Academia.edu uses cookies to personalize content, tailor ads and improve the user experience. By using our site, you agree to our collection of information through the use of cookies. To learn more, view our Privacy Policy.
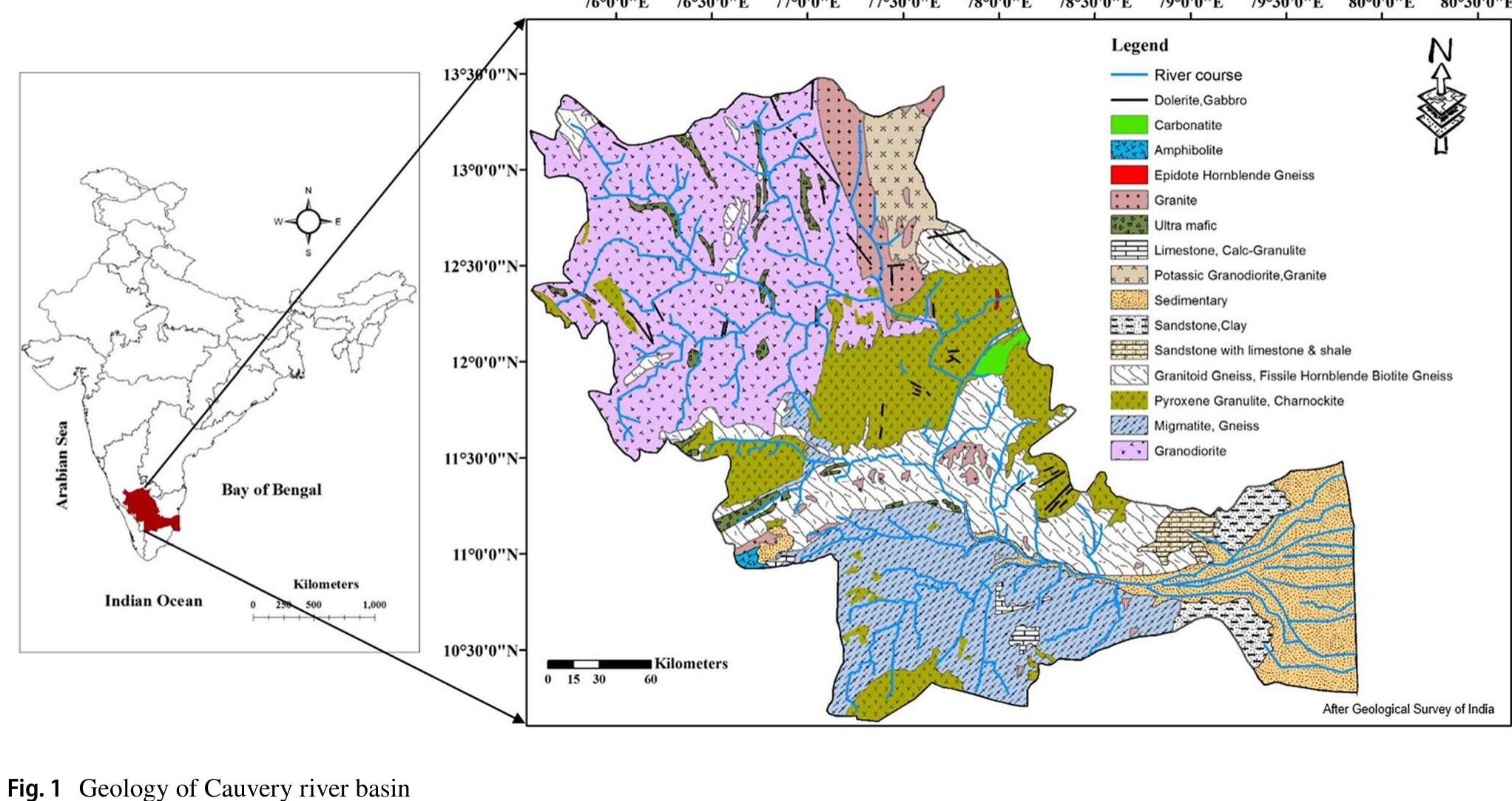
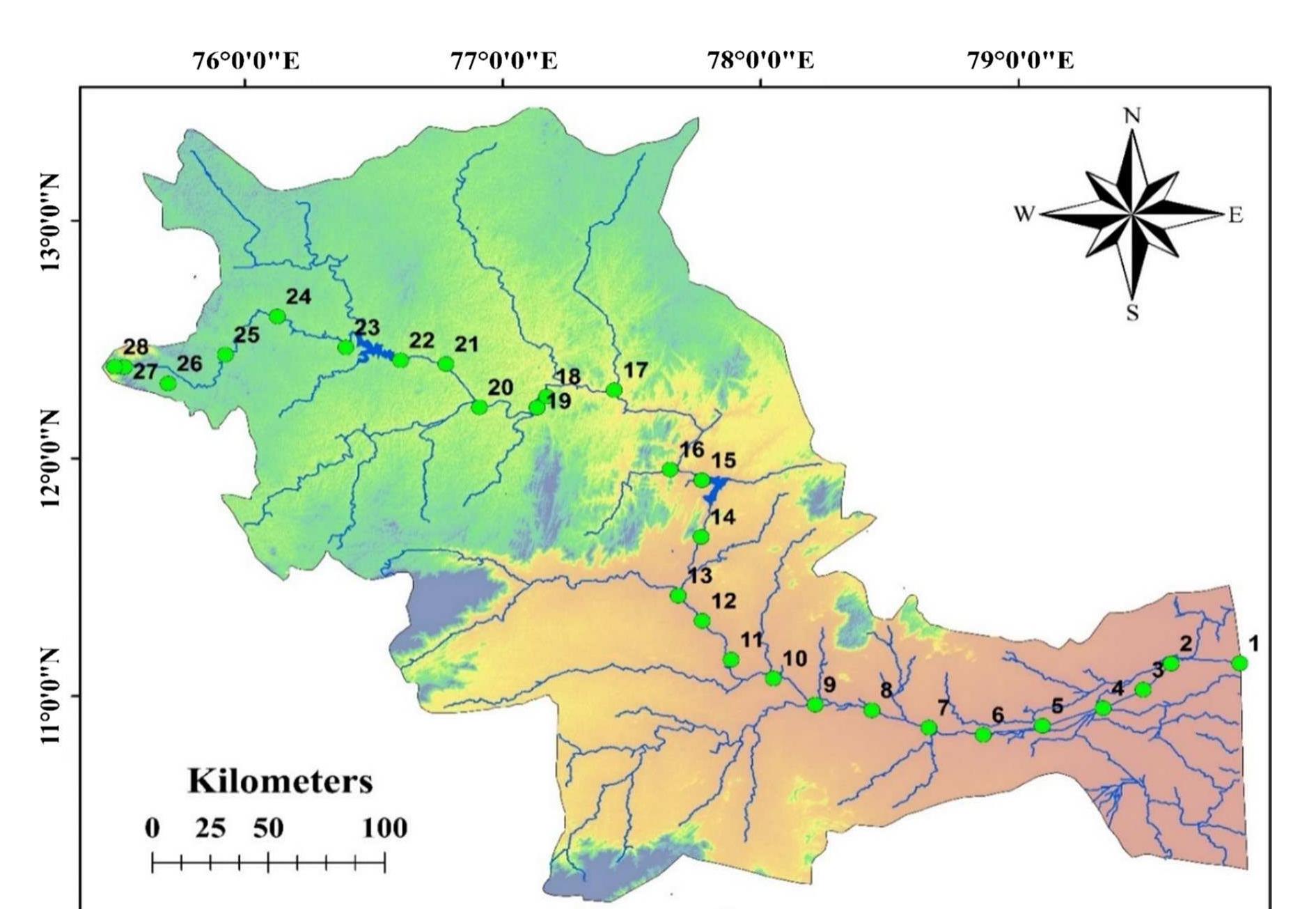
Evaluation of geogenic and anthropogenic impacts on spatio‑temporal variation in quality of surface water and groundwater along Cauvery River, India
https://doi.org/10.1007/S12665-017-7176-6…
17 pages

Sign up for access to the world's latest research
Abstract
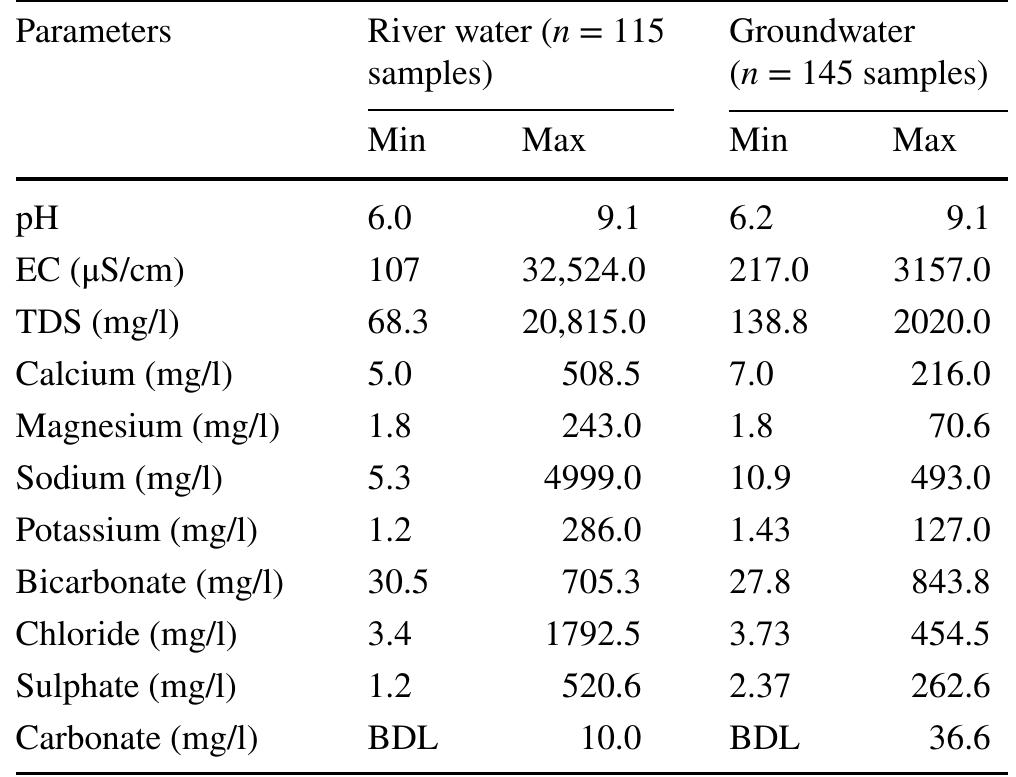
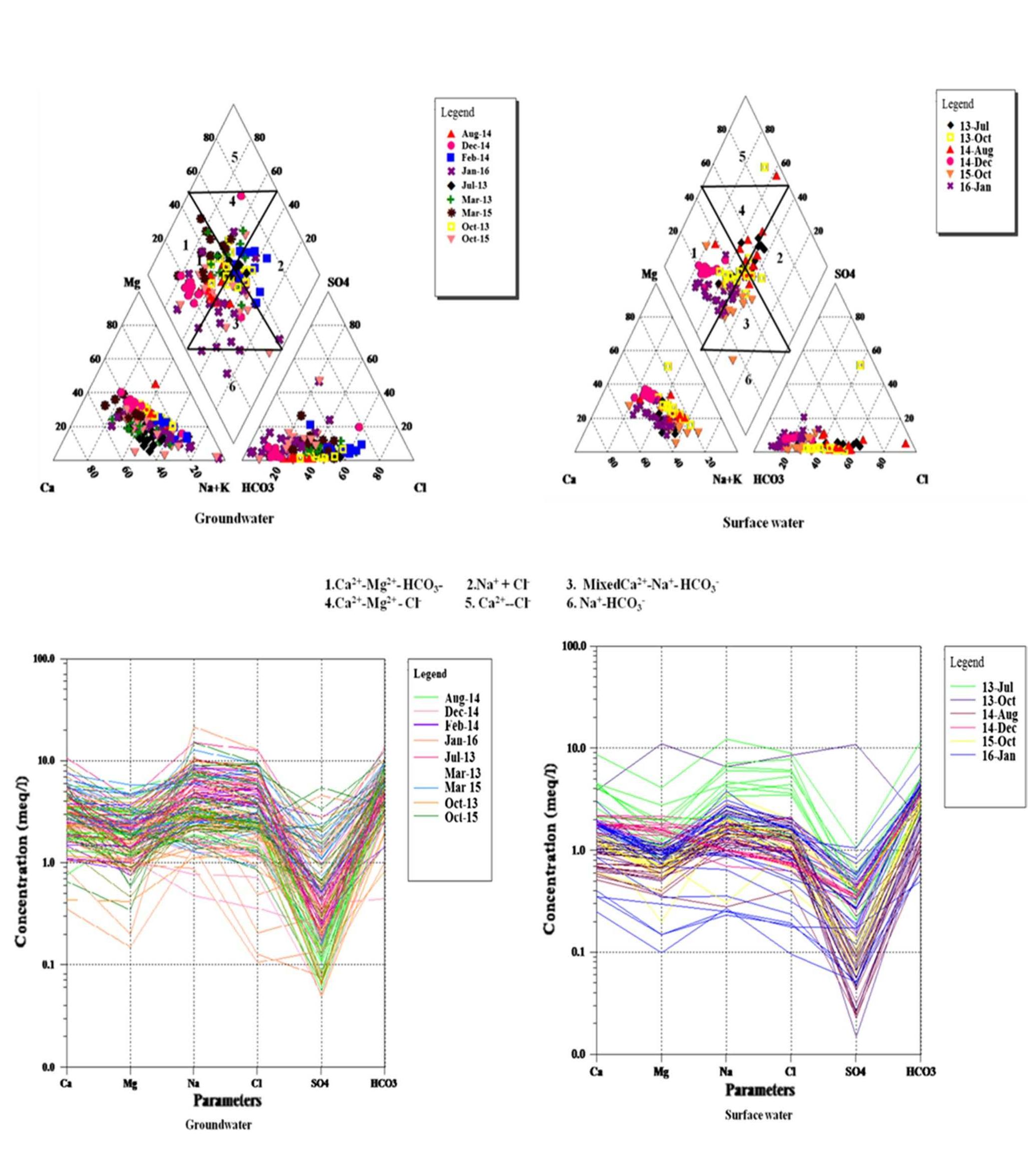
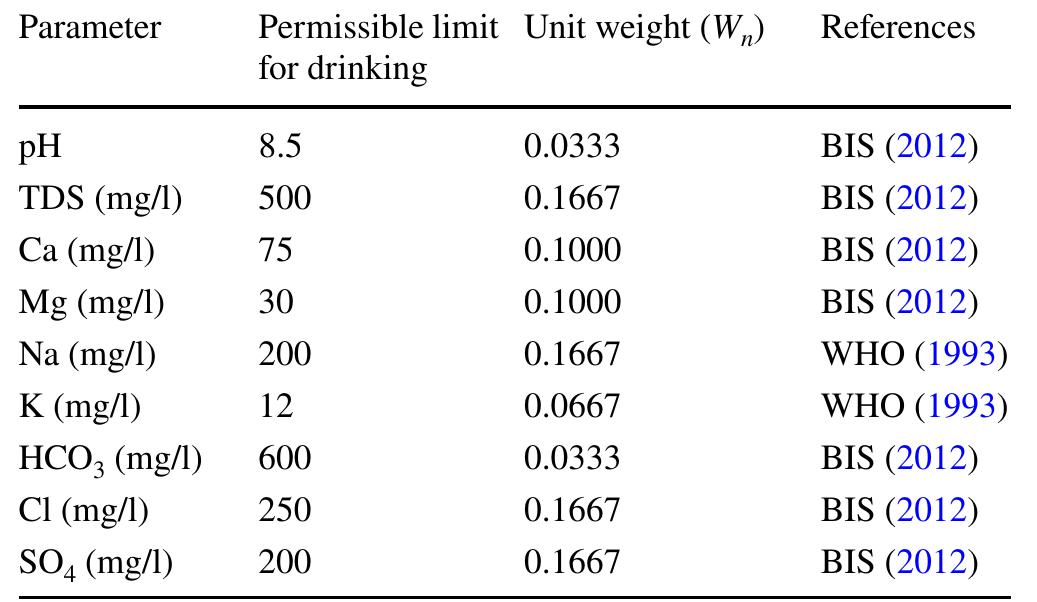
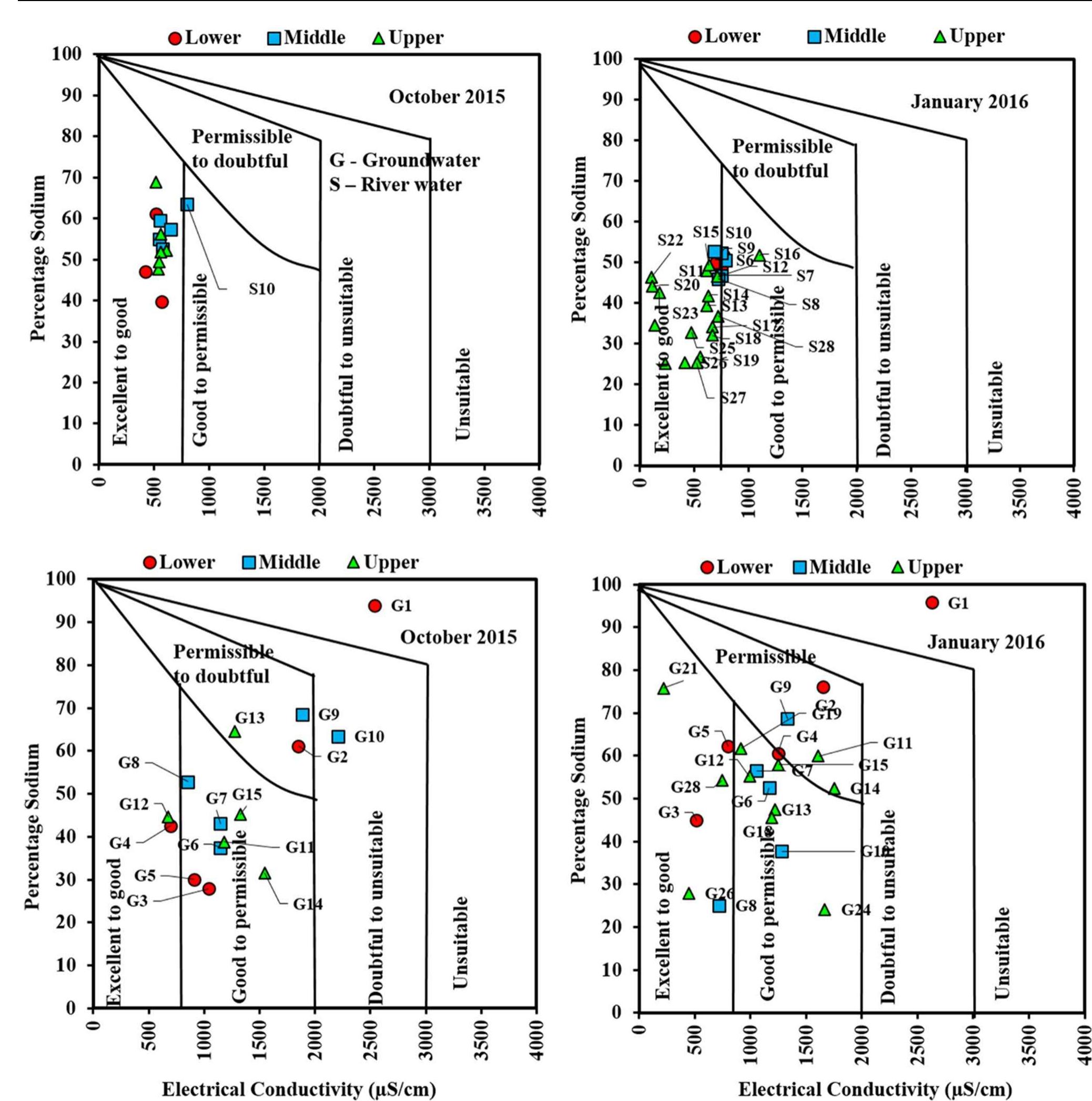
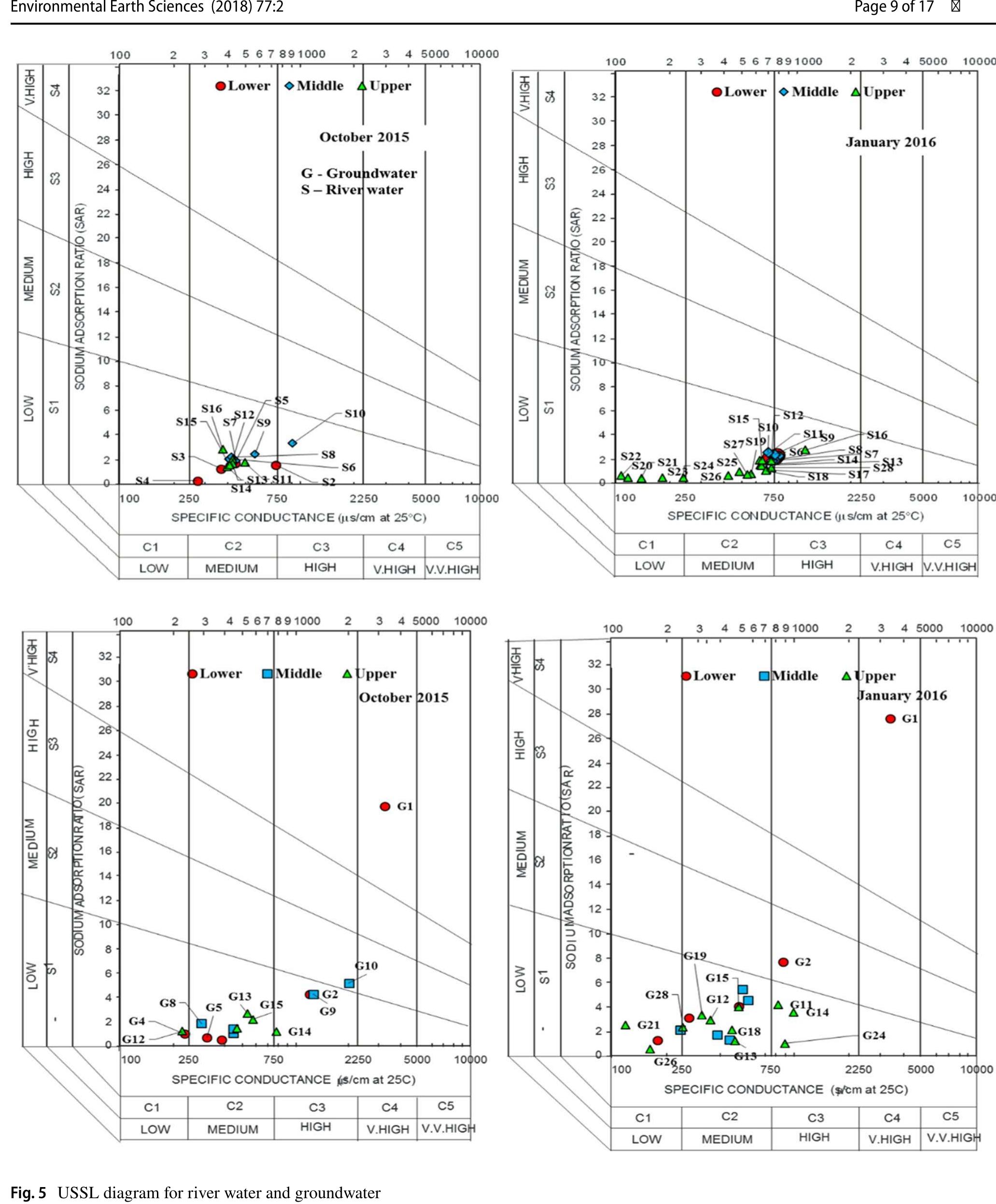
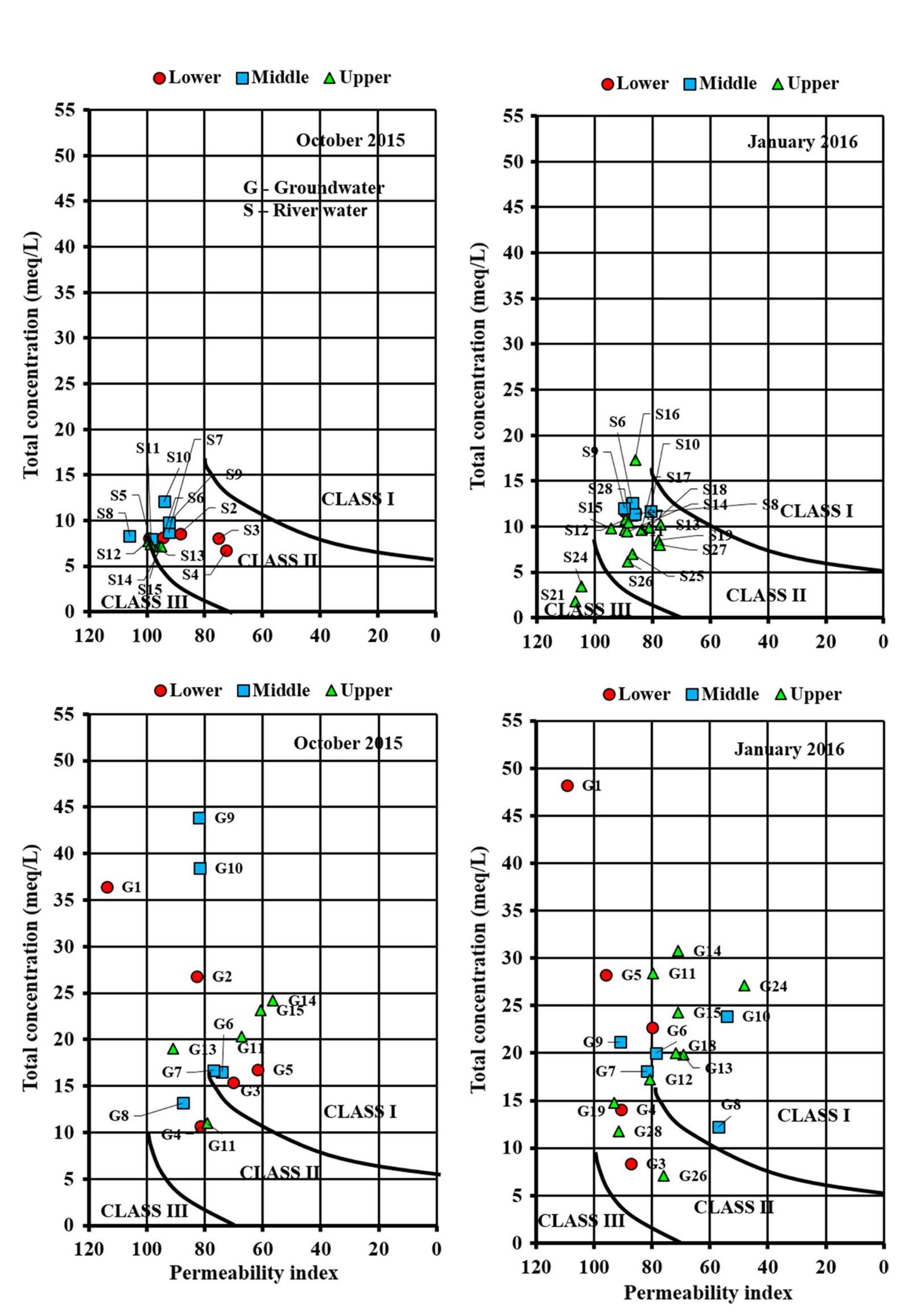
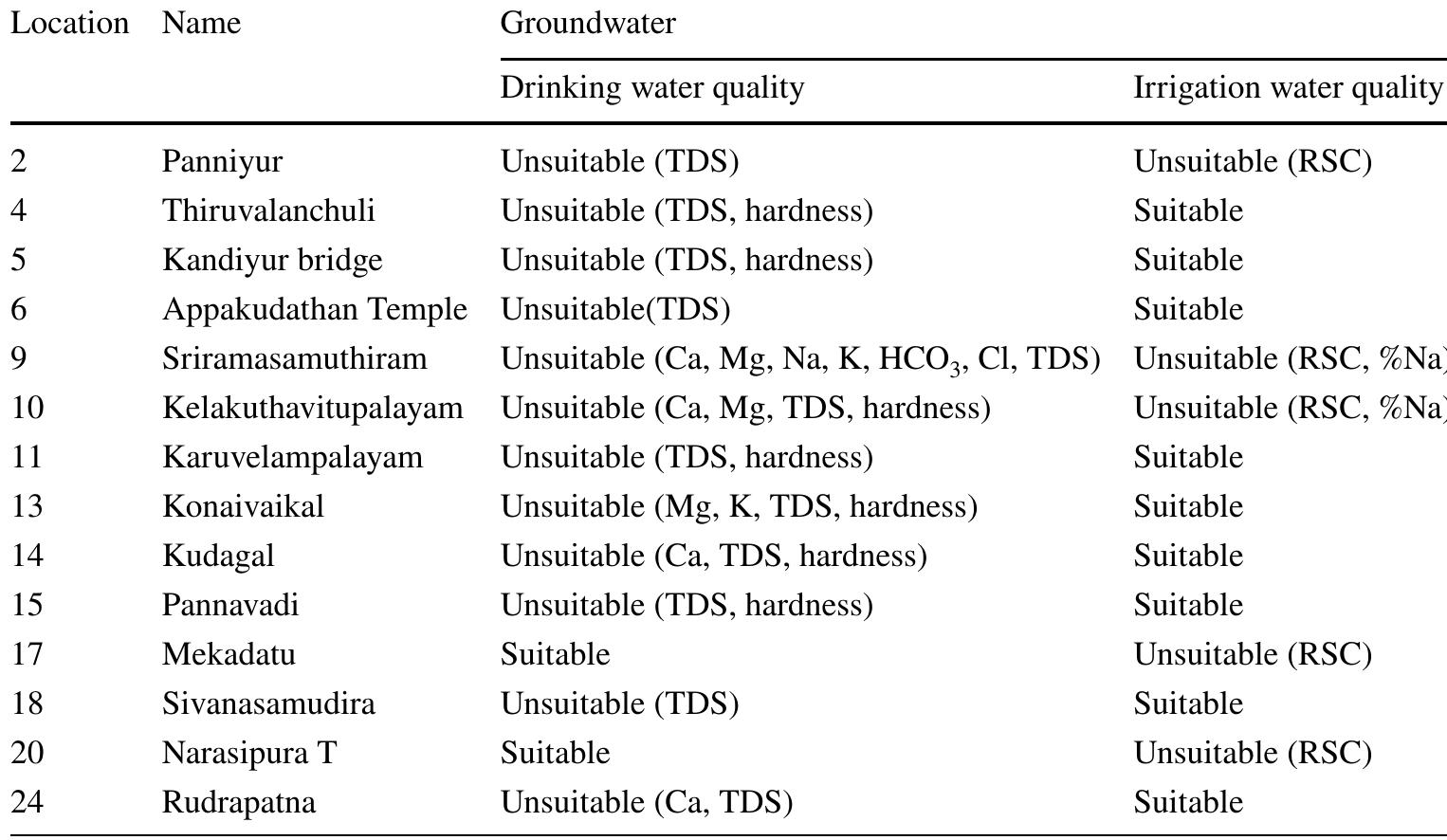
