paragraph (2)).
Comment: Several commenters recommended that CMS collaborate with AHRQ to establish a Consumer Assessment of Healthcare Providers & Systems (CAHPS) survey that specifically focuses on prenatal and childbirth care.
Response: We thank commenters for their feedback and will continue to collaborate with our HHS partners to improve maternal health care.
Comment: A commenter recommended that CMS promote alternative delivery sites, such as birthing centers, for low-risk pregnancies. Another commenter recommended that CMS ensure safety standards at these sites.
Response: In alignment with our National Quality Strategy (NQS) goals, we seek a person-centered approach to improving quality.[642] Therefore, we encourage patients to work with their clinicians to make the most appropriate choice for their individual situation, including selecting the delivery site most appropriate to their personal values and clinical needs. We understand that there are a variety of regulations that apply to alternative delivery sites, however CMS does not currently have the statutory authority under the CoPs to regulate entities such as birthing centers.
Comment: A few commenters recommended that CMS expand the CoPs to reduce the risk to patients of pregnancy criminalization. These commenters stated that by ensuring informed consent for drug testing and educating providers about treatment for pregnant patients with SUDs, the CoPs could be enhanced to increase patient trust in the health care system and increase use of prenatal care.
Response: We appreciate commenters' concern, however potential criminalization related to OB care with respect to drug testing and mandatory reporting and note that this is outside the scope of this final rule with comment period. We remind facilities that patients' reproductive care PHI is protected under the recently finalized Privacy Rule (89 FR 32976).
Comment: A few commenters recommended that CMS clarify policies related to telehealth for maternal health services, the use of which increased during the COVID-19 pandemic. These commenters stated telehealth has improved access to care without detrimental effects on care quality.
Response: Revisions to telehealth regulations are beyond the scope of these requirements, and we refer readers to HHS' Telehealth for Maternal Health Services guide [643] to support providers ( printed page 94474) in using telehealth for maternal health services. We note that for each State's Medicaid program, the State determines which services that can be provided through telehealth.[644]
2. Organization, Staffing, and Delivery of Services (§§ 482.59 and 485.649)
We proposed at new §§ 482.59 and 485.649 new CoPs for hospitals and CAHs offering obstetrical services outside of an ED. Specifically, we proposed to require that if a hospital or a CAH offers obstetrical services, the services must be well organized and provided in accordance with nationally recognized acceptable standards of practice for physical and behavioral health care of pregnant, birthing, and postpartum patients (inclusive of both mental health and substance use disorders). We proposed that, if outpatient obstetrical services are offered, the services must be consistent in quality with inpatient care in accordance with the complexity of services offered. We noted that nationally recognized acceptable standards of practice may be based on medical professional society and/or accrediting organization standards and that, while these CoPs would not require adherence to a specific organization's guideline or recommendations, we would expect that facilities be able to articulate their standards and the source(s) and demonstrate that their standards are based on evidence and nationally recognized sources. This overarching requirement for obstetric services is consistent with other hospital and CAH CoPs and is foundational to ensuring high-quality safe care.
At new §§ 482.59(a) and 485.649(a), we proposed that the organization of the obstetrical services be appropriate to the scope of services offered by the facility and integrated with other departments of the facility. At §§ 482.59(a)(1) and 485.649(a)(1), we proposed that the OB patient care units (that is, labor rooms, delivery rooms, including rooms for operative delivery, and post-partum/recovery rooms whether combined or separate) be supervised by an individual with the necessary education and training, and specified that that person be an experienced registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a doctor of medicine or osteopathy.
At §§ 482.59(a)(2) and 485.649(a)(2), we proposed that obstetrical privileges be delineated for all practitioners providing obstetrical care in accordance with the competencies of each practitioner. The obstetrical service would be required to maintain a roster of practitioners specifying the privileges of each practitioner. We noted that all hospitals are already required, at § 482.22(c)(6), to have medical staff bylaws that include criteria for determining the privileges to be granted to individual practitioners and a procedure for applying the criteria to individuals requesting privileges. Our proposed CoP would add additional specificity for an obstetrics service. The proposed obstetric services CoPs at §§ 482.59(a)(2) and 485.649(a)(2) recognize that practitioners other than physicians are important to delivering obstetric services. We again remind hospitals that existing CoPs allow for the privileging and credentialling of practitioners other than physicians, including nurse midwives (§ 482.12(a) and (c); § 482.22).
At new §§ 482.59(b) and 485.649(b), “Delivery of services”, we proposed to require that OB services must be consistent with the needs and resources of the facility and that policies governing obstetrical care be designed to assure the achievement and maintenance of high standards of medical practice and patient care and safety. We additionally proposed at §§ 482.59(b)(1) and 485.649(b)(1) that labor and delivery room suites have certain basic equipment readily available, including a call-in-system, cardiac monitor, and fetal doppler or monitor. We welcomed public comment on what is an appropriate minimum set of equipment that takes into account the varied levels of services provided for all hospitals offering obstetric services.
At §§ 482.59(b)(2) and 485.649(b)(2) we proposed that the service ensure that it has protocols, consistent with evidence-based, nationally recognized guidelines, that equipment and personnel be “readily available” (that is, necessary supplies and equipment are on the unit or are in close proximity and easily accessed by unit personnel) for obstetrical emergencies, complications, immediate post-delivery care, and other patient health and safety events as identified as part of the facility's QAPI program. While this requirement does not require any specific items, we would expect provisions to include, in addition to the equipment required under §§ 482.59(b)(1) and 485.649(b)(1), equipment, supplies, blood, and medication used in treating emergency cases. As discussed in section XXI.B.2 of this final rule with comment period, obstetric readiness is a concern in avoiding preventable maternal morbidity and mortality. Provisions and protocols, as we proposed to require, are one step towards addressing those concerns and improving perinatal outcomes.
We solicited public comments on these proposals, including whether these proposed requirements should be applicable to REHs. We address those comments, and our responses below:
Comment: Many commenters generally supported the establishment of CoPs for the organization, staffing, and delivery of obstetrical services. These commenters also supported the requirement that OB services be provided in accordance with nationally recognized standards of practice for maternal health care. A commenter supported the proposed CoPs to require obstetrical services to be organized appropriate to the scope of the services offered. The commenter stated that the proposed policy would also allow flexibility for hospitals and CAHs without creating undue burden. Other commenters also recommended that the standards cover the full continuum of maternity care, including prenatal, labor, delivery, and postpartum periods. Commenters also stated that it is necessary to integrate OB services with other departments to address chronic conditions such as cardiovascular disease, hypertension, substance use, mental health conditions, and kidney disease.
Response: We appreciate the commenters support of these proposals. We continue to believe that establishing standards for obstetrical services supports the provision of high-quality maternity care and protects the health and safety of patients. We agree that it is important for all Medicare and Medicaid participating hospitals and CAHs that offer these services to be held to a consistent set of requirements. With regard to the commenters recommendation that the standards cover the full continuum of maternity care, as previously noted in the CY 2025 OPPS/ASC proposed rule at 89 FR 59490, for the purposes of these requirements, obstetrical services include labor and delivery, prenatal and post-partum care, and care for newborn infants. This care may be provided in obstetrical units, inpatient units, emergency departments, hospital outpatient departments. We specifically require at §§ 482.59 and 485.649 that, if the hospital or CAH offers obstetrical services, and if outpatient obstetrical services are also offered, the outpatient services must be consistent in quality ( printed page 94475) with inpatient care in accordance with the complexity of services offered. In facilities that do not provide inpatient obstetric services, but rather only outpatient prenatal, maternity, and/or postpartum care, those hospital or CAH outpatient departments remain subject to all applicable existing hospital and CAH CoPs, including the hospital CoP for Outpatient Services at § 482.54. We remind facilities that the existing QAPI standards at §§ 482.21 (for hospitals) and 485.641 (for CAHs) require that the QAPI program involve “all hospital/CAH departments and services.” Finally, with regard to comments on the integration of OB services with other departments or services, we also agree that this is essential in order to provide quality, comprehensive care and ensure good communication and collaboration with other services. We included specific requirements for the integration of OB services at §§ 482.59(a) and 485.649(a).
Comment: We received several comments that did not support the establishment of CoPs related to organization, staffing, and delivery of obstetrical services. Some commenters stated that they did not believe that adopting new CoPs would be the most appropriate method to improve maternal health. A few commenters stated that the organization, staffing, and delivery of maternity services should be organized at a regional or state level, and that Federal regulations would be duplicative or conflict with existing regulations. Commenters also expressed concerns regarding enforcement actions, including potential loss of Medicare certification program if facilities are unable to comply with the proposed requirements.
Response: We thank the commenters for this input. As previously noted, we believe that the establishment of CoPs for the organization, staffing, and delivery of obstetric services is a critical step in addressing the maternal health crisis that the country continues to face. It is one part of a comprehensive strategy that HHS is undertaking to improve the quality of maternal health services. We agree that it is important for States and regions to develop standards and guidance for OB services that takes into account the unique circumstances of their respective environments, in ways that do not conflict with the national minimum requirements set forth in this rule. However, we continue to believe that establishing national minimum requirements for OB services helps to ensure that all patients receiving obstetrical care in Medicare and Medicaid participating hospitals and CAHs are receiving safe, quality care. We believe that the proposed standards are achievable, and we have provided flexibilities, including additional time to come into compliance, and clarifications in this final rule to facilitate compliance with these requirements. In addition, as noted above, if a surveyor identifies deficiencies during their survey, the initial step is for the hospital to develop a Plan of Correction (PoC) to remedy the deficiency. This activity is intended to ensure that the hospital or CAH becomes compliant with the CoPs prior to the loss of certification.
Comment: Some commenters requested that we provide clarification regarding the term “well organized” in reference to the proposed condition statement for hospitals and CAHs at §§ 482.59 and 485.649, respectively, that if the hospital or CAH offers obstetrical services, the services must be well organized and provided in accordance with nationally recognized acceptable standards of practice for the health care (including physical and behavioral health) of pregnant, birthing, postpartum patients.
Response: As stated in the CY 2025 OPPS/ASC proposed rule, the requirements that we proposed for the organization, staffing, and delivery of obstetrical services, are similar to or consistent with the current requirements for other services that hospitals and CAHs may opt to provide, such as surgical services. The interpretive guidance, published in the State Operation Manual (SOM), provides further detail regarding the expectations for “well-organized” surgical services that we believe can provides additional context for the proposed obstetrical services requirements. Similar to the description of the organization of surgical services provided in Appendix A of the SOM at A-0940,[645] obstetrical services should be organized and staffed in such a manner to ensure the health and safety of patients. Additionally, similar to the interpretive guidance provided in the SOM, acceptable standards of practice for obstetrical services would also include maintaining compliance with applicable Federal and State laws, regulations and guidelines governing obstetrical services or obstetrical service locations, as well as any standards and recommendations promoted by or established by nationally recognized professional organizations. We maintain that this overarching requirement for obstetrical services in hospitals and CAHs is foundational to ensuring high-quality, safe care. We also believe that providers need the flexibility to determine how to organize the obstetrical services in their facility in a way that best aligns with the services provided, characteristics of their facility, and needs of their patient population and therefore we do not require adherence to a specific set of standards. Lastly, we also expect to provide interpretive guidance specific to obstetrical services in the SOM in the future which would further detail how providers can assure compliance with these regulations.
Comment: A commenter stated that the proposed requirement that outpatient obstetrical services offered be consistent in quality with inpatient obstetrical care in accordance with the complexity of services offered would be difficult for some organizations to achieve because they believe that the proposal is ambiguous. The commenter stated that treatment could be very different between patients. The commenter provided an example, stating that gestational diabetes is not the same as type two diabetes, and treatment of hyper- and hypo-glycemic episode in a pregnant patient versus and 85-year-old male patient generally, cannot be treated similarly and may have different outcomes.
Response: The proposed requirement is specific to the provision of obstetrical services. If a hospital or CAH elects to provide inpatient obstetrical services, we will require that any obstetrical services provided in the outpatient setting must be consistent in quality with any obstetrical services that the hospital or CAH provides to hospital inpatients. While we understand that there may be some variation in the method or duration of treatment provided when an individual receives outpatient treatment versus inpatient treatment, we would expect that the quality of the services provided in either setting would be equivalent.
Comment: With regard to standards of practice for OB staffing, a few commenters recommended that CMS structure the obstetrical service requirements to ensure adequate staffing levels. These commenters suggested resources for establishing recommended staffing levels (specifically the resources developed by the Association of Women's Health, Obstetric and Neonatal Nurses or the ANA). A commenter recommended that staffing models include community health workers and care coordinators.
A commenter recommended that CMS ensure that the requirements do not ( printed page 94476) become overly burdensome, citing physician shortages and burnout as particular challenges. A few commenters urged CMS to study and understand barriers to implementing existing staffing standards prior to finalizing the proposal. A commenter recommended that CMS not refer to the ACOG 2019 Levels of Maternal Care when developing subregulatory guidance, maintaining that the requirement regarding anesthesiologists as opposed to CRNAs will cause negative effects and that CRNAs are proven to be the most cost-effective model for anesthesia delivery. A few commenters expressed concern with the lack of specificity regarding the appropriate guidelines to follow to meet the proposed CoPs, stating that some guidelines and standards will be overly burdensome and mandatory staffing ratios could cause undue hardship.
Response: We thank the commenters for their input. We note that we did not propose any specific standards for staffing levels for OB units in hospitals or CAHs. Both hospitals (at § 482.23(b)) and CAHs (at § 485.635(d)) are already required to provide nursing care to meet patient needs. We strive to balance the need for hospitals and CAHs to tailor the organization, staffing and policies and equipment for delivery of service of their obstetrical service to the unique populations, capabilities, and constraints of that hospital or CAH, while still meeting the overarching health and safety standards established by the CoPs. We agree that the standards mentioned by the commenters are the types of standards that we would expect hospitals and CAHs to reference in meeting these new CoPs. However, as we have done in other CoPs, CMS is not mandating a specific set of standards. We reference the recommended standards of practice for OB services, including OB staffing, in the CY 2025 OPPS/ASC proposed rule as examples (89 FR 59490).
Comment: Another commenter stated that the proposed requirement that obstetrical services be integrated with other departments would be challenging because effective coordination with other departments is not fully within the control of the OB department and that communication challenges often occur when pregnant or postpartum patients are admitted in another unit or department. The commenter recommended that CMS provide best practices and resources to help hospitals improve internal communications across departments.
Response: We thank the commenters for their input. We continue to believe that, while potentially challenging, in order to provide high quality and safe care, it is essential that the OB unit be integrated with other departments in the hospital as applicable. The integration with other departments must be such that the facility can make available the full extent of its patient care resources, such as laboratory, surgery, or anesthesia, to assess and render appropriate care for a patient receiving obstetrical services, particularly in emergency situations. We note that such integration is already specifically required of some of the services that an OB unit would be expected to coordinate with. For example, it is required for hospital outpatient services at § 482.54(a) and emergency services at § 482.55(a)(2). With respect to best practices, in the CY 2025 OPPS/ASC proposed rule, we provide references to the recommended standards for practice for OB services developed by several accrediting bodies and professional medical specialty societies, such as ACOG and Society of Maternal Fetal Medicine (SMFM), which also includes discussions of strategies related to collaboration and communication within the facility as well as with external facilities in order to best treat the patient.[646] We also refer the reader to our HHS resources on Strengthening Maternal Health,[647] the CMS Maternity Care Action Plan,[648] the Medicaid Maternal and Infant Health Initiative resources,[649] and HRSA's Maternal & Child Health Bureau resources.[650]
Comment: A few commenters supported the proposed requirements at §§ 482.59(a)(1) and 485.649(a)(1) for labor and delivery rooms/suites (including labor rooms, delivery rooms (including rooms for operative delivery), and post-partum/recovery rooms whether combined or separate) to be supervised by an experienced registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a doctor of medicine or osteopathy. These commenters appreciated that we included a broader range of providers to supervise OB units, noting that these providers play an integral role in providing high quality reproductive health care and improving outcomes. A commenter supported inclusion of other caregivers, such as certified nurse midwives, direct entry midwives, Tribal midwives, and doulas in the staffing of labor and delivery units as allowed by State law. Some commenters stated that expanding the role of midwives and doulas could reduce disparities in maternal health outcomes. Another commenter stated that only physicians should supervise OB units.
Other commenters did not support the proposal and stated that CMS underestimated the difficulties involved with recruiting, training, and retaining qualified clinicians to supervise OB units at the proposed levels, particularly for rural hospitals and CAHs, adding that these costs are likely to disproportionately impact facilities most at risk for eliminating labor and delivery services. These commenters stated that the difficulty in retaining staff could result in the closure of OB units and the loss of OB services.
Other commenters suggested that CMS provide an exemption for community hospitals, CAHs, and other small hospitals to the 24/7 supervision requirement and that CMS allow supervision of maternity departments or units as opposed to rooms and suites. A commenter stated that the proposed requirement for having an OB/GYN physically present at all times would reduce their facility's designation from an OB Level III Unit to an OB Level II Unit which could increase the risk of having to transfer high-risk patients.
Response: We appreciate the comments provided. We understand the staffing challenges experienced by a number of facilities, particularly with regard to recruitment and retention in rural and other underserved areas. We note that the proposed requirement does not alter any of the current requirements related to supervision of services in hospitals and CAHs. We also expect that many hospitals and CAHs that currently opt to provide obstetrical services and would therefore be required to comply with these obstetrical services requirements. Additionally, they would have to have practitioners on staff that would perform supervision duties for the OB unit such as overseeing staff, training, overall patient care, and supporting communications within the unit and across the facility. This requirement was intended to recognize that practitioners other than physicians are important to delivering obstetric services and that we considered them when developing these provisions. Also, to clarify, we did not propose a requirement for an OB/GYN or any other practitioner to be physically present at all times, that is, 24/7, in a hospital or CAH to provide supervision of obstetrical services. The current CAH ( printed page 94477) CoPs at § 485.618(e), however, require CAHs to establish procedures, in coordination with emergency response systems in the area, under which a doctor of medicine or osteopathy is immediately available by telephone or radio contact on a 24-hours a day basis to receive emergency calls, provide information on treatment of emergency patients, and refer patients to the CAH or other appropriate locations for treatment.
We reiterate that if not otherwise prohibited by State law, a hospital may elect to include advanced practice providers, such as advanced practice registered nurses, clinical nurse specialists, physician assistants, and nurse midwives) as part of their medical staff. Moreover, the hospital CoPs prohibit a hospital from granting staff membership or professional privileges in the hospital solely upon certification, fellowship, or membership in a specialty body or society (§ 482.12(a)(7)). In States that permit nurse midwives to admit patients (in accordance with hospital policy and practitioner privileges), section 1861(e)(4) of the Act requires only Medicare hospital patients of a nurse midwife to be under the care of an MD or DO (§ 482.12(c)(2)). The statute does not require Medicaid nor other non-Medicare patients admitted by a nurse midwife to be under the care of an MD or DO.376 For CAHs, CMS does not have the authority to remove the physician oversight requirement for inpatients at § 485.631(b)(1)(iv), as this is a statutory requirement and the physician oversight requirement for outpatients at § 485.631(b)(1)(v) is only applicable if required by State law.
With regard to including doulas in the staffing of an obstetrics unit, evidence demonstrates that doulas can improve outcomes, including reducing c-section frequency and improving the patient experience for mothers.[651] Nothing in these CoPs would restrict a hospital from utilizing doulas to support its obstetric patients, consistent with State law and acceptable standards of practice.
Comment: A few commenters requested clarification regarding the terms included in the proposed requirement. Specifically, commenters questioned what the term “supervision” means, including what minimum experience, education, or training is required for practitioners to supervise OB units. A few commenters stated that the term “experienced” clinician is not clearly defined and could create an arbitrary standard that small rural hospitals and CAHs may be unable to meet. One commenter recommended CMS provide more clarity, especially for smaller hospitals where staff often work in multiple specialties. Another commenter asked CMS to further define what is meant by “OB patient care unit supervisor,” noting that this role could include personnel from a shift-by-shift informal role to a formal unit supervisor or director role.
Response: We appreciate the commenters' input. We recognize that meeting this requirement may vary somewhat from facility to facility, based on the scope and complexity of the services offered. Similar to what is expected for surgical services, we would expect an OB unit to be supervised by an experienced registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a doctor of medicine or osteopathy. The individual supervising the obstetrical unit must demonstrate appropriate education, background working in obstetrical services, and specialized training in the provision of obstetrical services/management of obstetrical service operations. As noted above, we understand that this may vary somewhat based on the complexity and scope of the services offered by the facility. Thus, the hospital should address its required qualifications for the supervisor of the hospital's obstetrical unit in its policies and the supervisor's personnel file should contain information demonstrating compliance with the hospital's established qualifications.
Comment: Some commenters specifically opposed the proposed requirement at §§ 482.59(a)(2) and 485.649(a)(2) that obstetrical privileges be delineated for all practitioners providing obstetrical care in accordance with the competencies of each practitioner. Commenters also opposed the requirement for the obstetrical service to maintain a roster of practitioners and their specific obstetrical privileges, noting that a statement of the duties and privileges of medical staff are currently required in the CoPs regarding medical staff bylaws.
Response: The intent of the proposed regulation was to ensure that practitioners have the necessary education, training, and experience to provide safe, effective obstetrical care and appropriately perform specific procedures. We believed that the proposed CoP would provide additional specificity for obstetric service regarding the privileges of each practitioner on the service. While we anticipated that the proposed requirement would result in minimal additional burden, we appreciate the comments regarding the redundancy of this requirement with the medical staff bylaws at § 482.22(c) that require the inclusion of a statement of the duties and privileges of each category of medical staff as well as the inclusion of criteria for determining the privileges to be granted to individual practitioners and a procedure for applying the criteria to individuals requesting privileges. We agree that the information currently required in the medical staff bylaws meets the intended purpose of the proposed regulations. Therefore, we are finalizing the proposed CoP with a modification that references the requirements of the medical staff bylaws at § 482.22(c) for hospitals and the requirements for agreements for credentialing and quality assurance at § 485.616 for CAHs as opposed to finalizing a new requirement for the delineation of obstetrical privileges for all practitioners providing obstetrical care and the maintenance of a roster of practitioners and their specific privileges.
Comment: Some commenters supported the proposals at §§ 482.59(b)(1) and 485.649(b)(1) to require labor and delivery rooms to have certain basic equipment available to the room, including a call-in-system, cardiac monitor, and fetal doppler or monitor and recognized it as an effort to standardize equipment and supplies across facilities providing obstetrical care. One commenter specifically supported the requirements published in the Guidelines for Perinatal Care, 8th Edition, a joint publication by the American Academy of Pediatrics (AAP) and the American College of Obstetricians and Gynecologists (ACOG), in regards to equipment, in addition to the capabilities and equipment recommendations defined in the Levels of Maternal Care Consensus statement first published in 2015 by the American College of Obstetricians and Gynecologists (ACOG) and the Society for Maternal-Fetal Medicine (SMFM).
However, other commenters stated concerns about the proposal. Specifically, commenters requested clarity regarding what it means to have this equipment “available” and the evidence for proposing to require this equipment. These commenters noted concerns regarding the financial burden of providing the proposed equipment in every room, when it may not be necessary for the patient's treatment. They also stated that smaller or rural ( printed page 94478) hospitals and CAHs with lower patient volume may have this equipment available to the unit, but not in every labor and delivery room, and suggested that CMS instead allow facilities to have the flexibility to provide equipment in accordance with the scope, volume and complexity of services they offer. Commenters were particularly concerned with the requirement to have a cardiac monitor in each room, stating that cardiac monitors are not typically clinically necessary in all labor and delivery settings and that this requirement would impose significant costs for purchasing the devices and for additional staff training, as not all staff are trained in their use. A commenter also asked for clarification as to whether a call-in system described a call button or a telemetry system, stating that a telemetry system in each room would be overly burdensome.
Lastly, other commenters recommended expanding the proposed requirement to include specific additional emergency equipment such as adult and newborn oxygen supplies, infant resuscitation equipment, crash carts, hemorrhage carts, limited obstetric ultrasonography, and equipment that facilitates quantification of blood loss.
Response: We appreciate the comments regarding the proposed requirement for the availability of equipment for the care of obstetrical patients. We understand commenters' concerns regarding financial investment and potential burden associated with providing the proposed equipment to each labor and delivery room or suite, if the facility does not currently have it available. We also understand that certain facilities, particularly smaller facilities or those in underserved areas, including rural areas, may not have the volume of obstetric patients or provide the level or scope of obstetrical care to necessitate the availability of this equipment in every labor and delivery room in order to safely and effectively care for their patients. We would, however, expect that facilities that treat a large volume or high acuity obstetrical patients would have the aforementioned equipment available in every labor and delivery room to meet the needs of their patient population.
Based on these comments and our review of the proposed requirement, we believe that, as a minimum standard, facilities offering OB services must have basic equipment for the care of OB patients readily available to meet the patients' needs. Therefore, we are finalizing §§ 482.59(b)(1) and 485.649(b)(1) with a modification to clarify that basic equipment for treating OB patients (including a call-in-system, cardiac monitor, and fetal doppler/monitor) must be kept at the facility and be readily available to meet the needs of OB patients in accordance with the scope, volume, and complexity of services offered by the facility. This means a large-volume high-acuity OB unit may have this equipment in every L&D room, while a rural hospital with a low-volume of births may have this equipment readily available within the hospital. Of note, by “call-in system”, we mean a mechanism by which a patient and/or caregiver can alert staff of any emergencies or concerns. Examples may include a call-bell, alarm, or other notification device.
Additionally, at §§ 482.59(b)(2) and 485.649(b)(2), we will finalize the requirement that there must be adequate provisions and protocols, consistent with nationally recognized and evidence-based guidelines, for obstetrical emergencies, complications, immediate post-delivery care. Provisions include equipment (in addition to the equipment required under §§ 482.59(b)(2) and 485.649(b)(1)), supplies, and medication used in treating emergency cases. Such provisions must be kept in the hospital or CAH and be readily available for treating emergency cases. While this requirement does not require specific items, we would expect provisions to include equipment, supplies, blood, and medication used in treating emergency cases. In the proposed rule preamble, we provided examples of such supplies including: resuscitator, defibrillator, aspirator, and airways, endotracheal tubes, ambu bag/valve/mask, oxygen, tourniquets, nasogastric tubes, IV therapy supplies, suction machine, and defibrillator. Example emergency medications could include analgesics, local anesthetics, anti-arrhythmic, cardiac glycosides, antihypertensives, antiepileptics, uterotonics, anticoagulants, antifibrinolytics, electrolytes and replacement solutions. We note that hospitals and CAHs may maintain these and other obstetrical emergency supplies in “crash carts”, “obstetrical emergency carts/bags/boxes/kits”,[652] “OB hemorrhage carts”,[653] or other readily accessible method for use when and where needed.
In response to commenters asking for additional specific emergency equipment, we believe the above finalized emergency supply requirement is comprehensive while allowing facilities flexibility to provide care in line with their scope, volume, and complexity of services. CMS is not prohibiting hospitals from stocking commenters' suggested supplies, but likewise is not mandating specific items given facilities' diverse practice settings and scope of services. Rather CMS will require facilities to stock provisions necessary for the care their obstetrical and neonate patient populations consistent with (1) nationally recognized and evidence-based guidelines as well as (2) the facilities' scope, volume, and complexity of services. This approach is consistent with the new requirement for emergency services readiness (§ 482.55; § 485.618) and existing CAH (§ 485.618(b) and (c)) and REH (§ 485.516(c)(2)) emergency supply standards.
Comment: A commenter requested clarification regarding whether delivery rooms in hospitals and CAHs, specifically rooms for operative delivery, would be required to meet the obstetrical services CoP or continue to be required to meet the surgical services CoPs. The commenter noted that their facility already complies with the surgical services CoPs at § 482.51 for their operative delivery rooms. We note there are also requirements for surgical services provided at CAHs at § 485.639.
Response: In the CY 2025 OPPS/ASC proposed rule, we included proposals for requirements for operative delivery rooms. Specifically, at §§ 482.59(a)(1) and 485.649(a)(1), we proposed to require OB patient care units, described as labor rooms, delivery rooms, including rooms for operative delivery, and post-partum/recovery rooms whether combined or separate, to meet specific requirements regarding supervision. At §§ 482.59(b)(1) and 485.649(b)(1) we proposed that hospitals and CAHs must have specific equipment available to the labor and delivery room suite. The finalized policy requires this equipment to be kept at the hospital and be readily available for treating obstetrical cases to meet the needs of patients. As noted in the interpretive guidance provided in the SOM (A-0940), if a hospital provides any degree of surgical services to its patients, the hospital must comply with all the requirements of the surgical services CoP. Therefore, we expect hospitals and CAHs to continue to comply with the surgical services CoP. Additionally, we expect hospitals and CAHs to meet any requirements specific to obstetrical services that are not delineated in the surgical services CoP. For example, we would expect that ( printed page 94479) hospitals and CAHs providing obstetrical services would ensure that the OB patient care unit, including operative delivery rooms, have the basic equipment relevant to maternal care available in the unit.
Comment: There were supportive comments for the proposed requirement that the obstetric service have protocols and provisions to address obstetrical emergencies, complications, immediate post-delivery care, and other patient health and safety events as identified as part of the facility's QAPI program. One commenter recommended expanding the current list of equipment used in life-saving procedures to include an incubator and another commenter noting that equipment should be available for prenatal as well as neonatal emergencies. A few commenters recommended including protocols for transport as an important part of addressing emergencies, including transfer to a hospital with sufficient blood supply to address significant maternal hemorrhage and the capability to transport neonatal patients.
One commenter did not support the proposal stating that multiple protocols would be appropriate for complex patients, and it would be challenging to identify the appropriate protocol.
Response: We appreciate these recommendations. We reiterate that the proposed requirements at §§ 482.59(b)(2) and 485.649(b)(2) do not require hospitals and CAHs to have any specific items on the unit or in close proximity. The list of emergency equipment and supplies provided in the CY 2025 OPPS/ASC proposed rule (89 FR 59492) included examples of provisions that we would expect facilities to have available in order to quickly and effectively address obstetrical emergencies and other issues. Therefore, at this time, we do not believe that it is necessary to include any additional supplies or medications in this list. Additionally, while we acknowledge that some facilities may experience challenges in establishing protocols to address these issues that anticipate the complexities involved in this care, we believe that these protocols are an important step towards avoiding preventable maternal morbidity and mortality and improving maternal health outcomes. We believe this is an achievable minimum standard. We note that we discuss the policies regarding transfer protocols in section A.XXI.6 of this final rule with comment period.
Comment: In response to our comment solicitation regarding whether the proposed requirements for the organization, staffing, and delivery OB services should be applicable to REHs, we received a variety of comments. A commenter encouraged CMS to apply the organization, delivery, and training requirements to REHs. A few commenters stated that an REH should be staffed by an emergency medicine physician with experience and continuing education requirements in obstetric medicine, and a commenter suggested that emergency physicians who are competent in OB care should be credentialed to perform deliveries.
Response: With respect to applying these proposed requirements to REHs, we appreciate commenters insights and recommendations, as noted above. We will take these comments into consideration for future potential rulemaking.
Final Rule Action: After consideration of the public comments we received, we are finalizing these provisions as discussed below.
For hospitals, we are finalizing the following CoPs as proposed:
Section 482.59 (Condition of participation: Obstetrical services):
- Section 482.59(a) (Standard: Organization and staffing);
- Section 482.59(a)(1);
- Section 482.59(b) (Standard: Delivery of service); and
- Section 482.59(b)(2).
We are finalizing the following CoPs with modifications:
- Section 482.59(a)(2) with a modification to reference the requirements of the medical staff bylaws at § 482.22(c) for hospitals.
- Section 482.59(b)(1) with a modification to clarify that basic equipment, including a call-in-system (we note that for the purposes of this regulation, call-in system describes a call button), cardiac monitor, and fetal doppler or monitor must be kept at the hospital and be readily available for treating obstetrical cases to meet the needs of patients.
For Critical Access Hospitals, we are also finalizing the following CoPs as proposed:
Section 485.649 (Condition of participation: Obstetrical Services):
- Section 485.649(a) (Standard: Organization and staffing);
- Section 485.649(a)(1);
- Section 485.649(b) (Standard: Delivery of service); and
- Section 485.649(b)(2)
We are finalizing the following CoPs with modifications:
- Section 485.649(a)(2) with a modification to reference the requirements for agreements for credentialing and quality assurance at § 485.616 for CAHs.
- Section 485.649(b)(1) with a modification to clarify that basic equipment, including a call-in-system (we note that for the purposes of this regulation, call-in system describes a call button), cardiac monitor, and fetal doppler or monitor must be kept at the hospital and be readily available for treating obstetrical cases to meet the needs of patients.
3. Training for Obstetrical Staff in Hospitals and CAHs (§§ 482.59(c), 485.649(c))
We proposed at new §§ 482.59(c) and 485.649(c) that hospitals and CAHs with OB services would be required to develop policies and procedures that would ensure that relevant obstetrical services staff would be trained on select topics that reflect the scope and complexity of services offered, including but not limited to facility-identified evidence-based best practices and protocols to improve the delivery of maternal care within the facility. We also proposed that any additions, revisions, or updates to topics be informed by the hospital and CAH's QAPI program findings. Additionally, we proposed that governing body to identify and document which staff must complete annual training and that the hospital and CAH must document in the staff personnel records that the training was successfully completed. Further, we proposed that the hospital and CAH be able to demonstrate staff knowledge on the topics identified at §§ 482.59(c)(1) and 485.649(c)(1), respectively. We solicited public comment on these proposals, including whether these proposed staff training requirements should be applicable to REHs. We also solicited public comment on whether CMS should require specific training on person-centered care, trauma-informed care, cultural competency, and/or other topics as part of the evidence-based training.
In the following section we discuss the public comments received and our responses on proposed §§ 482.59(c) and 485.649(c), which would require hospitals and CAHs to train OB staff on key maternal health topics.
Comment: Several commenters supported requiring evidence-based trainings for staff at hospitals with OB services, noting that if properly implemented the proposal will likely lead to significant improvements in health outcomes by ensuring staff are well-trained and that a consistent minimum standard of care is delivered nationwide.
Many commenters also supported the requirement to train OB staff on select topics aimed at improving the delivery of maternal care and recommended several specific topics that OB staff ( printed page 94480) should be trained on. Several commenters supported requirements for training related to cultural competency, highlighting the negative health outcomes and health inequities that result from a lack of culturally competent care. For example, commenters shared that the lack of cultural competency among non-Tribal providers has contributed to poor quality care for pregnant and birthing Native women. Another commenter underscored the importance of combatting medical misinformation as it contributes to health inequities. For example, the commenters noted that it is still a common belief among medical professional students that Black patients have a higher pain tolerance than white patients. Because of this, Black patients often do not receive proper main management.
A few commenters also supported trauma-informed care (TIC) as a training topic, explaining that TIC training prepares staff to support patients who have experienced trauma. A commenter also supported inclusion of person-centered care training topics because these care models are associated with increased patient satisfaction and trust. Some commenters also recommended training on substance use disorders and related topics including neonatal abstinence, perinatal mood disorders, and managing substance use disorder within pregnant patients. Various other topics included health disparities, care for people with disabilities, and pregnancy criminalization. A few commenters also recommended clinical topics including severe hypertension/preeclampsia, hemorrhage, fetal monitoring, shoulder dystocia, and neonatal resuscitation.
Conversely, a few commenters recommended that CMS not specify training topics within the CoPs, explaining that because hospitals serve diverse patient populations, it is more appropriate for hospitals to determine the most relevant training topics.
Response: We thank commenters for their support and highlighting the improved health care outcomes staff training will lead to as well as providing recommendations on additional topics to improve the delivery of maternal care. These requirements will support clinicians in performing vital tasks consistently through evidence-based standardized practices, which can ultimately reduce errors.[654]
While CMS encourages facilities to train staff on the aforementioned topics, we also recognize that hospitals and CAHs serve diverse populations with unique needs. Therefore, we are providing facilities with the flexibility to select which evidence-based best practices and protocols to train on that reflect the scope and complexity of the services offered within their facility and the needs of their specific patient population. Based on the findings from a facility's QAPI program, a facility may determine that some of the aforementioned topics are important for staff to be trained on in order to improve patient outcomes or address disparities. For example, a CAH's QAPI program may reveal that a significant portion of their obstetrical patients have diagnosed substance use disorders (SUD). Subsequently, this CAH may decide to pursue training for their OB staff on behavioral health and/or medications for opioid use disorder. Another hospital's QAPI findings may reveal that a large percentage of their patient population does not speak English as their first language. To address this, the hospital may pursue training relevant staff on the culturally and linguistically appropriate care and/or person-centered care. While not required, facilities that wish to learn more about culturally competent care and person-centered care may wish to review the following resources among others:
- Think Cultural Health. Health and Human Services. Office of Minority Health, Department of Health and Human Services.https://thinkculturalhealth.hhs.gov/clas.
- Addressing Health Literacy. Health Resources and Services Administration.https://www.hrsa.gov/about/organization/bureaus/ohe/health-literacy/culture-language-and-health-literacy.
- Behavioral Health Implementation Guide for the National Standards for Culturally and Linguistically Appropriate Services in Health and Health. Office of Minority Health, Department of Health and Human Services.https://thinkculturalhealth.hhs.gov/assets/pdfs/resource-library/clas-standards.pdf.
- Fundamentals of the National Standards for Culturally and Linguistically Appropriate Services in Maternal Healthcare, Office of Minority Health, Department of Health and Human Services.https://thinkculturalhealth.hhs.gov/resources/presentations/1/fundamentals-of-the-national-standards-for-culturally-and-linguistical.
- Culturally and Linguistically Appropriate Services in Maternal Health Care, Department of Health and Human Services.https://thinkculturalhealth.hhs.gov/education/maternal-health-care.
Comment: A few commenters stated that requiring the governing body to identify and document which staff must complete annual training is inconsistent with governing body activities, which typically focus on oversight. Another commenter stated that hospitals' governing bodies may lack the medical expertise to identify which staff must complete annual training. These commenters noted that regulation implementation details are generally determined by service line leadership, and therefore, medical leadership should guide training decisions to ensure effective and relevant instruction.
Response: The hospital governing body performs a broad array of functions and is responsible for a variety of tasks, including but not limited to planning and budgeting, care of patients, managing contracted services, and participating in the QAPI program. Section 482.12(a)(10) requires the hospital governing body to consult directly with the individual assigned the responsibility for the organization and conduct of the hospital's medical staff. In addition, governing bodies have experience appointing individuals for certain roles and also have the ability to grant clinical privileges. For example, § 485.639 states, “If a CAH provides surgical services, surgical procedures must be performed in a safe manner by qualified practitioners who have been granted clinical privileges by the governing body.” The governing body is also responsible for appointing an individual or individuals to serve as the infection preventionist/infection control professional (§§ 482.42(a)(1), 485.640(a)(1)). Thus, we believe that determining which relevant staff must be trained on maternal health topics is not outside the scope of their capabilities and standard activities. Further, we note that the governing body has the ability to delegate tasks and responsibilities, including to medical staff, but maintains overall responsibility for compliance.
Comment: Some commenters requested CMS provide additional guidance regarding which staff will be required to complete OB training and provided further recommendations for staff that may require additional training. For example, a commenter requested clarification on training for staff without specific maternity credentials and privileges for midwives who do not have formal educational training, while another commenter ( printed page 94481) recommended that CMS develop guidance for the training of Nurse Practitioners, Physician Assistants, and direct entry midwives. Some commenters also suggested implementing standards that are developed for non-obstetrical staff, as appropriate, as they may still have interactions with OB patients. The commenter noted that these staff should have basic knowledge on the leading causes of maternal deaths ( e.g., hemorrhage, hypertension, and mood disorders).
Response: As outlined in the introductory paragraph of §§ 482.59 and 485.649, “Condition of participation: Obstetrical services,” if a hospital or CAH offers OB services, the services must be well organized and provided in accordance with nationally recognized acceptable standards of practice for the health care of pregnant, birthing, and postpartum patients. At §§ 482.59(c)(3) and 485.649(c)(3), we are requiring the governing body of hospitals and CAHs to identify and document which relevant staff must complete the training. While we are providing hospitals and CAHs with the flexibility to determine training topics based on the scope and complexity of their services and also with the flexibility to determine the format to administer the training, we expect facilities to provide high-quality training, consistent with, and tailored to the staff's expected role, with the goal of improving the delivery of obstetrical care.
We recognize that staff outside of OB units may also interact with OB patients. Thus, we are also requiring staff to be trained on transfer protocols (§ 482.43(c)) and emergency services readiness (§§ 482.55(c)(3), 485.618(e)(2)). We refer readers to sections A.XXI.5 and A.XXI.6 of this final rule with comment period for additional details on training on emergency services readiness transfer protocol training, respectively. This requirement does not preclude hospitals or CAHs from training additional staff on maternal health topics.
Comment: Some commenters supported that findings from QAPI programs should be used to inform training needs and updates on an ongoing basis. A commenter requested CMS more clearly define “ongoing basis” and instead suggested we incorporate different terminology, such as “annually.”
Response: Stratified data can produce meaningful measures that can be used to expose health disparities, develop interventions to reduce them, and monitor performance to ensure interventions aimed at improving care do not have unintended consequences for certain patients and improve patient outcomes.[655] Continuous quality improvement depends on a disciplined and well-defined data-driven process that constantly is monitored and improved.[656] Facilities must review findings from their QAPI programs on an “ongoing basis” to determine any additions, revisions, or updates to training concepts. That is, facilities should be able to verify that data analyses and collection are occurring over time and are not just one-time events. Hospitals and CAHs should perform follow-up analyses on its remedial actions to determine if its actions were effective in improving performance and quality. We also expect hospitals and CAHs to continuously study and improve their processes and service delivery and take a proactive approach to improve their performance (QSO-23-09-Hospital).[657] We refer readers to section A.XXI.3 of this final rule with comment period for further details on the updated QAPI requirements.
Comment: Some commenters provided recommendations regarding the modality in which staff be trained on pertinent maternal health topics. For example, a commenter recommended that training should be completed in an active learning style which involves staff collaboration. Another commenter recommended against requiring simulation training. Lastly, a few commenters recommended that staff be trained upon hire or complete an orientation program.
Response: While we acknowledge the benefits of training in a collaborative environment that encourages increased staff engagement, we are not requiring the training to be completed in a specific manner or format. These requirements permit facilities with the flexibility to determine the manner in which staff are trained on evidence-based best practices and protocols to advance the delivery of maternal care and other training topics as identified by the facilities' QAPI program. This flexibility allows facilities to test different approaches to conducting training over time, which could be informed by post-training evaluations or monitoring of outcomes.
High-quality orientation of trainees entering a new health care setting is imperative to support education and patient safety.[658] Therefore, we are revising the staff training requirements and will require staff to complete training when hired. Specifically, at new §§ 482.59(c)(2) and 485.649(c)(2) we are requiring hospitals and CAHs to provide relevant new staff with initial training. We are also requiring the governing body to identify and document what staff must complete the initial training and subsequent biannual training on evidence-based best practices at §§ 482.59(c)(3) and 485.649(c)(3). Research has demonstrated that a practical and well-structured nursing orientation program can have a positive impact on new nurses who are joining health care organizations, including improved competence.[659 660] Another study demonstrated that effective new employee orientation reduces role ambiguity and role conflict. Role ambiguity is negatively correlated to task performance and role conflict reflects job attitudes.[661] The Joint Commission describes an orientation program, “as an introductory program and/or activities intended to guide a person in adjusting to new surroundings, employment, policies/procedures, essential job functions, etc.[662] The hospital or CAH will be responsible for determining when and how long a person is considered to be in orientation.
Comment: Several commenters stated that financial, staffing, and infrastructure limitations would make training requirements burdensome for facilities. Commenters recommended modifying the proposed provisions to accommodate the specific needs of each facility and provide resources, technical assistance, and financial support/funding, particularly for hospitals in rural and/or underserved areas, to help facilities effectively implement the provisions. To ensure high-quality training, some commenters recommended specific resources that facilities may use as a guide to implement these requirements. Some examples include the HHS training “Culturally and Linguistically Appropriate Services (CLAS) in ( printed page 94482) Maternal Health Care [663] and ACOG's “Guidelines for Perinatal Care, 8th Edition.” [664]
Response: We understand the commenter's concerns about the potential burden that these training requirements could place on facilities. We therefore are revising the staff training requirements and reduced the frequency to every two years to ease any financial strain this requirement may place on facilities. While facilities will have upfront costs to implement these requirements, the benefits for patient outcomes outweighs the burden. Additionally, hospitals and CAHs have the flexibility to identify which evidence-based best practices and protocols they will train on to improve the delivery of maternal care within their facility. We are not prescribing a specific format the training be conducted in, allowing facilities to carry out the training in a manner that best fits their needs. In addition, we are also allowing for a staggered implementation. Further details on the implementation timeframe can be found in section A.XXI.7 of this final rule with comment period. During this time period, facilities will be able to gather resources and support to be prepared for the effective date.
To further mitigate burden concerns, we refer facilities to several resources facilities that may be utilized to achieve compliance with these requirements (although others may be used). For example, facilities will have support from Quality Improvement Organizations (QIOs), which serve people with Medicare, health care providers, and communities. The QIO Program data-driven initiatives bring stakeholders together at the local, community, and tribal levels to improve care coordination and the quality-of-care transitions and increase patient safety.[665] Perinatal quality collaboratives (PQCs) are State or multistate networks of teams that work to identify health care processes in need of improvement in order to advance the quality of care for mothers and babies.[666] Additionally, the Health Resource and Services Administration (HRSA) funds the Alliance for Innovation on Maternal Health (AIM) to establish patient safety bundles.[667 668] Perinatal quality collaboratives often support the implementation of maternal safety bundles, which have documented success in driving improvements, particularly with regards to obstetric hemorrhage, severe hypertension in pregnancy, and non-medically indicated Cesarean deliveries.[669 670 671 672]
As commenters outlined, facilities may use the credentialing published in the “Guidelines for Perinatal Care, 8th Edition” [673] or the HHS “Culturally and Linguistically Appropriate Services (CLAS) in Maternal Health Care” [674] training as a resource. Of note, these CoPs do not require adherence to a specific organization's guidelines or recommendations. We expect that facilities will be able to articulate their standards and the source(s) and to demonstrate that their staff training requirements are based on evidence-based best practices. As previously discussed in sections XXIA.1 and XXI.A.2 of this final rule with comment period, HHS and HRSA have established several initiatives to address the health care workforce shortage; CMS is not mandating a specific staffing standard.
Comment: Multiple commenters discussed existing staff training requirements that may be required through either accreditation standards and/or continuing education requirements. Some commenters recommended that CMS maintain flexibility in the training requirements to accommodate existing training requirements from national accreditation and credentialing bodies and to align with best practices from accreditation agencies (for example, TJC) which require a biannual training cycle. Other commenters questioned if the staff training requirement is in addition to continuing education requirements for clinical staff.
Response: We are aware that the majority of hospitals participate in the Medicare program through deemed status with an accrediting organization [675] and that accrediting organizations may have additional requirements that exceed Medicare CoP requirements as part of their CMS-approved deeming program. If a facility is in compliance with those requirements, this may also facilitate compliance with this requirement. Continuing medical education (CME) from local, State, hospital, specialty society, and accrediting organizations may be used by providers and facilities to meet these new requirements, subject to (1) facility's choice of “evidence-based best practices and protocols to improve the delivery of maternal care within the facility and (2) facility QAPI findings. For example, a specialty society may require that providers complete CME on maternal hypertension (HTN). If the hospital chooses maternal HTN as a training topic and identifies the same specialty society training, then it would align, and the provider may only have to do one training to meet both requirements. Conversely, if the hospital does not choose maternal HTN or creates its own training/protocol, then the provider would have to complete two trainings in this case.
Comment: A few commenters cautiously supported including REHs in the requirements for OB staff training, to the extent such training fits with the REH's scope of practice. A commenter explained that because these requirements allow for flexibility in designing the training curriculum and selecting appropriate trainers, this requirement should apply to REHs. However, another commenter expressed the concern that the ability to provide training would be diminished for REHs because of lack of experience with OB emergencies.
Response: While we recognize that OB staff training is important for REHs as patients utilizing these services may be some of the most high-risk populations, we are not extending the OB staff training requirement to REHs at this time. While not required, we still encourage REHs to train their staff on evidence-based best practices to improve the delivery of maternal care to their patients, within the scope and complexity of the services offered in their facility, to the extent the REH has the resources/capacity to do so. We acknowledge that some REHs may have already implemented these practices. We expect facilities to utilize organizations or individuals that are properly qualified or credentialed to ( printed page 94483) oversee the training and have demonstrated knowledgeable on the subjects.
Final Rule Action: After consideration of the public comments we received, we are finalizing the staff training requirements with modifications as outlined below.
For hospitals, we are finalizing the following CoPs as proposed:
- Section 482.59(c)(1).
- Section 482.59(c)(1)(i).
- Section 482.49(c)(1)(ii).
- Section 482.49(c)(4).
- Section 482.49(c)(5).
For hospitals, we are finalizing the following CoPs with modifications:
- Section 482.59(c) with a modification to clarify that the effective date of the staff training requirements for hospitals is January 1, 2027 (two years are the effective date of the final rule).
- Section 482.59(c)(2) with a modification to require the hospital to provide relevant staff with initial training.
- Section 482.59(c)(3) with a modification to require the governing body to identify and document which staff must complete an initial training and subsequent biannual training.
For CAHs, we are finalizing the following CoPs as proposed:
- Section 485.649(c).
- Section 485.649(c)(1)(i).
- Section 485.649(c)(1)(ii).
- Section 485.649(c)(4).
- Section 485.649(c)(5).
For CAHs, we are finalizing the following CoPs with modifications:
- Section 485.649(c) with a modification to clarify that the effective date of the staff training requirements for CAHs is January 1, 2027 (two years after the effective date of the final rule).
- Section 485.649(c)(2) with a modification to require the CAH to provide relevant staff with initial training.
- Section 485.649(c)(3) with a modification to require the governing body to identify and document which staff must complete an initial training and subsequent biannual training.
4. Quality Assessment and Performance Improvement (QAPI) Program (§ 482.21; § 485.641)
We proposed revisions to the existing QAPI standards (§ 482.21; § 485.641) for hospitals and CAHs that offer obstetrical services.[676] First, we proposed that a hospital or CAH that offers OB services would be required to use its QAPI program to assess and improve health outcomes and disparities among OB patients on an ongoing basis (§ 482.21(b); § 485.641(e)(2)). Specifically, the facility at a minimum must: (1) analyze data and quality indicators collected for the QAPI program by diverse subpopulations as identified by the facility among OB patients; (2) measure, analyze, and track data, measures, and quality indicators on patient outcomes and disparities in processes of care, services and operations, and outcomes among OB patients; (3) analyze and prioritize patient health outcomes and disparities, develop and implement actions to improve patient health outcomes and disparities, measure results, and track performance to ensure improvements are sustained when disparities exist among OB patients; and (4) conduct at least one performance improvement project focused on improving health outcomes and disparities among the hospital's population(s) of OB patients annually.
Next, under a new standard for Maternal Health QAPI activities for hospitals (§ 482.21(e)(1)) and CAHs (§ 485.641(d)(4)(i)), we proposed to require that for hospitals and CAHs that offer OB services, leadership must be engaged in the facility's QAPI activities. For purposes of this provision, leadership is defined as facility leadership, obstetrical services leadership, or their designate(s).
We further proposed that if a MMRC is available at the State or local jurisdiction in which the facility was located, hospitals (at § 482.21(e)(2)) and CAHs (at § 485.641(d)(4)(ii)) that offer OB services must have a process for incorporating MMRC data and recommendations into the facility's QAPI program.
Finally, we solicited public comments and evidence related to these proposals, including how these proposals would impact maternal health and safety, how facilities currently use their QAPI programs to address maternal health, best practices for data analysis and stratification in QAPI programs, best practices for sharing QAPI findings with impacted communities, and whether these proposed requirements should be applicable to REHs.
We received several comments on these proposals. We address those comments and our responses below:
Comment: Many commenters supported encouraging the use of QAPI programs to collect data, evaluate disparities, and address gaps in maternal health care to improve equity and outcomes for OB patients. Specifically, several commenters supported the QAPI revisions for OB patients related to data analysis, describing such QAPI work as important for improving maternal health. Several commenters stated that targeted data collection and analysis by diverse subpopulations is important for identifying disparities and enabling data-driven interventions. A few commenters also appreciated the CMS' permitting flexibility for hospitals to use the data analysis methodology most appropriate for their patient population.
However, other commenters expressed concern regarding the potential burden of these standards in terms of staff, technology, and data expertise, which could cause more hospitals to eliminate OB services and contribute to worse access to OB care and higher risks of maternal and infant morbidity and mortality, especially in rural and underserved communities. Multiple commenters recommended a phased implementation of the requirements to ensure facilities can successfully meet the new standards. Multiple commenters recommended that CMS offer resources (including financial) and technical assistance to support implementation, especially in rural facilities.
Response: We appreciate all input received on this topic. We agree that data analysis by diverse subgroup can drive improvements in maternal health and reduce associated disparities. As discussed in the CY 2025 OPPS/ASC proposed rule preamble, this new standard builds on other CMS quality improvement and data analysis initiatives, such as the “Birthing-Friendly” hospital designation and complements existing work by State Perinatal Quality Collaboratives.[677] Such initiatives can reduce the burden on facilities in meeting these revised requirements. Facilities may also choose to partner with QIOs as well as leverage integrated health system resources (as described in 42 CFR 482.21(f)) in order to reduce any burden of compliance. In addition to the resources cited in the CY 2025 OPPS/ASC proposed rule, CMS will develop guidance for facilities to support compliance with these new standards. We note that financial assistance or issues pertaining to payment policy are beyond the scope of ( printed page 94484) the CoPs. CMS will also allow for a phased in implementation timeline for these QAPI standards as further discussed in section XXI.A.7 of this final rule with comment period.
Comment: Multiple commenters noted concern for duplication and overlapping Federal quality efforts related to maternal health and these new CoPs. One commenter stated that the existing QAPI CoP requirements are comprehensive, making any new QAPI requirements unnecessary and duplicative.
Instead of the CoPs, several commenters suggested CMS use other policy levers, such as existing quality reporting and performance improvement programs, to incentivize improvements in maternal health care. Another commenter noted that hospitals are already involved in multiple OB quality efforts, including already having a QAPI program in their OB department, pursuing the “Birthing-Friendly” designation, and adopting maternal health focused quality measures. Additionally, a few commenters noted hospitals are already working to comply with CMS IQR program's new “Hospital Commitment to Health Equity” measure,[678] and that other programs exist to report data related to cesarean rates stratified by race and ethnicity. One commenter expressed concern that multiple projects addressing stratification may lead to confusing or duplicative requirements, which could increase burden on safety net hospitals which are already resource limited. Another commenter stated that the HHS Perinatal Improvement Collaborative is an existing example of using data to drive clinical quality improvement and advance health equity.
Commenters also flagged that the Federal Government is currently implementing new policies for the collection and use of race and ethnicity data. A few commenters recommended postponing requirements on the collection, reporting, and use of race and ethnicity data until OMB's revised race and ethnicity data collection standards are fully implemented. These commenters recommended focusing instead on other activities, such as implementation of HHS's Action Plan on Race and Ethnicity Data in preparation for the revised data collection standards.[679]
Overall, these commenters recommended that CMS support providers in these existing quality initiatives, as well as State Perinatal Quality Collaboratives, that foster the sharing of innovations and best practices, rather than pursue new CoPs. Multiple commenters recommended allowing hospitals flexibility to determine their own priorities for data collection, tracking, and analysis rather than new CoPs. A few commenters suggested that CMS provide incentives to facilities that demonstrate participation in evidence-based initiatives to improve maternal and infant health.
Response: CMS has developed a robust number of quality initiatives to spur improvements in maternal health, including the “Birthing-Friendly” designation [680] and hospital IQR measures [681] related to maternal health. We appreciate commenters highlighting CMS' ongoing efforts related to quality metrics and maternal health. CMS considers the new QAPI standards for OB services to be complementary to these quality initiatives, rather than duplicative. CMS is not seeking to create new burden nor have facilities repeat existing work, but rather ensure facilities' participation in quality initiatives in order to improve maternal morbidity and mortality while preserving flexibility for facilities in how they choose to go about doing so. Facilities may choose to develop their own quality initiatives/projects or join other local/state/Federal maternal health quality efforts as part of their QAPI program. As discussed in the CY 2025 OPPS/ASC proposed rule preamble, facilities may use data from State Perinatal Quality Collaboratives, CMS IQR maternal health quality measures, or other Federal or State maternal health quality initiatives in order to meet these new QAPI standards at reduced burden. CMS has also provided facilities with the flexibility to choose which maternal health topics and data analysis methods to use in meeting these standards so that their QAPI programs may be tailored to their unique patient populations.
Comment: Commenters also questioned whether new QAPI standards for OB services could cause unintended consequences due to low data quality and/or lack of data expertise. Specifically, a few commenters expressed concerns that analyses based on data which has not been validated may cause health care systems and hospitals to expend resources to identify trends or establish policies that fail to address the root cause of poor maternal health outcomes and inequities. A few commenters cautioned that misclassification or aggregation of data across diverse populations can mask disparities or inflate trends, leading to faulty conclusions, and that failing to account for systemic biases in data collection or analysis can perpetuate existing inequities. Another commenter expressed concern that many hospitals do not have staff sufficiently skilled in analyzing complex datasets to understand the relationship of SDOH data to clinical outcomes.
Response: As discussed in the preamble, hospitals are encouraged to utilize the data analysis methods that best fits their patient population and data analysis resources, and to utilize best practices in conducting their QAPI work. Partnering with QIOs, joining existing local/state/Federal quality efforts, and leveraging integrated health system resources can all be potential options for allowing facilities to comply with these new standards and access data analysis expertise and staff support, while being mindful of burden.
Comment: While several commenters appreciated CMS' permitting flexibility for hospitals to use the data analysis methodology most appropriate for their patient population, other commenters recommended that CMS consider the needs of providers with limited data collection resources or capabilities (staff, technology, data). Specifically, a few commenters stated that it is difficult to conduct statistically valid analyses of subpopulations due to the small number of deliveries in many rural hospitals, and that these data challenges complicate the efforts of smaller hospitals to assess and address disparities. Several commenters were concerned that smaller healthcare facilities may face privacy concerns due to the small size of certain subgroups. Additionally, another commenter recommended CMS ensure EHR vendors can support the required subpopulation analyses prior to adopting these requirements.
Response: CMS is committed to ensuring that all mothers, regardless of facility size or size of their patient demographic group within a particular facility, have access to high quality care. CMS is not requiring specific data analysis nor stratification methods. Rather, hospitals are encouraged to utilize the data analysis method that best fits their patient population and resources and to utilize best practices in conducting their QAPI work. Facilities ( printed page 94485) also have the flexibility to define diverse populations and set the time period for their analyses that best supports robust QAPI evaluations and patient privacy. The proposal does not require that facilities utilize EHRs to conduct their QAPI work, though CMS recognizes that electronic systems can greatly aid this work. We encourage facilities to adopt the analysis method that best suits their available resources, and leverage partnerships with QIOs, existing local/state/Federal quality efforts, and integrated health system resources in order to reduce burden and mitigate data challenges.
Comment: CMS received mixed comments regarding the effectiveness of quality improvement projects to address the maternal health crisis. Several commenters noted that targeted data collection and analysis by diverse subpopulations is important for identifying disparities and enabling data-driven interventions. In contrast, one commenter stated that CMS has not provided evidence that the proposed data analysis requirements are likely to improve maternal health outcomes. Another commenter stated that some hospitals have been working to improve maternal health outcomes without relying on regulatory requirements and have found that disparities persist. These commenters suggest that CMS consider policies addressing non-clinical factors (meaning the social determinants of health (SDOH), such as transportation access, housing quality, and insurance coverage), which have a significant impact on maternal mortality and morbidity, rather than establish new CoPs. On the other hand, several other commenters recommended CMS address the root causes of the maternal health crisis by requiring facilities to prioritize identifying and addressing unmet social needs in their QAPI programs.
Response: CMS recognizes the profound impact of the SDOH on maternal health outcomes. As discussed in the preamble, research indicates data stratification and analyses can allow facilities to identify when disparities occur and develop tailored interventions. Facilities have the flexibility to design their QAPI efforts to address SDOH, clinical factors, or both based on the needs of their patients and facility resources. While the proposal does not require facilities to do so, CMS believes facilities can play a critical role in connecting patients to resources that can address SDOH and improve outcomes. We encourage facilities to partner with existing community resources to assist patients with SDOH needs.
Comment: CMS received multiple comments regarding concerns from rural providers regarding the QAPI requirements. Specifically, several commenters urged CMS to exclude REHs and CAHs from the new QAPI requirements due to concerns regarding additional burden and compliance challenges faced by rural and low-volume providers compared to larger/urban facilities. A commenter recommended providing technical assistance to support these rural providers in effectively implementing QI initiatives, including integrating quality measurement with data reporting within their EHRs.
Response: CMS is committed to ensuring high quality OB care for all mothers regardless of geographic location, but is also sensitive to the burden concerns of rural providers. As will be discussed in section XXI.A.7 of this final rule with comment period, CMS will allow for a phased in implementation timeline for these QAPI standards in order to reduce burden. We will also issue subregulatory guidance regarding how surveyors will assess compliance with these requirements after publication of the final rule. We encourage facilities to utilize the data analysis method that best suits their available resources and leverage partnerships with QIOs, existing local/state/Federal quality efforts, and integrated health system resources in order to reduce burden and mitigate data challenges. While the proposal does not require that facilities use EHRs in their QAPI work, providers interested in doing so may wish to review the following resources from the Office of the National Coordinator for Health Information Technology (ONC) resources among others:
- ONC. “Clinical Quality and Safety.”https://www.healthit.gov/topic/clinical-quality-and-safety.
- ONC. “Quality Improvement Briefs.”https://www.healthit.gov/resource/quality-improvement-briefs.
- ONC. “Continuous Quality Improvement.”https://www.healthit.gov/faq/what-continuous-quality-improvement.
ONC. “Section 8: Quality and Patient Safety.” Health IT Playbook. https://www.healthit.gov/playbook/quality-and-patient-safety/.
Comment: We received several comments regarding the proposal to require facilities to conduct at least one maternal health performance improvement project (PIPs) annually. A few commenters supported the proposal, noting its potential to spur improvements in maternal morbidity and mortality. However other commenters stated that such projects may take longer than one year to implement and evaluate, and suggested CMS allow PIPs over a longer period (such as two or three years). A commenter supported allowing providers to choose PIPs that align with State or Federal quality initiatives and are specific to the needs of their facility or community but expressed concern that providers that do not participate in statewide maternal health initiatives may struggle to meet the new requirements. Two commenters suggested CMS allow hospitals to fulfill the goal of the PIP requirement through their participation in statewide quality improvement initiatives, such as the Perinatal Quality Collaboratives and the AIM patient safety bundles.
Response: We agree that PIPs can take longer than one year to fully implement and evaluate. We did not propose to require that facilities conduct a new/different maternal PIP each year. Rather facilities must be actively performing at least one PIP on maternal health a year, but this can be the same PIP over multiple years. As discussed in the preamble, facilities may choose to join existing local/state/Federal quality initiatives and utilize resources such as Perinatal Quality Collaboratives and the AIM patient safety bundles in order to comply with this requirement and reduce burden.
Comment: CMS received mixed comments regarding the proposal to require OB leadership to engage in QAPI. Several commenters stated that QAPI activities would be more successful with buy-in from facility leadership, which would ensure alignment with organizational goals and allocation of necessary resources. One commenter stated that the proposal is redundant because existing CoPs already outline who is responsible and accountable for ensuring the implementation of QAPI activities. One commenter recommended delaying the requirement to ensure leadership is engaged in QAPI activities by 12 months to allow time to train OB providers in quality improvement methods.
Response: We agree with commenters that leadership engagement is often a key ingredient for successful QAPI efforts. While the existing CoPs do set certain expectations for QAPI oversight, they do not specifically require OB leadership involvement as these requirements do. We believe this level of detail is needed in order to address the maternal health crisis. As discussed in section XXI.A.7 of this final rule with comment period, we will allow for a phased in implementation timeline for ( printed page 94486) these QAPI standards in order to reduce burden.
Comment: Many commenters supported the incorporation of MMRC data into facilities' QAPI programs. Commenters noted that MMRC data can inform hospital policies and patient safety practices and improve disparities. A commenter highlighted that the California Maternal Quality Care Collaborative (CMQCC) [682] already engages in these activities and is achieving success in targeting complications which disproportionately affect Black patients to drive better perinatal outcomes.
A few commenters did not support the proposal. One commenter said the proposal would be unnecessarily burdensome, particularly for rural hospitals with low volumes of births. Another noted lack of support for the proposal given that maternal deaths are rare, especially those that occur within the hospital. These commenters recommended allowing hospitals to focus on internal morbidity data instead of statewide data.
Several commenters also expressed concern regarding MMRC data itself, specifically that these data may not be relevant to all hospitals (particularly rural hospitals), that recommendations from MMRCs may be inconsistent across States, and that data collection is slow. These commenters recommended allowing flexibility. In particular, these commenters suggested allowing collaborations with other hospitals on performance improvement projects to satisfy the requirements, instead of relying on MMRCs.
Response: As discussed in the CY 2025 OPPS/ASC proposed rule preamble, evidence shows that data from MMRCs has been central in driving improvements in maternal morbidity and mortality across the country. While CMS is sensitive to concerns from rural providers, CMS disagrees that exclusion or modification of this MMRC proposal for rural providers is appropriate. CMS notes that State MMRCs often assess maternal deaths by region and/or by rurality. In this way, MMRC data can be relevant and specific to rural areas. Additionally, MMRC recommendations are written to allow facilities flexibility in their implementation rather than a one-size-fits-all approach, which allows rural facilities to adapt MMRC recommendations to their circumstances. Lastly, CMS notes that rural areas are not the only areas with limited resources or OB access. Urban hospitals, especially those in certain historically underserved communities, may also experience these challenges. CMS agrees that collaboration on QAPI work across different facilities and entities can enhance results and reduce burden. The proposal permits facilities to engage in such collaboration but does not require it. In particular, facilities may wish to join ongoing existing State Perinatal Quality Collaboratives, partner with QIOs, and/or leverage integrated health system resources (as described in 42 CFR 482.21(f)) in order to reduce any burden of compliance.
Comment: One commenter recommended that Tribal MMRCs be included alongside State and local MMRCs. This commenter noted that though Tribal MMRCs are not currently operating, their inclusion in the proposal could support their development. The commenter further asked that facilities be required to report data to any relevant Tribal MMRCs as requested by the Tribal MMRC.
Response: We appreciate this comment. To clarify, the current proposal does not include any requirement for facilities to report data to MMRCs (in deference to existing State laws on this subject).[683] We will not amend the proposal at this time related to data reporting, however we will amend the requirement on utilizing data from MMRCs to recognize Tribal MMRCs.
Comment: One commenter stated that requirements in the proposal are too vague and could be subject to disparate interpretations by CMS surveyors, suggesting CMS engage in provider listening sessions to add more specificity to the requirements before finalizing them.
Response: We will be issuing interpretive guidance following this final rule in order to assist facilities in complying with these new CoP standards.
Comment: Several commenters suggested CMS add specific additional topics and detail to the QAPI proposals. One commenter suggested a focus on pre-natal counseling and through post-partum follow-up as areas for the greatest potential to impact quality and safety. Another commenter recommended that CMS require stratification of maternal sepsis and neonatal infection as critical patient safety outcomes. This commenter stated that rates of sepsis during pregnancy are higher for Black patients, Asian/Pacific Islander patients, and Native American patients and stated that understanding maternal sepsis data, including time to initiation of appropriate therapy, through stratified data could inform performance improvement strategies. Another commenter stated that improving evidence-based best practices for C-section births could reduce surgical site infections, which are an area with potential care disparities due to lack of access to proven care, especially for patients with obesity. Another commenter recommended that QAPI work include mental health and substance use disorder metrics and require that QAPI data be made publicly available.
Response: We appreciate commenters sharing their expertise and highlighting key maternal health topics that facilities may wish to address in QAPI work. The proposal does not require facilities to address specific maternal health topics for quality improvement. Instead, facilities are free to choose the topic that best aligns with their patient populations and resources. Given that the current proposal did not include any requirement for facilities to report their maternal QAPI data nor make such data publicly available,[684] we are not revising the requirement at this time related to data reporting.
Comment: Several commenters recommended that CMS address a major cause of the maternal health crisis by requiring facilities to prioritize identifying and addressing unmet mental health and substance use disorders among OB patients. A commenter suggested that facilities collect data on maternal mental health and substance use disorder (SUD) specific metrics to evaluate the effectiveness of evidence-based practices, such as postpartum depression screening, follow-up, and initiation and engagement of SUD treatment. A few commenters further suggested that hospitals be required to collect, report, and incorporate data on rates of drug testing of pregnant people, medical justification for drug testing, demographic makeup of those who are drug tested, and health outcomes of both mother and fetus. A few commenters suggested that facilities further engage with stakeholders with lived experience in mental health and substance use disorder to determine which data and quality indicators are collected.
Response: We appreciate commenters sharing their expertise. CMS recognizes mental health and SUD as a critical area for maternal health. While the proposal does not require facilities to address specific maternal health topics for quality improvement, facilities that ( printed page 94487) wish to work on maternal health, SUD, and behavioral health within their QAPI programs may wish to review the following resources among others:
- SAMHSA. “Taskforce on Maternal Mental Health.”https://www.samhsa.gov/about-us/advisory-councils/acws/task-force-maternal-mental-health.
- SAMHSA. “Advisory: Evidence-Based, Whole Person Care of Pregnant People Who Have Opioid Use Disorder.”https://store.samhsa.gov/product/advisory-evidence-based-whole-person-care-pregnant-people-who-have-opioid-use-disorder.
- Alliance for Innovation on Maternal Health. “Perinatal Mental Health Conditions.”https://saferbirth.org/psbs/perinatal-mental-health-conditions.
- Alliance for Innovation on Maternal Health. “Maternal Mental Health.”https://saferbirth.org/psbs/archive-maternal-mental-health/.
- Alliance for Innovation on Maternal Health. “Care for Pregnant and Postpartum People with Substance Use Disorder.”https://saferbirth.org/psbs/care-for-pregnant-and-postpartum-people-with-substance-use-disorder/.
SAMHSA also has provided funding opportunities for a variety of maternal behavioral health initiatives.[685 686 687]
Comment: For CMS' questions on best practices for QAPI data analysis and stratification by diverse subgroup and as well as which data stratifications/subgroups/categories are key to ensuring the health and safety of all pregnant, birthing, and postpartum patients, we received several responses. One commenter recommended that CMS incorporate and share best practices to promote accuracy in demographic data collection, such as the ability for patients to self-identify, and to support privacy protections in the collection and reporting of aggregated demographic data. One commenter recommended accounting for patient preferences regarding sharing demographic data when adopting data collection requirements.
A commenter noted that current stratification practices vary according to site and are often impacted by resource constraints. Several commenters recommended CMS identify a minimum set of standardized data elements that impact maternal health outcomes, such as race, ethnicity, disability status, language, age, sexual orientation and gender identity (SOGI), geographic location (including rural versus urban; distance traveled), socioeconomic status (such as income level), immigration status, zip code, payor, gender identity, among other SDOH factors. A few commenters requested additional guidance to support SDOH Z-codes. Another commenter added that CMS should work with stakeholders to identify these common data elements. A few commenters recommended that CMS work with organizations that have already created comprehensive data processes to effectively design data collection practices that hospitals can use to meet nationally recognized standards. Specifically, one commenter further recommended collaboration with the private sector to adopt meaningful measures of maternal health outcomes and disparities such as demographic factors. One commenter suggested that CMS could identify disparities early by promoting the uniform collection of demographic data and consistent stratification of metrics by using the updated OMB Standards for Maintaining, Collecting, and Presenting Federal Data on Race and Ethnicity.
In terms of best practices for QAPI work, one commenter stated that multidisciplinary collaboration is necessary for effective QAPI processes, including the review of maternal health outcomes. This commenter recommended ensuring that QAPI meetings are led by physician and nurse leadership in collaboration with a range of other disciplines and services throughout the hospital system. Another commenter recommended including practicing OB-GYNs, other OB unit providers, non-medical staff, and patients in QAPI activities to ensure initiatives are practical, address working conditions, and minimize burden.
Response: We thank the commenters for their expertise and feedback on this comment solicitation. Following finalization of this rule, we will issue subregulatory guidance regarding how compliance with this requirement will be assessed. CMS is committed to providing technical assistance to support hospitals in coming into compliance with these requirements, including data analysis by diverse subpopulation. While CMS is requiring that “obstetrical services leadership” be involved in facility QAPI work (at §§ 482.21(e)(1) and 485.641(d)(4)(i)), facilities have flexibility regarding additional staff involvement in QAPI work. We agree with commenters that multidisciplinary collaboration is often critical to the success of QAPI activities. Payment policies (such as Z codes) are outside the scope of the CoPs, but we will share this feedback with the appropriate component within CMS.
Comment: Related to CMS' question on best practices for sharing QAPI findings with impacted communities, we received several responses. A commenter stated that community engagement is important to ensuring initiatives are leading to improvements in the community's actual needs. One commenter recommended using patient advisory councils to ensure patient involvement in QAPI initiatives.
A few commenters recommended sharing results of QAPI initiatives with the medical and scientific community, with other providers, and with the public that they serve. Other commenters recommended using existing communication channels, including Patient and Family Advisory Councils, websites, newsletters, community forums, and media outlets (including newspapers and television) to share these data. A few commenters recommended that the Federal Government collect these data and make them publicly available at a single central location.
A few commenters stated that engaging community members, especially in under-resourced communities, can be challenging and recommended establishing technical assistance to help hospitals with this engagement. One commenter stated that technical assistance could include strategies such as addressing power and knowledge differentials, providing materials accessible to lay audiences, incorporating community members from the outset, and compensating community members for their contributions.
Response: We thank the commenters for their input and will consider this feedback in possible future rulemaking. CMS encourages facilities to collaborate with communities to ensure the facility's QAPI work meets patients' needs, and to share QAPI outcomes with communities as applicable.
Comment: Related to CMS' question whether these proposed requirements should be applicable to REHs, we received several responses. A few commenters stated that all hospitals, including REHs, should meet consistent health and safety standards and suggested that CMS find ways to support REHs in providing quality care, including for QAPI. A commenter stated that requiring all levels of care, including REHs, to have baseline requirements regarding OB care delivery is critical to reducing disparities. A few ( printed page 94488) commenters suggested providing phase-in timelines as well as financial and technical support for REHs.
However, many commenters were opposed to applying these QAPI proposals to REHs. Several commenters remarked that several of the proposals are not applicable to REHs, and that coming into compliance would be overly burdensome. Several commenters stated that REHs have their own payment methodology and CoPs and should not be required to comply with CoPs meant for other hospital types. Some commenters expressed concern that requiring REHs to comply with these proposals would result in additional closures. A commenter stated that REHs should not be required to have a QAPI due to the resources required.
Response: We thank the commenters for their input. In light of the concerns raised, we will not extend these QAPI standards to REHs at this time.
Final Policy Decision: After consideration of the public comments we received, we are finalizing the proposals, without modification for hospitals and CAHs. CMS is not extending the new QAPI standards to REHs at this time.
5. Emergency Services Readiness (§ 482.55; § 485.618)
We proposed a new standard entitled “Emergency Services Readiness” within the existing Emergency Services CoP for hospitals (§ 482.55) and CAHs (§ 485.618) to set clear expectations as well as improve facility readiness in caring for emergency services patients, including pregnant, birthing, and postpartum patients. Notably, these requirements would apply to all hospitals and CAHs offering emergency services,[688] whether or not a hospital/CAH offers an additional specialty service line (such as OB services). This is done with the intention of ensuring baseline health, safety, and training standards for the care of patients with emergency conditions.
First for hospitals (§ 482.55(c)) and CAHs (§ 485.618(e)) that offer emergency services, we proposed that these facilities would be required to have adequate provisions and protocols to meet the emergency needs of patients in accordance with the complexity and scope of services offered. For protocols, hospitals (§ 482.55(c)(1)) and CAHs (§ 485.618(e)(1)) must have protocols consistent with nationally recognized and evidence-based guidelines for the care of patients with emergency conditions. While these CoPs would not require adherence to a specific organization's guideline or recommendations, we expect that facilities would be able to articulate their standards and the source(s) and to demonstrate that their standards are based on evidence and nationally recognized sources.
For hospitals (§ 482.55(c)(3)) and CAHs (§ 485.618(e)(2)) that offer emergency services, applicable emergency services personnel, as determined by the facility, must be trained on these protocols and provisions annually. Once staff are identified, hospitals and CAHs must document that applicable staff have successfully completed such facility-identified training and demonstrate staff knowledge on these topics.
For hospitals that offer emergency services, we further proposed at § 482.55(c)(2) that provisions include equipment, supplies, and medication used in treating emergency cases. Such provisions must be kept at the hospital and be readily available for treating emergency cases. The available provisions must include: (1) drugs, blood and blood products, and biologicals commonly used in life-saving procedures; (2) equipment and supplies commonly used in life-saving procedures; and (3) a call-in-system for each patient in each emergency services treatment area. These supply requirements are similar to existing CAHs (at § 485.618(b) and (c)) and REHs (at § 485.516(c)(2)) supply standards for emergency services, as well as the surgical services CoP supply requirements (§ 482.51(b)(3)). We did not propose any new emergency services equipment, supplies, or medication requirements for CAHs or REHs.
Finally, we solicited public comments and requested evidence related to these proposals, including how these proposals would impact patient (including maternal) health and safety, and whether these proposed requirements should be applicable to REHs.
We received several comments on these proposals. We address those comments and our responses below:
Comment: We received mixed comments on the proposal overall. Several commenters supported adopting requirements that address care in non-obstetric settings, such as emergency departments (EDs). One commenter stated that they felt that existing courses (such as Advanced Cardiovascular Life Support, Advanced Trauma Life Support, and Pediatric Advanced Life Support) are insufficient emergency training, noting that newly graduated NPs and PAs may be working in rural hospitals and CAHs with no or extremely limited emergency training, including in management of OB emergencies. This commenter supported additional training in OB emergencies for all rural hospital and CAH emergency services providers. One commenter stated high quality OB care in the ED is especially important in areas that are resource limited and do not have access to OB care. Another commenter added that training emergency clinical staff on obstetrical and gynecological emergencies would enhance patient safety in rural hospitals and CAHs because emergency providers often provide care in these settings due to a lack of OB specialists. One commenter further stated that ensuring EDs are equipped to treat OB emergencies can promote access even in areas without OB units. Another commenter noted that these requirements are particularly important as restrictions on reproductive care are exacerbating shortages of OB services in some locations.
In contrast, multiple commenters expressed opposition to the proposal. Several commenters stated that these requirements are duplicative to other CoPs. Specifically, a few commenters stated that emergency services protocols for OB patients are redundant of existing requirements because hospitals manage OB patients according to existing protocols and other relevant Conditions of Participation. Another commenter stated that emergency personnel are already trained in a breadth of diagnoses and recommended allowing hospitals flexibility to identify appropriate training based on their patient populations. Several commenters recommended that CMS implement the regulations carefully to avoid detrimental effects, especially for rural hospitals.
Response: We appreciate commenters' support for the proposal. The proposal aims to ensure the health and safety of all emergency services patients, including obstetrical patients, by requiring hospitals and CAHs to have appropriate emergency protocols and provisions in place. We agree that declines in obstetrical access across the country, including in rural areas, increase the importance of training in obstetrical and gynecological emergencies for emergency clinical staff.
We disagree that the proposal duplicates existing CoPs. There are currently no requirements related to emergency provisions (including for ( printed page 94489) obstetrical patients) under the existing hospital emergency services CoP. In contrast, CAHs (at § 485.618(b) and (c)) and REHs (at § 485.516(c)(2)) do have supply requirements under their emergency services CoP. The proposal therefore brings hospitals' emergency provisions expectations into alignment with the CAH and REH CoPs on this topic. Similarly for protocols, the existing hospital requirement (42 CFR 482.55(a)(3)) that “the policies and procedures governing medical care provided in the emergency service or department are established by and are a continuing responsibility of the medical staff” offers little detail of CMS' expectations for emergency readiness. The CAH emergency services CoP lacks such a provision entirely. For staff training, the hospital emergency services CoP requires that “there must be adequate medical and nursing personnel qualified in emergency care to meet the written emergency procedures and needs anticipated by the facility” (§ 482.55(b)(2)). The CAH standard (§ 485.618(d)) currently requires its personnel to have “training and experience in emergency care.” CMS believes clearer expectations surrounding “qualified/training and experience in emergency care” and maintenance of qualifications (that is, training) would improve facilities' readiness to care for patients with emergency conditions, enhancing patient health and safety.
The proposal therefore offers clarity regarding CMS' expectations for emergency readiness including protocols, provisions, and training for emergency staff. In response to providers' concerns, especially rural facilities, related to burden, CMS will institute an implementation delay as discussed in section A.XXI.7 of this final rule with comment period.
Comment: A few commenters stated that it is unclear whether the proposed updates were intended to be specific to OB emergencies or apply more broadly.
Response: We appreciate the opportunity to offer further clarity. The proposal is written broadly to ensure hospitals and CAHs meet the needs of all emergency services patients, including, but not limited to, patients with obstetrical emergencies, complications, and immediate post-delivery care needs. Specifically, the proposal requires that hospitals and CAHs develop protocols consistent with nationally recognized and evidence-based guidelines for the care of patients with emergency conditions. We further specify that these protocols must address obstetrical emergencies, complications, and immediate post-delivery care (in accordance with the complexity and scope of services offered at the facility), but the protocol requirement is not limited to these conditions. CMS offers hospitals and CAHs flexibility in determining what other protocols are needed to meet the needs of their specific emergency services patient populations. The same applies for the emergency provisions requirement for hospitals.
Comment: A commenter inquired regarding the scope of CMS' expectations regarding emergency protocols. In particular, this commenter asked if the proposal required facilities to develop protocols for all possible emergency conditions.
Response: We appreciate the opportunity to offer further clarity. We are not requiring that facilities develop a protocol or have provisions for every possible emergency scenario, but facilities should develop protocols and have available provisions to meet the emergency needs of their patients consistent with (1) the complexity and scope of services offered by the facility; and (2) nationally recognized and evidence-based guidelines. The final rule we are adopting specifies that protocols must address obstetrical emergencies, complications, and immediate post-delivery care, but are not limited to these conditions. CMS offers hospitals and CAHs flexibility in determining what other protocols are needed to meet the needs of their specific emergency services patient populations. For example, the American College of Emergency Physicians has issued multiple guidelines for best practices in managing common and critical emergency conditions and has developed a Geriatric Emergency Department Accreditation Program, which provides best practice standards for this at-risk population.[689 690] For obstetrical emergencies, the Alliance for Innovation on Maternal Health's (AIM; a partnership between HRSA and American College of Obstetricians and Gynecologists (ACOG) and other stakeholders) has developed resources which include example protocols and training resources for responding to obstetrical hemorrhage, severe hypertension, perinatal mental health conditions, sepsis, substance use disorder, and cardiac conditions, among others.[691] ACOG has also developed resources for Obstetric Emergencies in Nonobstetric Settings.[692] Similarly, the HRSA-supported Emergency Medical Services for Children (EMSC) Innovation and Improvement Center has resources for emergency departments seeking to improve “pediatric readiness.” [693] We expect hospitals to be able to identify the source of the nationally recognized and evidence-based guidelines utilized in their protocols.
Comment: EMTALA's relationship to the proposal was brought up by multiple commenters. One commenter supported the proposed rule and emphasized the requirement for hospitals with EDs to comply with EMTALA and Federal civil rights laws. In contrast, several commenters stated that these requirements duplicate requirements for hospitals under EMTALA.
Response: EMTALA requirements are separate and distinct from these requirements. As noted elsewhere, CMS expects that all Medicare-participating facilities with emergency departments comply with the EMTALA statute (42 U.S.C. 1395dd) and its implementing regulations (42 CFR 489.24). Specifically, under EMTALA, Medicare-participating hospitals and CAHs with emergency departments must provide individuals presenting to the emergency department with an appropriate medical screening exam and, if an emergency medical condition exists, offer stabilizing treatment or, under certain circumstances, appropriate transfer to receive stabilizing care.
However, EMTALA does not directly address emergency department readiness, which may have exacerbated gaps in access to care, including for obstetrical, geriatric, and pediatric populations.[694 695 696 697 698 699] This final rule therefore offers clarity regarding CMS' expectations for emergency readiness for all patients, inclusive of protocols, provisions, and training for emergency staff. The proposed rule preamble included multiple example ( printed page 94490) resources facilities may utilize to assist in compliance with the proposal.[700 701 702 703 704]
Comment: We received several questions requesting further guidance on the proposal. One commenter recommended that CMS provide technical resources that would facilitate identification and adoption of nationally recognized and evidence-based guidelines. This commenter also recommended adoption of guidance on the essential provisions required for emergency services or to allow hospitals to tailor their provisions to their specific needs. One commenter recommended that CMS compile a list of resources, including low-cost training resources, that could be used in complying with this regulation. A few commenters recommended allowing hospitals flexibility in selecting which resources and nationally recognized standards to use to meet the requirements, including QAPI and QA activities. Another commenter recommended ensuring that our policies not be prescriptive, which could limit flexibility during a surge beyond typical patient volume.
Response: We are not requiring that facilities adhere to a specific organization's guidelines or recommendations for emergency protocols and provisions. Facilities have flexibility to utilize national medical professional society, accrediting organization, credentialling body, or other national guidelines when determining appropriate protocols and provisions for their emergency services patient populations. We provide examples of such resources in the proposed rule preamble.[705 706 707 708 709] CMS encourages facilities to further inform their protocols and provisions choices based on the facility's QAPI program as appropriate. We agree that facilities should consider how their protocols may need to be adapted during surge periods. Following publication of this final rule with comment period, we will issue subregulatory guidance on how surveyors will assess compliance with this requirement, which can further assist facilities.
Comment: Several comments requested that CMS provide funding to support compliance with these new requirements. One commenter expressed concern regarding the initial cost to comply with these requirements, especially given the potential consequences for being non-compliant with CoPs. A few commenters recommended providing resources (such as funding and technical assistance).
Response: Funding resources are beyond the scope of the CoPs, but CMS continues to welcome stakeholder feedback on this topic and will share any such feedback with relevant CMS components as applicable. Following finalization of this rule, we will issue subregulatory guidance regarding how compliance with this requirement will be assessed. CMS is committed to providing technical assistance to support hospitals in coming into compliance with these requirements.
Comment: A few commenters supported flexibility for hospitals to define equipment used in lifesaving procedures. A few commenters expressed concern that requiring hospitals to have drugs, blood, blood products, and biologicals commonly used in emergency procedure could create a shortage of drugs and blood supplies.
Response: We are not requiring that facilities have any specific equipment, supplies, nor medication for emergency conditions. Facilities must determine (in accordance with the complexity and scope of services offered) what provisions are necessary to meet the emergency needs of their patients. Facilities may also want to consider what a resilient supply of specific equipment, supplies, and medication would be given their needs. We disagree that these polices would cause a shortage of drugs or blood supplies. In industry stakeholder listening sessions, stakeholders indicated hospitals already routinely have these provisions available. Moreover, CAHs (at § 485.618(b) and (c)) and REHs (at § 485.516(c)(2)) already have equipment, supplies, blood, and medication requirements under their emergency services CoP. The proposal therefore brings hospitals' emergency provisions expectations into alignment with the CAH and REH CoPs on this topic.
Comment: A few commenters requested interpretive guidance on the term “call-in system.” A commenter expressed concern that a call-in system would add financial and operational burden to hospitals.
Response: We proposed that each emergency services treatment area must have a call-in-system for each patient. The proposal is similar to the existing surgical services CoP (§ 482.51(b)(3)). By “call-in system”, we mean a mechanism by which a patient and/or caregiver can alert staff of any emergencies or concerns. Examples may include a call-bell, alarm, or other notification device. Following publication of this final rule with comment period, we will issue subregulatory guidance regarding how compliance with this requirement will be assessed. Given commenters' concern regarding burden, CMS will institute an implementation delay for this requirement as discussed in section A.XXI.7 of this final rule with comment period.
Comment: Several commenters noted the importance of behavioral health emergencies. One commenter recommended that hospitals and CAHs be required to develop evidence-based, person-centered, trauma-informed policies for the identification, treatment, and follow-up care of patients who present with an SUD-related condition, including medications for opioid use disorder and referrals to treatment and support services. The commenter recommended ensuring these protocols include policies to help address health disparities, as well as person-centered and trauma-informed care. Specific to maternal mental health, one commenter stated that evidence shows that mental health conditions (including substance use disorders and suicide) are the leading cause of pregnancy-related deaths. A few commenters recommended that CMS specifically address emergency readiness for OB patients having mental health or substance use emergencies.
Response: We appreciate commenters sharing their expertise and highlighting the importance of behavioral health emergencies, in particular as it relates to maternal health. The proposal does not require facilities to have protocols and provisions specific to behavioral health emergencies. Rather facilities may determine the protocols and provisions that best align with their patient populations. While not required, facilities that wish to work on behavioral health emergency services (including for OB patients) may wish to review the following resources among others:
- American College of Emergency Physicians. “Psychiatric Patient.”https://www.acep.org/patient-care/clinical-policies/Psychiatric-Patient.
- American Psychiatric Association. “Clinical Practice Guidelines.” https:// ( printed page 94491) www.psychiatry.org/psychiatrists/practice/clinical-practice-guidelines .
- Alliance for Innovation on Maternal Health. “Perinatal Mental Health Conditions.”https://saferbirth.org/psbs/perinatal-mental-health-conditions.
- Alliance for Innovation on Maternal Health. “Maternal Mental Health.”https://saferbirth.org/psbs/archive-maternal-mental-health.
- Alliance for Innovation on Maternal Health. “Care for Pregnant and Postpartum People with Substance Use Disorder.”https://saferbirth.org/psbs/care-for-pregnant-and-postpartum-people-with-substance-use-disorder.
- SAMHSA. “Taskforce on Maternal Mental Health.”https://www.samhsa.gov/about-us/advisory-councils/acws/task-force-maternal-mental-health.
- SAMHSA. “Advisory: Evidence-Based, Whole Person Care of Pregnant People Who Have Opioid Use Disorder.”https://store.samhsa.gov/product/advisory-evidence-based-whole-person-care-pregnant-people-who-have-opioid-use-disorder.
- SAMHSA. “Trauma and Violence.”https://www.samhsa.gov/trauma-violence.
- SAMHSA. “Clinical Guidance for Treating Pregnant and Parenting Women With Opioid Use Disorder and Their Infants.”https://store.samhsa.gov/product/clinical-guidance-treating-pregnant-and-parenting-women-opioid-use-disorder-and-their.
- SAMHSA. “Improving Cultural Competence.”https://store.samhsa.gov/sites/default/files/sma16-4931.pdf.
Comment: In offering their support for this new CoP, several comments also offered more specific recommendations for training and resources. One commenter specifically recommended addressing the risk of hemorrhage as part of OB emergency care. This commenter recommended ensuring a standard of assessing Quantitative Blood Loss (QBL) as part of early detection of maternal hemorrhage. One commenter recommended topics for training for non-OB emergency staff, including routine screening for risk factors and signs of complications in perinatal patients, stabilizing perinatal patients, and transfer protocols. The commenter noted that Association of Women's Health, Obstetric and Neonatal Nurses (AWHONN) has the courses “OB Triage” and “Maternal Fetal Triage Index” that can educate staff on common perinatal complications and appropriate escalation. A few commenters recommended using the regulations to encourage simulation-based training because this modality allows staff to practice decision-making and teamwork in a controlled environment.
Response: We thank the commenters for their expertise regarding best practices and resources. While the proposal does require facilities to ensure that relevant staff (as identified by the hospital) are trained annually on the emergency services protocols and provisions, facilities are not required to utilize any specific training modality, resources, nor metrics as part of training efforts. Rather facilities may determine the protocols, provisions, and training resources that best align with their specific situation. This is designed to enhance flexibility and reduce provider burden.
Comment: A commenter recommended that CMS adopt a requirement for hospitals to have protocols in place to reduce ED boarding, which the commenter defined as a “dangerous health system overload that puts patients in a holding pattern as they wait for an inpatient bed or transfer after their initial care.”
Response: We thank the commenter for this input. While ED boarding is beyond the scope the CY 2025 OPPS/ASC proposed rule, we will consider this input for possible future rulemaking.
Comment: We received comments regarding CAHs' standard for “Coordination with emergency response systems.” Some commenters expressed concern that these requirements for CAHs would require having a physician available on a 24/7 basis, which would be difficult to accomplish than the current requirement to have a practitioner available 24/7. A few commenters stated that the proposal to have a physician immediately available is more restrictive than a similar requirement for CAHs to have a practitioner available and would be more difficult for CAHs to meet.
Response: For clarity, we are not proposing any new standards or revisions to CAHs' “Coordination with emergency response systems” CoP (currently found at 42 CFR 485.618(e)). The text on this topic in this final rule is solely for amendatory purposes (that is, moving the current regulations text down to new 42 CFR 485.618(f)). We are not proposing any new staffing standards within CAHs' emergency services CoP.
Comment: We solicited comment on ways to reduce any burden associated with these proposals. One commenter recommended that the Federal Government create technical assistance programs for rural hospitals. These programs would be housed in the Health Resources and Services Administration (HRSA) and would support facilities with purchasing equipment and providing training and access to tele-consultation.
Response: We thank the commenter for their input. While directly providing financial support for equipment purchases and training is beyond the scope of the CoPs, we have shared this feedback with relevant CMS and HHS components.
Comment: To our question on whether these proposed requirements should apply to REHs, we received mixed responses. A few commenters supported adoption of emergency services readiness requirements for REHs. These commenters stated that many individuals giving birth may initially present to an REH and therefore these facilities should be able to recognize and address emergency situations to stabilize patients prior to transfer. Another commenter supported extending the proposed CoPs to REHs because these facilities should be prepared to provide high-quality care despite workforce and resource challenges, especially for REHs in rural, Black, and Indigenous communities which are most likely to be impacted by maternity ward closures. One commenter recommended that CMS use caution when extending the proposed updates to REHs.
Response: We thank the commenters for their input. In light of the mixed responses, CMS will not extend these new Emergency Services standards to REHs at this time.
Final Policy Decision: After consideration of the diversity of perspectives in public comments received, we are finalizing as proposed for hospitals and CAHs. CMS is not extending these new Emergency Services standards to REHs at this time.
6. Transfer Protocols (§ 482.43)
We proposed revisions to the hospital discharge planning regulations to include requirements for transfer protocols. We proposed at § 482.43(c) to require that hospitals have written policies and procedures for transferring patients under their care. This would be inclusive of hospital inpatients (for example, transfers from the emergency department to inpatient admission, transfers between inpatient units in the same hospital, as well as transfers between inpatient units at different hospitals). This would ensure that patients are transferred to the appropriate level of care promptly and without undue delay, in order to meet their needs. We also proposed to require the hospital to provide training to the relevant staff (as determined by the ( printed page 94492) facility) regarding the hospital policies and procedures for transferring patients under its care.
We received the following comments in response to this solicitation:
Comment: Some commenters supported the transfer protocols requirement. They stated requiring that hospitals have effective transfer processes was important for ensuring continuity of care, reducing the risks associated with care transitions, as well as ensuring that patients are transferred to the appropriate level of care in a timely manner. Some commenters stated that the transfer protocols, aligned with nationally recognized and evidence-based guidelines, will ensure that all pregnant women will receive timely and effective treatment.
However, some commenters opposed the transfer protocols requirement. A few commenters stated that the proposed requirement was duplicated existing transfer policies within the hospitals. One commenter suggested that CMS include only interhospital transfers (transfers between inpatient units at different hospitals) in the transfer protocols. Some commenters stated that the proposed requirement does not only focus on obstetrical or maternity care and there was insufficient evidence linking transfer protocols to better maternal outcomes. Additionally, a few commenters expressed concerns with the overly prescriptive nature of the proposed requirements, asserting that the proposal would not allow hospitals the flexibility to utilize clinical judgement to determine when and where a patient should be transferred. A few commenters suggested rather than implementing transfer protocols, hospitals should follow the guidelines for the transfers published in the Guidelines for Perinatal Care, 8th Edition. Numerous commenters expressed concerns that this would create additional burden and as a result, hospitals would close.
Response: We thank commenters for their feedback. While this requirement does not just focus on obstetrical patients, we note that effective transfer processes can improve transitions of care, increase a hospital's ability to transfer patients promptly to facilities able to appropriately care for them, as well as protect the health and safety of all patients, including pregnant, birthing, and postpartum women.
We also recognize that some hospitals might already have intrahospital (transfers within the same facility) policies in place, but hospitals that do not have an emergency department or are otherwise not covered by EMTALA, may also have a need to transfer patients to other facilities to receive needed services. Additionally, patients that are admitted as hospital inpatients may require transfer between facilities to meet the needs of the patient (for example, changing clinical condition, need for specialty and/or higher level of care). The transfer protocol requirement is meant to establish and standardize transfer protocol requirements of hospital inpatients (for example, transfers from the emergency department to inpatient admission, transfers between inpatient units in the same hospital, as well as transfers between inpatient units at different hospitals) for all hospitals.
We do not agree that the transfer protocol requirement is overly prescriptive. The regulatory flexibility and framework of the requirement will allow each hospital to establish and/or tailor its own policy parameters for inpatient transfers according to its specific patient population, individual institutional needs and resources, and own medical staff recommendations as long as the policies and procedures established and implemented meet or exceed the requirements finalized in this rule. The requirement also allows the hospital flexibility in selecting which staff are required to complete annual training regarding the hospital policies and procedures for transferring patients under their care. Lastly, to mitigate some of the potential burden, professional organizations (such as the American College of Obstetricians and Gynecologists and American Academy of Pediatrics) have resources available to support hospitals as they develop their transfer protocols. Therefore, we are finalizing that hospitals must have written policies and procedures for transferring patients under its care (inclusive of hospital inpatients) to the appropriate level of care (including to another hospital) as needed to meet the needs of the patient.
Comment: Multiple commenters suggested that staff be trained on transfer protocols annually. One commenter suggested that staff be trained upon hiring and then on an annual basis. This would ensure that staff are following the same procedures regarding patient transfers and are informed of any changes to the transfer policies and procedures.
Response: We agree with the commenters that requiring annual training on transfer protocols is essential. This will promote consistency in transfer processes and ensure that transfers are conducted safely by minimizing errors. We are therefore finalizing that hospitals are required to provide annual training to the relevant staff (as determined by the hospital) regarding the hospital policies and procedures for transferring patients under their care.
Comment: We did not receive any public comments regarding criteria for determining whether a transfer is carried out “promptly and without delay”. One commenter requested interpretive guidance on the term “promptly and without delay”. Another commenter stated that there are circumstances outside of a hospital's control that make it impossible to ensure that a transfer is carried out “promptly and without undue delay”. For example, rural hospitals often have trouble finding other hospitals willing to accept patients. In addition, ambulance availability can delay patients being transferred between facilities. Therefore, this can cause unavoidable delays.
Response: We thank commenters for their feedback We agree that transfers should be carried out as promptly as possible. Although it is not a requirement for the CoP finalized in this rule, CMS expects that hospitals are transferring patients as promptly as possible as staff and resources allow.
Comment: Some commenters expressed support for hospitals creating policies regarding their standards for accepting patients. This would ensure prompt care coordination for transferring patients and emphasize the shared responsibility of both transferring and receiving hospitals to ensure transfers are carried out promptly. However, another commenter expressed concerns that having policies that require hospitals to specify the conditions under which transfers will be accepted or not will diminish the ability of facilities, such as rural hospitals from transferring their patients to the appropriate level of care. For example, if a patient fails to meet all the criteria set forth by the selected recipient facility, that patient would be forced to wait for placement with a hospital whose acceptance criteria permitted transfer; further delaying care. One commenter recommended that CMS provide clarification of the required documentation for policies and procedures related to accepting transfers.
Response: We thank commenters for their feedback. We note that the comments we received may be taken into consideration in potential future rulemaking. To ensure that patients receive the care required for their medical conditions, we encourage hospitals to develop policies and ( printed page 94493) procedures regarding the acceptance of transfers.
Comment: Multiple commenters supported requiring all hospitals to have a documented partnership with another hospital that both provides OB services, as well as has a Medical Fetal Medicine (MFM) specialist available for consultations in urgent situations, if such service(s) are not already offered directly by the hospital. The partnerships would help hospitals build connections and establish relationships between hospitals and support coordinated care handoffs. Furthermore, such partnerships could reduce burdens for hospitals that do not provide obstetrical services. For example, hospitals would be informed of where appropriate specialists are available, therefore ensuring transfers are appropriately carried out. Multiple comments stated that hospitals having access to a robust telehealth OB network could provide real time support for pregnant patients and neonates, facilitate transfers, as well as prevent unnecessary transfers. Other commenters stated that it would be burdensome to establish and maintain these partnerships. A commenter stated that due to the shortage of Maternal Fetal Medicine (MFM) specialists, adding this requirement would exacerbate workforce challenges and increase staff burnout, which could contribute to a reduction in obstetrical services. Another commenter stated that facilities in rural and underserved areas may find it difficult to find and partner with hospitals with MFM specialists due to their limited availability in the area. To mitigate the burden of developing these partnerships, several commenters recommended technical support, funding, interpretive guidance and resources (for example, ACOG's Levels of Maternal Care), to support hospitals' efforts to build relationships within a region.
Response: We thank commenters for their feedback. We note that the comments we received may be taken into consideration in potential future rulemaking. We encourage hospitals to develop collaborative partnerships with other hospitals to improve transitions of care that will support better patient outcomes.
Comment: One commenter recommended that transfer protocols requirement be applied to REHs and CAHs.
Response: REHs provide outpatient services, emergency services, and observation care. They do not provide any acute inpatient hospital services, other than post-hospital extended care services provided in a distinct part unit licensed as a skilled nursing facility (SNF). The transfer protocols in this final rule are inclusive of hospital inpatients (for example, transfers from the emergency department to inpatient admission, transfers between inpatient units in the same hospital, as well as transfers between inpatient units at different hospitals). Furthermore, existing CoPs for CAHs and REHs include requirements related to the transfer of patients in the event that the facility is unable to furnish needed services for a patient or the patient requires a higher level of care. For example, the CAH CoPs at § 485.616(a) require CAHs that are members of a rural health network to have an agreement in place with at least one hospital that is also a member of the network for patient transfer. Additionally, the discharge planning requirements at § 485.642(b) require CAHs to discharge, transfer, or refer the patient, where applicable, with all necessary medical information pertaining to the patient's condition. We require similar actions regarding patient transfers for REHs at § 485.538. Therefore, the transfer protocols would not be necessary for REHs and CAHs, as similar requirements are already in place.
Comment: One commenter recommended that CMS require hospitals to include transfers from community settings to such hospitals in their transfer policies. They stated that, in some cases, community providers have encountered difficulties when transferring patients due to hospitals refusing to accept patients' medical records. Another commenter suggested that CMS considered policies that would facilitate better collaboration between hospitals and birthing facilities. They stated that birthing facilities face challenges when transferring pregnant or postpartum individuals and newborns to hospitals, including limited payment for stabilizing and transferring the patients. Furthermore, this would ensure safe and seamless transition for pregnant and postpartum patients. Another commenter stated that hospitals should focus on referral programs that leverage community resources to offer comprehensive care and address non-medical factors.
Response: While requiring hospitals to include transfers from community settings to the hospital in their transfer protocols is outside the scope of this rule, we encourage hospitals to foster relationships with local organizations in the community, including birthing facilities. By working together, hospitals, community organizations, and birthing facilities can share resources, enhance access to essential services, and improve maternal care.
Comment: One commenter requested that CMS include a list of resources in its interpretive guidance, that facilities can use when developing their transfer protocol policies and procedures. The commenter recommended Society of Maternal Fetal Medicine (SMFM)'s maternal transport briefing form and checklist.
Response: We thank the commenter for their recommendation. We agree there are a variety of resources to support hospitals in developing transfer protocol policies and procedures. For example, accreditation organizations (such as the Joint Commission) and professional organizations (such as the American College of Obstetricians and Gynecologists and Society for Maternal-Fetal Medicine) have such resources available. In addition, CMS will release interpretive guidance following the publication of the final rule.
Comment: A commenter requested that CMS fund statewide transportation programs in rural States where they are not currently available. This would enable patients from rural hospitals to be transported to a facility offering a higher level of care.
Response: We thank the commenter for their recommendation. However, funding for a statewide transportation program is outside the scope of this final rule with comment period.
Comment: A commenter expressed concern that the transfer protocols could overlap or conflict with existing transfer and stabilization policies under EMTALA.
Response: As noted above, EMTALA regulations are separate and distinct from these proposed CoPs. EMTALA (42 CFR 489.24) requires Medicare-participating hospitals with emergency departments to provide individuals presenting to the emergency department with an appropriate medical screening exam and stabilizing treatment if an emergency medical condition is found or under certain circumstances, appropriately transfer such individuals to receive stabilizing care. However, EMTALA does not address the transfer of hospital inpatients to a higher level of care. The proposed CoP require hospitals to develop policies and procedures for transferring patients under their care. The transfer protocols are inclusive of hospital inpatients (for example, transfers from the emergency department to inpatient ward, transfers between inpatient units in the same hospital, as well as transfers between inpatient units at different hospitals). ( printed page 94494)
Final Rule Action: After consideration of the comments, we are finalizing our proposal for transfer protocols with the following modification: Acute care hospitals are required to provide annual training to the relevant staff (as determined by the facility) regarding the hospital policies and procedures for transferring patients under its care. These requirements do not apply to CAHs and REHs.
7. Implementation Timeframe
In the CY 2025 OPPS/ASC proposed rule, we did not propose to delay the implementation of the new and revised CoP requirements. However, we received many comments and recommendations with respect to the effective date and the date of implementation of the requirements in hospitals and CAHs.
Comment: Given that the final requirements will be effective 60 days after publication of the final rule, many commenters expressed concerns that this period would not allow facilities enough time to meet the new and revised CoP requirements. Some commenters stated that many of the provisions would require possible extensive changes (for example, hiring additional staff, and purchasing additional equipment and supplies) to ensure full compliance. Commenters also expressed concern that requiring hospitals, especially small, rural hospitals, to comply with these requirements will result in unintended consequences, such as the closure of labor and delivery units. This would only exacerbate the existing maternal health care access challenges.
Some commenters recommended a period of discretionary enforcement to allow hospitals time to come into compliance. One commenter recommended postponing the implementation of the new CoPs until CMS had a better understanding of the potential impact of the proposed policies on providers and organizations. While some commenters recommended that we delay implementation until interpretive guidance was available to facilities, other commenters recommended that we delay implementation of the requirements for CAHs and other facilities with low-birth volumes.
Some commenters suggested that we implement a phase-in timeframe for the requirements as to not impose burden on the facilities or obstetrical workforce. However, various implementation timeframes were suggested. A few commenters suggested that we implement a phase-in timeframe of 1 year to 2 years. A couple of commenters recommended a phase-in timeframe of 4 years. Other commenters suggested that we implement different phase-in timeframes for hospitals and CAHs.
Response: We believe the new and revised OB services CoPs will help ensure that all pregnant, birthing, and postpartum women receive consistent, high quality maternal health care, regardless of their geographic location. However, we acknowledge that these proposed requirements would require a robust review of existing programs and would necessitate changes to ensure full compliance with the new and revised obstetrical CoP requirements. To reduce some of the burden of complying with these requirements, and to avoid any unintended consequences, we are finalizing an implementation approach in phases, that allows additional time for hospitals and CAHs to comply with the requirements of this rule.
Final Rule Action: After consideration of the public comments, we are finalizing a phased-in implementation for hospitals and CAHs in 3 phases, over a 2-year period. Specifically,
- Phase 1 would require facilities to comply with the following requirements 6 months following the effective date of the final rule:
○ Emergency services readiness for hospitals (§ 482.55) and CAHs (§ 485.618)
○ Transfer protocols for hospitals only (§ 482.43)
- Phase 2 would require facilities to comply with the following requirements 1 year following the effective date of the final rule:
○ Organization, staffing, and delivery of services for hospitals ((§ 482.59(a) and (b)) and CAHs (§ 485. 649(a) and (b))
- Phase 3 would require facilities to comply with the following requirements 2 years following the effective date of the final rule:
○ OB staff training in hospitals (§ 482.59(c)) and CAHs (§ 485.649(c))
○ QAPI program for OB services in hospitals (§ 482.21) and CAHs (§ 485.641)
We note that the final regulations are effective 60 days following publication of this final rule with comment period in the Federal Register . The implementation date for the specific requirements is listed in Table 174.
( printed page 94495)XXII. Modification to the Hybrid Hospital-Wide All-Cause Readmission and Hybrid Hospital-Wide All-Cause Risk Standardized Mortality Measures in the Hospital Inpatient Quality Reporting Program
A. Background
We refer readers to the following final rules for detailed discussions of the history of the Hospital Inpatient Quality Reporting (IQR) Program, including statutory history, and for the measures we have previously adopted for the Hospital IQR Program measure set:
- The FY 2010 Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long-Term Care Hospital Prospective Payment System (IPPS/LTCH PPS) final rule (74 FR 43860 through 43861);
- The FY 2011 IPPS/LTCH PPS final rule (75 FR 50180 through 50181);
- The FY 2012 IPPS/LTCH PPS final rule (76 FR 51605 through 61653);
- The FY 2013 IPPS/LTCH PPS final rule (77 FR 53503 through 53555);
- The FY 2014 IPPS/LTCH PPS final rule (78 FR 50775 through 50837);
- The FY 2015 IPPS/LTCH PPS final rule (79 FR 50217 through 50249);
- The FY 2016 IPPS/LTCH PPS final rule (80 FR 49660 through 49692);
- The FY 2017 IPPS/LTCH PPS final rule (81 FR 57148 through 57150);
- The FY 2018 IPPS/LTCH PPS final rule (82 FR 38326 through 38328 and 38348);
- The FY 2019 IPPS/LTCH PPS final rule (83 FR 41538 through 41609);
- The FY 2020 IPPS/LTCH PPS final rule (84 FR 42448 through 42509);
- The FY 2021 IPPS/LTCH PPS final rule (85 FR 58926 through 58959);
- The FY 2022 IPPS/LTCH PPS final rule (86 FR 45360 through 45426);
- The FY 2023 IPPS/LTCH PPS final rule (87 FR 49190 through 49310);
- The FY 2024 IPPS/LTCH PPS final rule (88 FR 59144 through 59203); and
- The FY 2025 IPPS/LTCH PPS final rule published on August 28, 2024 (89 FR 69515 through 69577).
We also refer readers to 42 CFR 412.140 for Hospital IQR Program regulations.
B. Updates to the Form, Time, and Manner Requirements for the Hybrid Hospital-Wide All-Cause Readmission (HWR) and Hybrid Hospital-Wide All-Cause Risk Standardized Mortality (HWM) Measures for the FY 2026 and the FY 2027 Payment Determinations
1. Background of the Hybrid HWR and Hybrid HWM Measures in the Hospital IQR Program
The Hospital IQR Program previously adopted two hybrid measures: (1) the Hybrid Hospital-Wide Readmission (HWR) measure; and (2) the Hybrid Hospital-Wide All-Cause Risk Standardized Mortality (HWM) measure. Hybrid measures use more than one data source for measure calculation; specifically for the Hybrid HWR and Hybrid HWM measures, they use core clinical data elements (CCDEs), linking variables, and claims data (80 FR 49698). CCDEs are a set of clinical variables derived from electronic health records (EHRs) that can be used to risk adjust hospital outcome measures (80 FR 49699). Linking variables are administrative data that can be used to link or merge the CCDEs and administrative claims data for measure calculation (80 FR 49703). These measures are designed to enhance risk adjustment of administrative claims-based outcome measures by utilizing patient clinical data captured in EHRs (80 FR 49698).
We initially solicited public comment on the potential future adoption of hybrid measures into the Hospital IQR Program in the FY 2016 IPPS/LTCH PPS final rule adoption (80 FR 49698 through 49704). In subsequent years, we adopted both the Hybrid HWR measure and the Hybrid HWM measure with initial voluntary reporting periods. A discussion of the measure history for both measures follows.
The Hybrid HWR was the first hybrid measure introduced into the Hospital IQR Program. The Hybrid HWR measure is designed to capture all unplanned readmissions that arise from acute clinical events requiring urgent rehospitalization within 30 days of discharge. The measure was adopted in a stepwise fashion starting with voluntary reporting periods. In the FY 2018 IPPS/LTCH PPS final rule, we finalized 6 months of voluntary reporting for the CY 2018 reporting period (82 FR 38350 through 38355). In the FY 2020 IPPS/LTCH PPS final rule, we finalized 2 additional years of voluntary reporting, followed by mandatory reporting impacting the FY 2026 payment determination (84 FR 42465 through 42479). Then, in the FY 2024 IPPS/LTCH PPS final rule, we modified the Hybrid HWR measure cohort to include both Medicare fee-for-service (FFS) patients and Medicare Advantage (MA) patients 65 years and older for the FY 2027 payment determination and for subsequent years (88 FR 59161 through 59168).
The Hybrid HWM measure was the second hybrid measure to be adopted into the Hospital IQR Program. The Hybrid HWM measure is an outcome measure that captures the hospital-level, risk-standardized mortality rate (RSMR) of unplanned, all-cause mortality within 30 days of hospital admission for any eligible condition. Like the Hybrid HWR measure, we adopted the Hybrid HWM measure in a stepwise fashion, starting with a period of voluntary reporting. We initially adopted the Hybrid HWM measure in the FY 2022 IPPS/LTCH PPS final rule and finalized one voluntary reporting period followed by mandatory reporting impacting the FY 2026 payment determination (86 FR 45365 through 45374). Then in the FY 2024 IPPS/LTCH PPS final rule, we modified the Hybrid HWM measure cohort to include both Medicare fee-for-service (FFS) patients and Medicare Advantage (MA) patients 65 to 94 years old for the FY 2027 payment determination and for subsequent years (88 FR 59161 through 59168).
As previously noted, these hybrid measures use data from claims in combination with CCDEs and linking variables pulled from hospital EHRs. We previously finalized that hospitals must submit linking variables on 95 percent of hospital discharges (84 FR 42470; 86 FR 45371). The previously finalized linking variables are: (1) CMS Certification Number; (2) Health and Insurance Claims Number or Medicare Beneficiary Identifier; (3) Date of Birth; (4) Sex; (5) Admission date; and (6) Discharge date (84 FR 42469; 86 FR 45371). The previously finalized CCDEs are vital signs and laboratory results (84 FR 42469; 86 FR 45371). We also previously finalized that hospitals would be required to report CCDEs on 90 percent of discharges in a given reporting period (84 FR 42469; 86 FR 45371). These submission requirements were finalized beginning with mandatory reporting for the FY 2026 payment determination (84 FR 42469 through 42470; 86 FR 45371). We refer readers to the FY 2020 IPPS/LTCH PPS final rule (84 FR 42465 through 42479) and FY 2022 IPPS/LTCH PPS final rule (86 FR 45365 through 45374) for more information regarding data sources, measure calculation, and risk adjustment requirements. We also refer readers to the FY 2020 IPPS/LTCH PPS final rule (84 FR 42506 through 42508), the FY 2021 IPPS/LTCH PPS final rule (85 FR 58941), the CY 2021 PFS final rule (85 FR 84472), and the FY 2022 IPPS/LTCH PPS final rule (86 FR 45421) for our previously adopted policies regarding certification, file format, and data submission requirements for hybrid measures in the Hospital IQR Program. For both hybrid measures, we previously finalized that hospitals must submit data for the mandatory reporting period impacting the FY 2026 payment ( printed page 94496) determination, based on performance data from July 1, 2023, through June 30, 2024, by October 1, 2024 (86 FR 45370). We finalized that data collected during the voluntary reporting periods would not be publicly reported (84 FR 42470; 86 FR 45371). We also finalized that we would begin public reporting of both hybrid measures' results, beginning with data collected from the July 1, 2023, through June 30, 2024, reporting period, impacting the FY 2026 payment determination (84 FR 42470 through 42471; 86 FR 45371).
2. Extension of Voluntary Reporting of CCDE and Linking Variable Data for the Hybrid HWR and Hybrid HWM Measures
Based on hospital performance during the most recent voluntary reporting period, it appeared that hospitals are unprepared for mandatory reporting of the Hybrid HWR and Hybrid HWM measures. As a part of measure maintenance, we routinely monitor hospital performance on the Hospital IQR Program's measures. We have been closely monitoring the results of voluntary reporting for both hybrid measures, including most recently the results of the second voluntary period for Hybrid HWR and the first voluntary period for Hybrid HWM. During these periods, approximately one-third of IPPS hospitals participated. The data indicated that three-fourths of the participating hospitals would not have met the reporting thresholds for the CCDEs and linking variables if the reporting requirement had been mandatory, and accordingly, would have been subject to a one quarter reduction to their annual payment update under the Hospital IQR Program for the given fiscal year.
The hospitals that participated in the voluntary reporting were mostly large, non-rural, non-critical access, and non-safety net. Based on our experience with implementing new types of digital measures, we understand that small and rural hospitals, as well as hospitals with fewer financial resources, may need additional time and flexibility to successfully implement new measure reporting requirements relative to larger, non-rural hospitals (82 FR 38357). We therefore believe the reporting failure rate may have been even higher if all IPPS hospitals participated.
In addition, we received feedback from hospitals (via email and help desk questions) raising various issues with reporting including issues related to CCDE collection timing and clinical workflow, issues with the types of units required for CCDE values, and achievability of the data submission requirement thresholds. We continued to investigate whether any of these issues or any other issues may be making it difficult for hospitals to meet CCDE and linking variable thresholds (that is, of 90 percent and 95 percent, respectively, of hospital discharges) and continued to analyze the root cause of these challenges.
We appreciate that, in light of the information discussed above, hospitals participating in the Hospital IQR Program may need additional time to remediate the issues and develop experience with reporting of CCDEs and linking variables before being subject to the associated program payment adjustments for noncompliance. In the CY 2025 OPPS/ASC proposed rule, we therefore proposed that for the FY 2026 payment determination (based on performance data from July 1, 2023, through June 30, 2024), the submission of CCDEs and linking variables would remain voluntary (89 FR 59502). We proposed that for the FY 2027 payment determination and subsequent years, the submission of CCDEs and linking variables would then become mandatory.
We explained in the CY 2025 OPPS/ASC proposed rule, that under our proposal, a hospital's annual payment determination for FY 2026 would not be affected by the voluntary reporting of CCDEs and linking variables, although we would still evaluate and assess the claims data portion of these measures (89 FR 59502). We proposed that the Hybrid HWR and Hybrid HWM measures would be publicly reported based on claims data for the FY 2026 Hospital IQR Program. We explained that the proposal would allow the Hospital IQR Program to publicly display hospital information on these important clinical areas and provide patients with visibility into hospital performance, while providing hospitals with more time to improve reporting on CCDEs and linking variables. Under the proposal, hospitals would continue to receive confidential hospital-specific reports in the Spring as a preview of public reporting. We noted that the hospital-specific reports would reflect the CCDEs and linking variables, should hospitals choose to submit them. We also stated in the proposed rule that we would continue to evaluate potential changes to the reporting requirements related to CCDEs and linking variables.
We invited public comment on our proposal.
Comment: Many commenters supported the proposal to extend voluntary reporting of the CCDEs and linking variables for the Hybrid HWR and Hybrid HWM measures, for the performance period of July 1, 2023, through June 30, 2024, impacting the FY 2026 payment determination for the Hospital IQR Program due to reporting challenges.
Response: We thank the commenters for their support.
Comment: Some commenters supported the proposed extended voluntary reporting for the performance period of July 1, 2023, through June 30, 2024, impacting the FY 2026 payment determination, but requested that voluntary reporting be extended further for both measures. The commenters' reasons for requesting additional years of voluntary reporting were due to challenges they identified with the current measure reporting requirements. Some of the challenges identified were:
- Many commenters noted issues specifically with capturing the weight value within the 24-hour timeframe as specified by the measure. Commenters noted challenges with this timeframe due to changes in patient status from observation to inpatient, data inconsistencies with patient transfers between facilities, labs being collected during pre-op appointments prior to the 24-hour timeframe for surgical patients, and prolonged emergency department stays. A few commenters noted while CMS has approved further revisions to the measure logic including updates to the timing of taking a patient's weight, these updates do not take effect until reporting that impacts the FY 2027 payment determination due to the existing eCQM Annual Update process. These commenters therefore noted that voluntary reporting should be extended until the FY 2027 payment determination when changes to the timing requirements for capturing the weight value will be implemented;
- Many commenters also noted challenges with the reporting of some of the CCDE required laboratory results. These challenges include difficulties related to the timing of when labs were drawn, including labs drawn outside of the 24-hour period currently specified by the measures, noting that this 24-hour requirement also encourages unnecessary duplication of lab tests. Many commenters also stated that some of the required laboratory results appear as “missing” because they were unnecessary to draw for the patient;
- Many commenters expressed concern regarding the lack of a standard unit of measurement for required CCDE data elements, specifically platelet values. A few commenters noted that units of measurement commonly used for platelets were counted as “missing” in their performance reports. A few commenters suggested additional ( printed page 94497) guidance is needed on whether the result for a non-standard unit can be converted;
- A few commenters stated that they received data submission errors related to linking variables, such as sex and date of birth;
- A few commenters stated that they need additional time to develop processes to improve collection of Health Insurance Claims Number or Medicare Beneficiary Identifier (HICN/MBI) for Medicare Advantage patients, which is one of the required linking variables for both measures;
- Several commenters representing specialty hospitals requested that the measures be refined to include a minimum relevant population threshold exemption for other patient populations such as surgical, hospice, psychiatric, and orthopedic patients; and
- Many commenters expressed the need for a longer voluntary reporting period to address clinical workflow and improve data collection processes by working with their hospitals' vendors before requiring reporting on all elements of these hybrid measures. A few commenters noted that small and rural hospitals require more time and flexibility to successfully implement new measure reporting requirements. Another commenter stated that hospitals need time to adjust time and date of admission status, as they found issues with misalignment between the CMS claims data and their EHR vendor. A few commenters expressed that variations in EHR systems present challenges in meeting measure thresholds. A commenter stated that these hybrid measures have been especially challenging during times of limited resources following the COVID-19 public health emergency.
Response: We thank the commenters for their feedback regarding the need for additional time before mandatory reporting of CCDEs and linking variables. Based on this feedback, in addition to finalizing our proposal to extend voluntary reporting of the CCDEs and linking variables for the Hybrid HWR and Hybrid HWM measures, for the performance period of July 1, 2023, through June 30, 2024, impacting the FY 2026 payment determination for the Hospital IQR Program, we will also finalize an additional year of voluntary reporting of the CCDEs and linking variables for these measures, for the performance period of July 1, 2024, through June 30, 2025, impacting the FY 2027 payment determination.
Specific to comments about the current timing requirements for CCDEs, in the eCQM Annual Update that will be posted in Spring 2025, we will extend the anchor timestamp requirement for CCDEs from the first CCDE beginning 24 hours before to 24 hours after the start of the inpatient admission for laboratory results and 24 hours before to 2 hours after the start of the inpatient admission for vital signs (except weight), to the first CCDE resulted after the start of the hospital encounter. This change may increase hospital success in reaching Hospital IQR Program threshold requirements, while maintaining the intent of the measure to collect the earliest laboratory test results and vital signs upon hospital arrival. The Spring 2025 Annual Update impacts the July 1, 2026, through June 30, 2027, performance period which is associated with the FY 2029 payment determination.
Specific to commenter concerns that platelets counted as “missing” in performance reports, we note that beginning with July 1, 2023, through June 20, 2024, performance period data, which is associated with the FY 2026 payment determination, platelet laboratory test values with the unit of Femtoliter (fL) will be accepted. The current specifications request hospitals to submit CCDE in Unified Code for Units of Measure (UCUM) units. To reduce hospital burden, any unit may be submitted by hospitals, as we convert to standard units during data cleaning. When the reported unit is not able to be converted to the requested UCUM units, the value will be set to missing and the median value reported for that CCDE will be imputed. In analyzing the data from the voluntary periods, we observed a large proportion of platelet values that were marked as missing. Within the platelet values reported as missing, a large portion of submitted values were unusable in measure calculation due to a unit, Femtoliter (fL), that cannot be converted to the standard unit (10*3/uL) without additional lab values from hospitals. This means that reported fL values were set to missing and the submission did not count towards hospitals' 90 percent reporting threshold for CCDEs. In reviewing these results, we confirmed that we are able to convert the values and/or apply a data imputation method to utilize values for platelets using data that are submitted in Femtoliters.
Regarding concerns about submitting sex and date of birth linking variables, we note that those linking variables are no longer required for submission.
Regarding the request for a population threshold exemption, in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49304), we stated that we do not believe that threshold exemptions are necessary for hybrid measures because the measures utilize the Initial Patient Population (IPP) and CMS confirms the measure cohort to determine whether a hospital has met the denominator criteria. However, we will continue to review hospital performance and consider ways to address the needs of specialty hospitals with a small number of relevant patients.
Comment: Many commenters described challenges meeting the 90 percent thresholds for CCDEs and the 95 percent thresholds for linking variables, and recommended reducing the required percentages. A few commenters recommended lowering the threshold of reporting laboratory results (included in the CCDEs) to no more than 80 or 85 percent for the first mandatory reporting period.
Response: We understand that some hospitals experienced difficulties with the submission requirements for CCDEs and linking variables for these measures during the voluntary reporting periods. We thank commenters for providing details of those challenges. We will continue to monitor hospital performance of these measures and intend to propose to lower the reporting thresholds in the upcoming FY 2026 IPPS/LTCH PPS proposed rule.
Comment: One commenter recommended that CMS create a specific hybrid measure Extraordinary Circumstances Exception (ECE) consideration based on the use of Certified Electronic Health Record Technology (CEHRT). The commenter stated that electronic Clinical Quality Measures (eCQMs) and hybrid measures are the only two collection types in the Hospital IQR Program that require the use of CEHRT and that CMS has specific ECE considerations for eCQMs, including issues related to the use of CEHRT. The commenter stated that CMS should have ECE considerations in the same manner as eCQMs when it comes to the use of CEHRT.
Response: We thank the commenter for this suggestion and will take it under consideration.
Comment: A few commenters requested additional clarity within measure specification documents. These commenters stated that the definitions in the specifications are not clear, that the specifications do not clearly describe the percentage thresholds required for measure submission, and that the measure specifications do not clearly describe how the measure results will be calculated. A few commenters requested clarification within the specifications on the surgical specialty cohort of the Hybrid HWR measure and the surgical divisions of the Hybrid HWM measure and sought additional ( printed page 94498) information regarding what group of surgical patients might be excluded.
Response: We thank the commenters for identifying topics within the measure specifications that could be clearer or where additional guidance would be useful. Regarding concerns that the specifications do not describe the percentage thresholds, we note that we do not typically include programmatic form, time, and manner requirements in the measure specifications.
Regarding measure calculation, for the Hybrid HWR measure, index admissions are assigned to one of five mutually exclusive specialty cohort groups consisting of related conditions or procedures. For each specialty cohort group, the standardized readmission ratio (SRR) is calculated as the ratio of the number of “predicted” readmissions to the number of “expected” readmissions at a given hospital. This approach is analogous to a ratio of “observed” to “expected” used in other types of statistical analyses. The specialty cohort SRRs are then pooled for each hospital using a volume-weighted geometric mean to create a hospital-wide combined SRR. The combined SRR is multiplied by the national observed readmission rate to produce the RSRR. Similarly, for the Hybrid HWM measure, index admissions are assigned to one of 15 mutually exclusive service-line divisions consisting of related conditions or procedures. For each service-line division, the standardized mortality ratio (SMR) is calculated as the ratio of the number of “predicted” deaths to the number of “expected” deaths at a given hospital. This approach is analogous to a ratio of “observed” to “expected” used in other types of statistical analyses. A hospital-wide composite SMR is then created by pooling the service-line SMRs for each hospital using an inverse variance-weighted mean. To produce the risk-standardized mortality rate (RSMR), the composite SMR is multiplied by the national observed mortality rate.
Additional information regarding measure calculation, including the surgical cohort and surgical divisions, is available in the measures' methodology reports available at https://www.cms.gov/medicare/quality/initiatives/hospital-quality-initiative/measure-methodology. We also note that although data submission requirements may differ by cohort or division ( i.e., hospitals must submit laboratory test results for at least 90 percent of discharges in non-surgical patients, meaning those not assigned to the surgical specialty cohort of the Hybrid HWR measure or the surgical divisions of the Hybrid HWM measure), no specialty cohort is excluded from the measure. We note that any updated guidance, or any future updates to measure specifications, will be made available at: https://qualitynet.cms.gov/inpatient/measures/hybrid/methodology.
Comment: Many commenters stated that they experienced issues with the quality and timing of the feedback reports provided by CMS. Many commenters expressed concern regarding the timing in receiving Hospital-Specific Reports (HSRs), stating that it prevented hospitals from being able to make improvements and correct data before the submission period closes. A few commenters stated that the HSRs were difficult to interpret. Several commenters expressed concern that CCDE and linking variable requirement threshold results reported in the voluntary reporting HSRs did not accurately reflect the true results due to calculations including surgical cohort patients, Medicare fee-for-service beneficiaries under the age of 65, and Medicare Advantage beneficiaries. One commenter noted that because the linking variables, vital signs, and laboratory test results are summated for all patients included in the initial population of the measure, patients that are excluded from the measure calculation are still being included in the summary Mandatory Reporting Data Submission Requirements table. The commenter stated that this makes it difficult to calculate whether their facility is meeting submission threshold requirements, as it is difficult to determine the specialty cohort the patient is in when they are excluded.
Response: We thank the commenters for sharing their experiences during the most recent voluntary reporting period and appreciate the helpful feedback and suggestions regarding ways to improve the feedback process for reporting of the Hybrid HWM and Hybrid HWR measures. Regarding timing of HSRs, Hospital IQR Program HSRs are usually released in the spring, approximately 8 months after the end of the hybrid data submission deadline. This reflects the length of time needed for the production process ( i.e., extracting and processing the data, updating the calculation code, running results, and updating and populating HSRs). We recognize that this year, the HSRs were not delivered until June 28, 2024, which was later than intended. We plan to release the HSRs in the spring next year. Regarding interpretation of the reports, we provided a HSR User Guide for the 2024 Voluntary Reporting to assist hospitals with interpreting the confidential HSR reports, available at: https://qualitynet.cms.gov/inpatient/measures/hybrid/reports , and the QualityNet Q&A tool [710] is also available for assistance if there are questions in interpreting HSRs.
Regarding concerns that patients that are excluded from the measure calculation were included in the voluntary reporting HSRs, Medicare FFS beneficiaries under the age of 65 were erroneously included in the threshold calculations in the 2023 voluntary reporting HSRs, but the 2024 voluntary reporting HSRs appropriately limited the age ranges. In the 2024 voluntary reporting HSRs, surgical cohort patients were erroneously included in the laboratory values thresholds, which should be based on the non-surgical patients only. The HSRs provided information that allowed hospitals to filter and remove these patients to derive their true laboratory values threshold result.
Regarding commenter concerns that the calculation of the inpatient population was incorrect due to inclusion of Medicare Advantage beneficiaries in the measure cohort, we are not aware of any hybrid HSRs including Medicare Advantage beneficiaries or data. However, we note that Medicare Advantage beneficiaries will be included in calculations for measure cohorts with the discharge data from July 1, 2024, through June 30, 2025 (88 FR 59161 through 59165 and 88 FR 59165 through 59168). We will review the commenter's concerns and review the initial patient population included in the next HSR release, which will include Medicare Advantage patients, for any issues.
Comment: Commenters provided many suggestions to improve the submission process. A few commenters recommended having a test submission process like that of eCQMs. One commenter recommended continuous monitoring and a formal feedback mechanism for ongoing improvement. A few commenters recommended CMS provide reports to support validation at the time of submission. Several commenters recommended providing technical assistance and resources to assist hospitals in meeting the thresholds. One commenter recommended best practice sharing and targeted support for small and rural hospitals. One commenter stated the style sheets used to evaluate the Quality Reporting Document Architecture (QRDA) I reports needed to be updated. ( printed page 94499) Additionally, a few commenters noted delayed or missing responses from CMS help desk and JIRA tickets.
Response: Regarding a test submission process, we note that the annual hybrid measures submission period is similar to the annual eCQM submission periods. Once we announce the HQR system is open to accept hybrid measure data, hospitals and/or vendors can test their submission files as often as needed and until the deadline. Once hospitals and/or vendors have validated their files are properly formatted and errors corrected, they can submit their files to “production.” Both “test” and “production” file submissions may be submitted once the portal opens through the submission deadline. We note that CMS does not maintain a QRDA style sheet; however, the HL7 QRDA I and III standards zip files direct users to use the Clinical Document Architecture stylesheet available at: https://hl7.org/permalink/?CDAStyleSheet.
We wish to note there are several additional ways for hospitals and EHR vendors to receive technical assistance to support implementation of these measures, which include CCSQ Support Central [711] and ONC JIRA.[712] Additional resources about the hybrid measures, including fact sheets, frequently asked questions, and webinar recordings, are available on our QualityNet website at: https://qualitynet.cms.gov/inpatient/measures/hybrid/resources. We will continue to identify opportunities to improve our responsiveness and quality of technical assistance.
Comment: One commenter did not support the proposal to delay mandatory reporting of the CCDEs and linking variables for the Hybrid HWR and Hybrid HWM measures for a year from the previously finalized FY 2026 payment determination. The commenter stated that while they appreciated CMS' analyses from recent voluntary reporting that 75 percent or more of hospitals are projected to not meet the reporting thresholds for key hybrid data elements, the commenter stated that CMS should not allow hospitals to delay mandatory reporting in these important measures due to the hospitals' own actions or inactions. The commenter stated that a reduction of annual payment update is a fair consequence given hospitals have had ample notification of CMS' plans for these measures. The commenter stated that allowing such delays sets a troubling precedent, as it implies that hospitals' actions or inactions during the voluntary reporting period can influence and potentially disrupt the established timeframes set by rulemaking.
Response: We thank the commenter and agree that these measures are important. Hybrid measures are a new mode of data collection for quality measures. They are unique compared to other quality measures in that they use more than one source of data and calculation requires that hospitals submit administrative data elements along with the CCDEs extracted from hospital EHRs. We understand that hospitals have faced a myriad of challenges with this new form of submission, as discussed in the comment summaries, and need additional time to successfully adapt to this method of reporting. Additionally, we will benefit from additional time to analyze the issues hospitals are facing to determine whether further refinements to the measures are warranted to enable reasonable levels of successful reporting for hospitals.
After consideration of the public comments we received, we are finalizing our proposal to continue voluntary reporting of the CCDEs and linking variables for both the Hybrid HWR and Hybrid HWM measures, for the performance period of July 1, 2023, through June 30, 2024, impacting the FY 2026 payment determination for the Hospital IQR Program. We are also finalizing our proposal that the Hybrid HWR and Hybrid HWM measures will be publicly reported based on claims data for the performance period of July 1, 2023, through June 30, 2024, impacting the FY 2026 payment determination.
Additionally, based on public comments regarding the need for additional time, we are further extending voluntary reporting of CCDEs and linking variables for the performance period of July 1, 2024, through June 30, 2025, impacting the FY 2027 payment determination for the Hospital IQR Program, for both the Hybrid HWR and Hybrid HWM measures. The Hybrid HWR and Hybrid HWM measures will be publicly reported using claims data for the performance period of July 1, 2024, through June 30, 2025, impacting the FY 2027 payment determination. We intend to make additional changes to the measures, including lowering the reporting thresholds, and will propose any substantive changes in future rulemaking.
XXIII. Individuals Currently or Formerly in the Custody of Penal Authorities
A. Medicare FFS No Legal Obligation To Pay Payment Exclusion and Incarceration (Revisions to 42 CFR 411.4)
1. Background
Section 1862(a)(2) of the Act prohibits Medicare payment under Part A or Part B for any expenses incurred for items or services for which the individual furnished such items or services has no legal obligation to pay, and which no other person (by reason of such individual's membership in a prepayment plan or otherwise) has a legal obligation to provide or pay for, except in the case of Federally qualified health center services. We refer to this payment exclusion as the “no legal obligation to pay” payment exclusion. The no legal obligation to pay payment exclusion is codified in regulation at § 411.4. The regulatory exclusion includes a general rule at § 411.4(a) that applies to all services (except as provided in § 411.8(b), which applies to services paid for by a government entity) and a special condition at § 411.4(b) for services furnished to individuals in custody of penal authorities.
In the 1989 final rule establishing the special condition at § 411.4(b) for services furnished to individuals in custody of penal authorities, we explained that the purpose of § 411.4(b) is to clarify how the no legal obligation to pay payment exclusion applies to services furnished to prisoners (see 54 FR 41716, 41723 (Oct. 11, 1989)) (“1989 final rule”). We explained that prisoners generally have the status of public charges and, as such, have no obligation to pay for the medical care they receive. Consequently, under the statutory no legal obligation to pay payment exclusion at section 1862(a)(2) of the Act, Medicare is prohibited for paying for such care. We noted, however, that in certain circumstances prisoners could have a legal obligation to pay for health care items or services they receive, and in such circumstances, Medicare may pay for the items or services.
As finalized in the 1989 final rule, the special rule at § 411.4(b) specifies the conditions that must be satisfied to establish that a prisoner has a legal obligation to pay for health care items or services and, thus, that Medicare may pay for such items or services. Specifically, § 411.4(b) provides that Medicare may pay for services furnished to individuals in the custody of police or other penal authorities or in the ( printed page 94500) custody of a government agency under a penal statute only if the following conditions are met: (1) State or local law must require individuals in custody to repay the cost of the medical services they receive while in custody; and (2) the State or local government must enforce the requirement to pay by billing all such individuals, whether or not covered by Medicare or any other health insurance, and by pursuing collection of the amounts they owe in the same way and with the same vigor that it pursues the collection of other debts.
In 2007, we added a description of “custody” to the special condition at § 411.4(b) for services furnished to individuals in the custody of penal authorities (see 72 FR 47130, 47405 through 47406 (Aug. 22, 2007)) (“2007 final rule”). We noted that CMS would not defer to a particular State or local government's definition or interpretation of what constitutes “custody.” Instead, we adopted a definition of “custody” that is consistent with how the term has been defined by Federal courts for purposes of the habeas corpus protections of the U.S. Constitution. As finalized, § 411.4(b) provides that individuals who are in custody include, but are not limited to, individuals who are under arrest, incarcerated, imprisoned, escaped from confinement, under supervised release, on medical furlough, required to reside in mental health facilities, required to reside in halfway houses, required to live under home detention, or confined completely or partially in any way under a penal statute or rule. We explained that, under this description, individuals who are on parole, probation, bail, or supervised release may be in custody for purposes of the payment exclusion. We also stressed that individuals who are under supervised release for the purpose of receiving medical services on so-called “medical furlough,” and who are required to return to a State or local government facility after the medical services are furnished, are considered to be in custody for purposes of the no legal obligation to pay payment exclusion.
In the 2007 final rule, we responded to several commenters who objected to the breadth of the description of custody that we adopted. The commenters maintained that the policy would place an unreasonable burden on hospitals, because hospitals often have no means of identifying whether an individual is in custody for purposes of § 411.4(b) if the individual is not physically confined in a correctional facility or brought to the hospital by government authorities. Another commenter added that a hospital has no way of knowing whether an individual who is in custody for purposes of § 411.4(b) has a legal obligation to pay for his or her medical care. In response, we stated that hospitals are not required to seek criminal histories or do background checks on all patients being registered. We explained that, if Medicare denies payment because the individual receiving care is in custody of penal authorities, the provider or supplier will be directed to seek payment from the State or local government that has custody of the individual. We concluded that, if the State or local government believes in such circumstances that it is not responsible for the care provided to the individual, it should be prepared to prove to Medicare either that the individual would not be considered to be in custody under Federal habeas corpus law or that the State or local government has no legal obligation to pay for the services because the conditions in § 411.4(b)(1) and (2) are satisfied.
There have been no further revisions to § 411.4 since it was revised in 2007.
2. Summary of the Proposed Provisions
The special condition at § 411.4(b) for services furnished to individuals in custody of penal authorities operates as a rebuttable presumption. The presumption is that individuals who are in custody, as the term is described in § 411.4(b), have no legal obligation to pay for health care items or services they receive while in custody; therefore, Medicare is prohibited from paying for such health care items or services under the statutory no legal obligation to pay payment exclusion. The presumption can be rebutted by a showing that: (1) the State or local government requires individuals in custody to repay the cost of the medical services they receive while in custody; and (2) the State or local government enforces the requirement to pay by billing all such individuals, whether or not covered by Medicare or any other health insurance, and by pursuing collection of the amounts they owe in the same way and with the same vigor that it pursues the collection of other debts.
In the CY 2025 OPPS/ASC proposed rule, we proposed to narrow the description of “custody” in § 411.4(b), because, as explained in greater detail below, we no longer believe that certain classes of individuals should be presumed to be in custody for purposes of the no legal obligation to pay payment exclusion. We also proposed to consider certain individuals who are required to reside in halfway houses not to be in custody for purposes of the no legal obligation to pay payment exclusion under specified conditions.
We proposed to redesignate the description of “custody” as § 411.4(b)(3), to remove individuals who are on supervised release and home detention from the current description of “custody,” and to strike the phrase “completely or partially in any way under a penal statute or rule.” Under the proposal, individuals who have been lawfully released from confinement in jail, prison, penitentiary, or similar institution, or released following arrest (that is, the individuals are no longer physically detained by law enforcement or penal authorities) on bail, parole, probation, or home detention would not be presumed to be in custody for purposes of the no legal obligation to pay payment exclusion, even if such individuals have been conditionally released and may be required to return to jail, prison, penitentiary, or similar institution at some later time (for example, due to conviction or failure to satisfy the conditions of their supervised release). However, individuals who are on “medical furlough” or similar arrangements (that is, the individuals are under the control of law enforcement or penal authorities and required to return to jail or prison after medical services have been provided) would still be considered in custody for purposes of § 411.4(b)(3). Given the differences in terminology used by various Federal, State, and local government penal authorities to refer to different levels of control and confinement in the criminal justice system, we invited comments on the appropriateness of the terminology in the existing description of “custody” in our regulations and whether additional or different terminology should be incorporated in the description of “custody” at § 411.4(b)(3). We also invited comment on whether we should explicitly state in regulatory text that individuals on bail, parole, probation, or home confinement are not considered to be in custody for purposes of proposed § 411.4(b).
In the CY 2025 OPPS/ASC proposed rule, we stated that we were proposing to narrow the description of custody in § 411.4(b)(3) for several reasons. First, we believe that individuals who have been released from jail or prison on bail, parole, probation, or home detention, or who have been sentenced to probation or home detention in lieu of incarceration, typically have a legal obligation to pay for the health care items or services they receive. We do not believe that such individuals have ( printed page 94501) the status of public charges, and we do not believe that Federal, State, or local government law enforcement or penal authorities are typically responsible for providing for the health care of such individuals. We stated that we, therefore, no longer believe that such individuals (and the providers and suppliers who furnish services to them) should have the burden to prove that the special conditions in existing § 411.4(b)(1) and (2) have been satisfied in order to receive payment from Medicare. We sought specific, detailed comments on the circumstances in which individuals who have been released from incarceration on bail, parole, probation, or home detention have a legal obligation to pay for some or all of the health care items or services they receive. We also sought comment on what specific health care items or services, if any, are typically furnished by Federal, State, or local governments at no cost to individuals who have been released from jail or prison on bail, parole, probation, or home detention.
We also proposed to narrow the description of “custody” in § 411.4(b) to remove barriers to access to Medicare by individuals who are returning to the community after incarceration. According to advocates for individuals returning to the community from incarceration, confusion about the applicability of the payment exclusion has led individuals released from incarceration to not apply for Medicare, even if they are eligible, or to apply only for Medicaid, because they believe that Medicare will not pay for items or services that they receive under the payment exclusion. Advocates have also maintained that certain providers or suppliers may be hesitant or refuse to treat individuals who are on bail, parole, probation, or home detention because the providers or suppliers believe that Medicare payment is not available for items or services provided to such individuals. As a result, according to advocates such individuals may delay or forgo necessary treatment, including treatment for substance use disorders, upon release from incarceration. We stated that the proposed changes to the description of “custody” were intended to clarify that Medicare may pay for health care items and services furnished to an individual while on bail, parole, probation, or home detention, provided the individual has a legal obligation to pay for such items or services, without having to prove that the special conditions in § 411.4(b)(1) have been satisfied. (See section XII.2 of this final rule with comment period for a discussion of proposed modifications to the special enrollment periods (SEP) for formerly incarcerated individuals under §§ 406.27(d) and 407.23(d) that would also increase access to Medicare for individuals returning to the community from incarceration.)
We also explained in the CY 2025 OPPS/ASC proposed rule that our proposal to narrow the description of “custody” in § 411.4(b) was intended to bring the Medicare no legal obligation to pay payment exclusion into greater alignment with certain related Social Security and Medicaid provisions. As we noted, CMS relies on data provided by the Social Security Administration to identify individuals who are in custody of penal authorities. However, under Social Security regulations, individuals who are released from incarceration on parole, probation, or home detention are generally not considered to be confined for purposes of a limitation on payment of certain benefits (see section 202(x)(1) of the Social Security Act (42 U.S.C. 402)), and the Social Security data do not track such individuals. Our proposal to narrow the description of “custody” was intended to improve the alignment between the no legal obligation to pay regulation at § 411.4(b) and the data that is used to help carry out the rule. In the proposed rule we also noted, however, that there would still be substantive differences between the Social Security definition of “confinement” and the proposed description of “custody” at § 411.4(b)(3). Most notably, the Social Security Administration suspends benefits if the individual has been convicted of a criminal offense and sentenced to a period of confinement, and based on that conviction remains confined for more than 30 days. In contrast, under our proposed description of “custody,” an individual would be considered to be in custody of penal authorities for purposes of the Medicare no legal obligation to pay payment exclusion if the individual is under arrest; confined in jail, prison, penitentiary, or similar institution while awaiting trial (that is, the person has not yet been convicted); or confined in jail, prison, penitentiary, or similar institution following conviction for any period of time, including sentences of less than 30 days. We noted that these differences are a result of Medicare's statutory no legal obligation to pay payment exclusion. Specifically, in the proposed rule, we explained our belief that individuals who are confined to jail while awaiting trial typically do not have a legal obligation to pay for health care items or services. We invited comments on whether such individuals confined to jail while awaiting trial typically pay for health care items or services they receive or have a legal obligation to do so.
Our proposal to narrow the description of “custody” in § 411.4(b)(3) was also intended to bring the Medicare no legal obligation to pay payment exclusion into closer alignment with Medicaid. As we explained in the CY 2025 OPPS/ASC proposed rule, under Medicaid regulations, Federal Financial Participation (FFP) is not available for services provided to individuals who are inmates of public institutions (see § 435.1009), and an “inmate of a public institution is defined generally as “a person living in a public institution” (see § 435.1010). According to Medicaid State Health Official Letter (SHO) # 16-007, however, individuals who are on parole, probation, or home confinement are generally not considered to be inmates for purposes of Medicaid (see SHO # 16-007 (RE: To facilitate successful re-entry for individuals transitioning from incarceration to their communities) (April 28, 2016) available at https://www.hhs.gov/guidance/sites/default/files/hhs-guidance-documents/sho16007_41.pdf). By no longer including such individuals in the Medicare description of “custody” at § 411.4(b)(3), our proposal was intended to improve dually eligible individuals' access to both Medicare and, potentially, Medicaid, including accessing benefits for an already-enrolled individual. As explained in the proposed rule, advocates shared anecdotally that some Medicaid agencies automatically suspend payment for services to an individual whose Medicare benefits are suspended due to a presumption that the individual is in custody. In addition, according to advocates, some individuals who are recently released from jail or prison on bail, parole, probation, or home detention have only enrolled in Medicaid, because they believe, even where they have a legal obligation to pay, that Medicare will not pay for their health care under the no legal obligation to pay payment exclusion. Our proposal to narrow the description of “custody” in § 411.4(b)(3) was intended to remove this real or perceived barrier to Medicare access.
We cautioned in the CY 2025 OPPS/ASC proposed rule, and we reiterate and underscore in this final rule with comment period, that the generally applicable no legal obligation to pay payment exclusion at § 411.4(a) continues to apply to services furnished to individuals on bail, parole, probation, or home detention in the same way it applies to any other Medicare ( printed page 94502) beneficiary who receives an item or service where there is no legal obligation to pay. The no legal obligation to pay payment exclusion in § 411.4(a) is a general rule that is applicable to all health care items or services (except Federally qualified health center services and as provided in § 411.8(b)) received by any Medicare beneficiary, regardless of whether the individual is in custody of penal authorities. Nothing in the proposed modification of the special condition at § 411.4(b) and the description of “custody” that we are finalizing in this final rule with comment period affects the scope of the general rule at § 411.4(a). Thus, if an individual on bail, parole, probation, or home detention has no legal obligation to pay for a health care item or service, the general rule at § 411.4(a) would continue to prohibit Medicare from paying for such a service, regardless of the scope of the description of “custody” in § 411.4(b)(3). For example, if a State or local government requires substance use disorder counseling as a condition of parole, and the State or local government does not charge all parolees for such services, then the parolee has no legal obligation to pay for such service under § 411.4(a); therefore, Medicare is prohibited under § 411.4(a) from paying for the service. In the proposed rule, we invited comments on what types of medically necessary health care items or services, if any, are typically provided at no cost to individuals on parole, probation, or home detention.
In addition to the proposed changes to the description of “custody,” we also proposed to reorganize and renumber the regulation at § 411.4(b). Currently, § 411.4(b) includes both a description of “custody” and a statement of the conditions that must be satisfied for payment to be made under Medicare for items or services furnished to individuals in custody of penal authorities. We proposed to separately codify the description of “custody” and the special conditions for payments. Specifically, at proposed § 411.4(b)(1)(i) through (iii), we proposed to state the conditions that must be satisfied for Medicare to pay for items or services furnished to an individual in custody of penal authorities; we also proposed certain non-substantive edits to the regulatory language adopted from current § 411.4(b). At proposed § 411.4(b)(2), we proposed a definition of “penal authority” that would apply generally to § 411.4(b). At § 411.4(b)(3), we proposed a description of “custody” that is narrower in scope than the current description.
To align the regulatory text with the statutory no legal obligation to pay payment exclusion at section 1862(a)(2) of the Act, we proposed certain non-substantive edits to § 411.4(a). Specifically, the statute refers to items and services but the current regulation refers only to services. Consistent with the statute, we proposed to refer to both items and services in the regulatory text. We also proposed to add a reference to Federally Qualified Health Center services at § 411.4(a) to align with the statute. Also, the current regulatory text at § 411.4(a)(2) states that no other person or organization has a legal obligation to pay, whereas the statute refers only to a person. Therefore, we proposed to delete “organization” from the regulation text because the term “person” includes both natural and non-natural persons, and the term “organization” is therefore superfluous. We also proposed to align the parenthetical in § 411.4(a)(2) with the statutory parenthetical and to replace the term “beneficiary” in § 411.4(a)(1) and (2) with “individual” to align with section 1862(a)(2) of the Act.
We also proposed to redesignate the special conditions that are specified in current § 411.4(b)(1) and (2) as § 411.4(b)(1)(i) through (iii). Under the proposal, the rebuttable presumption in § 411.4(b)(1) would apply to all items or services furnished to individuals in custody of penal authorities, regardless of who provides the items or services. We invited comments on whether the scope of the rebuttable presumption in proposed § 411.4.(b)(1) should be limited to items or services furnished by the penal authority or by a third party with which the penal authority has arranged to provide the items or services, noting that, if we limit the scope of the rebuttable presumption in this way, the rebuttable presumption in proposed § 411.4(b)(1) would not apply to items or services furnished to individuals in custody of penal authorities by third parties who do not have an arrangement or contract with the penal authority to provide the items or services. We also invited comments on whether individuals in custody of penal authorities are permitted to arrange for their own health care with third parties who do not have an agreement with the penal authority to provide the items or services. Lastly, we proposed at proposed § 411.4(b)(3)(v) to clarify that an individual who is required to reside in a mental health facility would only be considered to be in custody under the no legal obligation to pay payment exclusion if the individual is required to reside in such a facility under a penal statute or rule.
In the CY 2025 OPPS/ASC proposed rule, we addressed our particular concerns with the application of the no legal obligation to pay payment exclusion to individuals residing in halfway houses. We noted that, according to the National Institute of Justice (NIJ), a research, development, and evaluation agency of the U.S. Department of Justice, the term “halfway house” usually refers to temporary housing, provided in a community-based residential facility, which uses around-the-clock supervision and offers services to assist with the transition from incarceration to the community (see https://crimesolutions.ojp.gov/ratedpractices/90#1-0). The NIJ notes that, although the degree to which services are provided to residents varies significantly among halfway house programs, the following characteristics are common to most halfway houses: (1) constant supervision and daily contact between staff and returning individuals; (2) a requirement for participants to abide by rules (such as curfews and drug testing); and (3) access to employment, education, life skills training, and additional services as needed (such as substance use disorder treatment and counseling).
As the name suggests, halfway houses occupy a middle ground between complete custodial incarceration and unconditional release, and there appears to be wide variation in the degree of control exercised over halfway house residents in various State, local, and Federal facilities. Given this variability, we solicited comments on whether halfway house residents typically have a legal obligation to pay for health care items and services, and if so, to what extent (that is, do they have a legal obligation to pay for all or most health care items or services, or only certain items or services) and under what conditions. In particular, we solicited detailed comments, with examples as appropriate, focusing specifically on the legal obligation that halfway house residents have to pay for the health care items and services they receive.
To provide greater focus for our comment solicitation on halfway houses, we proposed regulatory text at § 411.4(b)(3)(vi) that is drawn from the Medicaid payment exclusion rule at § 435.1009. As explained in SHO # 16-007, FFP is available for services furnished to Medicaid-eligible individuals living in halfway houses, provided the following conditions are met: (1) residents are not precluded from working outside the facility in ( printed page 94503) employment available to individuals who are not under justice system supervision; (2) residents can use community resources (libraries, grocery stores, recreation, education, etc.) at will; and (3) residents can seek health care treatment in the broader community to the same or similar extent as other Medicaid enrollees in the state. SHO # 16-007 adds that “at will” is consistent with certain house rule restrictions and travel limitations, and stipulates that the State Medicaid agency must ensure that the halfway house meets the requirements enumerated above.
Consistent with Medicaid guidance, we proposed at § 411.4(b)(3)(vi) to consider an individual to be in custody for purposes of the special condition for payment at § 411.4(b) if the individual is required to reside in a halfway house under any of the following conditions: residents are precluded from working outside the facility in employment that is available to individuals who are not under penal authority supervision; residents may not use community resources (for example, libraries, grocery stores, recreation, or educational institutions) at will; or residents may not seek health care items and services in the broader community to the same or similar extent as individuals who are not under penal authority supervision. Like Medicaid, we proposed to interpret “at will” to be consistent with certain house rule restrictions and travel limitations. Our proposal to align the Medicare no legal obligation to pay payment exclusion at § 411.4(b) with Medicaid guidance was intended to facilitate access to Medicare for dually eligible individuals returning to the community from incarceration and residing in halfway houses.
Consistent with our discussion regarding bail, parole, probation, and home detention, we noted in the proposed rule, and we reiterate and emphasize in this final rule with comment period, that the general no legal obligation to pay payment exclusion at § 411.4(a) could apply to services furnished to individuals in halfway houses even if those individuals are not considered to be in custody for purposes of § 411.4(b). If an individual and the individual's insurance, if any, has no legal obligation to pay for a health care item or service, then Medicare may not pay for the item or service. In the CY 2025 OPPS/ASC proposed rule, we invited comments on whether individuals who reside in halfway houses under the conditions described in proposed § 411.4(b)(3)(vi) typically do not have a legal obligation to pay for health care items or services, or whether fewer, other, or additional factors compared to the factors described in the proposal would form a more appropriate basis for a presumption that the individual is in custody and has no legal obligation to pay for health care items or services.
In the CY 2025 OPPS/ASC proposed rule, we proposed to define “penal authority” at § 411.4(b)(2) as a police department or other law enforcement agency, a government agency operating under a penal statute, or a State, local or Federal jail, prison, penitentiary, or similar institution. We are aware that private contractors in some circumstances may be responsible for operating certain penal institutions or halfway houses, and we sought comment on whether such contractors should explicitly be included in the proposed definition of “penal authority.” As we stated in the CY 2025 OPPS/ASC proposed rule, the proposed definition was intended to be broad enough to include all agencies or institutions that might place or hold an individual in custody, as the term is described at proposed § 411.4(b)(3), regardless of whether the individual has been convicted of a crime. We stated in the proposed rule that the term “penal authority” does not appear to be commonly used outside of the Medicare context. We invited comments on whether other terminology would be more appropriate or would align more closely with terms commonly used in the criminal justice system. We also invited comments on whether the proposed definition of “penal authority” is too broad or narrow for purposes of the no legal obligation to pay payment exclusion.
We received 67 comments on our proposal to narrow the description of “custody” in § 411.4(b) and the other proposals described above. Commenters included advocacy organizations for justice involved-individuals, States, local government and law enforcement organizations, providers and suppliers, and provider organizations. With one exception, all of the commenters supported our proposal to narrow the description of “custody.” A summary of the comments and our responses follows.
Comment: With one exception, commenters unanimously supported our proposal to narrow the description of “custody” in § 411.4(b)(3)(i) by removing individuals who are on supervised release and home detention and striking the phrase “confined completely or partially in any way under a penal statute or rule” from the description. The vast majority of commenters agreed that individuals on supervised release, including individuals on bail, parole, probation, or home detention, are typically responsible for their own health care costs, including securing health insurance from an employer or Medicaid. One commenter representing an advocacy organization for justice-involved individuals surveyed its community-based partners in six states and reported that, universally, the people that they serve on pretrial release, probation, and parole do not receive health care from the justice system, prisons, or jails. Another commenter representing various law enforcement and correctional facility associations stated that, in almost all U.S. jurisdictions, individuals living under any form of community supervision are responsible for securing and paying for their own health care. Likewise, a commenter representing a State's community bail fund suggested that people living in the community under any form of supervised release are typically responsible for their own health care costs. A commenter representing hospitals explained that jails do not currently have an obligation to pay for care provided to formerly incarcerated individuals, so the care provided by hospitals to this population often is uncompensated. A commenter representing a State's department of corrections explained that, while people in their State living under supervised release (including bail, community supervision, re-entry centers, and electronic home monitoring) are responsible for their own health care costs, the department of corrections does pay for intensive inpatient substance use disorder treatment for people on community supervision and in partial confinement for a small number of individuals, and it also pays for this inpatient substance use treatment if an individual does not have insurance.
Most commenters suggested that our proposal to narrow the description of “custody” would also promote successful reentry and community integration for people in the criminal legal system. For example, a commenter representing a State agency for public counsel services explained that its clients reentering the community who do not qualify for Medicaid often face significant health care costs, needing to either pay out-of-pocket or find other insurance, which may lead individuals to delay or forgo treatment. The commenter further explained that its clients are often unable to access treatment that is required by conditions of probation, parole, and bail, due ( printed page 94504) simply to lack of access to health insurance, and that this causes violations to occur, which can lead to the individuals being returned to custody. Other commenters representing addiction and mental health advocacy groups noted that an individual is at the highest risk for an overdose during the first 2 weeks following release from incarceration. According to these commenters, equipping individuals with timely access to substance use, mental health, and other health-related services before release would facilitate the transition to care that is necessary to mitigate the risk of recidivism and to prevent death and other avoidable harms. Another commenter representing a team of researchers, physicians, and community health workers noted that there are significant health and racial disparities between individuals with and without a history of incarceration, as well as between those on probation or parole or those who are not. Citing these substantial disparities and barriers to health care access, the commenter suggested that the proposed revisions are a crucial step towards alleviating these inequities and improving healthcare access and health outcomes. The commenter further stated that increasing Medicare access for individuals on probation and parole has the capacity to increase preventive care utilization, improve health outcomes, and enhance racial health and healthcare equity.
Commenters also reported that the proposal to narrow the description of “custody” would improve dually eligible individuals' access to both Medicare and, potentially, Medicaid, including accessing benefits for already-enrolled individuals. One commenter provided an example of a dually enrolled individual on probation who was legally responsible for paying for his own medication and enrolled in Part D. According to the commenter, the individual was barred from submitting claims for the medication under Part D because the individual was considered to be in custody under the Medicare no legal obligation to pay payment exclusion, but was also denied coverage for the medication under Medicaid because the individual was enrolled in Part D. Another commenter, representing a nonpartisan research and policy organization, explained that some individuals who are recently released from jail or prison on bail, parole, probation, or home detention have only enrolled in Medicaid because they believe, even where they have a legal obligation to pay, that Medicare will not pay for their health care under the no legal obligation to pay payment exclusion.
Response: Based on our review of the comments, we are persuaded that the overwhelming majority of individuals who are on bail, parole, probation, or home detention have a legal obligation to pay for most or all of the health care items or services that they receive. We therefore no longer believe it is reasonable to presume that such individuals have no legal obligation to pay for their health care. We also agree with the commenters that narrowing the description of “custody” in the regulatory no legal obligation to pay payment exclusion will improve access to Medicare, including access by dually eligible individuals, thus facilitating the transition to the community for formerly incarcerated individuals, mitigating the risk of recidivism, and helping to prevent death and other avoidable harms. Therefore, using our authority in sections 1862(a)(2) and 1871 of the Act, we are finalizing our proposal to remove individuals who are on supervised release and home detention and to strike the phrase “confined completely or partially in any way under a penal statute or rule” from the description of “custody” at final § 411.4(b)(3)(i). As explained in further detail below, we are also finalizing an illustrative list at § 411.4(b)(3)(ii) of individuals who are not considered to be in custody for purposes of the payment exclusion.
Although we are narrowing the description of “custody” to no longer include individuals on bail, parole, probation, or home detention (and, as explained below, individuals who are required to reside in halfway houses), we reiterate that the generally applicable no legal obligation to pay payment exclusion at § 411.4(a) would continue to apply to services furnished to such individuals in the same way it applies to any other Medicare beneficiary who receives an item or service where there is no legal obligation to pay. The no legal obligation to pay payment exclusion in § 411.4(a) is a general rule that is applicable to all health care items or services (except Federally qualified health center services and as provided in § 411.8(b)) received by any Medicare beneficiary, regardless of whether the individual is in custody of penal authorities. Thus, if a State furnishes an item or service (or arranges for an item or service to be furnished), such as intensive inpatient substance use disorder treatment, to non-Medicare individuals who are on bail, parole, probation, or home detention or who are required to reside in a halfway house, and the individuals and the individuals' insurance (if any) are not pursued for payment with the same vigor that the State pursues the collection of other debts, then Medicare payment may not be made for similar items or services, because the Medicare beneficiaries, like the non-Medicare individuals, have no legal obligation to pay for the items or services.
Comment: One commenter did not support CMS' proposed changes for formerly incarcerated individuals. The commenter stated a concern that, under our proposal, Medicare would no longer pay for community health programs that support and assist individuals returning to the community from incarceration, in an effort to prevent recidivism. The commenter expressed concern that failure to pay for such medical services would cause more harm to communities, straining the healthcare system and leading to costly health care in emergency departments.
Response: We believe that the commenter misunderstood our proposed revisions. By narrowing the description of “custody” at § 411.4(b)(3)(i), we were not proposing that we no longer pay for Medicare-covered items or services furnished to these individuals. Rather, we were proposing to narrow the description of “custody” in § 411.4(b)(3)(i) because we no longer believe that certain classes of individuals should be presumed to be in custody for purposes of the no legal obligation to pay payment exclusion. We also proposed to narrow the description of “custody” to remove barriers to accessing Medicare by individuals who are returning to the community after incarceration. By adopting these revisions to the description of “custody,” we believe that we are adequately addressing this commenter's concerns regarding policies that facilitate successful re-entry for individuals transitioning from incarceration to their communities.
Comment: Many commenters encouraged CMS to explicitly state in the regulatory text that individuals on bail, parole, probation, or home confinement are not considered to be in custody. Commenters stated that explicit regulatory text would provide much needed clarity to individuals, providers, and advocates who are navigating these circumstances. One commenter representing an advocacy organization suggested that regulatory language set in the affirmative is easier to understand and will help further align the approaches of Medicare and Medicaid. The commenter also noted that it can be hard for individuals with criminal legal histories to find health care providers, and affirmative ( printed page 94505) regulatory language can help address that issue by providing reassurance to providers that their services will be reimbursed by Medicare. Additionally, the commenter expressed concern that, without explicitly stating who is not considered to be in custody under the payment exclusion, providers may not attempt to submit claims to Medicare or may not encourage Medicare enrollment for patients under community supervision because it is too complicated or may refuse to provide services to people on community supervision, citing Medicare's payment exclusion as the reason. Another commenter representing a state Medicaid and CHIP office agreed that expressly specifying in regulation when an individual is not considered to be in custody of a penal authority would help achieve consistency and clarity, reduce potential ambiguity, and ensure fair treatment of these individuals.
Response: We are persuaded by the commenters. Therefore, using our authority at sections 1862(a)(2) and 1871 of the Act, at § 411.4(b)(3)(ii), we are including in the regulatory text an illustrative list of individuals who are not considered to be in custody under the no legal obligation to pay payment exclusion, including individuals on bail, parole, probation, and home detention and individuals who are required to reside in halfway houses. We agree that this should help to achieve consistency and clarity, reduce potential ambiguity and uncertainty, and give providers and suppliers reassurance that their services may be reimbursed by Medicare. As finalized, § 411.4(b)(3)(ii) provides that individuals who are not considered to be in custody under the payment exclusion include, but are not limited to, those individuals who are released to the community pending trial (including those in pretrial community supervision and those released pursuant to cash bail), on parole, on probation, on home detention or home confinement, or required to live in a halfway house or other community-based transitional facility. The list is intended to be illustrative, not exhaustive. An individual is not deemed to be in custody merely because the terminology used by a particular penal authority to denote that individual's status does not align with the terminology used in final § 411.4(b)(3)(ii). Rather, the description of “custody” at § 411.4(b)(3)(i) states the sufficient conditions for determining whether an individual is considered to be in custody for purposes of the no legal obligation to pay payment exclusion. (We note also that, under the qualification “including, but not limited to” in final § 411.4(b)(3)(ii), individuals who have no involvement with the criminal justice system or who have been unconditionally released from the custody of penal authorities are also not considered to be in custody under the Medicare payment exclusion rule.) As a reminder, if an individual in any of the circumstances listed in final § 411.4(b)(3)(ii) does not have a legal obligation to pay for a health care item or service, the general rule at § 411.4(a) would continue to prohibit Medicare from paying for such an item or service, regardless of the scope of the description of “custody” in § 411.4(b)(3)(i), and regardless of the fact that such individuals are not considered to be in custody under § 411.4(b)(3)(ii).
Comment: Most commenters addressing the breadth or appropriateness of the terminology in our proposed description of “custody” suggested that we remove the term “under arrest.” Some commenters stated that this term is imprecise and overly broad, and could prevent people who are on bail or pre-trial release and whose services are not covered or provided by a carceral setting from accessing Medicare. Another commenter noted that home detention is sometimes referred to as “home arrest.” One commenter explained that there are typically two different periods of confinement following arrest and prior to conviction. According to the commenter, when an individual is arrested, they are often confined on site or in a local police station before they are arraigned. After the individual is arraigned, they may be held in jail awaiting trial in pre-trial detention. The commenter stated that, during the pre-arraignment period, the provision of health care services is typically minimal and any health care items or services furnished are billed to the individual receiving them. In contrast, according to the commenter, individuals who are confined in jail during pre-trial detention typically do not have a legal obligation to pay for their health care. The commenter indicated that, to the extent that the term “under arrest” refers to pre-arraignment confinement, it should not be included in the description of “custody,” because these individuals typically have a legal obligation to pay for their own health care, whereas, to the extent that the term “under arrest” refers to pre-trial detention in jail, the term is not necessary, because the proposed description of “custody” already includes individuals incarcerated in jail. Many other comments agreed that the term “incarcerated in jail” sufficiently captures the subset of individuals who have been placed under arrest and have no legal obligation to pay for their own health care.
Response: After reviewing the comments, we agree that the phrase “under arrest” is imprecise and overly broad, and we are therefore removing the term from the description of “custody” at final § 411.4(b)(3)(i). Specifically, based on the information provided by commenters regarding the typical obligation of individuals who are under arrest to have a legal obligation to pay for the health care items or services that they receive before they are arraigned, we do not believe that it is appropriate to presume that such individuals have no legal obligation to pay for their own health care. We are also concerned that the term “under arrest” may cause confusion for individuals post-arraignment, because it may be applied in some jurisdictions to individuals that we are explicitly excluding from the description of “custody,” including individuals who have been released to the community pending trial and individuals who are on home detention or confinement. Lastly, we agree that, insofar as individuals who are under arrest are incarcerated or physically detained in a jail or similar institution, these individuals would be considered to be in custody under the description of “custody” at final § 411.4(b)(3)(i)(A). We reiterate that, if an individual who is under arrest has no legal obligation to pay for a health care item or service, the general rule at § 411.4(a) would continue to prohibit Medicare from paying for such an item or service, regardless of the scope of the description of “custody” in § 411.4(b)(3). Given the breadth and imprecision of the term “under arrest,” as identified by the commenters, we are similarly not including the term “under arrest” in the list of individuals who are presumed not to be in custody at final § 411.4(b)(3)(ii).
Comment: One commenter representing those with lived experience of the criminal justice system asserted that careful consideration should be given to individuals who experience a very short period of incarceration related to a violation of the conditions of their probation or parole. According to the commenter, the sanctions for such violations of probation or parole often fall in a gray area between a defined sentence and a pre-hearing holder. The commenter expressed a concern that individuals who are temporarily incarcerated for violating the conditions ( printed page 94506) of their probation or parole may be denied Medicare payment for health care items or services for even a short period of time.
Response: An individual who is incarcerated is considered to be in custody of penal authorities under final § 411.4(b)(3)(i)(A), even if the individual is not convicted of a crime and even if the time spent in incarceration is relatively short. While the individual is incarcerated, the special condition at § 411.4(b) applies to health care items or services that the individual receives. We note, however, that once the individual is released to the community from incarceration for the probation or parole violation, the individual would no longer be considered to be in custody under final § 411.4(b)(3), and it would no longer be necessary to overcome the rebuttable presumption at § 411.4(b)(1) for Medicare payment to be made for items or services furnished after the individual is released from incarceration.
Similar principles apply to individuals who are intermittently incarcerated, such as individuals who are required to reside in jail over the weekend but are allowed to live at home during the week. For the time period when an individual is required to be in jail, the individual is considered to be in custody under § 411.4(b)(3)(i), and there is a presumption that the individual has no legal obligation to pay for health care items or services that they receive during this period. On the other hand, the individual is not considered to be in custody under § 411.4(b)(3)(i)(A) for those days in the week when the individual is not required to reside in jail, prison, penitentiary, or similar institution. Assuming the individual would also not be considered to be in custody under § 411.4(b)(3)(i)(B) through (D) (that is, the individual is not temporarily outside of a jail, prison, penitentiary, or similar institution; on medical furlough or similar arrangement; escaped from confinement by a penal authority; or required to reside in a mental health facility under a penal statute or rule), the rebuttable presumption at § 411.4(b) would not apply to health care items or services that the individual receives on those days that the individual is not in jail or prison.
Comment: A few commenters stated that those on “medical furlough” should also be removed from the description of “custody” at § 411.4(b)(3)(i). One commenter representing a hospital association expressed concern that, without including the furlough class of individuals, hospitals will continue to provide services that remain unreimbursed and generally unbillable. A commenter representing local law enforcement agencies asserted that individuals on medical furlough are responsible for securing and paying for their own health care. Another commenter representing a State department of health asserted that individuals on medical or geriatric parole in its State have the freedom to choose their own medical care and are financially responsible for that care. The commenter expressed a concern that the phrase “medical furlough or similar arrangements” could be interpreted broadly to preclude coverage for those on medical and geriatric parole. The commenter also objected to the phrase “temporarily outside of a jail, prison, penitentiary, or similar institution on medical furlough or similar arrangement” in the proposed description of “custody” because the commenter believed that bail, probation, parole, and home confinement, which we proposed to remove from the description of “custody,” are all instances where an individual is “temporarily outside of a jail, prison, penitentiary, or similar institution.”
Response: After considering the comments received, we do not believe it is appropriate to remove “medical furlough or similar arrangement” from the description of custody. The special condition at § 411.4(b) helps prevent improper Medicare payments for items and services furnished to individuals in custody of penal authorities, because such individuals typically have no legal obligation to pay for their own health care. We are concerned that categorically excluding medical furlough from the description of “custody” in § 411.4(b)(3) may lead to improper payments, as a penal authority would have an incentive to place Medicare beneficiaries on medical furlough to receive health care items or services that the penal authority furnishes to similarly situated non-Medicare individuals at no cost (that is, the penal authority does not pursue payment from the non-Medicare individuals and the individuals' insurance, if any). To prevent improper payments in such circumstances, we believe it is reasonable to require the penal authority to overcome the rebuttable presumption in § 411.4(b)(1) in order for Medicare payment to be made for health care items or services furnished to an individual on medical furlough. And, as explained in our response to other comments, if a penal authority has established a program or policy permitting medical furloughs or similar arrangements, we do not believe it is unreasonably burdensome for the penal authority to overcome the rebuttable presumption in § 411.4(b)(1).
We note also that we are not sure of the precise meaning of the terms “medical or geriatric parole” as used by the second commenter. In our regulations, the term “medical furlough or similar arrangement” refers to a situation where an individual is temporarily released from incarceration for the purpose of receiving medical services, and is unconditionally required to return to the jail or prison after the medical services are furnished. In contrast, the term “parole,” as we understand it, typically refers to a permanent release from incarceration, even if the individual has to meet certain conditions to retain their freedom. To the extent that “medical or geriatric parole” refers to “parole” in the latter sense, then, under final § 411.4(b)(3)(ii)(B), individuals who are released on medical or geriatric parole are not considered to be in custody for purposes of the payment exclusion. We note that the list of individuals who are not considered to be in custody under § 411.4(b)(3)(ii) is illustrative only and is not intended to apply only where penal authorities use specific terminology.
Comment: One commenter suggested that CMS clarify the difference between “medical furlough” and the practice of “compassionate release.” According to the commenter, “compassionate release” occurs where an individual is released from incarceration due to extraordinary circumstances, such as a severe or terminal illness. The commenter added that, because such individuals have completed their sentences, they typically have a legal obligation to pay for their own health care.
Response: As explained in our response to the previous comment, “medical furlough or similar arrangement” refers to a temporary release from incarceration for the purposes of obtaining health care with a requirement that the individual return to incarceration after treatment. In contrast, as described by the commenter, individuals who have received compassionate release are considered to have completed their sentences, and they are legally responsible for paying for their own health care. Under the circumstances described by the commenter, we agree that “compassionate release” is not an example of “medical furlough or similar arrangement,” and individuals who have received compassionate release as described by the commenter are not considered to be in custody under the ( printed page 94507) no legal obligation to pay payment exclusion. Although we do not believe that it is necessary to state specifically in regulation that individuals who have received compassionate release are not considered to be in custody, we reiterate that the list of individuals who are not considered to be in custody at final § 411.4(b)(3)(ii) is illustrative only, not exhaustive, and not intended to apply only where penal authorities use specific terminology.
Comment: One commenter representing an advocacy organization suggested that, instead of the term “bail” in the regulations at § 411.4(b), CMS use the phrase “released to the community pending trial (including those in pretrial community supervision and those released pursuant to cash bail).” This commenter expressed concern that, although bail technically refers to the process of pre-trial release, it is often used to only mean cash bail. According to the commenter, many individuals are released to the community prior to trial without cash bail and are responsible for paying for their health care.
Response: After considering the comments, we agree that the term “bail” may be too narrow, because it does not necessarily include individuals who have been released from jail prior to trial on their own recognizance. Therefore, we are using the phrase “released to the community pending trial (including those in pretrial community supervision and those released pursuant to cash bail)” rather than “bail” in final § 411.4(b)(3)(ii)(A).
Comment: The commenters that addressed the issue of individuals who reside in halfway houses supported narrowing the description of “custody” to exclude some or all individuals who are required to reside in halfway houses. We did not receive any comments in favor of considering all halfway house residents to be in custody for purposes of the no legal obligation to pay payment exclusion. Commenters reported that individuals who are required to reside in halfway houses are typically responsible for paying for their own health care. One commenter representing various law enforcement and correctional facility associations stated that, in almost all U.S. jurisdictions, individuals living in partial confinement facilities (such as halfway houses and work release facilities) are responsible for securing and paying for their own health care. Another commenter stated that most halfway house residents are responsible for their own health care, even when their freedom of movement is restricted, and gave as an example a person living in a halfway house with restricted freedom of movement setting up a mental health appointment with a therapist in the community as part of their re-entry plan. One commenter representing a State department of corrections stated that halfway house residents in its State are not responsible for their own health care if they are on “confined” status, while residents on “furloughed” status are legally responsible for the costs of that health care. A commenter representing a different State's department of corrections noted, without using the term “halfway house,” that individuals in its State under “partial confinement” are responsible for their own health care, with the exception of intensive inpatient substance use disorder treatment, which the State provides to certain individuals without charge. A third commenter representing a different State reported that individuals in its State are not required to live in halfway houses, and that individuals residing in the community under the supervision of the State's department of corrections are responsible for their own medical costs.
Response: We are persuaded by the commenters that individuals who are required to live in halfway houses typically have a legal obligation to pay for all or most of the health care items or services that they receive. Given this, we no longer believe that such individuals should be presumed to be in custody of penal authorities, with no legal obligation to pay for their own healthcare, and we no longer believe that the rebuttable presumption at § 411.4(b)(1) must be overcome in order for Medicare to pay for health care items or services furnished to such individuals. Therefore, using our authority in sections 1862(a)(2) and 1871 of the Act, in this final rule with comment period, we are not including individuals who are required to live in halfway houses in the description of custody at § 411.4(b)(3)(i). The regulation text at final § 411.4(b)(3)(ii)(E) expressly states that individuals who are required to reside in halfway houses or other community-based transitional facilities are not considered to be in custody for purposes of the no legal obligation to pay payment exclusion. We are using the phrase “community-based transitional facility” in the regulatory text at final § 411.4(b)(3)(ii) to make it clear that the residence where an individual resides need not be explicitly classified as a “halfway house” under State or local law for the exclusion from our description of “custody” to apply. We reiterate that § 411.4(b)(3)(ii), as finalized, is an illustrative, not exhaustive, list of individuals who are not considered to be in custody for purposes of the no legal obligation to pay payment exclusion.
With respect to work release programs, we understand that these programs differ from jurisdiction to jurisdiction. In some jurisdictions, individuals in work release programs reside in halfway houses or other community-based transitional facilities and leave the facility for limited time periods to work. Under § 411.4(b)(3)(ii)(E), these individuals are not considered to be in custody for purposes of the payment exclusion, because they reside in a halfway house or similar institution. In other jurisdictions, individuals who participate in work release programs are required to reside in a jail, prison, or similar institution while not working, even if they are permitted to leave the jail, prison, or similar institution to work for limited periods. For purposes of the no legal obligation to pay payment exclusion, these individuals are considered to be in custody under § 411.4(b)(3)(i)(A), even for the time period when they are outside of the jail or prison working, because they reside in, and remain inmates of, a jail, prison, or similar institution. We believe that the penal authority typically retains a legal obligation to pay for the health care of such individuals, even for the period when they are outside of the jail or prison working.
Finally, we note that, although commenters stated that halfway house residents typically have a legal obligation to pay for their own health care, some commenters also reported that, in certain instances, halfway house residents are not responsible for paying for some or all of their health care. We remind parties that, under the general no legal obligation to pay payment exclusion at § 411.4(a), if an individual who is required to live in a halfway house has no legal obligation to pay for a health care item or service, the general rule would continue to prohibit Medicare from paying for such an item or service, regardless of the scope of the description of “custody” in § 411.4(b)(3)(i), and regardless of the fact that halfway house residents are not considered to be in custody under final § 411.4(b)(3)(ii)(E).
Comment: Commenters generally supported our proposal to rely on Medicaid guidelines, drawn from SHO # 16-007, to determine if an individual who is required to reside in a halfway house should be considered to be in custody for purposes of the no legal ( printed page 94508) obligation to pay payment exclusion. Many commenters maintained that, if individuals have freedom of movement, then they should be entitled to have Medicare pay for their care. One commenter urged CMS to focus on freedom of movement and association as the determining factor, as opposed to focusing solely on the three factors drawn from SHO # 16-007 that are intended to demonstrate that an individual has freedom of movement and association (that is, freedom to seek work outside of the facility, use community resources, and procure health care in the community). The commenter expressed a concern that some halfway house residents with relatively high degrees of freedom of movement and association may be inappropriately considered to be in custody under the Medicare no legal obligation to pay payment exclusion if the facility where the individual resides fails to meet one or more of the three enumerated factors. Some commenters reported that, in some instances, halfway house residents may have their movement restricted but still have a legal obligation to pay for the health care items and services that they receive.
Many commenters explained that halfway houses are transitional facilities designed to support re-entry, where individuals typically only reside for a number of weeks. One commenter noted that a halfway house resident's degree of freedom of movement and association often increases over time as the individual advances through incentive-based phases. The commenter maintained that, under the proposed adoption of Medicaid guidelines from SHO # 16-007, on-going evaluation of an individual's degree of freedom of movement and association would be necessary to determine if the individual has the requisite degree of freedom necessary to no longer be considered in custody under the Medicare payment exclusion. The commenter recommended that CMS develop guidelines for individualized assessments of whether a halfway house resident is considered to be in custody, and include in the consideration a resident's access to health care both within and outside of the halfway house facility. Another commenter recommended that CMS focus on the reason for an individual's release to a halfway house—specifically, to facilitate successful reintegration into society—instead of the degree of freedom that the individual enjoys to determine whether the individual is in custody for purposes of the no legal obligation to pay payment exclusion.
Response: The special condition at § 411.4(b) for services furnished to individuals in custody of penal authorities is intended to clarify how the general no legal obligation to pay payment exclusion applies to services furnished to individuals in custody of penal authorities. Thus, the touchstone of our analysis is whether individuals who are required to live in halfway houses typically have a legal obligation to pay for the health care items or services that they receive. As noted in the response to comments above, we are persuaded that individuals who are required to reside in halfway houses typically have a legal obligation to pay for most or all of the health care items or services they receive. Therefore, we no longer believe that such individuals should be presumed to be in custody, with no legal obligation to pay for their own health care, under the Medicare no legal obligation to pay payment exclusion.
Although the Medicaid guidelines in SHO #16-007 offer a more targeted approach to halfway house residents, based on the comments, we are not convinced that the freedom of movement and association standard employed by Medicaid overlaps in all cases with the legal obligation to pay standard that is central to the Medicare payment exclusion. Given the typically short duration of halfway house residency, and the fact that individuals' level of freedom may increase during their residency, we are also concerned that the guidelines in SHO # 16-007 may be difficult to administer in the Medicare context, potentially adding burden and barriers to accessing Medicare payment for health care items and services. Rather than incorporate Medicaid guidelines in SHO #16-007, we are excluding halfway house residents from the description of “custody” at § 411.4(b)(3).
Comment: Numerous commenters suggested that we exclude from the rebuttable presumption any items or services furnished by a third party not under arrangement or contract with the penal authority. In response to our comment solicitation regarding whether an individual in custody of a penal authority is permitted to arrange for their own health care with third parties that do not have an arrangement or contract with the penal authority to provide the items or services, one commenter reported that the practice of arranging care with outside third parties varies from jurisdiction to jurisdiction. The commenter stated that, both for security purposes and to streamline staff operations, most jails and prisons use a centralized provider—either contracted or through direct hire—that provides a set of health care services and items. A commenter representing local law enforcement agencies stated that individuals in custody may be permitted to arrange for their own health care with third parties other than the health care entity that normally provides care to the penal authority facilities' residents. This commenter further explained that the incarcerated individual is typically required to pay out of pocket, not only for the health care services but also for the costs to the facility to transport the individual to the visit, including the associated custody staff costs. The commenter supported Medicare paying for the costs of such visits, including the associated transportation costs. One commenter representing a State department of corrections explained that, in the commenter's State, incarcerated individuals can request and access any health care items or services not covered by the State's health plan, but individuals are responsible for all costs related to the medical care, including transport, custody escort, mileage, medications, and any complications or follow up care indicated. The commenter explained that prison staff arrange for and schedule the care. Another commenter requested that we not presume that an individual in the custody of a penal authority who receives items and services from a third party not contracted with a penal authority has no legal obligation to pay for such items and services. This commenter went on to state that, alternatively, if such a presumption does continue to apply in these circumstances, information about how to rebut such a presumption should be amended to include circumstances where a non-contracted third party has provided health care items and services. The commenter also expressed concern that providers may not understand that the burden of rebutting the presumption is on the State or local government, not on the individual.
Several commenters representing mental health care providers requested that the rebuttable presumption not be limited to items or services furnished by the penal authority or by a third party with which the penal authority has arranged to provide the items or services. However, the commenters also suggested that the proposed regulatory description of when an individual is in “custody” and the requirements related to how to rebut the presumption that an individual has no legal obligation to pay ( printed page 94509) for their health care items and services would impose undue challenges to access and administrative burden for providers in confirming if someone is in custody, per the proposed definition. The commenters maintained that, by not finalizing our proposal, providers would be better positioned to serve individuals in what is often a particularly vulnerable and crucial time to access care, and individuals would have greater access to provider organizations, thereby strengthening their choice in the right treatment and care for them.
Response: After carefully considering the comments, we are not limiting the scope of the rebuttable presumption at § 411.4(b)(1) to only those items or services that are furnished by the penal authority or by a third party with which the penal authority has arranged to provide the items or services. The special condition at § 411.4(b) helps prevent improper Medicare payments for items and services furnished to individuals in custody of penal authorities, because such individuals typically have no legal obligation to pay for their own health care. We are concerned that limiting the scope of the rebuttable presumption at § 411.4(b)(1) to only items or services furnished by or on behalf of penal authorities may lead to improper payments, as penal authorities would have an incentive to arrange for Medicare beneficiaries to be treated by third party providers and suppliers, while furnishing similar items or services to similarly situated non-Medicare individuals without pursuing payment from the non-Medicare individual and the individual's insurance, if any.
To prevent improper payments in such situations, we believe it is reasonable and not unduly burdensome to require the penal authority to overcome the rebuttable presumption in § 411.4(b)(1). For example, assume that a penal authority has established a program or policy that permits individuals to receive health care items or services from third party providers or suppliers, and further that the health care items or services in question are not furnished to non-Medicare individuals at no cost (that is, the penal authority charges both Medicare beneficiaries and non-Medicare individuals for the items or services, and pursues payment in full for the items or services from both Medicare beneficiaries and non-Medicare individuals and the individuals' insurance, if any). To the extent that the penal authority is responsible for arranging such care and collecting amounts owed by incarcerated individuals for the health care items or services received, we do not believe it is unreasonably burdensome for the penal authority to overcome the rebuttable presumption in § 411.4(b)(1).
Comment: With respect to the terminology used to describe individuals who are required to reside in mental health facilities, one commenter requested that CMS replace “under a penal statute or rule” with “as a result of violations of the penal code.” According to the commenter, this terminology aligns with Medicare Part D guidance, which provides that individuals who are confined to mental health facilities as a result of violations of the penal code are considered to be incarcerated, while individuals who are confined to such facilities as a result of court orders not related to penal violations are not considered incarcerated.
Response: We decline to make the suggested edit because we believe it would make the description of “custody” too narrow for individuals who are required to reside in a mental health facility. In particular, the term “violation” suggests that the individual must be convicted of a criminal violation in order to be considered to be in custody under the no legal obligation to pay payment exclusion. In contrast, our description of “custody” is intended to capture those individuals who do not have a legal obligation to pay for their health care, which we believe also includes individuals who are required to reside in a mental health facility under a penal statute prior to conviction, while the individual is awaiting trial.
Comment: One commenter supported expanding the definition of “penal authority” to include private parties operating correctional facilities of any kind. The commenter stated that expanding the definition would facilitate access to the special enrollment period for individuals returning from incarceration.
Response: We are not persuaded that it is necessary to explicitly include private parties operating correctional facilities in the definition of penal authority at § 4114(b)(2). The definition of “penal authority,” as finalized, includes “a State, local or Federal jail, prison, penitentiary, or similar institution.” The definition does not specify that the facility must be owned and operated directly by a governmental authority in order to be a “penal authority.” Rather, under final § 411.4(b)(2), a jail, prison, or other correctional facility qualifies as a “penal authority” regardless of whether it is owned or operated by a governmental entity or by a private entity on behalf of a governmental entity.
Comment: Commenters variously suggested revising the term “penal authority” to “correctional authority,” “correctional facility,” “carceral facility,” or “correctional institution.” However, most commenters recommended we use the term “correctional facility,” which the commenters maintained is commonly used terminology in the criminal justice system, including by Medicaid. One commenter suggested we seek alternatives to the term “penitentiary.”
Response: After consideration of the public comments we received, we are not persuaded to change the term “penal authority” in the regulation text. The definition is intended to be broad enough to include all agencies or institutions that might place or hold an individual in custody, as the term is described at final § 411.4(b)(3)(i), regardless of whether the individual has been convicted of a crime. The terms suggested by commenters are not broad enough to accomplish this. Similarly, to ensure that the definition is sufficiently broad, we are not striking the term “penitentiary.” We are aware that the term “penal authority” does not appear to be commonly used outside of the Medicare context but nonetheless believe that this is the best term to implement our policies.
After consideration of the public comments we received, we are finalizing our proposal to narrow the description of custody at final § 411.4(b)(3)(i), with one modification: we are striking the term “under arrest.” We are also finalizing at § 411.4(b)(3)(ii) an illustrative list of individuals who are not considered to be in custody for purposes of the no legal obligation to pay payment exclusion. In addition, we are not including halfway house residents in the description of “custody” in final § 411.4(b)(3)(i), and we are clarifying at § 411.4(b)(3)(ii)(E) that individuals who are required to live in a halfway house or other community-based transitional facility are not considered to be in custody for purposes of the no legal obligation to pay payment exclusion. We are also finalizing our proposed non-substantive edits to § 411.4(a) and our proposal to reorganize and renumber the regulation at § 411.4(b). Lastly, we are finalizing the definition of “penal authority” at § 411.4(b)(2) as proposed. ( printed page 94510)
B. Revision to Medicare Special Enrollment Period for Formerly Incarcerated Individuals
1. Background
The Consolidated Appropriations Act, 2021 (Pub. L. 116-260) provided the authority to establish Medicare Part A and B special enrollment periods (SEP) for individuals due to exceptional conditions. In the final rule titled “Medicare Program; Implementing Certain Provisions of the Consolidated Appropriations Act, 2021 and Other Revisions to Medicare Enrollment and Eligibility Rules,” which appeared in the Federal Register on November 3, 2022 (87 FR 66454), CMS used this authority to establish an SEP for formerly incarcerated individuals. Individuals who use this SEP are able to enroll in Medicare Premium Part A and Part B and avoid potential gaps in coverage and late enrollment penalties (LEPs). As established in §§ 406.27(d)(1) and 407.23(d)(1), an individual is eligible to enroll in Medicare Parts A and/or B using this SEP so long as they demonstrate that they are eligible for Medicare and failed to enroll or reenroll in Parts A and/or B due to being in custody of penal authorities, and there is a record of release either through discharge documents or data available to the Social Security Administration (SSA). The SEP provisions at §§ 406.27(d) and 407.23(d) incorporate the description of `in custody of penal authorities' from the payment exclusion parameters already established in § 411.4(b). In the November 2022 final rule, we stated that it was important to align the scope of the SEP with the scope of individuals specified in § 411.4(b) as individuals in custody of penal authorities; alignment with § 411.4(b) provides a measure of assurance that individuals who are no longer subject to the payment exclusion are able to enroll in Medicare (Part B and, if necessary, premium Part A). We also noted that we would continue to assess the impact of alignment of the SEP with the scope of the payment exclusion. (87 FR 66464)
2. Summary of the Proposed Provisions, Public Comments and Responses to Comments
Section 202 of the Act generally provides the basis for SSA to determine an individual's eligibility for old age, survivors, and disability insurance (OASDI) benefits, also known as social security benefits, under Title II of the Act. For most Medicare beneficiaries, entitlement to Medicare Part A is based on entitlement to OASDI benefits under Title II per section 226 of the Act.[713] In addition, the SSA is responsible for determining entitlement to Part A and eligibility for Part B of Medicare. (See Pub. L. 103-296, sec. 105.) Section 202(x) of the Act suspends payment of OASDI benefits to prisoners, certain other inmates of publicly funded institutions, fugitives, probationers, and parolees, including when an individual is confined in a jail, prison, or other penal institution or correctional facility pursuant to conviction of a criminal offense for a period of more than 30 days. We proposed to amend the SEP at §§ 406.27(d)(1) and 407.23(d)(1) to align the SEP triggering event more closely with the bases on which an individual's OASDI benefit is reinstated or initiated rather than on the scope of the Medicare payment exclusion in § 411.4(b). We believe that these proposed amendments would streamline the administrative process for determining an individual eligible for this SEP and align eligibility for this SEP with an individual's obligation and ability to pay for services that would otherwise be covered by Medicare. Furthermore, we believe that the alignment of this SEP with the initiation or reinstatement of OASDI benefits is appropriate, as it allows a population facing many challenges reintegrating into society to enroll or reenroll in Medicare by having premiums deducted from their OASDI benefits, rather than paying out of pocket.