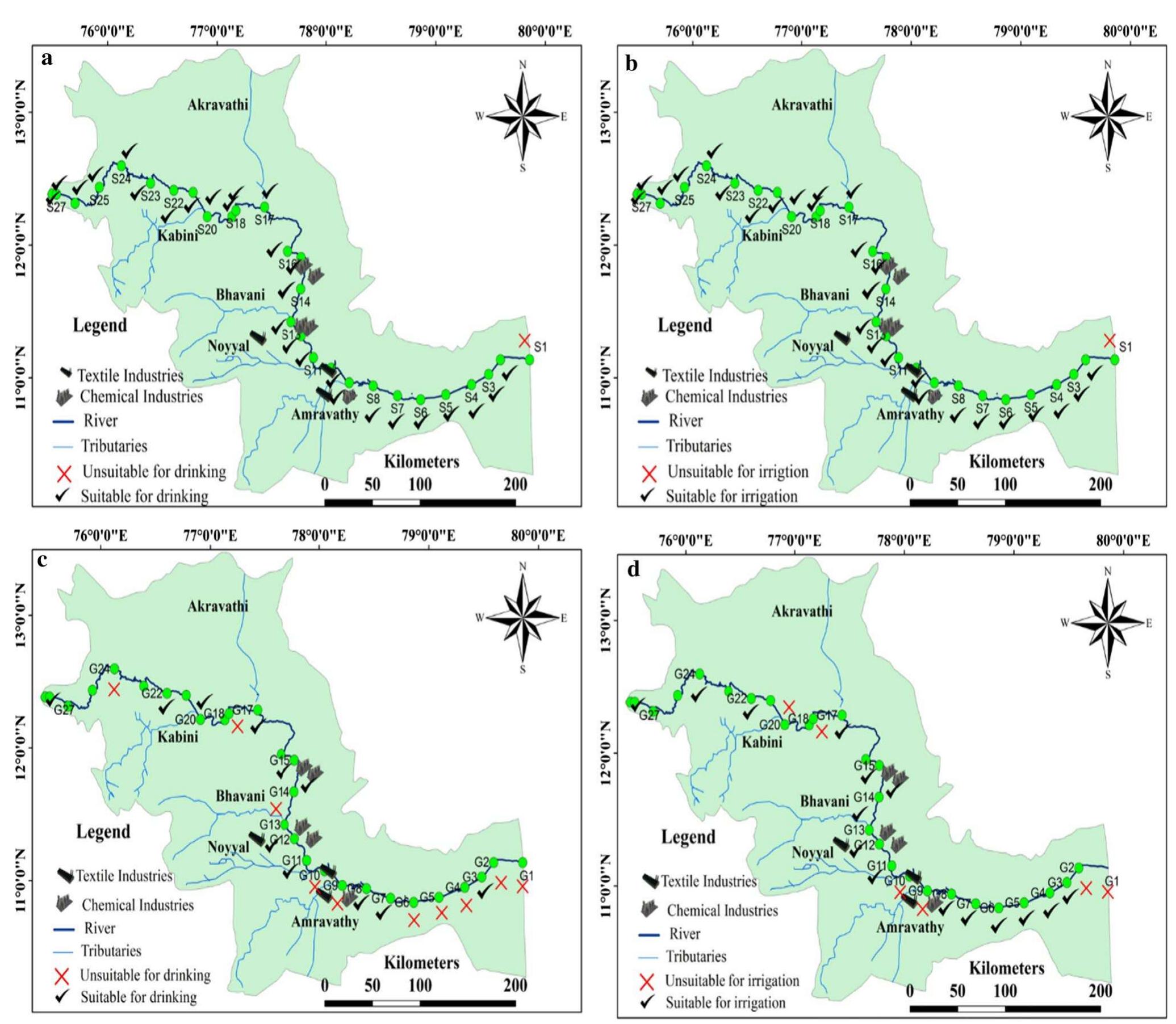
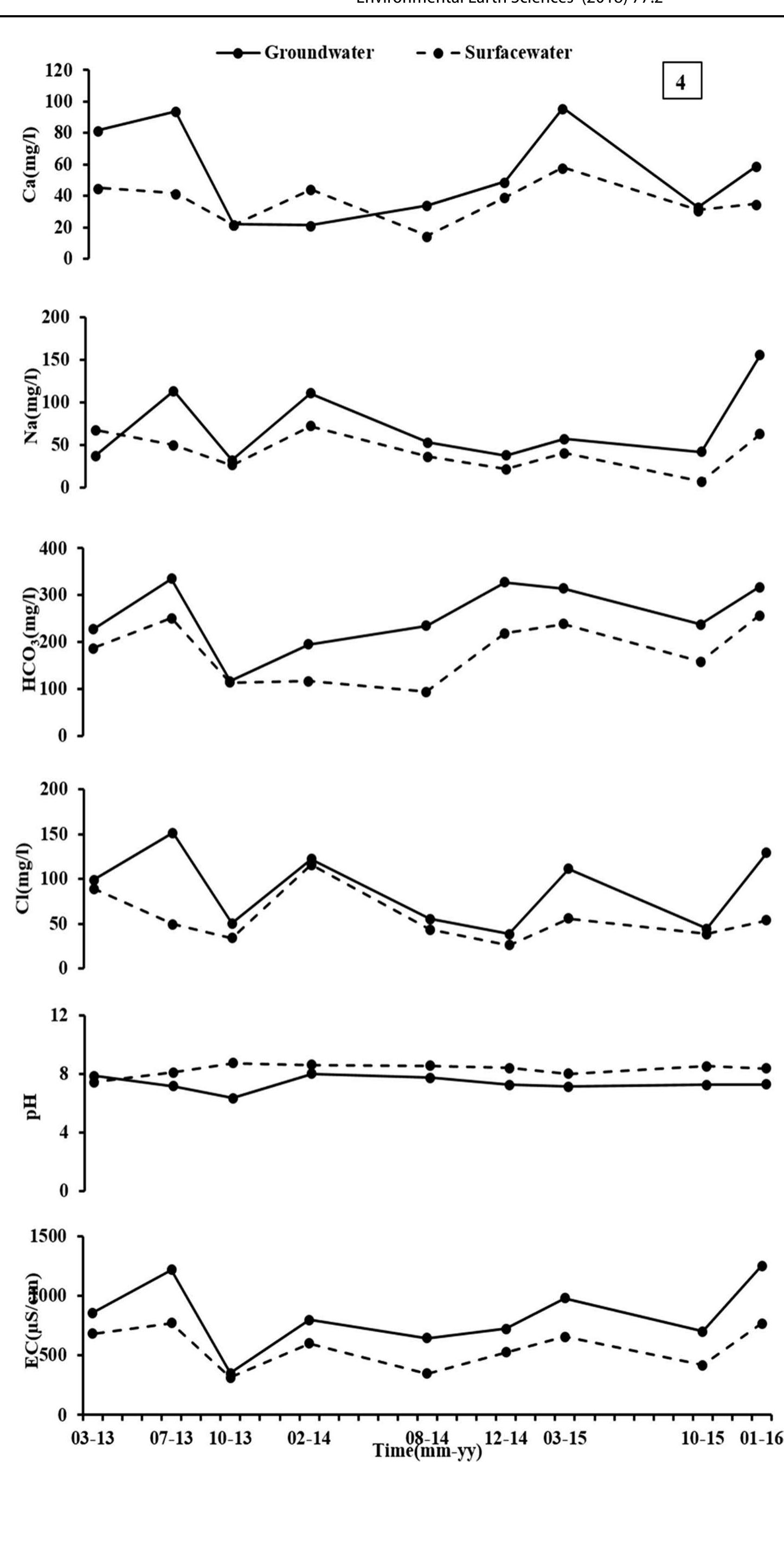
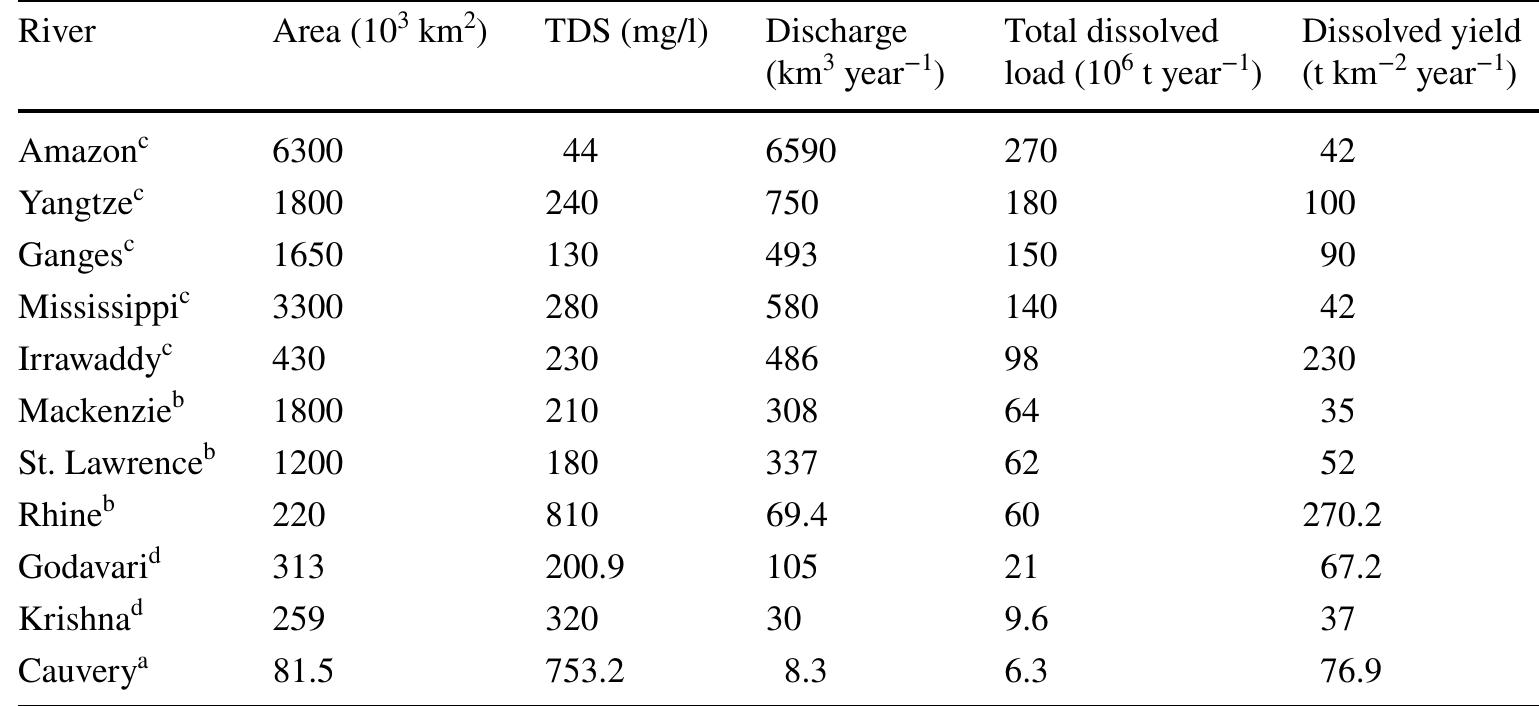
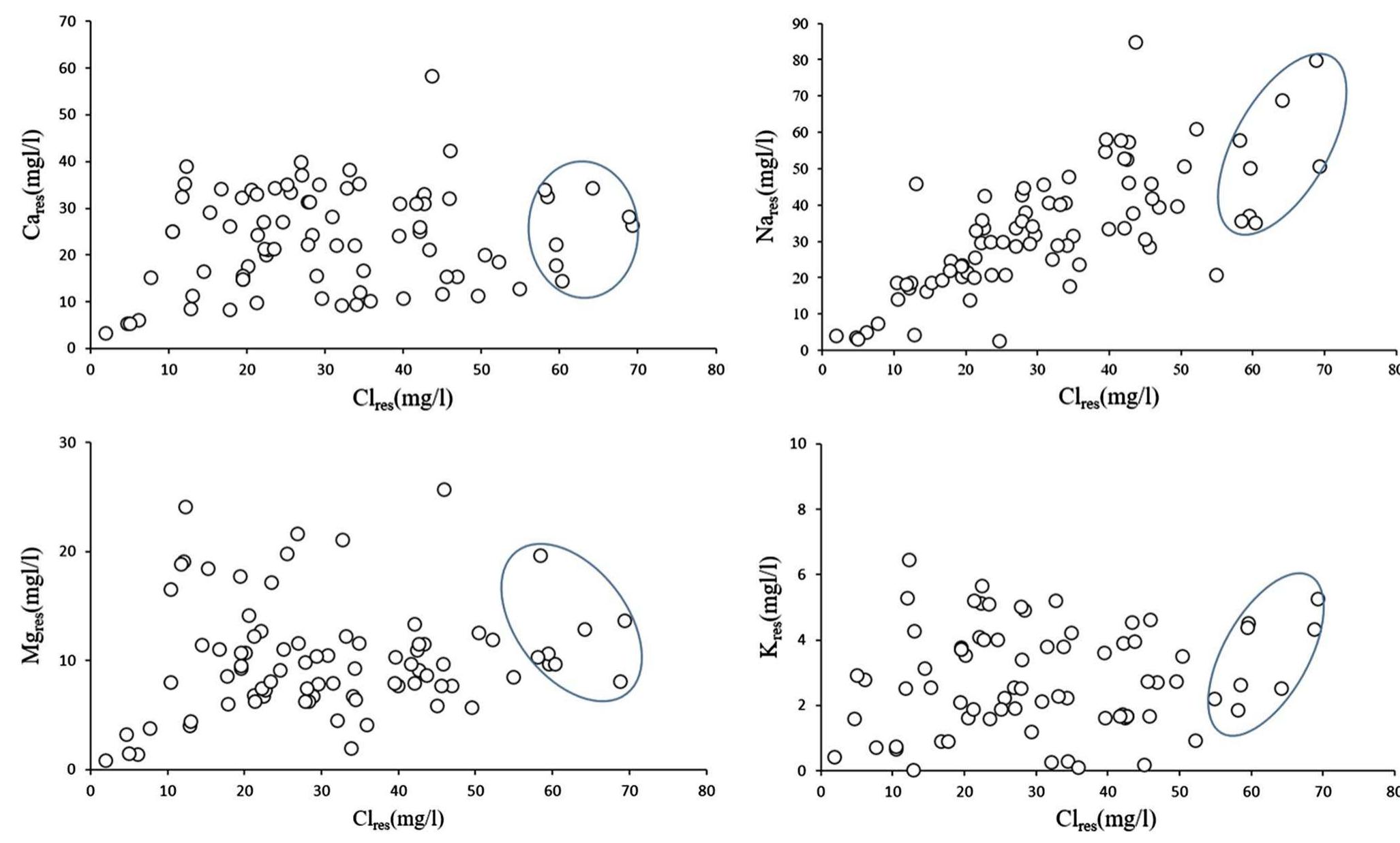
Assessment of groundwater and surface water quality along a river is important as it directly affects the agricultural, industrial activities and population. The objective of the study is to assess the quality of the Cauvery river water and adjacent groundwater for drinking and irrigational purposes and to identify the infuence of geogenic and anthropogenic sources. Groundwater and surface water samples were collected along the course of the river at approximate intervals of 25 km. The samples were analysed for electrical conductivity, pH, sodium, calcium, magnesium, potassium, bicarbonate, chloride and sulphate. Sodium was identified as the dominant cation and bicarbonate was the dominant anion for both river water and groundwater. These values were compared with limits recommended by the Bureau of Indian Standards for drinking purposes. The total dissolved solids were found to exceed the permissible limits for drinking water in most of the groundwater samples, and it was below the permissible limits in river water samples. Most of the river water samples were found to be suitable as per the drinking water quality standards, but most of the groundwater samples were unsuitable based on the concentration of major ions. Irrigation water quality was also assessed based on magnesium hazard, residual sodium carbonate, sodium percentage, sodium adsorption ratio, permeability index and salinity hazard. Most of the river water samples collected were suitable for irrigation, whereas most of the groundwater samples collected were doubtful for irrigation based on residual sodium carbonate and sodium percentage. Drinking water and irrigation water quality indices were also computed to assess the characteristics of water. Groundwater quality in locations nearer to the confluence of tributaries and industrial areas was of poor quality, while both river water and groundwater near the coast were poor, both for drinking and irrigation purposes. Comparison of the dissolved load with other rivers of the world was also made, which reveals that the Cauvery River yields comparatively higher dissolved load per area than most of the rivers. The chemical load in the river is due to natural and anthropogenic sources. Therefore, it is necessary to enforce the existing norms for the discharge of treated effluents by industries and townships along the river so as to reduce the chemicals contributed by anthropogenic sources.