The proposed amendments to § 411.4(b), discussed in section XXIII.A of this final rule with comment period, would narrow the list of settings where an individual is presumed to have no obligation to pay for Medicare-covered items and services—and, thus, Medicare is prohibited from paying for those services—because the individual is in custody of a penal authority. The rebuttable presumption at § 411.4(b) provides the individual an opportunity to indicate to CMS that the other entity did not cover certain items or services and ask that Medicare provide payment, but this option is moot if the individual is not able to enroll in Medicare. (See section XXIII.A of this final rule with comment period for more information about the rebuttable presumption and operation of the payment exclusion.) Under the current version of this SEP, beneficiary advocate groups raised concerns about the possibility for scenarios where an individual is not able to enroll when their items and services could be covered by Medicare, or they are able to enroll (and pay monthly premiums) but Medicare is not able to pay for their services.
In making determinations about the suspension of OASDI benefits due to an individual's incarceration, SSA uses data it collects from jails, prisons, other penal institutions or correctional facilities and certain mental health institutions regarding individuals confined in those and similar institutions for the reasons outlined in section 202(x)(1)(A) of the Act. See SSA System of Records Notice: Prisoner Update Processing System 60-0269, at 64 FR 11076 (Mar. 8, 1999) and updated at 72 FR 69723 (Dec. 10, 2007), 78 FR 40542 (Jul. 5, 2013), and 83 FR 54969 (Nov. 1, 2018). However, information about individuals within the full scope of the current provisions at § 411.4(b), including not only individuals who are confined in certain institutions but also individuals who are under arrest but not yet convicted, on medical furlough, or residing in half-way houses may not be as extensively or reliably available. As a result, making eligibility determinations for the SEP for formerly incarcerated individuals as it is currently drafted is operationally difficult. In addition, the current SEP for formerly incarcerated individuals incorporates the rebuttable presumption that is included in § 411.4(b) for situations where an individual (or healthcare provider seeking to bill Medicare for the services) can demonstrate that state or local law requires individuals to repay the cost of medical services while they are in custody and the state or local government entity enforces the requirement to pay by billing all such individuals, whether or not covered by Medicare or any other health insurance, and by pursuing collection of the amounts they owe in the same way and with the same vigor that it pursues the collection of other debts. However, SSA does not have a role in administering the Medicare payment exclusion in § 411.4(b). Therefore, there is a high likelihood of potential inconsistency and administrative burden with tying the implementation of the SEP at §§ 406.27(d)(1) and 407.23(d)(1) to the Medicare payment exclusion under § 411.4(b). Although the proposed changes to § 411.4(b) discussed in section XXIII.A to narrow the scope of the payment exclusion would flow through to §§ 406.27(d) and 407.23(d) without amendment to the SEP parameters, we believe that addressing the rebuttable presumption and potential administration burden is ( printed page 94511) appropriate and will reduce potential confusion.
Further, since the establishment of the SEP in January 2023, we have received feedback that raised concerns about tying SEP eligibility to the Medicare payment definition at § 411.4(b) and how SSA has begun administering the SEP using the data in SSA's systems. We have heard that the SEP eligibility parameters are confusing and potentially prevent or discourage eligible individuals from accessing the SEP. Conversely, situations may arise in which an individual is enrolled in Medicare using the SEP due to SSA data; however, due to the payment exclusion and limited exceptions for these settings, claims for health care may not be paid even if the specific state or local government does not provide health care in this type of setting.
Under our proposal, we intend that SSA would make a determination of an individual's eligibility to enroll using the Medicare SEP at §§ 406.27(d)(1) and 407.23(d)(1) based on the data SSA collects and keeps in its systems for determining OASDI benefit suspensions and any additional documentation provided by individuals to demonstrate that they have been released from incarceration. By more closely aligning the eligibility criteria for the SEP for formerly incarcerated individuals with the data used by SSA in applying the OASDI benefit suspension requirement in section 202(x)(1)(A) of the Act, we intend that the SEP can be more efficiently and accurately administered. With the proposed revisions, the SEP at §§ 406.27(d) and 407.23(d) will provide an opportunity, beginning January 1, 2025, for an individual to enroll in Medicare under the new parameters if the individual was released from incarceration on or after January 1, 2023, failed to enroll in Medicare (Premium Part A or Part B) due to being incarcerated, and is still within the 12-month SEP described in §§ 406.27(d) and 407.23(d).
Overall, we proposed to revise the eligibility requirements at §§ 406.27(d) and 407.23(d), beginning January 1, 2025, to remove the use of a release from the “custody of penal authorities as described in § 411.4(b)” and instead tie the eligibility for this SEP to whether an individual is “released from confinement in a jail, prison, or other penal institution or correctional facility,” which is phrasing that is more consistent with section 202(x)(1)(A)(i) of the Act. However, we did not propose that a criminal conviction or formal sentencing be required for an individual to have been confined in a jail, prison, or other penal institution or correctional facility because conviction of crime is not required for the payment exclusion in § 411.4(b) to apply. As this differs from the requirements under section 202(x)(1)(A) of the Act, we also solicited comment on what documentation an individual can provide to demonstrate they were confined and released without conviction to determine eligibility for the SEP for formerly incarcerated individuals under §§ 406.27(d) and 407.23(d). Further, individuals who have escaped confinement are not considered to be “released” from confinement. Under our proposal, both §§ 406.27(d) and 407.23(d) would use the terms “incarcerated” and “incarceration” as a general reference for individuals who meet either standard. The proposed change in the eligibility criteria for the SEP for formerly incarcerated individuals would align more closely with the standards SSA uses to determine whether an individual is within the scope of the limitation on payment of OASDI benefits established by section 202(x)(1)(A)(i) of the Act.
In the rulemaking to adopt and finalize §§ 406.27(d) and 407.23(d), we did not fully contemplate the implications of tying the SEP's eligibility criteria to the Medicare no legal obligation to pay payment exclusion (which includes a rebuttable presumption). A rebuttable presumption like the one available under the payment exclusion does not work well in enrollment context because although payment can be determined at the service level, an enrollment is either effectuated or not. It may be that for some services, the presumption that the individual has no legal obligation to pay can be rebutted while for other services it is not rebutted based on the scope of the state or local entity's policies and the individual's (or the healthcare providers) ability to provide sufficient evidence. However, enrollment in Medicare, including the obligation to pay Medicare premiums, would not vary with the specific service. While the proposed changes to § 411.4(b) would narrow the range of settings in which an individual is presumed to be in custody and another entity is responsible for the individual's health care coverage, we believe that the proposed revisions to §§ 406.27(d) and 407.23(d) would best address concerns about access to and confusion with the SEP for individuals who have been released from incarceration.
We proposed several changes to §§ 406.27(d) and 407.23(d) to significantly align the SEP eligibility criteria, beginning January 1, 2025, with the criteria used by SSA to determine whether an individual is incarcerated. Throughout, our proposed changes are largely to replace references to an individual being in custody of penal authorities as described in § 411.4(b) with references to an individual's confinement in a jail, prison, or other penal institution or correctional facility. First, we proposed to amend the introductory text in paragraph (d) of both §§ 406.27 and 407.23 to state the general rule that there is an SEP for Medicare eligible individuals who are no longer incarcerated after January 1, 2023. We use “incarcerated” and “incarceration” in this introductory language and in paragraph (d)(3), respectively, to include both being in custody of penal authorities as described in § 411.4(b) (and in the proposed revisions to paragraph (d)(1) of each regulation regarding the current scope of the SEP) and being confined as described in our proposed amendments to paragraph (d)(2) of each regulation. Using the term “incarcerated” is consistent with how these regulations were originally established and leads to more streamlined and less repetitive regulation text. We invited comments on the proposal, especially its implications for people in halfway houses, to ensure access to the SEP for formerly incarcerated individuals.
Second, we proposed to reorganize paragraphs (d)(1) and (2) to establish the rules for eligibility for and the duration of the SEP for releases from incarceration during the periods between (1) January 1, 2023, through December 31, 2024, and (2) on or after January 1, 2025. We proposed to revise §§ 406.27(d)(1) and 407.23(d)(1) to state the current parameters and duration for the SEP that are applicable to releases after January 1, 2023, and before January 1, 2025. The current eligibility requirements are proposed to be redesignated as paragraph (d)(1)(i) and the current duration of the SEP (from current §§ 406.27(d)(2) and 407.23(d)(2)) are proposed to be redesignated paragraph (d)(1)(ii), with clarifications that the date of release is used as part of the eligibility criteria. At §§ 406.27(d)(2) and 407.23(d)(2) we proposed to establish new parameters and duration for the SEP that would be applicable beginning January 1, 2025. Specifically, we proposed at new §§ 406.27(d)(2)(i) and 407.23(d)(2)(i) that an individual is eligible for the SEP if they are released on or after January 1, 2025, from confinement in a jail, prison, or other penal institution or correctional facility would be eligible for the SEP. The existing parameters ( printed page 94512) (currently at §§ 406.27(d)(1) and 407.23(d)(1)) that the individual must demonstrate that they are eligible for Medicare and failed to enroll or reenroll due to being incarcerated and there is a record of release either through discharge documents or data available to SSA would continue to be applicable and are therefore included in proposed §§ 406.27(d)(2)(i) and 407.23(d)(2)(i). At new §§ 406.27(d)(2)(ii) and 407.23(d)(2)(ii), we proposed that, beginning January 1, 2025, the SEP starts the day an individual is released from incarceration as determined by SSA and ends the last day of the 12th month after the month in which the individual is released. As noted above, individuals who use this SEP are able to enroll in Medicare Premium Part A and Part B without LEPs and this would continue to be the case whether individual uses this SEP before or after January 1, 2025.
Under the proposal, as originally intended with the SEP, individuals will have a clearer understanding for how to access this enrollment opportunity to ensure they do not have any gaps in coverage or any LEPs as they leave incarceration.
We received a number of public comments on our proposals. Our summaries and responses to the comments we received are discussed below:
Comment: Commenters unanimously supported our proposal to revise the eligibility requirements for the SEP for formerly incarcerated individuals. Commenters voiced appreciation for the changes and noted the health care access and continuity of care challenges facing formerly incarcerated individuals reintegrating into society and stated that CMS' proposal would reduce potential barriers to Medicare enrollment. Overall, commenters are pleased with the progress and dedication shown by CMS.
Response: We thank the commenters for their support.
Comment: CMS requested and received comments on the application of the SEP proposal for individuals residing in halfway houses. Commenters supported the extension of the SEP to individuals residing in halfway houses, noting that these individuals were responsible for paying for their health services and, while charity care may be available, it is often limited. Many commenters encouraged CMS to adopt the Medicaid program's approach to halfway houses, which extends Medicaid eligibility to individuals that have freedom of movement.
Response: We appreciate the overwhelming feedback received on this request. Based on the feedback received, we are revising the proposal to allow for individuals who have been recently released from incarceration or confinement and are residing in halfway houses to be eligible for this SEP. Specifically, we are revising and adopting the regulations at §§ 406.27(d)(2)(i) and 407.23(d)(2)(i) to explicitly state that individuals released from incarceration or confinement and transitioning to residence in halfway houses are not considered incarcerated or in confinement for the purposes of this SEP.
We note that section 202(x) of the Social Security Act prohibits OASDI benefit payments to beneficiaries that are confined. SSA, at GN 02607.001B.1, currently defines beneficiaries who are “transferred from the facility to a halfway house” as being confined, and therefore ineligible to receive OASDI benefits. Consequently, anyone utilizing this SEP and residing in a halfway house that falls under SSA's definition of confinement may be responsible for covering their Medicare premiums directly or through some other means such as Medicaid buy-in. We note that if an individual has Medicaid coverage, they will generally not use this SEP since states enroll most dually eligible individuals in Medicare and pay the premiums on their behalf under their state buy-in agreements with CMS. Further, as SSA systems may not identify individuals in halfway houses as no longer in confinement, these individuals will likely need to provide documentation of discharge to SSA proving that they have been released from incarceration to affirm eligibility for this SEP. However, as individuals in halfway houses are likely required to pay for their own healthcare and there are changes in this rule to § 411.4(b) to remove the exclusion of Medicare payment to individuals residing in halfway houses, we believe that it is critical to also extend eligibility to enroll in this SEP to individuals in halfway houses.
Comment: A few commenters recommended that CMS explicitly specify that individuals on parole, probation, under home confinement, or those in pre-trial status be eligible for the SEP.
Response: We appreciate the commenters' suggestion to further specify the categories of individuals eligible for this SEP. However, we believe the rule is inclusive of individuals on parole, probation, or under home confinement and additional categorization is unnecessary. Individuals on parole, probation, or home confinement will be able to demonstrate that they have been released from confinement and, thus, will be eligible for this SEP.
With respect to individuals in a pre-trial status, those individuals will have access to Medicare coverage and will not lose coverage due to a pending trial. Additionally, the current procedure for OASDI benefits provides that individuals in pre-trial status would not lose benefits, and therefore payment of Medicare premiums through their OASDI benefits would go uninterrupted. Therefore, we will not be making changes to extend the SEP to individuals on parole, probation, under home confinement, and those in pre-trial status.
Comment: Many commenters requested that CMS provide an option for incarcerated individuals to enroll via the SEP prior to being released from incarceration, citing concerns with access to continuity of care and gaps of coverage. Several commenters pointed to CMS' Medicaid Reentry Section 1115 Demonstration Opportunity that allows for Medicaid enrollment and coverage prior to release from incarceration as an example.
Response: We appreciate the feedback from commenters and understand the importance of mitigating the risk of gaps of coverage. However, we proposed changes specific to the eligibility requirements for the SEP for formerly incarcerated individuals and other changes to §§ 406.27(d)(2)(ii) and 407.23(d)(2)(ii), and as such, changing the duration of the SEP to allow for enrollment prior to release from confinement is not within the scope of this rulemaking.
We considered enrollment prior to release in previous rulemaking (CMS-4199-F; 87 FR 6643), but believed that our policy of allowing individuals the option of choosing an entitlement date retroactive to the first day of the month of their release from incarceration (not to exceed 6 months) addressed this issue.
In regard to the comment about applying a similar policy as the Medicaid Reentry Section 1115 Demonstration to Medicare, we note that individuals who are in confinement can enroll in Medicare during another available enrollment period, such as the IEP or GEP, and may maintain their enrollment if premium payments continue to be paid.
Comment: A few commenters suggested that CMS revise “discharge documents” in proposed §§ 406.27(d)(2)(i) and 407.23(d)(2)(i) to a more inclusive term, which would allow flexibility in the types of documentation accepted. ( printed page 94513)
Response: We thank the commenters for their suggestion and agree that the term can be revised to be more flexible. As a result, we are revising our language at proposed §§ 406.27(d)(2)(i) and 407.23(d)(2)(i) to replace the term “discharge documents” with “documentation of discharge.” This will allow individuals who may not have specific discharge documents, but have other proof of their discharge, the ability to use the SEP.
Comment: Many commenters supported the proposal to revise the SEP eligibility criteria and requested that CMS ensure that formerly incarcerated individuals who encountered barriers to accessing the current SEP prior to January 1, 2025, have an opportunity to enroll in Medicare coverage under equitable relief grounds or through applying overlapping effective dates of these SEP changes.
Response: We thank the commenter for their support and suggestion. The parameters for equitable relief, which is a mechanism to correct or eliminate inequity to the individual when their Medicare enrollment rights are prejudiced because of the error, misrepresentation, or inaction of the Federal Government, are defined in statute (section 1837(h) of the Social Security Act) and, as such, are out of scope for this regulation. We encourage individuals to contact SSA to determine the options available to them.
Comment: Several commenters expressed support but also requested that CMS increase outreach measures specifically for this SEP. They recommended that CMS collaborate with stakeholders to distribute and engage in targeted outreach to communities with high rates of incarceration and reentry. One commenter also recommended that CMS put forth detailed educational resources for medical providers as well as guidance for states on these new policies. Another commenter requested that CMS coordinate directly with Medicaid to enroll dually eligible individuals.
Response: We thank the commenters for their support and feedback. CMS plans to continue to collaborate with stakeholders to educate and inform the targeted populations. Since this Medicare SEP is relatively new CMS has been working on several initiatives to engage stakeholders and conduct outreach and education on the availability of this new enrollment opportunity. In collaboration with the Medicare Beneficiary Ombudsman, we have conducted multiple presentations to key groups and updated relevant CMS web pages and materials. We will continue to engage partners and coordinate in how best to reach this population.
Comment: A commenter stated that requiring enrollment in person at an SSA office for the SEP can be challenging for individuals experiencing transportation challenges.
Response: This comment does not pertain to the revision of the SEP eligibility criteria and, thus, is out of scope.
3. Technical Corrections
In the November 2022 final rule that established the SEP for formerly incarcerated individuals, we provided at §§ 406.27(d)(3) and 407.23(d)(3) that, generally, entitlement would begin the first day of the month following the month of enrollment. We also provided that an individual had the option to choose a retroactive entitlement date for a period not to exceed 6 months, provided that the individual pays the monthly premiums for the period of coverage. Upon further examination of the regulations, we have identified a number of technical errors in §§ 406.27(d)(3) and 407.23(d)(3) that we are taking this opportunity to propose to correct.
First, the language in § 407.23(d)(3)(ii) states that the individual has the option to request entitlement retroactive to the date of release from incarceration and this implies that coverage could start in the middle of the month. Entitlement for Medicare, regardless of the enrollment period being used or whether entitlement is prospective or retrospective, always begins on the first day of a month. As such, we proposed to revise the language above to state that coverage could begin retroactive to the beginning of the month of release from incarceration. We note that the payment exclusion in § 411.4(b) may continue to apply to any items and services furnished during the period between the first of that month and the actual date of release, provided that the individual or other person has no legal obligation to pay for such services as articulated in § 411.4(a).
Second, in §§ 406.27(d)(3)(ii) and 407.23(d)(3)(ii), we erroneously cited § 406.31 when referencing the requirement for individuals to pay monthly premiums for all periods of coverage. We proposed to correct the reference to § 406.31 in § 406.27(d)(3)(ii) with § 406.32(f) and the reference in § 407.23(d)(3)(ii) to § 408.4.
Third, we also stated at § 407.23(d)(3)(ii) that if the individual requests retroactive enrollment and the application is filed within the first 6 months of the SEP, the effective date could be retroactive to the release from incarceration. If the individual requests retroactive enrollment and the application is filed in the last 6 months of the SEP, the coverage effective date could be retroactive to 6 months after the date of release from incarceration. This provision results in the same coverage effective date regardless of when the individual applies during the last 6 months of the SEP, which we do not think is consistent with our policy goal of providing formerly incarcerated individuals the ability to make the healthcare decisions best suited to their needs and provide them the opportunity to avoid or minimize gaps of coverage (87 FR 66463). We believe the best way to remedy this situation is to link the retroactive period of coverage to the date when the individual applies for Medicare coverage, not when they are released from incarceration. As such, we proposed to revise § 407.23(d)(3)(ii) to state that if the individual requests retroactive enrollment and the application is filed in the last 6 months of the SEP, the coverage effective date is retroactive to the 6th month before the month of enrollment. We believe the proposed approach strikes an appropriate balance of reducing gaps in coverage without creating excessive (and potentially costly) retroactive periods of coverage. We also proposed to make similar changes at § 406.27(d)(3)(ii) for the sake of consistency and clarity.
Comment: One commenter recommended that CMS add the phrase “no earlier than” before the description of when coverage begins in § 407.23(d)(3)(ii). The commenter stated that the inclusion of the proposed phrasing would further clarify an individual's ability to make the best decision about the length of retroactive coverage they need.
Response: We appreciate the commenter's recommendation and understand the importance of clarifying language so that individuals can make the decision that best suits them. However, circumstances where enrollees have the ability to pick a length of retroactive Medicare coverage would be unprecedented and would provide additional options for this population that is not available for other retroactive Medicare enrollments. Further, it would lead to operational complexity for SSA and potential enrollees to select their retroactive coverage start time for only this SEP, which may generate unnecessary confusion for individuals using this SEP. Therefore, we are declining the commenter's suggestion and will be ( printed page 94514) adopting this technical amendment as proposed.
After consideration of the public comments we received, and as discussed above, we are adopting our proposals with the following modifications:
- Sections 406.27(d)(2)(i) and 407.23(d)(2)(i) are being revised to explicitly state that individuals released from incarceration or confinement and transitioning to residence in halfway houses are not considered incarcerated or in confinement for the purposes of this SEP.
- Sections 406.27(d)(2)(i) and 407.23(d)(2)(i) are being revised to replace the term “discharge documents” with “documentation of discharge.”
XXIV. Overall Hospital Quality Star Rating Modification To Emphasize the Safety of Care Measure Group: Request for Information (RFI)
A. Summary
In the CY 2025 OPPS/ASC proposed rule (89 FR 59509), we sought public input on potential methodologic modifications regarding the Safety of Care measure group within the Overall Hospital Quality Star Rating published on the provider comparison tool on Medicare.gov ( https://www.medicare.gov/care-compare/ ). Patient safety constitutes a fundamental component of the CMS National Quality Strategy, representing a sustained commitment to fostering optimal health outcomes and ensuring the safest possible care for all patients.[714] This RFI, hereinafter referred to as “Overall Star Rating RFI,” gathered broad public input on increasing the Safety of Care measure group's contribution to the Overall Hospital Quality Star Rating. We also noted our intention to potentially issue additional RFIs or undertake rulemaking on this topic in the future.
B. Background
The Overall Hospital Quality Star Rating provides a summary of certain existing hospital quality information on Medicare.gov based on publicly available quality measure results reported through CMS' hospital quality measurement programs, by assigning hospitals between one and five stars, a way that is simple and easy for patients to understand (85 FR 86193). The Overall Hospital Quality Star Rating methodology was developed and is maintained according to the guiding principles of scientific validity, maximizing inclusion of hospitals and measure information, accounting for heterogeneity of available measures and hospital reporting, accommodating changes in the underlying measures, aligning with CMS hospital quality measure programs to the extent feasible, transparency of the methodology, and responsiveness to input from interested parties. The Overall Hospital Quality Star Rating was first introduced and reported on our Hospital Compare website in July 2016 (now reported on Medicare.gov ) and has been refreshed multiple times, with the most current refresh in July 2024.[715]
In the CY 2021 OPPS/ASC final rule with comment period (85 FR 86193), we codified the Overall Hospital Quality Star Rating methodology, including several methodology refinements, intended to improve the simplicity and predictability of measure emphasis within the methodology over time, and comparability of ratings among hospitals. We also finalized the inclusion of Veterans Health Administration (VHA) hospitals and Critical Access Hospitals (CAHs) in the Overall Hospital Quality Star Rating. In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72233), we provided additional information on the previously finalized policy to incorporate VHA hospitals and finalized a proposal to amend 42 CFR 412.190 to revise how we would refresh the Overall Hospital Quality Star Rating annually.
C. Current Overall Hospital Quality Star Rating Methodology
Measures reported on the provider comparison tool on Medicare.gov ( https://www.medicare.gov/care-compare/) that meet the criteria for inclusion in the Overall Hospital Quality Star Rating are organized into five conceptually coherent measure groups: Safety of Care, Mortality, Readmission, and Patient Experience (all of which include outcome measures), and Timely and Effective Care (which includes a selection of process measures).
The current Overall Hospital Quality Star Rating methodology includes seven general steps. First, the direction of all included measures that indicate better performance with a lower score are reversed to uniformly indicate that a higher score indicates better performance for all the measures, and all measure scores are standardized to a single, common scale to account for differences in measure score units. Second, measures are arranged into measure groups. Each measure group contains a number of publicly reported measures to produce a robust measure group score, which are reflective of differences in hospital quality. Third, the measure group scores are calculated as a simple average of measure scores. Measure group scores are then standardized to a common scale making varying scores comparable. Fourth, the hospital summary score is calculated as a weighted average of measure group scores. Specifically, each measure group score is multiplied by the assigned weight for that group. The weighted measure group scores are then summated to generate the hospital summary score. If a hospital has no measure scores in a group (for example, by not achieving sufficient sample size in any of the measures), the weight is redistributed proportionally across the remaining groups. Fifth, minimum reporting thresholds are applied. To receive a Star Rating, hospitals must report at least three measures in at least three measure groups, one of which must be either the Mortality or Safety of Care measure groups. Sixth, peer grouping is applied. Hospitals are grouped into one of three peer groups based on the number of measure groups for which they report at least three measures: a three-measure peer group, a four-measure peer group, and a five-measure peer group. Seventh, a clustering algorithm is applied within each peer group to assign hospital summary scores to star ratings so that one star is the lowest and five stars is the highest.
For additional details regarding the methodology, we refer readers to § 412.190(d) and the Overall Hospital Quality Star Rating Methodology Reports, available at https://qualitynet.cms.gov/inpatient/public-reporting/overall-ratings/resources.
D. Safety of Care in Star Ratings
A foundational commitment of providing healthcare services is to ensure safety, as embedded in the centuries-old Hippocratic Oath, “First, do no harm.” Yet, the landmark reports “To Err is Human” and “Crossing the Quality Chasm” surfaced major deficits in healthcare quality and safety.[716 717] These reports resulted in widespread awareness of the alarming prevalence of patient harm and, over the past two decades, healthcare facilities implemented various interventions and ( printed page 94515) strategies to improve patient safety, with some documented successes.[718] Furthermore, the COVID-19 public health emergency (PHE) strained the healthcare system substantially, introducing new safety risks and negatively impacting patient safety in the normal delivery of care.[719 720]
Safety gaps and further risks in healthcare delivery were illuminated as a result of the COVID-19 PHE, revealing a lack of resiliency in the healthcare system.[721 722 723] Therefore, we are increasing efforts to emphasize the importance of patient safety for both patients and healthcare workers. To accomplish these goals, the Federal Government is taking a multi-pronged inter-Agency approach to improve safety. The Agency for Healthcare Research and Quality (AHRQ) on behalf of the Department of Health & Human Services (HHS) established the National Action Alliance to Advance Patient and Workforce Safety as a public-private collaboration to improve both patient and workforce safety and move towards zero harm in healthcare.[724] In September 2023, the President's Council of Advisors on Science and Technology (PCAST) published the “Report to the President: A Transformational Effort on Patient Safety,” with a call to action to renew “our nation's commitment to improving patient safety.” [725] The report put forth a recommendation as part of the call to action to “establish and maintain federal leadership for the improvement of patient safety as a national priority.” We also acknowledged a noticeable decline in patient safety measure scores during the COVID-19 PHE which reinforces the emphasis on patient safety established in several CMS initiatives, including the National Quality Strategy and Universal Foundation.[726 727] Additionally, hospitals report data on healthcare-associated infection (HAI) measures through a number of CMS quality programs, including the Hospital-Acquired Condition (HAC) Reduction and Hospital Value-Based Purchasing Programs. These programs are designed to improve patient quality of care and safety, as well as reduce complications and mortality, by rewarding hospitals that achieve high scores on measures, including HAI measures, and penalizing those that do not meet or exceed established performance standards.[728] However, it is possible in the current Overall Star Rating methodology for a hospital to score very low in the Safety of Care measure group yet still receive a high Star Rating due to their high performance in other measure groups. Therefore, in the CY 2025 OPPS/ASC proposed rule (89 FR 59510), we noted that we seek to explore potential adjustments to the Overall Hospital Quality Star Ratings methodology that would more greatly emphasize the measures within the Safety of Care measure group, in alignment with other CMS and HHS efforts to improve patient safety across all programs.
There are currently eight measures in the Safety of Care measure group, including six HAI measures (HAI-1 through HAI-6), one Complications measure after total hip or total knee replacement (Hip/Knee), and one composite adverse event measure (Patient Safety and Adverse Events Composite (PSI-90)). While this group of measures has been the same since the inception of the Overall Hospital Quality Star Rating, the specific safety measures included may be subject to change in the future. Measures reported on the provider comparison tool on Medicare.gov ( https://www.medicare.gov/care-compare/) undergo a rigorous development process which includes extensive measure testing, vetting by interested parties, evaluation by the Consensus-based Entity (which convenes the Partnership for Quality Measurement), and undergoing rulemaking for inclusion in CMS programs and public reporting. As such, the Overall Hospital Quality Star Rating methodology uses the measures as required under the CMS programs, with measure scores as reported on Medicare.gov at the time of the Overall Hospital Quality Star Rating calculation. Thus, any measures that are removed or suspended from one of the CMS hospital quality measure programs and not published on Medicare.gov would no longer be included. Similarly, any measures that are added to the CMS programs and displayed on Medicare.gov may be included in the Overall Hospital Quality Star Rating; for example, upcoming measures such as the Severe Obstetric Complication (87 FR 48780), Failure-to-Rescue (89 FR 35934), Hospital Harm-Severe Hypoglycemia (89 FR 35934), and Hospital Harm-Opioid-related Adverse Events (87 FR 48780) measures may be considered for inclusion in the Safety of Care measure group. The assessment presented here is based only on the current group of eight measures as listed above, but the Overall Hospital Quality Star Rating methodology is designed with the flexibility to accommodate such changes in the future.
The current methodology places the highest emphasis on the Safety of Care and Mortality measure groups. First, the measure group weights currently utilized in the Overall Hospital Quality Star Rating methodology are based on CMS policy and interested party feedback. Currently, the Safety of Care, Mortality, Readmission, and Patient Experience measure groups are each weighted 22 percent while the Timely and Effective Care measure group is weighted 12 percent (Table 177). Interested parties generally agreed that outcome measures should have more weight since they represent strong indicators of quality and are most important to patients in making healthcare decisions. Interested parties and stakeholders broadly considered the current weightings to be acceptable.
The Safety of Care and Mortality groups are further emphasized in the reporting threshold to receive a Star Rating: hospitals must report at least three measures in each of at least three measure groups, one of which must specifically be Safety of Care or ( printed page 94516) Mortality (85 FR 86228). This decision was partially informed by interested party feedback on the relative importance of patient safety and prevention of mortality.
Given the current ongoing efforts to advance patient safety, we investigated options to even further emphasize the patient safety measures in the Overall Hospital Quality Star Rating, above and beyond the emphasis of the current methodology.
We conducted an internal analysis utilizing data from the July 2023 refresh of the Overall Hospital Quality Star Rating to determine correlations between the Safety of Care measure group and performance in the Overall Hospital Quality Star Rating. There were 3,076 hospitals that met the criteria to receive a Star Rating. Among the 3,076 rated hospitals, 2,995 (97 percent) had at least one Safety of Care measure and therefore received a Safety of Care group score, while 2,615 (85 percent) had at least three Safety of Care measures. Our analysis showed a strong relationship between the Safety of Care measure group and the Star Rating. Hospitals that did well in Safety of Care tended to also do well on the Star Rating; however, there were a few hospitals that performed in the bottom quartile (lowest performing 25 percent) of the Safety of Care measure group that still received a 5-star rating. Of the 3,076 hospitals that received a Star Rating, 658 hospitals with at least three Safety of Care measures scored in the lowest quartile of the Safety of Care measure group and 19 hospitals received a 5-star rating, representing 0.6 percent of all rated hospitals (Table 175). An additional 94 hospitals fell into the lowest quartile of Safety of Care when the analysis was based on hospitals that reported just one or two Safety of Care measures. In general, these hospitals attained 5-star ratings despite poor Safety of Care performance by achieving high performance scores across the other measure groups.
We assessed reporting of individual Safety of Care measures and performance in the Safety of Care measure group by various hospital characteristics. We observed significant variation in the number of Safety of Care measures reported across different types of hospitals, typically with fewer measures for hospitals that have generally lower volume and so are less likely to reach sufficient case volume for individual measurements. Specifically: non-teaching hospitals, safety net hospitals,[729] critical access hospitals, smaller (<100 beds) hospitals, rural hospitals, and hospitals not qualifying for Medicare Disproportionate Share Hospital (DSH) payments were likely to report fewer Safety of Care measures compared to teaching, non-safety net, non-critical access, hospitals with 100+ beds, urban, and DSH-qualifying hospitals (Table 176). There was a broad distribution in performance scores across hospital types; however, certain hospital characteristics appeared to be associated with performance on the Safety of Care measure group. For example, smaller hospitals were more likely to fall toward the extremes of the performance score distribution while larger hospitals fell more toward the center, and safety net hospitals tended to fall into lower quartiles than non-safety net hospitals (Table 176).
( printed page 94517) ( printed page 94518)E. Potential Future Options to Greater Emphasize Patient Safety in the Overall Hospital Quality Star Rating
As part of the national commitment to improving patient safety, we sought feedback on whether hospitals that performed in the bottom quartile (lowest-performing 25 percent) in the Safety of Care measure group should be eligible to receive the highest 5-star rating. In the CY 2025 OPPS/ASC proposed rule, we noted that we are considering modifying the Overall Hospital Quality Star Rating methodology, specifically the Safety of Care measure group, to reinforce our dedication to emphasize patient safety across CMS. In this section, we discuss three options identified to modify the Overall Hospital Quality Star Rating methodology.
1. Reweighting the Safety of Care Measure Group
We conducted an internal analysis to explore the impact of modifying the weighting system for measure groups in the Overall Hospital Quality Star Rating utilizing data from the July 2023 refresh. Specifically, we explored increasing the weight assigned to the Safety of Care measure group from the current 22 percent to 30 percent while proportionally reducing the weights assigned to the other measure groups to examine the isolated effect of reweighting while otherwise adhering to the current methodology. The exact weighting values noted in the Overall Star Rating RFI are not prescriptive and could be adjusted based on interested party feedback to be more easily interpretable (for example, to 30 percent, 20 percent, and 10 percent), however, the results reported reflect this preliminary reweighting scenario that preserves proportionality between the remaining groups. Current and potential new weights for each measure group are detailed in Table 177.
( printed page 94519)Our analysis showed that by modifying the weight of the Safety of Care measure group to 30 percent, out of 3,076 hospitals, 213 hospitals would receive a higher Star Rating than when using the current weighting, while 233 hospitals would receive a lower Star Rating. Specifically, among the 752 rated hospitals in the lowest quartile of the Safety of Care measure group, 16 hospitals would achieve a higher Star Rating, while 133 hospitals would receive a lower Star Rating; only 3 of the 752 hospitals would receive a 5-star rating. Implementing this option would reduce the number of hospitals that perform poorly in Safety of Care yet still obtain the highest 5-star rating. However, reweighting the Safety of Care measure group would slightly reduce the influence of other measure groups on the Overall Hospital Quality Star Rating.
2. Policy-Based 1-Star Reduction for Poor Performance on Safety of Care
In the CY 2024 OPPS/ASC proposed rule (89 FR 59514), we explained that we are considering a post hoc policy-based adjustment that would reduce the Star Rating of any hospital in the lowest quartile of Safety of Care (based on at least three measure scores) by one star. Using 2023 Overall Hospital Quality Star Ratings data, applying a one-star reduction for all hospitals in the lowest quartile of Safety of Care with at least three safety measures would result in 530 hospitals, out of 3,076 hospitals, receiving a lower Star Rating. This option would emphasize safety through a new standard for all hospitals regardless of their Star Rating. Since the minimum Star Rating is one star, hospitals already getting one star would not get a further star reduction and therefore would effectively be exempt from this policy-based adjustment. Additionally, some hospitals that perform excellently in all other measure groups except the Safety of Care measure group would still receive a one-star reduction.
3. Reweighting the Safety of Care Measure Group Combined With a Policy-Based Star Rating Cap
In the CY 2024 OPPS/ASC proposed rule (89 FR 59514), we noted that we are also considering increasing the weight of the Safety of Care measure group to 30 percent (and proportionally reducing the weights assigned to the other measure groups, as described in Table 177) while also applying a policy that would limit hospitals in the lowest quartile of Safety of Care (based on at least three measure scores) to a maximum of four stars out of five. Using 2023 Overall Hospital Quality Star Ratings data, implementing a cap of four stars in the lowest quartile of Safety of Care with at least three safety measures combined with the reweighting for all hospitals would result in 235 hospitals, out of 3,076 hospitals, receiving a lower Star Rating and the reduction by 1 star for two hospitals in the lowest quartile of Safety of Care that would otherwise still receive a 5-star rating if only the reweighting solution was applied. This option provides a more targeted solution to the issue of hospitals performing poorly in Safety of Care receiving a 5-star rating and applies equally to all hospitals, reserving the 5-star rating for hospitals achieving a minimum threshold in Safety of Care.
We also explored alternative options for emphasizing patient safety, such as applying only the 4-star rating maximum or combining reweighting of the Safety of Care measure group with a policy-based 1-star reduction, however, these options did not effectively reach our goal of emphasizing patient safety. In our analysis, applying a 4-star rating maximum to hospitals in the lowest quartile of Safety of Care with at least three safety measures would have less impact, resulting in only 19 out of 3,076 hospitals receiving a lower Star Rating from five stars to four stars. Conversely, applying a combination of reweighting the Safety of Care measure group with a 1-star reduction may be considered an `over-correction', resulting in 635 out of 3,076 hospitals receiving a lower Star Rating with the greatest impact on hospitals already receiving two, three, or four stars in the current methodology.
Feedback solicited during fall 2023 from interested parties, including patients, patient advocates, technical experts, and clinicians, supported the increasing emphasis on Safety of Care in the Overall Hospital Quality Star Rating methodology.[730] However, there was varying feedback from interested parties on the methods to do so, with concerns including a decreased emphasis on the other measure groups, particularly Mortality, and the adequacy of the Safety of Care group measures as currently established to truly represent the experience of patient safety at a hospital.
F. Solicitation of Public Comment
We sought comments on potential modifications to the Safety of Care measure group in the Overall Hospital Quality Star Rating methodology. We requested input from interested parties on the following options: (1) reweighting the Safety of Care measure group; (2) applying a policy-based adjustment that reduces the Star Rating of any hospital in the lowest quartile of Safety of Care (based on at least three measures in the group) by one star; (3) reweighting the Safety of Care measure group combined with a policy-based 4-star rating maximum on Star Rating of any hospital in the lowest quartile of Safety of Care (based on at least three measures in the group). Specifically, we requested comment on the following questions:
- Do you support reweighting the Overall Hospital Quality Star Rating measure groups to give greater weight to Safety of Care as described in option 1? Do you agree with the potential new weights for each measure group (as shown in Table 177)? ( printed page 94520)
- Do you support reducing the Star Rating for hospitals with a low Safety of Care score as described in option 2? Do you agree with the potential policy to apply a 1-star reduction to all hospitals in the lowest quartile of Safety of Care?
- Do you support a combination of reweighting the Safety of Care measure group with a 4-star maximum on Star Rating as described in option 3?
- Do you have feedback or preference towards an approach of both up-scoring high performers and down-scoring poor performers as in options 1 and 3, or an approach of just down-scoring poor performers as in option 2?
- What are other methodological approaches that could be used to emphasize the Safety of Care measure group?
- With respect to the potential changes to the Overall Hospital Quality Star Rating methodology, are there any special considerations for small, rural or safety net hospitals (including Critical Access hospitals)?
Any modification to the Overall Hospital Quality Star Rating methodology would be addressed through future notice-and-comment rulemaking.
We received the following comments on this topic.
Comments on option 1, reweighting the Overall Hospital Quality Star Rating measure groups to give greater weight to Safety of Care are:
Several commenters supported the reweighting of the Safety of Care measure group, adding more emphasis on patient safety. One commenter supported the reweighting of the Safety of Care measure group and encouraged CMS to weigh it equally with the Mortality measure group. Another commenter also supported the reweighting of the Safety of Care measure group as it would allow the recalibration of this measure group, it would establish updated threshold benchmarks, and would better represent quality of care.
A few commenters expressed concern that increasing the weight of the Safety of Care measure group would de-emphasize the importance of other measure groups, particularly Patient Experience, Readmission, or Mortality. A commenter stated that equal weighting of the Mortality and Safety of Care measure groups aligns with the HVBP weighting and suggested using alternative weighting for the Safety of Care measure group could send conflicting signals.
A few commenters expressed concern that not all hospitals report on the same measures within the Safety of Care measure group. A commenter stated that hospitals that do not provide surgical services would not have the opportunity to report on certain measures within the measure group therefore, their score would rely solely on a few of the HAI's. The commenter stated increasing the weight of the Safety of Care measure group could reduce comparability between hospitals and produce less reliable ratings. Another commenter expressed concern that CMS did not take into consideration the consequences to hospitals that do not have data to report on the required number of safety measures.
Comments on option 2, reducing the Star Rating for hospitals with a low Safety of Care score with the potential policy to apply a 1-star reduction to all hospitals in the lowest quartile of Safety of Care are:
A commenter supported the removal of one star for low Safety of Care scores, and suggested hospitals with Safety of Care ratings in the lowest percentile be capped at no more than 4-stars. Another commenter agreed with the approach but believed hospitals with a 4-star rating still hold merit and only taking away one star does not significantly downgrade the ratings. Another commenter supported option 2, but suggested CMS add the new Patient Safety Structural Measure and assess if the current measures in the Safety of Care measure group truly captures the hospital's safety culture.
A few commenters stated that lowering the Star Rating by one star was unfair and overly punitive as well as oversimplified and would lead to hospitals receiving a double penalty. A few commenters noted that reducing the hospitals' Star Rating by one star would have negative impacts to the Star Rating distribution nationwide. Commenters noted that decreasing the Star Rating of hospitals performing poorly in the Safety of Care measure group by one star would not allow accurate comparison across hospitals and would not accurately reflect performance improvement, as there would always be a lowest quartile category, and CMS already penalizes for poor performance on safety measures in other programs. A commenter pointed to an in-house study which suggested this option would increase 1-star hospitals but not have any effect on 5-star hospitals. They noted this could create confusion for consumers and possibly cause reputational concerns for hospitals, especially if a once 5-star hospital was publicly reported as 4-stars. Commenters believed this approach would not take individual hospital nuances into account. A commenter suggested removing one star for poor performers would undermine the Star Rating, as a 4-star hospital that performed well across all measure groups would be compared with a 4-star hospital that did not perform well on the Safety of Care measure group.
Comments on option 3, reweighting the Safety of Care measure group and capping poor performers within the measure group at 4-stars are:
Several commenters supported this option. Commenters supported this option and noted that it is misleading to consumers if a hospital performing in the lowest quartile of this measure group could achieve 5-stars. Commenters believed that reweighting the Safety of Care measure group combined with a policy-based Star Rating cap would incentivize hospitals to invest in safety. A commenter suggested adding more patient safety measures but urged CMS to allow hospitals to test and validate any new measures before capping Star Ratings if this option is proposed. A commenter suggested increased performance and focus on patient safety would ultimately result in performance improvements in other measure groups such as Mortality and Readmissions.
A few commenters felt the reweighting of the Safety of Care measure group along with capping the Star Rating would have negligible impacts and would do a disservice to consumers when choosing a hospital as the Safety of Care measure group may not be as important to the consumer as other categories. A commenter believed this would cause confusion to consumers, as they may not understand why a hospital did not receive a 5-star rating.
We received the following comments on an approach of both up-scoring high performers and down-scoring poor performers as in options 1 and 3, or an approach of just down-scoring poor performers as in option 2:
A few commenters shared varying concerns with respect to an approach of both up-scoring high performers and down-scoring poor performers as in options 1 (reweighting the Safety of Care Measure Group) and option 3 (reweighting the Safety of Care measure group combined with a policy-based Star Rating cap), or an approach of just down-scoring poor performers as in option 2 (policy-based 1-Star reduction for poor performance on Safety of Care). One commenter expressed concern with an automatic “up-score” or “down-score” approach based on composite measure groupings noting that this approach would likely mislead consumers. Another commenter recommended a maximum of two stars ( printed page 94521) where there is poor performance in the Safety of Care measure group noting that subpar performance in safety should commensurately and inherently be reflected in a subpar Star Rating.
We received the following comments on special considerations for small, rural or safety net hospitals:
Several commenters were concerned with the impact on smaller hospitals, since at least three measures are required to score the Safety of Care measure group. A few commenters recommended CMS segment small, rural, and safety net hospitals, highlighting that smaller hospitals may report on fewer of the Safety of Care measures due to smaller populations, which could negatively impact their Star Rating. One commenter suggested adding additional safety measures relevant to rural hospitals to more accurately reflect patient safety performance and provide better comparisons among similar facilities.
We received the following comments on the composition of the Safety of Care measure group itself:
Several commenters shared concerns on the use of composite measures in the Safety of Care measure group, specifically PSI-90. One commenter suggested that the PSI-90 be split into individual measures and reweighted for patient safety focus; another commenter suggested removing PSI-90 and replacing it with other, non-composite PSI measures, such as Pressure Ulcer Rate (PSI-03), Iatrogenic Pneumothorax Rate (PSI-06), and Perioperative Hemorrhage and Hematoma Rate (PSI-09), which would allow more targeted and actionable information. A commenter stated the HAI measures should stand alone outside of the composite as their results get lost in the composite results and does not allow for easy comparison of like measures across hospitals. A commenter mentioned that the Safety of Care measure group has the most skewed measures among the five groups in the Overall Hospital Star Ratings and suggested a thorough review of the factors causing skewness in both patient safety and all other measures; another commenter recommended novel approaches to using broader safety measures that are not heavily affected by rare events. A commenter questioned whether the current Safety of Care measure group captures the safety culture of hospitals and was disappointed that CMS was only considering updating the methodology rather than assuring the safety culture of hospitals was taken into consideration; other commenters recommended ensuring all quality measures in the Safety of Care measure group accurately reflect patient and workforce safety.
We received the following comments on alternative methodological approaches that could be used to emphasize the Safety of Care measure group:
A few commenters were concerned that focusing on the Safety of Care measure group would result in excess penalties for hospitals doing well in other measure groups and would not reflect performance improvements in the Safety of Care measure group. One commenter recommended maintaining the current methodology and adding a special flag for hospitals in the lowest quartile for Safety performance, and a different flag for hospitals without a Safety score to highlight important information. Other commenters recommended equal weighting of the outcome-based Mortality, Readmission, and Safety of Care measure groups; incorporating social determinants of health as risk adjustment; and grouping hospitals on volume thresholds and surgical complexities rather the number of measure groups with at least three reported measures. A commenter suggested CMS explore placing more emphasis on patient safety in other programs, such as the Hospital-Acquired Condition Reduction Program (HACRP) and the Hospital Value-Based Purchasing (VBP) Program and was concerned that frequent changes to measures and methodologies could make it difficult for consumers to interpret Star Rating results.
We received the following general comments and recommendations for the Overall Hospital Quality Star Rating modification:
A commenter suggested adding comparison data, including percentile ranges to help identify and prioritize improvement efforts across the entire Star Rating program; another commenter recommended offering ratings by measure groups to give consumers more transparency about a provider's performance in specific areas. Other commenters recommended uniform application of a Star Rating cap across all measure groups, so poor performance in any measure group precludes hospitals from getting a 5-star rating; implementation of a simpler weighting system where the Safety of Care measure group is weighted at 30 percentage, Mortality, Readmission, and Patient Experience measure groups at 20 percentage each, and the Timely and Effective measure group at 10 percentage; delaying the proposal to modify the Overall Hospital Quality Star Rating and re-evaluating its options when more recent, non-COVID data are available; and reconsidering the methodology for determining peer groups.
Response: We thank commenters for their input and acknowledge their concerns and recommendations. We believe efforts to manage the issue of hospitals receiving a high Star Rating while performing in the lowest quartile of the Safety of Care measure group is critical to achieving our vision of emphasizing patient safety across CMS programs. We will take commenters' feedback into consideration as appropriate, in future potential rulemaking related to changes to the Overall Hospital Quality Star Rating.
XXV. Files Available to the Public Via the Internet
The Addenda to the OPPS/ASC proposed rules and final rules with comment period are published and available via the internet on the CMS website. In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59154), for CY 2019, we changed the format of the OPPS Addenda A, B, and C by adding a column titled “Copayment Capped at the Inpatient Deductible of $1,364.00” where we flag, through use of an asterisk, those items and services with a copayment that is equal to or greater than the inpatient hospital deductible amount for any given year (the copayment amount for a procedure performed in a year cannot exceed the amount of the inpatient hospital deductible established under section 1813(b) of the Act for that year). In the CY 2022 OPPS/ASC final rule with comment period (85 FR 86266), we updated the format of the OPPS Addenda A, B, and C by adding a column titled “Drug Pass-Through Expiration during Calendar Year” where we flagged, through the use of an asterisk, each drug for which pass-through payment was expiring during the calendar year on a date other than December 31. For CY 2025 and subsequent years, we proposed to retain these columns that are updated to reflect the drug codes for which pass-through payment is expiring in the applicable year.
In the CY 2023 OPPS/ASC final rule with comment period (87 FR 72250) for CY 2023, we changed the format of the OPPS Addenda A, B, and C by adding a column titled “Drug Pass-Through Expiration during Calendar Year” to include devices, so that the column reads: “Drug and Device Pass-Through Expiration during Calendar Year” where we flagged, through the use of an asterisk, each drug and device for which pass-through payment was expiring ( printed page 94522) during the calendar year on a date other than December 31.
For CY 2024, we deleted the column titled “Copayment Capped at the Inpatient Deductible” and instead added a new column for “Adjusted Beneficiary Copayment” to identify any copayment adjustment due to either the inpatient deductible amount copayment cap or the inflation-adjusted copayment of a Part B rebatable drug per section 1833(t)(8)(F) and section 1833(i)(9) of the Act, as added by section 11101 of the Inflation Reduction Act (IRA). We also added another column for notes. The “Note” column contains multiple messages including, but not limited to, inflation-adjusted copayment of a Part B rebatable drug, the copayment for a code capped at the inpatient deductible, or 8 percent of the reference product add-on applied for a biosimilar.
In addition, for CY 2024, we updated the format of the OPPS Addenda A, B, and C by adding another column for “IRA Coinsurance Percentage” to identify the percentage for the inflation-adjusted copayment of a Part B rebatable drug per section 1833(t)(8)(F) and section 1833(i)(9) of the Act, as added by section 11101 of the Inflation Reduction Act (IRA).
For CY 2025 and subsequent years we proposed to keep the same format for the addenda A, B, and C, and we did not propose any additional changes for CY 2025.
We did not receive any public comments related to the format of the OPPS Addenda A, B, and C and are adopting the addenda format as proposed.
To view the Addenda to this final rule with comment period pertaining to CY 2025 payments under the OPPS, we refer readers to the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient/regulations-notices; select “CMS-1809-FC” from the list of regulations. All OPPS Addenda to this final rule with comment period are contained in the zipped folder titled “2025 NFRM OPPS Addenda” in the related links section at the bottom of the page. To view the Addenda to this final rule with comment period pertaining to CY 2025 payments under the ASC payment system, we refer readers to the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ambulatory-surgical-center-asc/asc-regulations-and-notices; select “CMS-1809-FC” from the list of regulations. The ASC Addenda to this final rule with comment period are contained in a zipped folder titled “2025 NFRM Addendum AA, BB, DD1, DD2, EE, and FF” in the related links section at the bottom of the page.
XXVI. Collection of Information Requirements
Under the Paperwork Reduction Act of 1995, we are required to provide 60-day notice in the Federal Register and solicit public comment before a collection of information requirement is submitted to the Office of Management and Budget (OMB) for review and approval. In order to fairly evaluate whether an information collection should be approved by OMB, section 3506(c)(2)(A) of the Paperwork Reduction Act of 1995 requires that we solicit comment on the following issues:
- The need for the information collection and its usefulness in carrying out the proper functions of our agency.
- The accuracy of our estimate of the information collection burden.
- The quality, utility, and clarity of the information to be collected.
- Recommendations to minimize the information collection burden on the affected public, including automated collection techniques.
We solicited public comment on each of these issues for the following sections of this document that contain information collection requirements (ICRs):
A. ICRs for the Hospital Outpatient Quality Reporting (OQR) Program
a. Background
In section XV of this final rule with comment period, we discuss the requirements for the Hospital OQR Program. The Hospital OQR Program is generally aligned with the CMS quality reporting program for hospital inpatient services known as the Hospital Inpatient Quality Reporting (IQR) Program. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82131 through 82140) for detailed discussions of the previously finalized Hospital OQR Program ICRs which are currently approved under OMB control number 0938-1109 (expiration date February 28, 2025).
In this final rule with comment period, we are adopting four web-based measures as proposed that would impact previously approved burden estimates: (1) the Hospital Commitment to Health Equity (HCHE) measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period and mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (4) the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM), beginning with voluntary reporting for the CY 2026 reporting period and mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination.
We are also finalizing changes to the Hospital OQR Program that will not impact the previously approved burden estimates. We are removing two claims-based measures beginning with the CY 2025 reporting period/CY 2027 payment determination: (1) Magnetic Resonance Imaging (MRI) Lumbar Spine for Low Back Pain measure; and (2) Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure. We are further modifying the public reporting of data for the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients)—Psychiatric/Mental Health Patients stratification so that it may be published on the Compare tool hosted by HHS, available at: https://www.medicare.gov/care-compare/, or its successor website(s), in addition to the Provider Data Catalog downloadable files, available at: data.cms.gov/provider-data/, beginning in CY 2025. Lastly, we are requiring electronic health record (EHR) technology to be certified to all electronic clinical quality measures (eCQMs) available to report beginning with the CY 2025 reporting period/CY 2027 payment determination.
In the CY 2024 OPPS/ASC final rule with comment period, we calculated reporting burden estimates for the Hospital OQR Program by utilizing the Bureau of Labor Statistics (BLS) mean hourly wage rate for Medical Records Specialists (88 FR 82132). Specifically, we used the “general medical and surgical hospitals” industry to estimate the mean wage, as this categorization aligns the closest with the Hospital OQR Program care setting compared to other industries, such as “office of physicians” or “nursing care facilities.” The most recent data from BLS' May 2023 National Occupational Employment and Wage Estimates reflects a mean hourly wage of $27.69 per hour for medical records specialists ( printed page 94523) working in “general medical and surgical hospitals” (SOC 29-2072).[731] We calculated the cost of overhead, including fringe benefits, at 100 percent of the mean hourly wage, consistent with previous years. This is a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($27.69 × 2 = $55.38) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we calculate cost burden to hospitals using a wage plus benefits estimate of $55.38 per hour throughout the discussion in this section of this rule for the Hospital OQR Program.
In the CY 2024 OPPS/ASC final rule with comment period, our burden estimates were based on an assumption that approximately 3,350 hospital outpatient departments (HOPDs) would report data to the Hospital OQR Program (88 FR 82132). For this final rule with comment period, based on the most recent available data from the CY 2024 Hospital OQR Program payment determination, we estimate that 3,200 HOPDs will report data to the Hospital OQR Program for the CY 2025 reporting period/2027 payment determination.
b. Information Collection Burden Estimate for the Adoption of the Hospital Commitment to Health Equity Measure Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
As discussed in section XIV.B.1 of this final rule with comment period, we are adopting the web-based HCHE measure beginning with the CY 2025 reporting period/CY 2027 payment determination. For this measure, HOPDs will be required to report once annually on attestations of “yes” or “no” to a set of five domains related to organizational efforts towards health equity, using a CMS-designated information system, as described in section XIV.B.1.b of this final rule with comment period.
We estimate the reporting burden associated with this measure to be, on average across all 3,200 HOPDs, no more than 10 minutes per HOPD per year, as we believe the burden for HOPDs to report this measure will be very similar to the burden for hospital inpatient departments to report the same measure once annually under the Hospital IQR Program. We refer readers to the currently approved burden estimate for the HCHE measure in the Hospital IQR Program under OMB control number 0938-1022 (expiration date January 31, 2026) and as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49385).
Using an estimate of 10 minutes (or 0.167 hours) per HOPD per year, we estimate that this measure adoption will result in a total annual burden increase of 533 hours (0.167 hours × 3,200 HOPDs) at a cost of $29,518 (533 hours × $55.38/hr) across program-eligible HOPDs.
We received no comments on our burden estimates.
c. Information Collection Burden Estimate for the Adoption of the Screening for Social Drivers of Health (SDOH) Measure Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Payment Determination
In section XIV.B.2 of this final rule with comment period, we are adopting the web-based Screening for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination. For this measure, HOPDs will be required to report whether they screened patients for five health-related social need (HRSN) domains, as described in section XIV.B.2.a of this final rule with comment period.
HOPDs will be able to collect data for the measure using a self-selected screening tool. We expect that most HOPDs will likely collect data through a screening tool incorporated into their EHR or other patient intake process, such as those we describe as examples in section XIV.B.2.e of this final rule with comment period. We estimate the information collection burden related to conducting patient screening associated with this measure to be two minutes (0.033 hours) per patient. This is based on the currently approved burden estimate for the Hospital IQR Program under OMB control number 0938-1022 for the same measure with patient screening for the same HRSN domains and the same frequency of data reporting, as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49385 through 49386).
In the CY 2025 OPPS/ASC proposed rule, we estimated 498,843,518 patients would be screened annually when the measure becomes mandatory (89 FR 59517). In this final rule, we have revised our methodology to account for multiple HOPD visits per patient and the corresponding reduction in screenings per reporting period. To provide an estimate of patient volume for the purposes of calculating the information collection burden associated with this measure, we utilized data derived from the American Hospital Association which estimates 2,399 outpatient visits per 1,000 population in CY 2022 [732] and multiplied this by the estimated total U.S. population in CY 2022 [733] to estimate the total number of outpatient visits across the hospitals surveyed. We estimate that each year there are approximately 799,556,849 hospital outpatient visits ((2,399 hospital outpatient visits per 1,000 population in CY 2022 [734] ) × 333,287,557 total U.S. population in 2022). We then estimate a total of 412,651,290 HOPD patient visits potentially resulting in a patient needing to be screened when the measure becomes mandatory by multiplying the total 799,556,849 (hospital outpatient visits by a ratio of 3,200 HOPDs to the total of 6,200 hospitals surveyed [735] (799,556,849 hospital outpatient visits × 51.61 percent (3,200 HOPDs ÷ 6,200 hospitals surveyed)). We anticipate that the estimate of 412,651,290 is likely an overestimate due to the policy we are finalizing which allows for the use of screening information collected from other settings in the same reporting period, such as the hospital inpatient setting, and the expectation that some patients may have more than one outpatient visit in a reporting period at the same facility, and therefore, their information would have already been collected. To help mitigate the potentially significant impact to our estimates of repeat visits where re-screening would be unnecessary, such as in a follow-up visit, routine treatments, or multiple emergency department encounters, we divided the total number of HOPD patient visits by two. As a result, we estimate a total of ( printed page 94524) 206,325,645 HOPD patient visits (412,651,290 ÷ 2) per reporting period when the measure becomes mandatory. We intend to assess actual reported volumes of visits for future estimates. As submission rates among hospitals may vary, we conservatively estimate that for voluntary reporting for the CY 2025 reporting period, 50 percent of HOPDs will screen 50 percent of patients, and beginning with the first mandatory reporting period, 100 percent of HOPDs will screen 100 percent of patients.
We determine the cost for patients (or their representative) undertaking administrative and other tasks, such as filling out a survey or intake form, using a post-tax wage of $24.49/hr based on the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices,” which identifies the approach for valuing time when individuals undertake activities on their own time.[736] To derive the costs for patients (or their representatives), a measurement of the usual weekly earnings of wage and salary workers of $1,139 is divided by 40 hours to calculate an hourly pre-tax wage rate of $28.48/hr.[737] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre-and post-tax income,[738] resulting in the post-tax hourly wage rate of $24.49/hr. Unlike our state and private sector wage adjustments, we are not adjusting patient wages for fringe benefits and other indirect costs since the individuals' activities, if any, will occur outside the scope of their employment.
Measure data aggregated to the hospital level as a numerator and a denominator will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the Hospital OQR Program under OMB control number 0938-1109, we estimate a burden of 10 minutes per HOPD to report the measure data. Therefore, we estimate that each HOPD will spend 10 minutes (0.167 hours) annually to report the Screening for SDOH measure data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total burden increase for patients of 1,719,380 hours (206,325,645 patients × 50 percent response rate × 50 percent of HOPDs × 0.033 hours per patient) at a cost of $42,107,616 (1,719,380 hours × $24.49/hr). Beginning with the CY 2026 mandatory reporting period, we estimate an annual total burden increase for patients of 6,877,522 hours (206,325,645 patients × 0.033 hours per patient) at a cost of $168,430,514 (6,877,522 hours × $24.49/hr).
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase for program-eligible HOPDs of 267 hours (3,200 HOPDs × 50 percent of HOPDs × 0.167 hours per HOPDs) at a cost of $14,786 (267 hours × $55.38/hr). Beginning with the CY 2026 mandatory reporting period, we estimate a total collection and reporting burden increase for program-eligible HOPDs of 533 hours (3,200 HOPDs × 0.167 hours per HOPD) at a cost of $29,518 (533 hours × $55.38/hr).
We received no comments on our burden estimates in the proposed rule.
d. Information Collection Burden Estimate for the Adoption of the Screen Positive Rate for Social Drivers of Health (SDOH) Measure Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Payment Determination
In section XIV.B.3 of this final rule with comment period, we are adopting the Screen Positive Rate for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination. We refer readers to the currently approved burden estimate for the Screen Positive Rate for SDOH measure in the Hospital IQR Program under OMB control number 0938-1022 for the same measure and the same frequency of data reporting, as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49386). As discussed in section XIV.B.3.g of this final rule with comment period, if a hospital participates in both the Hospital OQR and Hospital IQR Programs, the hospital will need to submit data on this measure separately under each program. As such, we are estimating the burden separately under each program.
For this measure, HOPDs will be required to report on the number of patients who screened positive for one or more of the five domains (reported as five separate rates to reflect each of the five HRSN domains) divided by the total number of patients screened. We previously included the collection burden associated with screening patients in our discussion of the Screening for SDOH measure. Thus, for the Screen Positive Rate for SDOH measure, we estimate only the additional burden for HOPD reporting via the HQR system since patients will not need to provide, and HOPDs will not need to collect, any additional information for this measure. We continue to estimate that, for voluntary reporting for the CY 2025 reporting period, 50 percent of HOPDs will submit data, and beginning with the first mandatory reporting period, 100 percent of HOPDs will submit data.
Measure data aggregated to the hospital level as a numerator and a denominator will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the Hospital OQR Program under OMB control number 0938-1109, we estimate a burden of 10 minutes per HOPD to report the measure data. Therefore, we estimate that each HOPD will spend 10 minutes (0.167 hours) annually to report the Screen Positive Rate for SDOH measure data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase of 267 hours (0.167 hours × 3,200 HOPDs × 50 percent of HOPDs) at a cost of $14,786 (267 hours × $55.38), and beginning with the CY 2026 reporting period, we estimate a total annual collection and reporting burden increase for hospitals of 533 hours (0.167 hours × 3,200 HOPDs) at a cost of $29,518 (533 hours × $55.38/hr) across all program-eligible HOPDs.
We received no comments on our burden estimates. ( printed page 94525)
e. Information Collection Burden Estimate for the Adoption of the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM), Beginning With Voluntary Reporting for the CY 2026 Reporting Period and Mandatory Reporting Beginning With the CY 2027 Reporting Period/CY 2029 Payment Determination
In section XV.C.1.b of this final rule with comment period, we are adopting the Information Transfer PRO-PM beginning with voluntary reporting for the CY 2026 reporting period and mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination. The Information Transfer PRO-PM will use PRO data regarding recovery instructions, collected by HOPDs through a nine-item survey instrument administered to patients post-operatively. The modes of PRO data collection can include completion of the post-operative surveys electronically.
In the CY 2025 OPPS/ASC proposed rule, we estimated 498,843,518 patients would be screened annually when the measure becomes mandatory (89 FR 59518). In this final rule with comment period, we have revised our methodology to account for multiple HOPD visits per patient and the corresponding reduction in screenings per reporting period. To provide an estimate of patient volume for the purposes of calculating the information collection burden associated with this measure, we utilized data derived from the American Hospital Association related to hospital outpatient visits to estimate that each year there are roughly 799,556,849 hospital outpatient visits ((2,399 hospital outpatient visits per 1,000 population in CY 2022 [739] ) × 333,287,557 total U.S population in 2022 [740] ). We then estimate a total of 412,651,290 HOPD patient visits potentially resulting in a patient needing to be screened when the measure becomes mandatory by multiplying the total 799,556,849 hospital outpatient visits by a ratio of 3,200 HOPDs to the total of 6,200 hospitals surveyed [741] (799,556,849 hospital outpatient visits × 51.61 percent (3,200 HOPDs ÷ 6,200 hospitals surveyed)). However, as not all hospital outpatient visits are related to surgeries and procedures, and there are often multiple visits such as pre- and post-op visits associated with those that are, we estimate that 137,550,430 hospital outpatient visits (412,651,290 hospital outpatient visits ÷ 3 surgery or procedure-specific visits) would more realistically qualify for the cohort of this measure. We anticipate that this may be an overestimate and intend to assess actual reported volumes of visits for future estimates. As submission rates among hospitals may vary, we conservatively estimate that for voluntary reporting for the CY 2025 reporting period, 50 percent of HOPDs will survey 50 percent of patients, and beginning with the first mandatory reporting period, 100 percent of HOPDs will survey 100 percent of patients. While we are also allowing HOPDs to report a sample of at least 300 completed patient surveys, we are requiring all patients to be surveyed for this measure when mandatory reporting begins.
In the CY 2025 OPPS/ASC proposed rule, we estimated each patient would require an average of six minutes to complete the survey (89 FR 59518). In this final rule with comment period, we are revising our estimate to five minutes (0.083 hours) per patient based on more recent information.[742] As described in section XXIV.B.c of this final rule with comment period, for purposes of calculating patient burden, we determine the cost for patients (or their representatives) undertaking administrative and other tasks, such as filling out a survey or intake form, using a post-tax wage of $24.49/hr based on the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices,” which identifies the approach for valuing time when individuals undertake activities on their own time.[743] Unlike our state and private sector wage adjustments, we are not adjusting patient wages for fringe benefits and other indirect costs since the individuals' activities, if any, will occur outside the scope of their employment.
Measure data will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the Hospital OQR Program under OMB control number 0938-1109, we estimate a burden of 10 minutes per HOPD to report the measure data. Therefore, we estimate that each HOPD will spend 10 minutes (0.167 hours) annually to report the Information Transfer PRO-PM data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total burden for patients of 2,865,634 hours (137,550,430 patients × 50 percent response rate × 50 percent of HOPDs × 0.083 hours per patient surveyed) at a cost of $70,179,377 (2,865,634 hours × $24.49/hr). Beginning with the CY 2026 mandatory reporting period, we estimate an annual total burden for patients of 11,462,536 hours (137,550,430 patients × 0.083 hours per patient) at a cost of $280,717,507 (11,462,536 hours × $24.49/hr).
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden for program-eligible HOPDs of 267 hours (3,200 HOPDs × 50 percent of HOPDs × 0.167 hours per HOPDs) at a cost of $14,786 (267 hours × $55.38/hr). Beginning with the CY 2026 mandatory reporting period, we estimate a total collection and reporting burden for program-eligible HOPDs of 533 hours (3,200 HOPDs × 0.167 hours per HOPD) at a cost of $29,518 (533 hours × $55.38/hr).
We received no comments on our burden estimates in the CY OPPS/ASC proposed rule.
f. Information Collection Burden for the Removal of Two Claims-Based Measures
In sections XV.C.2.a and XV.C.2.b of this final rule with comment period, we are removing two claims-based measures beginning with the CY 2025 reporting period/CY 2027 payment determination: (1) MRI Lumbar Spine for Low Back Pain measure; and (2) Cardiac Imaging for Preoperative Risk ( printed page 94526) Assessment for Non-Cardiac, Low-Risk Surgery measure. Because these measures are calculated using Medicare fee-for-service (FFS) claims that are already reported to the Medicare program for payment purposes, removing these measures will not result in a change in burden associated with OMB control number 0938-1109.
We received no comments on our assumptions regarding burden for these measures calculated from administrative data.
g. Information Collection Burden To Publicly Report Data for the Median Time for Discharged ED Patients—Psychiatric/Mental Health Patients Stratification on Care Compare Beginning in CY 2025
In section XV.F.2 of this final rule with comment period, we are finalizing the public reporting of data for the Median Time for Discharged ED Patients—Psychiatric/Mental Health Patients stratification on Care Compare beginning in CY 2025. Because we are not requiring HOPDs to collect or submit any additional data for purposes of this public reporting, this policy will not result in a change in burden associated with OMB control number 0938-1109.
We received no comments on our assumptions regarding burden.
h. Information Collection Burden To Require EHR Technology To Be Certified to All eCQMs Available To Report Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
In section XV.E.2.b of this final rule with comment period, we are requiring EHR technology to be certified to all eCQMs available to report beginning with the CY 2025 reporting period/CY 2027 payment determination. We do not expect HOPDs will experience an increase in information collection burden associated with this policy because the use of EHR technology that is certified to all available eCQMs is already required for the Promoting Interoperability Program (83 FR 41672) and the Hospital IQR Program (84 FR 42604).
We received no comments on our assumptions regarding burden.
g. Summary of Information Collection Burden Estimates for the Hospital OQR Program
In summary, we estimate that the policies in this final rule with comment period will result in a total HOPD burden increase of 18,342,190 hours at a cost of $449,266,093 annually for all 3,200 program-eligible HOPDs from the CY 2025 reporting period/CY 2027 payment determination through the CY 2027 reporting period/CY 2029 payment determination. We will submit the revised information collection estimates to OMB for approval under OMB control number 0938-1109. (See Tables 178, 179, and 180.)
( printed page 94527) ( printed page 94528) ( printed page 94529)We requested comment on how we can reduce burden on HOPDs for both these new information collections as well as recommendations for the removal of existing information collections to offset these new burdens. We received no comments regarding the burden estimates associated with these new information collections; however, some commenters expressed their belief that CMS underestimates the burden of requirements for the Hospital OQR Program, in general. A few commenters recommended removal of measures in general to reduce information collections burden currently included in the Hospital OQR Program. We thank the commenters for their input but disagree that the burden of the Hospital OQR Program is underestimated, as this burden has been estimated in a manner aligned with other quality reporting programs, such as the Hospital IQR Program, and measure type burden has been approved under OMB control number 0938-1109. This burden accurately reflects the reporting burden estimate for the Hospital OQR Program. We appreciate their recommendations ( printed page 94530) and may consider them in future rulemaking.
B. ICRs for the Rural Emergency Hospitals Quality Reporting (REHQR) Program
a. Background
In section XVI of this final rule with comment period, we discuss the requirements for the REHQR Program. The REHQR Program is generally aligned with the CMS quality reporting program for HOPDs known as the Hospital OQR Program. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82148 through 82149) for detailed discussions of the previously finalized REHQR Program ICRs, which are currently approved under OMB control number 0938-1454 (expiration date April 30, 2027).
In this final rule with comment period, we are adopting three web-based measures that will impact previously approved burden estimates: (1) the Hospital Commitment to Health Equity (HCHE) measure, beginning with the CY 2025 reporting period/CY 2027 program determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination.
We are also finalizing our proposal to extend the reporting period for the previously adopted Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure beginning with the CY 2027 program determination. We believe this policy will not impact the previously approved burden estimates.
In the CY 2024 OPPS/ASC final rule with comment period, we calculated reporting burden estimates for the REHQR Program by utilizing the BLS mean hourly wage rate for Medical Records Specialists (88 FR 82148). Specifically, we used the “general medical and surgical hospitals” industry to estimate the mean wage, as this categorization aligns the closest with the REHQR Program care setting compared to other medical record specialist related industries, such as “office of physicians” or “nursing care facilities.” The most recent data from BLS' May 2023 National Occupational Employment and Wage Estimates reflects a mean hourly wage of $27.69 per hour for medical records specialists working in “general medical and surgical hospitals” (SOC 29-2072).[744] We calculated the cost of overhead, including fringe benefits, at 100 percent of the mean hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($27.69 × 2 = $55.38) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to REHs using a wage plus benefits estimate of $55.38 per hour throughout the discussion in this section of this rule for the REHQR Program.
In the CY 2024 OPPS/ASC final rule with comment period, our burden estimates were based on an assumption that approximately 746 hospitals could transition to REH status assuming that all eligible hospitals in states which have passed or amended necessary legislation enabling transition to occur as of March 2023 choose to do so and we stated that we would update our estimates once more information was made available (88 FR 82148). For this final rule with comment period, based on the actual number of acute care and critical access hospital conversions to REH status as of September 27, 2024, we estimate that 33 REHs will report data to the REHQR Program during the CY 2025 reporting period unless otherwise noted. While the exact number of REHs required to submit data may vary due to status changes to and from an REH, as reiterated in section XVI.A of this final rule with comment period, REHs are required by statute to submit quality data. Therefore, for purposes of estimating burden, we assume that all 33 REHs will submit data under the REHQR Program beginning with the CY 2025 reporting period.
b. Information Collection Burden for the Adoption of the Hospital Commitment to Health Equity (HCHE) Measure Beginning With the CY 2025 Reporting Period/CY 2027 Program Determination
In section XIV.B.1 of this final rule with comment period, we are adopting the web-based HCHE measure beginning with the CY 2025 reporting period/CY 2027 program determination. For this measure, REHs will be required to report on attestations of “yes” or “no” to a set of five domains related to organizational efforts towards health equity, as described in section XIV.B.1.b of this final rule with comment period.
We estimate the reporting burden associated with this measure to be, on average across all 33 REHs, no more than 10 minutes per REH per year, as we believe the burden that is annually reported by hospital inpatient departments under the Hospital IQR Program will be very similar to annual reporting by REHs on the same measure. We refer readers to the currently approved burden for the HCHE measure in the Hospital IQR Program under OMB control number 0938-1022 (expiration date January 31, 2026) and as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49385).
Using an estimate of 10 minutes (or 0.167 hours) per REH per year, we estimate that this measure adoption will result in a total annual burden increase of six hours (0.167 hours × 33 REHs) at a cost of $332 (6 hours × $55.38/hr) across all REHs.
We received no comments on our burden estimates.
c. Information Collection Burden for the Adoption of the Screening for Social Drivers of Health (SDOH) Measure Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Program Determination
In section XIV.B.2 of this final rule with comment period, we are adopting the Screening for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination. We refer readers to the currently approved burden for the Screening for SDOH measure for the Hospital IQR Program under OMB control number 0938-1022. For this measure, REHs will be required to report whether they screened patients for five health-related social need (HRSN) domains as described in section XIV.B.2.a of this final rule with comment period.
REHs will be able to collect data for the measure using a self-selected screening tool. We expect that most REHs will collect data through a screening tool incorporated into their EHR or other patient intake process, such as those we describe as examples in section XIV.B.2.e of this final rule with comment period. We estimate the information collection burden related to conducting patient screening associated ( printed page 94531) with this measure to be two minutes (0.033 hours) per patient. This is based on the currently approved burden estimate for the Hospital IQR Program for the same measure with patient screening for the same HRSN domains and the same frequency of data reporting, as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49385 through 49386).
To provide an estimate of patient volume for the purposes of calculating the information collection burden associated with this measure, we utilized data derived from a Definitive Healthcare survey which calculated that Medicare FFS patients account for 35.6 percent of hospital payer mix and a MedPAC report that determined hospitals which have converted to REH status average 4,200 outpatient visits for Medicare FFS beneficiaries to estimate that each year 11,798 (4,200 ÷ 35.6 percent) patients will be screened per REH when reporting on the measure becomes mandatory.[745 746] We therefore estimate a total of approximately 389,334 patients (11,798 patients × 33 REHs) will be screened across all 33 REHs. As submission rates among hospitals may vary, we conservatively estimate that for voluntary reporting for the CY 2025 reporting period, 50 percent of REHs will screen 50 percent of patients, and beginning with the first mandatory reporting period, REHs will screen 100 percent of patients.
We determine the cost for patients (or their representative) undertaking administrative and other tasks, such as filling out a survey or intake form, using a post-tax wage of $24.49/hr based on the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices,” which identifies the approach for valuing time when individuals undertake activities on their own time.[747] To derive the costs for patients (or their representatives), a measurement of the usual weekly earnings of wage and salary workers of $1,139 is divided by 40 hours to calculate an hourly pre-tax wage rate of $28.48/hr.[748] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income,[749] resulting in the post-tax hourly wage rate of $24.49/hr. Unlike our State and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs since the individuals' activities, if any, will occur outside the scope of their employment.
Measure data aggregated to the hospital level as a numerator and a denominator will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the Hospital OQR Program under OMB control number 0938-1109 (expiration date February 28, 2025), which REHs will have been eligible to report under prior to conversion to REH status, we estimate a burden of 10 minutes per REH to report the measure data. Therefore, we estimate that each REH will spend 10 minutes (0.167 hours) annually to report the Screening for SDOH measure data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase for patients of 3,343 hours (11,798 patients × 50 percent response rate × 17 REHs × 0.033 hours per patient) at a cost of $81,870 (3,343 hours × $24.49/hr). Beginning with the CY 2026 reporting period, we estimate a total collection and reporting burden increase for patients of 12,978 hours (389,334 patients × 0.033 hours per patient) at a cost of $317,831 (12,978 hours × $24.49/hr).
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase for REHs of three hours (33 REHs × 50 percent of REHs × 0.167 hours) at a cost of $166 (3 hours × $55.38/hr). Beginning with the CY 2026 reporting period, we estimate a total collection and reporting burden increase for REHs of six hours (33 REHs × 0.167 hours) at a cost of $332 (4 hours × $55.38/hr).
We received no comments on our burden estimates.
d. Information Collection Burden for the Adoption of the Screen Positive Rate for Social Drivers of Health (SDOH) Measure Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Program Determination
In section XIV.B.3 of this final rule with comment period, we are adopting the Screen Positive Rate for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 program determination. We refer readers to the currently approved burden estimate for the Screen Positive Rate for SDOH measure in the Hospital IQR Program under OMB control number 0938-1022 for the same measure and the same frequency of data reporting, as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49386).
For this measure, REHs will be required to report on an annual basis the number of patients who screened positive for one or more of the five domains (reported as five separate rates to reflect each of the five HRSN domains) divided by the total number of patients screened.
We previously included the burden associated with screening patients in our discussion of the Screening for SDOH measure. Thus, for the Screen Positive Rate for SODH measure, we estimate only the additional burden for a REH reporting via the HQR system since patients will not need to provide, and REHs will not need to collect, any additional information for this measure. We continue to estimate that, for voluntary reporting for the CY 2025 reporting period, 50 percent of REHs will screen 50 percent of patients, and beginning with the first mandatory period, REHs will survey 100 percent of patients.
Measure data aggregated to the hospital level as a numerator and a denominator will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the Hospital OQR Program under OMB control number 0938-1109, which REHs will have been eligible to report under prior to conversion to REH status, we estimate a burden of 10 minutes per REH to report the measure data. Therefore, we estimate that each REH will spend 10 minutes (0.167 hours) annually to report ( printed page 94532) the Screen Positive Rate for SDOH measure data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase of three hours (0.167 hours × 33 REHs × 50 percent of REHs) at a cost of $166 (3 hours × $55.38/hr), and beginning with the CY 2026 reporting period, we estimate a total annual collection and reporting burden increase for REHs of six hours (0.167 hours × 33 REHs) at a cost of $332 (6 hours × $55.38/hr) across all REHs.
We received no comments on our burden estimates.
e. Information Collection Burden To Extend the Reporting Period From for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery Measure Beginning With the CY 2027 Program Determination
In section XVI.C.2 of this final rule with comment period, we are extending the reporting period from one year to two years for the Risk-Standardized Hospital Visits within 7 Days after Hospital Outpatient Surgery measure, beginning with the CY 2027 program determination. We refer readers to a similar policy which was finalized for the claims-based Facility 7-Day Risk-Standardized Hospital Visit Rate After Outpatient Colonoscopy measure under the Hospital OQR Program in the CY 2019 OPPS/ASC final rule (83 FR 59106).
Because this claims-based measure is calculated using data that are already reported to the Medicare program for payment purposes, there is no burden associated with the collection and submission of data for this measure. Accordingly, our policy to extend the reporting period from one to two years will not result in additional burden for REHs.
We received no comments on our assumptions regarding burden.
f. Summary of Information Collection Burden Estimates for the REHQR Program
In summary, we estimate that the policies in this final rule with comment period will result in an increase of 12,996 hours at a cost of $318,827 for 33 REHs annually from the CY 2025 reporting period through the CY 2026 reporting period. We will submit these information collection estimates to OMB for approval under OMB control number 0938-1454. (See Tables 181 and 182.)
( printed page 94533)We requested comment on how we can reduce burden on REHs for both these new information collections as well as recommendations for the removal of existing information collections to offset these new burdens. We received neither comments regarding the burden estimates for these new information collections nor recommendations for removal of existing information collections under the REHQR Program.
C. ICRs for the Ambulatory Surgical Center Quality Reporting (ASCQR) Program
a. Background
In section XVII of this final rule with comment period, we discuss the requirements for the ASCQR Program. We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82140 through 82148) for detail regarding the previously finalized ASCQR Program ICRs which are currently approved under OMB control number 0938-1270 (expiration date August 31, 2025).
In section XIV.B of this final rule with comment period, we are adopting three measures that will impact previously approved burden estimates: (1) the Facility Commitment to Health Equity (FCHE) measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 ( printed page 94534) reporting period/CY 2028 payment determination. In section XVII.E.2.a of this final rule with comment period, we are finalizing that ASCs that ASCs will submit data annually for these measures using the CMS-designated information system (currently, the Hospital Quality Reporting [HQR] system).
In the CY 2024 OPPS/ASC final rule with comment period, we calculated reporting burden estimates for the ASCQR Program by utilizing the BLS mean hourly wage rate for Medical Records Specialists (88 FR 82140). Specifically, we used the “general medical and surgical hospitals” industry to estimate the mean wage, as this categorization aligns the closest with the ASCQR Program care setting compared to other medical record specialist related industries, such as “office of physicians” or “nursing care facilities.” The most recent data from BLS' May 2023 National Occupational Employment and Wage Estimates reflects a mean hourly wage of $27.69 per hour for medical records specialists working in “general medical and surgical hospitals” (SOC 29-2072).[750] We calculated the cost of overhead, including fringe benefits, at 100 percent of the mean hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, doubling the hourly wage rate ($27.69 × 2 = $55.38) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to ASCs using a wage plus benefits estimate of $55.38 per hour throughout the discussion in this section of this rule for the ASCQR Program.
Based on the most recent analysis of the CY 2024 payment determination data, we found that, of the 5,536 ASCs that were actively billing Medicare, 4,196 were required to participate in the ASCQR Program. Of the 1,340 ASCs not required to participate in the program, 279 ASCs did so and met full requirements. On this basis, we estimate that 4,475 ASCs (4,196 + 279) will submit data for the ASCQR Program for the CY 2025 reporting period/CY 2027 payment determination.
b. Information Collection Burden for the Adoption of the Facility Commitment to Health Equity (FCHE) Measure Beginning With the CY 2025 Reporting Period/CY 2027 Payment Determination
In section XIV.B.1 of this final rule with comment period, we are adopting the FCHE measure for the ASCQR Program beginning with the CY 2025 reporting period/CY 2027 payment determination. For this measure, ASCs will be required to report an attestation of “yes” or “no” to a set of five domains related to organizational efforts towards health equity, as described in section XIV.B.1.b of this final rule with comment period.
We estimate the reporting burden associated with this measure to be, on average across all 4,475 ASCQR Program eligible facilities, no more than 10 minutes per ASC per year, based on the currently approved burden for the same measure under the Hospital IQR Program under OMB control number 0938-1022 (expiration date January 31, 2026). This also aligns with our estimated burden per providers for HOPDs and REHs, discussed above.
Using an estimate of 10 minutes (0.167 hours) per ASC per year, we estimate that this measure adoption will result in a total annual collection and reporting burden increase of 746 hours (0.167 hours × 4,475 ASCs) at a cost of $41,313 (746 hours × $55.38/hr) across program-eligible ASCs.
We received no comments on our burden estimates.
c. Information Collection Burden for the Adoption of the Screening for Social Drivers of Health (SDOH) Measure Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Payment Determination
In section XIV.B.2 of this final rule with comment period, we are adopting the Screening for SDOH measure for the ASCQR Program beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination. For this measure, ASCs will be required to report whether they screened patients for five health-related social need (HSRN) domains as described in section XIV.B.2.a of this final rule with comment period.
As described in section XIV.B.2.e of this final rule with comment period, ASCs will be able to collect data for this measure using a self-selected screening tool. We expect that most ASCs will collect data through a screening tool incorporated into their EHR or other patient intake process, such as those we describe as examples in section XIV.B.2.e of this final rule with comment period. We estimate the information collection burden related to conducting patient screening associated with this measure will be 2 minutes (0.033 hours) per patient. This estimate is based on the currently approved burden for the Hospital IQR Program for the same measure, requiring the reporting of patient screening for the same HRSN domains and the same frequency of data reporting, as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49385 through 49386).
To provide an estimate of patient volume for the purposes of calculating the information collection burden associated with this measure, we utilized data derived from the ASC Quality Collaborative (ASCQC) related to ASC patient fall benchmarking data as this metric applies to all patients rather than a subset. In the CY 2025 OPPS/ASC proposed rule, we estimated approximately 10,427,619 patients would be screened annually (89 FR 59526). Based on public comments received, we are correcting our estimate. Since we expect that ASCs reporting data to the ASCQC will tend to be larger facilities with larger patient populations than non-reporting ASCs, we conservatively estimate that each year approximately 21,322,000 patients ((10,434,676 admissions [751] ÷ 2,190 ASCs reporting) × 4,475 ASCs) with an average of 4,765 patients per ASC (21,322,000 admissions ÷ 4,475 ASCs) will be screened annually when reporting on the measure becomes mandatory. As submission rates among facilities may vary, we conservatively estimate that, for voluntary reporting for the CY 2025 reporting period, 50 percent of ASCs will survey 50 percent of patients, and beginning with the first mandatory reporting period, ASCs will survey and report on 100 percent of patients.
We determine the cost for patients (or their representative) undertaking administrative and other tasks, such as filling out a survey or intake form, using a post-tax wage of $24.49/hr based on the report “Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices,” which identifies the approach for valuing time when ( printed page 94535) individuals undertake activities on their own time.[752] To derive the costs for patients (or their representatives), a measurement of the usual weekly earnings of wage and salary workers of $1,139 is divided by 40 hours to calculate an hourly pre-tax wage rate of $28.48/hr.[753] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income,[754] resulting in the post-tax hourly wage rate of $24.49/hr. Unlike our state and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs because the individuals' activities, if any, will occur outside the scope of their employment.
Measure data aggregated to the ASC level as a numerator and a denominator will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the ASCQR Program under OMB control number 0938-1270, we estimate a burden of 10 minutes per ASC to report the measure data. Therefore, we estimate that each ASC will spend 10 minutes (0.167 hours) annually to report the Screening for SDOH measure data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase for patients of 177,723 hours (21,322,000 patients × 50 percent response rate × 50 percent of ASCs × 0.033 hours per patient) at a cost of $4,352,436 (177,723 hours × $24.49/hr). Beginning with the CY 2026 reporting period, we estimate a total collection and reporting burden increase for patients of 710,733 hours (21,322,000 patients × 0.033 hours per patient) at a cost of $17,405,851 (710,733 hours × $24.49/hr).
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase for program-eligible ASCs of 373 hours (4,475 ASCs × 50 percent of ASCs × 0.167 hours) at a cost of $20,657 (373 hours × $55.38/hr). Beginning with the CY 2026 reporting period, we estimate a total collection and reporting burden increase for program-eligible ASCs of 746 hours (4,475 ASCs × 0.167 hours) at a cost of $41,313 (746 hours × $55.38/hr).
Comment: One commenter stated that the burden estimate for this information collection was calculated in error due to CMS incorrectly using the number of 4,475 ASCs to determine the average number of patients per ASC instead of the correct value of 2,190 ASCs as stated in the referenced data source.
Response: We thank the commenter and have clarified our rationale and process for the burden estimate. In the CY 2025 OPPS/ASC proposed rule, we estimated a total of 10,427,619 patients would be screened when the measure becomes mandatory based on a total number of ASC admissions over a 12-month period as reported by the ASCQC (89 FR 59526). In this final rule with comment period, we have revised our calculation of the number of ASC admissions using the ASCQC data, dividing by the 2,190 ASCs reporting to ASCQC to calculate an average number of annual admissions per ASC and multiplying the average per ASC by 4,475 total ASCs which could report the measure under the ASCQR Program.
d. Information Collection Burden for the Adoption of the Screen Positive Rate for Social Drivers of Health (SDOH) Measure Beginning With Voluntary Reporting for the CY 2025 Reporting Period Followed by Mandatory Reporting Beginning With the CY 2026 Reporting Period/CY 2028 Payment Determination
In section XIV.B.3 of this final rule with comment period, we are adopting the Screen Positive Rate for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination. We refer readers to the currently approved burden for the Screen Positive Rate for SDOH measure in the Hospital IQR Program under OMB control number 0938-1022 for the same measure and the same frequency of data reporting, as discussed in the FY 2023 IPPS/LTCH PPS final rule (87 FR 49386). For this measure, ASCs will be required to report annually the number of patients who screened positive for one or more of the five domains (reported as five separate rates to reflect each of the five HRSN domains) divided by the total number of patients screened.
We previously included the burden associated with screening patients in our discussion of the Screening for SDOH measure. Thus, for the Screen Positive Rate for SDOH measure, we estimate only the additional burden for an ASC reporting via the HQR system since patients will not need to provide, and ASCs will not need to collect, any additional information for this measure. We continue to estimate that for voluntary reporting for the CY 2025 reporting period, for the 4,475 ASCs estimated for reporting purposed, 50 percent of these ASCs will screen 50 percent of patients, and beginning with the first mandatory reporting period, 100 percent of these ASCs will submit data.
Measure data aggregated to the hospital level as a numerator and a denominator will be submitted via the HQR system annually. Similar to the currently approved burden estimate for web-based measures reported via the HQR system for the ASCQR Program under OMB control number 0938-1270 (expiration date August 31, 2025), we estimate a burden of 10 minutes per ASC to report the measure data. Therefore, we estimate that each ASC will spend 10 minutes (0.167 hours) annually to report the Screen Positive Rate for SDOH measure data to CMS.
For the CY 2025 voluntary reporting period, we estimate a total collection and reporting burden increase of 373 hours (0.167 hours × 4,475 ASCs × 50 percent of ASCs) at a cost of $20,657 (373 hours × $55.38). Beginning with the CY 2026 reporting period, we estimate a total annual collection and reporting burden increase for ASCs of 746 hours (0.167 hours × 4,475 ASCs) at a cost of $41,313 (746 hours × $55.38/hr) across program-eligible ASCs.
We received no comments on our burden estimates.
e. Summary of Information Collection Burden Estimates for the ASCQR Program
In summary, we estimate that the policies in this final rule with comment period will result in an increase of 712,971 hours at a cost of $17,529,790 for 4,475 program-eligible ASCs from the CY 2025 reporting period/CY 2027 payment determination through the CY 2026 reporting period/CY 2028 payment determination. We will submit the revised information collection estimates ( printed page 94536) to OMB for approval under OMB control number 0938-1270. (See Tables 183 and 184.)
( printed page 94537)D. ICRs Related to Medicaid Clinic Services Four Walls Exceptions
As discussed in section XVII of this final rule, we are finalizing our proposal for three additional exceptions to the four walls requirement under the Medicaid clinic services benefit at 42 CFR 440.90. Specifically, we are adding a mandatory four walls exception for IHS/Tribal clinics at § 440.90(c) and optional exceptions for behavioral health clinics and clinics located in rural areas at § 440.90(d) and (e). To attest to compliance with § 440.90(c) and to effectuate the options at § 440.90(d) and (e), States that cover the clinic services benefit will have to submit one or more Medicaid State plan amendments (SPAs).
The burden associated with submitting the SPAs implementing the proposed Medicaid clinic services four walls exceptions will be addressed as part of an associated SPA preprint being developed by CMS and submitted to OMB for approval under OMB control number 0938-1148 (CMS-10398).
E. ICRs for Changes to the Review Timeframes for Hospital Outpatient Department (OPD) Prior Authorization Process
In the CY 2020 OPPS/ASC final rule with comment period, we established a prior authorization process for certain hospital OPD services using our authority under section 1833(t)(2)(F) of the Act, which allows the Secretary to develop a method for controlling unnecessary increases in the volume of covered OPD services (84 FR 61142, 61446 through 61456).[755] As part of the CY 2021 OPPS/ASC final rule with comment period, we added additional service categories to the prior authorization process (85 FR 85866, 86236 through 86248). Through the CY 2023 OPPS/ASC final rule with comment period, we added an eighth service category to the prior authorization process for certain hospital OPD services (87 FR 71748, 72224 through 72233). The regulations governing the prior authorization process are located in subpart I of 42 CFR part 419, specifically at §§ 419.80 through 419.89.
In alignment with the CMS Interoperability and Prior Authorization final rule (89 FR 8758), we are finalizing our proposal to change the current review timeframes for provisionally affirmed or non-affirmed requests from 10-business days to 7-calendar days for standard reviews after receiving the prior authorization request for OPD services under Medicare FFS. The ICR associated with prior authorization requests for these covered outpatient department services is the required documentation submitted by providers. The prior authorization request must include all relevant documentation ( printed page 94538) necessary to show that the service meets applicable Medicare coverage, coding, and payment rules, and the request must be submitted before the service is provided to the beneficiary and before the claim is submitted for processing.
The burden associated with the changes in review timeframes for the OPD prior authorization process will be the time and effort necessary for the submitter to locate and obtain the relevant supporting documentation to show that the service meets applicable coverage, coding, and payment rules. The submitter will then forward the information to CMS or its contractor (MAC) for review and determination of a provisional affirmation. We expect that this information will generally be maintained by providers within the normal course of business and that this information will be readily available. We estimate that the average time for office clerical activities associated with this task will be 30 minutes, equivalent to normal prepayment or postpayment medical review. We anticipate that most prior authorization requests will be sent by means other than mail, such as electronically or by fax. However, we estimate a cost of $5 per request for mailing medical records. Based on data from 2019-2022, we estimate that there will be 127,397 initial requests mailed per year. In addition, we estimate there will be 41,806 resubmissions of a request mailed following a non-affirmed decision. Therefore, the total mailing cost is estimated to be $846,015 (169,203 mailed requests × $5). We also estimate that an additional 3 hours per provider will be required for attending educational meetings and reviewing training documents.
The average labor costs (including 100 percent fringe benefits) used to estimate the costs were calculated using data from the Bureau of Labor Statistics (BLS) and based on the 2022 median rate for Miscellaneous Healthcare Support Occupations.[756] Based on the BLS information, we estimate an average clerical hourly rate of $18.53 with a loaded rate of $37.06. The prior authorization program does not create any new documentation or administrative requirements. Instead, it just requires the same documents needed to support claim payments to be submitted earlier in the claim process. We use the clerical rate since we do not believe that clinical staff will need to spend more time completing the documentation than they would need to in the absence of the prior authorization policy. The hourly rate reflects the time needed for the additional clerical work of submitting the prior authorization request itself. We estimate that the total annual number of submissions will be 564,010 (394,808 submissions through fax or electronic means + 169,203 mailed submissions). Therefore, we estimate that the annual burden hours allotted across all providers will be 316,412 hours (.5 hours × 564,010 submissions plus 3 hours × 11,469 providers for education). The annual burden cost is $12,572,244 (316,412 hours × $37.06 plus $846,015 for mailing costs). We estimate the annual burden to be 316,412 hours and $12,572,244. The ICR approved under OMB control number 0938-1368 will be revised and submitted to OMB for approval of this extension.
Table 185 below is a chart reflecting the total burden and associated costs for the provisions included in this final rule with the comment period. The previously approved Paperwork Reduction Act package (CMS-10711) is currently undergoing the renewal process. We did not make any changes to the information collection, such as the number of respondents, responses, or other information collection requirements. However, there is a 1-hour change in the burden hours, from 316,413 to 316,412, likely due to rounding up in the previous year's calculations. The burden costs have increased from $11,561,950 to $12,572,244 due to an increase in the average clerical hourly rate from $17.13 in 2019 to $18.53 in 2022.
F. ICRs for the Hospital Inpatient Quality Reporting (IQR) Program
In section XXII.B.2 of this final rule with comment period, we are finalizing that for the FY 2026 payment determination, the submission of CCDEs and linking variables associated with the Hybrid Hospital-Wide Readmission (HWR) measure and the Hybrid Hospital-Wide All-Cause Risk Standardized Mortality (HWM) measure will remain voluntary. Additionally, based on public comment, we are extending voluntary reporting of CCDEs and linking variables for an additional year for the performance period of July 1, 2024, through June 30, 2025, impacting the FY 2027 payment determination for the Hospital IQR Program.
In the FY 2020 IPPS/LTCH PPS final rule and the FY 2022 IPPS/LTCH PPS final rule, we estimated the burden for voluntary reporting for the Hybrid HWR (84 FR 42603 and 42604) and Hybrid HWM measures (86 FR 45508), respectively. In both final rules, we stated that we encourage all hospitals to submit data for the Hybrid HWR and Hybrid HWM measures during the voluntary reporting period. Our previously finalized burden estimates assume that all hospitals will participate during the voluntary reporting period in order to not underestimate the burden on participating hospitals. Therefore, we estimate no changes to the burden currently approved for the Hospital IQR Program under OMB control number 0938-1022 (expiration date January 31, 2026).
We did not receive any public comments on our burden estimates.
G. ICRs for Continuous Eligibility (42 CFR 435.926 and 457.342)
In section XX of this final rule with comment period, we are finalizing our proposal to align the Medicaid and CHIP regulations with the continuous ( printed page 94539) eligibility requirements under section 5112 of Title V, subtitle B (hereafter, “section 5112”) of the CAA, 2023. To comply with section 5112 of the CAA, 2023, States must submit a CHIP SPA and a Medicaid SPA to provide continuous eligibility for children if they do not already do so in their CHIP or Medicaid State plans, or if their current continuous eligibility SPAs do not comply with the CAA, 2023 requirements. CMS has already received approval for the burden estimates for the CHIP continuous eligibility SPA under OMB control number 0938-1148 (CMS-10398) and for the Medicaid continuous eligibility SPA under OMB control number 0938-1188 (CMS-10434). States may provide extended continuous eligibility through an 1115 demonstration, which they would do by submitting a demonstration application or amendment.
We did not receive any public comments on the information collection requirements for our proposal and therefore, we are finalizing our provisions as proposed.
H. ICRs Regarding Organization, Staffing and Delivery of Services for Hospitals (§ 482.59(a) and (b)) and CAHs (§ 485.649(a) and (b))
We proposed that if a hospital or Critical Access Hospital (CAH) provides OB services, such services must be well-organized and in accordance with nationally recognized acceptable standards of practices for physical and behavioral health of pregnant, birthing, and postpartum patients. We also proposed that any outpatient OB services would be consistent in quality with inpatient OB services in accordance with the complexity of services offered. In addition, we proposed that the organization of the OB service be appropriate to the scope of services offered by the facility and integrated with other departments of the facility. We further proposed that the OB patient care units be supervised by an individual with the necessary education and training, and specify that person should be an experienced registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a doctor of medicine or osteopathy. In addition, we proposed that hospitals and CAHs must delineate and document obstetrical privileges for all practitioners providing obstetrical care in accordance with the competencies of each practitioner.
For delivery of services, we proposed that OB services must be consistent with the needs and resources of the facility. We proposed that policies governing OB care must be designed to ensure the achievement and maintenance of high standards of medical practice and patient care and safety. We additionally proposed that labor & delivery room suites have certain basic resuscitation equipment readily available, including a call-in-system, cardiac monitor, and fetal doppler or monitor. Furthermore, we proposed that the service must ensure that it has adequate provisions and protocols, consistent with nationally recognized and evidence-based guidelines for OB emergencies, complications, immediate post-delivery care, and other patient health and safety events as identified as part of the facility's QAPI program.
Comment: The Joint Commission (TJC) questioned our assumption that there would be no burden for TJC-accredited facilities to develop policies and procedures to ensure that OB services are well organized, provide high-quality care that is appropriate to the level of services provided, and integrated with other departments of the facility, and that facilities have internal standards and protocols to ensure compliance with nationally accepted guidelines for OB emergencies, complications, immediate post-delivery care, and other patient health and safety events. They indicated that the other proposed requirements would exceed their requirements in some areas and would impose additional costs on facilities.
Response: We appreciate the feedback on the burden for TJC-accredited facilities. Rather than assuming that TJC-accredited hospitals and CAHs will have no burden for these requirements, in this final rule with comment period we modify our cost estimate and assume that there is only a 50 percent overlap between the proposed organization, staffing, and delivery of service policy development requirements and current standards for TJC-accredited hospitals and CAHs. For all other proposed OB services requirements, we maintain the same approach we used in the proposed rule and do not assume any overlap between our proposed requirements and TJC requirements.
After consideration of the public comments we received and as indicated in section XXI.C.2 of this final rule with comment period, we are finalizing the proposed policies for hospitals (§ 482.59(a) through (b)) and CAHs (§ 485.649(a) through (b)) that offer obstetrical (OB) services with the modification to reference the requirements of the medical staff bylaws for hospitals and CAHs rather than requiring hospitals and CAHs with OB services to delineate and document obstetrical privileges for all practitioners. In addition, we are clarifying that basic equipment must be kept at the hospital and CAH and be readily available for treating obstetrical cases to meet the needs of patients. Below, we provide the burden estimate for the final requirements that include the following modifications: (1) a higher burden for TJC-accredited hospitals to develop policies and procedures to ensure that OB services are well organized and (2) the removal of the cost for facilities to delineate and document obstetrical privileges for all practitioners providing obstetrical care.
To identify the number of hospitals and CAHs subject to the finalized provisions, we utilized the Center for Medicare and Medicaid Services' Provider of Services File—Hospital and Non-Hospital Facilities. We excluded hospitals and CAHs that do not offer obstetric services, as well as Rural Emergency Hospitals that are not subject to these CoPs. Using this methodology, we obtained a total of 513 CAHs and 4,415 hospitals that offer obstetric services.[757]
We calculated the estimated hourly rates for staff members involved in developing these policies based upon the national mean salary for that particular position increased by 100 percent to account for overhead costs and fringe benefits using the May 2023 National Occupational Employment and Wage Estimates, Bureau of Labor Statistics (BLS) at https://www.bls.gov/oes/current/oes_nat.htm. The wage and salary data from the BLS do not include health, retirement, and other fringe benefits, or the rent, utilities, information technology, administrative, and other types of overhead costs supporting each employee. The HHS-wide guidance on preparation of regulatory and paperwork burden estimates states that doubling salary costs is a good approximation for including these overhead and fringe benefit costs.
Table 186 presents the BLS occupation code and title, the facility provider position, the estimated average or mean hourly wage, and the adjusted hourly wage (with a 100 percent markup of the salary to include fringe benefits and overhead costs).
( printed page 94540) ( printed page 94541)We expect that most hospitals and CAHs that offer OB services already have internal standards and protocols to ensure that OB services are well organized and to provide high-quality care that is appropriate to the level of services provided and integrated with other departments of the facility. We also expect that they have internal standards and protocols to ensure compliance with nationally accepted guidelines for OB emergencies, complications, immediate post-delivery care, and other patient health and safety events. Many hospital accrediting organizations also have specific requirements governing care for pregnant and postpartum patients that will meet part of these finalized requirements. For example, TJC has wide-ranging requirements for hospitals that provide perinatal care, covering everything from providing information to families in a way that is easy to understand to providing initial care for complications such as hemorrhage, hypertensive disorders, fetal heart rate abnormalities.[758] We expect, however, that all hospitals and CAHs will need to spend time ensuring that these standards and their sources are well-documented.
As outlined in 84 FR 51732, writing new policies related to patient care is estimated to take eight hours for each member of the staff involved in the care policy. We have estimated wages as indicated in Table 186 and included the involvement of a physician at $2,029.60 (8 × $253.70), a lawyer at $1357.44 (8 × $169.68), a registered nurse at $726.72 (8 × $90.84), a medical secretary at $333.6 (8 × $41.70), and a medical and health services manager at $1,034.24 (8 × $129.28) for a total estimated cost of $5,481.60 per policy. This estimate leads to an average hourly cost of $137.04 ($5,481.60 ÷ 40) per staff member involved in ensuring that these standards and their sources are well-documented. We assume that documentation will consist of one comprehensive policy per facility.
We do not expect that all facilities will need to spend 40 hours to meet these requirements. We expect that the burden will be lower for TJC-accredited facilities since the organization has wide-ranging requirements for hospitals and CAHs, with the requirements increasing as the complexity of OB care offered increases. We estimate that there is a 50 percent overlap between TJC requirements and our finalized requirements. To account for this reduction in the overall burden, we used CMS' CASPER (Certification and Survey Provider Enhanced Reports) [759] to identify TJC-accredited hospitals and CAHs. According to CASPER, approximately 72.2 percent of Medicare and Medicaid approved hospitals are accredited by TJC, as well as 25 percent of Critical Access Hospitals (CAHs). We then multiply the respective shares of TJC-accredited hospitals and CAHs by 0.5 to account for the 50 percent overlap in requirements.
To calculate the hourly burden for this requirement, in Table 187 we multiply the number of facilities by the number of responses per facility, applying the discount for hospitals and CAHs that are accredited by TJC, by the hourly burden estimate. To determine the associated cost, we multiply the revised hourly burden estimate by the average hourly labor cost. Using this formula, in Table 187 we estimate a total burden of 13,802 hours at a cost of $17,925,161.
Table 188 provides the annual burden estimate over a 10-year period for these finalized requirements. We do not estimate a burden for updating these policies and procedures after their initial development in year 1 since regularly reviewing and updating policies is a standard business practice for healthcare facilities that must comply with applicable Federal, state, and local laws, regulations and ordinances that periodically change. As such, the total estimate over 10 years is 130,802 hours at a cost of $17,925,161.
For the requirements that OB patient care units be supervised by an individual with the necessary education and training, as well as ensuring that OB patient care units have certain basic equipment readily available, and that the facility has adequate provisions for obstetrical emergencies, we provide the estimated cost in the regulatory impact analysis section below.
( printed page 94542)I. ICRs Regarding OB Staff Training for Hospitals (§ 482.59(c)) and CAHs (§ 485.649(c))
We proposed that hospitals and CAHs that offer OB services must develop policies and procedures to ensure that staff are trained on select topics related to improving the delivery of maternal care. The training must reflect the scope and complexity of services offered and must include, but is not limited to, facility-identified evidence-based best practices and protocols to improve the delivery of maternal care within the facility. They would also need to be trained in any additional topics as identified by the facility's QAPI program. We also proposed that the governing body must identify and document which staff must complete annual training on these topics. The facility must further document that training was successfully completed and must be able to demonstrate staff knowledge on these topics. Lastly, we proposed that the efficacy of the training must be reviewed and assessed on an ongoing basis, based on the results of data, measures, and quality indicators from the facility's QAPI program.
Comment: A commenter said that increased demand for training material for facilities to meet the proposed training requirement could cause vendors to raise their prices.
Response: We appreciate the comment regarding vendor prices. Facilities have the flexibility to determine which topics they train staff on to improve the delivery of maternal care and which vendors they use for their training. While some vendors could raise the prices for training materials in response to higher demand, others may choose to maintain or even lower it given the increased demand. Facilities may also choose to use free training resources such as those outlined in section XXI.C.3 of this final rule with comment period. Given the availability of free and low-cost resources for many training topics and likely variation across vendors, we are not including any additional burden related to vendor pricing.
Comment: A few commenters said that CMS should consider the disproportionate financial impact that the requirements will have on some provider types, particularly hospitals and CAHs that are most at risk of closing their OB units, which could lead to increased burden on other hospitals and result in patients having to travel greater distance to access maternal care.
Response: We appreciate commenters' suggestions to consider differences across providers. The CY 2025 OPPS/ASC proposed rule assessed likely variation in burden across different providers, taking into account variation in facility size, the number of patients, and staff size that we believe accurately reflects the likely burden for each facility type. We have also taken several steps to reduce burden and ensure that all hospitals and CAHs are able to successfully meet the requirements. First, we are implementing the requirements in three phases (see section XXI.A.7 of this final rule with comment period) to reduce potential implementation burden or unintended consequences. Second, as we discuss in more detail in the regulatory impact analysis, we are also reducing the frequency of the obstetrical services training requirement from annually to biannually (every other year), which leads to a large reduction in cost. Finally, we are not finalizing the requirement that hospitals and CAHs delineate and document obstetrical privileges for all practitioners providing obstetrical care.
After consideration of the public comments we received, and as indicated in section XXI.C.3 of this final rule with comment period, we are finalizing the policies for hospitals (§ 482.59(c)) and CAHs (§ 485.649(c)) that offer OB services with the modification that the effective date of the staff training requirement is January 1, 2027, and that the governing body in hospitals and CAHs must identify and document which staff must complete an initial training and subsequent biannual training. ( printed page 94543)
As outlined in 84 FR 51732, writing new policies related to patient care is estimated to take eight hours for each member of the staff involved in the care policy. We have estimated wages as indicated in Table 186 and included the involvement of a physician at $2,029.60 (8 × $253.70), a lawyer at $1357.44 (8 × $169.68), a registered nurse at $726.72 (8 × $90.84), a medical secretary at $333.6 (8 × $41.70), and a medical and health services manager at $1,034.24 (8 × $129.28) for a total estimated cost of $5,481.60 per policy. This estimate leads to an average hourly cost of $137.04 ($5481.60 ÷ 40) per staff member involved in ensuring that these standards and their sources are well-documented. We assume that documentation will consist of one comprehensive policy per facility. We do not estimate a burden for reviewing and assessing the efficacy of these efforts since we address this below in section XXVI.J. We do not estimate a burden for documenting that training was completed as updating employee records is also a customary business practice. As indicated in Table 189 and Table 190, we estimate that the development of the finalized OB staff training policies and procedures will take 197,120 hours to complete and cost $27,013,325. We discuss the burden for staff training in the regulatory impact analysis below.
J. ICRs Regarding Revisions to QAPI Standards for OB Services in Hospitals (§ 482.21) and CAHs (§ 485.641)
We proposed that hospitals and CAHs with OB services must use their QAPI program to address health disparities among OB patients on an ongoing basis. They must also measure and monitor for health disparities among OB patients and develop and implement actions to address these disparities and monitor subsequent results. Moreover, on an annual basis, they must conduct at least one performance improvement project focused on reducing maternal health disparities. In addition to the proposed QAPI requirements, we proposed that OB leadership be engaged in the facility's QAPI requirement. We further proposed that if a Maternal Mortality Review Committee (MMRC) is available at the state or local jurisdiction in which the facility is located, the facility must and have a process for incorporating MMRC data and recommendations into the facility's QAPI program.
Comment: One commenter said that full utilization of quality improvement activities often requires significant financial investment and administrative support to be successful. Another commenter noted that many metrics used in obstetrics cannot be assessed using administrative data and instead requires a time-consuming manual chart review. Some commenters indicated that the rule did not take into account increases in staff wages and salaries.
Response: We appreciate the comments regarding utilization of QAPI and that some obstetrical metrics could require manual chart reviews. We believe that the estimated costs in the proposed rule that included costs for data system modifications, data stratification, incorporation of MMRC recommendations into the QAPI program, and carrying out an annual project provide an accurate estimate of the likely average burden for facilities to engage in these activities. We agree that growth in staff wages and salaries needs to be considered when calculating costs. ( printed page 94544) According to the Bureau of Labor Statistics,[760] RNs in 2015 earned a mean hourly rate of $34.14 with the nominal mean hourly rate in 2023 [761] increasing to $45.42. Inflation,[762] however, accounts for 3.49 percentage points annually over these eight years, leaving a compound annual real growth rate of approximately 0.44 percent. To address concerns that wage costs are underestimated, we are including a 0.44 annual increase in real wage rates for staff involved in carrying out this requirement.
After consideration of the public comments we received, and as indicated in section XXI.C.4 of this final rule with comment period, we are finalizing the proposed revision to existing QAPI standards (§ 482.21; § 485.641) for hospitals and CAHs that offer obstetrical services without modifications. To account for growth in staff wage rates, we are including a 0.44 percent annual increase in real wage rates.
The costs associated with data collection will include the cost for facilities to modify their information technology infrastructure to ensure that they capture all features relevant for the diverse subpopulations that the facility identifies. Given that many facilities already collect some of these patient characteristics, such as race and ethnicity, we estimate that planning, programming, and performing quality checks will take 8 hours in the first year and 4 hours in all subsequent years. We anticipate a mixture of staff from computer and mathematical occupations will oversee these changes at an average hourly cost of $108.78. This leads to an average cost of $870.24 (8 × $108.78) per provider in the first year. For subsequent years, we estimate that the requirement will take 4 hours to complete and include a 0.44 percent annual increase in the average hourly cost to account for growth in real wage rates. As indicated in Table 191 we estimate that in the first year, updating infrastructure will cost a total of $4,288,543. In Table 192, we provide the estimated total 10-year cost which we estimate at $24,016,571.
Based on our experience working with healthcare data, we anticipate that stratification of data and quality indicators, together with monitoring the results after actions are taken to address these disparities, will take 8 hours annually. We anticipate that data scientists will oversee these efforts at an average hourly cost of $114.46. Since the requirements do not go into effect until year 2, we estimate that there will be no burden in year 1. For subsequent years, we increase the estimated burden to include a 0.44 percent annual increase in the average hourly cost to account for growth in real wage rates. For year 2, we estimate an average cost of $919.71 per provider. Table 193 provides the estimated cost in year 2 and Table 194 provides the estimated cost over 10 years. We estimate an average annual cost of annual cost of $4,532,326 with a total cost of $41,516,273 over 10 years.
( printed page 94545)K. ICRS Regarding Emergency Services Readiness in Emergency Services for Hospitals (§ 482.55(c)) and CAHs (§ 485.618(e))
We proposed a new standard for emergency services readiness and to improve staff readiness for providing emergency services to all patients, including pregnant and postpartum patients. The first proposed standard would require hospitals and CAHs with emergency services to have adequate provisions and protocols, consistent with nationally recognized and evidence-based guidelines, for the care of patients with emergency conditions (including but not limited to patients with OB emergencies, complications, immediate post-delivery care). Applicable staff would be required to be trained on these protocols and provisions. We also proposed that equipment, supplies, and medication used in treating emergency cases are kept at the hospital and are readily available for treating emergency cases.
Comment: A commenter said that the emergency services requirements may require additional investment by facilities, such as establishing proper storage, tracking, and protocols related to the necessary products.
Response: CMS expects that facilities would already have protocols in place for the storage and tracking of emergency products. As such, we do not estimate any additional burden for this requirement.
After consideration of the public comments we received and as indicated in section XXI.C.5 of this final rule with comment period, we are finalizing the proposed emergency services readiness standard for hospitals and CAHs without modifications. We are also finalizing the burden estimates as proposed.
As outlined in 84 FR 51732, writing new policies related to patient care is estimated to take eight hours for each member of the staff involved in the care policy. Since the finalized standard for emergency services involves adding a new standard to an existing policy, we estimate that it will take half the amount of time as writing a new policy, or 4 hours for each staff member involved. We have estimated wages as indicated in Table 186 and included the involvement of a physician at $1,014.80 (4 × $253.70), a lawyer at $678.72 (4 × $169.68), a registered nurse at $363.36 (4 × $90.84), a medical secretary at $166.80 (4 × $41.70), and a health services manager at $517.12 (4 × $129.28) for a total estimated cost of $2,740.80 per policy. This estimate leads to an average hourly cost of $137.04 ($2,740.80 ÷ 20) per staff member involved in developing this standard. We do not estimate a burden for updating standards since reviewing and updating policies and procedures is a customary business practice. As indicated in Table 195 and Table 196, we estimate that creating this standard will cost hospitals $17,294,448 with a total hourly burden of 126,200 hours.
( printed page 94546)L. Transfer Protocols in Discharge Planning (§ 482.43) for Hospitals
We proposed transfer protocol requirements for hospitals transferring patients under their care to the appropriate level of care, including to another hospital, as necessary to meet the needs of the patient and stabilize any emergency conditions (including but not limited to patients with OB emergencies, complications, immediate post-delivery care).
After consideration of the comments, and as indicated in section XXI.C.6 of this final rule with comment period, we are finalizing the proposed emergency services readiness standard for hospitals with the modification that acute care hospitals are required to provide annual training to the relevant staff (as determined by the facility) regarding the hospital policies and procedures for transferring patients under its care. We did not receive any comments on the estimated cost to develop transfer protocols and are finalizing our estimate for this requirement as proposed.
In 87 FR 40350, we estimated that for rural emergency hospitals (REHs), developing a transfer agreement with at least one hospital would require 2 hours of work from an administrator and a clerical person. We believe that hospitals will face a similar burden for this requirement. Using estimated wages as indicated in Table 186, we estimate that this requirement will include the involvement of a medical secretary at $83.40 (2 × 41.70) and a medical and health services manager at $258.56 (2 × 129.28) for a total estimate cost of $341.96 per hospital. This estimate leads to an average hourly cost of $85.49 ($341.96 ÷ 4) per staff member involved in developing this standard. We do not estimate a burden for updating transfer protocols since reviewing and updating policies and procedures is a customary business practice. As indicated in Table 197 and Table 198, we estimate that creating these protocols will cost hospitals $1,982,342 with a total hourly burden of 23,188 hours.
( printed page 94547)M. Total Costs for all ICRs Related to Maternal Health
In Tables 199 and 227, we provide the total hourly burden estimate and cost for all collection of information requirements related to maternal health as outlined in Tables 188, 190, 192, 194, 196, and 198. Overall, we estimate that the finalized requirements will have a total burden of 1,048,958 hours over 10 years at a cost of $129,748,120.
( printed page 94548)XXVII. Response to Comments
Because of the large number of public comments we normally receive on Federal Register documents, we are not able to acknowledge or respond to them individually. We will consider all comments we receive by the date and time specified in the DATES section of this preamble; and, when we proceed with a subsequent document, we will respond to the comments in the preamble to that document.
XXVIII. Economic Analyses
A. Statement of Need
This final rule with comment period is necessary to make updates to the Medicare hospital OPPS rates. It is also necessary to make changes to the payment policies and rates for outpatient services furnished by hospitals and CMHCs in CY 2025. We are required under section 1833(t)(3)(C)(ii) of the Act to update annually the OPPS conversion factor used to determine the payment rates for APCs. We also are required under section 1833(t)(9)(A) of the Act to review, not less often than annually, and revise the groups, the relative payment weights, and the wage and other adjustments described in section 1833(t)(2) of the Act. We must review the clinical integrity of payment groups and relative payment weights at least annually. We are revising the APC relative payment weights using claims data for services furnished on and after January 1, 2023, through and including December 31, 2023, and processed through June 30, 2024, and updated HCRIS cost report information.
This final rule with comment period is also necessary to make updates to the ASC payment rates for CY 2025, enabling CMS to make changes to payment policies and payment rates for covered surgical procedures and covered ancillary services that are performed in ASCs in CY 2025. Because ASC payment rates are based on the OPPS relative payment weights for most of the procedures performed in ASCs, the ASC payment rates are updated annually to reflect annual changes to the OPPS relative payment weights. In addition, we are required under section 1833(i)(1) of the Act to review and update the list of surgical procedures that can be performed in an ASC, not less frequently than every 2 years.
In the CY 2019 OPPS/ASC final rule with comment period (83 FR 59075 through 59079), we finalized a policy to update the ASC payment system rates using the hospital market basket update instead of the CPI-U for CY 2019 through 2023. In the CY 2024 OPPS/ASC final rule, we finalized a policy to extend the 5-year interim period by an additional 2 years, through CY 2024 and CY 2025, to enable us to more accurately analyze whether the application of the hospital market basket update to the ASC payment system resulted in a migration of services from the hospital setting to the ASC setting (88 FR 81960).
This final rule is also necessary to create three additional exceptions to the four walls requirement under the Medicaid clinic services benefit at 42 CFR 440.90. Specifically, we are finalizing our proposal to add a mandatory four walls exception for IHS/Tribal clinics at 42 CFR 440.90(c) and optional exceptions for behavioral health clinics and clinics located in rural areas at 42 CFR 440.90(d) and (e). As discussed in section XVIII.A of this final rule, our current regulation at 42 CFR 440.90(b) allows for an exception to the four walls requirement only for certain clinic services furnished to individuals who are unhoused.
This final rule is also necessary to codify the requirements of the CAA, 2023 for States to provide 12 months of continuous eligibility to children under the age of 19 in Medicaid and CHIP, with limited exceptions. Specifically, we are finalizing our proposal to remove the option to provide continuous eligibility to a subgroup of Medicaid and CHIP enrollees and for a time period of less than 12 months. For CHIP, we are finalizing removal of the option to disenroll children from CHIP during a continuous eligibility period for failure to pay premiums.
This final rule is necessary to make policy changes under the OPPS or ASC payment system for hospital outpatient ( printed page 94549) departments (HOPDs), rural emergency hospitals (REHs), and ASCs reporting data under the Hospital Outpatient Quality Reporting (OQR), REH Quality Reporting (REHQR), and ASC Quality Reporting (ASCQR) Programs, respectively. The primary objective of these quality reporting programs is to promote higher quality, more efficient health care for Medicare beneficiaries by collection and reporting on quality-of-care metrics. This information is made available to consumers, both to empower Medicare beneficiaries and inform decisionmaking, as well as to incentivize healthcare facilities to make continued improvements.
This final rule with comment period is also necessary to improve the quality of obstetrical services in hospitals and Critical Access Hospitals (CAHs). The United States has the highest maternal mortality rate among OECD countries.[763] This mortality rate has increased sharply in recent years rising from 17.4 deaths per 100 thousand live births in 2018, to 32.9 deaths per 100 thousand live births in 2021,[764] with most of the increased deaths in 2020 and 2021 being Covid-19 related deaths.[765] The causes of pregnancy-related deaths has shifted in recent years with a decline in traditional causes, such as hemorrhage, hypertensive disorders of pregnancy, and thromboembolism, and an increase in cardiovascular problems and other medical conditions.[766 767] Nearly a third of all pregnancy-related deaths occur between the day of delivery and the 6 days that follow, with another 20 percent of deaths occurring 7 to 42 days postpartum.[768]
Within the United States, there are widespread differences in maternal mortality rates based on age, race, and geographical location. According to the National Center for Health Statistics, the maternal mortality rates for women in the United States over 40 years of age in 2021 was nearly 8 times greater than for women under 25 years of age, with mortality rates for non-Hispanic black women over 40 years of age more than 21 times higher than the rate for Hispanic women under 25 years of age.[769] Similarly, pregnancy-related mortality rates are higher in rural areas vis-à-vis urban areas.[770] Beyond deaths, maternal morbidity, defined as “any health condition attributed to and/or aggravated by pregnancy and childbirth that has a negative impact on the woman's wellbeing,” [771] remains a common occurrence, with rates also varying by age and race.[772 773]
Pregnancy-related mortality and morbidity have large health and economic costs. One study estimates that between 2018 and 2020, pregnancy-related mortalities lead to nearly 114,000 years of potential life lost (YPLL) and cost more than $27.4 billion based on the value of statistical life (VSL).[774] Another study finds that severe maternal morbidity, as measured by 21 ICD-10 codes that the Centers for Disease Control and Prevention (CDC) identified, is associated with a 75 percent increase in costs for Medicaid patients and a more than doubling in costs for commercially insured patients during the prenatal to 30 days post-partum period.[775] Focusing specifically on nine maternal morbidities among the 2019 US birth cohort from birth to 5-years postpartum, researchers estimated they had a cost of $32.3 billion for birthing parents and their children, with $18.7 billion due to medical costs and $13.6 billion from non-medical costs.[776]
Although the studies vary in their methodology, time period pre-post birth analyzed, medical conditions analyzed, and cost estimates, they overall suggest that maternal morbidity and mortality impose a high health, safety, and economic cost on birth parents, children, and society.[777] Given these costs, we are implementing conditions of participation (COPs) that are designed to help reduce maternal mortality and morbidity.
We are finalizing requirements that hospital and CAH OB patient care units be supervised by an individual with the necessary education and training and have equipment at the hospital and CAH and readily available for treating obstetrical cases to meet the needs of patients. Facilities offering obstetrical services must also have adequate provisions that include equipment, supplies and medication used in treating emergency cases for obstetrical emergencies, complications, immediate post-delivery care, and other patient health and safety events as identified as part of the QAPI program. We also are finalizing requirements that staff involved with OB services be trained on key topics related to improving the delivery of maternal care. Hospitals and CAHs will also be required to utilize data from their QAPI program to implement one quality improvement project to address disparities in maternal care and to integrate information from MMRCs into their QAPI program. We are also finalizing requirements that hospitals and CAHs train their staff on emergency procedures for all patients and that hospitals have provisions including equipment, supplies, and medication used in treating emergency cases. Finally, we are finalizing the requirement that relevant acute care hospital staff receive annual training on proper transfer protocols.
B. Overall Impact of Provisions of This Final Rule With Comment Period
We have examined the impacts of this rule, as required by Executive Order 12866, as amended, on Regulatory Planning and Review (September 30, 1993), Executive Order 13563 on Improving Regulation and Regulatory Review (January 18, 2011), Executive Order 14094, titled “Modernizing Regulatory Review” (April 6, 2023), the Regulatory Flexibility Act (RFA) (September 19, 1980, Pub. L. 96-354), section 1102(b) of the Social Security Act, section 202 of the Unfunded Mandates Reform Act of 1995 (March 22, 1995, Pub. L. 104-4), Executive ( printed page 94550) Order 13132 on Federalism (August 4, 1999), and the Congressional Review Act (5 U.S.C. 804(2)).
Executive Orders 12866, as amended, and 13563 direct agencies to assess all costs and benefits of available regulatory alternatives and, if regulation is necessary, to select regulatory approaches that maximize net benefits (including potential economic, environmental, public health and safety effects, distributive impacts, and equity). Executive Order 14094 amends section 3(f) of Executive Order 12866. The amended section 3(f) of Executive Order 12866 defines a “significant regulatory action” as an action that is likely to result in a rule: (1) having an annual effect on the economy of $200 million or more in any 1 year (adjusted every 3 years by the Administrator of OMB's Office of Information and Regulatory Affairs (OIRA) for changes in gross domestic product), or adversely affect in a material way the economy, a sector of the economy, productivity, competition, jobs, the environment, public health or safety, or State, local, territorial, or tribal governments or communities; (2) creating a serious inconsistency or otherwise interfering with an action taken or planned by another agency; (3) materially altering the budgetary impacts of entitlement grants, user fees, or loan programs or the rights and obligations of recipients thereof; or (4) raise legal or policy issues for which centralized review would meaningfully further the President's priorities or the principles set forth in this Executive order, as specifically authorized in a timely manner by the Administrator of OIRA in each case.
A regulatory impact analysis (RIA) must be prepared for major rules with significant regulatory action/s and/or with significant effects as per section 3(f)(1) ($200 million or more in any 1 year). Based on our estimates, OIRA has determined this rulemaking is significant per section 3(f)(1) as measured by an effect on the economy of $200 million or more in any 1 year. Pursuant to Subtitle E of the Small Business Regulatory Enforcement Fairness Act of 1996 (also known as the Congressional Review Act), OIRA has also determined that this rule meets the criteria set forth in 5 U.S.C. 804(2). Accordingly, we have prepared an RIA that to the best of our ability presents the costs and benefits of the rulemaking. Therefore, OMB has reviewed this final rule with comment period, and the Departments have provided the following assessment of their impact.
We estimate that the total increase in Federal Government expenditures under the OPPS for CY 2025, compared to CY 2024, due to the changes to the OPPS in this final rule with comment period, will be approximately $1.98 billion. Taking into account our estimated changes in enrollment, utilization, and case-mix for CY 2025 we estimate that the OPPS expenditures, including beneficiary cost-sharing, for CY 2025 would be approximately $87.7 billion, which is approximately $4.7 billion higher than estimated OPPS expenditures in CY 2024. Table 201 of this final rule with comment period displays the distributional impact of the CY 2025 changes in OPPS payment to various groups of hospitals and for CMHCs.
We note that under our final CY 2025 policy, drugs and biologicals are generally paid at ASP plus 6 percent, WAC plus 6 percent, or 95 percent of AWP, as applicable.
We estimate that the final update to the conversion factor will increase total OPPS payments by 2.9 percent in CY 2025. The final changes to the APC relative payment weights, the final changes to the wage indexes, the final continuation of a payment adjustment for rural SCHs, including EACHs, and the final payment adjustment for cancer hospitals would not increase total OPPS payments because these changes to the OPPS are budget neutral. However, these updates would change the distribution of payments within the budget neutral system. We estimate that the total change in payments between CY 2024 and CY 2025, considering all budget-neutral payment adjustments, changes in estimated total outlier payments, the application of the frontier State wage adjustment, in addition to the application of the OPD fee schedule increase factor after all adjustments required by sections 1833(t)(3)(F), 1833(t)(3)(G), and 1833(t)(17) of the Act will increase total estimated OPPS payments by 3.0 percent.
We estimate the total increase (from changes to the ASC provisions in this final rule with comment period, as well as from enrollment, utilization, and case-mix changes) in Medicare expenditures (not including beneficiary cost-sharing) under the ASC payment system for CY 2025 compared to CY 2024, to be approximately $240 million. Tables 202 and 203 of this final rule with comment period display the redistributive impact of the CY 2025 changes regarding ASC payments, grouped by specialty area and then grouped by procedures with the greatest ASC expenditures, respectively.
We estimate that finalizing three additional exceptions to the Medicaid clinic services benefit four walls requirement for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas would cause total Medicaid transfers to increase by $1.18 billion for fiscal years 2025 through 2029. This includes a Federal impact of $1.15 billion and State impact of $30 million.
For the OB services provisions of this final rule with comment period, in Tables 231 and 232, we provide the total estimated cost and hourly burden of these finalized requirements both annually and over 10 years, excluding collection of information costs that we have already estimated above. Overall, we estimate that these finalized requirements will cost an average of approximately $410 million and take $2.24 million hours to complete. Over 10 years, we estimate that the total cost will be approximately $4.10 billion and take $22.4 million hours to complete. Below, we provide the cost estimates for each of the finalized requirements.
C. Detailed Economic Analyses
1. Estimated Effects of OPPS Changes in This Final Rule With Comment Period
a. Limitations of Our Analysis
The distributional impacts presented here are the projected effects of the final CY 2025 policy changes on various hospital groups. We post our hospital-specific estimated payments for CY 2025 on the CMS website with the other supporting documentation for this final rule with comment period. To view the hospital-specific estimates, we refer readers to the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient. On the website, select “Regulations and Notices” from the left side of the page and then select “CMS-1809-FC” from the list of regulations and notices. The hospital-specific file layout and the hospital-specific file are listed with the other supporting documentation for this final rule with comment period. We show hospital-specific data only for hospitals whose claims were used for modeling the impacts shown in Table 201 of this final rule with comment period. We do not show hospital-specific impacts for hospitals whose claims we were unable to use. We refer readers to section II.A of this final rule with comment period for a discussion of the hospitals whose claims we do not use for ratesetting or impact purposes.
We estimate the effects of the individual policy changes by estimating payments per service, while holding all other payment policies constant. We use the best data available but do not attempt to predict behavioral responses to our policy changes in order to isolate ( printed page 94551) the effects associated with specific policies or updates, but any policy that changes payment could have a behavioral response. In addition, we have not made any adjustments for future changes in variables, such as service volume, service-mix, or number of encounters.
b. Estimated Effects of OPPS Changes on Hospitals
Table 201 shows the estimated impact of the proposed rule on hospitals. Historically, the first line of the impact table, which estimates the change in payments to all facilities, has always included cancer and children's hospitals, which are held harmless to their pre-Balanced Budget Act (BBA) amount. We also include CMHCs in the first line that includes all providers. We include a second line for all hospitals, excluding permanently held harmless hospitals and CMHCs.
We present separate impacts for CMHCs in Table 201, and we discuss them separately below, because CMHCs have historically been paid only for partial hospitalization services under the OPPS and are a different provider type from hospitals. In the CY 2024 OPPS/ASC final rule with comment period (88 FR 81833), we finalized paying CMHCs for partial hospitalization services and intensive outpatient services under APCs 5851 through 5854. For CY 2025, we are maintaining the same APC structure and updating each APC payment rate to reflect the most recent available cost data.
The estimated increase in the total payments made under the OPPS is determined largely by the increase to the conversion factor under the statutory methodology. The distributional impacts presented do not include assumptions about changes in volume and service-mix. The conversion factor is updated annually by the OPD fee schedule increase factor, as discussed in detail in section II.B of this final rule with comment period.
Section 1833(t)(3)(C)(iv) of the Act provides that the OPD fee schedule increase factor is equal to the market basket percentage increase applicable under section 1886(b)(3)(B)(iii) of the Act, which we refer to as the IPPS market basket percentage increase. The final IPPS market basket percentage increase applicable to the OPD fee schedule for CY 2025 is 3.4 percent. Section 1833(t)(3)(F)(i) of the Act reduces that 3.4 percent by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act, which is 0.5 percentage point for CY 2025 (which is also the productivity adjustment for FY 2025 in the FY 2025 IPPS/LTCH PPS final rule (89 FR 69344)) resulting in the final CY 2025 OPD fee schedule increase factor of 2.9 percent. We are using the OPD fee schedule increase factor of 2.9 percent in the calculation of the final CY 2025 OPPS conversion factor. Section 10324 of the Affordable Care Act, as amended by HCERA, further authorized additional expenditures outside budget neutrality for hospitals in certain frontier States that have a wage index less than 1.0000. The amounts attributable to this frontier State wage index adjustment are incorporated in the estimates in Table 201 of this final rule with comment period.
To illustrate the impact of the CY 2025 changes, our analysis begins with a baseline simulation model that uses the CY 2024 relative payment weights, the FY 2024 final IPPS wage indexes that include reclassifications, and the final CY 2024 conversion factor. Table 201 shows the estimated redistribution of the increase or decrease in payments for CY 2025 over CY 2024 payments to hospitals and CMHCs as a result of the following factors: the impact of the APC reconfiguration and recalibration changes between CY 2024 and CY 2025 (Column 2); the wage indexes and the provider adjustments (Column 3); the combined impact of all of the changes described in the preceding columns plus the 2.9 percent OPD fee schedule increase factor update to the conversion factor (Column 4); the estimated impact taking into account all payments for CY 2025 relative to all payments for CY 2024, including the impact of changes in estimated outlier payments and changes to the pass-through payment estimate (Column 5).
We did not model an explicit budget neutrality adjustment for the rural adjustment for SCHs because we are maintaining the current adjustment percentage for CY 2025. Because the final updates to the conversion factor (including the update of the OPD fee schedule increase factor), the estimated cost of the rural adjustment, and the estimated cost of projected pass-through payment for CY 2025 are applied uniformly across services, observed redistributions of payments in the impact table for hospitals largely depend on the mix of services furnished by a hospital (for example, how the APCs for the hospital's most frequently furnished services would change), and the impact of the wage index changes on the hospital. However, total payments made under this system and the extent to which this final rule would redistribute money during implementation also will depend on changes in volume, practice patterns, and the mix of services billed between CY 2024 and CY 2025 by various groups of hospitals, which CMS cannot forecast.
Overall, we estimate that the final rates for CY 2025 will increase Medicare OPPS payments by an estimated 3.0 percent. Removing payments to cancer and children's hospitals because their payments are held harmless to the pre-OPPS ratio between payment and cost and removing payments to CMHCs results in an estimated 3.2 percent increase in Medicare payments to all other hospitals. These estimated payments will not significantly impact other providers.
Column 1: Total Number of Hospitals
The first line in Column 1 in Table 201 shows the total number of facilities (3,562), including designated cancer and children's hospitals and CMHCs, for which we were able to use CY 2023 hospital outpatient and CMHC claims data to model CY 2024 and CY 2025 payments, by classes of hospitals, for CMHCs and for dedicated cancer hospitals. We excluded all hospitals and CMHCs for which we could not plausibly estimate CY 2024 or CY 2025 payment and entities that are not paid under the OPPS. The latter entities include CAHs, IHS and tribal hospitals, and hospitals located in Guam, the U.S. Virgin Islands, Northern Mariana Islands, American Samoa, and the State of Maryland. This process is discussed in greater detail in section II.A of this final rule with comment period. At this time, we are unable to calculate a DSH variable for hospitals that are not also paid under the IPPS because DSH payments are only made to hospitals paid under the IPPS. Hospitals for which we do not have a DSH variable are grouped separately and generally include freestanding psychiatric hospitals, rehabilitation hospitals, and long-term care hospitals. We show the total number of OPPS hospitals (3,460), excluding the hold harmless cancer and children's hospitals and CMHCs, on the second line of the table. We excluded cancer and children's hospitals because section 1833(t)(7)(D) of the Act permanently holds harmless cancer hospitals and children's hospitals to their “pre-BBA amount” as specified under the terms of the statute, and therefore, we removed them from our impact analyses. We show the isolated impact on the 35 CMHCs at the bottom of the impact table (Table 201) and discuss that impact separately below. ( printed page 94552)
Column 2: APC Recalibration—All Changes
Column 2 shows the estimated effect of APC recalibration. Column 2 also reflects any changes in multiple procedure discount patterns or conditional packaging that occur as a result of the changes in the relative magnitude of payment weights. As a result of APC recalibration, we estimate that urban hospitals would experience a 0.1 increase, with the impact ranging from a decrease of 0.1 percent to an increase of 0.4, depending on the number of beds. Rural hospitals will experience an estimated decrease of 0.4 overall. Major teaching hospitals will experience an estimated decrease of 0.1 percent.
Column 3: Wage Indexes and the Effect of the Provider Adjustments
Column 3 demonstrates the combined budget neutral impact of the APC recalibration, the updates for the wage indexes with the FY 2025 IPPS post-reclassification wage indexes including the low wage index hospital policy, the rural adjustment, the frontier adjustment, and the cancer hospital payment adjustment. We modeled the independent effect of the budget neutrality adjustments and the OPD fee schedule increase factor by using the relative payment weights and wage indexes for each year and using a CY 2024 conversion factor that included the OPD fee schedule increase and a budget neutrality adjustment for differences in wage indexes.
Column 3 reflects the independent effects of the updated wage indexes, including the application of budget neutrality for the rural floor policy on a nationwide basis, as well as the final CY 2025 changes in wage index policy, discussed in section II.C of this final rule with comment period. We note that the final CY 2025 OPPS wage index includes the low wage index hospital policy, as proposed. We did not model a budget neutrality adjustment for the rural adjustment for SCHs because we proposed to continue the rural payment adjustment of 7.1 percent to rural SCHs for CY 2025, as described in section II.E of this final rule with comment period. We modeled a budget neutrality adjustment for the final cancer hospital payment adjustment because the proposed payment-to-cost ratio target for the cancer hospital payment adjustment in CY 2025 is 0.87, which is different from the 0.88 PCR target adopted in the CY 2024 OPPS/ASC final rule with comment period (88 FR 81589). We note that, in accordance with section 16002 of the 21st Century Cures Act, we are applying a budget neutrality factor calculated as if the cancer hospital adjustment target payment-to-cost ratio was 0.88, not the 0.87 target payment-to-cost ratio we are finalizing in section II.F of this final rule with comment period.
We modeled the independent effect of updating the wage indexes by varying only the wage indexes, holding APC relative payment weights, service-mix, and the rural adjustment constant and using the CY 2025 scaled weights and a CY 2024 conversion factor that included a budget neutrality adjustment for the effect of the changes to the wage indexes between CY 2024 and CY 2025.
Column 4: All Budget Neutrality Changes Combined With the Market Basket Update
Column 4 demonstrates the combined impact of all the final changes previously described and the update to the conversion factor of 2.9 percent. Overall, these changes would increase payments to urban hospitals by 3.2 percent and to rural hospitals by 3.3 percent. Rural sole community hospitals would receive an estimated increase of 3.3 percent while other rural hospitals would receive an estimated increase of 3.5 percent.
Column 5: All Changes for CY 2025
Column 5 depicts the full impact of the final CY 2025 policies on each hospital group by including the effect of all changes for CY 2025 and comparing them to all estimated payments in CY 2024. Column 5 shows the combined budget neutral effects of Columns 2 and 3; the OPD fee schedule increase; the impact of estimated OPPS outlier payments, as discussed in section II.G of final rule with comment period; the change in the Hospital OQR Program payment reduction for the small number of hospitals in our impact model that failed to meet the reporting requirements (discussed in section XIV of this final rule with comment period); and other rule adjustments to the CY 2025 OPPS payments.
Of those hospitals that failed to meet the Hospital OQR Program reporting requirements for the full CY 2024 update (and assumed, for modeling purposes, to be the same number for CY 2025), we included 98 hospitals in our model because they had both CY 2023 claims data and recent cost report data. We estimate that the cumulative effect of all changes for CY 2025 would increase payments to all facilities by 3.0 percent for CY 2024. We modeled the independent effect of all changes in Column 5 using the final relative payment weights for CY 2024 and the proposed relative payment weights for CY 2025. We used the final conversion factor for CY 2024 of $87.382 and the final CY 2025 conversion factor of $89.169 discussed in section II.B of this final rule with comment period.
Column 5 contains simulated outlier payments for each year. We used the 1-year charge inflation factor used in the FY 2025 IPPS/LTCH PPS final rule (89 FR 69660) of 4.1 percent (1.04118) to increase charges on the CY 2023 claims, and we used the overall CCR in the July 2024 Outpatient Provider-Specific File (OPSF) to estimate outlier payments for CY 2024. Using the CY 2023 claims and a 4.1 percent charge inflation factor, we currently estimate that outlier payments for CY 2024, using a multiple threshold of 1.75 and a fixed-dollar threshold of $7,750, would be approximately 0.83 percent of total payments. The estimated current outlier payments of 0.83 percent are incorporated in the comparison in Column 5. We used the same set of claims and a charge inflation factor of 8.5 percent (1.08406) and the CCRs in the July 2024 OPSF, with an adjustment of 1.015192 (89 FR 69960), to reflect relative changes in cost and charge inflation between CY 2023 and CY 2025, to model the proposed CY 2025 outliers at 1.0 percent of estimated total payments using a multiple threshold of 1.75 and a fixed dollar threshold of $7,175. The charge inflation and CCR inflation factors are discussed in detail in the FY 2025 IPPS/LTCH PPS final rule (89 FR 69955 through 69960).
Overall, we estimate that facilities will experience an increase of 3.0 percent under this final rule in CY 2025 relative to total spending in CY 2024. This projected increase (shown in Column 5) of Table 201 of this final rule reflects the final 2.9 percent OPD fee schedule increase factor, adding the 0.17 difference in estimated outlier payments between CY 2024 (0.83 percent) and CY 2025 (1.0 percent), minus 0.10 percent for the change in the pass-through payment estimate between CY 2024 and CY 2025. We estimate that the combined effect of all changes for CY 2025 would increase payments to urban hospitals by 3.2 percent. Overall, we estimate that rural hospitals would experience a 3.2 percent increase as a result of the combined effects of all the changes for CY 2025.
Among hospitals, by teaching status, we estimate that the impacts resulting from the combined effects of all changes include an increase of 2.7 percent for major teaching hospitals and an increase of 3.3 percent for nonteaching hospitals. Minor teaching hospitals will ( printed page 94553) experience an estimated increase of 3.5 percent.
In our analysis, we also have categorized hospitals by type of ownership. Based on this analysis, we estimate that voluntary hospitals will experience an increase of 3.1 percent, proprietary hospitals will experience an increase of 4.9 percent, and governmental hospitals will experience an increase of 2.6 percent.
c. Estimated Effects of OPPS Changes on CMHCs
The last line of Table 201 demonstrates the isolated impact on CMHCs, which historically have only furnished partial hospitalization services under the OPPS. As discussed in section VIII.C of this final rule with comment period, we are finalizing the proposal for CY 2025 to continue paying CMHCs using APCs 5851 through 5854. We modeled the impact of this APC policy, assuming CMHCs will continue to provide the same PHP care as seen in the CY 2023 claims used for ratesetting in the proposed rule. We note that the CY 2023 claims used for the CY 2025 final rule do not include any provision of IOP services. We did not exclude days with one or two services from our modeling for CY 2025, because our final rule policy would pay the per diem rate for APC 5853 for such days beginning in CY 2025. As a result of the final PHP APC changes for CMHCs, we estimate that CMHCs would experience a 11.9 percent increase in CY 2025 payments relative to their CY 2024 payments (shown in Column 5). For a detailed discussion of our final PHP policies, please see section VIII of this final rule with comment period.
Column 3 shows the estimated impact of adopting the final FY 2025 wage index values, which result in an estimated change of 0.0 percent to CMHCs. Column 4 shows that combining the OPD fee schedule increase factor, along with the proposed changes in APC policy for CY 2025 and the proposed FY 2025 wage index updates, will result in an estimated increase of 11.9 percent.
( printed page 94554) ( printed page 94555) ( printed page 94556)d. Estimated Effect of OPPS Changes on Beneficiaries
For services for which the beneficiary pays a copayment of 20 percent of the payment rate, the beneficiary's payment would increase for services for which the OPPS payments would rise and decrease for services for which the OPPS payments would fall. For further discussion of the calculation of the national unadjusted copayments and minimum unadjusted copayments, we refer readers to section II.H of this final rule with comment period. In all cases, section 1833(t)(8)(C)(i) of the Act limits beneficiary liability for copayment for a procedure performed in a year to the hospital inpatient deductible for the applicable year.
We estimate that the aggregate beneficiary coinsurance percentage would be approximately 18.0 percent for all services paid under the OPPS in CY 2025. The estimated aggregate beneficiary coinsurance reflects general system adjustments, including the final CY 2025 comprehensive APC payment policy discussed in section II.A.2.b of this final rule with comment period. We note that the individual payments, and therefore copayments, associated with services may differ based on the setting in which they are furnished. However, at the aggregate system level, we do not currently observe significant impact on beneficiary coinsurance as a result of those policies.
e. Estimated Effects of OPPS Changes on Other Providers
The relative payment weights and payment amounts established under the OPPS affect the payments made to ASCs, as discussed in section XIII of this final rule with comment period. Hospitals, CMHCs, and ASCs would be affected by the changes in this final rule. Additionally, the payment policies we established for IOP services affect RHCs and FQHCs. These providers of IOP are not paid under the OPPS and are not included in the impact analysis shown in Table 201. However, the final payment amount for OPPS APC 5861 would affect payments to RHCs and FQHCs since under sections 1834(o)(5)(A) and 1834(y)(3)(A) of the Act payment for IOP services in these settings is required to be equal to the payment determined for IOP services in the hospital outpatient department.
f. Estimated Effects of OPPS Changes on the Medicare and Medicaid Programs
The effect of the update on the Medicare program is expected to be an increase of $1.98 billion in program payments for OPPS services furnished in CY 2025. The effect on the Medicaid program is expected to be limited to copayments that Medicaid may make on behalf of Medicaid recipients who are also Medicare beneficiaries. We estimate that the changes in this final rule with comment period will increase these Medicaid beneficiary payments by approximately $165 million in CY 2025. Currently, there are approximately 11.5 million dual-eligible beneficiaries, which represent approximately 40 percent of Medicare Part B fee-for-service beneficiaries. The impact on Medicaid was determined by taking 40 percent of the beneficiary cost-sharing impact. The national average split of Medicaid payments is 58 percent Federal payments and 42 percent State payments. Therefore, for the estimated $165 million Medicaid increase, approximately $95 million will be from the Federal Government and $70 million will be from State governments.
g. Alternative OPPS Policies Considered
Alternatives to the OPPS changes we proposed and the reasons for our selected alternatives are discussed throughout this final rule with comment period.
h. Add-On Payment for High-Cost Drugs to the Indian Health Service (IHS) All-Inclusive Rate (AIR)
For CY 2025, we proposed to pay Indian Health Service (IHS) and tribal hospitals separately for high-cost drugs (Part B drugs with daily costs over $1,334) furnished in hospital outpatient departments through an add-on payment, in addition to the All-Inclusive Rate (AIR), using the IHS authority [778] under which the annual AIR is calculated. This policy is projected to increase Medicare program expenditures by approximately $30 ( printed page 94557) million in CY 2025. We refer readers to section X.C of this final rule with comment period for further discussion of this policy.
2. Estimated Effects of CY 2025 ASC Payment System Changes
Most ASC payment rates are calculated by multiplying the ASC conversion factor by the ASC relative payment weight. As discussed fully in section XIII of this final rule with comment period, we are setting the CY 2025 ASC relative payment weights by scaling the final CY 2025 OPPS relative payment weights by the finalized CY 2025 ASC scalar of 0.872. The estimated effects of the updated relative payment weights on payment rates are varied and are reflected in the estimated payments displayed in Tables 202 and 203.
Beginning in CY 2011, section 3401 of the Affordable Care Act requires that the annual update to the ASC payment system after application of any quality reporting reduction be reduced by a productivity adjustment. In CY 2019, we adopted a policy for the annual update to the ASC payment system to be the hospital market basket update for CY 2019 through CY 2023. In the CY 2024 OPPS/ASC final rule with comment period, we extended this 5-year interim period an additional 2 years through CYs 2024 and 2025. Section 1886(b)(3)(B)(xi)(II) of the Act defines the productivity adjustment to be equal to the 10-year moving average of changes in annual economy-wide private nonfarm business multifactor productivity (as projected by the Secretary for the 10-year period, ending with the applicable fiscal year, year, cost reporting period, or other annual period). For ASCs that fail to meet their quality reporting requirements, the CY 2025 payment determinations would be based on the application of a 2.0 percentage point reduction to the annual update factor, which is the hospital market basket update for CY 2025. We calculated the finalized CY 2025 ASC conversion factor by adjusting the CY 2024 ASC conversion factor by 0.9969 to account for changes in the pre-floor and pre-reclassified hospital wage indexes between CY 2024 and CY 2025, which includes our final policy to limit wage index declines of greater than 5 percent, and by applying the CY 2025 productivity-adjusted hospital market basket update factor of 2.9 percent (which is equal to the final inpatient hospital market basket percentage increase of 3.4 percent reduced by a productivity adjustment of 0.5 percentage point). The final CY 2025 ASC conversion factor is $54.895 for ASCs that successfully meet the quality reporting requirements.
a. Limitations of Our Analysis
Presented here are the projected effects of the final changes for CY 2025 on Medicare payment to ASCs. A key limitation of our analysis is our inability to predict changes in ASC service-mix between CY 2023 and CY 2025 with precision. We believe the net effect on Medicare expenditures resulting from the final CY 2025 changes would be small in the aggregate for all ASCs. However, such changes may have differential effects across surgical specialty groups, as ASCs continue to adjust to the payment rates based on the policies of the revised ASC payment system. We are unable to accurately project such changes at a disaggregated level. Clearly, individual ASCs would experience changes in payment that differ from the aggregated estimated impacts presented below.
b. Estimated Effects of ASC Payment System Policies on ASCs
Some ASCs are multispecialty facilities that perform a wide range of surgical procedures from excision of lesions to hernia repair to cataract extraction; others focus on a single specialty and perform only a limited range of surgical procedures, such as eye, digestive system, or orthopedic procedures. The combined effect of the final update to the CY 2025 payments on an individual ASC would depend on a number of factors, including, but not limited to, the mix of services the ASC provides, the volume of specific services provided by the ASC, the percentage of its patients who are Medicare beneficiaries, and the extent to which an ASC provides different services in the coming year. The following discussion includes tables that display estimates of the impact of the final CY 2025 updates to the ASC payment system on Medicare payments to ASCs, assuming the same mix of services, as reflected in our CY 2023 claims data. Table 202 depicts the estimated aggregate percent change in payment by surgical specialty or ancillary items and services group by comparing estimated CY 2024 payments to estimated CY 2025 payments, and Table 203 shows a comparison of estimated CY 2024 payments to estimated CY 2025 payments for procedures that we estimate would receive the most Medicare payment in CY 2024.
In Table 202, we have aggregated the surgical HCPCS codes by specialty group, grouped all HCPCS codes for covered ancillary items and services into a single group, and then estimated the effect on aggregated payment for surgical specialty and ancillary items and services groups. The groups are sorted for display in descending order by estimated Medicare program payment to ASCs. The following is an explanation of the information presented in Table 202.
- Column 1—Surgical Specialty or Ancillary Items and Services Group indicates the surgical specialty into which ASC procedures are grouped and the ancillary items and services group, which includes all HCPCS codes for covered ancillary items and services. To group surgical procedures by surgical specialty, we used the CPT code range definitions and Level II HCPCS codes and Category III CPT codes, as appropriate, to account for all surgical procedures to which the Medicare program payments are attributed.
- Column 2—Estimated CY 2024 ASC Payments were calculated using CY 2023 ASC utilization data (the most recent full year of ASC utilization) and CY 2024 ASC payment rates. The surgical specialty groups are displayed in descending order based on estimated CY 2024 ASC payments.
- Column 3—Estimated CY 2025 Percent Change is the aggregate percentage increase or decrease in Medicare program payment to ASCs for each surgical specialty or ancillary items and services group that is attributable to final updates to ASC payment rates for CY 2025 compared to CY 2024.
As shown in Table 202, for the six specialty groups that account for the most ASC utilization and spending, we estimate that the final update to ASC payment rates for CY 2025 will result in a 3 percent increase in aggregate payment amounts for eye and ocular adnexa procedures, a 3 percent increase in aggregate payment amounts for musculoskeletal system procedures, a 3 percent increase in aggregate payment amounts for nervous system procedures, a 5 percent increase in aggregate payment amounts for digestive system procedures, a 3 percent increase in aggregate payment amounts for cardiovascular system procedures, and a 3 percent increase in aggregate payment amounts for genitourinary system procedures. We note that these changes can be a result of different factors, including updated data, payment weight changes, and changes in policy. In general, spending in each of these categories of services is increasing due to the 2.9 percent payment rate update which is offset by roughly 0.3 percentage points as a result of the final CY 2025 ASC wage indexes and the final ASC wage index scalar of 0.9969, ( printed page 94558) resulting in a net 2.6 percent payment rate update. After the payment rate update is accounted for, aggregate payment increases or decreases for a category of services can be higher or lower than a 2.6 percent increase, depending on if payment weights in the OPPS APCs that correspond to the applicable services increased or decreased or if the most recent data show an increase or a decrease in the volume of services performed in an ASC for a category. For example, we estimate a 5 percent increase in gastrointestinal procedure payments. The increase in payment rates for gastrointestinal procedures is a result of relative increase in the OPPS relative weights for the Upper GI Procedures clinical family. These changes are further increased by the 2.6 percent ASC overall net payment rate increase for these procedures. For estimated changes for selected procedures, we refer readers to Table 202 provided later in this section.
Table 203 shows the estimated impact of the updates to the revised ASC payment system on aggregate ASC payments for selected surgical procedures during CY 2025. The table displays 30 of the procedures receiving the greatest estimated CY 2024 aggregate Medicare payments to ASCs. The HCPCS codes are sorted in descending order by estimated CY 2024 program payment.
- Column 1-CPT/HCPCS code.
- Column 2-Short Descriptor of the HCPCS code.
- Column 3-Estimated CY 2024 ASC Payments were calculated using CY 2023 ASC utilization (the most recent full year of ASC utilization) and the CY 2024 ASC payment rates. The estimated CY 2024 payments are expressed in millions of dollars.
- Column 4-Estimated CY 2025 Percent Change reflects the percent differences between the estimated ASC payment for CY 2024 and the estimated payment for CY 2025 based on the final update.
c. Estimated Effects of ASC Payment System Policies on Beneficiaries
We estimate that the CY 2025 update to the ASC payment system will be generally positive (that is, result in lower cost-sharing) for beneficiaries with respect to the new procedures to be designated as office-based for CY 2025. First, other than certain preventive services where coinsurance and the Part B deductible is waived to comply with sections 1833(a)(1) and (b) of the Act, the ASC coinsurance rate for all procedures is 20 percent. This contrasts with procedures performed in HOPDs under the OPPS, where the beneficiary is responsible for copayments that range from 20 percent to 40 percent of the procedure payment (other than for certain preventive services), although the majority of HOPD procedures have a 20-percent copayment. Second, in almost all cases, the ASC payment rates under the ASC payment system are lower than payment rates for the same procedures under the OPPS. Therefore, the beneficiary coinsurance amount under the ASC payment system will usually be less than the OPPS copayment amount for the same services. (The only exceptions will be if the ASC coinsurance amount exceeds the hospital inpatient deductible since the statute requires that OPPS copayment amounts not exceed the hospital inpatient deductible. Therefore, in limited circumstances, the ASC coinsurance amount may exceed the hospital inpatient deductible and, therefore, the OPPS copayment amount for similar services.) Beneficiary coinsurance for services migrating from physicians' offices to ASCs may decrease or increase under the ASC payment system, depending on the particular service and the relative payment amounts under the MPFS compared to the ASC. While the ASC payment system bases most of its payment rates on hospital cost data used to set OPPS relative payment weights, services that are performed a majority of the time in a physician office are generally paid the lesser of the ASC amount according to the standard ASC ratesetting methodology or at the nonfacility practice expense-based amount payable under the PFS. For those additional procedures that we are finalizing to designate as office-based in CY 2025, the beneficiary coinsurance amount under the ASC payment system generally will be no greater than the ( printed page 94560) beneficiary coinsurance under the PFS because the coinsurance under both payment systems generally is 20 percent (except for certain preventive services where the coinsurance is waived under both payment systems).
Accounting Statements and Tables for OPPS and ASC Payment System
As required by OMB Circular A-4 (available on the Office of Management and Budget website at: https://www.whitehouse.gov/wp-content/uploads/2023/11/CircularA-4.pdf), we have prepared accounting statements to illustrate the impacts of the OPPS and ASC changes in this final rule with comment period. The first accounting statement, Table 204, illustrates the classification of expenditures for the CY 2025 estimated hospital OPPS incurred benefit impacts associated with the final CY 2024 OPD fee schedule increase. The second accounting statement, Table 205, illustrates the classification of expenditures associated with the 3.1 percent CY 2025 update to the ASC payment system, based on the provisions of the proposed rule and the baseline spending estimates for ASCs. Both tables classify most estimated impacts as transfers. The third accounting statement, Table 206, contains the classification of the costs associated with the finalized health and safety standards for obstetrical services in hospitals and critical access hospitals. This includes the total cost, benefits and transfers as outlined in the collection of information section in Table 200, and the regulatory impact analysis as provided in Table 231. Since there are no transfers and we are not able to quantify the benefits of these provisions, we do not include them in the table. This statement provides our best estimate for the finalized health and safety standards for obstetrical services in hospitals and critical access hospitals provisions. Finally, the fourth accounting statement, Table 207, shows the impact of the Medicaid clinic services four walls exceptions included in this final rule. Table 207 illustrates the classification of expenditures associated with the addition of exceptions to the Medicaid clinic services four walls requirement for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas. The table classifies the estimated impacts as transfers and they are discounted at a rate of 2 percent.
( printed page 94561)3. Effects of Changes in Requirements for the Hospital Outpatient Quality Reporting (OQR) Program
a. Background
We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 81961 through 82012) for the previously estimated effects of changes to the Hospital OQR Program for the CY 2026 payment determination and subsequent years. Of the 3,062 hospital outpatient departments (HOPDs) that met eligibility requirements for the CY 2024 payment determination for the Hospital OQR Program, we determined that 109 HOPDs did not meet the program requirements to receive the full annual Outpatient Department (OPD) fee schedule increase factor while an additional 58 HOPDs elected not to participate. In the CY 2025 OPPS/ASC proposed rule, we erroneously stated that 117 HOPDs did not meet the program requirements to receive the full annual OPD fee schedule increase factor (89 FR 59553); we are correcting the number to 109 HOPDs in this final rule.
b. Impact of CY 2025 OPPS/ASC Final Rule Policies
In this final rule with comment period, we are adopting four measures: (1) the Hospital Commitment to Health Equity (HCHE) measure, beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for Social Drivers of Health (SDOH) measure, beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; (3) the Screen Positive Rate for SDOH measure, beginning with voluntary reporting for the CY 2025 reporting period and mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (4) the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM), beginning with voluntary reporting for the CY 2026 reporting period and mandatory reporting beginning with the CY 2027 reporting period/CY 2029 payment determination.
In addition, we are removing two claims-based measures beginning with the CY 2025 reporting period/CY 2027 payment determination: (1) the MRI Lumbar Spine for Low Back Pain measure; and (2) the Cardiac Imaging for Preoperative Risk Assessment for Non-Cardiac, Low-Risk Surgery measure.
We are further requiring electronic health record (EHR) technology to be certified to all electronic clinical quality measures (eCQMs) in the Hospital OQR Program measure set and available to report for the CY 2025 reporting period/CY 2027 payment determination and subsequent years.
We are modifying the public reporting of data for the Median Time from Emergency Department (ED) Arrival to ED Departure for Discharged ED Patients (Median Time for Discharged ED Patients)—Psychiatric/Mental Health Patients stratification so that it may be published on Care Compare in addition to the data.cms.gov downloadable files beginning in CY 2025.
Lastly, we are finalizing our proposal regarding our immediate measure suspension policy in the Hospital OQR Program such that in cases where we determine there is evidence that the collection and reporting of a measure raises potential patient safety concerns, we would suspend the measure from the program (as applicable) until potential removal can be proposed through the rulemaking process. This policy will not result in any additional burden or costs for hospitals as its impact would only be to reduce the number of measures hospitals are required to report, if necessary.
We refer readers to section XXVI.B (Collection of Information) of this final rule with comment period for a detailed discussion of the calculations estimating the changes to the information collection and reporting burden for finalized data requirements under the Hospital OQR Program for the estimated 3,200 program-eligible HOPDs. A summary table (see Table 180) shows an estimated total information collection and reporting burden increase of 18,342,190 hours at a cost of $449,266,093 annually associated with our policies for the CY 2027 reporting period/CY 2029 payment determination and subsequent years compared to our currently approved information collection burden estimates.
In section XIV.B.1 of this final rule with comment period, we are adopting the HCHE measure. For HOPDs For HOPDs to receive a point for each of the domains in the measure, affirmative attestations are required for each of the elements within a domain. To attest affirmatively to all the domains in the measure, HOPDs may incur costs associated with activities such as updating facility policies, engaging senior leadership, participating in new quality improvement activities, performing additional data analysis, and training staff. The extent of these costs will vary depending on what activities the HOPD is already performing, HOPD size, and the choices each HOPD makes in order to meet the criteria necessary to attest affirmatively.
In section XIV.B.2 of this final rule with comment period, we are adopting the Screening for SDOH measure. HOPDs that are not currently administering some screening mechanism and elect to begin doing so as a result of this measure adoption will ( printed page 94562) likely incur some non-recurring costs associated with changes in workflow and information systems to collect the data. The extent of these costs is difficult to quantify as different HOPDs may utilize different modes of data collection (for example, paper-based, electronically patient-directed, clinician-facilitated, etc.). In addition, depending on the method of data collection utilized, the time required to complete the screening may add a negligible amount of time to patient visits.
In section XIV.B.3 of this final rule with comment period, we are adopting the Screen Positive Rate for SDOH measure, which will not result in any additional economic impacts beyond those discussed for the associated Screening for SDOH measure or in section XXVI.B (Collection of Information) of this final rule with comment period.
In section XV.C.1.b of this final rule with comment period, we are adopting the Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (Information Transfer PRO-PM). For HOPDs that are not currently collecting these data and elect to begin doing so as a result of this measure there will be some costs associated with changes in workflow and information systems to collect the data. The extent of these costs is difficult to quantify as HOPDs may utilize different modes of data collection (collected by facilities or authorized third-party vendors post-discharge through a web-based survey instrument, distributed electronically) and have differing response rates influencing data volume. While we assume the majority of hospitals will report data for this measure directly to CMS, we assume some hospitals may elect to submit measure data via a third-party survey vendor, for which there are associated costs. Under OMB control number 0938-1240 for the Outpatient and Ambulatory Surgery Consumer Assessment of Healthcare Providers and Systems (OAS CAHPS) Survey (expiration date November 30, 2026), an estimate of approximately $4,000 per hospital is used to account for these costs.
In section XV.E.2.b of this final rule with comment period, we are requiring EHR technology to be certified to all eCQMs available to report. We do not expect HOPDs will experience an increase in information collection burden for the Hospital OQR Program as discussed in section XXVI.B (Collection of Information) of this final rule with comment period, because this policy does not require HOPDs to submit new data to CMS and the use of EHR technology that is certified to all available eCQMs is already required for the Medicare Promoting Interoperability Program (83 FR 41672) and the Hospital Inpatient Quality Reporting (IQR) Program (84 FR 42604). In addition, due to the differences in the build of respective CEHRT deployed in HOPDs, the mapping required to capture required data for measure calculation, and the range of HOPD participation in the development, implementation, and testing of new CEHRT functionality, an estimated cost impact of the policy is not quantifiable as it will vary by CEHRT and HOPD. For certifying a new eCQM in the measure set specifically, we expect some costs for HOPDs so that the eCQM is available for HOPDs to report.
In section XV.F.2 of this final rule with comment period, we are finalizing to publicly report data for the Median Time for Discharged ED Patients—Psychiatric/Mental Health Patients stratification on Care Compare, which will not result in any additional economic impacts because we are not requiring HOPDs to collect or submit any additional data for purposes of this public reporting.
4. Effects of Changes in Requirements for the Rural Emergency Hospital Quality Reporting (REHQR) Program
a. Background
We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82149) for the previously estimated effects of changes to the REHQR Program for the CY 2024 reporting period and subsequent years. For the CY 2025 reporting period, we have estimated there will be 33 REHs required to report under the REHQR Program based on hospital conversions as of September 27, 2024. We use this number of REHs for our impact analyses knowing that more jurisdictions will pass or amend necessary legislation enabling transitions, acknowledging that the number of conversions could be less than or significantly greater than this estimate with time.
b. Impact of CY 2025 OPPS/ASC Final Rule Policies
In this final rule with comment period, we are adopting three measures: (1) the Hospital Commitment to Health Equity (HCHE) measure beginning with the CY 2025 reporting period; (2) the Screening for Social Drivers of Health (SDOH) measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period; and (3) the Screen Positive Rate for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period. We are also extending the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure beginning with the CY 2025 reporting period.
We refer readers to section XXVI.C (Collection of Information) of this final rule with comment period for a detailed discussion of the calculations estimating the changes to the information collection and reporting burden for finalized data requirements under the REHQR Program for the estimated 33 REHs. A summary table (see Table 182) demonstrates an estimated total information collection and reporting burden for 33 REHs of 12,996 hours at a cost of $318,827 annually associated with our policies for the CY 2026 reporting period/CY 2028 program determination and subsequent years.
In section XIV.B.1 of this final rule with comment period, we are adopting the HCHE measure. For REHs to receive a point for each of the domains in the measure, affirmative attestations are required for each of the statements within a domain. To attest affirmatively to all of the domains in the measure, REHs may incur costs associated with activities such as updating facility policies, engaging senior leadership, participating in new quality improvement activities, performing additional data analysis, and training staff. The extent of these costs will vary depending on what activities the REH is already performing, and the individual choices each REH makes in order to meet the criteria necessary to attest affirmatively.
In section XIV.B.2 of this final rule with comment period, we are adopting the Screening for SDOH measure. REHs that are not currently administering some screening mechanism and elect to begin doing so as a result of this measure adoption will likely incur some costs associated with changes in workflow and information systems to collect the data. The extent of these costs is difficult to quantify as different REHs may utilize different modes of data collection (for example paper-based, electronically patient-directed, clinician-facilitated, etc.). In addition, depending on the method of data collection utilized, the time required to complete the screening may add a negligible amount of time to patient visits. ( printed page 94563)
In section XIV.B.3 of this final rule with comment period, we are adopting the Screen Positive Rate for SDOH measure, which will not result in any additional economic impacts beyond those discussed for the associated Screening for SDOH measure or in section XXVI.C (Collection of Information) of this final rule with comment period.
In section XVI.C.2 of this final rule with comment period, we are extending the reporting period for the Risk-Standardized Hospital Visits Within 7 Days After Hospital Outpatient Surgery measure from one to 2 years and establishing when an REH will be required to begin submitting data under the REHQR Program after converting to REH status, which will not result in any additional economic impacts because we are not requiring REHs to collect or submit any additional data.
5. Effects of Changes in Requirements for the Ambulatory Surgical Center Quality Reporting (ASCQR) Program
a. Background
We refer readers to the CY 2024 OPPS/ASC final rule with comment period (88 FR 82143) for the previously estimated effects of changes to the ASCQR Program for the CY 2027 payment determination. In section XVII of this final rule with comment period, we discuss our proposals affecting the ASCQR Program. Based on the most recent analysis of the CY 2024 payment determination data, we found that, of the 5,536 ambulatory surgical centers (ASCs) that were actively billing Medicare, 4,196 were required to participate in the ASCQR Program. Of the 1,340 ASCs not required to participate in the program, 279 ASCs did so and met full requirements. On this basis, we estimate that 4,475 ASCs (4,196 + 279) will submit data for the ASCQR Program for the CY 2025 reporting period unless otherwise noted. We note that this estimate is a decrease of 334 ASCs from our estimate of 4,809 provided in the CY 2024 OPPS/ASC final rule with comment period (88 FR 82143) due to results from more recent data analysis regarding numbers of eligible ASCs.
b. Impact of CY 2025 OPPS/ASC Final Rule Policies
In section XIV.B of this final rule with comment period, we are adopting three measures: (1) the Facility Commitment to Health Equity (FCHE) measure beginning with the CY 2025 reporting period/CY 2027 payment determination; (2) the Screening for Social Drivers of Health (SDOH) measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination; and (3) the Screen Positive Rate for SDOH measure beginning with voluntary reporting for the CY 2025 reporting period followed by mandatory reporting beginning with the CY 2026 reporting period/CY 2028 payment determination.
In addition, we are finalizing our proposal regarding our immediate measure suspension policy in the Hospital OQR Program such that in cases where we determine there is evidence that the collection and reporting of a measure raises potential patient safety concerns, we would suspend the measure from the program (as applicable) until potential removal can be proposed through the rulemaking process. This policy will not result in any additional burden or costs for hospitals as its impact would only be to reduce the number of measures hospitals are required to report, if necessary.
We refer readers to section XXVI.D (Collection of Information) of this final rule with comment period for a detailed discussion of the calculations estimating the changes to the information collection and reporting burden for finalized data requirements under the ASCQR Program for the estimated 4,475 program-eligible ASCs. A summary table (see Table 184) demonstrates an estimated total information collection and reporting burden increase for 4,475 ASCs of 712,971 hours at a cost of $17,529,790 annually associated with our policies for the CY 2026 reporting period/CY 2028 payment determination and subsequent years, compared to our currently approved information collection burden estimates.
In section XIV.B.1 of this final rule with comment period, we are adopting the FCHE measure. For ASCs to receive a point for each of the domains in the measure, affirmative attestations are required for each of the statements within a domain. To attest affirmatively to all of the domains in the measure, ASCs may incur costs associated with activities such as updating facility policies, engaging senior leadership, participating in new quality improvement activities, performing additional data analysis, and training staff. The extent of these costs will vary depending on what activities the ASC is already performing, ASC size, and the individual choices each ASC makes in order to meet the criteria necessary to attest affirmatively.
In section XIV.B.2 of this final rule with comment period, we are adopting the Screening for SDOH measure. ASCs that are not currently administering some screening mechanism and elect to begin doing so as a result of this measure adoption will likely incur some non-recurring costs associated with changes in workflow and information systems to collect the data. The extent of these costs is difficult to quantify as different ASCs may utilize different modes of data collection (for example paper-based, electronically patient-directed, clinician-facilitated, etc.). In addition, depending on the method of data collection utilized, the time required to complete the screening may add a negligible amount of time to patient visits.
In section XIV.B.3 of this final rule with comment period, we are adopting the Screen Positive Rate for SDOH measure, which will not result in any additional economic impacts beyond those discussed for the associated Screening for SDOH measure or in section XXVI.D (Collection of Information) of this final rule.
6. Effects of Changes in Requirements for the Hospital Inpatient Quality Reporting (IQR) Program
In section XXII of this final rule with comment period, we are finalizing that for the FY 2026 and FY 2027 payment determinations, the submission of core clinical data elements and linking variables associated with the Hybrid Hospital-Wide Readmission (HWR) measure and the Hybrid Hospital-Wide All-Cause Risk Standardized Mortality (HWM) measure will remain voluntary. This policy would not result in any economic impacts because we are not modifying either measure.
7. Effects of Changes for Individuals Currently or Formerly in the Custody of Penal Authorities
a. Medicare FFS No Legal Obligation To Pay Payment Exclusion and Incarceration (Revisions to 42 CFR 411.4)
The individuals currently or formerly in the custody of penal authorities provisions are discussed in section XXIII of this final rule with comment period. Section XXIII of this final rule with comment period describes the “no legal obligation to pay” payment exclusion codified in regulation at § 411.4. Specifically, we are narrowing the description of custody in § 411.4(b) and removing “under arrest” from the definition of “custody” because we no longer believe that certain classes of individuals should be presumed to be in custody for purposes of the no legal obligation to pay payment exclusion, ( printed page 94564) and we are including in regulation an illustrative list of individuals who are not considered to be in “custody.” We are also reorganizing and renumbering the regulation at § 411.4(b), making certain non-substantive edits to § 411.4(a) to align the regulatory text with the statutory no legal obligation to pay payment exclusion, and defining “penal authority.”
We expect that narrowing the description of “custody” will reduce burden for individuals on bail, parole, probation, home detention or who are required to reside in halfway houses and those providers and suppliers that treat them, because it will no longer be necessary to rebut the presumption that such individuals do not have a legal obligation to pay for their own healthcare in order for Medicare to pay for their health care items or services. These revised policies will ensure that Medicare properly pays for services for individuals who are on bail, parole, probation, home detention or who are required to reside in halfway houses. We believe that these policies will have a negligible impact on Medicare costs, as they do not add new covered services or benefits; rather, the revisions merely remove a real or perceived barrier so that individuals on bail, parole, probation, home detention or who are required to reside in halfway houses can more easily access the Medicare benefits for which they are legally entitled. We received no comments on the financial impact of our proposals.
b. Revision to Medicare Special Enrollment Period for Formerly Incarcerated Individuals
We proposed to revise the eligibility criteria for the Medicare special enrollment period (SEP) for formerly incarcerated individuals at §§ 406.27(d)(1) and 407.23(d)(1). Specifically, for releases on and after January 1, 2025, we proposed to base the determination of when an individual is no longer incarcerated on SSA's data collected in its systems for determining OASDI benefit suspensions in section 202(x)(1)(A) of the Act and any additional documentation provided by individuals to demonstrate that they have been released from incarceration. Our proposal would limit the current eligibility criteria for this SEP, which reference to the Medicare payment exclusion at § 411.4(b), to releases between January 1, 2023, and December 31, 2024.
The SEP for formerly incarcerated individuals at §§ 406.27(d)(1) (for Premium Part A) and 407.23(d)(1) (for Part B) starting in 2023 provides eligible individuals an opportunity to enroll in Medicare upon release from incarceration without waiting for the General Enrollment Period (GEP) and facing penalties for delayed enrollment. We anticipated that the proposed revisions to the SEP for formerly incarcerated individuals would provide clarity and make accessing this SEP easier upon release from incarceration, especially for a population facing many challenges reintegrating into society. However, we did not anticipate a significant impact on utilization of the SEP since there is no evidence that the current requirements have created barriers to those who want to use the SEP. As a result of this assumption, we expected a negligible impact on Medicare costs. We did not receive any comments related to this assumption.
8. Estimated Effects of Medicaid Clinic Services Four Walls Exceptions
a. Background
As discussed in more detail in section XVIII of this final rule, we are finalizing our proposal to add exceptions to the four walls requirement under the Medicaid clinic services benefit for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas. The exception for IHS/Tribal clinics will be mandatory for States that cover the clinic services benefit while the exceptions for behavioral health clinics and clinics located in rural areas will be at State option. We believe that these additional exceptions to the four walls requirement will help States strengthen and improve access to Medicaid clinic services for the populations served by IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas, and that they are responsive to the concerns we have heard from Tribes, the TTAG, the STAC, States, and other interested parties. In addition, we believe this final rule will advance health equity and improve health care access for the populations served by IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas.
b. Overall Estimated Effects of Medicaid Clinic Services Four Walls Exceptions
The aggregate economic impact of this final rule is estimated to be $1.18 billion in transfers for fiscal years 2025-2029. This includes a Federal impact of $1.15 billion and impacts to States of $30 million. For the purposes of this analysis, we estimated the impacts separately for Medicaid clinic services furnished outside of the four walls for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas. Uncertainties in the estimate result in an estimated range of $554 million to $1.82 billion in the Federal impact and a range of $7 million to $95 million in the State impact.
Current Medicaid clinic services expenditures were estimated using financial reporting for 2022. Estimated expenditures for IHS/Tribal Medicaid clinic services represent those expenditures not attributable to the following Medicaid services: inpatient hospital, outpatient hospital, prescription drugs, FQHC, and RHCs; in 2022, these expenditures included expenditures for IHS/Tribal Medicaid clinic services provided outside of the four walls (due to the grace period discussed below). We assumed that 15 percent of expenditures for Medicaid clinic services were related to behavioral health based on general behavioral health utilization and spending patterns. We assumed that 17 percent of remaining Medicaid clinic services expenditures were attributable to clinics in rural areas based on 17 percent of the Medicaid population residing in rural areas. Estimated baseline Federal Medicaid expenditures for Medicaid clinic services in 2025 are $934 million at IHS/Tribal clinics, $530 million for behavioral health services provided at Medicaid clinics, and $495 million for Medicaid clinic services provided in rural areas. The estimates for behavioral health services provided at Medicaid clinics and Medicaid clinic services provided in rural areas do not include Medicaid clinic services expenditures from IHS/Tribal clinics.
It is important to note that IHS/Tribal clinic services provided outside of the clinic's four walls are currently being paid for by Medicaid programs, under a CMS “grace period” that currently extends through February 11, 2025. For a more detailed discussion on this grace period please see section XVIII.A of this final rule. With the finalization of the exception for IHS/Tribal clinics in this final rule, States will be permitted to pay for Medicaid clinic services provided outside of the four walls of an IHS/Tribal clinic after February 11, 2025, when the grace period ends.
Table 208 demonstrates our estimates for the economic impact of an exception to the Medicaid clinic services four walls requirement for IHS/Tribal clinics. For the IHS/Tribal clinic exception at 42 CFR 440.90(c), we assumed that 19 percent of current total IHS/Tribal clinic services expenditures were for services provided outside of clinics, based on information provided by the Tribes. Allowing current claiming practices to continue, trended for changes in expected cost, utilization, and enrollment each year, we estimate that ( printed page 94565) Federal expenditures for services provided outside of clinics will be $1.09 billion for fiscal years 2025 through 2029. State expenditures on Medicaid clinic services provided to AI/AN Medicaid beneficiaries by IHS/Tribal clinics are Federally matched at 100 percent. State expenditures on Medicaid clinic services provided to Medicaid beneficiaries who are not AI/AN are matched at the otherwise applicable Federal matching percentage, which is generally less than 100 percent. The estimate assumes 100 percent Federal share for all IHS/Tribal clinic services expenditures, but we acknowledge that a very small portion of these IHS/Tribal clinic services expenditures may be attributed to Medicaid beneficiaries who are not AI/AN, resulting in some State expenditures. Data from which to estimate these State expenditures were unavailable for this analysis. We note that this impact estimate only reflects or accounts for the grace period through February 11, 2025; the baseline for the impact estimate does not reflect or account for the grace period for dates after the expiration of the grace period. From February 12, 2025, forward, the estimate compares projections under current law, which does not allow States to pay for Medicaid clinic services provided outside of clinics against projections under the final rule, which would permit States to pay for IHS/Tribal clinic services provided outside of clinics. When the grace period is factored into the analysis for dates after the expiration of the grace period and spending under the final rule is compared to expenditures under current practice, which allows payment for clinic services provided outside of IHS/Tribal clinics due to the grace period, we estimate little to no impact.
Tables 209 and 210 demonstrate our estimates for the economic impact of exceptions to the four walls requirement under the Medicaid clinic services benefit for behavioral health clinics and clinics located in rural areas that are not IHS/Tribal clinics. We acknowledge that we included in the final rule text that a State adopting the exception for clinics located in rural areas must include a definition of rural area in its State plan that must be either a definition adopted and used by a Federal governmental agency for programmatic purposes, or a definition adopted by a State governmental agency with a role in setting State rural health policy. For purposes of our estimates of the economic impact of our exception to the four walls requirement for clinics located in rural areas, our analysis defines rural areas using the RUCA classifications. We also acknowledge that our exception to the four walls requirement for behavioral health clinics includes any clinic services furnished outside of the four walls by a behavioral health clinic, including non-behavioral clinic services such as physical health services. However, for purposes of our economic impact we are unable to quantify the cost of non-behavioral clinic services. For our behavioral health clinic exception at 42 CFR 440.90(d) and clinics located in rural areas exception at 42 CFR 440.90(e), we assumed a 5 percent increase in current spending in each category due to increased payments for clinic services performed outside of the four walls. Growth in utilization and expenditures for clinic services provided both by behavioral health clinics and clinics in rural areas is expected to be limited by provider shortages in these areas of practice. Because the exceptions at 42 CFR 440.90(d) and (e) are at State option, we assume that States representing 25 percent of States providing coverage of the Medicaid clinic services benefit will implement one or both of the optional exceptions. Estimated expenditures are trended each year for changes in expected cost, utilization, and enrollment. We estimate that Federal expenditures will be $35 million for fiscal years 2025 through 2029 for clinic services furnished by behavioral health clinics, and $30 million for fiscal years 2025 through 2029 for clinic services furnished by clinics in rural areas.
( printed page 94566)Projected Medicaid clinic services expenditures may differ from our current estimates, including the amounts broken out for IHS/Tribal clinics, clinic services provided by behavioral health clinics, and clinic services provided by clinics in rural areas. There is uncertainty in how much current and projected IHS/Tribal Medicaid clinic services spending is attributable to Medicaid clinic services provided outside of the four walls. The IHS/Tribal clinic impact may range from $544 million to $1.63 billion over 5 years due to uncertainty in the level of spending for Medicaid clinic services provided outside of IHS/Tribal clinics. Uncertainty in provider availability and beneficiary demand result in uncertainty in the potential for changes in utilization and costs. The Federal impact for Medicaid clinic services furnished by behavioral health clinics may range from $5 million to $90 million and the Federal impact for clinic services furnished by clinics in rural areas may range from $5 million to $100 million over 5 years. State impacts over 5 years may range from $5 million to $45 million for clinic services furnished by behavioral health clinics and $2 million to $50 million for clinic services furnished by clinics in rural areas.
Table 211 demonstrates the total economic impact for our finalized exceptions to the four walls requirement under the Medicaid clinic services benefit for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas. The total estimated impact of these exceptions over 5 years is $1.18 billion, including Federal impact of $1.15 billion and State impact of $30 million. The impact may range from a low of $561 million to a high of $1.92 billion, including a range in the Federal estimate of $554 million to $1.82 billion and a range in the State impact of $7 million to $95 million.
( printed page 94567)c. Benefits of Medicaid Clinic Services Four Walls Exceptions
The changes to the Medicaid clinic services benefit are expected to benefit Medicaid beneficiaries, Tribes, and States by improving access to care for the populations served by IHS/Tribal clinics, behavioral health clinics, and clinics in rural areas. The exceptions to the four walls requirement under the Medicaid clinic services benefit for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas would help improve access to care for these clinics' patient populations by allowing services to be furnished where the beneficiary is located. We refer readers to section XVIII.B of this final rule for more robust discussions on how the populations served by these clinics might benefit from exceptions to the Medicaid clinic services benefit four walls requirement and how these exceptions would improve access to care. These potential benefits cannot be monetarily quantified at this time.
d. Alternative Medicaid Clinic Services Four Walls Exceptions Considered
We considered a few different alternatives in determining the best way to address the concerns we heard from Tribes, the TTAG, the STAC, States, and other interested parties about the four walls requirement under the Medicaid clinic services benefit. We considered including an exception to the four walls requirement only for the population served by IHS/Tribal clinics, but we viewed that alternative as too limited. As we discuss in detail in section XVIII.B of this final rule, we concluded that the patient populations served by behavioral health clinics and clinics in rural areas might also benefit from exceptions to the four walls requirement for those clinics.
We also considered proposing an exception, in addition to the three exceptions we are finalizing in this rule, for any other populations that are identified by States as likely to meet the four criteria described in this final rule that warrant an exception to the four walls requirement and that have no alternative access to services through Medicaid benefits that are not subject to a four walls requirement under Federal Medicaid law. Ultimately, it is our understanding that other populations are better able to access services through Medicaid benefits to which a four walls requirement does not apply under Federal Medicaid law (for example, FQHC services, RHC services, outpatient hospital services, etc.) than the populations targeted by the exceptions we are finalizing. As we indicate in section XVIII.B of this final rule, we invited comment in the proposed rule on our assumptions about other populations that may benefit from an exception to the four walls requirement under the Medicaid clinic services benefit. We are finalizing this rule without including exceptions to the four walls requirement for additional populations (beyond the exceptions for IHS/Tribal clinics, behavioral health clinics, and clinics located in rural areas), and a summary of the comments received that recommend additional exceptions and our response can be found in section XVIII.B of this final rule.
We also considered making the exceptions to the four walls requirement mandatory for behavioral health clinics and clinics located in rural areas, but, as we discuss in more detail in section XVIII.B of this final rule, it is our understanding that there is greater State variability in the degree to which the populations targeted by the behavioral health and rural exceptions meet the four criteria we identified than there is for the population served by IHS/Tribal clinics. We also invited public comment on these assumptions in section XVIII.B of the CY 2025 OPPS/ASC proposed rule. We are finalizing that the exceptions to the four walls requirement for behavioral health clinics and clinics located in rural areas are at state option, and a summary of the comments received on this topic and our response can be found in section XVIII.B of this final rule.
Finally, as we discuss in section XVIII.B of this final rule, we did not propose a specific definition of rural for our exception for clinics located in rural areas and in the proposed rule invited public comment on the alternative possible definitions we considered and described in that section of the proposed rule. The final rule provides that a State must include a definition of rural area in its State plan that must be either a definition adopted and used by a Federal governmental agency for programmatic purposes, or a definition adopted by a State governmental agency with a role in setting State rural health policy. A summary of the comments received on the options we considered for defining rural for clinics located in rural areas and our response can be found in section XVIII.B of this final rule.
9. Effects of Continuous Eligibility in Medicaid and CHIP
As discussed in section XX of this final rule with comment period, we are finalizing our proposal to codify the requirement of the CAA, 2023 for States to provide 12 months of continuous eligibility for children under age 19 enrolled in Medicaid and CHIP, with limited exceptions. In addition, we are finalizing our proposal to remove the option to disenroll children from CHIP during a continuous eligibility period due to failure to pay premiums. These regulation changes implement the statutory requirement in section 5112 of Title V, subtitle B of the Consolidated Appropriations Act, 2023, and the entirety of the economic impact ( printed page 94568) discussed in this section below follows from this statutory provision.
The requirement to provide 12-months of continuous eligibility in Medicaid and CHIP is estimated to increase annual average enrollment in Medicaid and CHIP by approximately 124,000 by 2028 (75,000 in Medicaid and 49,000 in CHIP). The total estimated impact of this requirement over 5 years is $2,466 million, including Federal impact of $1,592 million and State impact of $874 million. Enrollment may range from an increase of around 92,000 to an increase of around 159,000 by 2028. The total impact may range from a low of $1,837 million to a high of $3,154 million, including a range in the Federal estimate of $1,185 million to $2,039 million and a range in the State impact of $652 million to $1,115 million. (See Tables 212 and 213.)
10. Effects of Requirements for Obstetrical Services in Hospitals and Critical Access Hospitals (CAHs)
In response to the CY 2025 OPPS/ASC proposed rule, we received multiple comments regarding the cost estimates for the proposed requirements for obstetrical services in hospitals and critical access hospitals (CAHs). Most of the comments were from professional organizations, State agencies, and hospital systems. In this final rule with comment period, we provide a summary of the public comments received for each proposal and our responses to them, including relevant changes in the RIA methodology and estimate.
Comment: A few commenters said that CMS should consider the disproportionate financial impact that the requirements will have on some provider types, particularly hospitals and CAHs that are most at risk of closing their obstetrical services units, which could lead to increased burden on other hospitals and result in patients having to travel greater distances to access maternal care.
Response: We appreciate the suggestions to consider differences across providers. The proposed rule assessed likely variation in burden across different providers, taking into account variation in facility size, the number of patients, and staff size that we believe accurately reflects the likely burden for each facility type. In this final rule with comment period, we have also taken several steps to reduce burden and ensure that all hospitals and CAHs are able to successfully meet the requirements. First, we are implementing the requirements in three phases over the course of two years to ensure that facilities have adequate time to meet these requirements and to reduce any potential implementation ( printed page 94569) burden or unintended consequences (see section XXI.A.7 of this final rule with comment period). Second, we are reducing the frequency of the obstetrical services training requirement from annually to biannually (every other year). Finally, as we outline below, we are not finalizing the requirement that hospitals and CAHs with obstetrical services delineate and document obstetrical privileges for all practitioners.
a. Organization, Staffing and Delivery of Services for Hospitals (§ 482.59(a) Through (b)) and CAHs (§ 482.649(a) Through (b))
As indicated in section XXI.C.2 of this final rule with comment period, we are finalizing the policies for hospitals (§ 482.59(a) through (b)) and CAHs (§ 485.649(a) through (b)) that provide obstetrical (OB) services as proposed with the modification to reference the requirements of the medical staff bylaws for hospitals and CAHs rather than requiring hospitals and CAHs with OB services to delineate and document obstetrical privileges for all practitioners. In addition, we are clarifying that basic equipment must be kept at the hospital and CAH and be readily available for treating obstetrical cases to meet the needs of patients. Below, we provide the comments we received on the proposed requirements as well as our modifications to the final cost estimate based on these comments and the final policies.
Comment: Some commenters said that the proposed rule underestimated the obstetrical services' equipment costs since it did not consider the cost for maintaining and replacing required equipment. Other commenters said that the requirement for hospitals and CAHs with OB services to have cardiac monitors and call-in systems will result in a higher cost than estimated since specialized training is needed for nurses to correctly monitor cardiac monitors and facilities will also need to purchase central monitoring stations. Some commenters said that the requirement for each room/suite to have a call-in system, cardiac monitor, and a fetal doppler or monitor was too burdensome and would result in equipment going unused. Given this issue, they asked for flexibility to have the equipment available on the unit. Other commenters questioned the decision to mandate specific equipment for OB services, noting that evolving practice guidelines may lead necessary equipment to change and requiring specific equipment could prevent the adoption of new technology.
Response: We agree that replacing equipment will add additional burden for facilities. Based on our experience, we estimate that the required equipment will need to be replaced once every 5 years. As a result, in the regulatory impact analysis below, we include additional burden in year 6 for hospitals and CAHs with OB services to replace cardiac monitors, fetal monitor or dopplers, and call-in systems. As we noted in our comment response in section XXI.C.2 of this final rule with comment period, several commenters asked for clarity regarding the expected availability of basic equipment, noting that it would be costly to purchase equipment for every room/suite. In that section and in the regulation text, we have clarified that required equipment includes a call-in-system (we note that for the purposes of this regulation, call-in system describes a call button), cardiac monitor, and fetal doppler or monitor kept at the hospital and CAH and readily available for treating obstetrical cases to meet the needs of patients in accordance with the scope, volume, and complexity of services offered. Further, facilities must have adequate provisions that include equipment, supplies and medication used in treating emergency cases. We would note that while facilities may choose to purchase central monitoring equipment, they are not required to do so. In addition, as we have clarified in this final rule with comment period, we are requiring equipment to be readily available for treating obstetrical cases in accordance with the scope, volume, and complexity of services offered but are not requiring facilities to have equipment in every room/suite in the OB unit. We expect that hospitals and CAHs already have staff members in the facility who are able to correctly monitor the equipment and that if that expertise was not available in the OB unit or if facilities have special areas for patients utilizing cardiac monitors, patients would be moved to areas where staff already have that knowledge. As such, we are not including burden for training staff members to monitor equipment and purchasing central monitoring systems. Rather than maintaining our assumption in the proposed rule that facilities will have equipment for twice the average number of daily patients per facility, we have modified the estimated cost based on commenters indicating that facilities were concerned that they would have a lot of unused equipment. In our final estimates, we now assume that with an average daily birth volume of 2.04 per facility, facilities will ensure that they will have supplies for four patients. Finally, we disagree that requiring equipment will prevent hospitals and CAHs from adopting new technology designed to improve maternal care. The equipment we are requiring is basic equipment to ensure the health and safety of pregnant and postpartum patients. We expect that hospitals and CAHs that are planning on investing in new technology to advance maternal healthcare would likely ensure that basic equipment is available first even without our requirements.
Comment: A few commenters said that the CY 2025 OPPS/ASC proposed rule did not consider the cost for attracting and retaining qualified medical personnel.
Response: We disagree with the comment that we did not consider the cost for attracting qualified medical personnel. In Table 144 of the CY 2025 OPPS/ASC proposed rule, we estimated that hiring a qualified OB supervisor will cost, on average, $54,757 per hospital and CAH. In addition, as we noted in section XXI.C.2, many hospitals and CAHs already have qualified practitioners on staff and would not need to hire additional staff members. The CY 2025 OPPS/ASC proposed rule included no fixed minimum staffing requirements and instead allows facilities with flexibility to provide services that are well-organized and in accordance with nationally recognized acceptable standards of practices.
Based on the finalized policies and the comments received, we are making the following modification below to the estimated costs for these finalized proposals: including additional cost in year 6 to account for equipment replace costs.
In section XXVI of this final rule with comment period, we have estimated the cost for hospitals and CAHs to develop internal standards and protocols to ensure that services are well organized and to provide high-quality care that is appropriate to the level of services provided and integrated with other departments of the facility, as well as to ensure compliance with nationally recognized and evidence-based guidelines for OB emergencies, complications, immediate post-delivery care, and other patient health and safety events. Below, we estimate the cost for ensuring that OB patient care units ( i.e., labor rooms, delivery rooms, including rooms for operative delivery, and post-partum/recovery rooms whether combined or separate) are supervised by an individual with the necessary education and training, and specify that person should be an experienced ( printed page 94570) registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a doctor of medicine or osteopathy. We also estimate the cost for equipment that includes a call-in-system, cardiac monitor, and fetal doppler or monitor kept at the hospital and CAH and readily available for treating obstetrical cases to meet the needs of patients in accordance with the scope, volume, and complexity of services offered.
While hospitals and CAHs are likely to already have an individual supervising the OB patient care unit, there is variation across facilities regarding whether they have the necessary education and training related to OB patient care. Many facilities, especially larger hospitals that have large birth volumes, are likely to already have an experienced individual with the necessary education and training. Smaller facilities with lower birth volumes, in contrast, may be less likely to have an experienced individual and need to recruit a new individual to meet the proposed requirement.
Given uncertainty about the number of facilities that already have an experienced individual who will meet the requirement, we assume that each facility will need to hire one individual, who we assume will be a registered nurse, to meet the requirement. To estimate the cost of hiring this individual, we reviewed research related to the cost of registered nurse turnover. A review of academic literature found that each RN turnover cost employers between $21,514 and $88,000.[779] We take the midpoint of these two estimates, or $54,757 per individual hired. As shown, in Tables 214 and 215, we estimate that this requirement will cost facilities $268,842,496 in year 1 and over 10 years.
To estimate the cost for having basic equipment, including a call-in-system (we note that for the purposes of this regulation, call-in system describes a call button), cardiac monitor, and fetal doppler or monitor kept at the hospital and CAH and readily available for treating obstetrical cases to meet the needs of patients, we reviewed public data sources to estimate the cost of purchasing a call-in system, cardiac monitor, and a fetal doppler or monitor. While we were not able to identify public estimates for the price of call-in systems, based on our experience we estimate that they will cost $2,500 to $3,500 per system. Reviewing the web pages of various online suppliers, we found that fetal dopplers and fetal monitors cost between $502 and $8,995 [780 781] and cardiac monitors cost between $1,071 and $10,246.[782] For each of these systems, we use the mid-point of the price estimate and assume that each call-in system will cost $3,000, each fetal monitor or fetal doppler will cost $4,749, and each cardiac monitor will cost $5,659.
According to the Centers for Disease Control and Prevention there were a ( printed page 94571) total of 3,667,758 births in 2022.[783] With a total of 4,928 hospitals and CAHs with obstetrical units, this leads to an average of 744 (3,667,758 ÷ 4,928) births per hospital and CAH or an average of 2.04 births per facility per day. We estimate that each birth will take 1 day on average. To account for variation in birth volumes throughout the year, we assume that each facility will need to prepare for double the number of average births per day, or 4.08 patients. Although equipment cannot be divided, based on the public comments received and discussed above, we assume that cost-conscious facilities will have equipment available for 4 patients daily. We also assume that each facility already has one fetal monitor and cardiac monitor but do not assume that each facility has a call-in system.
Based on our experience working in obstetrical units, we estimate a fetal doppler or monitor and a call-in system will need to be available for each patient, a call-in system will need to be available for each patient, and cardiac monitors will need to be available for half the patients. As such, we estimate that each facility will need to purchase three fetal monitors or fetal dopplers at $14,247 ($4,749 × 3), one cardiac monitor at $5,659 ($5,659 × 1), and four call-in systems at $12,000 ($3,000 × 4) for an average per facility cost of $31,906. As indicate in Table 216 we expect that this requirement will cost $157,232,768 in year 1. We also expect that under normal use, this equipment will need to be replaced in five years. In Table 217, we estimate that this equipment requirement will cost a total of $314,465,536 over 10 years.
b. OB Staff Training for Hospitals (§ 482.59(c)) and CAHs (§ 485.649(c))
As indicated in section XXI.C.3 of this final rule with comment period, we are finalizing the policies for hospitals (§ 482.59(c)) and CAHs (§ 485.649(c)) that provide obstetrical (OB) services with the modification that the effective date of the staff training requirement is January 1, 2027 and that the governing body in hospitals and CAHs must identify and document which staff must complete an initial training and subsequent biannual training. Below, we provide the comments we received on the proposed requirements as well as our modifications to the final cost estimate based on these comments and the policies we are finalizing.
Comment: Several commenters expressed concern about the cost to train relevant OB staff. Other commenters said that the proposed rule underestimated training costs since it did not consider increases in wage and salaries for healthcare personnel. In addition, it did not consider staff turnover and the need to train new staff. One commenter thought that staff training could require more time than we have estimated.
Response: We appreciate the feedback regarding the cost estimates for OB staff training. As noted in section XXI.C.3 of this final rule with comment period, we are modifying the training requirements so that hospitals and CAHs will need to provide relevant OB staff with initial training upon hiring and then every other year, rather than every year. This reduction in training frequency reduces the training costs as outlined below in the regulatory impact analysis. We agree with the feedback regarding the need to ( printed page 94572) include wage growth for healthcare staff in the cost estimates. When estimating costs, registered nurses (RNs) are the largest group of staff members who we assume will likely receive training. According to the Bureau of Labor Statistics,[784] RNs in 2015 earned a mean hourly rate of $34.14 with the nominal mean hourly rate in 2023 [785] increasing to $45.42. Inflation,[786] however, accounts for 3.49 percentage points annually over these 8 years, leaving a compound annual real growth rate of approximately 0.44 percent. To address concerns that wage costs are underestimated, we are including a 0.44 annual increase in real wage rates in the cost estimates for the OB services training requirements starting in year 2. To address concern regarding staff turnover, we are including training costs for new staff members based on a 21 percent annual staff turnover rate.
We appreciate the comment regarding the time allocated for staff training. As noted in section XXI.C.3 of this final rule with comment period, hospitals and CAHs have flexibility to identify which evidence-based best practices and protocols they will train on to improve the delivery of maternal care within their facility. We are not prescribing a specific format the training be conducted in, allowing facilities to carry out the training in a manner that best fits their needs. While some facilities could choose training that takes more time than we have estimated, others could choose training that takes less time. As such, we maintain the estimated time burden for training requirements for hospitals and CAHs that offer OB services.
Based on the comments received and the finalized policies, we are making the following modifications to the cost estimates: (1) including a 0.44 percent annual increase in hourly rates for training costs starting in year 2 to take into account growth in wages and salaries for healthcare workers, (2) including additional burden to account for the cost of training new staff members, and (3) reducing the burden based on the modification that the governing body identify and document which staff must complete an initial training and subsequent biannual training rather than annual training.
In section XXVI of this final rule with comment period, we have estimated the cost for developing policies and procedures to ensure that staff are trained on key topics related to improving the delivery of maternal care, as well as documentating that training was completed and staff knowledge on these topics. We estimate that staff training on evidence-based best practices and protocols will take 2 hours per employee and that each facility will spend 1 hour training staff on additional topics identified by the facility's QAPI program. This leads to a total hourly burden of 3 hours per employee trained.
While hospitals and CAHs have flexibility regarding which OB staff will receive training, we expect that they will likely focus their training on medical staff who are working directly with OB patients. This includes surgeons, physicians, physician assistants, nurse practitioners, nurse midwives, nurse anesthetists, registered nurses (RNs), and Licensed Practical Nurses/Licensed Vocational Nurses (LPNs/LVNs).
To estimate the number of employees in CAHs and hospitals that are likely receive training, we first obtained data from the Bureau of Labor Statistics (BLS) on the number of surgeons, physicians, physician assistants, nurse practitioners, nurse midwives, nurse anesthetists, RNs, and LPNs/LVNs working in hospitals (NAICS 622000). Since the BLS does not provide separate employment statistics for CAHs and hospitals, we assume that the number of employees needing training and, henceforth, the cost to train them will be in proportion to the size of facilities, specifically the number of certified beds. We obtained information on the number of certified beds in hospitals and CAHs from CMS' Q1 2024 Provider of Services File—Hospital & Non-Hospital Facilities.[787] Using this database, we estimate that 98.6 percent of certified beds for facilities that provide OB services are in hospitals with the remaining 1.4 percent in CAHs.