Related papers
AbStRAct The ongoing degradation of the water quality of central Maharashtra's basaltic aquifer is of great concern for different authorities and agencies involved in the water sector in the Maharashtra, India. The Kham river, which is one of the major tributaries of the Godavari river, receives all domestic and industrial waste water from the Aurangabad city. The river, with no natural flow in the dry season, is extensively used for irrigation. In order to evaluate the quality of river and groundwater in the study area, eight river water and forty groundwater samples along right and left bank of the Kham river were collected and analyzed for various parameters. Physical and chemical parameters of the river and groundwater such as pH, TDS, EC, SO 4 , NO 3 and heavy metals like Pb, Cr, Cd, Zn, and Cu were determined. The results show that the river and groundwater of the area is generally unfit for domestic uses. Most of the physico-chemical parameters and heavy metals in the river and groundwater samples have higher value than the World Health Organization (WHO, 2006) and Bureau of Indian Standards (BIS, 2003) guidelines.
Environmental …, 2009
Groundwater quality of the Banana Plain (Mbanga, Njombe, Penja-Cameroon) was assessed for its suitability for drinking, domestic, and agricultural uses. A total of 67 groundwater samples were collected from open wells, springs, and boreholes. Samples were analyzed for physicochemical properties, major ions, and dissolved silica. In 95% of groundwater samples, calcium is the dominant cation, while sodium dominates in 5% of the samples. Eighty percent of the samples have HCO 3 as major anion, and in 20%, NO 3 is the major anion. Main water types in the study area are CaHCO 3 , CaMgHCO 3 , CaNaHCO 3 , and CaNaNO 3 ClHCO 3. CO 2-driven weathering of silicate minerals followed by cation exchange seemingly controls largely the concentrations of major ions in the groundwaters of this area. Nitrate, sulfate, and chloride concentrations strongly express the impact of anthropogenic activities (agriculture and domestic activities) on groundwater quality. Sixty-four percent of the waters have nitrate concentrations higher than the drinking water limit. Also limiting groundwater use for potable and domestic purposes are contents of Ca 2? , Mg 2? and HCO 3 and total hardness (TH) that exceed World Health Organization (WHO) standards. Irrigational suitability of groundwaters in the study area was also evaluated, and results show that all the samples are fit for irrigation. Groundwater quality in the Banana Plain is impeded by natural geology and anthropogenic activities, and proper groundwater management strategies are necessary to protect sustainably this valuable resource.
aes.asia.edu.tw
The water quality of river Cauvery in Tiruchirappalli district was monitored for a period of 3 months (January-March, 2009). Water samples were assessed by analyzing the various physicochemical parameters, such as pH, total dissolved solids, total hardness, total alkalinity, dissolved oxygen, biological oxygen demand, chloride, sulphate, nitrate and calcium. These 10 parameters were considered to compute the Water Quality Index (WQI). The WQI reveals the water of river Cauvery to be polluted moderately in the upstream of the city and unfit for human consumption towards the downstream. It needs sufficient treatment and management.
Journal of the Geological Society of India, 2021
Groundwater and surface water quality is an important factor that determines its usage for drinking and irrigational use. This study was carried out along a major irrigation water source—Uyyakondan channel in Tiruchirappalli, south India. Fourteen surface water samples along the channel and fifteen groundwater samples close to the surface water sampling locations were collected to determine its suitability for drinking and irrigational purposes. Electrical conductivity, pH and concentrations of calcium, magnesium, sodium, potassium, carbonate, bicarbonate, chloride, sulphate, fluoride and nitrate were determined in the water samples. The chemical composition of the water samples were compared with the drinking water standards of World Health Organisation and Bureau of Indian Standards. Groundwater from this area was suitable for drinking based on magnesium, sulphate, bicarbonate, fluoride and nitrate, while the concentration of calcium, sodium, potassium and chloride exceeded the maximum permissible limits at few locations. Surface water was within the permissible limits for magnesium, potassium, bicarbonate, sulphate, fluoride and nitrate, while calcium, sodium and chloride exceeded the highest desirable limits. Sodium chloride was the dominant groundwater and surface water type. Irrigation water quality was assessed based on magnesium hazard, residual sodium carbonate, sodium percentage, sodium adsorption ratio, permeability index and salinity hazard. Water was suitable for irrigation based on magnesium hazard and residual sodium carbonate. Most water samples were doubtful for irrigation use based on sodium percentage and good for irrigation depending on sodium adsorption ratio. Though 60 % of groundwater and 29 % of surface water samples were suitable for drinking based on water quality index, majority of the water samples were not suitable for irrigation. The water quality in this area needs to be monitored regularly and it is crucial to treat the water before consumption.
Environmental Geology, 2007
In the management of water resources, quality of water is just as important as its quantity. In order to know the quality and/or suitability of groundwater for domestic and irrigation in upper Gunjanaeru River basin, 51 water samples in postmonsoon and 46 in pre-monsoon seasons were collected and analyzed for various parameters. Geological units are alluvium, shale and quartzite. Based on the analytical results, chemical indices like percent sodium, sodium adsorption ratio, residual sodium carbonate, permeability index (PI) and chloroalkaline indices were calculated. The pre-monsoon waters have low sodium hazard as compared to post-monsoon season. Residual sodium carbonate values revealed that one sample is not suitable in both the seasons for irrigation purposes due the occurrence of alkaline white patches and low permeability of the soil. PI values of both seasons revealed that the ground waters are generally suitable for irrigation. The positive values of Chloroalkaline indices in post-monsoon (80%) and in premonsoon (59%) water samples indicate absence of base-exchange reaction (chloroalkaline disequilibrium), and remaining samples of negative values of the ratios indicate base-exchange reaction (chloroalkaline equilibrium). Chadha rectangular diagram for geochemical classification and hydrochemical processes of groundwater for both seasons indicates that most of waters are Ca-Mg-HCO 3 type. Assessment of water samples from various methods indicated that majority of the water samples in both seasons are suitable for different purposes except at Yanadipalle (sample no. 8) that requires precautionary measures. The overall quality of groundwater in post-monsoon season in all chemical constituents is on the higher side due to dissolution of surface pollutants during the infiltration and percolation of rainwater and at few places due to agricultural and domestic activities.
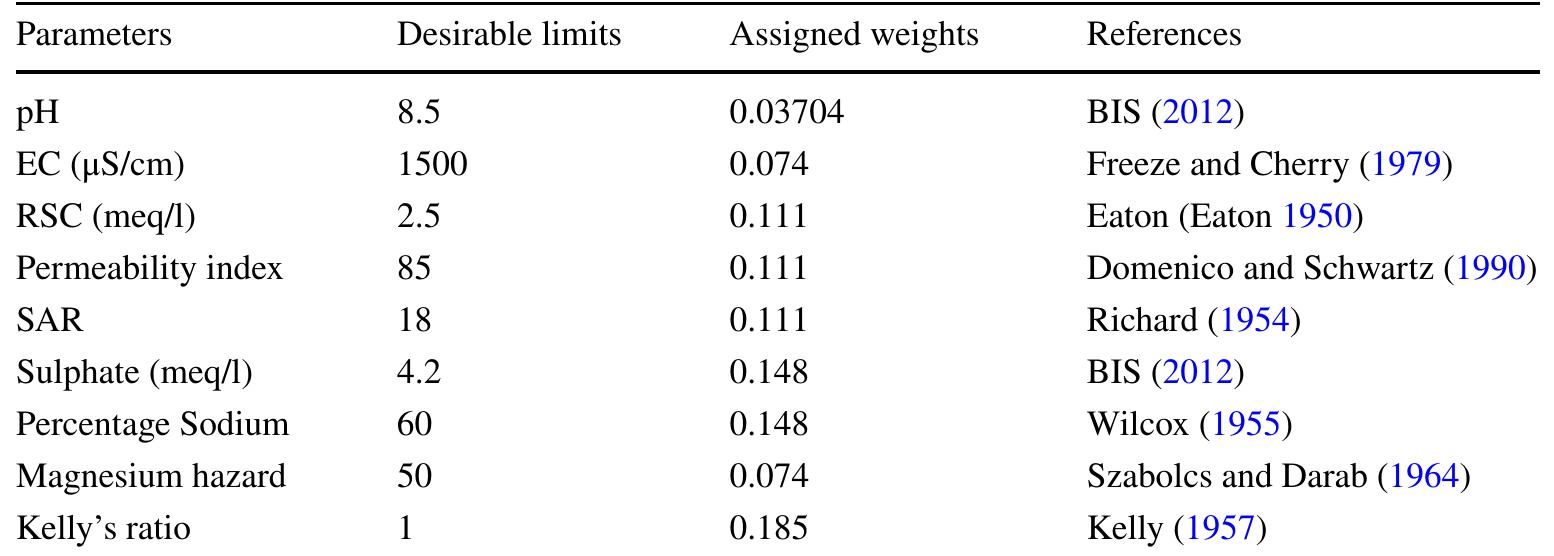
Groundwater is the major source of water supplies and irrigation, as also an important source Groundwater is the major source of water supplies and irrigation, as also an important source catering to industrial needs in the country. In the coming decades, groundwater considered as catering to industrial needs in the country. In the coming decades, groundwater considered as a sustainable and pure source of water, will be more and more in demand. But quality of a sustainable and pure source of water, will be more and more in demand. But quality of groundwater , which determines its usability for various purposes, is variable in both space groundwater , which determines its usability for various purposes, is variable in both space and time, Several factors control groundwater chemistry and quality like climate and rainfall, and time, Several factors control groundwater chemistry and quality like climate and rainfall, soil condition, geology, landuse, and water rock interaction. The geological set up of the soil condition, geology, landuse, and water rock interaction. The geological set up of the country is widely varied with two major hydrogeological zones, namely alluvial plains and country is widely varied with two major hydrogeological zones, namely alluvial plains and peninsular shield of crystallines, intrusives and volcanics. Mineralogical composition of the peninsular shield of crystallines, intrusives and volcanics. Mineralogical composition of the rocks, and soils being weathered products of rocks are crucial for ionic concentrations in rocks, and soils being weathered products of rocks are crucial for ionic concentrations in groundwater. Groundwater quality evolves during its passage through the rocks by groundwater. Groundwater quality evolves during its passage through the rocks by dissolution and chemical reactions. Bicarbonate type of water is prevalent in most parts of the dissolution and chemical reactions. Bicarbonate type of water is prevalent in most parts of the country, except in arid, semiarid and coastal tracts where bicarbonate-chloride and chloride-country, except in arid, semiarid and coastal tracts where bicarbonate-chloride and chloridebicarbonate types prevail. In the coastal areas interaction between seawater and freshwater, -bicarbonate types prevail. In the coastal areas interaction between seawater and freshwater,the delicate hydrochemical and hydrodynamic balance, determine distribution of saline and the delicate hydrochemical and hydrodynamic balance, determine distribution of saline and freshwater aquifers .Groundwater also possesses some special characteristics like high freshwater aquifers .Groundwater also possesses some special characteristics like high arsenic in Lower Bengal delta, high fluoride in arid and overexploited tracts of Rajasthan, and arsenic in Lower Bengal delta, high fluoride in arid and overexploited tracts of Rajasthan, and high nitrate in pockets from agricultural chemicals. Though groundwater is generally pure, high nitrate in pockets from agricultural chemicals. Though groundwater is generally pure, free from alien substances and suitable for all uses, various anthropogenic activities are free from alien substances and suitable for all uses, various anthropogenic activities are degrading its quality. The paper narrates the salient features of groundwater quality and degrading its quality. The paper narrates the salient features of groundwater quality and MultiSpectra Consultants White Paper MultiSpectra Consultants White Paper MultiSpectra Consultants White Paper MultiSpectra Consultants White Paper pollution in different parts of the country, reflecting on their significance in management pollution in different parts of the country, reflecting on their significance in management strategies. strategies.
Journal of Earth System Science, 2011
Water samples were collected from different formations of Gadilam river basin and analyzed to assess the major ion chemistry and suitability of water for domestic and drinking purposes. Chemical parameters of groundwater such as pH, electrical conductivity (EC), total dissolved solids (TDS), Sodium (Na + ), Potassium (K + ), Calcium (Ca + ), Magnesium (Mg + ), Bicarbonate (HCO $_{3}^{\ \,-}$ ), Sulphate (SO $_{4}^{\ \,-}$ ), Phosphate (PO $_{4}^{\ \,-}$ ) and Silica (H4SiO4) were determined. The geochemical study of the aquatic systems of the Gadilam river basin show that the groundwater is near-acidic to alkaline and mostly oxidizing in nature. Higher concentration of Sodium and Chloride indicates leaching of secondary salts and anthropogenic impact by industry and salt water intrusion. Spatial distribution of EC indicates anthropogenic impact in the downstream side of the basin. The concentration levels of trace metals such as Iron (Fe), Lead (Pb), Nickel (Ni), Bromide (Br), Iodide (I) and Aluminium (Al) have been compared with the world standard. Interpretation of data shows that some trace metals such as Al, Ni and Pb exceed the acceptable limit of world standard. Geophysical study was carried out to identify the weathered zone in the hard rock and contaminated zone by anthropogenic impact in the downstream of river Gadilam. A few of the groundwater samples in the study area were found to be unsuitable for domestic and drinking purposes.
Arabian Journal of Geosciences, 2013
A hydrogeochemical investigation was conducted in a coastal region of Cuddalore district to identify the influence of saltwater intrusion and suitability of groundwater for domestic and agricultural purposes. The geology of the study area comprises of sandstone, clay, alluvium, and laterite soils of Tertiary and Quaternary age. A total of 18 groundwater samples were analyzed for 14 different water quality parameters and the result indicates higher concentrations of ions like Cl (3,509 mg/l), Na (3,123 mg/l), and HCO 3 (998 mg/l) when compared with WHO, BIS, and ISI standards. A positive correlation (r 2 =0.82) was observed between Na and Cl, indicating its sources from salt water intrusion. Three factors were extracted with a total variance of 64% which indicates the sources of salinization, cation exchange, and anthropogenic impact to the groundwater. The Piper trilinear diagram indicates both Na-Cl and mixed Na-HCO 3 -Cl-type, indicating that groundwater was strongly affected by anthropogenic activities. The plot of (Ca+Mg)/(K+Na) indicates evidences of cation exchange and salt water intrusion. The (Ca-0.33*HCO 3 )/ SO 4 plot indicates salt water intrusion for elevated SO 4 levels rather than gypsum dissolution. The spatial distribution of total dissolved solid indicates the saline water encroachment along the SW part of the study area. As per sodium adsorption ratio (SAR), 50% of the samples with <10 SAR are suitable for irrigation and >10 SAR indicates that water is unsuitable for irrigation purposes. The residual sodium carbonate classification indicates that 50% of the samples fall in safe and 50% of the samples fall in bad zones and prolonged usage of this water will affect the crop yield. The Chloro Alkaline Index of water indicates disequilibrium due to a higher ratio of Cl>Na-K, indicating the influence of salt water intrusion. The Permeability Index of the groundwater indicates that the groundwater from the study area is moderate to good for irrigation purposes.
International Journal of Energy and Water Resources, 2022
Groundwater is the only freshwater in the region, as textile industry effluents pollute the Noyyal river. Therefore, a hydrogeochemical investigation was conducted in the western Coimbatore district of Tamil Nadu to examine the groundwater chemistry and its appropriateness for drinking and irrigation. For this purpose, 25 groundwater samples from a borewell were obtained in precleaned 1-L polythene bottles within the study area. The study found that almost all the analysed groundwater samples contained total hardness (TH), total dissolved solids (TDS), sodium (Na +), potassium (K +), calcium Ca 2+ , bicarbonate HCO − 3 , and chloride (Cl −) above the World Health Organization and the Bureau of Indian Standards recommendations for drinking water. In the investigated area, the groundwater chemistry is predominantly of Ca 2+ − HCO − 3 type, as shown in the Piper Trilinear diagram. Gibbs plot shows that groundwater chemistry is controlled by rock-water interaction. A geographic information system (GIS) platform was used to create a spatial distribution map of all the physical and chemical parameters. About 60% of the groundwater sample fall in the poor water class according to the Water Quality Index (WQI) assessment. In contrast, most groundwater samples are found suitable for irrigation purposes by computing sodium percentage, sodium adsorption ratio, salinity hazard, Kelly ratio, residual sodium carbonate and permeability index. These hydrogeochemical results could benefit stakeholders and farmers by managing the water resources in the study area.