In hospitals, which have a larger number of beds, there is likely to be a greater division of staff among units, with medical staff specifically designated to work in OB units and with pregnant patients, while other medical staff members will not work with pregnant patients. In contrast, critical access hospitals (CAHs), which are smaller in size, are likely to have medical staff that work across units. Based on our experience, we estimate that between 10 and 30 percent of medical staff in hospitals and 60 to 100 percent of medical staff in CAHs will receive the training. Given the variation for hospitals and CAHs, we take the midpoint of the two estimates and assume that 20 percent of hospital medical staff and 80 percent of medical staff in CAHs will receive training. As indicated in Table 218, if initial training for all staff who need the training began in year 1, it would cost approximately $151 million and take $1.43 million hours to complete annually. Our finalized requirement, however, is that applicable staff receive initial training and then every 2 years. Since hospitals have an annual turnover rate of approximately 21 percent,[788] we estimate that in addition to training all applicable staff as identified above, the number of staff members that hospitals and CAHs will need to train will increase by 21 percent due to turnover. Since the training requirement goes into effect in year 2, we expect there to be no burden for year 1. In year 2, we expect that facilities will need to train 121 percent of the applicable staff to account for initial training for all existing staff as well as staff turnover. In year 3, training will be needed for 21 percent of existing staff who will leave and be replaced by new staff. In year 4, 21 percent of employees hired in year 3 will leave and their replacements will need training. In addition, 79 percent of the employees who received training 2 years ago will need to receive their biannual training. For all subsequent years, the share of staff who need to receive training will be 21 percent of staff hired in the previous year to account for regular staff turnover and 79 percent of staff who received training 2 years prior.
To calculate the total hourly burden cost over 10 years, we multiply the share of staff that will need training each year by the baseline cost for training all staff members, taking into account an annual 0.44 percent increase in real wage rates. As indicated in Table 219, we estimate the requirements will have an average annual cost of approximately $85 million, with a total cost of $849,937,432 over 10 years. Over 10 years, the requirements are estimated to take $7,875,903 million hours to complete. ( printed page 94573)
This estimate is based on two assumptions. First, we assumed that when a staff member leaves, their replacement will be hired in the same year. If the replacement was not hired until the following year, then the cost would be lower than what we have estimated. Second, we assumed that employees who receive training and then quit 2 years after their training, would not receive their biannual training prior to leaving. If they did, then the actual cost would be higher than what we have estimated.
( printed page 94574) ( printed page 94575) ( printed page 94576)c. Quality Assessment and Performance Improvement Program (QAPI) for Hospitals (§ 482.21(b)(4)) and CAHs (§ 485.641(e)(2))
As indicated in section XXI.C.4 of this final rule with comment period, we are finalizing the proposed revision to existing QAPI standards (§ 482.21; § 485.641) for hospitals and CAHs that offer obstetrical services without modifications. Below, we provide the comments we received on the proposed requirements as well as our modifications to the cost estimates for the finalized revisions based on these comments.
Comment: Some commenters said that the proposed rule underestimated costs since it did not consider increases in wage and salaries for healthcare personnel. One commenter said that full utilization of quality improvement activities often requires significant financial investment and administrative support to be successful. Another commenter noted that many metrics used in obstetrical care cannot be assessed using administrative data and instead requires a time-consuming manual chart review.
Response: We appreciate and agree with the feedback regarding the need to include wage growth for healthcare staff in the cost estimates. As we noted in our comment response above regarding OB staff training costs, real wage costs for hospital staff are estimated to increase 0.44 percent annually so we are including a 0.44 annual increase in real wage rates starting in year 2 for all QAPI requirements. We also appreciate that full utilization of QAPI activities takes investment to be successful and that some obstetrical metrics could require manual chart reviews. We believe that the estimated costs in the proposed rule that included costs for data system modifications, data stratification, incorporation of MMRC recommendations into the QAPI program, and carrying out an annual project provide an accurate estimate of the average burden for facilities to engage in these activities.
Based on the comments received, we are making the following modification to the cost estimates: including a 0.44 percent annual increase in hourly rates for QAPI costs starting in year 2 to account for growing wages and salaries for healthcare workers.
In section XXVI of this final rule with comment period, we have already estimated the burden for collecting data and quality indicators for obstetric patients and their outcomes and disparities in processes of care and services and operations. We believe that these data will serve as the foundation to allow facilities to develop and implement actions to improve outcomes and reduce disparities when they exist. We expect that these data will likely be the focus of the required performance improvement project focused on improving health outcomes and reducing disparities among obstetrical patients.
To estimate the cost of tracking and implementing at least one quality improvement project, we utilized estimates from existing regulations governing QAPI program. Specifically, 81 FR 68688 estimates that collecting and analyzing data for all long-term care facilities' improvement projects will take 20 hours, with another 20 years annually spent on implementing and documenting improvement projects. Given that the requirement we are finalizing involves only a single improvement project and we have already accounted for the costs of collecting the data in the information collection requirement portion of this rule, we anticipate that the ongoing annual burden for each facility to analyze the data and implement and document their improvement project(s) will be 30 hours. Using loaded hourly wage rates from Table 186, we anticipate that this will include the participation of a hospital executive at $1,861.28 ($232.66 × 8 hours), an RN at $931.00 ($93.10 × 10 hours), a physician at $1,729.64 ($216.08 × 8 hours), and a data scientist at $368.96 ($92.24 × 4 hours). As indicated in Table 220, this would lead to a total per facility cost of $4,889.88 annually and an average hourly cost of $163 if the requirement went into effect in year 1. Since the requirement will not go into effect until year 2, we assume no burden in year 1. For years 2 to 10, we increase the estimated cost by 0.44 percent annually to account for growth in staff wage rates. As indicated in Tables 220 and 221, we estimate that this requirement will cost an average of $22,170,365 annually and $221,703,645 over 10 years.
( printed page 94577)d. Maternal Health QAPI Activities for Hospitals (§ 482.21(e)) and CAHs (§ 485.641(d)(4))
As indicated in section XXI.C.4 of this final rule with comment period, we are finalizing the proposed revision to existing QAPI standards (§ 482.21; § 485.641) for hospitals and CAHs that offer obstetrical services without modifications. Below, we provide the comments we received on the proposed requirement that when the MMRC provides information to hospitals and CAHs, they incorporate this information into the facility's QAPI program.
Comment: Some commenters said that the proposed rule underestimated costs since it did not consider increases in wage and salaries for healthcare personnel.
Response: We appreciate and agree with the feedback regarding the need to include wage growth for healthcare staff in the cost estimates. As we noted in our comment response above regarding OB staff training costs, real wage costs for hospital staff are estimated to increase 0.44 percent annually so we are including a 0.44 annual increase in real wage rates starting in year 2 for this requirement.
Based on the comments received, we are making the following modification to the cost estimates: including a 0.44 percent annual increase in hourly rates for QAPI requirements starting in year 2 to take into account growing wages and salaries for healthcare workers.
Using loaded hourly wage rates from Table 186, we expect that when the MMRC provides information to hospitals and CAHs, incorporating this information into the facility's QAPI program will include the participation of a physician at $864.32 ($216.08 × 4 hours) and an RN at $372.40 ($93.10 × 4 hours). As indicated in Table 222, this would lead to a total cost of $1,236.72 per facility if the requirement went into effect in year 1. Since the requirement will not go into effect until year 2, we assume no burden in year 1. For years 2 to 10, we increase the estimated cost by 0.44 percent annually to account for growth in staff wage rates. As indicated in table 223, we estimate that the average annual cost will be $5,607,200 and the 10-year total cost will be $56,071,996.
( printed page 94578)e. Emergency Services Readiness for Hospitals (§ 482.55(c)) and CAHs (§ 485.618(e))
As indicated in section XXI.C.5 of this final rule with comment period, we are finalizing the proposed emergency services readiness standard for hospitals and CAHs without modifications. Below, we provide the comments we received on the proposed requirement's cost as well as our modifications to the final cost estimate based on these comments.
Comment: Some commenters said that the proposed rule underestimated costs since it did not consider increases in wage and salaries for healthcare personnel, as well as staff turnover.
Response: We appreciate and agree with the commenters. We are including a 0.44 annual increase in real wage rates starting in year 2 for the emergency services training requirements. While the emergency training cost estimate in the proposed rule included costs for turnover in years 2 through 10, it did not include them in year 1. To address concern regarding staff turnover and training for new staff members, we are including training costs for new staff members based on a 21 percent annual staff turnover rate, which is the same rate we used for the OB staff training requirement.
Comment: Some commenters said that the proposed rule underestimated the equipment costs since it did not consider the cost for maintaining and replacing required equipment.
Response: We agree that replacing equipment will add additional burden for facilities. Based on our experience, we estimate that the required equipment will need to be replaced once every 5 years. As a result, we include additional burden in year 6 for hospitals with emergency services to replace all call-in systems.
Based on the comments received, we are making the following modifications to the cost estimates: (1) including a 0.44 percent annual increase in hourly rates for training costs starting in year 2 to take into account growing wages and salaries for healthcare workers, (2) including additional costs in year 1 to account for training new staff members, and (3) including additional costs to replace call-in systems in year 6.
In section XXVI of this final rule with comment period, we have already discussed the cost for hospitals to ensure that they have adequate protocols in place for emergency services, as well as to document that applicable staff have successfully completed the training and demonstrate their knowledge on these topics. The training requirement for hospitals and CAHs provides flexibility regarding which staff will receive training. We expect, however, that they will likely focus their training on medical staff within emergency departments. This staff includes surgeons, physicians, physician assistants, nurse practitioners, nurse midwives, nurse anesthetists, registered nurses, and LPNs/LVNs.
To estimate the number of employees in CAHs and REHs that will likely receive training, we first obtained data from the Bureau of Labor Statistics (BLS) on the number of surgeons, physicians, physician assistants, nurse practitioners, nurse midwives, nurse anesthetists, registered nurses, and LPNs/LVNs working in hospitals (NAICS 622000). Since the BLS does not provide separate employment statistics for CAHs and hospitals, we assume that the number of employees needing training and, henceforth, the costs will be in proportion to the size of facilities, specifically the number of certified beds. We obtained information on the number of certified beds in hospitals and CAHs from CMS' Q1 2024 Provider of Services File—Hospital & Non-Hospital Facilities.[789] Using this database, we estimate that 98.88 percent of certified beds for hospitals are in hospitals with the remaining 1.12 percent in CAHs.
Based on our experience, we expect that initial staff training will take approximately 3 hours per employee. Using data from Table 186 on loaded wage rates for each employee type, we estimated the cost for training all medical staff in hospitals and CAHs in year 1 using the following formula: loaded wage rate for medical staff (surgeons, physicians, physician assistants, nurse practitioners, nurse midwives, nurse anesthetists, registered nurses, and LPNs/LVNs) × total number of each medical staff type working in hospitals and CAHs × 3 hours per employee. Using this formula, we estimate that training all medical staff would cost $724,555,386.
In hospitals, which have a larger number of beds, there is likely to be a greater division of staff among units, with medical staff specifically designated to work in emergency departments. In contrast, CAHs, which are smaller in size, are likely to have medical staff that work across units given their small size. We assume, therefore, that 20 percent of medical staff in hospitals and all medical staff in CAHs will receive the training. To calculate the baseline year 1 cost for hospitals and CAHs, therefore, we use the following formula: Total cost for training all hospital/CAH medical staff × % hospital (CAH) medical staff ( printed page 94579) receiving training × Share of all Hospital and CAH Certified Beds. We then include the cost to train new employees who will need to receive 3 hours of training by multiplying our baseline cost estimates by 1.21 to account for an average hospital turnover rate of 21 percent annually.[790] As indicated in Table 224, we expect that the finalized requirement will cost approximately $9.8 million for CAHs and $173.4 million for hospitals in year 1.
For subsequent years, we expect that refresher training for medical staff, who received the full training in previous years, will take 1 hour to complete. In addition, new staff will need to receive the full 3-hour training. With an annual hospital turnover rate of approximately 21 percent,[791] we expect 21 percent of employees each year to be new employees who will need 3 hours of training and 79 percent of employees will need 1 hour of training. To calculate the burden for years 2 to 10, therefore, we use the following formula: (Total cost for training all hospital/CAH medical staff × % hospital(CAH) medical staff needing initial training × Share of all Hospital and CAH Certified Beds) + (Total cost for training all hospital/CAH medical staff × % hospital(CAH) medical staff receiving needing initial training × Share of all Hospital and CAH Certified Beds). To account for increases in real wage rates we increase the estimated cost by 0.44 percent annually. As indicated in Table 225, we estimate a total 10-year cost of $842,377,531.
To estimate the cost for hospitals to have certain basic equipment readily available, we consulted with medical experts on the requirements. Based on their experience, we expect that the most hospitals with emergency services already have drugs, blood and blood products, and biologicals commonly used in emergency procedures, as well as equipment and supplies commonly used in emergency procedures. As such, we do not estimate a burden for these requirements. There is likely, however, to be wide variation in hospitals that have call-in systems. Based on our experience, we estimate that 50 percent of hospitals already have call-in systems ( printed page 94580) while 50 percent will need to install them in their emergency departments.
As we noted above in estimating the cost for call-in systems for hospitals that offer obstetrical services, while we were not able to identify public estimates for the price of call-in systems, based on our experience we estimate that they will cost $2500 to $3500 per system, and we utilize the mid-point of the price estimate and assume each call-in system will cost $3000. We assume that 20 percent of hospital beds are allocated for emergency services and assume that there will need to be a call-in system for each bed. As indicated in Table 226, we estimate that this requirement will cost a total of $334,629,300 in year 1. We also expect that under normal use, call-in systems will need to be replaced in five years. As such, in Table 227 we estimate a total cost of $669,384,600 over 10 years.
f. Transfer Protocols in Discharge Planning for Hospitals (§ 482.43(c))
As indicated in section XXI.C.6 of this final rule with comment period, we are finalizing the proposed emergency services readiness standard for hospitals with the modification that acute care hospitals are required to provide annual training to the relevant staff (as determined by the facility) regarding the hospital policies and procedures for transferring patients under its care. Below, we provide the comments we received on the proposed requirement's cost as well as our modifications to the final cost estimate based on these comments.
Comment: Some commenters said that the proposed rule underestimated costs since it did not consider increases in wage and salaries for healthcare personnel, as well as staff turnover.
Response: We appreciate and agree with the commenters. We are including a 0.44 annual increase in real wage rates starting in year 2 for the transfer protocol training requirements. To address concern regarding staff turnover and training for new staff members, we are including training costs for new staff members based on a 21 percent annual staff turnover rate.[792]
After consideration of the comments, we are revising the transfer protocol staff training costs to include a 0.44 percent annual increase in real wage rates to account for wage growth among healthcare staff starting in year 2. We are also including additional burden for staff training to account for staff turnover and new staff members who will need transfer protocol training.
Since hospital inpatients are included in those who may need to be transferred, we believe that medical staff across hospitals, and not just those in emergency departments, will need to receive training on transfer protocols. Specifically, we expect that all surgeons, physicians, physician assistants, nurse practitioners, nurse midwives, nurse anesthetists in hospitals will receive this training. We do not expect, however, that LPNs will receive this training and similarly expect that most RNs will not receive this training. Rather, we expect that among RNs, only experienced RNs who serve as transfer coordinators will receive it and estimate that this is only 5 percent of RNs nationwide. We estimate that each employee will require 1 hour of training annually and assume that that this training will occur on an annual basis. As indicated in Table 228, after increasing the estimated burden by 21 percent to account for the average annual turnover in hospitals [793] that will require training new employees, we ( printed page 94581) expect the requirement to cost $86,207,786 in year 1. For subsequent years, we increase the estimated annual cost by 0.44 percent to account for real wage growth for staff. As indicated in Table 229, we expect that this requirement to cost an average of $87,934,883 annually and $879,348,830 over 10 years.
g. Summary of Regulatory Impact Analysis for Obstetrical and Emergency Services
In Tables 231 and 232 we provide an estimate of the total annual and 10-year financial and hourly burden for the requirements related to obstetrical and emergency services that include: (1) organization, staffing, and delivery of services for hospitals and CAHs as outlined in Table 215 and Table 217; (2) obstetrical services staff training for hospitals and CAHs as outlined in Table 219; (3) quality assessment and performance improvement program requirements for hospitals and CAHs as outlined in Table 221; (4) maternal health QAPI activity requirements for hospitals and CAHs as outlined in Table 223; (5) emergency services readiness requirements for hospitals and CAHs in Table 225 and Table 227; and (6) transfer protocols training for acute care hospitals as outlined in Table 229. These estimates exclude the cost for collection of information requirements that we have estimated above in Tables 199 and 200 to cost $129,748,120 million over 10 years and take 1,038,698 hours to complete. Overall, we estimate the total financial cost of the requirements will be approximately $4.10 billion and take $22.4 million hours to complete over 10 years.
We sought comments on several issues related to the regulatory impact analysis, including the following:
- Are there additional data sources that estimate the number of medical staff, who work with obstetrical patients?
- Are there additional data sources to estimate the number of hospital and CAH obstetrical rooms/suites?
- Are there any additional data sources to estimate the cost for the provisions of cardiac monitors, call-in systems, and fetal doppler or monitors?
- Are there additional data sources to estimate the number of medical staff who work with emergency care units?
- Are there data sources to estimate the number of hospital room/suites that are allocated for emergency services?
- Are there any additional staff members who are likely to receive training for emergency services and obstetrical services?
We did not receive responses to these comment solicitations.
( printed page 94582) ( printed page 94583)h. Benefits
We invited comments on additional benefits related to the proposed requirements for obstetrical services in hospitals and CAHs but did not receive any information about additional benefits. Since we are not finalizing the requirement that hospitals and CAHs delineate and document obstetrical privileges for all practitioners, we are removing the benefits for this policy. Below, we detail the benefits from the finalized policies.
First, there are the financial benefits. As noted above in the statement of need, research suggests that maternal mortality and morbidity have widespread negative effects on pregnant and postpartum patients and their families and high financial costs for payors. One study found that pregnancy-related mortality in the United States costs more than $27.4 billion and resulted in the loss of 114,000 years of potential life between 2018 and 2020.[794] Another study showed that from birth to 5 years postpartum, nine maternal morbidities among the 2019 US birth cohort cost birthing parents and their children $32.3 billion, with $18.7 billion due to medical costs and $13.6 billion related to non-medical costs.[795 796] A third study found that severe maternal morbidity during the prenatal to 30-day postpartum period was associated with a 75 percent increase in medical costs for patients utilizing Medicaid and a more than doubling in medical costs for commercially insured patients.[797] While these studies vary in their methodology, the pre-post birth time period analyzed, medical conditions analyzed, and cost estimates, they suggest that maternal morbidity and mortality impose high health and safety, as well as economic costs on birth parents, children, payors, and society.[798]
We believe that the policies we are finalizing will help reduce maternal morbidity and mortality and their associated costs for pregnant and postpartum patients and their families, as well as payors. Specifically, the finalized requirements that OB services are well-organized and in accordance with acceptable standards of practices, have adequate provisions and protocols for OB emergencies, complications, immediate post-delivery care and other patient health and safety events as identified as part of the facility's QAPI program, and that OB patient care units are supervised by an individual with the necessary education and training will provide the foundation for ensuring uniform high-quality OB services. The requirement that hospitals and CAHs that offer obstetrical services have equipment that includes a call-in-system, cardiac monitor, and fetal doppler or monitor kept at the hospital and CAH and readily available for treating obstetrical cases to meet the needs of patients in accordance with the scope, volume, and complexity of services offered will help ensure efficient and effective care that can help reduce patient morbidity and mortality. Similarly, OB staff training and appropriate transfer protocols can also help avert avoidable maternal complications and deaths.[799] Finally, engagement with recommendations from MMRCs and QAPI stratification of data can help facilities better identify unfavorable patient health and safety outcomes, which can allow them to ( printed page 94584) better tailor policies to address these issues.
Beyond reductions in maternal morbidity and mortality and their associated financial benefits, the finalized policies are likely also to reduce inequality among pregnant and postpartum women from different groups. For example, research shows that among women with any form of disability, there is a heightened risk for labor and delivery complications, as well as severe maternal morbidity and mortality. If hospitals and CAHs include training that helps health care practitioners better understand these risks and be more comfortable providing care to women with a disability, they may be able to better provide safe, high quality obstetric care, reducing obstetrical complications. Research also suggests that due to insufficient patient education by staff, women with limited English proficiency (LEP) experience disparities in obstetric care and are at risk for mental health conditions, including post-partum depression and substandard newborn care following neonatal ICU discharge.[800] If facilities engage in increasing language-concordant care and awareness among providers regarding the use of medical interpreters and materials in diverse languages, they may be able to improve patient satisfaction, decrease medical errors, and improve patient safety.[801 802] Similarly, stratification of patient data can produce insights into health disparities that allow facilities to develop interventions to reduce them, with research showing that data collection and analysis by patient subgroup within health care facilities has an important impact on improving patient care consistently across patient populations.[803 804 805 806 807]
Beyond the benefits for obstetrical patients, our requirements are likely to have positive effects on the health and safety for patients generally. Our requirements for hospitals to have written policies and procedures for transferring patients under their care and to train medical staff regarding transfer protocols can support hospitals in expediting transfers when necessary. Efficient transfers to hospitals that can treat complex conditions and provide higher levels of care for all patients as needed. Similarly, our requirement that hospitals with emergency services must have adequate provisions and protocols for the care of patients with emergency conditions and train applicable staff on these protocols and provisions, is also likely to improve patient health and safety. Additional obstetric training for emergency department staff improves staff competencies ( i.e., skills, knowledge, comfort, confidence, and effectiveness) in managing obstetric emergencies, supporting improved maternal health and safety,[808 809 810 811 812 813] while training in pediatric readiness,[814 815] and geriatric readiness [816 817 818 819 820] improves staff capabilities in caring for these populations.
i. Alternatives Considered
We considered a variety of approaches when developing the finalized obstetrical services requirements for hospitals and CAHs. One approach was to leave the development of policies to improve obstetrical services to accrediting agencies or individual States. We decided against this approach, however, since there is likely to be wide variation across States and accrediting agencies in their requirements, leading to variation in obstetrical services for patients depending on the facility or State where they are located.
We also considered requiring specific topics for the proposed OB services training requirement as well as for the requirement to train staff on the protocols for the care of patients with emergency conditions. We ultimately decided, however, to provide facilities with flexibility in how they approach these trainings so that they could provide it in a way that leads to the best improvements in and highest quality of care for pregnant and postpartum women. Similarly, we considered defining specific subpopulations that facilities must analyze when using their QAPI program to identify inequalities in health outcomes. Ultimately, however, we decided to provide facilities with flexibility regarding which subpopulations they analyze since features of patient populations are likely to vary greatly across different facilities.
D. Regulatory Review Cost Estimation
If regulations impose administrative costs on private entities, such as the time needed to read and interpret this final rule, we should estimate the cost associated with regulatory review. Due to the uncertainty involved with accurately quantifying the number of entities that will review the rule, we assume that the total number of unique commenters on this year's proposed rule will be the number of reviewers of this final rule. We acknowledge that this assumption may understate or overstate ( printed page 94585) the costs of reviewing this rule. It is possible that not all commenters reviewed this year's rule in detail, and it is also possible that some reviewers chose not to comment on the proposed rule. For these reasons we thought that the number of past commenters would be a fair estimate of the number of reviewers of this rule.
We welcomed any public comments on the approach in estimating the number of entities that would review the proposed rule. We did not receive any public comments specific to our solicitation.
Using the wage information from the BLS for medical and health service managers (Code 11-9111), we estimate that the cost of reviewing this rule is $129.28 per hour, including overhead and fringe benefits https://www.bls.gov/oes/current/oes_nat.htm. Assuming an average reading speed, we estimate that it would take approximately 8 hours for the staff to review half of this final rule. For each entity that reviews the rule, the estimated cost is $1,034.24 (8 hours × $129.28). Therefore, we estimate that the total cost of reviewing this regulation is $2,645,585.92 ($1,034.24 × 2,558).
E. Regulatory Flexibility Act (RFA) Analysis
The RFA requires agencies to analyze options for regulatory relief of small entities, if a rule has a significant impact on a substantial number of small entities. For purposes of the RFA, we estimate that many hospitals and CAHs are considered small businesses either by the Small Business Administration's size standards with total revenues of $41.5 million or less in any single year or by the hospital's not-for-profit status. Most ASCs and most CMHCs are considered small businesses with total revenues of $16.5 million or less in any single year. For details, we refer readers to the Small Business Administration's “Table of Size Standards” at http://www.sba.gov/content/small-business-size-standards.
Individuals and States are not included in the definition of a small entity. As its measure of significant economic impact on a substantial number of small entities, HHS uses a change in revenue of more than 3 to 5 percent. We believe that this threshold will be reached by the requirements in this final rule with comment period. Therefore, the Secretary has certified that this final rule with comment period will have a significant economic impact on a substantial number of small entities.
In addition, section 1102(b) of the Act requires us to prepare a regulatory impact analysis if a rule may have a significant impact on the operations of a substantial number of small rural hospitals. This analysis must conform to the provisions of section 604 of the RFA. For purposes of section 1102(b) of the Act, we define a small rural hospital as a hospital that is located outside of a metropolitan statistical area and has 100 or fewer beds. We estimate that this final rule with comment period will increase payments to small rural hospitals by approximately 3 percent; therefore, it should have a negligible impact on approximately 528 small rural hospitals. We note that the estimated payment impact for any category of small entity will depend on both the services that they provide as well as the payment policies and/or payment systems that may apply to them. Therefore, the most applicable estimated impact may be based on the specialty, provider type, or payment system.
For hospitals and CAHs, the finalized obstetrical services requirements will have an average annual cost of $423 million. According to the 2017 Economic Census,[821] revenues for general medical and surgical hospitals (NAIC 522110) exceeded $1 trillion in 2017, which is less than 0.50 percent of annual revenues.
The analysis above, together with the remainder of this preamble, provides a regulatory flexibility analysis and a regulatory impact analysis.
F. Unfunded Mandates Reform Act Analysis
Section 202 of the Unfunded Mandates Reform Act of 1995 (UMRA) also requires that agencies assess anticipated costs and benefits before issuing any rule whose mandates require spending in any 1 year of $100 million in 1995 dollars, updated annually for inflation. In 2024, that threshold is approximately $183 million. This final rule with comment period does not mandate any requirements for State, local, or tribal governments. This final rule with comment period will not impose a mandate that will result in the expenditure by State, local, and Tribal Governments, in the aggregate. Based on the cost estimates discussed in this section, we have assessed the various costs and benefits of the final updates to the requirements for participation for hospitals and CAHs. For the private sector facilities, the regulatory impact analysis, together with the remainder of the preamble, constitutes the analysis required under UMRA.
G. Federalism
Executive Order 13132 establishes certain requirements that an agency must meet when it promulgates a proposed rule (and subsequent final rule) that imposes substantial direct requirement costs on State and local governments, preempts state law, or otherwise has federalism implications. We have examined the OPPS and ASC provisions included in this final rule with comment period in accordance with Executive Order 13132, Federalism, and have determined that they would not have a substantial direct effect on State, local, or tribal governments, preempt State law, or otherwise have a federalism implication. As reflected in Table 201 of this final rule with comment period, we estimate that OPPS payments to governmental hospitals (including State and local governmental hospitals) would increase by 2.6 percent under this final rule with comment period. While we do not know the number of ASCs or CMHCs with government ownership, we anticipate that it is small. The analyses we have provided in this section of this final rule with comment period, in conjunction with the remainder of this document, demonstrate that this rule is consistent with the regulatory philosophy and principles identified in Executive Order 12866, the RFA, and section 1102(b) of the Act.
This final rule with comment period will affect payments to a substantial number of small rural hospitals and a small number of rural ASCs, as well as other classes of hospitals, CMHCs, and ASCs, and some effects may be significant. However, as noted in section XXIII of this final rule with comment period, this rule should not have a significant effect on small rural hospitals.
The obstetrical services provisions in this final rule with comment period will not have a substantial direct effect on State, local, or tribal governments, preempt State law, or otherwise have a federalism implication.
H. Conclusion
The changes we are finalizing will affect all classes of hospitals paid under the OPPS as well as both CMHCs and ASCs. We estimate that most classes of hospitals paid under the OPPS would experience a modest increase or a minimal decrease in payment for services paid under the OPPS in CY ( printed page 94586) 2025. Table 201 demonstrates the estimated distributional impact of the OPPS budget neutrality requirements that will result in a 3.0 percent increase in payments for all services paid under the OPPS in CY 2025, after considering all of the changes to APC reconfiguration and recalibration, as well as the OPD fee schedule increase factor, wage index changes, including the frontier State wage index adjustment, and estimated payment for outliers, changes to the pass-through payment estimate, and changes to outlier payments. However, some classes of providers that are paid under the OPPS would experience more significant gains or losses in OPPS payments in CY 2025.
The updates we are making to the ASC payment system for CY 2025 will affect each of the approximately 6,100 ASCs currently approved for participation in the Medicare program. The effect on an individual ASC would depend on its mix of patients, the proportion of the ASC's patients who are Medicare beneficiaries, the degree to which the payments for the procedures offered by the ASC are changed under the ASC payment system, and the extent to which the ASC provides a different set of procedures in the coming year than in previous years. Table 201 demonstrates the estimated distributional impact among ASC surgical specialties of the productivity-adjusted hospital market basket update factor of 2.9 percent for CY 2025.
The new conditions of participation and the modifications to current conditions of participation for hospitals and CAHs that offer obstetrical services and emergency services will have a total 10-year impact of approximately $4.23 billion as outlined in Tables 200 and 231. The average annual cost per hospital and CAH will be approximately $67,000.
Finally, the additional exceptions to the four walls requirement under the Medicaid clinic services benefit for IHS/Tribal clinics, and at State option, behavioral health clinics and clinics located in rural areas are estimated to have an $1.18 billion impact in transfers for fiscal years 2025-2029. Table 211 demonstrates the Federal and State share impacts on IHS/Tribal clinics, behavioral health clinics, clinics located in rural areas, and in aggregate. As explained earlier in this section of this final rule, there is uncertainty in the potential for changes in utilization and costs of clinic services because of uncertainty in provider availability and beneficiary demand.
I. Waiver Fiscal Responsibility Act Requirements
Please note that the Director of OMB has waived the requirements of section 263 of the Fiscal Responsibility Act of 2023 (Pub. L. 118-5) pursuant to section 265(a)(1) and (2) of that Act.
Chiquita Brooks-LaSure, Administrator of the Centers for Medicare & Medicaid Services, approved this document on October 23, 2024.
List of Subjects
42 CFR Part 406
- Diseases
- Health facilities
- Medicare
42 CFR Part 407
- Medicare
42 CFR Part 410
- Diseases
- Health facilities
- Health professions
- Laboratories
- Medicare
- Reporting and recordkeeping requirements
- Rural areas
- X-rays
42 CFR Part 411
- Diseases
- Medicare
- Reporting and recordkeeping requirements
42 CFR Part 416
- Health facilities
- Health professions
- Medicare
- Reporting and recordkeeping requirements
42 CFR Part 419
- Hospitals
- Medicare
- Reporting and recordkeeping requirements
42 CFR Part 435
- Aid to families with dependent children
- Grant programs—health
- Medicaid
- Reporting and recordkeeping requirements
- Supplemental Security Income (SSI)
- Wages
42 CFR Part 440
- Grant programs—health
- Medicaid
42 CFR Part 457
- Administrative practice and procedure
- Grant programs—health
- Health insurance
- Reporting and recordkeeping requirements
42 CFR Part 482
- Grant programs—health
- Hospitals
- Medicaid
- Medicare
- Reporting and recordkeeping requirements
42 CFR Part 485
- Grant programs—health
- Health facilities
- Medicaid
- Privacy
- Reporting and recordkeeping requirements
For the reasons set forth in the preamble, the Centers for Medicare and Medicaid Services amends 42 CFR chapter IV as set forth below:
PART 406—HOSPITAL INSURANCE ELIGIBILITY AND ENTITLEMENT
1. The authority citation for part 406 continues to read as follows:
2. Section 406.27 is amended by revising paragraph (d) to read as follows:
(d) SEP for formerly incarcerated individuals. An SEP exists for Medicare eligible individuals who are no longer incarcerated on or after January 1, 2023.
(1) SEP parameters and duration before January 1, 2025 —(i) Eligibility. An individual is eligible for this SEP if they are released from the custody of penal authorities between January 1, 2023, and December 31, 2024, as described in § 411.4(b) of this subchapter. The individual must demonstrate that they are eligible for Medicare and failed to enroll or reenroll in Medicare premium Part A due to being in custody of penal authorities and there is a record of release either through discharge documents or data available to SSA.
(ii) SEP duration. The SEP starts the day of the individual's release from the custody of penal authorities and ends the last day of the 12th month after the month in which the individual is released from the custody of penal authorities.
(2) SEP parameters and duration beginning January 1, 2025 —(i) Eligibility. An individual is eligible for this SEP if they are released from confinement in a jail, prison, or other penal institution or correctional facility on or after January 1, 2025, and demonstrate that they are eligible for Medicare and failed to enroll or reenroll in Medicare premium Part A due to being so confined, and there is a record of release, either through documentation of discharge or data available to SSA. Individuals released to and residing in halfway houses after incarceration are not considered incarcerated or in confinement for the purposes of this SEP.
(ii) SEP duration. The SEP starts the day an individual is released from confinement as determined by SSA and ends the last day of the 12th month after the month in which the individual is released from confinement in a jail, prison, or other penal institution or correctional facility.
(3) Entitlement —(i) General rule. Entitlement begins the first day of the ( printed page 94587) month following the month of enrollment, so long as the date is on or after January 1, 2023.
(ii) Special rule. An individual has the option of requesting entitlement for a retroactive period of up to 6 months provided the date does not precede the month of their release from incarceration, the date is on or after January 1, 2023, and the individual pays the monthly premiums for the period of coverage (as required under § 406.32(f)). If retroactive enrollment is requested and the application is filed within the first 6 months of the SEP, the effective date is retroactive to the beginning of the month of their release from incarceration. If retroactive enrollment is requested and the application is filed in the last 6 months of the SEP, the coverage effective date is retroactive to the 6th month before the month of enrollment.
PART 407—SUPPLEMENTARY MEDICAL INSURANCE (SMI) ENROLLMENT AND ENTITLEMENT
3. The authority citation for part 407 continues to read as follows:
4. Section 407.23 is amended by revising paragraph (d) to read as follows:
(d) SEP for formerly incarcerated individuals. An SEP exists for Medicare eligible individuals who are no longer incarcerated on or after January 1, 2023.
(1) SEP parameters and duration before January 1, 2025 —(i) Eligibility. An individual is eligible for this SEP if they are released from the custody of penal authorities between January 1, 2023, and December 31, 2024, as described in § 411.4(b) of this subchapter. The individual must demonstrate that they are eligible for Medicare and failed to enroll or reenroll in SMI due to being in the custody of penal authorities and there is a record of release either through discharge documents or data available to SSA.
(ii) SEP duration. The SEP starts the day of the individual's release from the custody of penal authorities and ends the last day of the 12th month after the month in which the individual is released from the custody of penal authorities.
(2) SEP parameters and duration beginning January 1, 2025 —(i) Eligibility. An individual is eligible for this SEP if they are released from confinement in a jail, prison, or other penal institution or correctional facility on or after January 1, 2025, and demonstrate that they are eligible for Medicare and failed to enroll or reenroll in SMI due to being so confined, and there is a record of release, either through documentation of discharge or data available to SSA. Individuals released to and residing in halfway houses after incarceration are not considered incarcerated or in confinement for the purposes of this SEP.
(ii) SEP duration. The SEP starts the day an individual is released from confinement as determined by SSA and ends the last day of the 12th month after the month in which the individual is released from confinement in a jail, prison, or other penal institution or correctional facility.
(3) Entitlement —(i) General rule. Entitlement begins the first day of the month following the month of enrollment, so long as the date is on after January 1, 2023.
(ii) Special rule. An individual has the option of requesting entitlement for a retroactive period of up to 6 months provided the date does not precede the month of their release from incarceration, the date is on or after January 1, 2023, and the individual pays the monthly premiums for the period of coverage (as required under § 408.4 of this subchapter). If retroactive enrollment is requested and the application is filed within the first 6 months of the SEP, the effective date is retroactive to the beginning of the month of their release from incarceration. If retroactive enrollment is requested and the application is filed in the last 6 months of the SEP, the coverage effective date is retroactive to the 6th month before the month of enrollment.
PART 410—SUPPLEMENTARY MEDICAL INSURANCE (SMI) BENEFITS
5. The authority citation for part 410 continues to read as follows:
6. Section 410.27 is amended by revising paragraph (a)(1)(iv)(B)( 1) to read as follows:
(a) * * *
(1) * * *
(iv) * * *
(B) * * *
( 1) For purposes of this section, direct supervision means that the physician or nonphysician practitioner must be immediately available to furnish assistance and direction throughout the performance of the procedure. It does not mean that the physician or nonphysician practitioner must be present in the room when the procedure is performed. For pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services, direct supervision must be furnished as specified in §§ 410.47 and 410.49, respectively. Through December 31, 2025, the presence of the physician or nonphysician practitioner for the purpose of the supervision of pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services includes virtual presence through audio/video real-time communications technology (excluding audio-only); and
7. Section 410.28 is amended by revising paragraph (e)(2)(iii) to read as follows:
(e) * * *
(2) * * *
(iii) Through December 31, 2025, the presence of the physician or nonphysician practitioner under paragraphs (e)(2)(i) and (ii) of this section includes virtual presence through audio/video real-time communications technology (excluding audio-only).
PART 411—EXCLUSIONS FROM MEDICARE AND LIMITATIONS ON MEDICARE PAYMENT
8. The authority citation for part 411 continues to read as follows:
9. Section 411.4 is revised to read as follows:
(a) General rule. Except in the case of federally qualified health center services and as provided in § 411.8(b) (for services paid by a governmental entity), Medicare may not pay for an item or service under Part A or Part B if—
(1) The individual has no legal obligation to pay for the item or service; and ( printed page 94588)
(2) No other person (by reason of such individual's membership in a prepayment plan or otherwise) has a legal obligation to provide or pay for the item or service.
(b) Special conditions for payment for items or services furnished to an individual in the custody of a penal authority. (1) An individual in the custody of a penal authority is considered to have a legal obligation to pay for items or services furnished to the individual only if the following conditions are met:
(i) State or local law requires the individual to pay the cost of items and services that the individual receives;
(ii) The penal authority enforces the requirement to pay for items or services by billing all individuals who receive such items or services, whether or not covered by Medicare or any other health insurance; and
(iii) The penal authority pursues collection of amounts owed for items or services received in the same way and with the same vigor that it pursues the collection of other debts.
(2) For purposes of this paragraph (b), a penal authority means a police department or other law enforcement agency, a government agency operating under a penal statute, or a State, local or Federal jail, prison, penitentiary, or similar institution.
(3) For purposes of this paragraph (b)—
(i) An individual is considered to be in the custody of a penal authority if the individual is:
(A) Incarcerated in a jail, prison, penitentiary, or similar institution;
(B) Temporarily outside of a jail, prison, penitentiary, or similar institution on medical furlough or similar arrangement;
(C) Escaped from confinement by a penal authority; or
(D) Required to reside in a mental health facility under a penal statute or rule.
(ii) Individuals who are not considered to be in the custody of a penal authority include, but are not limited to, individuals who are—
(A) Released to the community pending trial (including those in pretrial community supervision and those released pursuant to cash bail);
(B) On parole;
(C) On probation;
(D) On home detention or home confinement; or
(E) Required to live in a halfway house or other community-based transitional facility.
PART 416—AMBULATORY SURGICAL SERVICES
10. The authority citation for part 416 continues to read as follows:
11. Section 416.164 is amended by revising paragraphs (a)(4) and (b)(6) to read as follows:
(a) * * *
(4) Drugs and biologicals for which separate payment is not allowed under the hospital outpatient prospective payment system (OPPS);
(b) * * *
(6) Non-opioid pain management drugs, biologicals, and medical devices as determined by CMS under § 416.174.
12. Section 416.171 is amended by revising paragraph (b)(1) to read as follows:
(b) * * *
(1) Covered ancillary services specified in § 416.164(b), with the exception of radiology services and certain diagnostic tests as provided in § 416.164(b)(5) and non-opioid pain management drugs, biologicals, and medical devices as determined by CMS under § 416.174.
13. Section 416.174 is revised to read as follows:
(a) Eligibility for separate payment for non-opioid pain management drugs and biologicals. From January 1, 2025, through December 31, 2027, a non-opioid drug or biological is eligible for separate payment for an applicable calendar year if CMS determines it meets the following requirements through that year's rulemaking:
(1) The drug is approved under a new drug application under section 505(c) of the Federal Food, Drug, and Cosmetic Act (FDCA), under an abbreviated new drug application under section 505(j) of the FDCA, or, in the case of a biological product, is licensed under section 351 of the Public Health Service Act. The product also has a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors.
(2) The drug or biological does not have transitional pass-through payment status under § 419.64 of this subchapter. In the case where a drug or biological otherwise meets the requirements under this section and has transitional pass-through payment status that expires during the calendar year, the drug or biological will qualify for separate payment as specified in this paragraph (a) during such calendar year on the first day of the next quarter following the expiration of its pass-through status.
(3) The drug or biological has payment that is packaged into a payment for a covered outpatient department (OPD) service (or group of services) under a policy in this part.
(b) Eligibility for separate payment for non-opioid medical devices. From January 1, 2025, through December 31, 2027, a medical device is eligible for separate payment for an applicable calendar year if CMS determines it meets all of the following requirements through that year's rulemaking:
(1) The medical device is used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia, and has an application under section 515 of the FDCA that has been approved with respect to the device, has been cleared for market under section 510(k) of the FDCA, or is exempt from the requirements of section 510(k) of the FDCA pursuant to section 510(l) or (m) or 520(g) of the FDCA.
(2) The medical device has demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal.
(3) The medical device does not have transitional pass-through payment status under § 419.66 of this subchapter. In the case where a medical device otherwise meets the requirements under this section and has transitional pass-through payment status that expires during the calendar year, the medical device will qualify for separate payment as specified in this paragraph (b) during such calendar year on the first day of the next calendar year quarter following the expiration of its pass-through status.
(4) The medical device has payment that is packaged into a payment for a covered OPD service (or group of services) under a policy in this part.
(c) Payment amount. From January 1, 2025, through December 31, 2027, the amount of payment for a qualifying non-opioid treatment for pain relief is as follows:
(1) For a qualifying drug or biological as defined in paragraph (a) of this ( printed page 94589) section, the amount of payment is the amount determined under section 1847A of the Act for the drug or biological that exceeds the portion of the otherwise applicable Medicare OPD fee schedule amount, which is determined to be zero dollars for calendar year 2025, subject to paragraph (c)(3) of this section.
(2) For a qualifying medical device as defined in paragraph (b) of this section, the amount of payment is the amount of the hospital's charges for the device, adjusted to cost, that exceeds the portion of the otherwise applicable Medicare OPD fee schedule amount, which is determined to be zero dollars for calendar year 2025, subject to paragraph (c)(3) of this section.
(3) The payment amounts in paragraphs (c)(1) and (2) of this section shall not exceed the estimated average of 18 percent of the OPD fee schedule amount of the volume weighted average of the most frequent five OPD primary procedures into which a non-opioid treatment for pain relief would be packaged.
14. Section 416.320 is amended by revising paragraph (b) to read as follows:
(b) Immediate measure suspension. If CMS determines that the collection and reporting activities related to a measure potentially raise patient safety concerns, CMS will immediately suspend the measure from the ASCQR Program and promptly notify ASCs and the public of the suspension. CMS will propose to retain, modify, or remove the suspended measure in the next feasible rulemaking cycle.
PART 419—PROSPECTIVE PAYMENT SYSTEM FOR HOSPITAL OUTPATIENT DEPARTMENT SERVICES
15. The authority citation for part 419 continues to read as follows:
16. Section 419.2 is amended by revising paragraph (b)(15) to read as follows:
(b) * * *
(15) Drugs, biologicals, and radiopharmaceuticals that function as supplies when used in a diagnostic test or procedure (including but not limited to, diagnostic radiopharmaceuticals with per-day costs at or below the per-day diagnostic radiopharmaceutical packaging threshold for the applicable year, contrast agents, and pharmacologic stress agents);
17. Section 419.41 is amended by adding paragraphs (h) through (j) to read as follows:
(h) For a therapeutic radiopharmaceutical for which payment is not packaged into a payment for a covered outpatient department (OPD) service (or group of services) and that does have on transitional pass-through payment status as described in § 419.64, to calculate the program payment and copayment amounts CMS does the following:
(1) Determines the average sales price (ASP) for the therapeutic radiopharmaceutical for the quarter established under the methodology described by section 1847A of the Act. If that amount is not available, then CMS calculates the mean unit cost (MUC) using the most recently available claims data for that therapeutic radiopharmaceutical.
(2) Subtracts from the amount determined under paragraph (h)(1) of this section the amount of the applicable Part B deductible provided under § 410.160 of this chapter.
(3) Multiplies the amount determined under paragraph (h)(1) of this section (less any applicable deductible under paragraph (h)(2) of this section) by 20 percent. This is the beneficiary's copayment amount for the drug or biological.
(4) Subtracts the amount determined under paragraph (h)(3) of this section from the amount determined under paragraph (h)(1) of this section (less any applicable deductible determined under paragraph (h)(2) of this section). This amount is the preliminary program amount.
(5) Adds to the preliminary program amount determined under paragraph (h)(4) of this section the amount by which the copayment amount would have exceeded the inpatient hospital deductible for that year. This amount is the final Medicare program payment amount.
(i) For a diagnostic radiopharmaceutical for which payment is not packaged into a payment for a covered OPD service (or group of services) and that does not have transitional pass-through payment status as described in § 419.64, to calculate the program payment and copayment amounts CMS does the following:
(1) Calculates the MUC using the most recently available claims data for that diagnostic radiopharmaceutical, if claims data are available. If claims data are not available, determines the ASP for the diagnostic radiopharmaceutical for the quarter established under the methodology described by section 1847A of the Act. If ASP data are not available, then based on the wholesale acquisition cost (WAC), under the methodology described by section 1847A of the Act. If WAC data are not available, then based on 95 percent of the average wholesale price.
(2) Subtracts from the amount determined under paragraph (i)(1) of this section the amount of the applicable Part B deductible provided under § 410.160 of this chapter.
(3) Multiplies the amount determined under paragraph (i)(1) of this section (less any applicable deductible under paragraph (i)(2) of this section) by 20 percent. This is the beneficiary's copayment amount for the drug or biological.
(4) Subtracts the amount determined under paragraph (i)(3) of this section from the amount determined under paragraph (i)(1) of this section (less any applicable deductible determined under paragraph (i)(2) of this section). This amount is the preliminary program amount.
(5) Adds to the preliminary program amount determined under paragraph (i)(4) of this section the amount by which the copayment amount would have exceeded the inpatient hospital deductible for that year. This amount is the final Medicare program payment amount.
(j) For a drug covered as an additional preventive service (DCAPS), as defined at § 410.64 of this chapter, the program payment amount is 100 percent of the payment amount determined as follows:
(1) If ASP data is available for the drug, consistent with part 414, subpart J, of this chapter, then the payment amount is determined using the methodology set forth in section 1847A of the Act and according to the provisions in part 414, subpart K, of this chapter.
(2) If ASP data is not available, then the payment amount is determined according to the most recently published National Average Drug Acquisition Cost (NADAC) prices for the drug and is the lesser of the median NADAC price of all generic forms of the drug or the lowest NADAC price brand name product.
(3) If ASP data and NADAC prices are not available, then the payment amount ( printed page 94590) is determined according to the most recently published pharmaceutical pricing data for the drug as included in the Federal Supply Schedule (FSS), as managed by the Department of Veterans Affairs per 48 CFR part 38, and is the lesser of the median FSS price of all generic forms of the drug or the lowest FSS price brand name product.
(4) If ASP data, NADAC prices, and FSS pharmaceutical prices are not available, then, for the period beginning January 1, 2025, through December 31, 2025, the payment amount is WAC plus 6 percent, or 3 percent if in an initial sales period consistent with 1847A(c)(4) of the Act, and beginning January 1, 2026, the payment amount is the invoice price determined by the Medicare Administrative Contractor (MAC).
18. Section 419.43 is amended by adding paragraph (k) to read as follows:
(k) Payment for non-opioid pain management drugs and biologicals —(1) Eligibility for separate payment for non-opioid pain management drugs and biologicals. From January 1, 2025, through December 31, 2027, a drug or biological is eligible for separate payment for an applicable calendar year if CMS determines it meets the following requirements through that year's rulemaking:
(i) The drug is approved under a new drug application under section 505(c) of the Federal Food, Drug, and Cosmetic Act (FDCA), under an abbreviated new drug application under section 505(j) of the FDCA, or, in the case of a biological product, is licensed under section 351 of the Public Health Service Act. The product has a label indication approved by the Food and Drug Administration to reduce postoperative pain, or produce postsurgical or regional analgesia, without acting upon the body's opioid receptors.
(ii) The drug or biological does not have transitional pass-through payment status under § 419.64. In the case where a drug or biological otherwise meets the requirements under this section and has transitional pass-through payment status that expires during the calendar year, the drug or biological will qualify for separate payment as specified in this paragraph (k) during such calendar year on the first day of the next calendar year quarter following the expiration of its pass-through status.
(iii) The drug or biological has payment that is packaged into a payment for a covered OPD service (or group of services) under a policy in this section.
(2) Eligibility for separate payment for non-opioid medical devices. From January 1, 2025, through December 31, 2027, a medical device is eligible for separate payment for an applicable calendar year if CMS determines it meets the following requirements through that year's rulemaking:
(i) The medical device, is used to deliver a therapy to reduce postoperative pain, or produce postsurgical or regional analgesia, and has an application under section 515 of the FDCA that has been approved with respect to the device, been cleared for market under section 510(k) of the FDCA, or is exempt from the requirements of 510(k) of the FDCA pursuant to section 510(l) or (m) or 520(g) of the FDCA.
(ii) The medical device has demonstrated the ability to replace, reduce, or avoid intraoperative or postoperative opioid use or the quantity of opioids prescribed in a clinical trial or through data published in a peer-reviewed journal.
(iii) The medical device does not have transitional pass-through payment status under § 419.66. In the case where a medical device otherwise meets the requirements under this section and has transitional pass-through payment status that expires during the calendar year, the medical device will qualify for separate payment as specified in this paragraph (k)(2) during such calendar year on the first day of the next calendar year quarter following the expiration of its pass-through status.
(iv) The medical device has payment that is packaged into a payment for a covered OPD service (or group of services) under a policy in this section.
(3) Payment amount. From January 1, 2025, through December 31, 2027, the amount of payment for a qualifying non-opioid treatment for pain relief is as follows:
(i) For a qualifying drug or biological as defined in paragraph (k)(1) of this section, the amount of payment is the amount determined under section 1847A for the drug or biological that exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the drug or biological, subject to paragraph (k)(3)(iii) of this section.
(ii) For a qualifying medical device as defined in paragraph (k)(2) of this section, the amount of payment is the amount of the hospital's charges for the device, adjusted to cost, that exceeds the portion of the otherwise applicable Medicare OPD fee schedule that the Secretary determines is associated with the device, subject to paragraph (k)(3)(iii) of this section.
(iii) The payment amounts in paragraph (k)(3)(i) and (ii) of this section shall not exceed the estimated average of 18 percent of the OPD fee schedule amount of the volume weighted average of the most frequent five OPD primary procedures into which a non-opioid treatment for pain relief would be packaged.
19. Section 419.46 is amended by revising paragraph (i)(2) and adding paragraph (j) to read as follows:
(i) * * *
(2) Immediate measure suspension. If CMS determines that the collection and reporting activities related to a measure potentially raise patient safety concerns, CMS will immediately suspend the measure from the Hospital OQR Program and promptly notify hospitals and the public of the suspension. CMS will propose to retain, modify, or remove the suspended measure in the next feasible rulemaking cycle.
(j) Requirements for submission of electronic clinical quality measures (eCQMs) under the Hospital OQR Program. (1) Hospitals must utilize certified technology updated to be consistent with the Office of the National Coordinator for Health Information Technology's health information technology (IT) certification criteria, as adopted and updated in 45 CFR 170.315.
(2) Hospitals must use electronic health record technology certified to all eCQMs that are available to report under the Hospital OQR Program.
(3) Hospitals must use the most recent version of the eCQM electronic measure specifications for the applicable reporting period available on the Electronic Clinical Quality Improvement Resource Center website at https://ecqi.healthit.gov/, or another website as designated by CMS.
20. Section 419.47 is amended by revising the section heading and paragraph (a) introductory text to read as follows:
(a) Creation of a new HCPCS code for Category B IDE studies that have a treatment arm and a placebo control arm. CMS will create a new HCPCS code, or revise an existing HCPCS code, ( printed page 94591) to describe a Category B IDE study, which will include both the treatment and placebo control arms, related device(s) of the study, as well as routine care items and services, as specified under § 405.201 of this chapter, when CMS determines that:
21. Section 419.82 is amended by revising paragraph (d)(1)(iii) to read as follows:
(d) * * *
(1) * * *
(iii) The provisional affirmation or non-affirmation will be issued within 7-calendar days of receipt of the prior authorization request.
PART 435—ELIGIBILITY IN THE STATES, DISTRICT OF COLUMBIA, THE NORTHERN MARIANA ISLANDS, AND AMERICAN SAMOA
22. The authority citation for part 435 continues to read as follows:
23. Section 435.926 is amended by revising paragraphs (b) introductory text, (b)(1), (c)(1), and (d)(1) to read as follows:
(b) Eligibility. The agency must provide continuous eligibility for the period specified in paragraph (c) of this section for an individual who is:
(1) Under age 19; and
(c) * * *
(1) The length of the continuous eligibility period is 12 months.
(d) * * *
(1) The child attains age 19;
PART 440—SERVICES: GENERAL PROVISIONS
24. The authority citation for part 440 continues to read as follows:
25. Section 440.90 is revised to read as follows:
Clinic services means preventive, diagnostic, therapeutic, rehabilitative, or palliative services that are furnished by a facility that is not part of a hospital but is organized and operated to provide medical care to outpatients. The term includes the following services furnished to outpatients (services in paragraphs (a) through (c) of this section are a mandatory part of clinic services, while services in paragraphs (d) and (e) of this section are optional):
(a) Services furnished at the clinic by or under the direction of a physician or dentist.
(b) Services furnished outside the clinic, by clinic personnel under the direction of a physician, to an individual who does not reside in a permanent dwelling or does not have a fixed home or mailing address.
(c) Services furnished outside a clinic that is a facility of the Indian Health Service, whether operated by the Indian Health Service or by a Tribe or Tribal organization (as authorized by the Indian Self-Determination and Education Assistance Act (ISDEAA), Pub. L. 93-638), by clinic personnel under the direction of a physician.
(d) Services furnished outside of a clinic that is primarily organized for the care and treatment of outpatients with behavioral health disorders, including mental health and substance use disorders, by clinic personnel under the direction of a physician.
(e) Services furnished outside of a clinic that is located in a rural area and is not a rural health clinic (as referenced in section 1905(a)(2)(B) of the Social Security Act and § 440.20(b)) by clinic personnel under the direction of a physician. States must include a definition of rural area in their State plans. This definition must be either a definition adopted and used by a Federal governmental agency for programmatic purposes, or a definition adopted by a State governmental agency with a role in setting State rural health policy.
PART 457—ALLOTMENTS AND GRANTS TO STATES
26. The authority citation for part 457 continues to read as follows:
27. Section 457.342 is revised to read as follows:
A State must provide continuous eligibility for children under a separate CHIP in accordance with the terms of § 435.926 of this chapter, and subject to a child remaining ineligible for Medicaid, as required by section 2110(b)(1) of the Act and § 457.310 (related to the definition and standards for being a targeted low-income child) and the requirements of section 2102(b)(3) of the Act and § 457.350 (related to eligibility screening and enrollment).
PART 482—CONDITIONS OF PARTICIPATION FOR HOSPITALS
28. The authority citation for part 482 continues to read as follows:
29. Section 482.21 is amended by—
a. Adding paragraph (b)(4);
b. Redesignating paragraphs (e) and (f) as paragraphs (f) and (g), respectively; and
c. Adding new paragraph (e).
The additions read as follows:
(b) * * *
(4) Effective January 1, 2027, for hospitals that offer obstetrical services, the hospital must utilize its quality assessment and performance improvement (QAPI) program to assess and improve health outcomes and disparities among obstetrical patients on an ongoing basis. At a minimum, the hospital must:
(i) Analyze data and quality indicators collected for the QAPI program by diverse subpopulations as identified by the hospital among obstetrical patients.
(ii) Measure, analyze, and track data, measures, and quality indicators on patient outcomes and disparities in processes of care, services and operations among obstetrical patients.
(iii) Analyze and prioritize patient health outcomes and disparities, develop and implement actions to improve patient health outcomes and disparities, measure results, and track performance to ensure improvements are sustained among obstetrical patients.
(iv) Conduct at least one measurable performance improvement project focused on improving health outcomes and disparities among the hospital's population(s) of obstetrical patients annually.
(e) Standard: Maternal health QAPI activities. Effective January 1, 2027, for hospitals that offer obstetrical services, the following additional QAPI requirements apply:
(1) Obstetrical services leadership must engage in QAPI as specified in this section for obstetrical services, including but not limited to participating in data collection and monitoring as specified in paragraph (b) of this section. ( printed page 94592)
(2) If a maternal mortality review committee (MMRC) is available at the State, Tribal, or local jurisdiction in which the hospital is located, the facility leadership, obstetrical services leadership, or their designate(s) must further have a process for incorporating publicly available MMRC(s) data and recommendations into the hospital QAPI program as specified in paragraph (b) of this section.
30. Section 482.43 is amended by—
a. Redesignating paragraph (c) as paragraph (d); and
b. Adding new paragraph (c).
The addition reads as follows:
(c) Standard: Transfer protocols. Effective July 1, 2025, the hospital must have written policies and procedures for transferring patients under its care (inclusive of inpatient services) to the appropriate level of care (including to another hospital) as needed to meet the needs of the patient. The hospital must also provide annual training to relevant staff regarding the hospital policies and procedures for transferring patients under its care.
31. Section 482.55 is amended by adding paragraph (c) to read as follows:
(c) Standard: Emergency services readiness. Effective July 1, 2025, in accordance with the complexity and scope of services offered, there must be adequate provisions and protocols to meet the emergency needs of patients.
(1) Protocols. Protocols must be consistent with nationally recognized and evidence-based guidelines for the care of patients with emergency conditions, including but not limited to patients with obstetrical emergencies, complications, and immediate post-delivery care.
(2) Provisions. Provisions include equipment, supplies, and medication used in treating emergency cases. Such provisions must be kept at the hospital and be readily available for treating emergency cases to meet the needs of patients. The available provisions must include the following:
(i) Drugs, blood and blood products, and biologicals commonly used in life-saving procedures;
(ii) Equipment and supplies commonly used in life-saving procedures; and
(iii) Each emergency services treatment area must have a call-in-system for each patient.
(3) Staff training. Applicable staff, as identified by the hospital, must be trained annually on the protocols and provisions implemented pursuant to this section.
(i) The governing body must identify and document which staff must complete such training.
(ii) The hospital must document in the staff personnel records that the training was successfully completed.
(iii) The hospital must be able to demonstrate staff knowledge on the topics implemented pursuant to this section.
(iv) The hospital must use findings from its QAPI program, as required at § 482.21, to inform staff training needs and any additions, revisions, or updates to training topics on an ongoing basis.
32. Section 482.59 is added to subpart D to read as follows:
If the hospital offers obstetrical services, the services must be well organized and provided in accordance with nationally recognized acceptable standards of practice for the health care (including physical and behavioral health) of pregnant, birthing, and postpartum patients. If outpatient obstetrical services are offered, the services must be consistent in quality with inpatient care in accordance with the complexity of services offered.
(a) Standard: Organization and staffing. Effective January 1, 2026, the organization of the obstetrical services must be appropriate to the scope of the services offered. As applicable, the services must be integrated with other departments of the hospital.
(1) Labor and delivery rooms/suites (including labor rooms, delivery rooms (including rooms for operative delivery), and post-partum/recovery rooms whether combined or separate) must be supervised by an experienced registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a doctor of medicine or osteopathy.
(2) Obstetrical privileges must be delineated for all practitioners providing obstetrical care in accordance with the competencies of each practitioner in accordance with § 482.22(c).
(b) Standard: Delivery of service. Effective January 1, 2026, Obstetrical services must be consistent with needs and resources of the facility. Policies governing obstetrical care must be designed to assure the achievement and maintenance of high standards of medical practice and patient care and safety.
(1) The following equipment must be kept at the hospital and be readily available for treating obstetrical cases to meet the needs of patients in accordance with the scope, volume, and complexity of services offered: call-in-system, cardiac monitor, and fetal doppler or monitor.
(2) There must be adequate provisions and protocols, consistent with nationally recognized and evidence-based guidelines, for obstetrical emergencies, complications, immediate post-delivery care, and other patient health and safety events as identified as part of the QAPI program (§ 482.21). Provisions include equipment (in addition to the equipment required under paragraph (b)(1) of this section), supplies, and medication used in treating emergency cases. Such provisions must be kept in the hospital and be readily available for treating emergency cases.
(c) Standard: Staff training. Effective January 1, 2027, the hospital must develop policies and procedures to ensure that relevant staff are trained on select topics for improving the delivery of maternal care.
(1) Training concepts must reflect the scope and complexity of services offered within the facility, including but not limited to:
(i) Facility-identified evidence-based best practices and protocols to improve the delivery of maternal care within the facility; and
(ii) The hospital must use findings from its QAPI program, as required at § 482.21, to inform staff training needs and any additions, revisions, or updates to training topics on an ongoing basis.
(2) The hospital must provide relevant new staff with initial training.
(3) The governing body must identify and document which staff must complete initial training and subsequent biannual training on the topics identified at paragraph (c)(1) of this section.
(4) The hospital must document in the staff personnel records that the training was successfully completed.
(5) The hospital must be able to demonstrate staff knowledge on the topics identified at paragraph (c)(1) of this section.
PART 485—CONDITIONS OF PARTICIPATION: SPECIALIZED PROVIDERS
33. The authority citation for part 485 continues to read as follows:
34. Section 485.618 is amended by— ( printed page 94593)
a. Redesignating paragraph (e) as paragraph (f); and
b. Adding new paragraph (e).
The addition reads as follows:
(e) Standard: Emergency services readiness. Effective July 1, 2025, in accordance with the complexity and scope of services offered, there must be adequate provisions (as required under paragraphs (b) and (c) of this section) and protocols to meet the emergency needs of patients.
(1) Protocols. Protocols must be consistent with nationally recognized and evidence-based guidelines for the care of patients with emergency conditions, including but not limited to patients with obstetrical emergencies, complications, and immediate post-delivery care.
(2) Staff training. Applicable staff, as identified by the CAH, must be trained annually on the protocols and provisions implemented pursuant to this section.
(i) The governing body must identify and document which staff must complete such training.
(ii) The CAH must document in the staff personnel records that the training was successfully completed.
(iii) The CAH must be able to demonstrate staff knowledge on such training.
(iv) The CAH must use findings from its QAPI program, as required at § 485.641, to inform staff training needs and any additions, revisions, or updates to training topics on an ongoing basis.
35. Section 485.641 is amended by adding paragraph (d)(4) and revising paragraph (e) to read as follows:
(d) * * *
(4) Effective January 1, 2027, for CAHs that offer obstetrical services, the following additional QAPI requirements apply:
(i) Obstetrical services leadership must engage in QAPI as specified in this section for obstetrical services, including but not limited to participating in data collection and monitoring as specified in this paragraph (d) and paragraph (e) of this section.
(ii) If a maternal mortality review committee (MMRC) is available at the State, Tribal, or local jurisdiction in which the CAH is located, the facility leadership, obstetrical services leadership, or their designate(s) must further have a process for incorporating publicly available MMRC(s) data and recommendations into the CAH QAPI program as specified in this section.
(e) Standard: Program data collection and analysis. (1) The program must incorporate quality indicator data including patient care data, in order to achieve the goals of the QAPI program.
(2) Effective January 1, 2027, CAHs that offer obstetrical services, the CAH must utilize its QAPI program to assess and improve health outcomes and disparities among obstetrical patients on an ongoing basis. At a minimum, the CAH must:
(i) Analyze data and quality indicators collected for the QAPI program by diverse subpopulations as identified by the CAH among obstetrical patients.
(ii) Measure, analyze, and track health equity data, measures, and quality indicators on patient outcomes and disparities in processes of care, services and operations, and outcomes among obstetrical patients.
(iii) Analyze and prioritize identified patient health outcomes and disparities, develop and implement actions to improve patient health outcomes and disparities, measure results, and track performance to ensure improvements are sustained when disparities exist among obstetrical patients.
(iv) Conduct at least one measurable performance improvement project focused on improving health outcomes and disparities among the CAH's population(s) of obstetrical patients annually.
36. Section 485.649 is added to read as follows:
If the CAH offers obstetrical services, the services must be well organized and provided in accordance with nationally recognized acceptable standards of practice for the health care (including physical and behavioral health) of pregnant, birthing, postpartum patients. If outpatient obstetrical services are offered, the services must be consistent in quality with inpatient care in accordance with the complexity of services offered.
(a) Standard: Organization and staffing. Effective January 1, 2026, the organization of the obstetrical services must be appropriate to the scope of the services offered. As applicable, the services must be integrated with other departments of the CAH.
(1) Labor and delivery rooms/suites (including labor rooms, delivery rooms (including rooms for operative delivery), and post-partum/recovery rooms whether combined or separate) must be supervised by an experienced registered nurse, certified nurse midwife, nurse practitioner, physician assistant, or a Doctor of Medicine or a Doctor of Osteopathy (MD/DO).
(2) Obstetrical privileges must be delineated for all practitioners providing obstetrical care in accordance with the competencies of each practitioner, and consistent with credentialing agreements established under § 485.616(b).
(b) Standard: Delivery of service. Effective January 1, 2026, obstetrical services must be consistent with needs and resources of the CAH. Policies governing obstetrical care must be designed to assure the achievement and maintenance of high standards of medical practice and patient care and safety.
(1) The following equipment must be kept at the CAH and be readily available for treating obstetrical cases to meet the needs of patients in accordance with the scope, volume, and complexity of services offered: call-in-system, cardiac monitor, and fetal doppler or monitor.
(2) There must be adequate provisions and protocols, consistent with nationally recognized and evidence-based guidelines, for obstetrical emergencies, complications, immediate post-delivery care, and other patient health and safety events as identified as part of the QAPI program (§ 485.641). Provisions include equipment (in addition to the equipment required under paragraph (b)(1) of this section), supplies, and medication used in treating emergency cases. Such provisions must be kept in the CAH and be readily available for treating emergency cases.
(c) Standard: Staff training. Effective January 1, 2027, the CAH must develop policies and procedures to ensure that relevant staff are trained on select topics for improving the delivery of maternal care.
(1) Training concepts must reflect the scope and complexity of services offered within the facility, including but not limited to:
(i) Facility-identified evidence-based best practices and protocols to improve the delivery of maternal care within the facility; and
(ii) The CAH must use findings from its quality assessment and performance improvement (QAPI) program, as required at § 485.641, to inform staff training needs and any additions, revisions, or updates to training topics on an ongoing basis.
(2) The CAH must provide relevant new staff with initial training. ( printed page 94594)
(3) The governing body must identify and document which staff must complete initial training and subsequent biannual training on the topics identified at paragraph (c)(1) of this section.
(4) The CAH must document in the staff personnel records that the training was successfully completed.
(5) The CAH must be able to demonstrate staff knowledge on the topics identified at paragraph (c)(1) of this section.
Xavier Becerra,
Secretary, Department of Health and Human Services.
Footnotes
2. Status indicator “J1” denotes Hospital Part B Services Paid Through a Comprehensive APC. Further information can be found in CY 2025 Addendum D1.
Back to Citation3. Status indicator “J2” denotes Hospital Part B Services That May Be Paid Through a Comprehensive APC. Further information can be found in CY 2025 Addendum D1.
Back to Citation4. https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/bp102c15.pdf.
Back to Citation5. Zolgensma. FDA Package Insert. October 2023. https://www.fda.gov/media/126109/download?attachment.
Back to Citation6. FDA Package Insert. Yescarta. April 2024. https://www.fda.gov/media/108377/download?attachment.
Back to Citation9. Per the manufacturer's website: “Is Magtrace Radioactive? No. Magtrace is the world's first non-radioactive tracer to offer dual identification. This not only facilitates the eradication of radioactive products from the OR, but provides psychological benefits for the patient.” https://www.endomag.com/resources/faqs/magtrace/?product=magtrace.
10. Per the manufacturer's website: “Magtrace® is a uniquely designed non-radioactive liquid tracer used to map the potential spread of cancer to the axilla in sentinel lymph node biopsy procedures. The liquid is made up of tiny particles of iron oxide, coated in sugar and suspended in saline.” https://www.endomag.com/resources/faqs/magtrace/.
Back to Citation12. https://www.cms.gov/files/document/medicare-part-b-inflation-rebate-program-initial-guidance.pdf.
13. In addition, beginning with the April 2023 ASP Drug Pricing file, the file includes the coinsurance percentage for each drug and specifies “inflation-adjusted coinsurance” in the “Notes” column if the coinsurance for a drug is less than 20 percent of the Medicare Part B payment amount. Drug pricing files are available at https://www.cms.gov/medicare/medicare-fee-for-service-part-b-drugs/mcrpartbdrugavgsalesprice.
Back to Citation17. Currently approved under OMB control number 0938-0860; expires 07/31/2024.
Back to Citation18. Luxturna. FDA Package Insert. Available: https://www.fda.gov/media/109906/download.
Back to Citation19. LUXTURNA REIMBURSEMENT GUIDE FOR TREATMENT CENTERS. https://mysparkgeneration.com/uploads/2022/09/LUXTURNA-Reimbursement-Guide-for-Treatment-Centers-ISI-Update-April-2022-P-RPE65-US-320025.pdf.
Back to Citation20. ClinicalTrials.gov. “Randomized Controlled Pivotal Trial of Autologous Bone Marrow Cells Using the CardiAMP Cell Therapy System in Patients With Refractory Angina Pectoris and Chronic Myocardial Ischemia.” Accessed May 10, 2022. https://clinicaltrials.gov/ct2/show/NCT03455725?term=NCT03455725&rank=1.
Back to Citation21. ClinicalTrials.gov. “Randomized Controlled Pivotal Trial of Autologous Bone Marrow Mononuclear Cells Using the CardiAMP Cell Therapy System in Patients With Post Myocardial Infarction Heart Failure.” Accessed May 10, 2022. https://clinicaltrials.gov/ct2/show/NCT02438306.
Back to Citation22. Jelmyto Package Insert. 01/14/2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/211728s002lbl.pdf.
Back to Citation23. ClinicalTrials.gov. “The HistoSonics System for Treatment of Primary and Metastatic Liver Tumors Using Histotripsy (#HOPE4LIVER) (#HOPE4LIVER).” Accessed May 10, 2022. https://clinicaltrials.gov/ct2/show/study/NCT04573881.
Back to Citation24. FDA Package Insert. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/208742s007lbl.pdf.
Back to Citation25. The AMA's CPT Editorial Panel established CPT codes 36836 and 36837 during the January 2023 update and CMS deleted HCPCS codes G2170 and G2171, effective January 1, 2023.
Back to Citation27. ClinicalTrials.gov. “Trial to Evaluate the Safety and Effectiveness of Treatment With COMS One Device in Subjects With Diabetic Foot Ulcers (Mavericks).” Accessed September 24, 2024. https://clinicaltrials.gov/study/NCT05758545
Back to Citation28. Approved IDE Studies- “G220277-NCT05758545.” Accessed September 24, 2024. https://www.cms.gov/medicare/coverage/ide/approved-ide-studies/683545095/g220277-nct05758545
Back to Citation30. To apply for OPPS transitional device pass-through status, applicants complete an application that is subject to the Paperwork Reduction Act of 1995 (PRA) (44 U.S.C. 3501 et seq.). This information collection (CMS-10052) is currently approved under OMB control number 0938-0857 and has an expiration date of November 30, 2025.
Back to Citation31. Centers for Medicare & Medicaid Services (2017). Pub 100-04 Medicare Claims Processing (Transmittal 3941) in CMS Manual System. Accessed at https://www.cms.gov/regulations-and-guidance/guidance/transmittals/2017downloads/r3941cp.pdf.
Back to Citation32. HCPCS code 36902 (Introduction of needle(s) and/or catheter(s), dialysis circuit, with diagnostic angiography of the dialysis circuit, including all direct puncture(s) and catheter placement(s), injection(s) of contrast, all necessary imaging from the arterial anastomosis and adjacent artery through entire venous outflow including the inferior or superior vena cava, fluoroscopic guidance, radiological supervision and interpretation and image documentation and report; with transluminal balloon angioplasty, peripheral dialysis segment, including all imaging and radiological supervision and interpretation necessary to perform the angioplasty) and HCPCS code 36903 (Introduction of needle(s) and/or catheter(s), dialysis circuit, with diagnostic angiography of the dialysis circuit, including all direct puncture(s) and catheter placement(s), injection(s) of contrast, all necessary imaging from the arterial anastomosis and adjacent artery through entire venous outflow including the inferior or superior vena cava, fluoroscopic guidance, radiological supervision and interpretation and image documentation and report; with transcatheter placement of intravascular stent(s) peripheral dialysis segment, including all imaging and radiological supervision and interpretation necessary to perform the stenting, and all angioplasty within the peripheral dialysis segment).
Back to Citation33. We noted that the applicant originally utilized APC 5231 (Level 1 ICD and Similar Procedures) for the three tests of the cost criteria in the application. However, the applicant provided supplemental information indicating that, HCPCS codes 0795T and 0801T were assigned to APC 5224 (Level 4 Pacemaker and Similar Procedures) in the corrected Addendum B to the CY 2024 OPPS/ASC final rule with comment period and they believed that APC 5224 is currently the appropriate APC for the purposes of performing the cost significance calculations. We agreed with the applicant and selected APC 5224 for our calculation, which we believe is a more appropriate APC to use based on the assignment of HCPCS codes 0795T and 0801T to APC 5224 and the clinical similarity to other pacemaker insertion codes in APC 5224.
Back to Citation34. See Breakthrough Devices Program, Guidance for Industry and Food and Drug Administration Staff Document issued on September 15, 2023. https://www.fda.gov/media/162413/download.
Back to Citation35. We noted that the applicant selected the APC payment rate of $1,814.88 and the APC level device offset amount of $178.95 for APC 5302. However, the values selected are inconsistent with the APC payment rate and the APC level device offset amount found in CY 2024 OPPS APC Offset File, which were corrected as described in the CY 2024 OPPS/ASC final rule with comment period correction (89 FR 9002). The HCPCS/CPT code level device offset amounts for the HCPCS/CPT codes provided by the applicant are available in the corrected Addendum P to the CY 2024 OPPS/ASC final rule with comment period. For our calculation, we selected the APC payment rate of $1,812.99 and the HCPCS/CPT code level device offset amount of $14.50 related to HCPCS 43232 in APC 5302 found in the corrected Addendum P, which are the accurate values for these codes. Based on our initial assessment in the proposed rule, using the APC payment rate of $1,812.99 and the device offset amount of $14.50 would result in the EVSTM meeting the cost significance requirement.
Back to Citation36. July 2024 ASC code pairs file: https://www.cms.gov/license/ama?file=/files/zip/july-2024-asc-codepairs.zip.
Back to Citation37. Anthem CG-SURG-111: “Open sacroiliac joint fusion procedures are considered medically necessar y. . . during multisegment spinal constructs (for example, correction of deformity in scoliosis or kyphosis surgery) extending to the ilium. ” Ref: https://www.anthem.com/dam/medpolicies/abcbs/active/guidelines/gl_pw_e001136.html (accessed August 24, 2024).
Back to Citation38. The applicant referenced the Medicare Claims Processing Manual, Chapter 4 to support these assertions.
Back to Citation39. The applicant stated as neither HCPCS code 0338T nor 0339T had a device offset amount listed in Addendum P to the CY 2023 OPPS/ASC final rule with comment period, it selected a device offset amount of $0.00. However, for our calculation, we selected the CY 2023 APC level device offset amount of $1,491.08 for APC 5192 found in CY 2023 NFRM OPPS APC Offset File, as no CY 2023 HCPCS/CPT code level device offset amount was available at the time the application was received. Based on our initial assessment for the proposed rule, using the device offset amount of $1,491.08 would result in the Paradise® Ultrasound RDN System meeting the cost significance requirement.
Back to Citation40. According to the applicant, the current total cost of the Paradise® Ultrasound RDN System device is $23,235.00. For the cost criteria estimates, the applicant submitted an operating cost of $23,000.00 for the Paradise® Ultrasound RDN System device. Per the applicant, the individual component costs are as follows: Paradise® Remote (capital equipment) is $5.00; Paradise® Cart (capital equipment) is $5.00; Paradise® Generator (capital equipment) is $225.00; Paradise® RDN Catheter Kit (one time use) is $22,000.00; Paradise RDN Cable (one time use) is $250.00; and the Paradise® RDN Cartridge (one time use) is $750.00.
Back to Citation41. According to the Medicare Claims Processing Manual (60.4.3), the device pass-through category C1782 (Morcellator) is specific to devices used for cutting, coring, and extracting tissue in laparoscopic procedures. These are distinct from biopsy devices because morcellators are used for the laparoscopic removal of tissue.
Back to Citation42. Medicare Program; Changes to the Hospital Outpatient Prospective Payment System and Calendar Year 2006 Payment Rates, 70 FR 68516 (November 10, 2005).
Back to Citation43. We noted that the applicant selected the APC payment rate of $1,814.88 and the APC level device offset amount of $178.95 for APC 5302. However, the values selected are inconsistent with the APC payment rate and the APC level device offset amount found in CY 2024 OPPS APC Offset File, which were corrected as described in the CY 2024 OPPS/ASC final rule with comment period correction (89 FR 9002). The HCPCS/CPT code level device offset amounts for the HCPCS/CPT codes provided by the applicant are available in the corrected Addendum P to the CY 2024 OPPS/ASC final rule with comment period. For our calculation, we selected the APC payment rate of $1,812.99 and the HCPCS/CPT code level device offset amount of $23.75 related to HCPCS 43242 in APC 5302 found in the corrected Addendum P, which are the accurate values for these codes. Based on our initial assessment for the proposed rule, using the APC payment rate of $1,812.99 and the device offset amount of $23.75 would result in Precision GI meeting the cost significance requirement.
Back to Citation44. The applicant referenced CY 2013 OPPS FR (77 FR 68352) and Transmittal 2386, Change Request 7672 (Jan 13, 2012) to support these assertions.
Back to Citation45. The applicant referenced the Medicare Claims Processing Manual, Chapter 4, section 60.4.3 to support these assertions.
Back to Citation46. We noted that the applicant selected the APC payment rate of $5,451.51 and the HCPCS/CPT code level device offset amount of $3,365.76 for HCPCS 0339T in APC 5192. However, the values selected are inconsistent with the APC payment rate and the device offset amount found in the corrected Addendum P to the CY 2024 OPPS/ASC final rule with comment period. For our calculation, we selected the APC payment rate of $5,445.84 and the HCPCS/CPT code level device offset amount of $3,362.26 for HCPCS 0339T in APC 5192 found in the corrected Addendum P, which are the accurate values for these codes. Based on our initial assessment for the proposed rule, using the APC payment rate of $5,445.84 and the HCPCS/CPT code level device offset amount of $3,362.26 would result in the Symplicity SpyralTM Catheter meeting the cost significance requirement.
Back to Citation47. HCPCS procedure code 0338T (Transcatheter renal sympathetic denervation, percutaneous approach including arterial puncture, selective catheter placement(s) renal artery(ies), fluoroscopy, contrast injection(s), intraprocedural roadmapping and radiological supervision and interpretation, including pressure gradient measurements, flush aortogram and diagnostic renal angiography when performed; unilateral).
Back to Citation48. HCPCS procedure code 0339T (Transcatheter renal sympathetic denervation, percutaneous approach including arterial puncture, selective catheter placement(s) renal artery(ies), fluoroscopy, contrast injection(s), intraprocedural roadmapping and radiological supervision and interpretation, including pressure gradient measurements, flush aortogram and diagnostic renal angiography when performed; bilateral).
Back to Citation49. The Paradise® Ultrasound RDN System device category does not explicitly describe using an endovascular approach; however, it does describe renal nerve ablation via a catheter. We stated that we believe the device category describes the intravascular sonication (ultrasound treatment) delivered by the catheter. We noted that the proposed device description does not preclude an endovascular approach, and that, per the applicant, the Paradise® Ultrasound RDN system is a catheter-based endovascular based system.
Back to Citation50. Fengler, K., Rommel, K.-P., Kriese, W., Blazek, S., Besler, C., von Roeder, M., Desch, S., Thiele, H., & Lurz, P. (2023, February 13). Research Correspondence: 6- and 12-Month Follow-Up From a Randomized Clinical Trial of Ultrasound vs Radiofrequency Renal Denervation (RADIOSOUND-HTN). JACC: Cardiovascular Interventions, 16 (3),: 367-369. https://doi.org/10.1016/j.jcin.2022.10.058
51. Fengler, K., Rommel, K.-P., Blazek, S., Besler, C., Hartung, P., von Roeder, M., Petzold, M., Winkler, S., Höllriegel, R., Desch, S., Thiele, H., & Lurz, P. (2019). A Three-Arm Randomized Trial of Different Renal Denervation Devices and Techniques in Patients With Resistant Hypertension (RADIOSOUND-HTN). Circulation: 139 (5), 590-600. https://doi.org/10.1161/CIRCULATIONAHA.118.037654.
Back to Citation52. Fengler, et al. (2019). A Three-Arm Randomized Trial of Different Renal Denervation Devices and Techniques in Patients With Resistant Hypertension (RADIOSOUND-HTN). Circulation, 139 (5), 590-600. https://doi.org/10.1161/CIRCULATIONAHA.118.037654.
53. Fengler, et al. (2023). Research Correspondence: 6- and 12-Month Follow-Up From a Randomized Clinical Trial of Ultrasound vs Radiofrequency Renal Denervation (RADIOSOUND-HTN). JACC: Cardiovascular Interventions, 16 (3): 367—369. https://doi.org/10.1016/j.jcin.2022.10.058.
Back to Citation54. Fengler, et al. (2019).
55. Fengler, et al. (2023).
56. Symplicity Spyral renal denervation system Instructions for Use. Medtronic.
57. Azizi, M., Schmieder, R.E., Mahfoud, F., Weber, M.A., Daemen, J., Davies, J., Basile, J., Kirtane, A.J., Wang, Y., Lobo, M.D., Saxena, M., Feyz, L., Rader, F., Lurz, P., Sayer, J., Sapoval, M., Levy, T., Sanghvi, K., Abraham, J., Sharp, A.S.P., Fisher, N.D.L., Bloch, M.., Reeve-Stoffer, H., Coleman, L., Mullin, C., & Mauri, Laura on behalf of the RADIANCE-HTM Investigators (2018). Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomised, sham-controlled trial. Lancet, 391 (10137), P2335-P2345. https://doi.org/10.1016/S0140-6736(18)31082-1.
58. Azizi, M., Sanghvi, K., Saxena, M., Gosse, P., Reilly, J.P., Levy, T., Rump, L.C., Persu, A., Basile, J., Bloch, M.J., Daemen, J., Lobo, M.D., Mahfoud, F., Schmieder, R.E., Sharp, A.S.P., Weber, M.A., Sapoval, M., Fong, P., Pathak, A., Lantelme, P., Hsi, D., Bangalore, S., Witkowski, A., Weil, J., Kably, B., Barman, N.C., Reeve-Stoffer, H., Coleman, L., McClure, C.K., Kirtane, A.J. on behalf of the RADIANCE-HTN Investigators (2021). Ultrasound renal denervation for hypertension resistant to a triple medication pill (RADIANCEHTN TRIO): a randomised, multicentre, single-blind, sham-controlled trial. Lancet, 397(10293), P2476-P2486. https://doi.org/10.1016/S0140-6736(21)00788-1.
Back to Citation59. ReCor Paradise Ultrasound RDN Instructions for Use. https://mearis.cms.gov/public/publications/device-ptp/DEP231128137E1 (accessed July 26, 2024).
Back to Citation60. Symplicity Spyral Indications for Use. Available at: https://www.accessdata.fda.gov/cdrh_docs/pdf22/P220026D.pdf. Accessed Sept 2024.
Back to Citation61. Saxena, M., Schmieder, R.E., Kirtane, A.J., Mahfoud, F., Daemen, J., Basile, J., Lurz, P., Gosse, P., Sanghvi, K., Fisher, N.D.L., Rump, L.C., Pathak, A., Blakenstjn, P.J., Mathur, A., Wang, Y., Weber, M.A., Sharp, A.S.P., Bloch, M.J., Barman, N.C., Claude, L., Song, Y., Aziz, M. & Lobo, M.D. (2022). Predictors of blood pressure response to ultrasound renal denervation in the RADIANCE-HTN SOLO study. Journal of Human Hypertension, 36 (7), 629-639. https://doi.org/10.1038/s41371-021-00547-y.
Back to Citation62. Sato, Y., Kawakami, R., Jinnouchi, H., Sakamoto, A., Cornelissen, A., Mori, M., Kawai, K., Guo, L., Coleman, L., Nash, S., Claude, L., Barman, N.C., Romero, M., Kolodgie, F.D., Virmani, R., & Finn, A.V. (2001). Comprehensive Assessment of Human Accessory Renal Artery Periarterial Renal Sympathetic Nerve Distribution. JACC: Cardiovascular Interventions, 14 (3), 304-15. https://doi.org/10.1016/j.jcin.2020.09.043.
63. Struthoff, H., Lauder, L, Hohl, M., Hermens, A., Rami Tzafriri, A., Edelman, E.R., Kunz, M., Böhm, M., Tschernig, T., & Mahfoud, F. (2023). Histological examination of renal nerve distribution, density, and function in humans. EuroIntervention, 19 (7), 612-620. https://doi.org/10.4244/eij-d-23-00264.
Back to Citation64. Podium presentation. Mahfoud. Pooled, 12-month Blood Pressure Reductions Using the Symplicity Spyral Radiofrequency Renal Denervation Catheter. Transvascular Cardiotherapuetics Conference. October 2023.
Back to Citation65. Böhm, M., Kario, K., Kandzari, D.E., Mahfoud, F., Weber, M.A., Schmieder, R.E., Tsioufis, K., Pocock, S., Konstantinidis, D., Choi, J.W., East, C., Lee, D.P., Ma, A., Ewen, S., Cohen, D.L., Wilensky, R., Devireddy, C.M., Lea, J., Schmid, A., Weil, J., Agdirlioglu, T., Reedus, D., Jefferson, B.K., Reyes, D., D'Souza, R., Sharp, A.S.P., Sharif, F., Fahy, M., DeBruin, V., Cohen, S.A., Brar, S., & Townsend, R.R. on behalf of the SPYRAL HTN-OFF MED Pivotal Investigators (2020). Efficacy of catheter-based renal denervation in the absence of antihypertensive medications (SPYRAL HTN-OFF MED Pivotal): a multicentre, randomised, sham-controlled trial. Lancet, 395 (10234), 1444-1451. https://doi.org/10.1016/s0140-6736(20)30554-7.
Back to Citation66. Sakakura, K., Ladich, E., Cheng, Q., Otsuka, F., Yahagi, K., Fowler, D.R., Kolodgie, F.D., Virmani, R., & Joner, M. (2014). Anatomic assessment of sympathetic peri-arterial renal nerves in man. Journal of American College of Cardiology, 64 (7), 635-43. https://doi.org/10.1016/j.jacc.2014.03.059.
Back to Citation67. Wolf, M., Hubbard, B., Sakaoka, A., Rousselle, S., Tellez, A., Jiang, X., Kario, K., Hohl, M., Böhm, M., & Mahfoud, F. (2018). Procedural and anatomical predictors of renal denervation efficacy using two radiofrequency renal denervation catheters in a porcine model. Journal of Hypertension, 36 (12), 2453-2459. https://doi.org/10.1097/hjh.0000000000001840.
Back to Citation68. HCPCS codes C1826 (Generator, neurostimulator (implantable), includes closed feedback loop leads and all implantable components, with rechargeable battery and charging system), C1827 (Generator, neurostimulator (implantable), non-rechargeable, with implantable stimulation lead and external paired stimulation controller), C1821 (Interspinous process distraction device (implantable)), C1822 (Generator, neurostimulator (implantable), high frequency, with rechargeable battery and charging system), C1823 (Generator, neurostimulator (implantable), non-rechargeable, with transvenous sensing and stimulation leads), C1767 (Generator, neurostimulator (implantable), non-rechargeable), C1820 (Generator, neurostimulator (implantable), with rechargeable battery and charging system), and C1825 (Generator, neurostimulator (implantable), non-rechargeable with carotid sinus baroreceptor stimulation lead(s).
Back to Citation69. HCPCS codes C1730 (Catheter, electrophysiology, diagnostic, other than 3D mapping (19 or fewer electrodes)), C1731 Catheter, electrophysiology, diagnostic, other than 3D mapping (20 or more electrodes)), C1732 (Catheter, electrophysiology, diagnostic/ablation, 3D or vector mapping), C1733 (Catheter, electrophysiology, diagnostic/ablation, other than 3D or vector mapping, other than cool-tip), and C2630 (Catheter, electrophysiology, diagnostic/ablation, other than 3D or vector mapping, cool-tip).
Back to Citation70. Mancia, G., Kreutz, R., Brunström, M., Burnier, M., Grassi, G., Januszewicz, A., Muiesan, M.L., Tsioufis, K., Agabiti-Rosei, E., Algharably, E.A.E., Azizi, M., Benetos, A., Borghi, C., Hitij, J.B., Cifkova, R., Coca, A., Cornelissen, V., Cruickshank, J.K., Cunha, P.G., Danser, A.H.J., Pinho, R.M., Delles, C., Dominiczak, A.F., Dorobantu, M., Doumas, M., Fernández-Alfonso, M.S., Halimi, J.M., Járai, Z., Jelaković, B., Jordan, J., Kuznetsova, T., Laurent, S., Lovic, D., Lurbe, E., Mahfoud, F., Manolis, A., Miglinas, M., Narkiewicz, K., Niiranen, T., Palatini, P., Parati, G., Pathak, A., Persu, A., Polonia, J., Redon, J., Sarafidis, P., Schmieder, R., Spronck, B., Stabouli, S., Stergiou, G., Taddei, S., Thomopoulos, C., Tomaszewski, M., Van de Borne, P., Wanner, C., Weber, T., Williams, B., Zhang, Z.Y., & Kjeldsen, SE (2023). 2023 ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Hypertension. Journal of Hypertension, 41 (12), 1874-2071. https://doi.org/10.1097/HJH.0000000000003480.
71. Barbato, E., Azizi, M., Schmieder, R.E., Lauder, L., Böhm, M., Brouwers, S., Bruno, R.M., Dudek, D., Kahan, T., Kandzari, D.E., Lüscher, T.F., Parati, G., Pathak, A., Ribichini, F.L., Schlaich, M.P., Sharp, A.S.P., Sudano, I., Volpe, M., Tsioufis, C., Wijns, W., & Mahfoud, F. (2023). Renal denervation in the management of hypertension in adults. A clinical consensus statement of the ESC Council on Hypertension and the European Association of Percutaneous Cardiovascular Interventions (EAPCI). European Heart Journal, 44 (15), 1313-1330. https://doi.org/10.1093/eurheartj/ehad054.
72. Swaminathan, R.V., East, C.A., Feldman, D.N., Fisher, N.D., Garasic, J.M., Giri, J.S., Kandzari, D.E., Kirtane, A.J., Klein, A., Kobayashi, T., Koenig, G., Li, J., Secemsky, E., Townsend, R.R., & Aronow, H.D. (2023). SCAI Position Statement on Renal Denervation for Hypertension: Patient Selection, Operator Competence, Training and Techniques, and Organizational Recommendations. Journal of the Society for Cardiovascular Angiography & Interventions, 2 (6), 101121. https://doi.org/10.1016/j.jscai.2023.101121.
73. Kandzari DE, Townsend RR, Bakris G, Basile, J., Bloch, M.J., Cohen, D.L., East, C., Ferdinand, K.C., Fisher, N., Kirtane, A., Lee, D.P., Puckrein, G., Rader, F., Vassalotti, J.A., Weber, M.A., Willis, K., & Secemsky, E. (2021). Renal denervation in hypertension patients: Proceedings from an expert consensus roundtable cosponsored by SCAI and NKF. Catheterization & Cardiovascular Interventions, 98 (3), 416-426. https://doi.org/10.1002/ccd.29884.
Back to Citation74. Swaminathan, R.V., East, C.A., Feldman, D.N., Fisher, N.D., Garasic, J.M., Giri, J.S., Kandzari, D.E., Kirtane, A.J., Klein, A., Kobayashi, T., Koenig, G., Li, J., Secemsky, E., Townsend, R.R., & Aronow, H.D. (2023). SCAI Position Statement on Renal Denervation for Hypertension: Patient Selection, Operator Competence, Training and Techniques, and Organizational Recommendations. Journal of the Society for Cardiovascular Angiography & Interventions, 2 (6), 101121. https://doi.org/10.1016/j.jscai.2023.101121.
Back to Citation75. Mancia, G., Kreutz, R., Brunström, M., Burnier, M., Grassi, G., Januszewicz, A., Muiesan, M.L., Tsioufis, K., Agabiti-Rosei, E., Algharably, E.A.E., Azizi, M., Benetos, A., Borghi, C., Hitij, J.B., Cifkova, R., Coca, A., Cornelissen, V., Cruickshank, J.K., Cunha, P.G., Danser, A.H.J., Pinho, R.M., Delles, C., Dominiczak, A.F., Dorobantu, M., Doumas, M., Fernández-Alfonso, M.S., Halimi, J.M., Járai, Z., Jelaković, B., Jordan, J., Kuznetsova, T., Laurent, S., Lovic, D., Lurbe, E., Mahfoud, F., Manolis, A., Miglinas, M., Narkiewicz, K., Niiranen, T., Palatini, P., Parati, G., Pathak, A., Persu, A., Polonia, J., Redon, J., Sarafidis, P., Schmieder, R., Spronck, B., Stabouli, S., Stergiou, G., Taddei, S., Thomopoulos, C., Tomaszewski, M., Van de Borne, P., Wanner, C., Weber, T., Williams, B., Zhang, Z.Y., & Kjeldsen, SE (2023). 2023 ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Hypertension. Journal of Hypertension, 41 (12), 1874-2071. https://doi.org/10.1097/HJH.0000000000003480.
76. Barbato, E., Azizi, M., Schmieder, R.E., Lauder, L., Böhm, M., Brouwers, S., Bruno, R.M., Dudek, D., Kahan, T., Kandzari, D.E., Lüscher, T.F., Parati, G., Pathak, A., Ribichini, F.L., Schlaich, M.P., Sharp, A.S.P., Sudano, I., Volpe, M., Tsioufis, C., Wijns, W., & Mahfoud, F. (2023). Renal denervation in the management of hypertension in adults. A clinical consensus statement of the ESC Council on Hypertension and the European Association of Percutaneous Cardiovascular Interventions (EAPCI). European Heart Journal, 44 (15), 1313-1330. https://doi.org/10.1093/eurheartj/ehad054.
Back to Citation77. Please see the public posting at [ https://www.regulations.gov/document/CMS-2024-0199-0002/comment for a complete list of evidence and background documents submitted by the applicant during the public comment process. Please note, that as in the proposed rule, we do not summarize background documents, which are documents that do not support the applicant's assertions.
Back to Citation78. Centers for Medicare & Medicaid Services (2020). MLN Matters: July 2020 Update of the Ambulatory Surgical Center (ASC) Payment System (HHS-0938-2020-F-6667). U.S. Department of Health and Human Services. https://www.hhs.gov/guidance/sites/default/files/hhs-guidance-documents/MM11842.pdf.
Back to Citation79. Centers for Medicare & Medicaid Services (2020). MLN Matters: July 2020 Update of the Ambulatory Surgical Center (ASC) Payment System (HHS-0938-2020-F-6667). U.S. Department of Health and Human Services. https://www.hhs.gov/guidance/sites/default/files/hhs-guidance-documents/MM11842.pdf.
Back to Citation80. Centers for Medicare & Medicaid Services (2020). MLN Matters: July 2020 Update of the Ambulatory Surgical Center (ASC) Payment System (HHS-0938-2020-F-6667). U.S. Department of Health and Human Services. https://www.hhs.gov/guidance/sites/default/files/hhs-guidance-documents/MM11842.pdf.
Back to Citation81. Centers for Medicare & Medicaid Services (2020). MLN Matters: July 2020 Update of the Ambulatory Surgical Center (ASC) Payment System (HHS-0938-2020-F-6667). U.S. Department of Health and Human Services. https://www.hhs.gov/guidance/sites/default/files/hhs-guidance-documents/MM11842.pdf.
Back to Citation82. Muscarella, L.F. (2022). Contamination of Flexible Endoscopes and Associated Infections: A Comprehensive Review and Analysis of FDA Adverse Event Reports. LFM Healthcare Solutions, LLC. https://lfm-hcs.com/2022/01/contamination-of-flexible-endoscopes-and-associated-infections/.
Back to Citation83. Muscarella, L.F. (2023). Gastroscopes Have Been Linked to A Cluster of Resistant E. coli Infections—Is the Risk Sufficiently Recognized? LFM Healthcare Solutions, LLC. https://lfm-hcs.com/2023/01/gastroscopes-have-been-linked-to-a-cluster-of-resistant-e-coli-infections-is-the-risk-sufficiently-recognized/.
Back to Citation84. Ofstead, C.L., Smart, A.G., Hopkins, K.M., & Wetzler, H.P. (2023). The utility of lighted magnification and borescopes for visual inspection of flexible endoscopes. American Journal of Infection Control, 51 (1), 2-10. https://doi.org/10.1016/j.ajic.2022.08.026.
Back to Citation85. MAUDE Adverse Event Report: AIZU OLYMPUS CO., LTD. EVIS EXERA III GASTROINTESTINAL VIDEOSCOPE ( fda.gov).
Back to Citation86. Muscarella, L.F. (2022). Contamination of Flexible Endoscopes and Associated Infections: A Comprehensive Review and Analysis of FDA Adverse Event Reports. https://lfm-hcs.com/2022/01/contamination-of-flexible-endoscopes-and-associated-infections/.
87. Muscarella, L.F. (2023). Gastroscopes Have Been Linked to A Cluster of Resistant E. coli Infections—Is the Risk Sufficiently Recognized? https://lfm-hcs.com/2023/01/gastroscopes-have-been-linked-to-a-cluster-of-resistant-e-coli-infections-is-the-risk-sufficiently-recognized/.
88. MAUDE Adverse Event Report: AIZU OLYMPUS CO., LTD. EVIS EXERA III GASTROINTESTINAL VIDEOSCOPE ( fda.gov).
Back to Citation89. Muscarella, L.F. (2022). Contamination of Flexible Endoscopes and Associated Infections: A Comprehensive Review and Analysis of FDA Adverse Event Reports. https://lfm-hcs.com/2022/01/contamination-of-flexible-endoscopes-and-associated-infections/.
90. Muscarella, L.F. (2023). Gastroscopes Have Been Linked to A Cluster of Resistant E. coli Infections—Is the Risk Sufficiently Recognized? https://lfm-hcs.com/2023/01/gastroscopes-have-been-linked-to-a-cluster-of-resistant-e-coli-infections-is-the-risk-sufficiently-recognized/.
91. Ofstead, C.L., Smart, A.G., Hopkins, K.M., & Wetzler, H.P. (2023). The utility of lighted magnification and borescopes for visual inspection of flexible endoscopes. American Journal of Infection Control, 51(1), 2-10.
92. MAUDE Adverse Event Report: AIZU OLYMPUS CO., LTD. EVIS EXERA III GASTROINTESTINAL VIDEOSCOPE ( fda.gov).
Back to Citation93. Please see the public posting at https://www.regulations.gov/document/CMS-2024-0199-0002/comment for a complete list of background documents submitted [by the applicant] during the public comment process.
Back to Citation94. Haislip, I., Hoffman, D., Dehlholm-Lambertsen, E., & Cool, C. (20223) Single-Use Vs Reusable Endoscope Reprocessing: A Staff Survey on Safety and Effectiveness. Value in Health, 26 (12): S434. https://doi.org/10.1016/j.jval.2023.09.227.
95. Hoffman, D. & Cool, C. (2024). Gastrointestinal Endoscopy Volume, Efficiency, And Safety Issues: A Case Report. Ambu USA. https://www.ambuusa.com/Files/Files/Downloads/Ambu%20USA/Gastrointestinal-endoscopy-volume_-efficiency_-and-safety-issues-a-case-report-poster.pdf.
96. Wallace, M.M., Keck, T., Dixon, H., & Yassin, M. (2025). Borescope examination and microbial culture results of endoscopes in a tertiary care hospital led to changes in storage protocols to improve patient safety. American Journal of Infection Control, 51 (4): 361-366. https://doi.org/10.1016/j.ajic.2022.09.009.
97. Ofstead, C.L., Smart, A.G., Hopkins, K.M., & Wetzler, H.P. (2022). The utility of lighted magnification and borescopes for visual inspection of flexible endoscopes. American Journal of Infection Control, (22) 00660-5. https://doi.org/10.1016/j.ajic.2022.08.026.
98. Billy, H. & Pradhan, S. (2024). Superior Backward Articulation in Disposable Gastroscopes versus Reusable Gastroscopes. Surgery for Obesity and Related Diseases, 20 (6): S136. https://doi.org/10.1016/j.soard.2024.04.426.
Back to Citation99. Johnson, B.A., Raman, J.D., Best, S.L., & Lotan, Y. (2023). Prospective Randomized Trial of Single-Use vs Reusable Cystoscope for Ureteral Stent Removal. Journal of Endourology, 37 (10): 1139-1144. https://doi.org/10.1089/end.2023.0134.
100. Göger, Y.E., Özkent, M.S., Kılınç M.T., Taşkapu, H.H., Göger, E., Aydın, A., Sönmez, M.G., & Karalezli, G. (2021). Efficiency of retrograde intrarenal surgery in lower pole stones: disposable flexible ureterorenoscope or reusable flexible ureterorenoscope? World Journal of Urology, 39 (9): 3643-3650. https://doi.org/10.1007/s00345-021-03656-y.
101. Bowen, A.J., Macielak, R.J., Fussell, W., Yeakel, S., McMillan, R., Goates, A, Awadallah, A., & Ekbom, DC (2024). Single-use versus reusable rhinolaryngoscopes for inpatient otorhinolaryngology consults: Resident and patient experience. Laryngoscope: Investigative Otolaryngology, 9 (1): e1203. https://doi.org/10.1002/lio2.1203.
Back to Citation102. Wallace, M.M., Keck, T., Dixon, H., & Yassin, M. (2025). Borescope examination and microbial culture results of endoscopes in a tertiary care hospital led to changes in storage protocols to improve patient safety. American Journal of Infection Control, 51 (4): 361-366. https://doi.org/10.1016/j.ajic.2022.09.009.
103. Goyal, H., Larsen, S., Perisetti, A., Larsen, N.B., Ockert, L.K., Adamsen, S., Tharian, B., & Thosani, N. (2002). Gastrointestinal endoscope contamination rates—elevators are not only to blame: a systematic review and meta-analysis. Endoscopy: International Open, 10(06): E840-E853. https://doi.org/10.1055/a-1795-8883.
Back to Citation104. Goyal, H., Larsen, S., Perisetti, A., Larsen, N.B., Ockert, L.K., Adamsen, S., Tharian, B., & Thosani, N. (2002). Gastrointestinal endoscope contamination rates—elevators are not only to blame: a systematic review and meta-analysis. Endoscopy: International Open, 10(06): E840-E853. https://doi.org/10.1055/a-1795-8883.
Back to Citation105. Billy, H. & Pradhan, S. (2024). Superior Backward Articulation in Disposable Gastroscopes versus Reusable Gastroscopes. Surgery for Obesity and Related Diseases, 2 0 (6): S136. https://doi.org/10.1016/j.soard.2024.04.426.
Back to Citation106. Tomlinson L, Halipern N, Alexander M, Townsend S, Zabriskie K, Luper L. Modernizing Medical Device Instructions for Use (IFUs): Infection Preventionists Speak Up for Patient Safety. APIC_Modernizing-Medical-Device-IFUs_5_16_24.pdf ( sdapic.org).
Back to Citation107. Device Pass-through Requirements: https://mearis.cms.gov/public/resources.
Back to Citation108. We noted that the applicant selected a device offset amount of $21.55 for APC 5301 without selecting a specific HCPCS/CPT code. However, for the HCPCS/CPT codes provided by the applicant, we noted the HCPCS/CPT code level device offset amounts are available in Addendum P to the CY 2023 OPPS/ASC final rule with comment period. For our calculation, we selected the HCPCS/CPT code level device offset amount of $2.64 related to HCPCS 43239 in APC 5301 found in Addendum P to the CY 2023 OPPS/ASC final rule with comment period. Based on our initial assessment for the proposed rule, using the device offset amount of $2.64 would result in Ambu® aScopeTM Gastro meeting the cost significance requirement.
Back to Citation109. Medicare and Medicaid Programs: Hospital Outpatient Prospective Payment and Ambulatory Surgical Center Payment Systems and Quality Reporting Programs; Hospital Value-Based Purchasing Program; Organ Procurement Organizations; Quality Improvement Organizations; Electronic Health Records (EHR) Incentive Program; Provider Reimbursement Determinations and Appeals (CMS-1601-FC), 78 FR 74826 (Dec. 10, 2013) (amending 42 CFR parts 405, 410, 412, 419, 475, 476, 486, and 495).
Back to Citation110. Bell, D.P., et al. (2023). A Clinical Study Using Combination Therapy with Standard of Care for the Treatment of Diabetic Foot Ulcers: Interim Analysis. (poster presented at the 20th Annual Desert Foot Conference, December 6-9, 2023, Phoenix, AZ, USA).
111. Randomized Controlled Trial in Venous Leg Ulcers (NCT05291169) (no author or publication date given).
Back to Citation112. Black, G., Bakewell, S., & Bell, D. (2023). A Novel Combination Therapy Technology: Case Studies of Complete Closure of a Diabetic Foot Ulcer and a Charcot Foot Ulcer. (poster presented at the 2023 Diabetic Foot Conference, September 28-30, 2023, Anaheim, CA).
113. Barrett, C.L., Bakewell, S.J., & Bell, D. (2023). A Novel Combination Therapy Technology: Case Studies of Complete Closure of Diabetic Foot Ulcers. Presented at the 2023 Diabetic Foot Conference, September 28-30, 2023, Anaheim, CA.
Back to Citation114. Demographics for RCT in VLU (no author or publication date given).
Back to Citation115. Randomized Controlled Trial in Venous Leg Ulcers (NCT05291169) (no author or publication date given).
Back to Citation116. OCM treating Multiple Etiologies Final Trial Data (no author or publication date given).
Back to Citation117. Princeton Consumer Research Corp. (2019). Final Report. A safety study to assess the allergy potential of OMEZA collagen matrix in human subjects using the skin prick method.
Back to Citation118. The SweetBio Apis is FDA cleared and marketed under 510(k) since 2019 (FDA 510(k)) letter: https://www.accessdata.fda.gov/cdrh_docs/pdf21/K211972.pdf.
Back to Citation119. Bettle III, G., Bell, D.P., & Bakewell, S.J. (2024). A Novel Comprehensive Therapeutic Approach to the Challenges of Chronic Wounds: A Brief Review and Clinical Experience Report. Advances in Therapy, 41: 492-508. https://doi.org/10.1007/s12325-023-02742-4.
120. Simman, R., Bakewell, S.J., Bell, D., Shuman, S., & Cheney, M. (2024). A novel approach for the treatment of diabetic foot ulcers using a multimodal wound matrix: a clinical study. Journal of Wound Care. https://doi.org/10.12968/jowc.2024.0085.
121. Cole, W. (2024). Treatment of bacterially contaminated lower extremity ulcers with a fatty acid-containing wound matrix: a case series. Journal of Wound Care, 33 (8):554-559. https://doi.org/10.12968/jowc.2024.0101.
Back to Citation122. Sweetbio®. Citations. https://www.sweetbio.com/apis.
Back to Citation123. Note that while CMS refers to these materials as “skin substitutes” in its 2024 guidance Billing and Coding: Skin Substitutes Grafts/Cellular Tissue-Based Products for the Treatment of Diabetic Foot Ulcers and Venous Leg Ulcers, https://www.cms.gov/medicare-coverage-database/view/article.aspx?articleid=59625, the FDA collectively refers to this class of products as Dressings, Wound/Dressings, Wound, Collagen. see e.g. FDA (2022). Traditional 510(k), Kerecis, pg. 6. https://www.accessdata.fda.gov/cdrh_docs/pdf21/K213231.pdf.
Back to Citation124. The SweetBio Apis is FDA cleared and marketed under 510(k) since 2019 (FDA 510(k)) letter: https://www.accessdata.fda.gov/cdrh_docs/pdf21/K211972.pdf. Note that the FDA collectively refers to this class of products as Dressings, Wound or Dressings, Wound, Collagen. See e.g. FDA (2022). Traditional 510(k), Kerecis, pg. 6. https://www.accessdata.fda.gov/cdrh_docs/pdf21/K213231.pdf.
Back to Citation125. Dhillon, Y., Mulder, G., Patel, K., Moya, L., Boghossian, G., Swain, D., McLafferty, R., Perez, K., Nguyen, J., Wilkinson, N., Arragon, J., Contreras, L., Geiger, D., Cummings, R., LaVigne, B., Bell, D., Bakewell, S. (2024). An open-label, interventional, prospective, real-world evidence study to evaluate a multimodal wound matrix in patients with refractory wounds. Advances in Wound Care, Ed (pre-print).
Back to Citation126. Cole, W. (2024). Treatment of bacterially contaminated lower extremity ulcers with a fatty acid-containing wound matrix: a case series. Journal of Wound Care, 33 (8):554-559. https://doi.org/10.12968/jowc.2024.0101.
Back to Citation127. CMS approved an application for the Shockwave IVL System with Shockwave C2 Coronary IVL Catheter as a new device category for transitional pass-through payment status and established HCPCS code C1761 as a new device category effective July 1, 2021. We refer readers to the CY 2022 OPPS/ASC final rule with comment period (86 FR 63577 through 63583) for a full discussion the Shockwave IVL System with Shockwave C2 Coronary IVL Catheter application and decision.
Back to Citation128. Background articles are not included in the following table but can be accessed via the online posting for the technology.
Back to Citation129. The applicant indicated 35 atms of pressure as the rated burst pressure in the information included in the application.
Back to Citation130. We noted the applicant selected APC 5192 and an APC payment rate of $5,061.89 for the three tests of the cost criteria. However, for our calculation, we selected APC 5183, which we believe had the lowest applicable APC payment rate of $2,978.97 found in Addendum P to the CY 2023 OPPS/ASC final rule with comment period, among the APCs related to the HCPCS/CPT codes provided by the applicant. We selected the HCPCS/CPT code level device offset amount of $530.85 related to HCPCS 75625 in APC 5183. Based on our initial assessment in the proposed rule, using the APC payment rate of $2,978.97 and the device offset amount of $530.85 would result in OSCAR® meeting the cost significance requirement.
Back to Citation131. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation132. To apply for OPPS transitional Pass-Through Payment Status and New Technology Ambulatory Payment Classification (APC), applicants complete an application that is subject to the Paperwork Reduction Act of 1995 (PRA) (44 U.S.C. 3501 et seq.). This information collection (CMS-10008) is currently approved under OMB control number of 0938-0802 and has an expiration date of January 31, 2025.
Back to Citation133. We are finalizing our proposal to pay separately for diagnostic radiopharmaceuticals with per-day costs above a threshold. This category of policy-packaged drugs that function as supplies in a diagnostic test or procedure includes diagnostic radiopharmaceuticals with per-day costs below the threshold for the applicable year. Please refer to Section II.A.3.c. for more information regarding this policy.
Back to Citation134. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation135. We are finalizing our proposal to pay separately for diagnostic radiopharmaceuticals with per-day costs above a threshold. This category of policy-packaged drugs that function as supplies in a diagnostic test or procedure includes diagnostic radiopharmaceuticals with per-day costs below the threshold for the applicable year. Please refer to section II.A.3.c. for more information regarding this policy.
Back to Citation136. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation137. We are finalizing our proposal to pay separately for diagnostic radiopharmaceuticals with per-day costs above a threshold. This category of policy-packaged drugs that function as supplies in a diagnostic test or procedure includes diagnostic radiopharmaceuticals with per-day costs below the threshold for the applicable year. Please refer to section II.A.3.c. for more information regarding this policy.
Back to Citation138. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation139. In this rule we adopt a policy to pay separately beginning in CY 2025 for diagnostic radiopharmaceuticals with per-day costs above a threshold. This category of policy-packaged drugs that function as supplies in a diagnostic test or procedure would include diagnostic radiopharmaceuticals with per-day costs at or below the threshold for the applicable year. Please refer to section II.A.3.c. for more information regarding this policy.
Back to Citation140. In section II.A.3 of this rule, we adopt a policy to pay separately for diagnostic radiopharmaceuticals with per-day costs above a d threshold. This category of policy-packaged drugs that function as supplies in a diagnostic test or procedure includes diagnostic radiopharmaceuticals with per-day costs below the threshold for the applicable year. Please refer to section II.A.3.c. for more information regarding this policy.
Back to Citation141. Medicare Payment Advisory Committee. June 2005 Report to the Congress. Chapter 6: Payment for pharmacy handling costs in hospital outpatient departments. Available at: https://www.medpac.gov/wp-content/uploads/import_data/scrape_files/docs/default-source/reports/June05_ch6.pdf.
Back to Citation142. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation143. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation144. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation145. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation146. Please see section II.A.3.c. for more information regarding the payment of diagnostic radiopharmaceuticals.
Back to Citation147. As discussed in section IV.A.2 of this final rule with comment period, the DETOURTM System pass-through application was preliminarily approved for transitional pass-through payment under the alternative pathway effective January 1, 2024, and the AveirTM DR System pass-through application and the UDK-T component of the EVSTM pass-through application were preliminarily approved for transitional pass-through payment under the alternative pathway effective July 1, 2024. We are finalizing the approvals for device pass-through payment status for the DETOURTM System, the AveirTM DR System and the UDK-T component of the EVSTM in this final rule with comment period. Due to the timing of the preliminary approvals, the CY 2025 spending estimate for the DETOURTM System is included in the CY 2025 spending estimate for the first group of devices; and the CY 2025 spending estimate for the AveirTM DR System and the UDK-T component of the EVSTM is included in the CY 2025 spending estimate for the second group of devices.
Back to Citation148. The device application associated with HCPCS code C8000 will be included and discussed in the CY 2026 OPPS/ASC proposed and final rules.
Back to Citation149. https://www.cms.gov/medicare-coverage-database/view/article.aspx?articleid=56850&ver=23&.
Back to Citation150. Click on the link labeled “CY 2025 OPPS/ASC Notice of Proposed Rulemaking”, which can be found under the heading “Hospital Outpatient Prospective Payment System Rulemaking” and open the claims accounting document link at the bottom of the page, which is labeled “2025 NPRM OPPS Claims Accounting (PDF)”.
Back to Citation153. In the CY 2023 OPPS/ASC final rule with comment period, we removed § 410.27(a)(1)(iv)(D) in its entirety and added its language regarding pulmonary rehabilitation, cardiac rehabilitation, and intensive cardiac rehabilitation services and the virtual presence of a physician through audio/video real-time communications technology during the PHE to the newly designated § 410.27(a)(1)(iv)(B)( 1) (87 FR 72024).
Back to Citation154. Ibid.
Back to Citation155. Ibid.
Back to Citation156. Ibid.
Back to Citation158. 88 FR 87789 (December 19, 2023); https://www.federalregister.gov/documents/2023/12/19/2023-27815/reimbursement-rates-for-calendar-year-2024.
Back to Citation159. IHS Critical Access Hospitals (CAHs) are paid for covered outpatient services based on 101 percent of an all-inclusive facility specific rate rather than the national AIR rate. Consequently, they are excluded from the proposed separate payment policy.
Back to Citation160. Sections 321(a) and 322(b) of the Public Health Service Act (42 U.S.C. 248), Public Law 83-568 (42 U.S.C. 2001(a)), and the Indian Health Care Improvement Act (25 U.S.C. 1601 et seq.).
Back to Citation161. The OPPS provides outlier payments to hospitals to help mitigate the financial risk associated with high-cost and complex procedures, where a very costly service could present a hospital with significant financial loss. Outlier payments are provided on a service basis when the cost of a service exceeds the APC payment amount multiplier threshold (the APC payment amount multiplied by 1.75) as well as the APC payment amount plus a fixed-dollar amount threshold (the APC payment plus a certain dollar amount).
Back to Citation163. A biosimilar is a biologic medication. It is highly similar to a biologic medication already approved by FDA—the original biologic (also called the reference product).
Back to Citation167. https://www.cms.gov/medicare/payment/prospective-payment-systems/hospital-outpatient.
Back to Citation168. Health Insurance Marketplace® is a registered service mark of the U.S. Department of Health & Human Services.
Back to Citation170. https://www.cdc.gov/niosh/npptl/respirators/testing/NonNIOSHresults.html.
Back to Citation173. Glove Story Global Glove Production Amidst the COVID-19 Pandemic ( usitc.gov).
Back to Citation174. Glove Story Global Glove Production Amidst the COVID-19 Pandemic ( usitc.gov).
Back to Citation176. https://www.medicaid.gov/medicaid/prescription-drugs/retail-price-survey/index.html.
Back to Citation180. Coverage with Evidence Development: Guidance Document. August 7, 2024. https://www.cms.gov/medicare-coverage-database/view/medicare-coverage-document.aspx?mcdid=38.
Back to Citation181. https://www.cms.gov/medicare-coverage-database/view/medicare-coverage-document.aspx?mcdid=38.
Back to Citation182. Medicare Payment Advisory Committee. March 2024 Report to the Congress. Chapter 3: Hospital inpatient and outpatient services, p.49. Available at: https://www.medpac.gov.
Back to Citation183. Medicare Payment Advisory Committee. March 2024 Report to the Congress. Chapter 10: Ambulatory surgical center services: Status report, p.297. Available at: https://www.medpac.gov/wp-content/uploads/2024/03/Mar24_MedPAC_Report_To_Congress_SEC.pdf.
Back to Citation184. Caldolor. FDA Package Insert. May 2023. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/022348s024lbl.pdf.
Back to Citation185. Dexycu. FDA Package Insert. June 2020. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/208912s002lbl.pdf.
Back to Citation186. Ofirmev. FDA Package Insert. April 2018. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/022450s011lbl.pdf.
Back to Citation187. Ketorolac tromethamine Injection. FDA Package Insert. May 2014. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/074802s038lbl.pdf.
Back to Citation188. Exparel. FDA Package Insert. November 2023. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/022496s051lbl.pdf.
189. Omidria. FDA Package Insert. December 2017. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205388s006lbl.pdf.
190. Dextenza. FDA Package Insert. October 2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/208742s007lbl.pdf.
191. Xaracoll. FDA Package Insert. August 2020. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/209511s000lbl.pdf.
192. Zynrelef. FDA Package Insert. January 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/211988s013lbl.pdf.
193. Ketorolac tromethamine Injection. FDA Package Insert. May 2014. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/074802s038lbl.pdf.
194. On-Q Pump. FDA 510K. February 2019. https://www.accessdata.fda.gov/cdrh_docs/pdf18/K181360.pdf.
195. Ding DY, Manoli A 3rd, Galos DK, Jain S, Tejwani NC. Continuous Popliteal Sciatic Nerve Block Versus Single Injection Nerve Block for Ankle Fracture Surgery: A Prospective Randomized Comparative Trial. J Orthop Trauma. 2015;29(9):393-398. https://pubmed.ncbi.nlm.nih.gov/2616525.
Back to Citation196. FDA Approval Letter, July 31, 2018. https://www.accessdata.fda.gov/cdrh_docs/pdf18/K181422.pdf.
Back to Citation197. Ilfeld BM, Plunkett A, Vijjeswarapu AM, Hackworth R, Dhanjal S, Turan A, Cohen SP, Eisenach JC, Griffith S, Hanling S, Sessler DI, Mascha EJ, Yang D, Boggs JW, Wongsarnpigoon A, Gelfand H Percutaneous, Peripheral Nerve Stimulation (Neuromodulation) for Postoperative Pain: A Randomized, Sham-controlled Pilot Study, Anesthesiology, April 2021.
Back to Citation198. FDA Approval Letter, July 31, 2018. https://www.accessdata.fda.gov/cdrh_docs/pdf18/K181422.pdf.
Back to Citation199. Ilfeld BM, Plunkett A, Vijjeswarapu AM, Hackworth R, Dhanjal S, Turan A, Cohen SP, Eisenach JC, Griffith S, Hanling S, Sessler DI, Mascha EJ, Yang D, Boggs JW, Wongsarnpigoon A, Gelfand H Percutaneous, Peripheral Nerve Stimulation (Neuromodulation) for Postoperative Pain: A Randomized, Sham-controlled Pilot Study, Anesthesiology, April 2021.
Back to Citation200. See FDA Product Classification, Product Code ILO, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpcd/classification.cfm?id=5437 (identifying ILO devices as Class 2, 510(k) exempt devices, described further at 21 CFR 890.5720); see also FDA 510(k) Premarket Notification, K955057 (Mar. 8, 1996), https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPMN/pmn.cfm?ID=K955057 (recognizing motorized cold therapy devices used for the “application of ice after surgery or injury to reduce swelling and pain”).
201. FDA, Product Code Database, “ILO”, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPCD/classification.cfm?ID=5437; see also 21 CFR 890.5720.
202. https://www.accessdata.fda.gov/cdrh_docs/pdf/K955057.pdf.
Back to Citation203. Jones CA et al. Opioid-sparing pain management protocol after shoulder arthroplasty results in less opioid consumption and higher satisfaction: a prospective, randomized controlled trial. J Shoulder Elbow Surg. 2022 Oct;31(10):2057-2065. doi: 10.1016/j.jse.2022.05.029. Epub 2022 Jul 5. PMID: 35803549.
204. Barber FA, McGuire DA, Click S. Continuous-flow cold therapy for outpatient anterior cruciate ligament reconstruction. Arthroscopy. 1998 Mar;14(2):130-5. doi: 10.1016/s0749-8063(98)70030-1. PMID: 9531122.
205. P.B. Wyatt et al., The Role of Cryotherapy After Total Knee Arthroplasty: A Systematic Review, The Journal of Arthroplasty, Volume 38, 2023, Pages 950-956.
Back to Citation206. See FDA Product Classification, Product Code ILO, https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpcd/classification.cfm?id=5437 (identifying ILO devices as Class 2, 510(k) exempt devices, described further at 21 CFR 890.5720); see also FDA 510(k) Premarket Notification, K955057 (Mar. 8, 1996), https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPMN/pmn.cfm?ID=K955057 (recognizing motorized cold therapy devices used for the “application of ice after surgery or injury to reduce swelling and pain”).
207. FDA, Product Code Database, “ILO,” https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfPCD/classification.cfm?ID=5437; see also 21 CFR 890.5720.
208. https://www.accessdata.fda.gov/cdrh_docs/pdf/K955057.pdf.
Back to Citation209. Jones CA et al. Opioid-sparing pain management protocol after shoulder arthroplasty results in less opioid consumption and higher satisfaction: a prospective, randomized controlled trial. J Shoulder Elbow Surg. 2022 Oct;31(10):2057-2065. doi: 10.1016/j.jse.2022.05.029. Epub 2022 Jul 5. PMID: 35803549.
210. Barber FA, McGuire DA, Click S. Continuous-flow cold therapy for outpatient anterior cruciate ligament reconstruction. Arthroscopy. 1998 Mar;14(2):130-5. doi: 10.1016/s0749-8063(98)70030-1. PMID: 9531122.
211. P.B. Wyatt et al., The Role of Cryotherapy After Total Knee Arthroplasty: A Systematic Review, The Journal of Arthroplasty, Volume 38, 2023, Pages 950-956.
Back to Citation212. Jones CA, Throckmorton TW, Murphy J, Eason RR, Joyce M, Bernholt DL, Azar FM, Brolin TJ. Opioid-sparing pain management protocol after shoulder arthroplasty results in less opioid consumption and higher satisfaction: a prospective, randomized controlled trial. J Shoulder Elbow Surg. 2022 Oct;31(10):2057-2065. doi: 10.1016/j.jse.2022.05.029. Epub 2022 Jul 5. PMID: 35803549.
213. Barber FA, McGuire DA, Click S. Continuous-flow cold therapy for outpatient anterior cruciate ligament reconstruction. Arthroscopy. 1998 Mar;14(2):130-5. doi: 10.1016/s0749-8063(98)70030-1. PMID: 9531122.
214. Muaddi H, Lillie E, Silva S, Cross JL, Ladha K, Choi S, Mocon A, Karanicolas P. The Effect of Cryotherapy Application on Postoperative Pain: A Systematic Review and Meta-analysis. Ann Surg. 2023 Feb 1;277(2):e257-e265. doi: 10.1097/SLA.0000000000004987. Epub 2023 Jan 10. PMID: 34856580.
215. Wyatt PB, Nelson CT, Cyrus JW, Goldman AH, Patel NK. The Role of Cryotherapy After Total Knee Arthroplasty: A Systematic Review. J Arthroplasty. 2023 May;38(5):950-956. doi: 10.1016/j.arth.2022.12.004. Epub 2022 Dec 8. PMID: 36496048.
Back to Citation219. O'Connor LA, Dua A, Orhurhu V, Hoepp LM, Quinn CC. Opioid Requirements After Intercostal Cryoanalgesia in Thoracic Surgery. J Surg Res. 2022; 274:232-241.
220. Maxwell CM, Weksler B, Houda J, Fernando HC. Intercostal Cryoablation During Video-Assisted Lung Resection Can Decrease Postoperative Opioid Use. Innovations 2023 18(4):352-356.
221. Jaroszewski DE, Bostoros P, Farina JM, Botros MM, Aly MR, Peterson M, Lackey J, Pulivarthi KV, Smith B, Craner R, Stearns JD. Evolution of Pain Control for Adult Pectus Excavatum Repair. Ann Thorac Surg. 2024;117(4):829-837.
222. Graves CE, Moyer J, Zobel MJ, Mora R, Smith D, O'Day M, Padilla BE. Intraoperative intercostal nerve cryoablation during the Nuss procedure reduces length of stay and opioid requirement: A randomized clinical trial. J Pediatric Surg. 2019 Nov;54(11):2250-2256.
Back to Citation223. FDA 510(k) Clearance K052221; obtained in 2005 by and issued to Sorenson Medical, Inc., which was later acquired by Summit Medical Products, Inc. Avanos acquired substantially all assets of Summit Medical Products, including this FDA 510(k) Clearance K052221, in 2019.
Back to Citation224. Morkos M, DeLeon A, Koeckert M, Gray Z, Liao K, Pan W, Tolpin DA. The Use of Unilateral Erector Spinae Plane Block in Minimally Invasive Cardiac Surgery. J Cardiothorac Vasc Anesth. 2023 Mar;37(3):432-436.
Back to Citation225. FDA 510(k) No. K220656, available at https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K220656.
Back to Citation226. Urban JA, Dolesh K, Martin E. A Multimodal Pain Management Protocol Including Preoperative Cryoneurolysis for Total Knee Arthroplasty to Reduce Pain, Opioid Consumption, and Length of Stay. Arthroplast Today. 2021 Jul 12;10:87-92. doi: 10.1016/j.artd.2021.06.008. PMID: 34286056; PMCID: PMC8280475.
227. Mihalko WM, Kerkhof AL, Ford MC, Crockarell JR, Harkess JW, Guyton JL. Cryoneurolysis before Total Knee Arthroplasty in Patients With Severe Osteoarthritis for Reduction of Postoperative Pain and Opioid Use in a SingleCenter Randomized Controlled Trial. J Arthroplasty. 2021 May;36(5):1590-1598. doi: 10.1016/j.arth.2020.11.013. Epub 2020 Nov 14. PMID: 33279353.
228. Ilfeld BM, Finneran JJ, Swisher MW, Said ET, Gabriel RA, Sztain JF, Khatibi B, Armani A, Trescot A, Donohue MC, Schaar A, Wallace AM. Preoperative Ultrasound-guided Percutaneous Cryoneurolysis for the Treatment of Pain after Mastectomy: A Randomized, Participant- and Observer-masked, Sham-controlled Study. Anesthesiology. 2022 Nov 1;137(5):529-542. doi: 10.1097/ALN.0000000000004334. PMID: 35929983.
229. Dasa V, Lensing G, Parsons M, Harris J, Volaufova J, Bliss R. Percutaneous freezing of sensory nerves prior to total knee arthroplasty. Knee. 2016 Jun;23(3):523-8. doi: 10.1016/j.knee.2016.01.011. Epub 2016 Feb 10. PMID: 26875052.
Back to Citation230. Exparel. FDA Package Insert. November 2023. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/022496s051lbl.pdf.
231. Omidria. FDA Package Insert. December 2017. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205388s006lbl.pdf.
232. Payment for HCPCS Code 68841, is discussed in section III.E.6. of this final rule with comment period.
233. Dextenza. FDA Package Insert. October 2021. https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/208742s007lbl.pdf.
234. Xaracoll. FDA Package Insert. August 2020. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/209511s000lbl.pdf.
235. Zynrelef. FDA Package Insert. January 2024. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/211988s013lbl.pdf.
Back to Citation236. Ketorolac tromethamine Injection. FDA Package Insert. May 2014. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/074802s038lbl.pdf.
237. On-Q Pump. FDA 510K. February 2019. https://www.accessdata.fda.gov/cdrh_docs/pdf18/K181360.pdf.
238. Ding DY, Manoli A 3rd, Galos DK, Jain S, Tejwani NC. Continuous Popliteal Sciatic Nerve Block Versus Single Injection Nerve Block for Ankle Fracture Surgery: A Prospective Randomized Comparative Trial. J Orthop Trauma. 2015;29(9):393-398. https://pubmed.ncbi.nlm.nih.gov/2616525.
239. FDA Approval Letter, July 31, 2018. https://www.accessdata.fda.gov/cdrh_docs/pdf18/K181422.pdf.
240. Ilfeld BM, Plunkett A, Vijjeswarapu AM, Hackworth R, Dhanjal S, Turan A, Cohen SP, Eisenach JC, Griffith S, Hanling S, Sessler DI, Mascha EJ, Yang D, Boggs JW, Wongsarnpigoon A, Gelfand H Percutaneous, Peripheral Nerve Stimulation (Neuromodulation) for Postoperative Pain: A Randomized, Sham-controlled Pilot Study, Anesthesiology, April 2021.
241. https://www.accessdata.fda.gov/cdrh_docs/pdf18/K182565.pdf.
242. https://www.accessdata.fda.gov/cdrh_docs/pdf20/K200697.pdf.
243. https://www.accessdata.fda.gov/cdrh_docs/pdf23/K233170.pdf.
244. O'Connor LA, Dua A, Orhurhu V, Hoepp LM, Quinn CC. Opioid Requirements After Intercostal Cryoanalgesia in Thoracic Surgery. J Surg Res. 2022; 274:232-241.
245. Maxwell CM, Weksler B, Houda J, Fernando HC. Intercostal Cryoablation During Video-Assisted Lung Resection Can Decrease Postoperative Opioid Use. Innovations 2023 18(4):352-356.
246. Jaroszewski DE, Bostoros P, Farina JM, Botros MM, Aly MR, Peterson M, Lackey J, Pulivarthi KV, Smith B, Craner R, Stearns JD. Evolution of Pain Control for Adult Pectus Excavatum Repair. Ann Thorac Surg. 2024;117(4):829-837.
247. Graves CE, Moyer J, Zobel MJ, Mora R, Smith D, O'Day M, Padilla BE. Intraoperative intercostal nerve cryoablation during the Nuss procedure reduces length of stay and opioid requirement: A randomized clinical trial. J Pediatric Surg. 2019 Nov;54(11):2250-2256.
Back to Citation248. FDA 510(k) Clearance K052221; obtained in 2005 by and issued to Sorenson Medical, Inc., which was later acquired by Summit Medical Products, Inc. Avanos acquired substantially all assets of Summit Medical Products, including this FDA 510(k) Clearance K052221, in 2019.
249. Morkos M, DeLeon A, Koeckert M, Gray Z, Liao K, Pan W, Tolpin DA. The Use of Unilateral Erector Spinae Plane Block in Minimally Invasive Cardiac Surgery. J Cardiothorac Vasc Anesth. 2023 Mar;37(3):432-436.
250. FDA 510(k) No. K220656, available at https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpmn/pmn.cfm?ID=K220656.
251. Urban JA, Dolesh K, Martin E. A Multimodal Pain Management Protocol Including Preoperative Cryoneurolysis for Total Knee Arthroplasty to Reduce Pain, Opioid Consumption, and Length of Stay. Arthroplast Today. 2021 Jul 12;10:87-92. doi: 10.1016/j.artd.2021.06.008. PMID: 34286056; PMCID: PMC8280475.
252. Mihalko WM, Kerkhof AL, Ford MC, Crockarell JR, Harkess JW, Guyton JL. Cryoneurolysis before Total Knee Arthroplasty in Patients With Severe Osteoarthritis for Reduction of Postoperative Pain and Opioid Use in a SingleCenter Randomized Controlled Trial. J Arthroplasty. 2021 May;36(5):1590-1598. doi: 10.1016/j.arth.2020.11.013. Epub 2020 Nov 14. PMID: 33279353.
253. Ilfeld BM, Finneran JJ, Swisher MW, Said ET, Gabriel RA, Sztain JF, Khatibi B, Armani A, Trescot A, Donohue MC, Schaar A, Wallace AM. Preoperative Ultrasound-guided Percutaneous Cryoneurolysis for the Treatment of Pain after Mastectomy: A Randomized, Participant- and Observer-masked, Sham-controlled Study. Anesthesiology. 2022 Nov 1;137(5):529-542. doi: 10.1097/ALN.0000000000004334. PMID: 35929983.
254. Dasa V, Lensing G, Parsons M, Harris J, Volaufova J, Bliss R. Percutaneous freezing of sensory nerves prior to total knee arthroplasty. Knee. 2016 Jun;23(3):523-8. doi: 10.1016/j.knee.2016.01.011. Epub 2016 Feb 10. PMID: 26875052.
Back to Citation255. Centers for Medicare & Medicaid Services. (2022). CMS Framework for Health Equity 2022-2032. Available at: https://www.cms.gov/files/document/cms-framework-health-equity-2022.pdf.
Back to Citation256. Polyakova M, Udalova V, Kocks G, Genadek K, Finlay K, Finkelstein AN. (2021). Racial Disparities in Excess All-Cause Mortality During The Early COVID-19 Pandemic Varied Substantially Across States. Health Affairs, 40(2), 307-316. https://doi.org/10.1377/hlthaff.2020.02142.
257. Rural Health Research Gateway. (2018). Rural Communities: Age, Income, and Health Status. Rural Health Research Recap. Available at: https://www.ruralhealthresearch.org/assets/2200-8536/rural-communities-age-income-health-status-recap.pdf.
258. Heslin KC, Hall JE. (2021). Sexual Orientation Disparities in Risk Factors for Adverse COVID-19-Related Outcomes, by Race/Ethnicity—Behavioral Risk Factor Surveillance System, United States, 2017-2019. MMWR Morbidity Mortality Weekly Report, 70(5), 149. http://dx.doi.org/10.15585/mmwr.mm7005a1.
Back to Citation259. The Physicians Foundation. (2020). Survey of America's Patients, Part Three. Available at: https://physiciansfoundation.org/wp-content/uploads/2020/10/2020-Physicians-Foundation-Survey-Part3.pdf.
260. Office of the Assistant Secretary for Planning and Evaluation. (2020). Report to Congress: Social Risk Factors and Performance Under Medicare's Value-Based Purchasing Program (Second of Two Reports). Available at: https://aspe.hhs.gov/pdf-report/second-impact-report-to-congress.
Back to Citation261. Ibid.
262. Khullar D, Schpero WL, Bond AM, Qian Y, Casalino LP. (2020). Association Between Patient Social Risk and Physician Performance Scores in the First Year of the Merit-based Incentive Payment System. JAMA, 324(10), 975-983. https://jamanetwork.com/journals/jama/fullarticle/2770410.
Back to Citation263. Office of Disease Prevention and Health Promotion. (2021). Healthy People 2020: Disparities. Available at: https://wayback.archive-it.org/5774/20220414003754/https://www.healthypeople.gov/2020/about/foundation-health-measures/Disparities.
Back to Citation264. Centers for Medicare & Medicaid Services. (2021). A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights. Available at: https://www.cms.gov/priorities/innovation/media/document/ahcm-screeningtool-companion.
Back to Citation265. Kaiser Family Foundation. (2021). Racial and Ethnic Health Inequities and Medicare. Available at: https://www.kff.org/medicare/report/racial-and-ethnic-health-inequities-and-medicare/.
266. The Physicians Foundation. (2021). Viewpoints: Social Determinants of Health. Available at: https://physiciansfoundation.org/wp-content/uploads/2019/08/The-Physicians-Foundation-SDOH-Viewpoints.pdf.
Back to Citation267. Zhang Y, Li J, Yu J, Braun RT, Casalino LP. (2021), Social Determinants of Health and Geographic Variation in Medicare per Beneficiary Spending. JAMA Network Open, 4(6), e2113212. https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2780864.
268. Khullar D, Schpero WL, Bond AM, Qian Y, Casalino LP (2020). Association Between Patient Social Risk and Physician Performance Scores in the First Year of the Merit-based Incentive Payment System. JAMA, 324(10), 975-983. https://doi.org/10.1001/jama.2020.13129.
Back to Citation269. Centers for Medicare & Medicaid Services. (2021). Paving the Way to Equity: A Progress Report. Available at: https://www.cms.gov/files/document/paving-way-equity-cms-omh-progress-report.pdf.
270. Centers for Medicare & Medicaid Services Office of Minority Health. (2021). The CMS Equity Plan for Improving Quality in Medicare. 2015-2021. Available at: https://www.cms.gov/About-CMS/Agency-Information/OMH/OMH_Dwnld-CMSEquityPlanforMedicare_090615.pdf.
Back to Citation271. Alley DE, Asomugha CN, Conway PH, Sanghavi DM. (2016). Accountable Health Communities—Addressing Social Needs through Medicare and Medicaid. New England Journal of Medicine, 374(1), 8-11. Available at: https://www.nejm.org/doi/10.1056/NEJMp1512532.
272. Centers for Disease Control and Prevention. (July 2020). CDC COVID-19 Response Health Equity Strategy: Accelerating Progress Towards Reducing COVID-19 Disparities and Achieving Health Equity. Available at: https://www.cdc.gov/covid/media/pdfs/2024/07/CDC_COVID-19HealthEquityStrategy_English.pdf.
Back to Citation273. We use the term “healthcare facility” to refer to hospital outpatient departments (HOPDs), rural emergency hospitals (REHs), and ambulatory surgical centers (ASCs) collectively.
274. Smith SA, Yount N, Sorra J. (2017). Exploring Relationships Between Hospital Patient Safety Culture and Consumer Reports Safety Scores. BMC Health Services Research, 17(1), 143. https://doi.org/10.1186/s12913-017-2078-6.
275. Bradley EH, Brewster AL, McNatt Z, et al. (2018). How Guiding Coalitions Promote Positive Culture Change in Hospitals: A Longitudinal Mixed Methods Interventional Study. BMJ Quality & Safety, 27(3), 218-225. https://qualitysafety.bmj.com/content/27/3/218.
Back to Citation276. Smith SA, Yount N, Sorra J. (2017). Exploring Relationships Between Hospital Patient Safety Culture and Consumer Reports Safety Scores. BMC Health Services Research, 17(1), 143. https://bmchealthservres.biomedcentral.com/articles/10.1186/s12913-017-2078-6.
Back to Citation277. Centers for Medicare & Medicaid Services. (2024). CMS National Quality Strategy. Centers for Medicare and Medicaid Services. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy.
Back to Citation278. We use the phrase “payment determination” for the Hospital OQR and ASCQR Programs to represent our assessment of whether the 2-percentage point reduction in payment for failing to meet program requirements is warranted. We use the phrase “program determination” for the REHQR Program to represent our assessment of compliance with program requirements for an applicable year because the REHQR Program does not include an associated payment adjustment.
Back to Citation279. Bradley EH, Brewster AL, McNatt Z, Linnander EL, Cherlin E, Fosburgh H, Ting HH, Curry LA.(2018). How Guiding Coalitions Promote Positive Culture Change in Hospitals: A Longitudinal Mixed Methods Interventional Study. BMJ Quality & Safety, 27(3), 218-225. https://pubmed.ncbi.nlm.nih.gov/29101290/.
Back to Citation280. Agency for Healthcare Research and Quality. (September 2019). Leadership Role in Improving Patient Safety. Patient Safety Network. Available at: https://psnet.ahrq.gov/primer/leadership-role-improving-safety.
281. Joint Commission on Accreditation of Healthcare Organizations. (June 2021). The essential role of leadership in developing a safety of culture. Sentinel Event Alert. (57), 1-8. https://www.jointcommission.org/-/media/tjc/newsletters/sea-57-safety-culture-and-leadership-final3.pdf.
Back to Citation282. Mate KS, Wyatt R. (2017). Health Equity Must Be a Strategic Priority. NEJM Catalyst. Available at: https://catalyst.nejm.org/doi/full/10.1056/CAT.17.0556.
Back to Citation283. Centers for Medicare & Medicaid Services. (2021). Building an Organizational Response to Health Disparities [Fact Sheet]. U.S. Department of Health and Human Services. Available at: https://www.cms.gov/About-CMS/Agency-Information/OMH/Downloads/Health-Disparities-Guide.pdf.
Back to Citation284. We note that the term “hospital” includes HOPDs and REHs for the purposes of this measure.
Back to Citation285. CEHRT refers to the certified health IT requirements established by CMS. ONC health IT certification criteria referenced in the CEHRT definition can be found at 45 CFR 170.315. Please refer to the following for more details on CEHRT requirements: https://www.cms.gov/medicare/regulations-guidance/promoting-interoperability-programs/certified-ehr-technology. Please refer to the Measure Calculation section for more details on CEHRT and the HCHE Measure.
Back to Citation286. We define the term “EHR technology” as ONC's definition for Electronic Health Record, “a real-time patient health record with access to evidence-based decision support tools that can be used to aid clinicians in decision making. The EHR can automate and streamline a clinician's workflow, ensuring that all clinical information is communicated. It can also prevent delays in response that result in gaps in care. The EHR can also support the collection of data for uses other than clinical care, such as billing, quality management, outcome reporting, and public health disease surveillance and reporting,” at https://www.healthit.gov/topic/health-it-and-health-information-exchange-basics/glossary.
Back to Citation287. Taira A. (June 2021). ASCA Survey Shows Mixed Usage of EHR among ASCs. ASC Focus. Available at: https://www.ascfocus.org/ascfocus/content/articles-content/articles/2021/digital-debut/asca-survey-shows-mixed-usage-of-ehr-among-ascs.
Back to Citation288. Partnership for Quality Measurement. (September 2023). Guidebook of Policies and Procedures for Pre-Rulemaking Measure Review and Measure Set Review. Available at: https://p4qm.org/sites/default/files/2023-09/Guidebook-of-Policies-and-Procedures-for-Pre-Rulemaking-Measure-Review-%28PRMR%29-and-Measure-Set-Review-%28MSR%29-Final_0.pdf.
Back to Citation289. Partnership for Quality Measurement. 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
290. Partnership for Quality Measurement. Pre-Rulemaking Measure Review Measures Under Consideration: 2023 Recommendations Report. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-2023-MUC-Recommendations-Report-Final.pdf.
Back to Citation291. Ibid.
Back to Citation292. Centers for Medicare & Medicaid Services. (February 2022). Summary of Technical Expert Panel (TEP) Meeting #1, November 16, 2021: Health Equity Quality Measurement, Hospital Commitment to Health Equity Measure. Available at: https://mmshub.cms.gov/sites/default/files/HealthEquityQualityMeasurementTEP1SumReport.pdf.
Back to Citation293. Centers for Medicare & Medicaid Services (January 2024). Attestation Guidance for the Hospital Commitment to Health Equity Measure (v 1.2). Available at: https://qualitynet.cms.gov/files/659c609eca7fd3001b35edab?filename=AttstGdnceHCHEMeas_v1.2.pdf.
294. Centers for Medicare & Medicaid Services (January 2024). Frequently Asked Questions Hospital Commitment to Health Equity, HIQR. Available at: https://qualitynet.cms.gov/files/659c60afd4b704001df0af51?filename=FAQ_HCHE_HIQR.pdf.
Back to Citation295. Partnership for Quality Measurement. (2023). 2023 PRMR Final MUC Recommendation Spreadsheet. Available at: https://p4qm.org/PRMR.
Back to Citation296. Centers for Medicare & Medicaid Services. (February 2022). Summary of Technical Expert Panel (TEP) Meeting # 1 November 16, 2021: Health Equity Quality Measurement Hospital Commitment to Health Equity Measure. Available at: https://mmshub.cms.gov/sites/default/files/HealthEquityQualityMeasurementTEP1SumReport.pdf.
Back to Citation297. Measure specifications are posted at https://qualitynet.cms.gov/outpatient/oqr/proposedmeasures2#tab2 for the Hospital OQR and REHQR Programs, and https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 for the ASCQR Program and may be moved to other pages on QualityNet upon publication of the final rule.
Back to Citation298. Taira A. (June 2021). ASCA Survey Shows Mixed Usage of EHR among ASCs. ASC Focus. Available at: https://www.ascfocus.org/ascfocus/content/articles-content/articles/2021/digital-debut/asca-survey-shows-mixed-usage-of-ehr-among-ascs.
Back to Citation299. Office of the National Coordinator for Health Information Technology. (2023). National Trends in Hospital and Physician Adoption of Electronic Health Records. Available at: https://www.healthit.gov/data/quickstats/national-trends-hospital-and-physician-adoption-electronic-health-records.
Back to Citation300. Centers for Medicare & Medicaid Services (January 2024). Attestation Guidance for the Hospital Commitment to Health Equity Measure (v 1.2). Available at: https://qualitynet.cms.gov/files/659c609eca7fd3001b35edab?filename=AttstGdnceHCHEMeas_v1.2.pdf.
301. Centers for Medicare & Medicaid Services (January 2024). Frequently Asked Questions Hospital Commitment to Health Equity, HIQR. Available at: https://qualitynet.cms.gov/files/659c60afd4b704001df0af51?filename=FAQ_HCHE_HIQR.pdf.
Back to Citation302. Measure specifications are posted at https://qualitynet.cms.gov/outpatient/oqr/proposedmeasures2#tab2 for the Hospital OQR and REHQR Programs, and https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 for the ASCQR Program and may be moved to other pages on QualityNet upon publication of the final rule.
Back to Citation303. CMS Framework for Health Equity 2022-2032. (n.d.). https://www.cms.gov/files/document/cms-framework-health-equity-2022.pdf.
Back to Citation304. Centers for Medicare & Medicaid Services. (February 2022). Summary of Technical Expert Panel (TEP) Meeting # 1 November 16, 2021: Health Equity Quality Measurement Hospital Commitment to Health Equity Measure. Available at: https://mmshub.cms.gov/sites/default/files/HealthEquityQualityMeasurementTEP1SumReport.pdf.
Back to Citation305. Centers for Medicare & Medicaid Services (January 2024). Attestation Guidance for the Hospital Commitment to Health Equity Measure (v 1.2). Available at: https://qualitynet.cms.gov/files/659c609eca7fd3001b35edab?filename=AttstGdnceHCHEMeas_v1.2.pdf.
306. Centers for Medicare & Medicaid Services (January 2024). Frequently Asked Questions Hospital Commitment to Health Equity, HIQR. Available at: https://qualitynet.cms.gov/files/659c60afd4b704001df0af51?filename=FAQ_HCHE_HIQR.pdf.
Back to Citation307. Measure specifications are posted at: https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 for the ASCQR Program and may be moved to other pages on QualityNet upon publication of the final rule.
Back to Citation308. Witiw, C. D., Wilson, J. R., Fehlings, M. G., & Traynelis, V. C. (2020). Ambulatory Surgical Centers: Improving quality of operative spine care? Global Spine Journal, 10 (1_suppl). https://doi.org/10.1177/2192568219849391.
309. Chatterjee, A., Amen, T. B., & Khormaee, S. (2022b). Trends in geographic disparities in access to ambulatory surgery centers in New York, 2010 to 2018. JAMA Health Forum, 3(10). https://doi.org/10.1001/jamahealthforum.2022.3608.
Back to Citation310. Geier, A., & Smith, D. (2019). The role of electronic documentation in Ambulatory Surgery Centers. AORN Journal, 109(4), 444-450. https://doi.org/10.1002/aorn.12636.
Back to Citation311. Ibid.
Back to Citation312. Ibid.
Back to Citation313. Centers for Medicare & Medicaid Services (January 2024). Attestation Guidance for the Hospital Commitment to Health Equity Measure (v 1.2). Available at: https://qualitynet.cms.gov/files/659c609eca7fd3001b35edab?filename=AttstGdnceHCHEMeas_v1.2.pdf.
314. Centers for Medicare & Medicaid Services (January 2024). Frequently Asked Questions Hospital Commitment to Health Equity, HIQR. Available at: https://qualitynet.cms.gov/files/659c60afd4b704001df0af51?filename=FAQ_HCHE_HIQR.pdf.
Back to Citation315. Measure specifications are posted at https://qualitynet.cms.gov/outpatient/oqr/proposedmeasures2#tab2 for the Hospital OQR and REHQR Programs, and https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 for the ASCQR Program and may be moved to other pages on QualityNet upon publication of the final rule.
Back to Citation316. Newitt, P. (n.d.). HOPDs vs. ASCS: 10 considerations for 2024. Becker's ASC Review. https://www.beckersasc.com/asc-coding-billing-and-collections/hopds-vs-ascs-10-considerations-for-2024.html.
Back to Citation317. PRMR final MUC recommendation spreadsheet. PRMR Final MUC Recommendation Spreadsheet | Partnership for Quality Measurement. (n.d.). https://p4qm.org/prmr-final-muc-reco.
Back to Citation318. ASCA. (September 2024). What is an ASC? Ambulatory Surgery Center Association (ASCA). Available at: https://www.ascassociation.org/asca/about-ascs/surgery-centers#:~:text=Ambulatory%20surgery%20centers%2C%20or%20ASCs,including%20diagnostic%20and%20preventive%20procedures.
Back to Citation319. Assistant Secretary for Planning and Evaluation. (November 2023). Call to Action: Addressing Health-Related Social Needs in Communities Across the Nation. Available at: https://aspe.hhs.gov/sites/default/files/documents/3e2f6140d0087435cc6832bf8cf32618/hhs-call-to-action-health-related-social-needs.pdf.
Back to Citation320. Assistant Secretary for Planning and Evaluation. (September 2022). Reflections Accompanying a Report on Addressing Social Drivers of Health: Evaluating Area-level Indices. Available at: https://aspe.hhs.gov/sites/default/files/documents/474a62378abf941f20b3eaa74ca5721c/Area-level-Indices-ASPE-Reflections.pdf.
321. American Hospital Association. (December 2020). Health Equity, Diversity & Inclusion Measures for Hospitals and Health System Dashboards. Available at: https://ifdhe.aha.org/system/files/media/file/2020/12/ifdhe_inclusion_dashboard.pdf.
Back to Citation322. Fraze TK, Brewster AL, Lewis VA, Beidler LB, Murray GF, Colla, CH. (2019). Prevalence of Screening for Food Insecurity, Housing Instability, Utility Needs, Transportation Needs, and Interpersonal Violence by US Physician Practices and Hospitals. JAMA Network Open, 2(9), e1911514. https://doi.org/10.1001/jamanetworkopen.2019.11514.
Back to Citation323. Berkowitz RL, Bui L, Shen Z, Pressman A, Moreno M, Brown S, Nilon, A Miller-Rosales, Azar KM. (2021). Evaluation of a social determinants of health screening questionnaire and workflow pilot within an adult ambulatory clinic. BMC Family Practice, 22(1), 256. https://doi.org/10.1186/s12875-021-01598-3.
324. Schickedanz A, Hamity C, Rogers A, Sharp AL, Jackson A. (2019). Clinician Experiences and Attitudes Regarding Screening for Social Determinants of Health in a Large Integrated Health System. Medical Care, 57, S197-S201. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6721844/.
Back to Citation325. Centers for Medicare & Medicaid Services. (2021). A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights. Available at: https://innovation.cms.gov/media/document/ahcm-screeningtool-companion.
326. Centers for Medicare & Medicaid Services. (2021). Accountable Health Communities Model. Accountable Health Communities Model. Available at: https://www.cms.gov/priorities/innovation/innovation-models/ahcm.
Back to Citation327. Centers for Medicare & Medicaid Services. (2023). Accountable Health Communities (AHC) Model Evaluation: Second Evaluation Report. CMS Innovation Center. Available at: https://www.cms.gov/priorities/innovation/data-and-reports/2023/ahc-second-eval-rpt.
328. Billioux A, Verlander, K, Anthony S, Alley D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). https://doi.org/10.31478/201705b.
329. Centers for Medicare & Medicaid Services. (2021). Accountable Health Communities Model. Accountable Health Communities Model. Available at: https://innovation.cms.gov/innovation-models/ahcm.
Back to Citation330. Berkowitz SA, Seligman HK, Meigs JB, Basu S. (2018). Food Insecurity, Healthcare Utilization, and High Cost: A Longitudinal Cohort Study. The American Journal of Managed Care, 24(9), 399-404. http://www.ncbi.nlm.nih.gov/pmc/articles/pmc6426124/.
331. Seligman HK, Berkowitz, SA. (2019). Aligning Programs and Policies to Support Food Security and Public Health Goals in the United States. Annual Review of Public Health, 40(1), 319-337. https://www.annualreviews.org/doi/10.1146/annurev-publhealth-040218-044132.
332. National Academies of Sciences, Engineering, and Medicine. (2006). Executive Summary: Cost-Benefit Analysis of Providing Non-Emergency Medical Transportation. Washington, DC: The National Academies Press. Available at: https://nap.nationalacademies.org/catalog/23285/executive-summary-cost-benefit-analysis-of-providing-non-emergency-medical-transportation.
333. Berkowitz SA, Seligman HK, Meigs JB, Basu S. (2018). Food Insecurity, Healthcare Utilization, and High Cost: A Longitudinal Cohort Study. The American Journal of Managed Care, 24(9), 399-404. http://www.ncbi.nlm.nih.gov/pmc/articles/pmc6426124/.
334. Dean EB, French MT, Mortensen, K. (2020). Food insecurity, health care utilization, and health care expenditures. Health Services Research, 55(S2), 883-893. Available at: https://doi.org/10.1111/1475-6773.13283.
335. Hill-Briggs, F. (2020). Social Determinants of Health and Diabetes: A Scientific Review. Diabetes Care, 44(1), 258-279. https://pubmed.ncbi.nlm.nih.gov/33139407/.
336. Henry M, de Sousa T, Roddey C, Gayen S, Bednar T, Abt Associates. (January 2021). The 2020 Annual Homeless Assessment Report (AHAR) to Congress; Part 1: Point-in-Time Estimates of Homelessness. U.S. Department of Housing and Urban Development. Available at: https://www.huduser.gov/portal/sites/default/files/pdf/2020-AHAR-Part-1.pdf.
337. Baxter A, Tweed E, Katikireddi S, Thomson H. (2019). Effects of Housing First approaches on health and well-being of adults who are homeless or at risk of homelessness: systematic review and meta-analysis of randomized controlled trials. Journal of Epidemiology and Community Health, 73; 379-387. https://doi.org/10.1136/jech-2018-210981.
338. Centers for Medicare and Medicaid Services. (2023) Addressing Transportation Barriers: A User Case in Leveraging the Value-Based Insurance Design (VBID) Model. Available at: https://www.cms.gov/priorities/innovation/innovation-models/vbid.
339. Billioux A, Verlander, K, Anthony S, Alley D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). https://doi.org/10.31478/201705b.
340. Shier G, Ginsburg M, Howell J, Volland P, Golden R. (2013). Strong Social Support Services, Such as Transportation And Help For Caregivers, Can Lead To Lower Health Care Use And Costs. Health Affairs, 32(3), 544-551. https://www.healthaffairs.org/doi/10.1377/hlthaff.2012.0170.
341. National Alliance on Mental Illness. Medicaid: Non-Emergency Medical Transportation (NEMT). Available at: https://www.nami.org/Advocacy/Policy-Priorities/Supporting-Community-Inclusion-and-Non-Discrimination/Medicaid-Non-Emergency-Medical-Transportation.
342. Baxter A, Tweed E, Katikireddi S, Thomson H. (2019). Effects of Housing First approaches on health and well-being of adults who are homeless or at risk of homelessness: systematic review and meta-analysis of randomized controlled trials. Journal of Epidemiology and Community Health, 73; 379-387. https://doi.org/10.1136/jech-2018-210981.
343. Wright BJ, Vartanian KB, Li HF, Royal N, Matson JK (2016). Formerly Homeless People Had Lower Overall Health Care Expenditures After Moving into Supportive Housing. Health Affairs, 35(1), 20-27. https://www.healthaffairs.org/doi/10.1377/hlthaff.2015.0393.
344. Billioux A, Verlander K, Anthony S, Alley D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). https://doi.org/10.31478/201705b.
345. Henry M, de Sousa T, Roddey C, Gayen S, Bednar T, Abt Associates. (January 2021). The 2020 Annual Homeless Assessment Report (AHAR) to Congress; Part 1: Point-in-Time Estimates of Homelessness. U.S. Department of Housing and Urban Development. Available at: https://www.huduser.gov/portal/sites/default/files/pdf/2020-AHAR-Part-1.pdf.
Back to Citation346. Centers for Medicare & Medicaid Services. (2021). A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights. Available at: https://innovation.cms.gov/media/document/ahcm-screeningtool-companion.
Back to Citation347. The Screening for SDOH measure is identified on the MUC List as MUC2023-156.
Back to Citation348. Partnership for Quality Measurement. 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
Back to Citation349. Centers for Medicare and Medicaid Services. The Accountable Health Communities Health-Related Social Needs Screening Tool. Available at: https://www.cms.gov/priorities/innovation/files/worksheets/ahcm-screeningtool.pdf.
Back to Citation350. Centers for Medicare and Medicaid Services. (2023). A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights. Available at: https://www.cms.gov/priorities/innovation/media/document/ahcm-screeningtool-companion.
Back to Citation351. Ibid.
352. Billioux A, Verlander, K, Anthony S, Alley D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). https://doi.org/10.31478/201705b.
Back to Citation353. Social Interventions Research & Evaluation Network. (2019). Social Needs Screening Tool Comparison Table. Available at: https://sirenetwork.ucsf.edu/tools-resources/resources/screening-tools-comparison.
354. The Social Interventions Research and Evaluation Network (SIREN) at University of California San Francisco was launched in the spring of 2016 to synthesize, disseminate, and catalyze research on SDOH and healthcare delivery.
Back to Citation355. CEHRT refers to certified health IT requirements defined by CMS for certain programs which incorporate health IT certification criteria established by the Office of the National Coordinator for Health Information Technology (ONC) at 45 CFR 170.315.
Back to Citation356. Office of the National Coordinator for Health IT (ONC). United States Core Data for Interoperability. Available at: https://www.healthit.gov/isa/united-states-core-data-interoperability-uscdi.
Back to Citation357. The term “admitted patients” appears in the measure specifications and MUC documentation and is intended to refer to a person who receives ambulatory care in these designated settings.
Back to Citation358. Centers for Medicare & Medicaid Services. (2023). Addressing Social Needs (ASN) Electronic Clinical Quality Measure (eCQM) Specifications Document for Public Comment. Available at: https://mmshub.cms.gov/sites/default/files/Yale-CORE-ASN-measure-specs-for-public-comment.pdf.
Back to Citation359. Bensken WP, Alberti PM, Koroukian SM. (2021) Health-related social needs and increased readmission rates: findings from the nationwide readmissions database. J Gen Intern Med. 2021;36:1173-1180. https://doi.org/10.1007/s11606-021-06646-3.
360. Weaver RH, Bolkan C, Robbins SL, et al. (2021). Caring beyond health care: lessons learned from a community-based partnership to reduce hospital readmission among high-risk adults. J Comm Engag Scholarship. 2021;14(1). https://doi.org/10.54656/NWMT3812.
Back to Citation361. We refer ASCs to the following resource that match patients with community resources based on geographic location: https://www.hhs.gov/guidance/sites/default/files/hhs-guidance-documents/CMLN%20Social%20Determinants%20of%20Health%20Relevance%20and%20Resources%20Guide.pdf.
Back to Citation362. Office of the National Coordinator. National Trends in Hospital and Physician Adoption of Electronic Health Records. Available at: https://www.healthit.gov/data/quickstats/national-trends-hospital-and-physician-adoption-electronic-health-records.
Back to Citation363. On July 29, 2024, notice was posted in the Federal Register that ONC would be dually titled to the Assistant Secretary for Technology Policy and Office of the National Coordinator for Health Information Technology (89 FR 60903).
Back to Citation364. Geier, A., & Smith, D. (2019). The role of electronic documentation in Ambulatory Surgery Centers. AORN Journal, 109(4), 444-450. https://doi.org/10.1002/aorn.12636.
Back to Citation365. Ibid.
Back to Citation366. Ibid.
Back to Citation367. Centers for Medicare and Medicaid Services. (2023). A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights. Available at: https://www.cms.gov/priorities/innovation/media/document/ahcm-screeningtool-companion.
Back to Citation368. Centers for Medicare and Medicaid Services. (2020). Accountable Health Communities Mode (AHC) First Evaluation Report. Available at: https://www.cms.gov/priorities/innovation/data-and-reports/2020/ahc-first-eval-rpt-perspective.
Back to Citation369. Centers for Medicare and Medicaid Services. (2021). Accountable Health Communities Health-Related Social Needs Screening Tool Citation and Notification Information. Available at: https://www.cms.gov/priorities/innovation/media/document/ahcm-screening-tool-citation.
Back to Citation370. Centers for Medicare & Medicaid Services. (2021). A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights. Available at: https://www.cms.gov/priorities/innovation/media/document/ahcm-screeningtool-companion.
Back to Citation371. Assistant Secretary for Planning and Evaluation. (November 2023). Call to Action: Addressing Health-Related Social Needs in Communities Across the Nation. Available at: https://aspe.hhs.gov/sites/default/files/documents/3e2f6140d0087435cc6832bf8cf32618/hhs-call-to-action-health-related-social-needs.pdf.
Back to Citation372. Centers for Medicare & Medicaid Services. (April 2024). Hospital Quality Reporting Program Frequently Asked Question: Social Drivers of Health (SDOH) Measures. Available at: https://www.qualityreportingcenter.com/globalassets/2024/04/iqr/17.-sdoh-measure--faqs_april-2024_vfinal508.pdf.
373. Measure specifications are posted at https://qualitynet.cms.gov/outpatient/oqr/proposedmeasures2#tab2 for the Hospital OQR and REHQR Programs, and https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 for the ASCQR Program and may be moved to other pages on QualityNet upon publication of the final rule.
Back to Citation374. Ibid.
Back to Citation375. Measure specifications are posted at https://qualitynet.cms.gov/outpatient/oqr/proposedmeasures2#tab2 for the Hospital OQR and REHQR Programs, and https://qualitynet.cms.gov/asc/ascqr/proposedmeasures2#tab2 for the ASCQR Program and may be moved to other pages on QualityNet upon publication of the final rule.
Back to Citation376. The Gravity Project. Introducing the Gravity Project. Available at: https://thegravityproject.net/.
Back to Citation377. ONC certification criteria for health IT can be found at 45 CFR 170.315. Please refer to the following for more details on CEHRT requirements: https://www.cms.gov/medicare/regulations-guidance/promoting-interoperability-programs/certified-ehr-technology. Please refer to the Measure Calculation section for more details on CEHRT and the HCHE Measure.
Back to Citation378. Centers for Medicare & Medicaid Services. (April 2024). Hospital Quality Reporting Program Frequently Asked Question: Social Drivers of Health (SDOH) Measures. Available at: https://www.qualityreportingcenter.com/globalassets/2024/04/iqr/17.-sdoh-measure--faqs_april-2024_vfinal508.pdf.
Back to Citation379. Centers for Medicare & Medicaid Services. (2024). Quality Measurement and Quality Improvement. Available at: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/MMS/Quality-Measure-and-Quality-Improvement-#:~:text=Patient%20and%20families%20use%20quality,likelihood%20of%20desired%20health%20outcomes.
Back to Citation380. Centers for Medicare & Medicaid Services. (September 2021). Quality Measures: How They Are Developed, Used, & Maintained. Available at: https://www.cms.gov/sites/default/files/2021-09/Quality-Measures-How-They-Are-Developed-Used-Maintained.pdf.
Back to Citation381. For more information on the three HIPAA rules, we refer readers to the HIPAA for Professionals site at: https://www.hhs.gov/hipaa/for-professionals/index.html.
Back to Citation382. U.S. Department of Health and Human Services. What is PHI? Available at: https://www.hhs.gov/answers/hipaa/what-is-phi/index.html.
Back to Citation383. Centers for Medicare & Medicaid Services. (2024). Quality Measurement and Quality Improvement. Available at: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/MMS/Quality-Measure-and-Quality-Improvement-#:~:text=Patient%20and%20families%20use%20quality,likelihood%20of%20desired%20health%20outcomes.
Back to Citation384. Centers for Medicare & Medicaid Services. (September 2021). Quality Measures: How They Are Developed, Used, & Maintained. Available at: https://www.cms.gov/sites/default/files/2021-09/Quality-Measures-How-They-Are-Developed-Used-Maintained.pdf.
Back to Citation385. Billioux A, Verlander, K, Anthony S, Alley D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). https://doi.org/10.31478/201705b.
Back to Citation386. The Screen Positive Rate for SDOH measure is identified on the MUC List as MUC2023-171.
Back to Citation387. Centers for Medicare & Medicaid Services. List of Measures Under Consideration for December 1, 2023. Available at: https://mmshub.cms.gov/measure-lifecycle/measure-implementation/pre-rulemaking/lists-and-reports.
388. Centers for Medicare & Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf.
Back to Citation389. Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
Back to Citation390. Geier, A., & Smith, D. (2019). The role of electronic documentation in Ambulatory Surgery Centers. AORN Journal, 109(4), 444-450. https://doi.org/10.1002/aorn.12636.
Back to Citation391. Ibid.
Back to Citation392. Ibid.
Back to Citation393. Centers for Medicare & Medicaid Services. (2023). Addressing Social Needs (ASN) Electronic Clinical Quality Measure (eCQM) Specifications Document for Public Comment. Available at: https://mmshub.cms.gov/sites/default/files/Yale-CORE-ASN-measure-specs-for-public-comment.pdf.
Back to Citation394. Kang E, Gillespie BM, Tobiano G, et al. (2018). Discharge education delivered to general surgical patients in their management of recovery post discharge: A systematic mixed studies review. Int J Nurs Stud. 87:1-13. https://doi.org/10.1016/j.ijnurstu.2018.07.004.
395. Hoek AE, Anker SCP, van Beeck EF, et al. (2020). Patient Discharge Instructions in the Emergency Department and Their Effects on Comprehension and Recall of Discharge Instructions: A Systematic Review and Meta-analysis. Ann Emerg Med. 75(3):435-444. https://doi.org/10.1016/j.annemergmed.2019.06.008.
396. Downey E, Olds DM. (2021). Comparison of Documentation on Inpatient Discharge and Ambulatory End-of-Visit Summaries. J Healthc Qual. 43(3):e43-e52. https://doi.org/10.1097/JHQ.0000000000000269.
Back to Citation397. Ibid.
Back to Citation398. We use the term “recovery information” to mean the clinical care instructions provided to patients or their caregivers after the completion of surgery or a non-surgical procedure.
Back to Citation399. DeSai C, Janowiak K, Secheli B, et al. (2021). Empowering patients: simplifying discharge instructions. BMJ Open Quality;10(3)001419. http://doi.org/10.1136/bmjoq-2021-001419.
400. Malevanchik L., Wheeler M., Gagliardi K., Karliner L., Shah S.J. (2021). Disparities After Discharge: The Association of Limited English Proficiency and Postdischarge Patient-Reported Issues, The Joint Commission Journal on Quality and Patient Safety, 47(12):775-782. https://doi.org/10.1016/j.jcjq.2021.08.013.
Back to Citation401. Erlang AS, Schjødt K, Linde JKS, Jensen AL. (2021). An observational study of older patients' experiences of involvement in discharge planning. Geriatr Nurs 42(4):855-862. http://doi.org/10.1016/j.gerinurse.2021.04.002.
Back to Citation402. Choudhry AJ, Younis M, Ray-Zack MD, et al. (2019). Enhanced readability of discharge summaries decreases provider telephone calls and patient readmissions in the posthospital setting. Surgery. 165(4):789-794. https://doi.org/10.1016/j.surg.2018.10.014.
403. Mitchell JP. (2015). Association of provider communication and discharge instructions on lower readmissions. J Healthc Qual., 37(1):33-40. https://doi.org/10.1097/01.JHQ.0000460126.88382.13.
Back to Citation404. A copy of the survey instrument is available at: https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf.
Back to Citation405. The term “discharge” appears in the measure specifications and is intended to refer to the transition of a patient from the outpatient hospital setting to home or next level of care.
Back to Citation406. Centers for Medicare & Medicaid Services. (2024). Meaningful Measures 2.0: Moving to Measure Prioritization and Modernization. Available at: https://www.cms.gov/medicare/quality/cms-national-quality-strategy/meaningful-measures-20-moving-measure-reduction-modernization#:~:text=Meaningful%20Measures%202.0%20promotes%20innovation%20and%20modernization%20of,variety%20of%20settings%2C%20intereste.
Back to Citation407. Centers for Medicare & Medicaid Services. (2024). CMS Quality Strategy. Available at: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/Value-Based-Programs/CMS-Quality-Strategy.
Back to Citation408. Partnership for Quality Measurement. (2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
409. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation410. For more information on what the Cronbach alpha score determines and how it is used, we refer readers to: Tavakol M & Dennick R. (2011). Making sense of Cronbach's alpha. Int J Med Educ. 27;2: 53-55. www.doi.org/10.5116/ijme.4dfb.8dfd.
Back to Citation411. Ibid.
Back to Citation412. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation413. Partnership for Quality Measurement. 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
414. See also https://www.cms.gov/medicare/quality/initiatives/hospital-quality-initiative/measure-methodology.
Back to Citation415. Ibid.
Back to Citation416. Examples of survey administration and collection include email, text, and patient information portals. These examples are not exhaustive. By leaving the method of survey administration and collection to the HOPD, we allow facilities the flexibility to choose the most appropriate method for their current infrastructure and patient base.
Back to Citation417. The Information Transfer PRO-PM is identified on the MUC List as MUC2023-17.
Back to Citation418. Partnership for Quality Measurement. (2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
Back to Citation419. Ibid.
Back to Citation420. Centers for Medicare and Medicaid Services. Schedule of OAS CAHPs Contact Attempts by Survey Mode. Available at: https://oascahps.org/ScheduleOASCAHPSContactAttempts_7-20-21.pdf.
Back to Citation421. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation422. Engel KG, Buckley BA, Forth VE, et al. (2012). Patient understanding of emergency department discharge instructions: where are knowledge deficits greatest? Acad Emerg Med. 19(9):E1035-1044.
423. Engel KG, Heisler M, Smith DM, Robinson CH, Forman JH, Ubel PA. (2009). Patient comprehension of emergency department care and instructions: are patients aware of when they do not understand? Ann Emerg Med. 53(4):454-461 e415.
424. Erlang AS, Schjodt K, Linde JKS, Jensen AL. (2021). An observational study of older patients' experiences of involvement in discharge planning. Geriatr Nurs. 42(4):855-862.
425. Lin MJ, Tirosh AG, Landry A. (2015). Examining patient comprehension of emergency department discharge instructions: Who says they understand when they do not? Intern Emerg Med. 10(8):993-1002.
426. Makaryus AN, Friedman EA. (2005). Patients' understanding of their treatment plans and diagnosis at discharge. Mayo Clin Proc. 80(8):991-994.
427. Hastings S, Stechuchak K, Oddone E, et al. (2012). Older veterans and emergency department discharge information. BMJ Qual Saf. 21(10):835-842.
Back to Citation428. Clarke C, Friedman SM, Shi K, Arenovich T, Monzon J, Culligan C. (2005). Emergency department discharge instructions comprehension and compliance study. CJEM. 7(1):5-11.
429. Henderson A, Zernike W. A study of the impact of discharge information for surgical patients. (2001). J Adv Nurs. 35(3):435-441.
430. Karliner LS, Auerbach A, Napoles A, Schillinger D, Nickleach D, Perez-Stable EJ. (2012). Language barriers and understanding of hospital discharge instructions. Med Care. 50(4):283-289.
431. Makaryus AN, Friedman EA. (2005). Patients' understanding of their treatment plans and diagnosis at discharge. Mayo Clin PRac. 80(8):991-994.
Back to Citation432. Albrecht JS, Gruber-Baldini AL, Hirshon JM, et al. (2014). Hospital discharge instructions: comprehension and compliance among older adults. J Gen Intern Med. 29(11):1491-1498.
433. Coleman EA, Chugh A, Williams MV, et al. (2013). Understanding and execution of discharge instructions. AM J Med Qual. 28(5):383-391.
434. Flacker J, Park W. Sims A. (2007). Hospital discharge information and older patients; do they get what they need? J Hosp Med. 2(5):291-296.
Back to Citation435. Hastings SN, Barrett A, Weinberger M, et al. (2011). Older patients' understanding of emergency department discharge information and its relationship with adverse outcomes. J Patient Saf. 7(1):19-25.
Back to Citation436. Stull, D, Leidy, N, Parasuraman, B, et al. (2009). Optimal recall periods for patient-reported outcomes: Challenges and potential solutions. Current medical research and opinion. 25. 929-42. www.doi.org/10.1185/03007990902774765.
437. Peasgood T, Caruana JM, Mukuria C. (2023). Systematic Review of the Effect of a One-Day Versus Seven-Day Recall Duration on Patient Reported Outcome Measures (PROMs). Patient. 16(3):201-221. www.doi.org/10.1007/s40271-022-00611-w.
Back to Citation438. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation439. Surveys could be completed by patient or a caregiver.
440. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation441. The specifications for the newly finalized OQR Program measures can be found at https://qualitynet.cms.gov/outpatient.
Back to Citation442. Saraswathula A, Merck SJ, Bai G, et al. (2023). The Volume and Cost of Quality Metric Reporting. JAMA. 329(21):1840-1847. www.doi.org/10.1001/jama.2023.7271.
Back to Citation443. Centers for Medicare & Medicaid Services. (July 2021). Summary of Technical Expert Panel (TEP) Meeting, April 23, 2021: Patient Receipt of Key Information Following Outpatient Procedure Patient-Reported Outcome-Based Performance Measure (PRO-PM). Available at: https://www.cms.gov/files/document/patient-receipt-key-info-tep-summary-072621.pdf.
Back to Citation444. Ibid.
Back to Citation445. Centers for Medicare & Medicaid Services. (2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (PRO-PM) Version 1.0 Methodology Report. Available at: https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf.
Back to Citation446. Partnership for Quality Measurement. (September 2023). Guidebook of Policies and Procedures for Pre-Rulemaking Measure Review and Measure Set Review. Available at: https://p4qm.org/sites/default/files/2023-09/Guidebook-of-Policies-and-Procedures-for-Pre-Rulemaking-Measure-Review-%28PRMR%29-and-Measure-SetReview-%28MSR%29-Final_0.pdf.
Back to Citation447. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation448. Ahmad, H., & Halim, H. (2017). Determining Sample Size for Research Activities. Selangor Business Review, 2(1), 20-34. Retrieved from https://sbr.journals.unisel.edu.my/ojs/index.php/sbr/article/view/12.
449. Voorhis, C. & Morgan, B. (2007). Understanding Power and Rules of Thumb for Determining Sample Size. Tutorials in Quantitative Methods for Psychology. 3(2), 43-50. https://doi.org/10.20982/tqmp.03.2.p043.
Back to Citation450. Kang E, Gillespie BM, Tobiano G, et al. (2018). Discharge education delivered to general surgical patients in their management of recovery post discharge: A systematic mixed studies review. Int J Nurs Stud. 87:1-13. https://doi.org/10.1016/j.ijnurstu.2018.07.004.
451. Hoek AE, Anker SCP, van Beeck EF, et al. (2020). Patient Discharge Instructions in the Emergency Department and Their Effects on Comprehension and Recall of Discharge Instructions: A Systematic Review and Metaanalysis. Ann Emerg Med. 75(3):435-444. https://doi.org/10.1016/j.annemergmed.2019.06.008.
452. Downey E, Olds DM. (2021). Comparison of Documentation on Inpatient Discharge and Ambulatory End-of-Visit Summaries. J Healthc Qual. 43(3): e43-e52. https://doi.org/10.1097/JHQ.0000000000000269.
Back to Citation453. DeSai C, Janowiak K, Secheli B, et al. (2021). Empowering patients: simplifying discharge instructions. BMJ Open Quality;10(3)001419. http://doi.org/10.1136/bmjoq-2021-001419.
454. Malevanchik L., Wheeler M., Gagliardi K., Karliner L., Shah S.J. (2021). Disparities After Discharge: The Association of Limited English Proficiency and Postdischarge Patient-Reported Issues, The Issues, The Joint Commission Journal on Quality and Patient Safety, 47(12):775-782. https://doi.org/10.1016/j.jcjq.2021.08.013.
Back to Citation455. Reyes, G. (2016). Understanding non response rates: insights from 600,000 opinion surveys. Available at: https://thedocs.worldbank.org/en/doc/708511466183857404-0050022016/original/paperreyes.pdf.
Back to Citation456. Centers for Medicare & Medicaid Services. (2024). Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (PRO-PM) Version 1.0 Methodology Report. Available at: https://www.cms.gov/files/document/patient-understanding-key-information-related-recovery-after-facility-based-outpatient-procedure-or.pdf.
Back to Citation457. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/4210.
Back to Citation458. Partnership for Quality Measurement. 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf.
459. See also https://www.cms.gov/medicare/quality/initiatives/hospital-quality-initiative/measure-methodology.
Back to Citation460. Ibid.
Back to Citation461. Partnership for Quality Measurement. (2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-MeetingSummary-Final.pdf.
Back to Citation462. Flug JA and Lind KE. (2017). Public Reporting of MRI of the Lumbar Spine for Low Back Pain and Changes in Clinical Documentation, Journal of the American College of Radiology (14)12: 1545-1551. https://doi.org/10.1016/j.jacr.2017.07.012.
Back to Citation463. Blackmore CC. (2019). The Relationship Between Medicare Outpatient Efficiency Measure OP8 and Lumbar MRI Utilization, Journal of the American College of Radiology 16(3): 276-281. https://doi.org/10.1016/j.jacr.2018.10.026.
464. Lind KE and Flug JA. (2019). Sociodemographic Variation in the Use of Conservative Therapy Before MRI of the Lumbar Spine for Low Back Pain in the Era of Public Reporting, Journal of the American College of Radiology 16(4): 560-569. https://doi.org/10.1016/j.jacr.2018.12.047.
465. Martin BI and Jarvik JG. (2015). The Medicare Outpatient Imaging Efficiency Measure for Low Back Pain (“OP-8”), Radiology 276(1). https://doi.org/10.1148/radiol.2015150648.
Back to Citation466. Centers for Medicare & Medicaid Services, Public Comment Summary Report. Available at: https://www.cms.gov/files/document/lumbar-spine-imaging-low-back-pain-public-comment-summary-report.pdf.
Back to Citation467. Blackmore CC. (2019). The Relationship Between Medicare Outpatient Efficiency Measure OP8 and Lumbar MRI Utilization, Journal of the American College of Radiology 16(3): 276-281. https://doi.org/10.1016/ j.jacr.2018.10.026.
468. Lind KE and Flug JA. (2019). Sociodemographic Variation in the Use of Conservative Therapy Before MRI of the Lumbar Spine for Low Back Pain in the Era of Public Reporting, Journal of the American College of Radiology 16(4): 560-569. https://doi.org/10.1016/j.jacr.2018.12.047.
469. Martin BI and Jarvik JG. (2015). The Medicare Outpatient Imaging Efficiency Measure for Low Back Pain (“OP-8”), Radiology 276(1). https://doi.org/10.1148/radiol. 2015150648.
Back to Citation470. Flug JA and Lind KE. (2017). Public Reporting of MRI of the Lumbar Spine for Low Back Pain and Changes in Clinical Documentation, Journal of the American College of Radiology (14)12: 1545-1551. https://doi.org/10.1016/j.jacr.2017.07.012.
Back to Citation471. Ibid.
Back to Citation472. Centers for Medicare and Medicaid Services. Provider Data Catalog. Available at: https://data.cms.gov/provider-data/dataset/di9i-zzrc.
Back to Citation473. Revisions to the CEHRT definition are intended to incorporate ONC's approach of discontinuing references to yearly editions. For additional background, we refer readers to HTI-1 proposed rule (88 FR 23759).
Back to Citation474. On July 29, 2024, notice was posted in the Federal Register that ONC would be dually titled to the Assistant Secretary for Technology Policy and Office of the National Coordinator for Health Information Technology (89 FR 60903).
Back to Citation475. Ahmad, H., & Halim, H. (2017). Determining Sample Size for Research Activities. Selangor Business Review, 2(1), 20-34. Retrieved from https://sbr.journals.unisel.edu.my/ojs/index.php/sbr/article/view/12.
476. Voorhis, C., & Morgan, B. (2007). Understanding Power and Rules of Thumb for Determining Sample Size. Tutorials in Quantitative Methods for Psychology. 3(2), 43-50. www.doi.org/10.20982/tqmp.03.2.p043.
Back to Citation477. Chartis. Healthcare's uneven financial performance: Large hospitals bounce back while rural facilities face ongoing vulnerability concerns. Available at: https://www.chartis.com/insights/healthcares-uneven-financial-performance-large-hospitals-bounce-back-while-rural.
478. Chartis. Unrelenting Pressure Pushes Rural Safety Net Crisis into Uncharted Territory. Available at: https://www.chartis.com/sites/default/files/documents/chartis_rural_study_pressure_pushes_rural_safety_net_crisis_into_uncharted_territory_feb_15_2024_fnl.pdf.
479. Sherry TB, Damberg CL, DeYoreo M, et al. (2022). Is Bigger Better?: A Closer Look at Small Health Systems in the United States. Med Care. 60(7):504-511. www.doi.org/10.1097/MLR.0000000000001727.
Back to Citation480. Gardner, R.M., N.A. Friedman, M. Carlson, T.S. Bradham and T.W. Barrett. Impact of revised triage to improve throughput in an ED with limited traditional fast track population. Am J Emerg Med., 2017,36(1), 124-127.
Back to Citation481. Kelen GD, Wolfe R, D-Onofrio G, Mills AM, Diercks D, Stern SA, Wadman MC, Sokolove PE. Emergency Department Crowding: The Canary in the Health Care System. NEJM Catalyst. 2021; 5(2).
Back to Citation482. Ibid.
Back to Citation483. Nyce, A, Gandhi, S, Freeze, B, Bosire, J, Ricca, T, Kupersmith, E, Mazzarelli, A, Rachoin, J-S. Association of Emergency Department Waiting Times With Patient Experience in Admitted and Discharged Patients. 2021. J Pat Exp 8:1-7. https://doi.org/10.1177/23743735211011404.
Back to Citation484. Glickman SW, Boulding W, Manary M, Staelin R, Roe MT, Wolosin RJ. et al. Patient satisfaction and its relationship with clinical quality and inpatient mortality in acute myocardial infarction. Circ Cardiovasc Qual Outcomes. 2010; 3:188-95. Available at https://www.ahajournals.org/doi/10.1161/CIRCOUTCOMES.109.900597?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200pubmed.
Back to Citation485. Boulding W, Glickman SW, Manary MP, Schulman KA, Staelin R. Relationship between patient satisfaction with inpatient care and hospital readmission within 30 days. Am J Manag Care. 2011;17:41-8. Available at https://www.ajmc.com/view/ajmc_11jan_boulding_41to48.
Back to Citation486. Partnership for Quality Measurement. Submission Tool and Repository Measure Database. https://p4qm.org/measures/0496.
Back to Citation487. Nyce A, Gandhi S, Freeze B, et al. (2021). Association of Emergency Department Waiting Times With Patient Experience in Admitted and Discharged Patients. Journal of Patient Experience. 2021;8. www.doi.org/10.1177/23743735211011404.
488. Bucci S, de Belvis AG, Marventano S, et al. (2016). Emergency department crowding and hospital bed shortage: Is Lean a smart answer? A systematic review. Eur Rev Med Pharmacol Sci, 20(20), 4209-4219. https://www.europeanreview.org/article/11589.
489. Chang AM, Lin A, Fu R, et al. (2017). Associations of Emergency Department Length of Stay With Publicly Reported Quality-of-care Measures. Acad Emerg Med. 24(2):246-250. www.doi.org/10.1111/acem.13102.
490. Melton JD, Blind F, Hall AB, et al. (2016). Impact of a Hospitalwide Quality Improvement Initiative on Emergency Department Throughput and Crowding Measures. The Joint Commission Journal on Quality and Patient Safety. 42(12); 533-542. www.doi.org/10.1016/S1553-7250(16)30104-0.
Back to Citation491. NQF. Measure Worksheet. https://www.qualityforum.org/Projects/c-d/Cost_and_Efficiency/Measure_Worksheet_0496.aspx.
492. Mullins PM, Pines JM. National ED crowding and hospital quality: Results from the 2013 Hospital Compare data. Am J Emerg Med 2014; 32(6): 634-639. doi: 10.1016/j.ajem.2014.02.008.
493. Zocchi MS, McClelland MS, & Pines JM. Increasing Throughput: Results from a 42-Hospital Collaborative to Improve Emergency Department Flow. Jt Comm J Qual Patient Saf. 2015 Dec;41(12):532-42. doi: 10.1016/s1553-7250(15)41070-0.
494. Gardner RM, Friedman NA, Carlson M, Bradham TS, Barrett TW. Impact of revised triage to improve throughput in an ED with limited traditional fast track population. Am J Emerg Med. 2018 Jan;36(1):124-127. doi: 10.1016/j.ajem.2017.10.016.
495. Chang AM, Lin A, Fu R, McConnell KJ, Sun B. Associations of Emergency Department Length of Stay With Publicly Reported Quality-of-care Measures. Acad Emerg Med. 2017 Feb;24(2):246-250. doi: 10.1111/acem.13102.
Back to Citation496. We use the phrase “Program Determination” for the REHQR Program to represent our assessment of compliance with program requirements for an applicable year because the REHQR Program does not include an associated payment adjustment.
Back to Citation497. See section 1861(kkk)(7)(C)(iii) of the Act.
Back to Citation498. CMS, What is the CMS National Quality Strategy?. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy.
Back to Citation499. The Hospital Quality Reporting (HQR) system (formerly referred to as the QualityNet Secure Portal) is the only CMS-approved website for secure communications and health care quality data exchange to and within various CMS quality reporting programs. For more information regarding the HQR system, we refer readers to the CMS eCQI Resource Center ( https://ecqi.healthit.gov/tool/hospital-quality-reporting-hqr-system). In addition, we refer readers to the CY 2024 OPPS/ASC final rule (88 FR 82074 through 82075) for a discussion of data submission requirements for chart-abstracted measures under the REHQR Program, which includes submission of measure data via the HQR system.
Back to Citation500. In the CY 2025 OPPS/ASC proposed rule (89 FR 59471), we inadvertently noted that there were currently four specialty-specific, claims-based measures. As noted in Table 173 below, we inadvertently categorized the Endoscopy/Polyp Surveillance: Appropriate Follow-Up Interval for Normal Colonoscopy in Average Risk Patients measure as a claims-based measure and, on this basis, incorrectly attributed the number of claims-based, specialty-specific measures and non-claims-based, specialty-specific measures. We have corrected this error here in this final rule.
Back to Citation501. In the CY 2025 OPPS/ASC proposed rule (89 FR 59471), we inadvertently noted that there were currently four non-claims-based specialty-specific measures. As noted in Table 173 below, we inadvertently categorized the Endoscopy/Polyp Surveillance: Appropriate Follow-Up Interval for Normal Colonoscopy in Average Risk Patients measure as a claims-based measure and, on this basis, incorrectly attributed the number of claims-based, specialty-specific measures and non-claims-based, specialty-specific measures. We have corrected this error here in this final rule.
Back to Citation502. Public Law 100-203, enacted December 22, 1987, 100 Stat. 1330, 1330-147, https://www.congress.gov/100/statute/STATUTE-101/STATUTE-101-Pg1330.pdf.
503. This document contains links to non-United States Government websites. We are providing these links because they contain additional information relevant to the topic(s) discussed in this document or that otherwise may be useful to the reader. We cannot attest to the accuracy of information provided on the cited third-party websites or any other linked third-party site. We are providing these links for reference only; linking to a non-United States Government website does not constitute an endorsement by CMS, HHS, or any of their employees of the sponsors or the information and/or any products presented on the website. Also, please be aware that the privacy protections generally provided by United States Government websites do not apply to third-party sites.
Back to Citation504. An outpatient is defined at 42 CFR 440.2 as a patient of an organized medical facility, or distinct part of that facility who is expected by the facility to receive and who does receive professional services for less than a 24-hour period regardless of the hour of admission, whether or not a bed is used, or whether or not the patient remains in the facility past midnight.
Back to Citation505. U.S. Department of Health and Human Services, Health Care Financing Administration (HCFA), The State Medicaid Manual, Manual, (Baltimore, MD, 1985), Section 4320, https://www.cms.gov/regulations-and-guidance/guidance/manuals/paper-based-manuals-items/cms021927.
Back to Citation506. Medicare and Medicaid Programs; OBRA '87 Conforming Amendments, 56 FR 8832, 8835 (March 1, 1991) (hereinafter referred to as the 1991 rule). https://archives.federalregister.gov/issue_slice/1991/3/1/8829-8854.pdf.
Back to Citation507. CMS, Federal Funding for Services “Received Through” an IHS/Tribal Facility and Furnished to Medicaid-Eligible American Indians and Alaska Natives, State Health Official Letter (SHO) #16-002, (Baltimore, MD, 2016), https://www.medicaid.gov/sites/default/files/Federal-Policy-Guidance/Downloads/sho022616.pdf.
508. CMS, Frequently-Asked Questions (FAQs) Federal Funding for Services “Received Through” an IHS/Tribal Facility and Furnished to Medicaid Eligible American Indians and Alaska Natives (SHO #16-002), FAQ, (Baltimore, MD, 2017), https://www.medicaid.gov/media/40241.
Back to Citation509. We note that the Consolidated Appropriations Act, 2024, Division G, Title I, Section 209 (Pub. L. 118-42) amended section 1905 of the Act to establish a certified community behavioral health clinic (CCBHC) services benefit effective March 9, 2024. The CCBHC services benefit is distinct from the clinic services benefit and there is no four walls requirement for the CCBHC services benefit under Federal Medicaid law.
Back to Citation510. E.O. 14009, 86 FR 7793 (Jan. 28, 2021). See, https://www.Federalregister.gov/documents/2021/02/02/2021-02252/strengthening-medicaid-and-the-affordable-care-act.
Back to Citation511. E.O. 14070, 87 FR 20689 (Apr. 8, 2021). See, https://www.Federalregister.gov/documents/2022/04/08/2022-07716/continuing-to-strengthen-americans-access-to-affordable-quality-health-coverage.
Back to Citation512. E.O. 13175, 65 FR 67249 (Nov. 6, 2000). See, https://www.Federalregister.gov/documents/2000/11/09/00-29003/consultation-and-coordination-with-indian-tribal-governments.
Back to Citation513. President Joseph R. Biden to Heads of Executive Departments and Agencies, memorandum, “Tribal Consultation and Strengthening Nation-to-Nation Relationships,” January 26, 2021, Presidential Actions, https://www.whitehouse.gov/briefing-room/presidential-actions/2021/01/26/memorandum-on-tribal-consultation-and-strengthening-nation-to-nation-relationships/.
Back to Citation514. CMS, “Tribal Consultation,” CMS.gov, September 6, 2023, https://www.cms.gov/training-education/partner-outreach-resources/american-indian-alaska-native/tribal-consultation.
515. CMS, Tribal Consultation Policy, Policy (Washington, DC, 2015), https://www.cms.gov/outreach-and-education/american-indian-alaska-native/aian/downloads/cmstribalconsultationpolicy2015.pdf.
Back to Citation516. IHS, Indian Health Service Health Equity Report, Fact Sheet, (2024), https://www.ihs.gov/sites/newsroom/themes/responsive2017/display_objects/documents/factsheets/IHS_Health_Equity_Report_FactSheet_2024.pdf.
Back to Citation517. HCFA and IHS, “Memorandum of Agreement Between the Indian Health Services and the Health Care Financing Administration,” Memorandum of Agreement, December 19, 1996, https://www.cms.gov/Outreach-and-Education/American-Indian-Alaska-Native/AIAN/LTSS-TA-Center/pdf/memorandum-of-agreement.pdf.
Back to Citation518. Although Urban Indian Organizations that operate under Title V of the Indian Health Care Improvement Act are also part of the IHS delivery system, for purposes of our discussions of the proposed amendments to § 440.90, the terms IHS/Tribal facility and IHS/Tribal clinic do not include a facility operated by an Urban Indian Organization.
Back to Citation519. HCFA and IHS, Memorandum of Agreement.
Back to Citation520. CMS, Mental Health and Substance Use Disorder Action Plan, (2023), Action Plan, https://www.medicaid.gov/medicaid/benefits/downloads/cmcs-mntl-helth-substnce-disrdr-actn-plan.pdf.
Back to Citation521. Guth, Madeline, Heather Saunders, Lauren Niles, Angela Bergefurd, Kathleen Gifford, and Roxanne Kennedy Published. 2023. “How Do States Deliver, Administer, and Integrate Behavioral Health Care? Findings from a Survey of State Medicaid Programs.” KFF. May 25, 2023. https://www.kff.org/mental-health/issue-brief/how-do-states-deliver-administer-and-integrate-behavioral-health-care-findings-from-a-survey-of-state-medicaid-programs/.
Back to Citation522. CMS, CMS Framework for Advancing Health Care in Rural, Tribal, and Geographically Isolated Communities, (2022), Framework, https://www.cms.gov/files/document/cms-geographic-framework.pdf.
Back to Citation523. Medicaid and CHIP Payment and Access Commission (MACPAC), Medicaid and Rural Health, Issue Brief, (Washington, DC, 2021), 1, https://www.macpac.gov/wp-content/uploads/2021/04/Medicaid-and-Rural-Health.pdf.
Back to Citation524. IHS establishes the AIRs under the authority in sections 321(a) and 322(b) of the Public Health Service Act (42 U.S.C. 248 and 249(b)), Public Law 83-568 (42 U.S.C. 2001(a)), and the Indian Health Care Improvement Act (25 U.S.C. 1601 et seq.). IHS calculates AIRs on an annual basis and the rates are then published in the Federal Register . The AIRs are based on annual cost report analysis prepared by IHS's contractor. IHS reviews the cost report analysis and upon completion of the review, IHS submits recommended rates to the Office of Management and Budget (OMB) for final approval through HHS and CMS. Upon approval by OMB, the approved rates are published in the Federal Register . See https://www.ihs.gov/BusinessOffice/reimbursement-rates/. Calendar year 2024 rates and additional information can be found in the Federal Register published December 19, 2023 (88 FR 87789): https://www.Federalregister.gov/documents/2023/12/19/2023-27815/reimbursement-rates-for-calendar-year-2024.
Back to Citation525. CMS, Further Extension of Grace Period Related to the “Four Walls” Requirement under 42 CFR 440.90 for Indian Health Service and Tribal Facilities to February 11, 2025, CMCS Informational Bulletin, (Baltimore, MD, 2023), https://www.medicaid.gov/sites/default/files/2023-09/cib090823.pdf.
Back to Citation526. H.R. Rep. No. 100-391, at 523 (1987), reprinted in 1987 U.S.C.C.A.N. 2313-1, 2313-343.
Back to Citation527. U.S. Department of Housing and Urban Development (HUD), HUD 2022 Continuum of Care Homeless Assistance Programs Homeless Populations and Subpopulations, Summary Report, (Washington, DC, 2022), https://files.hudexchange.info/reports/published/CoC_PopSub_NatlTerrDC_2022.pdf.
Back to Citation528. Yale University, “Barrier to Care,” Hypertension Awareness & Prevention Program at Yale, accessed March 15, 2024, https://happy.sites.yale.edu/barriers-care.
529. Murphy, Erin Roark. 2019. “Transportation and Homelessness: A Systematic Review.” Journal of Social Distress and the Homeless 28(2): 1-10. https://doi.org/10.1080/10530789.2019.1582202.
Back to Citation530. Becker, Jenna N., and Karen J. Foli. 2021. “Health‐Seeking Behaviours in the Homeless Population: A Concept Analysis.” Health & Social Care in the Community 30(2). https://doi.org/10.1111/hsc.13499.
Back to Citation531. Baggett, Travis P., James J. O'Connell, Daniel E. Singer, and Nancy A. Rigotti. 2010. “The Unmet Health Care Needs of Homeless Adults: A National Study.” American Journal of Public Health 100(7): 1326-33. https://doi.org/10.2105/ajph.2009.180109.
532. Lin, Wen-Chieh, Monica Bharel, Jianying Zhang, Elizabeth O'Connell, and Robin E. Clark. 2015. “Frequent Emergency Department Visits and Hospitalizations among Homeless People with Medicaid: Implications for Medicaid Expansion.” American Journal of Public Health 105(S5): S716-22. https://doi.org/10.2105/ajph.2015.302693.
Back to Citation533. Logani, Ilina, Bruce Meyer, and Angela Wyse. 2023. The Mortality of the US Homeless Population. BF Becker Friedman Institute for Economics at UChicago. The University of Chicago. March 27, 2023. https://bfi.uchicago.edu/insight/research-summary/the-mortality-of-the-us-homeless-population/.
Back to Citation534. Candon, Molly, Stephen Zuckerman, Douglas Wissoker, Brendan Saloner, Genevieve M. Kenney, Karin Rhodes, and Daniel Polsky. 2018. “Declining Medicaid Fees and Primary Care Appointment Availability for New Medicaid Patients.” JAMA Internal Medicine 178 (1): 145. https://doi.org/10.1001/jamainternmed.2017.6302.
535. Holgash, Kayla, and Martha Heberlein. 2019. Physician Acceptance of New Medicaid Patients: What Matters and What Doesn't. Health Affairs Blog (blog). April 19, 2019. https://www.healthaffairs.org/content/forefront/physician-acceptance-new-medicaid-patients-matters-and-doesn-t.
Back to Citation536. Klein, Sarah, Martha Hostetter, and Douglas McCarthy. 2017. An Overview of Home-Based Primary Care: Learning from the Field. The Commonwealth Fund. The Commonwealth Fund. June 7, 2017. https://www.commonwealthfund.org/publications/issue-briefs/2017/jun/overview-home-based-primary-care-learning-field.
Back to Citation537. HCFA and IHS, Memorandum of Agreement.
Back to Citation538. Tipps, Robin T., Gregory T. Buzzard, and John A. McDougall. 2018. “The Opioid Epidemic in Indian Country.” The Journal of Law, Medicine & Ethics 46 (2): 422-36. https://doi.org/10.1177/1073110518782950.
Back to Citation539. Centers for Disease Control and Prevention (CDC), “Drug Overdose Prevention in Tribal Communities,” CDC, May 2, 2024, https://www.cdc.gov/overdose-prevention/health-equity/tribal-communities.html?CDC_AAref_Val=https://www.cdc.gov/drugoverdose/health-equity/tribal.html.
Back to Citation540. Congressional Research Service (CRS), Highways and Highway Safety on Indian Lands, CRS Report, (Washington, DC, 2016), https://www.everycrsreport.com/files/20160202_R44359_38af583fdef681edc7b5d4daeeeb5bc506a4f919.pdf.
Back to Citation541. Id.
542. CRS, Rural Highways, CRS Report, (Washington, DC, 2018), https://crsreports.congress.gov/product/pdf/R/R45250.
Back to Citation543. Government Accountability Office (GAO), Indian Health Service: Health Care Services Are Not Always Available to Native Americans, GAO-05-789, (Washington, DC, 2005), https://www.gao.gov/assets/gao-05-789.pdf.
Back to Citation544. CRS, Tribal Highway and Public Transportation Programs, In Focus, (Washington, DC, 2022), https://crsreports.congress.gov/product/pdf/IF/IF12129.
Back to Citation545. CDC, National Center for Health Statistics (NCHS), Lack of Reliable Transportation for Daily Living Among Adults: United States, 2022, by Amanda E. Ng, Dzifa Adjaye-Gbewonyo, and James Dahlhamer, NCHS Data Brief No. 490, (Hyattsville, MD, 2024), https://dx.doi.org/10.15620/cdc:135611.
Back to Citation546. U.S. Commission Civil Rights, Broken Promises: Continuing Federal Funding Shortfall for Native Americans, Briefing Report, (Washington, DC, 2018), https://www.usccr.gov/files/pubs/2018/12-20-Broken-Promises.pdf.
Back to Citation547. MACPAC, Medicaid's Role in Health Care for American Indians and Alaska Natives, Issue Brief, (Washington, DC, 2021), https://www.macpac.gov/wp-content/uploads/2021/02/Medicaids-Role-in-Health-Care-for-American-Indians-and-Alaska-Natives.pdf.
Back to Citation548. CDC, NCHS, Provisional Life Expectancy Estimates for 2021, by Elizabeth Arias, Betzaida Tejada-Vera, Kenneth D. Kochanek, and Farida B. Ahmad, Vital Statistics Rapid Release Report No. 23, (Hyattsville, MD, 2022), https://dx.doi.org/10.15620/cdc:118999.
Back to Citation549. IHS, “Disparities,” Indian Health Services, October 2019, https://www.ihs.gov/newsroom/factsheets/disparities/.
Back to Citation550. CMS, Biden-Harris Administration Takes Groundbreaking Action to Expand Health Care Access by Covering Traditional Health Care Practices, Press Release, (Baltimore, MD, 2024), https://www.cms.gov/newsroom/press-releases/biden-harris-administration-takes-groundbreaking-action-expand-health-care-access-covering.
Back to Citation551. Substance Abuse and Mental Health Services Administration (SAMHSA), Key substance use and mental health indicators in the United States: Results from the 2022 National Survey on Drug Use and Health, HHS Publication No. PEP23-07-01-006, NSDUH Series H-58, (Rockville, MD, 2023), https://www.samhsa.gov/data/report/2022-nsduh-annual-national-report.
Back to Citation552. Priester, Mary Ann, Teri Browne, Aidyn Iachini, Stephanie Clone, Dana DeHart, and Kristen D. Seay. 2016. “Treatment Access Barriers and Disparities among Individuals with Co-Occurring Mental Health and Substance Use Disorders: An Integrative Literature Review.” Journal of Substance Abuse Treatment 61 (61): 47-59. https://doi.org/10.1016/j.jsat.2015.09.006.
Back to Citation553. Wallace, Richard J, Paul Hughes-Cromwick, Hillary J Mull, and Snehamay Khasnabis. 2005. “Access to Health Care and Nonemergency Medical Transportation: Two Missing Links.” Transportation Research Record 1924 (January): 76-84. https://doi.org/10.3141/1924-10.
Back to Citation554. Knaak, Stephanie, Ed Mantler, and Andrew Szeto. 2017. “Mental Illness-Related Stigma in Healthcare.” Healthcare Management Forum 30 (2): 111-16. https://doi.org/10.1177/0840470416679413.
Back to Citation555. Iturralde, Esti, Natalie Slama, Andrea H. Kline-Simon, Kelly C. Young-Wolff, Don Mordecai, and Stacy A. Sterling. 2021. “Premature Mortality Associated with Severe Mental Illness or Substance Use Disorder in an Integrated Health Care System.” General Hospital Psychiatry 68 (January): 1-6. https://doi.org/10.1016/j.genhosppsych.2020.11.002.
Back to Citation556. Health Resources & Services Administration (HRSA), “What is Shortage Designation?” HRSA Health Workforce, June 2023, https://bhw.hrsa.gov/workforce-shortage-areas/shortage-designation.
Back to Citation557. HRSA, Designated Health Professional Shortage Areas Statistics, Designated HPSA Quarterly Summary, (Rockville, MD, 2024), https://data.hrsa.gov/Default/GenerateHPSAQuarterlyReport.
Back to Citation558. CDC, NCHS, “Drug Overdose Mortality by State,” CDC, March 1, 2022, https://www.cdc.gov/nchs/pressroom/sosmap/drug_poisoning_mortality/drug_poisoning.htm.
Back to Citation559. SAMHSA, “2022 NSDUH Detailed Tables,” SAMHSA, November 13, 2023, https://www.samhsa.gov/data/report/2022-nsduh-detailed-tables.
Back to Citation560. Morales, Dawn A., Crystal L. Barksdale, and Andrea C. Beckel-Mitchener. 2020. “A Call to Action to Address Rural Mental Health Disparities.” Journal of Clinical and Translational Science 4 (5): 1-20. https://doi.org/10.1017/cts.2020.42.
Back to Citation561. WWAMI Rural Health Research Center, University of Washington. Changes in the Supply and Rural-Urban Distribution of Psychologists in the U.S., 2014-2021. 2022. https://familymedicine.uw.edu/rhrc/wp-content/uploads/sites/4/2022/10/RHRC_DBOCT2022_PSYCHOLOGIST_Andrilla.pdf.
Back to Citation562. Nuako, Akua, Jingxia Liu, Giang Pham, Nina Smock, Aimee James, Timothy Baker, Laura Bierut, Graham Colditz, and Li-Shiun Chen. 2022. “Quantifying Rural Disparity in Healthcare Utilization in the United States: Analysis of a Large Midwestern Healthcare System.” Edited by Nickolas D. Zaller. PLOS ONE 17 (2): e0263718. https://doi.org/10.1371/journal.pone.0263718.
Back to Citation563. Arcury, Thomas A., John S. Preisser, Wilbert M. Gesler, and James M. Powers. 2005. “Access to Transportation and Health Care Utilization in a Rural Region.” The Journal of Rural Health 21 (1): 31-38. https://doi.org/10.1111/j.1748-0361.2005.tb00059.x.
Back to Citation564. GAO, Rural Hospital Closures, GAO-21-93, (Washington, DC, 2020), https://www.gao.gov/assets/gao-21-93.pdf.
Back to Citation565. Lister, Jamey J., and Paul J. Joudrey. 2022. “Rural Mistrust of Public Health Interventions in the United States: A Call for Taking the Long View to Improve Adoption.” The Journal of Rural Health 39 (1): 18-20. https://doi.org/10.1111/jrh.12684.
Back to Citation566. CDC, “About Rural Health,” CDC, May 16, 2024, https://www.cdc.gov/rural-health/php/about/?CDC_AAref_Val=https://www.cdc.gov/ruralhealth/about.html.
Back to Citation567. CDC, NCHS, Trends in death rates in urban and rural areas: United States, 1999-2019, by Sally C. Curtin and Merianne Rose Spencer, NCHS Data Brief No. 417, (Hyattsville, MD, 2021), https://dx.doi.org/10.15620/cdc:109049.
Back to Citation568. Smith, Laura Berrie, Michael Karpman, Dulce Gonzalez, and Sarah Morriss, “More than One in Five Adults with Limited Public Transit Access Forgo Health Care Because of Transportation Barriers,” Robert Wood Johnson Foundation, April 26, 2023, https://www.rwjf.org/en/insights/our-research/2023/04/more-than-one-in-five-adults-with-limited-public-transit-access-forgo-healthcare-because-of-transportation-barriers.html.
Back to Citation569. U.S. Department of Transportation (DOT), Federal Highway Administration (FHWA), Chapter 11: Rural America, Status of the Nation's Highways, Bridges, and Transit Conditions and Performance Report, 24th Edition (Washington, DC, 2021), https://www.fhwa.dot.gov/policy/24cpr/pdf/Chapter11.pdf.
Back to Citation570. Id.
Back to Citation571. Id.
Back to Citation572. CDC, Potentially Excess Deaths from the Five Leading Cause of Death in Metropolitan and Nonmetropolitan Counties—United States, 2010-2017, by Macarena C. Garcia, Lauren M. Rossen, Brigham Bastian, Mark Faul, Nicole F. Dowling, Cheryll C. Thomas, Linda Schieb, Yuling Hong, Paula W. Yoon, and Michael F. Iademarco, Morbidity and Mortality Weekly Report Vol. 68, No. 10, (Atlanta, GA, 2019), https://www.cdc.gov/mmwr/volumes/68/ss/pdfs/ss6810a1-H.pdf.
Back to Citation573. “Am I Rural?—Tool,” Rural Health Information Hub, accessed May 7, 2024, https://www.ruralhealthinfo.org/am-i-rural. This tool is not official and should not be relied upon as a formal Federal determination that a location is rural.
Back to Citation574. HRSA, “Defining Rural Population,” Health Resources & Services Administration, January 2024, https://www.hrsa.gov/rural-health/about-us/what-is-rural.
575. Response to Comments on Revised Geographic Eligibility for Federal Office of Rural Health Policy Grants, 86 FR 2418 (January 12, 2021). See, https://www.federalregister.gov/documents/2021/01/12/2021-00443/response-to-comments-on-revised-geographic-eligibility-for-federal-office-of-rural-health-policy.
Back to Citation576. Census Bureau, “Urban and Rural,” United States Census Bureau, September 2023, https://www.census.gov/programs-surveys/geography/guidance/geo-areas/urban-rural.html.
Back to Citation577. Urban Area Criteria for the 2020 Census-Final Criteria, 87 FR 16706 (Mar. 24, 2022). See, https://www.federalregister.gov/documents/2022/03/24/2022-06180/urban-area-criteria-for-the-2020-census-final-criteria.
Back to Citation578. Revised Geographic Eligibility for Federal Office of Rural Health Policy Grants, 85 FR 59806 (Oct. 23, 2020). See, https://www.federalregister.gov/documents/2020/09/23/2020-20971/revised-geographic-eligibility-for-federal-office-of-rural-health-policy-grants.
Back to Citation579. 2020 Standards for Delineating Core Based Statistical Areas, 86 FR 37770 (July 16, 2021). See, https://www.federalregister.gov/documents/2021/07/16/2021-15159/2020-standards-for-delineating-core-based-statistical-areas.
Back to Citation580. HRSA, Defining Rural Population.
Back to Citation581. CDC, NCHS, “NCHS Urban-Rural Classification Scheme for Counties,” CDC, June 1, 2017, https://www.cdc.gov/nchs/data-analysis-tools/urban-rural.html?CDC_AAref_Val=https://www.cdc.gov/nchs/data_access/urban_rural.htm.
Back to Citation582. Revised Geographic Eligibility for Federal Office of Rural Health Policy Grants, 85 FR 59806 (Oct. 23, 2020).
Back to Citation583. Response to Comments on Revised Geographic Eligibility for Federal Office of Rural Health Policy Grants, 86 FR 2418 (January 12, 2021).
584. HRSA, Defining Rural Population.
Back to Citation585. Proposed Inclusion of Terrain Factors in the Definition of Rural Area for Federal Office of Rural Health Policy Grants, 89 FR 32451 (April 26, 2024). See, https://www.federalregister.gov/documents/2024/04/26/2024-08931/proposed-inclusion-of-terrain-factors-in-the-definition-of-rural-area-for-federal-office-of-rural.
586. As of November 27, 2024, HRSA has not finalized these proposed changes to FORHP's definition of rural.
Back to Citation587. Response to Comments on Revised Geographic Eligibility for Federal Office of Rural Health Policy Grants, 86 FR 2418 (January 12, 2021).
Back to Citation588. U.S. Department of Agriculture (USDA), Economic Research Services (ERS), “Rural Classifications,” USDA, September 8, 2023, https://www.ers.usda.gov/topics/rural-economy-population/rural-classifications/.
Back to Citation589. Assistant Secretary of Planning and Evaluation (ASPE), How Increased Funding Can Advance the Mission of the Indian Health Service to Improve Health Outcomes for American Indians and Alaska Natives, Report No. HP-2022-21, (Washington, DC, 2022), https://aspe.hhs.gov/sites/default/files/documents/e7b3d02affdda1949c215f57b65b5541/aspe-ihs-funding-disparities-report.pdf.
Back to Citation591. See 87 FR 72225 and 72226.
Back to Citation592. See 85 FR 86237 and 86238.
Back to Citation594. The same requirement applies to state Medicaid agencies. 1902(a)(74) and (gg)(2) of the Act.
Back to Citation595. https://downloads.cms.gov/cmsgov/archived-downloads/SMDL/downloads/smd11001.pdf.
Back to Citation596. Georgetown University. (2021). Advancing Health Equity for Children and Adults with a Critical Tool: Medicaid and Children's Health Insurance Program Continuous Coverage. Retrieved from https://ccf.georgetown.edu/wp-content/uploads/2021/10/continuity-of-coverage-final.pdf.
Back to Citation597. Munira Gunja et al., Insights into the U.S. Maternal Mortality Crisis: An International Comparison (Commonwealth Fund, June 2024). https://doi.org/10.26099/cthn-st75.
Back to Citation601. Centers for Disease Control and Prevention. Pregnancy Mortality Surveillance System. Available at: https://www.cdc.gov/maternal-mortality/php/pregnancy-mortality-surveillance/index.html/ Accessed June 5th, 2024.
Back to Citation603. https://www.whitehouse.gov/wp-content/uploads/2022/06/Maternal-Health-Blueprint.pdf.
604. Taylor, J., Novoa, C., Hamm, K., & Phadke, S. (2021, December 3). Eliminating Racial Disparities in Maternal and Infant Mortality. Center for American Progress. https://www.americanprogress.org/article/eliminating-racial-disparities-maternal-infant-mortality/.
605. Hoffman KM, Trawalter S, Axt JR, Oliver MN. Racial bias in pain assessment and treatment recommendations, and false beliefs about biological differences between blacks and whites. Proc Natl Acad Sci U S A. 2016;113(16):4296-4301. doi:10.1073/pnas.1516047113 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4843483/.
Back to Citation606. White House Fact Sheet: Vice President Kamala Harris Announces Call to Action to Reduce Maternal Mortality and Morbidity. https://www.whitehouse.gov/briefing-room/statements-releases/2021/12/07/fact-sheet-vice-president-kamala-harris-announces-call-to-action-to-reduce-maternal-mortality-and-morbidity/.
Back to Citation607. Gleason JL, Grewal J, Chen Z, Cernich AN, Grantz KL. Risk of Adverse Maternal Outcomes in Pregnant Women With Disabilities. JAMA Netw Open. 2021;4(12):e2138414.doi:10.1001/jamanetworkopen.2021.38414.
608. Willi Horner-Johnson et.al. Perinatal Health Risks and Outcomes Among US Women With Self-Reported Disability, 41 Health Aff. 2011 (September. 2022), https://doi.org/10.1377/hlthaff.2022.00497.
Back to Citation611. CMS, CMS Announces New Actions to Help Hospitals Meet Obligations under EMTALA, January 22, 2024. Available at: https://www.cms.gov/newsroom/press-releases/cms-announces-new-actions-help-hospitals-meet-obligations-under-emtala#:~:text=Today%2C%20the%20Department%20of%20Health,meet%20their%20obligations%20under%20the.
Back to Citation612. https://www.cms.gov/priorities/your-patient-rights/emergency-room-rights/complaint-form.
Back to Citation615. U.S. Department of Health and Human Services, Strengthening Maternal Health. Available at: https://www.hhs.gov/healthcare/maternal-health/index.html.
Back to Citation616. Task Force on Maternal Mental Health, National Strategy to Improve Maternal Mental Health Care. Available at: https://www.samhsa.gov/sites/default/files/mmh-strategy.pdf.
Back to Citation617. U.S. Department of Health and Human Services, December 2021, HHS Announces Efforts to Help Expand Nationwide Access and Coverage for High-quality Maternal Health Services. Available at https://www.cms.gov/newsroom/press-releases/hhs-announces-efforts-help-expand-nationwide-access-and-coverage-high-quality-maternal-health.
Back to Citation618. U.S. Department of Health and Human Services, February 14, 2024, HHS Announces Final Phase Winners in Challenge to Address Racial Equity in Postpartum Care. Available at: https://www.hhs.gov/about/news/2024/02/14/hhs-announces-final-phase-winners-challenge-address-racial-equity-postpartum-care.html.
Back to Citation619. CMS, July 26, 2022, CMS Releases Maternity Care Action Plan to Implement Biden-Harris Maternal Health Blueprint; Launches Industry Call to Action. Available at: https://www.cms.gov/files/document/cms-maternity-care-action-plan.pdf.
Back to Citation620. CMS, August 1, 2022, New CMS Rule Increases Payments for Acute Care Hospitals and Advances Health Equity, Maternal Health. Available at https://www.cms.gov/newsroom/press-releases/new-cms-rule-increases-payments-acute-care-hospitals-and-advances-health-equity-maternal-health.
Back to Citation621. CMS, Transforming Maternal Health (TMaH) Model. Available at: https://www.cms.gov/priorities/innovation/innovation-models/transforming-maternal-health-tmah-model.
Back to Citation622. https://www.cms.gov/files/document/maternal-morbidity-structural-measure-specifications.pdf.
Back to Citation624. Medicaid.gov, Maternal & Infant Health Care Quality. Available at: https://www.medicaid.gov/medicaid/quality-of-care/quality-improvement-initiatives/maternal-infant-health-care-quality/index.html.
Back to Citation629. U.S. Department of Health and Human Services, Strengthening Maternal Health. Available at: https://www.hhs.gov/healthcare/maternal-health/index.html.
Back to Citation630. CMS, July 26, 2022, CMS Releases Maternity Care Action Plan to Implement Biden-Harris Maternal Health Blueprint; Launches Industry Call to Action. Available at: https://www.cms.gov/files/document/cms-maternity-care-action-plan.pdf.
Back to Citation631. Medicaid.gov, Maternal & Infant Health Care Quality. Available at: https://www.medicaid.gov/medicaid/quality-of-care/quality-improvement-initiatives/maternal-infant-health-care-quality/index.html.
Back to Citation632. The Joint Commission, Focus on Maternal Health to Help Reduce Mortality and Morbidity, January 17, 2023. Available at: https://www.jointcommission.org/resources/news-and-multimedia/news/2023/01/new-alerts-from-the-joint-commission-focus-on-maternal-health-to-help-reduce-mortality-and-morbidity/.
Back to Citation633. See, for example, ACOG, Levels of Maternal Care. Available at: https://www.acog.org/programs/lomc.
Back to Citation634. U.S. Department of Health and Human Services, Strengthening Maternal Health. Available at: https://www.hhs.gov/healthcare/maternal-health/index.html.
Back to Citation635. CMS, July 26, 2022, CMS Releases Maternity Care Action Plan to Implement Biden-Harris Maternal Health Blueprint; Launches Industry Call to Action. Available at: https://www.cms.gov/files/document/cms-maternity-care-action-plan.pdf.
Back to Citation636. Medicaid.gov, Maternal & Infant Health Care Quality. Available at: https://www.medicaid.gov/medicaid/quality-of-care/quality-improvement-initiatives/maternal-infant-health-care-quality/index.html.
Back to Citation637. For example, Alliance for Maternal Health. Available at: https://mchb.hrsa.gov/programs-impact/programs/alliance-innovation-maternal-health#:~:text=AIM%20%E2%80%9C%20patient%20safety%20bundles%20%E2%80%9D%20are%20sets,reduce%20preventable%20deaths%20and%20severe%20maternal%20morbidity%20%28SMM%29.
Back to Citation638. For example, the Maternal Mental Health Strategy. Available at: https://www.samhsa.gov/sites/default/files/mmh-strategy.pdf.
Back to Citation639. For example, the Challenge to Address Racial Equity in Postpartum Care. Available at: https://www.challenge.gov/?challenge=hhs-postpartum-equity-in-care-challenge.
Back to Citation640. CDC, Maternal Mortality from 2018 through 2022. Available at: https://www.cdc.gov/nchs/maternal-mortality/mmr-2018-2022-state-data.pdf.
Back to Citation642. CMS, The CMS National Quality Strategy: A Person-Centered Approach to Improving Quality, June 06, 2022. Available at: https://www.cms.gov/blog/cms-national-quality-strategy-person-centered-approach-improving-quality.
Back to Citation643. HHS, Telehealth for maternal health services. Available at: https://telehealth.hhs.gov/providers/ best-practice-guides/telehealth-for-maternal-health-services.
Back to Citation644. CMS, State Medicaid & CHIP Telehealth Toolkit, February 2024. Available at: https://www.medicaid.gov/medicaid/benefits/downloads/telehealth-toolkt.pdf.
Back to Citation651. See for example, Sobczak A, Taylor L, Solomon S, Ho J, Kemper S, Phillips B, Jacobson K, Castellano C, Ring A, Castellano B, Jacobs RJ. The Effect of Doulas on Maternal and Birth Outcomes: A Scoping Review. Cureus. 2023 May 24;15(5):e39451. doi: 10.7759/cureus.39451. PMID: 37378162; PMCID: PMC10292163.
Back to Citation653. https://saferbirth.org/wp-content/uploads/3-FINAL_AIM_OERRK_Readiness.pdf.
Back to Citation659. https://www.sciencedirect.com/science/article/pii/S2666142X24000419#bib0028.
660. https://www.sciencedirect.com/science/article/pii/S2352013221000028.
Back to Citation662. https://www.sciencedirect.com/science/article/pii/S2666142X24000419#bib0028.
Back to Citation664. https://www.acog.org/store/products/clinical-resources/guidelines-for-perinatal-care.
Back to Citation667. https://saferbirth.org/about-us/.
668. https://www.ihi.org/insights/what-is-a-bundle.
Back to Citation669. Jennifer A. Callaghan-Koru et al. Implementation of the Safe Reduction of Primary Cesarean Births safety bundle during the first year of a statewide collaborative in Maryland. Obstet Gynecol 2019;134:109-19.
670. Elliott K. Main et al. Reduction of severe maternal morbidity from hemorrhage using a state perinatal quality collaborative. Am J Obstet Gynecol 2017;216(3):298.e1-298.e11.
671. Patricia Lee King et al. Reducing time to treatment for severe maternal hypertension through statewide quality improvement. Am J Obstet Gynecol 2018;218:S4.
672. https://www.cms.gov/files/document/qso-22-05-hospitals.pdf.
Back to Citation673. https://www.acog.org/store/products/clinical-resources/guidelines-for-perinatal-care.
Back to Citation676. For purposes of the proposal, CMS considers a facility to “offer obstetrical services” when the facility “[holds itself] out to the public (by name, posted signs, advertising, or other means) as a place that provides care for obstetrical medical conditions.” This is similar to how emergency departments are defined in EMTALA (42 CFR 489.24(b), “Dedicated emergency department”, paragraph (2)).
Back to Citation680. https://data.cms.gov/provider-data/birthing-friendly-hospitals-and-health-systems.
Back to Citation688. For purposes of the proposal, CMS considered a facility to “offer emergency services” when it met the definition of “dedicated emergency department” as defined in EMTALA (42 CFR 489.24(b), “Dedicated emergency department”).
Back to Citation689. https://www.acep.org/patient-care/clinical-policies.
690. https://www.acep.org/geda.
Back to Citation692. https://www.acog.org/programs/obstetric-emergencies-in-nonobstetric-settings.
Back to Citation695. https://rhrc.umn.edu/wp-content/uploads/2021/09/UMN-emOB-Training-Needed_11.12.20_508.pdf.
696. https://www.cdcfoundation.org/sites/default/files/files/ReportfromNineMMRCs.pdf.
697. https://emscimprovement.center/domains/pediatric-readiness-project/.
699. https://forms.ihi.org/hubfs/Guide%20to%20Recognition%20for%20GEDA%20Sites_FINAL.pdf.
Back to Citation700. https://www.acep.org/patient-care/clinical-policies.
701. https://www.acep.org/geda.
702. https://saferbirth.org/.
703. https://www.acog.org/programs/obstetric-emergencies-in-nonobstetric-settings.
704. https://emscimprovement.center/domains/pediatric-readiness-project/.
Back to Citation705. https://www.acep.org/patient-care/clinical-policies.
706. https://www.acep.org/geda.
707. https://saferbirth.org/.
708. https://www.acog.org/programs/obstetric-emergencies-in-nonobstetric-settings.
709. https://emscimprovement.center/domains/pediatric-readiness-project/.
Back to Citation710. Available at: https://cmsqualitysupport.servicenowservices.com/qnet_qa.
Back to Citation711. Available at: https://cmsqualitysupport.servicenowservices.com/ccsq_support_central.
Back to Citation712. Available at: https://oncprojectracking.healthit.gov/olp/.
Back to Citation713. For more detailed information about entitlement and eligibility for Medicare, please refer to sections 226, 226A, 1818, 1818A, 1836, and 1881A of the Act and 42 CFR parts 406 and 407.
Back to Citation714. https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf.
Back to Citation715. Placeholder for 2024 Stars QUS.
Back to Citation716. Institute of Medicine (US) Committee on Quality of Health Care in America, Kohn, L.T., Corrigan, J.M., & Donaldson, M.S. (Eds.). (2000). To Err is Human: Building a Safer Health System. National Academies Press (US).
717. Institute of Medicine (US) Committee on Quality of Health Care in America. (2001). Crossing the Quality Chasm: A New Health System for the 21st Century. National Academies Press (US).
Back to Citation718. Agency for Healthcare Research and Quality. (February 2021). National Healthcare Quality and Disparities Report chartbook on patient safety. Rockville, MD. Available at: https://www.ahrq.gov/sites/default/files/wysiwyg/research/findings/nhqrdr/chartbooks/patientsafety/2019qdr-patient-safety-chartbook.pdf.
Back to Citation719. Lastinger LM, Alvarez CR, Kofman A, Konnor RY, Kuhar DT, Nkwata A, Patel PR, Pattabiraman V, Xu SY, Dudeck MA. Continued increases in the incidence of healthcare-associated infection (HAI) during the second year of the coronavirus disease 2019 (COVID-19) pandemic. Infect Control Hosp Epidemiol. 2023 Jun;44(6):997-1001. doi: 10.1017/ice.2022.116. Epub 2022 May 20. PMID: 35591782; PMCID: PMC9237489.
720. Patel, PR, Weiner-Lastinger, LM, Dudeck, MA, et al. Impact of COVID-19 pandemic on central-line-associated bloodstream infections during the early months of 2020, National Healthcare Safety Network. Infect Control Hosp Epidemiol 2021. doi: 10.1017/ice.2021.108.
Back to Citation721. Agency for Healthcare Research and Quality. (2021). AHRQ PSNet Annual Perspective: Impact of the COVID-19 Pandemic on Patient Safety. https://psnet.ahrq.gov/perspective/ahrq-psnet-annual-perspective-impact-covid-19-pandemic-patient-safety.
722. Fleisher, L.A., Schreiber, M.D., Cardo, D., and Srinivasan, M.D. (2022). Health care safety during the pandemic and beyond—building a system that ensures resilience. N Engl J Med, 386: 609-611. https://www.nejm.org/doi/full/10.1056/NEJMp2118285.
723. Implications of the COVID-19 pandemic for patient safety: a rapid review. Geneva: World Health Organization; 2022. Licence: CC BY-NC-SA 3.0 IGO.
Back to Citation724. AHRQ. (2023). National Action Alliance To Advance Patient and Workforce Safety. https://www.ahrq.gov/cpi/about/otherwebsites/action-alliance.html.
Back to Citation726. Fleisher, L.A., Schreiber, M., Cardo, D., Srinivasan, A. (2022). Health Care Safety during the Pandemic and Beyond—Building a System That Ensures Resilience. The New England Journal of Medicine, 386(7): 609-611. DOI: 10.1056/NEJMp2118285.
727. https://www.cms.gov/aligning-quality-measures-across-cms-universal-foundation.
Back to Citation729. Safety net hospitals are defined as those committed to caring for populations without stable access to care, specifically public hospitals or private hospitals with a Medicaid caseload greater than one standard deviation above their respective state's mean private hospital Medicaid caseload. https://www.cms.gov/newsroom/fact-sheets/data-brief-evaluation-national-distributions-overall-hospital-quality-star-ratings#_ftn3.
Back to Citation730. https://mmshub.cms.gov/sites/default/files/Star-Ratings-TEP-Summary-Report-Oct23.pdf.
Back to Citation731. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Medical Records Specialists. Accessed April 29, 2024. Available at: https://www.bls.gov/oes/current/oes292072.htm.
Back to Citation732. Kaiser Family Foundation, Hospital Outpatient Visits per 1,000 Population by Ownership Type. Available at http://www.kff.org/other/state-indicator/outpatient-visits-by-ownership/?currentTimeframe=&0sortModel=%7B%22colId%22:%22Location%22,%22sort%22:%22asc%22%7D.
Back to Citation733. Growth in U.S. Population Shows Early Indication of Recovery Amid COVID-19 Pandemic, December 22, 2022. Available at https://www.census.gov/newsroom/press-releases/2022/2022-population-estimates.html.
Back to Citation734. American Hospital Association, Fast Facts on U.S. Hospitals. Available at https://www.aha.org/statistics/fast-facts-us-hospitals.
Back to Citation735. American Hospital Association—Data and Insights, AHA Annual Survey DatabaseTM. Available at https://www.ahadata.com/aha-annual-survey-database.
Back to Citation736. Office of the Assistant Secretary for Planning and Evaluation, Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices, September 17, 2017. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework.
Back to Citation737. Bureau of Labor and Statistics, Usual Weekly Earnings of Wage and Salary Workers, First Quarter 2024. Available at https://www.bls.gov/news.release/pdf/wkyeng.pdf. Accessed April 16, 2024.
Back to Citation738. U.S. Census Bureau, End of Pandemic-Era Expanded Federal Tax Programs Results in Lower Income, Higher Poverty, September 12, 2023. Available at https://www.census.gov/library/stories/2023/09/median-household-income.html. Accessed April 16, 2024.
Back to Citation739. Kaiser Family Foundation, Hospital Outpatient Visits per 1,000 Population by Ownership Type. Available at http://www.kff.org/other/state-indicator/outpatient-visits-by-ownership/?currentTimeframe=0&sortModel=%7B%22colId%22%22Location%22%22sort%22:%22asc%22%7D.
Back to Citation740. Growth in U.S. Population Shows Early Indication of Recovery Amid COVID-19 Pandemic, December 22, 2022. Available at https://www.census.gov/newsroom/press-releases/2022/2022-population-estimates.html.
Back to Citation741. American Hospital Association—Data and Insights, AHA Annual Survey DatabaseTM. Available at https://www.ahadata.com/aha-annual-survey-database.
Back to Citation742. Yale New Haven Health Services Corporation—Center for Outcomes Research & Evaluation, Patient Understanding of Key Information Related to Recovery After a Facility-Based Outpatient Procedure or Surgery, Patient Reported Outcome-Based Performance Measure (PRO-PM) Version 1.0 Methodology Report, April 2024. Available at https://www.cms.gov/medicare/quality/initiatives/hospital-quality-initiative/measure-methodology.
Back to Citation743. Office of the Assistant Secretary for Planning an Evaluation, Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices, September 17, 2017. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework.
Back to Citation744. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Medical Records Specialists. Accessed April 29, 2024. Available at: https://www.bls.gov/oes/current/oes292072.htm.
Back to Citation745. Definitive Healthcare, Breaking down U.S. hospital payor mixes, July 14, 2023. Available at https://www.definitivehc.com/resources/healthcare-insights/breaking-down-us-hospital-payor-mixes.
746. Medicare Payment Advisory Commission, Medicare and the Health Care Delivery System, June 2021. Available at https://www.medpac.gov/wp-content/uploads/import_data/scrape_files/docs/default-source/reports/jun21_medpac_report_to_congress_sec.pdf.
Back to Citation747. Office of the Assistant Secretary for Planning an Evaluation, Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices, September 17, 2017. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework.
Back to Citation748. Bureau of Labor and Statistics, Usual Weekly Earnings of Wage and Salary Workers, First Quarter 2024. Available at https://www.bls.gov/news.release/pdf/wkyeng.pdf. Accessed April 16, 2024.
Back to Citation749. U.S. Census Bureau, End of Pandemic-Era Expanded Federal Tax Programs Results in Lower Income, Higher Poverty, September 12, 2023. Available at https://www.census.gov/library/stories/2023/09/median-household-income.html. Accessed April 16, 2024.
Back to Citation750. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Medical Records Specialists. Accessed April 29, 2024. Available at: https://www.bls.gov/oes/current/oes292072.htm.
Back to Citation751. ASC Quality Collaboration, ASC Quality Collaboration Quality Report. Available at https://ascquality.org/benchmarking/. Accessed September 12, 2024.
Back to Citation752. Office of the Assistant Secretary for Planning an Evaluation, Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices, September 17, 2017. Available at https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework.
Back to Citation753. Bureau of Labor and Statistics, Usual Weekly Earnings of Wage and Salary Workers, First Quarter 2024. Available at https://www.bls.gov/news.release/pdf/wkyeng.pdf. Accessed April 16, 2024.
Back to Citation754. U.S. Census Bureau, End of Pandemic-Era Expanded Federal Tax Programs Results in Lower Income, Higher Poverty, September 12, 2023. Available at https://www.census.gov/library/stories/2023/09/median-household-income.html. Accessed April 16, 2024.
Back to Citation755. See also Correction Notification issued January 3, 2020 (85 FR 224).
Back to Citation757. Provider of Services File—Hospital & Non-Hospital Facilities. Available at https://data.cms.gov/provider-characteristics/hospitals-and-other-facilities/provider-of-services-file-hospital-non-hospital-facilities/data. Accessed April 10, 2024.
Back to Citation758. The Joint Commission Edition. Program: Perinatal Care, “Chapter: Provision of Care, Treatment, and Services.” Available at https://e-dition.jcrinc.com/MainContent.aspx. Accessed April 10, 2024.
Back to Citation759. Quality, Certification & Oversight Reports (QCOR). CASPER (4/16/2024). qcor.cms.gov.
Back to Citation760. U.S. Bureau of Labor Statistics. Occupational Employment and Wages—May 2015. Accessed September 25, 2024. https://www.bls.gov/news.release/archives/ocwage_03302016.pdf.
Back to Citation761. U.S. Bureau of Labor Statistics. Occupational Employment and Wages, May 2023: 29-1141 Registered Nurses. Accessed September 25, 2024. https://www.bls.gov/oes/current/oes291141.html.
Back to Citation762. Federal Reserve Bank of Minneapolis. Inflation Calculator. Accessed February September 25, 2024. https://www.minneapolisfed.org/about-us/monetary-policy/inflation-calculator.
Back to Citation763. Tikkanen, R., et al., Maternal Mortality and Maternity Care in the United States Compared to 10 Other Developed Countries. 2020, Commonwealth Fund.
Back to Citation764. Hoyert, D.L., Maternal Mortality Rates in the United States, 2021. 2023, NCHS Health E-Stats.
Back to Citation765. United States Government Accountability Office, MATERNAL HEALTH: Outcomes Worsened and Disparities Persisted During the Pandemic. 2022.
Back to Citation766. Creanga, A.A., et al., Pregnancy-Related Mortality in the United States, 2011-2013. Obstetrics & Gynecology, 2017. 130(2): 366-373.
767. Wang, S., et al., Maternal Mortality in the United States: Trends and Opportunities for Prevention. Annual Review of Medicine, 2023. 74(1): 199-216.
Back to Citation768. Peterson, E.E., et al., Vital Signs: Pregnancy-Related Deaths, United States, 2011-2015, and Strategies for Prevention, 13 States, 2013-2017. Morbidity and Mortality Weekly Report, 2019. 68(18): 423-429.
Back to Citation769. Hoyert, D.L., Maternal Mortality Rates in the United States, 2021. 2023, NCHS Health E-Stats.
Back to Citation770. Merkt, P.T., et al., Urban-rural differences in pregnancy-related deaths, United States, 2011-2016. American Journal of Obstetrics and Gynecology, 2021. 225(2): 183.e1-183.e16.
Back to Citation771. Firoz, T., et al., Measuring maternal health: focus on maternal morbidity. Bull World Health Organ, 2013. 91(10): 794-796.
Back to Citation772. Liese, K.L., et al., Racial and Ethnic Disparities in Severe Maternal Morbidity in the United States. Journal of Racial and Ethnic Health Disparities, 2019. 6(4): p. 790-798.
773. Leonard, S.A., et al., Racial and ethnic disparities in severe maternal morbidity prevalence and trends. Annals of Epidemiology, 2019. 33: 30-36.
Back to Citation774. White Robert, S., et al., Economic burden of maternal mortality in the USA, 2018-2020. Journal of Comparative Effectiveness Research, 2022. 11(13): 927-933.
Back to Citation775. Black, C.M., et al., Costs of Severe Maternal Morbidity in U.S. Commercially Insured and Medicaid Populations: An Updated Analysis. Women's Health Reports, 2021. 2(1): 443-451.
Back to Citation776. O'Neil, S.S., et al., Societal cost of nine selected maternal morbidities in the United States. PLOS ONE, 2022. 17(10): e0275656.
Back to Citation777. Moran, P.S., et al., Economic burden of maternal morbidity—A systematic review of cost-of-illness studies. PLOS ONE, 2020. 15(1): e0227377.
Back to Citation778. Sections 321(a) and 322(b) of the Public Health Service Act (42 U.S.C. 248), Public Law 83-568 (42 U.S.C. 2001(a)), and the Indian Health Care Improvement Act (25 U.S.C. 1601 et seq.).
Back to Citation779. Bae, S.H., Noneconomic and economic impacts of nurse turnover in hospitals: A systematic review. Int Nurs Rev, 2022. 69(3): 392-404.
Back to Citation780. Medical, U. FETAL DOPPLERS. 2024 May 8, 2024 [cited 2024 May 8]; Available from: https://www.usamedicalsurgical.com/fetal-dopplers/.
781. CardiacDirect. Fetal Monitors. 2024 [cited 2024 May 8]; Available from: https://www.cardiacdirect.com/product-category/fetal-monitors/?utm_source=google&utm_medium=cpc&utm_term=fetal%20heart%20monitor&utm_content=!acq!v3!1163626993_kwd-295102856827__607346518010_g_c__&utm_campaign=FetalMonitor&gad_source=1&gclid=EAIaIQobChMIsLqpo8f-hQMVLyetBh3deAZuEAAYASAAEgIcx_D_BwE.
Back to Citation782. CardiacDirect. Patient Monitors. 2024 May 8, 2024 [cited 2024 May 8]; Available from: https://www.cardiacdirect.com/product-category/patient-monitors/?min_price=0&max_price=10250&page=1.
Back to Citation783. Osterman, M.J.K.H., Brady E., Martin, Joyce A.; Driscoll, Anne K.; Valenzuela, Claudia P., Births: Final Data for 2022, in National Vital Statistics Reports, U.S.D.o.H.a.H. Services, Editor. 2024, Centers for Disease Control and Prevention.
Back to Citation784. U.S. Bureau of Labor Statistics. Occupational Employment and Wages—May 2015. Accessed September 25, 2024. https://www.bls.gov/news.release/archives/ocwage_03302016.pdf.
Back to Citation785. U.S. Bureau of Labor Statistics. Occupational Employment and Wages, May 2023: 29-1141 Registered Nurses. Accessed September 25, 2024. https://www.bls.gov/oes/current/oes291141.html.
Back to Citation786. Federal Reserve Bank of Minneapolis. Inflation Calculator. Accessed February September 25, 2024. https://www.minneapolisfed.org/about-us/monetary-policy/inflation-calculator.
Back to Citation787. Centers for Medicare and Medicaid Services. Provider of Services File—Hospital & Non-Hospital Facilities, Q1 2024. https://data.cms.gov/provider-characteristics/hospitals-and-other-facilities/provider-of-services-file-hospital-non-hospital-facilities/data (Accessed April 30, 2024).
Back to Citation788. Nursing Solutions Incorporated, 2024 NSI National Health Care Retention & RN Staffing Report. 2024.
Back to Citation789. Centers for Medicare and Medicaid Services. Provider of Services File—Hospital & Non-Hospital Facilities, Q1 2024. https://data.cms.gov/provider-characteristics/hospitals-and-other-facilities/provider-of-services-file-hospital-non-hospital-facilities/data (Accessed April 30, 2024).
Back to Citation790. Nursing Solutions Incorporated, 2024 NSI National Health Care Retention & RN Staffing Report. 2024.
Back to Citation791. Nursing Solutions Incorporated, 2024 NSI National Health Care Retention & RN Staffing Report. 2024.
Back to Citation792. Nursing Solutions Incorporated, 2024 NSI National Health Care Retention & RN Staffing Report. 2024.
Back to Citation793. Nursing Solutions Incorporated, 2024 NSI National Health Care Retention & RN Staffing Report. 2024.
Back to Citation794. White Robert, S., et al., Economic burden of maternal mortality in the USA, 2018-2020. Journal of Comparative Effectiveness Research, 2022. 11(13): 927-933.
Back to Citation795. O'Neil, S.S., et al., Societal cost of nine selected maternal morbidities in the United States. PLOS ONE, 2022. 17(10): e0275656.
796. These nine conditions included the following: amniotic fluid embolism, cardiac arrest, gestational diabetes mellitus, hemorrhage, hypertensive disorders, mental health conditions, renal disease, sepsis, and venous thromboembolism.
Back to Citation797. Black, C.M., et al., Costs of Severe Maternal Morbidity in U.S. Commercially Insured and Medicaid Populations: An Updated Analysis. Women's Health Reports, 2021. 2(1): 443-451.
Back to Citation798. Moran, P.S., et al., Economic burden of maternal morbidity—A systematic review of cost-of-illness studies. PLOS ONE, 2020. 15(1): e0227377.
Back to Citation800. Togioka, Brandon & Seligman, Katherine & Delgado Upegui, Carlos. (2022). Limited English proficiency in the labor and delivery unit. Current Opinion in Anesthesiology. 35. 285-291.
Back to Citation801. Sentell, Tetine & Chang, Ann & Ahn, Hyeong Jun & Miyamura, Jill. (2015). Maternal Language and Adverse Birth Outcomes in a Statewide Analysis. Women & health. 56. 10.1080/03630242.2015.1088114.
802. https://www.ahrq.gov/sites/default/files/publications/files/lepguide.pdf).
Back to Citation803. Weinick, R.M. and R. Hasnain-Wynia, Quality Improvement Efforts Under Health Reform: How To Ensure That They Help Reduce Disparities—Not Increase Them. Health Affairs, 2011. 30(10): p. 1837-1843.
804. Bardach, N.S. and M.D. Cabana, The unintended consequences of quality improvement. Curr Opin Pediatr, 2009. 21(6): p. 777-82.
805. Perzynski, A.T., et al., Patient portals and broadband internet inequality. J Am Med Inform Assoc, 2017. 24(5): p. 927-932.
806. Antonio, M.G., O. Petrovskaya, and F. Lau, Is research on patient portals attuned to health equity? A scoping review. J Am Med Inform Assoc, 2019. 26(8-9): p. 871-883.
807. Sequist, T.D., et al., Effect of quality improvement on racial disparities in diabetes care. Arch Intern Med, 2006. 166(6): p. 675-81.
Back to Citation808. Burke SM, Schmitt T, Kennedy P, et al. Emergency Medicine Obstetrics and Gynecology: A Case-Based Curriculum for Residents. MedEdPORTAL. 2023;19:11330.
809. Espey E, Baty G, Rask J, Chungtuyco M, Pereda B, Leeman L. Emergency in the clinic: a simulation curriculum to improve outpatient safety. Am J Obstet Gynecol. Dec 2017;217(6):699.e1-699.e13.
810. Cooper MI, Papanagnou D, Meguerdichian M, Bajaj K. Emergency Obstetrics for the Emergency Medicine Provider. MedEdPORTAL. Oct 13 2016;12:10481.
811. Jacobs PJ. Using High-Fidelity Simulation and Video-Assisted Debriefing to Enhance Obstetrical Hemorrhage Mock Code Training. J Nurses Prof Dev. Sep/Oct 2017;33(5):234-239.
812. Hopmann P, Varre JS, Duncan G, Devoe WB, Gable BD. Multidisciplinary Simulation of Trauma in Pregnancy with Resuscitative Endovascular Balloon Occlusion of the Aorta (REBOA) Utilization. Cureus. Dec 2022;14(12):e32820.
813. Harrington J, Duncan G, D. Angelo K G. Multidisciplinary Simulation Improves Resident Confidence for Pregnant Patients Requiring Surgical Intervention. Cureus. Mar 2022;14(3):e23454.
Back to Citation814. Ames, S.G., et al., Emergency Department Pediatric Readiness and Mortality in Critically Ill Children. Pediatrics, 2019. 144(3).
815. Newgard, C.D., et al., Emergency Department Pediatric Readiness and Short-term and Long-term Mortality Among Children Receiving Emergency Care. JAMA Network Open, 2023. 6(1): p. e2250941-e2250941.
Back to Citation816. Keyes, DC, et al., Impact of a New Senior Emergency Department on Emergency Department Recidivism, Rate of Hospital Admission, and Hospital Length of Stay. Annals of Emergency Medicine, 2014. 63(5): p. 517-524.
817. Dresden, S.M., et al., Geriatric Emergency Department Innovations: The Impact of Transitional Care Nurses on 30-day Readmissions for Older Adults. Acad Emerg Med, 2020. 27(1): p. 43-53.
818. Foo, C.L., et al., Geriatric assessment and intervention in an emergency department observation unit reduced re-attendance and hospitalisation rates. Australas J Ageing, 2012. 31(1): p. 40-6.
819. Chong, E., et al., Emergency Department Interventions for Frailty (EDIFY): Front-Door Geriatric Care Can Reduce Acute Admissions. Journal of the American Medical Directors Association, 2021. 22(4): p. 923-928.e5.
820. Gettel, C.J., et al., An Outcome Comparison Between Geriatric and Nongeriatric Emergency Departments. Ann Emerg Med, 2023. 82(6): p. 681-689.
Back to Citation821. United State Census Bureau.622110: General medical and surgical hospitals. https://data.census.gov/profile/622110_-_General_medical_and_surgical_hospitals?n=622110&g=010XX00US. Accessed October 16, 2024.
Back to Citation
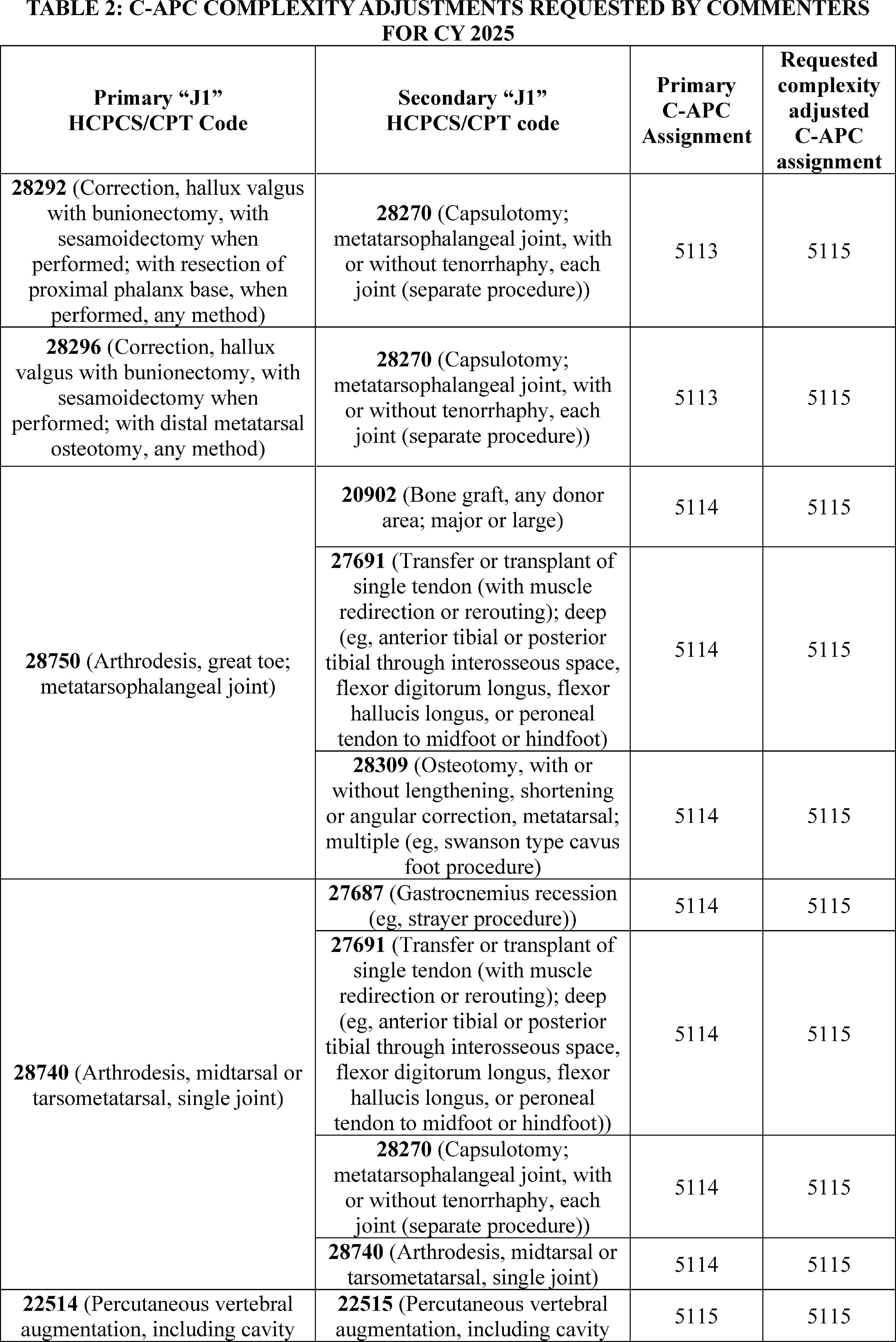



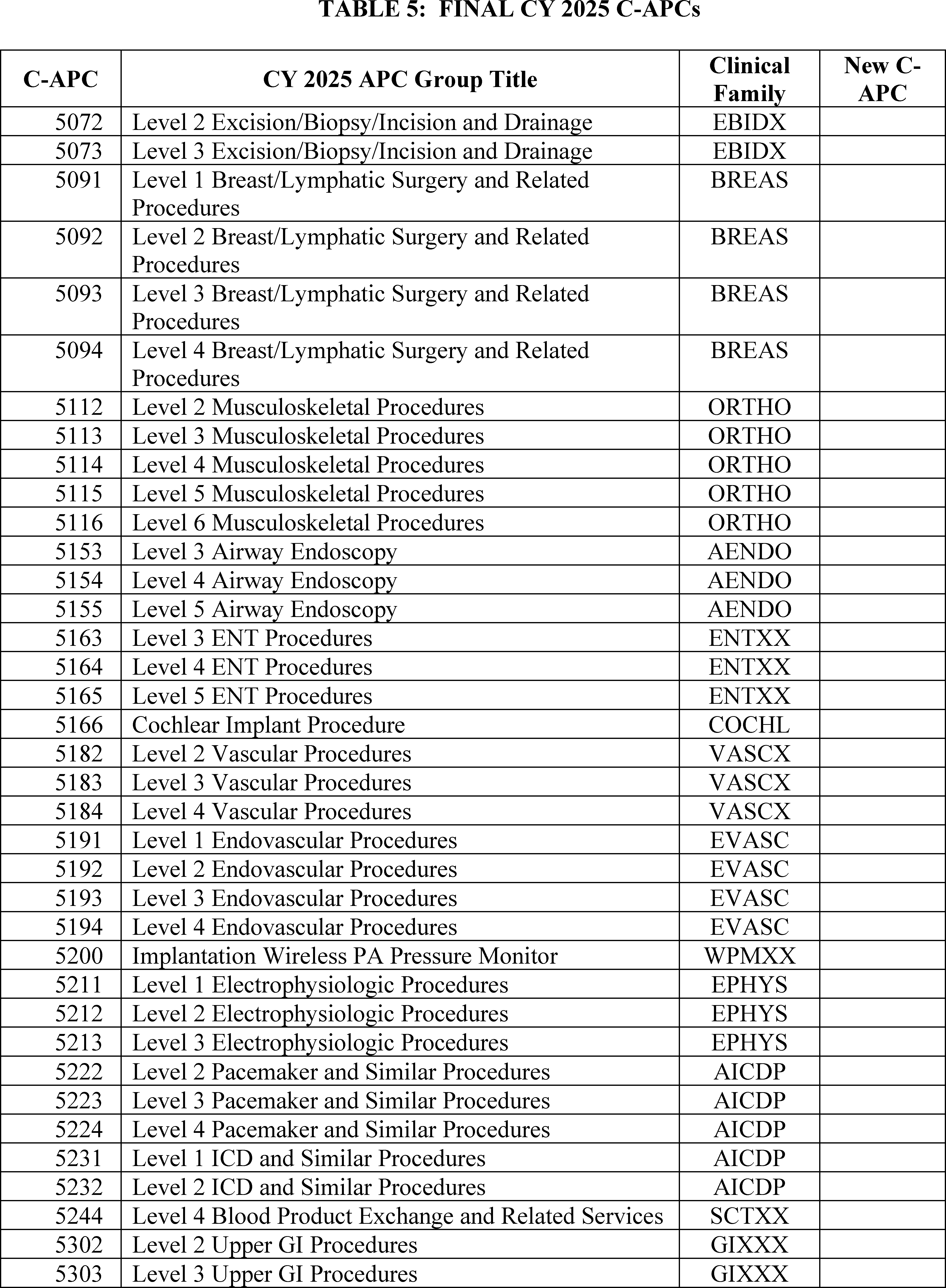
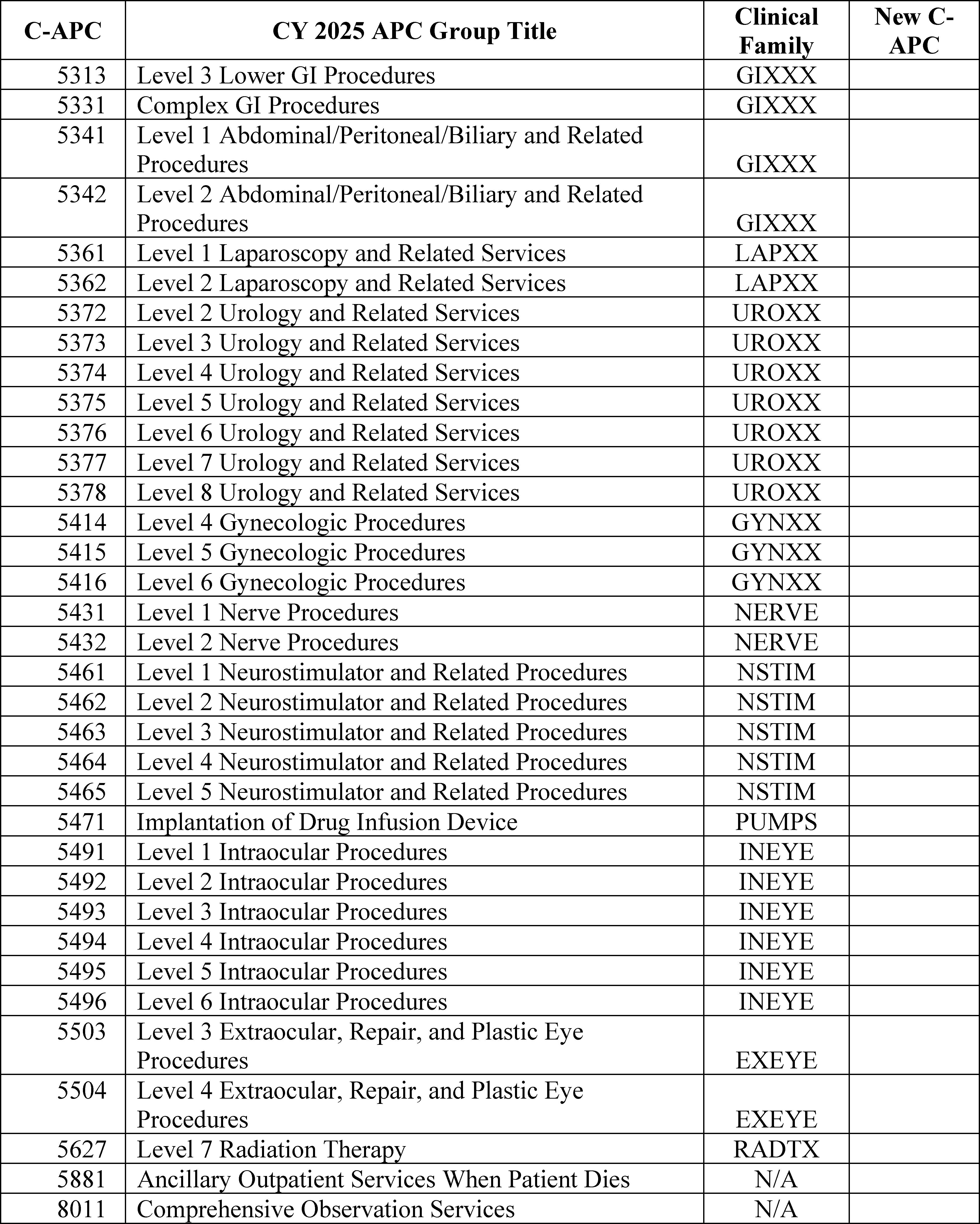