References (56)
- Afroz R, Banna H, Masud MM, Akhtar R, Yahaya SR (2016) House- hold's perception of water pollution and its economic impact on human health in Malaysia. Desalination and Water Treatment 57(1):115-123
- APHA (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association/ American Water Works Association/Water Environment Federa- tion, Washington, DC
- Ayers RS, Westcot DW (1985) Water quality for agriculture, vol 29. Food and Agriculture Organization of the United Nations, Rome
- Basu S, Lokesh KS (2012) Trend of temporal variation of Cauvery river water quality at KR Nagar in Karnataka. Int J Eng Sci Tech- nol 4(8):3693-3699
- Bhargava DS (1985) Water quality variations and control technology of Yamuna river. Environ Pollut A 37(4):355-376
- Bhutiani R, Khanna DR, Kulkarni DB, Ruhela M (2016) Assessment of Ganga river ecosystem at Haridwar, Uttarakhand, India with reference to water quality indices. Appl Water Sci 6(2):107-113 BIS (1982) Indian standard tolerance limits for Inland surface water subject to pollution, IS 2296:1982. Bureau of Indian Standards, New Delhi BIS (2012) Indian standard drinking water specification, second revi- sion IS 10500:2012. Bureau of Indian Standards, Drinking Water Sectional Committee, FAD25, New Delhi
- Brown RM, McCleiland NJ, Deiniger RA and O'Connor MFA (1972) Water quality index-crossing the physical barrier. In: Jenkis SH (ed) Proceedings in international conference on water pollution research. Jerusalem, pp 787-797
- Brown P, El Gohary F, Tawfic MA, Hamdy EI, Abdel-Gawad S (2003) Nile river water quality management study. Egypt Water Policy Reform, United States Agency for International Development, Egypt CPCB (Central Pollution Control Board) (2008) Status of water qual- ity in India-2007. http://www.cpcb.nic.in/upload/NewItems/ NewItem_129_NWMP-2007.pdf. Accessed 21 Dec 2016
- CPCB (Central Pollution Control Board) (2014) Status of water quality in India-2007. https://data.gov.in/catalog/status-water-quality- india-2012. Accessed 21 Dec 2016
- CWC (Central Water Commission) (2016) Integrated hydrologic data book, New Delhi. http://www.cwc.nic.in/main/downloads/ IHD2015_final.pdf. Accessed 21 Dec 2016
- Dahunsi SO, Owamah HI, Ayandiran TA, Oranusi SU (2014) Drinking water quality and public health of selected towns in South Western Nigeria. Water Qual Expo Health 6(3):143-153
- Domenico PA, Schwartz FW (1990) Physical and chemical hydrogeol- ogy. Wiley, New York, pp 410-420
- Doneen LD (1964) Water quality for agriculture. Department of Irriga- tion, University of California, Davis
- Eaton EM (1950) Significance of carbonates in irrigation waters. Soil Sci 69:123-133
- Freeze RA, Cherry JA (1979) Groundwater. Englewood Cliffs, New Jersey, p 604
- Gaillardet J, Dupre B, Louvat P, Allegre CJ (1999) Global silicate weathering and CO 2 consumption rates deduced from the chem- istry of large rivers. Chem Geol 159(1):3-30
- Garbarino JR, Antweiler RC, Brinton TI, Roth DA, Taylor HE (1995) Concentration and transport data for selected dissolved inorganic constituents and dissolved organic carbon in water collected from the Mississippi River and some of its tributaries, July 1991-May 1992. US Geol Surv Open File Rep 149:95-149
- Goher ME, Hassan AM, Abdel-Moniem IA, Fahmy AH, El-sayed SM (2014) Evaluation of surface water quality and heavy metal indices of Ismailia Canal, Nile River, Egypt. Egypt J Aquat Res 40(3):225-233
- Grosbois C, Négrel P, Grimaud D, Fouillac C (2001) An overview of dissolved and suspended matter fluxes in the Loire river basin: natural and anthropogenic inputs. Aquat Geochem 7(2):81-105
- Hart FG (1999) World delta database, Cauvery. http://www.geol.lsu. edu/WDD/ASIAN/Cauvery/cauvery.htm. Accessed 21 Dec 2016
- Huang X, Sillanpää M, Gjessing ET, Peräniemi S, Vogt RD (2011) Water quality in the southern Tibetan Plateau: chemical evalu- ation of the Yarlung Tsangpo (Brahmaputra). River Res Appl 27(1):113-121
- India Wris Wiki (2015) Cauvery. http://www.india-wris.nrsc.gov.in/ wrpinfo/index.php?title=Cauvery. Accessed on 21 Sept Jayananda M, Moyen JF, Martin H, Peucat JJ, Auvray B, Mahabaleswar B (2000) Late Archaean (2550 2520 Ma) juvenile magmatism in the Eastern Dharwar craton, southern India: constraints from geochronology, Nd Sr isotopes, and whole rock geochemistry. Precambrian Res 99:225-254
- John MM, Balakrishnan S, Bhadra BK (2005) Contrasting metamor- phism across Cauvery Shear Zone, south India. J Earth Syst Sci 114(2):1-16
- Kalpana L, Elango L (2013) Assessment of groundwater quality for drinking and irrigation purposes in Pambar river sub-basin, Tamil Nadu. Indian J Environ Prot 33(1):1-8
- Kelly WP (1957) Adsorbed sodium cation exchange capacity and per- centage sodium sorption in alkali soils. Science 84:473-477
- Lloyd JW, Heathcote JA (1985) Natural inorganic hydrochemistry in relation to groundwater. Clarendon Press, Oxford
- Meybeck M (1983) Atmospheric inputs and river transport of dissolved substances. Dissolved Loads Rivers Surf Water Quant/Qual Rela- tionsh 141:173-192
- Meybeck M, Ragu A (2012) GEMS-GLORI world river discharge database. Laboratoire De Géologie Appliquée, Université Pierre et Marie Curie, Paris
- Milliman J (2001) River inputs. In: Steele JH (ed) Encyclopedia of ocean sciences. Academic Press, London, pp 2419-2427
- Mohsin M, Safdar S, Asghar F, Jamal F (2013) Assessment of drinking water quality and its impact on residents health in Bahawalpur city. Int J Humanit Soc Sci 3(15):114-128
- Moosdorf N, Hartmann J, Lauerwald R, Hagedorn B, Kempe S (2011) Atmospheric CO 2 consumption by chemical weathering in North America. Geochim Cosmochim Acta 75(24):7829-7854
- Mukherjee D, Chattopadhyay M, Lahiri SC (1993) Water quality of the River Ganga (The Ganges) and some of its physico-chemical properties. Environmentalist 13(3):199-210
- Naqvi SM, Divakara Rao V, Satyanarayana K, Hussain SM (1974) Geochemistry of post-Dharwar basic dykes and the Precambrian crustal evolution of Peninsular India. Geol Mag 111:229-236
- Pattanaik JK, Balakrishnan S, Bhutani R, Singh P (2013) Estimation of weathering rates and CO 2 drawdown based on solute load: sig- nificance of granulites and gneisses dominated weathering in the Kaveri River basin, southern India. Geochim Cosmochim Acta 121:611-636
- Pichamuthu CS (1976) Some problems pertaining to the Peninsular Gneiss Complex. J Geol Soc India 17:1-16
- Pichamuthu CS (1978) Archaean geology investigations in southern India. Geol Soc India 19(10):431-439
- Piper AM (1944) A graphical procedure in the geochemical interpre- tation of water analysis. Trans Am Geophys Union 25:914-928
- Porcella DB, Sorensen DL (1980) Characteristics of nonpoint source urban runoff and its effects on stream ecosystems. Corvallis Envi- ronmental Research Laboratory, Office of Research and Devel- opment, US Environmental Protection Agency, United States of America
- Radhakrishna BP (1956) The closepet granite of Mysore State, India. Mysore Geologists' Association Special Publication, Bangalore, pp 1-110
- Rao KL (1975) India's water wealth. Orient Longman, New Delhi Rasool A, Xiao T, Farooqi A, Shafeeque M, Liu Y, Kamran MA, Kat- soyiannis IA, Eqani SAMAS (2017) Quality of tube well water intended for irrigation and human consumption with special emphasis on arsenic contamination in the area of Punjab, Pakistan. Environ Geochem Health 39(4):847-863
- Reynolds SE (1972) Water quality problem on the Colorado River. Nat Resour J 12:480
- Richards LA (1954) Diagnosis and improvement of saline and Alkali soils. USDA Handbook, Washington, DC
- Sawyer CN, McCarty PL (1978) Chemistry of environmental engineer- ing. Series in water resources and environmental engineering, 3rd edn. McGraw-Hill, New York
- Shiklomanov IA (1998) World water resources. A new appraisal and assessment for the 21st century. UNESCO, Paris
- Shio T, Maddocks A, Carson C, Loizeaux E (2015) 3 Maps explain India's growing water risks. http://www.wri.org/blog/2015/02/3- maps-explain-india%E2%80%99s-growing-water-risks. Accessed on 5 Oct
- Solaraj G, Dhanakumar S, Murthy KR, Mohanraj R (2010) Water qual- ity in select regions of Cauvery Delta River basin, southern India, with emphasis on monsoonal variation. Environ Monit Assess 166(1-4):435-444
- Subramanian KS, Selvan TA (2001) Geology of Tamil Nadu and Pondi- cherry, 1st edn. Geological Society of India, Bangalore, p 192
- Sundaram R, Rao PS (1981) Lithostratigraphy of Cretaceous and Pal- aeocene rocks of Tiruchirapalli District, Tamil Nadu, South India. GSI Rec 115(5):9-23
- Suresh M, Gurugnanam B, Vasudevan S, Dharanirajan K, Raj NJ (2010) Drinking and irrigational feasibility of groundwater, GIS spatial mapping in upper Thirumanimuthar sub-basin, Cauvery river, Tamil Nadu. J Geol Soc India 75(3):518-526
- Susheela FS, Srikantaswamy FS, Shiva Kumar FD, Gowda FA, Jagadish FK (2014) Study of Cauvery river water pollution and its impact on socio-economic status around KRS Dam, Karnataka, India. J Earth Sci Geotech Eng 4(2):91-109
- Szabolcs I, Darab C (1964). The influence of irrigation water of high sodium carbonate content of soils. Proceedings of 8th ISSS Trans, 2:802-812
- Todd DK (1959) Ground water hydrology, 2nd edn. Wiley, New York Vetrimurugan E, Elango L, Rajmohan N (2013) Sources of contami- nants and groundwater quality in the coastal part of a river delta. Int J Environ Sci Technol 10:473-486
- WBCSD (2006) Business in the world of water: WBCSD water sce- narios to 2025. World Business Council for Sustainable Devel- opment. http://www.wbcsd.org/Clusters/Water/Resources/Busi- ness-in-the-World-of-Water-WBCSD-water-scenarios-to-2025 Accessed 17 May WHO (1993) Guidelines for drinking water quality. World Health Organization, 2nd edn. Recommendations, WHO, Geneva WHO (2003a) Sodium in drinking water, background documents for development of WHO guidelines for drinking water quality. World Health Organisation. http://www.who.int/water_sanitation_health/ dwq/chemicals/sodium.pdf. Accessed 16 Oct WHO (2003b) Chloride in drinking water, Background documents for development of WHO guidelines for drinking water quality. World Health Organisation. http://www.who.int/water_sanitation_health/ dwq/chloride.pdf. Accessed 16 Oct WHO (2003c) Total dissolved solids in drinking water, Background documents for development of WHO guidelines for drinking water quality. World Health Organisation. http://www.who.int/ water_sanitation_health/dwq/chemicals/tds.pdf. Accessed 18 May WHO (2007) pH in drinking water, Revised background documents for development of WHO guidelines for drinking water quality. World Health Organisation. http://www.who.int/water_sanitation_health/ dwq/chemicals/ph_revised_2007_clean_version.pdf. Accessed 16 Oct Wilcox LV (1955) Classification and use of irrigation waters. USDA, Washington, DC
- Zhang J, Huang WW, Letolle R, Jusserand C (1995) Major element chemistry of the Huanghe (Yellow River), China-weathering pro- cesses and chemical fluxes. J Hydrol 168(1):173-203
- Zheng Q, Ma T, Wang Y, Yan Y, Liu L (2017) Hydrochemical char- acteristics and quality assessment of shallow groundwater in Xincai River Basin, Northern China. Procedia Earth Planet Sci 17:368-371
- Zhu B (2016) Natural water quality and its suitability for drinking and irrigation purposes in the Jungar Basin, Central Asia. J Civil Environ Eng 6:232
Professor L. Elango, Anna University, Chennai is a hydrogeologist with Masters Degree in Science (Applied Geology) from University of Madras(1984), Masters Degree in Engineering (Hydrology & Water Resources Engg.,)(1986) and Ph.D in Hydrogeology(1992) from Centre for Water Resources, Anna University. He has specialised in hydrogeochemical studies and groundwater modelling. He carried out his postdoctoral work at the University of Birmingham under Indian National Science Academy and The Royal Society, London fellowship programme. He has participated in various professional training programmes held in Danish Hydraulic Institute, Swiss Federal Institute of Technology, University of New Castle, UK and Ruhr University, Germany. He has carried out a number of sponsored research projects on various aspects of hydrogeology. He has published about 140 research papers in various journals. Professor Elango was a Vice President of International Association of Hydrological Sciences. He was an Associate Editor of International Journal of Environmental Geosciences and International Association of Hydrogeologist’s Hydrogeology Journal. He is/was on the editorial board of some international journals. He has organised many training programmes/workshops and conferences in the field of Hydrogeology. He coordinated a major capacity building programme for the officers of the State’s Water Resources Organisation under the World Bank funded Hydrology Project. He has organised four International workshops sponsored by UNESCO’s International Hydrology Programme. He has carried out a number of research projects. Some of the projects carried out include in collaboration with the British Geological Survey, Australian Research Council and Russian Academy of Sciences. He has also carried out a number of consultancy projects for major organisations such as Dept of Atomic Energy, PWD, MECON, Larson and Toubro, HPCC, Gammon India, Gimpex, .etc. He has travelled under academic and research assignments to Australia, Brazil, Canada, Denmark, France, Finland, Germany, Hong Kong, Hungary, Japan, Netherlands, Russia, Switzerland, Singapore, Sweden, Tunisia, UK and USA. He has supervised eighteen PhD studies until 2016. He received Tamil Nadu Scientist award in the year 2011. His technical paper won the first prize on the Ministry of Water Resources, Govt of India in the year 2016.