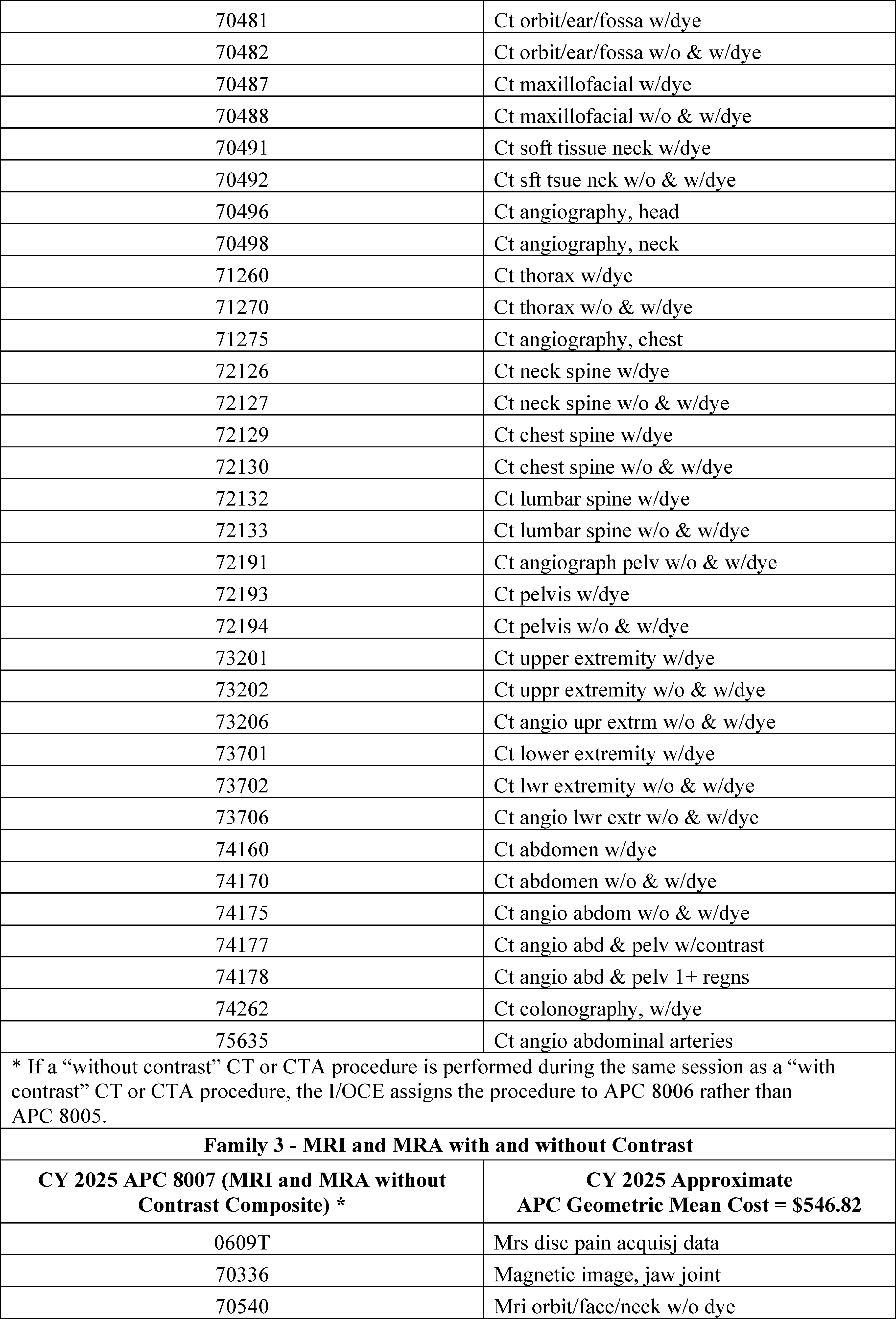


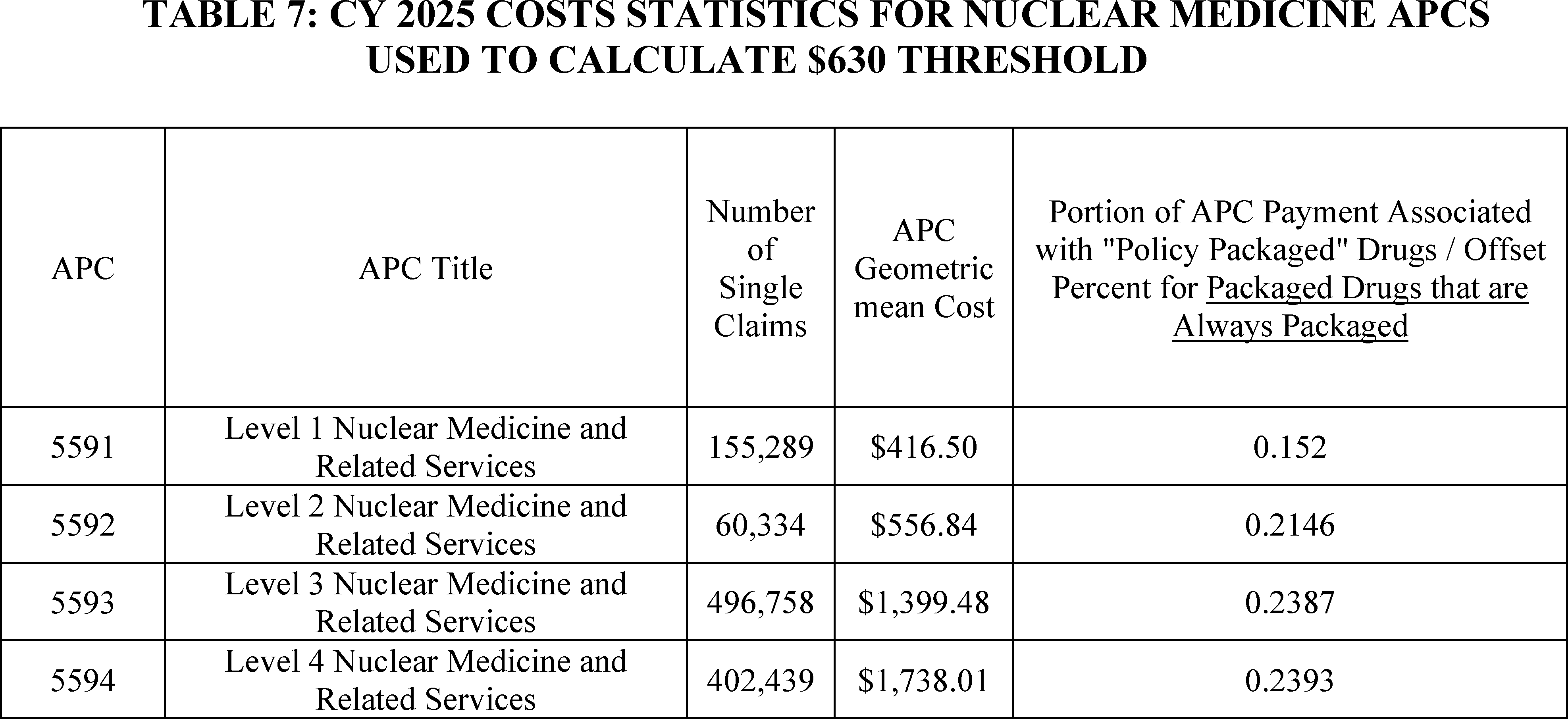
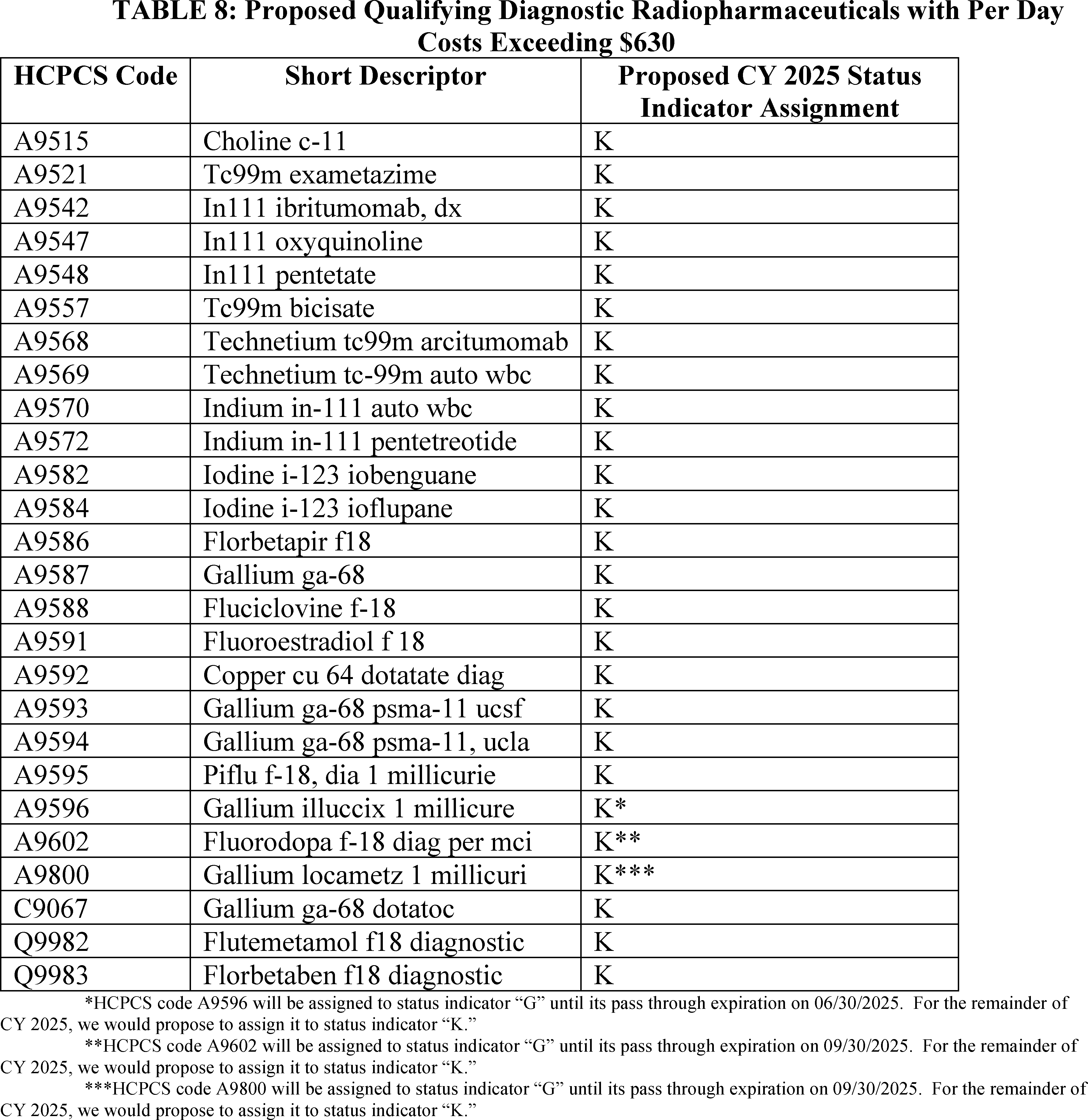
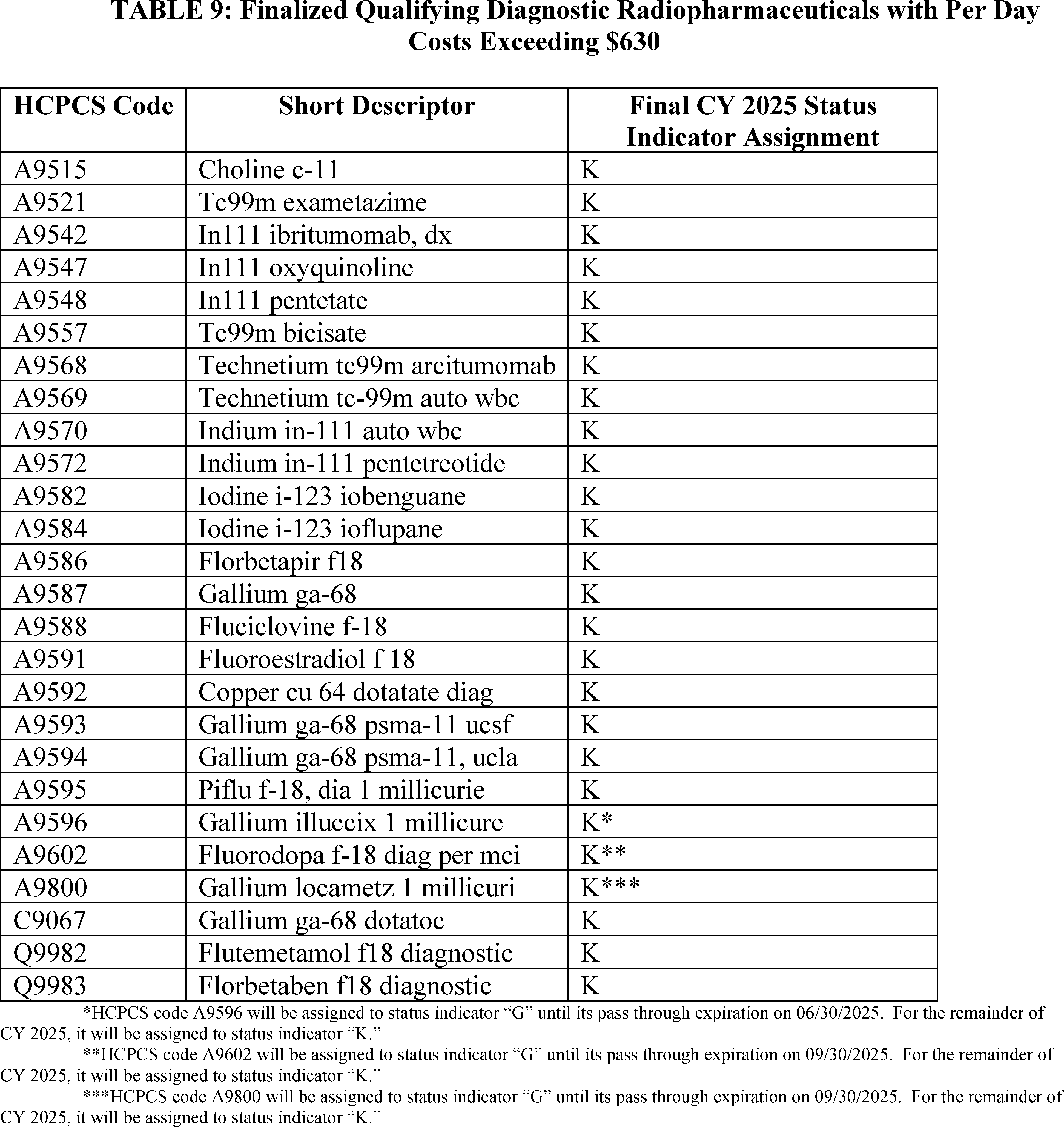


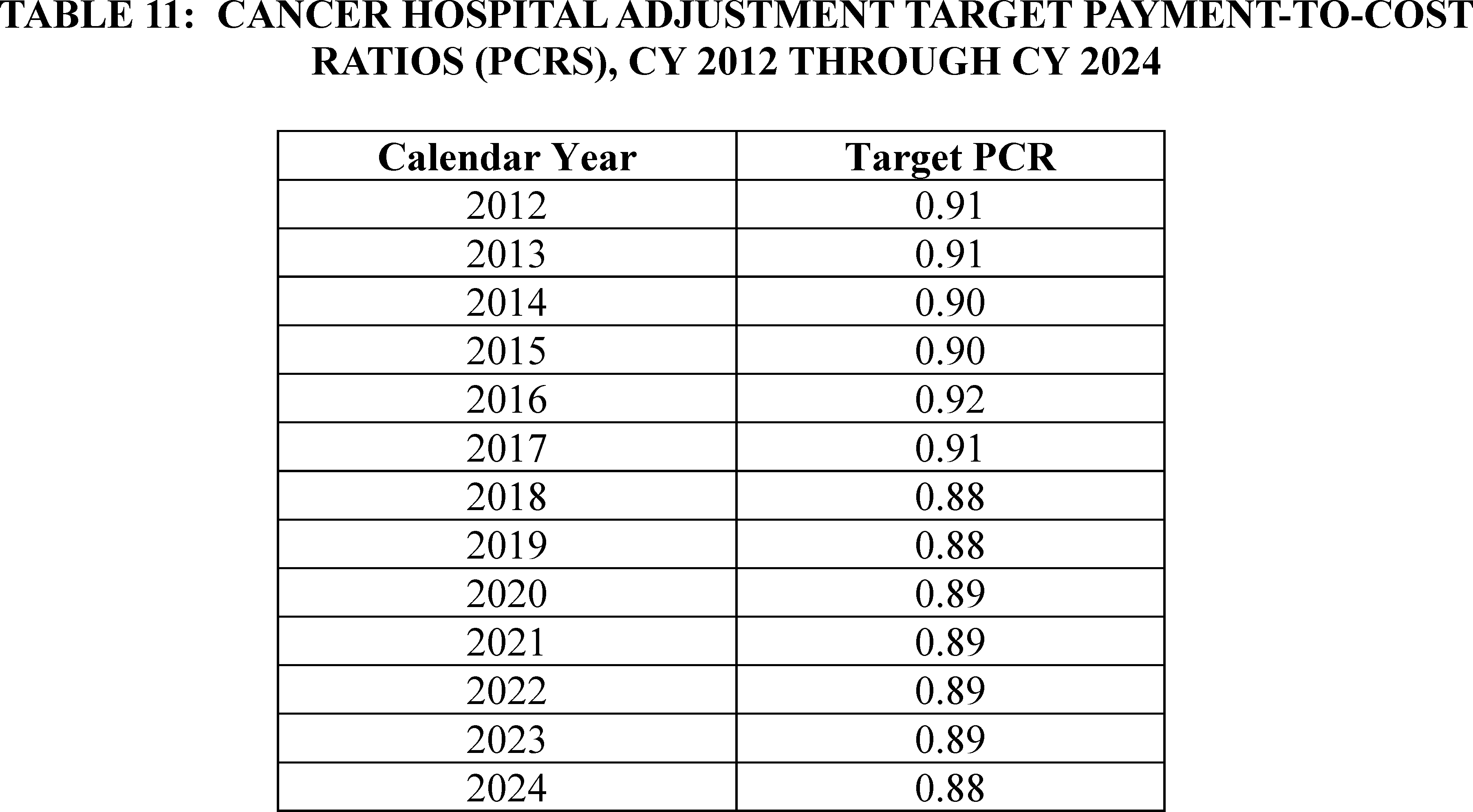
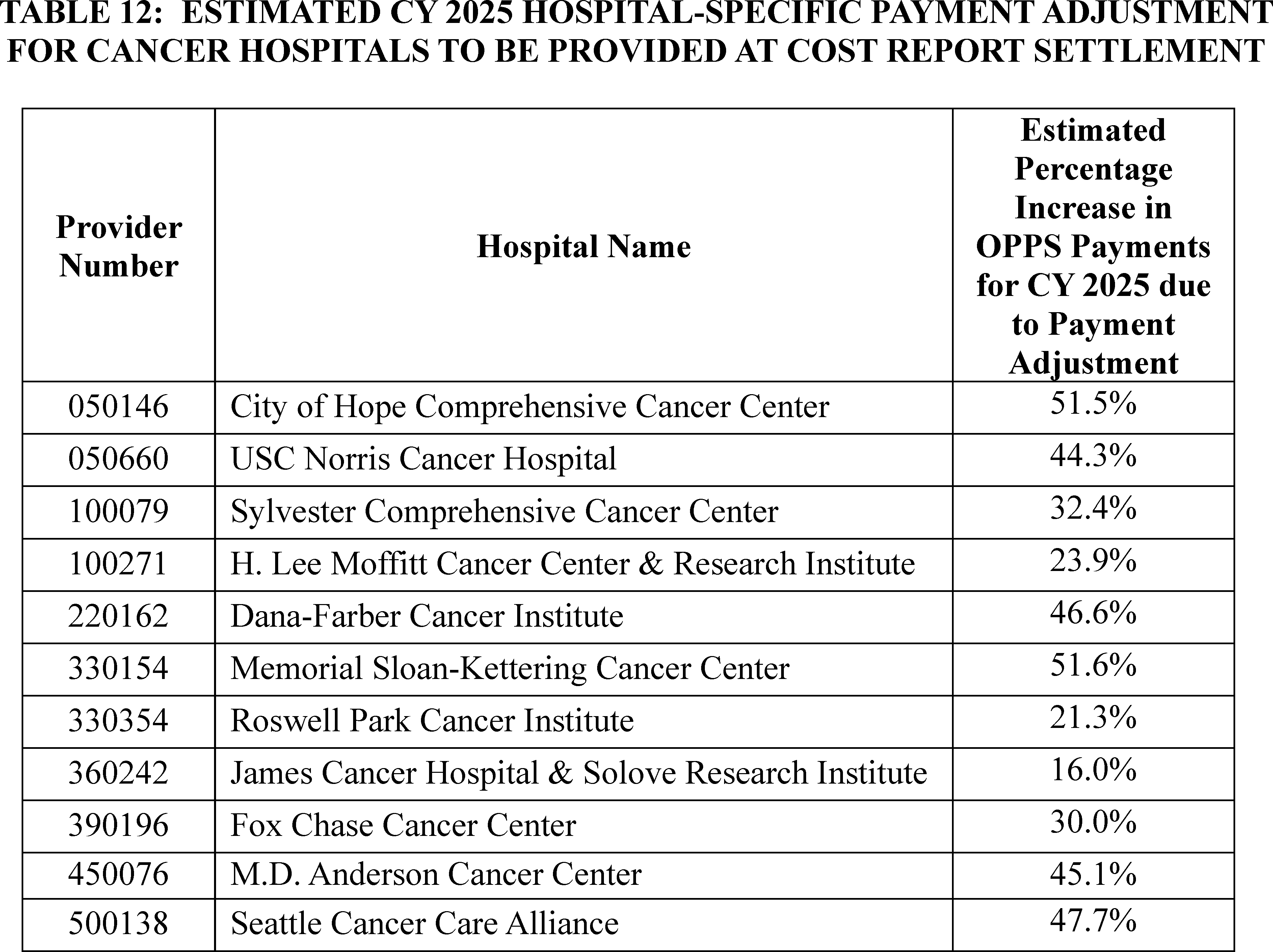

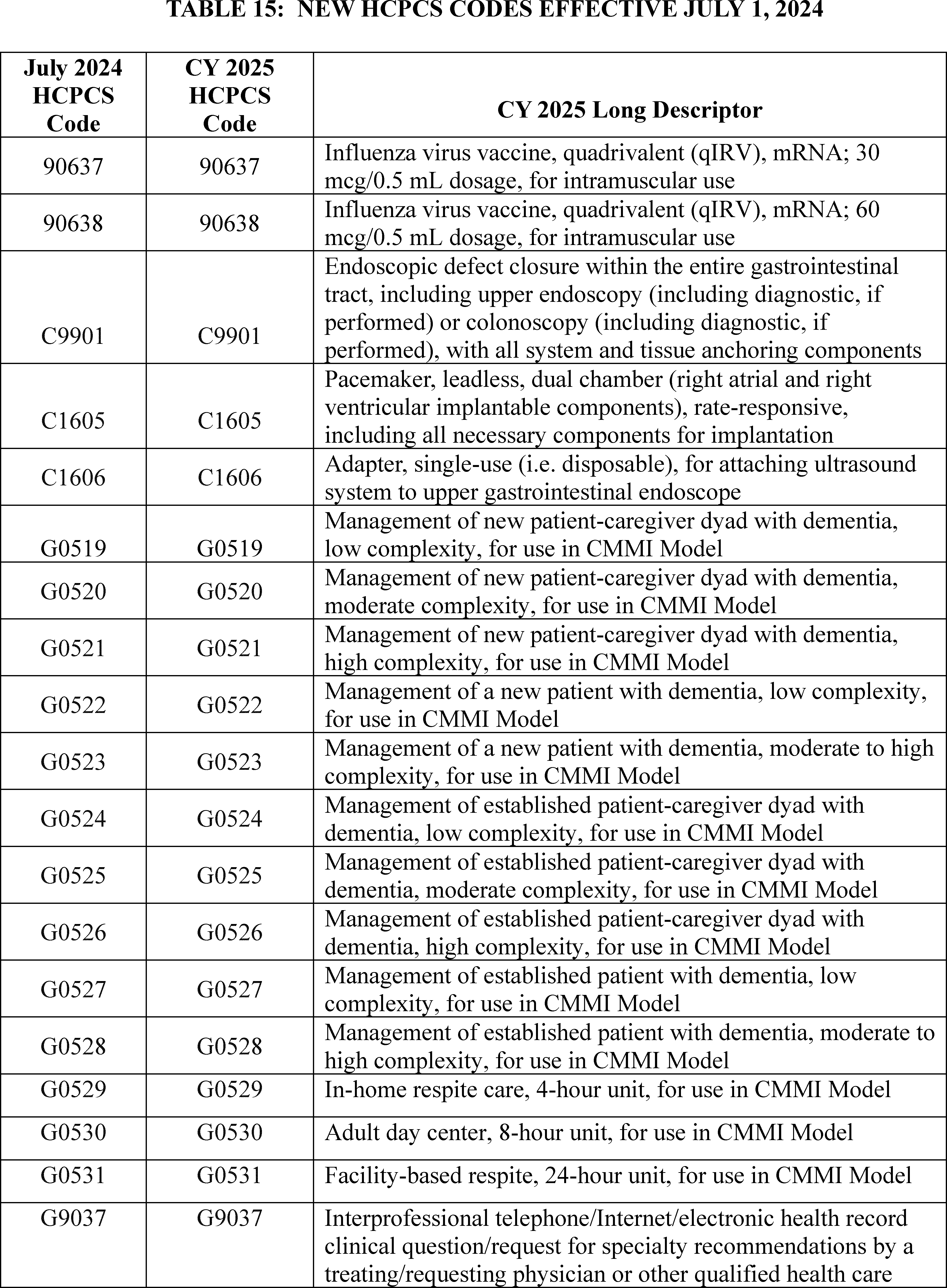
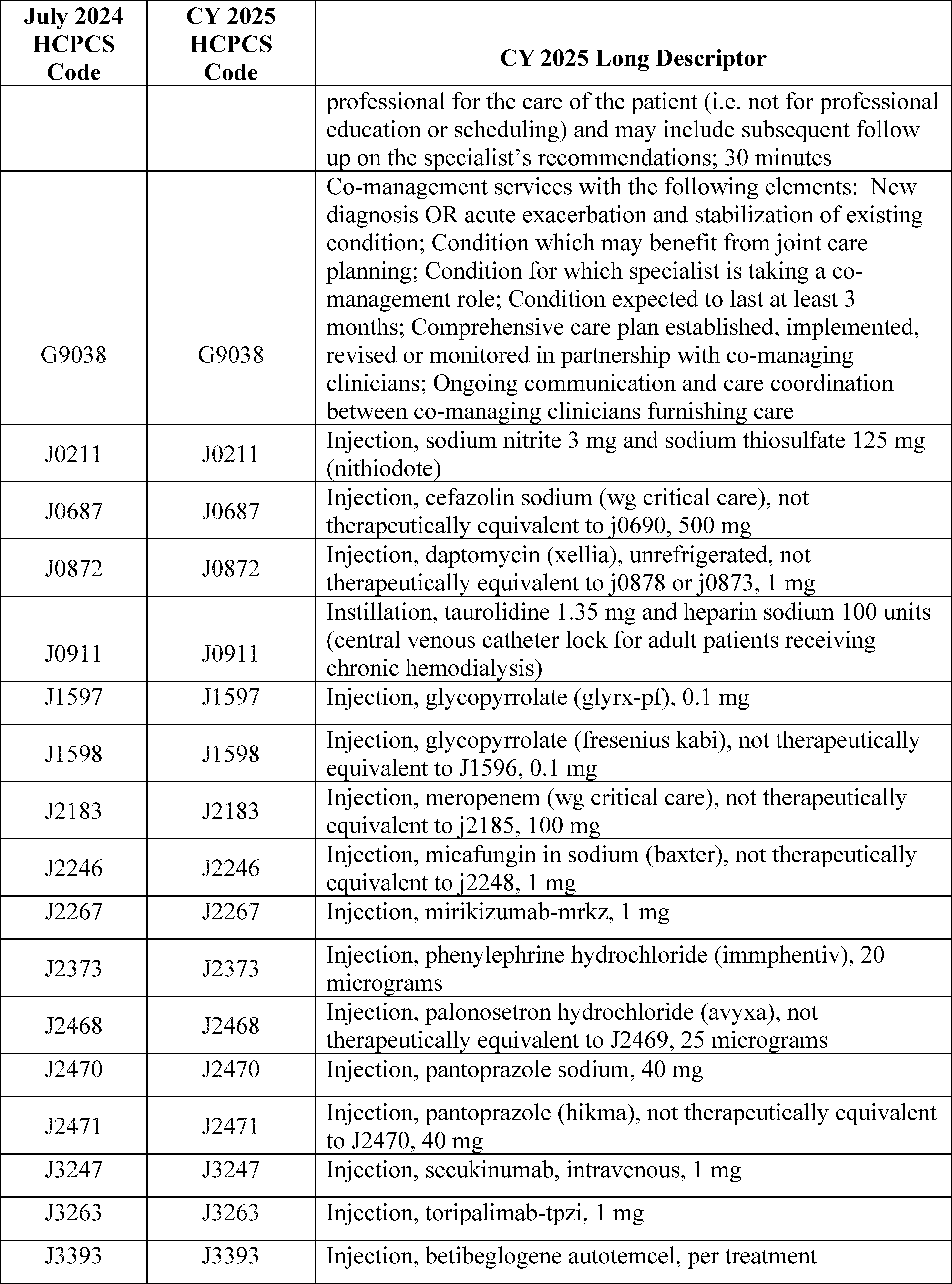
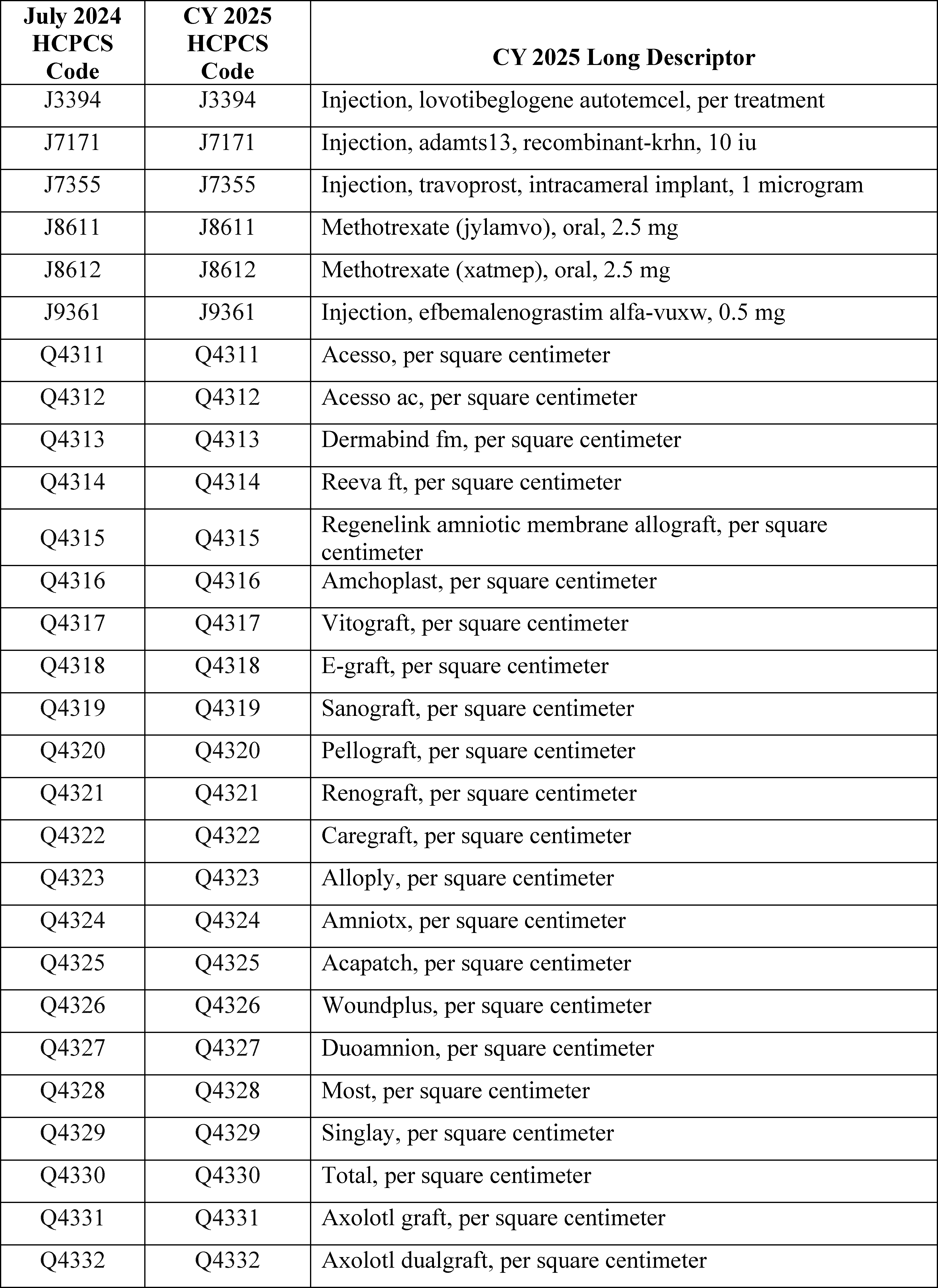
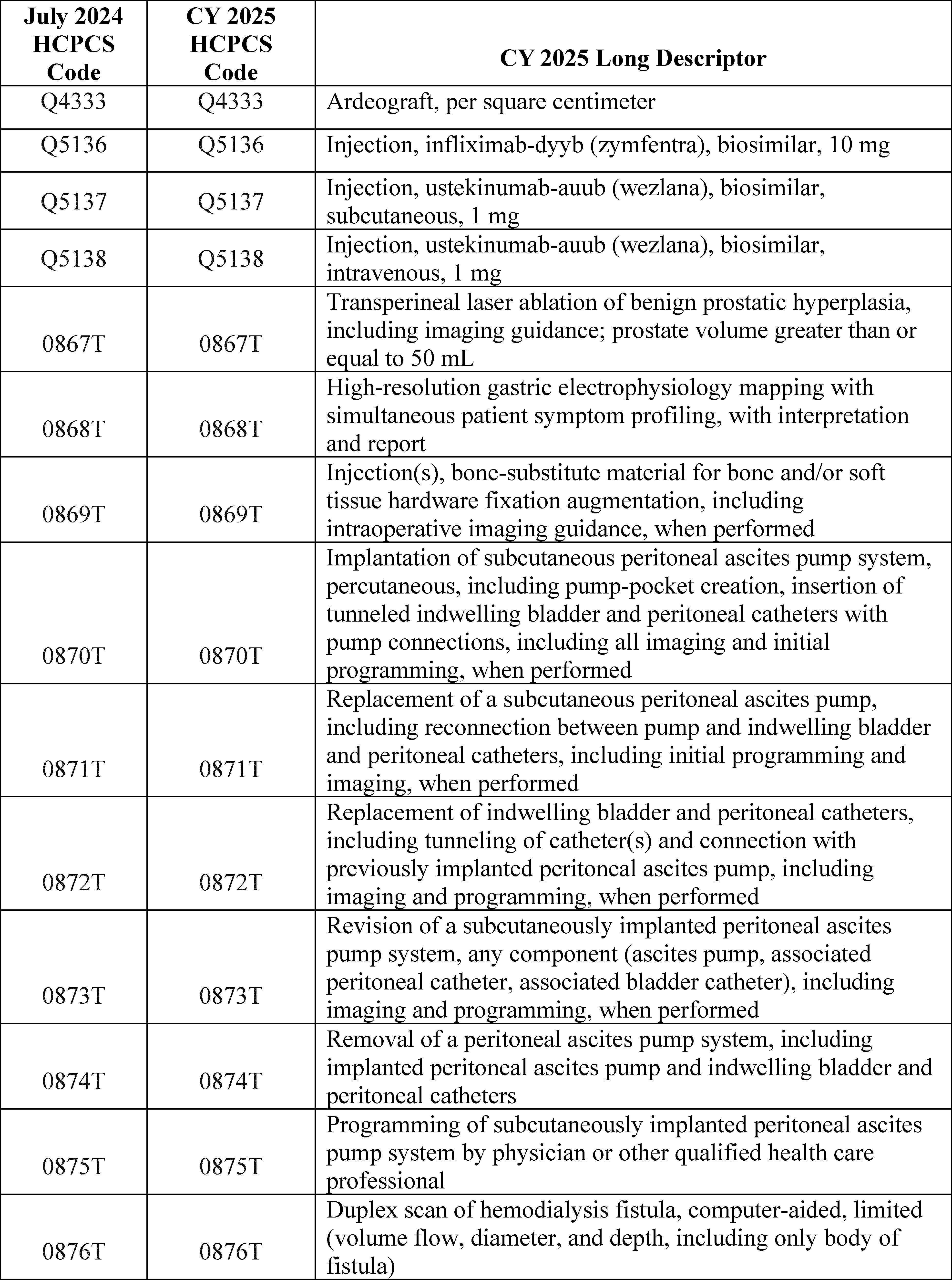
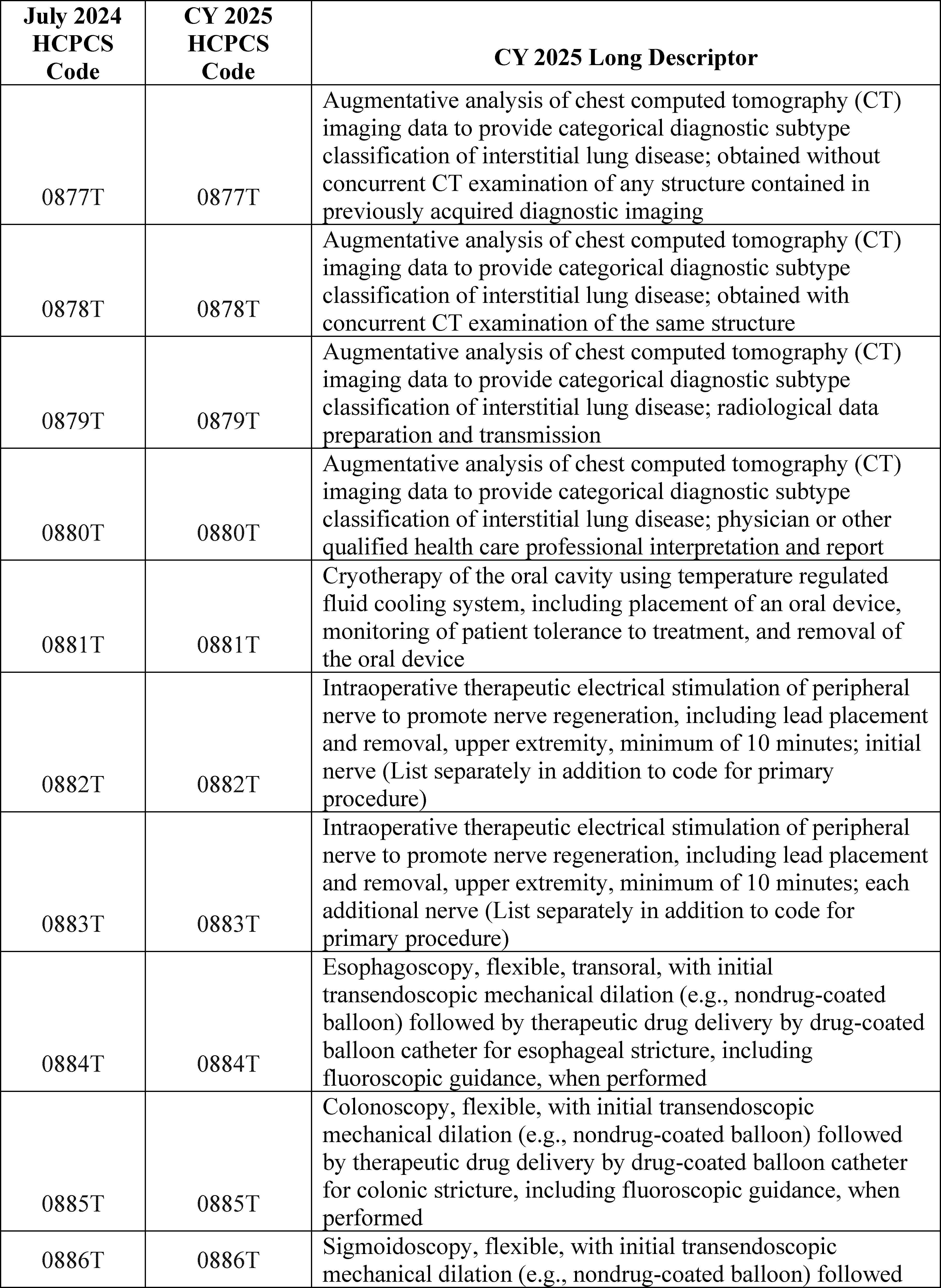
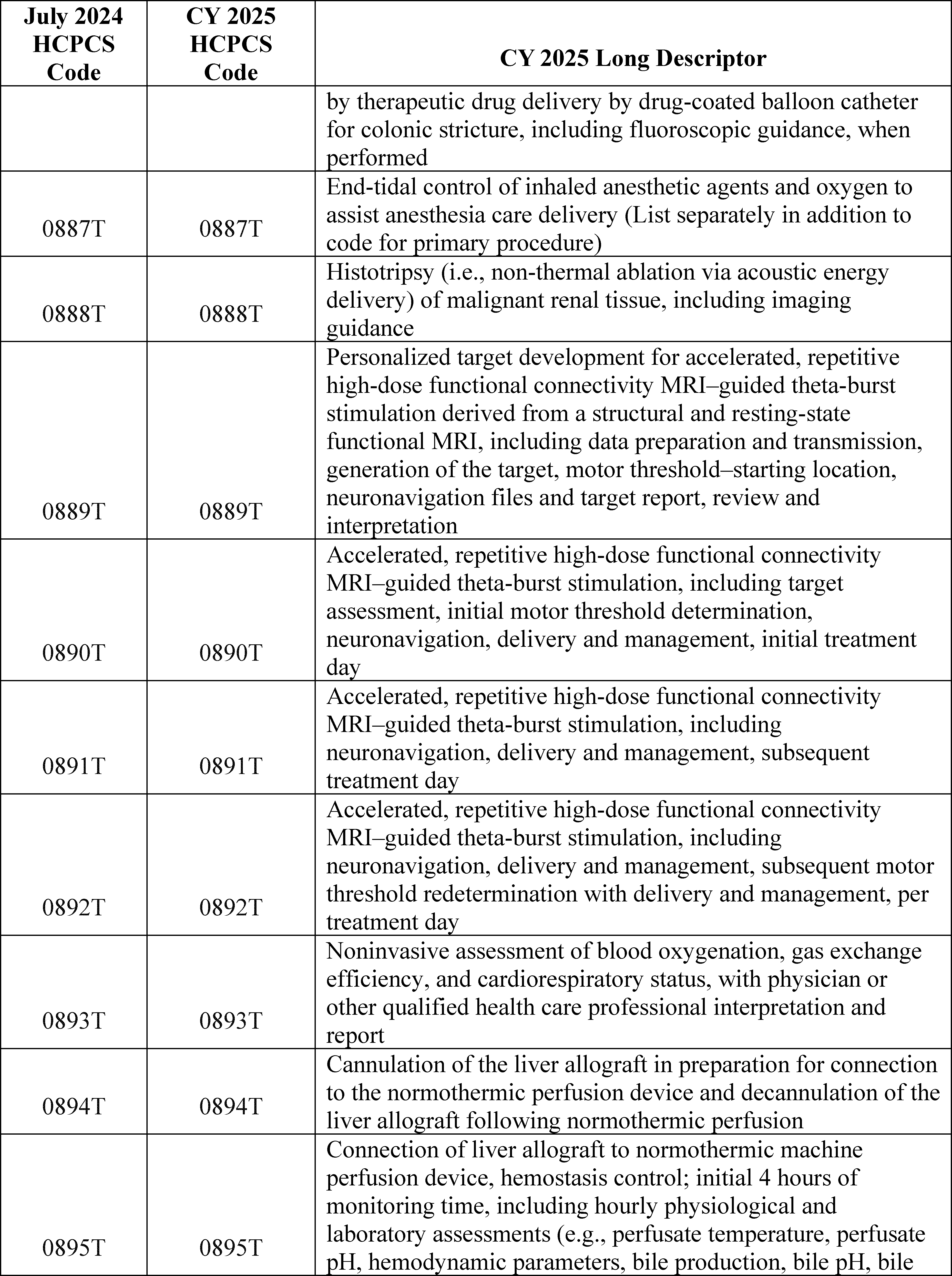
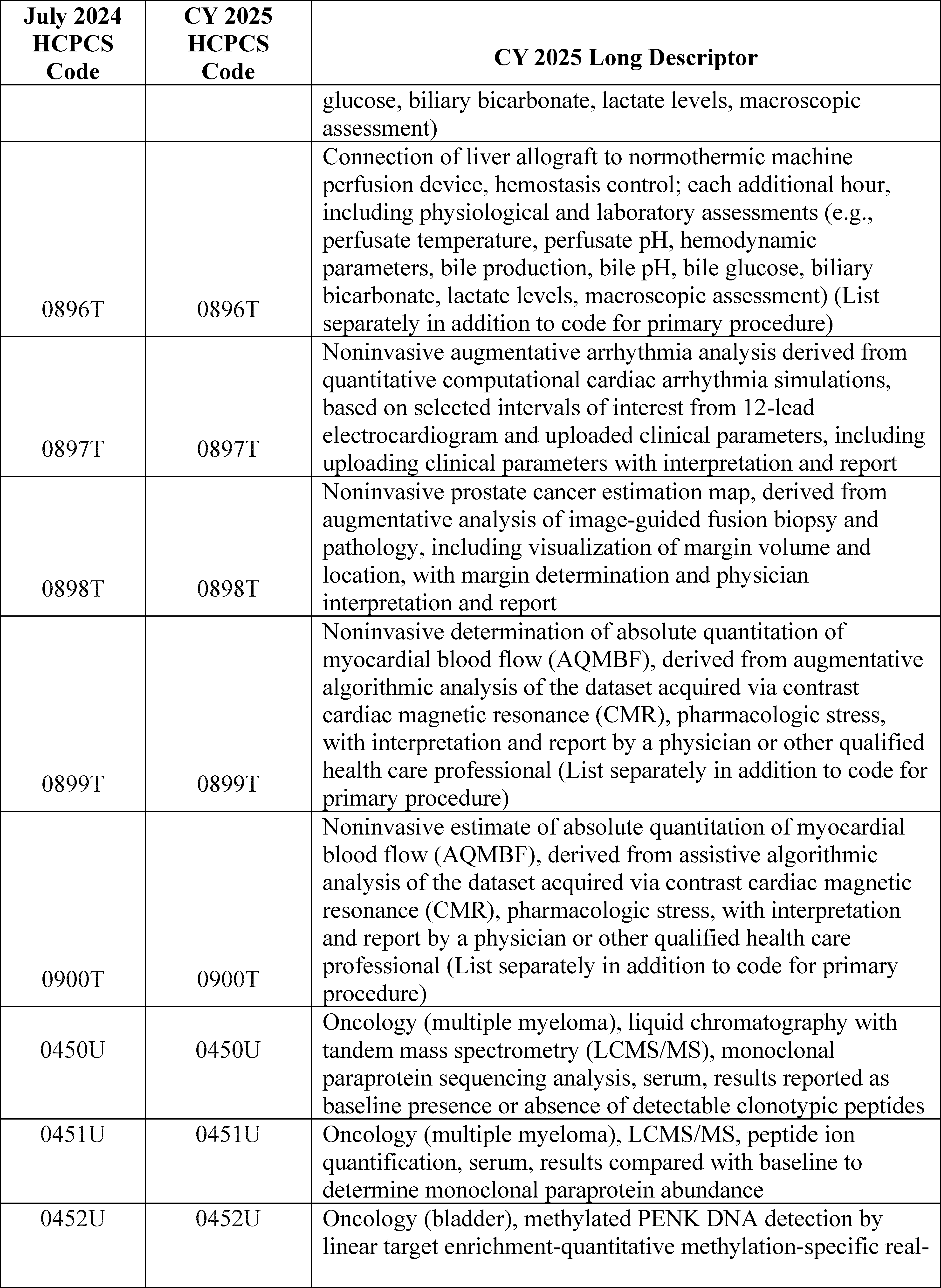
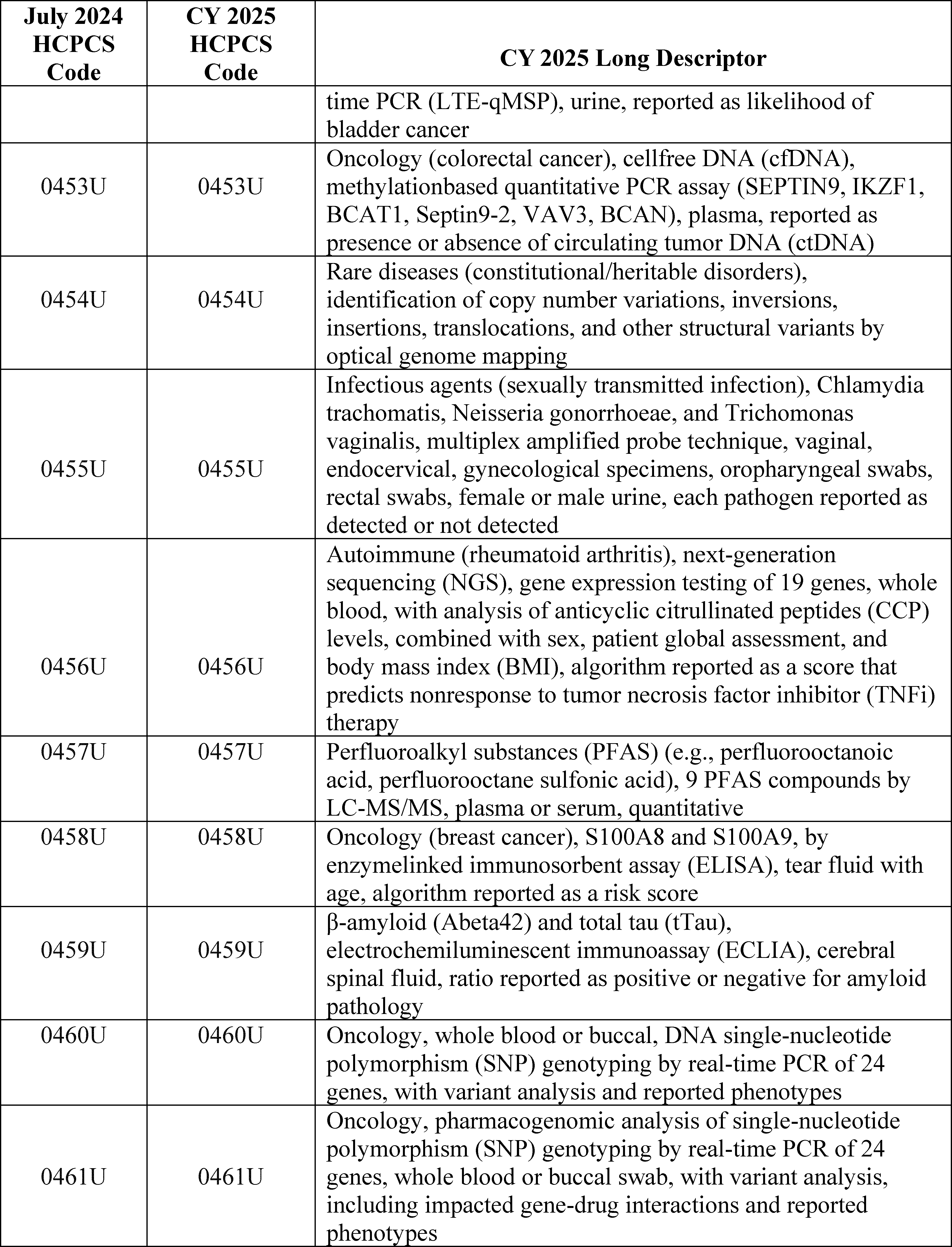
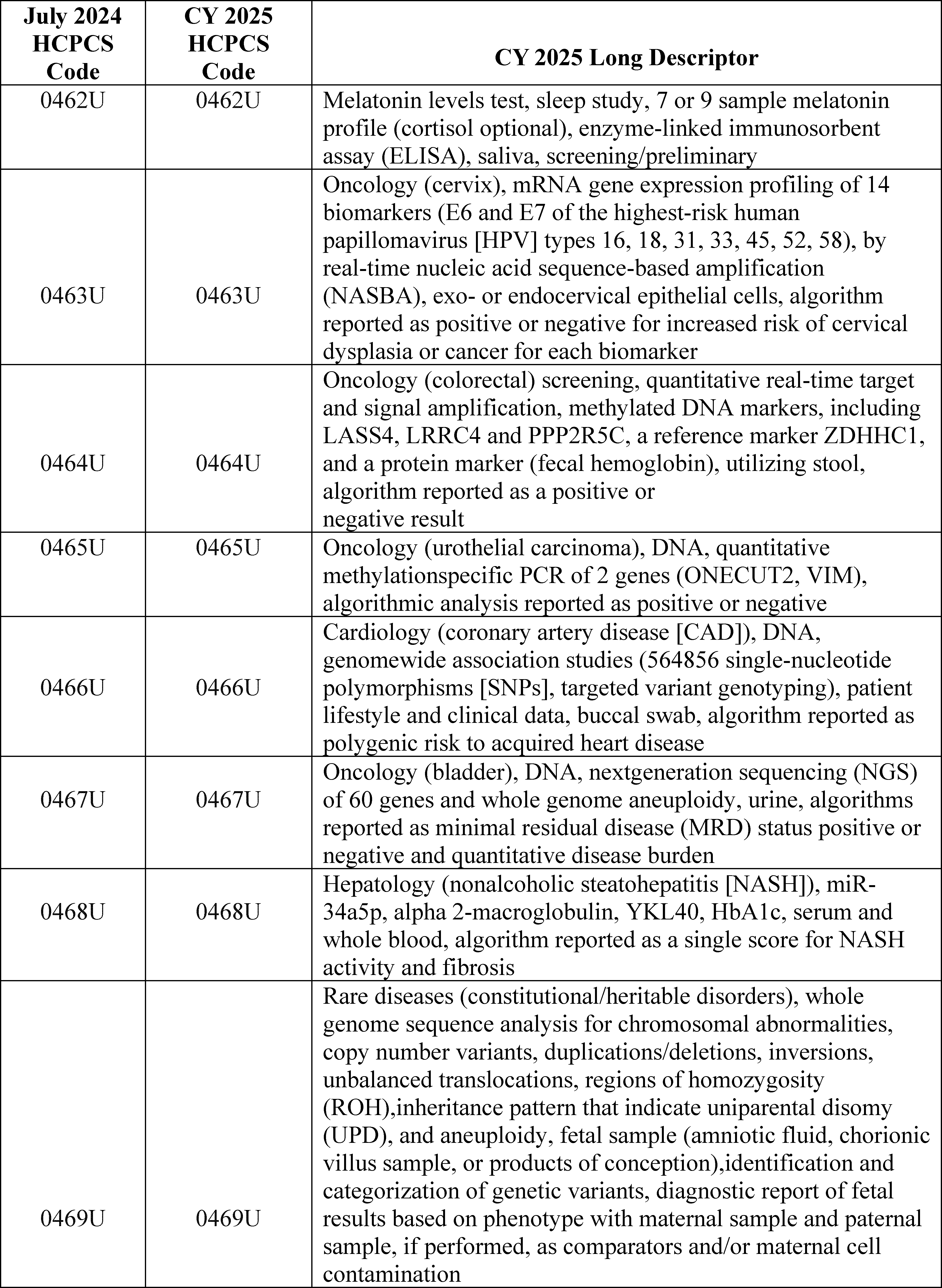
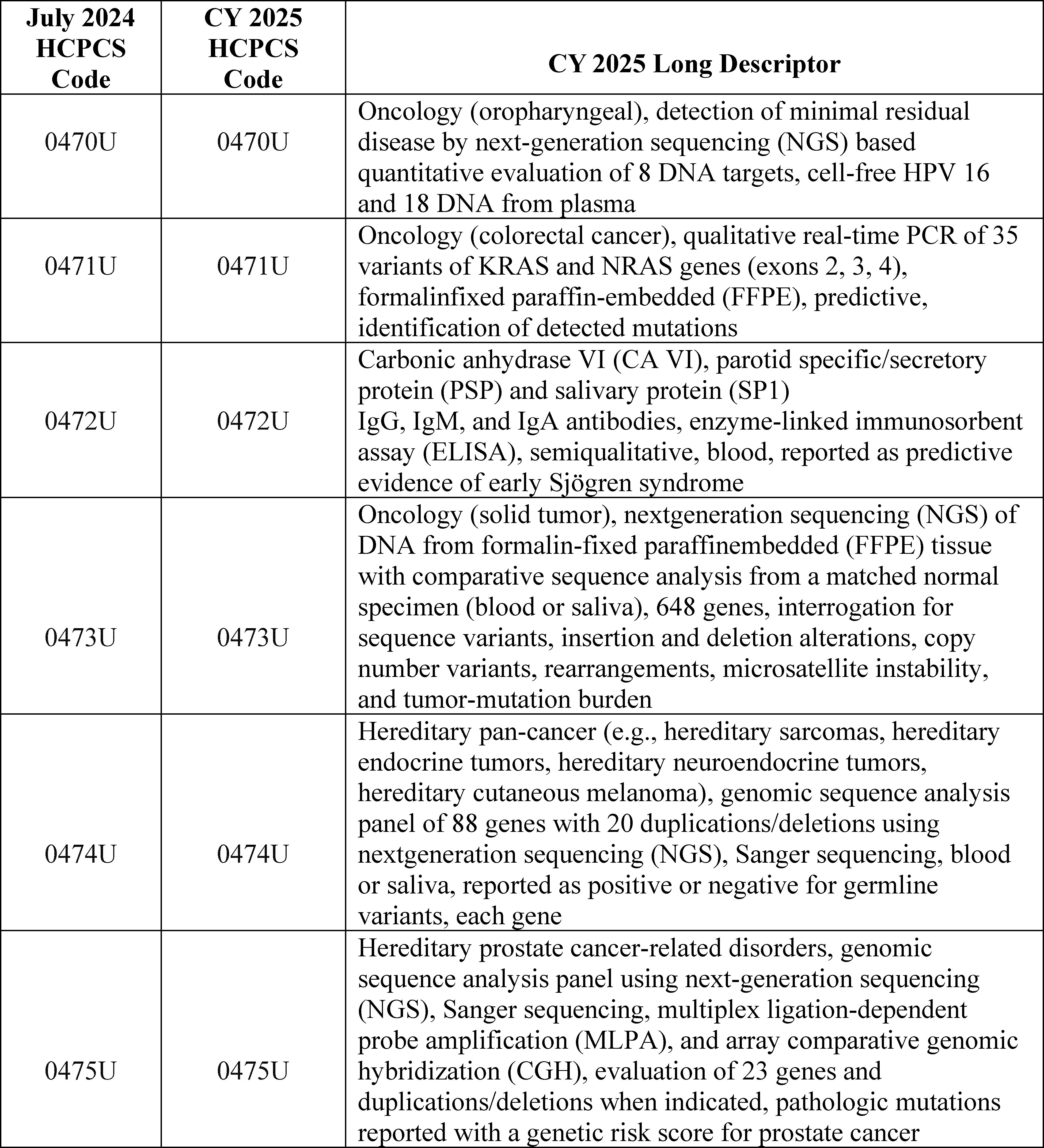
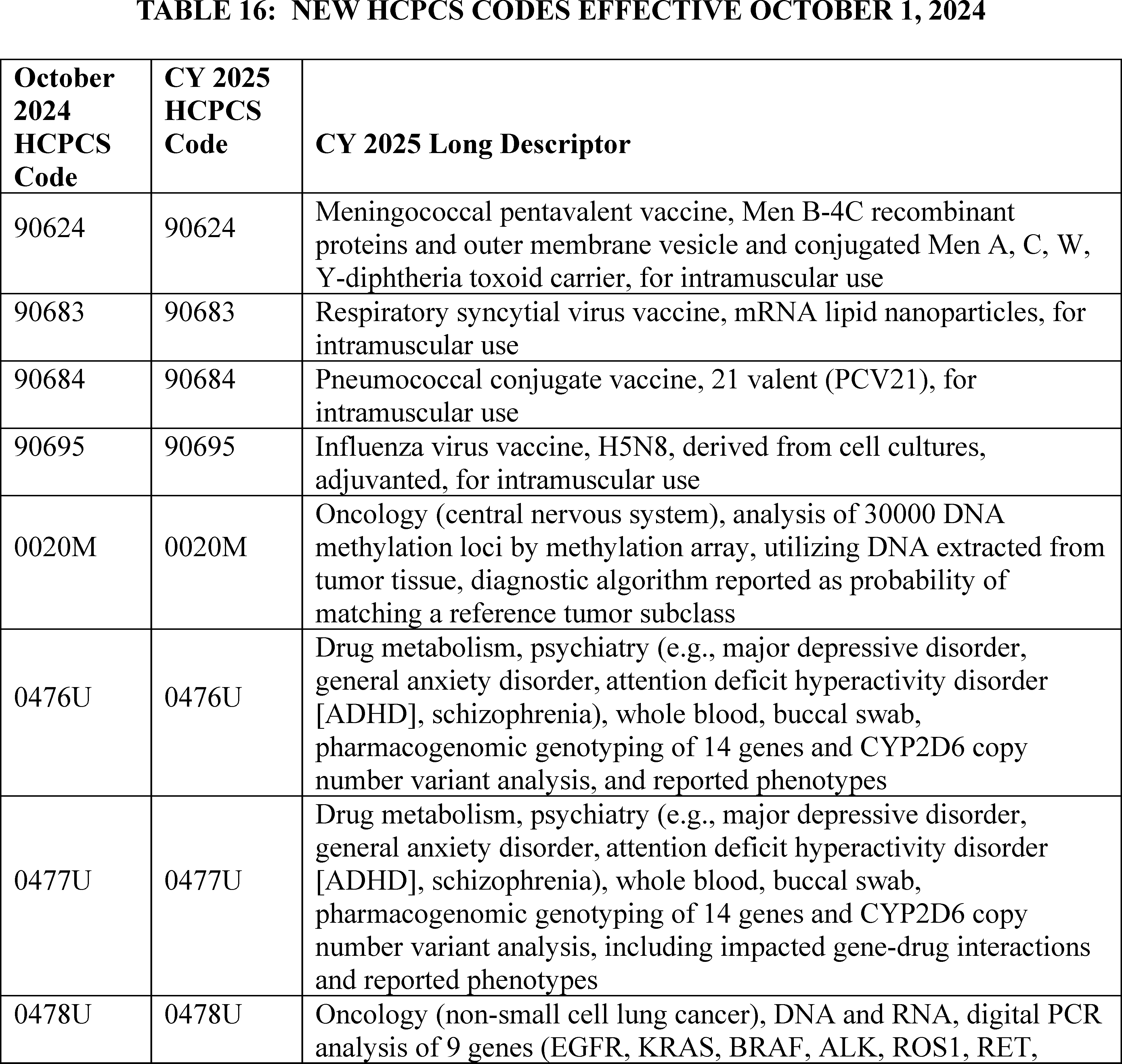
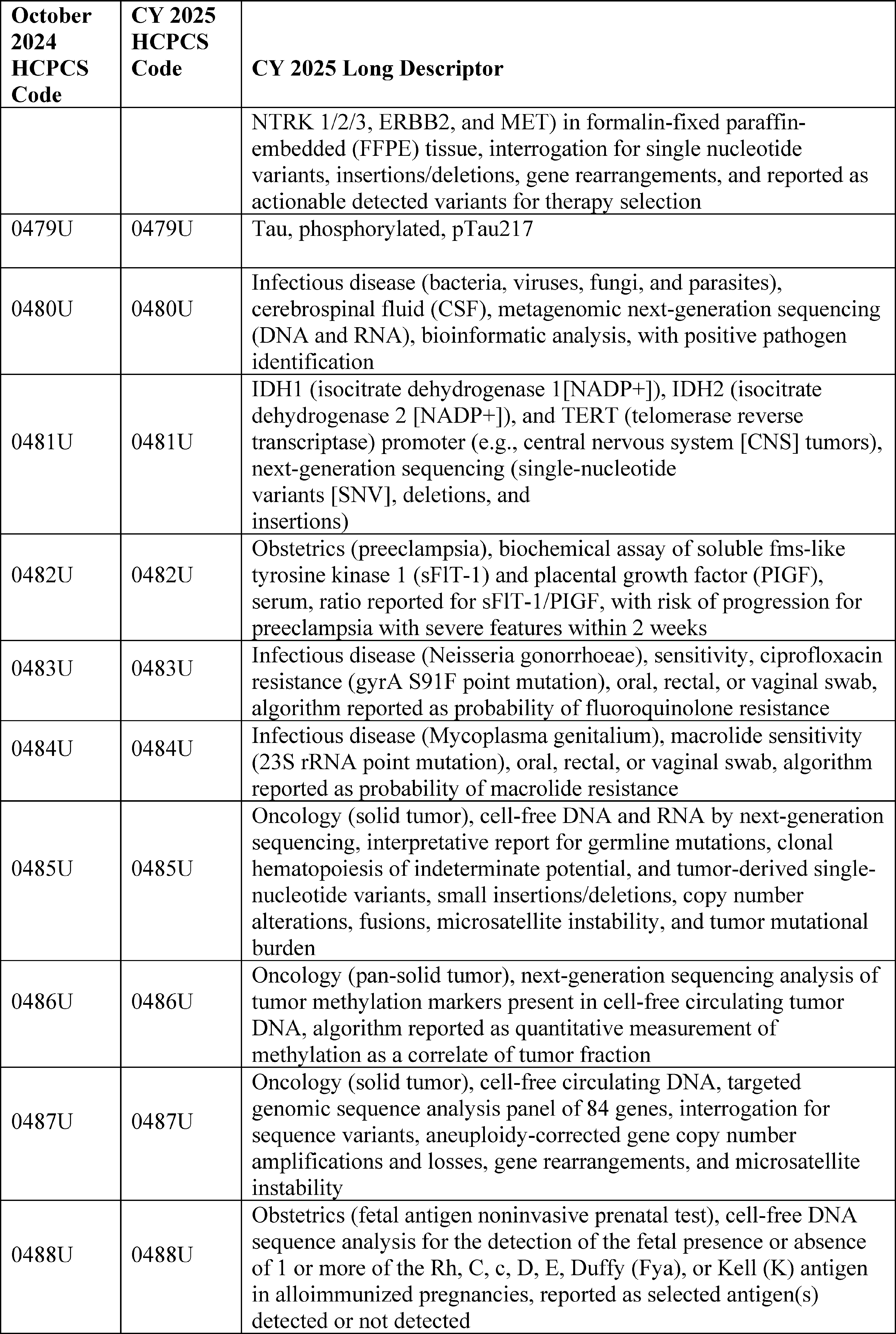
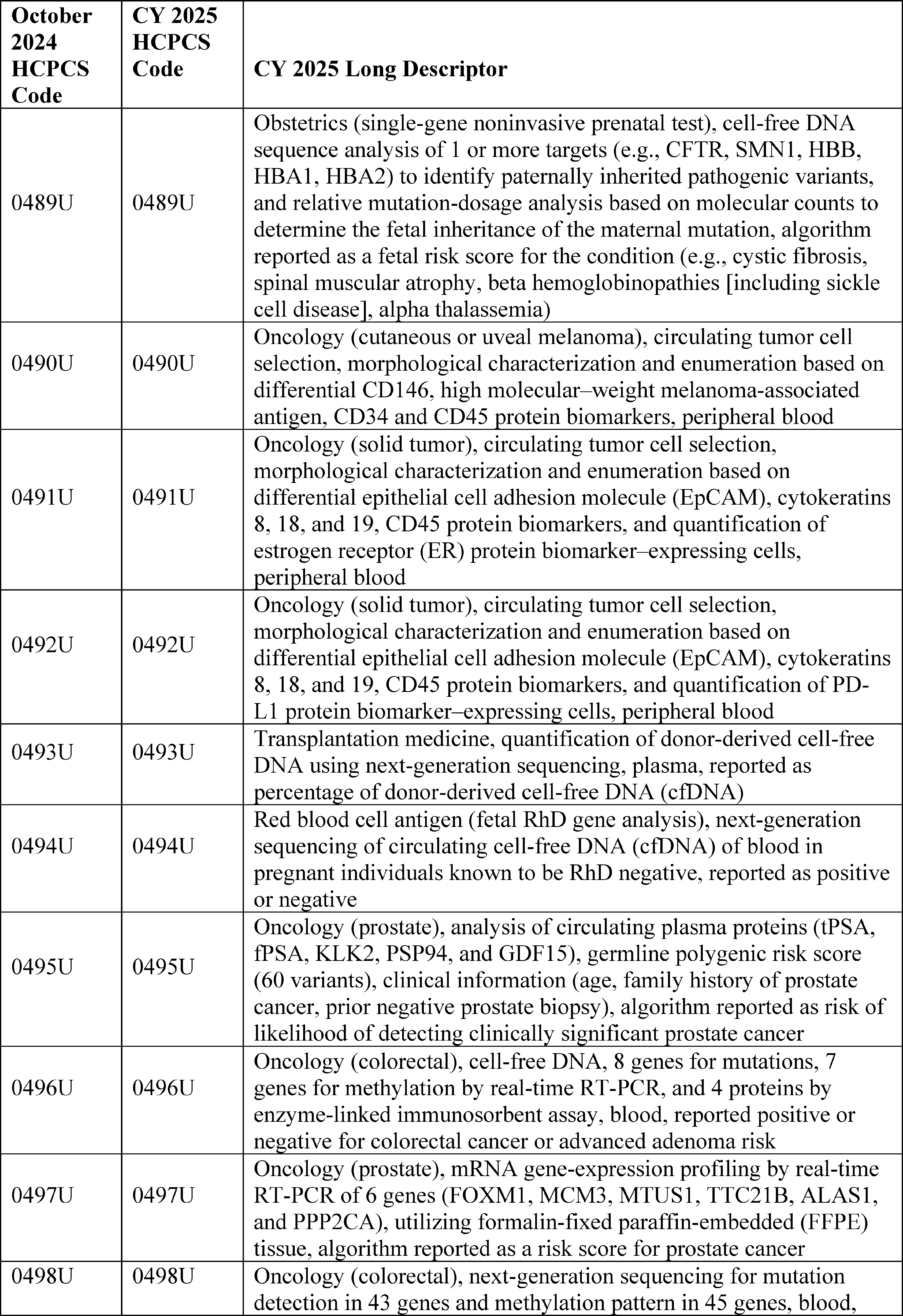
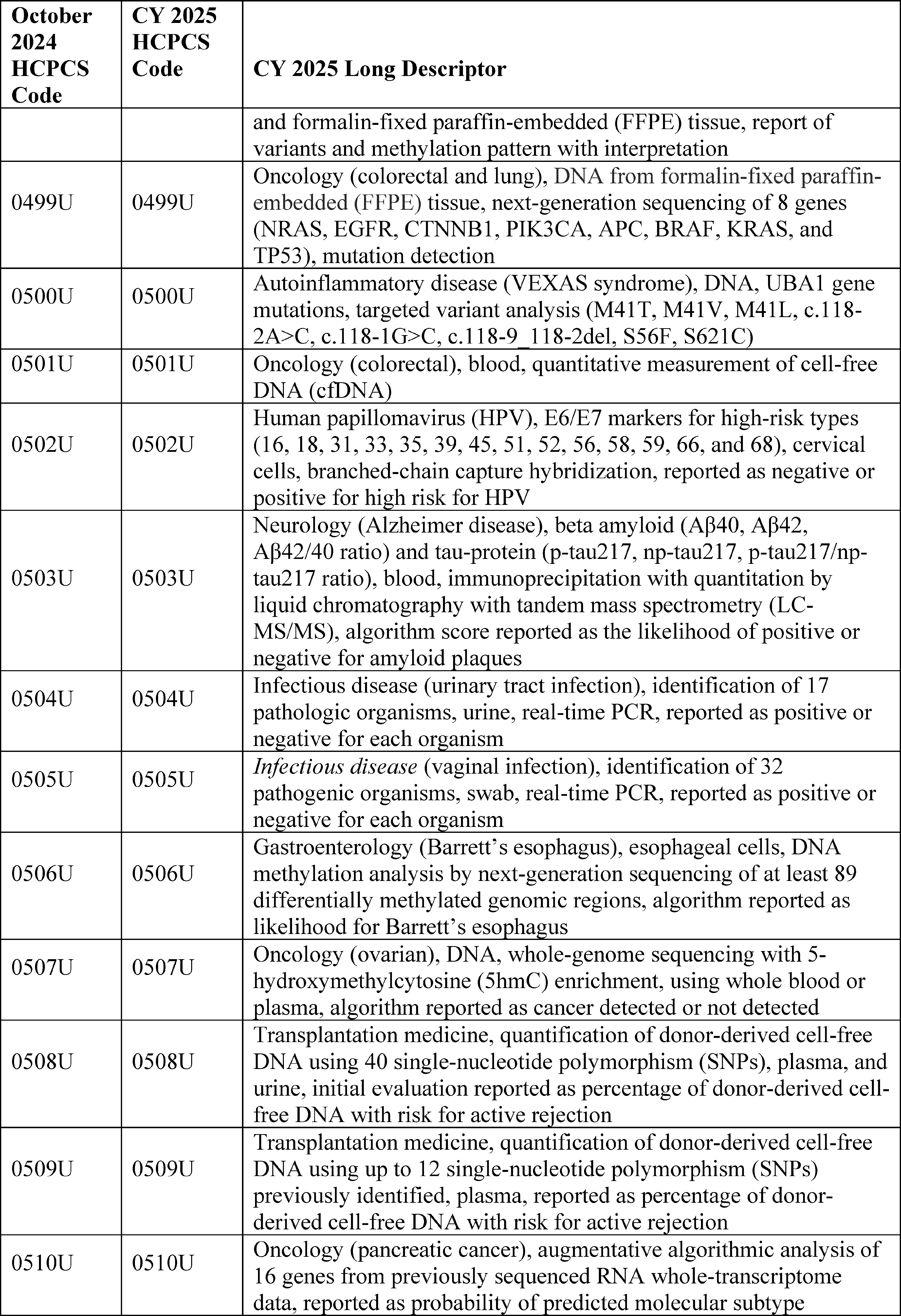
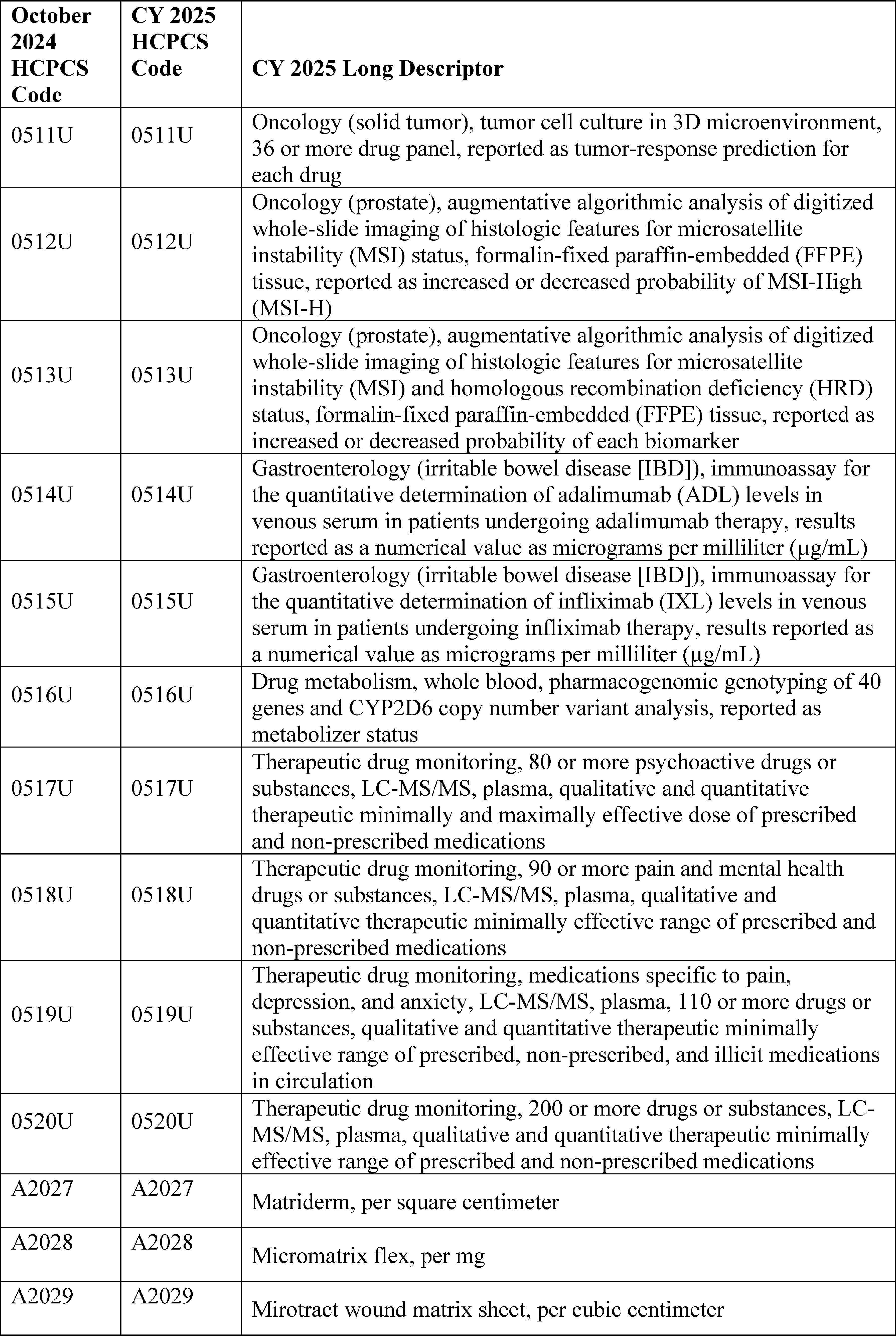
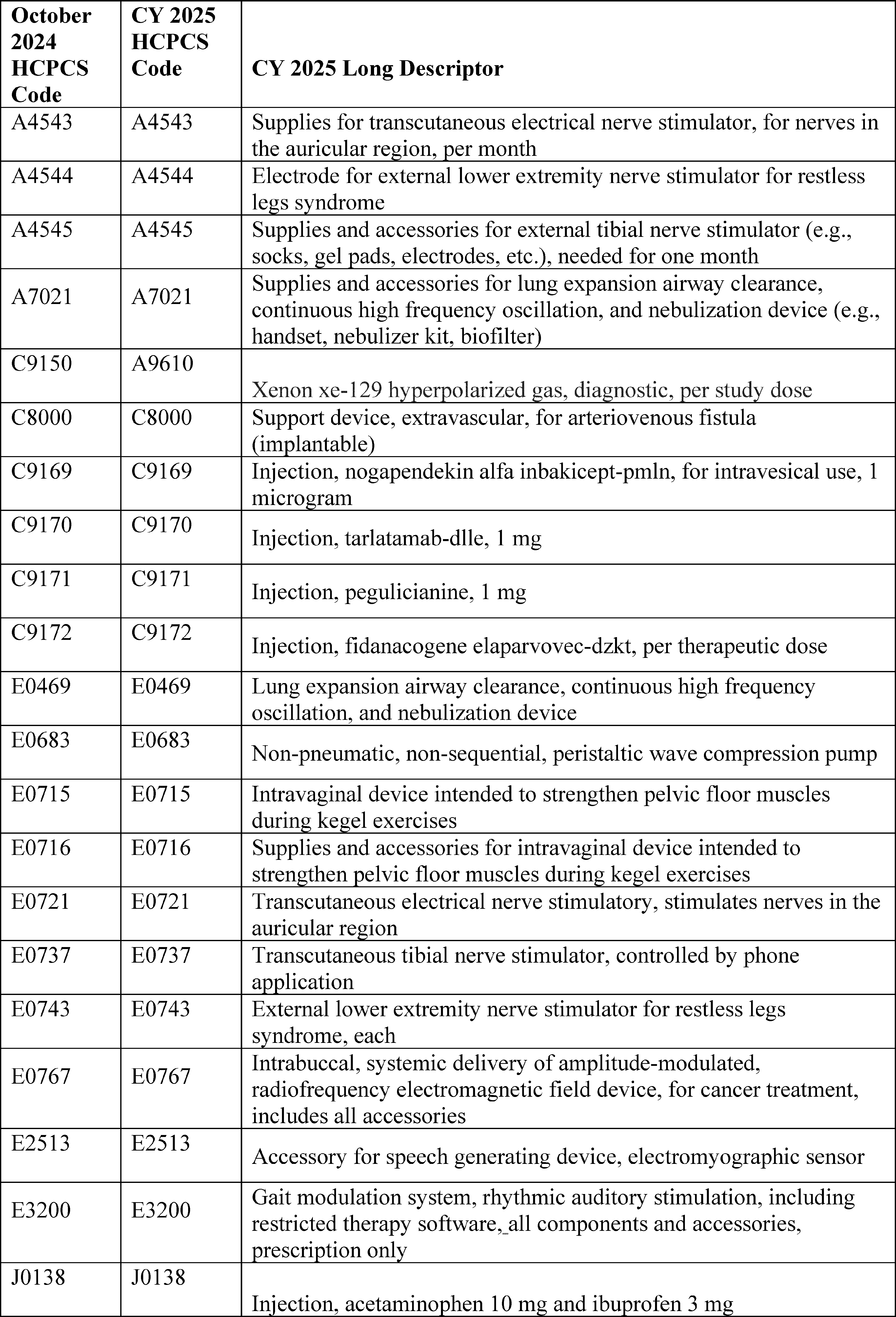
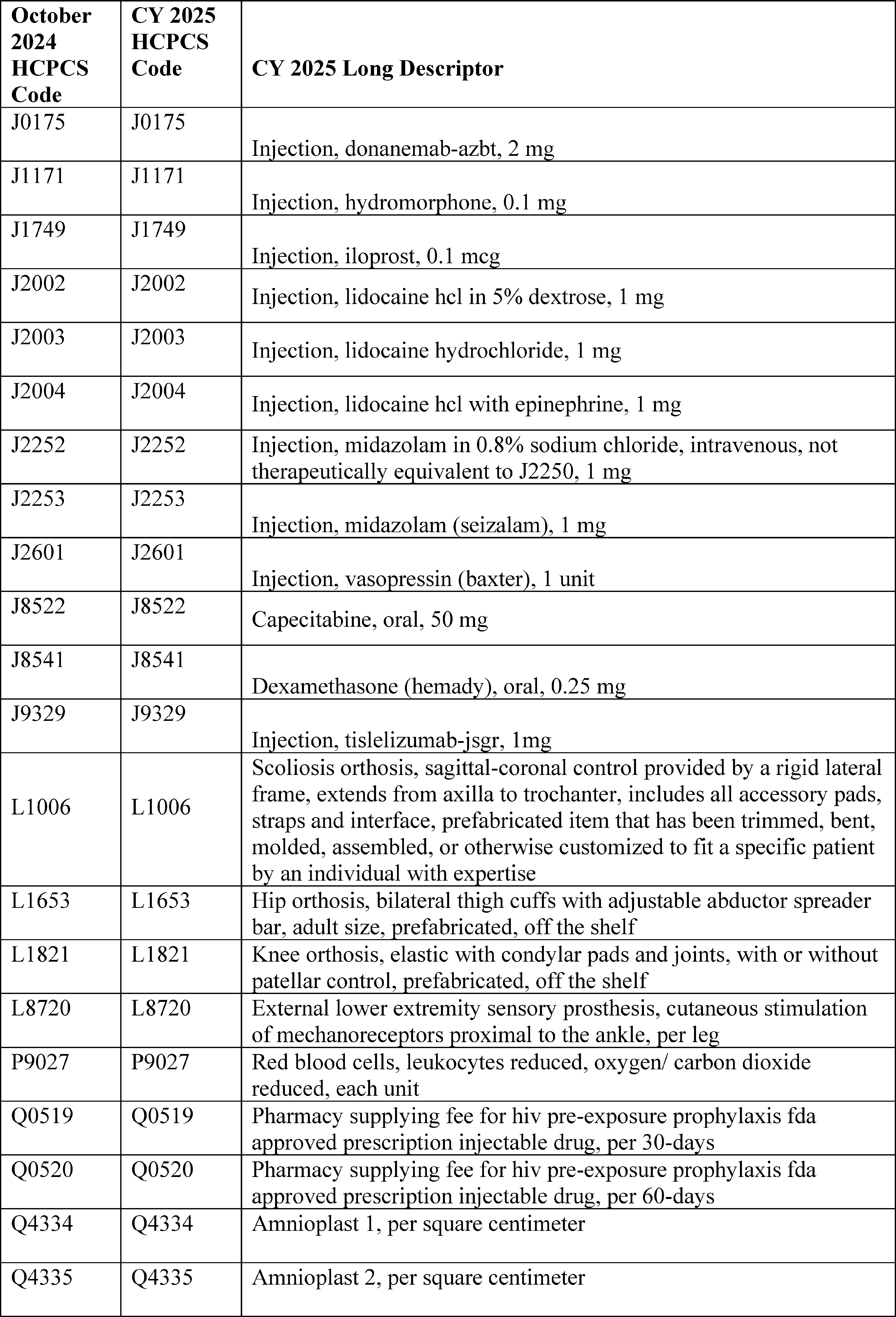

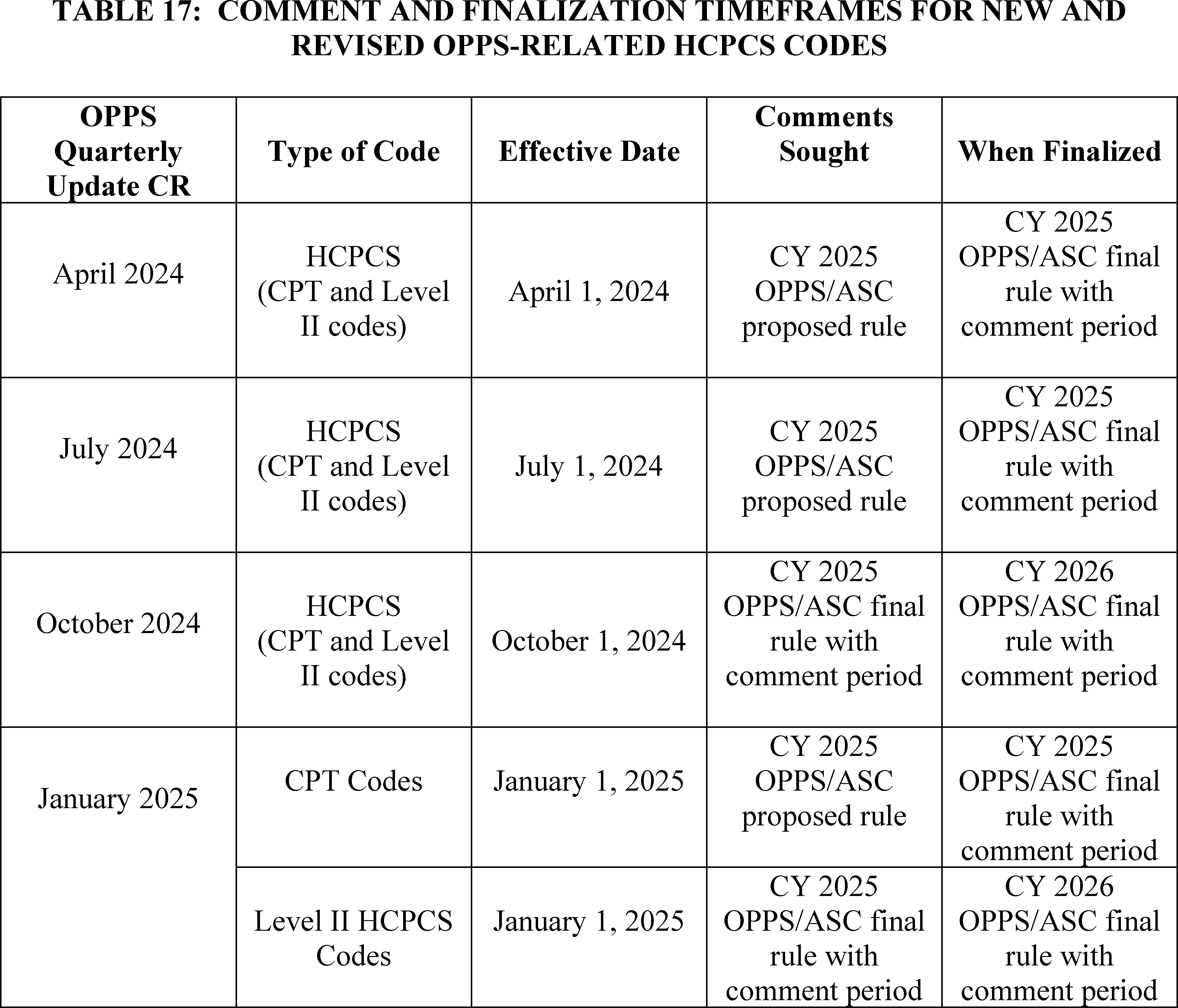









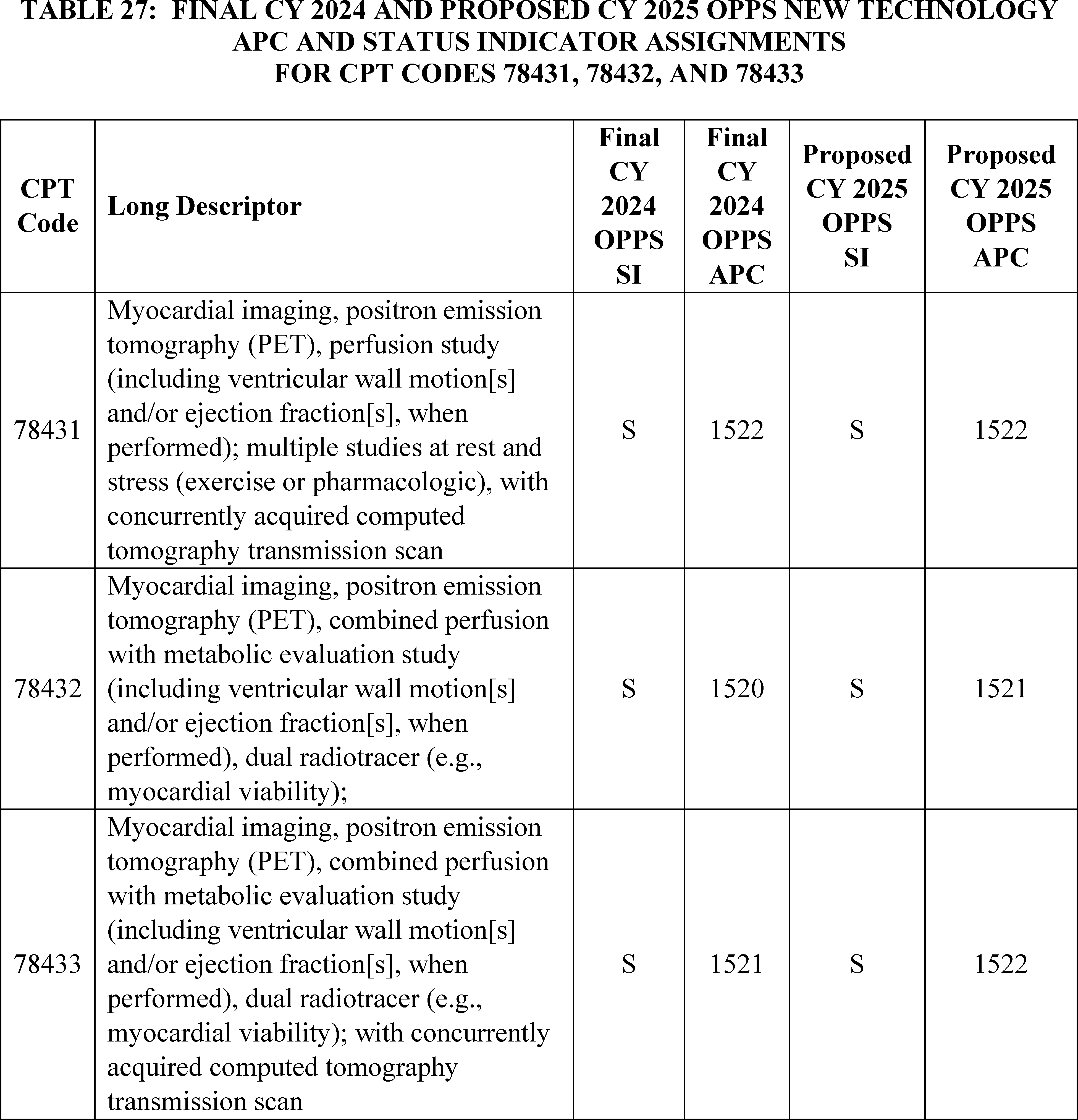
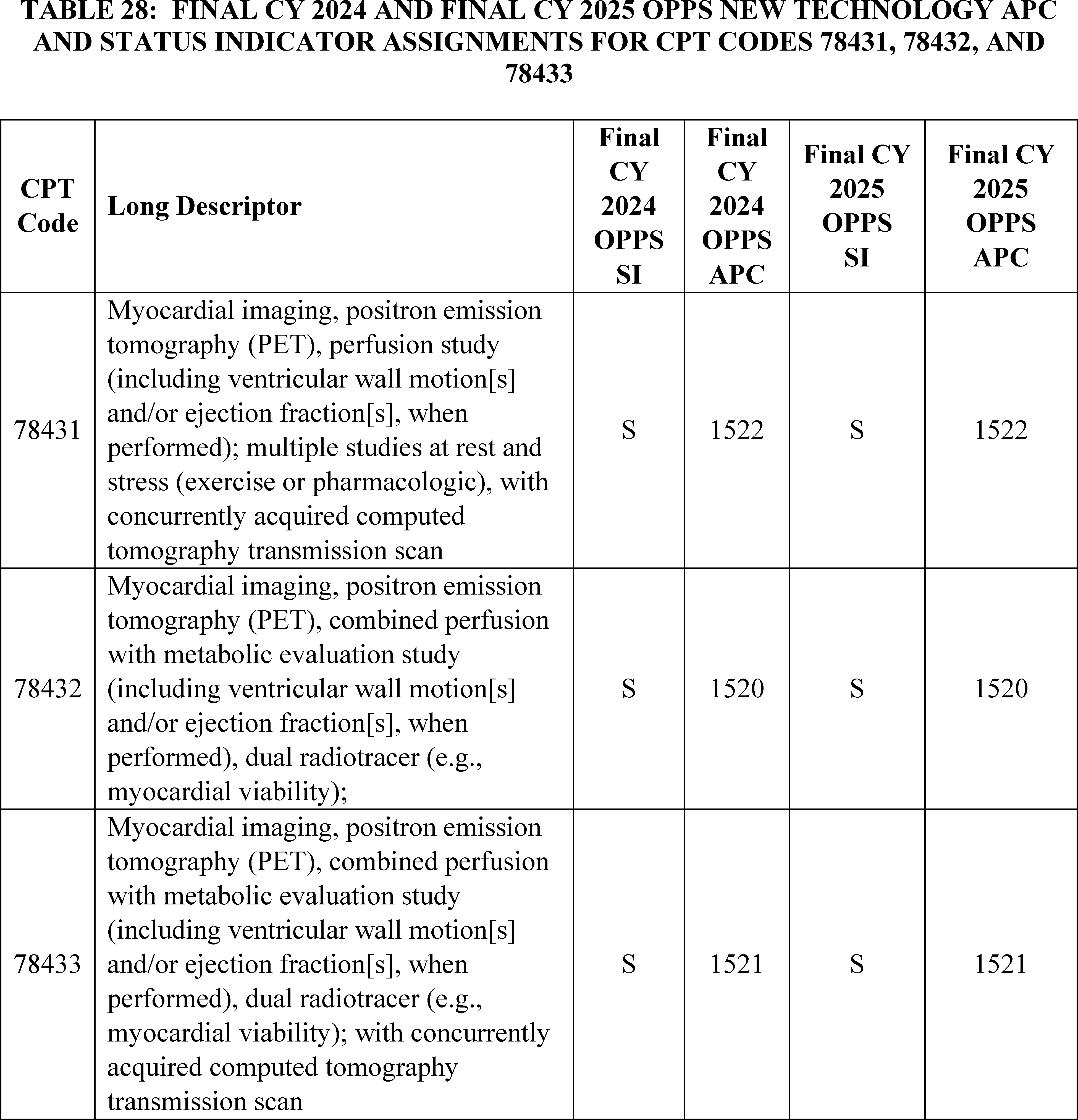







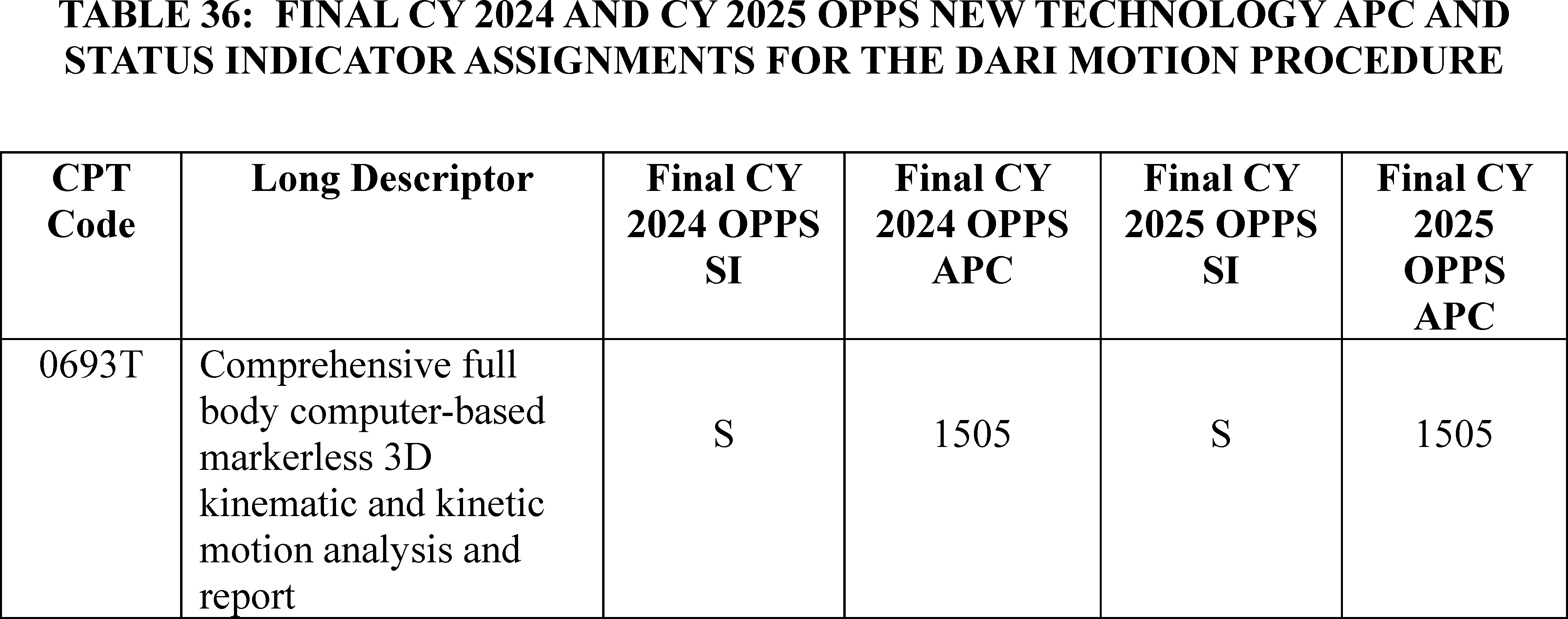


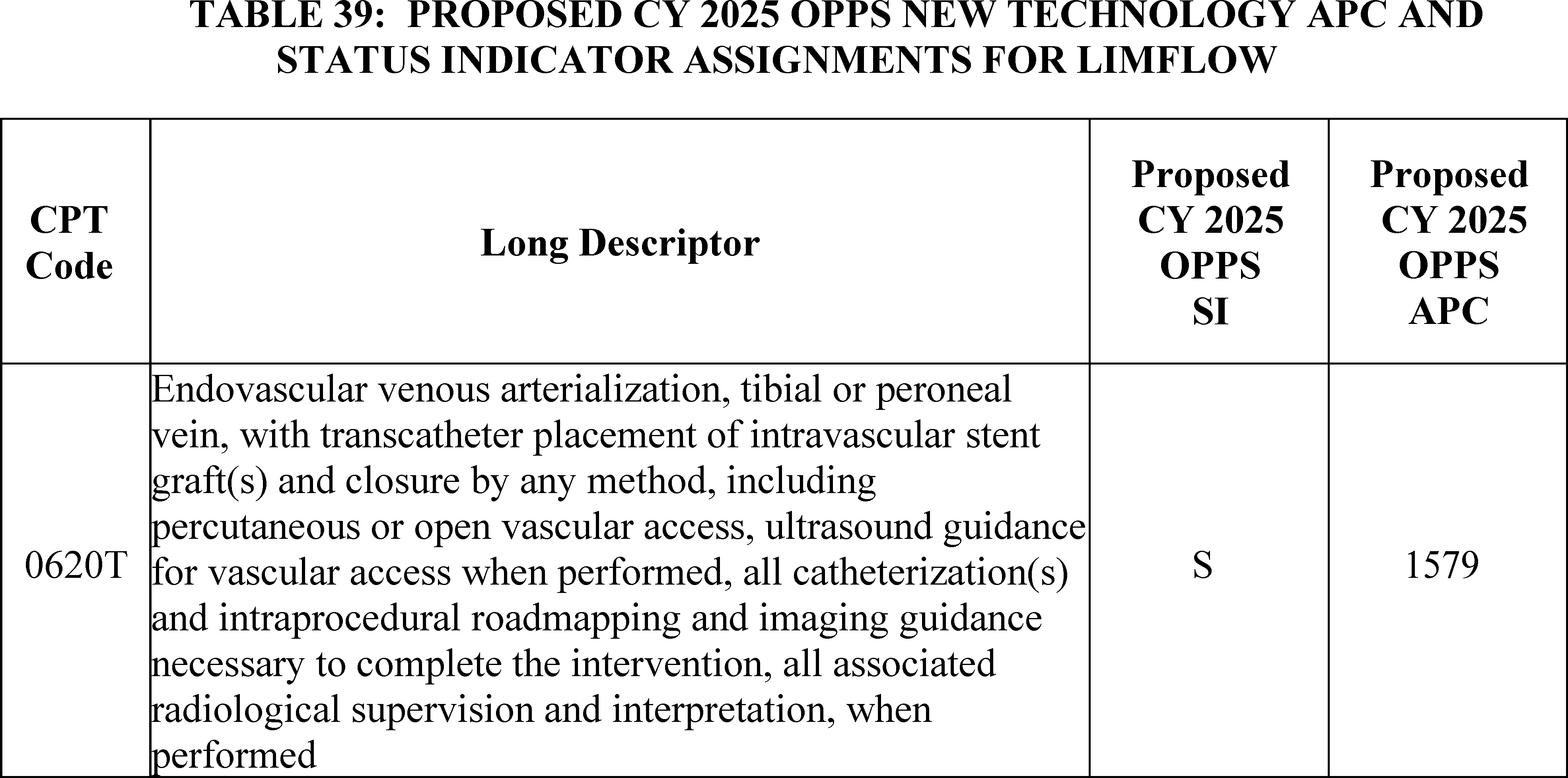
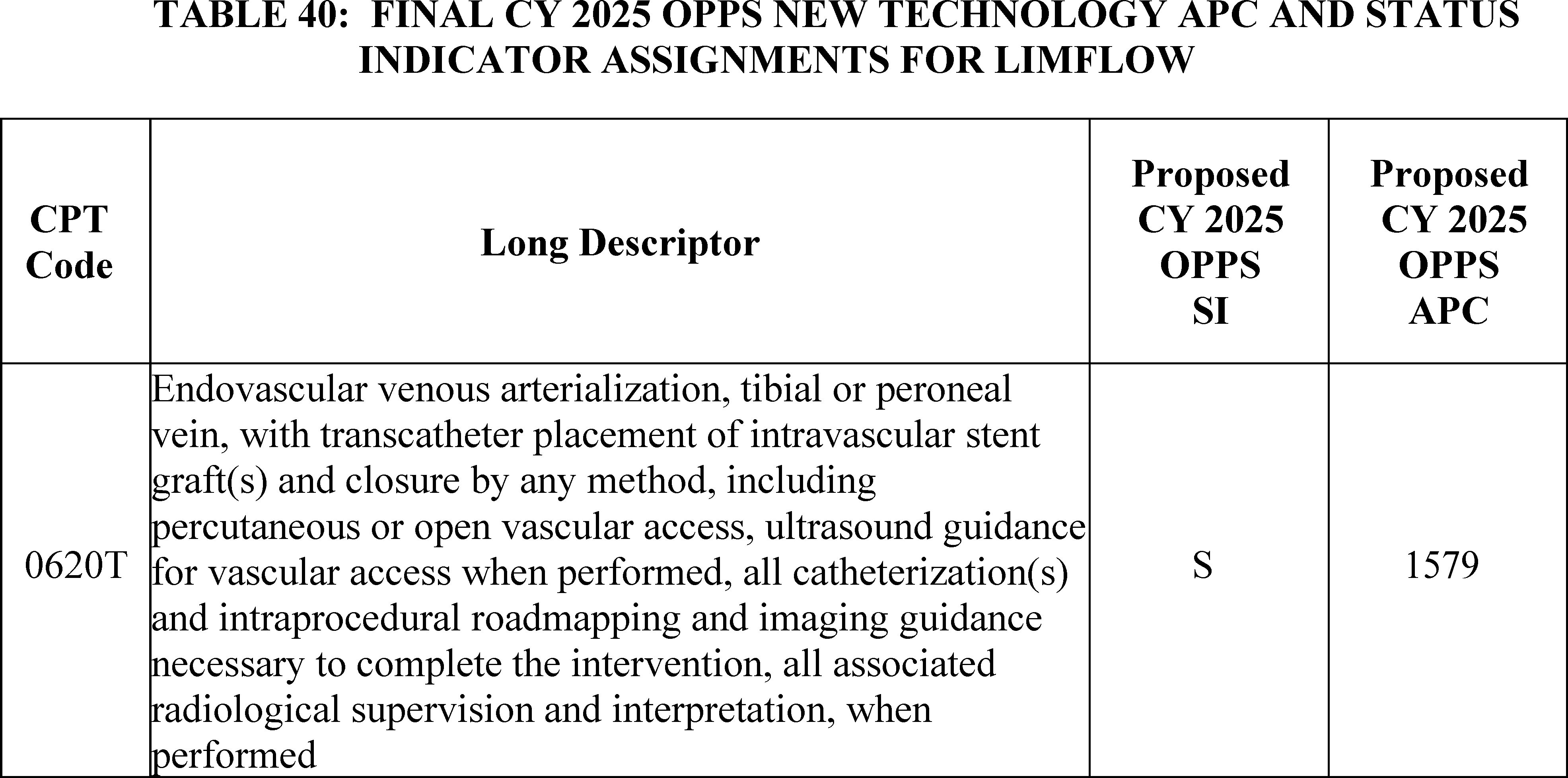










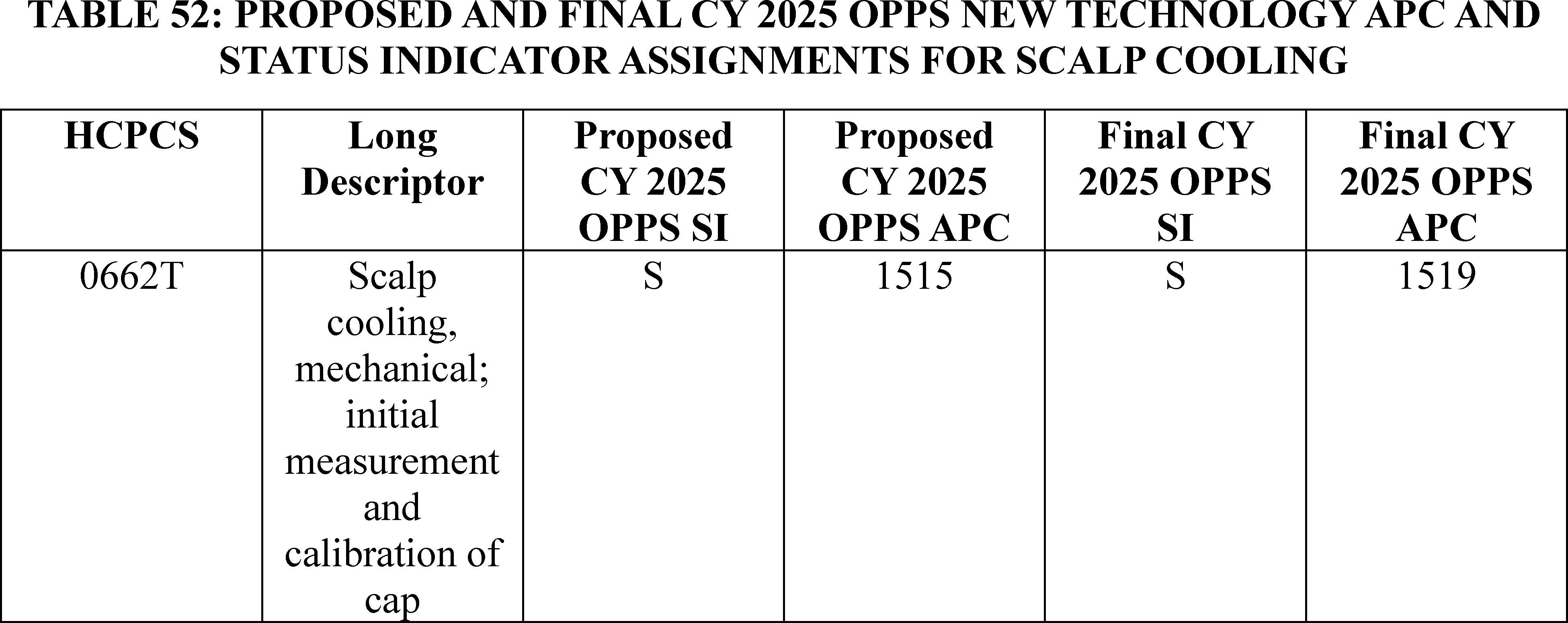









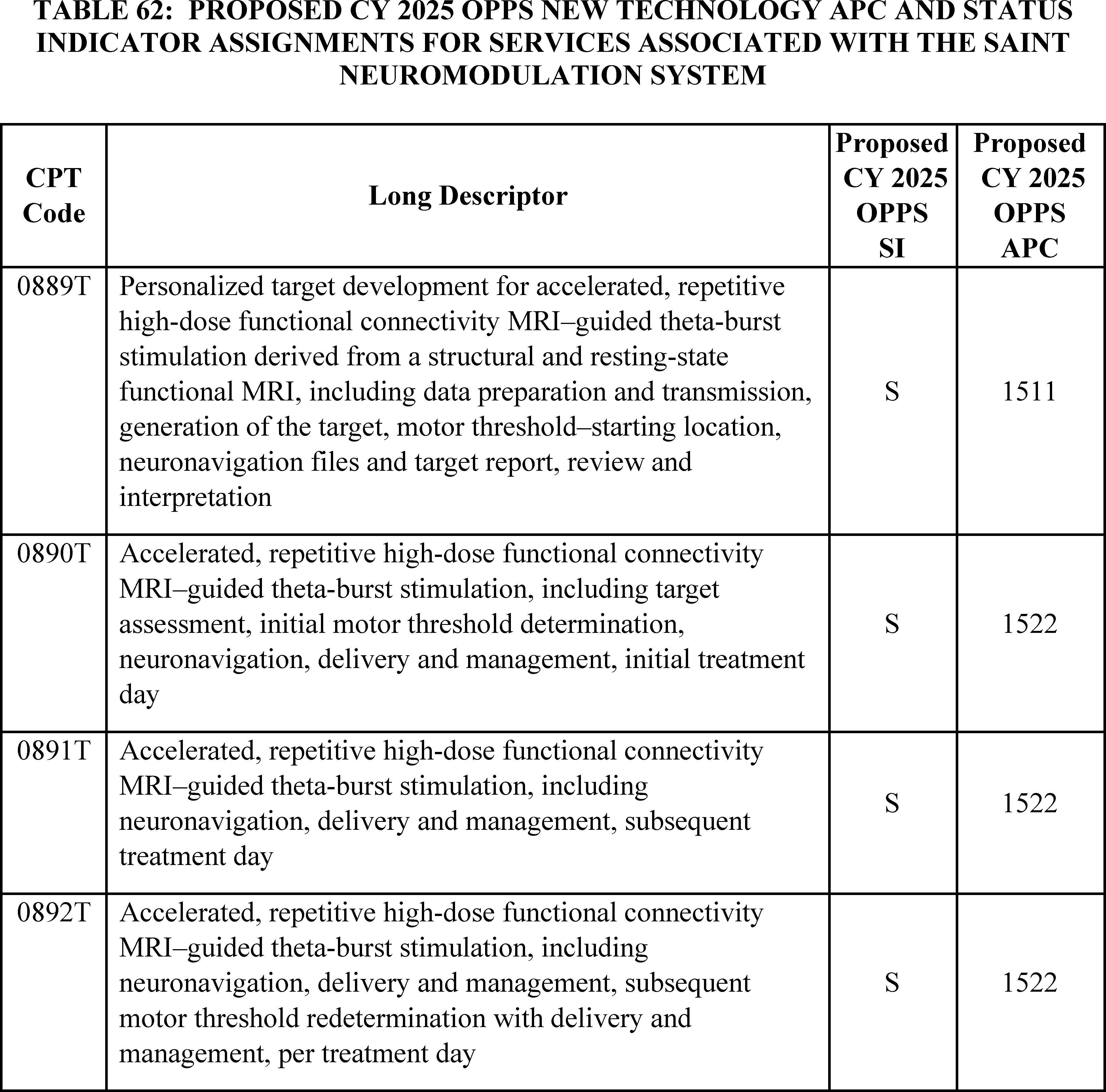
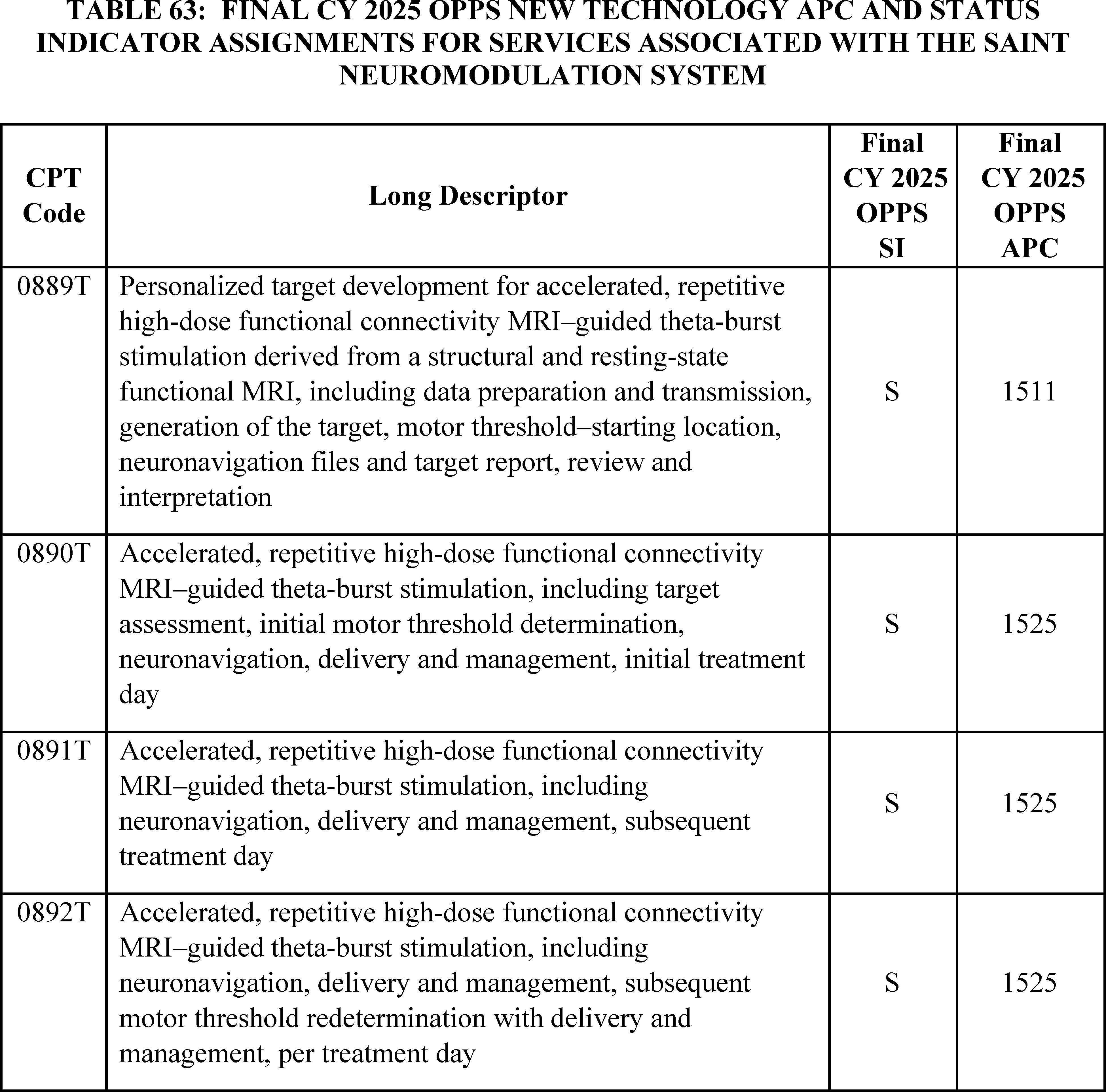

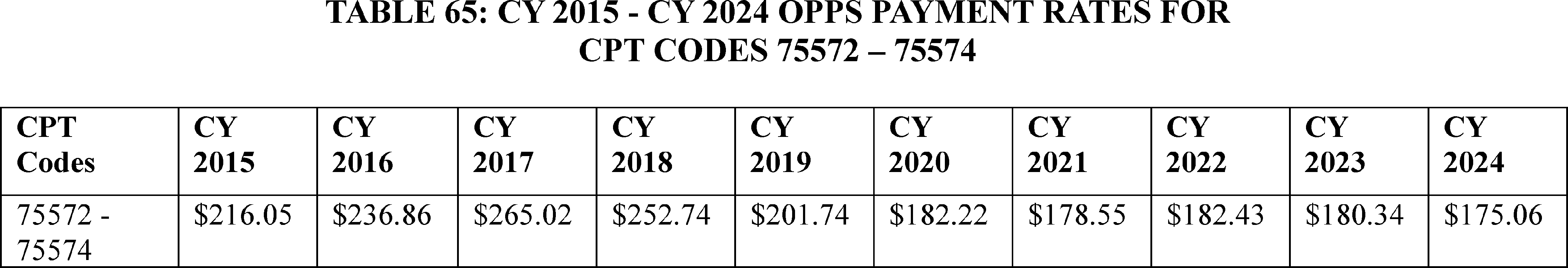
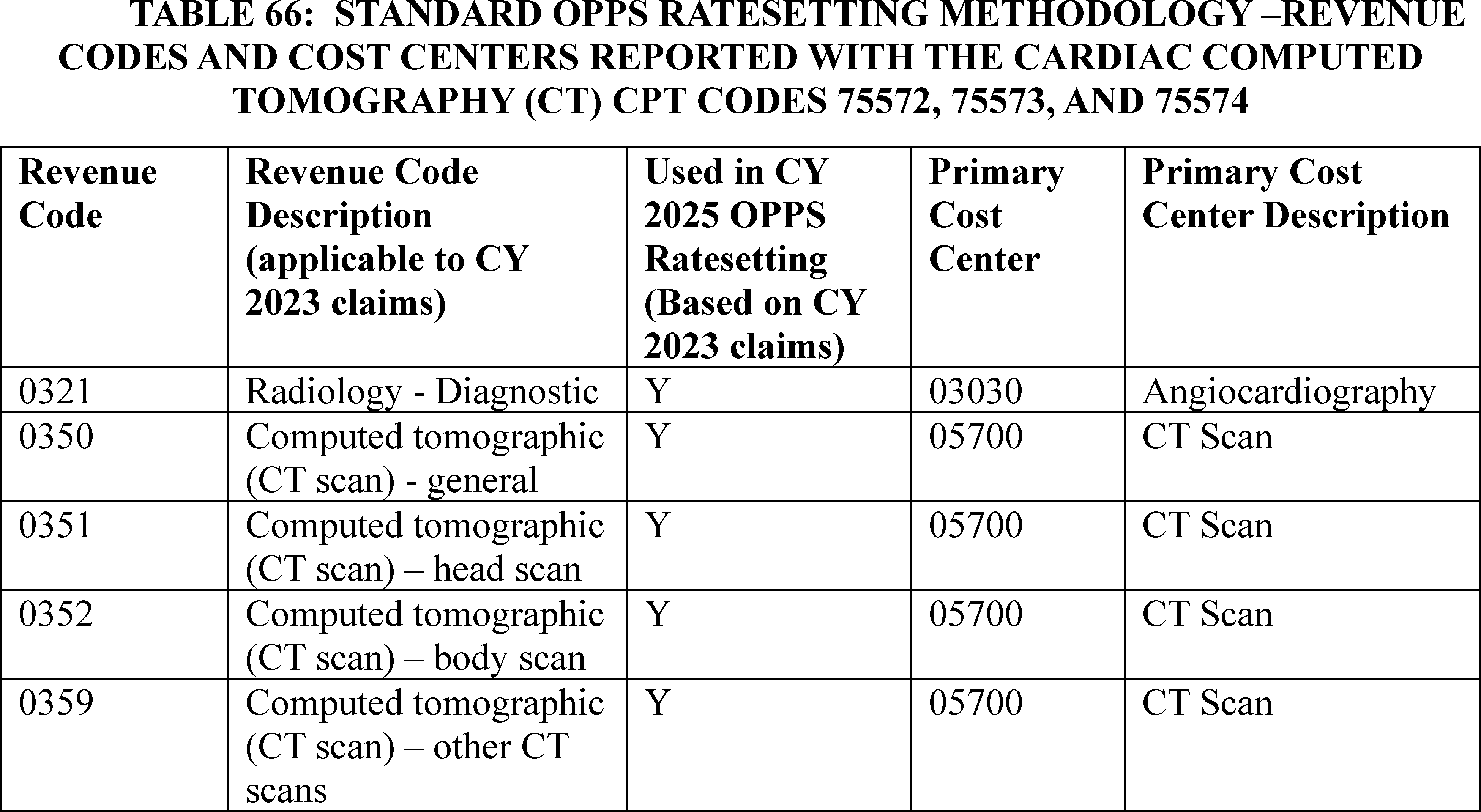

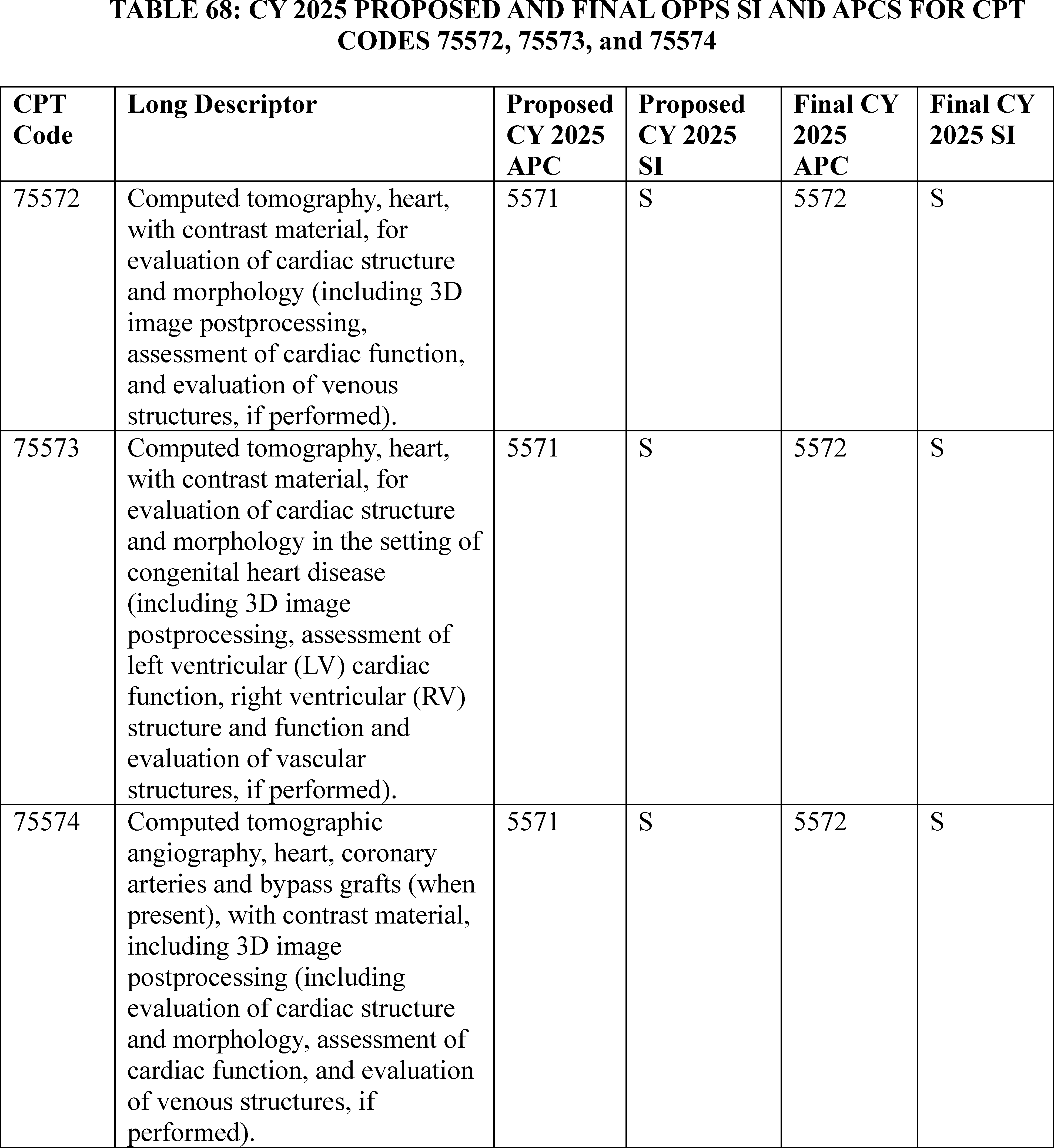
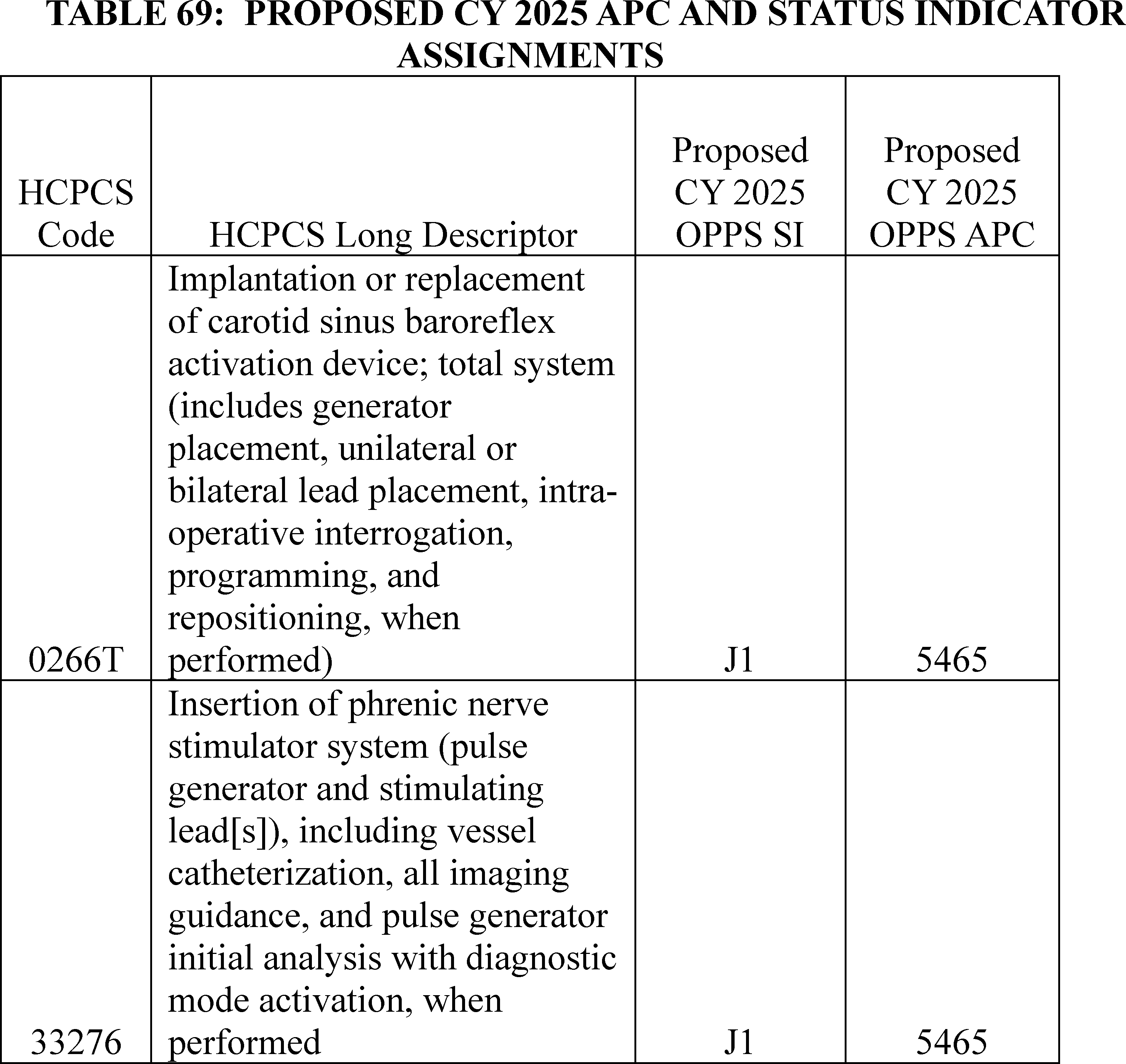
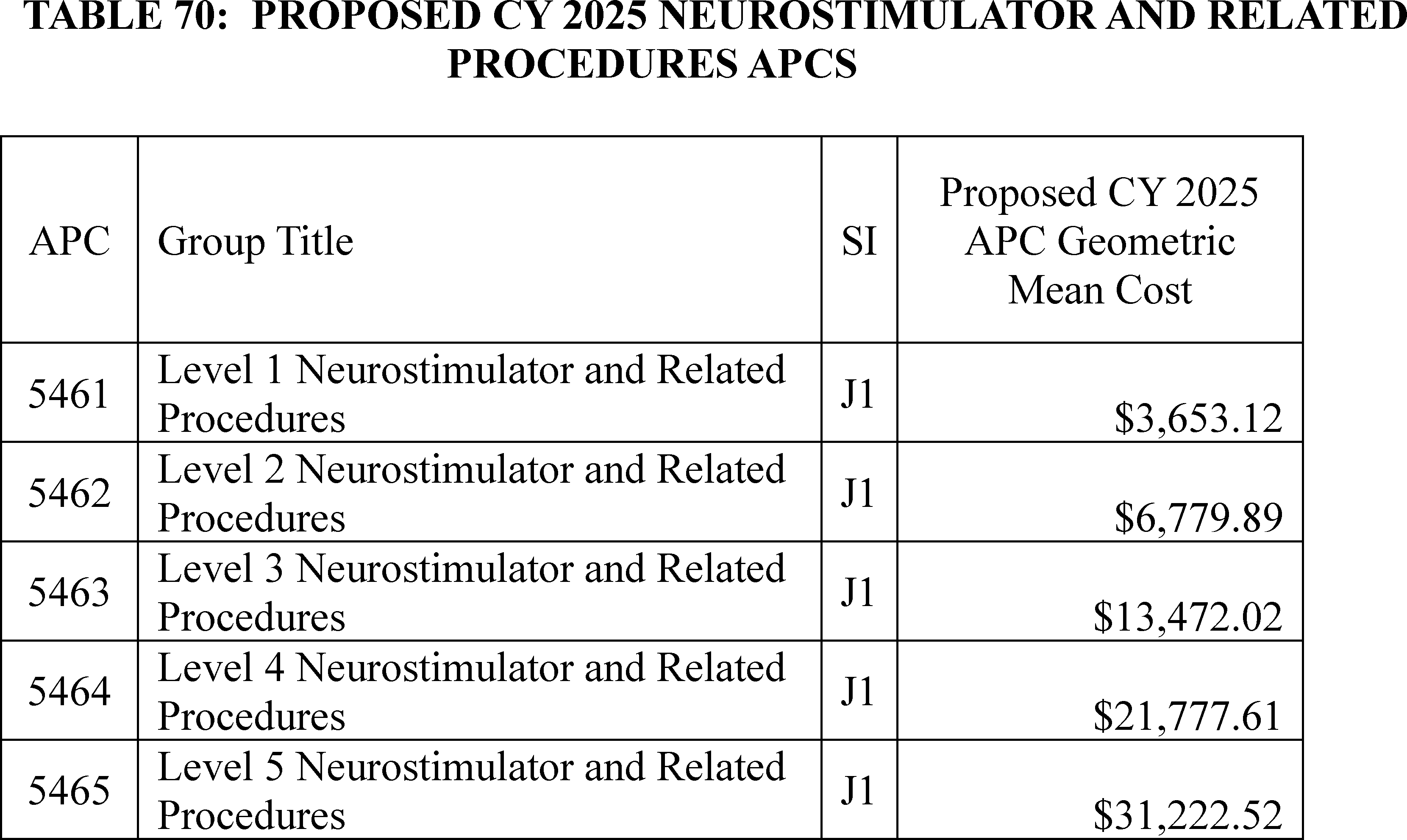









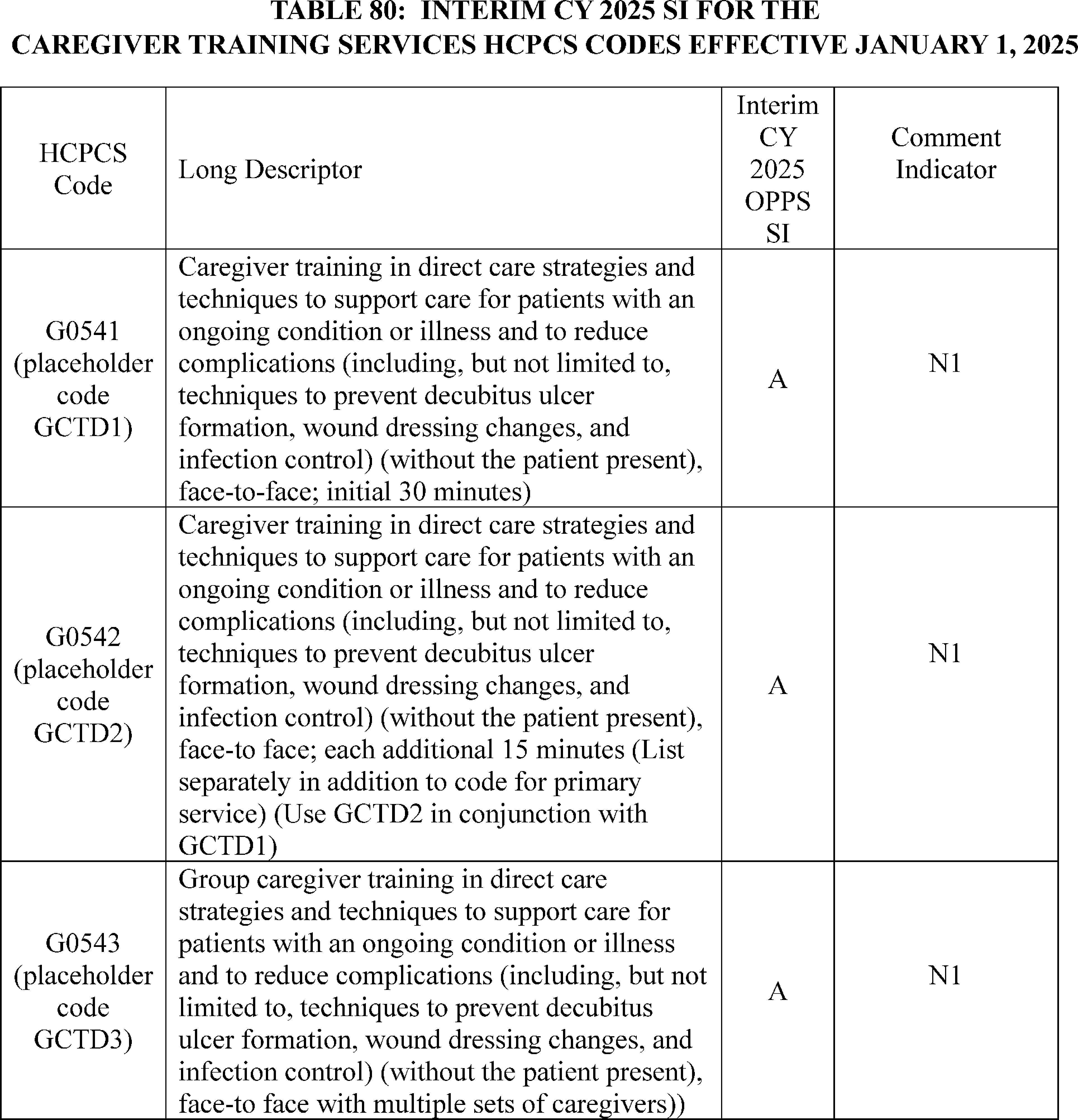
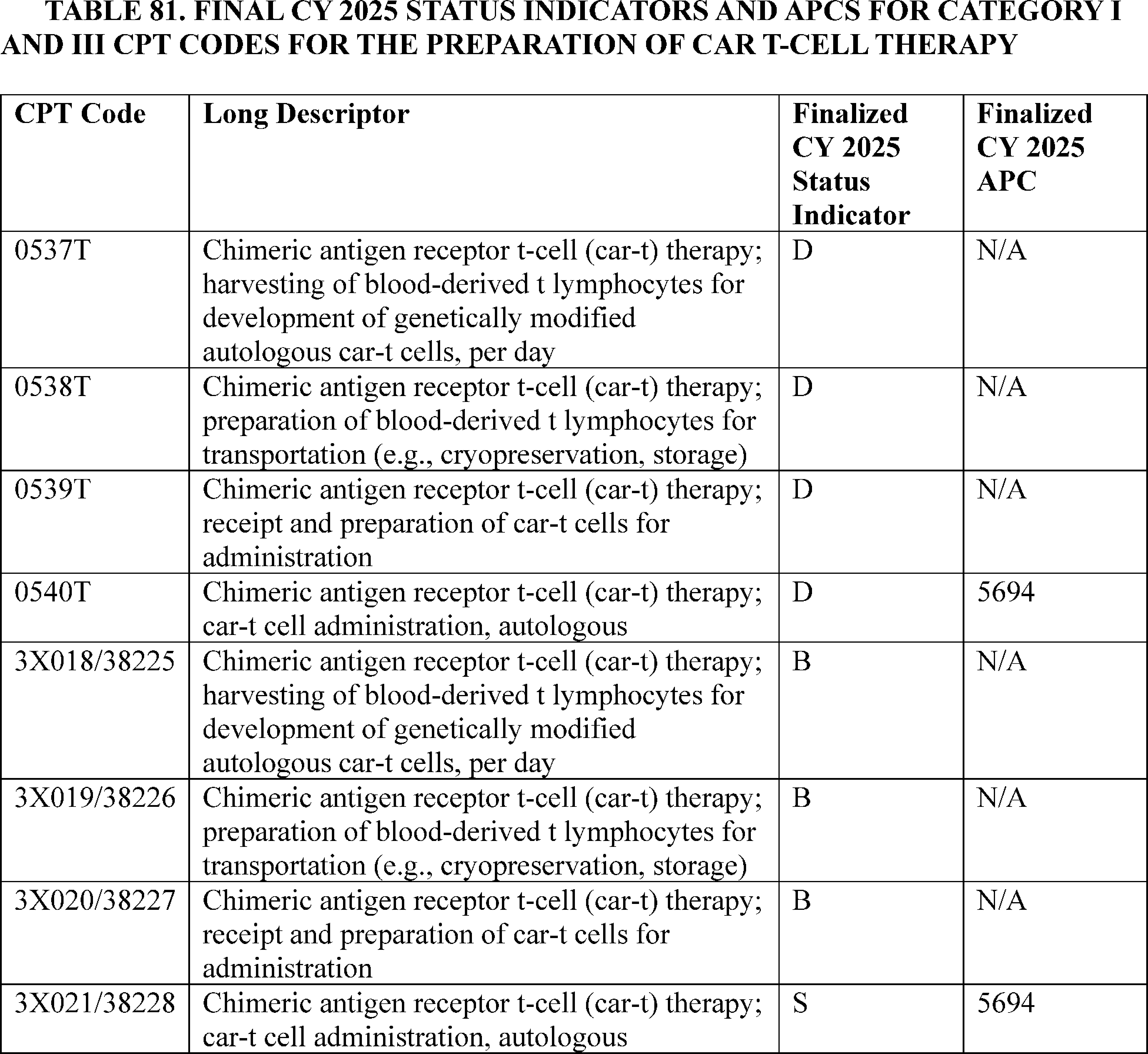

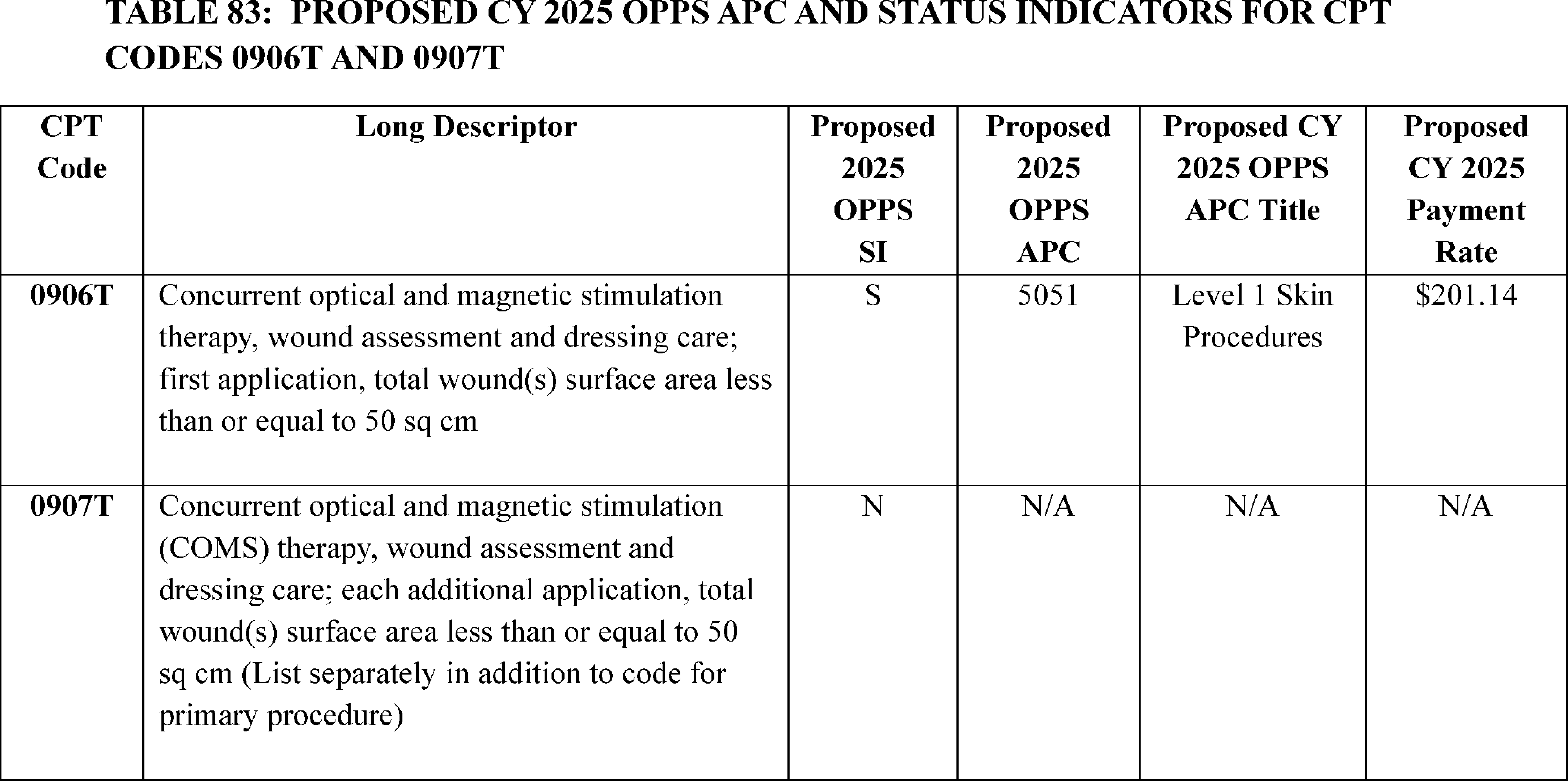
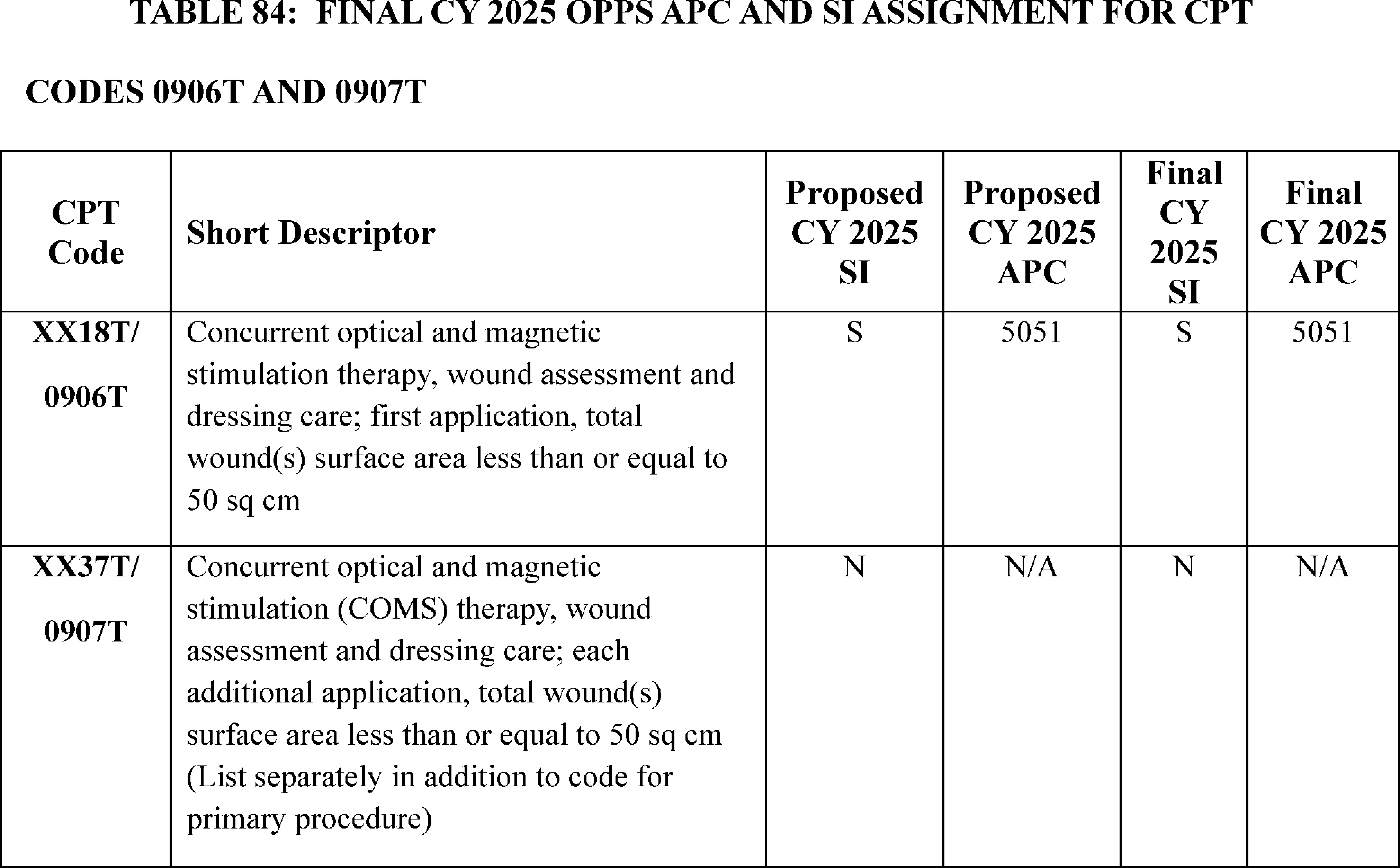


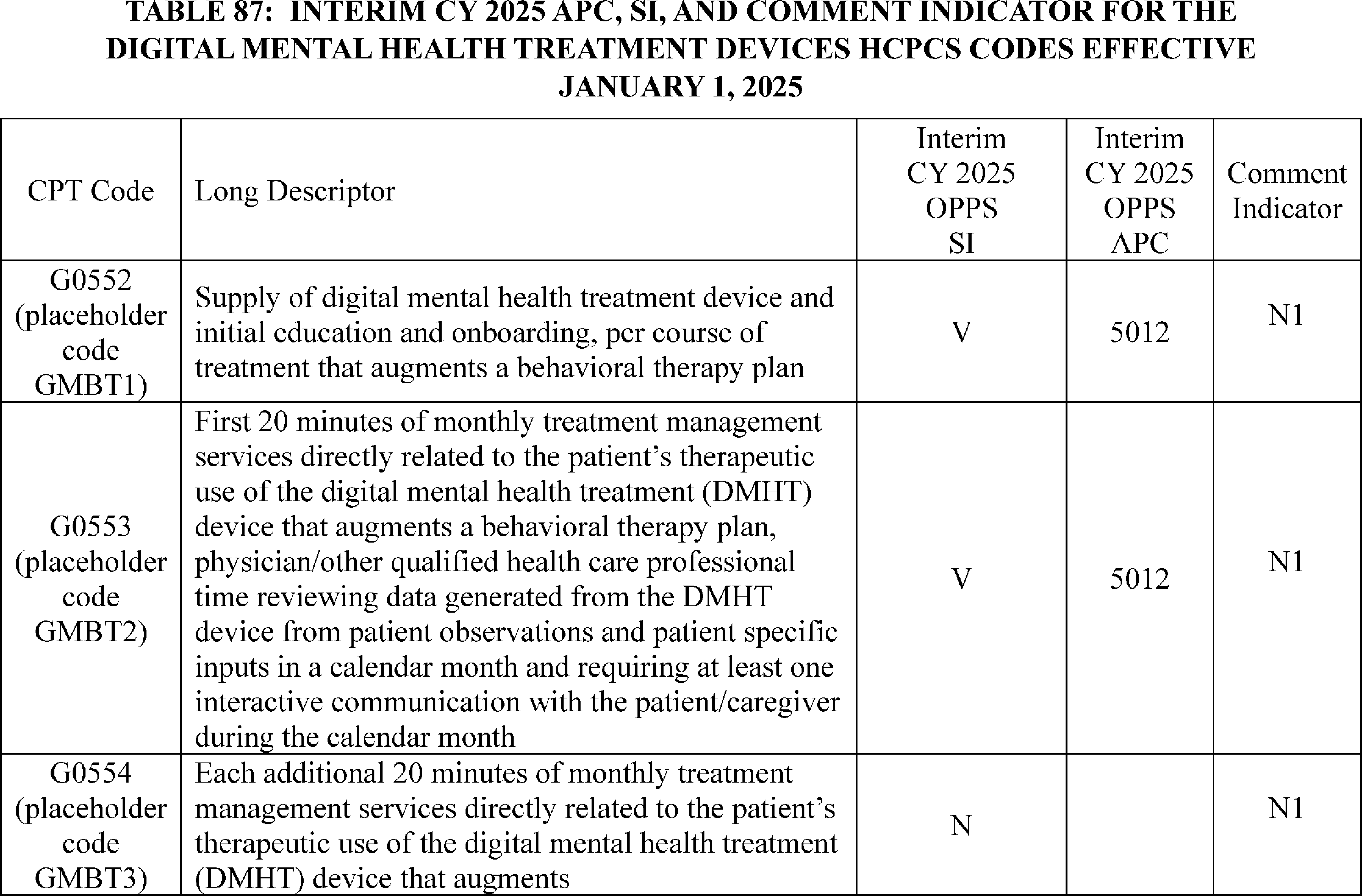
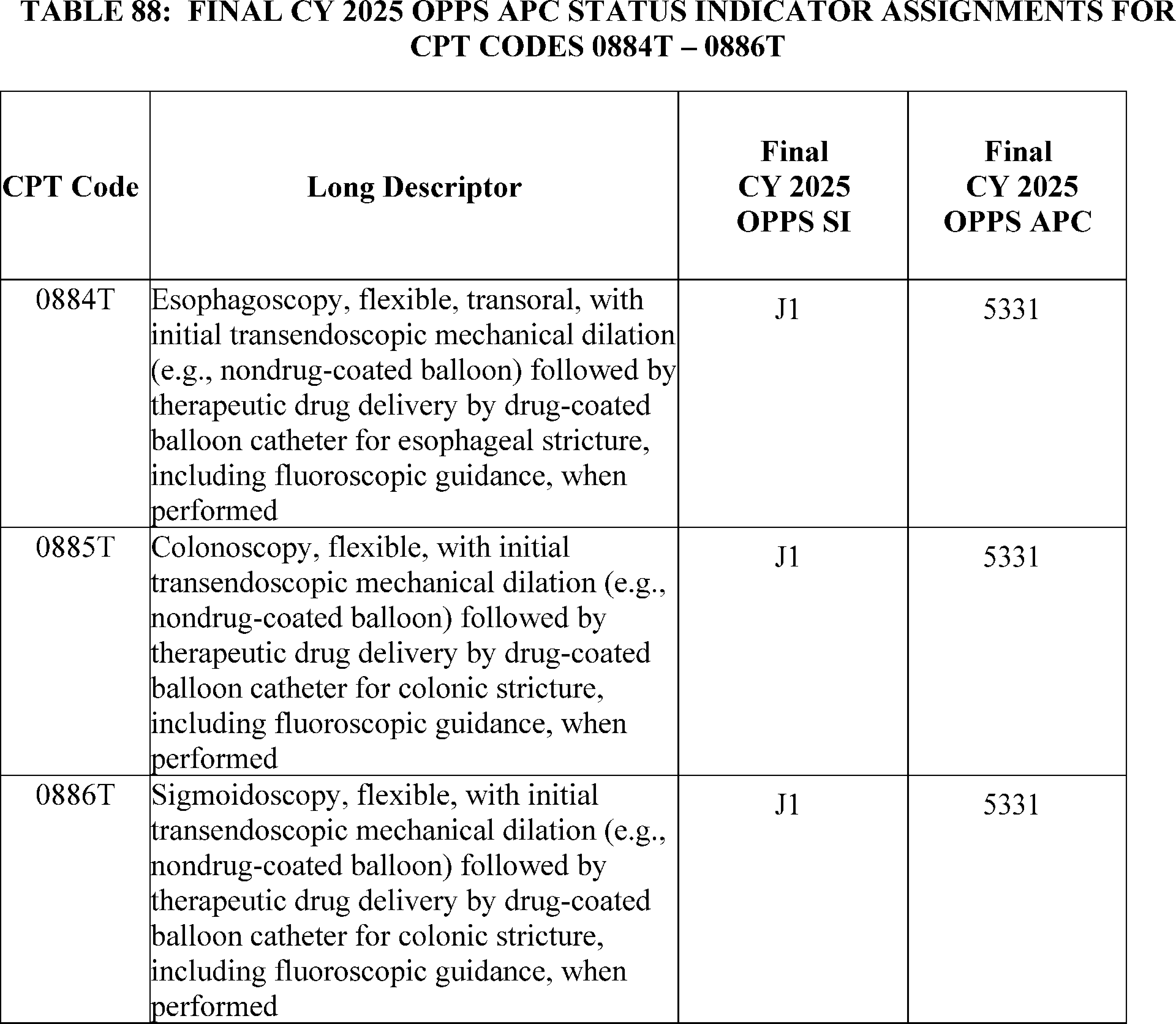







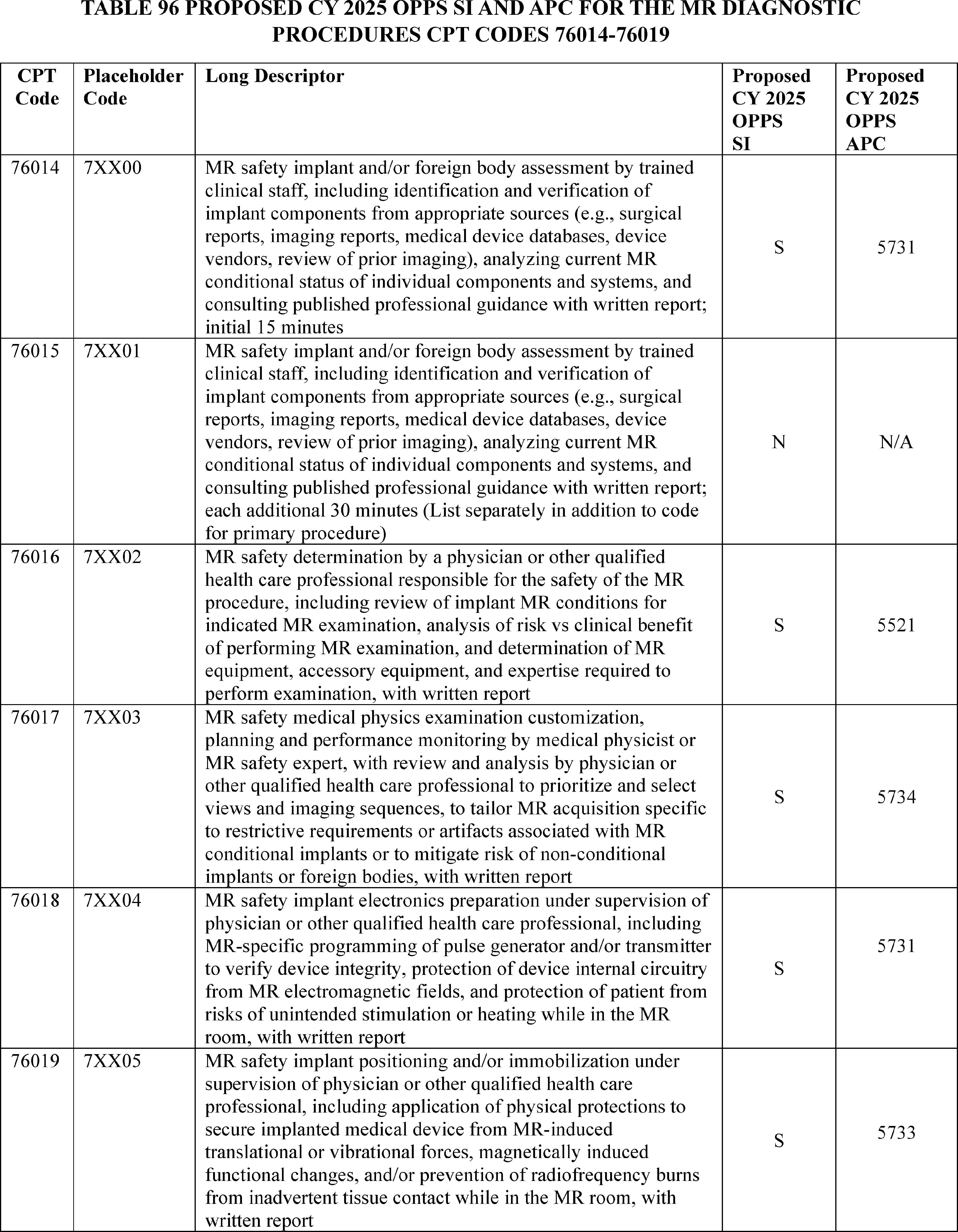
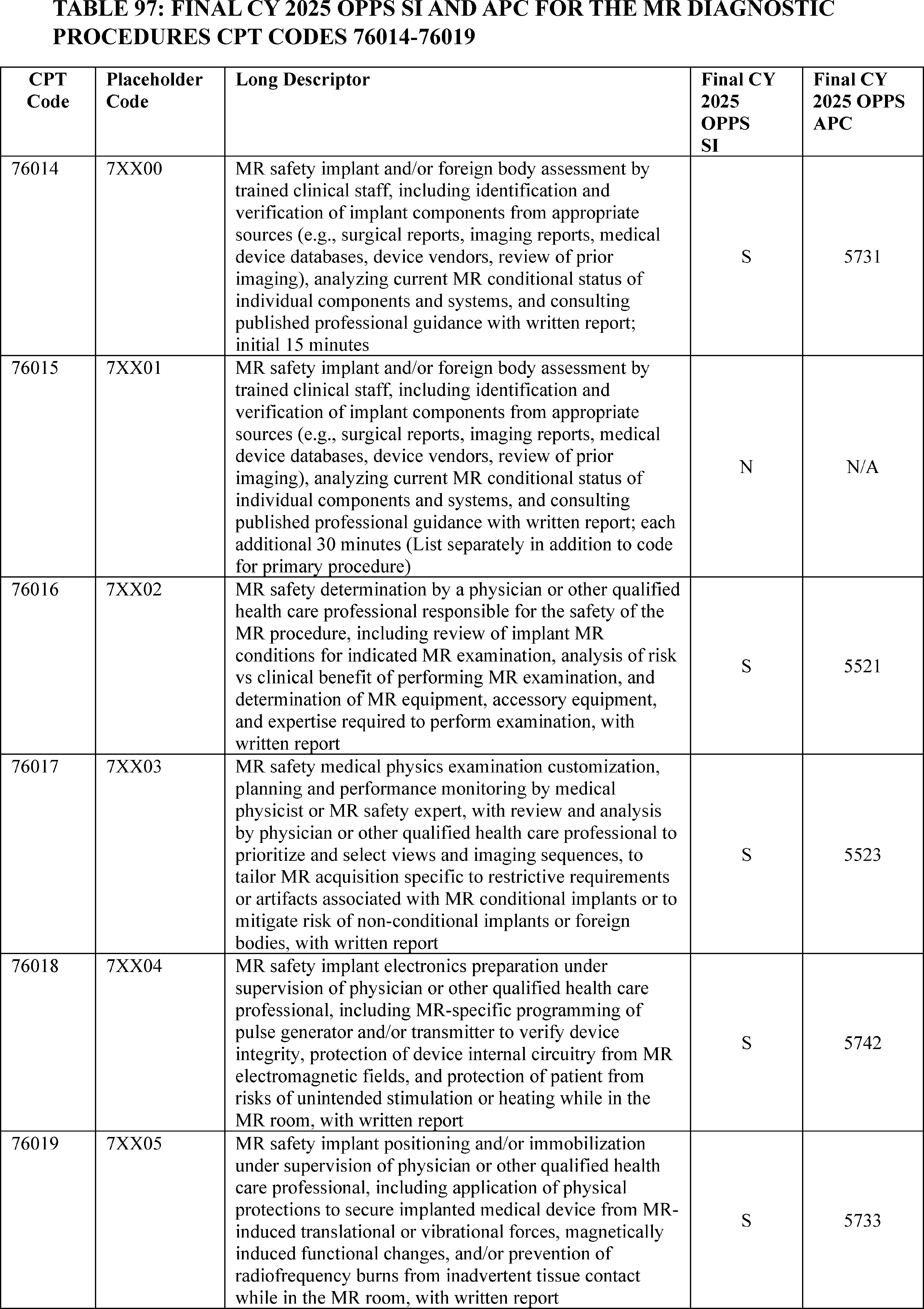



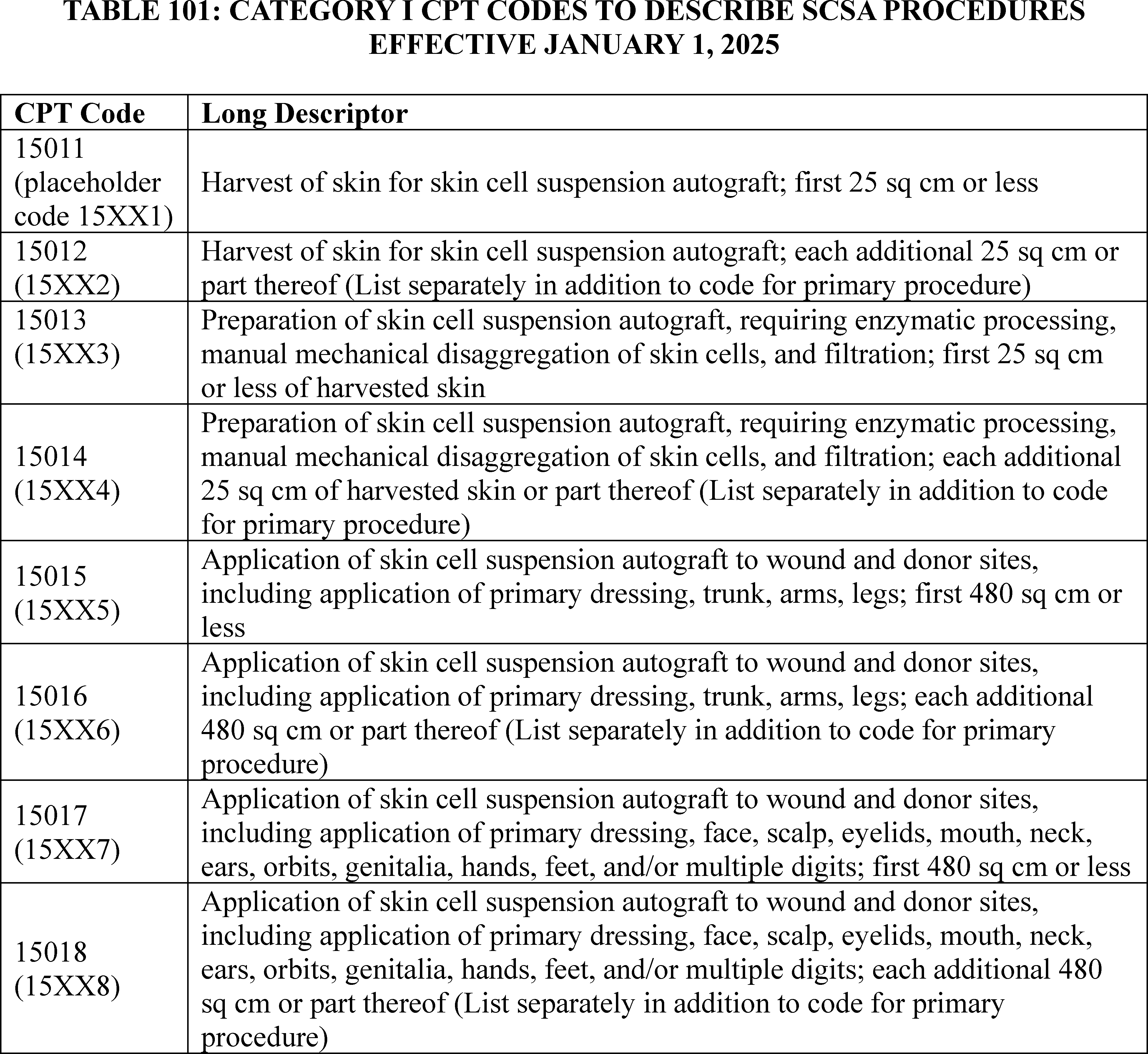
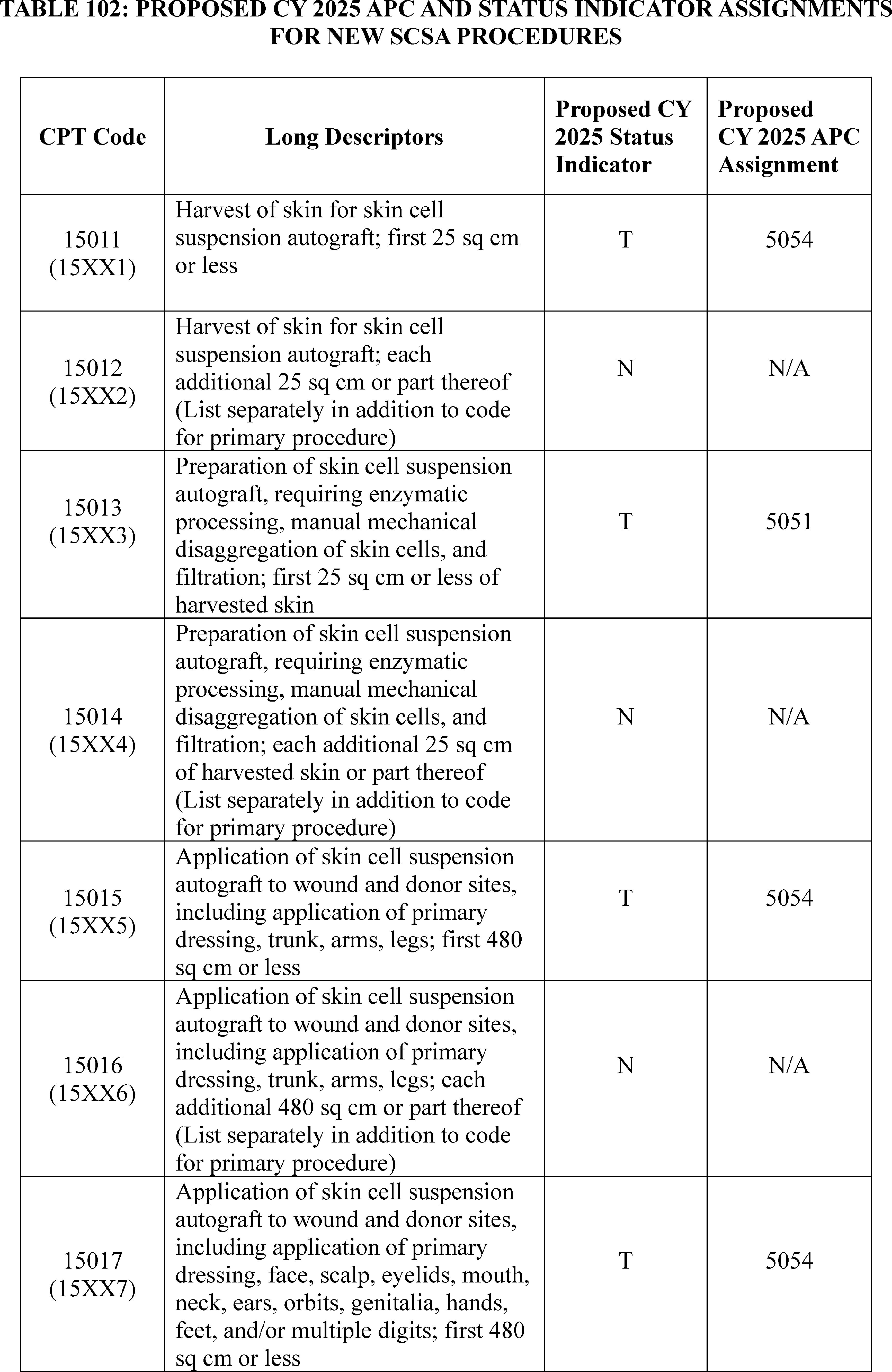








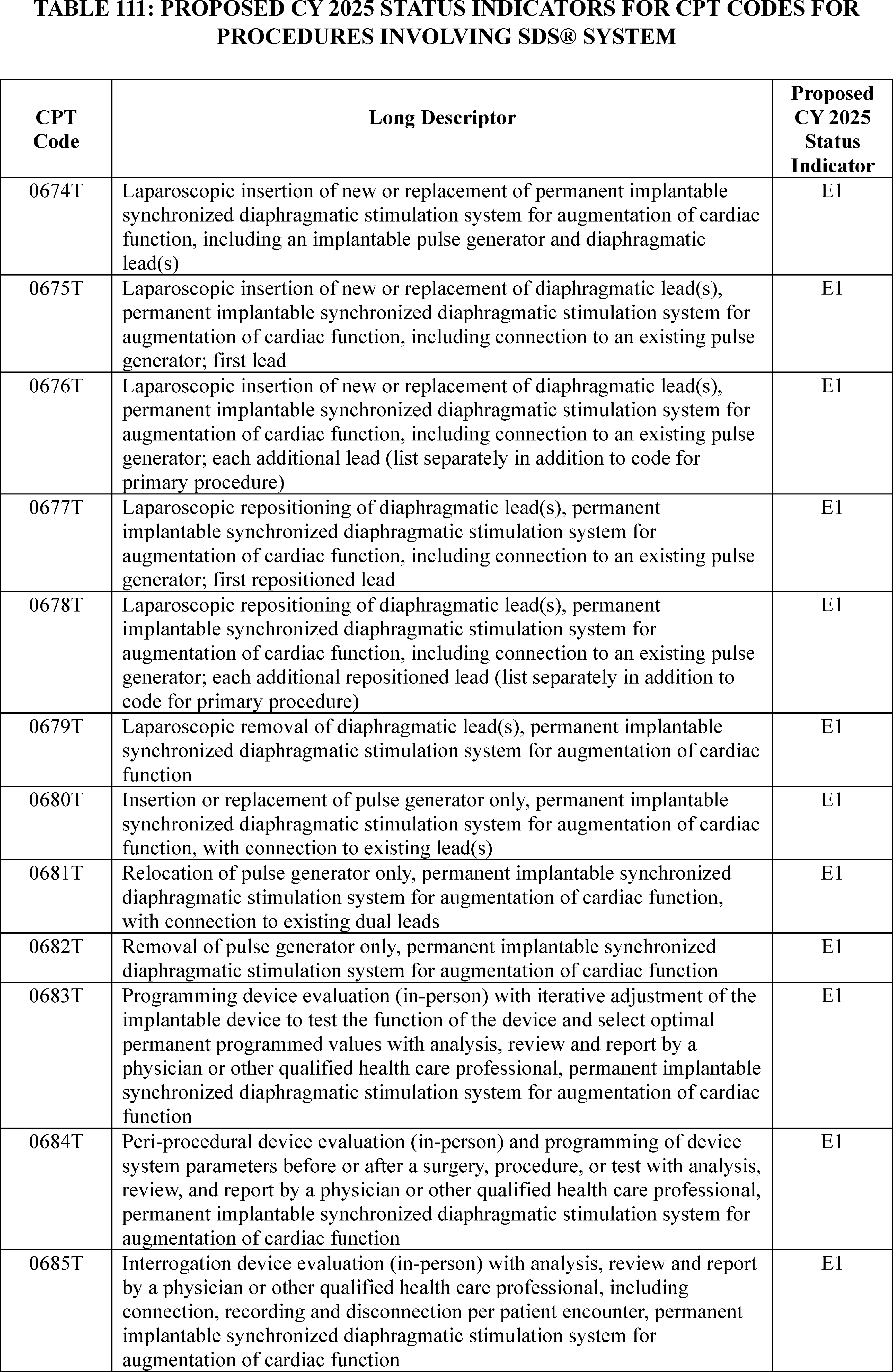
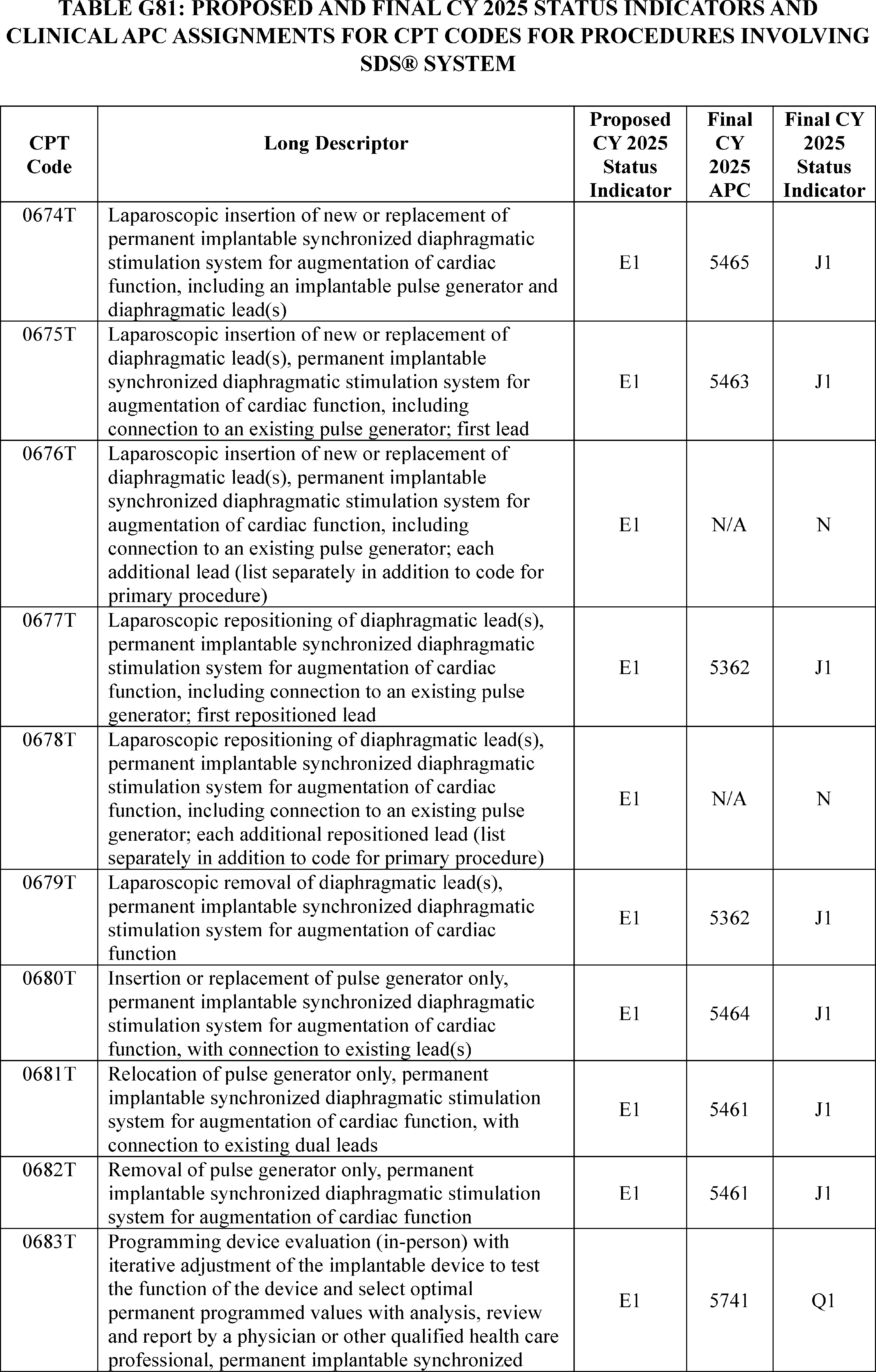

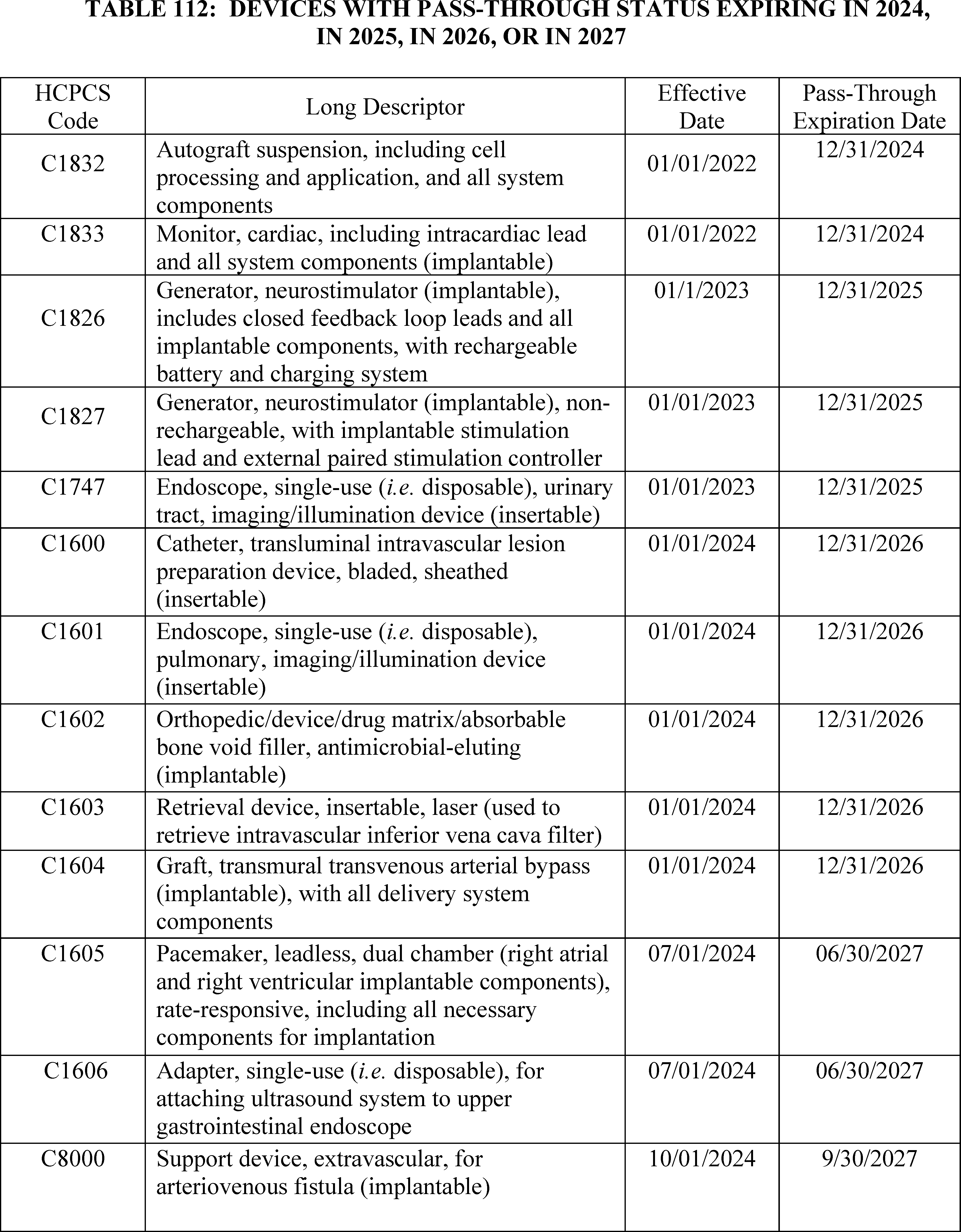


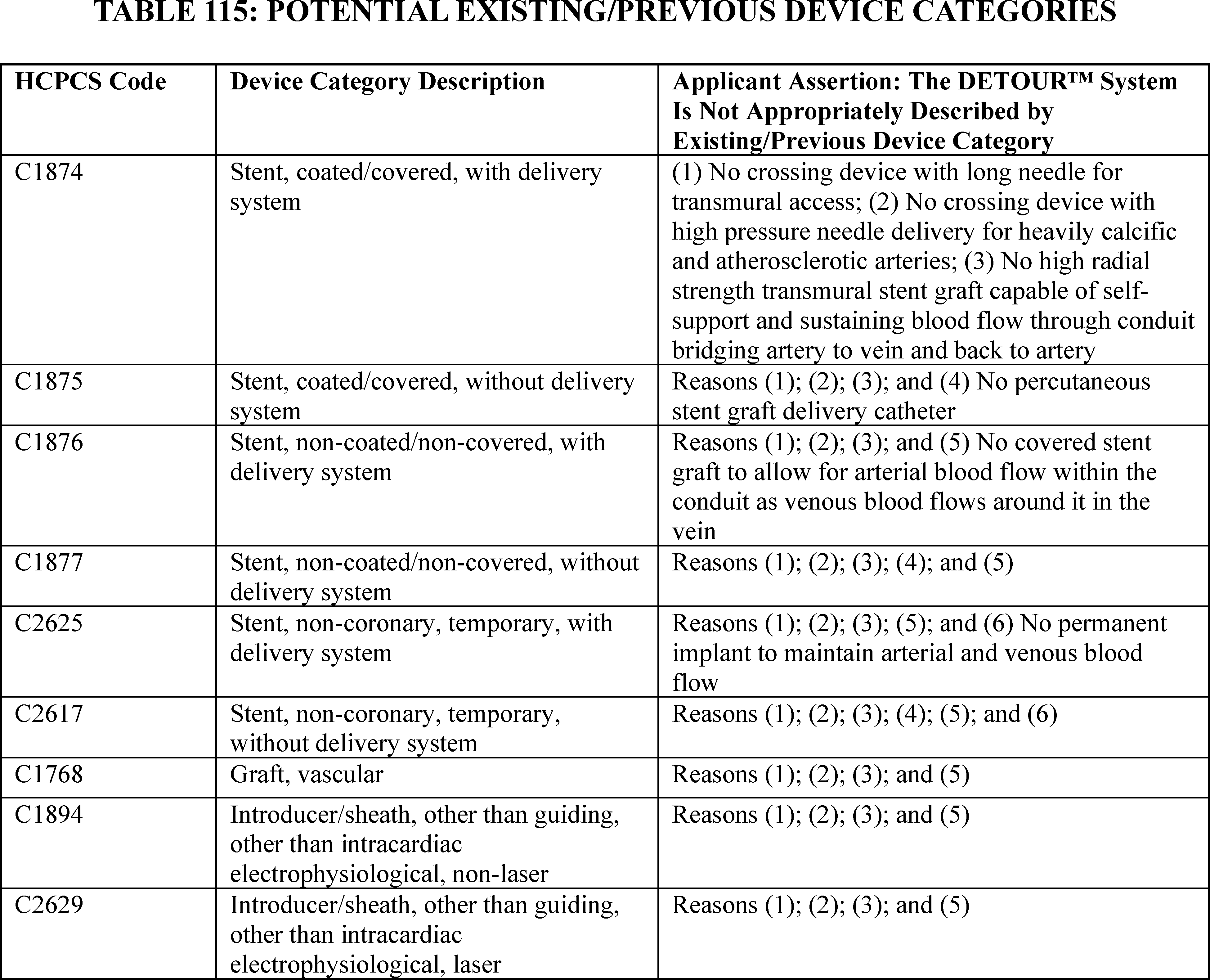

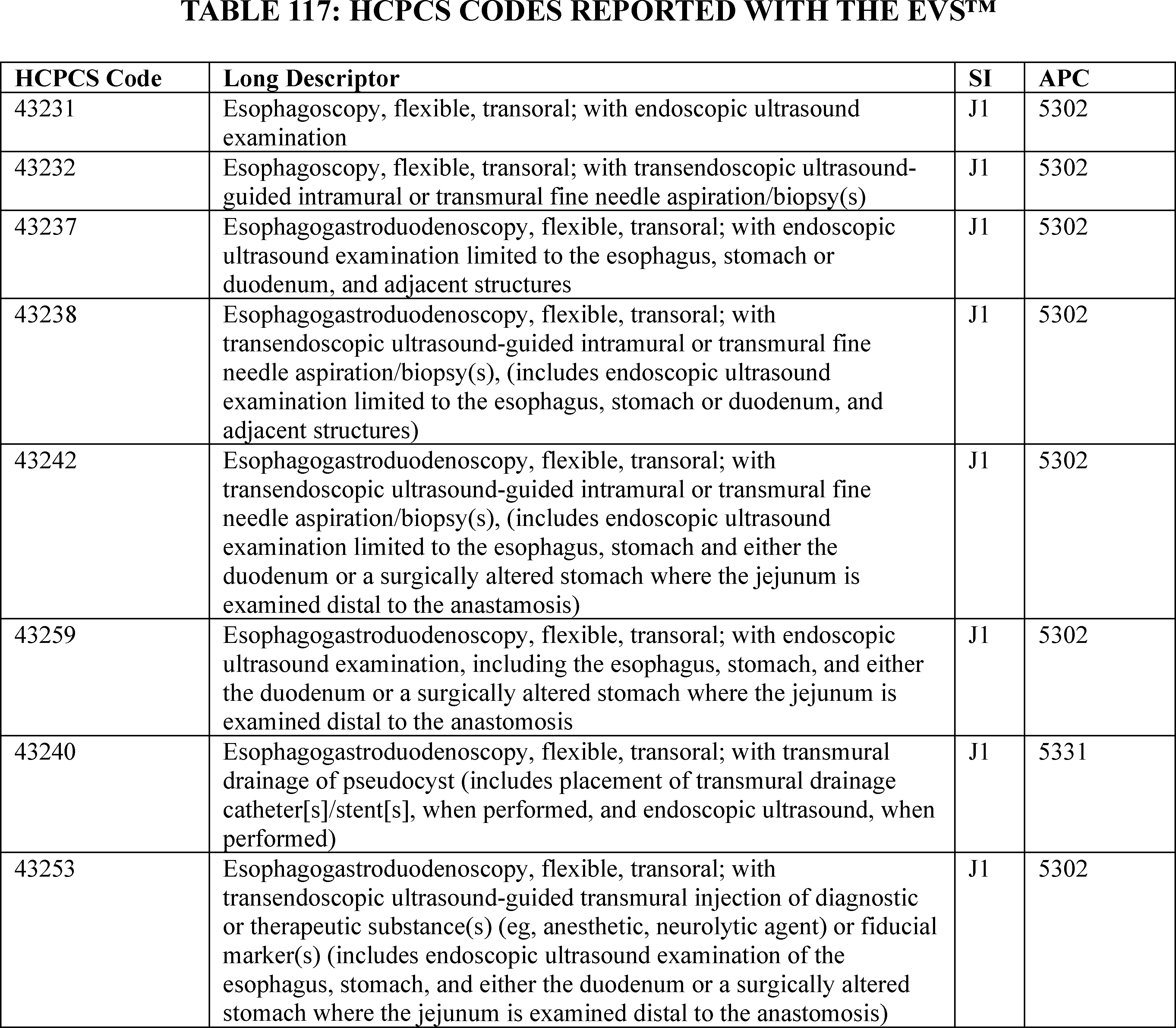




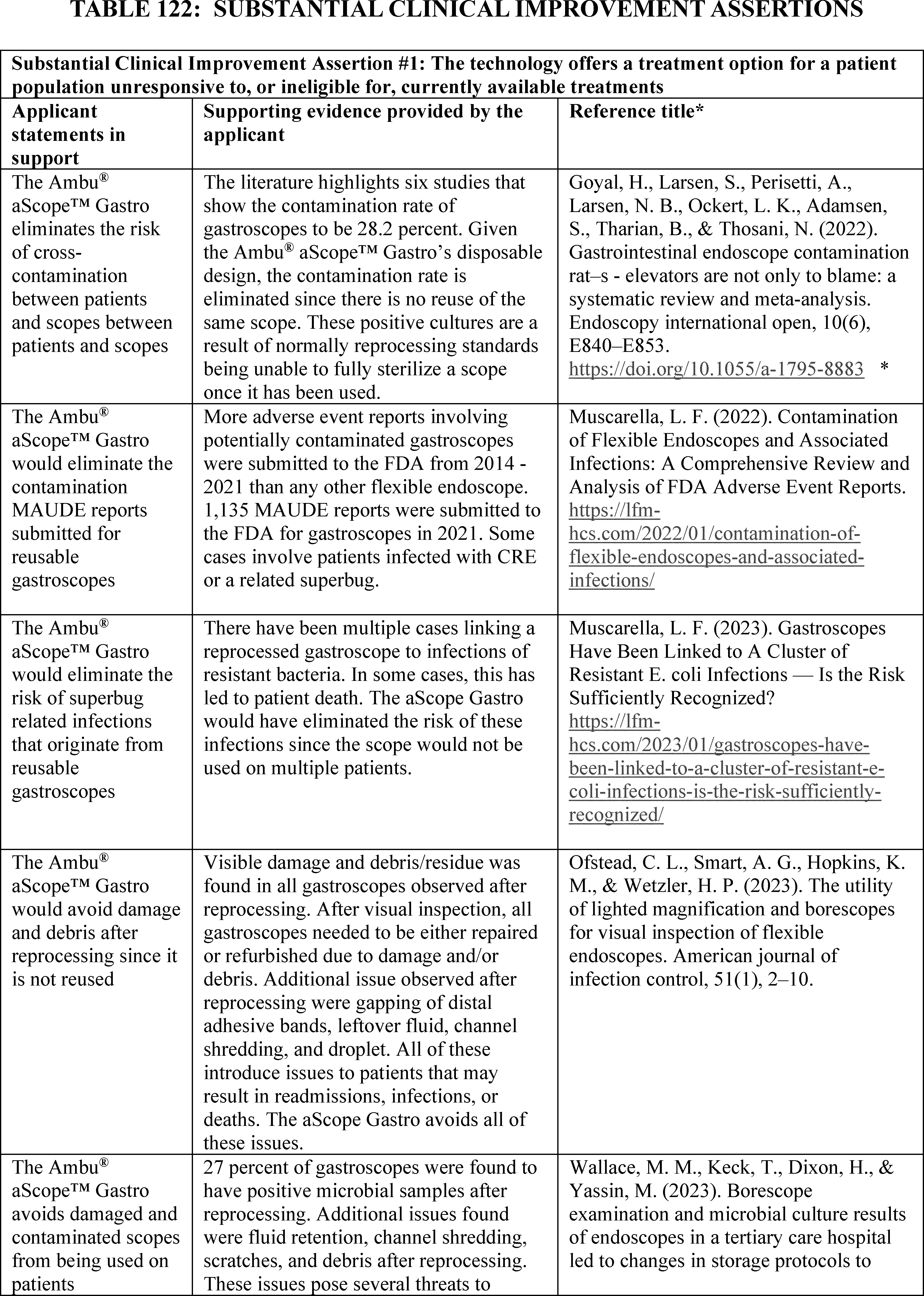

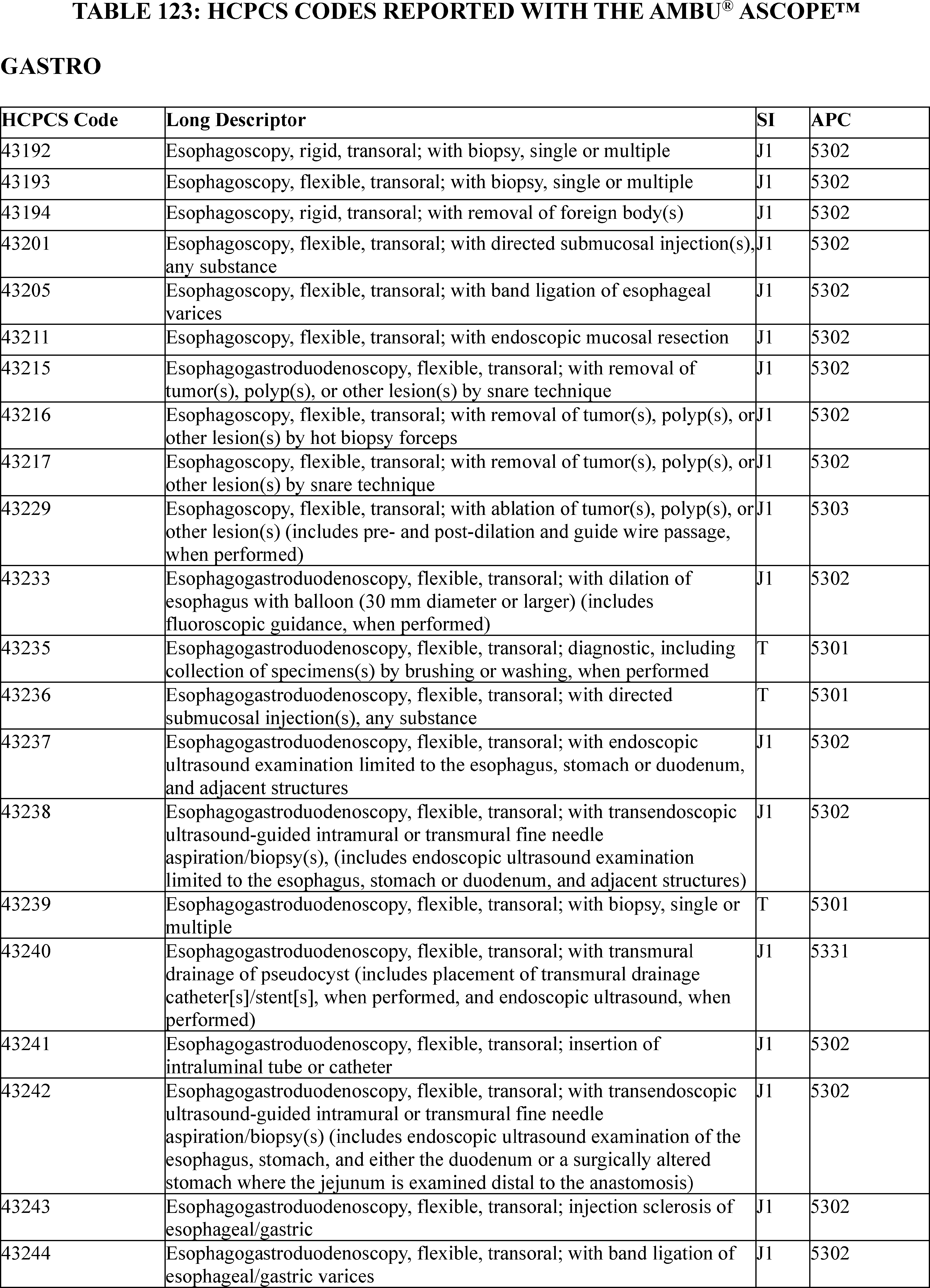


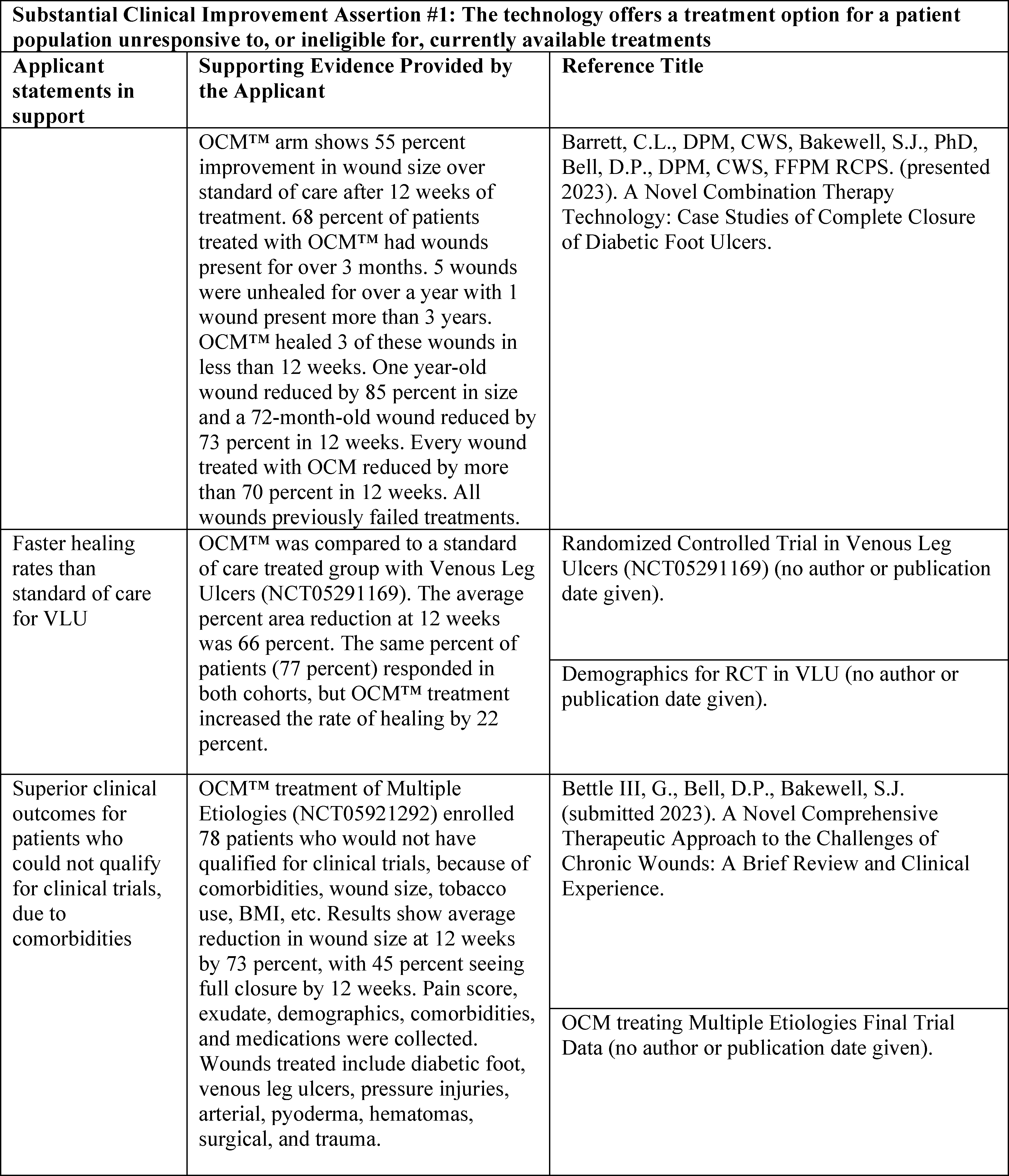
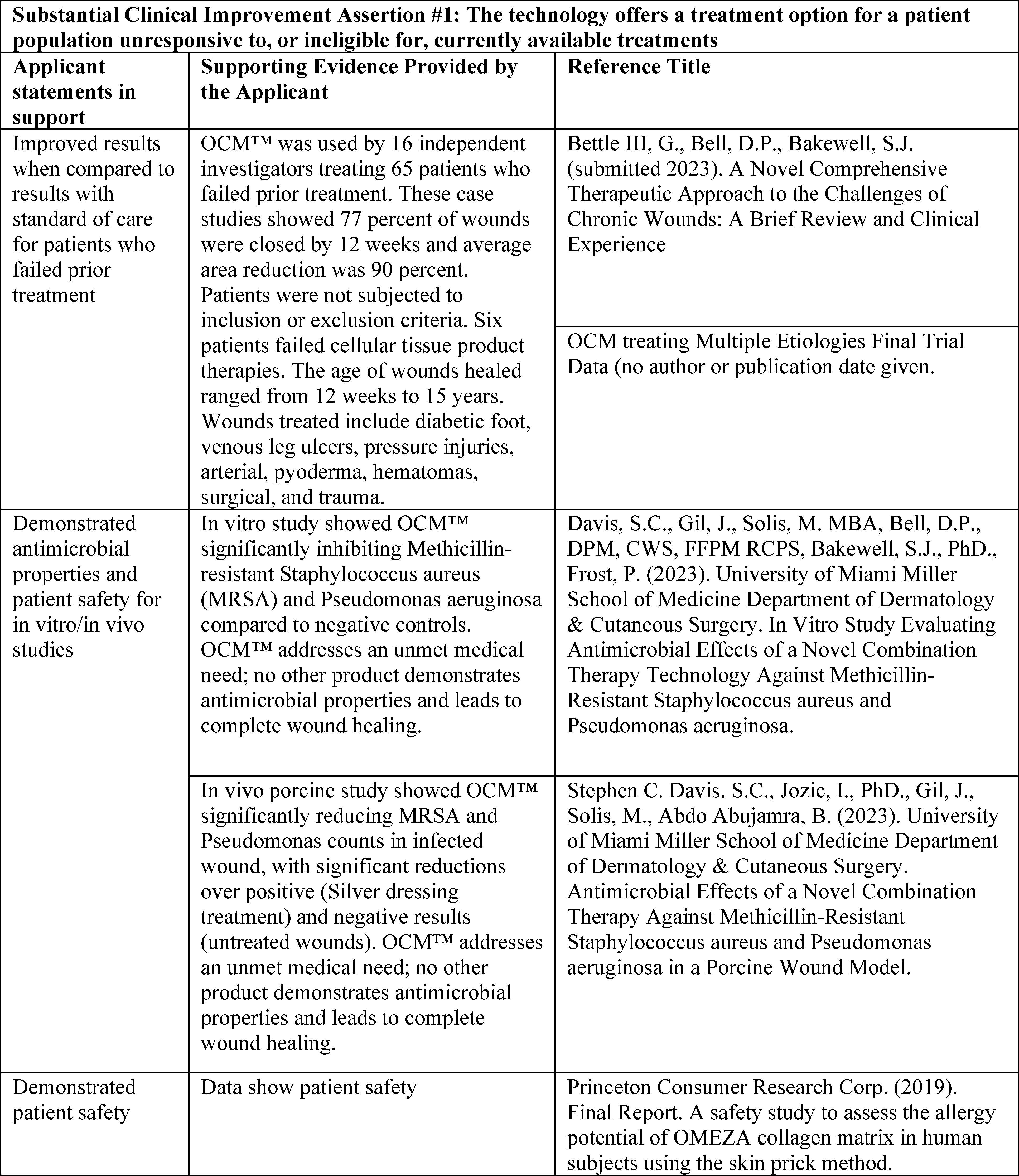