Related papers
In the global context water pollution is considered as a major issues pertaining to its pollution level from the anthropogenic activities. In this scenario the conservation strategies plays an important role in the conservation of water bodies as well as water quality. The quality of natural water in rivers, lakes and reservoirs and below the ground surface depends on a number of interrelated factors. Water has the ability to react with the minerals that occur in the soil and rocks and to dissolve a wide range of materials, so that its natural state is never pure. It always contains a variety of soluble inorganic, soluble organic and organic compounds. In addition to these, water can carry large amounts of insoluble materials that are held in suspension. Both the amounts and type of impurities found in natural water vary from place to place and by time of year and depends on a number of factors. In order to examine and evaluate the quality of Cauvery River, water samples were collected from different locations of the river basin. The results showed that the Cauvery river still in the purest water quality condition. Many research studies shown the water quality of the Cauvery River have become pollute and near to pollute, but in the present study it clearly indicates that the Cauvery river water sustains the normal quality form in the sampling location during the study period. It was observed that the impact and entering of human activity was very less in the sampling location of Kodagu District, India.
Present work deals with an assessment of some physico chemical parameters of the water bodies of two rivers (Cauvery and Arasalar) which is suitable for human consumption have been carried out during the period of one year (January 2010 to December 2010). Analysis of some physico-chemical characteristics like air temperature, water temperature, transparency, electrical conductivity, total solids, total dissolved solids, total suspended solids, pH, free carbon dioxide, dissolved oxygen, BOD and COD has been done during the investigation period. BOD values were not compiling with WHO guide lines in the River Cauvery and total solids and BOD values were not compiling with WHO guide lines in the River Arasalar. Study indicates the rivers were slightly polluted by anthropogenic performance due to local anthropogenic activities, agricultural runoff and discharge of untreated municipal sewage, religious credence and subject to amend owed to seasons, climate and flows and influx of waters from various tributaries. In addition present study points out that the river Arasalar facing severe pollution followed by the river Cauvery.
Golden Research Thoughts, 2014
The present study was undertaken to assess the water quality of the selected distributaries of river Cauvery in Tiruchirappalli district. Water samples were collected during 3 seasons (winter, southwest monsoon and northeast monsoon) and analysed for 14 physico-chemical parameters. While water from Cauvery was found fit for all uses (including domestic use) in all the three seasons, water from all the distributaries was found to be unfit for domestic use during winter; and water from Koolayar channel was unfit for domestic use in northeast monsoon too. However, water from all the channels in all the seasons were found suitable for irrigation and recreational uses. The sewage discharge, open defecation and agricultural run-off were the main sources of pollution.
At a local scale groundwater chemistry is influenced by natural mineralogical transformations while on basin scale, the chemistry changes with climatic inputs such as rainwater. Effect of rainfall on chemical behavior of groundwater along a climatic gradient is studied for a river basin by classifying the wells into shallow and deep. Semi-arid (500 -800 mm/year rainfall), sub-humid (1000 mm/year) and humid (1200 -1500 mm/year) zones along Upper Cauvery river in southern part of India are chosen for the analysis.
Sustainable water management in a river basin requires knowledge of the water availability in the basin and current and future demands. An attempt has been made to study determine the groundwater quality in Sarabanga sub basin, Cauvery river basin of crystalline terrain (Archaean age), Salem district, Tamil Nadu, south India. Groundwater quality analysis results were compared with the WHO standards of drinking water quality limits with the following the water quality parameters namely pH, Ec, TDS, Ca, Mg, Na, Cl, HCO3, SO4, TDS and TH etc., Hydrogeochemical facies of groundwater quality in study area reveals that fresh to brackish and alkaline in nature. Piper's plot shows that the groundwater samples fall in the field of CaHCO3, mixed CaMgCl, NaCl respectively, according to the order of their dominance. From the plot, it is observed that nearly 60% of samples fall in alkaline earths Ca2+, HCO3– exceed the other anions. The physical and chemical parameters of the Sarabanga sub-basin, results show that all the samples are under their commended limit for agricultural and drinking purposes.
Cited by
SN Applied Sciences
The present study aimed to assess the impact of municipal solid waste dumpsite on groundwater bodies at Hyderabad, India. Leachate and groundwater samples collected through pre-and post-monsoon analyzed the physicochemical, microbiological, biological and heavy metals. The analytical data were compared with Bureau of Indian Standards (BIS) drinking water quality standards. Water quality index (WQI), heavy metal pollution indices like heavy metal evaluation index (HEI) and degree of contamination (Cd) are calculated for groundwater samples. High total dissolve solids values in leachates revealed that they were highly contaminated with organic and inorganic salts. Biological oxygen demand values indicated that dumpsite was "old and stabilized" with decreasing biodegradability from time to time. According to WQI, about 75% of the water samples identified as "Poor" category that is not suitable for neither drinking nor domestic purposes as per BIS standards. Similarly, HEI and Cd results indicated that majority of the samples are labeled with low-metal pollution status. Spatial patterns obtained through geographic information systems using inverse distance weighted interpolation technique revealed that the concentrations of various parameters are high due to increased degradation of solid wastes during rainfall, especially during the post-monsoon. The study suggested that leachates have treated prior to disposal on land, and continuous monitoring of groundwater wells is required to minimize the pollution and potential health hazards.
Applied Water Science, 2018
Impacts of geogenic and anthropogenic sources change seriously quality of groundwater. Inferior groundwater quality directly affects the human health, agricultural output and industrial sector. The aim of the present study is to evaluate the groundwater quality for drinking purpose and also to identify the pollutants responsible for variation of chemical quality of groundwater, using pollution index of groundwater (PIG). Groundwater samples collected from a rural part of Telangana State, India, were analyzed for pH, total dissolved solids (TDS), calcium (Ca 2+), magnesium (Mg 2+), sodium (Na +), potassium (K +), bicarbonate (HCO − 3), chloride (Cl −), sulfate (SO 2− 4), nitrate (NO − 3) and fluoride (F −). The groundwater is characterized by Na + and HCO − 3 ions. The values of TDS
International Journal of Environmental Research and Public Health
Freshwater supply is essential to life on Earth; however, land use activities such as mining and agriculture pose a significant danger to freshwater resources and the wellbeing of aquatic environments. This study temporarily assesses the water quality characteristics of Mutangwi River. Physicochemical parameters (pH, temperature, total dissolved solids (TDS), salinity, electrical conductivity (EC), and turbidity) were determined in situ using an Extech multimeter and turbidity meter. The concentration of the selected metals (Mg, Cr, Fe, Cd, Mn, Pb, Ca, and Na) were analysed using an Atomic Absorption Spectrophotometer. Membrane filtration method was used to analyse microbiological parameters (Escherichia coli and Enterococci). The physicochemical water quality parameters as well as basic anions (fluoride, phosphate, sulfate, nitrate, and chloride) determined complied with the regulatory guideline of the World Health Organization (WHO) and the South Africa National Standards (SANS). ...
Water, 2019
In the race to enhance agricultural productivity, irrigation will become more dependent on poorly characterized and virtually unmonitored sources of water. Increased use of irrigation water has led to impaired water and soil quality in many areas. Historically, soil salinization and reduced crop productivity have been the primary focus of irrigation water quality. Recently, there is increasing evidence for the occurrence of geogenic contaminants in water. The appearance of trace elements and an increase in the use of wastewater has highlighted the vulnerability and complexities of the composition of irrigation water and its role in ensuring proper crop growth, and long-term food quality. Analytical capabilities of measuring vanishingly small concentrations of biologically-active organic contaminants, including steroid hormones, plasticizers, pharmaceuticals, and personal care products, in a variety of irrigation water sources provide the means to evaluate uptake and occurrence in cr...
Water
In order to evaluate and project the quality of groundwater utilized for irrigation in the Sahara aquifer in Algeria, this research employed irrigation water quality indices (IWQIs), artificial neural network (ANN) models, and Gradient Boosting Regression (GBR), alongside multivariate statistical analysis and a geographic information system (GIS), to assess and forecast the quality of groundwater used for irrigation in the Sahara aquifer in Algeria. Twenty-seven groundwater samples were examined using conventional analytical methods. The obtained physicochemical parameters for the collected groundwater samples showed that Ca2+ > Mg2+ > Na+ > K+, and Cl− > SO42− > HCO3− > NO3−, owing to the predominance of limestone, sandstone, and clay minerals under the effects of human activity, ion dissolution, rock weathering, and exchange processes, which indicate a Ca-Cl water type. For evaluating the quality of irrigation water, the IWQIs values such as irrigation water qual...
Water
Water quality monitoring is crucial in managing water resources and ensuring their safety for human use and environmental health. In the Al-Jawf Basin, we conducted a study on the Quaternary aquifer, where various techniques were utilized to evaluate, simulate, and predict the groundwater quality (GWQ) for irrigation. These techniques include water quality indices (IWQIs), geochemical modeling, multivariate statistical analysis, geographic information systems (GIS), and adaptive neuro-fuzzy inference systems (ANFIS). Physicochemical analysis was conducted on the collected groundwater samples to determine their composition. The results showed that the order of abundance of ions was Ca2+ > Mg2+ > Na+ > K+ and SO42− > Cl− > HCO3− > NO3−. The assessment of groundwater quality for irrigation based on indices such as Irrigation water quality index (IWQI), sodium adsorption ratio(SAR), sodium percent (Na%), soluble sodium percentage (SSP), potential salinity (PS), and res...
Water, 2023
This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY
International Journal of Environmental Research and Public Health
This study was a continuation of our investigation of the spatio-temporal variability of the Bzura River’s water chemistry. Our research is of particular importance in the context of the recent ecological disaster on the Oder River and concerns the international problem of surface water contamination. The study area was a 120 km section of the Bzura River. We tested more measurement points and with a higher sampling frequency than those used in the national monitoring of river water quality. During two hydrological years, 360 water samples were collected. The selected parameters: electrical conductivity, temperature, dissolved oxygen, dissolved organic carbon, nitrates, phosphates, bicarbonates, chlorides, sodium, potassium, calcium, and magnesium were determined. Numerous results exceeded the Polish threshold limits. Spatio-temporal variability and water quality were assessed using principal component analysis (PCA), cluster analysis (CA), and water quality index (WQI) approaches. ...
International Journal of Civil, Environmental and Agricultural Engineering
This research aims to determine the health consequences of fluoride contamination of groundwater in the Namakkal region in south India using the groundwater quality index (GWQI). Study area latitude and longitude: 11° 00' and 11° 30' in the north, and 77° 45' and 78° 15' in the east. Statewide, it is among the largest districts in the state. The study region occupies an area of 3406.37 km2. The geology of the studied area is mainly based on the Archaean crystalline and metamorphic complex. The district's major aquifer systems are composed of crystalline rocks that are weathered and fractured and of colluvial deposits. Alluvium and colluvium are examples of porous formations in the cross-section. Only the main river channels have alluvial deposits. The phreatic properties of groundwater Depending on the topography, these aquifers may reach 5 m saturation thickness. Groundwater samples were obtained from 58 bore well sites across the study area during the North-Ea...
 Elango L
Elango L