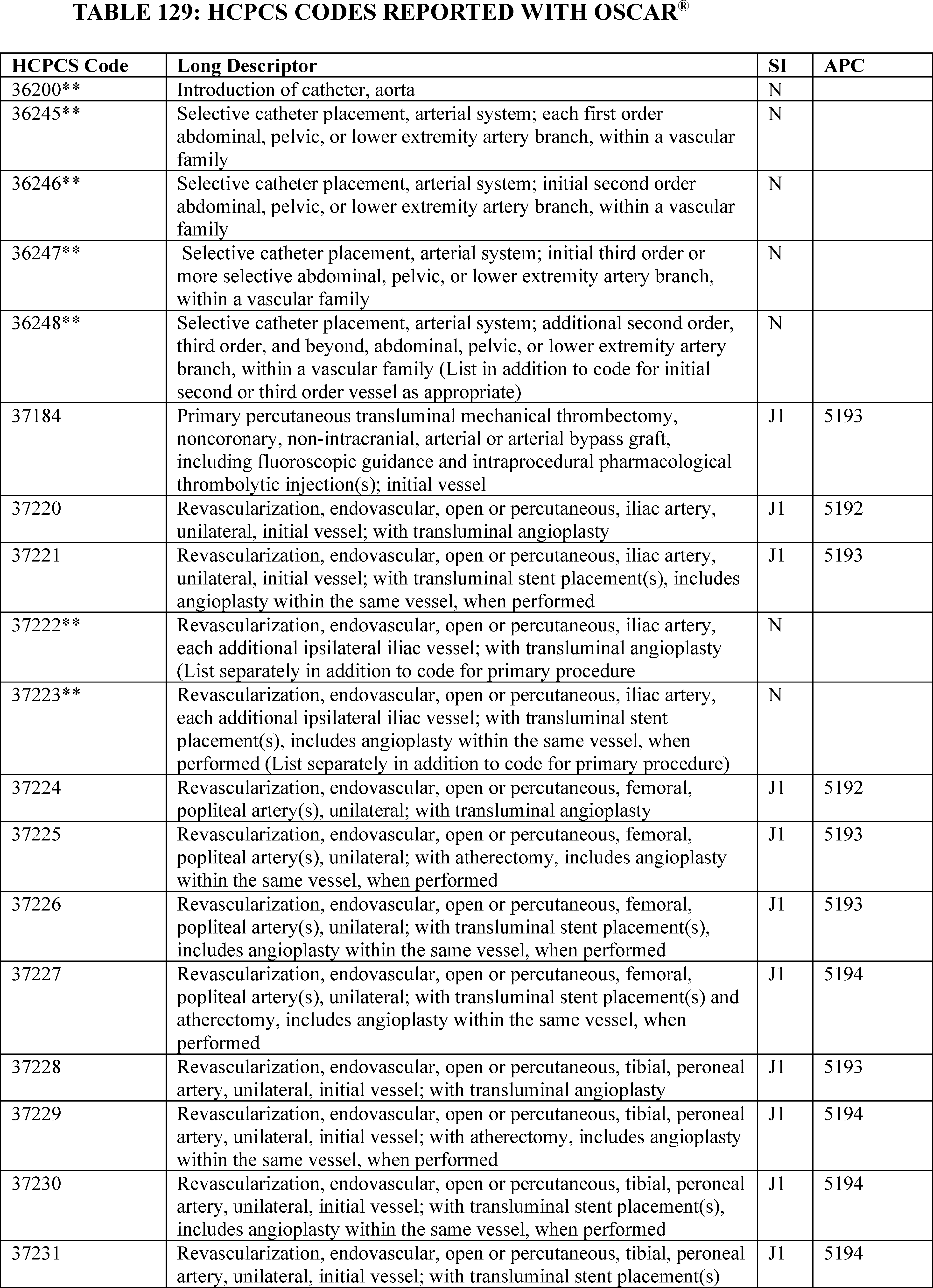
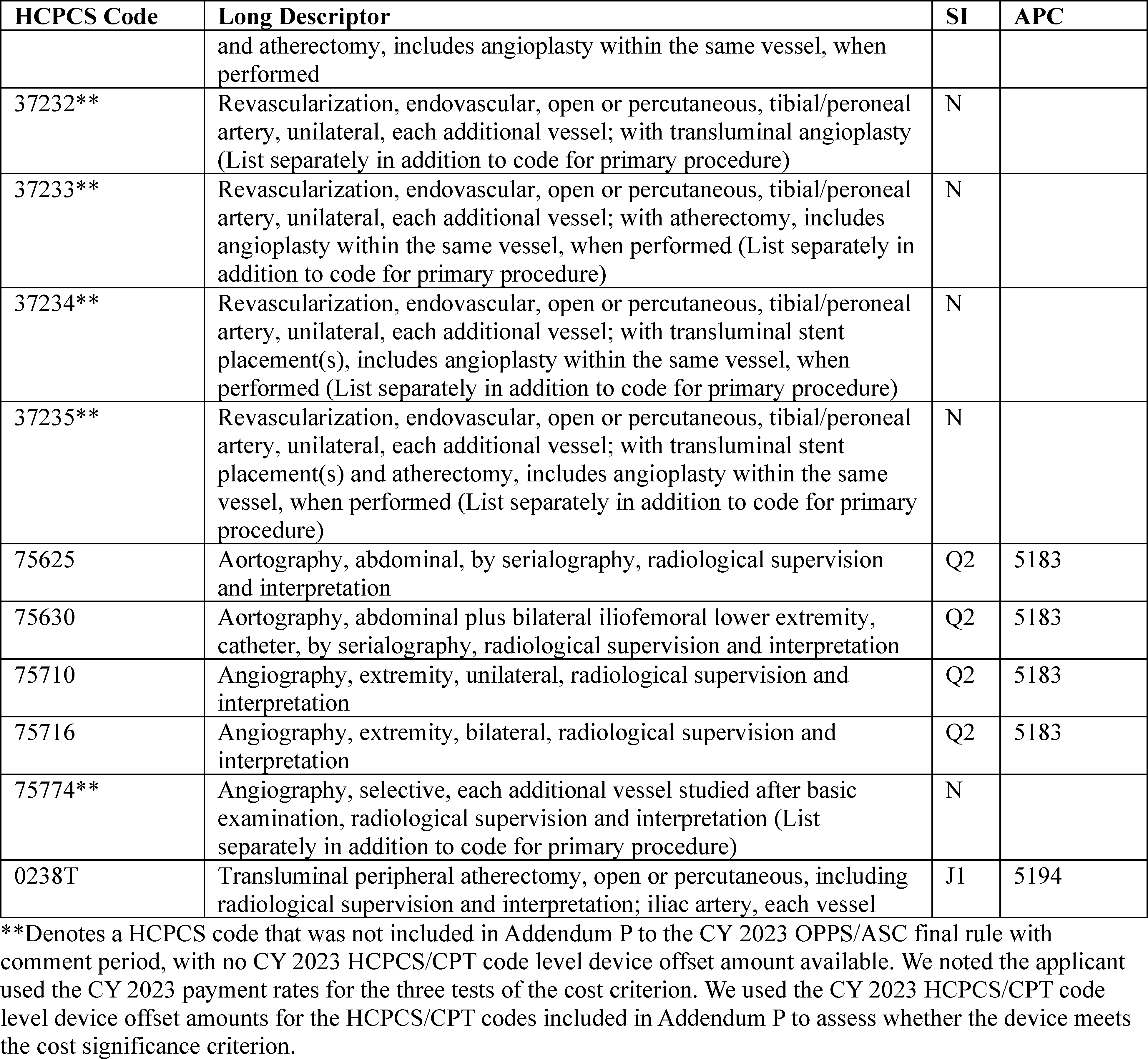
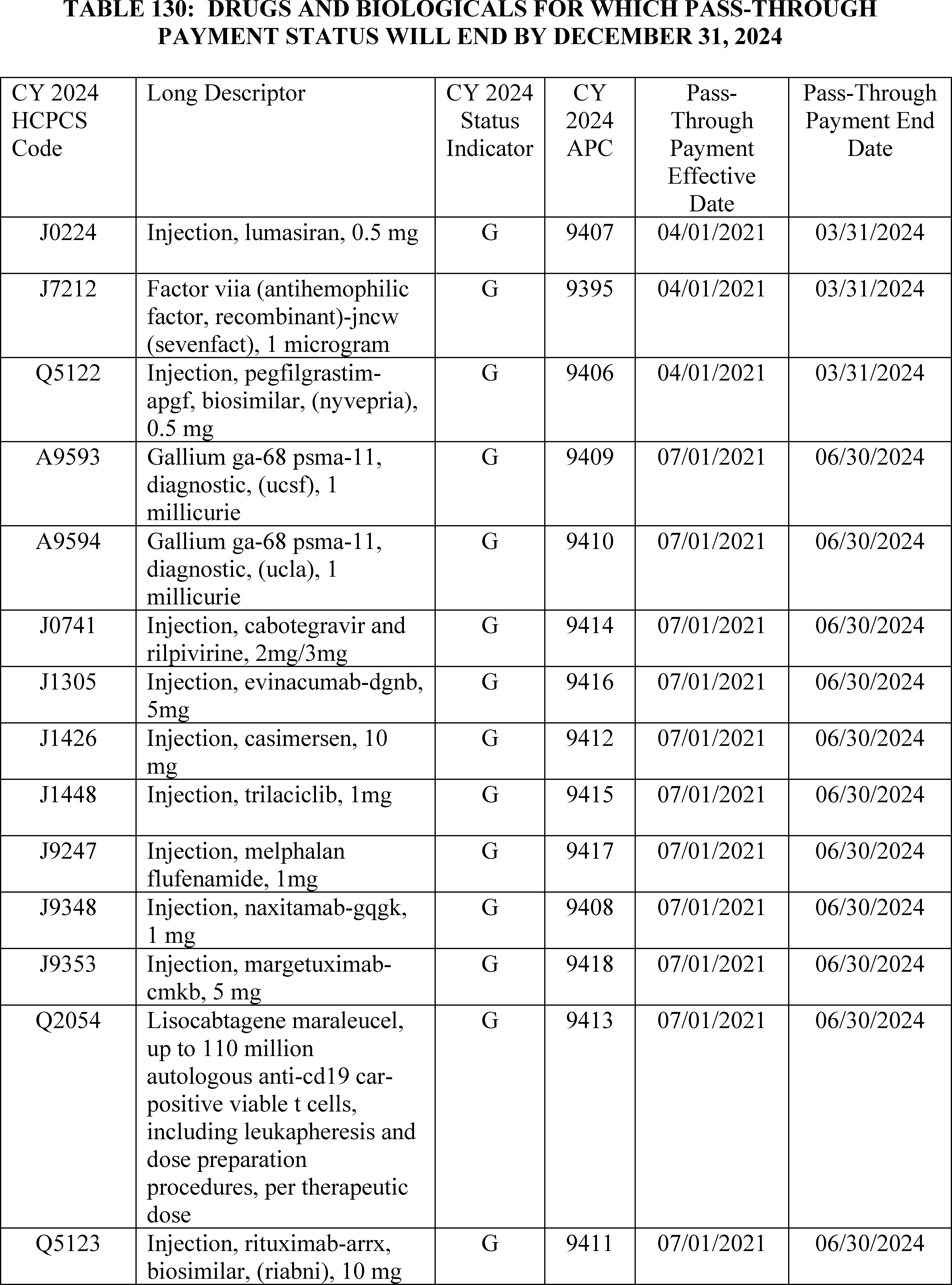
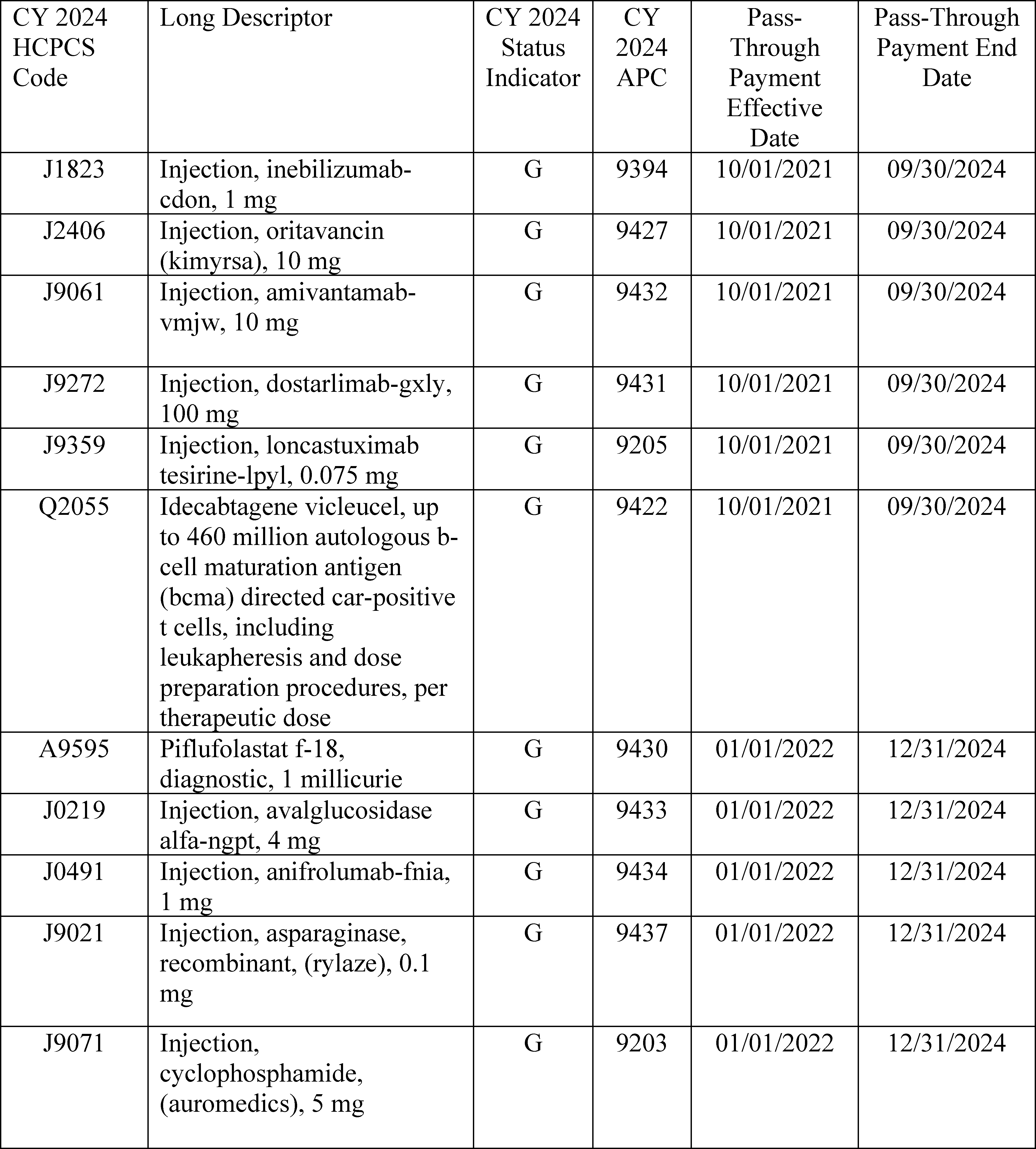
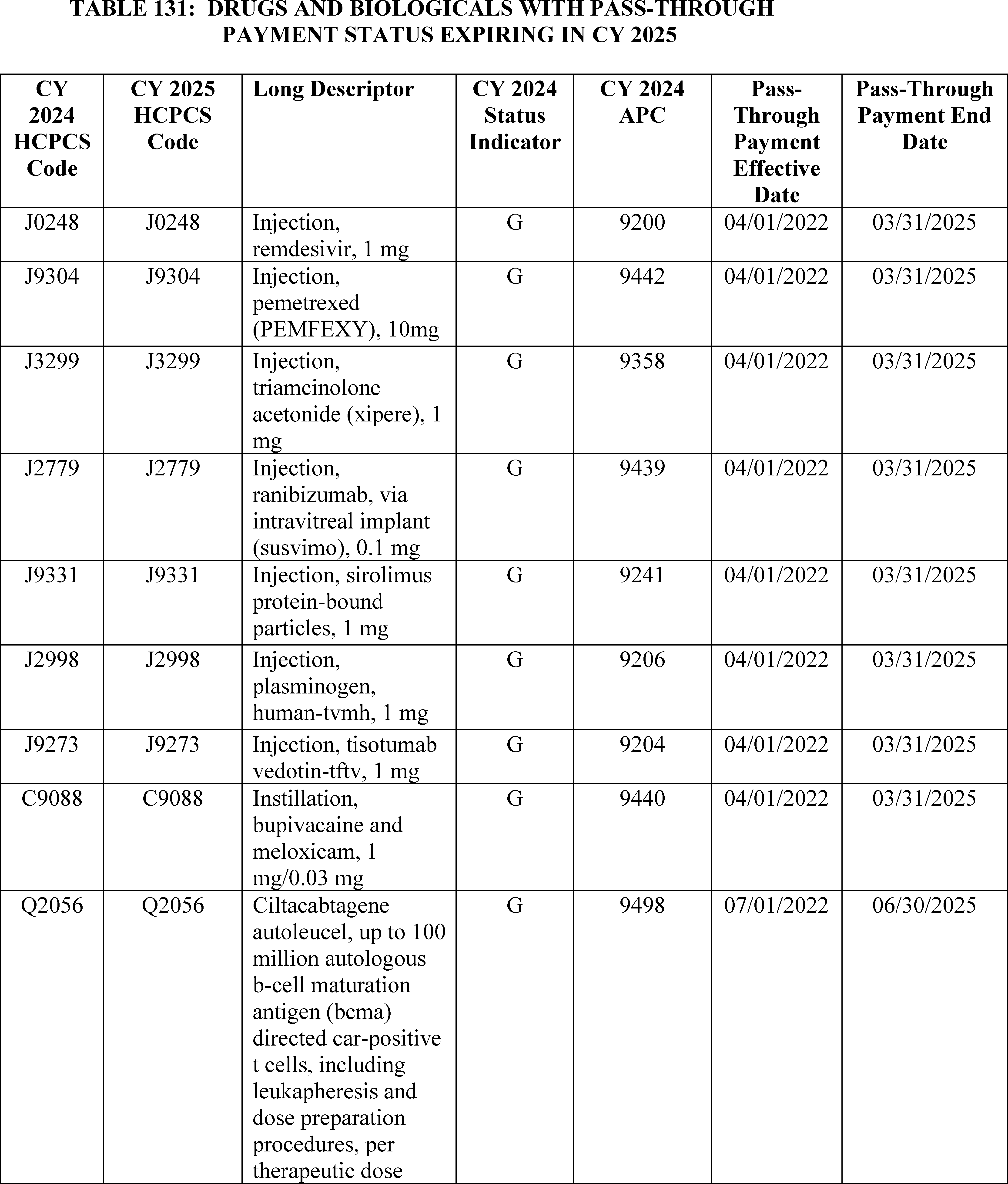
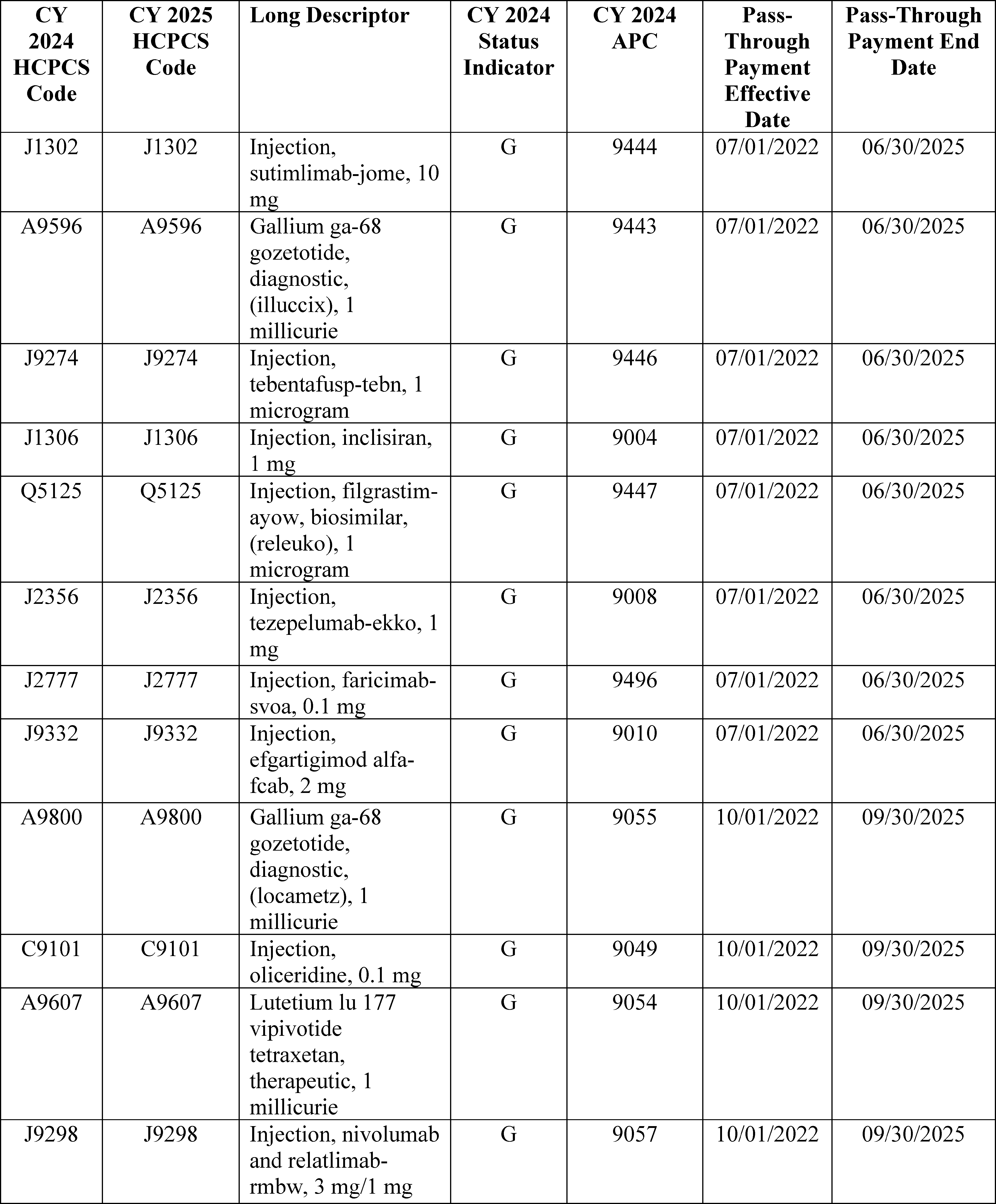
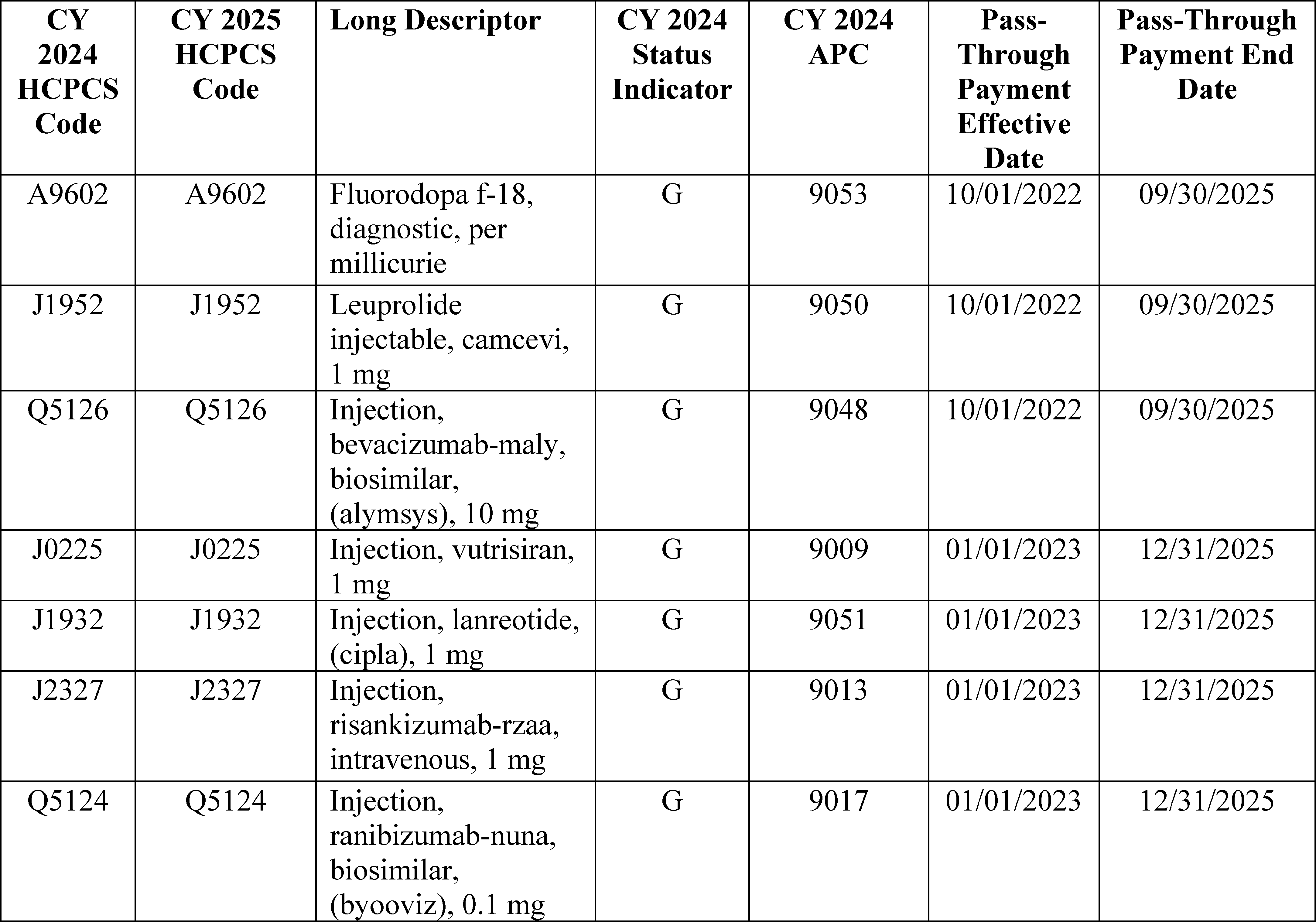
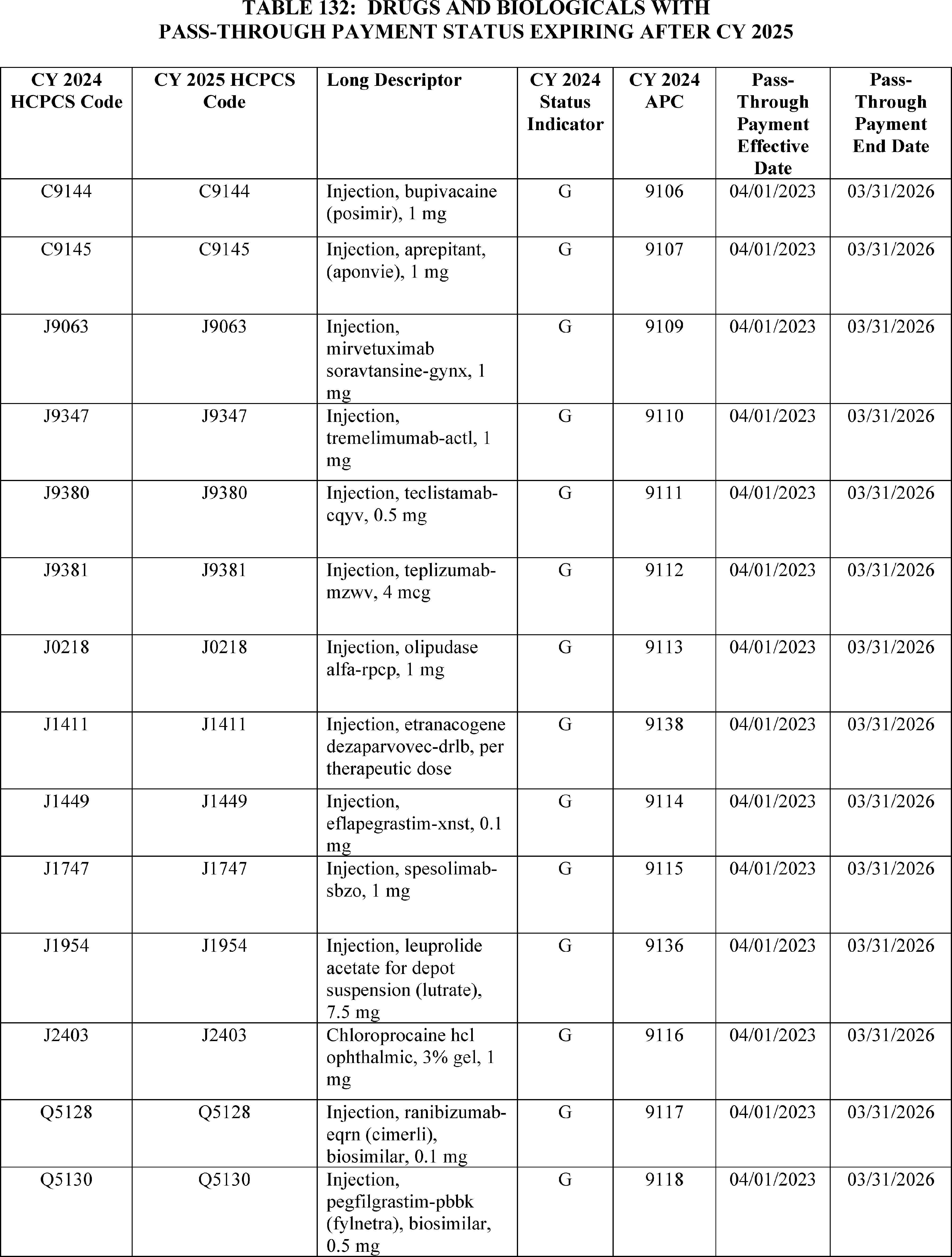
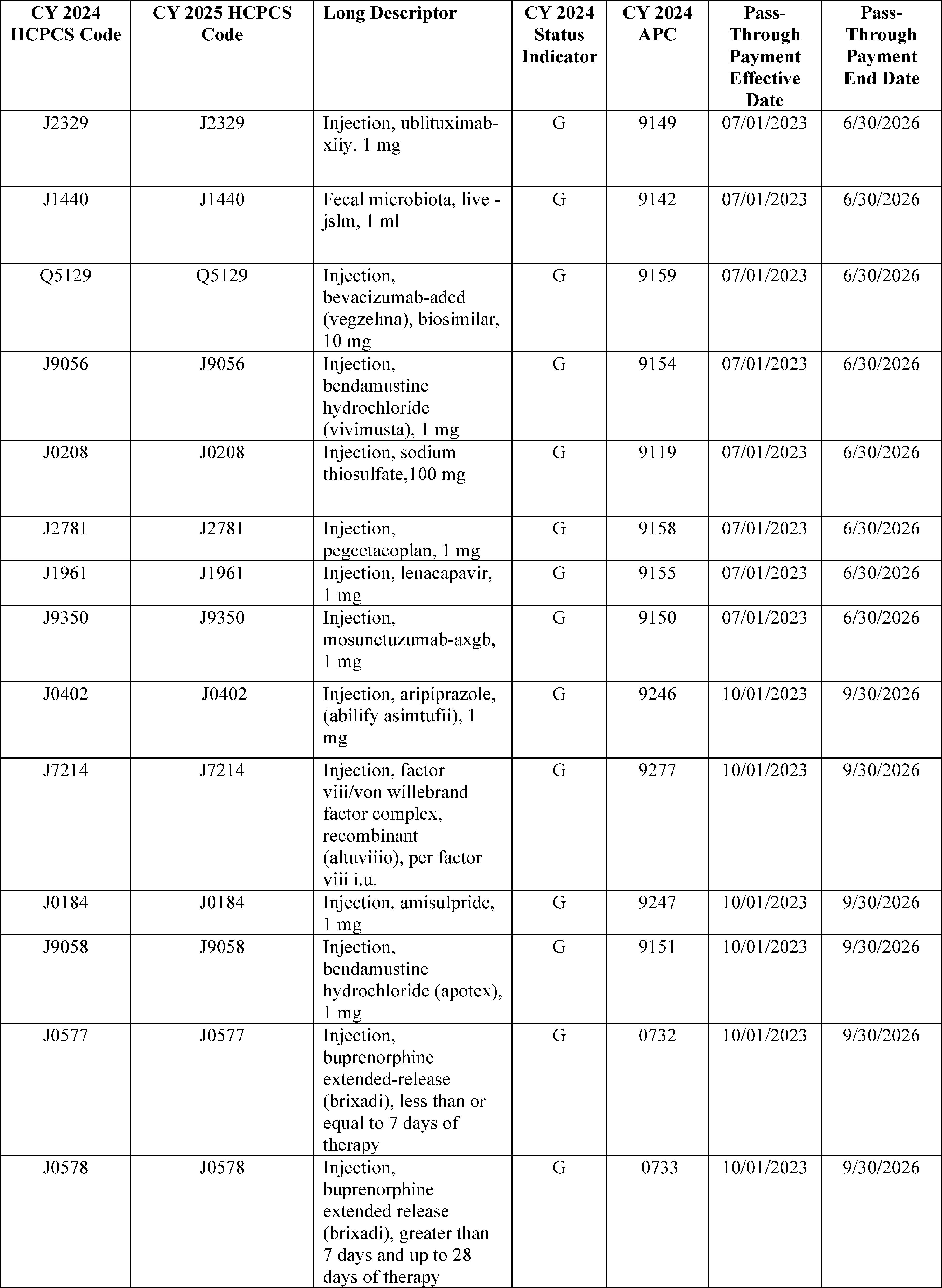
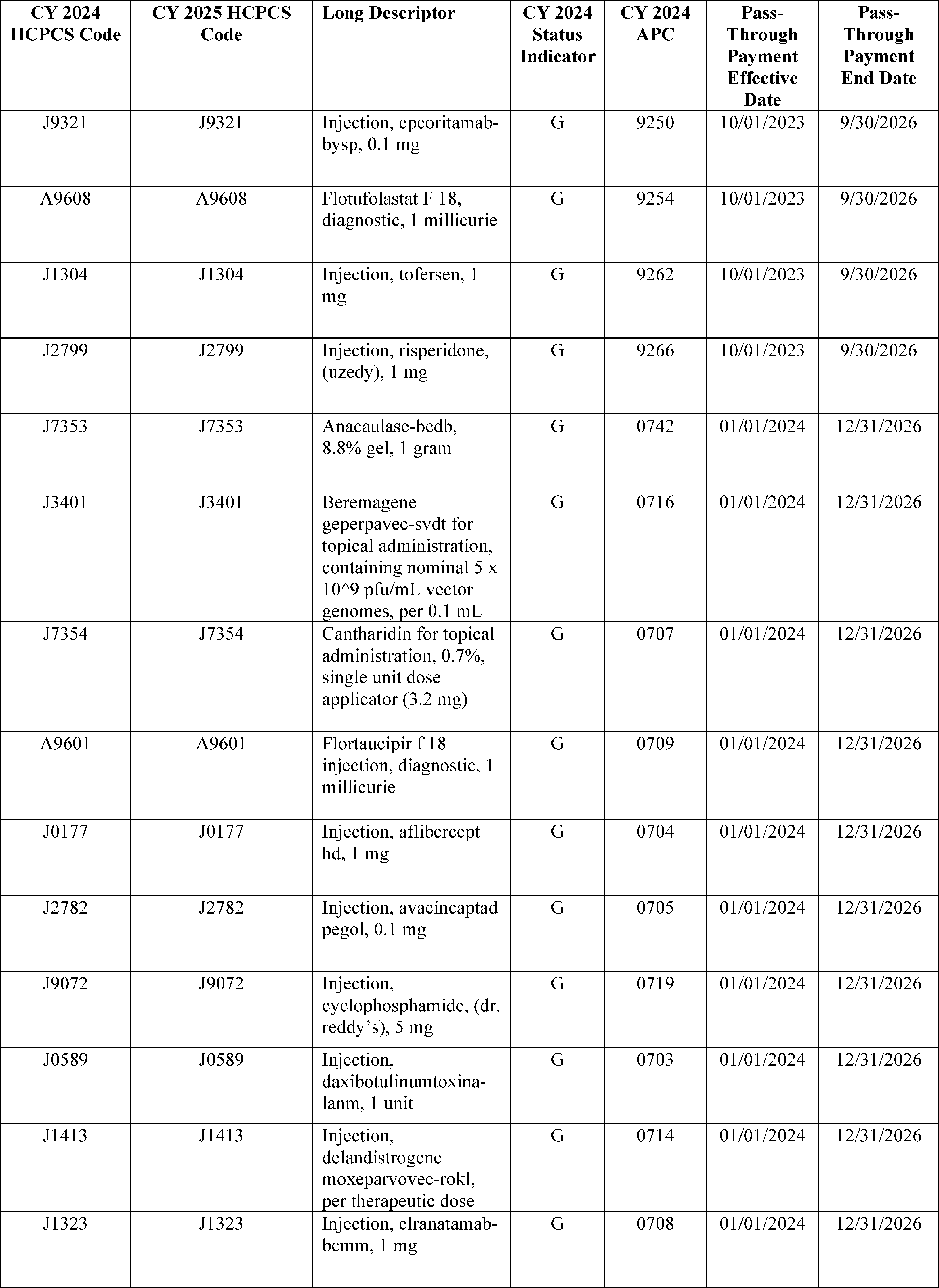
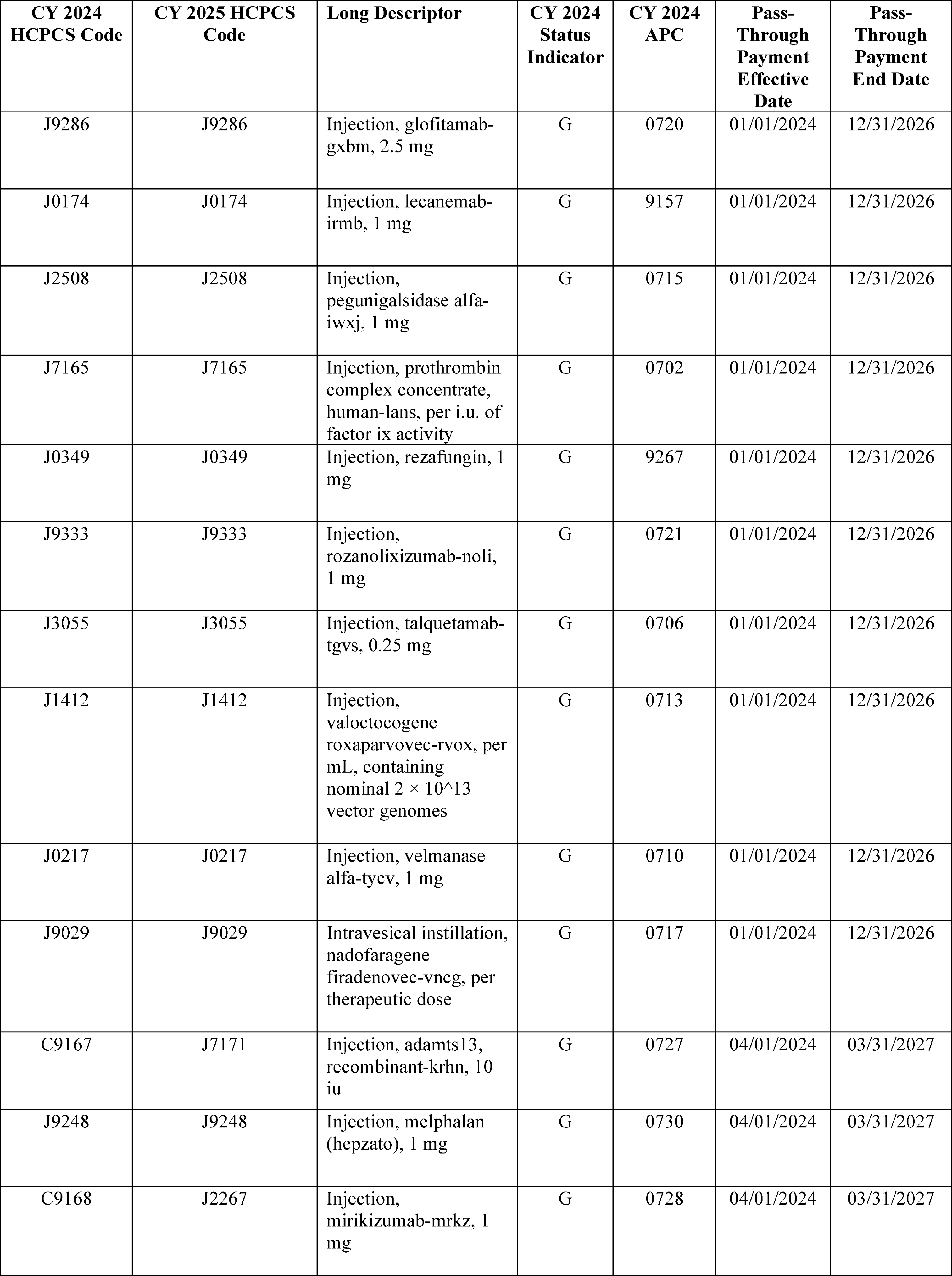
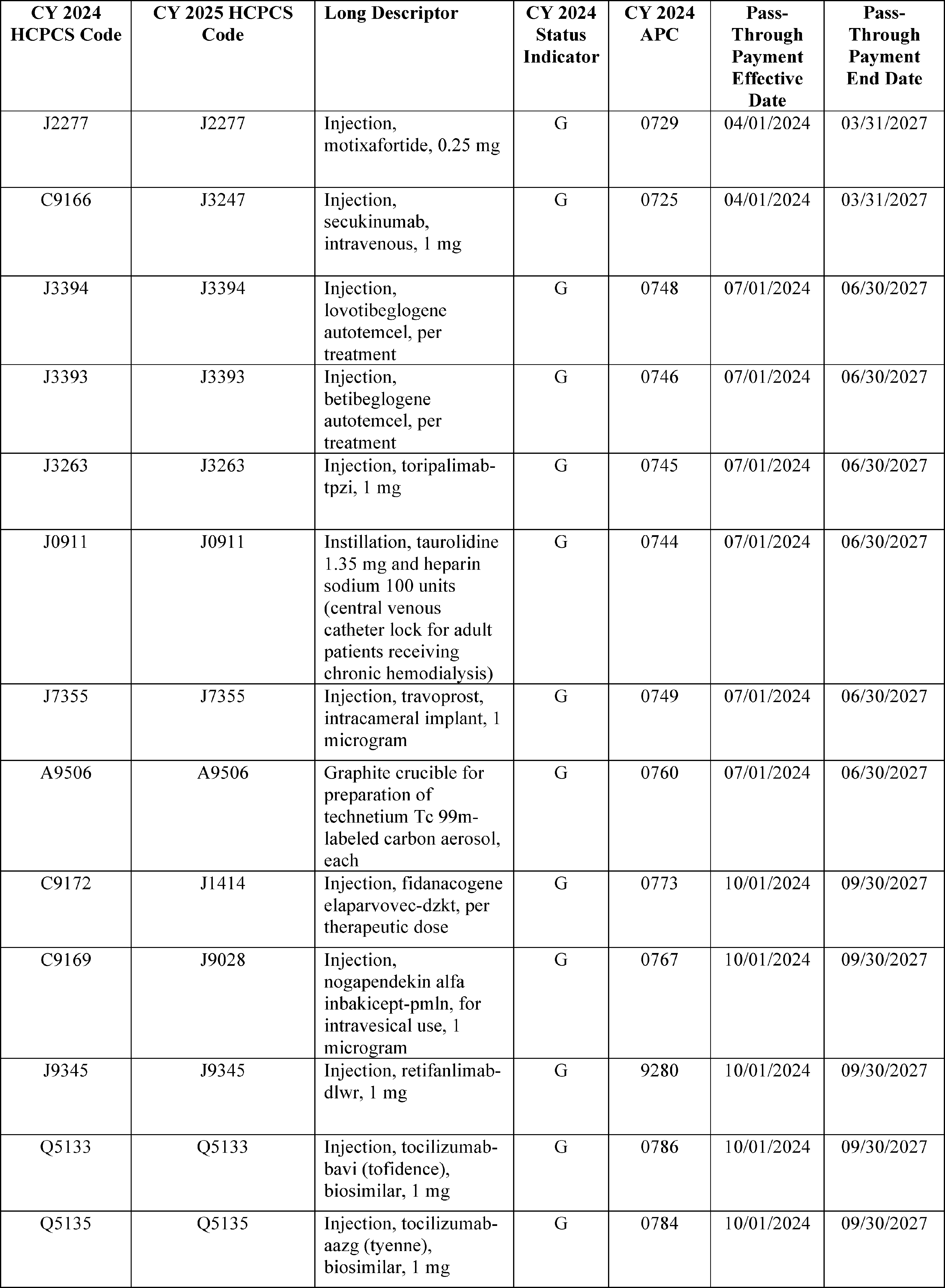
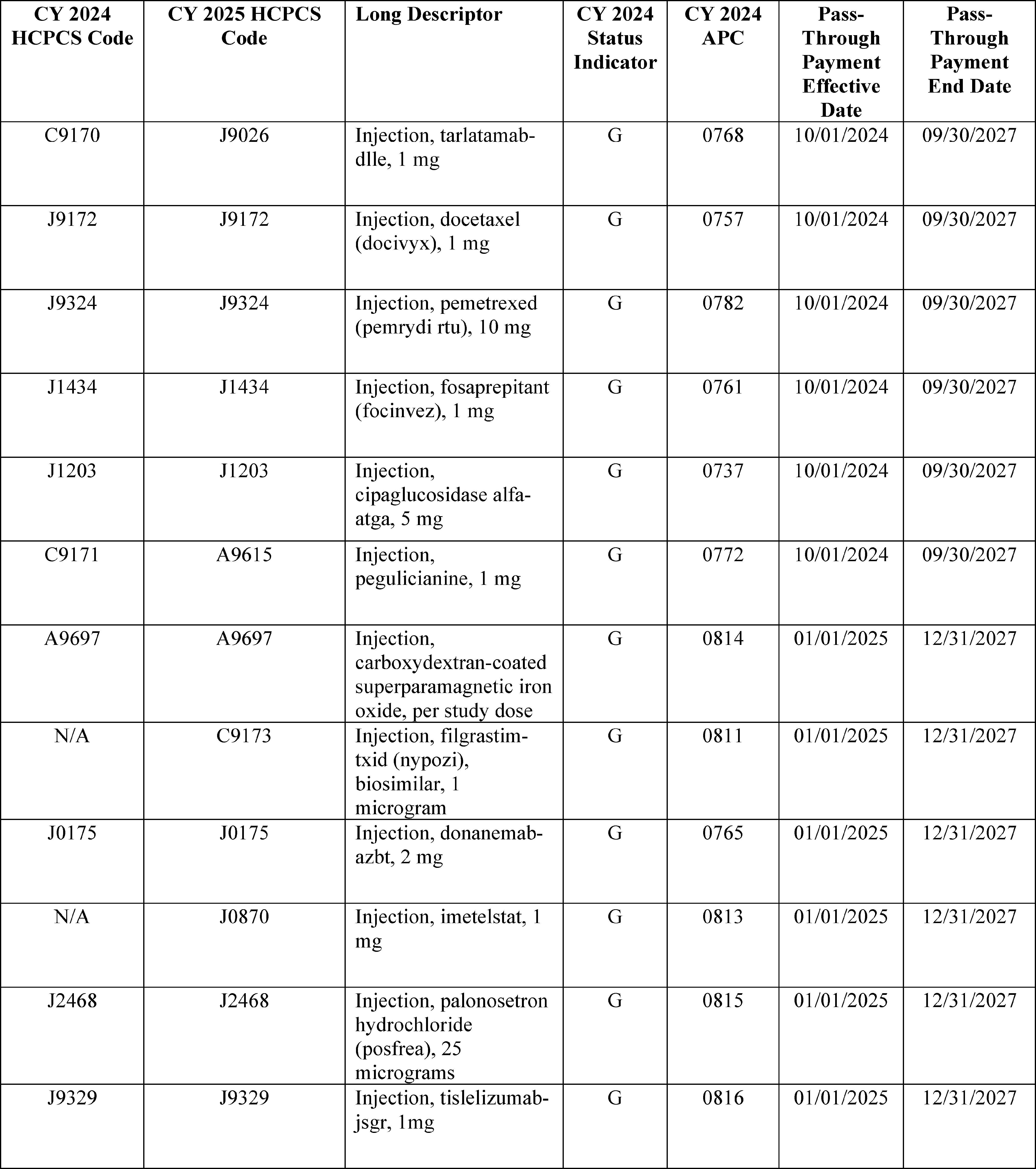


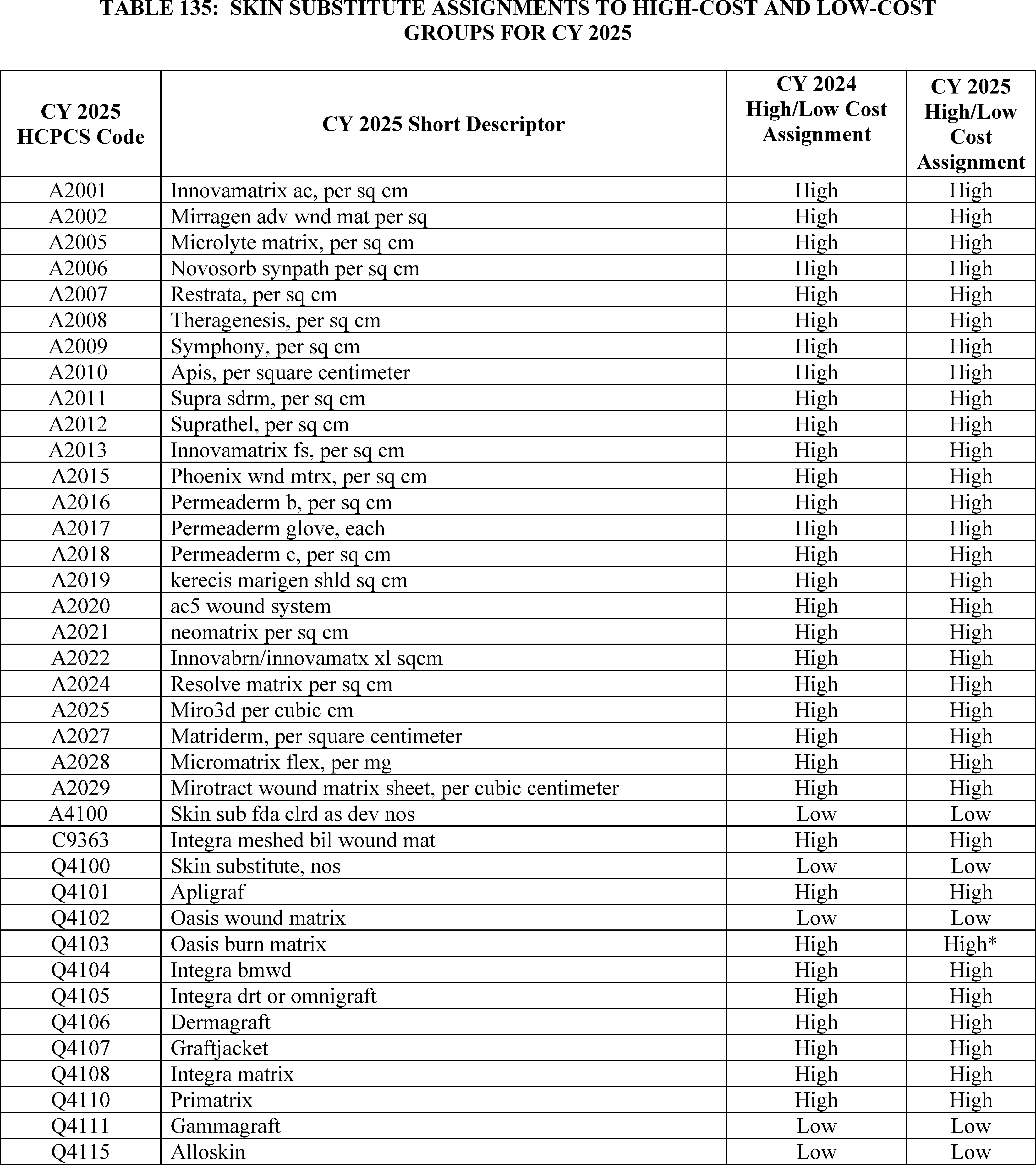
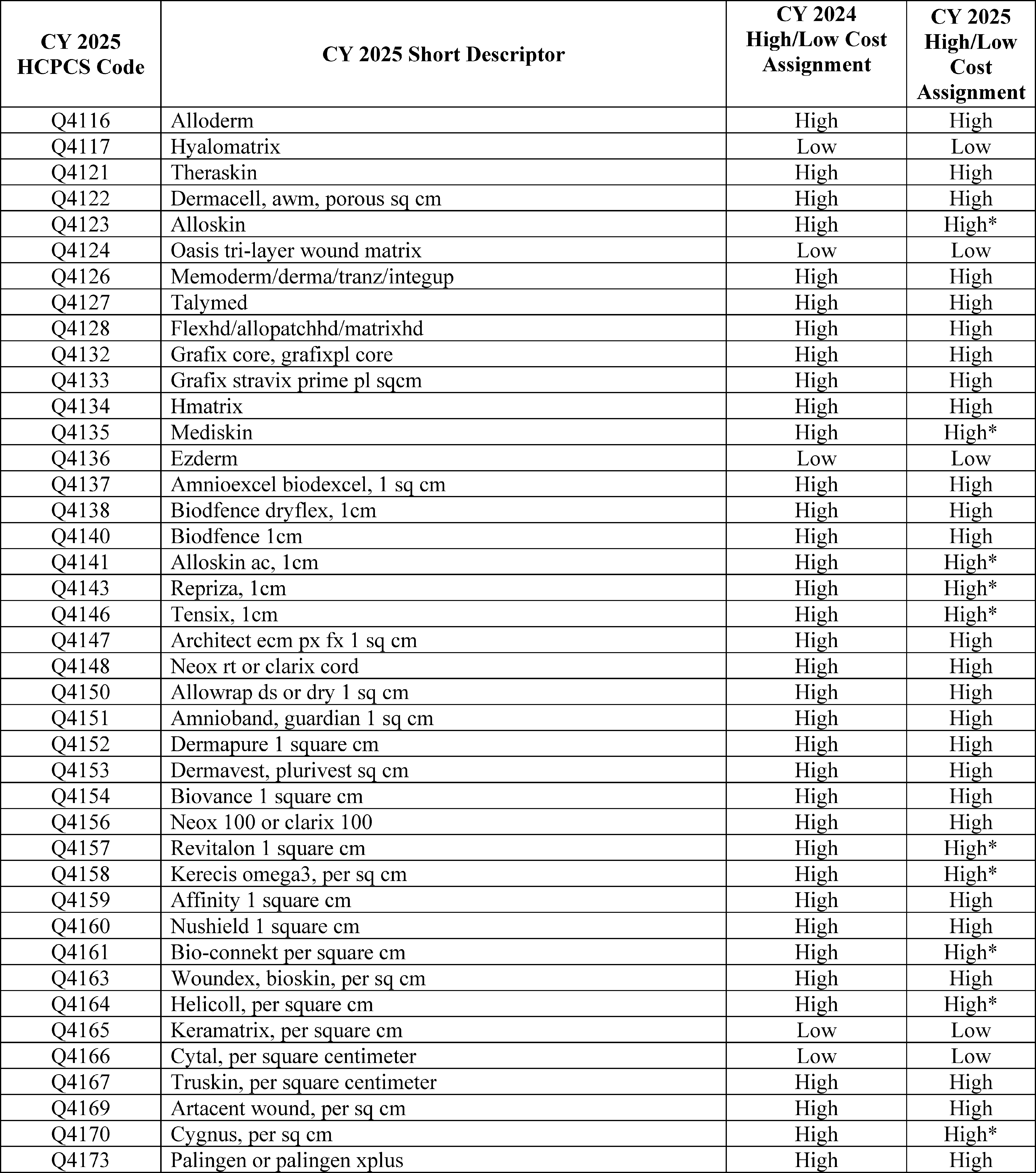
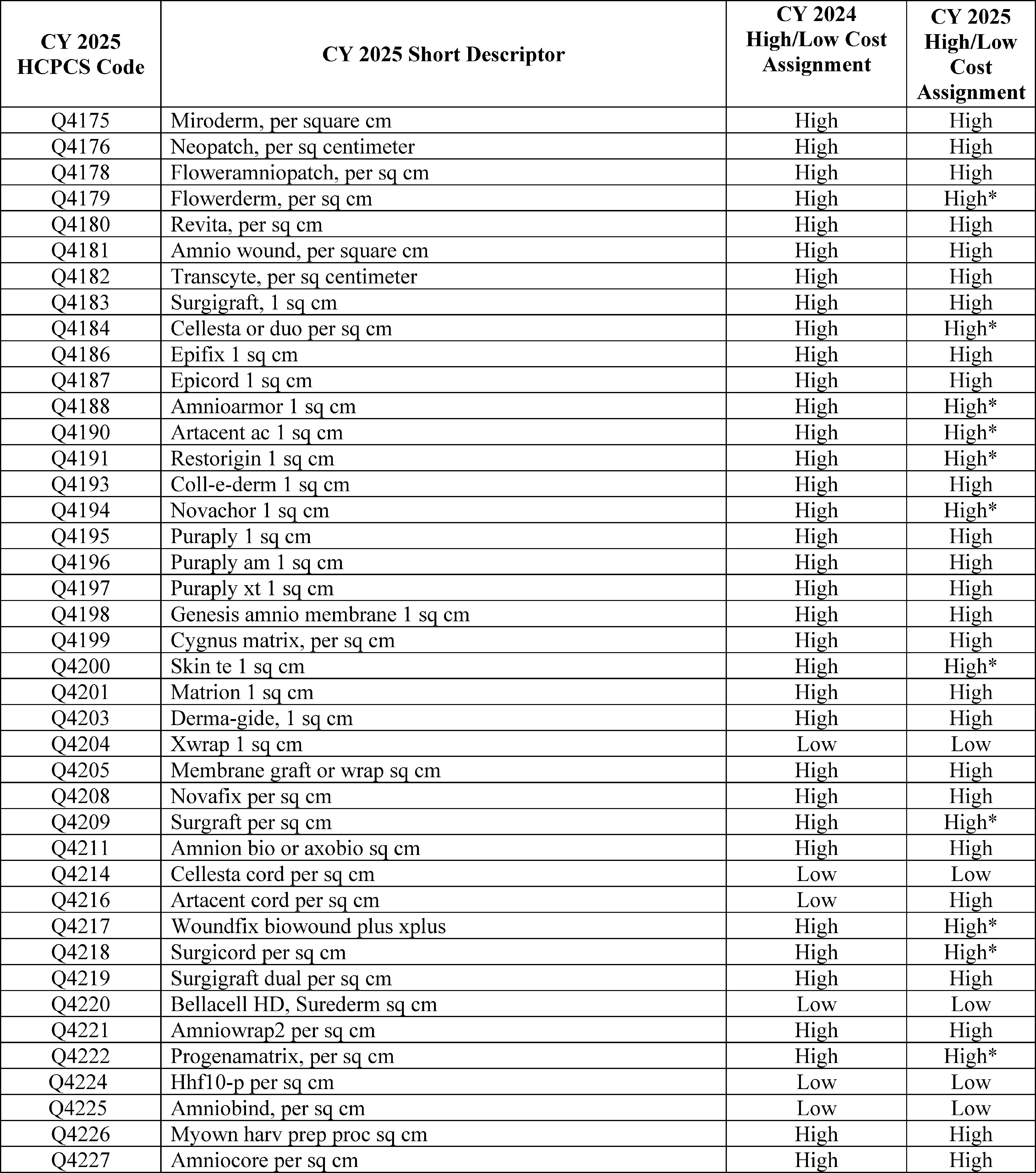
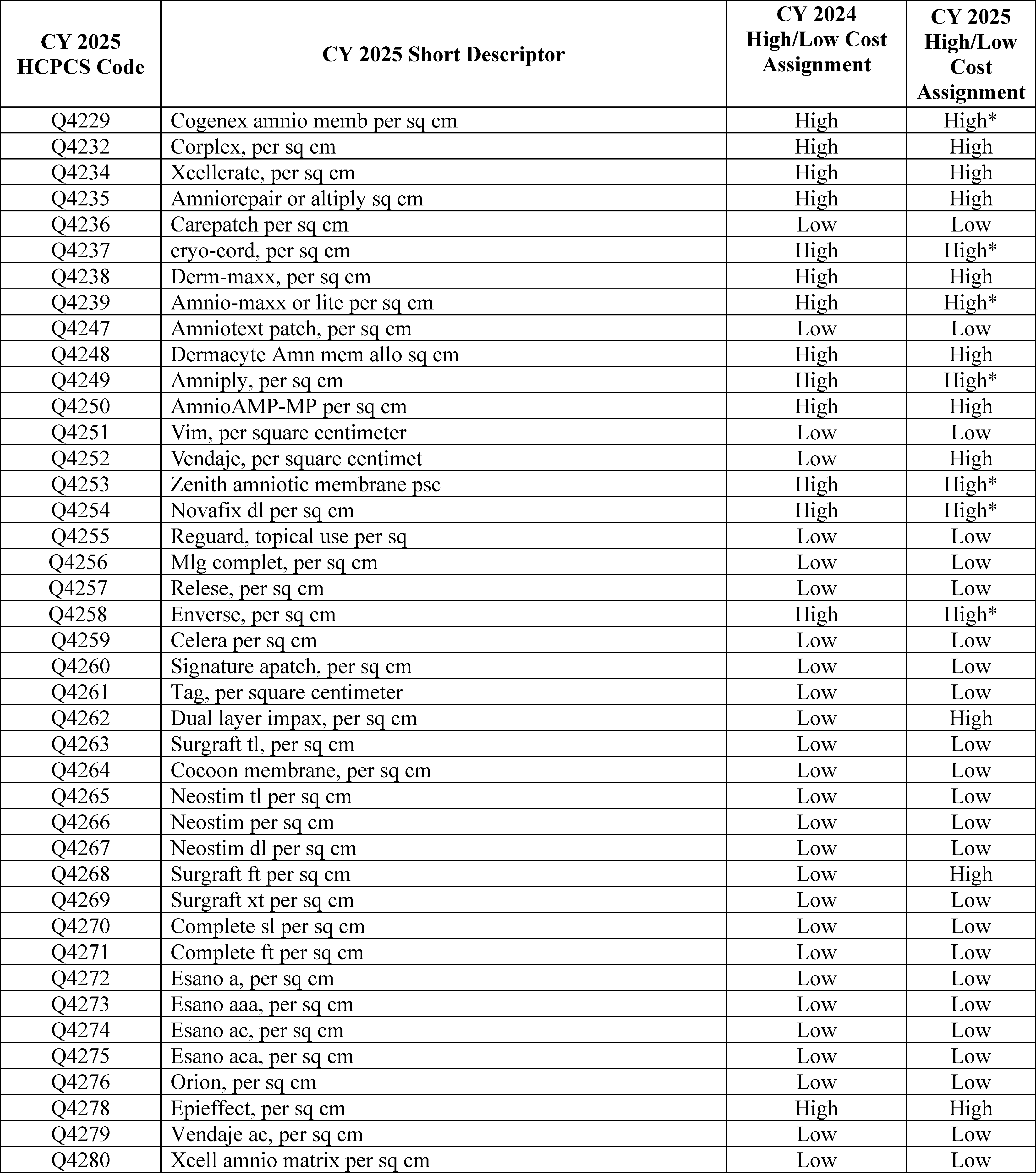
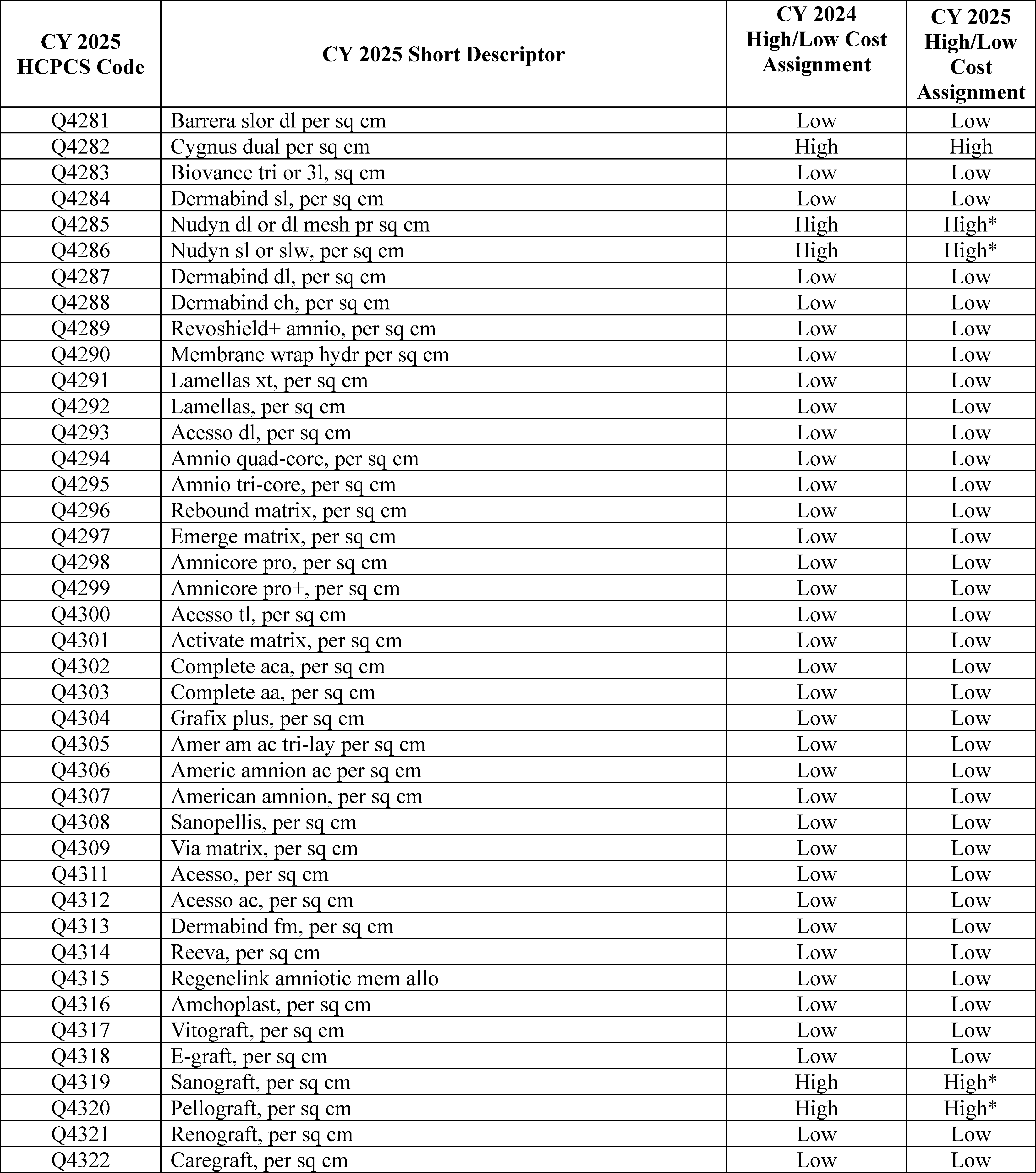

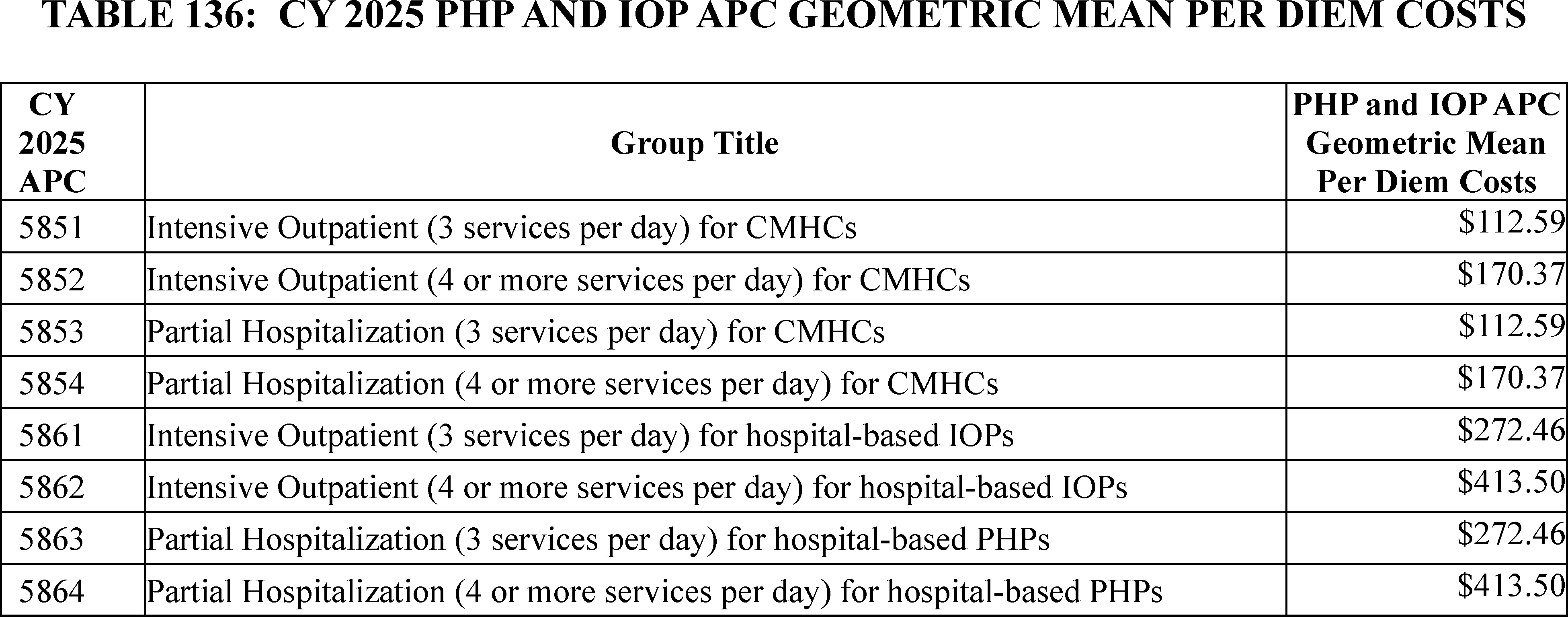

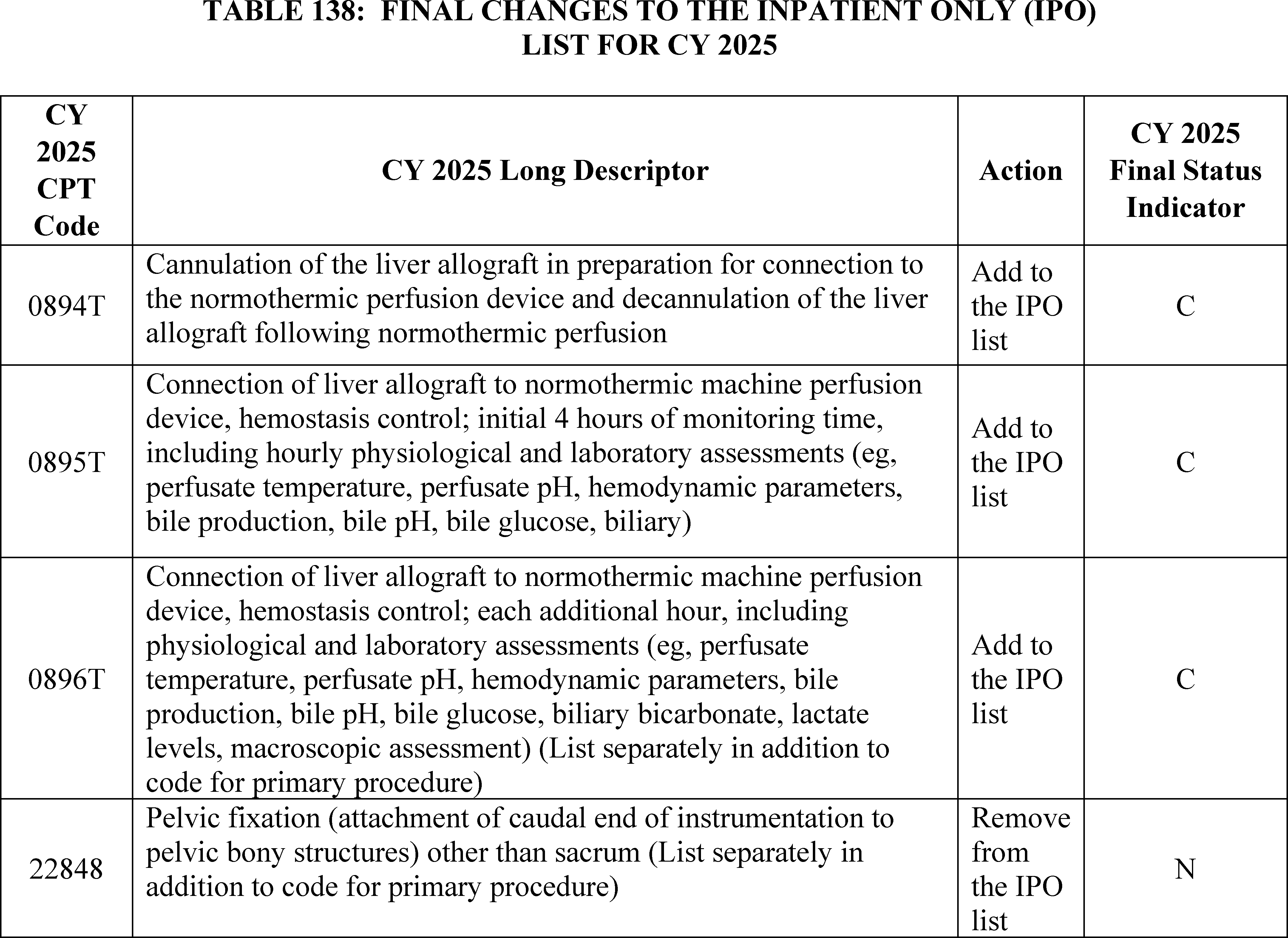
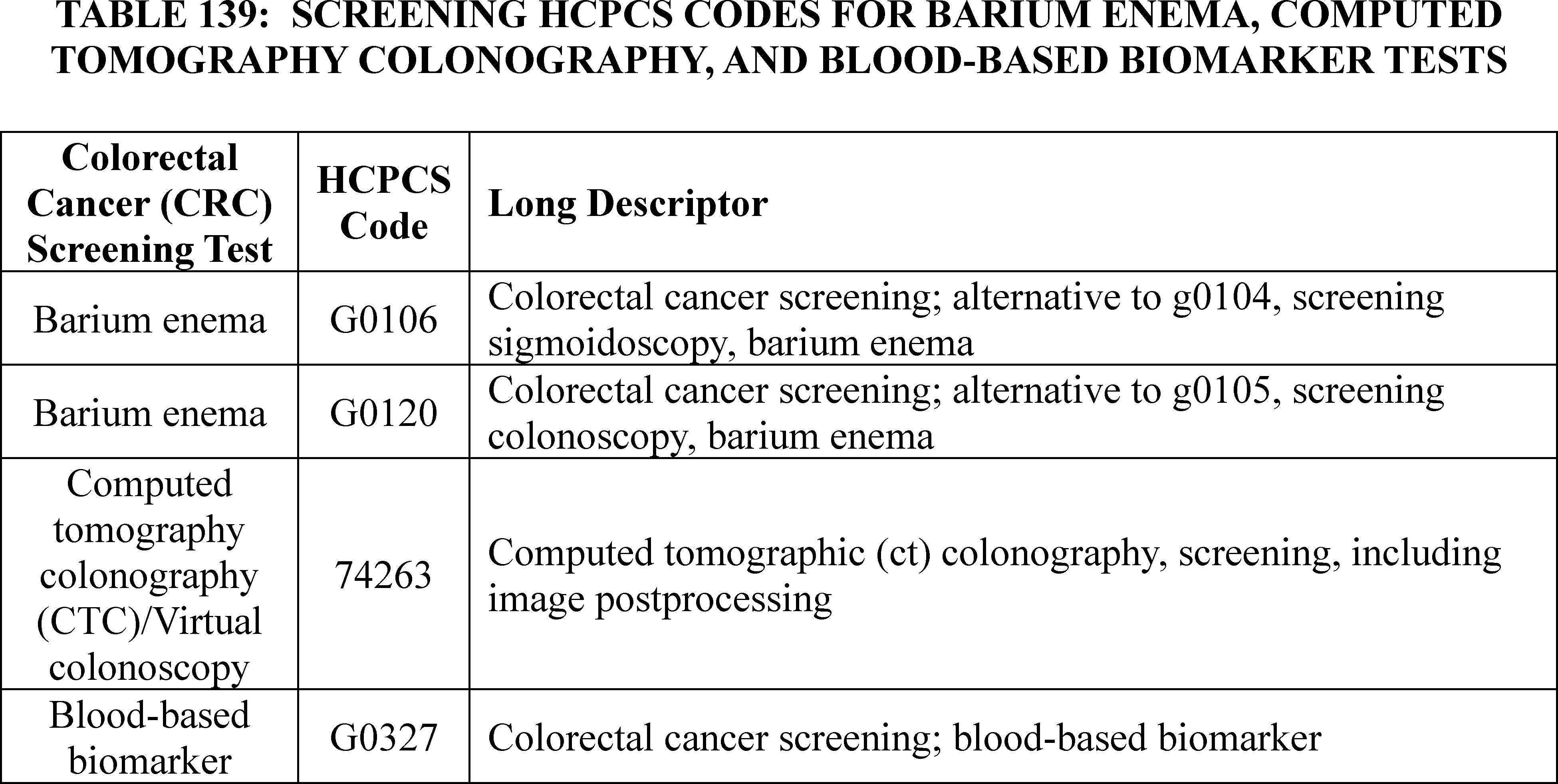



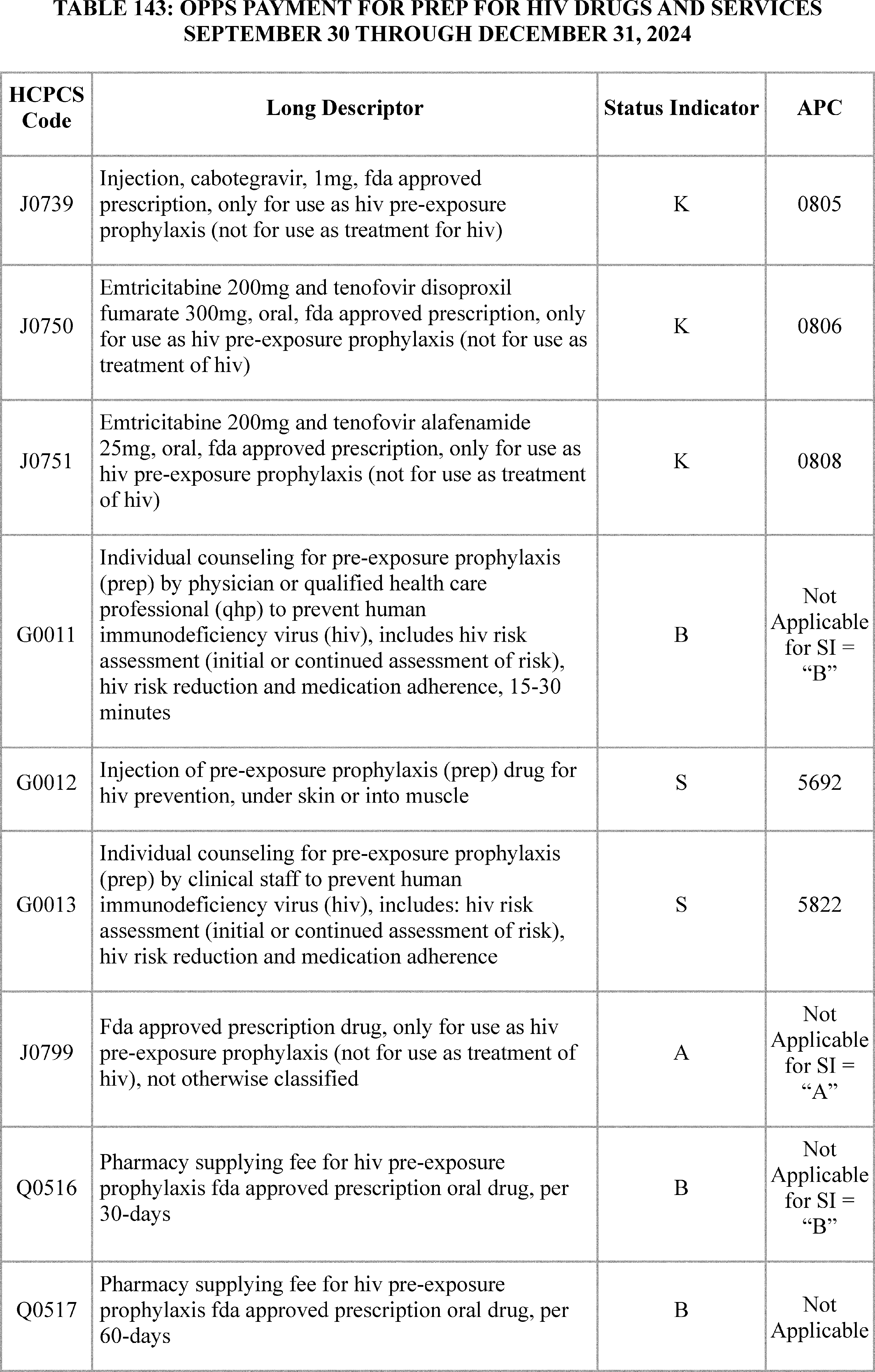
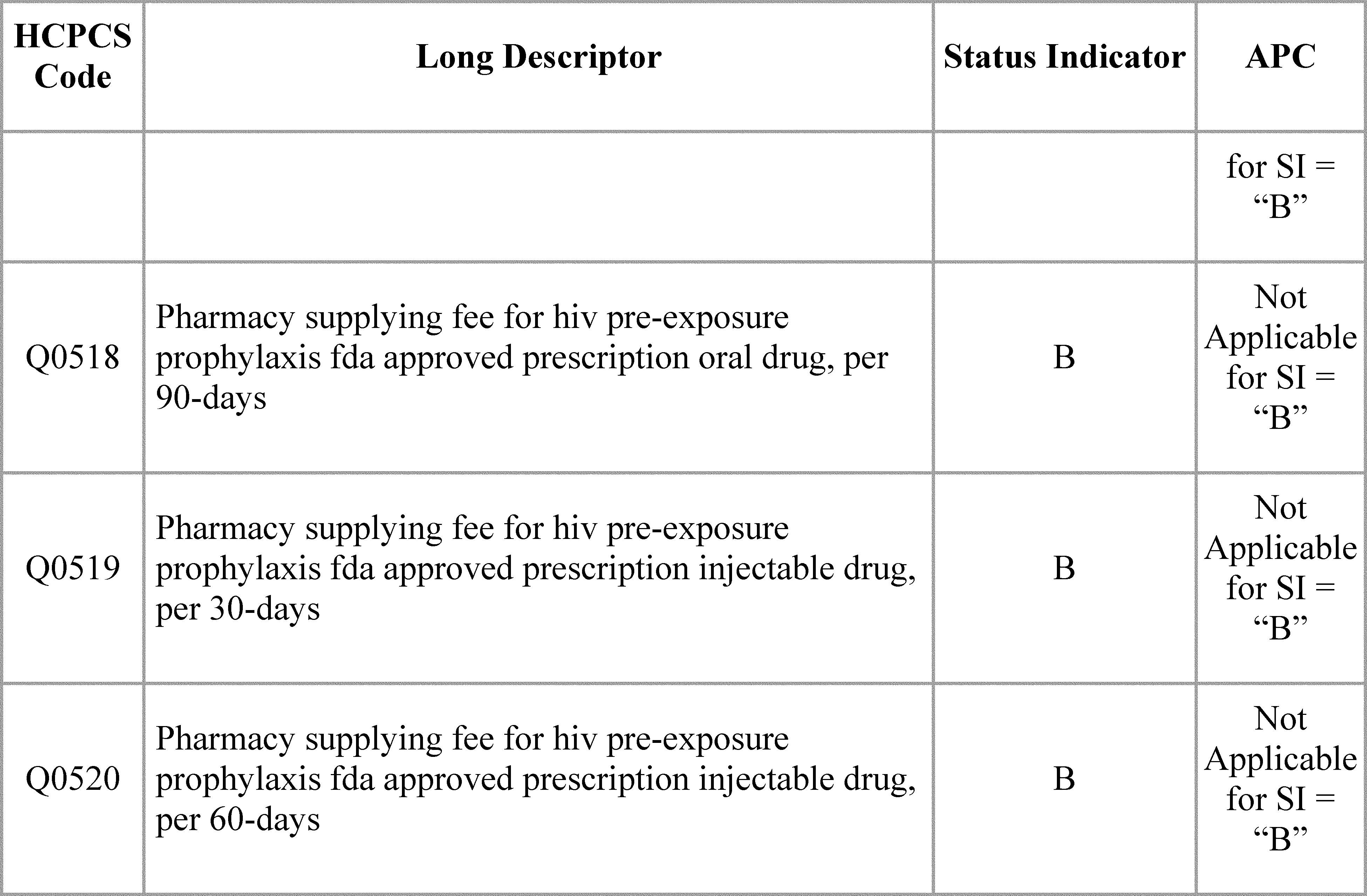
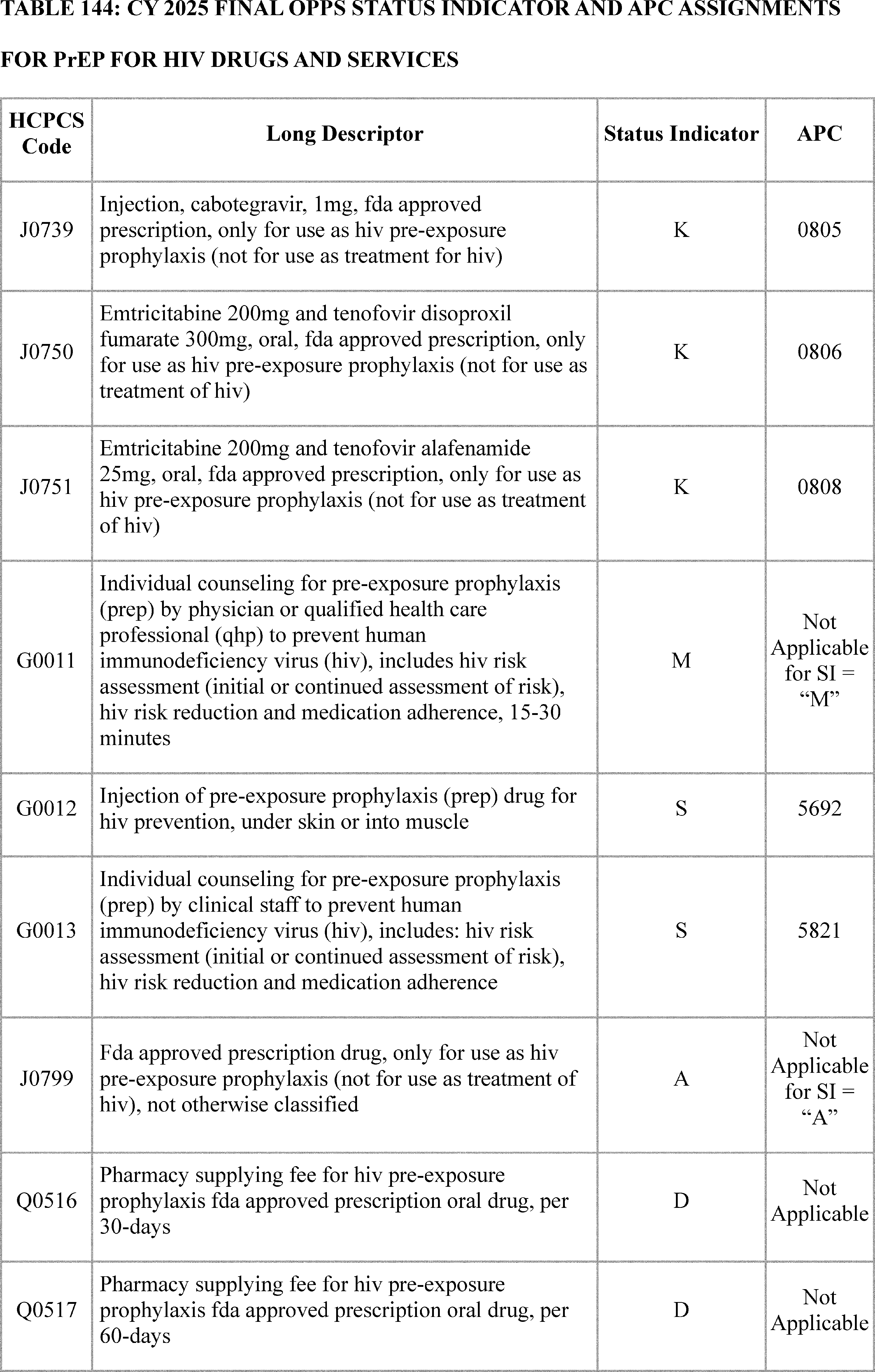



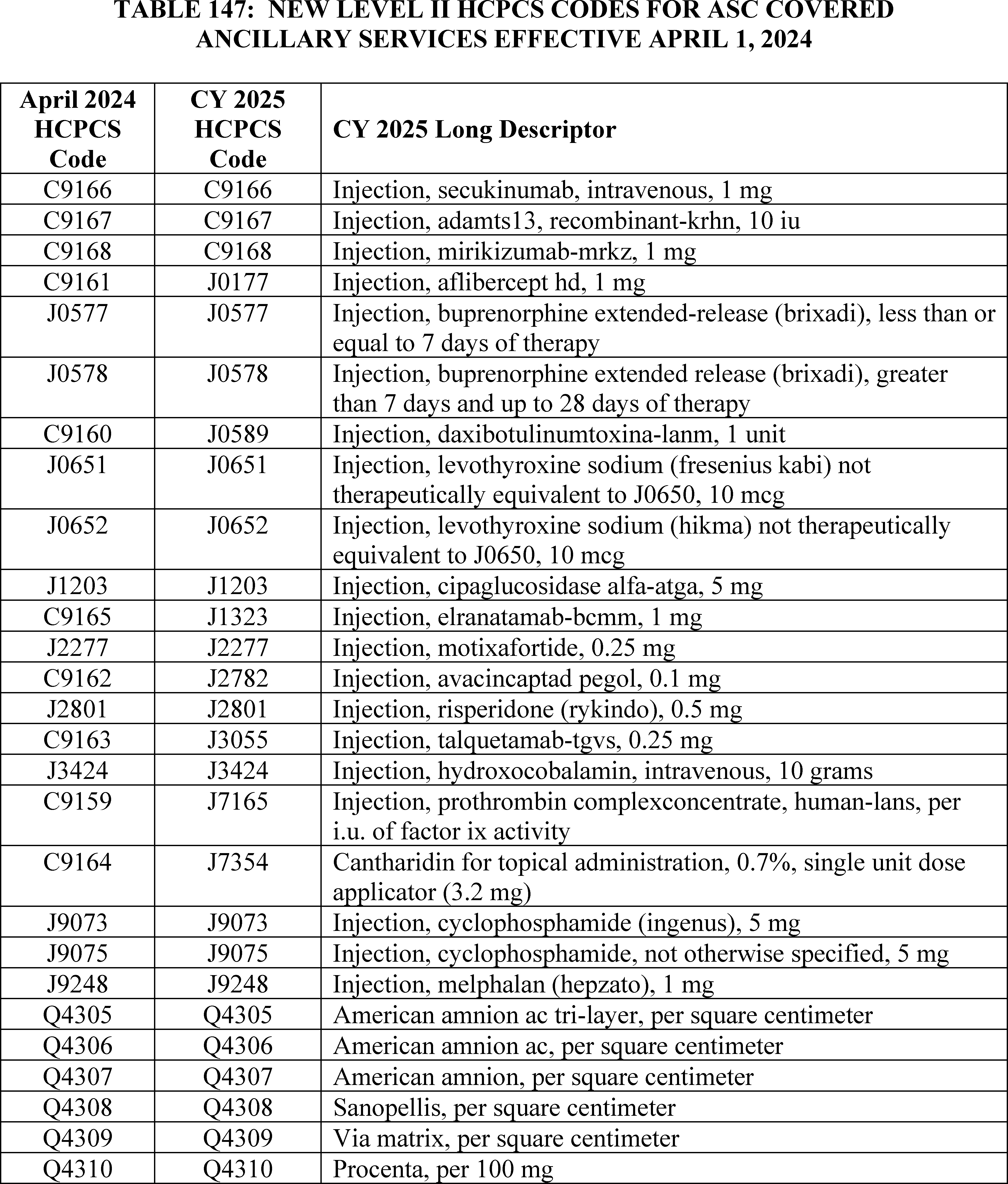
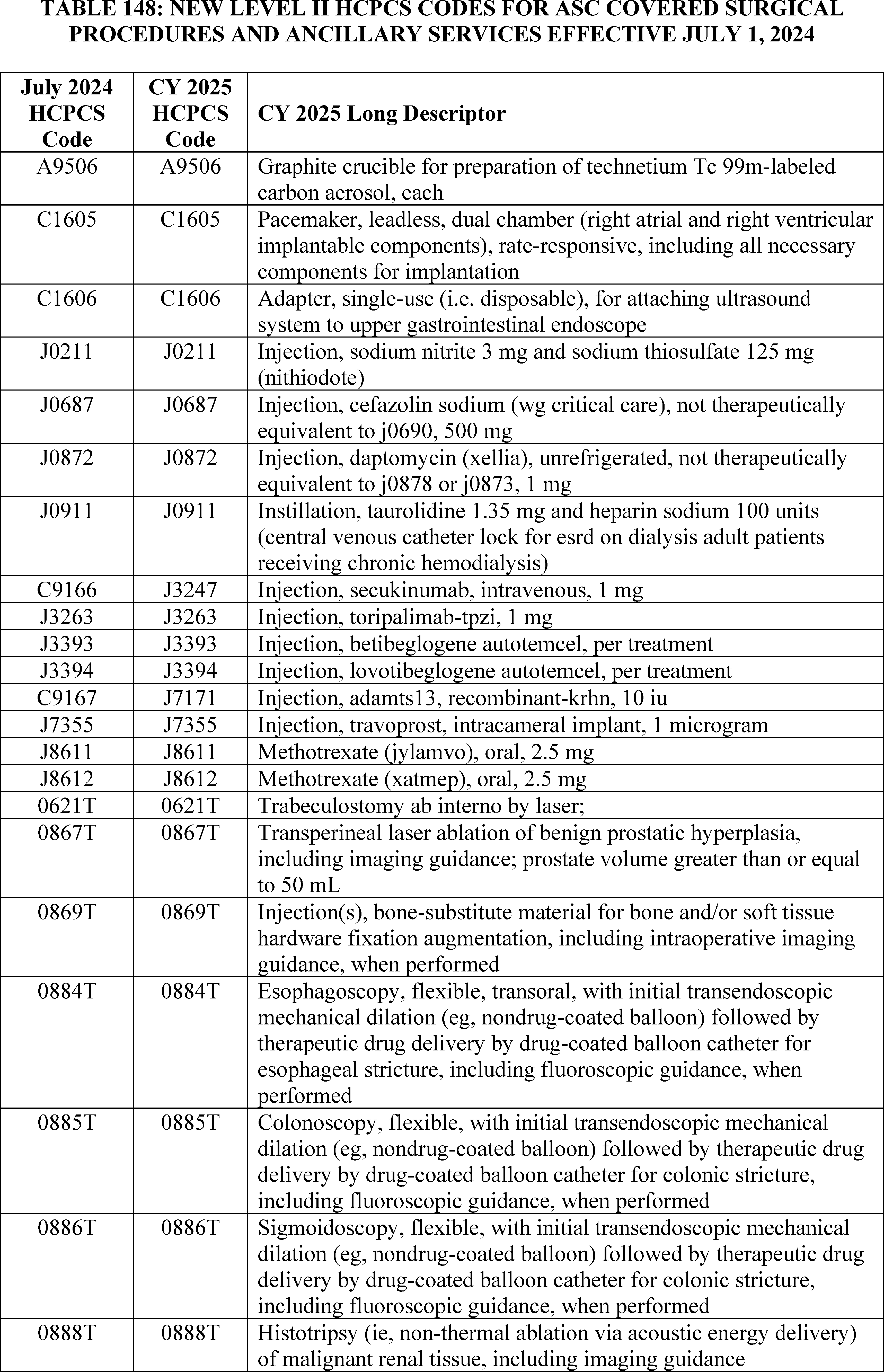
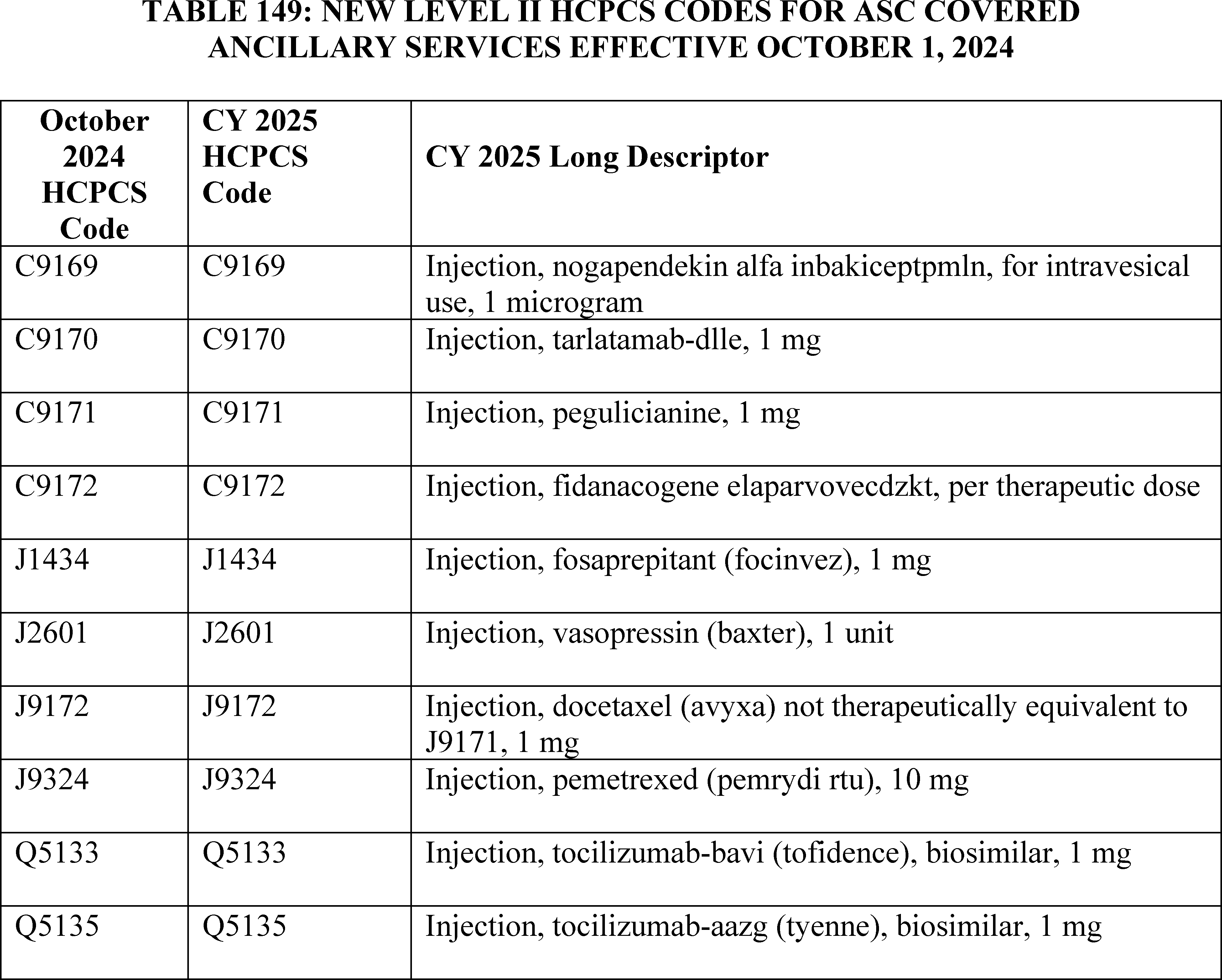
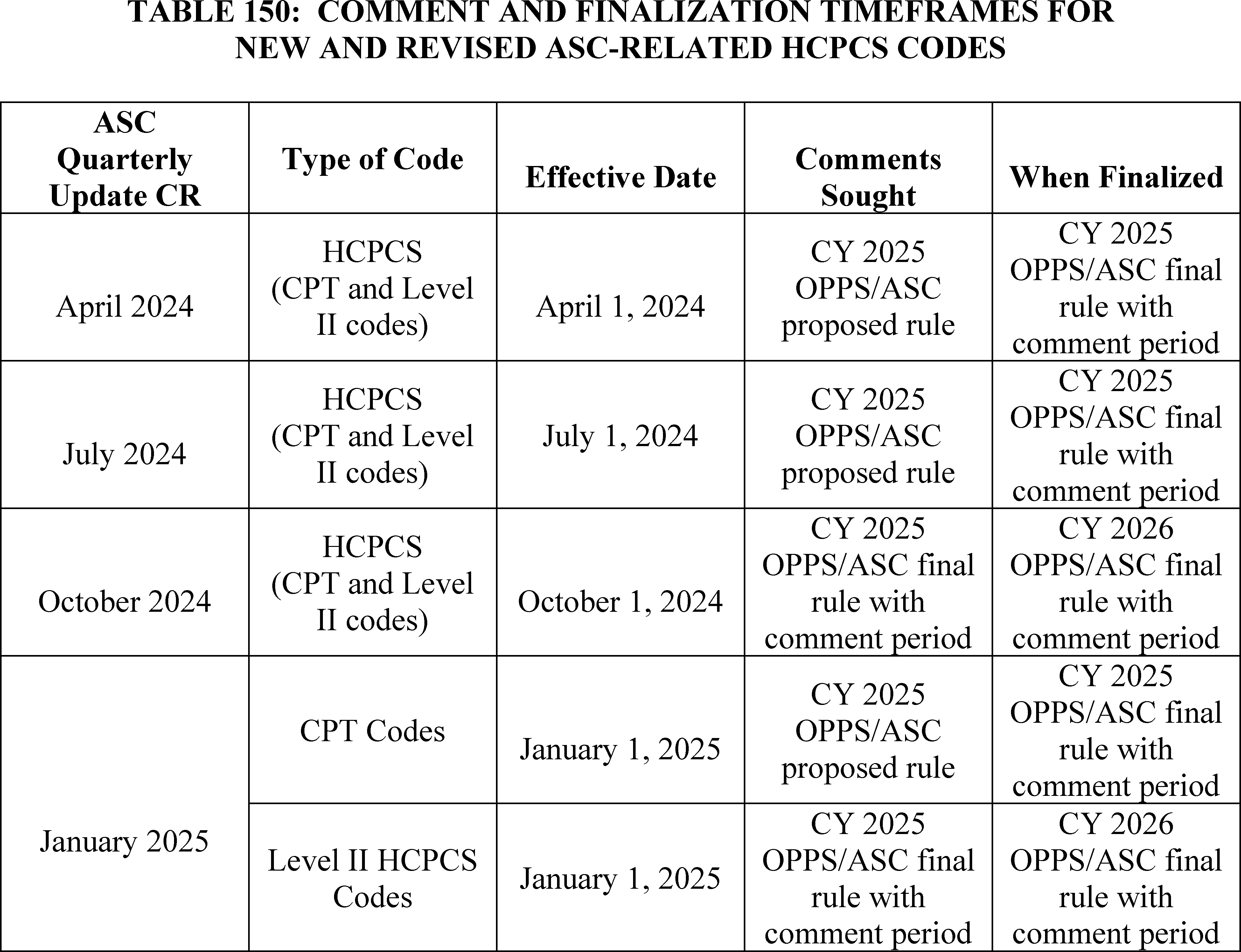


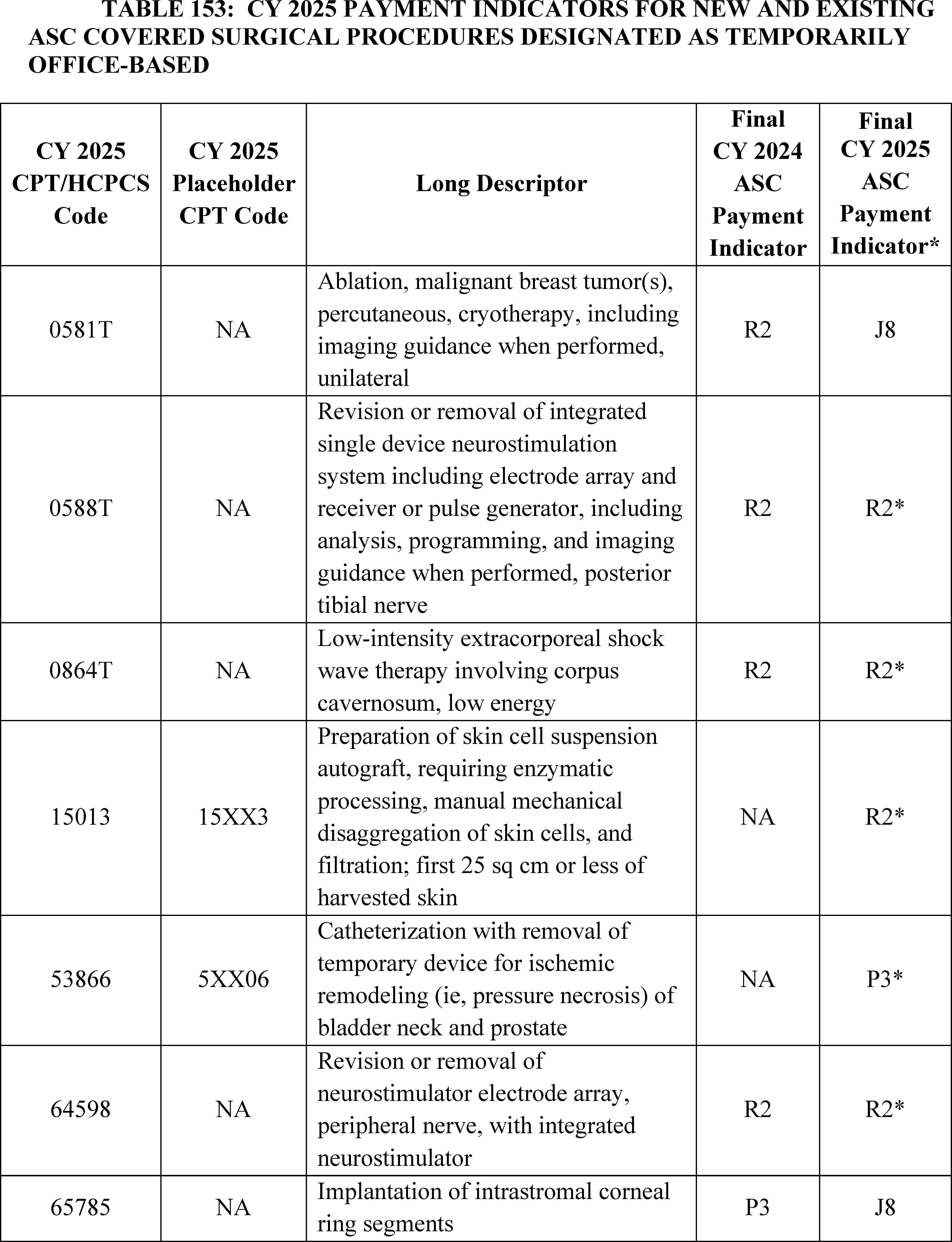
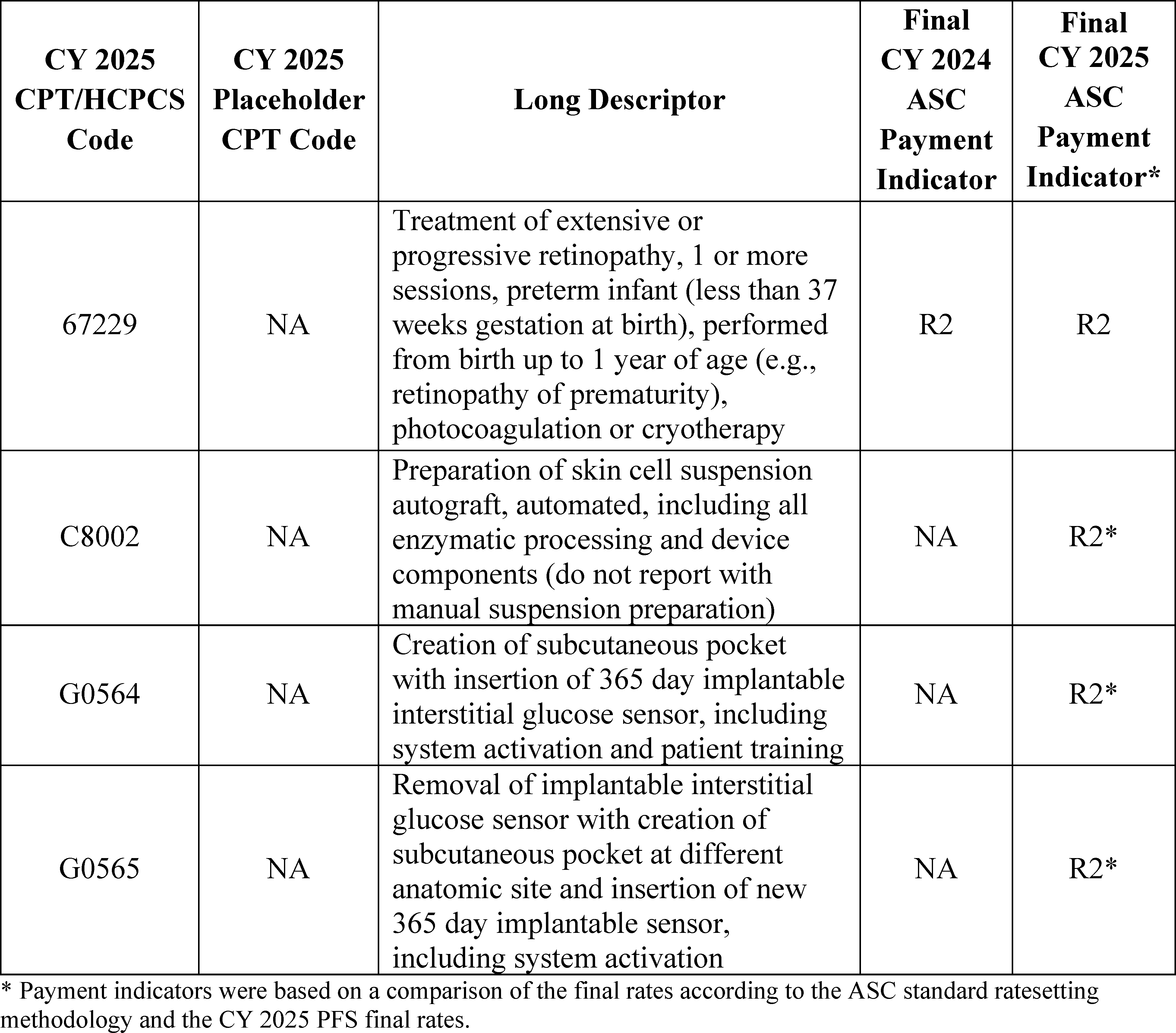
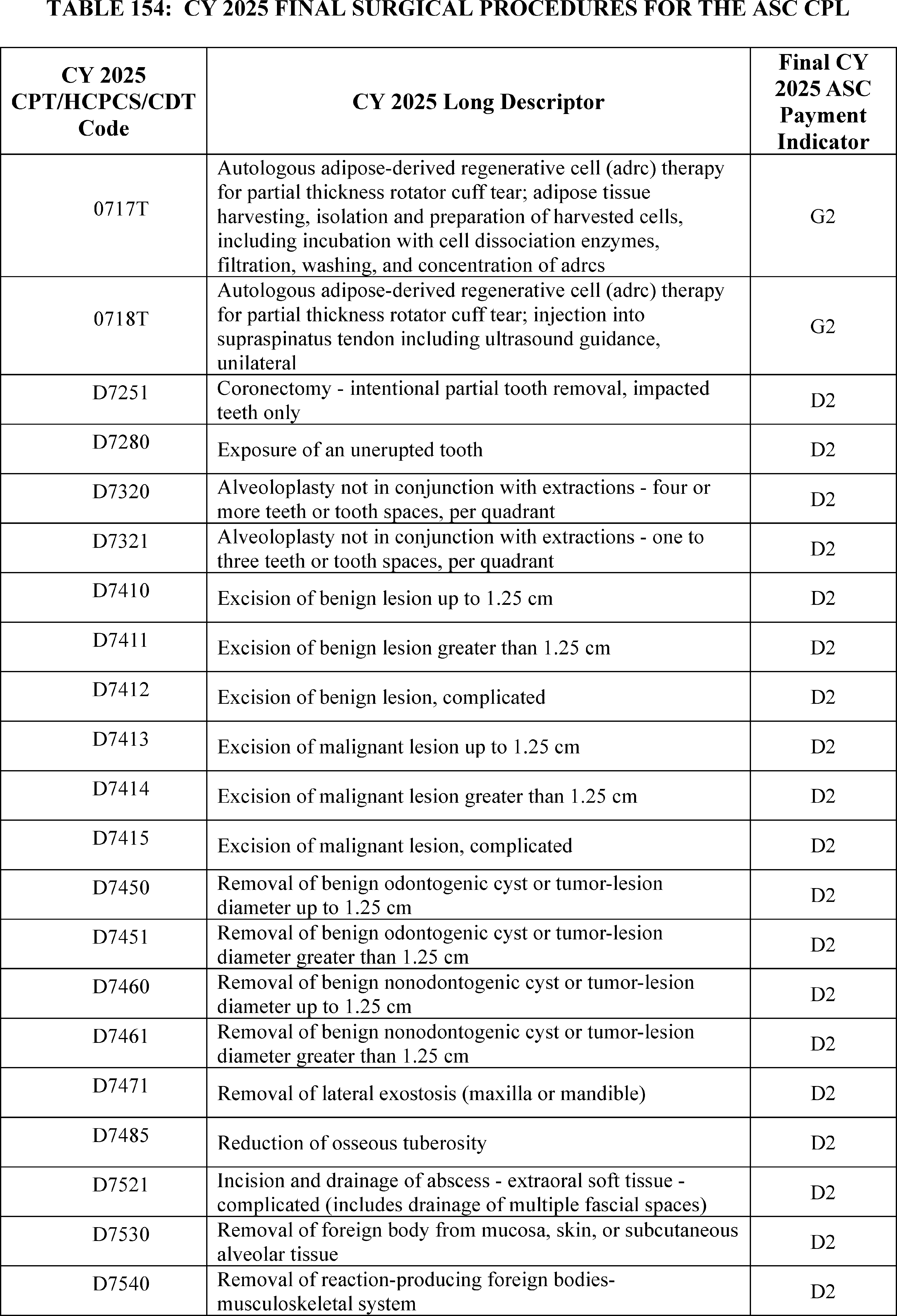
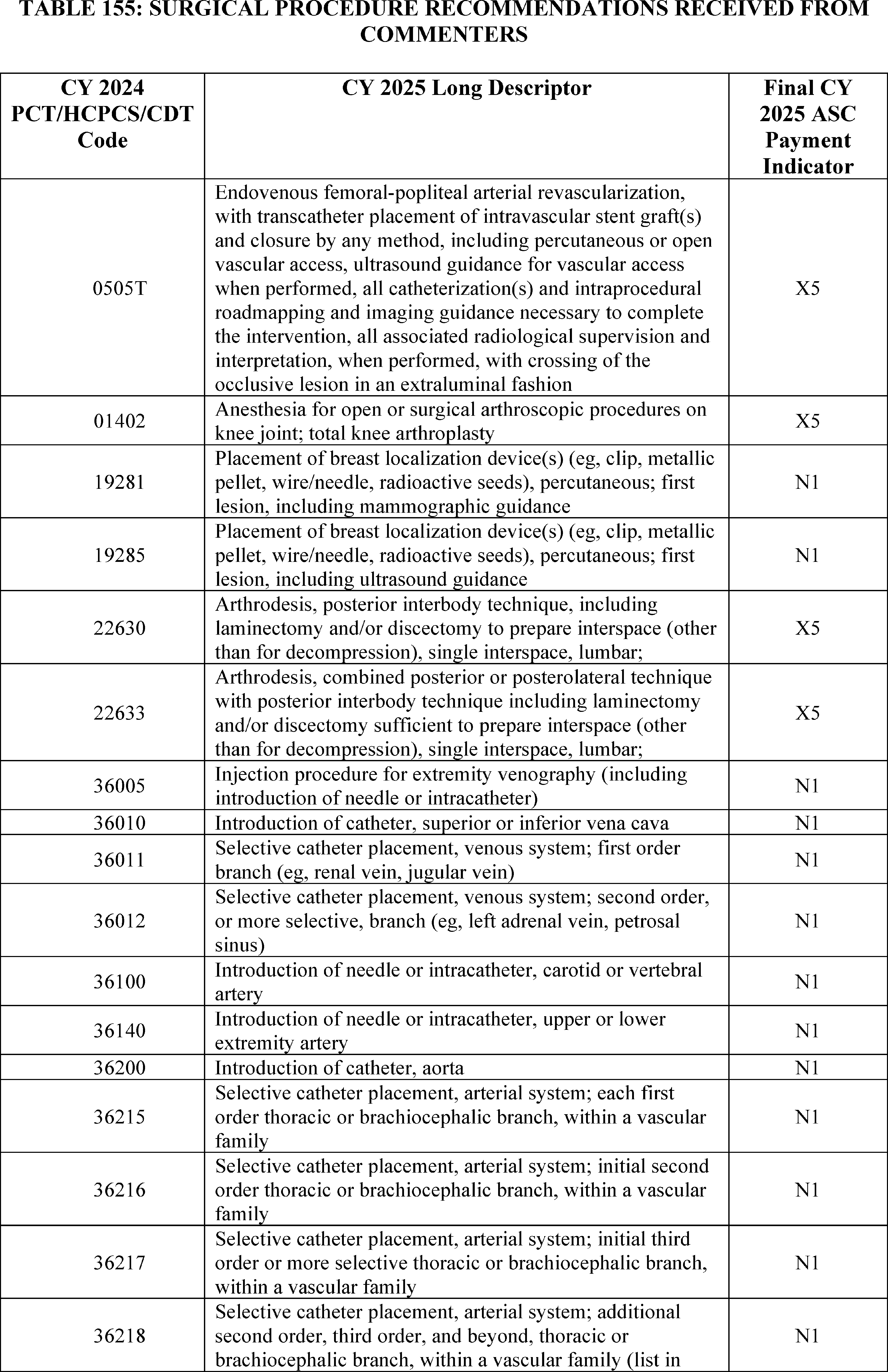
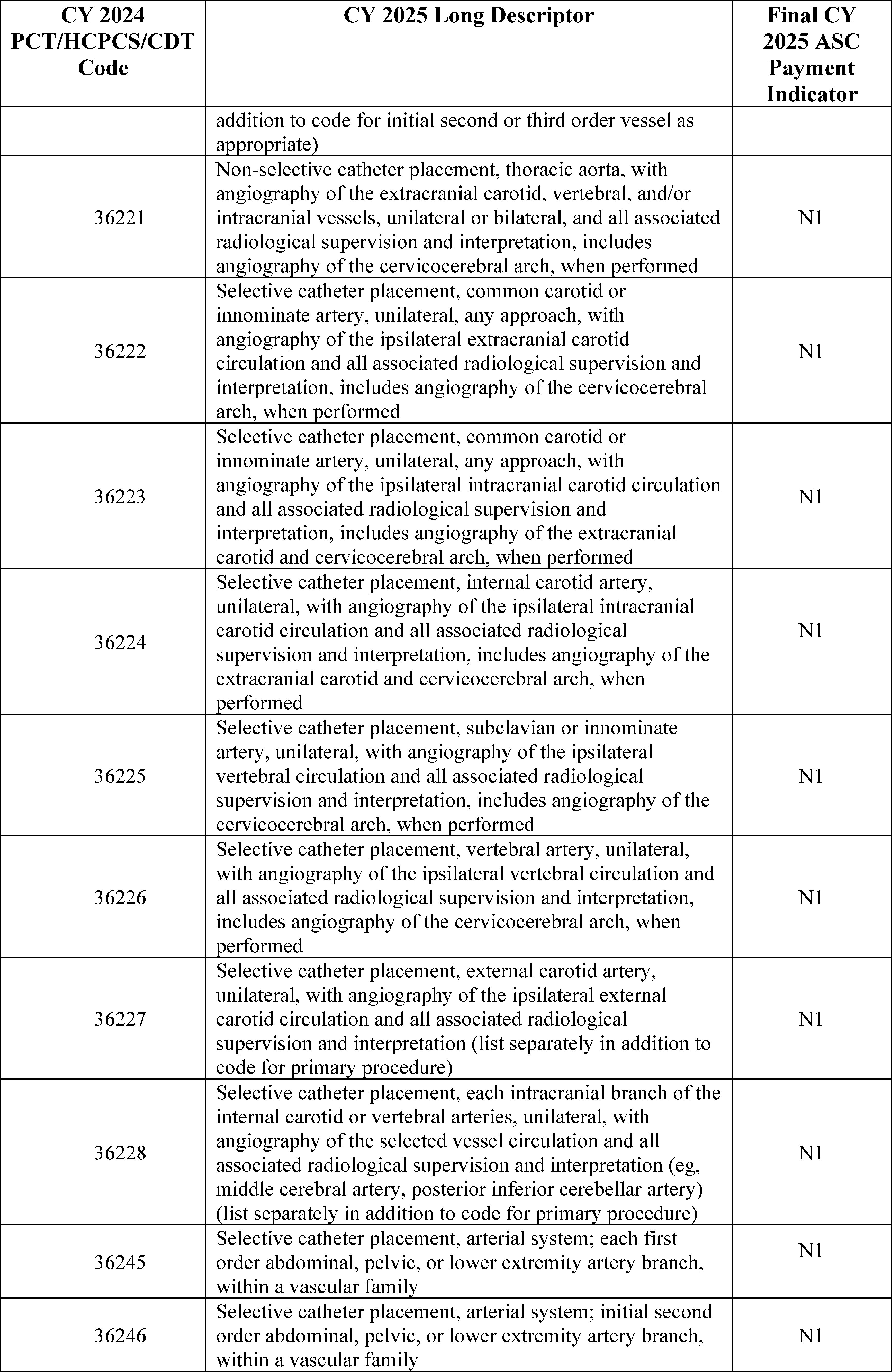

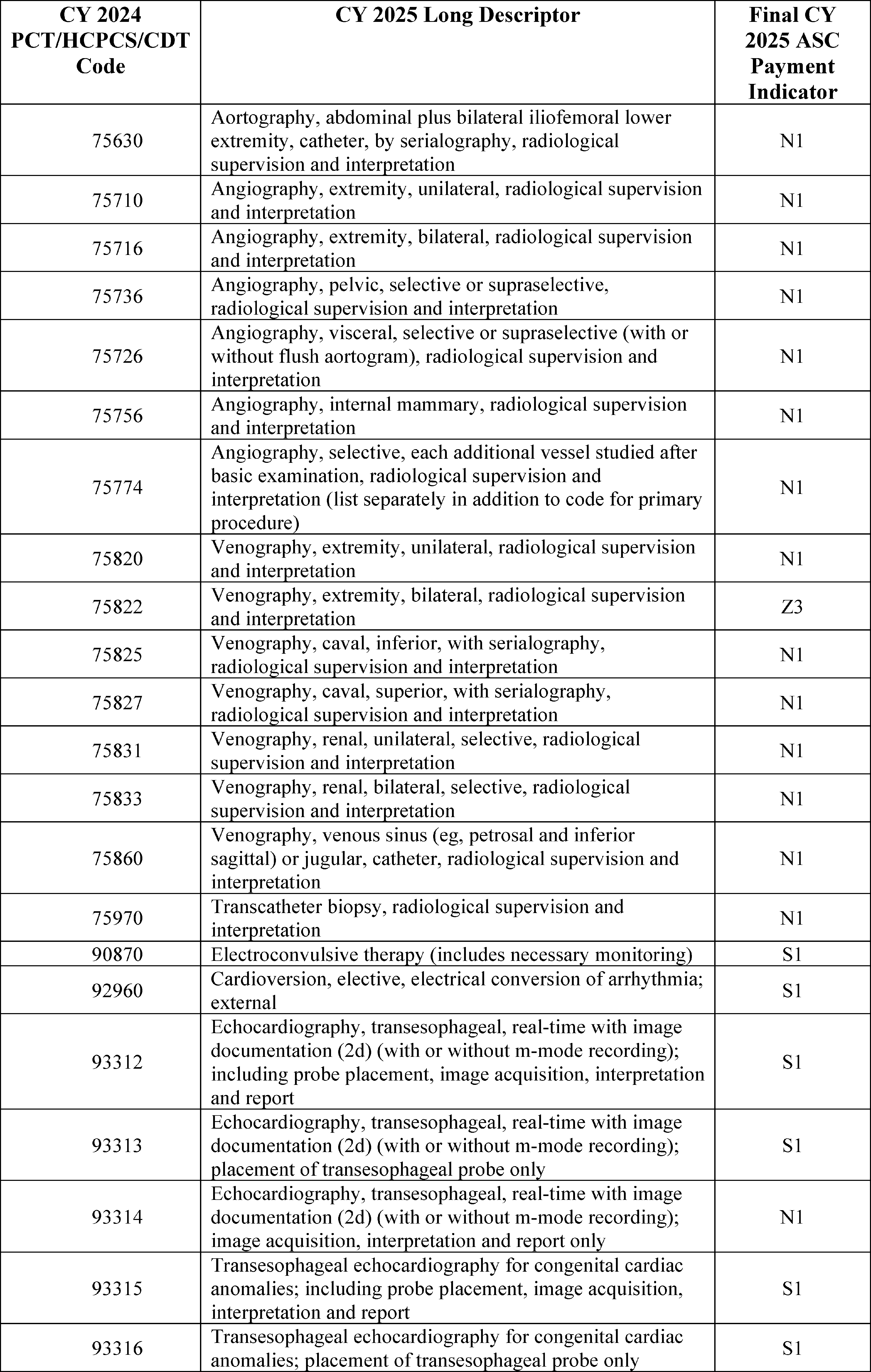



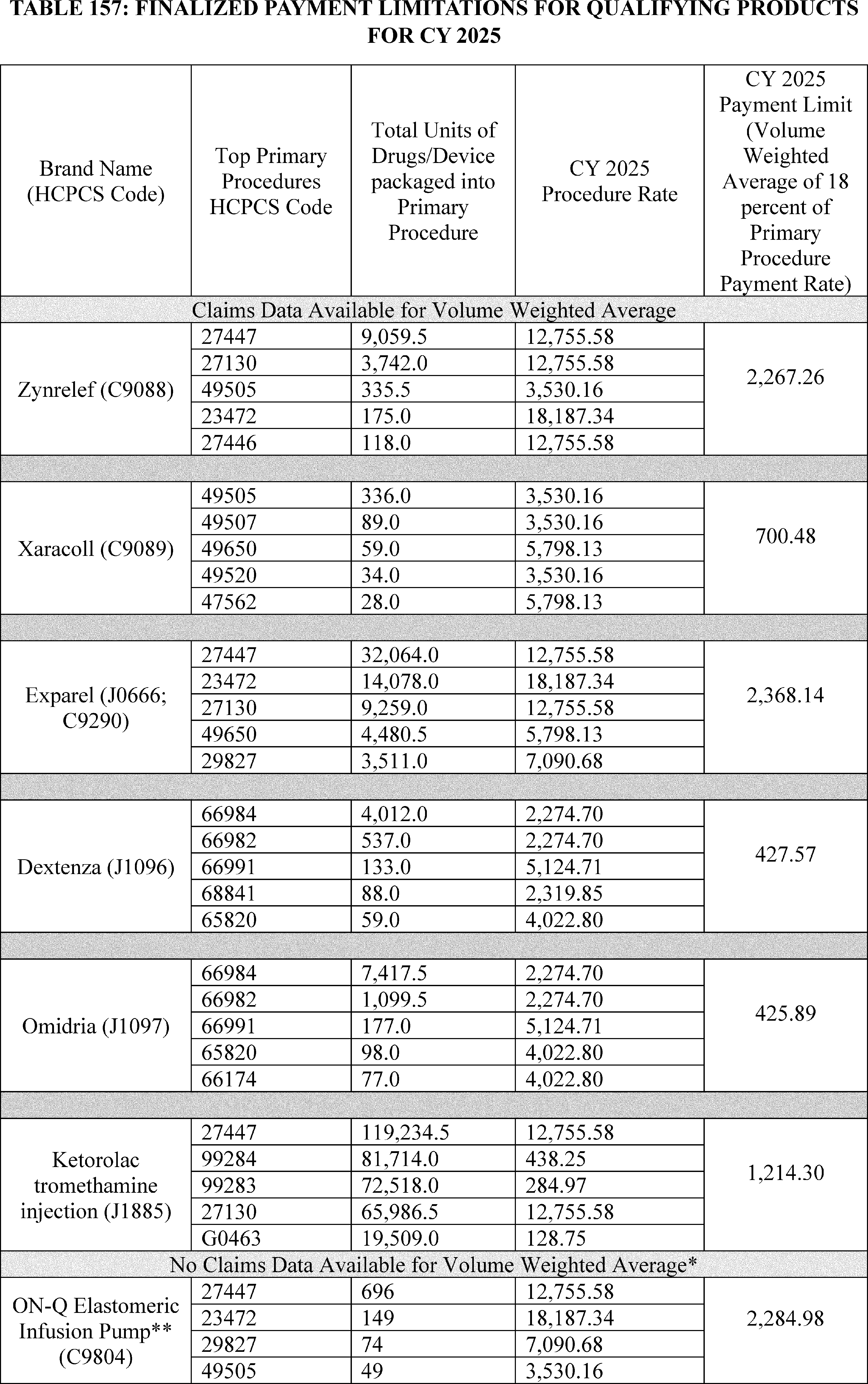
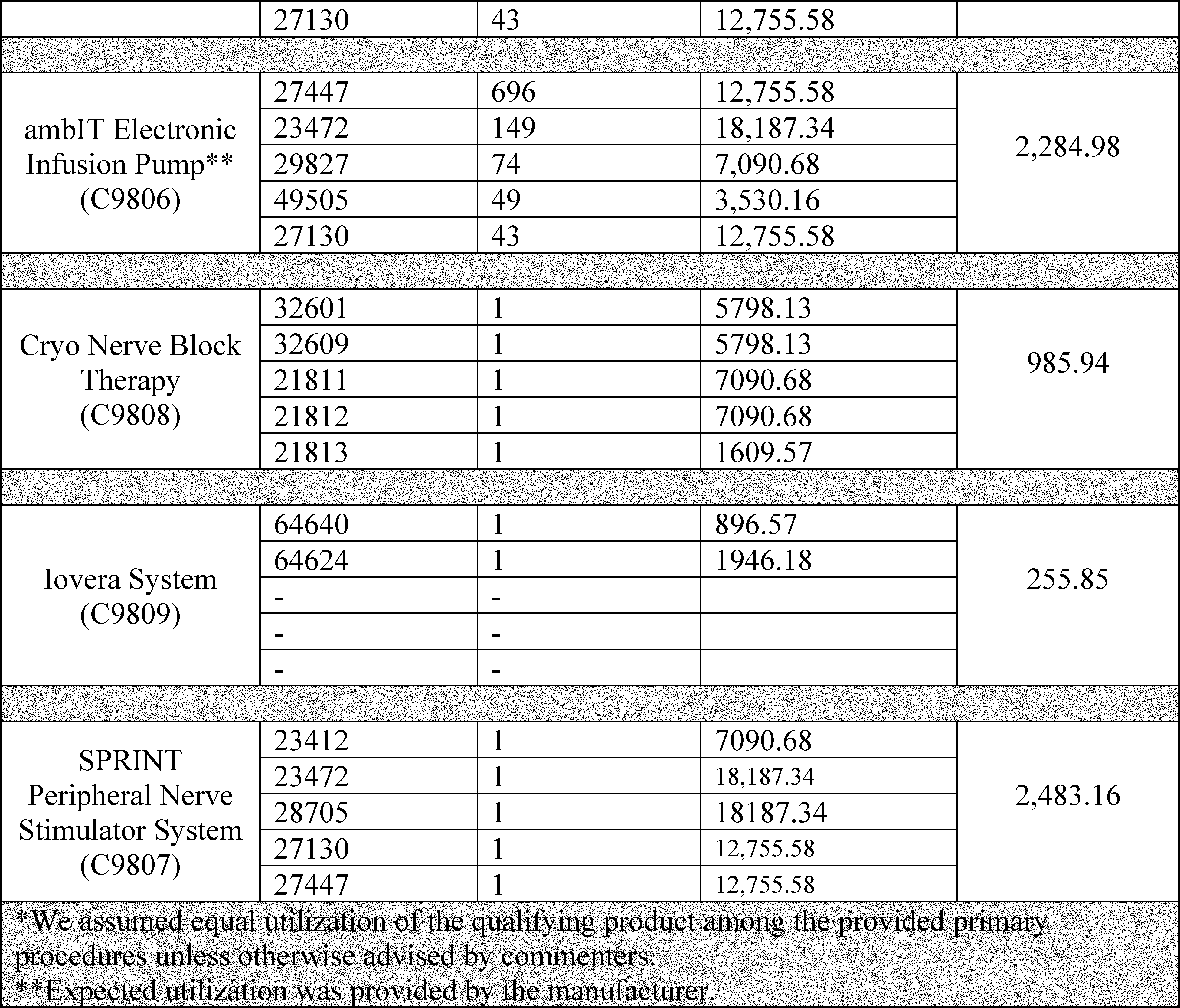

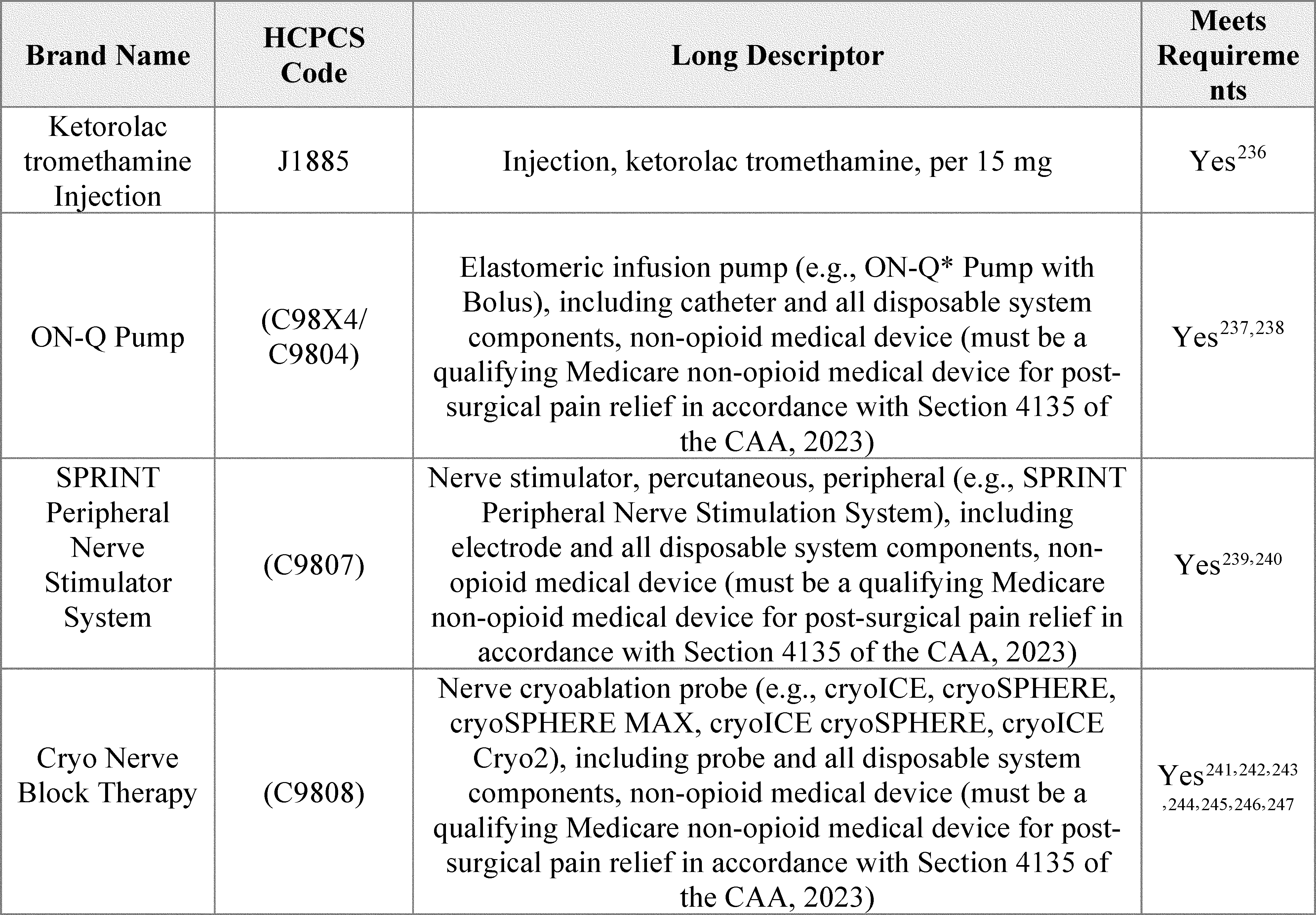

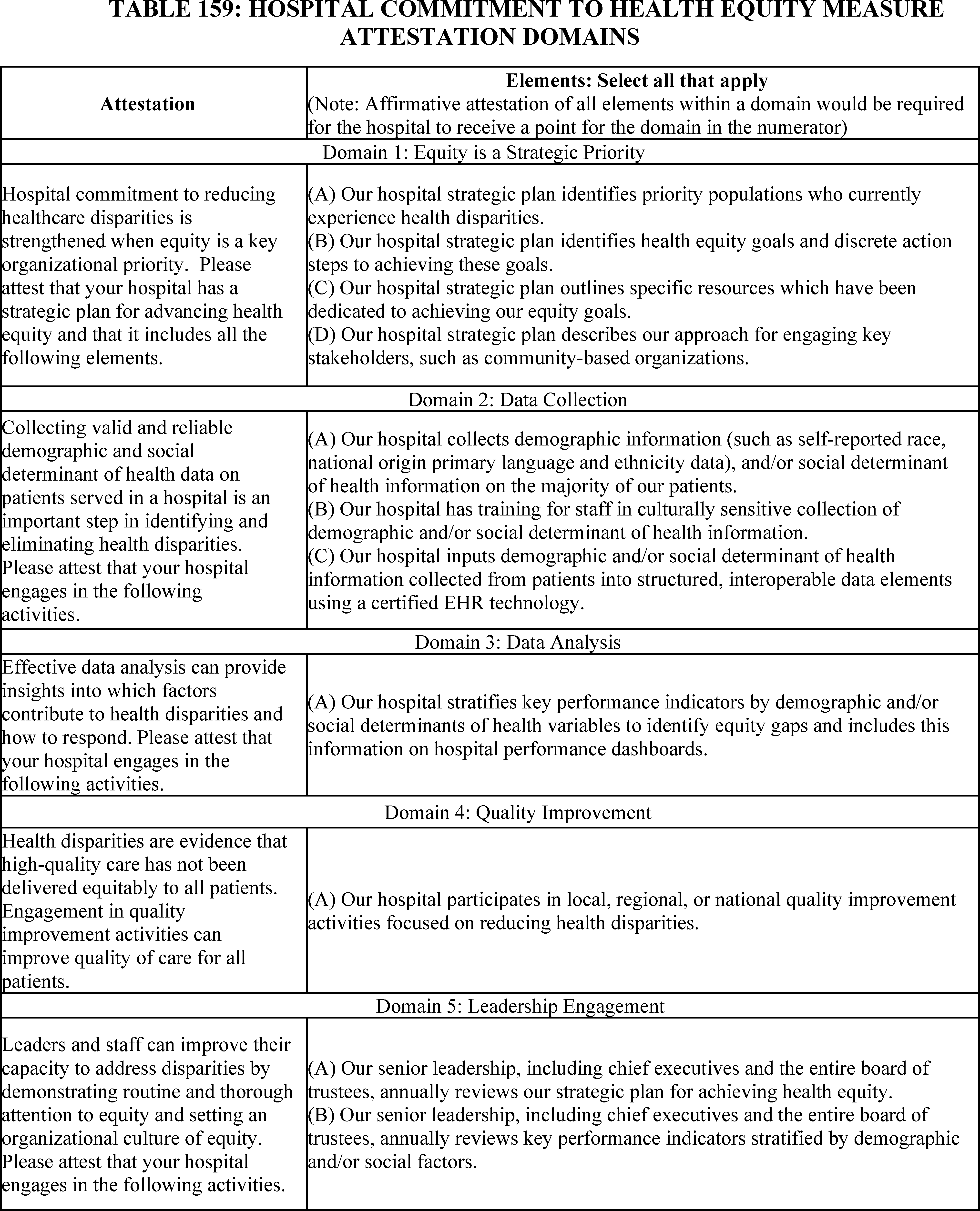
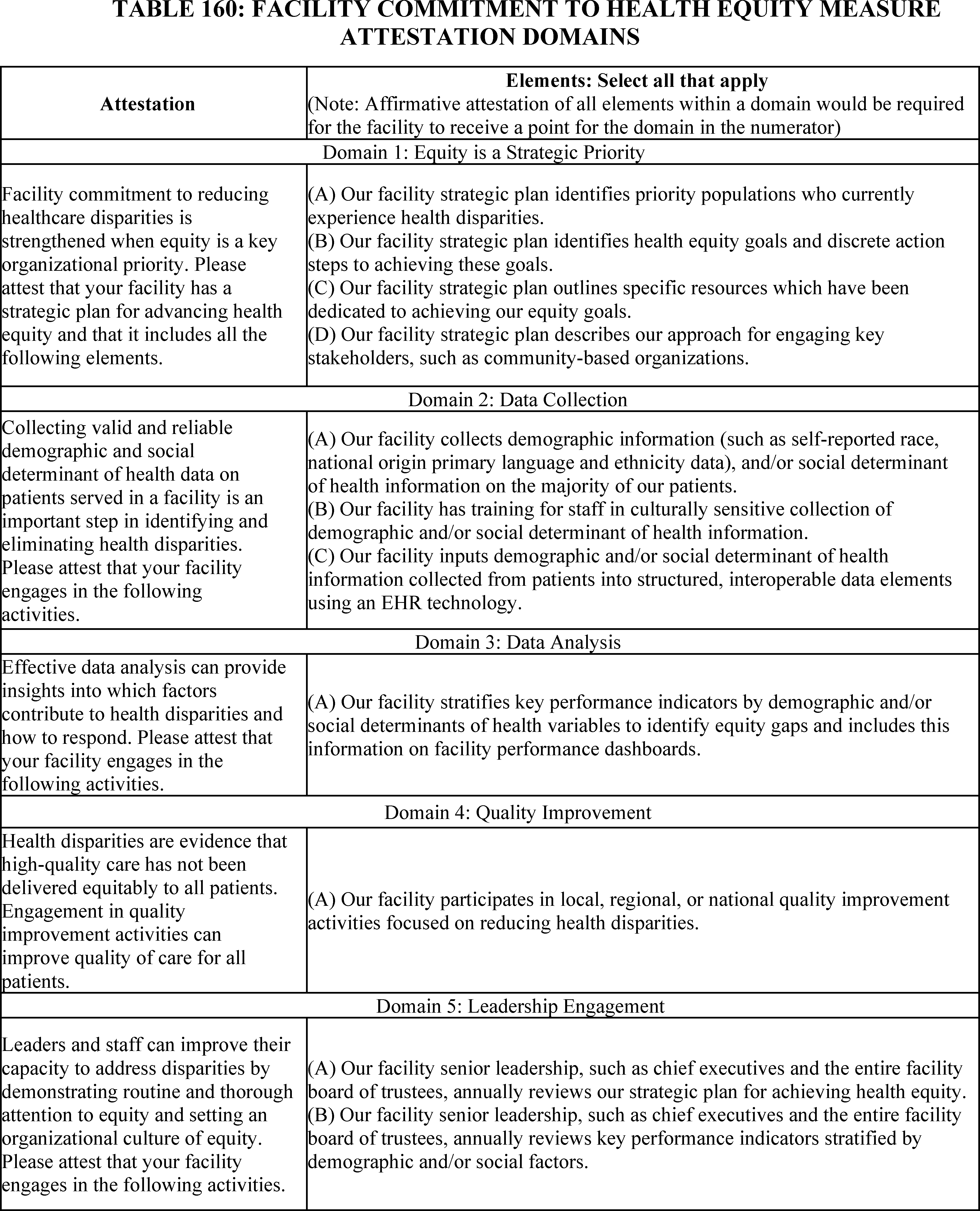
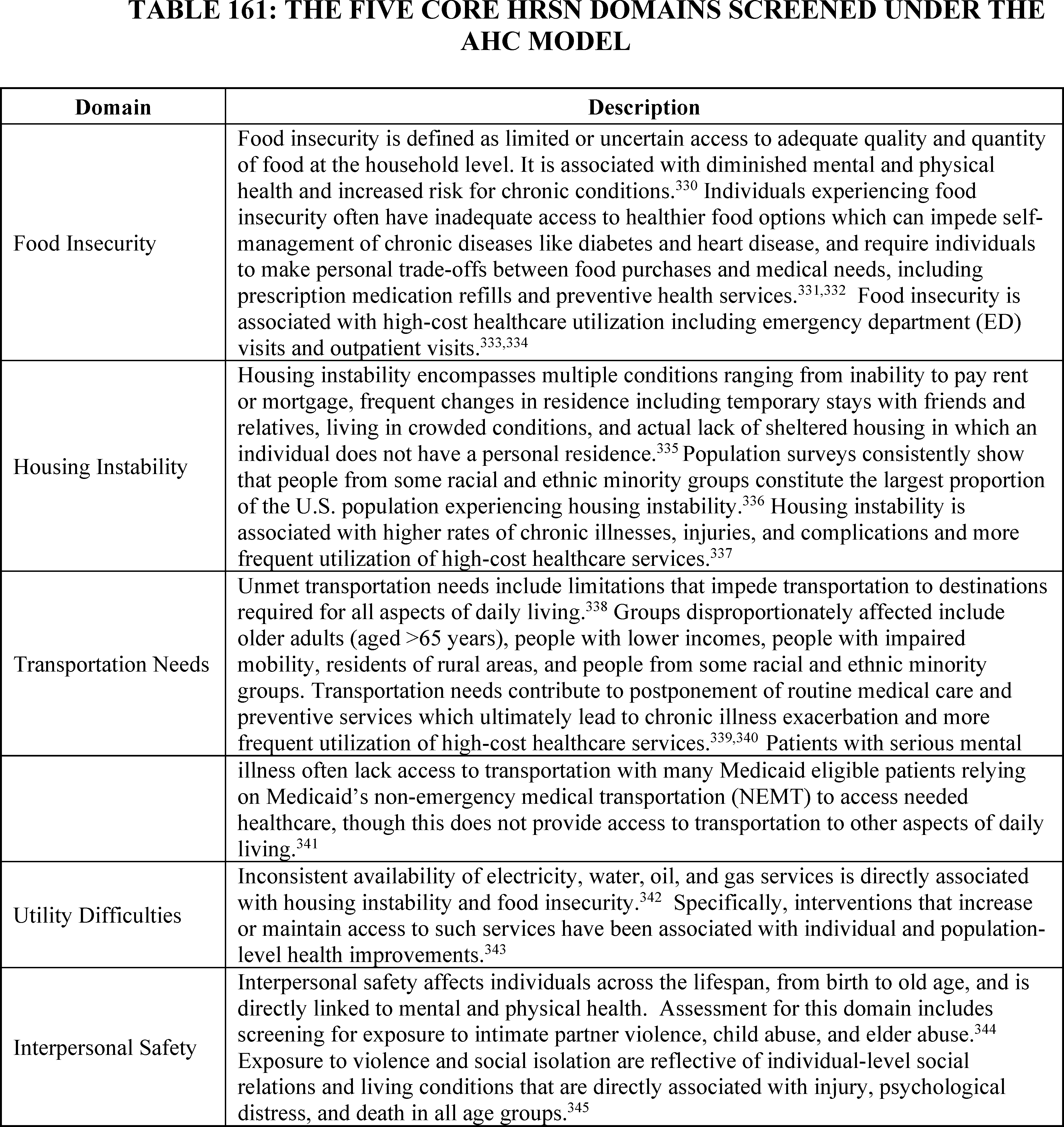
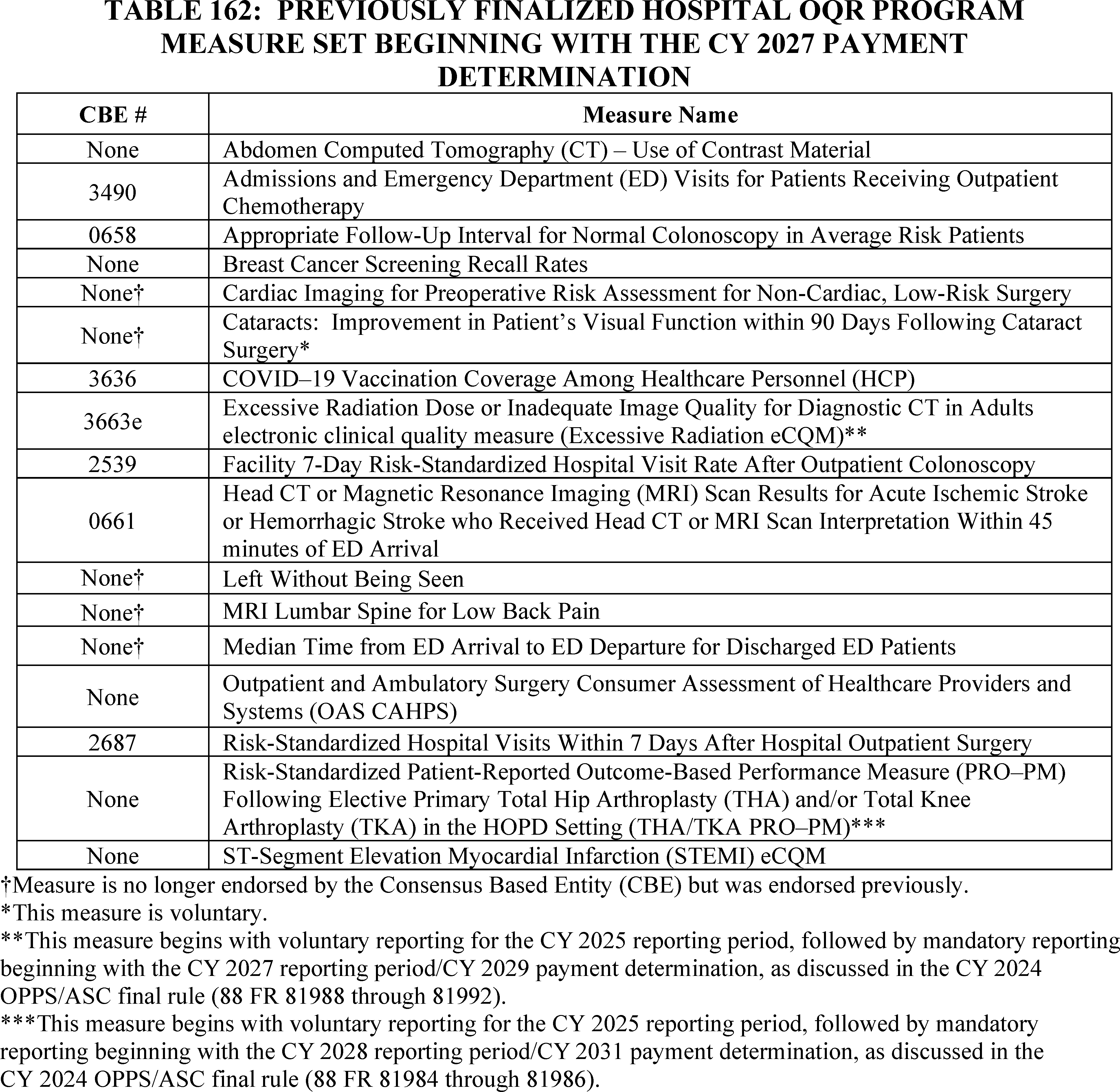
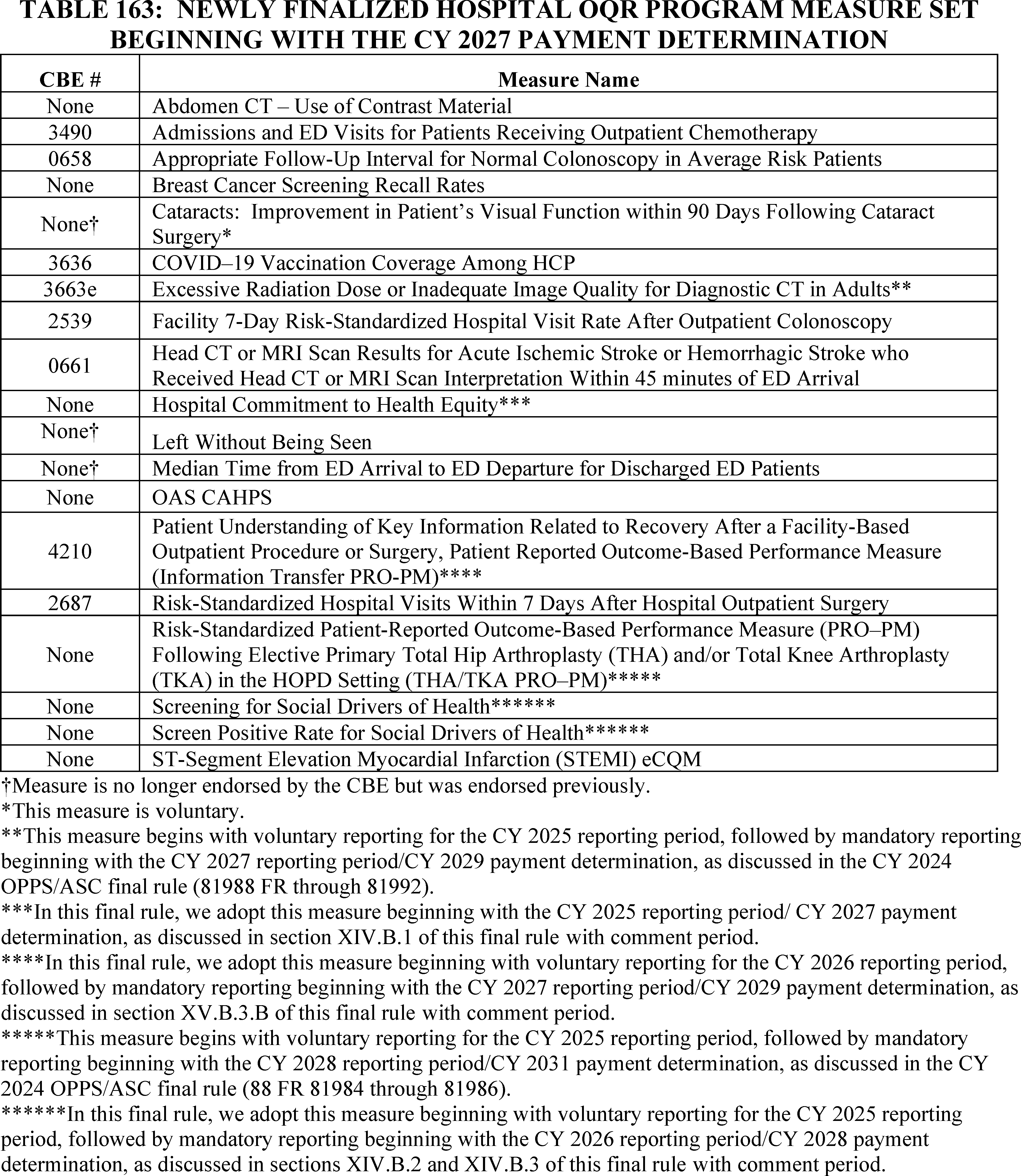


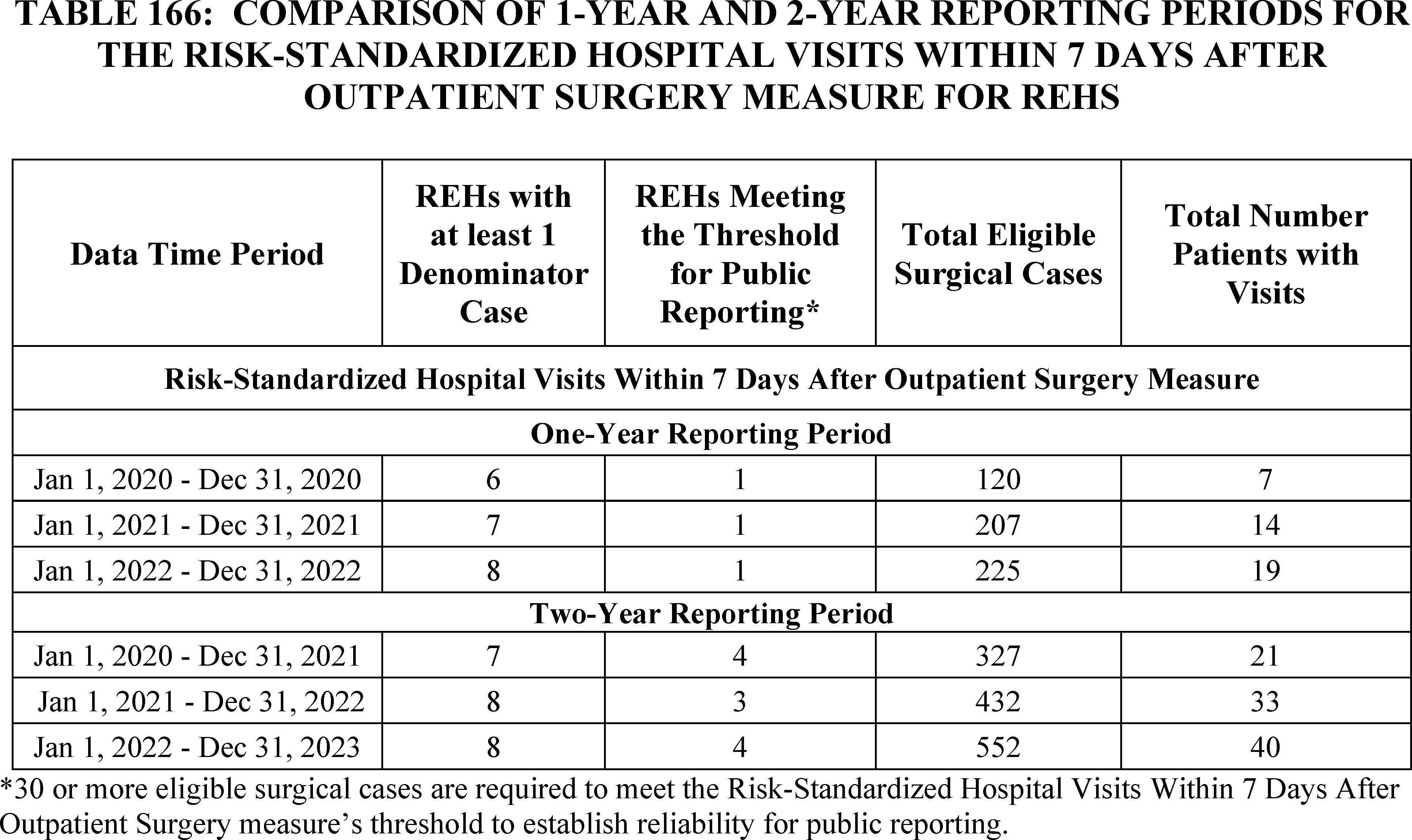






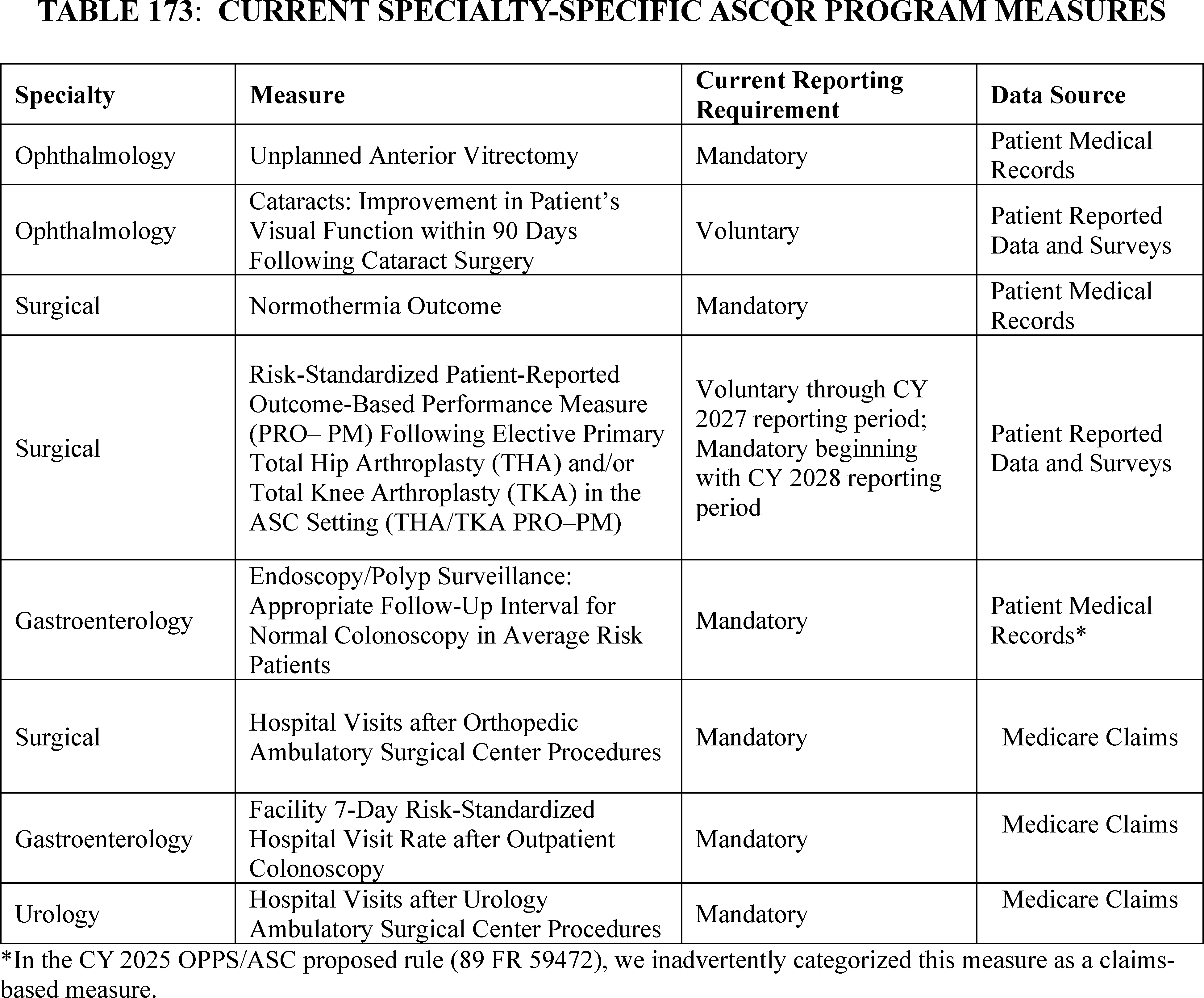
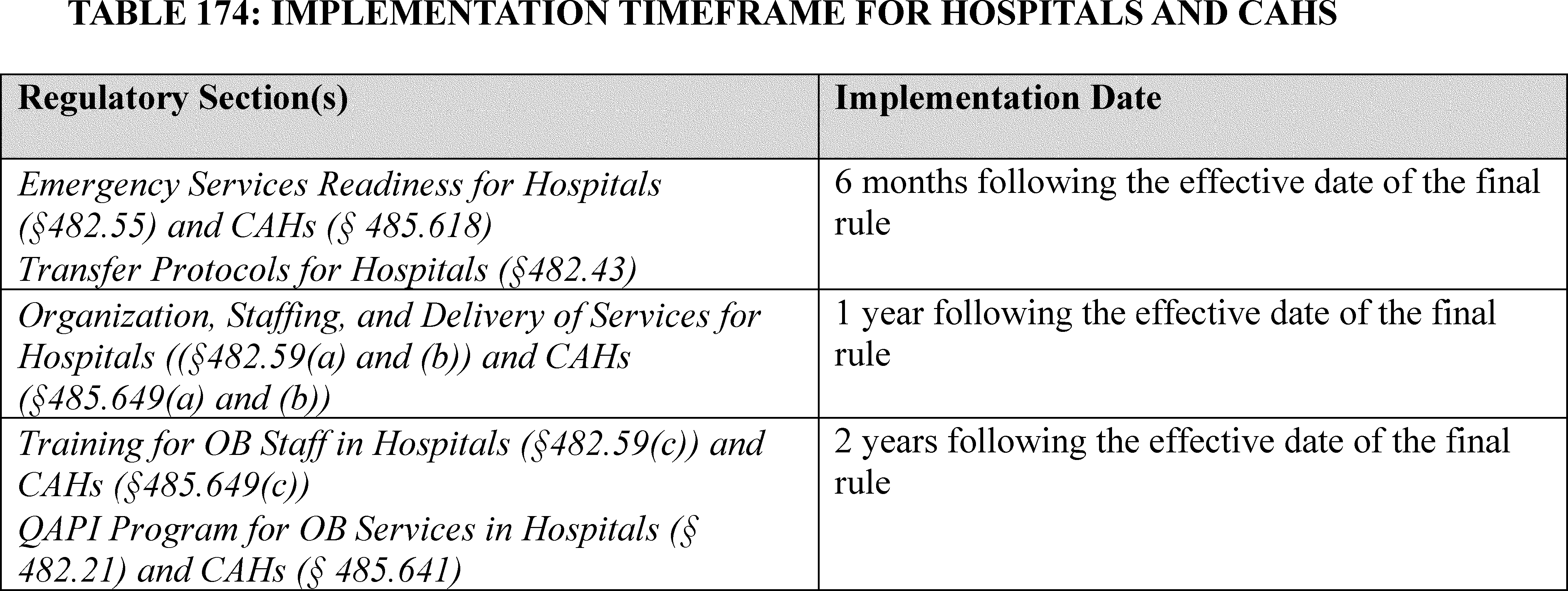
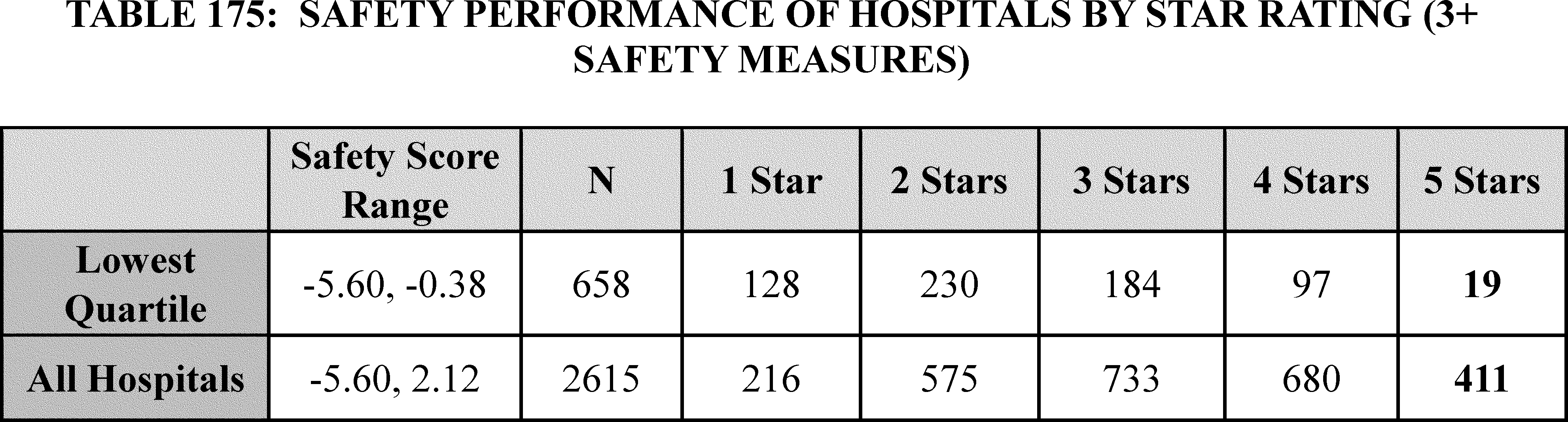
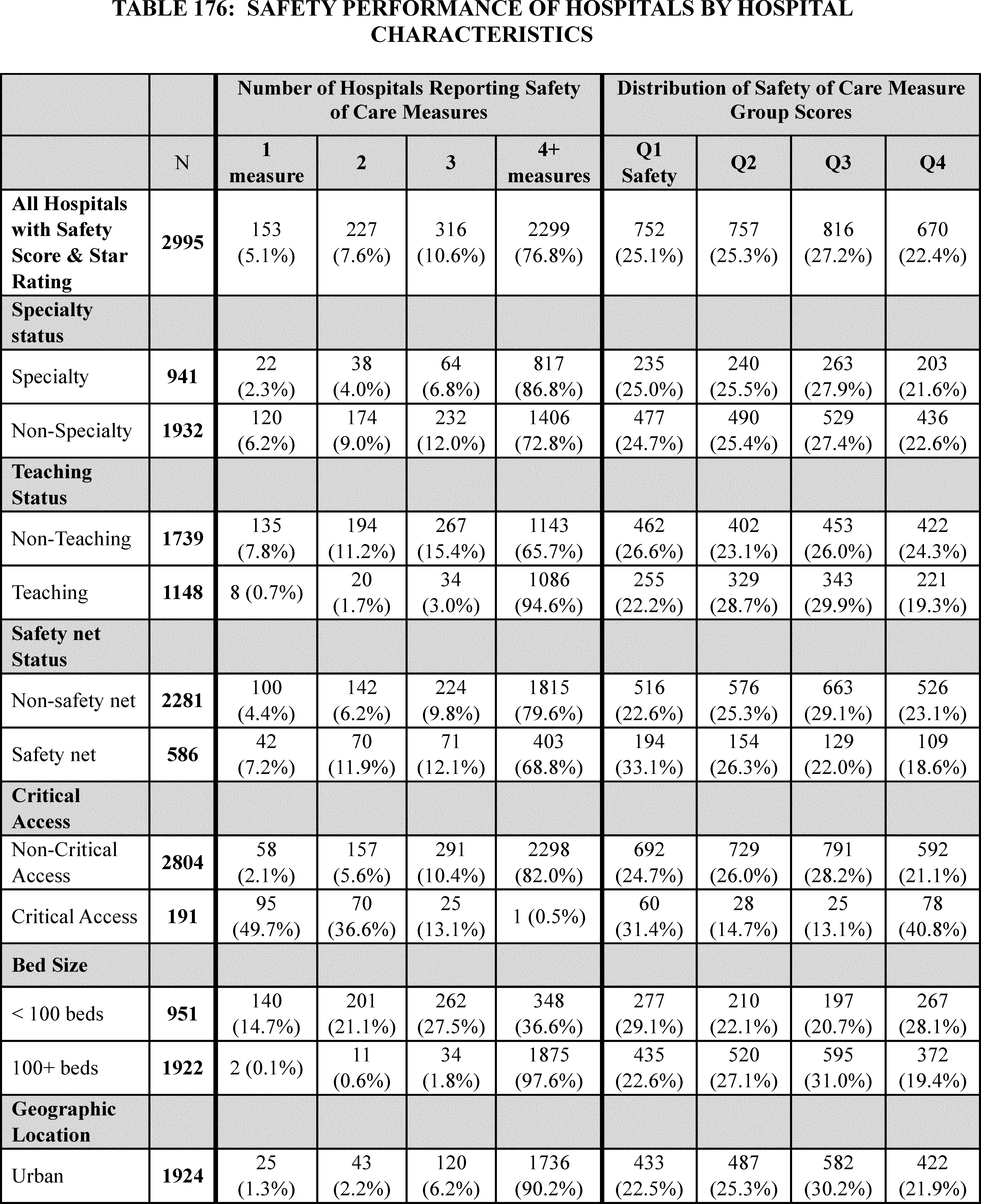
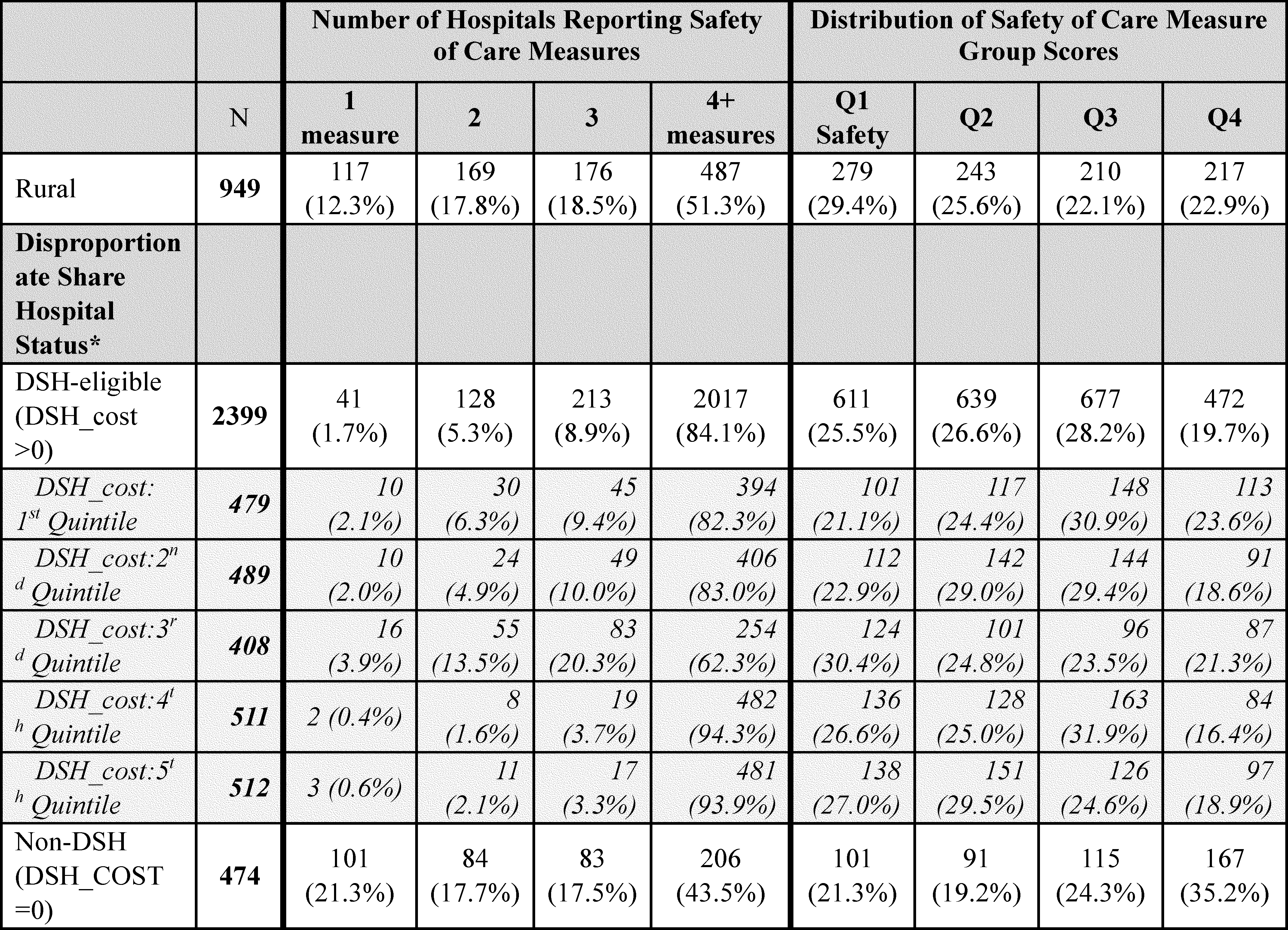
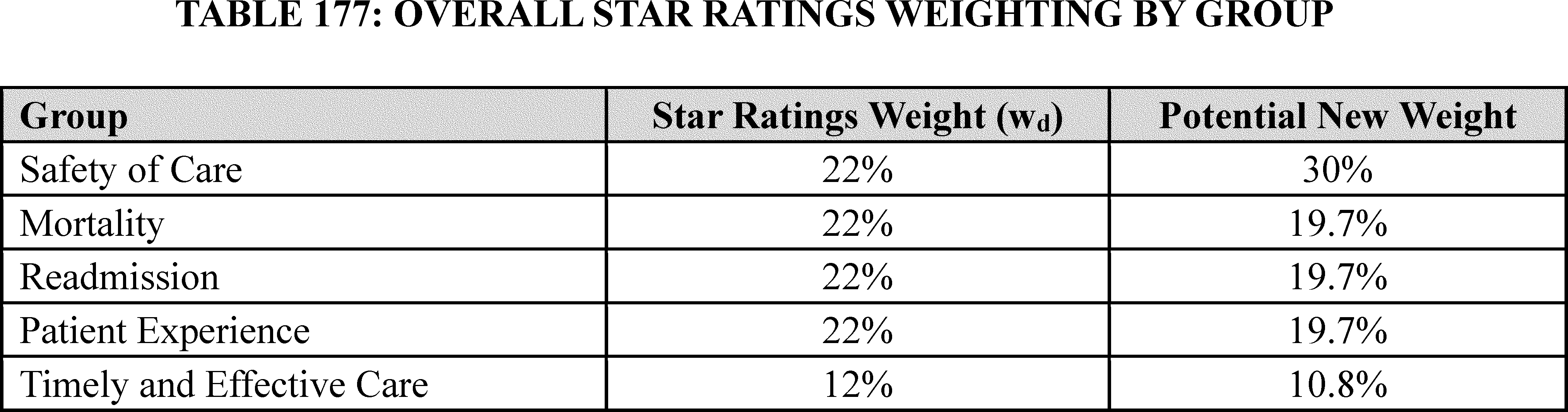
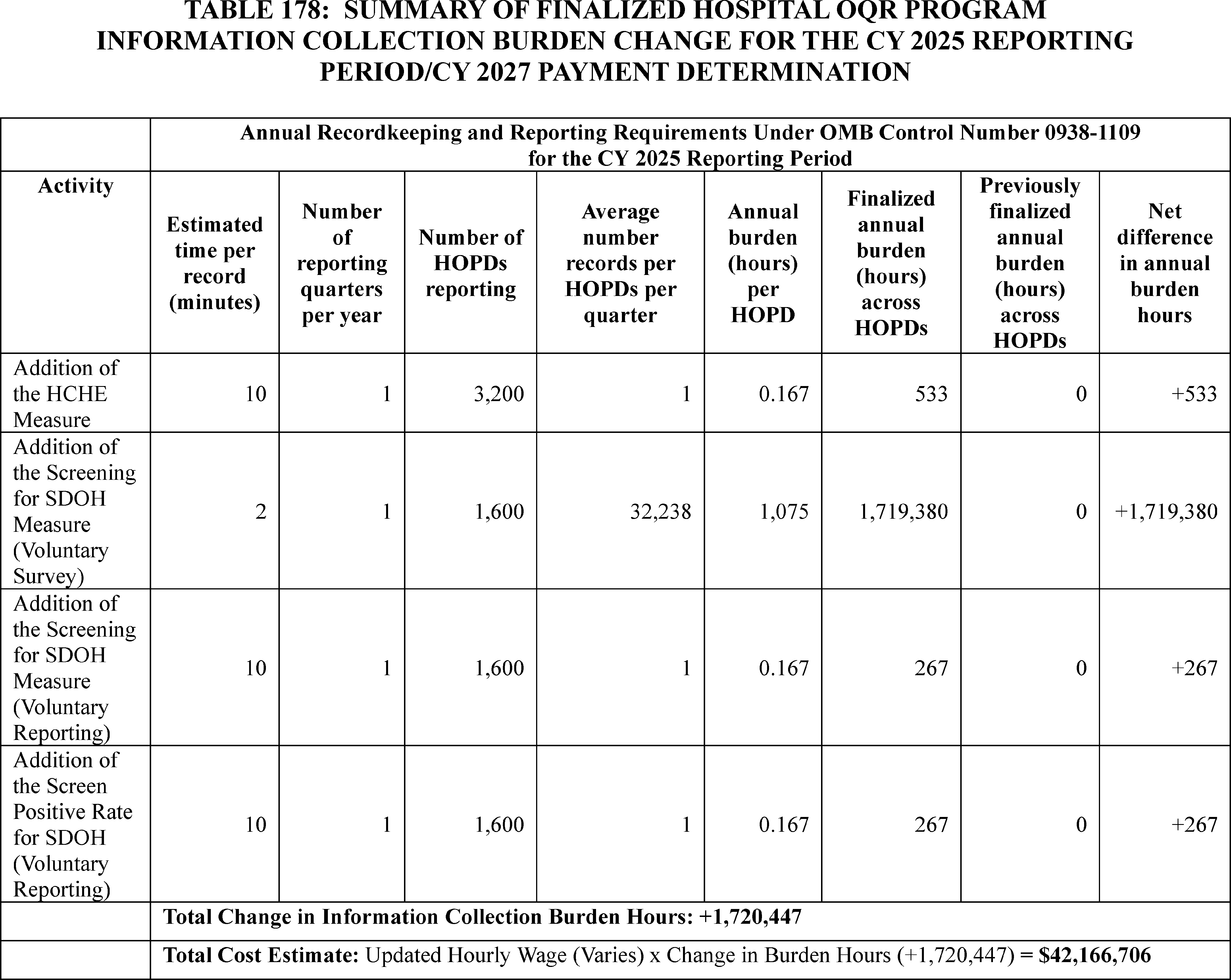
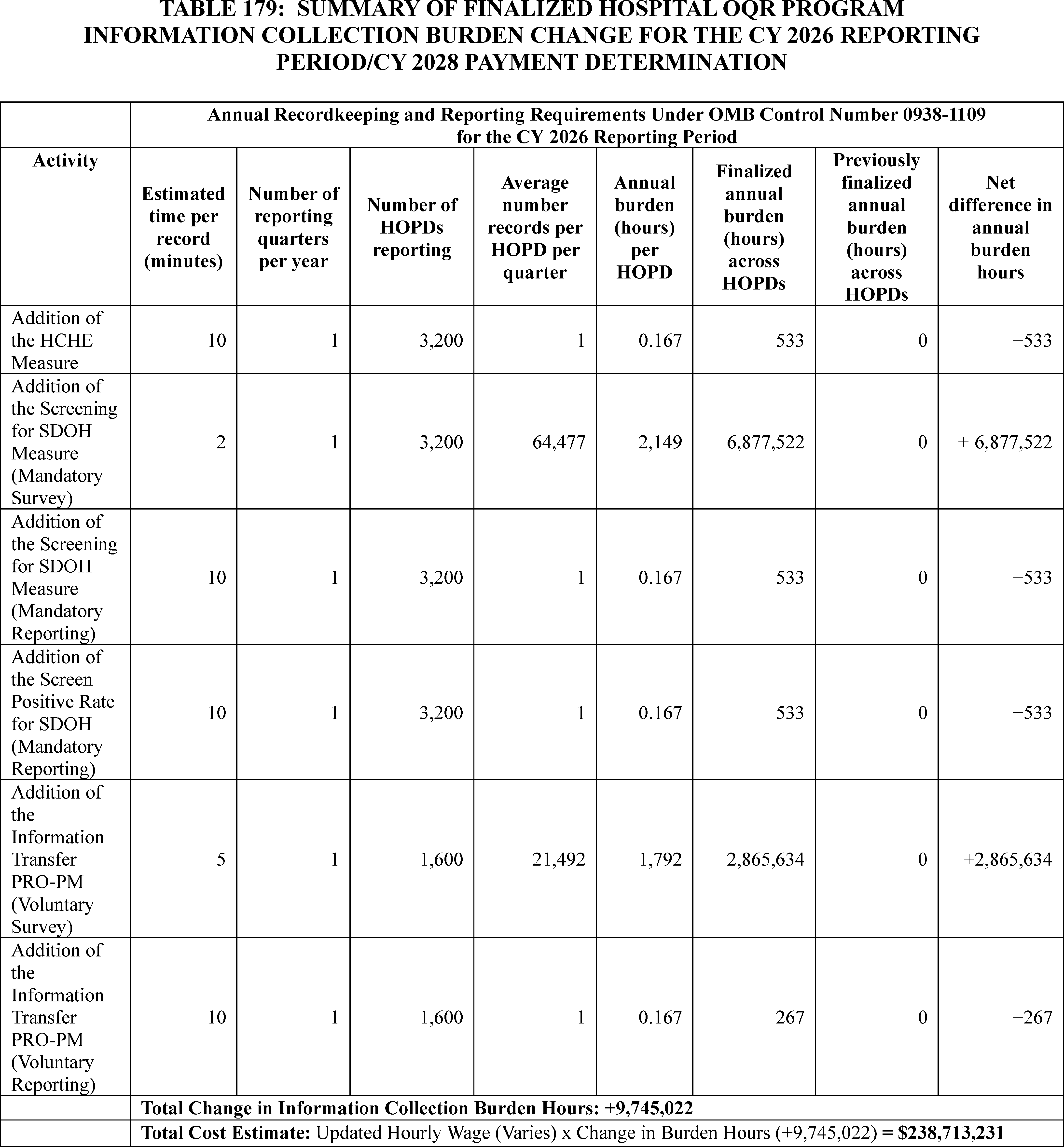
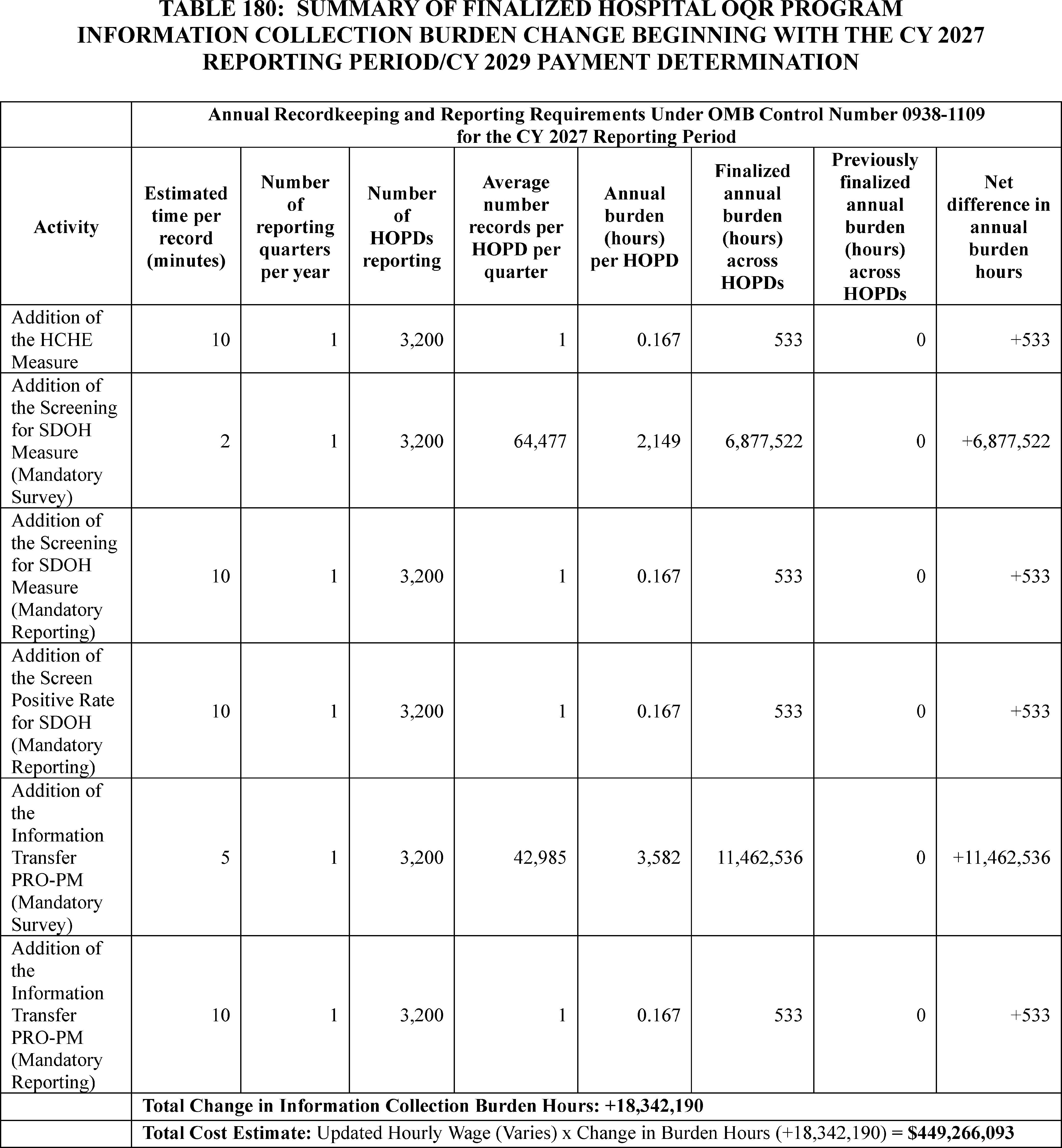

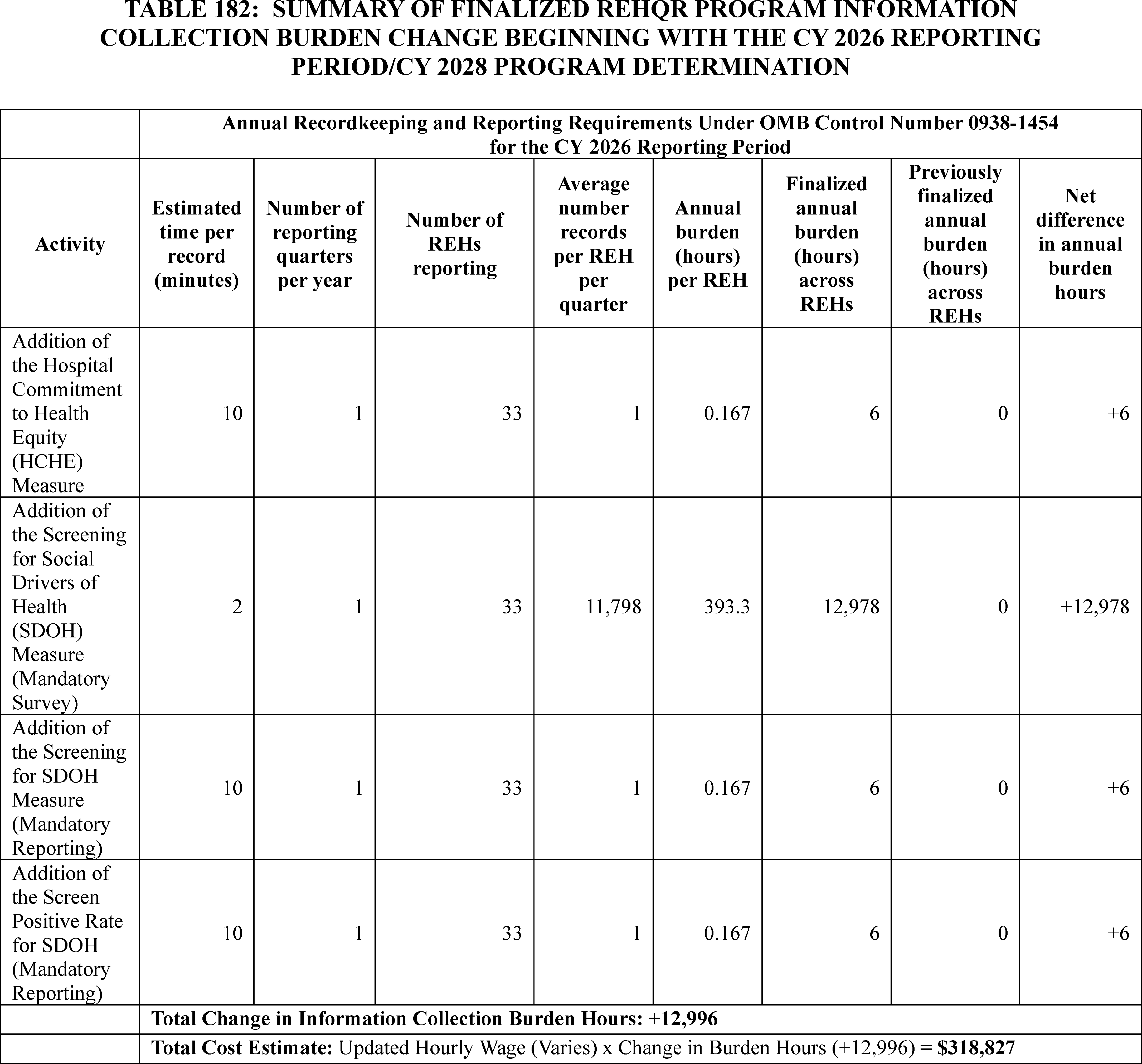
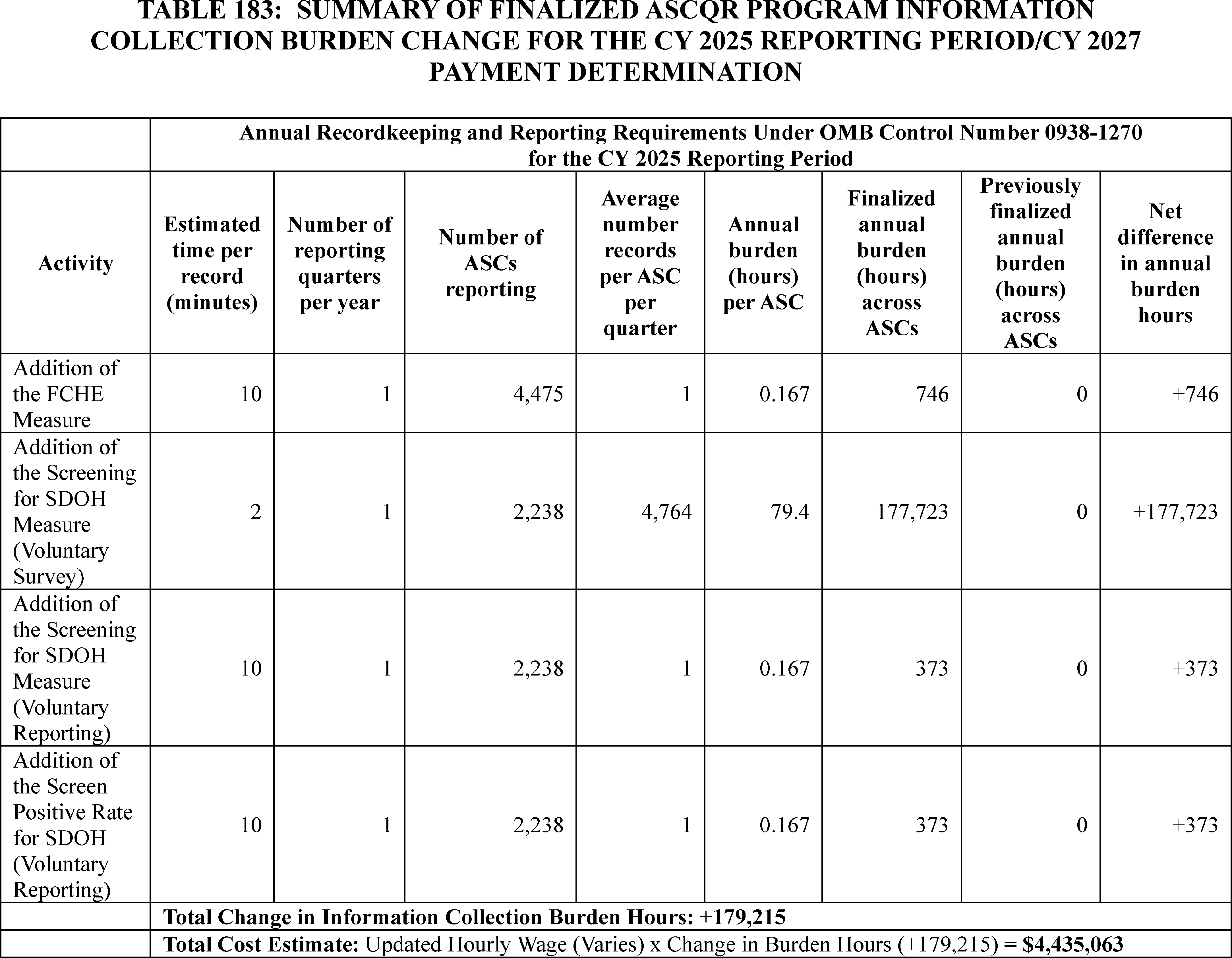
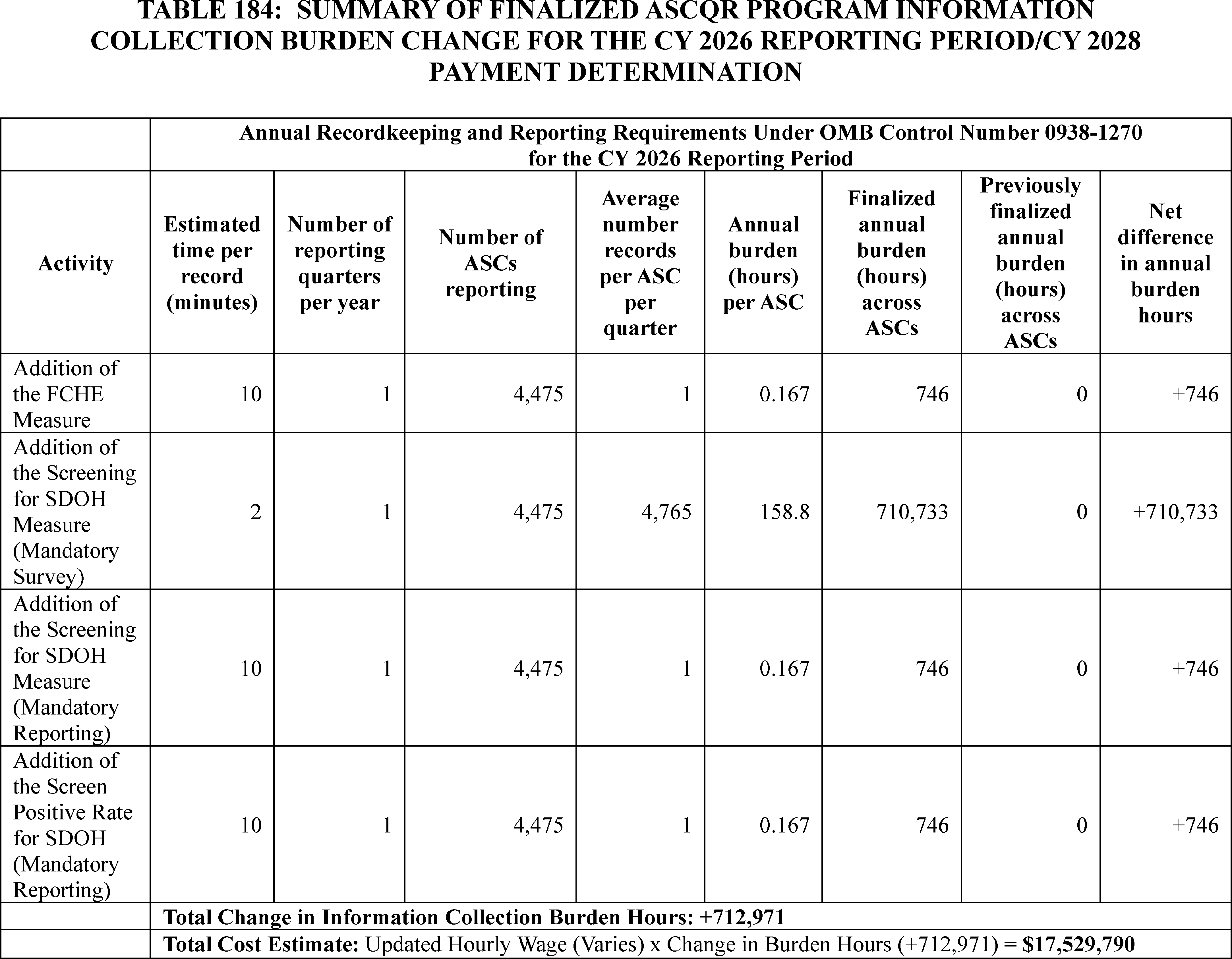

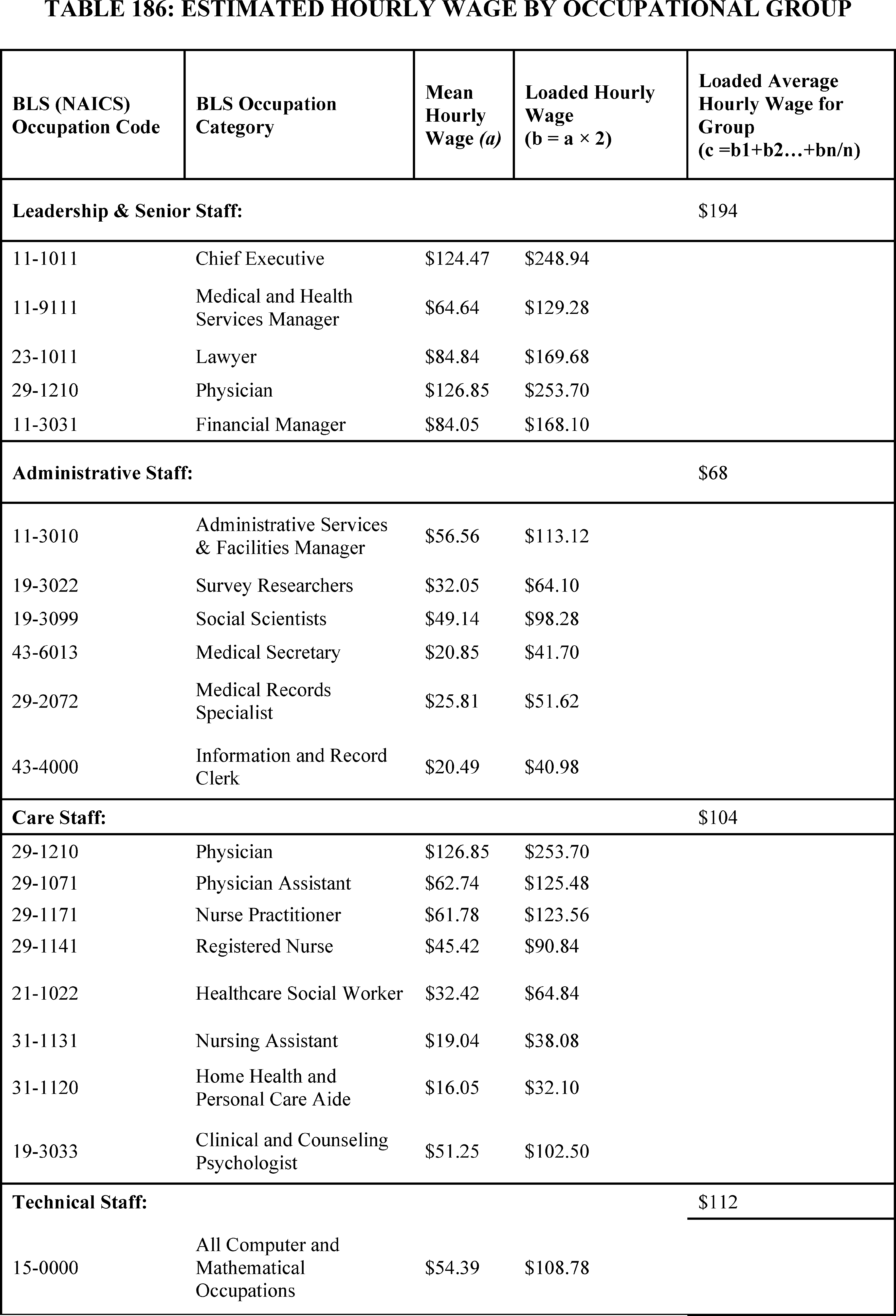
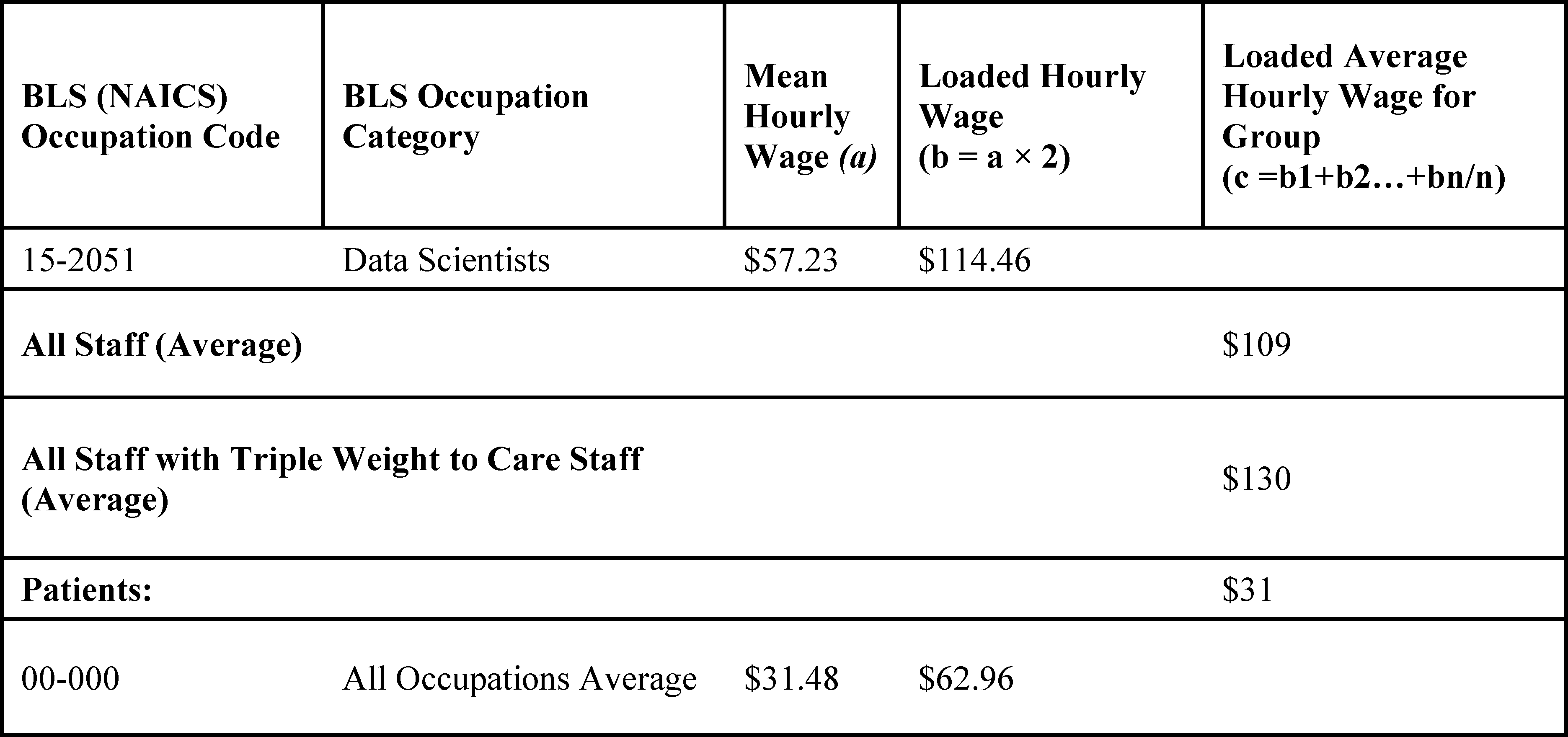
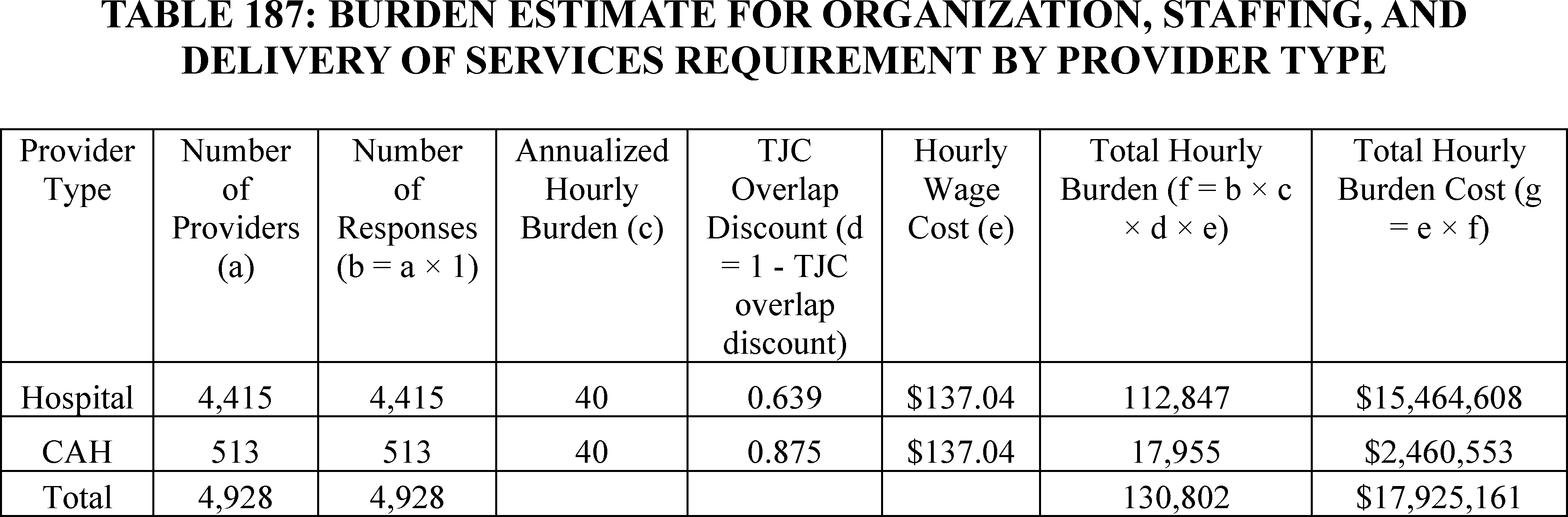

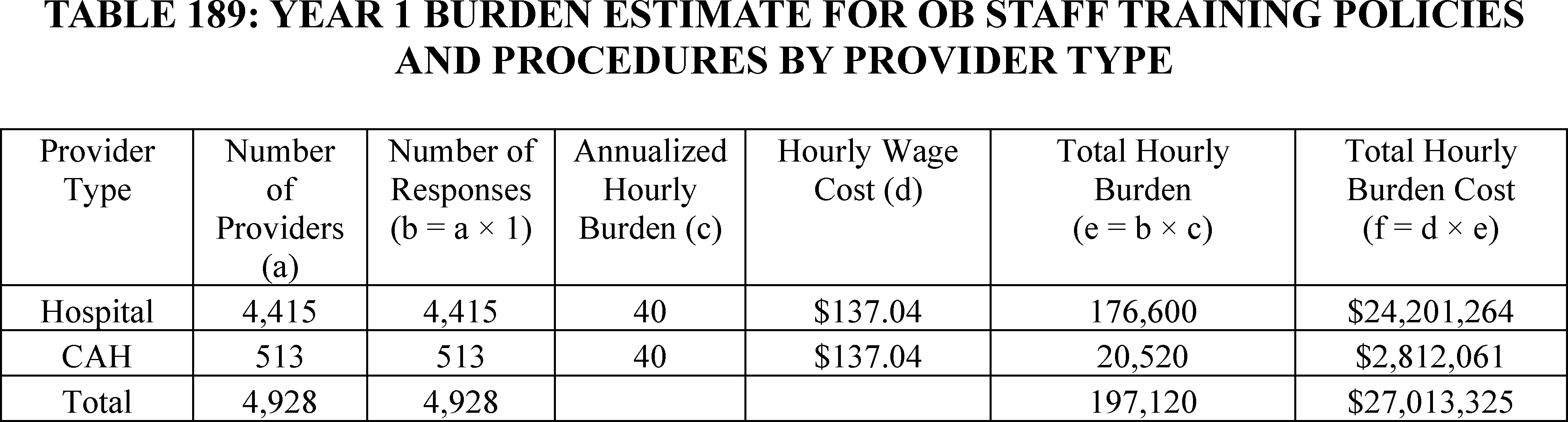

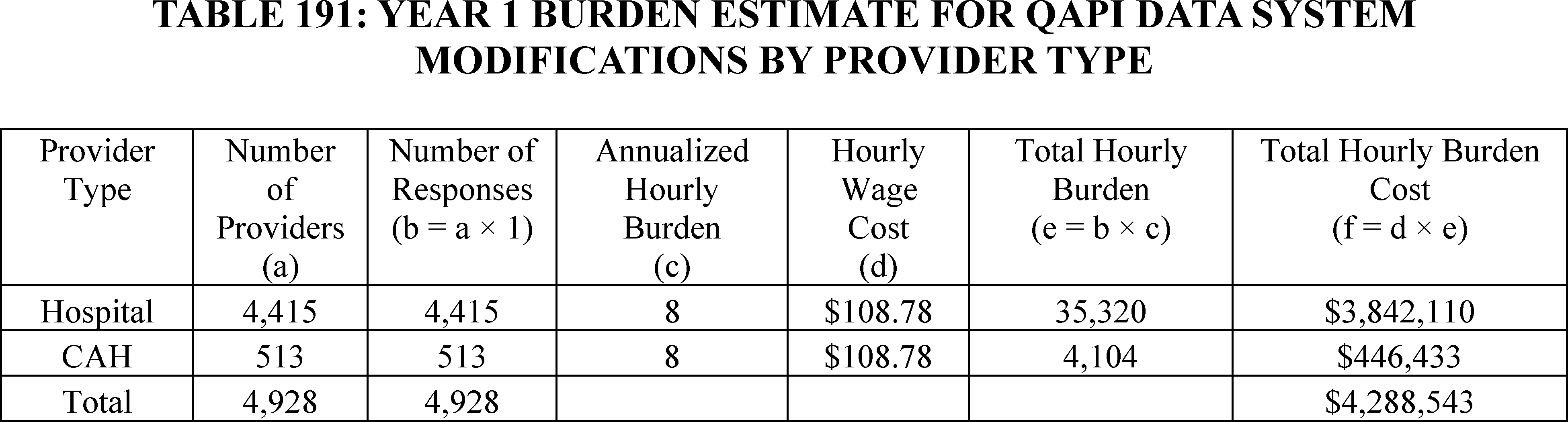




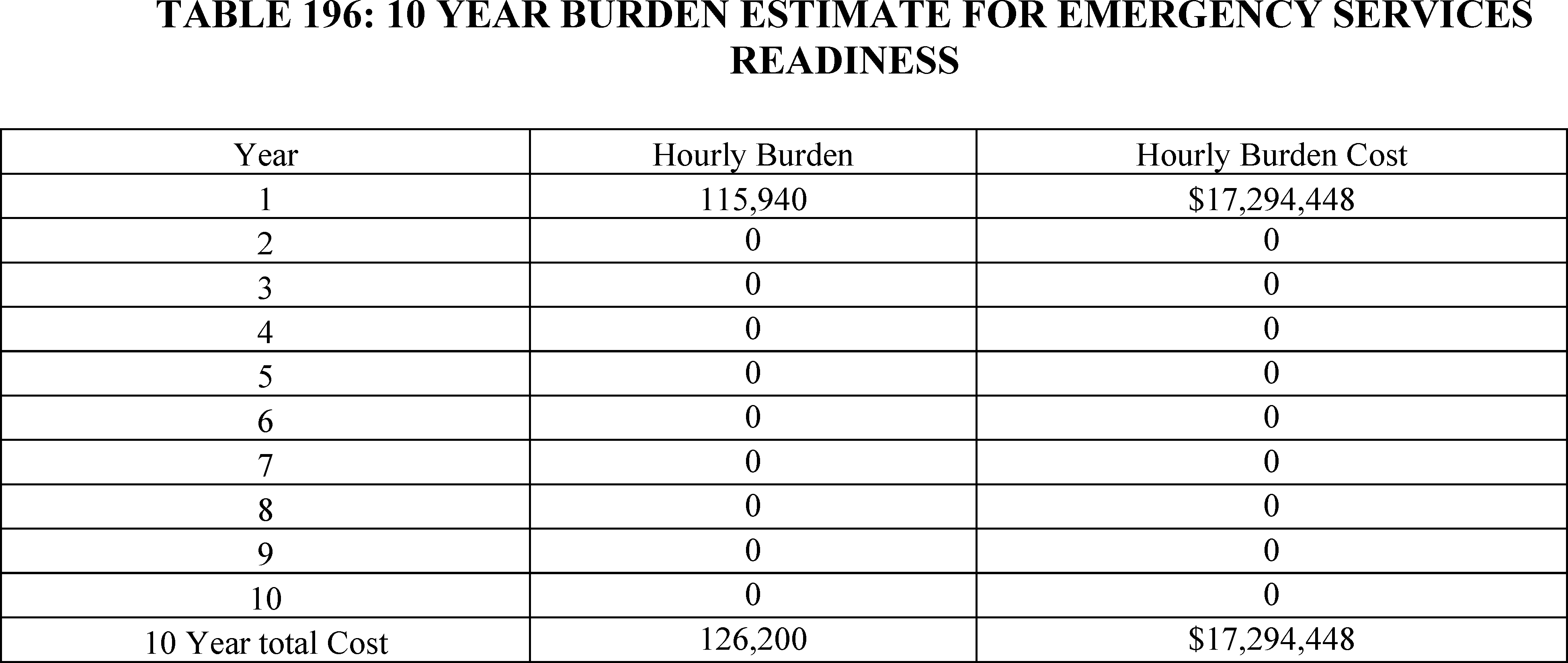

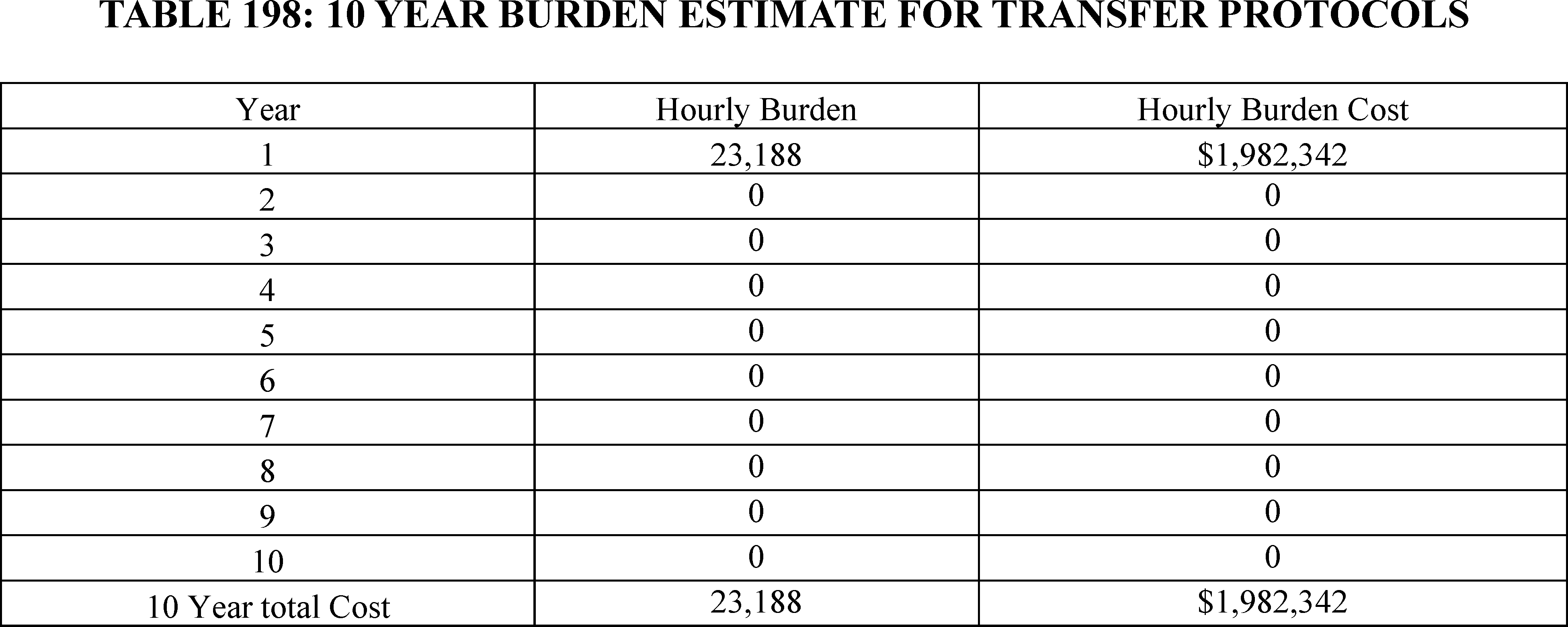
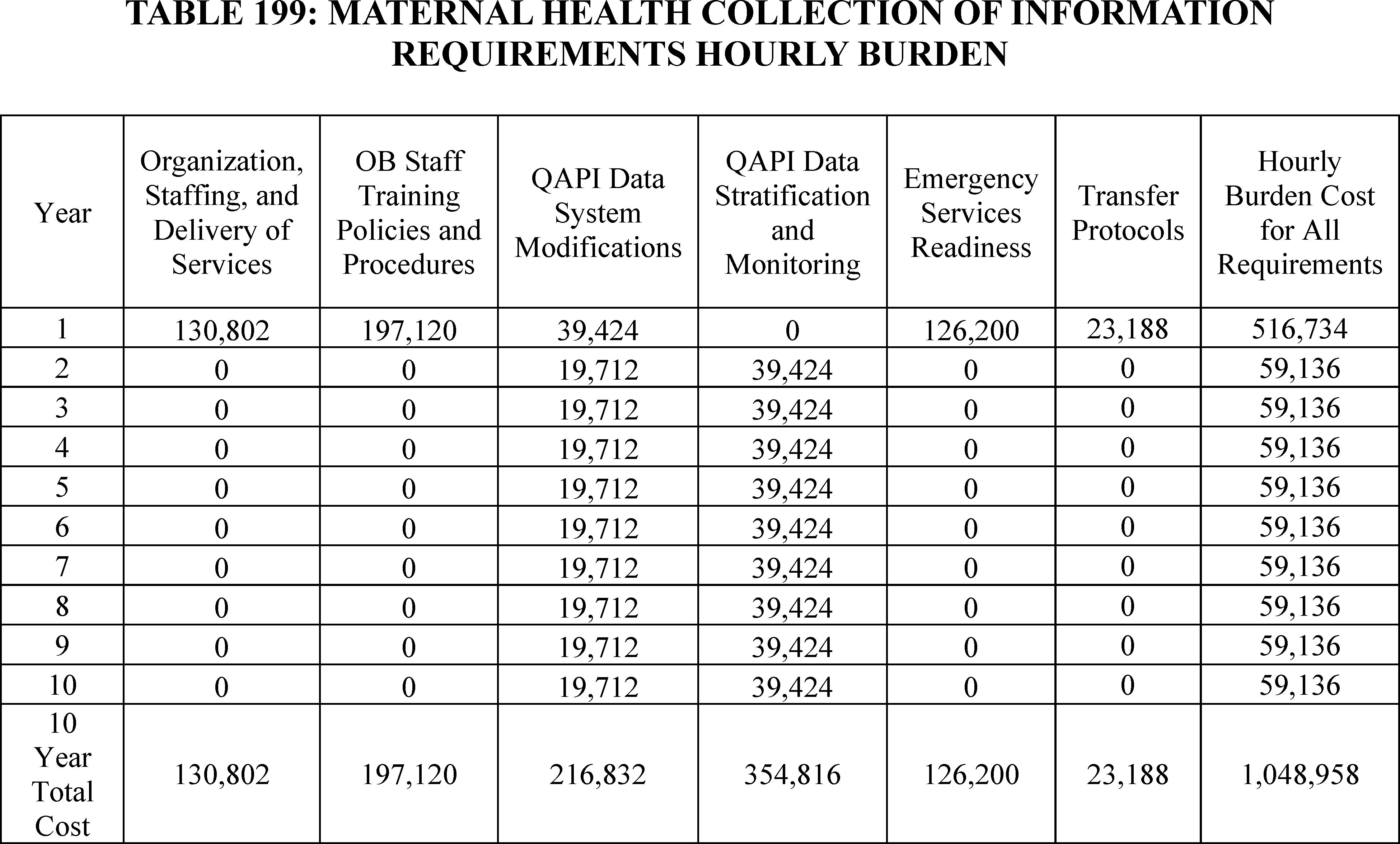
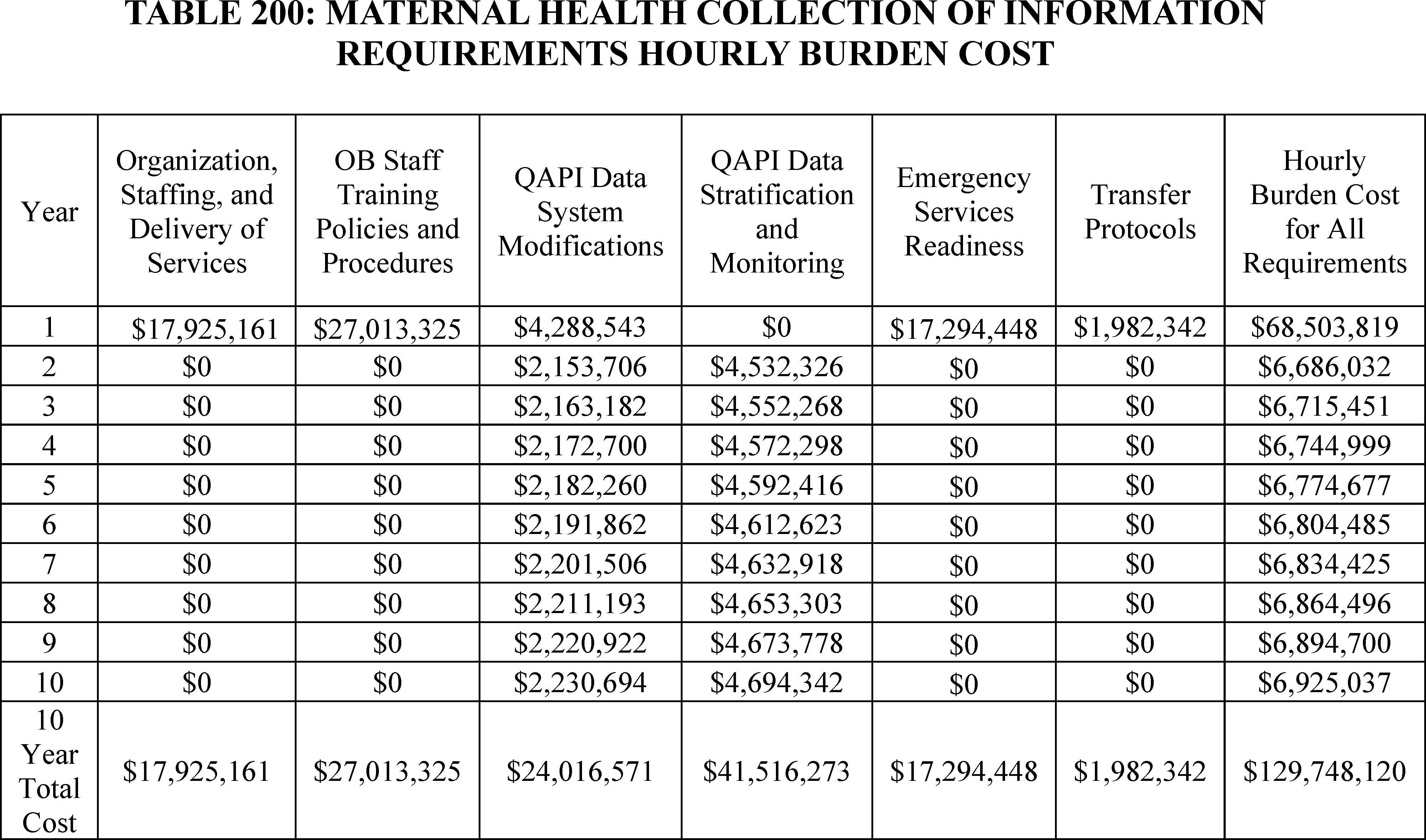
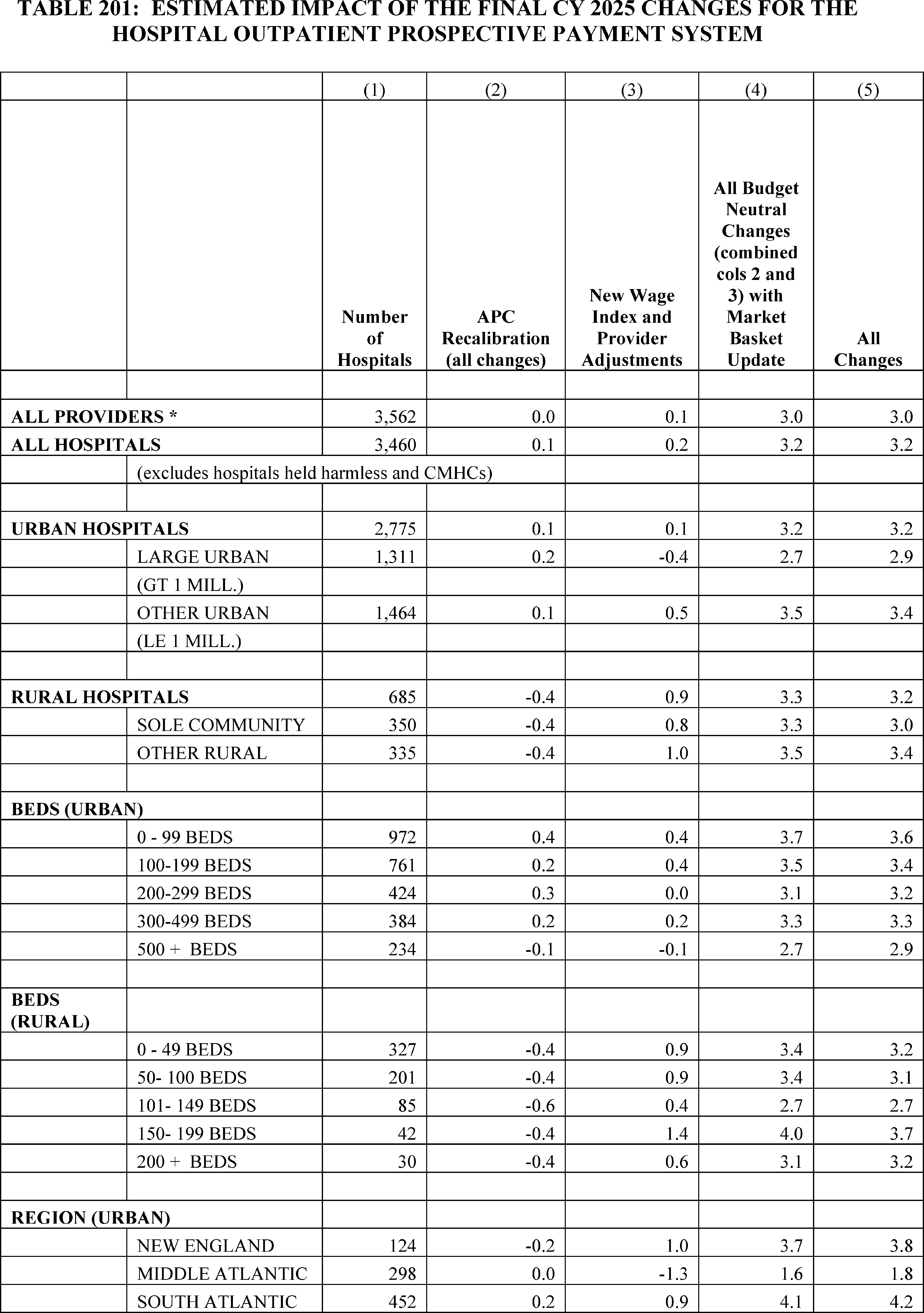
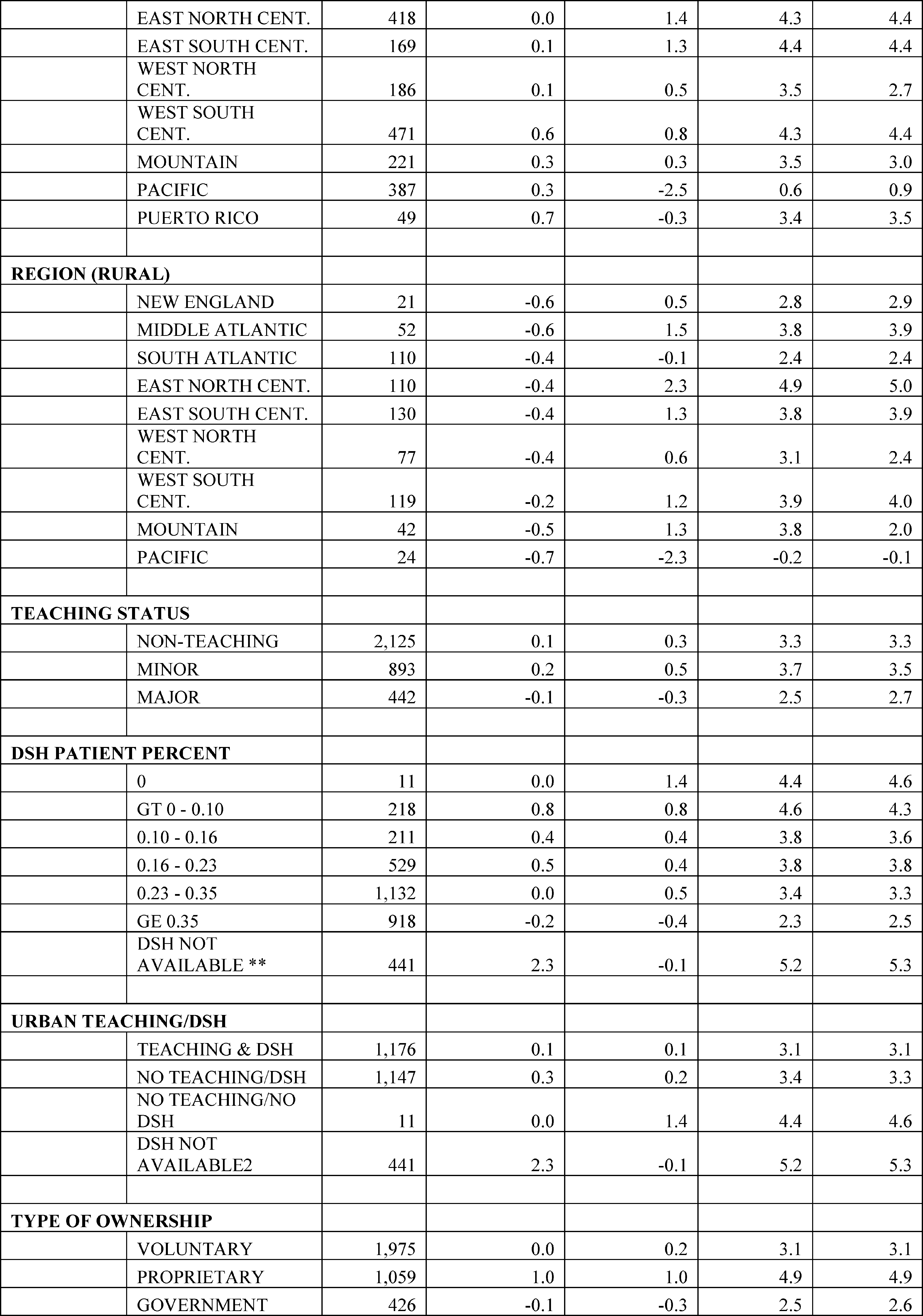
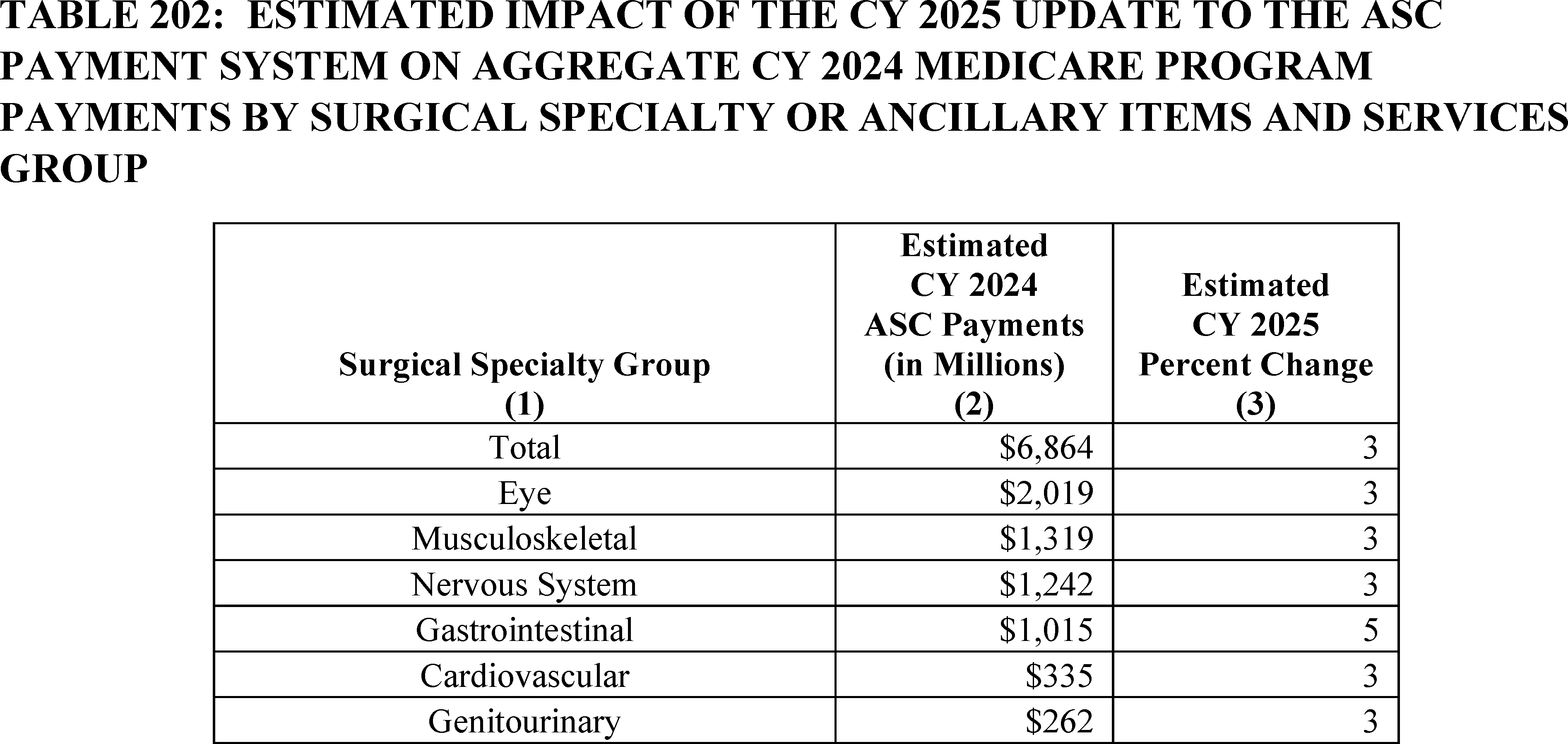
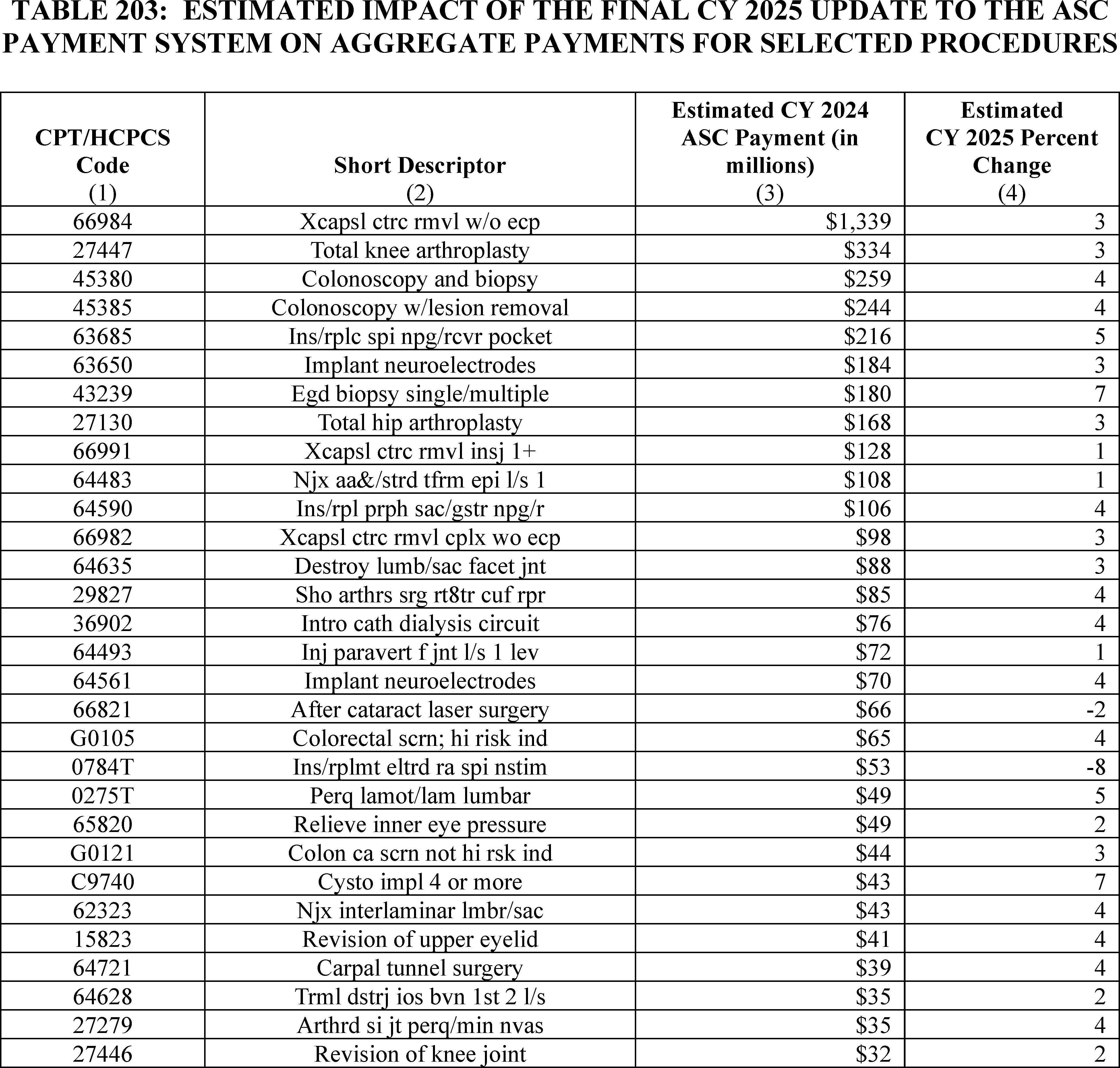




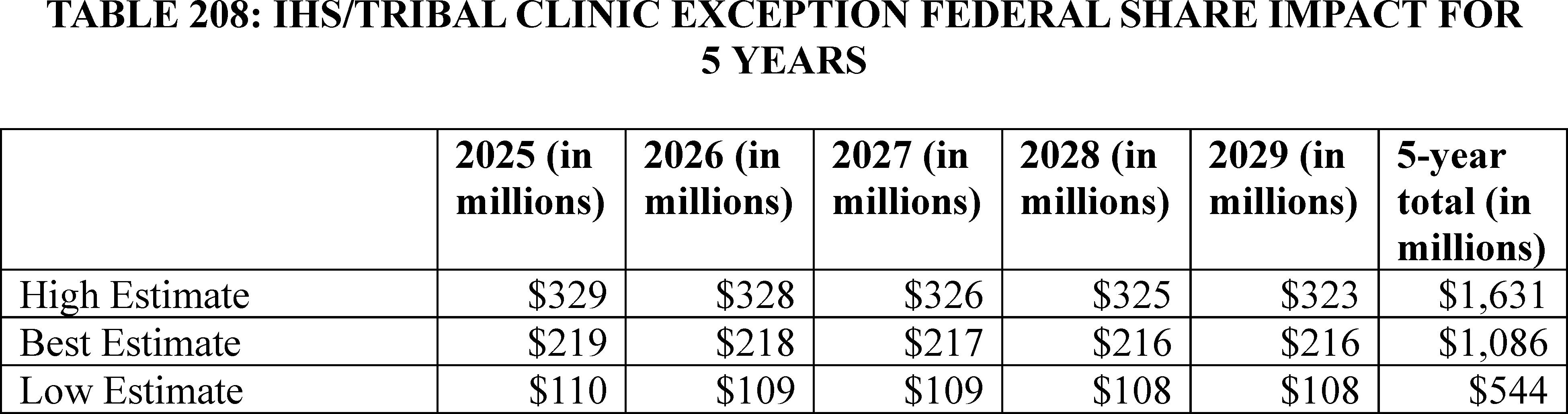
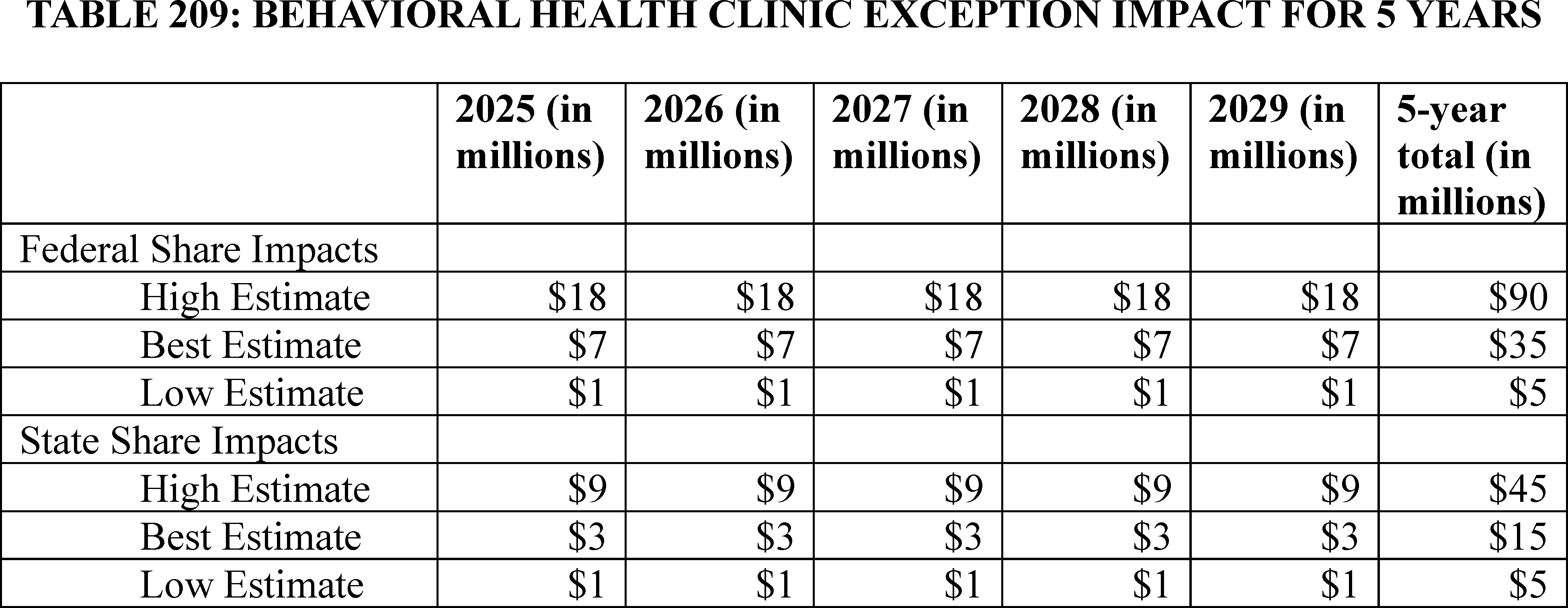
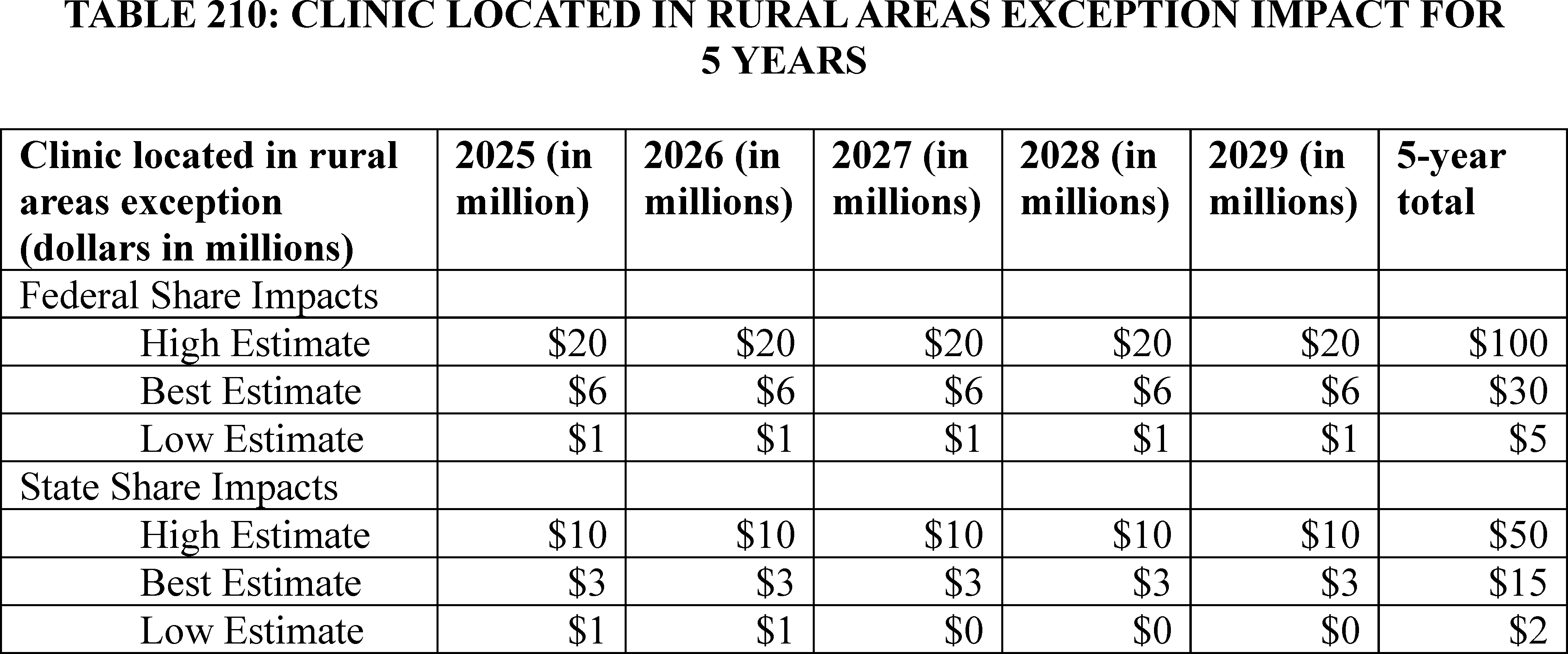
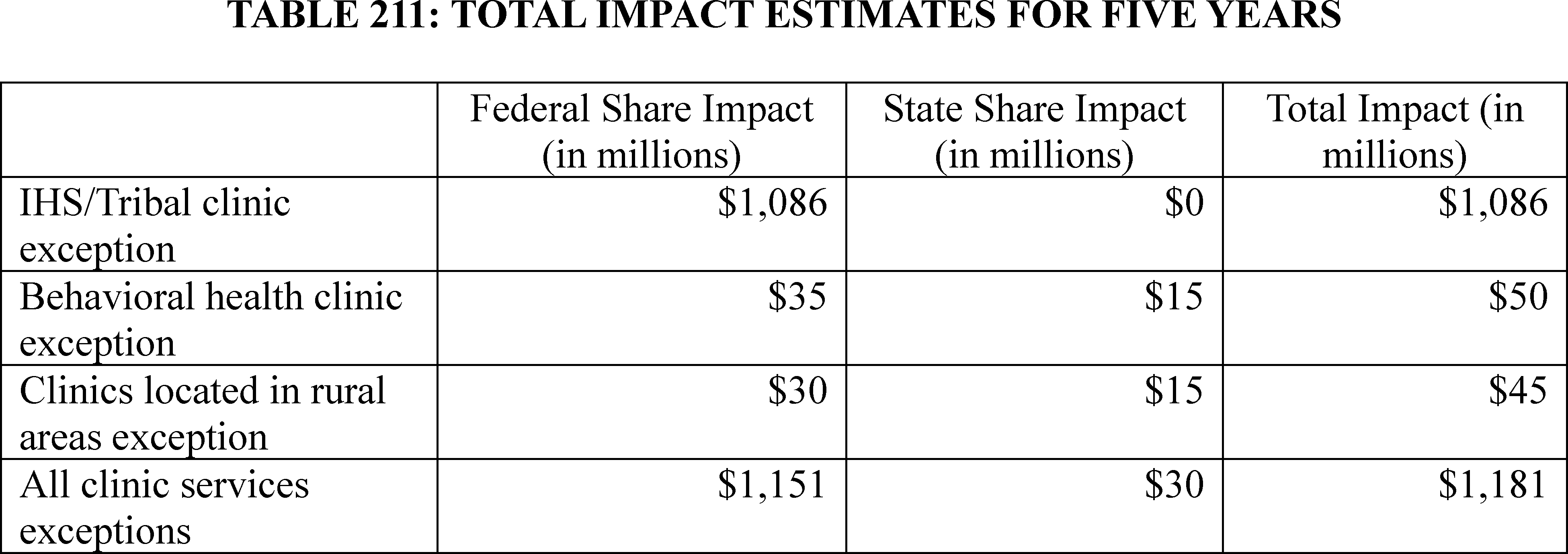
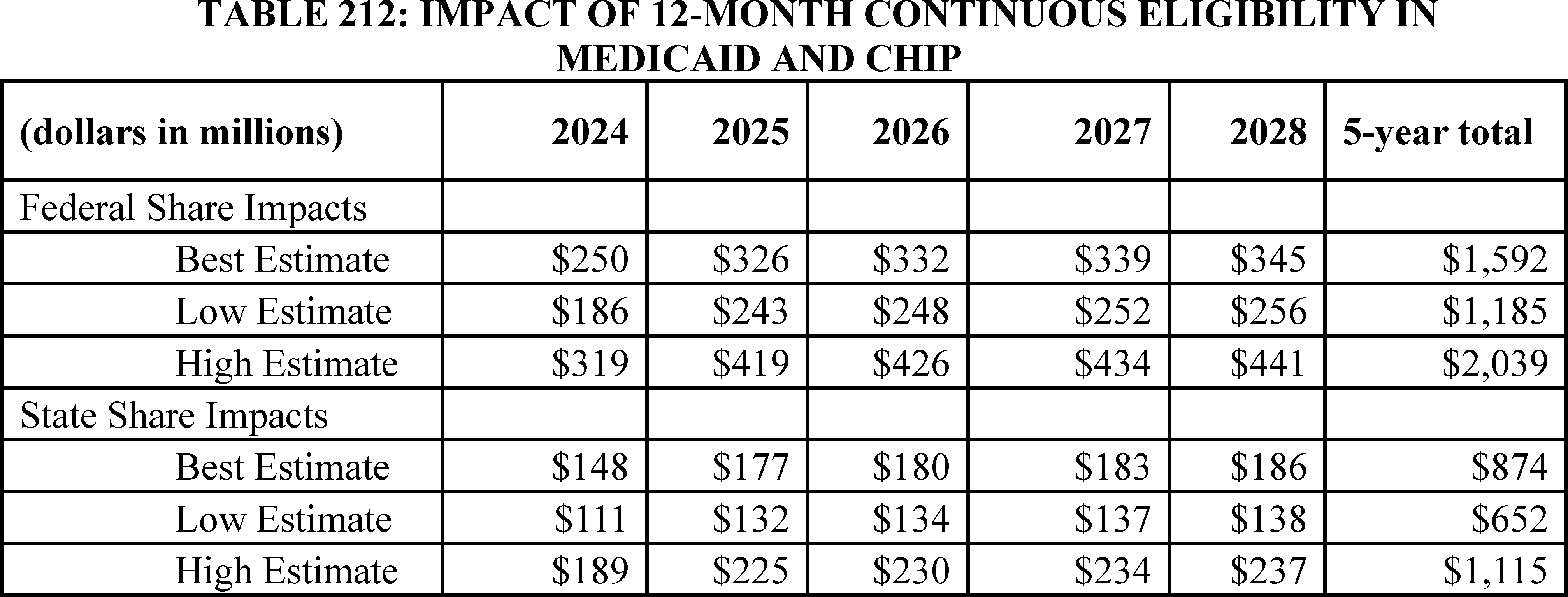

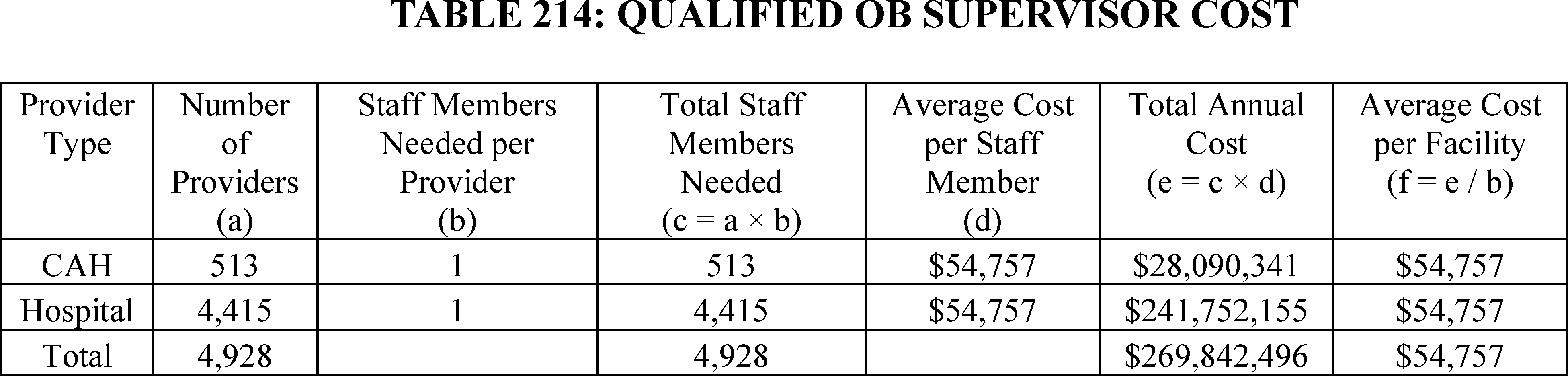
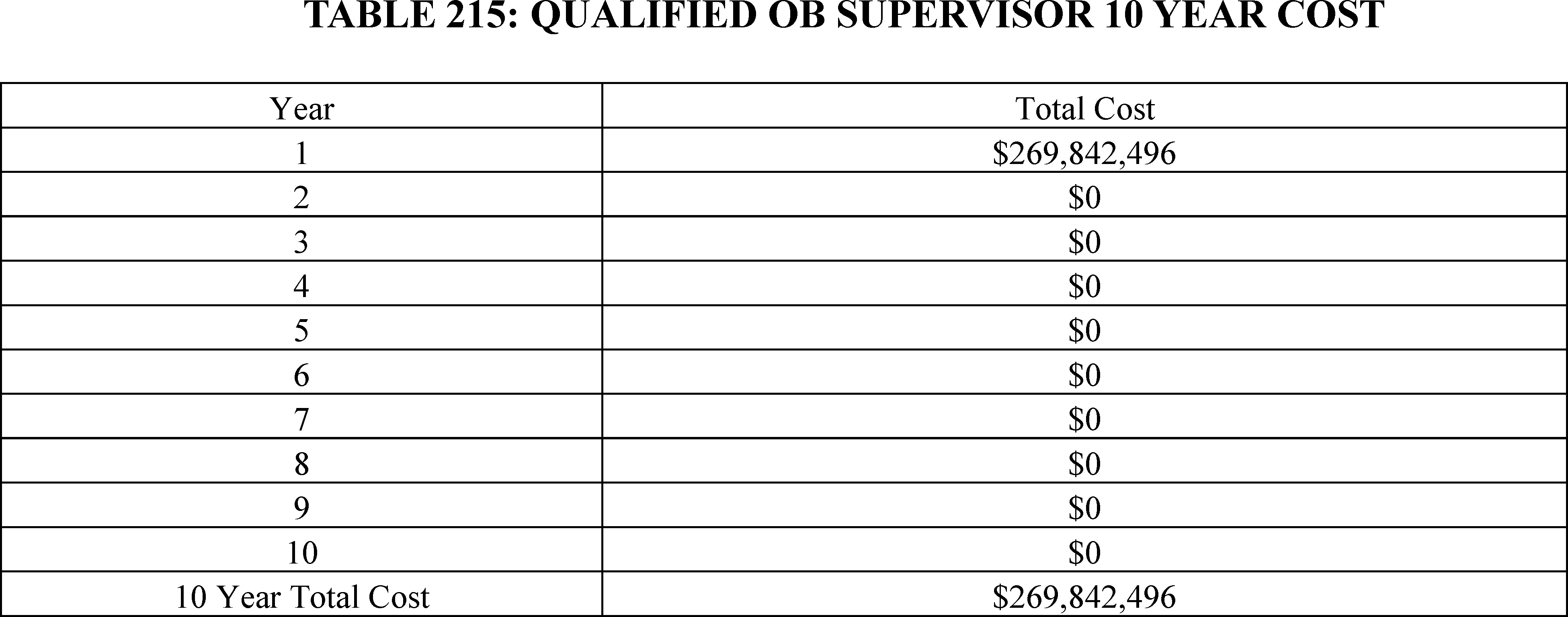
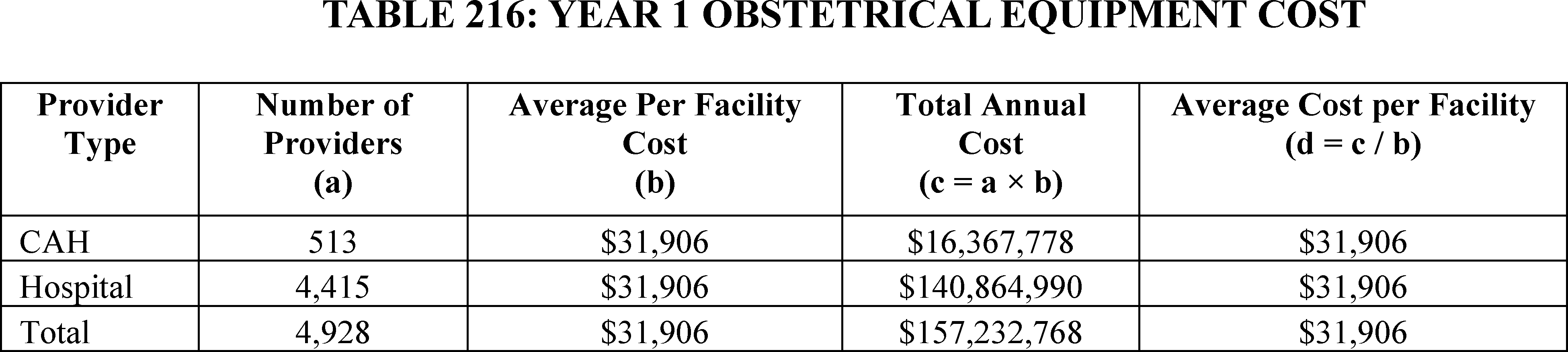
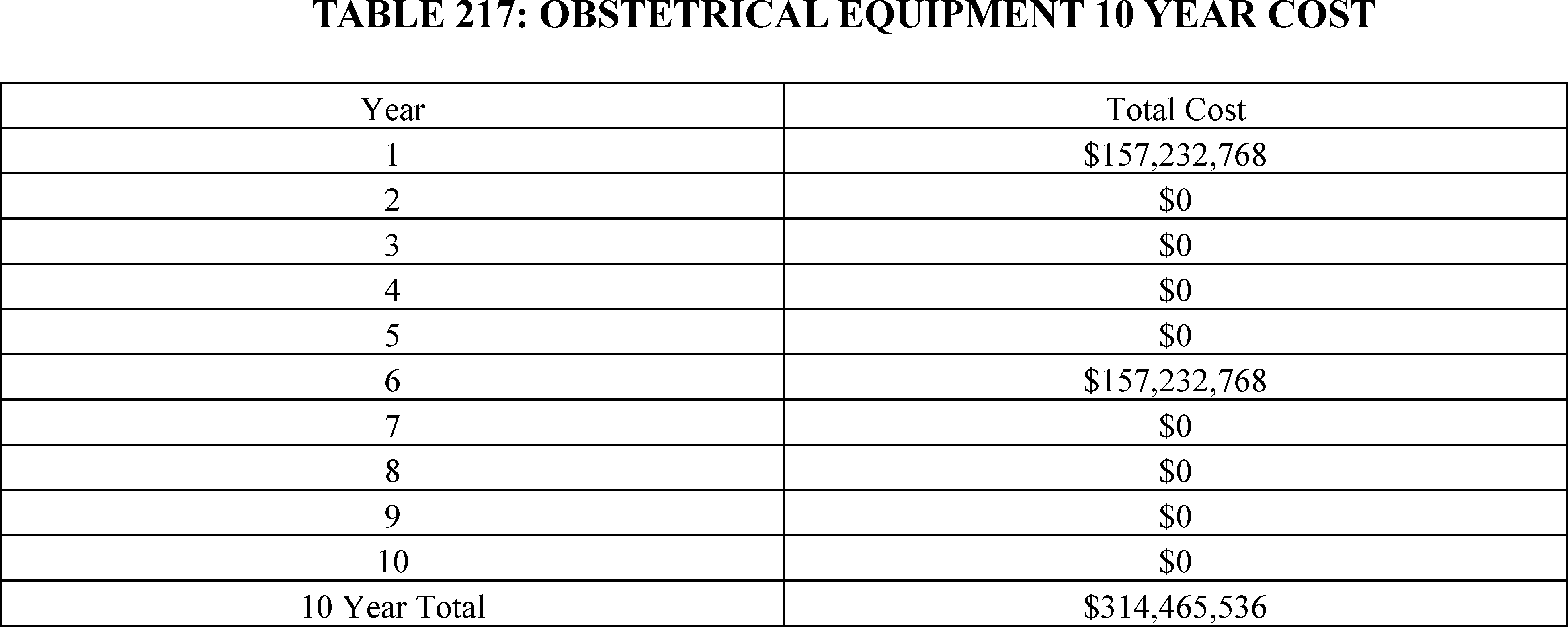
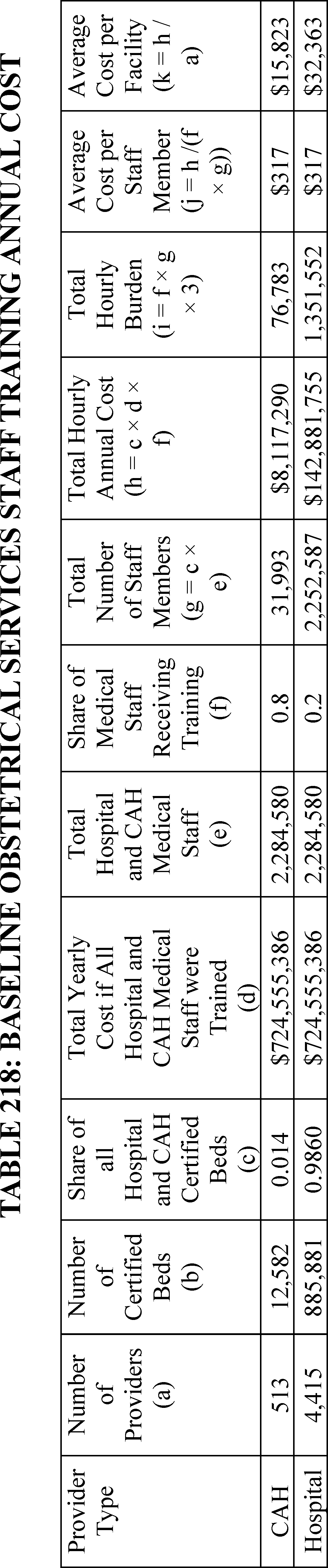

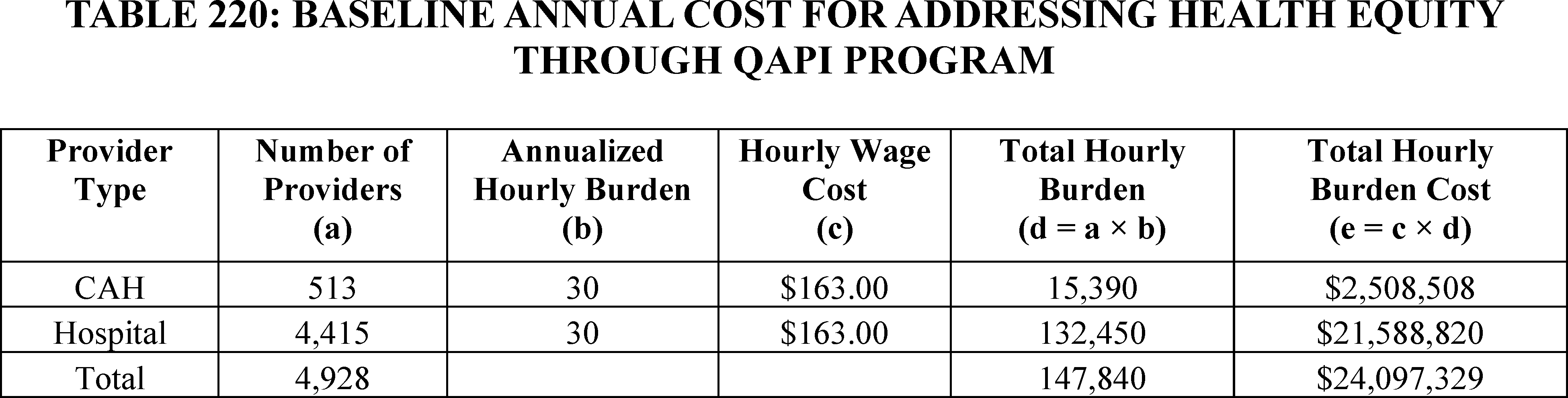
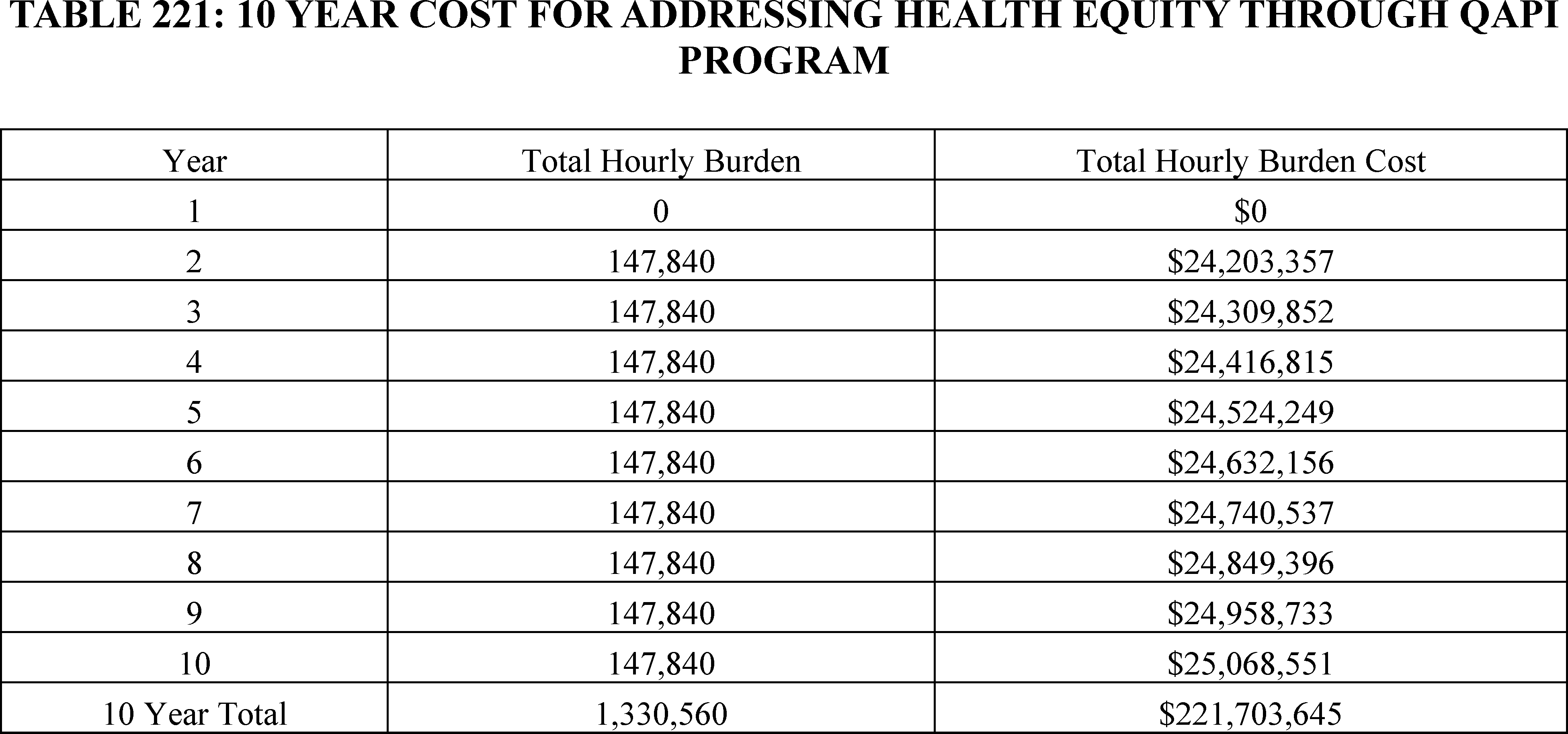


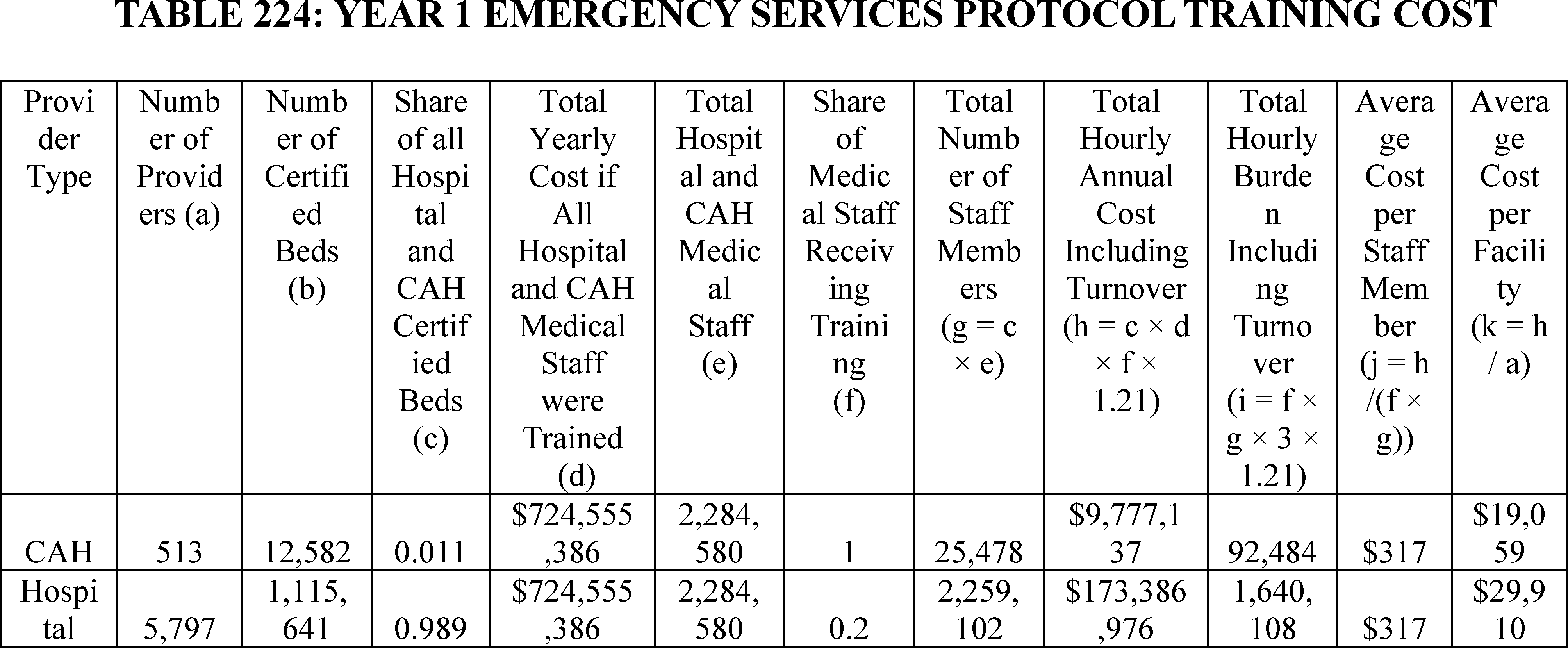
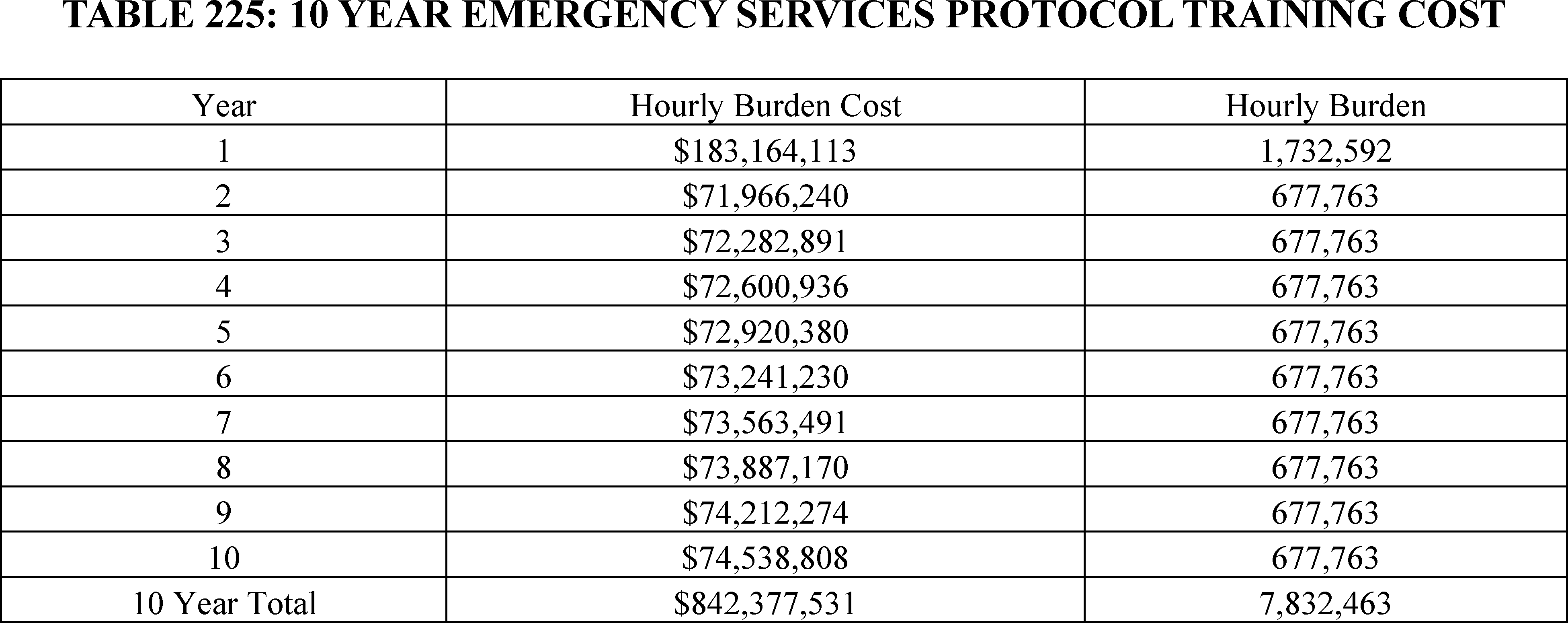
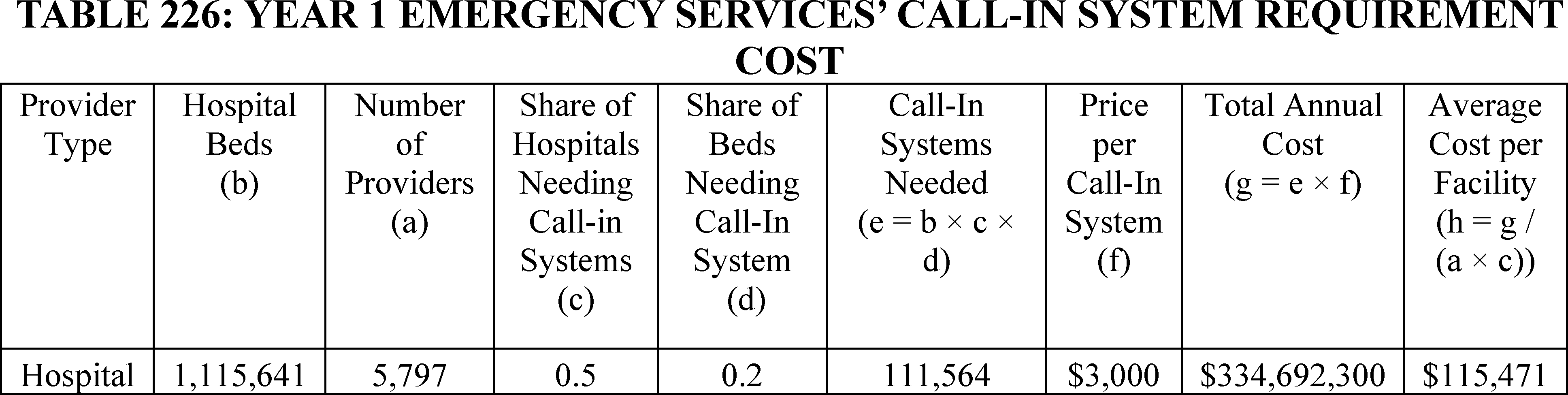
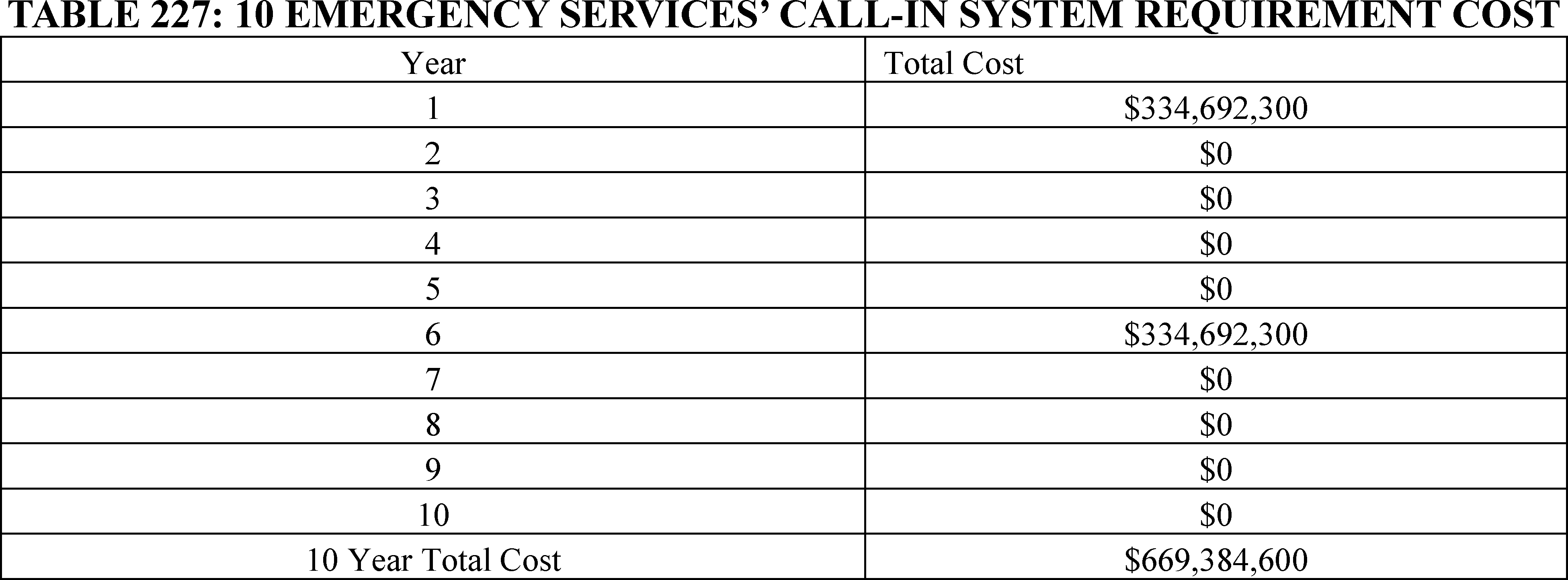
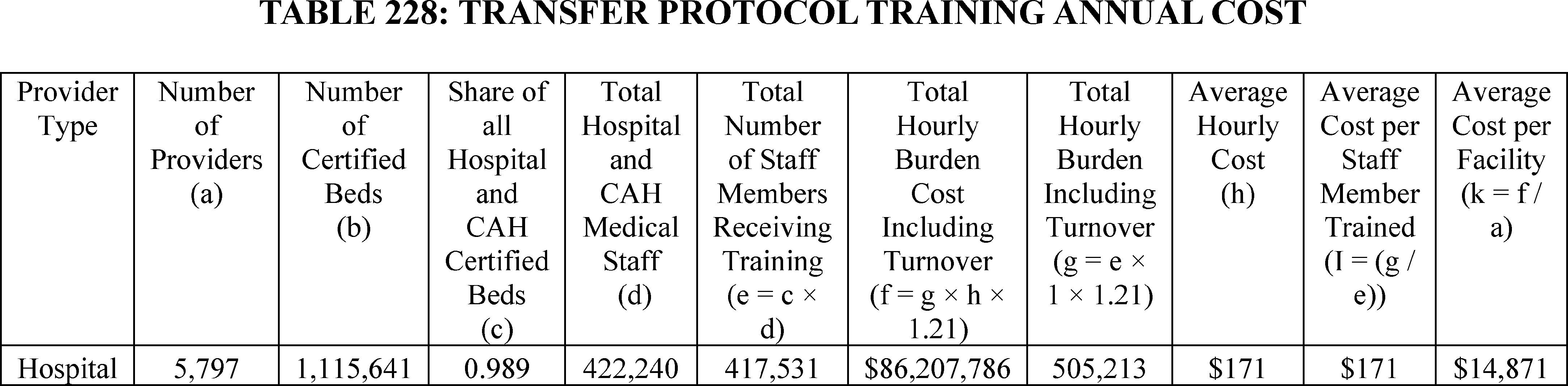
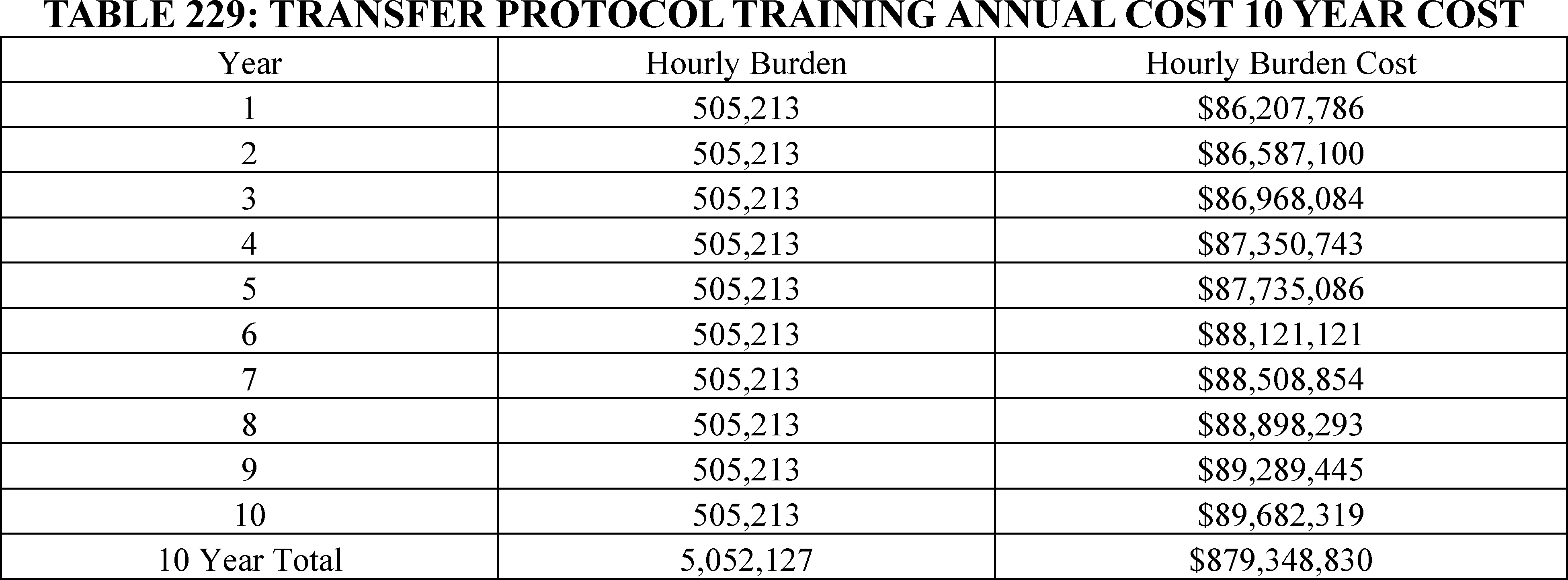
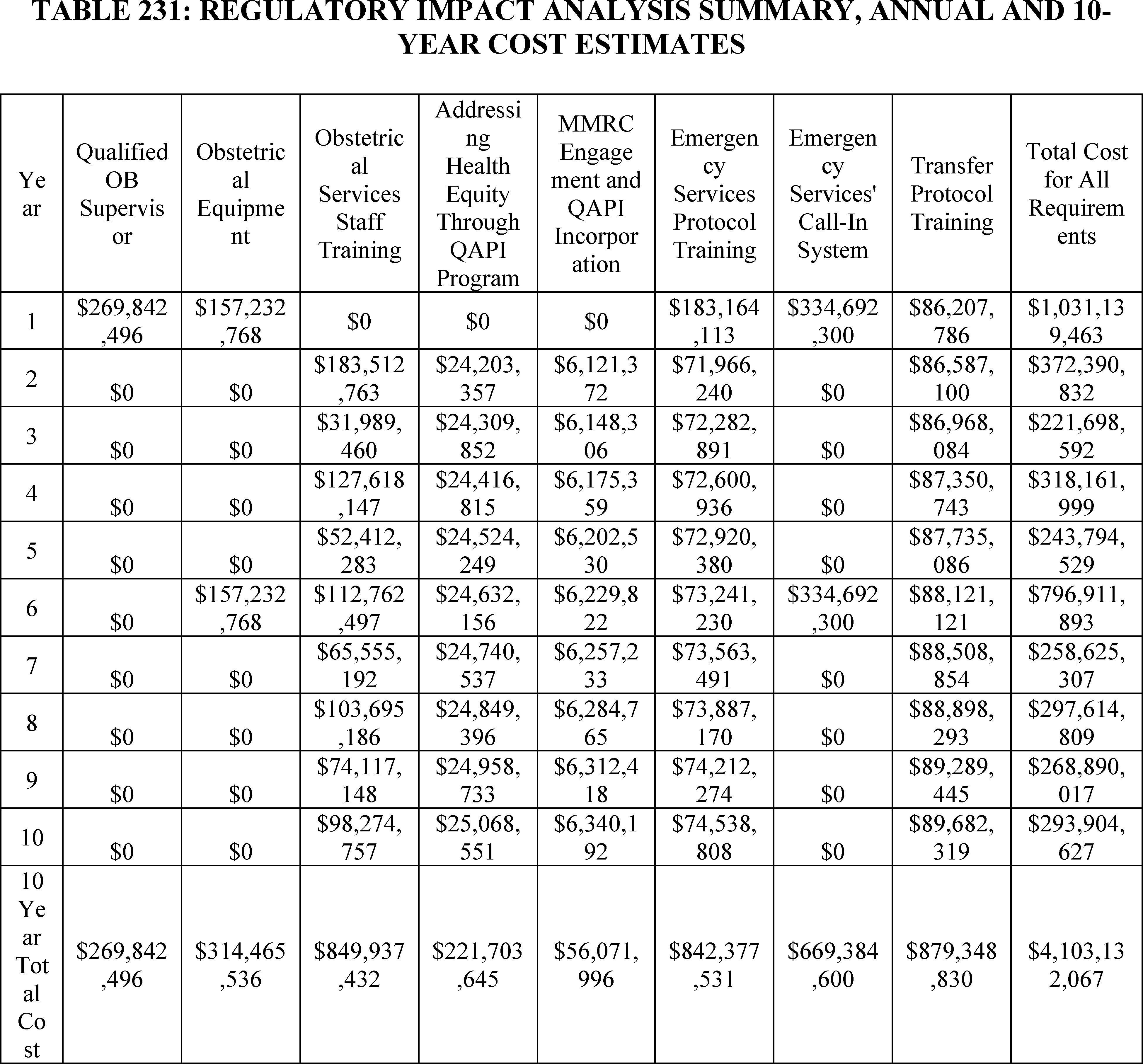
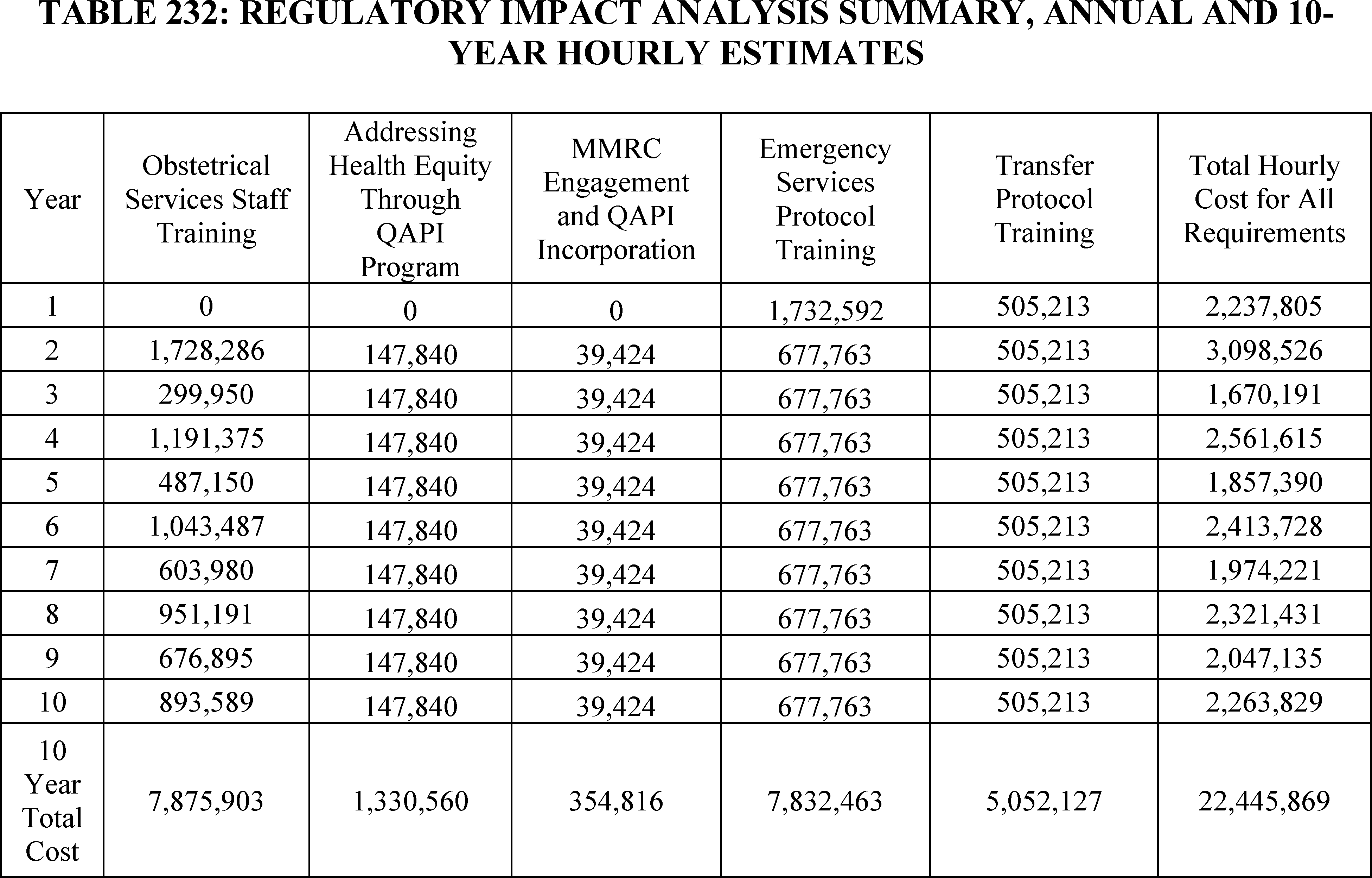
22 comments have been received at Regulations.gov.
Agencies review all submissions and may choose to redact, or withhold, certain submissions (or portions thereof). Submitted comments may not be available to be read until the agency has approved them.